CHAPTER THREE
Embryonic Stem Cells
Embryonic stem cells (ESCs) are found in the inner cell mass of the human blastocyst, an early stage of the developing embryo lasting from the 4th to 7th day after fertilization. In normal embryonic development, they disappear after the 7th day, and begin to form the three embryonic tissue layers. ESCs extracted from the inner cell mass during the blastocyst stage, however, can be cultured in the laboratory and under the right conditions will proliferate indefinitely. ESCs growing in this undifferentiated state retain the potential to differentiate into cells of all three embryonic tissue layers. Research involving human ESCs is at the center of the ethical debate about stem cell use and potential in regenerative medicine. Embryos from which ESCs are extracted are destroyed in the process.
Several scientific questions are important when considering the potential of stem cells for use in regenerative medicine and the policy and ethical issues that arise:
-
What properties of ESCs have promise for regenerative medicine?
-
What direct evidence supports ESCs’ effective use in regenerative medicine?
-
What obstacles and risks are associated with the use of ESCs in regenerative medicine?
PROPERTIES OF ESCs IMPORTANT FOR REGENERATIVE MEDICINE
Human ESCs were successfully grown in the laboratory for the first time in 1998 (Thompson et al., 1998). Under appropriate culture conditions, ESCs have demonstrated a remarkable ability to self-renew continuously, that is, to produce more cells like themselves that are multipotent. As indicated at the workshop by Thomas Okarma and Ron McKay, ESC lines established from single cells have been demonstrated to proliferate through 300-400 population-doubling cycles. Human ESCs that have been propagated for more than 2 years also demonstrate a stable and normal complement of chromosomes, in contrast to the unstable and abnormal complement of embryonic cancer cell lines used in the past to study early stages of embryonic development. Careful monitoring of the aging ESC lines will be needed to evaluate the significance of genetic changes that are expected to occur over time.
Because human ESCs have only recently become available for research, most of what is known about ESCs comes from studies in the mouse, which, as noted in Chapter 2, cannot be presumed to provide definitive evidence of the capabilities of human cells.
Nevertheless, ESCs derived from mouse blastocysts have been studied for 2 decades and provide a critical baseline of knowledge about the biology and cultivation of these cells (Torres, 1998; Wobus and Boheler, 1999). The factors that permit the mouse ESC to continue replicating in the laboratory without differentiation and methods to trigger differentiation into different cell types that exhibit normal function have been actively explored. Among the types of cells derived from cultured mouse ESCs are fat cells, various brain and nervous system cells, insulin-producing cells of the pancreas, bone cells, hematopoietic cells, yolk sac, endothelial cells, primitive endodermal cells, and smooth and striated muscle cells, including cardiomyocytes—heart muscle cells (Odorico et al., 2001).
Experience with mouse ESCs has provided clues to methods for culturing human ESCs and leading them to differentiate. Mouse ESCs
will proliferate in an undifferentiated state in the presence of a biochemical called leukemia inhibitory factor (LIF), but the culture conditions required to keep human ESCs from differentiating include growing them in petri dishes on a layer of mouse embryonic fibroblasts (referred to as “feeder cells”) in a medium containing serum from cows. The feeder cells are inactivated, so they are not dividing and expanding, but they produce growth factors that sustain the ESCs. The mechanism of how feeder cells maintain the proliferation of undifferentiated ESCs is unknown. Such in vitro culturing presents certain theoretical hazards to the use of stem cells for regenerative medicine, such as the spread of viruses and other infectious agents not normally found in humans. When removed from feeder cells and grown in suspension (in liquid), human ESCs form aggregated balls of cells called “embryonic bodies,” which have been reported to give rise to a multiplicity of cell types representing all three layers of embryonic tissue development (Itskovitz-Eldor et al., 2000; Reubinoff et al., 2000; Schuldiner et al., 2000). Evidence of the differentiation in culture includes detection of the products of genes associated with different cell types and in some cases by the characteristic shapes that are peculiar to different cell types. Cells derived from human embryonic bodies include “rhythmically contracting cardiomyocytes, pigmented and nonpigmented epithelial cells, and neural cells displaying an exuberant outgrowth of axons and dendrites” (Odorico et al., 2001). In other experiments, cells arising from human ESCs have been reported to express genes associated with liver and pancreas function (Schuldiner et al., 2000). Human ESCs grown in coculture with mouse bone marrow stromal cells have been reported to produce colonies of human hematopoietic precursors and ultimately cells from the blood (Kaufman et al., 1999).
Further evidence of the multipotent capability of human ESCs is based on studies in an in vivo setting. Human ESCs injected into mice form a type of benign tumor called a teratoma that is made up of tissues from all three embryonic layers. The tissues that arise in the tumor are often advanced, organized, and complex, and include teeth, gut, hair follicles, skin, epithelium, muscle, bone, cartilage, lung tissue, and neural
cells (Thompson et al., 1998). The experiments showed the capability of ESCs to produce a variety of tissues, but the results also highlight the complexity of the biological “program” of tissue development that can unfold in different biological environments. These results also emphasize the abnormal, potentially neoplastic potential of ESCs when placed into unnatural environments.
Major questions remain about the genetic or environmental factors in the body that control the fate of ESCs and about the importance of different factors during various stages of cell differentiation. Even on the basis of the limited findings, however, the ability to grow human ESCs in vitro and to have them differentiate in the laboratory makes them an important and unique tool with which to conduct the basic research that is critical for the foundation of future regenerative therapies. It has been possible, for example, to create a lineage of mouse ESCs that generate neural cell precursors (Li et al., 1998). Studies of the genes turned on and off as cells begin to differentiate, which are already under way with ESCs, will permit a better understanding of the genetic controls important in tissue differentiation (Duncan et al., 1998). In vitro studies of ESCs also provide an opportunity to explore the role of biochemicals produced in the normal cellular environment that induce stem cells to differentiate, to migrate to a site needing repair, and to assimilate into tissues (Schuldiner et al., 2000).
EVIDENCE SUPPORTING THE POTENTIAL OF ESCS FOR USE IN REGENERATIVE MEDICINE
At the workshop, James Thomson and Thomas Okarma suggested that human ESCs will someday provide a potentially unlimited source of cells, differentiated in vitro, for transplantation therapies involving the liver, nervous system, and pancreas. Irving Weissman alluded to the possible use of ESCs to enhance the success of whole-organ transplantation. If HSCs derived from human ESCs could be successfully transplanted into the blood system of a transplant recipient (by using immunosuppressive drugs), any further implant tissue (say kidney or pancreas)
developed with the same ESCs would not, in theory, be rejected by the recipient because the immune cells produced in the recipient’s blood by the HSCs would see the implant tissue as “self”.
But that is a long way off, as Marcus Grompe noted, in as much as no one has yet demonstrated any in vivo reconstitution of an organ’s function in either humans or experimental animals with cells derived from human ESCs. Moreover, ESCs in tissue culture give rise to a mixture of cell types all at once, and biochemical, tissue-culture, and molecular-biology techniques to control and limit differentiation require much further investigation.
Because human ESCs have only recently become available for research, and because public funding for such research has been limited, studies of how well ESCs or their differentiated tissues perform physiologic functions has been largely conducted with mouse models. Ron McKay described progress made in coaxing the in vitro differentiation of human ESCs into insulin-producing cells that might be useful in treating diabetes, but he also noted that studies have already been conducted with analogous mouse cells transplanted into mice that have diabetes and that partial restoration of insulin regulation was observed (Lumelsky et al., 2001). Other studies have demonstrated that mouse ESCs can be successfully transplanted into rodents that have Parkinson’s disease symptoms and partially relieve these symptoms (Studer et al., 1998). Similarly, studies suggest that mouse ESCs can be transplanted into animals that have spinal-cord injuries and partially restore neural function (McDonald et al., 1999).
Those studies provide promise, but not definitive evidence, that similar treatments could be effective in humans. Human ESCs will need to be tested in primate models, such as those for Parkinson’s disease and diabetes mellitus in the rhesus monkey. Methods for transplanting ESCs need to be developed, as do means of establishing whether the cells develop and function properly after transplantation. In some cases, it will be important to ensure that the transplanted cells or tissues are incorporated and positioned properly relative to existing tissues, such as in heart and neural tissue; the three-dimensional, cell-to-cell interactions
will play important roles in the functioning of an organ. Other cells, like pancreatic islet cells, or hematopoietic cells, will require less complex incorporation.
Also, the large-scale propagation of human ESCs in culture will require that they can be grown without feeder cells (Odorico et al., 2001). Research is needed to elucidate the mechanisms of feeder cells in repressing differentiation and to find alternatives to them, at the same time eliminating the potential that an animal virus from the feeder cells might be transferred to the ESCs.
Finally, it was noted earlier that the chromosomes of human ESCs have been shown to be stable in tissue culture. This does not mean however, that ESC lines will not be subject to the random mutations that affect all cell lines as they age. In cells from humans and other animals, approximately one mutation occurs every time a cell divides. A cell that has divided 200 times in culture therefore can be expected to harbor approximately 200 different mutations (Kunkel and Bebeneck, 2000). So far, there have been no studies published about the changes that may have occurred in existing stem cell lines. Vigilant monitoring of the integrity of existing cell lines is essential to allow understanding of the impact of long-term culture, and new stem cell lines may need to be developed in the future.
Obstacles and Risks Associated with the Use of ESCs
In addition to demonstrating the functional effectiveness of ESC transplants, it is necessary to identify and minimize, or eliminate, the risks that ESCs might pose. Two identifiable risks are tumor formation and immune rejection. As noted earlier, human ESCs injected into mice can produce a benign tumor made up of diverse tissues; this response is believed to be related to the multipotency of the undifferentiated cells in an in vivo environment. However, in a small number of short-term studies in mice, human ESCs that have been allowed to begin the process of differentiation before transplantation have not resulted in
significant tumor formation (Odorico et al., 2001). Obviously, this is a critical problem to understand and control.
It is too early to tell, therefore, whether it will be appropriate to use human ESCs directly in regenerative medicine. A great deal obviously must be elucidated about how the body controls the differentiation of stem cells, and this has yet to be reliably reproduced in vitro. Also, the behavior of ESCs implanted in a specific organ has not been well studied. It might someday be possible to add growth factors with a transplant to stimulate the production of a particular cell type or multiple cell types. “Inducer tissues” that interact with stem cells might be cotransplanted with ESCs to achieve a similar result. Those possibilities are still in experimental investigation.
In another respect, the possible problems associated with ESC transplantation are common to all transplantation, such as the risk of infection and the risk of tissue rejection. As discussed in Chapter 2, rejection is a serious obstacle to successful transplantation of stem cells and tissues derived from them. It has been suggested that ESCs provoke less of an immune reaction than a whole-organ transplant, but it is unclear whether that will be true of the regenerated tissues derived from ESCs. Some types of cells (such as dendritic cells, immune system cells, and vascular endothelial cells) carry more of the histocompatibility antigens that provoke immune reactions than other cells. Those types are present in the tissues of whole organs; they connect an organ with the bloodstream and nervous system. However, tissue derived in vitro from ESCs, such as liver tissue, would not contain such cells and therefore would theoretically trigger a milder immune response; this assumes that techniques for controlling differentiation of ESCs will be available. In addition, the liver cells likely would not be devoid of all surface antigens, and so, in the absence of other techniques to reduce transplant rejection, the use of immunosuppressive drugs will still have to be used, with attendant risks of infection and toxicity.
Although difficult to conceive, the creation of a very large number of ESC lines might be one way to obtain a diversity of cells that could
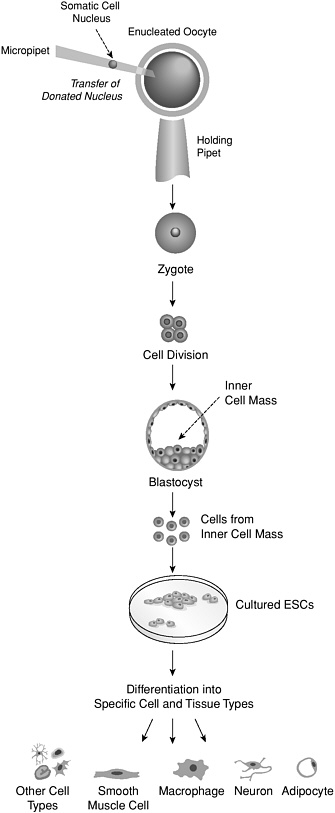
theoretically increase the chances of matching the histocompatibility antigens of a transplant recipient. It has also been suggested that ESCs could be made less reactive by using genetic engineering to eliminate or introduce the presence of surface antigens on them (Odorico, 2001). An exact genetic match between a transplant recipient and tissue generated from ESCs could also, in theory, be achieved by using somatic cell nuclear transfer to create histocompatible ESCs (Figure 4). Cells created with this technique would overcome the problem of immune rejection. However, it might to not be appropriate to transplant such cells in a person with a genetically based disease, since the cells would carry the same genetic information. In any case, an understanding of how to prevent rejection of transplanted cells is fundamental to their becoming useful for regenerative medicine and represents one of the greatest challenges for research in this field.