4
Review of Steller Sea Lion Biology
BIOGEOGRAPHY AND EVOLUTION
There are three extant families of pinnipeds—the otariids (sea lions and fur seals), phocids (true seals), and odobenids (walruses). Steller sea lions are members of the Otariidae family. The otariids arose from arctoid carnivores during the Oligocene, presumably by way of an ursid (bear)-like ancestor (Romer, 1966). The otariids themselves originated during the Miocene in the northeastern Pacific Ocean (Repenning et al., 1979), after which they diversified rapidly. Late Cenozoic polar cooling, and the resulting increase in marine production from cold, nutrient-rich seas, likely drove these radiations and many others.
In contrast with phocids, which range from the tropics to polar regions of the Arctic and Antarctica, otariids are generally restricted to temperate latitudes (King, 1983). Hence, unlike phocids, which successfully managed both transarctic and transtropical interchanges, early otariids were confined to the North Pacific Ocean by continental land masses in the east and west, polar sea ice in the north, and the tropics in the south (Repenning, 1976). They remained in this region until Pliocene closure of the Central American Seaway produced cool upwelled conditions in the eastern tropical Pacific Ocean, thus providing an avenue of dispersal along the west coast of the Americas to the southern hemisphere. From southern South America, and aided by the westwind drift, the otariids spread eastward to southern Africa, Australia, New Zealand, and many of the subarctic islands (Repenning et al., 1979). Fur seals first crossed the tropi-
cal Pacific into the southern hemisphere some 5 million years ago; sea lions followed about 2 million years later (Wynen et al., 2001). Otariids have never occupied the North Atlantic Ocean, apparently because they were unable to breach the tropical Atlantic.
Traditionally the otariids have been separated into two subfamilies—the fur seals and sea lions. However, this classification is not supported by recent molecular phylogenetic analyses (Wynen et al., 2001). These analyses indicate that the northern fur seal (Callorhinus ursinus) has a basal relationship, diverging from a lineage leading to the remaining otariid species about 6 million years ago. Precise relationships among these species are unclear because various clades radiated rapidly at the same time they were diverging from one another. In particular, it is unclear whether the modern sea lions are mono- or multiphyletic.
Seventeen species of otariids are recognized (Gentry, 2002)—10 fur seals (2 genera: Arctocephalus and Callorhinus) and 7 sea lions (5 genera: Eumetopias, Neophoca, Otaria, Phocarctos, and Zalophus). Two species (Zalophus wollebaeki and Arctocephalus galapogoensis) are equatorial, occurring in the upwelled waters of the Galapagos; five species occur in the northern hemisphere (Z. californianus, Z. japonicus, E. jubata, C. ursinus, and A. townsendi) and the remaining 10 species occur in the southern hemisphere. Otariids vary greatly in overall abundance, from the comparatively rare (more than 7,000 individuals) Guadalupe fur seal (Arctocephalus townsendi) to the extremely abundant (about 3 million) Antarctic fur seal (Arctocephalus gazella). According to Gentry, only Steller sea lions and southern sea lions (Otaria flavescens) are in overall decline, although northern fur seals in the eastern North Pacific Ocean could be added to this list.
In contrast with the phocids, which display a wide array of foraging behaviors and mating systems (Riedman, 1990), both of these features are more highly conserved in the otariids. All species of otariids are sexually dimorphic, with males being larger than females, and all species have polygynous mating systems in which males aggressively defend breeding areas or groups of females. This extreme sexual dimorphism and polygyny probably arose from the combined selective effects of (1) high population densities in productive temperate seas; (2) a dependency on land for birth and reproduction; (3) the limited availability of suitable land-based habitats (e.g., predator-free islands) at temperate latitudes; (4) the resulting aggregation of individuals on land; and (5) the consequent ability of males to compete for and acquire large numbers of females (Bartholomew, 1970). The postpartum behavior of adult females is also highly stereotyped—during this period, all otariid species alternate between at-sea foraging and on-land suckling until their pups are weaned.
POPULATION STRUCTURE OF STELLER SEA LIONS
Steller sea lions are distributed throughout most of the North Pacific from the coast of California to eastern Russia and northern Japan. Major rookeries and haulouts in the Alaskan part of the Steller sea lion range are shown in Figure 4.1. As indicated in Box 4.1, Steller sea lions are polygynous: males come ashore to establish breeding territories and guard harems of females. Immature animals occupy haulout sites, usually in the vicinity of the breeding and pupping rookeries. Thus, the sex and age structure observed on the breeding rookeries does not really represent the sex and age structure of the population. Existing data suggest sex ratios at birth are near 50-50; thus, there is little reason to believe any bias exists in population sex ratios except possibly in older age groups when males may show higher mortality rates than females. Higher mortality rates for older males are suggested because the competition for breeding among males creates wounds that might contribute to shorter lives, but there are very little data on survival rates at older ages.
During the period of rapid decline of Steller sea lions in western Alaska, the population in southeastern Alaska showed a slow but steady increase. This prompted consideration of the western stock as a distinct population segment as defined by the Endangered Species Act. In 1995 the National Marine Fisheries Service (NMFS) proposed listing the western stock as an endangered population separate from the eastern stock. This consideration was based on genetic studies, tagging and branding studies, and telemetry studies (60 Federal Register 192, Oct. 4, 1995, pp. 51968-51978).
With respect to behavior at the colony level that supports separation of the two stocks, Raum-Suryan et al. (2002) carried out a complete analysis of branded and tagged individuals from past marking studies. They found that the observed breeding fidelity to the rookery of birth for females branded in 1987-1988 was 67% for the western stock and 81% for females branded in 1994-1995 in the eastern stock. Sighting of marked females showed that they generally remain within 500 km of the natal rookery. In general, some straying occurred, but homing by females to their natal areas was quite strong. Males tended to use more haulout sites and rookeries and to disperse greater distances. However, there was little evidence of exchange of individuals between stocks. No adults were observed breeding in the opposite stock, but some adults of breeding age did move between stocks.
Genetic studies have mostly used mitochondrial DNA (mtDNA) sequences to characterize the stock structure of sea lions throughout their range. The mitochondrial genome is maternally inherited as a discrete unit and can be used to analyze female lineages. The distribution of female
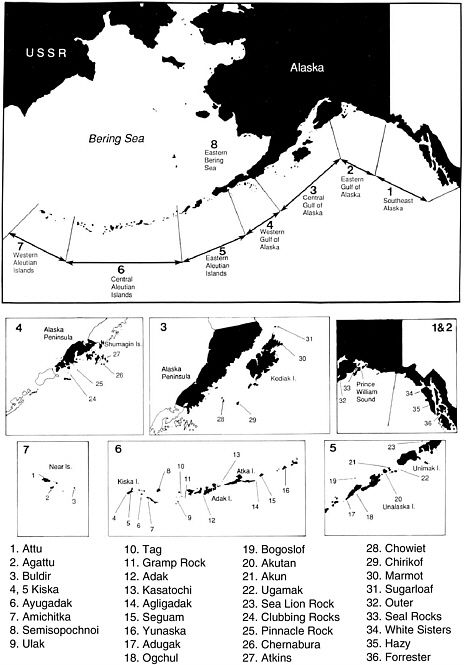
FIGURE 4.1 Distribution of the Steller sea lion in Alaska. The eight major geographic divisions of the population are shown on the map of Alaska above. The locations of individual rookeries and haulouts are indicated on the lower maps of each division and identified in the key by number.
SOURCE: Sease et al. (2001, Figure 1).
|
BOX 4.1 Steller Sea Lion Life History The Steller sea lion (Eumetopias jubatus) is the largest otariid species. Adult males and females average 566 and 264 kg in weight, respectively (Calkins and Pitcher, 1982). They are found along the northwest coast of North America and extend to the eastern Russian and Japanese coasts. The Steller sea lion is a gregarious, polygynous species with an annual seasonal reproductive cycle. They form large rookeries during late spring on isolated islands and rocky shorelines throughout their range. Males arrive at rookeries in May to establish and aggressively defend territories. The largest bulls typically control the prime territories and nonbreeding (bachelor) males remain at peripheral sites or haulouts. Females arrive at rookeries soon after the males and give birth to a single pup within a few days (Pitcher and Calkins, 1981). The majority of pupping occurs between mid-May and mid-July, and breeding occurs approximately 10 days postpartum (Pitcher and Calkins, 1981). There is a period of delayed implantation with fetal development beginning in September. Pups usually remain with their mothers for slightly less than a year before weaning, although some will nurse for a second year (Pitcher and Calkins, 1981; Ono et al., 2001). Females become sexually mature at about 4.5 years of age. Males mature at about 7 years of age but usually cannot maintain a territory until they are larger and older, about 9 to 13 years of age. SOURCE: Harmon (2001). |
lineages throughout the population range provides an indication of the degree of female migration. The mtDNA analysis identified a distinct break in genetic haplotypes between the western and eastern stocks, defined as populations separated at Cape Suckling (144° W; Bickham et al., 1998). Hence, the mtDNA haplotype distribution confirmed earlier tagging studies that showed females tend not to migrate but return to their site of birth to breed (natal homing). NMFS concluded that natal site fidelity made it unlikely that declining rookeries in the western stock would be supplemented by recruitment from more distant healthy populations in southeastern Alaska.
Existing evidence from microsatellite DNA supports the previous tagging data, suggesting that males do not have as high site fidelity as females. Examination of microsatellite DNA markers,1 found in the nuclear
genome, shows less genetic differentiation of eastern and western stocks than the mtDNA (Loughlin and York, 2001). Since the nuclear DNA of an individual includes both maternal and paternal genes, this indicates that males have migrated among the rookeries over larger distances than the females.
POPULATION TRENDS
Counts of Steller sea lions began as early as 1959 at rookery sites from islands just north of Kodiak Island out the Aleutians to Attu (see Tables 4.1 and 4.2). At this time there was no reason to believe that any decline in the number of sea lions would be possible since it was reported that there were probably over 300,000 in the world. In the late 1970s a group of biologists surveyed the Aleutians via boat and declared the “stock to be healthy and robust” (Loughlin, 1998). Although counts in those early days were not repeated at frequent intervals, they do provide some baseline data that indicate when the western stock of sea lions began its most rapid declines and in what area the declines were initially the most dramatic. There was likely a considerable difference in the quality of the counts between the early days and current counts, mostly because of improved technology and logistics. The early counts were made using several techniques, some from boats moving along the shore, others from shore counts by people on the rookeries, and still others from fixed-wing aerial observations. These counts were considered to be a total tally of individual sea lions observed. At present, the number of sea lions observed in the surveys represents the minimum population estimate and there is no attempt to correct for animals that were at sea when the counts were taken. Therefore, these values represent an index, not an exact count of the number of sea lions at any given site. Techniques have improved over time, and the most recent counts were made using standardized techniques with experienced observers from fixed-wing aircraft. Also, new imaging procedures have enhanced the applications of aerial photography.
The exact timing of declines in all areas of the range has been difficult to establish because frequent (on a schedule of about every 2 years) range-wide counts did not begin until 1989 (Loughlin et al., 1992). Braham et al. (1980) identified a decline in the eastern Aleutians by comparing aerial surveys carried out during the summers of 1975, 1976, and 1977 with surveys conducted between 1957 and 1968. These surveys covered the rookeries and haulout areas in the vicinity of Umnak and Unalaska Islands and documented a decline of 50% in this region between the survey periods. Loughlin et al. (1984) summarized existing data for the entire Steller sea lion range and noted declines in two specific areas between 1956-1960 and 1975-1980 (see Figure 4.2).
TABLE 4.1 Estimates of the Number of Nonpup Steller Sea Lions by Year and Geographical Subarea. Each subarea is characterized by the count data, the fraction of world population and the change in the size of the population from 1960 to 1989.
|
Area |
1960 |
Percentagea |
1977 |
Percentage |
1985 |
Percentage |
1989b |
Percentage |
Changec |
|
Russia |
52,000 |
21 |
29,000d |
15 |
18,000d |
13 |
13,310 |
15 |
-74% |
|
Bering Sea (U.S.) |
7,000 |
3 |
2,000 |
1 |
1,000 |
<1 |
887 |
<1 |
-87% |
|
Aleutian Islands |
99,000 |
40 |
90,000 |
46 |
61,000 |
45 |
19,033 |
22 |
-81% |
|
Gulf of Alaska |
69,000 |
27 |
55,000 |
28 |
38,000 |
28 |
31,600 |
34 |
-54% |
|
Southeastern Alaska |
7,000 |
3 |
8,000 |
4 |
8,000 |
6 |
12,303 |
14 |
+70% |
|
British Columbia |
8,000 |
3 |
5,000 |
3 |
5,000 |
4 |
8,131 |
9 |
00% |
|
Oregon and California |
8,000 |
3 |
5,000 |
3 |
4,000 |
3 |
5,357 |
6 |
-33% |
|
Total |
250,000 |
|
194,000 |
|
135,000 |
|
90,621 |
|
-64% |
|
aPercentage of the total for that year. bThe counts obtained in 1989 multiplied by 1.331 (pup correction method), except for British Columbia for which 1987 was used. cPercentage change from 1960 to 1989. dThe 1977 and 1985 values for Russia were calculated assuming the population declined by a constant number of animals each year from 1960 to 1989. Actual values were not available. SOURCE: Loughlin et al. (1992), Table 8, p. 237. |
|||||||||
TABLE 4.2 Population Trends at Rookery Sites. Trends are presented as regression correlation coefficients (r) where a positive value indicates an increasing slope and a negative value a decreasing slope. Distance refers to nautical miles measured from Outer Island off the Kenai Peninsula. This table indicates that the period of most rapid decline at many rookeries occurred between 1985 and 1989. See also Figure 4.3.
|
Rookery |
Distance (nm) |
1959-1975 |
1976-1985 |
1985-1989 |
1989-1994 |
1991-2000 |
|
Outer |
0 |
0.0158 |
-0.1354 |
|
-0.2070 |
|
|
Sugarloaf |
64 |
-0.0436 |
-0.0620 |
-0.1186 |
-0.1129 |
-0.077 |
|
Marmot |
83 |
0.0493 |
-0.0759 |
-0.1899 |
-0.1358 |
-0.104 |
|
Chirikof |
283 |
0.0181 |
-0.0021 |
-0.1519 |
-0.2117 |
-0.183 |
|
Chowiet |
298 |
-0.0579 |
0.0032 |
0.2568 |
-0.0523 |
-0.053 |
|
Atkins |
405 |
-0.0303 |
-0.0696 |
-0.1818 |
-0.0446 |
-0.034 |
|
Chernabura |
420 |
-0.0635 |
-0.1202 |
0.0277 |
0.0463 |
0.005 |
|
Pinnacle Rock |
484 |
-0.0310 |
-0.0105 |
-0.0376 |
-0.0682 |
-0.024 |
|
Clubbing Rocks |
506 |
-0.0129 |
0.0031 |
-0.0949 |
0.0074 |
-0.021 |
|
Sea Lion Rocks |
506 |
-0.0127 |
-0.1776 |
-0.1118 |
0.0774 |
0.015 |
|
Ugamak |
590 |
-0.0494 |
-0.1601 |
-0.3015 |
0.1096 |
|
|
Akun |
610 |
-0.0077 |
-0.1232 |
-0.2662 |
0.1234 |
0.007 |
|
Akutan |
632 |
-0.0347 |
-0.1062 |
-0.1966 |
0.0909 |
-0.041 |
|
Bogoslof |
698 |
0.0043 |
-0.0741 |
-0.1588 |
-0.1084 |
-0.079 |
|
Ogchul |
737 |
-0.0508 |
-0.0907 |
-0.2311 |
-0.0117 |
-0.060 |
|
Adugak |
762 |
0.0184 |
-0.0821 |
-0.2226 |
-0.0435 |
-0.046 |
|
Yunaska |
812 |
0.1329 |
-0.1236 |
-0.2080 |
-0.0098 |
-0.063 |
|
Seguam |
882 |
0.0499 |
-0.1319 |
-0.3966 |
-0.0034 |
-0.027 |
|
Agligadak |
900 |
0.0070 |
-0.1098 |
-0.3340 |
-0.5936 |
-0.155 |
|
Kasatochi |
978 |
0.0047 |
-0.1026 |
-0.1435 |
-0.1801 |
-0.013 |
|
Adak/Lake Pt. |
1039 |
-0.0496 |
0.0069 |
-0.2780 |
0.0955 |
0.030 |
|
Gramp Rock |
1086 |
0.0445 |
-0.0465 |
-0.1366 |
-0.0650 |
-0.032 |
|
Tag |
1092 |
0.0735 |
-0.1019 |
-0.1175 |
-0.1259 |
-0.027 |
|
Ulak |
1109 |
0.0185 |
0.0382 |
-0.2220 |
-0.0647 |
-0.057 |
|
Ayugadak |
1179 |
0.0446 |
-0.1224 |
-0.1476 |
-0.0706 |
-0.076 |
|
Lief Cove |
1213 |
0.2498 |
-0.1768 |
-0.3032 |
-0.0884 |
-0.056 |
|
Cape St. Stephen |
1218 |
0.0723 |
-0.0814 |
-0.2672 |
-0.1773 |
-0.066 |
|
Buldir |
1251 |
0.0349 |
|
-0.2196 |
-0.126 |
|
|
Agattu |
1324 |
0.0033 |
-0.1379 |
-0.0777 |
-0.0766 |
-0.096 |
|
Attu |
1354 |
0.0037 |
-0.0600 |
-0.2239 |
-0.0114 |
|
|
SOURCE: Modified from Table 12.2 of York et al. (1996); additional data for 1991-2000 came from Anne York, National Marine Fisheries Service, National Marine Mammal Laboratory, Seattle, personal communication, 2002. |
||||||
As depicted in Figure 4.2, a rather dramatic decline occurred in the region between about 157° W and 165° W longitude (western Gulf of Alaska to eastern Aleutians) and between 152° W and 153° W (central Gulf of Alaska). The region between 157° W and 165° W corresponds to
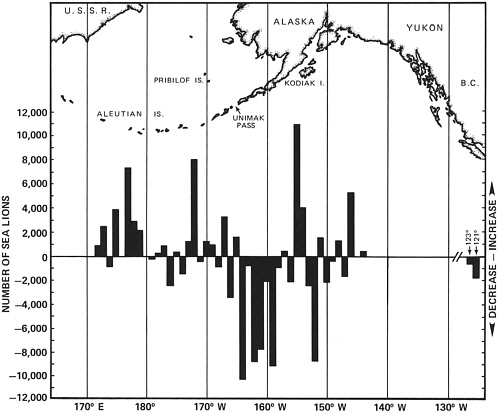
FIGURE 4.2 Changes in site counts (comparison of 1956-1960 and 1975-1980 counts) for which data are available by 1° longitude throughout the central range of Steller sea lions. An upper bar denotes an increase and a lower bar a decrease in the number of Steller sea lions.
SOURCE: Loughlin et al. (1984, Figure 2).
the area where Braham et al. (1980) noted a 50% decline, and the other region corresponds to the population in the vicinity of Kodiak Island. During the late 1980s several publications continued to document declines. Table 4.1, taken from Loughlin et al. (1992), indicates large declines since the 1960s, and the declines are range-wide except in southeastern Alaska.
The homing behavior of female Steller sea lions suggests that rookery sites may represent a metapopulation (Raum-Suryan et al., 2002). A metapopulation is comprised of local populations with low levels of exchange with one another. Each local population may fluctuate or approach extinction in response to local or global conditions. York et al. (1996) considered this possibility and analyzed the count data from the trend rookeries between Kenai and Kiska (see regions 3, 4, 5, and 6 in Figure 4.1). While
they were interested in predicting the probability of persistence over time, they summarized data that provide a look at site-specific declines over time. Table 4.2 presents the results of regression analysis on the trend data to calculate rates of change and includes information from more recent censuses. Bar graphs of these data are shown in Figure 4.3. The regression analysis shows that the most dramatic decline occurred between 1985 and 1989 and that this rapid decline included the entire western stock (Table 4.2; Figure 4.2).
The pattern suggests some initial cause(s) that started in the Gulf of Alaska to the eastern Aleutian Islands from Chowiet to Ogchul some time between 1959 and 1975 and spread across the entire western stock by 1989. During the 1989-1994 period, it seems the general area from Chernabura Rocks to Akutan had begun to recover, but then between the 1996 and 2000 censuses this pattern of recovery did not continue. The most recent count data (National Marine Fisheries Service, 2002) showed about a 5% increase in the adult count in the western population above what was found during the 2000 census. This increase should be interpreted cautiously because very few rookeries showed increases in pup counts, and population gains were mostly limited to the Gulf of Alaska with counts in the western Aleutians still in decline. The heterogeneity in population trends since 1990 suggests there may have been a change in the predominant sources of mortality from the earlier to the current pattern of decline.
VITAL RATES
All Steller sea lion population models use vital rates data derived from samples taken near Marmot Island from 1975 to 1978 and again from 1985 to 1986 (Calkins et al., 1998; Pitcher et al., 1998). Fecundity was estimated by shooting adult females on rookeries, at haulouts, and in coastal Gulf of Alaska (Pitcher and Calkins, 1981; Pitcher et al., 1998). Age estimates were based on bands in the cementum laid down each year of life, and thus the animal’s age was estimated by counting these bands from thin sections of the tooth. The second upper premolar was the tooth usually used for sea lions. Pitcher et al. (1998) also measured mass, length, and thickness of blubber, as well as pregnancy and lactation status. Nearly all females were pregnant in both time periods, and heavier females were more likely to be pregnant than lighter females. Standard length, axillary girth, and mass were lower in the 1980s than in the 1970s—most of these differences were manifest in the juvenile stage (Calkins et al., 1998). Sample sizes for the reproductive parameters were 46 and 62 adult females in the 1970s and 1980s, respectively. Age and growth were based on more animals (80 and 102 animals, respectively), but overall sample
sizes were not large for either measure. Calkins and Pitcher (1982) used the data on ages of animals shot from 1975 to 1978 to estimate age-specific survival rates for females age 1 to 13+ and adult females collected to estimate fecundity (Pitcher and Calkins, 1981) for a preliminary life table (Calkins and Pitcher, 1982).
It is on these data that all subsequent population modeling has been based—it is unknown whether the relatively small numbers of animals taken were representative of the extant population at the time of sampling. Despite the limitations of the 1970s data, no new vital rates data have been collected since then. Killing animals to obtain new data is unlikely to be permitted given the current population status. Current pup branding protocols allowing long-lasting marks coupled with intense monitoring for resightings should yield critical juvenile survival data. However, it will take some time and a consistent resighting effort to produce reliable new estimates for the older age classes.
Juvenile live captures would give additional information on this vulnerable life stage, and juvenile branding could contribute to survival estimates. No reliable new data on adult survival, at least to ages 10 to 15, will emerge from the pup branding effort until this period of time has passed. Age at first reproduction for females should start to become available after 5 or 6 years. Some data on sexual maturity for branded females have been given by Raum-Suryan et al. (2002), but the sample sizes were small. Significant sample sizes will only become available if sufficient numbers of pups are marked and monitored over long time periods. New age structure data for the present population will be required, possibly from a combination of ages of animals taken in subsistence takes, incidental captures in fisheries, or other sources of mortality.
PHYSIOLOGY, BEHAVIOR, AND FEEDING ECOLOGY
To understand the potential impacts of food limitation or changes in food quality on the biology of Steller sea lions, it is necessary to consider their physiology, nutritional demands, and behavioral feeding patterns. This is usually conducted by equating their metabolic demands with the amount or quality of food available and the costs of obtaining that food. However, despite the apparent simplicity of this question, obtaining robust data on metabolic expenditures in wild animals and the quality or quantity of food consumed is exceedingly difficult in practice. Some of this is because the animals live in remote and difficult-to-reach locations.
Because nutritional stress is central to many of the hypotheses concerning the decline of Steller sea lions, it is essential to understand the assumptions used in models for evaluating such a stress. If one breaks down the cost of living at sea into the two major components of metabolic
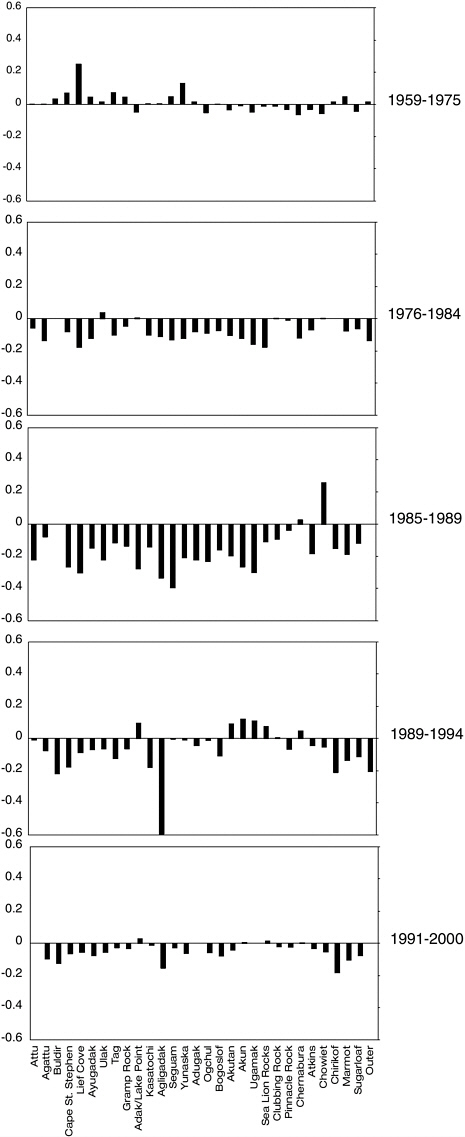
FIGURE 4.3 Relative population trends during various time intervals at rookeries oriented from west to east (Attu to Outer). Values were taken directly from Table 4.2.
SOURCES: Data for 1957-1994 from York et al. (1996); data from 1996-2000 from Sease et al. (2001).
demand and supply, there are distinct biochemical, physiological, and behavioral issues to each of those that can be addressed. Some of these factors can be measured in free-living species but others cannot and must be estimated or modeled. In all cases the goal is to obtain data that can be used to estimate the nutritional requirements of Steller sea lions.
A critical factor in any study of nutrition for pinnipeds is that these animals are well adapted to fasting as a routine part of their annual breed-
ing cycle. Northern elephant seals (Mirounga angustirostris), for example, fast from food and water for several months during the breeding season. They metabolize their blubber during this period and use water obtained as a by-product of fat metabolism. They also reduce their metabolic rate in order to minimize metabolic costs. Otariids are fasting adapted, though not for periods as long as most of the seals. Adult female Steller sea lions will fast for about 2 weeks when they come to shore to pup and breed. Adult males will fast for the entire breeding season if they are holding a territory. Therefore, standard mammalian medical, biochemical, and physiological models of starvation do not apply to these fasting-adapted species (Castellini and Rea, 1992). Fasting must be distinguished from starving in Steller sea lions to effectively study food limitation as a cause of population decline. There have been several laboratory studies of fasting in this species that have been designed to describe the body condition and metabolic changes associated with short-term (up to 2-week) periods of food deprivation in Steller sea lions (Rea and Nagy, 2000; Rea et al., 2000, 2001). These fasting-associated changes have been used to estimate feeding status in wild populations as a test of the food limitation hypothesis (Rea et al., 1998).
Behavior and Feeding Ecology
Steller sea lions consume a variety of fish species and generally dive in the top 250 m to catch their prey. There are developmental aspects to their diving patterns, with young animals diving neither as long nor as deep as older animals (Merrick and Loughlin, 1997). Recent data indicate that young-of-the-year (5 to 6 months old) dive to about 70 m, while pups slightly less than a year old dive in the 140-m range (Rehberg et al., 2001). Routine dive time for young animals remaining underwater is less than 1 minute and for older animals up to about 2 minutes (Rehberg et al., 2001). These data are important for defining critical habitat and understanding if changes in prey base move some fish stocks out of sea lion foraging range. For example, male northern elephant seals migrate into the same area of the North Pacific to feed but do so at depths far exceeding those of routine depths by Steller sea lions (Le Boeuf, 1994). Adult Steller sea lion females with newborn pups will go to sea to feed and return to nurse their pups in 24- to 48-hour cycles. Changes in trip time have been hypothesized to be an indicator of feeding success, as discussed in Chapter 6. Biological Opinion #4 used these types of dive data to prescribe areas of critical habitat and fishing regulation zones.
STELLER SEA LION DIET
As with most pinniped species, the earliest records of diet for Steller sea lions came from the stomachs of killed animals or from direct observations of animals capturing prey at the surface (Mathisen et al., 1962; Thorsteinson and Lensink, 1962). Given their decline, Steller sea lions can no longer be shot, except by subsistence hunters in certain areas. Current methods of diet analysis include collection and analysis of scat samples from rookeries or haulouts and analysis of chemical signatures in Steller sea lion tissues that are indicative of certain prey items (stable isotope and fatty acid analysis). Methods under development include the use of video dive recorders and measurements of prey ingestion with stomach temperature sensors.
Most of the diet information from stomach collections is for adult animals. Information about the diets of young animals, especially during the weaning transition, is sparser and is based mainly on newer methods using chemical signatures and scat analysis.
Data on the temporal variation in Steller sea lion diets is confined almost exclusively to scat and chemical analysis. The extensive stomach diet data of the 1970s and 1980s was obtained from animals collected during the spring and summer, with few collections made during the haulout periods. There are sporadic notes of unusual diet items for Steller sea lions, including birds, otters, and small pinnipeds, but these are not considered further in this report.
History of Diet Studies
Early Stomach Analysis
Imler and Sarber (1947) collected the stomachs of 23 animals from southeastern Alaska and from the northern Gulf of Alaska (near Kodiak and Seward) in 1945-1946. Of these, 15 (7 from the Gulf of Alaska and 8 from southeastern Alaska) contained food. Pollock was the principal item by volume in 6 of the 8 animals from southeastern Alaska and in 3 of the 7 from the Gulf of Alaska. Salmon were the only fish remains found in 2 animals collected near Seward. Overall, pollock constituted 55% of the volume of fish remains. Wilke and Kenyon (1952) noted that most sea lions collected in the Bering Sea from 1949 to 1951 had empty stomachs. Of 23 animals collected, only 2 had food in their stomachs. One contained only sand lance, and the other had 75% flatfish by volume, 15% cod, and 10% pollock.
In the stomachs of 382 males, primarily harem bulls, from five major rookeries in the western Gulf of Alaska in 1959, Thorsteinson and Lensink
(1962) found that the leading prey by frequency of occurrence were cephalopods (36%), bivalve and gastropod invertebrates (29%), sand lance (18%), and rockfish (11%). Interestingly, they noted no pollock in these stomachs; however, unidentified fish remains were found 18% of the time. In sea lions collected during 1960-1962, Fiscus and Baines (1966) examined the stomach contents of 10 Bering Sea animals and found only one with pollock; the rest had primarily capelin. In 8 animals collected from the Gulf of Alaska, they found salmon, rockfish, capelin, and herring. From 250 animals collected in 1975-1978 in the Gulf, Pitcher (1981) found the dominant food item was pollock (58% of volume, present in two-thirds of the stomachs).
Animals off Oregon were considered “opportunistic feeders, and the stomach contents usually reflected the relative abundance of prey in the area” (Maser et al., 1981). Calkins and Goodwin (1988) analyzed the stomach contents of 170 sea lions collected in southeastern Alaska and around Kodiak Island in 1985-1986. Eighty-eight animals had food in their stomachs (14 from the southeast, 74 from Kodiak). At both sites, pollock was the dominant item by frequency of occurrence (about 60%). Pollock was the largest by volume at Kodiak (42%); unidentified flatfish were second at 25%. In southeastern Alaska, Pacific cod was the largest by volume (57%), and pollock was 32% of the volume. Both studies indicated that the animals fed on a wide variety of fish.
In the proposed designation of Steller sea lion critical habitat, the National Oceanic and Atmospheric Administration summarized the diet studies as follows:
Data on SSL [Steller sea lion] prey consumption are fairly limited . . . studies in Alaska since 1975 indicate that walleye pollock has been the principal prey in all areas over this time period . . . Few data are available in Alaska prior to 1975 . . . but indicate that pollock may have been a less important component in previous years. (58 Federal Register 61, April 1, 1993, p. 17184)
In the 1992 Steller sea lion recovery plan, only stomach content analysis was summarized, with no mention of scat samples. The recovery plan notes that there may be seasonal, spatial, and individual variations in diet but concludes that “diet studies over the last 15 years show that Steller sea lions eat a variety of fishes and invertebrates; demersal and off-bottom schooling fishes predominate.” A summary table of stomach content studies can be found in the 1999 Biological Opinion (BiOp #2; Table 5 of National Marine Fisheries Service, 1999) and in condensed form in an overview report prepared by the Alaska Department of Fish & Game (Kruse et al., 2000, Table 3.2).
When concern about Steller sea lions intensified in the early 1990s,
information on the types of food and the spatial and temporal variations in their diet became more critical for listing and legal purposes but was harder to obtain because animals could not be taken for stomach content analysis. Therefore, more emphasis has been placed on scat collections and chemical analysis of Steller sea lion tissues. Scats are relatively easy to collect but may not accurately reflect the species composition and abundance of consumed prey because the efficiency of digestion varies with different prey sizes and types. Species composition of scat samples can indicate spatial and temporal patterns, but these require careful interpretation. Large carnivores like sea lions can simply strip flesh from larger prey without consuming any hard parts. Finally, scat analysis cannot be used to assess whether or not a stock is starving or flourishing because it does not reflect caloric consumption. Residence time in the digestive tract, whether a prey species contains hard parts that appear in scats and can be identified, and whether the size and numbers consumed can be inferred from this are essential for determining an energy budget. Thus, scat analyses by themselves have little to offer for resolving issues of food limitation in Steller sea lions.
Chemical analysis of sea lion tissues, such as blubber, provides an indication of what animals have been eating over longer time periods than either stomach contents or scats, but this method is limited because animals must be captured to obtain a sample. Furthermore, chemical analyses (such as stable isotope patterns and fatty acid analysis) are not yet capable of identifying individual prey items in the diet, although ongoing controlled diet studies may make this possible in the future (Iverson, 2001). Development of techniques to obtain blubber samples from dart biopsies, such as used in cetacean studies, could be useful in collecting large numbers of blubber samples without having to capture animals. At present, research on the sea lion’s diet is restricted to scat samples and the limited number of tissue samples available for chemical analyses. Bowen et al. (2001) criticize the November 2000 Biological Opinion (BiOp #3; National Marine Fisheries Service, 2000) for concluding that scats are a “reliable tool for monitoring seasonal and temporal trends in predator diets” and recommend greater use of techniques such as fatty acid signatures. However, Bowen et al. do not mention that the relatively small sample size will limit interpretation of the results.
Stable isotope patterns can be used in retrospective studies by utilizing samples from past collections. Hirons et al. (2001) examined bone samples from Steller sea lions, northern fur seals, and harbor seals collected in the Bering Sea and the Gulf of Alaska from 1951 to 1997 for possible changes in stable isotope ratios of nitrogen (d15N, which varies with trophic level) and carbon (d13C, which varies with the carbon isotope composition of primary producers). Change in d13C might thus reflect
shift at the base of the oceanic food web and dietary shifts by these apex predators might be reflected in d15N variation. The d13C analyses of Steller sea lions suggested that a subtle change might have occurred at the base of the food web. Although similar patterns were not found in harbor seal or northern fur seal, on all three pinniped species the d15N analyses indicated large differences in diet among individuals but no trends over time.
Scat Collections
As noted above, collections of scat samples increased dramatically in the early 1990s. By spring 2002, thousands of scat samples were collected range wide and analyzed for seasonal and temporal shifts in prey parts, hormone and contaminant residues, and genetic composition. Based on about 400 scat samples, Merrick et al. (1997) concluded that the “high correlation between area-specific diet diversity and population changes supports the hypothesis that diet is linked with the Steller sea lion population decline in Alaska.” This study forms the basis for the “diet diversity” hypothesis (see Chapter 6).
CHALLENGES/LOGISTICS OF WORKING WITH SEA LIONS
Because of their seasonal, behavioral, and spatial distributions, this species is particularly difficult to study with a hands-on approach. During the nonbreeding season, the animals congregate at haulout locations that are distinct from rookery sites, although a limited number of animals may remain at a rookery location through the winter. There are many more haulout sites than rookery sites throughout their range and the animals disperse during the late summer from the rookery sites to the haulout sites. However, if adult females are not going to pup, they may remain at sea and not return to rookery sites. This attendance behavior is a critical limitation in monitoring pup production rates and female condition. Essentially, only those females healthy enough to produce pups arrive at the rookeries.
-
Pups. For studies that focus on newborn pups, these young animals are captured on their natal beaches up to about 3 to 4 weeks of age. After that, they are too fast, strong, wary, and easily able to move into the water to be captured in large numbers. Therefore, hands-on studies of newborn pups (branding, physiology, nursing behavior, etc.) must take place from mid-June to early/mid-July. By working within this time limitation, hundred of pups have been captured and handled on their natal beaches with large field teams.
-
Juveniles. Only 26 individuals less than 2 years of age were captured
-
and tagged from 1990 to 1996 (Alaska Department of Fish & Game and National Marine Fisheries Service, 2001). This is because until the very late 1990s it was difficult to capture juveniles (postpup, prebreeding) on the winter haulouts given the wariness of the animals and the difficulty of stalking and darting them in poor weather conditions. However, beginning in 1998, the Alaska Department of Fish & Game refined an underwater technique that allowed capture of as many as five young animals per day. There are now routine year-round expeditions by both the Alaska Department of Fish & Game and NMFS that capture young animals up to 2 to 3 years of age. As of the spring 2002, over 309 animals had been captured by the two agencies using this method (Thomas Gelatt, Alaska Department of Fish & Game, Anchorage, personal communication, 2001). This capture technique has been a major breakthrough in the study of young sea lions and has allowed initiation of several large programs of juvenile biology and behavior to be initiated.
-
Adults. Adult females can be captured on rookeries when they are moving back and forth from the sea while nursing their pups. Adult female Steller sea lions, like all otariids, make foraging trips to sea and then return to the beaches to nurse their pups. Among Steller sea lions, the first foraging trip by the female occurs about 7 days postpartum. The at-sea duration is typically less than 48 hours. Using this behavioral handle, it is possible to capture females when they are onshore with their pups so as to attach instruments and then have a reasonable chance of recapturing the females on a return trip. The caveat to such an approach, however, is that the sample size is low for the effort required. This method requires long stalking periods to find females on the rookeries that are good candidates for darting and large field teams consisting of anesthesiologists, animal handlers, boat drivers, and technical personnel. In addition, once a female is darted and the field team moves onto the rookery, the beach is heavily disturbed and other females typically leave the site and move into the water, leaving their pups onshore. The effort is such that no more than 5 or 6 females have been captured and handled in a single breeding season. This is not a method that is easily scaled up to large datasets and is a serious limitation for studies that focus on adult breeding females.
Working with adult females on their winter haulouts to gain data on condition, pregnancy status, or behavior during the nonbreeding season is extremely difficult because the animals are wary during this time, they have no pups waiting for them on the beach, and the winter weather makes procedures such as darting much more difficult. Consequently, no adult females have been captured during the winter among the eastern stock and only five have been captured from the Gulf and the Aleutians (Alaska Department of Fish & Game and National Marine Fisheries Service, 2001). Finally, fewer than six adult males have ever been captured at
-
any time of the year because of their aggressive behavior, large size, and less accessible locations on the beaches.
-
Location and behavior. An important aspect of sea lion behavior is that many sea lions haul out on rocky ledges or pinnacles on which it is impossible to land a small boat. Thus, while sea lions may be routinely visible in large numbers to tour boats and seem to be easy to study, biologists cannot capture them. This behavior is also critical to the susceptibility of the species to shooting. Sea lions on ledges, rookeries, or haulouts do not react strongly to nearby boats and though it may be difficult or impossible to land on the beach and capture live animals, it is very easy to shoot at them from boats or a nearby beach. This susceptibility to shooting has been noted before as a particular issue with this species (Thorsteinson and Lensink, 1962). However, on a positive note, this behavior may favor new techniques that use darting methods to mark or take small biopsy samples from animals that otherwise could not be handled.
-
Permit issues. A final caveat is that the endangered status of Steller sea lions and increased legal, political, and economic ramifications of research associated directly with the species have made the Marine Mammal Protection Act permitting process cumbersome. Many hands-on research projects approved during the spring 2001 in the multimillion dollar Steller Sea Lion Research Initiative had still not been approved for permitting as of spring 2002.
-
Demographic and temporal limitations. Collecting biological information about the species (e.g., mass at age for adult females) is difficult because of the biological, handling, and legal issues addressed above. Thus, basic information that is necessary to define current conditions of the animals or to define lifetables must be collected indirectly, for example, with branding and resight studies with pups. Therefore, despite over $80 million allocated to collect such data, even an ideal experiment designed to define the survivorship of pups would take at least 7 to 8 years until the animals matured and survived to reproductive age. The basic biology of the species cannot be altered by legislation, money, or research. Any experimental or adaptive management method proposed must consider the time necessary to see the results. There must be a commitment to a multiyear approach because of the basic life history of the species.




















