Appendix B
Estimating Animal Numbers
ESTIMATING ANIMAL NUMBERS FOR BREEDING COLONIES
(Some parts of this section are reprinted from ARENA-OLAW, 2002.)
Investigators maintain breeding colonies for a variety of reasons. For example, a breeding colony may be required for an established animal model because the animal model is not commercially available, young animals with specific age or weight that cannot be provided by a commercial breeding colony are required, or the physiologic status of a mutant animal is too severely affected for it to survive shipment.
Investigators developing a new spontaneous or induced mutant animal model need to maintain their own breeding colony because there is no alternative source for the mutant. While trying to establish a breeding colony for a new mutant model, the investigator is also working to determine phenotype, to identify affected physiologic systems, and to define inheritance pattern.
Review of protocols for breeding colonies can be a challenge for the IACUC for several reasons. There may be questions about colony management, for example, the number of breeders and the number of young per cage, the breeding system (including number of females per male or continuous versus interrupted mating), the weaning age, or methods for identification of individual animals.
Large numbers of animals are required to maintain a breeding colony. The number of animals can be only approximated because it is impossible to predict the exact number and sex of offspring. There also can be confusion about whether an estimate of number of animals distinguishes between breeders, young that cannot be used in experiments because they are of the wrong genotype or sex, and animals that are actually subjected to experimental manipulations.
Determining which animals to include in the estimated number of animals in an animal-use protocol can be confusing to the investigator in the absence of IACUC-developed guidelines. The estimated number of animals that are kept for breeding purposes and not subjected to any experimental manipulations should be part of the animal-use protocol. That is in keeping with requirement to include animals “maintained but not yet used in experiments” in the USDA Annual Report of Research Facility. Each IACUC needs to develop practical guidelines about when to include young animals in the estimated number of animals. Instructions for USDA Annual Report of Research Facility do not specifically address breeding colonies except to note that animals that are used in experiments must be reported in the appropriate category.
If a study requires fertilized one-cell eggs, embryos, or fetuses, then the experimental-design section of the protocol should indicate the number of eggs, embryos, or fetuses that are required. But the estimated number of experimental animals should be limited to the number of female animals that are mated and euthanized or surgically manipulated to collect the required eggs, embryos, or fetuses. In this situation, males would be listed as breeders because they are not subjected to any experimental manipulation.
At what age to include suckling animals in the estimated number of animals is the next question. Requiring an investigator to include all animals born fails to recognize factors that result in stillbirths. Counting all live-born animals fails to recognize normal preweaning mortality. If a suckling animal will be subjected to any manipulation—such as thymectomy, toe-clipping or ear-notching for identification, tail-tip excision for genotyping, or behavioral tests—the estimated number of manipulated sucklings must be included in the number of animals used. If suckling animals will be euthanized at or before weaning because they are of the wrong genotype or sex for the experiment, they should be included as animals held but not subject to experimental manipulation.
One alternative is to instruct investigators to include all preweaning animals subjected to experimental manipulation in the estimated number of animals or for the IACUC to request estimated animal numbers as follows:
|
Estimated number of weaned and adult animals to be subjected to experimental manipulation |
____* |
|
Estimated number of suckling animals to be subjected to experimental manipulation |
____* |
|
TOTAL |
____ |
|
Estimated number of breeders held but not subjected to experimental manipulation |
____ |
|
Estimated number of suckling animals to be euthanized at or before weaning and not subjected to experimental manipulation |
____ |
|
*Estimated numbers should be subdivided on the basis of invasiveness of procedures according to institutional criteria. |
|
If a specified number of young animals are required within a 1- or 2-week period, the number of breeders required can be estimated with the mathematical formulas shown below. Mice in pair matings (1 female to 1 male) may produce more offspring than mice in trio matings (2 female to 1 male), but if the investigator needs young mice of a specific genotype or sex, trio matings may be more productive on a per-cage basis. However, there are no guarantees that the animals will breed when expected, produce the number of offspring expected, or produce animals of the expected genotypes or sex. Thus, more or fewer than the estimated number of animals may be required.
(1)

a) Sex Correction - correction when animals of a specific sex are required:
|
|
Multiply by: |
|
Either sex can be used |
1 |
|
Female required |
0.5* |
|
Male required |
0.5* |
|
*Multiplier assumes 50:50 female-to-male sex ratio in offspring. If unusual sex ratio is known in advance, multiplier is modified accordingly. |
|
b) Mutant Correction—correction when animals of a specific genotype are required:
|
Inheritance pattern of mutant gene |
Mating scheme* |
Genotype required for experiment** |
Multiply by: |
|
Not applicable |
Not applicable |
No preference |
1.00 |
|
Recessive |
Incross |
m/m |
1.00 |
|
Recessive |
Intercross |
m/m |
0.25** |
|
Recessive |
Intercross |
m/+ |
0.50 |
|
Recessive |
Backcross |
m/m |
0.50*** |
|
Recessive |
Backcross |
m/+ |
0.50 |
|
Dominant |
Incross |
M/M |
1.00 |
|
Dominant |
Intercross |
M/M |
0.25*** |
|
Dominant |
Intercross |
M/+ |
0.50 |
|
Dominant |
Backcross |
M/M |
0.50*** |
|
Dominant |
Backcross |
M/+ |
0.50 |
|
*Definitions of mating schemes: Incross Homozygous animals of the same genotype are mated together. Cross Homozygous animals of different genotypes are mated together. Intercross Heterozygous animals are mated together. Backcross Homozygous animal is mated to heterozygous animal. |
|||
c) Infertility Factor—correction for infertility:
|
Proportion infertile matings |
Multiply by: |
|
5 % |
1.05 |
|
10 % |
1.11 |
|
15 % |
1.18 |
|
20 % |
1.25 |
|
25 % |
1.33 |
|
30 % |
1.42 |
|
35 % |
1.53 |
|
40 % |
1.66 |
(2) The number of male breeders needed will depend on the ratio of females to males. It may be 1 female to 1 male (pair matings), 2 females to 1 male (trio mating), or 3 or more females to 1 male (harem mating).
Example:
A study requires a group of 50 homozygous mutant female mice with a 1- or 2-week age range. Homozygous mutant mice are fully viable but sterile. The mutation is maintained by intercross mating. The mutation is maintained on inbred strain X. Strain X female breeders wean an average of 6 pups per litter. Approximately 20% of strain X matings are infertile.
All 84 female mice are mated at the same time to synchronize litters. The number of male breeders required will vary depending on the female-to-male ratio. Depending on the strain of mice, the number of pups weaned by females in pair matings may exceed the number of pups weaned by females in trio or harem matings.
In addition to the desired 50 homozygous mutant female mice, the 84 breeders will produce on the average 50 homozygous mutant male mice, 200 heterozygous mice of both sexes, and 100 homozygous wild-type mice of both sexes. These additional 350 mice should be listed in the protocol as animals produced but not subjected to experimental manipulation.
If the study requires a sustained production over time of a specified number of pups of a specified age per week, pups weaned per female per week (or a similar productivity index) is used to estimate the number of breeder females required. The number of pups weaned per female per week is determined as follows:
Once the number of pups weaned per female per week is determined, the number of female breeders is estimated as follows:
Example:
Fifty homozygous mutant female mice are required once a month with a 2-week age range. Homozygous mutant mice are fully viable but sterile. The mutation is maintained by intercross mating on inbred strain B. Strain B female breeders wean an average of 0.6 pup, regardless of genotype, per female per week. Approximately 20% of strain B matings are infertile.
If productivity is to be sustained for many months, the average age of the breeding population should remain constant from month to month. That is achieved by retiring the oldest breeders and replacing them with young breeders regularly, monthly in small colonies, and weekly in very large colonies. The table below shows percentage of the colony to be replaced assuming a reproductive “life span” of 5, 6, 7, 8, 9, or 10 months. The number of replacement breeders required each month must be considered in estimating the number of female breeders required. If nonmutant-bearing animals are to be produced, the number of breeders is increased to produce the required number of replacement breeders. If mutant animals are being produced, the number of breeders may or may not need to be increased to accommodate replacement breeders, depending on the genotypes of breeders and the genotypes desired for experimental use.
|
Effective reproductive life of breeders |
Percentage of colony retired monthly |
|
5 months |
20.0 % |
|
6 months |
16.7 % |
|
7 months |
14.3 % |
|
8 months |
12.5 % |
|
9 months |
11.1 % |
|
10 months |
10.0 % |
Studies involving genetic analysis are animal-intensive. Genetic analysis can involve determining whether a single gene has dominant or recessive inheritance, identifying different genes involved in a quantitative (polygenic) trait, or fine mapping to determine chromosomal location of a mutant gene. It is possible for the investigator to estimate the number of animals required but difficult for the IACUC to evaluate the estimate in the absence of experience.
1,200 mice can be required to map a single gene with recessive inheritance and full penetrance and have adequate numbers of progeny for developmental studies, phenotyping, and linkage analysis. That number assumes a breeding colony of 10-12 pair matings with a 6- to 8-month reproductive life span, around 90% productive matings, replacement of breeders, and no unusual mutant infertility or mortality.
1,100 mice can be required for quantitative trait loci analysis using analysis of F2 progeny. That number assumes small breeding colonies of two inbred parental strains (four to six pairs) and two reciprocal F1 hybrids (two to four pairs), no unusual infertility, replacement of breeders at 6- to 8-month intervals, and generation of 500-1,000 F2 mice for genotyping.
750 mice can be required to construct a congenic strain using “speed” congenic genotyping methods. That number assumes a breeding colony of 10-12 breeding pairs, replacement of breeders, and progeny for phenotyping and genetic linkage. If the homozygous mutant does not breed and the congenic strain must be developed by using intercross matings, the estimated number of mice increases to 1,200.
After founder transgenic or targeted mice have been identified, 80-100 mice may be needed to maintain and characterize a line. The number assumes up to five breeder pairs per line, breeder replacement, no unusual infertility, and adequate numbers of weanlings for genotyping and phenotyping characterization.
References
ARENA-OLAW. 2002. Insitutional Animal Care and Use Committee Guidebook. (2nd ed.). Washington DC: US Government Printing Office.
Festing, M.F.W. 1987. Animal production and breeding methods. In The UFAW Handbook on the Care and Management of Laboratory Animals. 6th ed. T.B. Poole (ed). Churchill Livingstone, Inc. New York. pp. 18-34.
Fox, R.R. and B.A. Witham (eds). 1997. Handbook on Genetically Standardized JX Mice. 5th edition. The Jackson Laboratory. Bar Harbor. Pp. 43-44, 120-125.
Standel, P.R. and Corrow, D.J. 1988. Model index of specific breeding productivity in inbred mouse colonies. Poster 48. National Meeting AALAS.
ESTIMATING ANIMAL NUMBERS TO DEVELOP AN INDUCED MUTANT
Creation of a genetically modified mouse requires four groups of mice: stud males of the same strain or stock as the desired new model, recently weaned females of the same strain or stock as the desired new model to donate embryos for genetic modification, vasectomized males from a strain or stock with good “sex drive,” and young sexually mature females from a strain or stock with good maternal characteristics. The stud males and donor females provide fertilized eggs or early embryos. Creation of a transgenic requires fertilized eggs. Creation of a targeted mutant requires blastocysts. A male mouse can be successfully mated to one or two female mice every few days to once a week, depending on the strain of mouse. A naturally ovulating inbred female mouse yields between six to eight two-cell embryos per female (Mobraaten, 1981). A naturally ovulating hybrid or outbred female mouse will usually yield more fertilized eggs or embryos. Immature female mice given hormones to induce ovulation ovulate larger numbers of eggs—16-24 eggs per inbred female (Mobraaten, 1981) to 30 or more eggs per outbred Swiss female (Wilson, 1962; Zarrow, 1961). There are marked strain differences in response to hormone injections (Hogan et al., 1986).
Generally speaking, the number of fertilized eggs collected per mouse will be higher than the number of blastocysts collected per mouse. If 100 fertilized eggs or blastocysts are to be collected, four to six female donors or 13-17 female donors could be required, depending on the genetic background of the mice, whether naturally ovulated or induced ovulated eggs are used, and whether fertilized eggs or blastocysts are collected. One report (Kovacs et al., 1993) indicated no difference in the percentage of live births between blastocysts developed from naturally ovulated donors and those developed from superovulated donors.
The number of males required depends on whether the female-to-male ratio is 1:1, 2:1, or 3:1 and higher. It is unlikely that every prospective female donor will mate at the appropriate time, conceive after mating, or respond to hormone injections. Depending on the genetic background of the mice, environmental factors, and health status, the number of unmated females or females with unfertilized eggs could be very low (less than 10%) or very high (40-50%). The number of prospective donor females must increase to compensate for females that do not mate or conceive.
Microinjection of cDNA into 100 fertilized eggs does not guarantee 100 two-or four-cell embryos to be surgically transferred. The loss will depend on technical skill, the genetic background of the mice, and environmental factors. Likewise, injection of embryonic stem cells (ES cells) into 100 blastocysts will be associated with some losses.
Vasectomized males and young sexually mature females are used to produce pseudopregnant females. Pseudopregnant female mice are required for surgical transfer of the microinjected two-cell embryos or ES cell-injected blastocysts.
The females are mated to vasectomized males. The number of vasectomized males depends on female-to-male ratios. The number of embryos surgically transferred to a pseudopregnant female is usually 8-15. If 90 microinjected two-cell embryos or blastocysts are available for surgical transfer, six to eight pseudopregnant females are required. As with collection of fertilized eggs, additional females must be mated to the vasectomized males to compensate for females that do not mate at the appropriate time.
Not all surgically transferred microinjected two-cell embryos or blastocysts undergo further cell division, implantation in the uterus, or development into viable liveborn pups. Losses depend on the genetic background of the mice, surgical skill, and environmental factors. One study reported approximately 60% births after embryo transfer of ES-cell-injected blastocysts (Kovacs et al., 1993). Blood, tail-tip, or other tissue from each liveborn pup is tested either shortly after birth or at weaning to determine whether the transgene (or targeted gene) is present in the tissues. The testing process is referred to as genotyping and is usually done with polymerase chain reaction (PCR) or Southern blot techniques. The transgene is typically present in 15-30% of the mice that develop from microinjected embryos (Gordon, 1990). Although gene alteration can be confirmed in ES cells before injection into a blastocyst, there is no guarantee that the ES cells will be incorporated into that blastocyst and produce a targeted mutant mouse.
Some young mice normally die between birth and weaning. Mortality between birth and weaning could be less than 5% or markedly higher, depending on the genetic background, induced mutation, and possible interactions between genetic background and mutation. Injection of ES cells into 100 fertilized eggs or blastocysts may not yield more than a small number of confirmed transgenic or “knock-out” weaned mice, depending on losses along the way.
Each new transgenic (or targeted) mouse must be mated to a normal mouse to determine whether the transgene (or targeted gene) is incorporated into germ cells. Each offspring is genotyped. If the first litter includes offspring that carry the transgene (or targeted gene), germline transmission has been demonstrated, and further breeding to establish the new line can began. If no offspring carrying the transgene (or targeted gene) are found, the number of offspring to be genotyped will depend on how certain one wants to be about whether the transgene (or targeted gene) is or is not incorporated into some germ cells of the parent. For example, if the new transgenic (or knockout) mouse carries the transgene in 50% of its germ cells, only half its offspring inherit the transgene (or targeted gene). Four to five offspring must be genotyped and shown not to carry the transgene (or targeted gene) before it can be concluded with 95% certainty that the new transgenic (or targeted) parent does not carry the transgene (or targeted gene) in 50% or more of its germ cells. For 99% certainty, the number of offspring increases to seven or eight. Likewise, if the transgene (or targeted gene) is present in 25% of the founder’s germ cells, one-fourth of its offspring inherit the transgene (or targeted gene). Then 10 or 11 offspring must be genotyped and shown not to
carry the transgene (or targeted gene) for 95% certainty. The number of offspring increases to approximately 16 for 99% certainty. If the new transgene is present in 12.5% of the founder’s germ cells, one-eighth of its offspring inherit the transgene (or targeted gene). Then 22 or 23 offspring must be genotyped for 95% certainty. The number of offspring increases to approximately 34 for 99% certainty.
After a founder and first-generation offspring have been identified, mating is continued as brother x sister or backcross to a selected inbred to determine whether the transgene (or targeted gene) will transmit to later generations and can be made homozygous. As this new transgenic (or targeted) line expands from founder to first, second, and later generations, phenotypes associated with hemizygous or homozygous transgenic mice (or heterozygote or homozygote targeted mice) will become apparent. At this point, protocol review becomes identical with review for breeding colonies.
The numbers of animals required to make a new induced mutant model can be estimated by using the tables below. Any numbers generated with this table are best-guess approximations. The actual number of animals required will not be known until after germline transmission and stable inheritance have been established.
|
|
Number of animals |
|
Donor females to produce desired number of naturally ovulated fertilized eggs (or blastocysts) |
(i) |
|
Stud males to mate with donor females |
(ii) |
|
Pseudopregnant females to receive manipulated embryos |
(iii) |
|
Vasectomized or sterile males to mate with pseudopregnant females |
(ii) |
|
Estimated number of nontransgenic animals to be weaned from original number of eggs (or blastocysts) |
(iv) |
|
Estimated number of transgenic (knockout) animals to be weaned from original number of eggs (or blastocysts) |
(iv) |
|
Number animals to be mated to transgenic (knockout) animals |
(v) |
|
Number of offspring to be genotyped |
(vi) |
|
Total |
|
-
N equals number of donor females needed to produce desired number of naturally ovulated fertilized eggs (blastocysts). Estimate as follows:
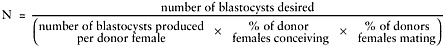
-
Will depend on whether pair (1 female to 1 male), trio (2 female to 1 male) or harem mating is used.
-
N equals number of pseudopregnant females to receive manipulated embryos. Estimate as follows:
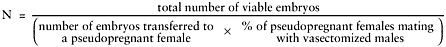
number of viable embryos = number of blastocysts injected × expected % of blastocysts viable after injection
-
N equals estimated ratio of nontransgenic to transgenic animals to be weaned from original number of blastocysts. Estimate as follows:
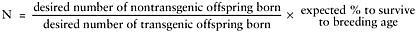
-
N equals number of animals to be mated to transgenic animals. Estimate as follows:
N = desired number of transgenic founder × number of animals to be mated to each transgenic animal to reach breeding age
The desired number of potential founders may be predetermined by the investigator. A common number is 6-10 potential founders for a single cDNA construct.
-
N equals number of offspring to be genotyped. Estimate as follows:
N = number of offspring per transgenic animal × expected number of transgenic animals actually bred
References
Beamer, W. “Use of Mutant Mice in Biological research.” 12/8/89, SCAW Conference: Guidelines for the Well-being of Rodents in Research, Proceedings edited by H. Guttman.
Gordon, J.W.. 1990. Transgenic animals. Lab Animal 19(3):27-30.
Hogan, B, F Constantini and L Lacey. 1986. Manipulating the Mouse Embryo: A Laboratory Manual. Cold Springs Harbor Laboratory Press. Cold Springs Harbor, NY.
Kovacs, M.S., L Lowe and M.R. Kuehn. 1993. Use of superovulated mice as embryo donors for ES cell injected chimeras . Lab Anim Sci 43:91-93
Mobraaten, L.E.. 1981. The Jackson Laboratory Genetics Stocks Resource Repository. In Frozen Storage of Laboratory Animals. Zeilmaker, GD (ed). Gustav, Fisher, Verlag. New York. Pp. 165-1177.
Wilson E.D., and M.X. Zarrow. 1962. Comparison of superovulation in the immature mouse and rat. J Reprod. Fert. 3:148-158.
Zarrow, M.Z. and E.D. Wilson. 1961. The influence of age on superovulation in the immature rat and mouse. Endocrinology. 69:851-855.










