Page 15
assess the usefulness of this device in breast cancer screening or diagnosis.
For example, pathologists could examine cells taken from breast fluid for any abnormalities that might indicate breast cancer. Measurement of genetic or protein markers of breast cancer in such fluids is also being considered. The tests developed for breast fluid so far are not sensitive enough for breast cancer screening, and it is not clear how to intervene if abnormalities are found.
Researchers are now exploring a number of potential markers of breast cancer in blood, including proteins made by genes linked to cancer (oncogenes), growth factors, antibodies associated with tumors, and markers of blood vessel development, which is common in tumors. Researchers are also developing tests to detect the tumor cells themselves in blood. Although some of these markers and tests are sometimes used to monitor breast cancer progression, they are currently not sensitive enough to be useful for breast cancer screening.
In summary, the committee felt that studying the basic biology of breast cancer in order to develop molecular markers of breast cancer should be a high priority because molecular markers show promise for breast cancer screening, diagnosis, and prognosis. But these molecular markers require much more study and development before their usefulness in breast cancer detection can be evaluated.
BARRIERS TO THE DEVELOPMENT OF BREAST CANCER DETECTION TECHNOLOGY
The process of developing new technologies for breast cancer detection is quite complex, and several hurdles must be overcome to bring budding technologies to fruition. For example, sponsors of new technologies must have adequate resources for gathering the evidence necessary for FDA approval and for obtaining medical insurance coverage. Acceptance and use of the new technology by women and their doctors is also essential.
Research Resources
Once a new strategy for detecting cancer has been developed, a great deal of research is required to refine and test it. The complex nature of biomedical research requires enormous financial investments to undertake such testing and refinement.
Traditionally, private companies and other investors have played a major role in developing medical technology. More recently, resources are also being provided through newly developed government programs aimed at translating research findings into practical clinical applications. Other resources are shared
Page 16
through collaborations between industry, academia, and government agencies, such as the National Cancer Institute (NCI), the U.S. Army, NASA, and others. For example, some of these agencies joined forces to adapt defense technologies used in missile and target recognition for breast cancer detection. This collaboration also assisted in the development of the current commercially available CAD software for mammography. Other government agencies that fund research on breast cancer detection include the Department of Defense and the National Institute of Standards and Technology.
As noted previously, a major goal of breast cancer research is to identify molecular changes in the various breast cancer stages and precursors. Such markers could help doctors determine which abnormalities detected in screening are likely to become life-threatening and thus require treatment. The committee noted that this was an important area of research and recommended that the government continue to fund the development of breast cancer markers, as well as research aimed at determining the appropriate clinical use of such markers. The committee noted that such research should focus on use of markers in screening as well as diagnosis and treatment of breast cancer.
Two important resources for this research avenue are automated genetic or protein testing devices, and specimen banks, which collect pre-cancerous as well as cancerous breast tissues, breast fluid, and blood serum. By using these devices to analyze breast specimens, researchers hope to pinpoint differences in biochemistry or gene activity that could serve as molecular red flags for all types of breast tissue, ranging from normal to malignant.
But most breast specimen banks, which are run by academic, government, and/or private institutions, are inadequately supplied and staffed. Additional problems for researchers using the specimen banks may include issues related to informed consent, privacy, and patents.
Women who donate tissue or blood samples to a specimen bank usually sign a general consent form to allow future unspecified research, but occasionally some research projects on the specimens are not approved because the donors did not specifically consent to the given research project.
Some genetic research that is conducted on patient specimens could potentially indicate the donor's inherent genetic susceptibility to breast or other cancers. Some women choose not to donate specimens because they are concerned that the genetic information gathered from studies will not be kept private and could lead to discrimination by health insurers or others. The NCI recently proposed methods for protecting the identity of specimen donors, although these methods have not been put into place by all specimen banks.
Some specimen banks require a share of any profits from technologies that
Page 17
stem from research conducted on their samples. Companies that operate automated genetic or protein analytical devices may also claim patent rights on future products based on discoveries made using their technology. Such profit sharing and limitations on patent rights can be a financial disincentive for researchers conducting studies on molecular markers of breast cancer. In addition, the patents on the specific gene sequences used in some automatic genetic testing devices raise the price of such technology too high for many investigators.
Recognizing these problems, the committee recommended that breast cancer specimen banks be expanded and that researcher access to their samples be improved. They suggested that these specimen banks be a funding priority of the government and that the NCI devise and enforce strategies to make it easier to use patient samples in specimen banks. More specifically, the committee recommended that funding for specimen banks include support for the costs incurred by sharing specimen samples with collaborators, and that government-funded specimen banks not place excessive restrictions on the use of the specimen with regard to potential future patent rights. The committee also recommended that health care professionals and patient advocacy groups educate women about the importance of building specimen banks and encourage women to provide consent for research on specimen samples. However, stronger laws should also be passed at the national level to prevent genetic discrimination and to ensure the protection of patients who donate specimens for biomedical research.
In addition, the committee recommended that the government provide funding for the purchase and operation of automated technologies for the study and assessment of genetic and protein markers. Noting that the ability to draw conclusions from such research depends on innovative computerized data analysis techniques that are often lacking, the committee also recommended that the government fund the development of new approaches for analyzing large amounts of biological data.
FDA Regulations
In order for a medical device to be put into widespread clinical use, its manufacturer must provide the data needed for the Food and Drug Administration (FDA) to assess its safety and effectiveness. If the FDA determines that the device meets the appropriate standards, it approves the device for specific uses, such as breast cancer screening or diagnosis.
The FDA was originally established to evaluate the safety and effectiveness of drugs and other therapeutics. Since medical devices came under its domain in
Page 18
1976, the FDA has reviewed relatively few cancer screening technologies. This relative lack of precedent, combined with the complexity involved in assessing the safety and effectiveness of screening or diagnostic devices as opposed to a drug, has caused some difficulties in FDA's evaluation of new breast cancer detection devices, the committee noted. Drug treatments directly generate clear-cut endpoints that can be measured, such as the ability to reduce illness or to cause various side effects. In contrast, screening and diagnostic tests generate information, which is subject to interpretation and has only an indirect effect (mediated by subsequent therapy) on a person's health.
Depending on the nature of the medical device, FDA may require companies to show that it is equivalent to devices currently in use, or they may require more involved studies designed to show the safety and effectiveness of the device. The effectiveness of these devices is generally determined by their ability to accurately detect breast tumors. Such accuracy depends on two factors. One factor is sensitivity, or the ability to detect all tumors present. The other factor is specificity, which is the ability to rule out cancer in people who do not have a tumor.
Accuracy comparisons between two detection devices can be very difficult, however, because of the inherent variability of technologies like mammography. Mammograms can be quite difficult to interpret. Moreover, the recommendations of radiologists can vary depending on their level of experience and their tendency to order invasive biopsy procedures as a follow-up to moderately suspicious findings. As a result, different radiologists can interpret the same mammograms differently. Furthermore, subtle differences in the placement of the breast during the different testing procedures can alter the resulting images and how those images are interpreted. Such variability can obscure the differences between breast cancer detection devices.
In fact, accuracy comparisons were found to be meaningless when companies tried to show the equivalence of their new digital mammography devices to conventional film mammography devices. This led to a long and costly approval process, during which the FDA changed its requirements several times. For breast MRI, in contrast, the FDA only required companies to show that the breast coil used in the imaging was equivalent to MRI devices used to image other parts of the body. Little consideration was given to the accuracy of interpreting the images generated by the breast coil.
In order to reduce some of these difficulties in the future, the committee recommended that the FDA generate more consistent and clearly stated criteria for the approval of screening and diagnostic devices and tests, and thus speed the process. Given the complexity of assessing new technologies, the committee
Page 19
also suggested that FDA improve the external advisory panels that make recommendations to the FDA by including more experts in biostatistics, technology assessment, and epidemiology. For “next generation devices,” which have a technical adaptation to improve a device already in use, the committee recommended that advantages in addition to accuracy, such as patient comfort or ease of data acquisition and storage, be considered in the approval process. The committee also recommended a more coordinated approach for assessing new technologies when making decisions about both FDA approval and insurance coverage, as will be described in more detail in the next section.
Insurance Coverage
FDA approval is only the first hurdle that new technologies face once they have been developed. Although both the public and physicians commonly perceive that FDA approval means technologies “work” and should be reimbursed, health care coverage decisions are rarely that simple. FDA approval does not mean that a new device is better than its predecessor, or that it is useful for applications that have not been evaluated. For example, once devices have been FDA-approved for diagnostic tests and are thus available to general clinicians, health care providers may opt to use them for screening purposes, even though they have not been tested for that purpose (good diagnostic tests are not necessarily good screening tests). Ideally, breast screening devices would be deemed effective if they reduced the number of deaths from breast cancer. But FDA approval for screening technologies generally does not require such studies, which are very large and last for many years.
FDA approval simply allows the device to be sold. Ultimately, though, coverage decisions by insurers are likely to determine whether the technology becomes widely used. Developers of new technologies face a major challenge in seeking coverage and reimbursement. Insurers are increasingly basing their coverage and reimbursement rates on evidence of whether a procedure reduces illness or prolongs life. If such improved “patient outcomes” cannot be shown, then coverage may be legitimately denied and/or reimbursement may be low. For example, the additional costs of new technologies such as computer-assisted detection and digital mammography, which have not yet been definitively proven to improve patient outcome or detection accuracy, are currently not reimbursed.
However, technology development is often a process that continues after a given device enters the clinic. Most technologies that ultimately achieve widespread use go through successive stages of development, variation, and appraisal of actual experience in the market, as clinicians using new devices
Page 20
provide valuable feedback to manufacturers. Thus, assessment at early stages of development may not recognize the full potential of a new medical device. Because of this conundrum, the concept of “conditional coverage” has been explored as a potential way to allow new medical technologies to enter the market before making a final and definitive yes/no decision about coverage. Conditional coverage refers to limited, temporary coverage under specified conditions to allow for collection of data that can be used in determining the value of a technology and for setting a definitive coverage policy
The committee advocated a more coordinated approach than the current system for testing new screening technologies. They propose that FDA approval and insurance coverage decisions for new screening tests should depend on evidence of improved patient outcome from clinical trials. These studies should be designed, approved, and conducted with input and support from FDA, NCI, the Healthcare Financing Administration (HCFA, which oversees Medicare), private insurers, and breast cancer advocacy groups. The committee suggested that if a new device already approved for breast cancer diagnosis shows promise for accurate screening, an “investigational device exemption” (which allows the device to be used in clinical trials for FDA approval) should be granted for this use. Conditional coverage should then be provided within the context of approved clinical trials. Trial data should be reviewed at appropriate intervals, and the results would determine whether FDA grants the device approval for screening, and also whether coverage is extended to general use (outside of clinical trials).
This coordinated assessment approach would help prevent new technologies approved only for diagnosis from being widely adopted as screening tools before their effectiveness for screening is proven. At the same time, support by NCI and medical insurers would make it easier for technology sponsors to conduct the clinical studies needed to assess whether a new breast cancer screening technology reduces breast cancer deaths. For example, health insurers would cover the costs of performing tests in approved clinical studies, and the NCI and the technology manufacturers would share the other costs involved with the studies. Participation by private insurers would be particularly important for assessing new technologies intended for use in younger women who are not yet eligible for Medicare coverage. While this expense may initially seem burdensome to private insurers, the cost of providing tests within a clinical trial would be much less than the costs associated with broad adoption by the public (and the associated pressure to provide coverage) in the absence of experimental evidence for improved clinical outcome.
The committee also recommended that NCI create a permanent system for
Page 21
testing the effectiveness of new technologies for early cancer detection. The committee noted that the NCI's Breast Cancer Surveillance Consortium or the American College of Radiology Imaging Network, with their extensive databases, tissue samples and other resources, may provide a useful model or platform for such an undertaking.
Dissemination
Once a new technology receives FDA approval and insurers agree to reimburse the costs associated with its use, adoption of this technology will ultimately depend on whether consumers and their health care providers fred it acceptable, necessary, affordable, and accessible. Experience with x-ray film mammography screening suggests that these factors can be important barriers to a new technology being used by the public. Only 69 percent of women 50 years old and over reported having a recent mammogram in 1998 ( figure 8 and figure 9). In addition, not all women who receive mammograms do so at the recommended intervals. One study indicated that only about a quarter of women are screened at the intervals appropriate for their age.
A number of factors that hinder mammography screening are likely to also play a role in limiting the adoption of new breast cancer screening technologies. The most important factor is whether a woman's doctor recommends mammography screening, even though a physician referral is not needed for a screening exam. A significant fraction of women between the ages of 50 and 75 do not receive recommendations for such screening from their doctors.
A number of scientific and professional organizations provide guidelines for breast cancer screening, but these guidelines lack consistency as to when to begin screening, how often to screen, and when to discontinue screening. The guidelines have also changed over time as the results of new studies on screening mammography were published.
There currently is no universal consensus on the value of screening for women under 50 or over 70. The committee noted the lack of studies on whether screening mammography benefits women over the age of 70, despite the fact that breast cancer is most prevalent among women in this age group. The committee recommended that the NCI, through the American College of Radiology Imaging Network or the Breast Cancer Surveillance Consortium, sponsor a large clinical study to define more accurately the benefits and risks of screening mammography in women over the age of 70.
The committee also recommended that NCI sponsor large randomized clinical studies every 10–15 years to reassess the benefits of screening techniques. These studies would compare two technologies in current use, and
Page 22
assess how each reduces the number of breast cancer deaths among the women who undergo the screening. This would address the continually evolving nature of breast cancer screening and treatment and detect any changes in benefits that would affect screening recommendations.
A lack of health insurance that reimburses the costs of mammography clearly hinders this form of screening. To overcome this barrier, the 1990 Breast and Cervical Cancer Mortality Prevention Act mandated the establishment of the National Breast and Cervical Cancer Early Detection Program. This program, which is run by the Centers for Disease Control and Prevention (CDC), targets women who lack health insurance, with a focus on encouraging screening at recommended intervals. Approximately 60 percent of the budget is allocated for screening services, with the remaining 40 percent devoted to education and
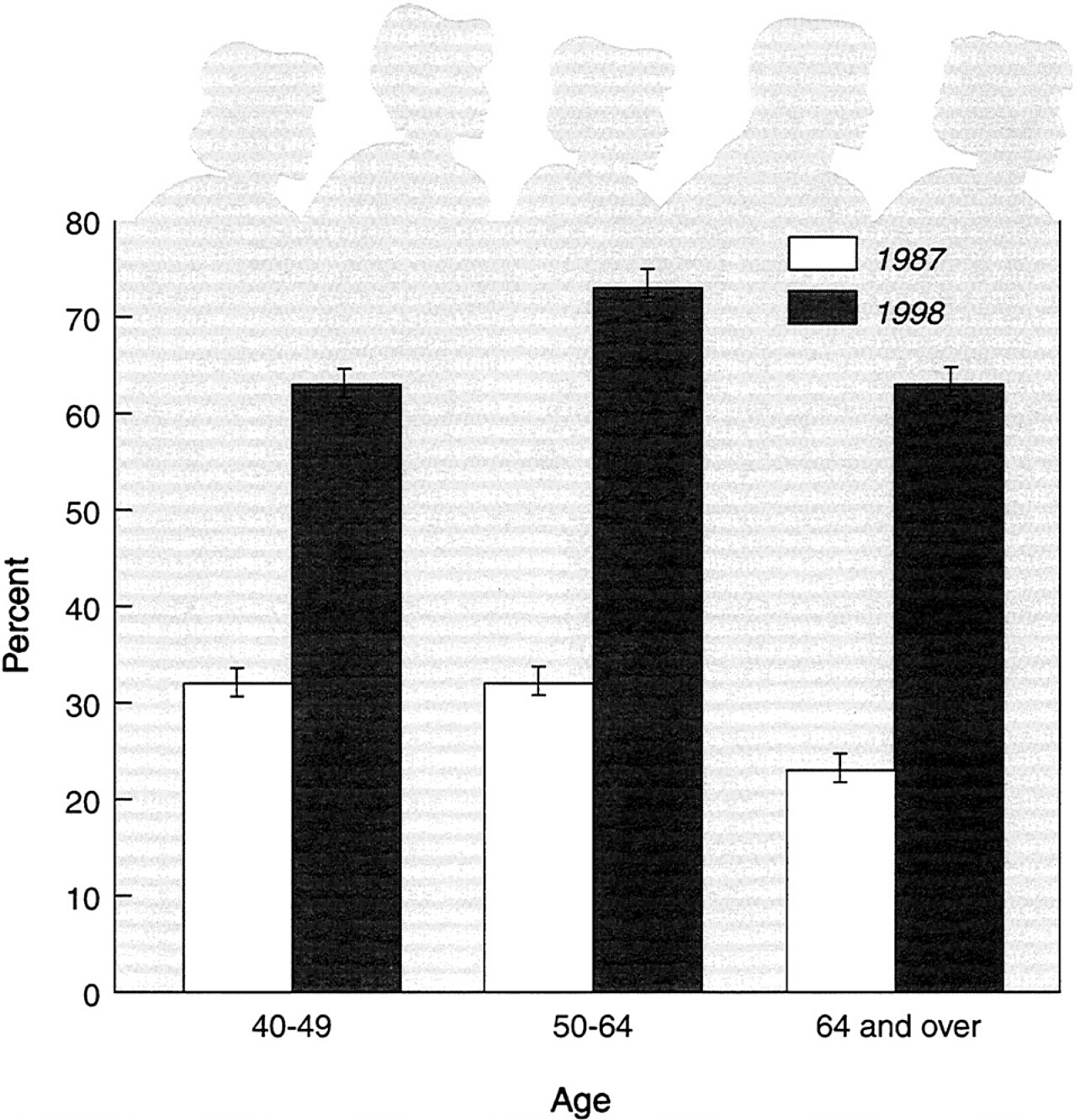
~ enlarge ~
FIGURE 8 Use of mammography by women 40 years of age and over, 1987 and 1998. The percent of women having a mammogram within the last two years. SOURCE: U.S. Department of Health and Human Services, 2000. ( www.cdc.gov/nchs/products/pubs/jubd/hus/hus.htm). Data are based on the National Vital Statistics System, National Center for Health Statistics.
Page 23
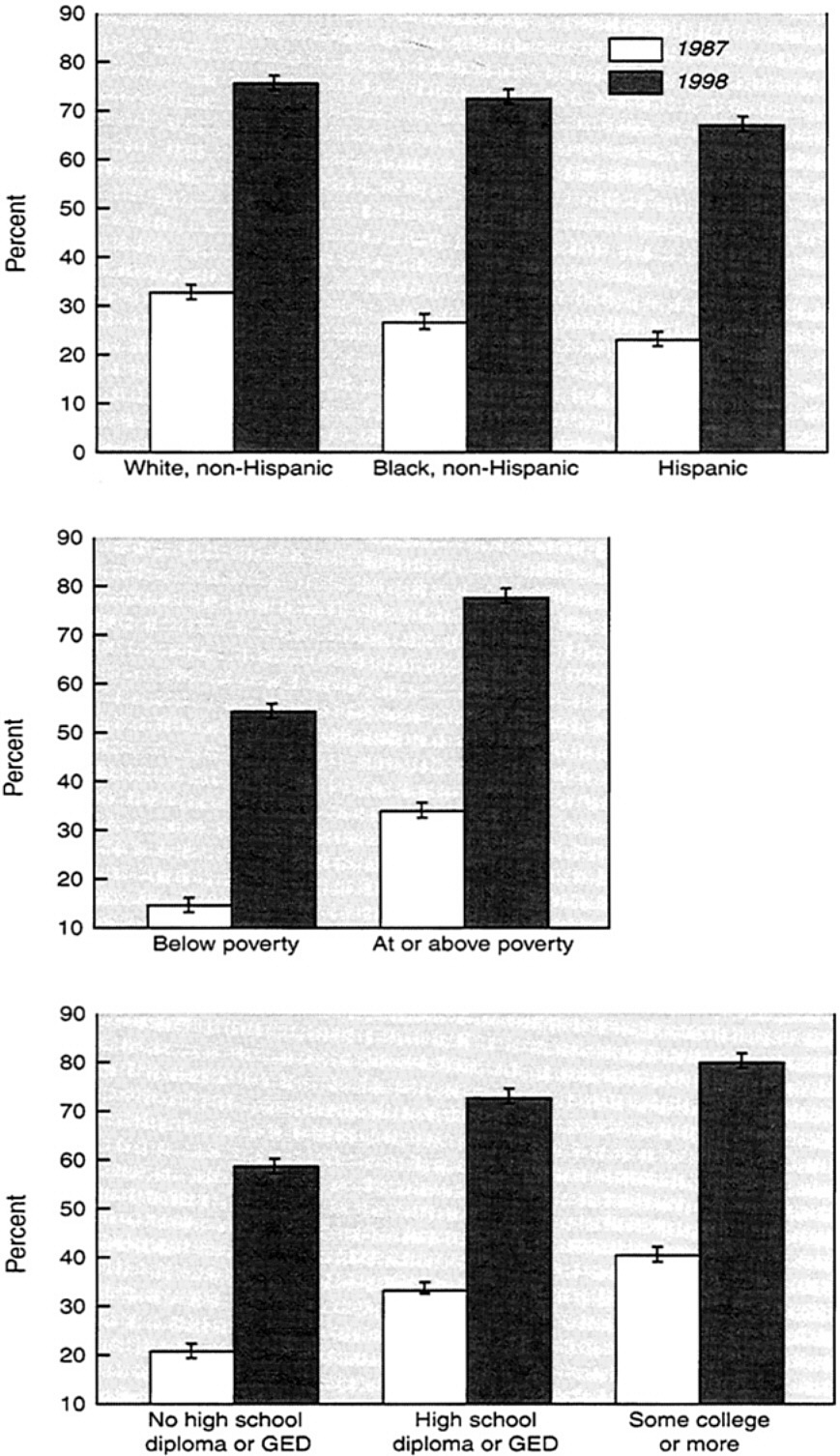
~ enlarge ~
FIGURE 9 Use of mammography by women aged 50 to 64, according to various attributes (1987, 1998). Percent of women having a mammogram in the last two years. SOURCE: U.S. Department of Health and Human Services, 2000. ( www.cdc.gov/nchs/products/pubs/jubd/hus/hus.htm). Data are based on the National Vital Statistics System, National Center for Health Statistics.
outreach. Currently, only 12–15% of eligible women are served by the program, largely because of a lack of adequate resources. The committee recommended that the CDC screening program be expanded to reach more eligible women.
The services available through the program include mammography and such follow-up diagnostic procedures as ultrasound, fine needle aspiration, and breast biopsy. (One or more of these procedures may be needed if a mammogram shows a suspicious finding.) Federal legislation recently provided funding through Medicaid for treatment of cancers detected through the program. But Medicaid funds are provided by both federal and state governments. (A certain amount of federal Medicaid dollars is allocated for each
Page 24
Medicaid dollar spent by a state government for the program.) The committee urged states to provide Medicaid funds for treatment of cancers diagnosed through this early cancer detection program.
Another possible barrier to mammography screening is that the capacity of screening facilities may not be keeping pace with the increasing demand for mammography services. Because our nation's population is aging, the number of women eligible for screening in this country is increasing each year. A recent survey of the Society of Breast Imaging indicated that waiting times for screening appointments have been increasing over the last two years.
There are anecdotal reports that low reimbursement rates for x-ray mammography may have prompted some facilities to close or decrease their volume of breast cancer screening. Radiologists have argued that the reimbursement for mammography is too low for the time, effort, and interpretive skill it requires compared to other imaging procedures. In addition, the Mammography Quality Standards Act (MQSA), which was enacted in 1994, may boost the cost of providing mammography services. MQSA requires all mammography facilities to meet minimum quality standards for equipment and health care professionals, and requires extensive records to show they meet such standards. Since its inception, the quality of mammograms has improved. But this regulation, which is unique to mammography, also increases costs to facilities, and MQSA does not require reimbursement levels to cover those costs.
The committee recommended that the Heath Care Financing Administration (HCFA) analyze the current Medicare and Medicaid reimbursement rates for x-ray mammography. This analysis should include a comparison with other radiological procedures to determine whether they adequately cover the total costs of providing the x-ray mammography.
Concerns have also been raised about a possible shortage of radiologists and technologists trained in mammography, although to date these claims have not been substantiated with careful studies. The number of mammography training fellowships for radiologists has decreased by about one-quarter over the last five years. Radiologists may be deterred from specializing in mammography by the relatively low reimbursement for mammography, combined with the rising number of malpractice suits linked to breast cancer screening, although these connections have not been documented. The committee recommended that the Health Resources and Services Administration (HRSA) analyze trends in specialty training for breast cancer screening among radiologists and technologists, as well as try to pinpoint the factors affecting decisions to enter or remain in the field. If trends suggest there will be a shortage of trained experts, HSRA should seek input from professional societies such as the American










