1
Sources and Effects of Carbon Monoxide Emissions
THE PROBLEM
Carbon monoxide (CO)—a colorless, odorless, tasteless, and toxic air pollutant—is produced in the incomplete combustion of carbon-containing fuels, such as gasoline, natural gas, oil, coal, and wood. The largest anthropogenic source of CO in the United States is vehicle emissions. Breathing the high concentrations of CO typical of a polluted environment leads to reduced oxygen (O2) transport by hemoglobin and has health effects that include headaches, increased risk of chest pain for persons with heart disease, and impaired reaction timing. In the 1960s, vehicle emissions led to increased and unhealthful ambient CO concentrations in many U.S. cities. With the introduction of emissions controls, particularly automotive catalysts, estimated CO emissions from all sources decreased by 21% from 1980 to 1999 (EPA 2001a). Average ambient concentrations decreased by about 57% over the same period (EPA 2001a).
The locations that continue to have high concentrations of CO tend to have topographical or meteorological characteristics that exacerbate pollution; for example, strong temperature inversions or the existence of nearby hills that inhibit wind flow may limit pollutant dispersion. Because of the limited dispersion, many of those areas also have unhealthful concentrations of sum-
mer ozone (O3) and year-round particulate matter (PM).1 Low temperatures also contribute to high CO concentrations. Engines and vehicle emissions-control equipment operate less efficiently when cold: Air-to-fuel ratios are lower, combustion is less complete, and catalysts take longer to become fully operational. The result is that products of incomplete combustion, including CO, are formed in higher concentrations. Sometimes, topography, meteorology, and emissions combine to cause high concentrations of CO. Compliance with the health-based National Ambient Air Quality Standards (NAAQS) for CO has proved difficult under those circumstances. The question arises as to whether unique methods are necessary to manage CO in such problem areas, or whether the current policies will ultimately achieve good air quality.
CHARGE TO THE COMMITTEE
In response to the challenges posed for some areas by having to come into compliance with the NAAQS for CO, a committee was established by the National Research Council (NRC) to investigate the problem of CO in areas with meteorological and topographical problems. The committee’s statement of task is as follows:
An NRC committee will assess various potential approaches to predicting, assessing, and managing episodes of high concentrations of CO in meteorological or topographical problem areas. The committee will consider interrelationships among emissions sources, patterns of peak ambient CO concentrations, and various CO emissions-control measures in such areas. In addition, the committee will consider ways to better understand relationships between episodes of high ambient CO and personal exposure, the public-health impact of such episodes, and alternative ways to measure progress in controlling ambient CO. An interim report dealing with Fairbanks, Alaska, as a case study is to be completed. A final report, including other CO problem areas, will be completed by the end of the study.
The committee will address the following specific issues:
-
Types of emissions sources and operating conditions that contribute most to episodes of high ambient CO.
-
Scientific bases of current and potential additional approaches for developing and implementing plans to manage CO air quality, including the possibility of new catalyst technology, alternative fuels, and cold-start technology as well as traffic and other management programs for motor-vehicle sources. Control of stationary source contributions to CO air quality will also be considered.
-
Assessing the effectiveness of CO emissions-control programs, including comparisons among areas with and without unusual topographical or meteorological conditions.
-
Relationships between monitored episodes of high ambient CO concentrations and personal human exposure.
-
The public-health impact of such episodes.
-
Statistically robust alternative methods to assist in tracking progress in reducing CO that bear a relation to the CO concentrations considered harmful to human health.
This study is expected to provide scientific and technical information potentially helpful to the development of state implementation plans; however, the committee will not provide prescriptive advice on the development of specific state implementation plans for achieving CO attainment. In addition, it will not suggest changes in regulatory compliance requirements for areas in nonattainment of the NAAQS, and it will not recommend changes in the NAAQS for CO.
Fairbanks, Alaska, was chosen as a case study for this interim report because its meteorological and topographical characteristics make it susceptible to severe winter inversions that trap CO and other pollutants near ground level. The combination of low temperatures and low solar radiation during the winter decreases the rate at which CO is ventilated from the area, while Fairbanks’s location in a river valley with hills on three sides further inhibits pollutant dispersion. In addition, Fairbanks experiences relatively high winter emissions per vehicle because of its low temperatures.
In this interim report, the committee addresses those parts of the charge pertinent to Fairbanks. Specifically, the committee reviewed Fairbanks in terms of the meteorological and topographical conditions that foster pollution episodes, CO emissions from mobile sources in severe winter conditions, air
quality management options, and statistical methods for tracking progress. Because no exposure data were available in Fairbanks and the small population size makes epidemiological studies difficult, the committee was unable to directly evaluate the public-health impact of high ambient CO concentrations there. Nevertheless, after reviewing the evidence on the health effects of CO presented in the literature and summarized by EPA in their most recent criteria document (EPA 2000a), the committee agrees that meeting the NAAQS for CO will protect human health with an adequate margin of safety. It is important to note that the committee was specifically instructed not to suggest changes to the levels or the form of the NAAQS for CO.
The final report, which will be issued in early 2003, will look at the characteristics of the CO problem in other meteorological and topographical problem areas and at the applicability of the various CO-control measures in these areas. One subject of particular interest will be the extent to which areas other than Fairbanks would benefit from stricter vehicle-certification standards that control emissions produced by vehicle starts at low temperatures.
HEALTH EFFECTS OF CO
Clinical and Epidemiological Studies of CO Effects
CO affects human health by impairing the ability of the blood to bring O2 to body tissues. When CO is inhaled, it rapidly crosses the alveolar epithelium to reach the blood, where it binds to hemoglobin to form carboxyhemoglobin (COHb), a useful marker for predicting the health effects of CO. Because CO has an affinity for hemoglobin more than 200 times greater than does O2, the presence of CO in the lung will displace O2 from the hemoglobin. In other words, when CO is present in the lungs, the hemoglobin will be unable to reach 100% O2 saturation. In addition, the presence of COHb increases hemoglobin’s affinity for O2, thereby inhibiting release of O2 from the hemoglobin to body tissues. The effect of COHb is illustrated by a leftward shift of the O2-hemoglobin dissociation curve (Figure 1–1). As shown in Figure 1–1, once COHb forms, the hemoglobin is unable to reach 0% O2 saturation. This second effect continues until the COHb dissociates, typically several hours after CO exposure. CO not only decreases the O2-carrying capacity of the blood, but also decreases the ability of the tissues to extract O2 from the blood during circulation. CO has also been shown to bind to myoglobin and may affect O2 transport to muscle (EPA 2000a).
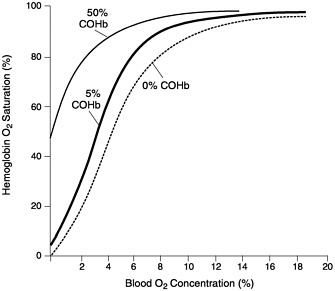
FIGURE 1–1 Diagram of hemoglobin response to the presence of COHb. The concentration of O2 in the environment surrounding the hemoglobin is shown on the x-axis. The O2 saturation, or how much of the hemoglobin’s capacity for storing O2 is used, is shown on the y-axis. At higher O2 concentrations, as are found in the lungs, the hemoglobin can be more O2 saturated. Likewise, at lower O2 concentrations, as are found in other parts of the body, O2 will dissociate from the hemoglobin to achieve O2 saturations as indicated by the curve. The presence of COHb shifts this curve to the left. For a given O2 concentration, the hemoglobin will require a higher O2 saturation and allow less O2 to be released to body’s tissues. Source: Adapted from Shephard 1983.
COHb levels in healthy individuals not recently exposed to high concentrations of ambient CO are 0.3 to 0.7%. Exposure to high concentrations of ambient CO can result in concentrations of COHb of 2% or higher if the exposure lasts long enough (hours). For those who smoke, cigarette-smoking is typically the most significant source of personal CO exposure. COHb concentrations, which are generally less than 1% in nonsmokers, average about 5% in smokers and are up to 10% or even higher in some very heavy smokers (Beckett 1994).
The CO health standards set by EPA are intended to keep COHb concentrations for nonsmokers below 2% in order to protect the most susceptible members of the population. EPA last published the Air Quality Criteria for
Carbon Monoxide, which summarizes health research findings and their implications for setting the NAAQS, in June 2000 (EPA 2000a). That document provides a comprehensive review of the literature pertaining to the health effects of CO for typical environmental exposures that would be associated with COHb levels less than 10%. The major findings presented by EPA (EPA 2000a) are summarized below; the committee accepts these findings as sufficient evidence of the health effects caused by exposure to CO at concentrations of 9 parts per million (ppm) and above for an extended period of time.
The acute affects of CO poisoning are well understood (Raub et al. 2000). Generally, in otherwise healthy people, headache develops when COHb concentrations reach 10%; tinnitus (ringing in the ear) and lightheadedness at 20%; nausea, vomiting, and weakness at 20–30%; clouding of consciousness and coma at around 35%; and death at around 50% (Coburn 1970). However, the outcomes of long-term, low-concentration CO exposures are less well understood. Because of the critical nature of blood flow and O2 delivery to the heart and brain, these organ systems, as well as the lungs (the first organ to come into contact with the pollutant), have received the most attention.
In patients with known coronary artery disease, COHb concentrations as low as 3% exacerbate the development of exercise-induced chest pain (Allred 1989a). Concentrations as low as 6% are associated with an increase in the number and frequency of premature ventricular contractions during exercise in patients with severe heart disease (Allred 1989b; Sheps et al. 1990). Large environmental-exposure cohort studies have confirmed that daily increases in ambient CO concentrations are associated with statistically significant increases in the numbers of hospital admissions for heart disease (Poloniecki et al. 1997; Schwartz 1999) and congestive heart failure (Morris et al. 1995) and with increases in deaths from cardiopulmonary illnesses (Prescott et al. 1998).
Neuropsychiatric (neurological and psychiatric) disorders and cognitive impairments due to long-term, low-concentration CO exposures have been hypothesized in part on the basis of extrapolation from the known acute effects of high-dose CO poisoning and the concomitant subacute and delayed neuropsychological sequelae. In clinical experiments on healthy volunteers, controlled CO exposure was associated with subtle alterations in visual perception when COHb concentrations were above 5% (McFarland 1970; Horvath et al. 1971). However the significance of this finding remains unknown. Similar studies have shown measurable but small effects on auditory perception, driving performance, and vigilance (Beard and Wertheim 1967; McFarland 1973; Benignus et al. 1977).
The role of CO in pulmonary disease is unclear. In the Seattle area, a single-pollutant model showed a 6% increase in the rate of hospital admis-
sions for asthma with each 0.9-ppm increase in CO, but that was concomitant with increases in other air pollutants (Sheppard et al. 1999). In Minneapolis and Toronto, CO concentrations showed only weak and inconsistent associations with total admissions for respiratory diseases (Burnett et al. 1997; Moolgavkar et al. 1997).
A fetus is more susceptible to CO than an adult; the O2-hemoglobin dissociation curve is to the left of that in the adult. It is shifted even further to the left by CO exposure. Also, because the half-life of fetal COHb is longer than that of adults, it may take up to five times longer to reduce the concentrations to normal. Other studies have shown that exposure to high concentrations of CO during the last trimester of pregnancy may increase the risk of low birth weights and that exposures to CO and PM during pregnancy may trigger preterm births (Ritz and Yu 1999; Ritz et al. 2000). A recent study linked CO and O3 exposure during pregnancy to birth defects such as cleft lip and defective heart valves (Ritz et al. 2002). However, the lead author of the study cautioned that the real culprit may be other pollutants that are dispersed with CO in tailpipe emissions (Beate Ritz, personal communication, January 31, 2002).
Public-health laws are designed to protect the most susceptible people in the population. People with coronary artery disease or other cardiopulmonary diseases, fetuses, infants, and athletes who exercise heavily in high-CO atmospheres are particularly susceptible to experiencing adverse health effects from CO. The evidence summarized above, and described more fully by EPA (2000a), indicates that attainment of the ambient-CO standards can decrease morbidity and mortality from atherosclerotic heart disease. Although less conclusive, there is evidence that attainment of the CO standards will also decrease morbidity from pulmonary disease, neurological disease, fetal loss, and childhood developmental abnormalities. These health benefits translate into economic savings associated with avoided health care and avoided work-time losses as well as intangible savings in life quality.
CO Exposure
Motor-vehicle emissions are the primary source of CO in outdoor air in populated areas and are associated with the highest outdoor CO exposure in nonsmokers. Outdoor concentrations of CO tend to be higher in urban areas and to increase with the density of vehicles and miles driven. Measurements of ambient CO typically exhibit a bimodal diurnal pattern, with the highest concentrations generally occurring on weekdays during the commuting hours
of 7:00–9:00 a.m. and 4:00–6:00 p.m. (EPA 2000a).2 CO also accumulates in the rider compartments of motor vehicles. Studies have shown that when the concentration near roadways averages 3–4 ppm, the average concentration in the cab is typically 5 ppm (Akland et al. 1985; Flachsbart et al. 1987).
Most people spend a majority of their time indoors; this is particularly true in Fairbanks and other cold climates during the winter, when ambient CO concentrations tend to be highest. That leads to the question of the relationship between indoor and outdoor concentrations. Air pollution in buildings can come from indoor sources and from air exchange with outdoor ambient pollution. Air exchange may be active, as in the case of a mechanical ventilation system, or passive, as in the case of infiltration associated with temperature or pressure differences between the outside and the interior of a building. CO penetrates freely with infiltration air from the outside and is not removed by building materials or ventilation systems. Furthermore, there are no effective indoor chemical or physical processes for lowering CO on the time scales of interest for exposure and toxic effects.
The relationship between indoor and outdoor CO concentrations can be evaluated with a simple differential mass-balance model (Shair and Heitner 1974) that has the following steady-state solution when we combine active ventilation and passive infiltration into a single air-exchange term:
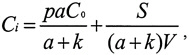
(1)
where
Ci=indoor concentration, μg/m3;
Co=outdoor concentration, μg/m3;
p=penetration coefficient, 0–1;
a=air exchange rate, h−1;
k=decay rate, h−1;
S=mass flux of the indoor source, μg/h; and
V=building volume, m3.
For CO, the relationship is simpler because the penetration coefficient (p) is unity and the decay rate (k) is effectively zero. Therefore, the solution is
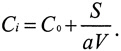
(2)
It is clear that in the absence of indoor sources (S), the steady-state indoor concentration of CO will equal the outdoor concentration. When a source of CO is present indoors (for example, from a faulty furnace, an underground parking garage, a kerosene heater, or a smoker), the indoor source adds to the background concentration from the outdoor air (EPA 2000a). Therefore, buildings do not provide protection from high outdoor concentrations of CO. The idea that buildings provide protection from high outdoor CO concentration is a common misconception.3
Related Pollutants
The incomplete combustion of fossil fuels, which is responsible for CO emissions, also causes emissions of fine particles (PM2.5) and toxic organic air contaminants. Epidemiological studies have linked exposure to PM2.5 with various adverse health effects, including premature mortality, exacerbation of asthma and other respiratory tract diseases, and decreased lung function (NRC 1998; EPA 2001b). Because of these adverse health effects, EPA issued NAAQS regulating ambient concentrations of PM2.5 (EPA 1997). In addition to the six criteria air pollutants previously regulated, the 1990 amendments of the Clean Air Act (CAAA90) designated 189 toxic air contaminants. Incomplete combustion in mobile sources is estimated to contribute a substantial fraction to the emissions of several toxic air pollutants, including benzene, 1,3-butadiene, and aldehydes (EPA 2001c). Each of these toxic air pollutants poses some carcinogenic risk. In addition, chronic exposure to benzene is associated with blood disorders; chronic exposure to 1,3-butadiene is associated with cardiovascular disease; and chronic exposure to aldehydes is associated with respiratory problems and eye, nose, and throat irritation (EPA 1994). Many emissions-control strategies for CO will also cause reductions in these copollutants and their associated adverse health effects.
CO may be a good indicator gas for other pollutants that are emitted at the same time but are not widely measured. The concentrations and spatial distributions of the copollutant species are generally not as well known as for CO. In particular, little data are available about exposure to air toxics present in the ambient environment. CO could be especially useful as an indicator of mobile-source emissions of PM2.5 and air toxics, which some studies have shown to be strongly correlated with CO. In one study of emissions from in-use vehicles sampled in Denver, San Antonio, and the Los Angeles area, strong correlations were found between CO and particle emissions (R2=0.65) and between particle and total hydrocarbon (HC) emissions (R2=0.78) (Cadle et al. 1999). The same study demonstrates that emissions of the pollutant species increase with vehicle age and during cold starts. For individual vehicles, however, the correlation among the pollutants is weaker, reflecting the complex mechanisms of formation of the related combustion products.
In another study, three vehicles from Fairbanks were evaluated with the Federal Test Procedure (FTP) for the Urban Dynamometer Driving Schedule (Mulawa et al. 1997). Particle emissions were higher at lower temperatures; the average PM emissions from the three tested automobiles (using regular gasoline) increased from 14.2 to 44.2 mg/mile as the temperature decreased from 20°F to −20°F. Most of the particles were released during Phase I of the FTP, representing the first 505 seconds during cold start. The particle emissions also correlated with HC and CO emissions and appeared to be related to rich-operating conditions. Mechanisms of PM formation may include oil consumption.
NATIONAL AIR QUALITY STATUS FOR AMBIENT CO
NAAQS
In recognition of the adverse health effects of CO, the U.S. Environmental Protection Agency (EPA), as directed by the Clean Air Act (CAA), established health-based air quality standards for CO in 1971. Recognizing that exposure can have both acute and chronic effects, the primary NAAQS for CO has two criteria with different averaging periods: 35 ppm averaged over 1 hour (h), and 9 ppm averaged over 8 h. Each standard is not to be exceeded more than once per year; the second exceedance and each subsequent exceedance
|
BOX 1–1 Recommendations: Health and Exposure Health Benefits from Meeting CO Standards To reduce the potential adverse health effects of CO, the borough will need to continue to make progress in meeting NAAQS for CO. Public-awareness campaigns regarding the public-health issues associated with ambient CO should be enhanced. Exposure During Episodes Additional efforts should be made to monitor personal exposures to CO in buildings and garages near high-CO areas, outside in residential locations, and in motor vehicles. Sampling of human exposure with personal monitoring, blood carboxyhemoglobin (COHb) measurements, or breath samples is recommended. Monitoring of CO concentrations in microenvironments and of human exposure to CO should be performed in conjunction with improved ambient monitoring to better characterize the CO problem in Fairbanks. Copollutants The expected adverse health effects of exposures to copollutants is another reason to consider enhanced CO controls. To assess the relationship between CO and copollutants in Fairbanks during winter, Alaska and the borough should implement an ambient monitoring program for toxic organic compounds and expand the monitoring of PM2.5. |
within a year are considered violations of the standard.4 The standards have been periodically reviewed on the basis of new scientific findings as mandated by the CAA. The most recent review was published in 2000, when the standards were reaffirmed (EPA 2000a).
The 8-h standard of 9 ppm is the more difficult to attain. EPA originally designated an area as “nonattainment” if the second highest 8-h CO concentration measured during a calendar year (termed the “design value”) was greater than 9 ppm. After the CAAA90, the EPA administrator designated each area that had previously been in nonattainment as “serious” if the design value was 16.5 ppm or greater, “moderate” if the design value was 9.1–16.4 ppm, “not classified” if recent data were insufficient to determine whether the standard was met, or in attainment. Moderate areas that did not reach attainment by July 1996 could be reclassified as serious by EPA. Each nonattainment area is required to submit to EPA a state implementation plan (SIP) that includes a characterization of pollutant concentrations and emissions, a description of the emissions reductions the area plans to make, and an “attainment demonstration” showing how the emissions reductions will enable the area to attain and maintain compliance with the NAAQS. To be eligible to request reclassification from nonattainment to attainment status, an area must have air quality monitoring data to show that it did not violate the standard for the previous 2 years (y).
Areas in Nonattainment for CO
Following the CAAA90, 42 areas were designated as nonattainment for the 8-h standard for CO. As of this time, seven areas remain in serious nonattainment status for the 8-h standard, although only three areas failed to meet the CO NAAQS in 1999–2000: Fairbanks, Alaska; Lynwood, California, in the South Coast Air Basin; and Calexico, California, near the Mexican border (EPA 2000b). All areas are in attainment with respect to the 1-h standard. Monitoring in Fairbanks in 2000 and 2001 did not show an exceedance, so the borough is now eligible to apply for attainment status. Lynwood apparently suffers from an older vehicle fleet with higher emissions and inhibited dispersion caused by strong inversions combined with low windspeeds at night (Bowen et al. 1996). Calexico suffers from cross-border pollutant transport and is not considered to be primarily a meteorological or topographical problem area. Continued reductions in emissions are expected in Fairbanks and Lynwood over the next decade as newer, cleaner vehicles replace older ones. However, unusual meteorological conditions or unexpected increases in emissions (in the case of Fairbanks, construction of a trans-Alaska natural-gas pipeline or substantial missile defense facilities) could prevent those areas from maintaining compliance.
The seven areas classified as being in serious nonattainment as of 2001 are listed in Table 1–1. In addition, for each area, the year from 1995 to 2000 that had the most exceedances is shown with the highest and second-highest 8-h average CO concentrations measured during that year. The highest 8-h average CO concentrations occurred early in the 6-y period in each area; this is consistent with the general decline in CO concentrations across the country during that period. The highest number of exceedances and the highest 8-h averages are associated with the Los Angeles South Coast Basin, and the Lynwood area in particular. Fairbanks ranks second and is unusual for such a small metropolitan area in having such a serious air quality problem. CO concentrations measured in Calexico, California, exceed the 8-h standard, but that area has not yet been classified by EPA.
METEOROLOGY AND TOPOGRAPHY IN CO NONATTAINMENT AREAS
When ventilation in a region with high CO emissions from a nearby source is restricted, CO can accumulate in the air near the ground. The air can be trapped vertically, by temperature inversions, and horizontally, by topography or stagnant meteorological conditions. Stagnation is characterized by very low windspeeds. Figure 1–2a,b illustrates air-stagnation cases in the continental United States (Wang and Angell 1999). The Southwest and parts of the Gulf Coast are clearly susceptible to air stagnation.
Vertical ventilation depends on how air temperature varies with altitude. The average temperature in the troposphere decreases with altitude at about 6.5°C/km, as shown in Figure 1–3 (Huschke 1959). Under some meteorological conditions, temperature in the lower atmosphere may increase with altitude for short distances. Such temperature inversions inhibit vertical mixing be cause less-dense warm air rests above colder, denser air. The temperature inversion therefore defines the vertical limit of mixing. For example, as shown in Figure 1–3b, pollutants emitted at the surface could be mixed up to about 600 m. The inversion strength depends on the rate at which temperature increases with altitude; stronger inversions have a more rapid increase of temperature with altitude.
As illustrated in Figure 1–3a,b, inversions can be surface-based and due generally to surface cooling or can be at a high altitude and due generally to horizontal advection of warm air aloft or subsidence (downward motion of air). An existing inversion can be strengthened by atmospheric subsidence in
TABLE 1–1 U.S. Areas Classified As Serious Nonattainment for CO As of 2000
|
|
During the Worst Year in 1995–2000 |
||||||
|
Area Name |
Population (1990) |
Areas (mi2) |
Number of Monitors |
Worst Year in 1995–2000 |
Highest 8-h Average CO (ppm) |
2nd Highest 8-h Average CO (ppm) |
Number of exceedancesa |
|
Fairbanks, AK |
|||||||
|
City |
30,000 |
32 |
3 |
1995 |
15.2 |
11.8 |
9 |
|
Borough |
|||||||
|
|
80,000 |
7,366 |
|
||||
|
Anchorage, AK |
222,000 |
43 |
3 |
1996 |
11.0 |
9.6 |
3 |
|
Las Vegas, NV |
258,000 |
1,320 |
7 |
1996 |
10.3 |
10.1 |
3 |
|
Spokane, WA |
279,000 |
161 |
7 |
1995 |
13.1 |
11.2 |
4 |
|
Denver-Boulder, CO |
1,800,000 |
1,500 |
7 |
1995 |
11.0 |
9.5 |
2 |
|
Phoenix, AZ |
2,006,000 |
2,580 |
9 |
1995 |
10.2 |
9.9 |
3 |
|
LA-South Coast, CA |
13,000,000 |
6,000 |
14 |
1996 |
17.5 |
14.5 |
22 |
|
aNumber of times 8-h average CO concentrations in the area were greater than of equal to the standard of 9 ppm. Source: Larry Elmore, EPA, Office of Air Quality Planning and Standards. |
|||||||
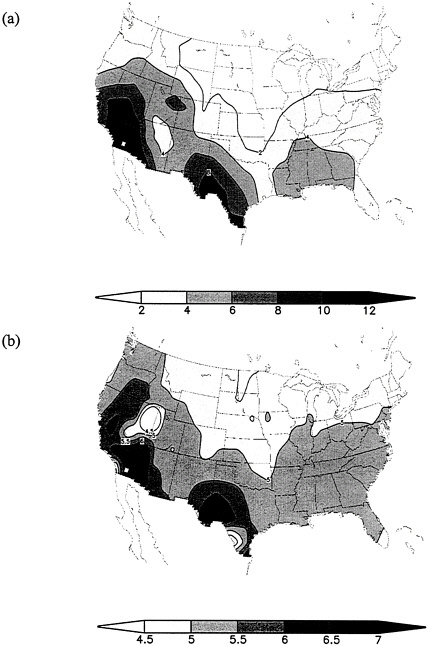
FIGURE 1–2a,b (a) Air stagnation annual mean cases for 1948–1998 for the contiguous 48 states, (b) Mean duration of stagnation cases, in days. Air is considered stagnant if for at least 4 d sea level geostrophic windspeeds are less than 8 m/s, there is no precipitation, and the winds at 500 hPa are less than 13 m/s.
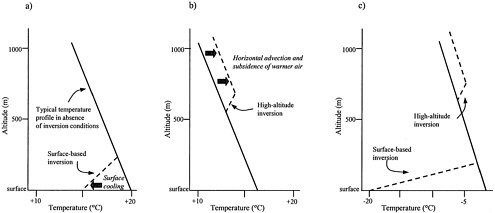
FIGURE 1–3 Schematics of (a) a surface-based inversion, (b) a high-altitude inversion, and (c) a typical Alaskan temperature profile with both surface-based and high-altitude inversions. Solid lines indicate the temperature profile in the absence of inversions. Dashed lines indicate temperature profiles affected by inversions.
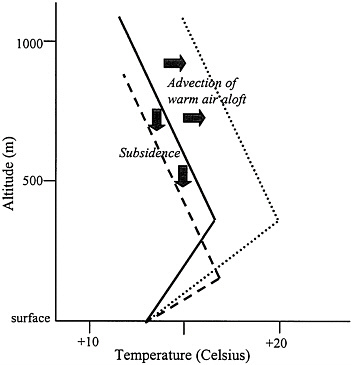
FIGURE 1–4 Schematic of how an existing surface-based inversion (solid line) can be strengthened by subsidence (dashed line) or by advection of warm air aloft (dotted line).
the region or by the transport into the region of air aloft that is warmer than the air at the surface (Figure 1–4). Indeed, regions under high-pressure systems, where subsidence and the advection of warm air aloft can both be present (Figure 1–5), typically experience inversion conditions. Thus, urban locations that have frequent high-pressure systems often suffer from serious air pollution because inversions limit the vertical mixing of pollutants (Pielke et al. 1987; Stocker et al. 1990).
In central Alaska, the combination of clear skies, light surface winds, snow cover, and the absence of solar heating during the winter often produces exceptionally strong surface inversions (Figure 1–3c). Radiation of heat to space produces low surface temperatures that can trap air and pollutants during high-pressure subsidence or warm-air advection from the south. It is likely that warm-air advection aloft from the south is necessary to produce CO exceedances in Fairbanks (S.A.Bowling, Fairbanks Meteorology, in prepara-
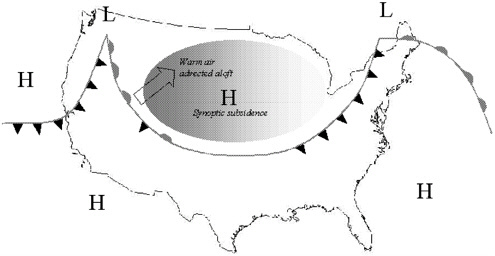
FIGURE 1–5 Schematic of how major synoptic weather features can strengthen inversions. Areas of high pressure (indicated by “H”) experience synoptic subsidence. At the same time, warm fronts (indicated by semicircles) advect warm air above the surface. The shaded area indicates a region where existing inversion conditions can be strengthened by both subsidence and advection of warm air aloft.
tion, 2002). In contrast, southwestern California experiences inversions at higher altitudes because of strong subsidence associated with descending air in the subtropical eastern Pacific Ocean high-pressure system. Inversions in southwestern California are high because, in contrast with the winter situation in Fairbanks, sunshine on the surface permits vertical mixing in the atmospheric layer near the surface. Topographical features, such as the presence of an urban area in a valley, can inhibit horizontal dispersion of air. Unless the large-scale wind is strong enough to enable horizontal dispersion, the local air mass will remain confined to the valley (see Figure 1–6). Los Angeles is subject to that problem because it has mountains on three sides. Similarly, Fairbanks has high terrain to its west, north, and east. Because cities tend to develop at lower elevations, often in river valleys, terrain trapping is a concern for many urban areas. Valleys well above sea level have an added problem in that it is easy to lose heat to space at high elevations. Pielke et al. (1991) proposed a method to estimate worst-case air quality in complex terrain.
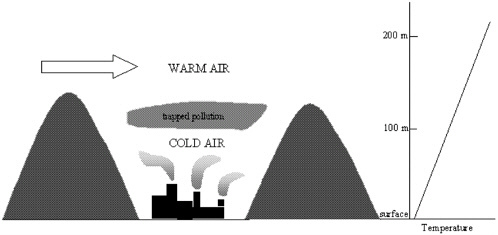
FIGURE 1–6 Schematic of a trapping valley. The temperature profile in the valley is shown on the right.
NATIONAL INVENTORY OF CO EMISSIONS
An emissions inventory is an accounting of all emissions of a pollutant in a defined area in a specified period. It is common to calculate the annual emissions of CO in nonattainment areas; the estimates are used to compare emissions in various years and locations, assess the effectiveness of air pollution policies, and predict the impact of future projects on emissions. Determining an emissions inventory requires that all major sources of a pollutant be identified, that the releases from the sources be quantified, and that the releases be summed to determine the total mass emitted in the defined area. Major sources are usually quantified with the aid of emissions rates, which are multiplied by an activity rate to yield the mass of emissions released.
For example, annual highway vehicle emissions are commonly determined by multiplying the emissions rate, in grams per mile, by the number of miles traveled in a year. Likewise, emissions from oil heaters are usually determined by multiplying the emissions rate, in grams per gallon of heating oil, by the number of gallons of heating oil used.
Table 1–2 is an inventory of CO emissions in the United States during 1999 (EPA 2001a). An estimated 77% of the anthropogenic CO emissions are
from mobile sources, including onroad vehicles (51% of the total) and nonroad engines and vehicles (26% of the total). The remaining CO emissions are from area and point sources, including fuel combustion and industrial processes. In urban areas, mobile sources may contribute relatively more or less than the national average to the mix of emissions. For example, mobile sources contributed 89% of CO emissions in Anchorage, Alaska, but contributed only 76% of CO emissions in Denver, Colorado. On the basis of its Mobile Source Emissions Factor (MOBILE) model, EPA suggested that vehicles may contribute 90% or more of CO emissions in cities with serious air pollution (EPA 1993). As described in NRC’s report (NRC 2000), however, studies evaluating the MOBILE model have shown substantial inaccuracies in its estimates of fleet emissions and the effectiveness of control strategies.
CO EMISSIONS FROM VEHICLES
The primary source of CO from vehicles is the incomplete combustion of gasoline in engine cylinders. The fuel-oxidation process (combustion) is the conversion of the fuel to lower-molecular-weight intermediate HCs (including olefins and aromatics) and their conversion to aldehydes and ketones, then to CO, and finally to carbon dioxide (CO2). The initial reactions are faster than the final conversion of CO to CO2. Incomplete conversion of fuel carbon to CO2 results in part from insufficient O2 in the combustion mixture—known as fuel-rich5 conditions—and insufficient time to oxidize fuel carbon fully to CO2. CO emissions by diesel vehicles are minimal, primarily because of the excess air used in the diesel combustion cycle. Hence, the following discussion is limited to gasoline vehicles.
Vehicle Technologies
Before the 1980s, carburetors were used to meter fuel in proportion to the intake air of gasoline engines. The design of the carburetor typically ensured
TABLE 1–2 National CO Emissions Inventory Estimates for 1999
|
Source Category |
Thousands of Short Tons |
|
Point- or Area-source fuel combustion |
5,322 |
|
Electric utilities |
445 |
|
Industry |
1,178 |
|
Residential wood burning |
3,300 |
|
Other |
399 |
|
Industrial processes |
7,590 |
|
Chemical and allied product manufacturing |
1,081 |
|
Metals processing |
1,678 |
|
Petroleum and related industries |
366 |
|
Waste disposal and recycling |
3,792 |
|
Other industrial processes |
599 |
|
Onroad vehicles |
49,989 |
|
Light-duty gas vehicles and motorcycles |
27,382 |
|
Light-duty gas trucks |
16,115 |
|
Heavy-duty gas vehicles |
4,262 |
|
Diesels |
2,230 |
|
Nonroad engines and vehicles |
25,162 |
|
Recreational |
3,616 |
|
Lawn and garden |
11,116 |
|
Aircraft |
1,002 |
|
Light commercial |
4,259 |
|
Other |
5,169 |
|
Miscellaneous |
9,378 |
|
Slash or prescribed burning |
6,152 |
|
Forest wildfires |
2,638 |
|
Other |
588 |
|
Total |
97,441 |
|
Source: EPA 2001a. |
|
that the mixture of air and fuel was adequate to provide vehicle performance, but the carburetor could never be truly optimized for emissions control across the entire operating range of the engine. Carbureted engines featured an automatic choke that increased the fueling rate during cold start and initial operation. Depending on choke calibration, CO and HC emissions could be substantial during cold starts. This type of nonfeedback fuel metering is termed an open-loop system.
Since the middle 1980s, modern computer-controlled engines have used electronic fuel injectors rather than carburetors to deliver fuel to cylinders in automobiles and most light-duty trucks. Using closed-loop control, the engine computer system reads the signal from an O2 sensor in the exhaust system and adjusts the air-to-fuel ratio to help maintain stoichiometric combustion. That feedback provides just enough air to combust the fuel but maintains the maximal catalytic-converter efficiency for control of CO, HC, and nitrogen oxides (NOx).
During hard acceleration and high-speed operations, however, engine computers often use fuel-enrichment strategies to enhance engine performance for short periods and to protect sensitive engine components from high-temperature damage. Likewise, fuel-enrichment strategies are often used during cold starts (as discussed later). Thus, in modern engines, CO emissions are prominent primarily during enrichment associated with heavy loads, hard accelerations, and cold starts.
The onboard diagnostics system installed in model year 1996 and newer vehicles, known as the OBDII system, can help to detect problems during vehicle operations that increase CO emissions. The OBDII system uses sensors to monitor and modify the performance of the engine and emissions-control components. The onboard computer detects signals from the sensors to identify sensor and control-system failures, illuminating the malfunction indicator light on the vehicle dashboard and storing the fault codes (know as diagnostic trouble codes) for later analysis. In a garage setting, mechanics can download the OBDII fault codes from the onboard computer with a diagnostic analyzer (“scan tool”). The codes identify emissions-control systems and components that are malfunctioning. However, some components of OBDII systems (such as exhaust-gas recirculation and O2 sensors) are often disabled by the engine computer under conditions in which the manufacturer cannot guarantee the components’ performance (J.Cabaniss, Association of International Automobile Manufacturers, personal communication, July 10, 2001). That tends to be the case for vehicles operating at temperatures below 20°F. It may be prudent to take steps to ensure that manufacturers certify their OBDII systems to lower operating temperatures than currently required. This
is especially true if many northern locations begin adopting OBDII inspection and maintenance (I/M) systems, where the OBDII system is relied upon to determine whether a vehicle fails or passes an emissions test. When a significant number of sensors become inoperative, the OBDII system will also have less ability to alert vehicle owners of potential emissions-system failures.
Cold-Start Emissions
Under cold-start conditions, the engine computer commands the fuel injectors to add excess fuel to the intake air to ensure that enough fuel evaporates to yield a flammable mixture in the engine cylinders. A typical engine-computer strategy injects several times the stoichiometric amount of fuel during the first few engine revolutions, using a fixed fueling schedule to reach idling conditions. Excess fuel continues to be injected until the engine and O2 sensor are warmed up and the exhaust-catalyst inlet temperature reaches about 250–300°C (482–572°F), sufficient for the catalyst to oxidize CO to CO2. This open-loop operation, before the catalyst reaches peak efficiency, can continue for several minutes at low ambient temperatures. It is responsible for most of the emissions of CO, air toxics, and unburned HCs from properly operating modern vehicles. Once the engine and emissions-control systems are warmed up, combustion becomes stoichiometric, and CO is converted to CO2 in the catalyst, keeping CO emissions very low under typical operating conditions. Typical warmup times under mild ambient conditions, around 70– 80°F, can be about 1 minute (min) for modern catalysts and even as short as a few seconds for modern close-coupled catalysts (catalysts close to the engine). When ambient temperatures are −20°F or lower, however, catalyst and engine warmup times can exceed 5 min (Sierra Research 1999).
The amount of fuel enrichment required for a cold start of the engine is a function of engine design and ambient air and coolant temperatures. As the temperature in the combustion chamber gets lower, gasoline vapor pressure decreases, and additional fuel is required to ensure ignition. Gasoline sold during the winter in cold climates has a higher vapor pressure than gasoline sold in the summer to assist cold-start ignition. In summer, the vapor pressure is reduced to minimize evaporative emissions.
Below about 0°F, engine starting becomes more difficult, so external means, such as adjusting the fuel vapor pressure, must be devised to ensure that enough fuel vaporizes. A popular method for assisting engine startup is to use engine-block heaters, which heat the engine coolant to allow easier cold
starting. These plug-in heaters reduce the time for engine and catalyst warmup under cold conditions and so help to avoid the extremely high enrichments used by fuel-management control units at low temperatures. Use of plug-in heaters in very cold conditions is also an effective method of reducing CO emissions (Sierra Research 1999).
New-Vehicle Certification Programs
Emissions of CO during cold-start conditions and hot stabilized operations are most efficiently controlled through engineering design, that is, by the original equipment manufacturers, in new vehicles. The most important and effective vehicle emissions-control strategy for CO has been the nationwide introduction in 1980 and 1981 of more stringent vehicle certification requirements designed to reduce exhaust emissions of CO, HCs, and NOx. In 1981, EPA introduced emissions certification standards for passenger cars that provided a 3.4-g/mile (mi) limit for CO. That limit is still in place. The new-vehicle certification program appears to have contributed to reductions of about 36% in CO emissions from mobile sources from 1980 to 1999 (EPA 2001a).
Though the emissions certification standards for CO for passenger cars have not changed, there have been myriad regulations resulting in reductions in CO from the vehicle fleet. HC emissions were greatly reduced by Tier 1 vehicle emissions certification standards (introduced in model year 1994) and the National Low Emissions Vehicle (NLEV) standards (introduced in model year 1999). Because CO is readily reduced by the same vehicle emissions-control technology that reduces HC, more stringent HC standards have resulted in concomitant reductions in CO. The Tier 1 standards also required emissions-control system durability to increase to 100,000 mi (from 50,000 mi), which improved the robustness of emissions-control systems and tightened CO standards for some light-duty trucks. Tier 2 vehicle emissions certification standards, which will be introduced in model year 2004, will require that all light-duty trucks meet the CO standards for passenger cars. In addition, the Tier 2 standards also greatly reduce the sulfur content of gasoline. As discussed in Chapter 2, reducing fuel sulfur content improves catalyst performance, thereby reducing CO emissions. Thus, the Tier 1, NLEV, and Tier 2 vehicle-emissions and fuel standards have provided and will continue to provide improvements to vehicle CO emissions performance as the fleet turns over.
Since 1994, new cars have been required to meet a CO limit of 10 g/mi on the same FTP cycle conducted at 20°F (40 CFR Subpart C 86.201–94, pages 544–554). However, it is uncertain whether these vehicles achieve the same low emissions at substantially lower operating temperatures. The lower-temperature cold-start test represents only the upper boundary of the temperature range of concern in Fairbanks. Furthermore, some emissions-control-system sensors and actuators are inaccurate at very low temperatures, and the engine computer might ignore their input signals until the engine warms up (J. Cabaniss, Association of International Automobile Manufacturers, personal communication, July 10, 2001). Additional studies on the emissions behavior of vehicles at temperatures below 20 °F should be conducted to establish the effect of these extreme temperatures on emissions and control devices. Studies on after-market technologies that could provide benefits similar to those that would be achieved with an enhanced cold-start test should also be undertaken. It should be noted that EPA does not perform in-use vehicle testing to confirm that vehicles certified to the low-temperature standards are meeting those standards.
Reducing the 10 g/mi limit for the 20°F cold-start test or reducing the test temperature may provide additional CO emissions reductions for cold northern regions, such as Fairbanks. Indeed, CAAA90 mandated that, if six or more areas were designated as nonattainment as of July 1, 1997, EPA require cars to meet a cold-start emissions limit of 3.4 g/mi. EPA has yet to make a formal determination of the number of nonattainment areas as of July 1, 1997. A question to be addressed is whether requiring all automobiles sold nationwide to meet a more stringent cold-start test may be an ineffective use of resources, in that it would impose additional emissions controls on vehicles that might never be used in cold climates.
Strategies to Address Emissions-Control Failures
Although progress has been made in controlling CO emissions from vehicles, some problems remain. One concern is that failures in the operation of emission-control systems typically default to fuel-rich conditions that produce higher CO emissions while allowing engine performance to be maintained. For example, when O2 or temperature sensors are defective, engine computers may default to fuel-rich conditions. Similarly, defective fuel injectors may result in higher CO emissions. A weak spark ignition can cause hard starting, misfires, and poor performance and result in increased CO emissions. Other
common failures in the emissions-control system that affect CO emissions are O2-sensor deterioration, air-injection system defects, and catalyst deterioration. Because engine failures are unavoidable and many such failures cause higher CO emissions, a strategy is needed to identify vehicles with unacceptably high emissions.
Many urban areas use inspection and maintenance (I/M) programs to identify the highest-emitting vehicles and require that they be repaired. High emitters exhibit increased emissions under almost all onroad operating conditions because of failure in emissions-control or fuel-control systems. Not surprisingly, cold-start emissions from high-emitting vehicles are typically also substantially increased. Thus, identification of vehicles as high emitters during the summer months and their repair before winter can yield significant reductions in cold-start emissions. However, some emissions-control system failures may go undetected during an I/M test of a vehicle’s tailpipe emissions under idle conditions. Placing a vehicle under a driving load, such as by testing it on a dynamometer, as is done in the IM240 test, has a higher probability of revealing an O2-sensor malfunction and other defects. Remote-sensing systems, which determine CO emissions by measuring absorption of infrared radiation from a beam directed across the roadway, can also be used to identify high emitters (NRC 2001).
AIR QUALITY MODELS
Air quality modeling is an essential element of air quality management. Models can be used to demonstrate attainment of the NAAQS, evaluate the effects of new construction projects, and conduct further research into what causes pollution episodes and how to predict them. A number of modeling techniques—requiring various levels of scientific expertise, input data, and computing resources—are available for those purposes. The simplest models assume a direct correlation between emissions and ambient pollutant concentrations; the most complicated models resolve temporal and spatial variations in pollutant concentrations and include the effects of meteorology, emissions, chemistry, and topography. Models are also characterized by the size of the problem they address: microscale models simulate pollution from an intersection or point source, mesoscale models simulate metropolitan or multistate pollution, and large-scale models simulate continental or global pollution.
In the attainment demonstration presented in their SIPs, states are required by EPA to model how emissions reductions will lead to the desired air quality
improvements. Three types of models have been used to demonstrate attainment of the CO NAAQS: statistical rollback, Gaussian dispersion, and numerical predictive models. The simplest is a statistical rollback model in which the needed reduction in emissions is assumed to be proportional to the required reduction in ambient CO concentrations (ADEC 2001a):
where
CObaseyear=the second highest 8-h average in the base year;
CONAAQS=the NAAQS of 9 ppm (or sometimes 9.4 ppm); and
CObackground=an average regional background CO in the absence of emissions.
Because no information is needed about meteorology or the spatial distribution of emissions in a nonattainment area, EPA has allowed states to use rollback models to demonstrate attainment in smaller cities, rather than the more resource-intensive dispersion and urban-airshed models described below. Although easy to implement, rollback models do not explicitly consider the role of meteorology or the spatial heterogeneity of CO emissions and concentrations.
A second type of model that has been used for CO-attainment demonstrations is a Gaussian dispersion model, which is typically used to simulate CO concentrations for microscale analysis in the vicinity of intersections or along major traffic corridors (EPA 1992). These models simulate how a pollutant is dispersed into the immediately surrounding atmosphere. They assume that the atmospheric concentration of the pollutant is proportional to its emissions and inversely proportional to windspeed and that the resulting spatial distribution of the pollutant is Gaussian (Wayson 1999). Inputs for dispersion models include meteorological data, such as windspeed and inversion strength in the vicinity of the pollutant source, and temporally resolved emissions. Because predicted concentrations are directly proportional to emissions, the accuracy of emissions measurements is crucial to the modeling process. In the case of modeling of intersections, the emissions inventory may be derived from information about traffic patterns, mean speeds, and vehicle-fleet composition. Larger cities have also used Gaussian dispersion models to evaluate the air quality effects of increasing road capacity or other large construction projects (EPA 1992).
Box models, another tool available for microscale analysis of air pollution,
have not been used extensively for SIP attainment demonstrations. The “box” is some volume of air into which emissions are injected and in which chemical transformation may take place and air is exchanged with the environment. The air in the box is assumed to be well mixed, so spatial variations in emissions or pollutant concentrations on scales smaller than the box model are not resolved. Box models are particularly useful for understanding how various emissions scenarios and meteorological conditions affect pollutant concentrations. For example, a box model for CO in Anchorage, Alaska, has been used to quantify how mechanical turbulence from roadway traffic might increase the mixing height and reduce CO concentrations on severe-stagnation days (Morris 2001). Limitations of the box-model approach include an inability to include spatial variations and a dependence on assumptions to represent meteorological parameters. Chapter 2 presents a simple box model developed for the Fairbanks nonattainment area.
The most complicated models used for attainment demonstrations simulate how a pollutant concentration varies with time and space over an entire urban area. These numerical predictive models, generally intended for mesoscale analysis, can simulate emissions from multiple sources and the dispersion, advection, and photochemical reactions of gaseous pollutants in the atmosphere. Numerical predictive models, such as the Urban Airshed Model (UAM), have been used for many years to simulate O3, which is an areawide or mesoscale pollutant. The UAM has also been adapted to simulate CO in Denver, Colorado (Colorado Department of Public Health and Environment 2000). Because of the local nature of high-CO episodes, extensive modeling of the entire urban airshed may be unnecessary for CO-attainment demonstrations. Airshed modeling is resource-intensive, requiring detailed knowledge of an area’s meteorology (usually based on the output of a mesoscale weather model constrained by observations), spatially and temporally resolved emissions inventories, and measurements of the pollutant at several locations to allow model evaluation. Highly trained personnel are needed to conduct the simulations. However, a simplified approach of this method may be appropriate in some cases.
More complicated models are not always appropriate for attainment demonstrations, but they can be valuable in improving our understanding of the interactions among atmospheric processes. Even better research tools than the numerical predictive models describe above (such as the UAM) are process numerical models, which allow coupling between processes specific to air quality modeling and meteorology. Process numerical models typically are formulated by adding pollutant emissions, chemistry, and transport into an existing meteorological model rather than simply using the meteorological
data as a model input. The relatively nonreactive behavior of CO makes it an ideal chemical species for simulation in a weather model. Predictions of CO, for example, can be straightforward in the National Weather Service Eta Model, which has a horizontal grid framework of 12×12 km over the contiguous United States.
Despite advances in air quality modeling capabilities over the last 30 y, many improvements are still possible and needed, particularly in the numerical predictive models, which are used more widely than process numerical models. One problem is that the vertical and horizontal resolution of both types of models is too coarse to capture the variability in pollutant concentrations, which is necessary to identify local hotspots. Most numerical predictive and process numerical models are based on statistical representations of atmospheric motion on scales smaller than the spatial resolution of the models. When unusual meteorological conditions occur, the validity of these representations becomes questionable and could lead to errors in the prediction (Pielke 2002). Models used for regulatory purposes can suffer the loss of realism as a result of such shortcomings.
Various models have been applied to predict future pollutant concentrations, particularly with the goal of identifying conditions that might create an episode. Numerical predictive models can be used, as can simpler empirical models, which attempt to identify statistically significant relationships between specific air quality variables and a set of predictors. Empirical models typically use regression or neural-network techniques to develop a relationship based on observations of meteorological variables and pollutant concentrations. Future air quality can be predicted by using the output of weather-forecast models as values for the predictors. Meteorological forecasts are disseminated daily by the National Weather Service (Hooke and Pielke Jr. 2000), but only a few areas in the country provide short-term air quality forecasts, usually only for O3, which is an areawide pollutant.
SUMMARY
CO is a pollutant that impairs the ability of blood to carry O2 to body tissues. Exposure to CO at sufficiently high concentrations can cause headaches, exacerbate heart problems, lengthen reaction times, and affect fetal development. On the basis of a compilation of scientific knowledge about the relationship between various concentrations of ambient CO and their adverse health effects, EPA set CO standards of a maximum 1-h average concentration of 35 ppm and a maximum 8-h average concentration of 9 ppm. Even though
the standards are for outdoor air, they help to improve indoor air. CO remains in the outdoor air long enough to penetrate the indoor environment and is not removed by filtration. Because the measures taken to reduce CO emissions typically result in reduced emissions of copollutants, such as PM2.5 and air toxics, the health benefits of CO reductions can be greater than those associated with reduced CO concentrations alone.
Controls on CO emissions, particularly in the form of improved vehicle technology, have led to significant reductions in ambient CO concentrations throughout the United States. However, a few locations still experience concentrations that approach or exceed the CO 8-h health standard. Most of those areas have meteorological conditions (such as frequent inversion or stagnation conditions) or topography (such as being situated in a mountain valley) that inhibit ventilation and allow CO to accumulate at high concentrations near the surface. One such location is Fairbanks, Alaska. The task of the Committee on Carbon Monoxide Episodes in Meteorological and Topographical Problem Areas was to assess approaches for predicting, assessing, and managing episodes of high CO concentrations in meteorological or topographical problem areas. The committee’s charge called for Fairbanks to be the focus of this interim report. The rest of this report describes the CO problem there, including its physical characteristics, the emissions-control strategies used there, and the prospects for the area to remain in attainment with the NAAQS for CO.






























