AMO Science Expanding the Frontiers
Discovery
This chapter presents some vignettes of research at the frontiers of AMO science. The prefix ultra and the adjective extreme appear frequently, for these frontiers really are astonishing: Ultracold temperatures are a billion times colder than interstellar space. Ultraintense light is strong enough to rip apart atoms and even space itself. Ultrashort light pulses are so fast that they can “stop” atoms as they move within a molecule. Ultraprecise atomic clocks are so accurate that they vary by less than a second in a hundred million years.
The goals of AMO science, however, are not just to generate “ultras,” but to understand atoms and light at their deepest level, to probe the fundamental laws of physics, to push the frontiers of our understanding, and to develop new techniques for measuring and manipulating light and matter. AMO research studies the basic symmetries of nature; the properties of space and time; unexplored aspects of quantum mechanics; the interaction of matter and light under extreme conditions; the structure and interactions of atoms and molecules; new forms of matter such as coherent atoms and superfluid atomic gases; and the deep connections between physics and information. Basic research often leads to totally unexpected applications, and while it is too early to judge the ultimate payoffs that may result from the advances and developments reported in this chapter, the joyful quest for knowledge and the promise of new technologies continue to drive and animate AMO science.
Theory Versus Hypothesis
Theories are the means by which physicists understand and describe the world. Some theories are limited in scope and seek only to describe a narrow class of physical phenomena. Other the-ories are more ambitious and seek to describe such a wide range of physical phenomena that they are called fundamental theories or physical laws. But no matter what the scope of a theory is, all theories have one feature in common: They make predictions that are open to experimental verification. Indeed, before a theory’s predictions can be experimentally verified, it is only a hypothesis, a speculation that over time and after rigorous experimental scrutiny will come to be either widely accepted, or rejected.
Physical theories are not immutable—they only represent our current understanding of the world. As this understanding matures, theories change, become more complete, or are supplanted by other, “better” theories. It is a dynamical process that has occurred several times in the last century. Finding the limits of a theory, and pushing it outward just a little bit to discover something new and different, is part of the excitement and joy physicists feel when doing research.
Practically Perfect
A mathematical theorem is either perfectly true or it is false; a circle, by definition, is perfectly round. Physical theories, in contrast, are never exactly true. One finds new physics by probing the limits of presently known physical laws.
These ideas thread through an area of atomic and optical physics that is loosely described as fundamental tests and high-precision measurements. Its origins can be traced to the 19th century, when Michelson developed an interferometer to detect a predicted spatial anisotropy of the speed of light. His failure to find any effect was actually a success: It is often taken as the starting point for explaining Einstein’s Special Theory of Relativity.
The tradition of fundamental tests leading to new science and new technologies is alive today. For example, experiments on the spectroscopy of hydrogen have long been a testing ground for basic theory and a driving force for new experimental techniques. The development of the needed laser technology has made it possible to measure the frequency of an absorption line of atomic hydrogen (even though light waves oscillate with a corresponding period of about a femtosecond, a billionth of a millionth of a second) to better than 2 parts in 1014 (1 followed by 14 zeros)— about the same as one drop of ink in 10,000 swimming pools! This new capability has potentially revolutionary implications for the science of measurement and atomic clocks, as well as in the communication and navigation applications discussed in the chapter “AMO Science Impacting the Economy.”
Another example of fundamental tests is related to theoretical predictions about the nature of the force between the nucleus of an atom and its orbiting electrons. In addition to the dominant electrostatic force, there is the small contribution of a short-range force called the weak interaction. According to the Standard Model of high-energy physics, this interaction has handedness—a property that distinguishes your hand from its mirror image.
In addition to its color and direction of propagation, light is characterized by its polarization, which can be conveniently thought of as an arrow rotating either clockwise or counter clockwise. A consequence of the handedness of the weak interaction is that atoms absorb light that rotates in one direction more easily than they absorb light rotating in the other direction. This effect is minuscule, no more than a part in 1012 (one drop of ink in 100 pools).Yet it not only has been detected but also has been measured accurately using laser spectroscopic techniques. Thus, a tabletop atomic physics experiment has provided a test of a fundamental theory; such tests normally require gigantic particle accelerators.
Another puzzle that physicists are interested in is the observation that the basic laws of physics do not seem to depend on the direction of time. To test this observation, AMO scientists are looking at the shape of the neutron. If physical laws do depend on the direction of time, the neutron would behave as if it were not perfectly spherical. The test of this physical law, performed with AMO techniques, is among the most sensitive ever carried out in physics. It shows that if a neutron could be enlarged to the size of Earth, it would be perfectly round, to within the thickness of a human hair. As a sphere, the neutron seems to be practically perfect. When any imperfection is found, physics will take another step forward, and our understanding of the physical world will deepen.
Ultrashort Times
Amazing progress has been achieved in the last 150 years in the study of fast phenomena. Figure 14 shows a sequence of pictures taken by the British artist, photographer, and inventor Muybridge. In the 1870s he was first to capture motion through photography, with a resolution of a fiftieth of a second or so.

FIGURE 14 Muybridge’s horse in motion.
Figure 15 is a famous 1957 photograph by Edgerton, the inventor of stroboscopic flash photography, which allowed for resolutions on the order of millionths of a second (microseconds). It shows the splash of a milk drop “stopped” by light from a stroboscopic flash, providing a totally new way to see a familiar event. The exposure time was 0.3 microseconds, several hundred times faster than possible with a mechanical shutter.

FIGURE 15 Edgerton’s milk drop.
Today, we can view things on a time scale so short that it would make the fastest stroboscopic flash seem interminable. Light pulses of a few millionths of a billionth of a second (femtoseconds) have been generated using ultrafast laser techniques. Femtosecond light pulses are so fast that they can reveal the inner workings of the fastest electronics circuits and “stop” chemical reactions.
Physicists and chemists are now using femtosecond lasers to unravel the complex dance that atoms perform during chemical reactions. The bonds that bind atoms within molecules are made and broken on the femtosecond time scale. For example, the protein bacteriorhodopsin absorbs light to provide energy for a cell. Femtosecond pulses allow scientists to probe and understand how the protein absorbs light, important information when investigating how our eyes work or how plants absorb energy from sunlight during photosynthesis.
As usually occurs when a new technology is created, many new applications suddenly appear. This brochure highlights several such applications in various fields. Shorter and shorter pulses are being exploited in communications. They may be used to monitor water content in plants and food and will form the basis for the next generation of ultraprecise clocks and GPS systems. They might also find application in drilling the smallest, most precise holes for use in microsurgery and micromachining.
Ultrahot and Intense
Earth is a tranquil, relatively nonviolent part of our universe—a veritable backwater near the edge of an average spiral galaxy. If we look outward, we see astrophysical environments completely outside of our experience—hot, violent, and totally inhospitable. At very high temperatures, atoms are stripped of many or all of their electrons. The resulting hot soup of ions, electrons, and photons, which is found, for example, in supernova remnants, is called plasma.

AMO scientists and engineers invent tools that produce ultraintense pulses of light, which can create environments hotter and denser than the interior of the Sun. They also use accelerators and state-of-the-art synchrotron light sources to study the processes that occur in high-temperature plasmas. This allows them to understand how ultrahot electrons, ions, and photons interact and collide with one another in these extreme environments.
Imagine a laser beam focused onto a spot on a solid surface smaller than the diameter of a human hair. As we begin to increase the laser pulse energy, we first vaporize the spot and create a crater (Figure 16). At still higher energies, the laser continues to heat the vapor from the crater until atoms and molecules explode into electrons and ions, forming an ultrahot, ionized plasma with a temperature of millions of degrees, similar to a star’s interior. We can turn up the laser pulse energy even higher, so that the laser light pushes the electrons and ions around so violently that they accelerate to relativistic velocities close to the speed of light, hardly interacting with each other at all. In this regime, even the concept of temperature loses its meaning.
Finally, we have lasers with such incredibly high energies that we can literally rip apart empty space to form new matter and light where none previously existed! The ultrashort pulses of light that can accomplish these wonderful phenomena are generated with equipment that would fit on your kitchen table.
As well as providing new tools with which to understand our universe, ultraintense lasers have many practical applications. The craters formed by laser beams have edges smooth enough to be used in eye surgery or for the drilling of microscopic holes or the cutting of materials in industry. Laserlike beams of x rays can be generated and used to “see” atoms and molecules as they move about in chemical reactions. Finally, intense lasers can be used to accelerate beams of electrons to the ultrahigh energies required to study the fundamental constituents of matter. These new x-ray and matter beams may be used in the future for applications such as building smaller, faster electronic circuits or higher-resolution microscopes.

FIGURE 16 Ultraintense laser pulses vaporizing a target.
One at a Time . . .
Atoms are so small that we usually encounter them in astronomical numbers. A cubic centimeter of a solid, for instance, typically contains about 1023 atoms (1 followed by 23 zeros); the smallest speck discernable with an optical microscope contains more than a trillion atoms. In view of such numbers, the idea of carrying out an experiment on a single atom seems most improbable. Nevertheless, AMO experimentalists can isolate and manipulate not only single atoms, but also single electrons, single ions, single molecules, and even single photons (see Figure 17).
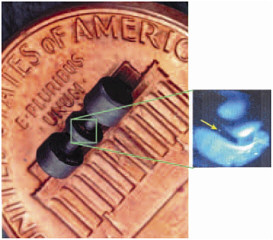
FIGURE 17 Left: electrodes used to trap a single atomic ion. Right: The arrow points to a single mercury ion held in the trap.
Physicists have dreamed of experiments with single particles for a long time. The use of single particles eliminates unwanted interference by other particles. It also emphasizes the quantized nature of light and particles and reveals the quantum effects that are washed out in large ensembles.
A lone electron, trapped and observed for months, has provided the most precise test of theory in the history of physics: Quantum electrodynamics was verified to a precision of 2 parts in 1012. A single photon, generated in a one-atom maser, has been trapped in a superconducting cavity for times approaching a second. If free, it would have traveled 300,000 km during this time. These trapping methods are being applied to new types of quantum systems, advancing our understanding in areas of quantum communication and quantum information processing. Likewise, a single ion or several ions can be held indefinitely in a trap, providing essentially ideal conditions to monitor their behavior.
Single molecules have been observed in crystals, polymers, and liquids, and even in biomolecular environments where proteins and DNA are found. The magic key is to use a molecule that emits light when pumped by a laser beam. Light from the single molecule can then be detected while the possible interfering signal from nearly one trillion other molecules of the nearby host material is rejected. This represents truly ultrasensitive detection.
Single-molecule investigations are opening new areas of biology, physics, and chemistry. They challenge scientists to develop a far more accurate picture of how an ensemble of molecules works. These insights will be crucial in the future drive toward molecular machines at the nanoscale.
Ultralarge: AMO Science in Astrophysics
It is perhaps surprising to realize that much of our understanding about the universe, its origins, and evolution relies heavily on our knowledge of atomic and molecular science. Almost all of the information that we receive about the vast array of astronomical objects in the universe comes to us through the light waves detected by ground-based telescopes and telescopes orbiting high above Earth. Thus, spectroscopy is an important fundamental tool in astrophysics. It is through spectroscopic observations of the early universe, galaxies, stars, supernovae, molecular clouds, and planets, for instance, that one can deduce temperatures, element abundances, and gas densities in such objects—provided that the necessary atomic and molecular studies have been carried out to obtain the relevant spectroscopic data.

FIGURE 18
New and exciting observations often point to the need for new atomic data. In 1996 astronomers detected x-ray emission from comet Hyakutake. Comets are known to radiate light, either scattered sunlight or solar radiation first absorbed and then re-emitted by gas in their atmospheres, but the origin of these x rays was a mystery. The theory that was developed suggests that x rays are emitted by highly charged ions in the solar wind excited in collisions with neutral species in the comet’s atmosphere. The Chandra x-ray satellite, carrying instruments with improved resolution, has confirmed the general predictions of the theory. However, to identify individual x-ray features that are now observed, further laboratory experiments on these ion-molecule collisions will be essential. Meanwhile, x-ray emission has now been observed from several more comets.
In another example, in the last few decades a variety of unusual molecules have been discovered through their radio-emission spectra to exist in interstellar clouds and circumstellar shells. A vast majority of these molecules are hydrocarbons, and a few are the same as simple organic molecules that form the basis of life on Earth. Figure 18, a picture taken by the Hubble Space Telescope, shows a glowing sky surrounding dark regions where there is no visible radiation. These regions are dark because starlight is obscured by large quantities of dust. They do, however, emit radiation at infrared and radio frequencies. It is in such dusty, dark clouds that astronomers have detected a rich array of molecules; in some cases molecules that had never before been seen on Earth were identified in interstellar clouds. Laboratory scientists have since been able to produce them. Aided by computational studies, they have confirmed the composition and structure of these molecules. Further work on atomic and molecular processes contributing to molecular formation will allow scientists to understand the fascinating physics that controls the dynamics and evolution of these interstellar and circumstellar regions. Because interstellar clouds are the birthplace of stars, knowledge of molecular physics will help to unlock the secrets of star formation and stellar life cycles.
Ultracold—Approaching Absolute Zero
Whenever the frontiers of temperature are pushed downward, new physics emerges. Thus, for physicists it is never too cold. When temperatures of a few kelvin were reached (room temperature is about 300 kelvin, or 300 degrees above absolute zero) amazing quantum phenomena were discovered—superconductivity, in which an electrical current flows with zero resistance, and superfluidity, in which liquid helium flows with zero friction.
In a series of developments starting in the mid-1980s, novel techniques based on laser cooling and evaporation moved the low temperature frontier into the nanokelvin (billionth of a degree) regime.
Low temperature means low energy, for temperature is a measure of the energy in a system. The lower the temperature, the less random thermal motion is present. For this reason, access to the ultralow temperature regime has provided many payoffs. For example, atomic motion is an impediment to many precise measurements. However, at ultralow temperatures, atoms move very slowly or can even be made to stand practically still. This slow motion has already been exploited to develop a new type of atomic clock with unprecedented precision. In another application, laser cooling was used to create atomic gyroscopes that have the potential of exceeding the best gyroscopes ever made.
At ultralow temperatures, energy can be so scarce that a gas of atoms forms a new state of matter—a quantum gas. In particular, one class of atoms called bosonic atoms can undergo a transition to what is known as a Bose-Einstein condensate.
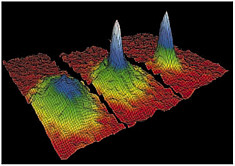
FIGURE 19 The velocity distributions in this figure illustrate the transition from a normal, albeit ultracold, gas to a Bose-Einstein condensate. The broad distribution at the left is characteristic of an ordinary collection of atoms. The narrow, peaked distribution at the center and right represent the condensate, some of the coldest matter in the universe. The vertical axis in the distributions corresponds to the number of atoms, while the horizontal axes correspond to two of the velocity components.
Bose-Einstein condensation (BEC) is one of the few known phenomena where quantum mechanical behavior—usually only relevant on a microscopic scale—manifests itself macroscopically (see Figure 19). Research on BEC has provided new insight into the quantum world and has led to a new technology for atoms and atom optics. The 2001 Nobel Prize in Physics was awarded to three physicists for the first demonstration of BEC in 1995.
Quantum Weirdness
Computers operate by manipulating sequences of elementary objects, called bits, which can take one of two values, zero or one. By contrast, the equivalent quantum object, a qubit, can be in any superposition of these two values. This is a bizarre consequence of an aspect of quantum mechanics that allows a quantum system to be in two different states at once. For instance, a quantum particle can find itself in two positions, or have two velocities, at the same time. This is the origin of the famous paradox of Schrödinger’s cat, which can be simultaneously alive and dead!
Even stranger are the properties of pairs of quantum particles, such as the light produced by an optical parametric amplifier, illustrated in Figure 20. This device emits photons in pairs, for instance a blue and a red photon, or two green photons. Photons, the elementary particles of light, are characterized by a polarization, which can be thought of as an arrow pointing up or down along some direction. If a pair of green photons—the two bright green spots on the picture—is emitted by that optical parametric amplifier, it is fundamentally impossible to know with certainly the polarization of either one. However, if we determine that one of the photons has “up” polarization, we can be sure that the other has “down’’ polarization. Pairs of quantum particles with this type of correlated properties are said to be entangled. They are so intermixed that there is no way to describe either separately.
Entangled pairs of particles lead to apparently paradoxical and counterintuitive effects, such as the “spooky action at a distance” that so disturbed Einstein. The distinction in the behavior of pairs of classical and quantum particles was quantified in a mathematical form called Bell’s inequalities, which are satisfied classically but violated in the quantum world. This violation has been unambiguously demonstrated in a series of beautiful atomic physics experiments.
At the fundamental level, these recent advances have shed light on the mind-bending transition from the microscopic to the quantum world. On the practical side, they are opening up the way to a technological revolution: quantum information technology. They have led to the first demonstrations of quantum teleportation, which allows a quantum system to be exactly reproduced at a distant location. Another application, quantum cryptography, allows the development of unbreakable protocols to secretly exchange information. And someday, quantum computers, which are based on qubits instead of bits, could implement the quantum algorithms being developed for tackling problems that are unsolvable using classical computers.

FIGURE 20 Light from an optical parametric amplifier. The bright green spots correspond to the emission of a pair of entangled photons.








