7
The Higher Actinides
The United States’ massive program to produce nuclear materials, which lasted for over 50 years, was halted in 1992. With the closure of its production reactors and separations facilities the Department of Energy (DOE) no longer has the capability for large-scale production of transuranic isotopes (see Chapter 2 and Appendix A). DOE’s inventory, however, contains substantial amounts of the higher actinides,1 many of which were produced in special campaigns that involved multiple irradiation and separation steps. Currently there is little or no use foreseen for these isotopes and for the most part they are considered a liability by DOE’s Office of Environmental Management (EM), which is charged with cleaning up the former production sites. In addition, the facilities for handling and storing these isotopes are being closed as part of the cleanup. Consequently, EM plans to dispose of many unique materials as waste (e.g., by adding them to high-level tank waste). This route will foreclose all other options and risks future regret of an irrevocable action.
Starting with uranium, the higher actinide isotopes are built up by successive, non-fission-producing neutron capture in the parent nucleus followed by ß-particle emission (see Figure 7.1). Because neutron capture usually splits the nucleus (fission), the yield of the higher actinides decreases rapidly as atomic number and mass increase. As noted in Chapter 3, DOE produced about 100 metric tons of plutonium-239. By contrast, kilogram quantities of americium-243 and gram quantities of californium-252 are considered large amounts. Known uses for some of the higher actinides will consume only a fraction of the available inventory.
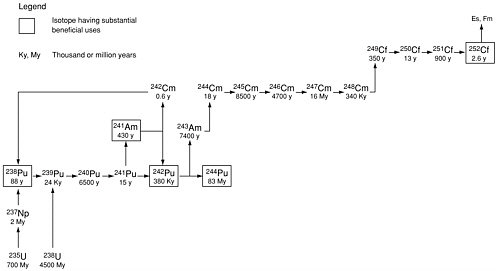
Figure 7.1 The higher actinides were built-up through multiple neutron captures by target isotopes under intense irradiation in DOE’s production reactors. Because production operations have ceased, the relatively large quantities of higher actinide isotopes in DOE’s inventory cannot be replaced. Source: Croff et al., 2002.
As is the case with DOE’s other legacy nuclear materials discussed in this report, there has been comparatively little research toward beneficial uses of the higher actinides. The committee regards this as a knowledge gap rather than an intrinsic shortcoming of the materials.
The committee recommends that the EMSP should support research to preserve and stabilize the inventory of higher actinide iso topes, identify beneficial new uses, and develop a better understand ing of their radiological and chemical health effects.
There is need and opportunity for the Office of Science to play a lead role in ensuring that the United States maintains both the facilities and the skilled personnel necessary to preserve and to conduct research with DOE’s unique separated isotopes. EMSP-funded research can play an important role in this effort.
Neptunium-237 and Plutonium-238
Plutonium-238 for use in thermoelectric power systems in the space program is produced by reactor irradiation of Np-237. During reactor irradiation, neutron capture by Np-237 produces Np-238, which beta-decays with a half-life of 2.1 days to Pu-238. Neptunium-237, a fissile isotope, is obtained by reprocessing reactor fuels.
About a third of the thermal neutron captures by U-235 lead to U-236 rather than fission of the U-235. During production operations enriched uranium fuel at Savannah River Site (SRS) was recovered, converted to metal at Oak Ridge, and recycled back to SRS fuel elements to build the U-236 in successive irradiation cycles. As U-236 increases, further neutron capture gives U-237, which decays to Np-237 by beta emission with a short half-life of 6.75 days. A lesser production route has been by (n, 2n) reactions on U-238 by the relatively small flux of fast neutrons in the production reactors. Neptunium-237 has a half-life of about 2 million years and decays by alpha emission.
About 100 to 200 kg of Np-237 will be needed to support the space program’s requirement for Pu-238 for the next 35 years (Croff et al., 2002). This supply is assured if DOE is able to convert the main Np-237 inventory, stored in solution at SRS, to a solid for shipment to Oak Ridge and if adequate reactor and processing infrastructure remains at Oak Ridge. Oak Ridge researchers (Collins et al., 2002) have also proposed to produce Pu-238 from Am-241 that will be recovered from weapons-grade plutonium to be converted into mixed oxide fuel at SRS (see Chapter 3). This would also require maintaining reactor and processing infrastructure at Oak Ridge.
Mossbauer spectroscopy is an especially useful method for basic research on the chemical properties of Np-237, such as electronic and magnetic bonding data. Many studies have been reported in the chemical literature, and a substantial experimental database exists for Np-237 Mossbauer parameters as a function of oxidation state and chemical species (Greenwood and Gibb, 1971; Long, 1984). There are research opportunities to use this technique for fundamental studies that may lead to more efficient separation processes or potentially new uses for neptunium.
Americium-243 and Curium-244
Americium-243 and curium-244 were produced in extensive campaigns at SRS and Oak Ridge from 1961 to 1975. Cm-244 was expected to become a substitute for Pu-238 in thermoelectric power units for space missions. Cm-244, with an 18-year half-life, provides 2.8 watts (thermal) per gram versus 0.57 watts per gram for Pu-238 with its 86-year half-life. A second objective was to use the Cm-244 as a target for building up to Cf-252, which can be used as a point source of neutrons, as discussed in the next section. Am-243 and Cm-244 are essential feedstocks for production of the heavier curium isotopes, californium, and higher isotopes.
The major campaign to produce americium and curium began at SRS by long irradiations of about 100 kg of Pu-239 to enhance the Pu-
240 content. This was followed by fabrication of plutonium-aluminum alloy targets and re-irradiations for two and a half years in a production reactor specially reconfigured to produce an extremely high neutron flux.2 Overall the program encompassed ten different processing campaigns with up to three target irradiation histories per campaign.
This effort produced about 10 kg of Am-243 and 2.6 kg of Cm-244. The expected markets for Cm-244 and Cf-252 did not materialize, and these materials were left in solution in the SRS F-Canyon. At the time the committee completed its work, in fall 2002, DOE had closed F-Canyon and had firm plans to dispose of its Am/Cm solution into the million-gallon-capacity high-level waste tanks at SRS. An additional 750 grams of curium are in unreprocessed targets (Mark 18A targets), which were also left as a legacy of the campaign. Besides these legacy materials, about 800 grams of curium are held at Oak Ridge National Laboratory (ORNL) and SRS for use by the DOE Office of Science to produce higher actinides. Disposing of the SRS F-Canyon solution will result in the loss of most of the U.S. feedstock for producing higher actinides.
Plutonium-244
Plutonium-244 is the product of years of intense neutron irradiation of plutonium in nuclear reactors.3 It is used in very small quantities in isotope dilution mass spectrometry to precisely measure the isotopic composition of plutonium being safeguarded by U.S. and international organizations such as the International Atomic Energy Agency. Only gram quantities were made, and the current supply of separated Pu-244 is essentially exhausted. The aforementioned Mark 18A targets, however, contain about 20 grams of unseparated Pu-244. This Pu-244 is now viewed as a national resource, mainly because of new concerns over weapons proliferation, and it is likely that the legacy Mark 18As will be retained, shipped to ORNL, and reprocessed as needed (Croff et al., 2002).
Californium-252
Californium-252 was first identified in the debris from thermo-nuclear test explosions. Currently ORNL produces about 0.25 grams of this isotope per year from SRS feedstock. The only other production is at Dmitrovgrad, Russia, which has a capacity of about one-tenth that of ORNL.
The primary use of Cf-252 is in brachytherapy for the treatment of certain cancers.4 In addition, during the past 45 years, Cf-252 has been used successfully for neutron radiography of objects ranging from flowers to entire aircraft; startup sources for nuclear reactors; and elemental analysis of coal, nuclear fuel, explosives, and the human body, among other uses (Osborne-Lee and Alexander, 1995). As noted earlier, the large demand for Cf-252 envisioned in the mid-1970s did not materialize. Recently, in treating muscular sarcomas; skin, head and neck tumors; and gynecological tumors, brachytherapy has shown high cure rates and lower recurrence rates compared to other treatment options such as surgery (Fontanesi et al., 1999; Maruyama, 1984 and Maruyama et al., 1986; Rivard et al., 1999; Taeev et al., 2001). Medical applications for radioisotopes are expanding rapidly (see Sidebar 7.1), and there appears to be a good possibility that there will be resurgence in the use of Cf-252.
Challenges and Opportunities for Preserving the Inventory
The higher actinides in the DOE inventory represent material that may be useful in its present form, may be suitable for target material, or may be essential for research into developing new materials. However, the inventory is part the legacy of former U.S. weapons production, which is generally considered to be undesirable, expensive, and hazardous. The committee concluded that there are three principal challenges to preserving the inventory:
|
SIDEBAR 7.1MEDICAL USES OF ISOTOPES There are many radioisotopes in use for medical purposes. The DOE Office of Isotopes for Medicine and Science Programs has published a list of major isotopes and their uses (http://www.ne.doe.gov/isotope/isotop.html#top). New developments continue to arise, such as radioactively tagged monoclonal antibody treatments for leukemia using bismuth-213 being studied at New York City’s Sloan Kettering Cancer Center, at ORNL, and at the National Cancer Institute (DOE, 2001c; Wu, 1997). Isotope demand versus availability has been addressed previously by an Institute of Medicine report (IOM, 1995). More recently, the demand and availability of medical isotopes was discussed in a joint meeting between the DOE and the National Cancer Institute. The DOE supply of isotopes (Lowe, 2002) in particular was discussed, including near-term and long-term plans. Five facilities are available for isotope production. Two accelerators will continue to operate (Brookhaven and Los Alamos); and three reactors will continue production (Oak Ridge, Idaho, and Sandia). The Isotope Production Facility at Los Alamos is expected to come on line in 2004. A new production cyclotron will come on line in 2008, possibly at Brookhaven National Laboratory. The acquisition of a dedicated, single-mission, isotope production and processing facility that will be fully operational by 2010 is planned to include a cyclotron and a reactor both dedicated to isotope production. Projections of demand for medical isotopes forecast a sharp rise for both therapeutic and diagnostic isotope products, as shown inFigure 7.2(Burns, 2002). While there is some debate about specific nuclides and rates of growth (Wagner and Reba, 1999), a recent expert panel reported general agreement on the following trends: (http://www.ne.doe.gov/nerac/finalisotopereport.pdf):
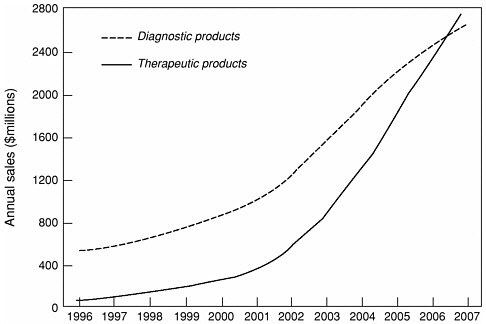
Figure 7.2The medical use of radioisotopes is expected to continue growing. In addition to Cf-252, other higher actinides may have beneficial applications in medicine. Source:Burns, 2002. |
-
Facilities capable of handling or storing the materials are being closed.
-
Few new nuclear scientists are being trained.
-
Accumulated knowledge, both documentation and personal expertise, is being lost.
The committee believes that the Office of Science’s mission, which includes stewardship of human resources, essential scientific disciplines, institutions, and scientific facilities, and fostering dissemination of information (Dehmer, 1998), is well suited to meet these challenges.
The Office of Science has an opportunity to lead other DOE offices and industrial partners in establishing a “center of excellence” to ensure that the United States has a continuing capability to handle and store large inventories of higher actinides for research, beneficial use, or as feedstock. A precedent for such a center was the ORNL Heavy Isotope Working Group, which was funded by DOE-Oak Ridge and EM’s Office of Nuclear Material and Spent Fuel (EM-21) in FY 2001. The center’s scope need not be limited to the higher actinides, rather its function could include lighter radioisotopes and cross-cut research for characterizing and stabilizing the plutonium, depleted uranium, and spent nuclear fuel described earlier in this report.
A similar center of excellence concept was offered by a previous National Academies’ committee, which recommended that EMSP consider establishing field sites for conducting subsurface contamination research to “attract new researchers to the program, encourage both formal and informal multidisciplinary collaborations among researchers, and facilitate the transfer of research results into application” (NRC, 2000, p. 8). A field research site is being established at the Idaho National Engineering and Environmental Laboratory.5
The committee also noted the tremendous amount of research, design, test, and evaluation work that was conducted at the DOE production sites. Much of the research and development needed for managing DOE’s nuclear materials has already been done at the production sites. Reports have been either archived or classified to the extent that many are neither searchable nor easily retrievable, and hence likely to be lost. Another function of the center would be to bring this documentation into the modern world of information management.
Radiation Effects on Organisms
Most of the motivation for EM to dispose of the higher actinides is the perception that they are a hazard to people or the environment.6 However, the scientific basis for determining the biological and environmental effects of the higher actinides and consequently their risks and disposal requirements is limited (see, for example, Sontag, 1983). Understanding their risks is also essential for developing beneficial future uses.
The risks presented by radionuclides have largely been dictated by their potential impact on people. For the past 50 years, what and how much can be released has been derived from acceptable doses to affected persons. First, an acceptable dose limit (rem or Sv) to humans was specified. Then, an allowable annual intake of a particular radionuclide to achieve that limit was calculated (Ci or Bq per year). All the routes (pathways) the radionuclide(s) could take from the source to the receptor were identified, and the fractional transport through each environmental medium was estimated. Finally, the allowable contamination level was back calculated—based on the exposure scenarios, length of time from release to receptor, and other factors. While this methodology has been an integral part of managing radioactive materials for several decades, there is substantial room for improvement in the process. Importantly, any reduction in conservatism in any of the assumptions could lead to a substantive impact on how radionuclides are handled and ultimately disposed.
All of our current models and standards are based on protecting humans. The general adequacy of the models has been shown with regard to food chain transport to people. Consequences to nonhuman biota have largely been ignored. It has been assumed (ICRP, 1991) that protection of the general public automatically confers an adequate level of protection for the environment. Recent developments have shown that models used to assess the potential impacts on humans from radionuclide releases to the environment may not adequately predict the potential impacts to biota (DOE, 2002b; IAEA, 1992, 2002a, 2002b; Stone, 2002). According to the IAEA (2002b, p. 6),
For biota, the concept of dose is not yet fully developed. In principle, the calculation of absorbed dose, including the distribution of absorbed dose between different organs or tissues, is feasible.
However, in practice, the ability to do so is limited by the lack of data on the uptake of radionuclides by the wide range of organisms, which are of interest, and on the distribution of radionuclides between different internal organs and tissues. Currently, most assessments of radiation doses to biota have estimated absorbed doses averaged throughout the whole organism. There is no consensus on the use of radiation weighting factors (values ranging from 5 to 40 for alpha radiation have been suggested, and used in dose assessments). An international consensus on this issue would be valuable.
Specific areas of research recommended by the IAEA include:
-
long-term radioecological studies (many of these have been abandoned or mothballed at a number of major DOE facilities— some of these studies had been in place for 30-40 years and could be resumed);
-
biotic uptake factors by species, environment, and nuclide;
-
dosimetry calculations for reference organisms;
-
biological elimination functions and rates by species;
-
environmental transfer rates;
-
dose-effect studies;
-
radiation weighting factors; and
-
development of environmental models that focus on pathways other than those leading to humans.
Stabilization and Reuse
EMSP research toward assuring safe, inexpensive, long-term storage of the higher actinides can play an important role in preserving them for beneficial future uses. Medical uses of Cf-252 appear to be increasing, as discussed in the previous section. Medical uses of other actinides, including the trans-californium isotopes, may be discovered. There are opportunities for research on both the radiological and the chemical effects of the higher actinides in biological systems. This research can lead to better understanding of both the harmful effects these materials might have in the environment and their potentially beneficial uses in medicine.
There are opportunities for research toward new uses for the higher actinides in industry. In Russia, californium has been used mainly in industrial applications rather than in medicine. Uses have included continuous neutron activation analyses in mining operations to sort and grade various ores, reducing production costs. One variation was on ships recovering undersea manganese or other nodules, to monitor intake and discard low-grade material continuously instead of hauling it to shore.
Neutron moderation measurements were used to detect water condensed or frozen inside airframe walls, which adversely affects aircraft performance—particularly in Siberia. A recent National Research Council report (NRC, 2002) on retrieving buried waste noted new technologies being developed by the U.S. Department of Defense that utilize Cf-252 in land mine detection.
Cm-244 with its half-life of 18 years constitutes a relatively rapidly decreasing stockpile of feed for higher elements. Continuing irradiation to yield Cm-246 and Cm-248 with half-lives of 4,730 years and 340,000 years would give essentially a non-decaying stockpile. There are research opportunities in basic physics, chemistry, and materials science to determine the feasibility of this transformation, which would be essential if significant amounts of curium are to be preserved for more than about 50 years.
The high flux reactor at the Institute at Dimitrovgrad has a modified neutron energy spectrum that enhances the capture to fission ratio markedly, giving improved yields of heavy nuclides. Use of this reactor to produce the more stable curium isotopes may have practical benefits as well as encouraging more international collaboration in research on the higher actinides.
Because the higher actinides are stored as relatively pure, separated materials—rather than in the highly heterogeneous mixtures that are characteristic of wastes—they are good candidates for long-term storage or disposal in specifically tailored ceramic forms. There are research opportunities to develop ceramics with lattice configurations and radiation response that will ensure that they are permanently immobilized. See Chapter 3 for a detailed discussion of ceramics for plutonium.
A variety of analytical techniques are available to study the basic chemistry of the higher actinides and for developing immobilization matrices. One of the most broadly applicable is X-ray photoelectron spectroscopy (XPS). Survey scans using the technique can be used to determine the identity and composition of the material with respect to the elements present, while high-resolution spectra can be obtained to get a detailed understanding of the chemistry of the materials, chemical alterations, and impurities. Detailed chemical information can be obtained, including oxidation states, chemical functional groups in which the elements are contained, and chemical species of the elements. The reaction chemistry involving the materials can be followed, especially reactions with water, gases, and other fluids. XPS has been used extensively to study uranium, thorium, plutonium, and other actinides with respect to both their electronic and magnetic properties and their reaction chemistries.
Auger electron spectroscopy (AES) also has been used widely in materials science studies, including its extensive use in corrosion studies. In addition to giving basic spectral data, it can be used as a microprobe-based characterization technique to image and analyze the surface of
materials. Auger spectra may be produced by both electron and X-ray beams. With the latter source also producing XPS spectra, one can obtain combined XPS-AES spectra and take advantage of additional spectral parameters on chemical species that cannot be obtained by either XPS or AES alone. The element of imaging adds the possibility of obtaining morphological data concerning interfacial reactions on material surfaces.
Both XPS and AES are highly useful in looking at the detailed chemistry of actinides in crystalline matrices. The two techniques can be coupled with microscopy to look at virtually any materials chemistry problem that these forms pose. Phenomena such as elemental segregation, redox chemistry, grain boundary problems, dissolution, and chemisorption can be addressed with these two techniques (Briggs and Seah, 1990).











