5
Nutrients as Regulators of Gene Expression
DONALD B. JUMP
Michigan State University
Macronutrients (carbohydrate, lipids, and protein) play a fundamental role in mammalian growth and development by serving as a source of energy as well as components for the synthesis of structural and regulatory components of cells. Certain macronutrients also affect cell function through changing circulating hormones (e.g., glucose regulation of insulin release from pancreatic β-cells). We have known for many years that certain micronutrients, like vitamins A and D, have dramatic effects on gene expression through the regulation of intracellular receptors that bind promoters of specific genes.
The notion that macronutrients, or their metabolites, might also affect gene expression is a new concept that has emerged over the last decade. It is now clear that certain macronutrients (or their metabolites) affect gene expression and lead to changes in the abundance of key proteins that function at critical steps in metabolic pathways or control cell division or differentiation (Figure 5-1). Here, I briefly describe how three macronutrients (i.e., cholesterol, glucose, and dietary fat) affect gene expression to change cell metabolism.
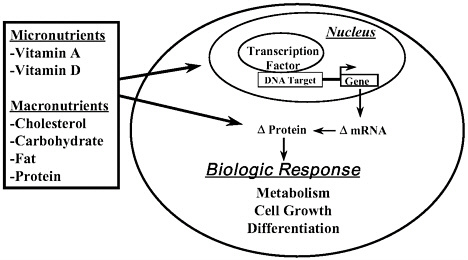
FIGURE 5–1. Overview of nutrients as regulators of gene expression.
CHOLESTEROL REGULATION
Pioneering work of Brown and Goldstein (1997) over the last 10 years has clearly shown that cholesterol regulates its own metabolism. Cholesterol plays an important role in growth because it is a component of cell membranes and steroids. The cell has the capacity to regulate its uptake of cholesterol through the low density lipoprotein (LDL) receptor (LDLR), as well as to regulate de novo synthesis of cholesterol. (Figure 5–2). Low intracellular cholesterol levels prompt a cellular response that leads to an induction in both the LDLR and the enzymes responsible for cholesterol synthesis. As intracellular cholesterol levels increase, this process is reversed. These changes in cholesterol metabolism are due, at least in part, to the effect of cholesterol on the nuclear content of a family of specific transcription factors called sterol regulatory element binding proteins (SREBP). Three SREBP subtypes have been described (i.e., SREBP1a, SREBP1c, and SREBP2). SREBPs are helix-loop-helix transcription factors that bind at specific cis-regulatory elements called sterol regulatory elements (SRE) in the promoters of several genes involved cholesterol synthesis, cholesterol uptake, and fatty acid synthesis. Binding of SREBP to SRE induces transcription of specific genes, leading to an increase in the mRNA and corresponding protein.
SREBPs are synthesized as ~125 kd precursor proteins (pSREBP) tethered to the endoplasmic reticulum and golgi membranes. Specific proteases digest the precursor to generate a 65 kd form of SREBP (nSREBP) that travels to the nucleus where it binds SREs. Cholesterol regulates the nuclear content of
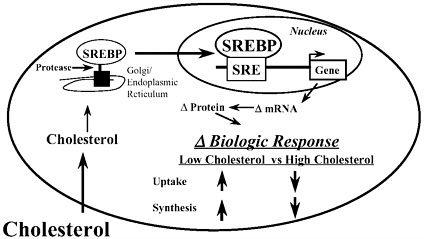
FIGURE 5–2. Cholesterol regulation of cholesterol metabolism.
SREBP by controlling the proteolysis step. Cholesterol controls the passage of SREBPs from the endoplasmic reticulum to the golgi. When cholesterol levels are low, proteolysis of pSREBP occurs and generates nSREBP and specific SREBP-regulated genes are activated. As intracellular cholesterol levels rise, SREBP proteolysis declines leading to a fall in the nSREBP and suppression of gene transcription. Thus, cholesterol is a feedback regulator for its own synthesis by controlling the nuclear content of SREBPs.
GLUCOSE REGULATION
Glucose effects on gene expression have traditionally been ascribed to its regulation of insulin release from the pancreatic β-cell. However, studies with primary hepatocytes and pancreatic β-cells have shown that glucose acts independently of insulin to control gene expression. When glucose concentrations increased in liver cells, enzymes like pyruvate kinase and acetyl-CoA carboxylase increase. This leads to increased flux of glucose metabolites into fatty acid synthesis. In the pancreatic β-cell, transcription of pyruvate kinase and the insulin gene is induced by glucose. In both cases, glucose stimulates transcription of specific genes that augment the cellular content of the corresponding mRNA and protein. These glucose-regulated genes contain in their promoters specific cis-regulatory elements, called carbohydrate (or glucose)
response elements. Unfortunately, the identity of the transcription factors binding these elements and the mechanism for glucose regulation of these factors is unknown. Nevertheless, the overall effect of this regulatory mechanism is to promote glucose uptake from the circulation, its intracellular metabolism and assimilation into lipid.
FATTY ACID REGULATION
Dietary fat and the resulting fatty acids have emerged as major regulators of gene expression through at least three distinct mechanisms: 1) as precursors to eicosanoids, 2) as ligands for nuclear receptors, and 3) as a controller of the nuclear content of SREBP1c.
Eicosanoids are oxidative products of arachidonic acid, a polyunsaturated fatty acid (PUFA). These products include prostaglandins, leukotreines, and thromboxanes and involve two enzymatic pathways, the cyclooxygenase (COX) and lipoxygenase pathways. Arachidonic acid is released from cell membrane phospholipids by the action of phospholipase A2 and is converted to eicosanoids by COX or lipoxygenases. These bioactive lipids, like prostaglandin E2 (PGE2), are secreted from cells where they act locally on plasma membrane-associated G-protein linked receptors (GPR) on target cells (Figure 5-3). These receptors control intracellular second messenger levels, like cAMP and free calcium, which, in turn, control numerous cellular processes through changes in protein phosphorylation. Consequently, eicosanoid binding to GPR rapidly stimulates protein phosphorylation, leading to changes in metabolism, cytokine production, and production of adhesion molecules. Some of these effects involve changes in gene expression through controlling the activity of specific transcription factors, like cFos, cJun, NFκB, and cMyc.
Essential fatty acid deficiency is associated with a decline in arachidonic acid phospholipid content and the production of eicosanoids. Eicosanoid production is associated with inflammatory responses and host defense. Interestingly, certain dietary fats, particularly the highly unsaturated n-3 fatty acid, are poor substrates for COX. This leads to a decline in eicosanoid production as well as a diminished inflammatory response.
A second route for fatty acids to affect gene expression is through the regulation of a family of nuclear receptors called peroxisome proliferator activated receptors, (PPAR). Four PPAR subtypes have been identified (i.e., α, β, γ1, and γ2). These are members of the steroid superfamily of nuclear receptors that bind DNA motifs, called peroxisome proliferator regulatory element (PPRE). PPARs bind PPRE in association with a second receptor called retinoid X receptor (RXR). PPARs were first identified as the molecular targets for peroxisome proliferators. Peroxisomes are subcellular organelles involved in β-oxidation of fatty acids and cholesterol metabolism. Peroxisome proliferators are
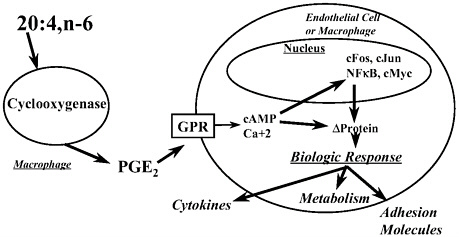
FIGURE 5-3. Eicosanoid regulation of gene expression.
a structurally diverse class of hydrophobic compounds that lead to peroxisomal proliferation in rodent liver. However, certain drugs have as their molecular targets specific PPARs. For example, the hypolipemic drugs may target PPARα, while PPARγ is the molecular target for the insulin sensitizing drugs.
PPARs have been associated with the regulation of expression of genes involved in nearly all facets of fatty acid metabolism (i.e., fatty acid uptake, fatty acid binding, fatty acid oxidation, and adipocyte differentiation). In addition, these receptors are reported to participate in inflammation as well as cell growth and differentiation. Interestingly, fatty acids, their metabolites and certain drugs bind to and activate PPARα, much like a steroid binds to a steroid receptor. For example, in the rodent liver, hypolipemic drugs and highly unsaturated n-3 fatty acids activate PPARα, leading to enhanced peroxisomal and microsomal fatty acid oxidation. In preadipocytes, thiazolidinediones and possibly eicosanoids bind to and activate PPARγ2. This accelerates the rate of adipocyte differentiation and increases insulin sensitivity of the adipose depot.
While PPARs have attracted considerable attention as molecular targets for fatty acid regulation of gene expression, it appears that these factors are not the sole targets for fatty acid effects on the genome. Recent studies indicate that one of the SREBPs, namely SREBP1c, is particularly sensitive to fatty acid regulation. Several reports appearing in the last 2 years have suggested that SREBP1c plays a major role in both hepatic and adipocyte lipogenesis, i.e., the synthesis of fatty acids and triglycerides. Feeding animals diets supplemented with polyunsaturated fatty acids suppress the mRNA encoding SREBP1c as well
as both the precursor and nuclear forms of SREBP1c. Because SREBP1c is a key factor in the transcription of several lipogenic genes, its decline leads to a reduction in lipogenic gene expression and de novo lipogenesis.
Clearly, fatty acid effects on cell function go far beyond serving as sources of energy and structural components of membranes. Fatty acids enter cells, undergo metabolism, and can serve as ligands for both membrane and nuclear receptors. Alternatively, fatty acids or their metabolites can regulate the nuclear abundance of SREBP1c, a key transcription factor in the synthesis of fatty acids and triacylglycerols.
SUMMARY
I have highlighted some of the recent advances in macronutrient regulation of gene expression, and I have provided the detail needed to understand the roles and effects of these nutrients. In addition to their role as an energy source, as structure elements or precursors to signaling molecules, macronutrients clearly have profound effects on gene expression. This nutrient-genome interaction interfaces with other signaling networks to allow integration of cellular control between dietary intake and internal regulatory mechanisms. It reflects an adaptive response, allowing cells to adjust to changes in the type, quantity, and duration of nutrients ingested for efficient growth.
While pharmacologic agents have been developed to control cholesterol synthesis (i.e., statins) and lipid synthesis (i.e., fibrates or thiazolidinediones), a better understanding of these regulatory processes will allow for the design of more effective agents to modify metabolism in both man and animals. This understanding and ability to design agents to modify metabolism will have benefit to human health, as well as animal production and health.
REFERENCES
Brown, M.S., and J.L. Goldstein. 1997. The SREBP pathway: Regulation of cholesterol metabolism by proteolysis of a membrane-bound transcription factor. Cell 89: 331-340.
Jump, D.B., and S.D. Clarke. 1999. Regulation of gene expression by dietary fat. Annu. Rev. Nutr. 19: 63-90.






