3
Epidemiology of Malaria
A familiarity with the technical aspects of malaria is necessary for decisions to be made about how to devise a locally appropriate malaria control strategy. Such familiarity can aid in determining whether malaria might pose an important public health risk in a given situation, how large a problem it might be, which drugs would be expected to be effective and which probably would not, and which groups within a displaced population might be at increased risk of malaria and therefore need special attention.
The following section presents information on areas at risk of malaria transmission, the mechanics of how malaria is transmitted within a population, the range of clinical manifestations of malaria, antimalarial drug resistance, and the mosquito vector. The information presented is by no means exhaustive but should aid in basic decision making or in recognizing when expert assistance is needed.
AREAS AT RISK
Malaria occurs primarily in tropical and some subtropical regions of Africa, Central and South America, Asia, and Oceania (Figure 2-1). In areas where malaria occurs, there is tremendous variation in the intensity of transmission and risk of infection. For example, over 90 percent of clinical malaria infections and deaths occur in sub-Saharan Africa (World Health Organization, 1996a). However, even there the risk varies widely. Highland (>1,500 m) and arid areas (<1,000 mm rainfall/year) typically have
less malaria, although these areas are prone to epidemic malaria if climactic conditions become favorable to mosquito development (World Health Organization, 1996a). Although urban areas have typically been at lower risk, explosive unplanned population growth has been a major factor in making urban or peri-urban transmission an increasing problem (Knudsen and Sloof, 1992).
Human malaria is caused by one or more of four parasites: Plasmodium falciparum, P. vivax, P. ovale, and P. malariae. Distribution of these parasites varies geographically, and not all species of malaria are transmitted in all malarious areas. P. falciparum, the species most commonly associated with fatal malaria, is transmitted at some level in nearly all areas where malaria occurs. It accounts for over 90 percent of all malaria infections in sub-Saharan Africa, for nearly 100 percent of infections in Haiti, and causes two-thirds or more of the malaria cases in Southeast Asia. P. vivax is a relatively uncommon infection in sub-Saharan Africa. Duffy antigens, which are required by the parasite to invade red blood cells, are lacking in many ethnic groups, especially in West Africa. Vivax malaria, however, is the predominant species in Central America, most of malarious South America, and the Indian subcontinent (Miller et al., 1977).
MECHANISMS OF INFECTION AND TRANSMISSION
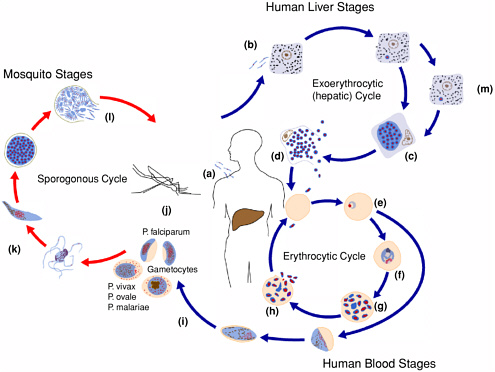
Malaria is typically transmitted by the bite of an infective female Anopheles mosquito; transmission can also occur transplacentally, as a result of blood transfusion, or by needle sharing. Infective mosquitoes inject sporozoites into the bloodstream during feeding (see Figure 3-1). These sporozoites infect liver cells (b) where they undergo asexual reproduction (exoerythrocytic schizogony), producing schizonts (c). In 6 to 14 days (sometimes longer), the schizonts rupture, releasing merozoites into the bloodstream (d). Merozoites invade red blood cells and undergo a second phase of asexual reproduction (erythrocytic schizogony), developing into rings (e), trophozoites (f), and finally blood stage schizonts (g). The schizonts rupture, destroying the red blood cell and releasing more merozoites into the bloodstream, starting another cycle of asexual development and multiplication (h). This erythocytic cycle will continue until the infected individual is successfully treated, mounts an immune response that clears the infection, or dies. During this cycle, sexual forms called gametocytes are produced (i) and can be ingested by a mosquito during a
blood meal (j). Sexual reproduction occurs in the mosquito (k). Sporozoites are formed (l), which migrate to the salivary glands, making the mosquito infective to humans.
The timing of events in the life cycle of malaria parasites and the number of merozoites produced during schizogony vary by species. Additionally, two species of malaria, P. vivax and P. ovale, have a form, “hypnozoites” (m), that can persist in the liver for months to years, causing periodic relapses of peripheral parasitemia and illness (see Table 3-1).
MALARIA VECTORS AND VECTOR BEHAVIOR
Human malaria is transmitted by the bite of female mosquitoes belonging to the genus Anopheles. Of the 400 or so species of Anopheles in the world, approximately 60 are important vectors of malaria. However, a particular species of Anopheles may be an important vector in one area of the world and of little or no consequence in another.
Different species of Anopheles can behave differently. Mosquito behavior can differ in terms of breeding or larval habitat (e.g., fresh vs. brackish water; flowing streams, still pools, or man-made habitats; shaded or sunny sites), feeding preferences (e.g., time of day when peak biting occurs, preferences for people over animals, feeding indoors or outside), and resting habits (resting indoors after feeding or leaving the house before resting). These differences in mosquito behavior can affect both the epidemiology of malaria and the choice of malaria control strategy used. For example, An. dirus is an important vector in Southeast Asia and is primarily a forest dweller. People at greatest risk are, therefore, those who enter the forest for whatever reason, while those who stay closer to home (such as small children) are at less risk. This also means that malaria control strategies aimed at preventing mosquito biting in the home (such as residual spraying or insecticide-treated bed nets) would be of little value in preventing exposure and infection. An. gambiae, the most important vector in much of sub-Saharan Africa, breed in small temporary pools of water (even as small and temporary as cattle hoofprints). Therefore, vector control strategies aimed at reducing or eliminating breeding sites will likely have little impact. For these reasons, expert advice relevant to the primary malaria vectors in a given area is essential for making sound decisions regarding control options.
TABLE 3-1 Characteristics of the Four Species of Human Malaria
|
|
P. falciparum |
P. vivax |
P. ovale |
P. malariae |
|
Exoerythrocytic cycle |
6-7 Days |
6-8 Days |
9 Days |
14-16 Days |
|
Prepatent period |
9-10 Days |
11-13 Days |
10-14 Days |
15-16 Days |
|
Incubation period (mean) |
9-14 (12) Days |
12-17 (15) Days to 6-12 months |
16-18 (17) Days or longer |
18-40 (28) Days or longer |
|
Severity of primary attack |
Severe |
Mild to severe |
Severe |
Severe |
|
Duration of primary attacka |
16-36 Hours or longer |
8-12 Hours |
8-12 Hours |
8-10 Hours |
|
Duration of untreated infectiona |
1-2 Years |
1.5-5 Years |
1.5-5 Years |
3-50 Years |
|
Relapse |
No |
Yes |
Yes |
No |
|
Central nervous system complicationsa |
Frequent |
Infrequent |
Infrequent |
Infrequent |
|
Anemiaa |
Frequent |
Common |
Infrequent |
Infrequent |
|
Renal insufficiencya |
Common |
Infrequent |
Infrequent |
Infrequent |
|
Effects on pregnancya |
Frequent |
Infrequent |
Unknown |
Unknown |
|
Hypoglycemia |
Frequent |
Unknown |
Unknown |
Unknown |
|
a Influenced by immunity. Documentation of complications for species other than P. falciparum is limited. SOURCE: Adapted from Bruce-Chwatt (1985). |
||||
VECTOR LIFE CYCLE
There are four stages in the life cycle of the mosquito: egg, larva, pupa, and adult. Eggs are deposited singly on water in suitable breeding sites, where the developing embryo hatches as a larva after 2 or more days. During the aquatic period of development, the larva sheds its skin four times. The fourth larval molt gives rise to a pupa. At this stage the mosquito undergoes a complete metamorphosis, emerging as an adult. When parasites are ingested during a blood meal, they undergo further development in the mosquito’s stomach, and during the next 10 to 20 days the parasite passes through a number of stages, eventually multiplying and penetrating all parts of the mosquito body. Parasites that end up in the salivary glands can be transmitted to humans when the mosquito takes another blood meal.
The length of each developmental stage depends on temperature and humidity. The life span of adults under natural conditions is difficult to determine but averages 10 to 14 days or longer (Service, 1993). The time for an egg to develop to an adult in the tropics can be as short as 5 to 7 days. Development of malaria parasites in the mosquito host is also temperature dependent; as ambient temperatures decrease, the time needed for parasites to develop increases. Malaria transmission stops when the time needed for development of infective sporozoites exceeds the life span of the mosquito (Gilles, 1993).
MALARIA ILLNESS
The normal incubation period from infective mosquito bite to onset of clinical symptoms is 9 to 30 days or longer, depending on such parameters as species of parasite, immune status, infecting dose, and use of antimalarial drugs. The clinical symptoms associated with malaria parasites are produced by increases in cytokines (particularly tumor necrosis factor) in response to merozoites, pyrogens, and cellular debris released when red blood cells rupture at the end of schizogony (Kwiatkowski, 1990). Onset of illness is associated with the initial rupture of erythrocytic schizonts; exoerythrocytic forms (sporozoites, exoerythrocytic schizonts, and hypnozoites) and gametocytes do not cause clinical symptoms.
Typical symptoms among nonimmune individuals include fever, chills, myalgias and arthralgias, headache, diarrhea, and other nonspecific signs. Other findings may include splenomegaly, anemia, thrombocytopenia,
pulmonary or renal dysfunction, changes in mental status, and coma. Most cases of malaria-related severe illness and death are associated with P. falciparum infection. This is due, in part, to the parasite’s ability to infect both mature and immature red blood cells, its rapid rate of asexual reproduction, and a broad range of poorly understood pathological processes associated with its ability to sequester in postcapillary venules, especially in the central nervous system. Cyclical fevers seen in nonimmune individuals with synchronous infections (occurring when a majority of schizonts rupture at the same time) are frequently absent among individuals with immunity.
In general, immunity to malaria is acquired after repeated exposure to the malaria parasite; those individuals who survive their initial infections develop some degree of immunity. In highly endemic areas, most clinical malaria and malaria-associated mortality occurs in children less than 5 years old, whose immunity has not yet fully developed. Although malaria prevalence in endemic areas decreases with increasing age, suggesting an acquired ability to clear malaria infections, immunity to malaria typically also involves the development of a tolerance for the presence of malaria parasites in the blood with a minimum of symptoms and relative protection from severe illness and death.
EPIDEMIOLOGY OF CLINICAL MALARIA
Malaria transmission intensity, levels of acquired immunity in a population, and manifestations of malaria illness are intimately linked (see Table 3-2; Snow et al., 1994; Slutsker et al., 1994). Understanding this relationship should help in estimating the likely impact of malaria in a given population. An important additional consideration is understanding the implications of differences between the environment from which a displaced population comes and the environment in which that population settles, even if only temporarily.
The overall level of immunity to malaria is highest in areas where malaria transmission is the most intense. The first exposure to malaria occurs very early in childhood, and with repeated exposures the likelihood of severe illness or death lessens. In Africa, where the majority of malaria-associated deaths occur, the highest mortality rates occur in children less than 5 years old. It has been estimated that in the Gambia, malaria accounts for 25 percent of all deaths among children less than 5 years old. Malaria-associated mortality decreases rapidly with increasing age (Greenwood et
TABLE 3-2 Malaria Transmission Characteristics, Immunity, and Clinical Features
|
Areas Where P. falciparum Malaria Is Predominant or Represents a Significant Proportion of Infecting Malaria Species |
Areas Where Nonfalciparum Malaria Is the Predominant Infecting Species |
||
|
Areas of Intense Perennial Malaria Transmission and/or Populations with High Levels of Immunity Originating in Those Areas |
Areas of Low-to-Moderate Malaria Transmission |
Areas of Very Low, Highly Seasonal, or Epidemic Malaria Transmission and/or Low Levels of Immunity in the Population |
Examples: Central America, some parts of the Indian subcontinent, parts of Central Asia, parts of eastern Europe. Typical epidemiology of clinical illness: Nonfalciparum malaria can be a significant cause of morbidity but is rarely associated with severe illness or death. Spontaneous splenic rupture can occur in chronic P. vivax infections, but cerebral malaria has only rarely been attributed to this parasite. Infections caused by P. malariae have been associated with nephrotic syndrome. In areas where both falciparum and nonfalciparum malaria occur, species-specific treatment is advisable. Because drugs used for the treatment of acute infections do not typically affect the hypnozoites of P. ovale and P. vivax, these infections can relapse months to years later. |
|
Example: Most of tropical equatorial Africa. Typical epidemiology of clinical illness: Bulk of clinical disease occurs in young (<5 years old) children. While adults and older children are often parasitemic, they are more likely to be asymptomatically infected or to have only mild symptoms. Severe anemia tends to be more common than cerebral malaria in young children. |
Examples: Parts of southern Africa, coastal eastern Africa, Indian subcontinent, Southeast Asia. Typical epidemiology of clinical illness: Bulk of disease occurs in broader range of ages. Cerebral malaria becomes more common. |
Examples: Sahelian and highland areas of Africa, Central America. Typical epidemiology of clinical illness: Disease can occur in all age groups and is more likely to progress to severe disease if not promptly treated. In some areas of very low transmission (e.g., Haiti), mortality rates associated with malaria can be paradoxically low, probably due to high incidence of clinical symptoms, rapid treatment-seeking behavior of the population, and the high efficacy and ready availability of CQ. |
|
al., 1987; Campbell, 1991). In areas with very high levels of transmission, severe malaria tends to manifest itself more frequently as anemia than as cerebral disease. As transmission intensity decreases and population immunity is lessened, illness is seen more frequently in all age groups and the incidence of cerebral disease increases relative to severe anemia.
Displaced populations coming from areas of little or no malaria transmission to areas with intense transmission are therefore at greatest risk of severe illness and death due to a lack of acquired immunity. Until they themselves acquire a protective level of immunity to malaria, these populations will probably require a much more intensive intervention effort to achieve and maintain low rates of morbidity and mortality.
Malaria also has important effects in the way of chronic or repeated infections, including anemia (especially among individuals with underlying malnutrition), and poor pregnancy outcomes. Anemia, itself an important cause of mortality associated with malaria, is also a common reason for blood transfusion.
MALARIA DURING PREGNANCY
Among nonimmune women, acute malaria during pregnancy carries a high risk of maternal and fetal death if not treated promptly and adequately. Among semiimmune pregnant women, however, malaria parasites preferentially sequester in the placenta, with minimal increase in overt clinical disease. Malaria during pregnancy is a cause of both maternal anemia and low birth weight, accounting for as much as 35 percent of preventable low birth weight in malarious areas (Steketee et al., 1996a). Low birth weight, in turn, is a well-recognized risk factor for infant mortality (McCormick, 1985). In most populations living in areas of high malaria transmission, this is an issue primarily affecting women in their first and second pregnancies.
MALARIA AND HIV/AIDS
Malaria and HIV infection can occur at high frequencies in the same population, especially in sub-Saharan Africa, raising concerns that interactions between these two diseases could greatly complicate control of both (Corbett et al., 2002). Malaria and HIV have indeed been shown to interact in important ways. Peripheral malaria infection has been shown to occur more frequently and parasite densities have been shown to be higher
among HIV-positive pregnant women compared with HIV-negative pregnant women (Steketee et al., 1996b). HIV-infected women are at increased risk of placental malaria, even during later pregnancies when placental sequestration is less of a problem among HIV-negative women (Steketee et al., 1996b). Similarly, HIV-infected, nonpregnant individuals are at increased risk of both malaria infection and illness (Whitworth et al., 2000). There is also evidence that acute malaria infection can increase viral load among HIV-infected individuals, an increase that is reversed with effective malaria therapy (Hoffman et al., 1999). Finally, unscreened blood transfusions for malaria-associated anemia remain an important source of HIV transmission in malarious areas (Corbett, et al., 2002).
ANTIMALARIAL DRUG RESISTANCE
Simply defined, antimalarial drug resistance occurs when malaria parasites gain the ability to survive what should be an effective dose of antimalarial drugs. Resistance occurs because of naturally occurring mutations that affect the susceptibility of the parasite to a given drug. For some drugs, resistance can occur after a single-point mutation (as for the drug atovaquone); with others a number of mutations may be required. Factors that facilitate intensification of drug resistance include poor adherence to recommended treatment regimens, inadequate dosing, use of poor-quality drugs, presumptive treatment, and use of drugs that have a long half-life. Because of rapidly developing and spreading resistance to antimalarials and the relatively slow process of developing new antimalarials, the number of useful drugs is dwindling (Bloland and Ettling, 1999; Winstanley, 2000). The most commonly available antimalarial drugs are described in Appendix A and Table 3-3, although not all are practical or appropriate for use in any given situation.
Chloroquine-resistant P. falciparum (CRPF) was first recognized almost simultaneously in Thailand and South America in the late 1950s. It was first identified on the east coast of Africa in 1978. In the past 20 to 25 years, CRPF has spread and intensified to the point that only Central America northwest of the Panama Canal, the island of Hispaniola (Haiti and the Dominican Republic), and limited regions of the Middle East are free of chloroquine (CQ) resistance. All other endemic areas have malaria that is, to varying extents, resistant to CQ. In some regions, CQ resistance has intensified to the point where the drug no longer has an optimal effect
TABLE 3-3 Antimalarial Drugs for Uncomplicated Malaria
|
|
Dosing (all per os)a |
|
||||
|
Drug Name |
Use |
Adult |
Pediatric |
Contra-indications |
Cost (US$)b |
Comments |
|
Combination Therapy |
||||||
|
MQ + artesunate |
Treatment of nonsevere falciparum infections thought to be CQ and SP-resistant. |
15 mg/kg MQ base on day 2 of treatment, followed by 10 mg/kg MQ base on day 3. Usual adult dose is 750 mg on day 2 followed by 500 mg on day 3. |
15 mg/kg MQ on day 2 of treatment followed by 10 mg/kg MQ on day 3. |
See under MQ monotherapy. |
3.90 |
Safety of artemisinins and MQ during first trimester of pregnancy not established. Vomiting after MQ can be reduced by administering it on the second and third days after an initial dose of artesunate. Artemisinin (20 mg/kg initially followed by 10 mg / kg once daily for 2 more days) can be substituted can besubstituted for artesunate. |
|
|
4 mg/kg artesunate daily for 3 days. |
4 mg/kg artesunate daily for 3 days. |
|
|||
|
SP + artesunate |
Treatment of nonsevere falicparum infections thought to be CQ resistant. |
As for SP monotherapy (see below). |
As for SP monotherapy (see below). |
Known SP allergy |
1.12 |
This combination has not been evaluated as extensively as MQ + artesunate. Safety of artemisinin during first trimester of pregnancy not established. Artemisinin (as described above) can be substituted for artesunate. |
|
|
4 mg/kg artesunate daily for 3 days. |
4 mg/kg artesunate daily for 3 days. |
|
|||
|
CQ or amodiaquine + artesunate |
Treatment of CQ-resistant malaria. |
Treatment: 25 mg base/kg divided over 3 days. |
Treatment: 25 mg base/kg divided over 3 days. |
|
Combination of amodiaquine + artesunate generally more useful due to widespread, typically high-level CQ resistance. In some areas, |
|
|
|
Dosing (all per os)a |
|
||||
|
Drug Name |
Use |
Adult |
Pediatric |
Contra-indications |
Cost (US$)b |
Comments |
|
|
Average adult dose: 2.5 gm (salt) divided over 3 days. |
|
neither would be expected to be particularly effective. See note below on possible toxicity associated with amodiaquine. |
|||
|
|
4 mg/kg artesunate daily for 3 days. |
4 mg/kg artesunate daily for 3 days. |
|
|||
|
Lumefantrine/ artemether also known as: co-artemether Trade names: Coartem, Riamet |
Treatment of multidrug-resistant malaria. |
Adult: 4 tablets per dose at 0, 8, 24, 36, 48, and 60 hours. |
Pediatric: 10-14 kg: 1 tablet/dose 15-24 kg: 2 tablets/dose 25-35 kg: 3 tablets/dose >35 kg: as for adult given at 0, 8, 24, 36, 48, and 60 hours. |
Hyper-sensitivity to component drugs. |
2.40 (WHO price only) |
Commercially available fixed-dose combination of 20 mg artemether and 120 mg lumefantrine. Not recommended for <10 kg or pregnant women Available in 2 packaging schemes: 24 tablet/6 doses (Riamet) and 16 tablets/ |
|
|
4 doses. (Coartem)— 4 dose regimen not as effective especially for nonimmunes. |
|||||
|
|
Also available from WHO in specially designed blister packs. |
|||||
|
CQ (or amodiaquine) + sulfadoxine/ pyrimethamine |
Treatment of CQ-resistant malaria. |
Use routine doses for both CQ/ amodiaquine and SP. |
Use routine doses for both CQ/ amodiaquine and SP. |
Same as for CQ/ amodiaquine and SP monotherapy. |
|
Primarily useful only in areas where CQ resistance is low to moderate and SP resistance is low. |
|
|
See notes of caution regarding use of amodiaquine. |
|||||
|
Single-Agent Therapy |
||||||
|
CQ Trade names: Nivaquine, Malaraquine, Aralen, many others |
Treatment of nonfalciparum infections. Treatment of P. falciparum infections in areas where CQ remains effective. |
Treatment: 25 mg base/kg divided over 3 days. Average adult: 1.5 gm base divided over |
Treatment: 25 mg base/kg divided over 3 days. |
|
0.08 |
Widespread resistance in P. falciparum in most regions. Resistance in P. vivax occurs in some areas. Can cause pruritus in dark-skinned patients, reducing compliance. |
|
|
Dosing (all per os)a |
|
||||
|
Drug Name |
Use |
Adult |
Pediatric |
Contra-indications |
Cost (US$)b |
Comments |
|
|
Chemoprophylaxis in areas where CQ remains effective. |
3 days. |
|
Preparations differ in amount of base (100 or 150 mg tablets, 50 mg syrup). |
||
|
|
Prophylaxis: 5 mg/kg base per week. |
Prophylaxis: 5 mg base/kg once per week. |
|
|||
|
Amodiaquine Trade names: Camoquine, others |
Treatment of nonsevere falicparum infections thought to be CQ resistant. |
Treatment: 25 mg base/kg divided over 3 days. Single dose of 25 mg (sulfa)/kg |
Treatment: 25 mg base/kg divided over 3 days. Single dose of 25 mg (sulfa)/kg |
|
0.14 |
Cross-resistance with CQ limits usefulness in areas with high rates of CQ resistance. Has been associated with toxic hepatitis and agranulocytosis when used as prophylaxis; risk when used for treatment unknown. |
|
SP Trade name: Fansidar, others |
Treatment of nonsevere falicparum infections thought to be CQ resistant. |
Average adult: 3 tablets as a single dose. |
By age: • <1 year, 1/2 tablet • 1-2 years, 3/4 tablet • 3-5 years, 1 tablet |
Known sulfa allergy |
0.12 |
Efficacy for vivax infections may be poor. Widespread resistance in P. falciparum in some regions. |
|
Sulfalene/ pyrimethamine (Metakelfin) |
|
• 6-8 years, 1.5 tablets • 9-11 years, 2 tablets • 12-13 years, 2.5 tablets. •>14 years, 3 tablets |
|
Can cause severe skin disease when used prophylactically; risk when used as treatment unknown but likely to be very low. |
||
|
MQ Trade names: Larium, Mephaquine |
Treatment of nonsevere falicparum infections thought to be CQ and SP resistant. Chemoprophylaxis in areas with CQ resistance. |
Treatment: 750 to 1,500 mg base depending on local resistance patterns. Larger doses (>15 mg/kg) best given in split doses over 2 days. |
Treatment: 15-25 mg base/kg depending on local resistance patterns. Larger doses (>15 mg/kg) best given in split doses over 2 days. |
Known or suspected history of neuro-psychiatric disorder, history of seizures, concomitant use of halofantrine. |
1.92 |
Vomiting can be a common problem in young children. In some populations (e.g., very young African children), unpredictable blood levels, even after appropriate dosing, can produce frequent treatment failure. |
|
|
Prophylaxis: 250 mg once per week. |
Prophylaxis: 5 mg base/kg once per week. |
|
|||
|
|
Dosing (all per os)a |
|
||||
|
Drug Name |
Use |
Adult |
Pediatric |
Contra-indications |
Cost (US$)b |
Comments |
|
Halofantrine Trade name: Halfan |
Treatment of suspected multidrug-resistant falciparum. |
8 mg base/kg every 6 hours for 3 doses. Average adult: 1,500 mg base divided into 3 doses as above. |
8 mg base/kg every 6 hours for 3 doses. |
Preexisting cardiac disease, congenital prolongation of QTc interval, treatment with MQ within prior 3 weeks, pregnancy. |
5.31 |
Cross-resistance with MQ has been reported. Reported to have highly variable bioavailability. Risk of fatal cardiotoxicity. |
|
Quinine |
Treatment of severe malaria. Treatment of multidrug-resistant P. falciparum. Treatment of malaria |
Nonsevere malaria: 8 mg base/kg 3 times daily for 7 days. |
Nonsevere malaria: 8 mg base/kg 3 times daily for 7 days. |
|
1.51 |
Side effects can greatly reduce compliance. Used in combination with tetracycline, doxycycline, clindamycin, or SP (where effective) and in areas where |
|
|
during first trimester of pregnancy. |
Average adult: 650 mg 3 times daily for 7 days. |
Severe: see section on treatment of severe malaria. |
|
quinine resistance is not prevalent, duration of quinine dosage can be reduced to 3 days. |
|
|
|
Severe: see section on treatment of severe malaria. |
|
||||
|
Tetracycline (tetra)/ doxycycline (doxy) |
In combination with quinine, can increase efficacy of treatment in areas with quinine resistance and/or reduce likelihood of quinine-associated side effects by reducing duration of quinine treatment. Prophylaxis. |
Tetra: 250 mg/ 4 times per day for 7 days. |
Tetra: 5 mg/kg 4 times per day for 7 days. |
Age less than 8 years, pregnancy. |
0.14-0.20 |
Used only in combination with a rapidly acting schizonticide such as quinine. |
|
Doxy: 100 mg 2 times per day for 7 days. |
Doxy: 2 mg/kg twice per day for 7 days. |
|||||
|
Prophylaxis: 100 mg doxy per day. |
Prophylaxis: 2mg/kg doxy per day up to 100 mg. |
|||||
|
|
Dosing (all per os)a |
|
||||
|
Drug Name |
Use |
Adult |
Pediatric |
Contra-indications |
Cost (US$)b |
Comments |
|
Clindamycin |
For patients unable to take tetracycline. In combination with quinine, can increase efficacy of treatment in areas with quinine resistance and /or reduce likelihood of quinin-associated side effects by reducing duration of quinine treatment. |
300 mg 4 times per day for 5 days. |
20 to 40 mg/kg per day divided into 3 daily doses for 5 days. |
Severe hepatic or renal impairment, history of gastrointestinal disease, especially colitis. |
15.00 |
Not as effective as tetracycline, especially in nonimmune patients. Used only in combination with a rapidly acting schizonticide such as quinine. |
|
Atovaquone/ proguanil Trade name: Malarone |
Treatment of multidrug-resistant P. falciparum infections. |
4 tablets (1,000 mg atovaquone + 400 mg proguanil) daily for 3 days. |
No pediatric formulation currently available, but for patients 11-40 kg in body weight: |
|
35.00 |
Fixed-dose combination. Reportedly safe in pregnancy and young children. Drug donation program exists. Pediatric formulation in development. |
|
|
11-20 kg 1/4 adult dose 21-30 kg 1/2 adult dose 31-40 kg 3/4 adult dose |
|
|||
|
Artesunate |
Treatment of multidrug-resistant P. falciparum infections. |
4 mg/kg on day 1 followed by 2 mg/kg daily for total of 7 days. |
4 mg/kg on day 1 followed by 2 mg/kg daily for total of 7 days. |
0.50-1.20 |
A high number of counterfeit artemisinin products have been found in Southeast Asia. |
|
Artemisinin |
|
20 mg/kg on day 1 followed by 10 mg/kg daily for total of 7 days. |
20 mg/kg on day 1 followed by 10 mg/kg daily for total of 7 days. |
0.50-2.10 |
Use of artemisinin derivatives not recommended during first trimester of pregnancy. |
|
Dihydro-artemisinin |
|
4 mg/kg on day 1 followed by 2 mg/ kg daily for total of 7 days. |
4 mg/kg on day 1 followed by 2 mg/kg daily for total of 7 days. |
? |
Rectal preparations of artemisinin and artesunate are available for initial treatment of severe malaria in facilities unable to administer parenteral therapy. |
|
|
Dosing (all per os)a |
|
||||
|
Drug Name |
Use |
Adult |
Pediatric |
Contra-indications |
Cost (US$)b |
Comments |
|
Artemether |
|
4 mg/kg on day 1 followed by 2 mg/kg daily for total of 5 to 7 days. |
4 mg/kg on day 1 followed by 2 mg/kg daily for total of 5-7 days. |
|
3.60 |
|
|
Primaquine |
Treatment of P. vivax infections (reduce likelihood of relapse). Gametocytocidal agent. |
14 mg base per day for 14 days. 45 mg once per week for 8 weeks. |
0.3 mg (base)/kg daily for 14 days. |
G6PD deficiency, pregnancy. |
0.06-0.24 |
Primaquine has also been investigated for prophylaxis use. See text for additional cautions. |
|
aNote: “p.o.” or “per os” is latin for “by mouth.” bCost is given for a full adult (60-kg) treatment course. Prices derived from individual reports, personal communications, McFayden (1999), Medical Economics Co. (1999) and WHO (2001b). Prices reflect best prices or best estimates; local prices may differ greatly. |
||||||
on P. falciparum malaria parasites and can no longer be relied on to provide effective treatment or prophylaxis.
Drug resistance is not limited to chloroquine. In some areas of Southeast Asia, the situation is deteriorating to the point where few effective therapies exist. Chloroquine was abandoned as first-line therapy for malaria in Thailand in 1972 in preference to sulfadoxine/pyrimethamine (SP). Drug resistance develops rapidly to dihydrofolate reductase inhibitors (such as pyrimethamine and proguanil) when used alone or in combination with sulfa drugs (such as SP) (Björkman and Phillips-Howard, 1990; Sibley et al., 2001). In 1985, in response to declining SP efficacy, SP was combined with mefloquine (Thaithong et al., 1988). After a few years of widespread use of mefloquine (15 mg/kg), greater than 50 percent of P. falciparum infections showed resistance to it in some areas of Thailand (Mockenhaupt, 1995). Cure rates were improved to 70 to 80 percent by increasing the dose of mefloquine to 25 mg/kg, but the efficacy of this higher dose also declined rapidly in some areas (Nosten et al., 2000). In Southeast Asia, parasitological response to quinine has also been deteriorating (Bunnag and Harinasuta, 1987; Wongsrichanalai, et al., 2002). Resistance to newer antimalarials, such as halofantrine, has also been reported, especially in areas with mefloquine resistance (Wongsrichanalai et al., 1992; ter Kuile et al., 1993).
Currently, multidrug-resistant malaria in Thailand is being treated with a combination of an artemisinin derivative and mefloquine; efficacy of this combination has remained high (Nosten et al., 2000). To date, no confirmed cases of resistance to the artemisinin drugs have been reported. However, reports of decreasing sensitivity in vitro in some areas, as well as a number of case reports of potential (but unconfirmed) treatment failures, raise concern that this class of antimalarial drug is not “immune” to the development of resistance (Wongsrichanalai et al., 1999; Sahr et al., 2001).
In many areas where population displacement occurs, national treatment policies of the host and source countries may not necessarily reflect the drug resistance patterns of a given region or population. In many cases this is due to a lack of current efficacy data, a lack of funds for implementation of a new policy, and a variety of other concerns (Bloland and Ettling, 1999). For example, CQ is still the recommended first-line treatment for P. falciparum in much of Africa, despite the high prevalence of CRPF. Choice of the most appropriate antimalarial drug should be based, whenever possible, on actual evaluation of the efficacy of possible therapeutic options using standard methods (see Appendix B).
Drug resistance is not an all-or-nothing phenomenon. In any given area, a wide range of parasitological responses can be found, from complete sensitivity to high-level resistance (see Table 3-4). In general, malaria parasites in western sub-Saharan Africa are less resistant to drugs like CQ and SP than malaria parasites in eastern or southern Africa. In Southeast Asia the distribution of drug resistance is highly variable and focal. While some very well-publicized areas (such as the refugee camps along the Thai-Burmese border) are faced with highly resistant malaria, requiring complicated combination therapy approaches, nearby areas reportedly have had a much longer period of success with MQ alone (K. Thimasarn, Malaria Control Programme, Thai Ministry of Public Health, 1998, personal communication; Singhasivanon, 1999; Wongsrichanalai et al., 2000).
The policy response to increasing evidence of antimalarial drug resistance has been variable as well. In parts of East Africa, parasitological resistance to CQ is very high, with 80 to 90 percent of P. falciparum infections being moderately to highly resistant (Bloland et al., 1993). In response to these high rates of resistance, Malawi switched from CQ to SP for first-line therapy for P. falciparum in 1993. A number of countries in eastern and southern Africa (including Tanzania, Kenya, Democratic Republic of Congo, Rwanda, Uganda, Ethiopia) have made similar policy changes to SP alone or in combination (with either CQ or amodiaquine) on a national or provincial/district level. After a long period of disinclination to change treatment policies, many more countries in sub-Saharan Africa are now reevaluating their national treatment guidelines and considering policy changes to locally effective regimens. Although the drugs being used differ, similar efforts are under way in the Amazon region and Southeast Asia.
VECTOR CONTROL
Attempts to interfere with the entomological link in malaria transmission are an essential and integral part of many malaria control programs. However, a vector control measure that is appropriate in one setting may be totally inappropriate elsewhere (see earlier section on Malaria Vectors and Vector Behavior). It is therefore strongly advised that the assistance of an experienced medical entomologist who is familiar with existing malaria vector data for the area in question be obtained. Decisions based on expert advice and firsthand data are the most certain route to cost-effective vector control.
The importance of understanding vector biology and behavior prior to initiating control measures is evident when one considers the key questions that must be answered to determine which type of control measure is best for a given situation. Identification of the mosquito species responsible for most malaria transmission involves surveillance, collection, and species identification from various parts of the affected area. There can also be a number of anophelines in a given area. However, not all anopheline mosquitoes transmit malaria and not all anophelines that do transmit malaria are efficient vectors. Therefore, the most commonly collected adult anopheline species captured in a given area will sometimes, but not always, be responsible for most malaria transmission. Control activities aimed at one important vector species may not be effective against another vector species. For example, insecticide-treated nets may be useful for reducing exposure to mosquitoes biting inside houses but may have no effect on mosquitoes biting primarily outside.
Information about the breeding places of vectors is required. Mosquitoes have diverse breeding habits, some of which may be targeted for control purposes. Mosquitoes generally do not range farther than 2 to 3 km from their breeding sites, unless carried by winds or some other vehicle. When one site or a few produce most of the vectors responsible for transmission, larval control may be appropriate and effective. In situations where breeding sites are small, dispersed, or not easily identified, however, larval control would be inappropriate. As mentioned previously, some malaria vectors (such as An. gambiae in Africa) can breed in pools as small and temporary as an animal’s hoofprint. Identifying breeding areas also requires considerable skill. When done correctly, larval control has been very effective in certain circumstances, such as special projects, urban areas, and land-based business ventures.
Differences in the behavior patterns of adult mosquitoes have a marked effect on their capacity to transmit malaria as well as the choice of control methods used. Preferred time of biting, for example, can vary from daytime to late evening. The efficacy of many control measures varies, depending on the mosquito activity cycle. Insecticide-treated bed nets or other control measures that are used indoors, such as space spraying with insecticides, would obviously have little effect on malaria transmission occurring at times when people are not indoors. For example, malaria transmission in much of Southeast Asia is associated with exposure to mosquitoes during activities conducted in the forest.
TABLE 3-4 Distribution of Drug-Resistant P. falciparum Malaria.
|
|
Resistance Reported1 |
|||
|
Region |
CQ |
SP |
MQ |
Others |
|
Central America (Mexico, Belize, Guatemala, Honduras, El Salvador, Nicaragua, Costa Rica, Northwestern Panama) |
N |
N |
N |
|
|
Carribean (Haiti and Dominican Republic only) |
N |
N |
N |
|
|
South America (Southeastern Panama, Columbia, Venezuela, Ecuador, Peru, Brazil, Bolivia) |
F |
F |
R |
QN |
|
West Africa |
F |
R |
R |
|
|
East Africa |
F |
R |
R |
|
|
Southern Africa |
F |
F/R |
N |
|
|
Indian subcontinent |
F |
R |
N |
|
|
Southeast Asia and Oceania |
F |
F |
F |
HAL, QN |
|
East Asia (China) |
F |
F |
F |
|
|
NOTES: 1Reports of resistance to a given agent occurring in an area do not necessarily mean that occurrence is frequent enough to pose a significant public health risk. “F” indicates agents to which frequent resistance occurs in a given area (although actual risk may be highly focal—e.g., Southeast Asia, where MQ resistance, while very frequent in |
||||
|
Comments |
|
Northwest of Panama Canal only. |
|
Resistance to MQ and QN, although reported, is considered to occur infrequently. |
|
Incidence of resistance to CQ is variable but very common in most areas. |
|
Incidence of resistance to SP is highly variable, with some reports of high levels occurring focally but generally low to moderate. |
|
Resistance to SP is considered to be generally low, except for KwaZulu Natal Province, South Africa; MQ resistance is probably very rare (reported from Malawi). |
|
Border areas of Thailand, Cambodia, and Myanmar at highest risk for multidrug-resistant infections; in other areas, incidence of resistance to SP and MQ is highly variable or even absent. |
|
Greatest problem with resistance in southern China where bulk of falciparum transmission occurs. |
|
some limited areas, is infrequent or absent in most others). “R” indicates that, while resistance to agent has been reported and can be focally common, it is not believed to occur frequently enough to warrant complete avoidance of that particular drug. “N” indicates that resistance has not been reported to date or occurs at a rate low enough to not present a substantial public health threat. |
|
Technical Overview of the Epidemiology of Malaria: Key Points
|
|