2
Protecting Animal Health and Well-being: Nutrition and Immune Function
KIRK C. KLASING
University of California, Davis
The immune system protects animal health and contributes to animal well-being. Nutrition is an important modulator of immune function and can often tip the balance between health and disease. Current Committee on Animal Nutrition (CAN) reports provide important recommendations on nutrient requirements that account for desired or expected animal performance. Industry professionals and university scientists would like to examine other major functional endpoints. If animal health was selected as the functional endpoint instead of growth rate or reproductive performance, nutrient recommendations might change (Figure 2-1). In most cases, National Research Council (NRC) recommendations for estimated requirements are probably adequate for optimal animal health and well being, but the goal of CAN committees should be to look for exceptions.
Animal nutrition research often relies on information from human studies to point to nutrients that may require more in-depth examination. Whereas information from human studies is important (Gershwin et al., 2000), laboratory animal studies give us a better understanding of mechanistic and quantitative aspects of the immune system, such as how much of a specific nutrient is needed for the immune system to do its job and how insufficiencies or excesses of nutrients affect the immune system. It is also important to determine what
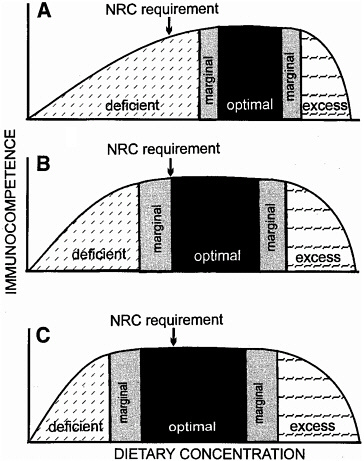
FIGURE 2-1. Dietary requirements set by the National Research Council (NRC) (shown as arrows) are usually based on concentrations that maximize growth and reproduction and prevent known deficiency pathologies. Optimal immunocompetence may occur at nutrient levels that are: higher than the NRC requirement (panel A); equal to the NRC requirement (panel B); or less than the NRC requirement (panel C).
components within the immune system should be measured and what functional endpoints are the best indicators of animal health.
Eventually target animal species need to be examined to determine specific concentrations of nutrients for optimal immunocompetence and health. Field tests and epidemiologic studies in production facilities will be necessary to verify the results with authentic disease challenges. Achieving this goal will be complicated because each species of animal has potential pathogens because and the defense to these organisms may respond differently to nutritional
manipulations. Most veterinarians track 20 diseases and nutritionists are concerned with 45 or more nutrients at any given time. So, the number of combinations in one species is formidable. Sorting out these interactions between nutrition and disease can be simplified by focusing on the immune system and the modulation of this system by dietary components (Klasing, 1998).
ASSUMPTIONS REGARDING IMMUNOLOGY RESEARCH
When examining the immune system, a correlation between immunity and disease resistance is assumed. This is true for most infectious diseases, but sometimes other physiologic systems, such as membrane integrity and types of receptors on epithelial cells, are the major deciding factors.
It is also assumed that scientists know what components of the immune system to measure. This is not necessarily true. In the last 15–20 years, major advances in immunology have been driven by interest in heart disease, cancer, and basic science. These studies have given scientists a better understanding of what components to measure, but uncertainty still remains when attempting to measure animal health and susceptibility to disease. Recent efforts by toxicologists designed to find endpoints of immunocompetence that best correlate with disease resistance (Dean, 1997) should be noted by animal nutritionists as they design their research.
DIETARY INFLUENCES ON THE IMMUNE SYSTEM
Fundamental mechanisms through which diet influences the immune system and various mechanistic aspects need to be examined to determine how and why diet affects the immune system (Table 2-1). Among some of the areas to be examined are substrate needs, nutritional immunity, direct regulatory effects, hormone balance, pathology, and non-nutrient components of feeds. Examining these areas closely may provide answers to immunologic and other questions as well.
Substrate Needs
Surprising little information is available that indicates how big the immune system is, what it needs to do its job, or even its priorities for nutrient use when they become limiting. The immune system is relatively small when estimated as a percentage of body weight. The weight of all the leukocytes and their products involved in immune function are probably less than 2 percent of body weight. Although the immune system is relatively small, it is a vital system whose requirements must be met.
TABLE 2-1. Mechanisms by which nutrition modulates immunocompetence and disease resistance.
|
Mechanism |
Nutrients |
|
The supply of substrates to the immune system |
All nutrients |
|
Deprivation of nutrients from pathogens (nutritional immunity) |
Iron, biotin, manganese |
|
Direct regulatory effects on cells of the immune system |
Fatty acids, vitamins A, D, E |
|
Changing the balance of hormones that regulate immunity |
Energy, protein, meal patterns |
|
Reduction of pathology induced by an immune response |
Antioxidants |
|
Physical and chemical actions of feeds in the intestines |
Non-starch polysaccharides, lectins, sugars |
The diet supplies substrates, such as energy and amino acids, that contribute to the development, maintenance and use of the immune system. Substrates are necessary for the anabolic activity of immune system’s cells (leukocytes), such as proliferation and antibody production as well as the secretion by the liver of large quantities of immunologically active molecules, the acute phase proteins. In young animals, a severe deficiency of virtually any nutrient impairs many indices of immunocompetence (Cook, 1991). Such a situation is rare in modern animal husbandry, and questions that consider the needs of the immune system relative to other systems (e.g., growth) are most relevant.
Though an understanding of how the cells of the immune system obtain their nutrients is incomplete, it is beginning to appear that the immune system uses many of the same types of glucose and amino acid transporters that nervous and other high priority tissue use. When leukocytes become activated, they express high levels of nutrient transporters, which allows them to easily obtain necessary nutrients even when they are at low concentrations. The immune system can also appropriate nutrients from muscle and other tissues. When leukocytes become stimulated by pathogens, they release a series of pro-inflammatory cytokines like interleukin 1, tumor necrosis factor and interleukin 6 that go throughout the body and redistribute nutrients—especially those from skeletal muscle. The small size of the immune system, its capacity to appropriate nutrients from other tissues, and its endowment with high priority nutrient transporters generally indicate that the immune system can usually obtain many of the nutrients that it needs to do its job over a wide range of dietary levels. However, some trace nutrients such as iron, copper, and zinc are problematic because of their low concentration in muscle and their relatively high need within the immune system. Evidence is accumulating that the dietary requirement for some trace nutrients may be higher for optimal immune function than it is for maximal growth or reproductive
performance. Clearly more work is needed to determine the quantitative nutrient requirements of the immune system for its development in the young animal, its maintenance at times of good health, and its use during a challenge by a virulent pathogen.
Nutritional Immunity
Nutritional immunity is the process whereby the body withholds essential nutrients from pathogens to reduce their rate of replication. For example, it is well documented that injecting or orally feeding baby pigs iron provides additional amounts of this limiting nutrient that enhance the growth of pathogens. In this case, pathology takes the form of increased severity and duration of diarrhea. These pigs are also more likely to die. In birds, a similar situation exists within the egg to deprive bacteria of nutrients so that they are unable to colonize the albumen and infect the developing embryo. Immune cells sequester trace minerals, such as manganese and iron, when they engulf pathogens, and this action serves to starve pathogens and prevent their replication. As there are situations where high levels of specific dietary nutrients compromise immune function, the idea that “more is better” should be applied with caution.
Direct Regulatory Effects
Cellular communication within the immune system is critical because this system is one of the most complex, dynamic, and potentially destructive systems within the body. The number of communication molecules utilized by the immune system rivals that of the brain. Nutrients in the diet can directly affect the regulatory functions of leukocytes altering the type, duration, and vigor of the immune response.
For example, dietary factors such as type of fat can change the proportion of prostaglandins and other eicosanoids that are released by leukocytes to coordinate their responses to disease challenges. This response is because the type of dietary fat changes the composition of the phospholipids in the membranes of leukocytes, and these membrane fatty acids are the precursors for the synthesis of eicosanoids. Thus, the potency and specific regulatory properties of the eicosanoids released during immune responses change with the composition of dietary fat. Fish oil is high in eicosapentaenoic acid, which causes macrophages to be predisposed to release interleukins that drive T helper cells toward a Th2 type of response and less predisposed to a Th1 type of response, especially the inflammatory response (Fritsche et al., 1999; Korver and Klasing, 1997). These divergent responses are important in defense against different pathogens. It is important to note that nutrients that effect communication within the immune system “modulate” or “change” the response, accentuating some components of the response while decreasing others; the
dietary manipulations do not “boost” the entire immune system. Thus, host resistance to specific pathogens shifts - with better resistance to some pathogens, but greater susceptibility to others. In the case of dietary fish oil, the prevalence of those diseases in which protection is mediated by a Th2 response is diminished, whereas the incidence of those where protection is afforded by the inflammatory response is increased.
Many other examples demonstrate how diet can affect the communication and regulatory decisions made by cells in the immune system. While lipids are one of the best demonstrated areas, there is developing literature on the regulatory actions of vitamins A, D, and E, xanthophylls, as well as some amino acids and bioactive minerals.
Hormone Balance
Diet also affects the balance of various hormones that modulate the immune system. Feeding regimes markedly affect insulin, glucagon, glucocorticoid, and IGF levels, which can change the type and duration of the immune response. For example, chronic severe feed restriction results in elevated levels of glucocorticoids, which impinge on T-cell function and decrease many indices of immunocompetence. However, very moderate restriction of food intake can increase immunocompetence and decrease the incidence of infectious diseases. When broiler chickens are restrictively fed, insulin levels are decreased and glucagon levels are increased. Changes in these hormone levels affect the chicken’s ability to mobilize neutrophils, which affects their resistance to various types of disease. Other dietary factors that impact immunity through their effects on hormone levels include protein to calorie ratios and presenting food ad libitum versus in a few large daily meals.
Pathology
The immune system releases a variety of noxious substances at the site of infection in order to kill invading pathogens. Immune systems respond in a measured way so lethality is localized to the pathogen and not on surrounding tissues. However, collateral damage to healthy cells in the area surrounding the site of infection is evident in many infections. Nutritional factors that minimize the extent of pathology induced by immune responses mitigate the nutritional costs for repair and convalescence. For example, reactive oxygen intermediates released at the site of infection can cause damage to the cell membranes of healthy host cells and adequate levels of dietary antioxidants minimize this pathology (Chew, 1995).
Non-Nutrient Components of Feeds
Feeds have specific effects on immunocompetency when they contain non-nutrient substances that influence the function of leukocytes, the integrity of the intestinal epithelia, or the population of commensal microflora found in the intestines. Feed components with these activities include sugars, lectins, and other mitogens, lignins and silicas. Scientists are just beginning to understand what non-nutrient components in feeds are important and what levels of these components cause beneficial and detrimental immunomodulation.
CONCLUSIONS
The immune function of all animals should be optimized to ensure health and welfare of the individual and of the group. An optimal immune response can be measured and can occur only under appropriate conditions. Optimizing the immune system is important because responses with the wrong leukocyte populations or under-responsiveness can increase the incidence of infectious diseases, whereas over-exuberant responses result in a variety of problems in an animal. In fact, human diseases of aging such as arthritis and arteriosclerosis are caused by the immune system. Production animals can develop anorexia, impaired growth, and other systemic stress responses that reaffirm that more is not always better and what is optimal is not necessarily maximal. An effective method of assessing and maximizing the health of animals is necessary.
The ultimate goal of research efforts should be to minimize the nutrition-disease cycle, where poor nutrition causes poor immunocompetency. Poor immunocompetency, in turn, can result in greater incidence and duration of infections, which cause decreased food intake, nutrient losses, and impaired animal health and well-being.
REFERENCES
Chew, B.P. 1995. Antioxidant vitamins affect food animal immunity and health. J. Nutr. 125:1804S-1808S.
Cook, M.E. 1991. Nutrition and the Immune Response of the Domestic Fowl. Cr. Rev. Poult. Biol. 3:167-190.
Dean, J.H. 1997. Issues with introducing new immunotoxicology methods into the safety assessment of pharmaceuticals. Toxicology 119:95-101.
Fritsche, K.L., M. Byrge, and C. Feng. 1999. Dietary omega-3 polyunsaturated fatty acids from fish oil reduce interleukin-12 and interferon-gamma production in mice. Immunol. Let. 65:167-173.
Gershwin, M.E., J.B. German, and C.L. Keen. 2000. Nutrition and immunology: principles and practice. Humana Press, Totowa, New Jersey.
Klasing, K.C. 1998. Nutritional modulation of resistance to infectious diseases. Poultry Sci. 77:1119-1125.








