3
Coordinating the Roles of the Federal Government to Enhance Quality of Care
|
Summary of Chapter Recommendations The federal government has the central role in shaping all aspects of the health care sector. Strong federal leadership, a clear direction in pursuit of common aims, and consistent policies and practices across all government health care functions and programs are needed to raise the level of quality for the programs’ beneficiaries and to drive improvement in the health care sector overall. RECOMMENDATION 2: The federal government should take maximal advantage of its unique position as regulator, health care purchaser, health care provider, and sponsor of applied health services research to set quality standards for the health care sector. Specifically: a. Regulatory processes should be used to establish clinical data reporting requirements applicable to all six major government health care programs. b. All six major government health care programs should vigorously pursue purchasing strategies that encourage the adoption of best practices through the release of public domain comparative quality data and the provision of financial and other rewards to providers that achieve high levels of quality. c. Not only should health care delivery systems operated by the public programs continue to serve as laboratories for the development of innovative 21st-century care delivery models, but much greater emphasis should be placed on the dissemination of findings and, in the case of information technology, the creation of public-domain products. d. Applied health services research should be expanded and should emphasize the development of knowledge, tools, and strategies that can support quality enhancement in a wide variety of settings. |
OVERVIEW OF FEDERAL ROLES
The federal government plays a number of different roles in the American health care arena, including regulator; purchaser of care; provider of health care services; and sponsor of applied research, demonstrations, and education and training programs for health care professionals. Each of these roles can support the accomplishment of somewhat different objectives along the spectrum from quality assurance to quality improvement to quality innovation.
As discussed in Chapter 1, research demonstrates wide variability in the quality of health care. As illustrated in Figure 3-1, some proportion of care is poor and unsafe; no patient should be exposed to this care. Some care is adequate, but not as good as it should be. Most of the services received by patients are effective, but the benefits are not as great as they could be, and resource use is unnecessarily high. Some fraction of patients receive very good care that is consistent with best practices, and an even smaller fraction probably receives excellent care employing state-of-the-art practices. Efforts to improve quality seek to shift the curve to the right and to truncate the left tail of the distribution.
It is through its regulator role that the federal government establishes minimal health care standards. Effective regulatory requirements protect beneficiaries from incompetent, impaired, and inadequately trained clinicians and from health care organizations that lack the requisite capabilities and processes to provide a minimal level of quality. Although regulatory “floors” can continually be raised, thus tightening the distribution of services by quality, regulatory approaches most often seek to cull substandard providers—to truncate the left tail of the distribution. Regulatory requirements have generally been set at levels that nearly all providers could satisfy. Regulatory requirements can have adverse impacts as well, by creating unnecessary reporting burdens, conveying conflicting objectives, and omitting essential elements of quality.
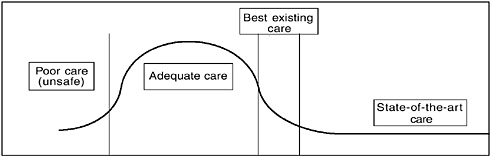
FIGURE 3-1 Distribution of care by level of quality, a conceptual scheme.
Although the federal government has for decades relied extensively on regulatory strategies to address quality concerns, there has been considerable evolution in the types of regulatory requirements. Traditionally, regulatory requirements focused on quality assurance—structural or competency requirements for hospitals (e.g., all hospitals must have well-defined infection control processes) or health care professionals (e.g., physicians and nurses must attain a given level of training and maintain current state licenses). Over decades, regulatory strategies, especially those applicable to the government programs that deliver care through the private sector (i.e., Medicare, Medicaid, and the State Children’s Health Insurance Program [SCHIP]), have incorporated quality improvement approaches that focus more on demonstrating improvement in care processes and patient outcomes. For example, Medicare+Choice (M+C) health plans must collect data on specific performance measures and demonstrate improvement over time. Regulatory strategies that focus on quality improvement offer some potential to shift the quality distribution to the right, although very little is known about which of these approaches works best.
It is this transition from quality assurance to quality improvement strategies that has also broadened the potential for the government to strengthen its roles as purchaser and health care provider. Quality improvement strategies emphasize direct measurement of the clinical quality of care and of patient perceptions and outcomes, and these data then enable differentiation of various levels of quality.
In its purchaser role, the government could reward providers that achieve high levels of quality. Purchasing strategies can raise the quality of care provided by the majority of providers thus shifting the curve to the right. Such strategies include public disclosure of comparative quality data on providers and health plans, and financial and other rewards for high levels of quality.
The disclosure of comparative performance data on hospitals, health plans, physicians, and other providers draws attention to best practices in hopes of encouraging other providers to adopt them. To the extent that consumers act on this information when making choices, health care providers have incentives to improve their performance, thus increasing demand for their services and their market share. Public disclosure of comparative quality data may spur action on the part of providers themselves or professional groups, with steps being taken to encourage poor performers to enhance their knowledge and skills or limit the scope of their practice. Furthermore, public disclosure may stimulate public support for the exercising of regulatory authority by federal or state governments to address persistent poor performance.
The purchaser role also relies on linkages between payment and per-
formance. The federal government (acting on behalf of program beneficiaries) might engage in selective contracting with the highest-quality providers. Providers (e.g., hospitals, physicians, and plans) that achieved exemplary performance might receive higher fees, diagnosis related group (DRG) payments, capitation rates, or bonuses. Proper risk adjustment (see Chapter 2) is critical to payment strategies that reward quality, as public recognition also attracts patients with more complex care needs.
In its provider role, the federal government assumes all the responsibilities of ownership of health care institutions, employer of the health care workforce, and manager and operator of comprehensive delivery systems. In this capacity, it has an opportunity to serve as a laboratory in which to test new financing, delivery, and information dissemination models, while experimenting with various quality measurement and improvement strategies. Just as performance measurement activities have proliferated within the regulatory requirements for Medicare, Medicaid, and SCHIP, performance measurement and improvement have become an integral component of the clinical management processes of the Veterans Health Administration (VHA), Department of Defense (DOD) TRICARE, and Indian Health Service (IHS) programs. As discussed in Chapter 5, VHA and DOD have also led the way in building clinical information systems to support care delivery, quality improvement, patient safety, surveillance and monitoring, and many other applications.
As a major sponsor of applied health services research, the federal government provides support for the development of the knowledge and creation of the tools needed to carry out more effectively the regulator, purchaser, and health care delivery roles. In recent years, the focus of state of the art quality enhancement has shifted toward the measurement of clinical quality (i.e., medical care processes and outcomes) and consumer perceptions. Through the Agency for Healthcare Research and Quality (AHRQ) and other applied research programs sponsored by the National Institutes of Health (NIH), VHA, the Food and Drug Administration, and the Centers for Disease Control and Prevention, the federal government can assist in the development of quality measures, survey instruments, and public reporting tools to enhance federal and state regulatory functions and public and private purchasing activities. The federal government also supports applied health services research that addresses many of the broader health care financing and delivery issues important to creating an environment that supports quality. For example, AHRQ conducts applied research and demonstrations on payment approaches and quality incentives, health care delivery models, and clinical decision-support systems.
The committee recognizes that the federal government influences the health care sector in numerous other ways that are outside the immediate
scope of this project. NIH provides extensive support for clinical research designed to expand the scientific knowledge base and develop new medical technology. The federal government provides extensive support for the education and training of health care professionals, and these programs offer yet another opportunity for the federal government to influence quality and safety. The Medicare program provides about two-thirds of the approximately $18 billion spent annually to educate medical residents (Anderson et al., 2001). In fiscal year 2001, the Bureau of Health Professions and the Bureau of Primary Health Care devoted about $460 million to health professions training, including physicians, nurses, dentists, allied health professionals, and public health practitioners (MedPAC, 2001). NIH provides support for the training of researchers through a variety of mechanisms (Association of American Medical Colleges, 2001). The federal and state governments provide further support for the health care sector through tax policy, including the exclusion of employers’ contributions to group health insurance from taxable income for employees, granting of tax exempt status to many health care institutions, and individual tax deductions for certain health care expenditures (Arnett, 1999).
This chapter is not intended to provide a comprehensive assessment of all of these government roles, but rather a focused review of how the government might better employ some of them in carrying out quality enhancement processes. Specifically, the remainder of this chapter examines some of the ways in which the government health care programs could employ regulatory, purchasing, and care delivery strategies in their quality enhancement processes. Current efforts to standardize and coordinate those processes across the six programs are reviewed, and an overall approach to quality enhancement that maximally leverages the various government roles is outlined. Chapter 6 provides a discussion of the role of applied health services research in strengthening quality enhancement processes.
REGULATORY STRATEGIES
Each of the government health care programs has pursued a regulatory approach to some degree. In general, the programs that pay for services provided through the private sector (i.e., Medicare, Medicaid, SCHIP, and DOD TRICARE) rely more on regulatory approaches than do those that provide most services directly through government-owned provider organizations (i.e., VHA, DOD TRICARE, and IHS). The latter programs have the option of encouraging quality improvement through internal quality management activities (see the discussion of the government’s care delivery role below).
All government programs rely to some extent on a patchwork of both
federal and state laws and enforcement programs that are intertwined. Historically, state laws have established certain requirements for the licensing of clinicians and institutional providers, and form the bedrock of the regulatory system. Layered over state laws are various federal regulatory requirements, many of which are tied to state or local law.
A sizable body of federal regulatory requirements pertaining to Medicare and, to a lesser degree, Medicaid has slowly but steadily accumulated over the years (American Hospital Association, 2002). It is through these two government programs, which contract with the majority of private providers, that the federal government has the greatest impact on the nation’s health care delivery system. Other government health care programs tend to apply many of Medicare’s regulatory requirements.
In most government programs, regulatory standards focus on institutional providers, clinicians, and health plans that seek to receive payment from or deliver care under an identified program (see Tables 3-1 and 3-2). In the Medicaid and SCHIP programs, however, regulatory requirements are the responsibility of the state governments that administer the programs, and it is the responsibility of the state to ensure that providers and health plans satisfy federal requirements.
In general, regulatory standards fall into two groups—standards of participation and external review processes—although the lines of distinction are not always clear. Most standards of participation are aimed at ensuring that providers have and/or maintain certain key competencies, while external review refers to the assessment of provider performance (e.g., care processes and patient outcomes) by an independent organization, usually a quality improvement organization (QIO).
Standards for Participation
A good deal of the regulation in each government program’s portfolio is intended to ensure that program participants possess minimal levels of competence and comply with health and safety requirements. For institutions, these include requirements pertaining to physical safety and sanitation, as well as such organizational competencies as governance, internal quality review, credentialing of medical staff, and medical records management.
Typically, these minimal participatory standards require compliance with state and local licensing laws as a threshold requirement for participation, with some variations. In the DOD TRICARE and IHS programs, clinicians can be licensed in any one of the states. In addition to licensure, most clinicians must comply with state scope-of-practice rules. Such rules are an important determinant of (1) the availability and choice of clinicians (e.g., a broad scope-of-practice for nonphysician providers increases
TABLE 3-1 Overview of Regulatory Requirements in Medicare, Medicaid, and SCHIP
|
|
MEDICARE |
MEDICAID |
|
||
|
Fee-for-Service (FFS) |
Managed Care |
Medicaid FFS |
Medicaid Managed Carea |
SCHIP |
|
|
Target entities |
Institutional providers and clinicians that receive Medicare reimbursement. |
Medicare+Choice plans |
Institutional providers and clinicians that receive Medicaid reimbursement |
State Medicaid programs and managed care plans that enroll Medicaid beneficiaries |
State SCHIP programs |
|
Requirements |
Must meet standards for physical structure, governance, quality assurance, staff credentialing, infection control, etc. Participation in external review projects is voluntary; other health care institutions must respond to data requests from QIOs. |
Must implement a quality improvement process and show results using the Medicare HEDIS, CAHPS, and the Health Outcomes Survey. Participation in external review is mandatory. |
Medicare rules apply for institutional providers. |
The Medicaid program must ensure that managed care plans use a quality improvement process that collects, assesses, and reports performance data to clinicians. States must contract with an EQROb for annual quality reviews and conduct annual medical audits of each managed care contractor. |
The State Child Health Plan must describe performance goals; how progress will be assessed; and assure CMS that the state will collect, assess, and report standardized data. External review requirements are at states’ discretion. |
TABLE 3-2 Overview of Regulatory Requirements: VHA, DOD TRICARE, and IHS
|
|
VHA |
DOD TRICARE |
IHS |
|
Target entities |
Institutional providers and clinicians that provide care to VHA beneficiaries |
Institutional providers, clinicians, and networks that serve TRICARE beneficiaries |
IHS-funded institutional providers and clinicians |
|
Requirements |
VHA hospitals, facilities, and other providers must be accredited by JCAHO or some other accrediting group. Clinicians must be credentialed according to VHA policies and JCAHO standards. An external review program covers all VHA facilities. The current contractor is the West Virginia Medical Institute, Inc. |
Institutional providers, clinicians, and networks must be Medicare-approved (where relevant). Except for operational ambulatory clinics (treating active-duty personnel only), all “fixed” hospitals and freestanding ambulatory clinics must be accredited by JCAHO or some other applicable accrediting group A national external review program is carried out by KePRO, Inc. |
Most IHS facilities are accredited by JCAHO or the Accreditation Association for Ambulatory Health (AAAHC), or certified by CMS (whichever is relevant). Must meet the external review requirements of the Medicare and Medicaid programs. |
|
Enforcement |
Failure to comply disqualifies clinicians from serving VHA beneficiaries. Deficiencies in compliance generally lead to corrective authorized provider. |
Failure to meet the quality standards and certification requirements may result in termination of payments and identification as a non-action initiatives. |
Deficiencies in compliance generally lead to corrective action initiatives. |
|
SOURCES: Department of Defense, 1995, 2001; Indian Health Service, 2001b; Pittman, 2002; and Veterans Administration, 2001. |
|||
the supply of primary care providers); (2) the degree of interdependence and authority of various types of health care professionals (Cooper et al., 1998); (3) the ability to deliver care through multidisciplinary teams (e.g., proscriptive state scope-of-practice acts limit innovation in redefining roles and functions performed by nonphysician health care professionals) (Sage and Aiken, 1997); and (4) the development of approaches to care
delivery and organization that cross state lines (e.g., provision of care through use of the Internet and multistate provider groups) (Finocchio et al., 1998; Rosenfeld et al., 2000; Sage and Aiken, 1997). Participation standards reflect a good deal of consistency among programs. Most of the federal programs require that providers conform to Medicare standards of participation, but there are some exceptions. For example, SCHIP programs that do not operate as Medicaid expansions are required to conform only to state-established standards of participation.
Enforcement of compliance is generally delegated to a web of private organizations and state agencies that conduct inspections and certify that standards have been met. The Joint Commission on Accreditation of Healthcare Organizations (JCAHO) has statutory authority under Medicare to certify hospitals, ambulatory surgical centers, clinical laboratories, home health agencies, and hospices as being in compliance with federal regulations. This authority is based on the concept that organizations meeting JCAHO standards are “deemed” to meet federal standards. The Centers for Medicare and Medicaid Services (CMS) is required to monitor the performance of JCAHO, as well as that of other organizations with deemed status to ensure that equivalency is maintained. JCAHO accreditation is also accepted in the VHA, DOD TRICARE, and IHS programs. The National Committee for Quality Assurance (NCQA) was recently granted deeming authority for certain requirements pertaining to M+C plans and has similar authority for health plans in TRICARE and in some states for Medicaid (NCQA, 2002). Deeming is one way to reduce the burden of repetitive inspections, but there must be adequate oversight to ensure that accrediting entities carry out this responsibility properly (MedPAC, 2000).
Very little work has been done to assess the effect of conditions of participation, as currently structured and enforced, on processes of care or patient outcomes. In addition, the minimal standards are updated infrequently, and little evaluation is done to streamline standards to ensure that they focus on requirements that actually improve patient safety and quality of care (MedPAC, 2000).
External Review
External review is used most extensively by Medicare and Medicaid. External review under Medicare started in the early 1970s,1 and is currently carried out by a network of 37 private-sector QIOs (formerly known as peer review organizations), under contract with CMS. Other govern-
ment programs, state governments, and private-sector groups also contract with QIOs. External review focuses on measurement of care processes and patient outcomes through such means as abstraction of samples of medical records (conducted by QIO staff or the providers); screening of hospital discharge abstracts and claims data to identify such events as nosocomial infections, unscheduled returns to surgery, and deaths; and conduct of a wealth of focused studies in selected clinical areas (discussed further in Chapter 4).
Under Medicare fee-for-service (FFS), QIO review is mandatory for hospitals and other institutions, and there are some QIO activities for ambulatory care in which physicians may voluntarily choose to participate. Starting in 1985, quality review (by QIOs or QIO-like entities) became mandatory for health plans (Consolidated Omnibus Budget Resolution Act [COBRA] 1985); today, the review processes for M+C plans are more extensive than those conducted for FFS Medicare.
During the late 1970s and 1980s, quality review programs were developed and applied within state Medicaid programs. These efforts are difficult to characterize because federal quality requirements and activities differ by type of health care program, which include FFS programs, primary care case management programs, capitated full-risk managed care, Section 1915(b) waiver programs, Section 1115 waiver demonstrations, home and community-based services waiver programs, and programs of all-inclusive care for elderly beneficiaries (Shalala, 2000).
Federal law pertaining to the Medicaid program requires that states adopt procedures to evaluate the utilization of care and services and establish a plan for reviewing the appropriateness and quality of care. The federal government pays states an enhanced federal financial participation rate of 75 percent (as opposed to an average closer to 50 percent) to help cover the costs of reviews conducted by QIOs or QIO-like entities, and most states have pursued this option (Verdier and Dodge, 2002). States, however, have considerable latitude in how they choose to define, implement, and enforce quality review; the level and degree of external review vary widely among the states.
With the growth of Medicare and Medicaid managed care options in the 1990s and in response to concerns about burden and conflicting quality requirements, CMS developed the Quality Improvement System for Managed Care (QISMC), which is based on technical performance measurement (Centers for Medicare and Medicaid Services, 2001a). The system is mandatory for M+C plans and voluntary for Medicaid managed care. QISMC relies to a great extent on measures in the Health Plan Employer Data and Information Set (HEDIS); the standardized quality measurement set of the NCQA; and the Consumer Assessment of Health Plans (CAHPS), a survey instrument and reporting system developed to help
consumers and purchasers choose among health plans (see Chapters 4 and 6). QISMC also includes fairly extensive requirements pertaining to the internal quality assurance and improvement processes of health plans.
In addition to Medicare and Medicaid, other government health care programs rely to varying degrees on external review to safeguard quality. The DOD TRICARE program contracts with the Keystone Peer Review Organization to review the appropriateness of care for about 1,500 medical, surgical, and mental health cases per month; to certify mental health facilities; and to handle patient and provider appeals. VHA has traditional regulatory programs, including an external peer review program. But in these government programs that own and operate their own delivery systems, external review activities are overshadowed by the quality management and improvement programs embedded in the health care delivery function (discussed below) (Institute of Medicine, 2001c).
While external review in all the programs relies on performance measurement of various types to assess the quality of care being delivered, these assessments are necessarily limited by the absence of supportive tools and infrastructure. In the absence of computer-based record keeping on elements of care, quality-of-care studies are confined to manual extractions from paper medical records, resulting in time-consuming analysis of small samples, or to the sparse clinical information available on claims. Moreover, the lack of consistent standards among states and review organizations, the lack of consistent datasets, and the inadequacy of the data in general create substantial obstacles to establishing quality benchmarks or making valid cross-program comparisons of the quality of care received. As discussed in Chapter 5, some progress has been made in addressing these issues in recent years, but the pace of progress is too slow in light of the gravity of the quality and safety shortcomings.
PURCHASING STRATEGIES
There have been very few attempts to expand upon traditional purchasing regulatory mechanisms and engage in what is called “value-based purchasing.” Two different strategies are at the heart of value-based purchasing: (1) disclosure of comparative quality information to encourage consumers and purchasers to choose the highest-quality providers, and (2) selective purchasing or payment incentives to providers and beneficiaries. The purpose of value-based purchasing is to promote market forces that encourage and reward (through higher market share and/or higher payments) providers that achieve higher levels of quality. Both information disclosure and payment incentives, however, are dependent upon the availability of comparative quality data on providers and few such data are available. Moreover, some comparisons of performance require
risk adjustment for differences in patient mix. Such comparisons, too, require richer clinical information than is currently available in most administrative datasets.
In the Medicare program, the federal government has taken some steps consistent with its purchaser role by facilitating disclosure of comparative quality data in the public domain. In 1998, the National Medicare Education Program—an initiative to educate beneficiaries about Medicare health care options—was launched. Under this program, CMS makes available on the World Wide Web limited comparative quality data for M+C plans from CAHPS and HEDIS to help beneficiaries select an M+C plan. For the nearly 87 percent of beneficiaries enrolled in Medicare FFS, the primary decisions to be made are whether to shift from FFS to an M+C plan and what clinician to select. Current information does not permit a comparison to support the former decision, because most performance data are available only for M+C plans. Few if any performance data are available to help beneficiaries choose a doctor or other clinician.
CMS provides beneficiaries with comparative data on kidney dialysis centers, as required by the Balanced Budget Act of 1998. CMS funded the development of clinical practice measures, based on the practice guidelines of the National Kidney Foundation’s Dialysis Outcome Quality Initiative and awarded the development contract to Pro-West (Centers for Medicare and Medicaid Services, 2001c). The measures were developed collaboratively with providers, and dialysis facilities were given the opportunity to review their data prior to public release (American Association of Kidney Patients, 2001). There is a strong commitment to public disclosure, and the CMS website provides a rating of dialysis centers as average, below average, or above average (Centers for Medicare and Medicaid Services, 2002a). CMS recently announced its intent to make similar comparative quality information available on nursing homes. Data from a pilot project conducted in six states (Colorado, Florida, Maryland, Ohio, Rhode Island, and Washington) using the Minimum Data Set measures were recently released (Centers for Medicare and Medicaid Services, 2001b).
At present, CMS has very limited authority to link payment to performance for traditional Medicare, other than through demonstration projects designed to test alternative purchasing approaches (MedPAC, 1999). For example, under the Centers of Excellence demonstration, Medicare contracts selectively with a limited number of hospitals or other organizations to provide comprehensive services for specific procedures (e.g., heart transplants, total joint replacement procedures) under a bundled payment scheme (Centers for Medicare and Medicaid Services, 2002b). Providers compete for these contracts on the basis of quality, as well as other factors, such as geographic accessibility, organizational ca-
pacity, and price. CMS is also conducting disease management demonstration projects that focus on Medicare FFS beneficiaries with congestive heart failure, diabetes, and coronary heart disease. These demonstrations involve innovative care management approaches, expanded coverage for prescription drugs, and the assumption of financial risk by providers (Centers for Medicare and Medicaid Services, 2002c). In addition, CMS has awarded 15 grants for coordinated care demonstration projects focused on Medicare fee-for-service beneficiaries with complex chronic conditions, and these, too, involve care delivery innovations and alternative payment models (Department of Health and Human Services, 2001).
In 1997, DOD initiated a Centers of Excellence program to select, on the basis of a rigorous evaluation process, a limited number of providers to deliver highly specialized services in selected clinical areas (TRICARE, 2002). This program is not yet operational, but a great deal of work has been done to identify the selected clinical areas and the criteria for selection. The selected areas are bone marrow and solid organ transplants, burn care, cardiac care, complex general surgery, cranial and spinal procedures, gynecologic oncology, head and neck oncology, neonatal and prenatal medicine, and total joint replacement. The criteria for selection emphasize the ability to measure various aspects of quality, adjust for severity, measure outcomes, and report externally on clinical processes and outcomes.
In 1998, DOD began reporting some information on quality and access to beneficiaries (Department of Defense, 2001). The Military Treatment Facility Report Card includes information on waiting times for major services; patient satisfaction; and summary scores from JCAHO accreditation surveys applicable to credentialing, provider/staff competence, infection control, and nursing care.
Although beyond the immediate scope of the present study, it should be noted that the federal government has pursued a purchaser approach in carrying out its responsibilities under the Federal Employees Health Benefits Program. For health plans participating in this program, federal employees can access CAHPS and HEDIS data and summary results from NCQA accreditation surveys (Office of Personnel Management, 2002).
CARE DELIVERY
VHA and IHS have comprehensive care delivery programs. For the most part, the federal government owns and operates the health care facilities and employs the workforce necessary to provide comprehensive services to beneficiaries in these programs. The DOD TRICARE program also has a large delivery system component—just over half of health care services are provided through DOD’s treatment facilities (located mainly on military bases), with the remainder being delivered through private-
sector providers. Each of these government programs has pursued a variety of quality measurement and improvement activities as an integral part of its quality management activities (see Table 3-3).
The VHA program stands apart from most health care programs, both public and private, in its commitment to building the strong organizational supports necessary to provide safe and effective care. In the late 1970s, VHA recognized the important role of clinical decision-support systems in improving quality. During the 1980s and 1990s, VHA created the Veterans Health Information Systems and Technology Architecture (VistA), a computerized patient records system that now extends throughout all 1,100 VHA facilities (including 172 hospitals) in the United States (Institute of Medicine, 2001c). Since 1997, VHA has taken steps to make the automated clinical information more accessible and meaningful at the point of care (see Chapter 5).
VistA serves as the foundation for an extensive program of quality measurement and improvement and clinical decision support, including ongoing benchmarking across a wide range of preventive, acute, and chronic care quality measures; automated entry of medication orders; a notification system that alerts clinicians about clinically significant events identified through the use of integrated laboratory, radiology, pharmacy, progress notes, and other data; a clinical reminder system to promote evidence-based practice; and use of bar codes for medication administration and verification of blood type prior to transfusion.
The DOD TRICARE program conducts numerous quality measurement and improvement projects, including ones that use the HEDIS measurement set and beneficiary surveys such as CAHPS. In recent years, DOD has made progress in developing a computerized clinical information system (see Chapter 5). IHS has emphasized improving diabetes care across the various regions using a standardized measurement set.
CURRENT EFFORTS TO STANDARDIZE AND COORDINATE
Important efforts have been made in recent years to coordinate the quality enhancement activities of the various government health care programs. AHRQ has played a central role in many of these efforts. Its contribution to the development of CAHPS and other standardized tools and techniques for quality measurement and improvement is noteworthy. CAHPS is now used by DOD TRICARE, state Medicaid agencies, private-sector purchasers (e.g., Ford Motor Company, Vermont Employer’s Health Care Alliance), Federal Employees Health Benefit Program (FEHBP), and accrediting bodies (e.g., NCQA) (Agency for Healthcare Research and Quality, 2000). To facilitate widespread use and public disclosure of comparative results, AHRQ established a National CAHPS
TABLE 3-3 Internal Quality Management Activities: VHA, DOD TRICARE, and IHS
|
|
VHA |
DOD TRICARE |
IHS |
|
Target entities |
Institutional providers, clinicians, and facilities that provide direct patient care to VHA beneficiaries |
Institutional providers, clinicians, and networks that serve active duty military personnel and other DOD TRICARE beneficiaries |
IHS providers, hospitals, health centers, and clinics |
|
Internal Quality Management Activities |
An internal Performance Measurement Program (QUERI), which evaluates in-house outcome measures as well as HEDIS and JCAHO measures adopted by the VHA external review contractor and other sources. Two patient surveys: the Veteran Satisfaction Survey and the American Consumer Satisfaction Index. The National Surgical Quality Improvement Program, an ongoing effort to evaluate and improve surgical outcomes. The National Center for Patient Safety, used for evaluating “close calls” and adverse events. |
Numerous special quality studies, including ones that use HEDIS measures and various beneficiary satisfaction surveys |
The Performance Evaluation System, which includes a patient database and information system for identifying health problems and needs among the IHS population. The Indian Health Diabetes Care and Outcomes Audit, which involves chart reviews to determine compliance with the IHS standards of care for diabetes. |
|
SOURCES: Code of Federal Regulations, 2001; Department of Defense, 2001; Indian Health Service, 2000, 2001a; Institute of Medicine, 2001b; KePRO, 2001; Kizer, 1995; and Veterans Health Administration, 2001. |
|||
Benchmarking Database in which survey sponsors participate on a voluntary basis. The database currently includes data from over 900 health plans (Agency for Healthcare Research and Quality, 2001c).
AHRQ plays an important role in various interagency collaborative efforts. The agency is mandated by statute to provide coordination of quality improvement programs and activities among the various government health care programs. The primary vehicle for this purpose is the Quality Interagency Coordinating Committee (QuIC).2 The QuIC was established in 1998 to ensure that all federal agencies involved in regulating, purchasing, providing, or studying health care services coordinate their activities with the common goal of improving quality. The membership of the QuIC includes representation from within the Department of Health and Human Services (DHHS) (i.e., CMS, IHS and AHRQ), DOD, VHA, and numerous other federal agencies (see Chapter 4). The QuIC has work groups on issues such as providing consumer information, measuring quality, improving clinical quality, developing the work force, and improving information systems (Agency for Healthcare Research and Quality, 2001a).
Lastly, AHRQ leads an interagency initiative started in 2000 to address patient safety concerns (Agency for Healthcare Research and Quality, 2001b). The Patient Safety Task Force includes representatives of AHRQ, CMS, the Centers for Disease Control and Prevention, and the Food and Drug Administration. The goals of the task force are to (1) coordinate the collection and analysis of safety-related data across various government programs; (2) exchange information on patient safety reporting and practices with other public-and private-sector initiatives; (3) disseminate analyses to health care providers and others; and (4) carry out research, programs, and projects that will improve patient safety.
LEVERAGING THE GOVERNMENT ROLES
There are, of course, important differences across the government health care programs in the roles played by the federal government and the degree of emphasis placed on any individual role in influencing qual-
ity. This is the case in part because the programs have very different historical underpinnings and statutory enabling authorities. In general, the programs vary in terms of the degree of responsibility the federal government assumes for quality; the political and other barriers to addressing quality concerns; and the incentives, tools, and data available to measure and improve quality.
On one end of the spectrum is Medicaid, in which the federal government has a very small role in quality enhancement. Federal regulatory requirements are minimal, and the states, which administer the program, have a great deal of latitude in carrying out quality oversight responsibilities. For the most part, both federal and state governments have pursued regulatory approaches to quality enhancement for the program.
On the other end of the spectrum are the federal health care delivery programs, such as the VHA, DOD TRICARE, and IHS programs. In these programs, the federal government assumes nearly total responsibility for quality. In these programs, there is less distinction, and certainly less conflict, among the various types of federal roles. In the case of VHA, and more recently DOD, the federal government has used its formidable influence and resources to build an information infrastructure capable of supporting a wide range of quality measurement, clinical decision-support, and care delivery applications. The accomplishments of these programs represent best practices that the federal government should actively share with other public- and private-sector health care programs.
Somewhere in the middle of this spectrum is Medicare. Established as a traditional indemnity insurance program, Medicare has emphasized the use of regulatory mechanisms to protect beneficiaries from poor-quality providers. The transition to producing comparative quality data has been a gradual one, and attempts at public disclosure of these data have been sporadic and infrequent.
There is little doubt that the federal government could play its various roles more effectively to both promote improvements in quality within each of the government health care programs and drive improvement in the health care sector overall. The federal government should exercise appropriate influence through each of its roles to the maximum extent possible to promote quality improvements. Specifically, the committee encourages the leadership of the various government health care programs to ensure that their quality enhancement processes adhere to the following guiding principles:
-
Government health care programs should establish consistent quality expectations and requirements and apply them fairly and equitably to all financing and delivery options within a program.
-
Government health care programs should promote and encourage provid-
-
ers to strive for excellence by providing financial and other rewards and public recognition to providers who achieve superior levels of quality.
-
Government health care programs should actively collaborate with each other and private-sector quality enhancement organizations with regard to all aspects of quality enhancement—including use of standardized measures and sharing of data—where doing so will likely result in greater gains in quality or reduced provider burden.
-
Government health care programs should encourage and enable active consumer participation in efforts to enhance quality through such means as the following:
-
Raising consumer awareness of the magnitude of quality and safety shortcomings and the means of addressing these problems
-
Seeking consumer input into the design and evaluation of quality enhancement processes
-
Including patient assessments of quality and service in the portfolio of performance measures
-
Providing patients with health information necessary to evaluate treatment options and participate in care management
-
Providing consumers with comparative performance data on providers and health plans
-
-
Government health care programs, in collaboration with the Agency for Healthcare Research and Quality (AHRQ), should pursue a rich agenda of applied research and demonstrations focusing on tools, techniques, and approaches to quality enhancement.
There are many variations in quality enhancement requirements across the government health care programs, some rooted in differences among the needs of the populations served, but most stemming from the fact that the programs have developed their quality enhancement processes independently. In the absence of compelling reasons for differences in quality enhancement requirements, the federal government should strive to provide the same minimal level of quality protection to all populations served. Efforts should also be made to streamline the implementation of quality enhancement processes so as to minimize the burden on providers, especially those in the private sector, who typically have relationships with multiple third-party payers.
The federal government has far less experience in pursuing purchasing strategies to enhance quality than in establishing regulatory requirements. Given the seriousness of current safety and quality shortcomings in the health care system, it is imperative that the government be given the flexibility and resources necessary to explore value-based purchasing. Regulations alone cannot solve the problem. Purchasing initiatives should be carefully evaluated to determine whether they are effective. Purchas-
ing strategies should be aimed at creating an environment that will encourage and reward exemplary performance. Purchasing strategies are less rigid than regulatory requirements, and their expected effects are more difficult to predict and to quantify. Yet such strategies have the potential to motivate the majority of health care providers to improve quality, thus encouraging widespread change in the health care sector that would complement regulatory efforts.
One purchasing strategy—public disclosure of comparative quality data on providers—also has the potential to engage numerous other stakeholders in the quality debate. Government health care programs provide little if any information to the public on variations in quality and outcomes across hospitals, provider groups, and treatment programs; yet, there is a strong evidence base to substantiate that such variations are large (Schuster et al., 1998). Not all beneficiaries are capable of or interested in incorporating information on quality into their decisions when selecting providers or treatment programs, but not all need do so to provide incentives to improve performance. Other stakeholders, including group purchasers, professional leaders, governing boards of hospitals and other institutions, peer review organizations, and state and federal regulators would find comparative quality data useful in carrying out their responsibilities.
Finally, the federal government must provide leadership for the development of the infrastructure needed to support quality oversight and improvement. All of the federal health care programs have made a stronger commitment in recent years to technical quality measurement. Nearly all of the programs have focused a good deal of attention on quality measures pertaining to leading chronic conditions in response to the needs of the populations they serve. The increased emphasis on clinical quality measurement in all of the federal health care programs is a positive development, highlighting the potential for the federal government to encourage greater coordination and standardization of performance measurement efforts across government programs and, indeed, throughout the health care sector overall. In the absence of strong federal leadership and a clear strategy for coordinating the efforts of various government health care programs, the likely outcome will be a duplicative and burdensome patchwork of quality measurement data of limited utility to end users (as discussed further in Chapter 4).
In general, there is a lack of recognition of the dependence of effective quality oversight and improvement efforts on computerized clinical data (as discussed further in Chapter 5). Despite numerous reports in recent years calling attention to the importance of computerized clinical data (Institute of Medicine, 2001a; National Committee for Quality Assurance,
2002), the development of an information technology infrastructure to support quality enhancement processes has been very slow.
The challenge for federal quality enhancement processes is to harness their potential to drive and facilitate quality improvement throughout the health care delivery system. Without coordination, standardization, dissemination of information, and incentives, fulfillment of that potential will be impossible to attain.
REFERENCES
Agency for Healthcare Research and Quality. 2000. “CAHPS-User Story: AHRQ Publication No 00-P023.” Online. Available at http://www.ahrq.gov/qual/cahps/cahpuser.htm [accessed May 6, 2002].
———. 2001a. “Quality Interagency Coordination Task Force (QuIC) Fact Sheet, AHRQ publication No. 00-P027.” Online. Available at http://www.ahcpr.gov/qual/quicfact.htm [accessed June 18, 2001a].
———. 2001b. “Patient Safety Task Force Fact Sheet.” Online. Available at http://www.ahrq.gov/qual/taskforce/psfactst.htm [accessed May 20, 2001b].
———. 2001c. “Annual Report of the National CAHPS ® Benchmarking Database 2000:What Consumers Say About the Quality of Their Health Plans and Medical Care.” Online. Available at http://ncbd.cahps.org/pdf/NCBD2000AnRpt.pdf [accessed May 6, 2002c].
American Association of Kidney Patients. 2001. “What’s new at HCFA.” Online [accessed Aug. 14, 2002].
American Hospital Association. 2002. Patients or Paperwork? The Regulatory Burden Facing America’s Hospitals. Washington DC: PricewaterhouseCoopers for American Hospital Association.
Anderson, G. F., G. D. Greenberg, and B. O. Wynn. 2001. Graduate Medical Education: The Policy Debate. Annu Rev Public Health 22:35-47.
Arnett, G-M. 1999. Emporwering Health Care Consumers Through Tax Reform. An Arbor MI: The University of Michigan Press.
Association of American Medical Colleges. 2001. AAMC Data Book: Statistical Information Related to Medical Schools and Teaching Hospitals. 102.
Centers for Medicare and Medicaid Services. 2001a. Medicaid Program, Medicaid Managed Care. Final Rule. Federal Register.
———. 2001b. “Nursing Home Compare - Home.” Online. Available at http://www.medicare.gov/NHCompare/home.asp [accessed May 6, 2002b].
———. 2001c. “Quality of Care ~ National Projects, ESRD Clinical Performance Measures Project (2000 Annual Report).” Online. Available at hcfa.gov/quality/3m8.htm [accessed Jan. 9, 2002c].
———. 2002a. “Medicare.gov - Dialysis Facility Compare Home.” Online. Available at http://www.medicare.gov/Dialysis/Home.asp [accessed Aug. 14, 2002a].
———. 2002b. “Medicare Partnerships for Quality Services Demonstration.” Online. Available at http://cms.hhs.gov/healthplans/research/mpqsdem.asp [accessed Oct. 14, 2002b].
———. 2002c. Medicare Program; Solicitation for proposals for the demonstration project for disease management for severely chronically ill Medicare beneficiaries with congestive heart failure, diabetes, and coronary heart disease: Notice for Solicitation of Proposals. Fed Regist 67 (36):8267-70.
Code of Federal Regulations. 2001. “32CFR199.15: Quality and Utilization Review Peer Review Organization Program.” Online. Available at http://www.tricare.osd.mil/CFR/C15.PDF [accessed May 9, 2002].
Cooper, R., T. Henderson, and C. Dietrich. 1998. Roles of Non-physician Clinicians as Autonomous Providers of Patient Care. JAMA 280 (9):795-802.
Department of Defense. 1995. DoD Directive Number 6025.13 (July 20): Clinical Quality Management Program (CQMP) in the Military Health Services System (MHSS).
———. 2001. “Healthcare Quality Initiatives Review Panel (HQIRP) Report.” Online. Available at http://www.tricare.osd.mil/downloads/FinalReport123.pdf [accessed June 26, 2002].
Department of Health and Human Services. 2001. Medicare Fact Sheet: Providing Coordinated Care to Improve Quality of Care for Chronically Ill Medicare Beneficiaries. Washington DC: U.S. Department of Health and Human Services.
Finocchio, L., C. Dower, N. Blick, and The Taskforce on Health Care Workforce Regulation. 1998. Strengthening Consumer Protection: Priorities for Health Care Workforce Regulation. San Francisco CA: Pew Health Professions Commission.
Health Care Financing Administration. 1998. Medicaid Program; Medicaid Managed Care; Proposed Rule August 20, 2001. 42 CFR §400, 430, et al.
———. 2000. “Quality Improvement System for Managed Care (QISMC) for Organizations Contracting with Medicare or Medicaid. Introduction: Year 2000 Standards and Guidelines.” Online. Available at www.hcfa.gov/quality/3a1.htm [accessed Nov. 9, 2001].
———. 2002. “Medicare Managed Care Manual.” Online. Available at www.hcfa.gov/pubforms/86_mmc/mc86toc.htm [accessed Mar. 24, 2002].
Indian Health Service. 2000. “Welcome to the IHPES web site.” Online. Available at http://www.ihs.gov/NonMedicalPrograms/IHPES/index.cfm?module=content&option=home [accessed Feb. 15, 2002].
———. 2001a. “Welcome to the Indian Health Service National Diabetes Program.” Online. Available at http://www.ihs.gov/medicalprograms/diabetes/ihsndpnewpage.asp [accessed May 9, 2002a].
———. 2001b. “Accreditation Achievement.” Online. Available at http://info.ihs.gov/QualityAccount/Quality4.pdf [accessed Mar. 20, 2002b].
Institute of Medicine. 2001a. Crossing the Quality Chasm: A New Health System for the 21st Century. Washington DC: National Academy Press.
———. 2001b. An Overview of Major Federal Health Care Quality Programs: A Technical Report for the Institute of Medicine Committee on Enhancing Federal Healthcare Quality Programs. E. K. Swift (ed.). Washington DC: Institute of Medicine.
———. 2001c. An Overview of Major Federal Health Care Quality Programs: Appendix B. Washington DC: IOM.
KePRO. 2001. “KePRO: TRICARE.” Online. Available at http://www.kepro.org/tricare.htm [accessed Nov. 28, 2001].
Kizer, K. W. 1995. Vision for Change: A Plan to Restructure the Veterans Health Administration. Washington DC: U.S. Department of Veterans Affairs.
MedPAC. 1999. Chapter 2: “Influencing Quality in Traditional Medicare”. Report to Congress: Selected Medicare Issues. Washington DC: MedPAC.
———. 2000. Report to the Congress: Selected Medicare Issues. Washington DC: MedPAC.
———. 2001. Report to the Congress: Medicare Payment for Nursing and Allied Health Education. Washington DC: MedPAC.
———. 2002. “Report to Congress: Applying Quality Improvement Standards in Medicare.” Online. Available at http://www.medpac.gov/publications/congressional_reports/jan2002_QualityImprovement.pdf [accessed Oct. 2, 2002].
National Committee for Quality Assurance. 2002. HEDIS 2002, Volume 3: Specifications for Survey Measures Information. Washington DC: NCQA.
NCQA. 2002. “Medicare+Choice (M+C) Deeming Program.” Online. Available at http://www.ncqa.org/programs/m+c/m-cmain.htm [accessed May 16, 2002].
Office of Personnel Management. 2002. “Federal Employees Health Benefits Program Home Page.” Online. Available at http://www.opm.gov/insure/health/ [accessed Mar. 26, 2002].
Pittman, R., Principal Pharmacy Consultant (Indian Health Service). 26 March 2002. IHS Conditions of Participation, Roles of the Federal and State Governments, and Quality Improvement. Personal communication to Jill Eden.
Rosenfeld, B., T. Dorman, and M. Breslow, et al. 2000. Intensive Care Unit Telemedicine: Alternative Paradigm for Providing Continuous Intensive Care. Crit Care Med 28 (12):3925-31.
Sage, W., and L. Aiken. 1997. Chapter 4: Regulating Interdisciplinary Practice. In Regulation of Healthcare Professions. Chicago, IL: Health Administration Press.
Schuster, M. A., E. A. McGlynn, and R. H. Brook. 1998. How good is the quality of health care in the United States? Milbank Q 76 (4):517-63.
Shalala, D. 2000. Report to Congress: Safeguards for Individuals with Special Health Care Needs Enrolled in Medicaid Managed Care. Washington DC: Department of Health and Human Services.
TRICARE. 2002. “DoD Centers of Excellence.” Online. Available at http://www.tricare.osd.mil/coe/default.cfm [accessed May 6, 2002].
Verdier, J., and R. Dodge. 2002. Other Data Sources and Uses, Working Paper in the Informed Purchasing Series. Lawrenceville NJ: Center for Health Care Strategies.
Veterans Administration. 2001. “VHA Handbook 1100.19: Credentialing and Privileging.” Online. Available at http://www.va.gov/publ/direc/health/handbook/1100-19HK(3-6-01).pdf [accessed May 8, 2002].
Veterans Health Administration. 2001. Major quality improvement and evaluation programs in the Department of Veterans Affairs: a summary prepared for the Institute of Medicine. Washington DC: U.S. Department of Veterans Affairs.























