Page 6
THEORY AND PRINCIPLES OF CANCER SCREENING AND DIAGNOSIS
Screening tests are performed on people who have no physical signs of the disease being tested for. The goal of cancer screening is to detect tumors at an early enough stage so that they will be curable when treated. It is important to keep in mind, however, that even if a screening device is effective at detecting small tumors, it may not detect them early enough to make a difference in reducing the number of cancer deaths. For example, chest x-ray screening for lung cancer did not decrease the number of lung cancer deaths among the people screened for the disease, even though tumors identified by screening were smaller than those found in the absence of screening. By the time lung tumors were detectable on an x-ray, they usually had already advanced to a stage that was incurable by spreading beyond the lung.
According to guidelines established by the World Health Organization, screening should be used only if it provides a net benefit to those screened. For example, a test should reduce the number of deaths from a particular condition without excessively harming people without the condition. But it can be difficult to assess the real benefit of any cancer screening technology or program.
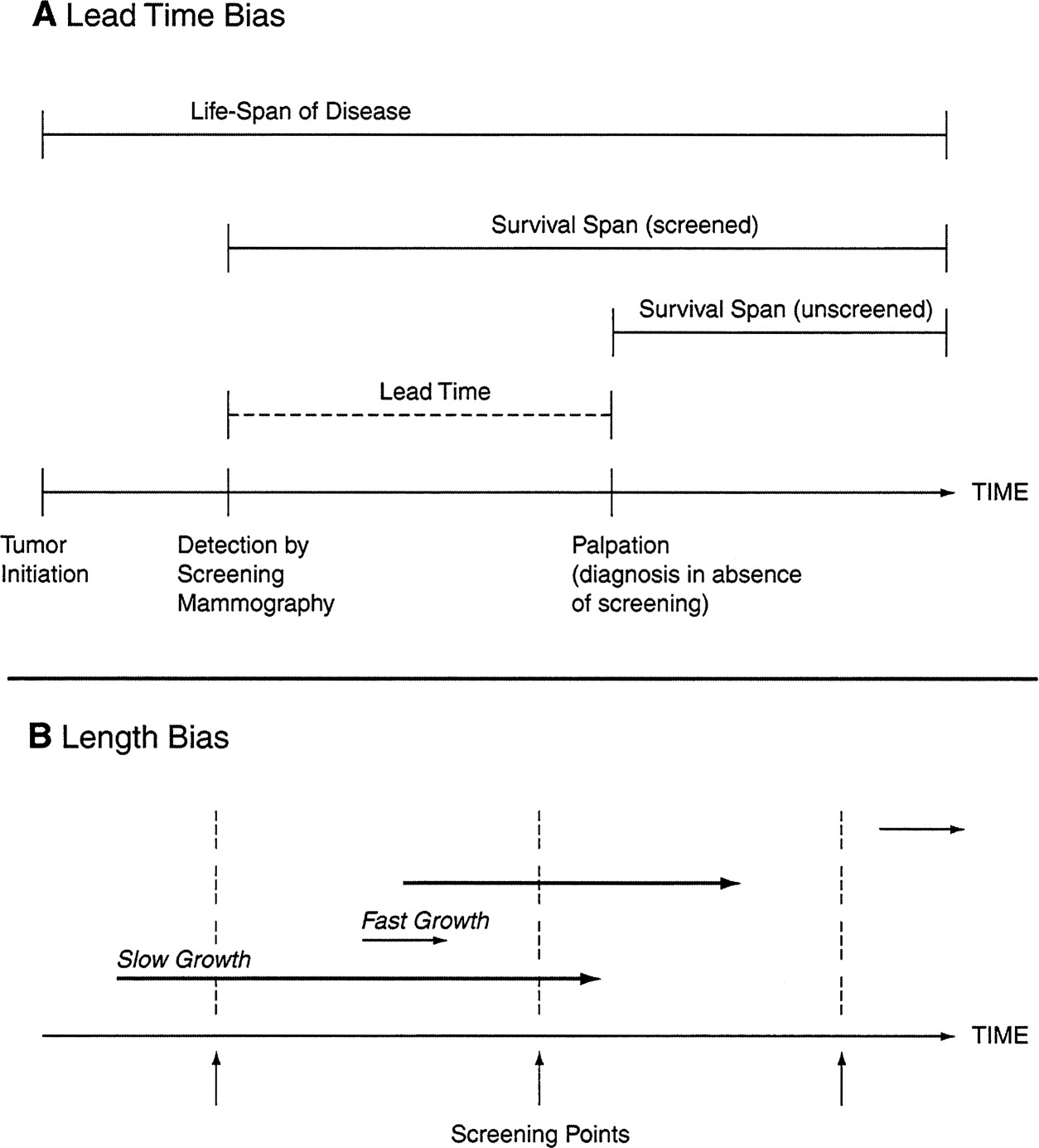
It is often assumed that any improved survival time observed among people who undergo a particular screening program is beneficial, but this may not actually be true ( Figure 4). For example, a woman who dies two years after she finds a lump in her breast may not necessarily have survived longer if she had undergone mammogram screening two years earlier. Some aggressive cancer cells can spread beyond the breast even when the tumor is relatively small and undetectable. Similarly, it is impossible to know whether a woman who survives four years after her cancer is detected with mammography has truly gained an extension of her life. It may be that she has simply been aware of her diagnosis for a longer period of time. Because of this phenomenon of “lead-time bias,” measuring the length of survival after diagnosis is not a valid way of assessing the effectiveness of a screening test.
Screening tests done on a regular basis are also more likely to detect a disproportionate number of people with slow-growing tumors, a phenomenon known as “length bias.” This is because a cancer that takes many years to reach a size that can be felt on a breast exam is more likely be detected as a smaller tumor by regular mammography screening than one that grows to the same size in a much shorter period of time. If an aggressive, fast-growing tumor is more likely to become life-threatening than a slow-growing tumor, then many women whose tumors were identified through a screening program will inherently have
Page 7
a more favorable outcome following treatment. Consequently, a higher survival rate among women whose tumors are detected by breast cancer screening may not actually result from the screening itself. That is, the tumors may have responded well to treatment even if they weren't detected until they grew large enough to be felt on a breast exam.
Additional difficulties encountered in assessing breast cancer screening programs include selection bias and overdiagnosis. Selection bias assumes that women who are at higher risk for breast cancer will be more likely to participate
~ enlarge ~
FIGURE 4 Lead-time (A) and length (B) bias. In part B, the length of the arrows represents the time required for the tumor to reach a palpable size. For a more detailed description of lead-time and length bias, see the accompanying text in the summary.
Page 8
in breast cancer screening studies and will comply more with the recommended guidelines for the screening. Since such screening may be more beneficial and cost-effective in high-risk populations than in the general population, a selection bias may result in overestimating the value of using a screening program for the general population.
“Overdiagnosis” results from labeling some abnormalities as cancer or pre-cancer when in fact these abnormalities may never have progressed to a life-threatening disease if left undetected and untreated. In such cases, some of the “cures” following early detection may not be real. In addition, overdiagnosis prompts some patients to undergo expensive, uncomfortable, and potentially damaging treatments that may not be necessary.
The best way to counter some of these problems inherent in assessing the value of cancer screening techniques is to conduct large studies in which participants are randomly selected to receive a screening test. Researchers then need to study these participants over a long enough period of time to determine whether the screening test reduces the number of deaths from breast cancer. Such studies may require hundreds of thousands of women and take 10 to 15 years to complete. (For example, over the course of nearly 20 years, approximately 500,000 women participated in 8 studies to assess the benefits of mammography screening, among which about 2,500 deaths from breast cancer occurred.) Because of the extensive costs and time involved, researchers have yet to conduct the large, randomized screening studies required to assess the value of using new breast cancer screening technologies.
Once a breast abnormality has been detected by screening mammography or physical exam, the abnormality must be diagnosed as benign or malignant using additional imaging techniques and/or biopsy and microscopic examination of the tissue. Many new breast cancer detection technologies are being studied as diagnostic tools, often as an addition to diagnostic mammography, in the hopes of avoiding unnecessary biopsy of benign abnormalities.
The assessment of diagnostic technologies differs from that of screening technologies. Diagnostic studies primarily involve an evaluation of a test's accuracy in determining which women with a suspicious breast abnormality have cancer and which do not. Such studies do not examine differences in clinical endpoints such as death from breast cancer. Consequently, studies to evaluate diagnostic technologies are usually much smaller, shorter in duration, and less expensive.



