Microbiological Problems in Nonhuman Primates Used in Research
Gary Baskin, DVM
MICROBIOLOGICAL AGENTS
The following microbiological agents are concerns in relation to research with and management of captive nonhuman primates: viruses, bacteria, parasites, and fungi. I will discuss their different types briefly.
Viruses
Simian Retrovirus (SRV), Type D Retrovirus. SRV is a natural infection of many species of macaques, which causes anemia, immunodeficiency, and retroperitoneal fibromatosis. SRV is very difficult to control because animals may be intermittently seropositive and/or viremic and may be asymptomatic for long periods. One must test for both antibody and virus (culture or polymerase chain reaction) at least three times before determining an animal is SRV negative. Even this testing is sometimes insufficient to detect positive monkeys. There are multiple serotypes that complicate testing protocols.
Simian Immunodeficiency Virus (SIV). SIV is a natural infection of many African primates. SIV does not appear to cause disease in its natural hosts, but it may cause an immunodeficiency disease similar to AIDS in
Tulane National Primate Research Center, Covington, Louisiana
macaques. It is also potentially zoonotic. Serology and viral isolation may be used, but there are many different isolates from many different primate species, making detection of unknown strains problematic. SIV is not a natural infection of macaques. SIV infects primarily CD4+ T-cells and macrophages.
Simian T-Lymphotrophic Virus (STLV-1). STLV is a natural infection of many Asian and African monkeys. It is apparently nonpathogenic in Asian monkeys but is sometimes associated with leukemia and lymphoma in African species, especially baboons. STLV infects T-cells.
Herpesvirus simiae (Herpes B, B-virus, Cercopithicine herpesvirus 1). Herpes B is endemic in most captive macaque colonies where the incidence often approaches 100% in adults. Herpes B is the macaque homologue of herpes simplex I in humans, in that it seldom causes serious disease in macaques (except in M. radiata and M. fascicularis). The ability of herpes B to cause fatal encephalomyelitis in humans makes the zoonotic potential of this virus its most significant aspect.
Herpes papio. Herpes papio is endemic in many baboon colonies. It is the baboon homologue of herpes simplex II in humans, in that it often affects the external genitalia. The lesions are sometimes serious and may disrupt the breeding capacity of affected individuals.
Lymphocryptovirus. Lymphocryptoviruses are γ-herpes viruses that are endemic in many monkey species. They are homologues of Epstein-Barr virus in humans and have been associated with B-cell lymphomas in immunodeficient macaques. They infect B-cells and various epithelial cells.
Rhadinovirus. Rhadinoviruses are γ-herpes viruses that are homologues of HHV-8 (KSHV) in humans. Some research colonies of rhesus and pigtailed macaques have a high incidence of seropositivity. Rhadinoviruses have been associated with retroperitoneal fibromatosis and lymphoid hyperplasia. They produce an interleukin-6-like protein.
Cytomegalovirus (CMV). CMV is endemic in most research colonies. It is a common opportunistic infection in immunodeficient animals.
Adenovirus. There are numerous adenovirus serotypes endemic in nonhuman primate colonies. Adenoviruses are common opportunistic infections.
B-19-like Parvovirus. This parvovirus is a homologue of B-19 in humans and is endemic in many macaque colonies. It can cause fatal anemia in immunodeficient monkeys.
SV40. SV40 is a papovavirus that is endemic in rhesus monkeys. It is a homologue of JC virus in humans and causes progressive multifocal leukoencephalopathy, pneumonia, and nephritis in immunodeficient monkeys.
Orthoreovirus. Orthoreovirus is endemic in some baboon colonies and can cause encephalitis.
Simian Varicella Virus (SVV). SVV is a herpesvirus that is a homologue of human chicken pox. It causes sporadic outbreaks of generalized disease in several African and Asian species. SVV can be carried latently in ganglia.
Encephalomyocarditis Virus (EMCV). EMCV is a virus of rodents that is endemic in wildlife. It causes myocarditis in several species of nonhuman primates.
Measles. Measles is a human virus to which many species of nonhuman primates are highly susceptible.
Simian Hemorrhagic Fever (SHFV). Erythrocebus patas and possibly other African monkeys carry SHFV asymptomatically. It causes highly transmissible hemorrhagic fever in macaques.
Ebola. Asian strains of Ebola-like viruses have been found in feral macaques and have caused epizootics of hemorrhagic fever in captive macaques.
Foamy Viruses. Spumaviruses are endemic in many species of nonhuman primates. They are nonpathogenic, but frequently cause cytopathic effect (CPE) in cell cultures. Spumaviruses are also zoonoses.
Bacteria
Shigella. Shigella is endemic in many primate colonies. Shigella commonly causes ulcerative, hemorrhagic colitis.
Campylobacter. Campylobacter is endemic in many primate colonies and commonly causes proliferative colitis.
Helicobacter. Helicobacter pylori and heilmanni commonly infect the stomach of macaques, where they cause chronic gastritis.
Yersinia. Rodents and birds carry Yersinia. It causes enteritis and septicemia in nonhuman primates.
Moraxella. Moraxella is commonly carried in the nasal sinuses of macaques, where it sporadically causes acute hemorrhagic sinusitis.
Streptococcus pneumoniae (Diplococcus). S. pneumoniae is commonly carried in the nasal sinuses of many nonhuman primate species. It occasionally causes fibrinopurulent serositis, especially in young monkeys.
Bordetella, Klebsiella, Escherichia coli. These bacteria are common environmental bacteria that sometimes cause pneumonia and other infections in many species of nonhuman primates.
Mycobacteria. M. tuberculosis and bovis may cause tuberculosis in nonhuman primates and are usually acquired from humans. M. avium/ intracellularae is a common environmental bacteria that frequently causes
opportunistic infections in immunodeficient animals. M. leprae may also spontaneously infect some nonhuman primates.
Parasites
Strongyloides. Strongyloides are endemic and impossible to control in animals group housed on dirt.
Cryptosporidium, Trichomonas, Balantidium, and Giardia. These protozoa are endemic in many primate colonies and cause sporadic and opportunistic enteric disease.
Sarcocystis. Sarcocystis is commonly found in the muscles of wild-caught monkeys.
Babesia. Babesia is endemic in many baboon colonies and may cause anemia in stressed animals.
Plasmodium. Many species of Plasmodium are endemic in wild and captive populations of many species of monkeys. Most are quiescent until animals are stressed.
Toxoplasma and Trypanosoma. Both protozoa are endemic in wildlife and occasionally infect outdoor-housed primates.
Fungi
Pneumocystis, Enterocytozoon, and Candida. These common fungi are endemic in most primate colonies and frequently cause opportunistic infections in immunodeficient animals.
Chronic Colitis Syndrome. The most common cause of spontaneous death in captive macaques is the chronic colitis, arthritis, amyloidosis syndrome that causes diarrhea, wasting, and death. The cause is unknown, but the syndrome may result from a dysregulated immune response to bacterial antigens in the gut.
FACTORS THAT CONTRIBUTE TO THE DIFFICULTY OF CONTROL
Documentation
It is sometimes difficult to obtain accurate and complete health reports from the source colonies. Health reports should include all testing that has been done (including the dates and types of tests and the results) as well as all previous research, diseases, treatments, and so forth. Colony health reports that detail all diseases, infections, viruses, bacterial isolations, and parasites within the entire colony are also important. The ro
dent supply industry provides some excellent examples of these types of detailed health reports.
Skilled Personnel
There is a general shortage of laboratory animal veterinarians and veterinary pathologists with training and experience in primate diseases. Most training programs do not provide much exposure to nonhuman primates. Training programs that focus on a PhD often do not produce trainees with the interest and the hands-on medical and diagnostic knowledge and skills that are needed.
Housing
Monkeys in breeding colonies are usually group housed, often outdoors, which makes the control of infectious diseases very difficult. Many primate facilities do not have an adequate number of small enclosures for separating smaller groups of monkeys that carry or do not carry particular agents.
Testing
Viral testing is very expensive and time consuming. Adequate resources for accomplishing systematic testing (e.g., money, reagents, laboratory capacity, quarantine space, labor) are often not available.
Diseases
Investigators are usually experts in their own field of research, but few are knowledgeable about the natural diseases of nonhuman primates. They sometimes do not insist on monkeys that are free of defined agents because they are not aware that the agents exist and could compromise their research, because the monkeys are not available, or because of the cost. No one knows how the various microbial agents might affect particular types of research. None of these agents would be acceptable in rodents used for research. Monkey studies usually use small numbers of animals. Any factor that contributes to variability makes the interpretation of results problematic, especially in small groups.
Although systems have been developed to eliminate some selected viral infections from colonies, there are still no proven methods for eliminating many other infectious agents, particularly bacteria. Most of these types of agents were eliminated from commercial rodent colonies by caesarian derivation and barrier maintenance. This process would be very
difficult in nonhuman primates due to their reproductive physiology. Monkeys raised in isolation develop abnormal behavior. A caesarian-derived infant would require 4 to 5 years to reach sexual maturity, and then would produce one infant per year. The time, cost, and effort required to produce completely clean monkeys is therefore daunting.
RECOMMENDATIONS
As research funding has increased, with particular emphasis on AIDS, and now bioterrorism, the demand for nonhuman primates for research has steadily increased. Unfortunately, funding to support the infrastructure that is required to produce the monkeys has not increased. We are now in the entirely predictable situation that there are not nearly enough animals available for the research that needs to be done. The same level of intellectual and financial commitment that is currently being applied to research also needs to be applied to the infrastructure that supports that research.
Some obvious solutions to this problem are to (1) make better use of the available monkeys by rigorous study design, (2) conserve available monkeys by improving disease definition and control, and (3) increase the supply of monkeys by developing new sources and expanding breeding capacity.
Additional funding to support research into the pathogenesis, epidemiology, treatment, and control of common infectious diseases of nonhuman primates would be very useful. Providing funds for adequate disease surveillance in breeding colonies (necropsy, serology, virology, bacteriology) is important. Additional training programs in laboratory animal medicine and veterinary pathology that emphasize nonhuman primates are needed. Additional emphasis on training of the husbandry staff that cares for nonhuman primates should be given as well. Funds to support the breeding infrastructure, such as corrals, indoor enclosures, clinics, and associated facilities, are needed. All of these activities are very labor intensive and require a knowledgeable, well-trained, and well-supervised technical staff. Guidelines for animal allocation based on prioritized research needs would be very helpful.
Nonhuman Primate Importation and Quarantine: United States, 1981-2001
Tom DeMarcus, MS
In this presentation, I will review the importation and quarantine of nonhuman primates (NHPs) into the United States during the previous 21 years as one component of the international perspective on the future of NHP resources. Specifically, I will (1) describe the US Centers for Disease Control and Prevention’s (CDC’s) NHP import and quarantine requirements; (2) review the CDC NHP import quarantine program activities for fiscal year 2001 (FY01; October 2000 through September 2001); (3) provide US Fish and Wildlife Service (USFWS) NHP import data for 1981 through 2000 (USFWS 2002); and, (4) describe microbiological and transportation issues related to the importation and quarantine of NHPs in the United States.
CDC NHP IMPORT AND QUARANTINE REQUIREMENTS
To prevent the introduction and spread of communicable diseases capable of causing serious outbreaks in humans (particularly monkeypox, yellow fever, Marburg/Ebola disease, and tuberculosis), importers of NHPs must (Foreign Quarantine Regulations 1985)
Division of Global Migration and Quarantine, National Center for Infectious Diseases, Centers for Disease Control and Prevention, US Department of Health and Human Services, Atlanta, Georgia
-
Register with CDC;
-
Certify that imported NHPs will be used only for bona fide science, education, or exhibition;
-
Implement disease control measures to minimize human exposure to the animals during transportation, isolation, and quarantine;
-
Isolate each shipment of imported NHPs for 31 days, monitor the animals for illness, test them for tuberculosis, test them for filovirus infection (if necessary), and maintain records regarding illness and death;
-
Report suspected zoonotic illness to CDC; and
-
Maintain records regarding the distribution of each shipment.
Before registration and periodically thereafter, CDC inspects importer facilities and reviews their procedures and record-keeping systems. In addition, CDC
-
Reviews proposed plans for each shipment itinerary;
-
Monitors handling of arriving shipments at the ports of entry and at the quarantine facilities;
-
Assesses transportation and disease control measures;
-
Reviews animal health records; and
-
Investigates reports of zoonotic illness.
CDC NHP IMPORT QUARANTINE PROGRAM ACTIVITIES FOR FY01
In FY01, 28 facilities were registered with CDC as importers of NHPs. Registrants included seven commercial importers, six zoos, four regional primate research centers, five universities, five private research facilities, and one pharmaceutical company. These facilities imported a total of 14,710 NHPs in 133 shipments, including
-
11,915 cynomolgus macaques (81% of total), in 89 shipments;
-
1867 rhesus macaques (13% of total), in 21 shipments;
-
456 marmosets (< 3% of total), in three shipments;
-
135 owl monkeys (< 1% of total), in three shipments;
-
120 baboons (< 1% of total), in three shipments;
-
113 African green monkeys (< 1% of total), in three shipments;
-
44 squirrel monkeys (< 1% of total), in one shipment;
-
40 pigtail macaques (< 1% of total), in one shipment; and
-
20 various other species (< 1% of total), in nine shipments.
Cynomolgus, rhesus, and pigtail macaques constituted 94% (13,822 animals) of the NHPs imported into the United States in FY01, including
-
2766 cynomolgus and 1855 rhesus macaques from China;
-
3440 cynomolgus macaques from Mauritius;
-
3120 cynomolgus macaques from Vietnam;
-
1653 cynomolgus and 40 pigtail macaques from Indonesia;
-
900 cynomolgus macaques from the Philippines; and
-
36 cynomolgus and 12 rhesus macaques from Canada.
Other NHP species imported into the United States in FY01 included
-
451 marmosets from the United Kingdom and five from Brazil;
-
135 owl monkeys and 44 squirrel monkeys from Peru;
-
120 baboons from Tanzania; and
-
89 African green monkeys from St. Kitts and 24 from Tanzania (CDC 2002).
Historically, mortality during transit and quarantine among NHPs imported into the United States has been reported to be as high as 20% (CDC 1989). Since 1990, imported NHP mortality rates have been comparatively low. Of the 14,710 NHPs imported in 133 shipments in FY01, a total of 107 (0.7%) animals in 41 shipments were reported to have died during transit or quarantine. Eighteen (0.12%) of these animals in four shipments were reported dead on arrival at the port of entry. Also during FY01, a total of 526 (3.5%) animals in 38 shipments were reported to have exhibited signs of illness (predominantly stress, diarrhea, or bloody diarrhea) during transit or quarantine. None of these dead or ill animals were reported to have been positive for filovirus antibody or antigen. All 14,710 NHPs imported during FY01 were tested for tuberculosis (TB). A total of 28 (0.19%) individual animals, in seven shipments arriving from five countries were identified as TB suspects. One shipment had 16 suspect animals, one had seven, and five had one each. Five of the seven shipments had positive laboratory reports (histology, culture, polymerase chain reaction, or a combination of these) for at least one animal.
USFWS NHP IMPORT: 1981-2000
This presentation includes a series of 11 charts presenting available USFWS NHP import data for 1981 through 2000 to illustrate trends in US NHP importation for the period. (Space herein does not permit inclusion of these charts. Electronic copies of the slides for this presentation, including the charts, have been provided to the Institute for Laboratory Animal Research [ILAR].) USFWS data for 1993 were not available. CDC FY93 NHP import data for three major NHP species (cynomolgus, rhesus, and African green monkeys) were inserted into the charts in an effort to ap
proximate continuity in the trends for those species. No CDC FY93 data were available for other NHP species, so that the charts for these species indicate zero imports for 1993.
Some highlights from the combined USFWS and CDC data for the years 1981 through 2001 include the following:
-
Total US NHP imports averaged 13,276 per year for the 20-year period; 16,731 per year for the period 1981 through 1990; and 9821 per year for the period 1991 through 2000. A total of 14,710 were imported in FY01. An upward trend in total NHP imports has emerged since 1998.
-
Total US cynomolgus macaque imports averaged 9335 per year for the 20-year period; 10,857 per year for the period 1981 through 1990; and 7813 per year for the period 1991 through 2000. A total of 11,915 were imported in FY01.
-
Total US rhesus macaque imports averaged 725 per year for the 20-year period; 589 per year for the period 1981 through 1990; and 862 per year for the period 1991 through 2000. A total of 1795 were imported in FY01.
-
Total US African green monkey imports averaged 554 per year for the 20-year period; 937 per year for the period 1981 through 1990; and 171 per year for the period 1991 through 2000. A total of 113 were imported in FY01.
MICROBIOLOGICAL ISSUES RELATED TO NHP IMPORTATION AND QUARANTINE IN THE UNITED STATES
Microbiological issues related to NHP importation and quarantine in the United States include monkeypox, yellow fever, Marburg/Ebola disease, and tuberculosis because of their potential to cause outbreaks of serious diseases in humans. Laboratory testing and health documentation before importation is not a requirement. Rather, importers are required to handle animals as though they are infectious during transportation and through completion of import quarantine. Importers are required to monitor imported NHPs for these and other zoonotic diseases and to report suspected cases among animals or workers.
Importers of cynomolgus, rhesus, and African green monkeys are required to perform laboratory tests for filovirus antigen on animals that die during quarantine. In addition, importers of those species are required to perform laboratory tests for filovirus antibodies on animals that are ill during quarantine with symptoms suggestive of filovirus infection. On rare occasions when positive animals have been found (1989, 1990, and 1996), CDC has worked closely with the importers to implement control measures.
Importers are required to screen all imported NHPs for tuberculosis. Each imported NHP must complete at least three negative tuberculosis skin tests at not less than 2-week intervals, using mammalian old tuberculin licensed by the US Department of Agriculture, in accordance with ILAR standards. When animals are found to be positive or suspect for TB, all remaining animals in the shipment must complete at least five additional negative TB skin tests at not less than 2-week intervals after removal of the suspect animal. This process of isolation and repeated testing is an inexact science and does not always prevent the introduction of TB infection into an NHP colony or into a group of animals that are part of an ongoing study protocol. Such introductions can cause significant morbidity and mortality that can have devastating effects on both the NHP resources and the scientific studies. Promising new TB screening tools to improve the process are being investigated.
Upon completion of quarantine, CDC reviews the veterinary medical report on each shipment and, if appropriate, concurs with its release. CDC may require extension of the quarantine period pending resolution of zoonotic illness issues.
TRANSPORTATION ISSUES RELATED TO THE IMPORTATION OF NHPs INTO THE UNITED STATES
A major transportation issue related to the importation and quarantine of NHPs in the United States is the limited number of international airlines that will transport NHPs to the United States. Before 1990, NHPs were transported to the United States on numerous international airlines; however, since 1990, the number of international airlines that will carry NHP to the United States has been declining. Currently, no major US airlines will carry NHPs to the United States. The number of foreign international airlines that will carry NHPs to the United States is limited.
A number of factors might have contributed to the decline in the number of international airlines that will carry NHPs to the United States. One factor is concern about human health. Although there have been no reported cases of serious illness in humans associated with exposure to imported NHPs in international transit, the potential for exposure is clear. In 1990, CDC required importers to implement disease control measures to minimize exposure to imported NHPs during transit. Before the implementation of these requirements, importers had little direct involvement in the handling of NHPs during international transit. The CDC requirements focused airline attention on NHP shipments.
Another factor is increased emphasis on the International Air Transportation Association regulations. Since 1990, importers and the USFWS have placed increased emphasis on ensuring that NHP shipments comply
with these requirements. Other factors contributing to the decline might include publicity surrounding human Ebola outbreaks (in Africa); human B virus cases (in the United States); and books, newspapers, magazines, media articles, and movies on these subjects. Airlines have also reported that cargo handler unions have made NHP cargo a safety issue in contract negotiations. Finally, animal activists have initiated a letter writing campaign to pressure international airlines not to carry NHPs. It is also important to note that shipment of NHPs represent a tiny fraction of the total volume of international air cargo.
The impact of the limited number of international airlines that will carry NHPs to the United States is significant. For example,
-
Available flights more often require more stops en route, increasing air transport distance, time, and crate handling.
-
Available flights more often arrive at airports distant from quarantine facilities, increasing ground transport distance, time, and crate handling.
-
Longer flight itineraries might increase shipping stress on animals.
-
Available flights are more likely to require aircraft changes en route, increasing shipping stress on animals and requiring more handling of shipping crates.
-
The necessity for more, expensive charters is increased.
CONCLUSION
Information regarding the importation and quarantine of NHPs into the United States over the previous 21 years offers one component of the international perspective on the future of NHP resources. CDC implements U.S. NHP quarantine requirements to prevent the introduction of human disease. In FY01, nearly 15,000 NHPs were imported in 133 shipments; 94% were macaques. The USFWS maintains official NHP import data, which show that total US NHP imports were fewer in the 1990s than in the 1980s, although an upward trend appears to have emerged since 1998. CDC places microbiological emphasis on preventing zoonotic disease capable of causing serious outbreaks in humans. TB has the potential to also affect US NHP colonies and research. The limited number of US and international airlines that will carry NHP to the United States is a major transportation issue.
REFERENCES
CDC [Centers for Disease Control and Prevention]. 1989. Ebola virus infection in imported primates—Virginia, 1989. MMWR 38:831-832.
CDC, National Center for Infectious Disease, Division of Global Migration and Quarantine, Field Operations Branch, program files, March 14, 2002.
Foreign Quarantine Regulations, 42 CFR Part 71.53, January 1985.
USFWS [US Fish and Wildlife Service], Division of Management Authority, hard copy and electronic documents transmitted via personal correspondence from Mark Albert, March 1, 2002.
Diagnosis of Tuberculosis in Nonhuman Primates
S. L. Motzel, DVM, MS, PhD, R. D. Schachner, R. W. Kornegay, M. A. Fletcher, B. Kanaya, J. A. Gomez, D. T-W Ngai, W. J. Pouch, M. V. Washington, L. A. Handt, J. L. Wagner and H. J. Klein, VMD
Tuberculosis (TB) is of great importance in humans and monkeys. In humans, approximately one third of the world’s population is infected. Eight million people are infected annually with Mycobacterium tuberculosis, and two million die from TB each year (Mustafa 2001). There are limitations to current nonhuman primate, guinea pig, and rodent animal models, and there is continued need for research into preventives and therapeutics.
TB is also very important in nonhuman primates. Unfortunately, necropsy of an acutely dead nonhuman primate all too frequently reveals grossly visible miliary or granulomatous lesions consistent with disseminated TB. Special staining of lesioned tissue then highlights the presence of the causative acid-fast organisms. Much of the information we have on TB in nonhuman primates is anecdotal—verbal reports on what has occurred in colleagues’ facilities. There is a comparative paucity of published literature on TB in nonhuman primates, and the information is often contradictory. Some reports outline the diagnostic value of the enzyme-linked immunosorbent assay (ELISA), lymphocyte stimulation tests, and erythrocyte sedimentation rates; and other reports conclude that they are of no value. The same confusing picture exists regarding the predictive value of the polymerase chain reaction (PCR) technique, radi
Merck Research Laboratories, West Point, PA
ography, and the classic intradermal test. From the clinical reports that exist primarily for macaques, it is difficult to extrapolate to other species regarding the course of infection and value of diagnostic tests.
In an effort to clarify the conflicting information, we completed a study to evaluate commonly used TB diagnostics. The objectives of our study were to determine the efficacy and predictability of current methods for diagnosis of TB in three species of cercopithecines and to follow the course of the disease in species in which little is known regarding susceptibility to infection. Six cynomolgus, six rhesus, and five vervet monkeys were quarantined and extensively prescreened for the absence of TB infection prior being placed on study. The research was approved by the Institutional Animal Care and Use Committee and conducted in accordance with the Guide for the Care and Use of Laboratory Animals (NRC 1996). Animals were given intrathecal injection of 100 colony forming units of Erdman strain (35801 obtained from the American Type Culture Collection) M. tuberculosis, and studies were conducted under Biosafety Level 3 conditions.
Animals were monitored daily for behavior, appetite, and general health; and any clinical signs such as coughing were noted. Intradermal tuberculin testing was conducted every 2 weeks using mammalian old tuberculin (MOT) in the palpebrum and abdomen and purified protein derivative (PPD) in the abdomen. Animals were radiographed monthly. Tracheal washes were analyzed by culture and PCR every 2 weeks for the first month and then monthly thereafter. Blood was collected every other week, and hematology, clinical chemistries, ELISA, and lymphocyte proliferation assays were performed. The animals had complete physical examinations performed and body weights recorded every 2 weeks. Animals were euthanized at the predetermined study end of 7 1/2 months postinoculation (PI) or earlier, due to pre-established euthanasia guidelines including persistent clinical signs such as coughing, anorexia, or weight loss. A complete necropsy with culture, PCR, and histopathological analysis of collected tissue was performed on each animal at the conclusion of the study.
Surprisingly, vervets were exquisitely sensitive to this experimental TB infection, with all five being euthanized at days 44 to 52 PI. Rhesus monkeys had a variable course of disease, with animals euthanized at 2, 4, 5, and 6 months PI and at study termination. Cynomolgus displayed a more chronic duration of infection, with two animals euthanized at 5 1/2 and 7 months, respectively, and the other four at study termination. On necropsy, all animals had gross evidence of tuberculosis. M. tuberculosis infection was confirmed postmortem in all animals via culture and PCR of various tissues, although results of the two methods did not always correlate. Pathology was generally related to the length of time the dis
ease progressed, with all animals displaying varying degrees of severity of generalized disseminated TB. Acute multifocal necrosis was seen in tissues from the vervet, granulomas with multinucleate giant cells were present in rhesus tissues, and caseous granulomas with mature fibrous connective tissue capsules were apparent in the cynomolgus.
Intradermal tests using MOT were uniformly negative in all species 2 weeks PI; most were predictive between 4 and 12 weeks PI but then were uniformly false-negative for the remainder of the study. Interestingly, the intrapalpebral tests were often positive at the 24- or 48-hour reading but were consistently graded one to two grades lower (and therefore negative) at the definitive 72-hour reading. Intraabdominal testing was consistently less sensitive than intrapalpebral. Intradermal abdominal testing with PPD was routinely negative in all species throughout the study. Thoracic radiography was found to be predictive in all species. Radio-graphic evidence of pulmonary disease preceded most clinical signs, and lesion severity progressed with time. All vervets had radiographic evidence of pulmonary consolidation by 6 weeks PI and all rhesus by 3 months PI. Two cynomolgus monkeys had radiographic evidence of pulmonary disease by 1 month PI, two by 4 to 5 months PI, and two were radiographically unremarkable throughout the study period. No spondylitis or extrathoracic lesions were evident. Tracheal washes were rather predictive of infection as well. M. tuberculosis was identified by PCR or recovered by culture from tracheal washes of all animals, except for three cynomolgus, from at least one sampling point throughout the study. Frequent (in this case, every other week) sampling was necessary, however, as more individual samples were negative than positive.
Erythrocyte sedimentation rates (ESRs) were predictive in the vervets, with all five having prolonged rates at 1 month PI. ESR was not predictive in rhesus or cynomolgus. Hematology and clinical chemistry was not predictive of disease in any of the species. ELISA results were intriguing, but inconsistent. Three vervets seroconverted by 4 weeks PI and two did not. Five rhesus seroconverted by 6 weeks PI; one was not seropositive until 5 months PI. Cynomolgus seroconverted from 4 to12 weeks PI. Not all animals remained consistently seropositive after seroconversion, with five of the cynomolgus having biphasic or triphasic responses. Results of the lymphocyte proliferation assay also were somewhat interesting and tended to mirror the ELISA results. Coughing was the most predictive clinical sign noted in this study. Vervets displayed intermittent to chronic coughing from about 4 weeks PI until terminated; rhesus coughed from 1 to 3 months PI to around 5 months PI, and three of six cynomolgus coughed starting at 3 to 4 months PI continuing through 5 to 6 months PI.
In conclusion, we found that vervet monkeys are more sensitive to pulmonary M. tuberculosis than rhesus monkeys, which are more sensi
tive than cynomolgus. We also found that it is important not to rely on intradermal tuberculin testing with MOT as the sole diagnostic method for tuberculosis because animals had many false-negative reactions. Furthermore, intraabdominal testing with PPD had no predictive value in this study. We found that thoracic radiography and assessment of coughing may have predictive value in diagnosing tuberculosis, as does culture or PCR testing of tracheal washes. The ELISA and lymphocyte proliferation assay may hold promise as adjunct diagnostic methods for tuberculosis.
Finally, it is clear that no single diagnostic test is absolutely predictive of TB in these Old World nonhuman primates. It is our recommendation instead to rely on a battery of tests to diagnose infection and to utilize a herd health approach to analyzing information instead of relying on individual animal data. It is clear that research is needed to discover and validate diagnostic methods that are sensitive, specific, and robust and utilize samples obtained in a minimally invasive manner.
REFERENCES
Mustafa A.S. 2001. Biotechnology in the development of new vaccines and diagnostic reagents against tuberculosis. Curr Pharmaceut Biotechnol 2:157-173.
NRC [National Research Council]. 1996. Guide for the Care and Use of Laboratory Animals. 7th ed. Washington, D.C.: National Academy Press.
Specific Pathogen-free Rhesus Macaques
Keith Mansfield, DVM
INTRODUCTION
According to the World Health Organization, the human immunodeficiency virus (HIV) is recognized as the leading infectious cause of human morbidity and mortality worldwide. More than 30 million individuals have contracted this agent, with the vast majority of infections occurring in underdeveloped regions of the world. Development of effective strategies for control, prevention, and treatment remains a significant biomedical objective that must be addressed as we move into the next millennium. Rhesus macaques infected with simian immunodeficiency virus (SIV) represent one important tool with which to advance our knowledge of this disease, and these animals have been used extensively to investigate aspects of viral pathogenesis and host immunity. For reasons of biosafety and to eliminate confounding variables, such investigations are best carried out in animals that are free of a number of viral agents commonly found in conventional colonies, including simian retrovirus type D (SRV-D), simian T-lymphotropic virus (STLV-1), SIV, and B virus (CDC 1987; Holmes and others 1995). In addition to these requirements, investigators increasingly request animals of defined major histocompatibility complex (MHC) type or animals free of additional in
New England National Primate Research Center, Harvard Medical School, Southborough, MA
fectious agents in pursuit of their research goals. MHC-defined macaques have been utilized to investigate cytotoxic T-lymphocyte (CTL) responses during SIV infection through the use of tetramer technology. Animals free of additional agents have been utilized to investigate novel vaccine strategies and the pathogenesis of opportunistic infections during AIDS. Such requirements are likely to increase with further refinements of the model.
Animals specific pathogen free (SPF) of these agents are both costly and not readily available. The establishment of SPF colonies has proven difficult, and current production from domestic sources is not adequate to meet demand from private and academic institutions. The limited availability of such animals is an impediment to the continued progress and success of animal model-based AIDS-related research.
NEW ENGLAND NATIONAL PRIMATE RESEARCH CENTER SPF COLONY
A rhesus macaque breeding colony free of the specific pathogens B virus (BV), STLV-1, SRV-D, and SIV was established at the New England National Primate Research Center (NENPRC) in 1988. Stringent virological testing to identify SPF foundation animals in NENPRC breeding groups and among macaques purchased from other domestic sources has formed the basis of this successful colony. Virological testing has been performed on site with techniques originally developed and used extensively by investigators at the Center. Through careful management, the colony has grown to 550 adult breeding and juvenile animals and approximately 150 infant macaques less than one year of age. Juvenile animals are sold and assigned to investigators at 2 to 2 1/2 years of age, and this colony has produced 1220 offspring to date.
Virological assessment of the SPF colony is performed on site and is integrated with the colony preventative health care program. All colony animals are examined, TB tested, and bled once every 3 months. Serum obtained at this time is banked and evaluated by enzyme-linked immunosorbent assay (ELISA) for antibodies to B viruses, SRV-D, STLV-1, and SIV. Viral antigen and ELISA plates are produced on-site through techniques previously described, and herpes simplex is used in lieu of B virus antigen for biosafety reasons (Daniel and others 1988).
On-site testing is utilized both because it is cost effective and because it has reduced turnaround time compared with commercial laboratories. Positive and negative ELISA values are set using commercially available software (Dynatech Laboratories). All indeterminate or positive ELISA readings are sent for conformational testing to the Simian Retrovirus Laboratory (N. Lerche, Davis, CA) or to the National B Virus Reference
Laboratory (J. Hilliard, Atlanta, GA). Viral isolation, PCR, and immuno-histochemistry are available on site and are used in the investigation of selected cases. The colony has remained negative to simian retroviruses since 1990. As a result of this success, annual testing for the simian retroviruses was initiated in 1999. A single break in BV SPF status occurred in 1996, and BV testing continues quarterly. The testing algorithm used for derivation of SPF macaques may be seen in Figure 1.
SPF DEFINITIONS
SPF macaques may be defined as offspring arising from breeding programs in which selected agents have been eliminated through an extensive test-and-remove strategy of founder animals. SPF founders may
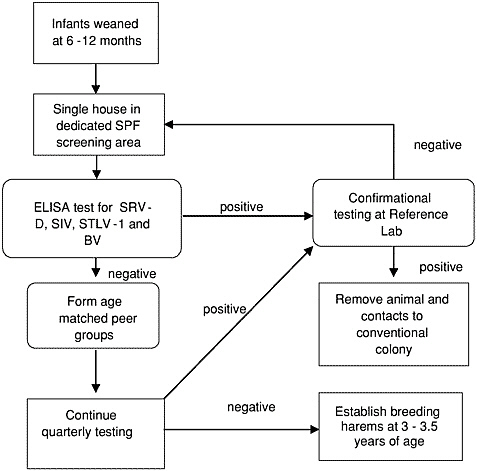
FIGURE 1 Testing algorithm for macaque SPF derivation.
also be known as level 1 SPF animals and offspring as level 2 SPF. By definition, SPF animals have been housed either individually or only with contact animals of similar virological status. “Conventional” animals are those in which no testing and segregation strategy has been employed. A “seronegative” designation is given to animals that arise from conventional colonies, lack antibodies to a particular agent, and have not been housed with other potentially infected animals. An animal lacking antibodies to a particular agent but housed with other infected animals should also be considered conventional.
SPF TARGET VIRUSES
The SPF target viruses may differ from colony to colony. The minimum target viruses for colonies supported by the National Institutes of Health/National Center for Research Resources are B viruses, STLV, SIV, and SRV-D.
SIV
SIVs are a group of related primate lentiviruses that have been investigated extensively as an animal model of human HIV infection (Gardner and others 1994). Numerous African primate species are naturally infected with specific strains of SIV with little clinical effect. Cross-species transmission from these species to Asian macaques results in a disease process remarkably similar to AIDS in humans (Baskin and others 1988; Lackner 1994). During the 1970s and 1980s, investigation of spontaneously occurring immunodeficiency in colony macaques housed at several National Primate Research Centers revealed that these animals had become infected with SIV (Lowenstine and others 1992; Mansfield and others 1995). Presumably, infection had resulted from cross-species transmission between captive animals. Although an antibody-negative viremic state exists, a simple test and removal strategy using standard whole virus ELISAs was successful in eliminating this virus from colonies (Daniel and others 1988). Whereas SIV is a target virus of most SPF colonies, spontaneous SIV infection of macaques has not been recognized in domestic colonies since the mid-1980s.
SRV-D
In spite of the extensive effort that has been devoted to the study of SIV, relatively little work has been conducted on SRV-D. The virus readily infects T cells (CD4 and CD8), B cells, macrophages, and epithelial cells (Lackner and others 1990). SRV-D is the principal cause of immunodefi
ciency in domestically bred macaques and is a significant cause of morbidity and mortality when present in captive colonies. The natural host population for the SRV-D serotypes is unknown. SRV-D may be isolated from saliva of healthy carrier animals, and biting with inoculation of saliva or blood is the most likely method of horizontal transmission (Lerche and others 1986). Mother-to-infant transmission may occur in the perinatal and postnatal period. Clinical disease is characterized by chronic diarrhea and acquisition of opportunistic infections. Retroperitoneal fibromatosis and noma are clinical conditions associated with chronic SRV-D infection. As with SIV, a test and remove strategy has been successful in eliminating SRV-D (Daniel and others 1988). An antibody-negative viremic state occurs not uncommonly after in utero or perinatal transmission. Although this antibody-negative state may complicate testing strategies based on the detection of host immune responses, such animals generally present with clinical disease early in life, which can be eliminated by maintaining a high index of clinical suspicion and routinely attempting viral SRV-D isolation of ill animals. Alternatively molecular-based assays such as PCR or routine viral isolation can be incorporated in the initial screening program.
B VIRUS
B virus is an alphaherpes virus, which is similar to herpes simplex in humans and causes widespread infection of all species of macaques. B virus infection of macaques usually results in self-limiting clinical disease, with the establishment of latency after primary infection (Weigler and others 1993). The virus is readily transmitted, with most animals seroconverting by 2 years of age (Kessler and Hilliard 1990; Weigler and others 1990). Although consequences in the natural host are limited, zoonotic transmission of B virus to human handlers may result in severe clinical disease and often death. Even though it is a rare occurrence, with fewer than 40 fatal cases described in literature since 1932, many facilities will not accept B virus antibody-positive animals for reasons of biosafety (Holmes and others 1995). Of the four target viruses, B virus has been the most problematic to eliminate from SPF colonies (Weigler and others 1990), probably for multifactorial reasons. A phenomenon of delayed seroconversion has been recognized clinically in SPF macaque colonies since their inception. Delayed seroconversion can be defined as cases in which animals that have repeatedly tested negative for B virus for more than 1 year develop B virus antibodies (Freifeld and others 1995; Ward and Hilliard 1994). The phenomenon is poorly understood, but one hypothesis is that animals that were infected early in life may establish
latency without an adequate antibody response. These animals may then develop a primary humoral immune response during periods of viral reactivation. Regardless of their SPF status, it is common policy to treat all macaques as if they potentially harbor B virus and take appropriate precautions.
STLV
STLV is a type C retrovirus related to human T-lymphotropic virus type-1 (Franchini and Reitz 1994; Watanbe and others 1985). STLV appears to be less readily transmitted in macaque colonies with reported seroprevelance rates of 0 to 20%. Serological surveys indicate an increasing prevalence with age, and although the mechanism of natural transmission is unknown, parenteral and sexual routes are suspected of being of greater importance than perinatal transmission (Ishikawa and others 1987). Seronegative virus-positive animals are uncommon. STLV appears to have limited health consequences in immunologically normal macaques, but it has been associated with lymphoproliferative disorders such as lymphoma in AIDS (Homma and others 1984). STLV has limited zoonotic potential but has been selected as a target SPF virus due to its potential as a confounding variable in immunological studies, particularly those investigating other simian retroviruses. Commercially available ELISA reagents to detect antibodies to HTLV-1 also detect antibodies to STLV and can be used in a test and remove strategy (Daniel and others 1988). All positive tests should be verified by Western blot at a reference laboratory because the test used in this setting may lack specificity.
OTHER POTENTIAL SPF TARGET AGENTS
In addition to these four SPF target viruses, nonhuman primates may be infected with a variety of other viral bacterial and parasitic agents (Ward and Hilliard 1994). Such agents may adversely affect colony health or represent a significant zoonotic risk. Pathogen-free animals may also be required for specific research protocols. Examples of such agents are listed in Table 1. Many facilities have developed or are developing “expanded” SPF program in which animals are free of other agents in addition to the original four target viruses. Such expanded programs should be based on the present and anticipated research needs of facilities being supplied by the breeding program. In establishing new colonies, it may be educational to screen a subset of the source colony for additional agents. If the seroprevalence level of such additional agents is low or nonexistent, it may be cost effective to include these agents in the SPF program.
TABLE 1 Potential Additional Specific Pathogen-free Target Agents
|
Agent |
Zoonotic Potential |
Comment |
|
Viral |
||
|
Rhesus rhadinovirus (RRV) |
Unknown |
RRV-free animals may be required for specific research protocol |
|
Lymphocryptovirus (LCV) |
Unknown |
LCV-free animals may be required for specific research protocol |
|
Rhesus cytomeglovirus (RhCMV) |
Unknown |
RhCMV-free animals may be required for specific research protocol |
|
Simian foamy virus (SFV) |
Yes |
May confound studies; zoonotic potential |
|
Simian virus 40 (SV40) |
Yes |
SV40-free animals may be required for specific research protocols; zoonotic potential |
|
Bacterial |
||
|
Helicobacter pylori |
Yes |
H. pylori-free animals may be required for specific research protocols |
|
Parasitic |
||
|
Cryptosporidia parvum |
Yes |
Potential zoonosis. C. Parvum-free animals may be required for specific research protocol |
|
Enterocytozoon bieneusi |
Yes |
E. Bieneusi-free animals may be required for specific research protocols; zoonotic potential |
VIRAL TESTING
A test-and-remove strategy based on detection of viral-specific antibodies is the foundation for establishing colonies free of target agents. Often screening and confirmational tests are utilized. The ideal screening test would be inexpensive, sensitive, specific, and technically simple to perform. Antibody detection-based ELISA procedures fulfill these criteria and have been used extensively in the establishment of SPF colonies.
On-site screening of samples by ELISA provides some advantages over sending all samples to reference laboratories for analysis. On-site testing is less expensive and has a shorter turnaround time. With proper planning, tests can be run within 24 to 48 hours of collection and can greatly expedite colony management decisions. Delays in obtaining re
sults may be extensive, particularly if samples must be sent from the country of origin for testing. Such delays may adversely affect husbandry decisions.
ELISA plates may be made for the four target viruses utilizing purified whole virus preparations (Daniel and others 1988). HSV-1 may be used in lieu of B virus for biosafety reasons. HSV-1 and HTLV-1 plates are also available commercially and can be adapted for use in a surrogate testing program for B virus and STLV-1, respectively. SRV-D and SIV plates are not currently available commercially and must be prepared from purified virus. A summary of viral preparation and ELISA techniques currently in use at the NENPRC is presented in Table 2.
Antibody testing for B virus may lack sensitivity in certain instances. Seroconversion of SPF founder animals has previously been described and represents a problem in the establishment of SPF colonies. It is suspected that delayed antibody response plays a role in the virus-positive result of tested animals (Ward and Hilliard 1994). Whether such animals shed virus and remain a threat to other colony animals and humans remains unknown. In 1996, it was noted that indeterminate values utilize the HSV ELISA in a NENPRC female SPF founder. Previously this animal had been negative on repeated HSV ELISAs performed at NENPRC and on a Western blot analysis performed at the National B Virus Reference Laboratory in 1992. Following recognition of the indeterminate ELISA, confirmational testing was requested from the National B Virus Reference Laboratory that confirmed B virus antibody by Western blot. This animal
TABLE 2 Parameters in Specific Pathogen-free Testing at NENPRCa
|
Viral purification • Cells for virus production grown in 600-mL T-150 flasks • Harvested by centrifugation 72 hr after splitting • Cell-free supernatants pelleted by ultracentrifugation (48,000 g at 4°C for 3 hr) • Purified by sepharose 4B column chromatography • ELISAa plates coated with viral antigen diluted 1:1,000 in Triton ×-100-PBSa and blocked with 0.3% BSAa |
|
ELISA • 1:20 dilution of test serum (1hr) • 1:100 dilution of goat anti-human immunoglobulin G (Fc) conjugated with APa (1 hr) (Kirkegaard & Perry Laboratories) • Developed with p-nitrophenylphosphate substrate (30 min) • Absorbance read at 410 nm with Dynatech ELISA reader |
|
aAP, alkaline phosphatase; BSA, bovine serum albumin; ELISA, enzyme-linked immunosorbent assay; NENPRC, New England National Primate Research Center; PBS, phosphate-buffered saline. |
and contacts within the breeding harem were removed from the colony. Further testing did not reveal seroconversion of additional animals in the group, and the colony has remained negative to B virus based on continued quarterly testing. This case illustrates the problem experienced in attempting to eradicate B virus from macaque colonies. Although of concern, this is the only such case recognized at the NENPRC colony since 1992, and it represents more than 3000 macaque-years of observation. As in other institutions, the NENPRC maintains a policy that all macaques should be treated as if they harbor B virus and appropriate precautions taken.
TESTING STRATEGIES
Testing strategies may vary depending on the founding population and target SPF agents, and they should be tailored to the individual facility. A testing strategy is outlined in Figure 1. In general, juvenile animals between 6 and 12 months of age are the best SPF candidate animals. Maternal immunity offers a level of protection, and most animals do not seroconvert to the target viruses until 2 to 3 years of age. Although animals may be used at a younger age, this approach increases husbandry requirements and may result in increased health problems and behavioral disorders. Healthy juvenile animals are weaned from founder breeding groups at 6 to 12 months of age. At weaning a blood sample is obtained and routine preventative health care performed. Animals are housed individually until initial virological testing is available. Seropositive animals are returned to the conventional colony. Seronegative animals are placed in small age-matched peer groups of 2 to 4 animals. Small peer groups allow for normal behavioral development but limit contact to prevent spread of infectious agents. In general, a 1:8 male to female ratio is used in the harvest. Animals are examined quarterly, and blood is drawn for serological assessment. Indeterminate or positive ELISA tests obtained at this stage should be confirmed at a reference laboratory. If seroconversion is detected, the entire group should be removed to the conventional colony. Quarterly testing should continue for a minimum of 2 years, and animals should be placed in breeding harems at 3 to 3 1/2 years of age. Due to difficulties experienced in eliminating BV, quarterly testing for this agent should continue indefinitely.
HOUSING STRATEGIES
Housing strategy is an important component of SPF colony management and may directly affect program success. A variety of strategies have been used to house SPF breeding colonies including time mating,
corral breeding, and harem breeding configurations. All strategies offer distinct advantages and disadvantages (summarized in Table 3). Corral housing is a common configuration used in areas of the world in which year-round climatic conditions allow housing of animals outdoors. Corrals generally contain 15 to 20 breeding males and 100 to 125 adult females. Corral configurations have reduced fixed and variable costs including reduced construction, labor, and supply costs. Reproductive efficiency is usually greater than with other housing configurations. Corral housing presents several distinct disadvantages from the standpoint of establishing SPF colonies. The main disadvantage is that pathogen containment is more difficult in large group settings. A single break in SPF status puts all contact animals at risk. When this occurs in breeding harems, potential contacts have been limited to the 8 to 10 animals housed together. Testing of the affected group can be intensified or the entire group can be eliminated. A break in SPF status in corral housing may put 150 to 200 animals at risk, and a subsequent test-and-remove strategy is difficult or impossible to perform.
In contrast, breeding harems consist of one male and 8 to 10 adult females. Animals are housed in indoor pens or out-door “corn cribs.” This smaller size effectively contains breaks in SPF status. Breeding harems also allow for the more accurate construction of breeding pedigrees and enhance the ability to observe sick or injured animals. Smaller group size increases the ability to observe and treat injured animals and to perform routine preventative health care.
Time mating strategies offer similar advantages to breeding harems in pathogen containment but have high caging costs, are labor intensive, and often have reduced reproductive performance. They allow the production of timed pregnancies for research purposes.
TABLE 3 Advantages and Disadvantages of Breeding Strategies in Production of Specific Pathogen-free Macaques
|
Breeding Corrals |
Breeding Harems |
|
Increased reproductive efficiency |
Decreased reproductive efficiency |
|
Decreased initial construction costs |
Increased initial construction costs |
|
Decreased husbandry costs |
Increased husbandry costs |
|
Difficult to pedigree |
Reliable pedigrees |
|
Increased overall mortality |
Decreased overall mortality |
|
Difficult pathogen containment |
Ease of pathogen containment |
PREVENTIVE HEALTH PROGRAM AND DISEASE SURVEILLANCE
A strong preventative health care program is critical to the success of SPF colonies. Such programs allow not only the tracking of target SPF agents but also the control of other potential primate pathogens that affect colony health. Components of a preventative program should include quarterly routine physical examination of all animals. At that time, intradermal tuberculin testing and phlebotomy for viral testing can be performed. Serum and, optimally, DNA should be obtained and stored. Vaccinations based on risk assessment for measles, rabies, and tetanus may be administered. Standard operating procedures for these procedures should be developed and followed. Individual animal medical records should be maintained.
A rigorous diagnostic program for ill or sick animals should be developed to help in the identification of potential disease outbreaks that may affect colony health. A production-animal herd-health approach should govern the decision making process. Disease surveillance is an important component of a preventative health program. All animals that have died or are euthanized for poor health should be necropsied, and tissue should be examined histologically. Results should be entered in a computerized database to allow tracking of disease trends. Necropsies allow not only identification of cause of death in individual animals but also surveillance of the colony and identification of subclinical disease.
Strict separation of SPF colony animals from conventional colony animals, indigenous primates, and other wild or feral populations must also be maintained. Indigenous primates may represent a potential exogenous source of the SPF target agents. Wild or feral populations such as rodents and dogs may transmit such important pathogens as leptospirosis, encephalomyocarditis virus, or rabies virus. Once colonies are established, introduction of animals from other sources should be discouraged. If required to maintain genetic diversity, extended quarantines and repeated testing for a period of at least 1 year is recommended.
EXPANDED SPF PROGRAMS
When the NENPRC SPF colony was first established, four viruses were targeted for elimination: B virus, STLV-1, SRV-D, and SIV. Since then, additional viral agents have been recognized as causing widespread infection in most rhesus macaque colonies. With refinement of the SIV AIDS model, investigators have requested animals free of additional agents to study the impact of concurrent infections on disease pathogenesis and to investigate novel antigen delivery systems. Particular colonies
free of SV40 and SFV may be required for some experimental protocols and would represent a valuable SPF asset. Furthermore, in some cases (e.g., simian foamy viruses), these agents may pose a newly recognized zoonotic risk to personnel. In response to such requests, the NENPRC has rederived animals from the SPF colony by cesarean section. These animals were hand reared in isolation from the remainder of the colony and tested for the presence of additional viral and nonviral pathogens. Quarterly testing by ELISA or PCR has revealed that animals are free of the following agents:
-
Simian virus 40 (SV40), based on ELISA;
-
Lymphocryptovirus (LCV), based on PCR/peptide ELISA
-
Rhesus rhadinovirus (RRV), based on ELISA
-
Cytomegalovirus (CMV), based on ELISA and PCR
-
Simian foamy virus (SFV), based on ELISA
To perform this testing, additional ELISA procedures were developed using purified virus for SV40, RRV, and CMV as previously described (Desrosiers and others 1997; Kaur and others 1996). Testing for LCV was initially performed by PCR on DNA isolated from peripheral blood mononuclear cells as previously described. A peptide ELISA for antibody testing has also been developed utilizing two peptides representing the carboxyterminal domains of the rhesus LCV VCAp18 (Wang 2001).
SUMMARY AND RECOMMENDATIONS
-
Consider minimum SPF target viruses: B virus, SIV, STLV, and SRV-D. Consideration should also be given to the inclusion of other agents such as RRV, CMV, LCV, SV40, and SFV based on perceived research requirements and seroprevalence in the source colony.
-
Select on-site serological testing capabilities/off-site confirmational testing. For cost effectiveness and rapid responsiveness on-site serologic testing capabilities may be warranted. Colonies should have access to reference laboratories for off-site confirmational testing.
-
Perform initial serological surveys. Serological surveys should be performed during the SPF planning process to assess the prevalence of the target agents and to define other agents that may or may not be present in the colony.
-
Select candidate SPF animals at 6 to 12 months of age. Most macaques will seroconvert to the four standard target SPF viruses between 1 and 3 years of age. Initial testing of animals at 6 to 12 months will increase the seronegative selection pool.
-
House animals initially in small peer groups and test quarterly for target viruses for 2 years. After initial tests, seronegative animals should be formed into small peer groups of 2 to 4 animals, and testing should continue quarterly until negative for 2 years. Animals that seroconvert and contacts should be removed from the colony.
-
Establish breeding harems at 3 to 3 1/2 years of age and begin testing for retroviruses annually and BV quarterly. There are advantages and disadvantages to the various possible SPF breeding configurations. Breeding harems provide the best opportunity at pathogen containment early in SPF colony development.
-
Separate SPF colonies from conventional and indigenous primates. Strict separation of SPF colonies from conventional and indigenous primates as well as feral and wild animal populations should be maintained. Such primates represent a potential source of breaks in SPF status.
-
Establish a vigorous preventative health care and diagnostic program. This program should include quarterly physical examinations, blood and DNA banking, viral screens, vaccinations, and individual animal medical records.
-
Maintain genetic diversity. SPF animals should be managed to preserve genetic diversity and maintained as out-bred colonies. Detailed pedigrees and breeding records should be kept and the use of software for the management of genetic, pedigree and demographic data considered.
REFERENCES
Baskin G. B., M. Murphey-Corb, E. A. Watson, and L. N. Martin. 1988. Necropsy findings in rhesus monkeys experimentally infected with cultured simian immunodeficiency virus (SIV)/delta. Vet Pathol 25:456-467.
CDC [Centers for Disease Control and Prevention]. 1987. Guidelines for prevention of Herpesvirus simiae (B virus) infection in monkey handlers. MMWR 36:680-689.
Daniel M. D., N. L. Letvin, P. K. Sehgal, D. K. Schmidt, D. P. Silva, K. R. Solomon, F. S. Hodi, Jr., D. J. Ringler, R. D. Hunt, N. W. King, and R. C. Desrosiers. 1988. Prevalence of antibodies to 3 retroviruses in a captive colony of macaque monkeys. Int J Cancer 41:601-608.
Desrosiers, R. C., V. G. Sasseville, S. C. Czajak, X. Zhang, K. G. Mansfield, A. Kaur, R. P. Johnson, A. A. Lackner, and J. U. Jung. 1997. A herpesvirus of rhesus monkeys related to the human Kaposi’s sarcoma-associated herpesvirus. J Virol 71:9764-9769.
Franchini, G. and M. S. Reitz. 1994. Phylogenesis and genetic complexity of nonhuman primate retroviridae. AIDS Res Hum Retroviruses 10:1047-1060.
Freifeld, A. G., J. Hilliard, J. Southers, M. Murray, B. Savarese, J. M. Schmitt, and S. E. Strauss. 1995. A controlled seroprevalencee survey of primate handlers for evidence of asymptomatic herpes B virus infection. J Infect Dis 171:1031-1034.
Gardner, M. B., M. Endres, and P. Barry. 1994. The simian retroviruses: SIV and SRV, p. 133-276. In J. A. Levy (ed.), The retroviridae volume 3. New York: Plenum Press.
Holmes, G. B., L. E. Chapman, J. A. Stewart, J. A. Straus, S. E. Straus, J. K. Hilliard, and D. S. Davenport. 1995. Guidelines for the prevention and treatment of B-virus infections in exposed persons. Clin Infect Dis 20:421-439.
Homma, T., P. J. Kanki, R. D. Hunt, M. J. O’Connell, N. L. Letvin, R. C. Desrosiers, R. C. Yang, and M. Essex. 1984. Lymphoma in macaques: association with the virus of human T lymphotropic family. Science 225:716-718.
Ishikawa, K. I., M. Fukasawa, H. Tsujimoto, J. G. Else, M. Isahakai, N. K. Ubhi, T. Ishida, O. Takenaka, Y. Kawamoto, Y. Spriatna, K. Abe, and K. Yamamoto. 1987. Serological survey and virus isolation of simian T-cell leukemia in their native countries. Int J Cancer 40:233-239.
Kaur, A., M. D. Daniel, D. Hempel, D. Lee-Parritz, M. S. Hirsch, and R. P. Johnson, 1996. Cytotoxic T-lymphocyte responses to cytomegalovirus in normal and SIV-infected rhesus macaques. J of Virol 70, 7725-7733.
Kessler, M. J. and J. K. Hilliard, 1990. Seroprevalence of B virus (Herpesvirus simiae) antibodies in a naturally formed group of rhesus macaques. J Med Primatol 19, 155-160.
Lackner, A. A. 1994. Pathology of simian immunodeficiency virus induced disease. Current Top Microbiol Immunol 199:35-64.
Lackner, A. A., P. F. Moore, P. A. Marx, R. J. Munn, M. B. Gardner, and L. J. Lowenstine. 1990. Immunohistochemical localization of type D retrovirus serotype 1 in the digestive tract of rhesus monkeys with simian AIDS. J Med Primatol 19:339-349.
Lerche, N. W., K. G. Osborn, P. A. Marx, S. Prahalda, D. H. Maul, L. J. Lowenstine, R. J. Munn, M. L. Bryant, R. V. Henrickson, L. O. Arthur, R. V. Gilden, C. S. Barker, E. Hunter, and M. B. Gardner. 1986. Inapparent carriers of simian AIDS type D retrovirus and disease transmission with saliva. J Natl Cancer Inst 77:489-496.
Lowenstine, L. J., N. W. Lerche, J. L. Yee, A. Uyeda, M. B. Jennings, R. J. Munn, H. M. McClure, D. C. Anderson, P. N. Fultz, and M. B. Gardner. 1992. Evidence for a lentiviral etiology in an epizootic of immune deficiency and lymphoma in stump-tailed macaques (Macaca arctoides). J Med Primatol 21:1-14.
Mansfield, K. G., Lerche, N. W., Gardner, M. B., and Lackner, A. A. 1995. Origins of simian immunodeficiency virus infection in macaques at the New England Regional Primate Research Center. J Med Primatol 24, 116-122.
Wang, F. 2001. A new animal model for Epstein-Barr virus pathogenesis. Curr Top Microbiol Immunol 258:201-219.
Ward, J. A. and J. K. Hilliard. 1994. B virus-specific pathogen-free (SPF) breeding colonies of macaques: issues, surveillance, and results in 1992. Lab Anim Sci 44:222-228.
Watanbe, T., M. Seiki, H. Tsujimoto, I. Miyoshi, M. Hayami, and M. Yoshida. 1985. Sequence homology of the simian retrovirus genome with human T-cell leukemia virus type 1. Virology 144:59-65.
Weigler, B. J., D. W. Hird, J. K. Hilliard, N. W. Lerche, J. A. Roberts, and L. M. Scott. 1993. Epidemiology of Cercopithecine herpesvirus 1 (B virus) infection and shedding in a large breeding cohort of rhesus macaques. J Infect Dis 167:257-263.
Weigler, B. J., J. A. Roberts, D. W. Hird, N. W. Lerche, and J. K. Hilliard. 1990. A cross sectional survey for B virus antibody in a colony of group housed rhesus macaques. Lab Anim Sci 40:257-261.
Session 5:
Panel Discussion
Participants:
Hilton Klein—Session Chair, Merck Research Laboratories, USA
Gary B. Baskin—Tulane National Primate Research Center, USA
Tom DeMarcus—CDC/National Center for Infectious Diseases, USA
Sherri L. Motzel—Merck Research Laboratories, USA
Keith Mansfield—New England National Primate Research Center, USA
QUESTIONS AND ANSWERS
PARTICIPANT A: Dr. Motzel, did you see anything interesting on the bronchial lymph nodes on those monkeys at post mortem examination and were they routinely stain-free acid-fast bacteria? If so, what did you see?
DR. MOTZEL (Sherri L. Motzel, Merck Research Laboratories): They were routinely stained for acid-fast bacteria. We also did polymerase chain reaction (PCR), and we had variable results. We saw acid-fast organisms in some cases, and we did see positive PCR on some. I think that of the tissues that were most predictive for recovering tuberculosis, they were probably in the top three along with lung and, in our case, the liver.
PARTICIPANT B: Mr. DeMarcus, do you know which species were the 18 DRAs on your list?
MR. DEMARCUS (National Center for Infectious Disease): I do not recall, but I can get that information for you. They were macaques.
PARTCIPANT B: Thank you. Do you perhaps know which origin or which country?
MR. DEMARCUS: I am sorry, I do not have that information with me.
DR. BAUDOIN (Mario Baudoin, Ministry of Sustainable Development and Planning): Dr. Mansfield, could you tell me the age of the female animal that was ADA negative until 1992, and then in 1995 was separated from the source colony.
DR. MANSFIELD (Keith Mansfield, New England National Primate Research Center): She was an adult animal when she was recruited to the colony, not a juvenile.
DR. BAUDOIN: Is this then probably the longest incubation time we have ever heard of herpes B?
DR. MANSFIELD: It is fairly long, but I think there are other cases of 6, 7, or 8 years after separation from the source colony.
I would like to add a quick comment: someone asked whether we treat our SPF for superclean animals any differently from the conventional animals, and we do not. We treat all animals as if they potentially harbor B virus and wear the same PPE. Part of that approach is to prevent the acquisition of B virus from an animal such as this. The other aspect is that these animals do carry a wide variety of other infectious agents that are potentially harmful to humans.
DR. BAUDOIN: Just out of curiosity, is it true that none of the other animals were in close proximity in your harems?
DR. MANSFIELD: None of the animals share a converted status; and as far as I am aware, no one has demonstrated that these animals that showed delayed seroconversion, if that is what it is, pose a risk to other animals in the colony—that they, in fact, shed virus.
DR. BAUDOIN: An additional matter of curiosity to me is whether we are certain this is herpes B and not something that cross-reacts with herpes?
DR. MANSFIELD: The sample was sent off to the National B-Virus Laboratory, and they distinguished HSB from herpes B. As reliable as that can be, it appears that whatever it was was closely related to herpes B.
PARTICIPANT C: Mr. DeMarcus, the importation information was very interesting, and I noted that the peak importation in the United States, 1988-1989, corresponded with the Ebola outbreak in Reston. Other than testing for the viruses, was there anything different about the quarantine procedures in 1988 versus what you are doing today?
MR. DEMARCUS: Certainly, most of the monitoring and surveillance that CDC was doing of primate quarantined facilities prior to the
Reston Ebola outbreak in 1989 and 1990 was passive. We maintained a registry. We required those registrations to be renewed every 2 years. We reviewed the information that was submitted in the registration applications. We routinely visited facilities, monitored their handling procedures, and reviewed their records. Beyond making additional recommendations for disease control, we required importers to document their compliance with those recommendations for continued registration. We monitored the handling of the animals on arrival at the airport, on arrival at the facility, and periodically; and we reviewed their compliance with the implementation of the standard operating procedures. So yes, it was dramatically different before and after.
PARTICIPANT C: As a follow-up question, has any one agency historically looked at the time period to try to trace back the origin of the infection in the Reston animals to where they might have been contaminated or whether they were contaminated at the source of origin?
MR. DEMARCUS: Yes, I think there are several papers in the literature, and there is also some special pathogen personnel still present at the CDC who could probably answer that question better than I can.
PARTICIPANT C: Dr. Mansfield, when you do SRV testing and on Western blot, your confirmation test, what do you do when you obtain an indeterminate, particularly on epitose 27s?
DR. MANSFIELD: We do all our confirmational testing at the National Retrovirus Laboratory, and we do have occasional animals that are in the SPF colony that show indeterminate tests on Western blots. When we follow those animals, often they revert to a negative test over a period of time. I believe those animals are negative because none of the animals’ contacts, have ever seroconverted to D virus in the past 12 years.
PARTICIPANT D: First, Mr. DeMarcus, I was somewhat surprised that the CDC does preshipment diagnostic testing given the many properties these monkeys have and the fact that the animal handlers at the airport may be exposed to these conditions. I am wondering whether the CDC revealed these requirements to actually make it compulsory for some sort of preshipment testing to try and minimize that danger of infection at the airport or for the animal handlers.
MR. DEMARCUS: I think that the question of preshipment testing is a complicated one. It would require standardization, oversight, and monitoring, which the CDC really does not have the resources to implement. We believe that if the recommended precautions regarding separation and isolation during transit and quarantine are implemented properly, that the risk during transit is very low and the screening done in quarantine is more reliable to make certain that the animals are at least free of disease before they are released from quarantine.
PARTICIPANT D: Dr. Motzel, with regard to the animals that were experimentally infected with Mycobacteria, did you attempt to treat them and if so, was the treatment regime successful?
DR. MOTZEL: We did not treat any of the animals.
DR. MCGREAL (Shirley McGreal, International Primate Protection League): Mr. DeMarcus, I am thinking of the reports in 1989 when they had facilities with 49 (between 13 and 49) deficiencies of monkeys in twotier cages urinating on top of those below, which was totally appalling. Do you think these kinds of conditions have stopped, and secondly, how can one justify allowing macaques into the pet trade? Anybody can answer this question because I think the CDC is probably the one agency that can stop it rather than trying to obtain restrictive state-by-state laws, because someone with a pet macaque will die, which we want to prevent.
MR. DEMARCUS: The conditions in primate quarantine facilities comply with the CDC recommendations that were implemented in 1989 and 1990 without exception. As I mentioned in my presentation with respect to specific disease, CDC’s regulatory mandate is aimed at those diseases that have the capability of causing serious outbreaks—person-to-person spread in humans. Although B virus is a potentially deadly disease in humans, it is very rare and rare indeed in its reported ability to be able to spread person-to-person. Again, CDC’s mandate is to prevent serious outbreak of human disease associated with nonhuman primates. CDC believes that the 31-day quarantine is adequate to do that. Certainly macaques do not make good pets. They present serious hazards to advocationists, but it is beyond the scope of CDC’s import quarantine mandate to attempt to regulate the possession and interstate distribution of nonhuman primates.
PARTICIPANT E: Dr. Mansfield, is anyone developing SPF colonies of Chinese rhesus?
DR. MANSFIELD: Not that I am aware of. All the OAR funded-breeding programs are Indian-origin rhesus macaques.
PARTICIPANT E: Do you think it would be worthwhile to do so?
DR. MANSFIELD: Yes, I think it would be worthwhile.
DR. ROBERTS (Jeffrey A. Roberts, University of California—Davis): I can respond to that question because we have a base grant SPF colony and a colony supported through the grant mechanism, and our base grant colony does include Chinese SPF animals in our stock. I have two questions, one very specific and one more general. Dr. Mansfield, in terms of the rabies vaccine, what was the product and what was the rationale for indoor-housed animals?
DR. MANSFIELD: To clarify, I am saying that the vaccination program needs to be based on risk assessment. So for an indoor-housed
animal in New England, rabies is relatively low on my differential list. However, certainly in other places in the world, rabies may be much more problematic. Then I think there should be a risk assessment study performed to determine whether that vaccine might be worthwhile to use. A variety of vaccines that have been used in nonhuman primates; however, the efficacy is unknown. I do not think anyone has done studies to show that they are efficacious.
DR. ROBERTS: The second question is for all of the pathologists on the panel. In this session, we have talked about diseases and about the importance of record keeping and allocation of resources. What do you do in terms of banking tissue samples; how long do you keep them? It is a tremendous information database, but it also takes up a lot of space and requires a lot of inventory. Is there any feedback you would like to give on your policies?
DR. BASKIN (Gary Baskin, Tulane National Primate Research Center): We keep blots and slides forever. We keep wet tissues until the case is signed out. On routine animals, animals that are on specific projects, we ask the investigator whether to store the wet tissues after the pathology work is complete. Our problem is storage capacity. We perform approximately 900 micropsies per year. You can imagine the storage problems with those kinds of samples, and we do not keep them.
DR. MANSFIELD: We have a smaller colony and probably do about 300 to 350 micropsies per year. We keep all paraffin-embedded, wet, and frozen tissue, at least as far back as 1988. All animals’ micropsy have both formal and fixed and frozen tissue on them, but it is becoming a space issue for us also.
DR. MOTZEL: We keep all blots and slides, but we routinely dispose of wet tissues after a period of a few years.
PARTICPANT F: I have been working with John Robertson at the Regional College of Veterinary Medicine at Virginia Tech to begin developing a tissue archive for primates. Hopefully it would be possible to develop a repository there, in a relatively low cost area, where additional slides, tissues, and specimens could be stored for any of you. Those of you who have just mentioned some of the problems with repositories eventually will probably be filled. This project is pursuant to our development of a great ape brain bank, which is now being expanded into a CNS tissue repository beyond great apes. I mention it because it has already been very productive, particularly in alliance with some of the human brain banks that are supported under the Alzheimer’s program.






































