2
Improving Chemical Oceanographic Data
Consistently comparable data collected on a global scale and over time are essential for the global monitoring programs and process studies described in the previous chapter. Individual investigators involved in mechanistic studies will also benefit from consistent and comparable analytical results. Although the oceanographic community has a strong record of emphasis on analytical quality control focused on precision, knowledge of the accuracy is often missing due to a lack of reference materials. Routine use of appropriate reference materials allows systematic analytical offsets to be recognized and related to known procedures or sample matrices. Availability of reference materials may actually encourage development and adaptation of new analytical methods as it will allow accurate and consistent comparisons. Bringing about community-wide use of reference materials on a routine basis is in part a matter of education. A key first step is to bring the issue to the attention of the oceanographic community, and to make the many advantages of comparative analysis clear. Example and peer pressure, especially from small groups of analysts that are already associated by common measurements, should also help. In addition, peer review of proposals and publications should also place more emphasis on the use of reference materials.
MATRIX DEPENDENCE OF REFERENCE MATERIALS
The subsequent chapters of this report are organized on the basis of matrix (e.g., seawater versus sediment), as opposed to analyte or mea-
surement method. This choice is based on several considerations. Most importantly, seawater matrices must be sampled, processed, and analyzed differently than either marine sediments or particulate matter. Seawater is dominated by dissolved salts that comprise 3.5 percent of the fluid by weight and profoundly affect its physical properties (e.g., density, viscosity, and refractive index) as well as the types and rates of chemical reactions in solution. In contrast, marine sediment and other particulate matrices are dominated by minerals (typically calcium carbonates, opal or detrital aluminosilicates). Whereas seawater exhibits relatively constant compositions of the major ions (those present in amounts greater than 1 mg/kg), sediments are extremely variable in the types of minerals they contain and the chemical conditions encountered among and within deposits. Because it is seldom possible to quantitatively separate an analyte from its seawater or sediment matrix, the background ions and minerals must be carried through a major portion of most preparation and analytical procedures, where they can cause a broad range of complications and compromises.
A final rationale for the report’s matrix-based organization is that seawater or sediment samples are often analyzed for different reasons by different types of oceanographers who may have little interest in another matrix. In general, analyses of seawater components address biological (e.g., photosynthesis/respiration), chemical (e.g., scavenging, reactions and mixing), and/or physical (e.g., circulation/mixing) processes imprinted over time scales of less than 1000 years, the mixing time of the global ocean. Particulate matter reflects biological and chemical processes in the water column corresponding to time periods significantly less than 100 years. Sediments are typically studied as temporal records of events occurring in the water column over time by sampling the accumulating deposit and can represent much longer time periods, extending up to millions of years. These contrasting oceanographic perspectives share a susceptibility to artifacts deriving from the unique matrices (whether seawater or sediment) from which chemical information is extracted.
HOW REFERENCE MATERIALS WORK
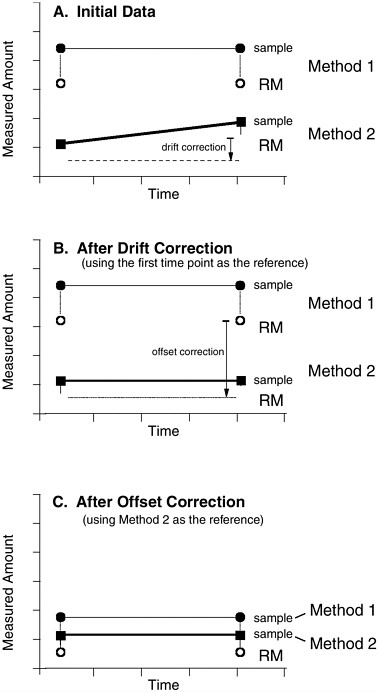
Routine use of reference materials can help tie together data sets, allowing us to answer key biogeochemical questions, even when the mechanisms of matrix effects and other analytical complications are not fully understood. This fundamental advantage can be illustrated by considering a scenario (Fig. 2.1) in which two different analytical methods for the same analyte are used to characterize an environment at two different times. In this example, the original data set (Fig. 2.1A) is ambiguous regarding whether the measured environmental variable has changed
over time or whether the measurement method itself caused this shift. Method 1 indicates the concentration has remained constant with time, whereas Method 2 suggests it has changed. Parallel analysis of a reference material (whose composition remains unchanged over time) shows not only that Method 2 has drifted but also the extent of that shift. The drift-corrected data obtained by both methods now indicate that the concentration of the target analyte has not varied in the sample over time (Fig. 2.1B). Even after drift correction, the two methods measure very different analyte concentrations. Because the sample measurements are linked by simultaneous analyses of the reference material, however, the paired data show that the different concentrations measured by Methods 1 and 2 largely result from a systematic offset between these two procedures. Once this offset is removed (in this case, by adjusting all measurements made by Method 1 downward until the reference material values for both methods coincide), the sample measurements agree much more closely between methods (Fig. 2.1C). The overall result is that data obtained using the two methods can be confidently used together to constrain the value of the sample over time.
Reference materials can also provide a convenient means of identifying troublesome matrix effects and for probing the possible mechanisms for a given analytical method. For example, measurements of the same analyte in a variety of samples with different commonly-encountered matrices provide a powerful means of identifying unusually large offsets between different analytical methods. Such sample-dependent inconsistencies serve as a warning that at least one of the compared methods is unusually sensitive to some characteristic of the sample matrix. Because sample matrix effects can be extremely complex and analyte-specific, it is highly advantageous for the oceanographic community to investigate these complications using a small number of widely-shared reference materials. In turn, these reference materials should represent the major matrix types encountered in natural samples, and they should contain a wide array of compounds in various physical forms and stages of degradation.
LIMITATIONS OF REFERENCE MATERIALS
The availability of reference materials or standards will not solve all the analytical problems faced by the marine community. In addition to using reference materials, the use of agreed-on common collection and analytical methods can also improve the chemical data being collected by oceanographers. Standardization of these methods minimizes the variability that may result from differences in laboratory procedure. A major disadvantage of method standardization, however, is that it can discour-
age the development of improved analytical methods. In the specific case of oceanographic studies, where accurate analyses of major elements in many commonly encountered matrices are not yet routine, it is therefore premature to standardize measurement methods for most analytes. Nevertheless, even in the absence of reference materials, there are well-developed approaches to the quality control of measurements. For example, careful attention needs to be paid to (1) blanks, (2) calibration, (3) replicate analyses, and (4) where practical, the use of “spike recovery” to assess the quality of analyses.
Different sampling methods often result in collection of different components of the element or compound of interest. Most seawater samples are collected in bottles, filtered to remove particles and analyzed directly or after preconcentration of minor components. Particulate matter from the water column is collected by filtration or with sediment traps. Sediment samples are collected in cores that recover intact chronological sequences and are commonly subsampled, dried and ground to a powder. Pore waters are extracted from sediments by squeezing or suction, dialysis, and centrifugation.
One example of a collection issue that cannot be solved using reference materials is particulate matter filtration. This widespread pretreatment has led to the operational definition that “particulate” material is retained by a filter (commonly between 0.2 and 0.7 µm pore-size), whereas “dissolved” material passes through the filter. Although this definition was arbitrarily based on the minimum pore size filters available to early oceanographers, it allows most living organisms (except smaller bacteria and viruses) and particles of sufficient size to sink to be concentrated and separated. The choice of filters and even the filtering technique can greatly affect the composition of the sample that is analyzed. Studies of colloidal matter and its importance in aqueous processes are still at an early stage, but will have additional impacts on the separation into “dissolved” and “particulate” materials.
A current example of these filtering artifacts is the measurement of particulate organic carbon (POC) and chlorophyll concentrations in the water column. Because organisms vary in size and fragility, the choice of filter can determine the types of organisms that pass through or are retained, and thus the measured concentrations of both the particulate and dissolved species of interest, while filtration pressure can influence whether the organism is ruptured during the process. The size of the water sample can affect the analysis for materials that adsorb onto the filter; for example, dissolved organic carbon (DOC) can adsorb onto filters and may bias particulate organic carbon analysis in very small samples. Standardization of collection methods allows many laboratories to measure analytes in the same particulate component, but certain ques-
tions may require measurement of a different component. The best way to resolve this type of problem is for authors to report in detail their collection methods so that later investigators will know what component was being measured and can draw their own conclusions. Journals should be encouraged to recognize the need for publication of such information.
BENEFITS OF REFERENCE MATERIALS
The following boxes present four case studies to illustrate how the introduction of reference materials has decreased the uncertainty of the chemical oceanographic measurement of salinity (Box 2.1), DOC (Box 2.2), and dissolved inorganic carbon (DIC) (Box 2.3). Box 2.4 illustrates the acute need for pigment reference materials, which are currently unavailable.
|
Box 2.1 Case Study: Salinity (S) Early chemists found that the relative composition of the major components of seawater were constant (Marcet, 1819), meaning that the measurement of one component of seawater could yield values for many of the remaining elements (Millero, 1996). It also meant that one could determine the total dissolved salts or salinity (S, parts per thousand, ‰) in seawater from the measurement of one element. The element of choice was chlorine. Knudsen and co-workers defined chlorinity (Cl) as “the chlorine equivalent of the total halide concentration in parts per thousand by weight, measured by titration with silver nitrate (AgNO3) solution” (Forch et al., 1902). The salinity determined from the evaporation of seawater (“Salinity is to be defined to be the weight of inorganic salts in one kilogram of sea water, when all the bromides and iodides are replaced by an equivalent amount of chlorides, and all carbonates are replaced by an equivalent quantity of oxides.”) was directly related to chlorinity Cl (‰) as measured above. For over 60 years researchers used that relationship, as expressed in the following equation, to determine the salinity and subsequent density of ocean waters: S (‰) = 0.03 + 1.805 Cl (‰). Early scientists recognized that standards were needed to determine reliable values of the chlorinity and salinity of seawater. The IAPSO Standard Sea Water Service (originally based in Copenhagen) collected and distributed seawater from the North Atlantic with a known, measured chlorinity. This sample was supplied to oceanographers to standardize the AgNO3 solutions used to determine chlorinity in various laboratories. In the 1960s more and more oceanographers were trying to determine the salinity of seawater using conductivity measurements. Attempts were made to develop correlations of the conductivity ratio of ocean samples with respect to seawater with a known chlorinity. The values of chlorinity determined in this manner were then converted to salinity using the Knudsen equation given above. Studies revealed, however, that the conductivity ratio of standard seawater varied for waters of the same salinity or chlorinity, which led to the development of the practical salinity scale that related the conductivity ratio to a fixed mass of potassium chloride (KCl) in a given mass of water. The seawater used to fix the scale had a salinity equal to 35 (S = 35.000) that was consistent with the Knudsen equation (Cl = 19.374) and thus with earlier measurements. Conductivity derived salinity is thus no longer directly connected to the chlorinity of the seawater; standards now have a specified conductivity ratio that can be used to calibrate salinometers with a sample of known salinity. The conductivity standard has made it possible for ocean chemists worldwide to make a routine determination of seawater salinity that can be compared with great precision and accuracy. |
|
Box 2.2 Case Study: Dissolved Organic Carbon (DOC) The measurement of DOC in seawater has long been a challenge to oceanographers and marine scientists (Hansell and Carlson, 2002). Indeed, ever since Natterer (1892) and Putter (1909) made the first measurements of DOC using different methods, there has been a continuing controversy over the levels of organic carbon in seawater. Resolution of this issue appeared to be imminent in the late 1970s following the publication of several comparison studies (e.g., MacKinnon, 1978; Gershey et al., 1979). In 1988, Sugimura and Suzuki reported dramatically higher DOC levels along with a remarkable correlation of these new observations with apparent oxygen utilization (AOU). The findings sparked a re-evaluation of the various methods used to determine DOC and opened a discussion regarding the validity of widely held notions regarding the nature, concentration, and distribution of dissolved organic matter in the ocean (Williams and Druffel, 1988). It would take another decade before the issue was settled. An early attempt to resolve the discrepancy between the high values of Sugimura and Suzuki (1988) and more traditional analyses failed to reach a definitive conclusion (Williams, 1992). The start of the Joint Global Ocean Flux Study (JGOFS) field program with the North Atlantic Bloom Experiment in 1989 put additional pressure on the various groups to resolve this issue quickly. The National Science Foundation (NSF) and the National Oceanic and Atmospheric Administration (NOAA) funded a workshop held in Seattle in July 1991 to resolve the issue. Prior to the Seattle Workshop, several batches of seawater from Hawaii were distributed to attendees for analysis. It became immediately clear to workshop participants that the key to making valid comparisons was both a common reference material and a uniform blank solution (Hedges et al., 1993; Sharp, 1993). The primary source of discrepancy among analysts was poor blank control, not oxidative capacity. During the JGOFS EqPac cruises (1992) a smaller group was supplied with intercomparison samples and a uniform blank solution (Sharp et al., 1995). This effort led to a broad community intercalibration exercise for the accurate determination of DOC concentrations (Fig. 2.2). Conducted over the past five years, 62 laboratories from 17 nations participated in these intercalibration exercises. Most groups of analysts showed comparability better than 10 percent, while the most experienced analysts, sharing reference materials, showed reproducibility at the 2 percent level (Sharp et al., 2002). At the Seattle Workshop, DOC concentrations had varied by as much as a factor of three, and there was little common agreement regarding background levels for DOC in deep-ocean waters. Dennis Hansell has continued the production of a seawater DOC reference material and blank water reference material that is now being distributed internationally at minimal cost to the investigators (Hansell, 2001). Continued support of this program (currently funded by NSF) is essential to maintaining the high quality of research results; omission of the use of these standards is slowly becoming grounds for rejection of research papers by some peer-reviewed journals. |
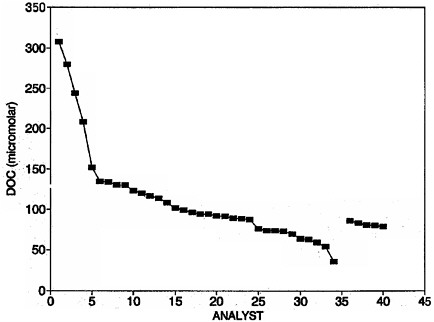
FIGURE 2.2 Example of the use of reference materials on the agreement between analysts. Analysts 1-34 represent data from all analyses of the Pacific Ocean surface sample from the Seattle Workshop (Hedges et al., 1993). These analyses were purposely performed without common reference materials or uniform blank correction in order to assess the state-of-the-art. Analysts 36-40 represent a select group of JGOFS investigators analyzing the EqPac interlaboratory comparison sample using a common blank water and reference standard. The improvement in precision is immediately apparent.
|
Box 2.3 Case Study: Dissolved Inorganic Carbon (DIC) High quality measurements of ocean CO2 have been an integral part of the JGOFS and World Ocean Circulation Experiment (WOCE) programs. Despite their importance for understanding the oceanic carbon cycle, past measurements made by different groups were rarely comparable. A significant contribution of the JGOFS effort has been to produce and distribute reference materials for oceanic measurements. With funding from the NSF and Department of Energy (DOE) since 1989, Andrew Dickson’s laboratory at the Scripps Institution of Oceanography has prepared over 50 separate batches of DIC reference material and has distributed more than 25,000 bottles of this material to scientists in over 25 countries. During a recent expedition in the Indian Ocean, as part of the WOCE Hydrographic Program, members of the U.S. DOE CO2 Survey Science Team used these reference materials extensively for the quality control of measurements of total DIC and total alkalinity. Two manuscripts detailing the results of these measurements on reference materials were published describing the CO2 measurements made on that expedition and how reference materials were used to assess the overall data quality (Johnson et al., 1998; Millero et al., 1998). Another indication that the use of reference materials has improved oceanographic data quality can be seen by examining the degree of agreement between measurements for deep water masses obtained where two separate cruises intersect. Lamb et al. (2002) examined this in detail for cruises in the Pacific Ocean and showed that the measurements of total DIC (for cruises where reference materials were available) typically agreed to within 2 µmol/kg (Fig. 2.3). This is in sharp contrast to the required adjustments to previous oceanic carbon data sets over the years. A comparison of the concentration of 13C1 over a time span of 10 to 20 years can be used to calculate the uptake rate of anthropogenic CO2 (Quay et al., 1992). Although extensive sampling occurred during the GEOSECS program in the 1970s, it has been impossible to use the 13C data because of concerns about their reliability (Kroopnik, 1985). The lack of 13C standards during the 1970s therefore has rendered much of the historical isotopic data useless.
|
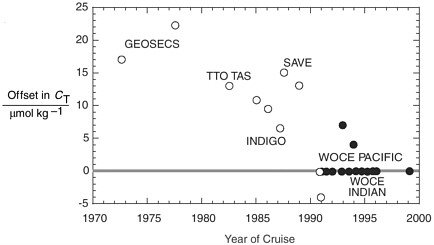
FIGURE 2.3 Estimated data offsets for total DIC measurements on various cruises (Gruber et al., 1996; Gruber, 1998; Sabine et al., 1999; Lamb et al., 2002). Certified reference materials for CO2 became available in 1991 and were used on cruises represented by filled circles. (Note: The period 1991-2000 includes 35 cruises, most with no offset.) Labels refer to oceanographic research programs. Printed with permission of A. Dickson.
Other cruises beginning in the 1970s on which δ13C was measured provided data that were used to calculate CO2 uptake rates (Quay et al., 1992). Since no reference materials were available during that time, these calculations relied on comparison of measurements made on samples from depths greater than 2000 meters with the assumption that the δ13C content below this depth would not change over the decadal time scale. Originally it appeared that there was agreement between the δ13C from the 1970s values and those measured in the late 1980s and early 1990s.
It is possible to use the increased sampling and greater geographic coverage provided by the JGOFS and WOCE δ13C programs in the 1990s (McNichol et al., 2000; Quay et al., submitted), to make more deep water comparisons at stations that are geographically closer. Using this new data it is possible to show that the deep water data from the 1970s is offset from that of the 1990s by 0.14 to 0.20 ‰ (Lerperger et al., 2000; Quay et al., submitted). Applying this offset to the data would significantly alter the calculated uptake rates. There is anecdotal evidence, however, that surface water samples were collected and analyzed differently from the deep water samples, making it unclear whether the observed offset should be subtracted from the surface water data.
|
Box 2.4 Case Study: Pigments Pigment intercalibration exercises have been performed in support of NSF’s JGOFS program (Latasa et al., 1996) and for NASA’s Sea-viewing Wide Field-of-view Sensor (SeaWiFS) project (Hooker et al., 2000). For the first exercise, pure individual and mixed pigment standards were distributed to eight JGOFS pigment laboratories. Results from three separate intercalibration exercises documented a better agreement for spectrophotometric analyses than for high pressure liquid chromatography (HPLC) analyses. For the spectrophotometric comparisons, 90 percent of the pigments analyzed by participant laboratories were within six percent of mean consensus values. By comparison, 65 and 85 percent of the laboratories agreed to within 10 and 20 percent, respectively, on HPLC analyses. Chlorophyll absorption measurements obtained with diode array-type spectrophotometers were six to nine percent lower than those obtained with monochromator-type spectrophotometers. Furthermore it was determined that the use of HPLC methods incapable of separating monovinyl chlorophyll a from divinyl chlorophyll a can result in the overestimation of total chlorophyll a concentration in Prochlorococus-dominated oceanic waters by 15 to 25 percent. A simple dichromatic approach was suggested for eliminating this variable source of error, which is caused by coelution of these structurally-related pigments (Latasa et al., 1996). For the second exercise, marine particulate matter collected at twelve stations in the Mediterranean Sea was distributed in triplicate to each of four SeaWiFS validation laboratories. Each laboratory used a different HPLC method for determining the concentrations of 15 different pigments or pigment associations. The four methods for determining concentrations of total chlorophyll a (i.e., monovinyl plus divinyl chlorophyll a) and the full set of pigments agreed within 7.9 percent and 19.1 percent respectively. In addition, accuracy was reduced by approximately 12.2 percent for every order-of-magnitude decrease in pigment concentration. Standards and samples prepared for these two intercomparisons were consumed during those studies and were not available for JGOFS Antarctic Environment and Southern Ocean Process Study (AESOPS) cruises. Comparisons of chlorophyll a concentrations measured by fluorometry (ChlF) and by HPLC (ChlH) during AESOPS indicated large discrepancies for three of the four Polar Front cruises. The large overestimates in ChlF observed may have been caused by interference associated with chlorophyllide a and chlorophyll c (Trees et al., 2000). The measurement disparities and probable causes described above required several months of additional data analysis and interpretation. The availability of pigment reference materials during the AESOPS program would have minimized the time required to sort out the observed discrepancies through the identification (or elimination) of potential analytical problems (e.g., operator error, accessory pigment interference, poor extraction efficiencies, calibration problems, etc.) during data collection. Standardization of pigment methodology for use with the HPLC technique would have resulted in more accurate and precise data. |