1
Introduction
For the last 30 years, the nation has been trying to assess, remediate, and otherwise manage thousand of acres of soil and sediment1 contaminated with chemicals produced during the industrial age. Of primary concern has been the risk that these contaminated media pose to humans and ecological receptors. Evaluation of exposure is a key component of chemical risk assessment, and understanding the factors that influence exposure enables decision-makers to develop solutions for addressing environmental contamination. This report of the National Research Council examines the bioavailability of contaminants in soil and sediment, focusing on those factors that influence the percentage of total contaminant levels to which humans and ecological receptors are exposed. The extent to which chemicals are bioavailable has significant implications for the cleanup of contaminated media.
National attention on bioavailability stems from a growing awareness that soils and sediments bind chemicals to varying degrees, thus altering their availability to other environmental media (surface water, groundwater, air) and to living organisms (microbes, plants, invertebrates, wildlife, and humans). It is also recognized that the physiological characteristics or “niche” of plant and animal species influence the availability of chemicals, such that exposure to the same contaminated material may be very different from one species to another. The altered availability of chemicals associated with soils or sediments has been variously described by such terms as partitioning, reduced desorption rates,
|
1 |
The terms “soil” and “sediment” are defined in detail in Chapter 3. |
reduced biodegradation rates, geochemical binding, sequestration, and limited absorption through biological membranes—to name but a few descriptors. While these descriptors may all involve different chemical, physical, and biological processes, they all describe the phenomenon that chemicals in soils and sediments behave differently than when those chemicals are present in other media, notably water and air.
“Bioavailability processes” are defined as the individual physical, chemical, and biological interactions that determine the exposure of plants and animals to chemicals associated with soils and sediments. One reason for adopting the term “bioavailability processes” in this document is the realization that “bioavailability” has been defined in different ways that are often discipline-specific. Instead of redefining the term “bioavailability,” the committee has chosen to recognize the value of various definitions and to focus instead on the interacting biological, chemical, and physical processes particular to the presence of chemicals in soils and sediments that influence exposure. The term “bioavailability processes” captures this idea.
Currently, “bioavailability” is used in risk assessment most frequently as an adjustment or correction factor that accounts for the ability of a chemical to be absorbed by an organism—an approach that makes a number of assumptions regarding individual bioavailability processes. Unfortunately, contemporary risk assessment practice does a poor job of identifying and explaining these assumptions, such that it is generally not clear how bioavailability processes are incorporated into risk assessments. It can be difficult to know whether all of the relevant processes are addressed and whether assumptions are based on valid concepts and reliable data. In fact, there is ample reason to suspect that many bioavailability processes are dealt with inadequately or inaccurately. In order to improve this aspect of risk assessment, it will be necessary to identify relevant bioavailability processes in a more transparent way, to gain greater mechanistic understanding of these processes, and to evaluate the ability of various tools to offer information on bioavailability processes. Over the long term, such a process-based approach will improve exposure assessment, resulting in greater consistency, reliability, and defensibility in measurement, modeling, and prediction.
BIOAVAILABILITY PROCESSES FOR CONTAMINANTS IN SOILS AND SEDIMENTS
Several definitions for the term “bioavailability” are listed Table 1-1. Depending on the context, bioavailability may represent the fraction of a chemical accessible to an organism for absorption, the rate at which a substance is absorbed into a living system, or a measure of the potential to cause a toxic effect. Often, environmental scientists consider bioavailability to represent the accessibility of a solid-bound chemical for assimilation and possible toxicity (Alexander, 2000), while toxicologists consider bioavailability as the fraction of chemical
TABLE 1-1 Definitions of “Bioavailability” and Related Terms
|
Definition |
Source |
|
Bioavailability |
|
|
A chemical element is bioavailable if it is present as, or can be transformed readily to, the free-ion species, if it can move to plant roots on a time scale that is relevant to plant growth and development, and if, once absorbed by the root, it affects the life cycle of the plant. |
Sposito, 1989 |
|
Generally used to describe the extent and rate of absorption for a xenobiotic which enters the systemic circulation in the unaltered (parent) form from the applied (exposure) site. |
Hrudy et al., 1996 |
|
The availability of a chemical to an animal, plant, or microorganism. It may be assayed by measurement of uptake, toxicity or biodegradability. |
Linz and Nakles, 1997 |
|
A concept that describes the ability of a chemical to interact with living organisms. |
NEPI, 1997 |
|
The accessibility of contaminants to microbes from the standpoint of their metabolism, their ability to grow on these chemicals, to change cellular physiology, and perhaps modulation of genetic response. |
Sayler et al., 1998 |
|
A measure of the fraction of the chemical(s) of concern in environmental media that is accessible to an organism for absorption. |
ASTM, 1998 |
|
A measure of the potential for entry into ecological or human receptors. It is specific to the receptor, the route of entry, time of exposure, and the matrix containing the contaminant. |
Anderson et al., 1999 |
|
The extent to which a substance can be absorbed by a living organism and can cause an adverse physiological or toxicological response. |
Battelle and Exponent, 2000 |
|
Bioavailable: For chemicals, the state of being potentially available for biological uptake by an aquatic organism when that organism is processing or encountering a given environmental medium (e.g., the chemicals that can be extracted by the gills from water as it passes through the respiratory cavity or the chemicals that are absorbed by internal membranes as the organism moves through or ingests sediment). In water, a chemical can exist in three different forms that affect availability to organisms: (1) dissolved, (2) sorbed to biotic or abiotic components and suspended in the water column or deposited on the bottom, and (3) incorporated (accumulated) into the organisms. |
EPA, 2000a |
|
The fraction of an administered dose that reaches the central (blood) compartment, whether from gastrointestinal tract, skin, or lungs. Bioavailability defined in this manner is commonly referred to as “absolute bioavailability.” |
NEPI, 2000a |
|
Definition |
Source |
|
In the environment, only a portion of the total quantity of chemical present is potentially available for uptake by organisms. This concept is referred to as the biological availability (or bioavailability) of a chemical. |
Casarett and Doull’s, 2001 |
|
A measure of the potential of a chemical for entry into, or interaction with, ecological or human receptors. It is specific to the receptor, the route of entry, time of exposure, and the matrix containing the contaminant. |
Lanno, 2001 |
|
A term used to indicate the fractional extent to which a dose reaches its site of action or a biological fluid from which the drug has access to its site of action. |
Wilkinson, 2001 |
|
The degree to which a drug or other substance becomes available at the physiological site of activity after administration. |
American Heritage Dict., 3rd Ed. |
|
The degree and rate at which a substance (as a drug) is absorbed into a living system or is made available at the site of physiological activity. |
Webster’s Dictionary, 10th Ed. |
|
Absolute Bioavailability |
|
|
The fraction or percentage of an external dose which reaches the systemic circulation, that is, the ratio of an internal dose to an applied dose. This ratio is called the bioavailability factor (BF). |
Hrudy et al., 1996 |
|
The percentage of an external exposing mass that reaches the systemic circulation (the internal dose). et al., |
Paustenbach 1997 |
|
The fraction of an administered dose that reaches the central (blood) compartment from the gastrointestinal tract. Bioavailability defined in this manner is equal to the oral absorption fraction. |
Ruby et al., 1999 |
|
The fraction or percentage of a compound which is ingested, inhaled, or applied on the skin that actually is absorbed and reaches the systemic circulation. |
Battelle and Exponent, 2000 |
|
The fraction of an administered dose that reaches the central (blood) compartment, whether from gastrointestinal tract, skin, or lungs. |
NEPI, 2000a |
|
Relative Bioavailability |
|
|
The absolute bioavailability of an external exposing mass divided by the absolute bioavailability of the chemical compound under the conditions used to derive the toxicity criterion. |
Paustenbach et al., 1997 |
|
Definition |
Source |
|
Refers to comparative bioavailabilities of different forms of a chemical or for different exposure media containing the chemical (e.g., bioavailability of a chemical from soil relative to its bioavailability from water) and is expressed as a fractional relative absorption factor. |
NEPI, 2000a; Ruby et al., 1999 |
|
A measure of the extent of absorption among two or more forms of the same chemical (e.g., lead carbonate vs. lead acetate), different vehicles (e.g., food, soil, water), or different doses. In the context of environmental risk assessment, relative bioavailability is the ratio of the absorbed fraction from the exposure medium in the risk assessment (e.g., soil) to the absorbed fraction from the dosing medium used in the critical toxicity study. |
Battelle and Exponent, 2000 |
|
Other Definitions |
|
|
Bioaccumulation is the total accumulation of contaminants in the tissue of an organism through any route, such as food items as well as from the dissolved phase in water. Bioconcentration is accumulation of a chemical directly from the dissolved phase through the gills and epithelial tissues of an aquatic organism. Biomagnification is the process by which bioaccumulation causes an increase in tissue concentrations from one trophic level to the next from food to consumer. |
Rand and Petrocelli, 1985; Schnoor, 1996; EPA, 2000a |
|
Bioavailable fraction is that portion of the bulk concentration that is available to be accumulated into an organism under a defined set of conditions. For instance, for a metal it could be the freely dissolved ion of the metal. Other forms of the metal bound in precipitates or covalent or hydrogen bonded to other ions would not be available. The available fraction is a proportion ranging from 0.0 to 1.0. The available fraction determines the reactive portion of the total mass of material, much like the activity coefficient relates activity to concentration. |
EPA, 2000a |
|
Bioaccessibility describes the fraction of the chemical that desorbs from its matrix (e.g., soil, dust, wood) in the gastrointestinal tract and is available for absorption. The bioaccessible fraction is not necessarily equal to the RAF (or RBA) but depends on the relation between results from a particular in vitro test system and an appropriate in vivo model. |
Paustenbach et al., 1997; Ruby et al., 1999 |
|
Relative absorption factor (RAF) describes the ratio of the absorbed fraction of a substance from a particular exposure medium relative to the fraction absorbed from the dosing vehicle used in the toxicity study for that substance (the term relative bioavailability adjustment (RBA) is also used to describe this factor.) |
Ruby et al., 1999 |
|
Definition |
Source |
|
Absorption describes the transfer of a chemical across the biological membrane into the blood circulation.a |
Paustenbach et al., 1997 |
|
Biostabilization refers to the biodegradation of the more labile HOC (hydrophobic organic compound) fraction leaving a residual that is much less available and mobile. |
Luthy et al., 1997 |
|
aIn this report, “absorption” is used generically for non-mammalian organisms to be synonymous with “uptake.” |
|
absorbed and able to reach systemic circulation in an organism. Another view of bioavailability is represented by a chemical crossing a cell membrane, entering a cell, and becoming available at a site of biological activity. Others might think of bioavailability more specifically in terms of contaminant binding to or release from a solid phase. These different viewpoints of bioavailability create a semantic stumbling block that can confound use of the term across multiple disciplines—hence the reason that “bioavailability processes” is used in this report.
Figure 1-1 is a depiction of bioavailability processes in soil or sediment; it incorporates exposure by release of solid-bound contaminant and subsequent transport, direct contact of a bound contaminant, uptake by passage through a membrane, and incorporation into an organism. “A”—contaminant binding and release—refers to the physical and [bio]chemical phenomena that bind/unbind, expose, or solubilize a contaminant associated with soil or sediment. This may include geological processes like weathering and scouring, chemical processes like redox reactions or complexation, and biochemical processes through the action of biosurfactants or hydrolytic enzymes. Binding may occur by adsorption on solid surfaces, by absorption within a phase like natural organic matter, or by a change in form as in covalent bonding. “B” in Figure 1-1 involves the movement of a released contaminant to the membrane of an organism. Transport may result from diffusion and advection to target receptors such as microbes, plants, and humans. Thus, bioavailability processes A and B comprise exposure via various chemical and biochemical phenomena that affect release and subsequent transport of dissolved contaminants. “C” involves the movement of contaminants still bound to the solid phase, which can play a role in dermal contact of soils, oral ingestion of soil or sediment, or exposure to burrowing organisms in soil or sediment. It should be noted that processes A, B, and C can occur internal to an organism such as in the gut lumen, although they are depicted in Figure 1-1 as occurring in the external environment.
The bioavailability process depicted as D in Figure 1-1 entails movement across membranes. Here the contaminant passes from the external environment through a physiological barrier and into a living system. An example is transport
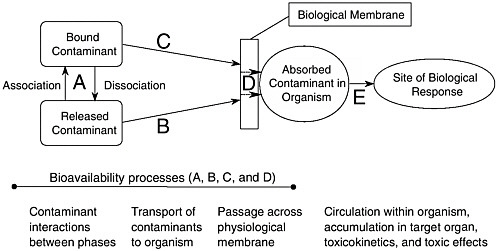
FIGURE 1-1 Bioavailability processes in soil or sediment. Note that A, B, and C can occur internal to an organism such as in the lumen of the gut.
through the gut membrane of an organism (e.g., the intestinal epithelium of a mammal). Exposure to both dissolved and solid-bound contaminants can lead to chemical interaction with the membrane of an organism and subsequent uptake or absorption (these terms are used synonymously). “E” in Figure 1-1 refers to paths taken by the chemical following uptake across a membrane. For example, after passage across a biological membrane the chemical can exert a toxic effect within a particular tissue (among many possibilities).
It should be noted that A, B, and C in Figure 1-1 are sometimes considered to be fate and transport processes (which they are) rather than bioavailability processes. On the other hand, process D is more traditionally associated with bioavailability in contemporary risk assessment. The committee’s definition of “bioavailability processes” incorporates all the steps that take a chemical from being bound or isolated in soil or sediment to being taken up into an organism (A through D). Figure 1-1 makes it clear that soils and sediments can affect exposure in various ways, both external and internal to the organism. For example, solid phases influence the extent of contaminant transfer from one medium to another, thereby determining soluble chemical concentrations. There is also differential uptake of contaminants into animals and plants depending on whether they are solubilized or solid-bound. Although of great importance in determining the overall effect of a contaminant on an organism, E processes—the toxic action or metabolic effect of a chemical—are not defined as bioavailability processes per se because soil and sediment are no longer a factor. However, because E processes are often measured endpoints, they are described at length in Chapters 3 and 4.
Bioavailability processes have definable characteristics that provide the foundation for this report. First, in the broadest sense, bioavailability processes describe a chemical’s ability to interact with the biological world. Second, bioavailability processes are quantifiable through the use of multiple tools. Third, bioavailability processes incorporate a number of steps (see Figure 1-1), not all of which would be applicable for all compounds or all settings. Indeed, it is because the term implies several individual interactions and processes that the committee prefers the term “bioavailability processes” to “bioavailability.” Fourth, there are barriers that change exposure at each step. Thus, bioavailability processes modify the amount of chemical in soil or sediment that is actually taken up and available to cause biological responses.
HISTORICAL PERSPECTIVE
That soils and sediments can impact chemical interactions with plants and pests has been known for some time by farmers and those involved in agricultural services (e.g., manufacturers of fertilizers, pesticides, and herbicides). However, in the past few decades the phenomenon has gained attention with respect to releases of hazardous chemicals to the environment. First, interest in bioavailability has been driven by a desire to reduce the uncertainties in estimating exposures as part of human and ecological risk assessment. That is, a better understanding of bioavailability processes could help identify sediment- or soil-specific factors that might influence exposure. A second impetus comes from the remediation of contaminated sites, including observations that the effectiveness of bioremediation and other treatment technologies can be limited by the availability of chemicals in soils or sediments. In some cases, the greatest opportunity for risk reduction may be to treat or contain the bioavailable fraction of the hazardous chemicals in soils and sediments and then to rely on natural attenuation approaches to treat the long-term, slow release of residual contaminants. Thus, there is considerable interest in setting cleanup goals based on the bioavailable amount rather than the entire contaminant mass. The brief history below acknowledges the varied use of the term and the extent to which bioavailability processes have been considered in different contexts.
Toxicological, Pharmacological, and Nutritional Use of Bioavailability
Although coinage of the term “bioavailability” is relatively recent, an appreciation of bioavailability concepts in the context of toxicology is ancient, particularly with regards to the treatment and prevention of poisoning. For example, pre-Columbian natives in South America were known to extract a powerful muscle-paralyzing agent—curare—from various Strychnos plants. They had no means of knowing that this alkaloid possesses a quaternary nitrogen atom, and that the charge on this nitrogen atom prevents its movement across the gas-

trointestinal epithelium. They understood quite well, however, that this poison was harmless when ingested, but very effective when injected. As a result, they could immobilize prey with curare-tipped arrows, dispatch the prey, and safely eat the meat.
From the fifth century BC to the fifteenth century AD, red clay from a specific hill on the Greek island of Lemnos was regarded as a sacred antidote for poisoning (Thompson, 1931). Called terra sigillata, it was considered effective against all poisons, no doubt acting as an adsorbent and preventing uptake in the gastrointestinal tract. The use of charcoal as an adsorbent to reduce the effect of poisons can be traced back to even earlier times, with its mention recorded in the Egyptian Papyrus of 1550 BC. In the nineteenth century, when toxicologists had the fortitude to serve as their own experimental subjects, P. F. Tourney demonstrated the effectiveness of charcoal before the French Academy of Medicine by ingesting ten times the lethal dose of strychnine combined with charcoal, and surviving (Holt and Holz, 1963).
One of the most fundamental concepts in toxicology is that an adverse effect is dependent upon the dose of the toxic substance (or toxicant) reaching a target organ or tissue. With the exception of chemicals that react with the organism on contact, such as corrosive agents, the toxicant must be absorbed into the systemic circulation to reach its biological target. From a toxicological perspective then, bioavailability implies movement of a chemical into the systemic circulation because to a large extent this is a good indication of the biologically effective dose. This view is reflected in the definition of bioavailability given in toxicology texts; for example, Casarett and Doull (2001) define bioavailability as the “frac-
tion of dose absorbed systemically.” From the toxicologist’s perspective, this definition applies to virtually all circumstances of chemical exposure, including exposure to chemicals in soils and sediments.
Because the disciplines of toxicology and pharmacology share many basic principles, this is essentially the same way bioavailability has been defined in medicine, except of course that the focus is on the absorption of drugs from dosage forms instead of chemicals from environmental media. The tenth edition of the classic pharmacology text, Goodman and Gilman (2001), defines bioavailability as “the fraction of dose of a drug reaching the systemic circulation or site of action.”
Both toxicologists and medical doctors are cognizant of the importance of events outside the body and that physical–chemical properties of the toxicant or drug and its interactions with its surroundings can affect the rate and extent of absorption. In fact, much of what is termed pharmaceutics involves an understanding of these phenomena as they pertain to drugs and manipulation of drugs and their microenvironment to therapeutic advantage. Also, toxicologists are well aware that a variety of events in the environment can affect the rate and form in which chemicals are delivered to the body. Nevertheless, the defining aspect of bioavailability, as the term is used in both toxicology and medicine, is the movement of chemical from outside the body into the systemic circulation.
Bioavailability is also an important consideration in nutrition. Here the focus is on absorption of nutrients from the gastrointestinal tract, and the term bioavailability can have different meanings in different situations. For example, nutrients such as amino acids in proteins must be liberated through digestive enzyme activity in the gut. In this context, bioavailability may become synonymous with digestibility. Other nutrients, such as most vitamins, require metabolic activation in order to have nutritional value. For these substances, bioavailability is sometimes defined to include both absorption and the metabolic activation process. For still other nutrients that do not require digestion or metabolic activation, bioavailability is regarded simply as the process of absorption of the substance from the gut into the systemic circulation, as in toxicology and medicine.
In considering the toxicological use of the term, it is important to recognize that systemic absorption is not necessarily equivalent to general uptake or absorption into the body, particularly from the gastrointestinal tract. Mammalian anatomy is responsible for this complication. Chemicals absorbed from the gastrointestinal tract enter hepatic portal circulation and must pass through the liver before reaching the general circulation. The liver (and to some extent, the gastrointestinal epithelium) may metabolize the chemical, converting it to substances with greater, lesser, or qualitatively different biological activity. This view of bioavailability, in terms of what reaches the systemic circulation (as opposed to just crossing a biological membrane), includes both absorption and metabolism components, and components both internal and external to the body. It can also lead to some ambiguity in how bioavailability is operationally defined for a
particular chemical. Often bioavailability in toxicology is described in terms of the chemical itself, ignoring metabolites that are formed during the chemical’s transit from the gut to the general circulation. However, in some instances it is important to describe bioavailability in ways that include metabolites, such as when metabolites are formed that contribute significantly to the biological dose of the chemical. This is analogous to the expanded definition of bioavailability in nutrition to include metabolic activation of vitamins. Regardless of how it is defined, a clear articulation of the basis for the bioavailability determination (with or without metabolites) is required in order to interpret the results.
Bioavailability in Agriculture
Nutrient Phytoavailability
The recognition that total soil concentration of a compound is not equivalent to bioavailable or effective concentration is well established in the agricultural sciences. This is well known not only for plant nutrients but also for water, where physical processes such as water tension or matrix potential control the fraction of total water that is plant-available. Attempts to maximize yields and optimize economic return have resulted in extensive research to describe the behavior of necessary plant nutrients in soil systems. Methods to determine total concentration as well as the plant-available (“phytoavailable”) fraction of the 18 required plant macro- and micronutrients (including water) have been developed across a range of soil types (Bartels and Sparks, 1996). These have been validated with field trials for multiple crops under varied soil, climate, and moisture regimes.
The bioavailable nutrient pool varies significantly by soil type and by plant species (Chaney, 1994). This reflects the different complexing capacities of different soil orders as well as different plant mechanisms for accessing soil nutrients (Marschner, 1995). Availability can also depend on the source of the nutrient. For example, nitrogen can be added to soils as manure N, ammoniacal N, nitrate N, and N–P materials; each of these sources will have different release characteristics that vary by soil type, soil moisture, plant growth stage, and soil microbial activity (Pierzynski et al., 2000).
The range of factors that affect nutrient availability and the methods that have been developed to predict effective nutrient concentrations potentially can be used as a model for the development of appropriate protocols to assess bioavailability processes for contaminants in soils and sediments. Although the majority of these protocols have been developed to predict phytoavailability of nutrients in potentially deficient conditions, there is a direct correlation to the development of an understanding of the bioavailable fraction of soil contaminants. In many cases, however, plants are aggressively attempting to alter the rhizosphere environment to facilitate nutrient uptake, during which they may inadvertently access soil-bound contaminants.
While this research has significantly increased knowledge of bioavailability processes and led to the development of tools to measure the bioavailable fraction, it is not yet at the point where the phytoavailability of nutrients across a range of soils and crops can always be accurately predicted. Heterogeneity in soil colloids and adsorption surfaces and differences in soil pH, organic matter, and pore spaces preclude the ability to definitively predict the fate of nutrients in soil systems. This is further complicated by differences in uptake efficiencies across plant species. Nonetheless, the factors involved in nutrient uptake may help to clarify the processes that are involved in determining the bioavailability of contaminants in soil systems.
Pesticide Bioavailability
The concept of bioavailability also has a history in the application of pesticides, particularly herbicides, to agricultural soils. As with the uptake of nutrients by plants, the efficacy of an applied herbicide, fungicide, or insecticide depends on a range of soil properties, primarily soil organic matter content and texture. Specific properties of the pesticide will also affect its behavior in the soil system, including the size of the molecule, its structure and functional groups, its polarity, and resulting dissociation constants and partitioning coefficients (e.g., Ka, and Koc). Thus, different application rates are recommended for different soil types and compounds. In addition, the potential for herbicide residues to damage successive croppings will vary because of changes in the persistence of the compound in different soils. This has been understood and incorporated in product development for several decades (Hance, 1967; Bailey and White, 1970; Walker et al., 1982).
Generally, herbicides must be dissolved in soil solution to be effective. As the soil organic matter concentration or soil clay content increases, the portion of the herbicide that is sorbed also increases (Stevenson, 1994). In soils of high organic matter such as peats, herbicides may be completely ineffective when applied at typical economic rates. For soils with very low organic matter concentrations, application may not be recommended because too much of the compound may be present in soil solution, increasing the potential for crop damage as well as leaching. Other factors, such as moisture content, soil texture, and timing of rainfall after application will also affect the efficacy of the compound (Mueller-Warrant, 1999). These factors have been sufficiently recognized within the industry that compound labels will generally recommend different application rates based on soil type. For example, application rate recommendations for S-metalochlor are based on soil texture and percent organic matter, with recommended rates varying from 0.8 kg active ingredient (ai) ha–1 to 1.4 kg ai ha–1 depending on specific soil characteristics (Blumhorst et al., 1990).
Bioavailability is also an issue when dealing with residues of agricultural chemicals applied in the past. In particular, the bioavailability of insecticides
|
BOX 1-1 Persistence and Bioavailability of Pesticides Owing to the widespread use and economic importance of pesticides, their long-term persistence in soil has been studied for more than half a century. Methods to assess pesticide concentrations in soil have evolved to recover as much added compound as possible with ever-increasing precision and accuracy. Today there is a debate as to whether analytical methods designed to measure the total concentration adequately reflect the risk from such pesticides. Early evidence showed that pesticides persist in soil for a long time. In 1949 and 1951, plots were established to study the long-term persistence and rates of disappearance of several chlorinated hydrocarbon insecticides applied to soil, including dieldrin, chlordane, and DDT (Nash and Woolson, 1967). Their results showed that 39 percent of the original DDT remained after 17 years. These soil plots gave an upper-limit persistence owing to the amount and means of pesticide added and management of the test plots with minimal tillage. Nash and Woolson’s persistence data for DDT, heptaclor, dieldrin and five other pesticides are presented in semi-logarithmic fashion, implying long-term, steady decline. Alexander (2000) arithmetically plotted selected data sets of Nash and Woolson for DDT, heptaclor, and dieldrin to suggest gradual decrease in the rate of reduction of contaminant mass for which some latter data points do not change much with time. Thus, depending on one’s presentation of such data, two views emerge—either a “hockey stick” curve where concentrations rapidly level off with time, or a first-order plot where concentrations progressively decline, albeit slowly. Other evidence for the long-term persistence of DDT and its residues in soil is presented by Boul et al. (1994), who report the longevity of DDT and its residues over a 30-year period for conditions typical for pastoral agriculture. Their data suggest gradual DDT decline since the last application in 1965, and that appreciable levels of DDT residues, mainly DDE, will remain. Other studies have tried to demonstrate a link between persistence and bioavailability by measuring contaminant assimilation into animals or effects on crops for soils with aged compounds versus soils with freshly added compounds. For example, Morrison et |
applied years ago has received attention in the environmental engineering arena, with the intent of determining whether these residues pose a present-day risk to humans or ecological receptors. Box 1-1 describes a series of studies on pesticide persistence in soil and resulting bioavailability.
Bioavailability in Evaluating and Managing Hazardous and Solid Wastes
The attention given to bioavailability in the environmental arena is relatively recent compared to disciplines like toxicology and agronomy. This attention has been driven in large part by hazardous materials and site cleanup legislation and concerns about the exposure to and risk from hazardous chemicals. For example, chemicals that are encapsulated, insoluble, or strongly bound to solids may not be prone to biological uptake or exert a biological response, while chemicals that are
|
al. (2000) studied earthworm assimilation from soils that had been treated with DDT and dieldrin in 1949, using the same soils prepared originally by Nash and Woolson (1967). Soil from Dahlgren, Virginia, contaminated with DDT approximately 30 years ago was studied as well. Comparison was made with soil freshly spiked with pesticide. Their data showed that although aging reduced uptake into earthworms, some of the pesticide was still assimilated by the earthworms even after an aging period of 49 years. For example, based on the differences in concentrations in the worms exposed to unaged and aged compounds with DDT and dieldrin spiked into unaged soil samples at concentrations found in the field after 49 years, 32 percent of the DDT and 28 percent of the dieldrin from the 49-year old samples was “available” relative to the unaged samples. Thus, while a compound’s aging in soil reduced its uptake into earthworms, some exposure remains. In an analogous study, Robertson and Alexander (1998) showed a significant reduction in mortality of insects to DDT- and dieldrin-amended soils aged for 30 days compared to freshly added insecticides. Toxicity decreased with further aging for 180 or 270 days, showing no mortality. About 85 and 92 percent of the contaminant was recovered from the soil by extraction after 180 and 270 days, respectively. The authors concluded that pesticides residing in soil became “less bioavailable” with time. Similar results are reported for herbicides, where the toxicity was less than anticipated based on total sample analysis. Scribner et al. (1992) assessed the bioavailability of simazine residues from a cornfield where simazine had been applied continuously for 20 years. Aged simazine residues were shown to be biologically unavailable to sugarbeets and to microbial degraders, whereas recently added simazine caused damage to sugarbeets and was substantially degraded by microbes. In summary, pesticides can persist in soils for up to 50 years and perhaps much longer. Based on tests with microorganisms, worms, insects, and plants, pesticides may or may not exhibit greatly reduced bioavailability (as measured by degradation, uptake, or toxicity) over the long term. |
dissolved may be readily available. Typically, modern analytical methods are designed to report the total amount of all forms of a compound present in a sample. Thus, the difference between the total amount of a compound detectable using modern analytical techniques and the bioavailable amount of the compound has become a central issue in the environmental arena.
The earliest studies of contaminant bioavailability from soil for the purposes of refining human exposure assessment focused on dioxins and furans in the mid-1980s (Bonaccorsi et al., 1984; McConnell et al., 1984; Lucier et al., 1986; Umbreit et al., 1986; Shu et al., 1988). These were soon followed by similar studies on the oral bioavailability of polychlorinated biphenyls (PCBs) and polyaromatic hydrocarbons (PAHs) from soil (Fries et al., 1989; Goon et al., 1990). The dermal bioavailability of dioxins and furans, PCBs, and PAHs in soil was also under study during this time frame. Starting in the late 1980s, bioavailability
research for human exposures shifted to address inorganics, primarily lead because of the size of many Superfund mining sites in the Rocky Mountain West where childhood exposure to lead in soil was a significant concern. This prompted the development of lead bioavailability models in rats (Freeman et al., 1992) and swine (Casteel et al., 1997a), which were subsequently used to assess lead bioavailability from soil at approximately 20 sites. The success of this approach for lead resulted in the development of analogous models for arsenic in swine and monkeys (Freeman et al., 1995; Casteel, 1997b) and the use of these models to assess arsenic bioavailability from soil and house dust at more than ten sites. Mercury bioavailability also has been the subject of recent investigations (as reviewed in Davis et al., 1997; Paustenbach et al., 1997; Schoof and Nielsen, 1997). Several review documents compile the results from these site-specific bioavailability studies (Battelle and Exponent, 1999, 2000; NEPI, 2000a, b).
Despite this work, for many scenarios there is limited agreement on how to quantify all relevant bioavailability processes at hazardous waste sites, partly because too few compounds have been tested to make generalizations. A large body of information comes from empirical observations suggesting that bioavailability processes are important for assessing the risk of compounds in soil. In particular, for organic chemicals a pattern of chemical disappearance composed of a more rapid initial phase followed by a period in which little or no degradation of chemical can be detected is commonly observed (e.g., Linz and Nakles, 1997). In the case where the compounds are known to be biodegradable, the lack of disappearance in the second phase is taken to mean the compounds are unavailable to microorganisms. In addition, it is argued that the observed slowing in the biodegradation rate of organic compounds in aged samples imposes a limit on what may be achieved by bioremediation. Indeed, in many cases it has been observed that organic–solid partitioning or the aging of organic pollutants in soil and sediment systems results in residues that are recalcitrant to further microbial attack despite favorable environmental conditions (Mihelcic and Luthy, 1991; Alexander, 1995; Ramaswami and Luthy, 1997).
Beyond empirical observations, more quantitative attempts to document bioavailability processes at hazardous waste sites use a variety of techniques including mass transfer measurements, geochemical analyses, microbial responses, extractants that mimic the digestive action of organisms, accumulation or uptake tests (as in the lead model discussed above), and bioassays of acute and chronic responses (for a detailed discussion of tests see Chapter 4). Accumulation into earthworms (e.g., ASTM, 1998) is a relatively simple test that has been widely applied to contaminated soils and sediments for mainly ecological risk assessment purposes. Toxicity bioassays in use for ecological risk assessment (EPA, 1991; EPA/USACE, 1991; Ingersoll, 1995) have generally relied on acute toxicity tests. Where concern has focused on the potential risk associated with longer exposures to low levels of contamination, tests that measure sublethal endpoints such as growth and reproduction have been applied (Dillon et al., 1993;
Benoit et al., 1997; Moore et al., 1997). These approaches offer the advantage of providing a closer link to effects on higher levels of biological organization (e.g., populations and communities), which represent the focus of most ecological risk assessments (Suter, 1993; Bridges et al., 1996). Although bioassays of uptake and effect are most applicable to the test organism (usually microorganisms, clams, worms, and plants), the results may also be relevant to other animals and humans. There is not a long history of developing such surrogates; thus, it should not be surprising that bioavailability has infrequently “been considered in devising or interpreting toxicological tests of higher organisms or in assessments of risks from organic toxicants in soil” (Alexander, 1997).
In addition to the metals-contaminated mining sites mentioned previously, bioavailability also has been seriously considered at former manufactured gas plant (MGP) facilities, which made gaseous fuels from coal and oil prior to the widespread distribution of natural gas following WWII. These plants operated from 50 to 150 years ago, and wastes remain at thousands of sites around the world. Bioavailability processes have emerged as important for assessing environmental exposures and for remediating contaminated soils and sediments at MGP sites (e.g., Luthy et al., 1994; Stroo et al., 2000). The focus has been primarily on the bioavailability of coal tar constituents—specifically PAHs. The implications of bioavailability for biological treatment of these materials also have been evaluated. For example, some treatment technologies have focused on methods of increasing the availability of coal tar constituents (e.g., Ali et al., 1995). In other cases, the goal has been to demonstrate that contaminants in the treated soils or sediments are no longer in an available form and thus pose less risk. This is the case for MGP purifier waste, which contains elevated levels of cyanide compounds that happen to be much less bioavailable than simple cyanide salts (Ghosh et al., 1999). One state—Massachusetts—has tried to account for these differences by developing a method for determining the “physiologically available cyanide” present in soils (MA DEP, 2001).
Another area where bioavailability processes are a primary focus of environmental risk assessment is in the management of coal ash. Ash is one of the largest solid waste residuals associated with energy production from fossil fuels. The Electric Power Research Institute (1983, 1993) has conducted a substantial amount of the research associated with these materials, including development of geochemical models for predicting leaching and transport behavior of the metals in ash. These recent assessments include evaluations of exposure to ecological receptors and incorporate bioavailability processes as reflected by biological uptake factors.
Finally, bioavailability processes are an important component of U.S. Environmental Protection Agency (EPA) regulations concerning the beneficial use of biosolids, which are the residual materials generated by municipal wastewater treatment and applied to land for their fertilizer value. As discussed in greater detail in Chapter 2, the Part 503 Sludge Rule contains risk-based standards de-
signed for land application of biosolids. Initially, the proposed regulations called for limits on the amount of sludge that could be applied to land, based on metal toxicity to certain plants (Marks et al., 1980). As more studies using biosolids were conducted (e.g., Bingham et al., 1975; Dowdy, 1975; Latterell et al., 1978), evidence mounted to suggest that there may be concentrations below which there are no adverse effects from the metals or organics in biosolids—that is, below which the contaminants are not bioavailable (Page et al., 1987). For all exposure pathways other than human ingestion of biosolids, Part 503 regulations currently permit the use of data from such field studies to determine these concentration thresholds and set application rates of biosolids such that metal limits are not exceeded.
Bioavailability in Risk Assessment
Risk assessments provide the foundation for decisions about exposure to chemicals and cleanup of soils and sediments at contaminated sites. Bioavailability processes are important for evaluating exposures of humans and ecological receptors to persistent compounds. Indeed, risk management decisions related to judging the acceptability of dioxins in soils can be traced back to evaluations that explicitly considered bioavailability (Kimbrough et al., 1984). Since that time, some progress has been made in explicitly incorporating bioavailability concepts into risk assessment, particularly for lead contamination of soils and for dermal exposure pathways (see Chapter 2). In general, though, most bioavailability processes are not transparently dealt with during risk assessment, and are instead part of certain assumptions, adjustments, or correction factors, which may or may not be based on experiments. Following is a brief overview of how bioavailability concepts are incorporated into human health and ecological risk assessment. A more thorough examination of the topic is given in Chapter 2.
Human Health Risk Assessment
In human health risk assessment, the term “bioavailability” is specifically used in reference to systemic absorption. This is consistent with the toxicological use of the term “bioavailability,” as explained previously, and is understandable given that human health risk assessment was developed from basic toxicological principles. Bioavailability processes leading up to absorption (processes A–C in Figure 1-1) are also included in human health risk assessments, but typically are not identified as such. Instead, they often are described using other terms, such as “environmental fate and transport” processes.
When bioavailability is considered as the fraction of the chemical that is absorbed into systemic circulation, two operational definitions are important— absolute and relative bioavailability. The amount of chemical that is ingested, lies on the surface of the skin, or is inhaled is called the applied dose. The amount
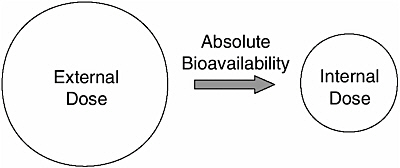
FIGURE 1-2 Absolute bioavailability determines the fraction of the external or applied dose that reaches the systemic circulation (internal dose).
that is absorbed and reaches the systemic circulation is called the internal dose; it is dependent upon the absolute bioavailability of the chemical, i.e., the fraction of the applied dose that is absorbed (Figure 1-2). Clearly, absolute bioavailability can never be greater than 100 percent.
Relative bioavailability represents a comparison of absorption under two different sets of conditions. Examples might include absorption of a chemical from two different routes of exposure, or from the same route of exposure but from two different types of environmental samples. Relative bioavailability says nothing directly about the amount of chemical absorbed into the body; it only describes the relationship between the amount absorbed under two different circumstances. For example, if a chemical is absorbed equally well through the skin as from the gut, the relative bioavailability (dermal versus ingestion) for these exposure routes is 100 percent, even though the fraction absorbed (absolute bioavailability) from each of the routes may be only 5 percent. Relative bioavailability can be greater than or less than 100 percent.
Human health risk assessment involves combining an estimate of exposure with a toxicity value to derive a risk. Most toxicity values are based on applied dose, meaning for example that an acceptable oral daily intake for a chemical is based on the amount ingested per day (usually per unit body weight). Although using applied-dose toxicity values is convenient, the disadvantage is that the toxicity of most chemicals is related more directly to their internal dose. As a result, comparing applied doses to gain inferences on risks can be misleading if the relationship with their corresponding internal doses is not consistent (i.e., if they have different absolute bioavailabilities).
Figure 1-3 shows two circumstances in which a comparison of applied (external) doses is not a valid reflection of the size of the internal doses because the relative bioavailability in each case is not 100 percent (that is, the absolute bioavailability in each case is different than in the test case). It is not difficult to imagine circumstances in which absolute bioavailabilities are not equal, for example when extrapolating from animals to humans, from fasted subjects to fed subjects, or from studies conducted with the test substance in a highly-absorbable
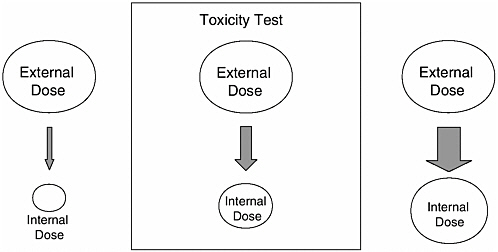
FIGURE 1-3 Comparing applied (external) doses can be problematic if relative bioavailability is not 100 percent. The size of each arrow indicates the magnitude of absolute bioavailability. The upper circles represent the magnitude of each applied (external) dose. If the conditions that prevailed in the toxicity test (center) are different than the actual situation such that relative bioavailability is much greater than (right-hand side) or much less than (left-hand side) 100 percent, comparing applied doses will lead to an overestimating or underestimating of the risk.
form versus environmental exposure to the chemical in a complex matrix. If the absolute bioavailability is less under conditions of environmental exposure than in the critical study used to develop the safe dose, the conclusion that a health risk exists would be in error, as indicated by the left side of Figure 1-3. Comparisons of applied doses still can be useful in these situations if a correction is applied in the form of a relative bioavailability term, which is the major reason that studies to determine relative bioavailability are much more common in human health risk assessment than absolute bioavailability studies. As described in Chapter 2, the results of relative bioavailability studies are used to refine risk calculations that would otherwise assume a relative bioavailability of 100 percent.
Chapter 2 discusses the methodology for estimating doses for ingestion, dermal contact, and inhalation routes of exposure, and the manner in which certain bioavailability processes have been explicitly included in exposure assessment for these routes. Incidental ingestion of contaminated soils is often the most important exposure pathway for human health risk assessment and drives many of the generic and site-specific cleanup criteria for soils contaminated with organic chemicals and metals. The exposure intake equation for incidental ingestion of soil invokes an adjustment factor if the absolute bioavailability for the case of concern is known to differ from the absolute bioavailability implicit in the toxicity value used (i.e., if relative bioavailability is something other than 100
percent). Such calculations are more difficult for dermal exposure pathways, because there are very few toxicity values available specifically for dermal exposure. This necessitates the use of toxicity values developed for other routes of exposure (ingestion or inhalation) and additional relative correction factors to account for bioavailability processes. Default assumptions for dermal bioavailability are often specified by EPA or state agencies (see Chapter 2). The inhalation pathway presents even more complexity because both the doses and the toxicity values are often expressed in terms of concentration in air, rather than an amount of chemical per unit body weight. There are few examples of situations where a bioavailability adjustment factor has been used to refine an inhalation risk assessment. In the absence of information on a specific chemical (which is exceedingly limited), absolute bioavailability from inhalation is usually assumed to be 100 percent.
Ecological Risk Assessment
Bioavailability processes are equally important to ecological risk assessment, and have actually been afforded greater attention here than in human health risk assessment because of the need to encompass an enormous number of exposure pathways. This is evident in long-standing ecotoxicology terms that are related to bioavailability, such as bioconcentration (i.e., accumulation of contaminants from the dissolved phase for aquatic organisms), bioaccumulation (i.e., accumulation of contaminants from all food sources as well as the aqueous phase), and biomagnification (i.e., increase in body burden of contaminants through the food web) (described in greater detail in Chapter 3). The science of ecological risk assessment has grown with extensive research on many bioavailability processes, such as transport of chemicals in the environment and environmental modeling. The number of factors that may alter exposure assessments is enormous, including species-specific criteria, interactions between competing organisms, ecosystem structure, interactions among communities of organisms, and other factors not specifically considered in human health risk assessment.
Bioavailability concepts can be explicitly considered in ecological risk assessments in many ways. With regard to the specific process of absorption, as with human health risk assessment there may be site-specific estimates of relative bioavailability that can be derived either from measurements, from modeling, or from a combination of the two and used in exposure assessment for certain pathways. A limited number of studies using highly bioavailable forms of chemicals have been conducted in organisms of interest. Exposure pathways for ecological risk assessment often involve food-chain models, particularly for bioaccumulative compounds such as PCBs, dioxins, pesticides, and methyl mercury. Thus, site-specific tests and models to determine the bioaccumulation of compounds into the tissues of plants and lower-order animals can be used to evaluate exposures to higher trophic levels such as fish and wildlife.
The importance of bioavailability processes is also acknowledged in screening-level ecological risk assessments. For example, it is standard practice to consider the partitioning of chemicals between sediment, sediment pore water, and animals when evaluating exposures to certain groups of organic chemicals (EPA, 2000b). The degree of partitioning is influenced by the organic content of the sediments, such that solid-phase chemistry data can be used to generate relative bioavailability factors or make other refinements to the ecological risk assessment. In the case of soils, EPA has recently decided to explicitly consider soil properties that influence bioavailability processes in setting screening levels for soil contamination (EPA, 2000c). The various methods by which bioavailability processes are explicitly included in exposure assessment for several common ecological risk assessment pathways are discussed in greater detail in Chapter 2.
IMPLICATIONS OF BIOAVAILABILITY PROCESSES
With regard to solid waste management, there is no doubt that interest in bioavailability processes has been fueled by the recognition that cleanup levels expressed as bulk concentrations in soils and sediments may not correlate with actual risk. The hypothetical example illustrated in Table 1-2 reflects the concern that many remedial engineers have about cleanup decisions based solely on bulk chemical measurements. In this table, contaminant bioavailability (as measured by an unspecified method) decreases in order from Site 1 to 5. Although Site 5 has the highest total contaminant levels, it has the lowest effective contaminant concentration because of limited bioavailability. This illustration shows that it is conceptually possible to reverse the order of importance for dealing with sites when the bioavailable chemical concentration rather than the total chemical concentration is considered.
A consideration of bioavailability processes offers the potential for reducing the volume of soil or sediment requiring remediation. If it can be demonstrated that greater levels of contamination can be left in soil or sediment without risk, decreased cost may be realized and an opportunity for less intrusive remedial approaches exists. Box 1-2 discusses the importance of quantifying the difference
TABLE 1-2 Hypothetical Illustration of How Bioavailability Processes Could Influence Exposure and Remedial Decisions
|
Site # |
Total Contaminant Concentration (ppm) |
Percent Contaminant Bioavailability |
Effective Contaminant Concentration (ppm) |
|
1 |
200 |
100 |
200 |
|
2 |
250 |
75 |
188 |
|
3 |
300 |
50 |
150 |
|
4 |
400 |
33 |
133 |
|
5 |
500 |
20 |
100 |
|
BOX 1-2 Total Concentration vs. Bioavailable Concentration: Metals in Sediment It is important to understand the magnitude of error involved if bioavailability is not considered when evaluating sediment or soil contamination. Significant, even strong, correlations between bioavailability (measured by uptake into tissues or toxicity) and total metal concentration can be found among geochemically similar environments (Bryan, 1985) or within experiments using a single type of sediment (Lee et al., 2000). However, poor correspondence between total metal concentration and bioavailability is common when experiments are conducted with sediments or soils that differ widely in critical geochemical characteristics (Luoma and Jenne, 1977; DiToro et al., 1990, 1991). For example, in a large data set from English estuaries, metal concentrations in fine grained surface sediments (judged to be oxidized by appearance) were compared to concentrations in the tissues of a bivalve and a polychaete that lived within the sediments and ingested sediments with their food (Luoma and Bryan, 1981; Bryan, 1985; Bryan and Langston, 1992). The estuaries included a wide range of physical, biogeochemical, and pollution conditions, and co-variance among geochemical variables was rare. Some sources of variability, such as particle size, large redox differences, or dilution of tissue concentrations by reproductive tissue, were carefully controlled. The results displayed the typical variability of correspondence between metal concentrations in organisms (bioaccumulation) and metal concentrations in sediments. For example, no significant correlation was observed between cadmium in sediments and in the polychaete Neries diversicolor or between copper in sediment and copper in the bivalve Scrobicularia plana. Bioavailability in these cases was completely unpredictable from total metal concentrations in sediments. In contrast, copper in sediments predicted over 50 percent of the variance in copper in the polychaete, and cadmium in sediment predicted over 50 percent of the variance in bivalve cadmium (Bryan, 1985). Silver and lead concentrations in sediments explained about half the variance in bioaccumulation in three species, especially when these elements were extracted from sediments with 0.1M HCl. Clearly, factors that influence bioavailability can differ among metals, species, and environmental factors, and differ with different combinations of these three variables. In the above example, bioavailability processes add variance to the relationship between total concentration and bioaccumulated metal, so the importance of considering bioavailability depends upon how much variance is acceptable (Luoma, 1983, 1989; Landrum et al., 1992). In general, predictions of metal bioavailability from total concentration in sediment alone were outside the two-fold criteria for accuracy suggested by Landrum et al. (1992). If a higher threshold for variance is acceptable, then consideration of bioavailability is less important. Total concentration does appear to provide a first-order control on bioavailability. This control is (statistically) most evident if a large concentration gradient is considered. In the example, total concentration in sediment would be a feasible indicator of the exposure of deposit feeders to most metals if 2- to 50-fold uncertainty were acceptable (the implicit criteria employed by Long et al., 1995, for example). However, because the need to assure less than 50-fold uncertainty exists in many instances, much effort has gone into developing tools and techniques to better relate environmental concentrations and bioavailability. |
between total and bioavailable concentrations of contaminants in soils and sediments, and also the variability of these differences and their dependence upon such factors as the geological materials, the contaminant species present, exposure pathways, and the potential receptors.
Despite the fact that bioavailability has gained popularity as a justification for leaving some contamination in place at hazardous waste sites, in fact the integration of bioavailability processes into risk-based cleanup has the potential to either increase or decrease currently accepted cleanup requirements for residual contamination. To understand this, it should be noted that the term “bioavailability” is often used to refer specifically to uptake or absorption. It is true that absorption efficiency can never be greater than 100 percent, and thus assessments that focus exclusively on absorption efficiency would seem to have the potential to measure only “reduced” bioavailability. However, when other bioavailability processes are taken into account, then it is possible for overall exposure to increase or decrease. That is, although one bioavailability process may suggest that less contaminant is available to a receptor, other bioavailability processes may act as counterbalances, such that the actual dose is not reduced. This is illustrated by the example in Box 1-3, where the overall dose received by an organism is dependent on many factors, including the presence of multiple exposure pathways, ingestion rates, total concentration, and other bioavailability processes. Thus, an examination of all relevant bioavailability processes may actually increase the cost of remediation or alter the remedial technology implemented.
A few points can be made with the example presented in Box 1-3 and Table 1-3. First, many definitions of “bioavailability” are limited to the term in the last column of Table 1-3 (uptake efficiency or absorption). This is somewhat analogous to the terms “absolute bioavailability” and “relative bioavailability” commonly used in human health risk assessment. In the absence of compound-specific data, assumptions about absolute and relative bioavailability are made, with a common assumption being that relative bioavailability is 100 percent (see Chapter 2). Part of the goal of this report is to suggest that experiments be conducted to better define the numbers used in the final column of such a table, numbers that often are based on limited data and may not be applicable in all situations. For example, the default for the relative bioavailability of soil-bound lead via oral ingestion is 60 percent, which may be too low or high in certain situations and for certain soils. Indeed, for most compounds and soil- or sediment-types, absolute and relative bioavailability numbers are not available.
Second, it should be clear from the above discussion that the committee’s concept of bioavailability processes encompasses not only the uptake term in Table 1-3, but also the concentration term and the term dealing with ingestion rates. Gaining a better understanding of all bioavailability processes can help manage contaminated sediments and soils in a way that not only protects the environment but also considers other issues such as costs, permanence, future
land or water use, and community acceptance. As discussed in Box 1-3, management guidelines derived from the viewpoint of a single process can underestimate risk if other important processes are not considered, just as likely as they might overestimate risk.
TASK STATEMENT AND REPORT ROADMAP
Growing interest in bioavailability processes has generated numerous questions among scientists, engineers, risk assessors, managers, regulatory agencies, and other interested parties. It has highlighted a need for better understanding such processes in terms of specific pathways, contaminated media, biological receptors, and even routes of entry. This report seeks to address the most pressing issues and to contribute toward developing common frameworks and language to build a mechanistic-based perspective of bioavailability processes. Several key questions served to guide the work of the committee:
-
What scientific understanding is missing that would provide confidence in the use of bioavailability factors for different contaminant classes? That is, what mechanisms and processes require better understanding? What are the highest priority research needs? For which contaminant classes, environmental settings, and organism classes are bioavailability assessments most important?
-
What tools (biological, chemical, and physical methods) are available to characterize and measure bioavailability for different contaminant classes, and what new tools are needed? What criteria should be used to validate these tools?
-
How do treatment processes affect bioavailability for different contaminant classes? How does bioavailability affect treatment processes that rely on microbial degradation of contaminants?
-
How and when should bioavailability information be used? What are its implications for relevant regulations? How can information on bioavailability be reliably communicated, especially to the public?
This report assesses our current understanding of processes that affect the degree to which chemical contaminants in soils and sediments are bioavailable to humans, animals, microorganisms, and plants. Chapter 2 discusses how the bioavailability concept is used today in solid and hazardous waste management. The legal and regulatory framework for considering bioavailability during soil, sediment, and biosolids management is evaluated as well as the technical methods devised for use in human health and ecological risk assessment. Case studies are presented that illustrate where bioavailability adjustment factors have been used to refine risk assessment calculations.
Because the concept of bioavailability incorporates multiple physical, chemical, and biological processes that affect the concentration and transformation of chemicals in soils, sediments, and aquatic systems, Chapter 3 describes these processes in greater detail and weighs their relative importance in certain envi-
|
BOX 1-3 Multiple Bioavailability Processes Affect Contaminant Intake Several environmental processes affect how a contaminant in soil or sediment is taken into an organism. Viewing bioavailability as a single factor, and then making implicit assumptions about the link between the single process and incorporation of the chemical into an organism, can lead to false conclusions. The example below illustrates how a mix of processes can be relevant to bioavailability of a contaminant in sediments, such as:
Influx rate at the membrane is an unambiguous indicator of incorporation into an animal. Mathematically, influx into an organism (say a sediment dwelling, deposit feeding animal) from a dissolved source is defined as: Influxwater = C × R × A where C is concentration in water (μg/g water), R is the rate at which the animal passes water across the gills (gwater/ganimal/d) and A is the absorption efficiency (what proportion of the total concentration is absorbed into the organism) (Wang et al., 1996). A similar equation defines other exposure routes such as from food, where C is concentration in food (μg/g), R is ingestion rate (g/ganimal/d) and A is the absorption efficiency (what proportion of the total concentration ingested is absorbed into the organism). This equation illustrates the interplay among contaminant concentration, biology, and factors modifying absorption, whatever the exposure route. The importance of considering all three in combination is illustrated in the table below. Table 1-3 presents a hypothetical example using reasonable concentrations from a natural system. The goal is to compare intake from two sources with very different absorption efficiencies (often assumed to define bioavailability). The biological processes are typical of a sediment (deposit) feeding animal, like a bivalve. The feeding rate is 1 g sediment per g tissue per day; the filtration rate is 1000 g water per g tissue per day. The concentrations are typical of a moderate cadmium contaminated sediment: 4 μg Cd/g dry wt in sediment; 0.0002 μg Cd/gpore water in pore water (again, units are converted). Absorption efficiency from water is taken as 0.99 because it is often assumed that absorption from solution is highly efficient. Absorption efficiency from food is typical of cadmium availability for a bivalve |
|
(20 percent). An analysis of the values could lead to the statement that cadmium is more “bioavailable” from water than sediment (because efficiency of absorption is much higher). But if all bioavailability processes are considered, intake is similar between the sources because concentrations are much higher in sediment. Filtration rate and feeding rates can also make great differences in the ultimate exposure. TABLE 1-3 Hypothetical Intake Rates of Cadmium given Two Different Exposure Pathways
The point illustrated by this example has important implications for setting cleanup standards. Determination of the environmental toxicity of chemicals for regulatory purposes is typically based upon bioassay exposures of surrogate organisms to a dissolved chemical, under circumstances that maximize the efficiency of bioavailability process D in Figure 1-1. For example, selenium toxicity was first determined using exposure of fish or invertebrates to selenite in solution, recognizing that selenite is the “most bioavailable” of the oxidation states (the standard condition is assumed to be close to 100 percent absolute bioavailability). Tests typically reported selenite toxicities at concentrations > 70 μg/L (Lemly, 1998). The first case studies of selenium toxicity in nature, however, showed that selenium was responsible for the elimination of most fish species in Belews Lake, but that selenite concentrations were less than 5mg/L (Lemly, 1985). Clearly, in this system “bioavailability” was greater than predicted from the (originally implied) maximum bioavailability, and the standard test had underestimated risk. Interpretation of the lake data and later experimental studies showed that an additional process was responsible for the enhanced risk. Selenium exposure was found to occur primarily from diet, but dietary exposure was not considered in the tests that set the standard (Lemly, 1985; Luoma et al., 1992). The most recent analyses suggest that understanding of selenium risks in nature requires consideration of multiple additional processes (Lemly, 1995; Luoma and Presser, 2000). |
||||||||||||||||||||
ronmental settings. Solubility and sorption, burial and encapsulation, diffusion and advection, microbial transformation and degradation, and uptake into organisms are considered, among other processes.
Chapter 4 of this report describes and evaluates the myriad of methods and techniques for measuring different bioavailability processes for both metal and organic contaminants in soils and sediments. For each method, the report considers what bioavailability process(es) it addresses, for what chemicals and contaminated media it can be used, what endpoint is considered, its cost, and the extent to which it has been validated. Suggestions are given for improving our ability to quantitatively assess bioavailability.
The implications of more explicitly considering bioavailability processes in environmental cleanup constitute Chapter 5. In particular, the chapter discusses for which contaminants and environmental settings measurements of bioavailability are needed and likely to be most beneficial for the protection of human health and ecosystems. A section is devoted to exploring the complex relationship between contaminant bioavailability and success of bioremediation. Finally, it asks how more explicit consideration of bioavailability can be moved into the regulatory arena and also into practice. Because of the importance of regulatory and public buy-in prior to the refinement of risk assessment and the alteration of cleanup goals, the report discusses ways to effectively communicate bioavailability concepts.
REFERENCES
Alexander, M. 1995. How toxic are chemicals in soil? Environ. Sci. Technol. 29:2713-2717.
Alexander, M. 1997. Sequestration and bioavailability of organic compounds in soil. Chapter 1 In: Environmentally Acceptable Endpoints in Soil. D. G. Linz and D. V. Nakles, Eds. Annapolis, MD: American Academy of Environmental Engineers.
Alexander, M. 2000. Aging, bioavailability, and overestimation of risk from environmental pollutants. Environ. Sci. Technol. 34:4259-4265.
Ali, M. A., D. A. Dzombak, and S. B. Roy. 1995. Assessment of in situ solvent extraction for remediation of coal tar sites: process modeling. Water Environment Research 67(1):16-24.
American Society for Testing and Materials (ASTM). 1998. Standard guide for conducting laboratory soil toxicity or bioaccumulation test with the lumbricid earthworm Eisenia foetida. E 1676-97. Philadelphia, PA: ASTM.
Anderson, W. C., R. C. Loehr, and B. P. Smith. 1999. Environmental availability of chlorinated organics, explosives, and metals in soils. Annapolis, MD: American Academy of Environmental Engineers.
Bailey, G. W., and J. L. White. 1970. Factors influencing the adsorption, desorption, and movement of pesticides in soil. Residue Rev. 32:29-92.
Bartels, J. M., and D. L. Sparks (eds.). 1996. Methods of soil analysis, part 3: chemical methods. Madison, WI: Soil Science Society of America, Inc.
Battelle and Exponent. 1999. Guide for incorporating bioavailability adjustments into human health and ecological risk assessments at U.S. Navy and Marine Corps facilities (draft final). Part 1: overview of metals bioavailability. Port Hueneme, CA: Naval Facilities Engineering Command.
Battelle and Exponent. 2000. Guide for incorporating bioavailability adjustments into human health and ecological risk assessments at U.S. Navy and Marine Corps facilities. Part 2: technical background document for assessing metals bioavailability. Port Hueneme, CA: Naval Facilities Engineering Command.
Benoit, D. A., P. K. Sibley, J. L. Juenemann, and G. T. Ankley. 1997. Chironomus tentans life-cycle test: design and evaluation for use in assessing toxicity of contaminated sediments. Environ. Toxicol. Chem. 16:1165-1176.
Bingham F. T., A. L. Page, R. J. Mahler, and T. J. Ganje. 1975. Growth and cadmium accumulation of plants grown on a soil treated with a cadmium enriched sewage sludge. J. Environ. Qual. 4(2):207-211.
Blumhorst, M. R., J. B. Weber, and L. R. Swain. 1990. Efficacy of selected herbicides as influenced by soil properties. Weed Technology 4:279-283.
Bonaccorsi, A., A. di Domenico, R. Fanelli, F. Merli, R. Motta, R. Vanzati, and G.A. Zapponi. 1984. The influence of soil particle adsorption on 2,3,7,8-tetrachlorodibenzo-p-dioxin biological uptake in the rabbit. Arch. Toxicol. Suppl. 7:431-434.
Boul, H. L., M. L. Garnham, D. Hucker, D. Baird, and J. Aislabie. 1994. Influence of agricultural practices on the levels of DDT and its residues in soil. Environ. Sci. Technol. 28:1397-1402.
Bridges, T. S., R. B. Wright, B. R. Gray, A. B. Gibson, and T. M. Dillon. 1996. Chronic toxicity of Great Lakes sediments to Daphnia magna: elutriate effects on survival, reproduction, and population growth. Ecotoxicology 5:83-102.
Bryan, G. W., and W. J. Langston. 1992. Bioavailability, accumulation and effects of heavy metals in sediments with special reference to United Kingdom estuaries: a review. Environ. Pollutn. 76:89-131.
Bryan, G. W. 1985. Bioavailability and effects of heavy metals in marine deposits. Pp. 42-79 In: Wastes in the Ocean: Nearshore Waste Disposal. B. H. Ketchum, J. M. Capuzzo, W. V. Burt, I. W. Duedall, P. K. Park and D. R. Kester (eds.). New York: John Wiley and Sons.
Casarett and Doull’s. 2001. Toxicology. The Basic Science of Poisons, Sixth Edition. C. D. Klaassen (ed.). New York: McGraw-Hill.
Casteel, S. W., R. P. Cowart, C. P. Weis, G. M. Henningsen, E. Hoffman, W. J. Brattin, R. E. Guzman, M. F. Starost, J. T. Payne, S. L. Stockham, S. V. Becker, J. W. Drexler, and J. R. Turk. 1997a. Bioavailability of lead to juvenile swine dosed with soil from the Smuggler Mountain NPL site of Aspen, Colorado. Fund. Appl. Toxicol. 36:177-187.
Casteel, S. W., L. D. Brown, M. E. Dunsmore, C. P. Weis, G. M. Henningsen, E. Hoffman, W. J. Brattin , and T. L. Hammon. 1997b. Relative bioavailability of arsenic in mining wastes. Prepared for U.S. Environmental Protection Agency, Region VIII, Denver, CO. Doc. Control No. 4500–88–AORH.
Chaney, R. L. 1994. Diagnostic practices to identify iron deficiency in higher plants. J. Plant Nutr. 7:47-67.
Davis, A., N. S. Bloom, and S. S. Que Hee. 1997. The environmental geochemistry and bioaccessibility of mercury in soils and sediments: a review. Risk Analysis 17(5):557-569.
Dillon, T. M., D. W. Moore, and A. B. Gibson. 1993. Development of a chronic sublethal bioassay for evaluating contaminated sediment with the marine polychaete worm Nereis (Neanthes) arenaceodentata. Environ. Toxicol. Chem. 12:589-605.
DiToro, D. M., J. D. Mahony, D. J. Hansen, K. J. Scott, M. B. Hicks, S. M. Mayr, and M. S. Redmond. 1990. Toxicity of cadmium in sediments: the role of acid volatile sulfides. Environ. Toxicol. Chem. 9:1487-1502.
DiToro, D. M., C. S. Zarba, D. J. Hansen, W. J. Berry, R. C. Swartz, C. E. Cowan, S. P. Pavlou, H. E. Allen , N. A. Thomas, and P. R. Paquin. 1991. Technical basis for establishing sediment quality criteria for nonionic organic chemicals using equilibrium partitioning. Environ. Toxicol. Chem. 10:1541-1583.
Dowdy, R. 1975. The availability of sludge-borne metals to various vegetable crops. J. Environ. Qual. 4(3-2):278-282.
Electric Power Research Institute (EPRI). 1983. Demonstration of saturated and unsaturated groundwater flow modeling at a coal ash and dry FGD disposal site. EPRI Report CS-3173. 110 p.
Electric Power Research Institute (EPRI). 1993. FASTCHEM, MYGRT V2.0 used to track coal pile and ash leachates. EPRI Product IN-103051.
Environmental Protection Agency (EPA). 1991. Methods for measuring the acute toxicity of effluents and receiving waters to freshwater and marine organisms. EPA/600/4-90-027. Cincinnati, OH: EPA Environmental Monitoring Systems Laboratory.
EPA. 2000a. Bioaccumulation testing and interpretation for the purpose of sediment quality assessment: status and needs. EPA-823-R-00-001. Washington, DC: EPA Office of Water and Office of Solid Waste.
EPA. 2000b. Technical basis for the derivation of equilibrium–partitioning sediment guidelines (ESGs) for the protection of benthic species: nonionic organic contaminants. EPA-822-R-00-001. Washington, DC: EPA Office of Science and Technology and Office of Research and Development.
EPA. 2000c. Ecological soil screening level guidance. Draft. Washington, DC: EPA Office of Emergency and Remedial Response.
EPA/U.S. Army Corps of Engineers. 1991. Evaluation of dredged material proposed for ocean disposal (testing manual). Implementation manual for section 103 of Public Law 92-532 (Marine Protection, Research, and Sanctuaries Act of 1972). Vicksburg, MS: U.S. Army Engineer Waterways Experiment Station.
Freeman, G. B., J. D. Johnson, J. M. Killinger, S. C. Liao, P. I. Feder, A. O. Davis, M. V. Ruby, R. L. Chaney, S. C. Lovre, and P. D. Bergstrom. 1992. Relative bioavailability of lead from mining waste soil in rats. Fund. Appl. Toxicol. 19:388-398.
Freeman, G. B., R. A. Schoof, M. V. Ruby, A. O. Davis, J. A. Dill, S. C. Liao, C. A. Lapin, and P. D. Bergstrom. 1995. Bioavailability of arsenic in soil and house dust impacted by smelter activities following oral administration in cynomolgus monkeys. Fund. Appl. Toxicol. 28:215-222.
Fries, G. F., G. S. Marrow, and C. J. Somich. 1989. Oral bioavailability of aged polychlorinated biphenyl residues contained in soil. Bull. Environ. Contam. Toxicol. 43:683-690.
Ghosh, R. S., D. A. Dzombak, and R. G. Luthy. 1999. Equilibrium precipitation and dissolution of iron cyanide solids in water . Environmental Engineering Science 16:293-313.
Goodman and Gilman. 2001. The pharmacological basis of therapeutics, Tenth Edition. J. G. Hardman, L. E. Limbird, and A. Goodman Gilman (eds). New York: McGraw-Hill.
Goon, D., N. S. Hatoum, J. D. Jernigan, S. L., Schmidt, and P. J. Garvin. 1990. Pharmacokinetics and oral bioavailability of soil-adsorbed benzo(a)pyrene (B(a)P) in rats. Toxicologist 11:1356.
Hance, R. J. 1967. Relationship between partition data and the adsorption of some herbicides by soils. Nature 214:630-31.
Holt, L. E., and P. H. Holz. 1963. The black bottle: A consideration of the role of charcoal in the treatment of poisoning in children. J. Pediatrics 63:306-314.
Hrudy, S. E., W. Chen, and C. G. Rousseaux. 1996. Bioavailability in environmental risk assessment. Boca Raton, FL: CRC Press.
Ingersoll, C. G. 1995. Sediment tests. Pp. 231-255 In: Fundamentals of Aquatic Toxicology, 2nd edition. G. M. Rand (ed.). Washington, DC: Taylor and Francis.
Kimbrough, R. D., H. Falk, and P. Stehr. 1984. Health implications of 2,3,7,8-tetrachlorodibenzodioxin (TCDD) contamination of residential soil. J. Toxicol. Environ. Health 14:47-93.
Landrum P. F., H. Lee II, and M. J. Lydy. 1992. Toxicokinetics in aquatic systems: model comparisons and use in hazard assessment. Environ. Toxicol. Chem. 11:1709-1725.
Lanno, R. 2001. Presentation to the NRC Committee on Bioavailability of Contaminants in Soils and Sediments. April 23, 2001.
Latterell J. J., R. H. Dowdy, and W. E. Larson. 1978. Correlation of extractable metals and metal uptake of snap beans grown on soil amended with sewage sludge. J. Environ. Qual. 7:435-440.
Lee, B.-G., S. B. Griscom, J.-S. Lee, H. J. Choi, C.-H. Koh, S. N. Luoma, and N. S. Fisher. 2000. Influence of dietary uptake and reactive sulfides on metal bioavailability from aquatic sediments. Science 287:282-284.
Lemly, A. D. 1985. Toxicology of selenium in a freshwater reservoir: Implications for environmental hazard evaluations and safety. Ecotoxicol. Environ. Safety 10:314-338.
Lemly, A. D. 1995. A protocol for aquatic hazard assessment of selenium. Ecotoxicol. Environ. Safety 34: 223-227.
Lemly, A. D. 1998. Pathology of selenium poisoning. Pp. 281-296 In: Environmental Chemistry of Selenium. W. Frankenberger and R. A. Engberg (eds.). New York: Marcel Dekker Inc.
Linz, D. G., and D. V. Nakles. 1997. Environmentally acceptable endpoints in soil. Annapolis, MD: American Academy of Environmental Engineers.
Long, E. R., D. D. Macdonald, S. L. Smith, and F. D. Calder. 1995. Incidence of adverse biological effects within ranges of chemical concentrations in marine and estuarine sediments. Environ. Mgt. 19:81-97.
Lucier, G. W., R. C. Rumbaugh, Z. McCoy, R. Hass, D. Harvan, and P. Albro. 1986. Ingestion of soil contaminated with 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) alters hepatic enzyme activities in rats. Fund. Appl. Toxicol. 6:364–371.
Luoma, S. N., and E. A. Jenne. 1977. The availability of sediment-bound cobalt, silver, and zinc to a deposit-feeding clam. Pp. 213-230 In: Biological Implications of Metals in the Environment. R. W. Wildung and H. Drucker (eds.).
Luoma, S. N., and G. W. Bryan. 1981. Statistical assessment of the form of trace metals in oxidized estuarine sediments employing chemical extractants. Sci. Total Environ. 17:165-196.
Luoma, S. N. 1983. Bioavailability of trace metals to aquatic organisms—a review. Sci. Total Environ. 28:1-22.
Luoma, S. N. 1989. Can we determine the biological availability of sediment-bound trace elements? Hydrobiologia 176/177:379-396.
Luoma, S. N., and T. S. Presser. 2000. Forecasting selenium discharges to the San Francisco Bay-Delta estuary: ecological effects of a proposed San Luis drain extension. USGS Professional Paper 00-416-RTS. 210 pp.
Luoma, S. N., C. Johns, N. Fisher, N. A. Steinberg, R. S. Oremland, and J. R. Reinfelder. 1992. Determination of selenium bioavailability to a benthic bivalve from particulate and solute pathways. Environ. Sci. Technol. 26:485-491.
Luthy, R. G., D. A. Dzombak, C. A. Peters, S. B. Roy, A. Ramaswami, D. V. Nakles, and B. R. Nott. 1994. Remediating tar-contaminated soils at manufactured gas plant sites. Environ. Sci. Technol. 28(6):266A-276A.
Luthy, R. G., G. R. Aiken, M. L. Brusseau, S. D. Cunningham, P. M. Gschwend, J. J. Pignatello, M. Reinhard, S. Traina, W. J. Weber, Jr., and J. C. Westall. 1997. Sequestration of hydrophobic organic contaminants by geosorbents. Environ. Sci. Technol. 31(12):3341-3347.
Marks, M. J., J. H. Williams, and C. G. Chumbley. 1980. Field experiments testing the effects of metal contaminated sewage sludges on some vegetable crops. Pp. 235-251 In: Inorganic pollution and agriculture : proceedings of a conference organised by the Agricultural Development and Advisory Service April 1977. London: H.M.S.O.
Marschner, H. 1995. Mineral nutrition of higher plants. London: Academic Press.
Massachusetts DEP. 2001. Quality assurance and quality control requirements and performance standards for SW-856 Method 9014 (modified), total and physiologically available cyanide (PAC), when utilized in support of assessment and evaluation decisions at disposal sites regulated under M.G.L. c. 21E and 310 CMR 40.0000, the Massachusetts Contingency Plan (MCP). Boston, MA: MA Department of Environmental Protection.
McConnell, E. E., G. W. Lucier, R. C. Rumbaugh, P. W. Albro, D. J. Harvan, J. R. Hass, and M. W. Harris. 1984. Dioxin in soil: bioavailability after ingestion by rats and guinea pigs. Science 223:1077–1079.
Mihelcic, J. R., and R. G. Luthy. 1991. Sorption and microbial degradation of naphthalene in soil-water suspensions under denitrification conditions. Environ. Sci. Technol. 25(1):169-177.
Moore, D. W., T. S. Bridges, B. R. Gray, and B. M. Duke. 1997. Risk of ammonia toxicity during sediment bioassays with the estuarine amphipod Leptocheirus plumulosus. Environ. Toxicol. Chem. 16:1020-1027.
Morrison, D. E., B. K. Robertson, and M. Alexander. 2000. Bioavailability to earthworms of aged DDT, DDE, DDD, and dieldrin in soil. Environ. Sci. Technol. 34: 709-713.
Mueller-Warrant, G. W. 1999. Duration of control from preemergence herbicides for use in non-burned grass seed crops. Weed Tech. 13:439-449.
Nash, R. G., and E. A. Woolson. 1967. Persistence of chlorinated hydrocarbon insecticides in soils. Science 157:924-927.
National Environmental Policy Institute (NEPI). 1997. Bioavailability: the policy impact of emerging science. Washington, DC: NEPI.
NEPI. 2000a. Assessing the bioavailability of metals in soil for use in human health risk assessments. Washington, DC: NEPI.
NEPI. 2000b. Assessing the bioavailability of organic chemicals in soil for use in human health risk assessments. Washington, DC: NEPI.
Page, A. L., T. J. Logan, and J. A. Ryan (eds.). 1987. Land application of sludge: food chain implications. Chelsea, MI: Lewis Publishers.
Paustenbach, D. J., G. M. Bruce, and P. Chrostowski. 1997. Current views on the oral bioavailability of inorganic mercury in soil: implications for health risk assessments. Risk Analysis 17(5):533-544.
Pierzynski, G., J. T. Sims, and G. F. Vance. 2000. Soils and environmental quality. Boca Raton, FL: CRC Press.
Ramaswami, A., and R. G. Luthy. 1997. Measuring and modeling physicochemical limitations to bioavailability and biodegradation. Chapter 78 In: Manual of Environmental Microbiology. C. J. Hurst (ed.). Washington, DC: ASM Press.
Rand, G. M., and S. R. Petrocelli, Eds. 1985. Fundamentals of aquatic toxicology: effects, environmental fate, and risk assessment, 2nd Ed. New York: Hemisphere Publishing Co.
Robertson, B. K., and M. Alexander. 1998. Sequestration of DDT and dieldrin in soil: disappearance of acute toxicity but not the compounds. Environ. Toxicol. Chem. 17:1034-1038.
Ruby, M., R. Schoof, W. Brattin, M. Goldade, G. Post, M. Harnois, D. E. Mosby, S. W. Casteel, W. Berti, M. Carpenter, D. Edwards, D. Cragin, and W. Chappell. 1999. Advances in evaluating the oral bioavailability of inorganics in soil for use in human health risk assessments. Environ. Sci. Technol. 33(21):3697-3705.
Sayler, G. S., U. Matrubutham, F.-M. Menn, W. H. Johnson, and R. D. Stapleton, Jr. 1998. Molecular probes and biosensors in bioremediation and site assessment. Pp. 385-434 In: Bioremediation: Principles and Practice. Volume 1 Fundamentals and Applications. Subhas K. Sikdar and Robert L. Irvine (eds.). Lancaster: Technomic Pub. Co.
Schnoor, J. L. 1996. Environmental modeling: fate and transport of pollutants in water, air, and soil. New York: J. Wiley and Sons.
Schoof, R. A., and J. B. Nielsen. 1997. Evaluation of methods for assessing the oral bioavailability of inorganic mercury in soil. Risk Analysis 17(5):545-555.
Scribner, S. L., T. R. Benzing, S. Sun and S. A. Boyd. 1992. Desorption and bioavailability of aged simazine residues in soil from a continuous corn field. J. Environ. Qual. 21:115-120.
Shu, H., D. Paustenbach, F. J. Murray, et al. 1988. Bioavailability of soil-bound TCDD: oral bioavailability in the rat. Fund. Appl. Toxicol. 10:648-654.
Sposito, G. 1989. The chemistry of soils. New York: Oxford University Press.
Stevenson, F. J. 1994. Humus chemistry: genesis, composition, reactions. New York: John Wiley and Sons.
Stroo, H. F., R. Jensen, R. C. Loehr, D. V. Nakles, A. Fairbrother, and C. B. Liban. 2000. Environmentally acceptable endpoints for PAHs at a manufactured gas plant site. Environ. Sci. Technol. 34(18):3831-3836.
Suter, G. W. 1993. Ecological risk assessment. Boca Raton, FL: Lewis Publishers.
Thompson, C. J. S. 1931. Poisons and poisoners with historical accounts of some famous mysteries in ancient and modern times. London: Harold Shaylor.
Umbreit, T. H., E. J. Hesse, and M. A. Gallo. 1986. Comparative toxicity of TCDD contaminated soil from Times Beach, Missouri, and Newark, New Jersey. Chemosphere 15(9-12):2121-2124.
Walker, A., G. G. Briggs, M. P. Greaves, R. J. Hance, and A. R. Thompson. 1982. Herbicides in soil. Pp 87-105 In: Weed Control Handbook: Principles. H. A. Roberts (ed.). England: Blackwell Scientific Publications.
Wang, W.-X., N. S. Fisher, and S. N. Luoma. 1996. Kinetic determinations of trace element bioaccumulation in the mussel, mytilus edulis. Marine Ecology Prog. Ser. 140:91-113.
Wilkinson, G. R. 2001. Pharmacodynamics. The dynamics of drug absorption, distribution, and elimination. In: Goodman & Gilman’s The Pharmacological Basis of Therapeutics, Tenth Edition. J. G. Hardman, L. E. Limbird, and A. Goodman Gilman (eds.). New York: McGraw-Hill.
































