Page 287
7
Occupational Stress
As indicated in the review of the epidemiologic literature in Chapter 4, a number of workplace psychosocial factors can affect work-related back and upper extremity disorders. The literature provides strong evidence for the role, in low back disorders, of job satisfaction, monotonous work, social support at work, high work demands, job stress, and emotional effort at work. The perception of one's ability to return to work was also positively associated with future back pain. While the literature on upper extremity disorders is not so extensive as with back disorders, higher levels of perceived job demands and job stress were the psychosocial factors most consistently linked to upper extremity disorders. The reviews of the epidemiologic literature also indicated that certain psychosocial factors that are not work-specific (e.g., general worry/psychological tension, depression/anxiety, general coping style, and response to pain) were also associated with both back and upper extremity disorders. Nonwork-related variables tend to be more commonly related to back than to upper extremity disorders.
Given that the emphasis of this report is on work-related factors, this chapter reviews various models of occupational stress and discusses how exposure to stresses at work can impact the physiology of musculoskeletal pain in the spine and upper extremities. Nonworkplace psychosocial stressors can exert similar effects, but are not discussed here.
The study of occupational stress is a difficult endeavor because of the many factors that can influence the development, exacerbation, and maintenance of job stress and the highly subjective nature of measures of exposure and outcomes used in this area. In addition, the various biological correlates of stress exposure and, more specifically, the proposed models of how job stress may affect occupational musculoskeletal disor-
Page 288
ders, are speculative. Also, if biological pathways linking job stress to work-related musculoskeletal disorders exist, it is currently unknown whether they are specific to these disorders or, more likely, represent the final common pathway by which exposure to both work-related and nonwork-related stressors exert an effect on a number of health disorders (e.g., cardiovascular disease). That is, the specificity of these pathways is unknown. It is generally accepted that musculoskeletal pain can be experienced in the absence of evident physiological change or tissue damage (Melzack, 1999) and that such pain is modulated primarily by cognitive processes.
This chapter reviews general models of occupational stress, biological correlates of stress exposure, selected theories related to how occupational stress might impact musculoskeletal disorders, and hypothesized pathways that may account for the relationship.
GENERAL MODELS OF OCCUPATIONAL STRESS
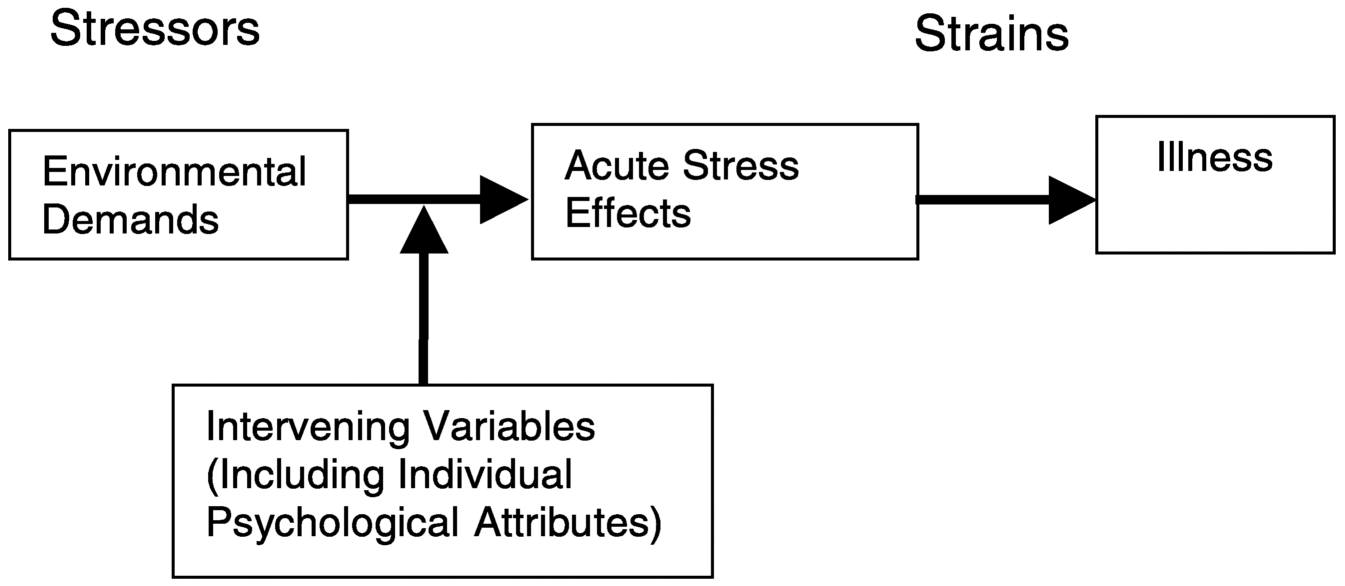
Several general models of occupational stress have emerged that define job stress and explain how certain aspects of work can contribute to the experience of stress. An early model proposed by Levi (1972) includes the components of most models of occupational stress. A simplified version of the major features of this and most models of stress is depicted in Figure 7.1.
The Levi model describes a process by which a worker is in constant interchange with his or her work environment; these interactions require
~ enlarge ~
FIGURE 7.1 Generic psychosocial stress model. From Sauter and Swanson (1996:6). Reprinted with permission.
Page 289
continuous adaptation by the worker. When these transactions are perceived as uncontrollable, the event or situation generates a condition of psychological distress that, if persistent or repeated, can lead to negative health outcomes. The first component of this stress model includes social structures (such as workplace, family) and social processes (events that take place within the social structure). These structures and events are continuously perceived, appraised, and evaluated by the individual. When there is a persistent discrepancy between the worker's abilities, personal needs, expectations, environmental demands, and the opportunities, potential outcomes, social structures, and/or events, or when transactions are perceived as threats, then a complex emotional, cognitive, behavioral, and physiological reaction is evoked.
This response can result in transitory disturbances in mental and physical function (stress response) that, if prolonged, can eventually lead to persistent feelings of distress (frustration, anger, anxiety, dysphoria) and subsequently to physical disorders. Presumably, this response to stress is affected by early environmental influences and genetic factors. It is proposed that this cascade of events can be modified by a number of “interacting variables,” including, for example, a worker's coping repertoire and skills, and the availability and use of social support. These models typically propose the existence of a set of stressors, which are generally defined as environmental demands, and responses to these stressors, often referred to as strains. These strains are the acute effects of the exposure to stressors. A set of intervening factors plays a role in modulating the effect of the demands on strain. As indicated earlier, these intervening factors can include coping skills, problem solving abilities, past learning and exposure, and biological predisposition to react to stress. It is important to note that there are other models of occupational stress (e.g., Hurrell, 1987; Karasek and Theorell, 1990) that have not been reviewed in this chapter. The purpose of this section is not to provide an exhaustive review of models but rather to provide a general overview of the components of most models.
BIOLOGICAL RESPONSE
Work can trigger a complex set of behavioral, cognitive, and physiological responses or strains, commonly referred to as the stress response. The stress response is typically associated with systemic and localized physiological changes that are intended to reestablish a biological state of homeostasis (Selye, 1956). It is postulated that recurrent or chronic exposure to a wide range of intrinsic or extrinsic stressors or demands repeatedly evokes a stress response, which, in turn, contributes to the etiology, exacerbation, and maintenance of a number of prevalent health problems
Page 290
(Chrousos and Gold, 1992; Baum and Posluszny, 1999; Kiecolt-Glaser et al., 1995).
Many models of human stress propose that the presence and severity of a stress response are proportional to an individual's appraisal of the extent of threat represented by the specific stressor (Lazarus, 1974). Psychological processes (e.g., emotions, behaviors, memory) involved in stress appraisal or evaluation and subsequent response are assumed to trigger the biological consequences of stress. Pathways linking psychological and biological processes are often referred to as “biobehavioral.” In an effort to understand the potential physiological effects of stress on adverse health outcomes, various biobehavioral pathways have been proposed (Kiecolt-Glaser et al., 1995; Glaser and Kiecolt-Glaser, 1998; Baum and Posluszny, 1999).
Two common pathways through which exposure to stressors can affect health are the corticotropin-releasing hormone and the locus ceruleus-norepinephrine/autonomic (sympathetic) systems. These two systems are regulated both centrally (brain) and peripherally; they can exert their effects on target tissues and bodily systems through substances released in response to exposure to stressful conditions. These substances include adrenocorticotropic hormone, glucocorticoids, and catecholamines (epinephrine and norepinephrine). Recent research on neuro-immunomodulation (the bidirectional signaling between both the central and the peripheral nervous systems and the cells and organs of the immune system) has demonstrated that the brain can send neuroendocrine signals to cells within the immune system; the immune system, in turn, through the release of cytokines, can signal neurons in the brain and the peripheral nervous system (Felten and Maida, 2000; Watkins, Maier, and Goehler, 1995; Chrousos and Gold, 1992).
The significance of these findings is that this bidirectional communication system can serve as one pathway by which exposure to stressful events in the environment can translate to profound changes in immune function, thereby affecting a range of health outcomes. Specifically, cytokines or molecules released from various cells throughout the body following injury can interact not only with other cells within the immune system, altering their intracellular capabilities, but can also be directly affected by the brain's processing of the meaning of external events, such as stressors on the job and problems at home. While the complex biological processes involved in the stress response are just beginning to be revealed, current knowledge regarding the biological response to stress provides a foundation for understanding how psychological stressors may modulate activity within the nervous, endocrine, and immune systems, thereby exerting an effect on health.
Page 291
OCCUPATIONAL STRESS AND WORK-RELATED MUSCULOSKELETAL DISORDERS: SOME WORKING HYPOTHESES
A number of nonspecific biobehavioral mechanisms have been hypothesized to delineate how stress may affect the physiological processes involved in common musculoskeletal disorders (Bongers et al., 1993; Smith and Carayon-Sainfort, 1989; Smith and Carayon, 1996; Melin and Lundberg, 1997; Sauter and Swanson, 1996; Feuerstein, 1996; Turk and Flor, 1999; Marras et al., in press; Sjøgaard, Lundberg, and Kadefors, 2000). At present, these efforts have not identified specific pathways linking stress to back or upper extremity pain, but they do provide general information that offers a preliminary framework for identifying such pathways in the future.
The peripheral pain receptors (nociceptors) thought to be responsible for the initiation of back pain have been identified. Nociceptors responsive to mechanical deformations have been identified in facet joint capsules, spinal ligaments, bone, and the outermost fibers of the disc annulus fibrosus, but not in the disc nucleus. The response of spinal nerve roots and dorsal root ganglia to compression, vibration, and chemical stimuli has been analyzed in part. A possible role for the nerve root vascular supply in mediating the nerve root pain associated with spinal stenosis has also been identified. Afferent pathways in the spinal cord have been mapped. In contrast, the role of central mechanisms (i.e., the brain) that are involved in the cognitive interpretation of work as stressful, and in the concomitant emotional response to such an appraisal, remains largely undetermined with respect to the pathophysiology of various back disorders. Similarly, the role of afferent feedback loops (signals back to the brain from the periphery) is largely unknown.
One biobehavioral hypothesis for how exposure to work-related stressors may be associated with physiological processes involved in low back pain is that certain individuals possess a predisposition to respond to a stressor with increased paraspinal muscle activity, which may lead to ischemia, reflex muscle spasm, oxygen depletion, and the release of pain-producing substances (e.g., histamine, substance P) (Flor and Turk, 1989; Turk and Flor, 1999). It has been proposed that this increased reactivity to stress results in a feedback loop that involves subsequent increased pain, triggering further increases in muscle activity, psychological distress, and pain (Flor and Turk, 1989). Attempts to confirm this model in individuals with chronic low back pain have resulted in inconsistent findings (Flor and Turk, 1989). Investigations have typically measured surface paraspinal EMG in a laboratory setting in response to various nonwork-related stressors. These studies have demonstrated that individuals with
Page 292
chronic low back pain have baseline EMG activity similar to normal controls and show no consistent pattern of accentuated activity different from that of controls. This research does indicate that individuals with low back pain demonstrate a delayed muscle recovery to prestress levels, suggesting that the recovery following exposure to stress may be deficient in those with back pain. However, it is difficult to determine whether this delayed recovery predated the onset of the disorder. It is possible that increased muscle reactivity may be observed in addition to the delayed recovery in individuals with less chronic back pain, or that increased reactivity may be observed only during actual work exposure rather than in response to exposure to a simulated laboratory stress (i.e., during stress exposures with greater ecological validity).
Identification of EMG reactivity to stress may require the use of more sophisticated measurement approaches and laboratory protocols. A recent investigation observed that exposure to psychosocial stress while performing a lifting task resulted in greater cocontraction of a number of spinal muscles and significant increases in spine compression and lateral shear (Marras et al., in press). This study also found that how an individual interacts with his or her environment (i.e., personality style) was further predictive of spinal loading. Those who were more internally focused or introverted generated higher spinal loads (greater trunk musculature coactivation and alterations in movement patterns) in response to stress during an identical lifting task than did those with a more extroverted style. These findings suggest a biomechanical pathway in which a worker's response to stress interacts with work demands and individual psychological style to generate increases in spinal loading. This study highlights the utility of testing models of occupational stress and low back pain within the context of the performance of work tasks.
There is a need to develop more sophisticated models of how occupational stress can affect occupational back pain and to identify the physiological processes involved. In the past, most models of low back pain were based on clinical observations with chronic patients. Models and related research on chronic low back pain do not provide information on the more immediate or shorter-term processes by which stress at work can affect the onset of back pain.
Upper Extremity Disorders
Models Linking Occupational Stress to Work-Related Musculoskeletal Disorders
In contrast to the back literature, over the past decade a number of work-specific models have been proposed to help explain how stress at
Page 293
work could affect upper extremity disorders. Four representative models that attempt to link job stress to work-related upper extremity disorders are the balance theory of job design and stress, the biopsychosocial model of job stress, the ecological model of musculoskeletal disorders, and the workstyle model.
The balance theory of job design and stress provides a framework for examining the relationships and interactions among work organization, ergonomic exposure, job stress, and work-related upper extremity disorders. Smith and Carayon (1996) identify three general domains of the human stress response: biophysiological, behavioral, and psychological or cognitive. As with the generic models of stress, this model proposes that job stressors produce short-term emotional (e.g., adverse mood states), behavioral (e.g., smoking, excessive use of force in work tasks), and physiological (e.g., increased muscle tension, elevated blood pressure, elevated cortisol and catecholamine levels) reactions. For individuals who are chronically exposed to job stress, these reactions can lead to increased risk for adverse health outcomes, which may include—but are not limited to—work-related upper extremity disorders. This model also incorporates individual characteristics, such as age and personality, which may influence the stress response. In addition, the model emphasizes feedback loops among disease or illness, stress reactions, and stressors, and uniquely considers the experience of symptoms as stressors, in and of themselves, which can increase stress reactivity and lead to further adverse mental and physical health outcomes (Smith and Carayon, 1996).
The biopsychosocial model of job stress and musculoskeletal disorders (Melin and Lundberg, 1997) was developed for application to individuals performing light physical work, such as data entry or other types of computer-related work. In this model, job stress (biomechanical or psychological) is defined as any task or situational demand that creates a condition of over- or under stimulation (Frankenhaeuser and Gardell, 1976). Both of these conditions can evoke physiological responses, including increased muscle tension and secretion of cortisol and catecholamine. This model also addresses the potential effects of nonwork-related demands (e.g., household work, child care) and their effect on recovery from stress experienced while at work. It is argued that individuals with high levels of total workload (work and home) are at increased risk because they remain at higher levels of physiological arousal or experience a delayed recovery due to prolonged work demands.
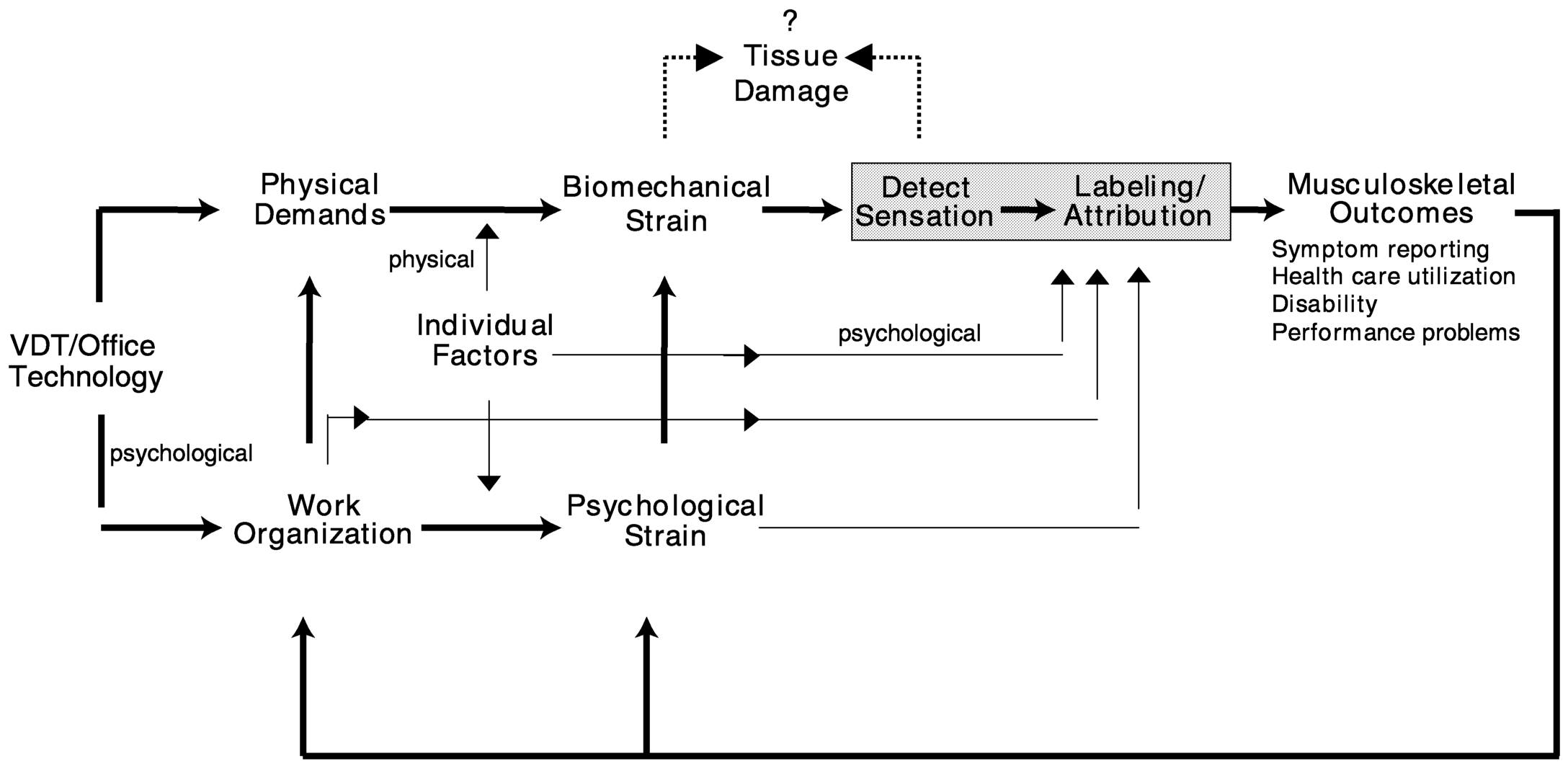
The ecological model of musculoskeletal disorders (Figure 7.2) (Sauter and Swanson, 1996) addresses the interaction of psychosocial and biomechanical stressors. According to this model, work-related musculoskeletal disorders can be ultimately traced to work technology, which includes tools and work systems. In addition, this model proposes a direct path
Page 294
~ enlarge ~
FIGURE 7.2 An ecological model of musculoskeletal disorders. From Sauter and Swanson (1996:8). Reprinted with permission.
Page 295
among work organization, psychosocial stressors, and musculoskeletal outcomes via two routes. First, psychological strain is hypothesized to produce muscle tension and autonomic effects that, in turn, compound biomechanical strain induced by task-related physical demands. Second, psychological strain is hypothesized to moderate the relationship between biomechanical strain and the appearance of symptoms, by means of perception, attribution, and appraisal of symptoms, without directly affecting physical pathology (i.e., cognitive appraisal or interpretation). For example, in the execution of dull, routine, or repetitive tasks, the need to attend to the details of the work may be reduced, increasing the probability that symptoms, which might have gone unnoticed under more stimulating circumstances, will be detected. As with the appraisal of any internal stimulus, once the symptom is perceived, explanations are then sought (Schacter and Singer, 1962). In a stressful work environment, such symptoms or discomfort may be more likely to be attributed to one's job. In this manner, job stress and the psychosocial work environment may increase the probability of help seeking and injury reporting without exerting a direct influence on the underlying pathology. Finally, the model suggests that the experience of musculoskeletal symptoms themselves can influence stress at work.
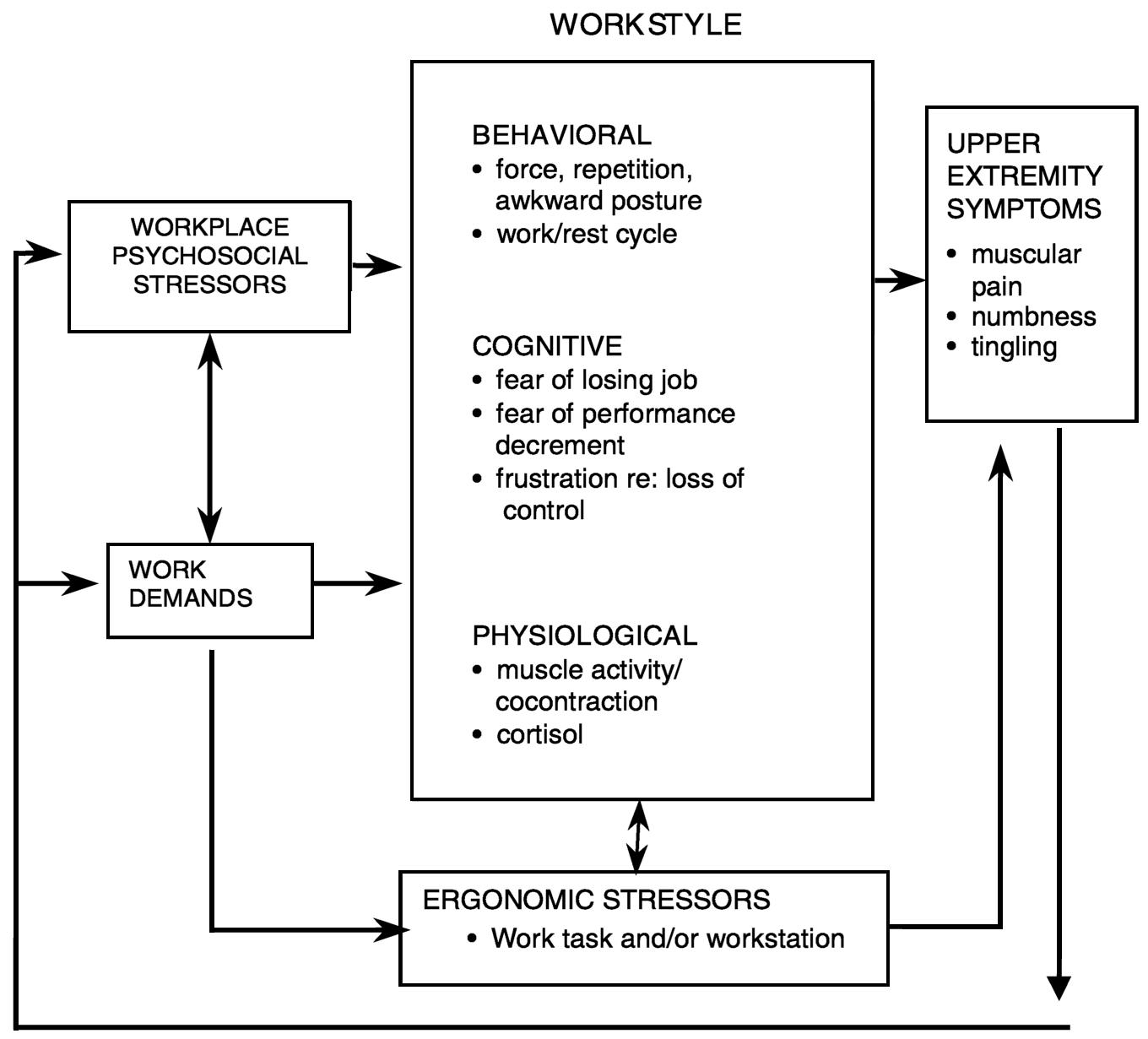
The workstyle model of job stress and musculoskeletal disorders (Feuerstein, 1996) was proposed to explain the relationship among job stress, ergonomic exposure, and work-related upper extremity disorders (illustrated in Figure 7.3). This model posits the importance of workstyle, that is, how an individual performs work in response to work demands. Workstyle consists of cognitive, behavioral, and physiological components that are consistent with the frequently reported multicomponent stress response. The behavioral component represents the overt manifestations of movement, posture, and activity. These behaviors can interact with workplace exposure to ergonomic risk factors, potentially increasing the risk of musculoskeletal pain (Macfarlane, Hunt, and Silman, 2000). The cognitive component refers to the worker's thoughts, feelings, appraisals, and evaluation of the success or failure of his or her responses to the work demands. The physiological component represents the biological changes that accompany the behavioral and cognitive reactions; these changes include increased muscle tension, tendon force, catecholamine or cortisol release, and stress-induced changes in immune function (Feuerstein, Huang, and Pransky, 1999). In a work environment perceived as stressful or demanding, individuals with a “high-risk” workstyle may continually exert excessive levels of effort to cope with such an environment, thereby exposing themselves to recurrent or chronic physiological, cognitive, and behavioral consequences of stress. This continuous arousal or reactivity can then set the stage for the development of symptoms,
Page 296
~ enlarge ~
FIGURE 7.3 Workstyle model. Adapted from Feuerstein (1996).
disorders, and, if this process persists, disability. High-risk workstyle has been shown to be associated with symptom severity, functional limitations, and work disability in individuals exposed to hand-intensive work (Feuerstein et al., 1997; Haufler, Feuerstein, and Huang, 2000).
While these models have some components in common, each has a unique contribution, such as a focus on work systems, the integration of work demands and unpaid domestic workload, the labeling of or attributions for symptoms experienced in the process of work, and the potential impact of workstyle. Also, each of these models hypothesizes a psycho-physiological substrate linking occupational stress to various health outcomes, in an effort to explain how job stress may affect work-related upper extremity disorders. The biological plausibility of a link between
Page 297
job stress and work-related upper extremity disorders is a critical element in determining the validity and credibility of such an association.
As with generic stress and health models, the majority of models attempting to describe how exposure to job stress may exert its effects on the physiology of upper extremity disorders focus their attention on musculoskeletal and neuroendocine pathways. These pathways are common to each of the models discussed above.
Potential Biological Pathways
One pathway by which exposure to occupational stress results in pain may be through changes in musculoskeletal activity or reactivity (Melin and Lundberg, 1997; Sjøgaard et al., 2000). Increases in muscular activity have been associated with tasks involving greater psychological demands (Melin and Lundberg, 1997). It has also been observed that muscle activation can be triggered by mental stress independent of physical effort (Melin and Lundberg, 1997). Spectral changes in forearm EMG, increased forearm tremor, and increased musculoskeletal discomfort have been observed in response to stress (Gomer et al., 1987). In the absence of a quantifiable increase in work demands, the perception of an increase in work demands is sufficient to increase forearm muscular tension during task performance (Arndt, 1987). When exposure to psychological stressors co-occurs with physical stressors, levels of EMG, blood pressure, and self-reported stress tend to be greater than in response to either exposure alone (Lundberg et al., 1994). An individual's psychological style, or how he or she characteristically responds to a challenge, has been related to the degree of muscle coactivation during completion of a simple motor task. This finding suggests that muscle activation can vary depending on how one characteristically perceives his or her environment (Glasscock et al., 1999). Taken as a whole, these studies suggest that while a perception of work as demanding is sufficient to increase musculoskeletal activity, the addition of physical exposures and a certain style or personality can further increase this activity. It is possible that such recurrent or chronic forearm muscle tension, secondary to psychosocial stressors, places sustained loads on the tendons in the wrist and elbow.
The fatiguing of low-level motor neurons in the upper trapezius and forearm, secondary to stress-induced muscle contraction, may also lead to musculoskeletal discomfort or injury via the fatiguing of small motor units (Westgaard and Bjorklund, 1987; Waersted, Bjorklund, and Westgaard, 1991; Westgaard and de Luca, 1999). This discomfort or injury may encourage alternative muscle recruitment and exposure to increased biomechanical risks (e.g., compromised postures, increased exertion of less suitable muscles) (Sjøgaard, 1996).
Page 298
Another potential pathway linking occupational stress to work-related upper extremity disorders may involve the biochemical consequences of the stress response, specifically the release of catecholamines and cortisol. Although serum levels of cortisol, epinephrine, and norepinephrine typically rise and fall throughout the day (Melin et al., 1999; Kirschbaum and Hellhammer, 1989), investigations of psychological and environmental correlates of stress hormone levels have determined that the sympathetic nervous system stimulates the release of these hormones differentially, based on the qualitative interpretation of the exposure to stress. On one hand, in general, it has been reported that the catecholamines appear to be related to the “positive” (i.e., the motivating or challenging) mental and physical demands of a stressor (Lundberg and Johansson, in press). On the other hand, cortisol appears to be generally related to negative aspects of the stress response, including negative emotions, the anticipation of negative consequences, and the perception of events as novel, uncontrollable, or unpredictable (Brantley et al., 1988; Van Eck et al., 1996; Kirschbaum and Hellhammer, 1989; Smyth et al., 1998).
The psychosocial characteristics of the work environment, such as task repetitiveness and rigid work arrangements, also appear to affect circulating catecholamines. Lundberg et al. (1993) reported that “deactivation” (i.e., recovery to baseline) of catecholamines, especially epinephrine, was slower after a repetitive data entry task than after a stimulating, self-learning task. Melin et al. (1999) reported that catecholamine levels in male and female assembly workers who were given the opportunity to work in autonomous groups and to influence their work pace decreased more rapidly after work than did catecholamine levels in individuals who worked in the “traditional” work organization (with fixed workstations and short, repetitive work cycles). In this study, the pattern of heightened reactivity and delayed recovery was observed in female workers in contrast to males and the pattern was even more marked in female workers who had children at home. Luecken et al. (1997) reported that 24-hour cortisol secretion was higher in women with at least one child living at home than in women without children. These cortisol levels were not affected by marital status or social support. These studies suggest a potential interaction among gender, paid and unpaid work demands, and stress reactivity and recovery (Melin et al., 1999; Lundberg and Frankenhaueser, 1999).
Chronic elicitation of the stress response, and the concomitant increases in catecholamine and cortisol release, could also directly affect the structure and function of muscles, tendons, and ligaments (Johnson et al., 1992). In addition to the direct effect these substances can exert on soft tissues, norepinephrine release can, theoretically, affect behavior, result-
Page 299
ing in an increased rate at which an individual performs tasks under stress. This, in turn, could result in more rapid and forceful responses during work tasks, thereby potentially increasing exposure to biomechanical risks.
Release of neurotransmitters may also play a role in the exacerbation of muscle pain. It has been observed that serotonin, which is released in response to stress (Stratakis, Gold, and Chrousos, 1995), potentiates the effects of endogenous pain mediators, such as bradykinin (Babenko et al., 1999). The infusion of both serotonin and bradykinin into the tibialis anterior muscle in humans has been associated with elevated pain intensity and prolonged pain in response to mechanical stimulation. This preliminary evidence suggests that exposure to stress could exert a nociceptor sensitization effect on muscle. In addition to the peripheral effect of stress on pain, data exist to support a direct role of the central nervous system (Fields and Basbaum, 1994). This may help explain how psychological processes, such as attention and emotion, influence pain and pain tolerance. In addition, the peripheral vasoconstriction induced by circulating catecholamines could further inhibit blood flow to a potentially compromised nerve in the case of carpal tunnel syndrome.
It has also been shown that exposure to stress exerts an inhibitory effect on inflammatory or immune responses (Chrousos and Gold, 1992). Glucocorticoids (cortisol) decrease the production of cytokines and other mediators of inflammation and inhibit the effects of these agents on target tissues. It is possible that the repeated elicitation of the stress response does not allow for the pain-sensitive tissues to recover following mechanical insult. Although the exact mechanism of injury differs from most work-related upper extremity disorders, it has recently been demonstrated that recovery from an oral puncture wound is significantly delayed following exposure to stress (Marucha, Kiecolt-Glaser, and Favagehi, 1998). Production of interleukin-1Ï, a proinflammatory cytokine important in cell recruitment and activation of fibroblasts, was noted to decrease by 68 percent following exposure to a stressor.
As indicated earlier, the potential pathways described above are speculative and represent a series of hypotheses that require rigorous scientific scrutiny. A critical element in validating these models is the determination of the time course between job stressor exposure, physiological changes, and symptom expression. Given the role that job stress plays in work-related symptoms in both the back and upper extremities, it is critical to identify the biobehavioral pathways underlying this effect, in order to understand how job stress can result in the symptom expressions characteristic of these disorders and in order to develop effective prevention and management strategies. The models reviewed need to be carefully tested using both laboratory and workplace studies.
Page 300
SUMMARY
Several distinct work-related psychosocial factors have been identified conceptually and can be measured reliably. They are associated with work-related low back pain and, less consistently, with work-related upper extremity disorders. The extent to which these variables interact with physical stressors remains to be determined. Also in terms of intervention, it is currently unclear what impact reducing physical stressors may have on psychological distress. The models of job stress presented in this chapter provide an important conceptual link between work-related psychosocial factors and work-related musculoskeletal disorders. They share many core features, providing additional (face) validity. These features include an association between stressful workplace conditions and a set of physiological responses, which, in turn, are associated with musculoskeletal disorders. These models need to be further validated.
While the precise biological mechanisms through which these physiological responses to stress lead to musculoskeletal disorders have not been fully elucidated, work to date suggests that musculoskeletal, neuroendocrine, and immunological pathways may be prominent. Further work in this area is needed to better define these biologic mechanisms. In addition, the knowledge currently available on psychosocial stress should be incorporated into interventions to ameliorate work-related musculoskeletal disorders.














