2
AEA SILVER II™ Technology Process
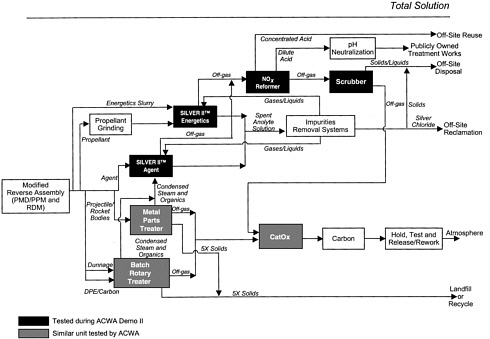
The AEA SILVER II™ technology is based on the highly oxidizing nature of silver II ions (Ag2+), which are generated by passing an electric current through a solution of silver nitrate and nitric acid in a standard electrochemical cell. Figure 2–1 shows a block diagram of the AEA SILVER II™ total system solution. The first step in the system is a modified reverse-assembly process in which the energetics, agents, and metal parts are separated. The energetic materials are then reduced in size to less than one-eighth-inch particles before further treatment. In the case of the high-pressure washout of bursters, the particle size was reduced to less than 500 microns (µm). The agent and energetic components are destroyed in separate electrochemical processing units. Metal parts and dunnage are thermally treated in a metal parts treater. The solid, liquid, and gaseous effluents are separated and treated to remove reagents so that they can be recycled and to clean the emissions prior to discharge. The AEA Demo II program tested two SILVER II™ processing plants. One used a 2 kW electrochemical processing unit and the other used a 12 kW unit. The 2 kW SILVER II™ plant was used to test the destruction of agents and agent simulants, and the 12 kW plant was used to test destruction of energetic materials and agent simulants.
The 2 kW SILVER II™ system was at the Edgewood Chemical and Biological Center (ECBC) facility and the 12 kW SILVER II™ system was at the Aberdeen Test Center Firebox. More detailed descriptions of the total system solution and the unit operations can be found in the original NRC ACW I report (NRC, 1999).
DESCRIPTION OF THE SYSTEMS
2 kW SILVER II™ System
The 2 kW SILVER II™ demonstration unit installed in the toxic chamber at ECBC (Figure 2–2) consists of an Imperial Chemical Industries (ICI) FM-21 electrochemical cell with a single in-cell flow channel and a single electrode pair. This unit and the associated gas treatment system are not as complex as the 12 kW unit, which is described in detail later in this chapter.
The system was designed to validate the capability of the SILVER II™ process to destroy chemical agents VX, GB (both are nerve agents), and HD (distilled mustard). It was also intended to demonstrate the effective destruction of agent simulants chloroethyl ethyl sulfide (CEES) and dimethyl methylphosphonate (DMMP) for comparison with results from the 12 kW system.
This system was also tested for its ability to handle and treat the silver chloride (AgCl) formed during the processing of HD and CEES to a 5X level. The chlorinated feeds from which the silver chloride was formed were either agents or agent simulants, which were introduced into a premix vessel from which they were metered into the anolyte vessel. A single hydrocyclone was installed in the anolyte circuit to remove the solids and prevent their accumulation in the electrochemical cell. During part of the test, the hydrocyclone discharge was sent back to the anolyte vessel; the rest of the time, discharges were sent to the settling/collection vessel. AgCl crystals that were removed from the recirculating anolyte circuit were later transferred to a 5X treatment unit.
The silver chloride 5X treatment unit was an electrically heated oven with forced-air circulation to prevent cold spots. The temperature of the silver chloride was measured with a thermocouple mounted in the unit. The vent air from the unit containing nitric acid vapors was passed through a condenser prior to flowing into the main plant off-gas system.
12 kW SILVER II™ System
The full-scale unit for the treatment of energetics and agent simulant operates at 300 to 750 kW; thus, the 12 kW test unit is only 2 to 4 percent of the size of the full-scale unit. The process flow diagram for the test unit is shown in Figure 2–3. The 12 kW test unit, which was operated by Army personnel from the Aberdeen Test Center, is a larger
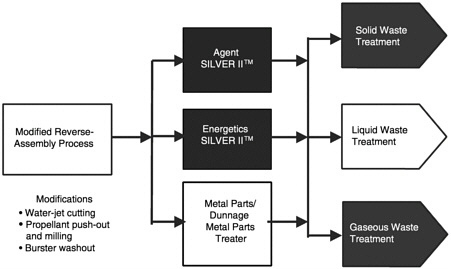
FIGURE 2–1 AEA SILVER II™ total system solution. Solid boxes represent unit operations for demonstration. SOURCE: Adapted from AEA (2000).
version of the 2 kW unit and consists of an ICI titanium electrolytic cell (2 V/6,000 A) with a Nafion™ membrane. The pressure drop across the cell is 0.5 bar during operation. The solution in the anode compartment circulates around a closed loop through a titanium anolyte vessel that has a volume of 600 L. The solution in the cathode compartment circulates around a separate closed loop. Both the anolyte and catholyte circuits are made of either glass or Teflon-lined components. The anolyte circuit, as designed, includes three hydrocyclones in parallel to remove AgCl particles (formed when chlorine-containing compounds are treated) from the liquid stream. The feed system for energetics is charged with premixed water slurries of fine (<500 µm) M28, Composition B1 (an energetic material), and tetrytol (TNT and tetryl) in batches. These slurries are continuously fed to the anolyte vessel at a rate matching the destruction rate. To determine when the organics are completely destroyed, sensors measure the flow rates, CO (carbon monoxide), CO2 (carbon dioxide), and the total organic carbon (TOC) content in the anolyte vessel.
The catholyte circuit contains 4M (molar) nitric acid that is reduced to NOx (mixed oxides of nitrogen) during the process. The NOx is reoxidized to nitric acid in a reformer that consists of two columns: a water (or dilute acid) absorption column and a distillation column to separate the acid from the water before recycling. The NOx is absorbed in a column fed with the cooled acid condensate stream from the top of the distillation column. Oxygen is added to the system through the catholyte vessel to oxidize the NOx to nitric acid. The liquid stream from the absorption column is passed to the distillation column, where the acidic condensate and the concentrated acid are separated.
The concentrated acid is removed from the base of the column and the distillate from the top of the column. The nitric acid produced is recycled to the SILVER II™ system. For the destruction of compounds containing nitrogen (e.g., energetics), a net excess of nitric acid is produced that is removed as concentrated acid from the base of the column. For organic feeds that do not contain nitrogen, all of the concentrated acid must be returned to the catholyte vessel to replace the acid reduced to NOx in the cell. The anticipated nitric acid recovery is 99.5 percent.
The reformer off-gas is directed to a hypochlorite caustic scrubber. A continuous emission monitor (CEM) is used to monitor scrubber off-gas. The composition of this gas is expected to be primarily carbon dioxide (CO2) and oxygen (O2) with small amounts of water, nitrogen oxides (NOx), sulfur oxides (SOx), hydrogen (H2), and carbon monoxide (CO).
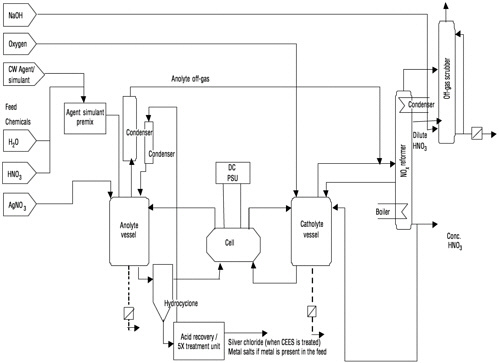
FIGURE 2–2 Process flow diagram of the AEA 2 kW demilitarization process. SOURCE: Adapted from AEA (2000).
TESTING
Agent Tests
Demo II testing of the AEA SILVER II™ 2 kW system with agent and agent simulant had the following objectives (DoD, 2001):
-
Validate the ability to achieve a destruction and removal efficiency (DRE) of 99.9999 percent for HD, GB, and VX.
-
Determine the impact of operations on materials of construction.
-
Demonstrate the operation and performance of key process components for future scale-up.
-
Develop the operational data for comparison with the 12 kW system.
-
Characterize the silver-bearing residuals and determine potential silver recovery and disposal options for residuals from the silver recovery operation (HD only).
-
Characterize gas, liquid, and solid process streams.
Sufficient agent or agent simulant was added to a premix vessel to complete an entire test.
Demo II testing of the SILVER II™ 12 kW system with agent simulant had the following objectives (DoD, 2001):
-
Validate the ability to achieve a DRE of 99.9999 percent for agent simulants.
-
Determine the impact of operations on materials of construction.
-
Demonstrate the operation and performance of key process components.
-
Develop operational data for comparison with the 2 kW system.
-
Demonstrate the ability/inability to recycle, reuse, or dispose of nitric acid.
-
Characterize gas, liquid, and solid process streams.
One agent simulant, DMMP, was tested in the 12 kW test unit (no agent was tested). For the DMMP validation, the organic feed was premixed with deionized water. This was to replace, in part, water lost from the anolyte during processing due to transfer across the membrane and consumption in oxidation reactions. Because of scheduling difficulties, the test originally planned for agent simulant CEES
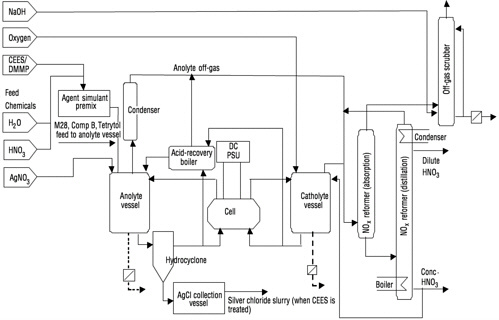
FIGURE 2–3 Process flow diagram of the AEA 12 kW demilitarization plant. SOURCE: Adapted from AEA (2000).
in the 12 kW unit was not performed. Once the DMMP had been premixed for 24 hours, it was discharged into a second vessel, from which it was continually mixed and metered into the anolyte vessel.
Energetics Tests
Demo II testing of the SILVER II™ 12 kW system with energetics had the following objectives (DoD, 2001):
-
Validate the ability to achieve a DRE of 99.999 percent for tetrytol, Composition B, and M28 propellant.
-
Determine the impact of operations on materials of construction.
-
Demonstrate the operation and performance of key process components for future scale-up.
-
Demonstrate the ability/inability to recycle, reuse, or dispose of nitric acid.
-
Characterize gaseous, liquid, and solid process streams.
The tests on energetic material were conducted with premixed water slurries in the 12 kW unit. The average size of the energetics particles was 500 µm. The slurries were prepared in batches and kept well stirred in a storage vessel until they were pumped into the anolyte vessel for destruction. The feed rate was adjusted to maintain a minimum level of energetic material in the anolyte circuit at any time. The TOC in the anolyte vessel was monitored online to estimate the level of energetic in the anolyte vessel.
TEST RESULTS
The next sections summarize the results of the 2 kW and 12 kW tests and their relation to the stated objectives. These sections include discussions and comments from the committee on destruction rates, materials of construction, and gaseous, liquid, and solid process discharge. Other important committee observations germane to the operation and performance of key process components for future scale-up are also included.
Destruction and Removal Efficiency
In both the 2 kW and 12 kW systems, the overall instantaneous destruction rate was calculated from the volumetric flow rate and the measured composition of the anolyte offgas. Analysis of the anolyte off-gas by continuous emission monitors provided the composition (volume percent, equivalent to mole percent) of the gas. The volumetric flow, corrected for ambient temperature and pressure, provided the total molar flow rate.
The DRE (in percent) for a feed of agent or energetic was defined by AEA as follows:
100×[(total amount fed to plant)−(amount remaining in plant+discharges after campaign)]/ [total amount fed to plant]
where the amounts are expressed in moles.2
In Appendix D of the AEA Draft Final Report (AEA, 2000), AEA gave a detailed calculation of the DRE (e.g., Table D-11 for DMMP), and it is clear that AEA calculated the discharges based on the flow rate of gas through the scrubber, the concentration observed in the scrubber off-gas, and the duration of the run. Post-test sampling of the anolyte, catholyte, and dilute and concentrated nitric acid and caustic scrubber solution was used to determine the amount remaining in the plant.
The goal for the destruction of agent simulant was 99.9999 percent; for energetics it was 99.999 percent. Determination of the DRE (as defined by AEA) depends on the detection limit of the analytical procedure for a particular agent or energetic and on the actual amount of material that was fed into the system. The total amount of agent, simulant, or energetic fed to the plant is known accurately from run records. The Environmental Protection Agency (EPA) reference test methods were used to determine concentrations of organics in solutions.
The 2 kW tests successfully demonstrated the destruction of HD, GB, and VX, as well as the destruction of simulant DMMP to 99.9999 percent DRE or greater. The amount of material treated, steady-state current efficiency, and DREs are shown in Table 2–1.
Tests originally planned for the simulant CEES were not conducted because of schedule constraints (the other demonstration tests took longer than had been expected, as discussed in more detail below).
In the 12 kW facility, 40 kg of the simulant DMMP was destroyed with a DRE similar to that in the 2 kW unit (99.99997 percent) and the same electrochemical efficiency (40 to 50 percent). Thus, AEA concludes that the two systems appear to be operating in a similar fashion.
All tests on energetics were conducted in the 12 kW plant. The Comp B was not tested owing to schedule constraints. Tests on M28 propellant successfully demonstrated high DREs of nitrocellulose (99.9999 percent) and nitroglycerine (99.99999 percent) and very high electrochemical efficiency (80 to 100 percent). More than 159 kg of M28 were treated in 8 days of operation.
In the tests conducted with tetrytol, solid material built up on the walls of the anolyte and catholyte circuits and forced operation at slower feed rates than anticipated. The
TABLE 2–1 Destruction Efficiency in the 2 kW Test Unit
|
Agent or Simulant |
Amount Treated (kg) |
Current Efficiency (%) |
Destruction Efficiency (%) |
|
DMMP |
10 |
40–50 |
99.9998957 |
|
VX |
4 |
70–90 |
99.9999886 |
|
HD |
16 |
40–60 |
99.9999914 |
|
GB |
15.7 |
60–80 |
99.999996 |
|
SOURCE: AEA (2000). |
|||
difficulties encountered in processing tetrytol were attributed to the formation of recalcitrant intermediate products that either crystallize in the anolyte circuit or migrate through the membrane to the catholyte circuit before crystallizing. The accumulation of these solids, which obstructed filters and sample lines and accumulated in tanks, valves, and piping, forced periodic change-outs of the solutions in both circuits. This slowed the feed rate of tetrytol. Very low electrochemical efficiencies (20 to 30 percent) were measured for tetrytol due to the small amount of organic material that could be tolerated in the anolyte vessel. When the concentrations of organics in the anolyte solution were lowered, their level of oxidation was reduced. The measured DREs of the tetrytol components were relatively low—for example, 99.7 percent for TNT and 99.8 percent for tetryl. Though the tetrytol runs were scheduled to last 7 days, they actually lasted 18 days, because the feed rate had to be lowered, and only 73 kg of tetrytol was processed. AEA has suggested several changes to the plant design to reduce the accumulation of this intermediate product. These suggestions are discussed later in this chapter.
Preliminary results from gas chromatography/mass spectrometry and high-performance liquid chromatography analysis of a solid intermediate recovered from the tetrytol testing identified several energetic compounds, including trinitrobenzoic acid (TNBA) and trinitrobenzene (TNB) (Winkler, 2001). Also, picric acid (PA) was identified in the solid intermediate (personal communication between John Coffey, senior environmental chemist, CH2M Hill, Inc., and Darren Dalton, PMACWA, February 21, 2001).
Currently, AEA considers the indicators of complete reaction to be the absence of TNT and tetryl (compounds that are present in the original feed) and a low value of TOC in the anolyte solution. However, the presence of TNBA, PA, and TNB crystals in both the anolyte and catholyte circuits implies that the absence of TNT and tetryl is in and of itself not a valid indicator of the total destruction of all energetic compounds in the system. Also, the measurement of TOC in the anolyte solution is not equivalent to the measurement of total organics in the system, because there is no accounting for the solids that precipitate out or otherwise accu
mulate in the system. The explosive hazard should not be assumed to be eliminated as the reaction proceeds, because TNBA or other energetic compounds may still remain.
Only a small amount of PA was observed in the solids formed during the tetrytol testing (O’Neil, 2001). However, if a mixed feed stream containing an aromatic nitro compound (Composition B and tetryl or tetrytol) is processed with a propellant that contains lead, lead picrate (a very sensitive primary explosive) can be formed. During the M28 propellant tests, lead dioxide precipitated on the cell membrane, so it is possible that the precipitation of lead with the small amount of picric acid that might be present does not compete with the formation of lead dioxide in the electrochemical cell. However, the possibility of lead picrate precipitation cannot be dismissed a priori. A determination of the relative solubilities of lead dioxide and lead picrate in nitric acid solutions could provide some insight into whether the formation of lead picrate is likely to be a problem for mixed feeds.
Materials of Construction
Even given the relatively short duration of the Demo II tests, it was apparent that corrosion is a serious problem. Notable problems encountered during the Demo II tests are listed below:
-
anolyte pump failures in the 2 kW tests
-
leakages in the glass-to-glass joints in the 2 kW facility
-
failure of the glass nonreturn valves in the 2 kW tests
-
damage to glass during the processing of GB in the 2 kW test
-
failure of glass components and joints in the 12 kW unit
-
cell gasket failure in the 12 kW unit
Materials of construction must be carefully selected to avoid leaks and failures that could interfere with full-scale operation.
The results of coupon tests updated by AEA (Table 2–2) demonstrate that serious issues remain concerning the selection of materials of construction. In these tests, coupons of different materials were exposed in the anolyte chamber during the Demo II program and were reweighed and photographed after exposure to the SILVER II™ environment for all of the runs. The approximate exposure times were just over 34 days. All materials tested showed significant weight losses during exposure to these environments. Corrosion was probably caused by the formation of hydrofluoric acid (HF) when treating GB. Even polytetrafluoroethylene (PTFE)-coated stainless steel showed high rates of corrosion. However, the technology provider has indicated that PTFE-lined stainless steel is still a viable alternative based on previous tests results and industrial experience. The technology provider attributed the coupon failure to micropores in the PTFE coatings, which it said should not be present in the PTFE-lined piping.
Characteristics of Gaseous, Liquid, and Solid Process Streams
All mass balances were obtained using the volumes and composition of input reactants and output products. Routine on- and off-line samplings were used to determine the concentrations of the intermediate species in the process streams. The inventory of materials in the system was estimated from the volume of material in each vessel. All streams leaving the process were analyzed and this, in conjunction with volume measurements, gave the total inventory of the species for the mass balance.
The organic feed material was well characterized and quantified, but the organic intermediates in the process were diverse and not quantified individually. However, the anolyte, catholyte, and reformer liquors were analyzed regularly to determine their TOC content. This TOC value, along with volume readings, yielded the inventory of dissolved organics in the plant. The organic feed rate; TOC determinations; and off-gas analyses for CO, CO2, and volatile organic compounds (VOCs) were used to derive a continuous mass
TABLE 2–2 Anolyte Coupon Weights Before and After Testing
|
Coupon |
Starting Weight (g) |
Final Weight (g) |
Change in Weight (g) |
Change in Weight (%) |
|
PTFE/304L #2 |
22.074 |
19.306 |
2.768 |
12.54 |
|
PTFE/304L #1 |
22.118 |
19.275 |
2.843 |
12.85 |
|
Titanium #4 |
20.086 |
16.343 |
3.743 |
18.64 |
|
Titanium #3 |
20.290 |
16.197 |
4.093 |
20.17 |
|
Glass #2 |
22.861 |
16.475 |
6.386 |
27.94 |
|
Glass #1 |
23.538 |
15.532 |
8.006 |
34.01 |
|
Zirconium #5 |
18.554 |
2.317 |
16.237 |
87.51 |
|
Zirconium #4 |
18.617 |
2.268 |
16.349 |
87.82 |
|
SOURCE: AEA (2000). |
||||
balance for carbon. Organic sulfur and phosphorus were determined from analysis of the sulfate and phosphate salts formed in the anolyte circuit after oxidation. All organic chlorine was assumed to be converted to AgCl at the end of a campaign. Any silver remaining in solution was neglected, because the solubility of silver chloride is less than 0.001M.
The following gaseous effluents were analyzed:
-
the anolyte gases for CO and SO2 (sulfur dioxide) by CEM; VOCs by EPA TO15; semivolatile organic compounds (SVOCs) by EPA 0010; agents by the depot area air monitoring system (DAAMS); and Schedule 2 decomposition compounds by EPA TO14
-
the pre-reformer off-gas for O2 and NOx by CEM
-
the post-reformer off-gas for O2 and NOx by CEM
-
the discharged off-gas for CO2, O2, CO, N2 (nitrogen), N2O (nitrous oxide), H2, SOx, and NOx by CEM and gas chromatography; VOCs by EPA TO15; SVOCs by EPA 0010; agents by DAAMS; and Schedule 2 decomposition compounds by EPA TO14
The Demo II tests revealed the presence of VOCs in the offgas stream. AEA has indicated that the design will be changed to include a catalytic oxidation (CATOX) unit on the off-gas vents to control the emissions of these organics. In the EDS phase of the program it is important to evaluate the performance of the proposed CATOX unit with particular focus on how impurities such as phosphorus and fluorine affect the catalyst’s oxidative reactivity.
The liquid discharges from the anolyte circuit, catholyte circuit, NOx reformer, and caustic scrubber were sampled and analyzed for metals and organics. At the time of this report, not all of the data were available to the committee. The tests apparently validated that this technology did not generate Schedule 1 compounds or significant quantities of Schedule 2 compounds regulated under the Chemical Weapons Convention (CWC). The Army has concluded that the characterization of the products of agent and propellant destruction showed that acceptable treatment of most hazardous intermediates (to relatively low levels) was achieved and validated for this process. Additional treatment steps that should effectively destroy the remaining hazardous intermediates were proposed but not demonstrated.
AEA’s effluent management system proposes to send the dilute nitric acid waste streams to a publicly owned treatment works (POTW) under a pretreatment exemption. Although the availability of a POTW has not been confirmed, the Demo II tests indicated that the concentrations of hazardous material in the liquid streams were sufficiently low to qualify for treatment at such a facility.
Solids from the scrubber filter and from the anolyte bag filter were analyzed at the end of each run. They were analyzed for agents, agent simulants, Schedule 2 compounds, other decomposition products, metals, SVOCs, VOCs, TOC, sulfates and phosphates, dioxins, and furans. Hazardous properties of the solids (ignitability, corrosivity, reactivity) were also examined. At the time the committee completed its evaluation for this report, the solid product characterization had not been completed.
Electrochemical Efficiency
AEA defines electrochemical efficiency as 100 times the ratio of current utilized to oxidize organic compounds in the feed to the sum of this current and the current used to electrolyze water. The current utilized to oxidize organic compounds is inferred from the amounts of CO and CO2 produced; the current used for the electrolysis of water is inferred from the amount of oxygen produced. Thus,
Ico, ![]() and
and ![]() are the currents (in amperes) used to form CO, CO2, and O2, respectively. This provides a measure by which to determine the relative amount of the current used to destroy the agent or the energetic versus the amount used in parasitic reactions.
are the currents (in amperes) used to form CO, CO2, and O2, respectively. This provides a measure by which to determine the relative amount of the current used to destroy the agent or the energetic versus the amount used in parasitic reactions.
The currents are not measured directly but are determined from the relative molar amounts of CO, CO2, and O2 measured in the off-gas. Using HD as an example, equations for the anodic reactions producing CO and CO2 are:
C4H8Cl2S+8H2O→→4CO+24H++2Cl− +S04−2+20e−
C4H8Cl2S+12H2O→→4CO2+32H++2Cl− +SO4−2+28e−
Thus, each mole of CO produced requires (20/4) faradays (F), or 5F, of charge to flow, while each mole of CO2 requires (28/4)F, or 7F. The electrochemical inefficiency is related to the fraction of the current that results in the production of O2:
2H2O→→O2+4H++4e−
Each mole of O2 thus requires 4F of charge to pass.
The currents required to calculate the efficiency are therefore determined from measurements of the off-gas composition and solution of the following equations:
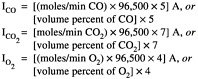
The committee believes that this definition of electrochemical efficiency cannot be taken as an indicator of the amount of energy that will be required to run the plant. Non-electrochemical oxidations, such as direct oxidation by nitric acid rather than Ag2+, may contribute to the destruction of the feed, thus reducing the need for electrochemically driven reactions and consequently the energy requirements. On the other hand, other dissipative processes, such as resistive heating and electrochemical oxidation of impurities in the system, will increase the required energy. The only utility of the electrochemical efficiency as defined by AEA is to reflect the competition between oxidation of the feed and parasitic reactions.
AEA DESIGN CHANGES BASED ON TEST RESULTS
Major changes have been proposed to the process flow diagram based on the Demo II test results. Figure 2–4 shows the revised AEA process flow diagram. Some of the changes are listed below:
-
addition of a CATOX unit to control VOCs in offgases
-
use of hydrocyclones in both the anolyte and catholyte circuits
-
addition of a return flow of material (both solids and liquids) from the catholyte to anolyte to return organics that have migrated across the cell membrane
-
addition of a high-shear vortex mixer in the anolyte circuit to achieve better mixing of the tetrytol to promote the oxidation of intermediate products that precipitate
-
addition of chemical cleaning processes for electrodes to remove the buildup of lead and AgCl
-
addition of an energetic rotary deactivator for the destruction of separated fuzes and supplemental charges
AEA proposes adding an impurities removal system (IRS) as a bleed stream on the anolyte circuit. The IRS would withdraw anolyte solution to control concentrations of impurities such as phosphates and sulfates and to remove solid precipitates (i.e., AgCl). In the IRS, liquids and gases would be separated from solids. The liquids and gases would be recycled back to the SILVER II™ anolyte circuits, and the solids would constitute a solid waste stream. The IRS includes five operations: (1) removal of solids, (2) fractional distillation of the solution, (3) separation of lower portions of the distillation column containing nitric acid enriched with phosphoric acid and sulfuric acid, (4) release of off-gases through a CATOX unit, and (5) recirculation of the condensate to the catholyte unit. The process flow diagram for the IRS was being modified when this report was written. Because the IRS is so complicated, further refinements and development will be necessary prior to its full-scale deployment.
The sizing of the IRS and the frequency of bleeds will be determined by the expected impurities in the feeds. AEA considers the lead in the burn rate modifiers in propellants to be an impurity that must be treated by the IRS. It has indicated that most of the lead oxide produced from lead in the propellants will be spalled to produce a suspension that will also need to be removed in the IRS. A smaller amount of lead precipitates out as lead oxide on the cell membrane, which keeps it from reaching the IRS. Fluoride, sulfate, and phosphate are expected to be present from agents but not energetics.
One of AEA’s main concerns to be addressed by the IRS is the presence of small pieces of metal in the energetics originating from resonance rods, which in turn come from the rocket propellant removal operation. The resonance rods sometimes become dislodged when the propellant is being removed, and unless they are removed, they will be ground up with the propellant. These metal grindings will be present in the slurry that is fed into the anolyte vessel. Even if the grindings do not short out the cell electrodes, a metallic conductor placed between two electrodes from which it is electrically isolated (no electronic connectivity) will itself become a bipolar electrode—the surface of the metal facing the anode will become cathodic and the surface of the metal facing the cathode will become anodic. In the extreme, the potentials can be strong enough to drive faradaic processes. If this happens, productivity could be lost as a result of parasitic side reactions and reduced voltage on the principal electrodes themselves.
At the time of the committee’s review, the IRS system was not well developed. The IRS system AEA proposes for agent is different from the system for energetics, and the design of the full-scale system is expected to be much different from the one that has already been tested. Because the IRS was not tested in Demo II, no other data were available on the characterization of solids. AEA plans to put the solids in containers and send them to a RCRA-approved disposal facility.
REEVALUATION OF STEPS REQUIRED FOR IMPLEMENTATION
The ACWI committee identified several key steps that would have to be implemented before the AEA SILVER II™ technology could be fully implemented (NRC, 1999). These steps are reevaluated on the basis of the results of the Demo II tests.
-
Modified shearing locations for M55 rockets and a new shearing machine must be tested to show routine segregation of components and reduction in particle sizes to less than 1 inch in diameter.
This still needs to be evaluated.
-
The modified mine shearing approach must be tested.
This is not applicable for the Blue Grass Stockpile, which has no land mines.
-
The dissolution of fuzes and mine bodies in nitric acid and SILVER II™ solution must be evaluated.
This is no longer relevant, because an energetic rotary deactivation unit has been added to the design.
-
All effluents must be characterized in detail when treating agents contaminated with metals from disassembled chemical weapons (i.e., potential trace species and reaction by-products, such as nitrated hydrocarbons, partially oxidized products, and metals, must be identified) and their environmental impacts evaluated.
The Demo II test evaluated all of the major effluent streams for a full suite of trace species and reaction by-products. Intermediate reaction products were found in tests with tetrytol, as described above. However, at the time this report was being prepared, not all of the data were available to the committee.
-
Demonstrations of the scale-up, development, and integration of hardware with real materials of construction must focus on the robustness of the parallel flow in multiple-cell reactors. The issues of cell blockage, hydrocyclone performance, and NOx reformer performance must be addressed.
The ACW II committee repeats this recommendation and stresses its importance in light of the increased complexity of the process once changes have been made to address problems revealed in the Demo II tests, which were discussed in the preceding section.
-
The efficacy of high pressure-jet wash-out of agent and gelled agent from M55 sheared pieces must be tested.
This recommendation was not addressed in the Demo II tests, so it remains valid.
-
The treatment of burster charges and M28 propellant in the SILVER II™ reactor must be tested, and the material preparation required to ensure reasonable treatment times with no energetic events must be evaluated. This testing must also determine what happens to the lead stearate in the propellant during SILVER II™ treatment.
The tests with the 12 kW system in the Demo II confirmed that this technology is capable of destroying the components (nitrocellulose and nitroglycerine) of M28 propellant. For tetrytol, the destruction of TNT and tetryl was good. However, recalcitrant intermediate products were formed during the treatment of tetrytol; AEA was still evaluating them as this report was being written. Given the preliminary indication that the intermediates were energetics, the finding of the need to demonstrate that no energetic events can occur remains valid. The committee is also not aware of any new data on the fate of lead stearate from these Demo II tests.
-
The process must be developed and tested for the efficacy of submerged-bath dilute nitric acid treatment for metals parts, including the effects of agitation and temperature.
This recommendation is no longer relevant, because the current design uses a metal parts steam treater; the condensed steam and organics produced by this unit are introduced into the agent SILVER II™ circuit. Off-gas is directed to a CATOX unit.
-
The treatment of shredded dunnage material must be tested in a prototype-scale SILVER II™ reactor.
This is no longer relevant because the dunnage and demilitarization protective ensemble (DPE)/carbon will be treated in a batch rotary treater. The resulting condensed steam and organics are sent to the SILVER II™ agent circuit, and the off-gas from the batch rotary dunnage treater is passed through a CATOX unit.
-
Techniques for controlling particulate matter to prevent plugging of SILVER II™ electrolytic cell channels must be developed and demonstrated.
The committee’s concern at the time of this recommendation was plugging in the cell channels due to formation of AgCl particles. Although no significant plugging problems were encountered in Demo II in the cell channels themselves due to AgCl particles, there was evidence of other types of particulate buildup in other components. For example, lead oxide precipitated in the cells. A wide range of unanticipated particulate and material blockages was encounered in the vessels and piping in the anolyte circuit. There was also a buildup of particulates in the catholyte circuit, caused by the migration of organics across the membrane. The potential for plugging as the units are scaled up remains a concern for the committee.
-
Materials of construction must be evaluated under corrosive and oxidizing conditions.
Severe corrosion was found in several materials, particularly those used to contain fluorine-bearing streams. This recommendation remains valid.
-
The realistic potential for off-site recycling/reuse of silver salts and concentrated nitric acid must be evaluated, including recyclers’ ability to accept, handle, and treat these materials.
This issue was addressed in the Demo II tests through an analysis of SILVER II™ concentrated acid by Ensign Bickford Company. Additional confirmation of the capabilities of commercial facilities should be solicited.
REVIEW OF THE ACW I COMMITTEE’S FINDINGS
It may be worth noting that many of the same problems were identified in the NRC study Review and Evaluation of Alternative Chemical Disposal Technologies (NRC, 1996) and the ACW I report (NRC, 1999). The ACW I committee’s earlier findings are updated below:
Finding AEA-1. Significant barriers to the development of the sophisticated equipment and processes for segregation, materials handling, and size reduction (to reduce materials to less than 1 inch in diameter) must be overcome.
These barriers remain a concern to the ACW II committee. The tests with energetics in the 12 kW unit were done with material reduced to less than 500 µm (AEA, 2000) and then premixed into slurries. The full-scale design calls for one-eighth-inch (~3,000-µm) pieces in slurries. The equipment and process for segregating, handling, and size-reducing to this target size will have to be defined and evaluated for the full-scale equipment.
Propellant grinding has not been successfully demon-strated. Another concern is that increasing the particle size from 500 µm to 3,000 µm could substantially increase the processing time, decrease electrochemical efficiency, and increase the formation of by-products and associated blockage.
The description for AEA’s energetic process is as follows (AEA, 2000).
Using the Army’s existing Projectile/Mortar Demil Machine (PMD), 155mm and 8-inch projectiles will be disassembled, but we propose to replace the existing Multipurpose Demil Machine (MDM) with a new punch, drain, and washout machine. Projectiles will be received in the unpack area and loaded into the existing feed equipment for transportation into the explosive containment room. There will be two identical disassembly equipment lines. For rockets, the PMD will remove the nose closure, burster, fuze, supplemental charge and miscellaneous parts. Fuzes and supplemental charges will be conveyed to the detonation chamber for deactivation. The bursters will be extracted and conveyed to a stand-alone burster washout machine to fluid jet out the burster, using conventional fluid jet technology. This will result in an energetic slurry with a nominal maximum particle size of oneeighth inch and a slurry concentration not to exceed 20 percent by weight. The Rocket Demilitarization Machine directs the rocket motor section to the propellant processing station. The propellant is mechanically extracted from the motor section. The propellant grain is then deposited into the energetics grinder for size reduction. The grinder design will be optimized during EDS testing. Blade configuration and speed will be selected to process the propellant, which has been described as having a consistency of “tire rubber.” The grinding operation will be done “wet” to reduce the possibility of fire and aims to produce a one-eighth inch (maximum) particle size.
Wet cutting and grinding/shredding have been used safely by industry. However, there is always a possibility that metal shards or large chunks of propellant will become wedged in the cutting or shredding equipment and ignite. The final design is expected to address this possibility, and the cutting area will be designed to withstand propellant ignition.
Finding AEA-2. Potential problems associated with plugging of the passages in the electrolytic cells will have to be addressed.
No blockages of the electrolytic cell were reported during normal operation in either the 2 kW or the 12 kW unit during Demo II. However, there were buildups of silver chloride particles and blockage in other areas: (1) the carryover from the 5X treatment unit and sample lines in the 2 kW unit and (2) failure of the cell gasket in the 12 kW unit. For this reason, the ACW II committee remains concerned that cell blockages are likely to occur with the equipment scale-up.
Other blockages occurred outside the electrochemical cells during the treatment of tetrytol. As was discussed above, these were apparently caused by the formation and precipitation of intermediate decomposition products with low solubility. The blockages were severe enough in the 12 kW unit to prevent completion of the demonstration tests for tetrytol. In addition, there was deposition of solids and blockage in the catholyte as a result of the movement of intermediate products across the cell membrane. AEA has proposed a number of changes in the process design to prevent this: (1) the addition of a hydrocyclone in the catholyte circuit and anolyte circuits, (2) the addition of a high-shear vortex mixer in the anolyte and catholyte circuits, (3) the addition of an IRS using a bleed from the anolyte, and (4) provisions for a return flow from the catholyte to the anolyte circuit. The ACW II committee reiterates that buildup of solid materials in the circuits and the potential for blockages is an area of high technical risk that will require continued attention during scale-up and engineering design.
The committee further notes that the formation of intermediate decomposition products was not anticipated. Until the unit is operated successfully with all possible feeds, this will remain another area of high technical risk. Furthermore, some of the feeds are expected to vary in composition, which will complicate the challenge to system operability.
AEA has determined that TNBA did form in the anolyte vessel and that solids formed on the membrane and sides of the anolyte vessel during the treatment of tetrytol. Some of the TNBA crystals were 0.25 inch across. AEA said that when the particles reached a certain size, they broke away from the walls and fell to the bottom of the anolyte vessel. In its EDS, AEA proposes using a high-shear mixer in the anolyte vessel to break up these crystals. The mixer would break up other particles and is expected to reduce most of the solids to less than 100 µm before they enter the cell where the Ag2+ is formed and destruction takes place. AEA does not plan to do a mass balance but will determine when the destruction is complete by measuring the amount of starting material left in the anolyte vessel. Although this will indicate when the tetryl is destroyed, it will not indicate when all of the intermediate energetics has been destroyed.
Finding AEA-3. The proposed chemical dissolution of fuzes is a complete unknown because no data on this process are available. This approach is complex in comparison to more conventional techniques of separation and detonation.
The dissolution of fuzes was not tested in the Demo II test program. The preliminary design now calls for the separation of fuzes followed by destruction in an energetics rotary deactivation unit, a common approach for destroying fuzes. Chemical dissolution of fuzes is no longer part of the design.
Finding AEA-4. Data are not available to assess the efficacy of the treatment of energetics at larger scales.
The 12 kW pilot-scale tests conducted during the Demo II tests specifically tested for the DRE of components in M28 propellant and tetrytol. These tests provided evidence that high DREs (99.9999 percent) could be achieved for components of M28 propellant (nitrocellulose and nitroglycerine). However, lower DREs (~99.7 percent) were achieved with the components of tetrytol.
Processing time for the destruction of energetics, particularly M28 propellants, in the SILVER II™ unit at full scale will certainly increase as a result of larger particles (3,000 µm). In addition, processing times for all energetics will increase as a result of slower mixing in the anolyte vessel, the need to treat intermediate by-products, and the impacts of recycling untreated material from the catholyte. The committee reiterates its concerns about the impact of scale-up on DREs for energetics.
Finding AEA-5. The ability to obtain a representative sample of gaseous effluents retained in the holding tanks prior to release has not been demonstrated.
The hold, sample, and release option was not included in the Demo II tests. All gas analyses were performed on samples taken from the flowing off-gases over 2-hour periods. Techniques for sampling and ensuring that no agents are present prior to release will have to be developed and confirmed in subsequent programs.
Finding AEA-6. Several issues need to be addressed during the scale-up of the process into a fully integrated system, including temperature control, reaction times, efficiency of the NOx reformer, cell flow management, efficiency of the hydrocyclone, and the tolerance of cells to particulate matter. These issues are potentially serious enough to create processing problems in larger scale systems.
The 2 kW and 12 kW demonstration units were successfully operated, showing that small-scale units could operate properly with adequate process control. However, critical gaps in knowledge remain with respect to performance of the scale-up process. Several process features are expected to become more difficult to control and manage as the scale is increased from 12 kW to more than 300 kW in a full-scale system. The most pressing issues are the following:
-
longer reaction times for larger particles
-
mixing limitations in larger anolyte vessels
-
less effective performance of larger hydrocyclones
-
formation of additional soluble by-products
-
need for recirculation of catholyte solution to the anolyte vessel to control organics passing through the cell membrane
-
control issues associated with a fully integrated pilot plant
Although none of these issues is likely to preclude the use of this technology, resolving the uncertainties will require comprehensive, time-consuming, integrated, pilot-scale development and systemization.
Finding AEA-7. Identifying which corrosion-resistant materials would be compatible with HNO3 and HNO3/HF will require a significant development program.
The committee reiterates and reinforces the initial finding that corrosion from fluorine-containing agents—GB, for instance—will be a serious problem in the full-scale operation of the SILVER II™ process. AEA has recognized this issue and is considering other materials of construction and exploring techniques for removing fluorine from agents prior to their introduction to the anolyte circuit. The committee believes that further assessments will be necessary to determine the viability of these techniques and notes that a fluorine removal step further complicates the process.
Finding AEA-8. Limiting or controlling reactions between nitric acid and agent and energetics must be demonstrated.
The Demo II results show that reactions between nitric acid and agent and energetics can be safely controlled.
Finding AEA-9. Conversion of excess nitric acid into ammonium nitrate fertilizer may be complicated because of the potential for contamination and the liabilities this would entail.
Conversion of nitric acid was not tested or evaluated in this study. However, the excess nitric acid was analyzed and found to be free of organics, agents, Schedule 2 compounds, and decomposition products. Therefore, conversion of nitric acid to ammonium nitrate, a commercially viable process, appears to be feasible.
Finding AEA-10. A large inventory of silver is required for processing of chemical weapons, and finding an off-site recycler to accept the potentially contaminated materials could be a problem.
AEA provided a letter from an outside vendor indicating that the dried silver chloride product is suitable for recy-
cling. However, the silver chloride that was acceptable to the vendor was produced from a well-controlled feed material. Impurities in the feed from assembled chemical weapons may be present in the silver chloride produced during their destruction and may affect the acceptability of this product to off-site recyclers.
SUPPLEMENTAL FINDINGS
Finding DII AEA-1. The overall process flow has been further complicated by major design changes in response to the Demo II testing. These changes include the addition of the IRS, CATOX units, and a flow return circuit from the catholyte to the anolyte circuit. All three changes require small-scale and pilot-scale testing. Such modifications further complicate the interfaces between process units, which increases the time required for development, start-up, and commissioning of the full-scale system. Integration of the operating units will make achievement of a viable total solution very difficult.
Finding DII AEA-2. The discovery of organic material migration across the electrochemical cell membrane will require major modifications in design and operation, such as recycling of the catholyte material to the anolyte circuit and the addition of hydrocyclones in the catholyte circuit.
Finding DII AEA-3. The formation of intermediate oxidation by-products raises operational issues, including slower processing rates and reduced electrochemical efficiency. During the testing with tetrytol in the 12 kW unit, the problems were severe enough to cause the runs to be extended well beyond the planned processing times.
Finding DII AEA-4. The generation of new energetic compounds (TNBA, PA, TNB) in the course of processing increases the complexity and hazards of the SILVER II™ process. Although the explosion hazard is reduced as the energetic feed is consumed, it is not completely eliminated until all energetic intermediates are destroyed.
Finding DII AEA-5. During the treatment of M28 in the Demo II test, lead oxide and other materials accumulated on cell anodes. The committee believes that a maintenance procedure for routine cleaning of the anodes will be required.
Finding DII AEA-6. Low steady-state electrochemical efficiencies (20 to 30 percent) were observed during treatment of tetrytol. These low efficiencies will decrease the throughput per cell and increase processing time and energy consumption.
Finding DII AEA-7. VOCs were detected in the off-gas of the AEA process technology. AEA has now included a CATOX unit in the preliminary design. The committee believes that the introduction of this additional unit operation will further complicate the scale-up and integration.
Finding DII AEA-8. The IRS for removing salts (sulfates, phosphates, silver fluoride), excess water, and any metals that may be present requires extensive development and integration. The IRS has not yet been described in sufficient detail to allow for a meaningful assessment.
SUPPLEMENTAL RECOMMENDATIONS
Recommendation DII AEA-1. The possible formation of lead picrate when mixed energetic feeds are treated must be investigated before any processing of lead-containing propellant, TNT-based energetics, or tetryl is undertaken.
Recommendation DII AEA-2. The IRS, the CATOX units, the return flow, and all other major modifications to the system must be tested and proven during the EDS design phase.
Recommendation DII AEA-3. AEA must validate complete destruction of all energetic intermediates during the EDS design phase.
Recommendation DII AEA-4. AEA must conduct additional tests to identify suitable materials of construction to overcome corrosion problems encountered owing to the formation of HF in the treatment of GB.