Executive Summary
BACKGROUND FOR THE STUDY
Genetic engineering and other new technologies are among many advances made to traditional breeding practices in plants, animals, and microbes to enhance food quality and increase productivity. Genetic engineering, the targeted manipulation of genetic material, and nontargeted, nontransgenic methods—including chemical mutagenesis and breeding—are components of the entire range of genetic modification methods used to alter the genetic composition of plants, animals, and microorganisms. (For more comprehensive definitions of key terms used throughout this report, please see Appendix A: Glossary.)
In this report, genetic engineering refers only to recombinant deoxyribonucleic acid (rDNA) methods that allow a gene from any species to be inserted and subsequently expressed in a food crop or other food product. Although the process involving rDNA technology is not inherently hazardous, the products of this technology have the potential to be hazardous if inserted genes result in the production of hazardous substances.
Nongenetic engineering methods of genetic modification include embryo rescue, where plant or animal embryos produced from interspecies gene transfer, or crossing, are placed in a tissue culture environment to complete development. Other methods include somatic hybridization, in which the cell walls of a plant are removed and the “naked” cells are forced to hybridize, and induced mutagenesis, in which chemicals or irradiation are used to induce random mutations in DNA. The development of these approaches has enhanced the array of techniques that can be used to advance food production. However, as with all other technologies for genetic modification, they also carry the potential for introducing unintended compositional changes that may have adverse effects on human health.
Preventing adverse health effects by maintaining a safe food supply requires the application of appropriate scientific methods to problems of predicting and identifying unintended compositional changes that may result from genetic modification of plants, animals, and microbes intended for consumption as food. To address this need, the U.S. Department of Agriculture, the U.S. Department of Health and Human Services’ Food and Drug Administration, and the U.S. Environmental Protection Agency asked the National Academies to convene a committee of scientific experts to outline science-based approaches for assessing or predicting the unintended health effects of genetically engineered (GE) foods and to compare the potential for unintended effects with those of foods derived from other conventional genetic modification methods.
COMMITTEE CHARGE AND APPROACH
This report is intended to aid the sponsoring agencies in evaluating the scientific methods to assess the safety of GE foods before they are sold to the public. The task presented to the committee by the sponsors was to outline science-based approaches to assess or predict unintended health effects of GE foods in order to assist in their evaluation prior to commercialization. The committee was charged to focus on mechanisms by which unintended changes in the biochemical composition of food occur as a result of various conventional and genetic engineering breeding and propagation methods, the extent to which these mechanisms are likely to lead to significant compositional changes in foods that would not be readily apparent without new or enhanced detection methods, and methods to detect such changes in food in order to determine their potential human health effects. The committee was further charged to identify appropriate scientific questions and methods for determining unintended changes in the levels of endogenous nutrients, toxins, toxicants, allergens, or other compounds in food from genetically engineered organisms (GEOs) and outline methods to assess the potential short- and long-term human consequences of such changes.
The committee was charged to compare GE foods with foods derived from other genetic modification methods, such as cross breeding, with respect to the frequency of compositional changes resulting from the modification process and the frequency and severity of the effects of these changes on consumer health. As part of this comparison, the likelihood that elevated toxin or allergen levels would occur in domesticated animals or plants that are modified by different methods was to be considered. Based on this analysis, the committee was charged to discuss whether certain safety issues are specific to GE foods, and if so, recommend approaches for addressing these issues. In addition, the committee was to separately evaluate methods to detect potential unintended compositional changes and health effects of foods derived from cloned animals. The evaluation is presented in a short subreport, separate from, but designed to accompany, the committee’s full-length report on foods derived from genetic modification methods.
MECHANISMS BY WHICH UNINTENDED COMPOSITIONAL CHANGES IN FOOD OCCUR AS A RESULT OF BREEDING OR PROPAGATION METHOD
Conventional Breeding
The oldest approach to plant genetic modification is simple selection, where plants exhibiting desired characteristics are selected for continued propagation. Modern technology has improved upon simple selection with the use of molecular analysis to detect plants likely to express desired features. Plants that are selected for desired traits, such as reduced levels of chemicals that produce unpalatable taste, may diminish the ability of plants to survive in the wild because they are also more attractive to pests. Selection for other traits, such as chemicals that increase the resistance of plants to disease, may also be harmful to humans.
Another approach, crossing, can occur within a species or between different species. For example, the generation of triticale, a crop used for both human food and animal feed, arose from the interspecies crossing of wheat and rye. Because most crops can produce allergens, toxins, or antinutritional substances, conventional breeding methods have the potential to produce unintended compositional changes in a food crop.
Genetic Modification
Hazards associated with genetic modifications, specifically genetic engineering, do not fit into a simple dichotomy of genetic engineering versus nongenetic engineering breeding. Not only are many mechanisms common to both genetic engineering as a technique of genetic modification and conventional breeding, but also these techniques slightly overlap each other. Unintentional compositional changes in plants and animals are likely with all conventional and biotechnological breeding methods. The committee assessed the relative likelihood of compositional changes occurring from both genetic engineering and nongenetic engineering modification techniques and generated a continuum to express the potential for unintended compositional changes that reside in the specific products of the modification, regardless of whether the modification was intentional or not (Figure ES-1).
METHODS TO DETECT UNINTENDED CHANGES IN FOOD COMPOSITION
Important advances in analytical methodology for nucleic acids, proteins, and small molecules have occurred over the past decade as a result of concurrent advances in technology and instrumentation; however, there is a need for improvement in all of these areas.
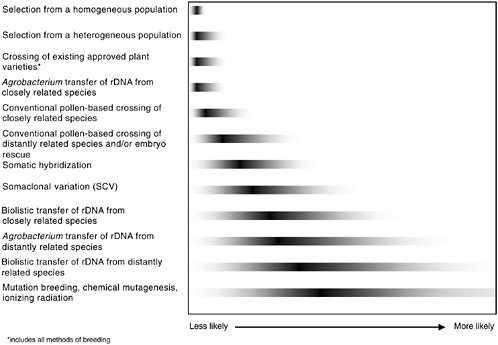
FIGURE ES-1 Relative likelihood of unintended genetic effects associated with various methods of plant genetic modification. The gray tails indicate the committee’s conclusions about the relative degree of the range of potential unintended changes; the dark bars indicate the relative degree of genetic disruption for each method. It is unlikely that all methods of either genetic engineering, genetic modification, or conventional breeding will have equal probability of resulting in unintended changes. Therefore, it is the final product of a given modification, rather than the modification method or process, that is more likely to result in an unintended adverse effect. For example, of the methods shown, a selection from a homogenous population is least likely to express unintended effects, and the range of those that do appear is quite limited. In contrast, induced mutagenesis is the most genetically disruptive and, consequently, most likely to display unintended effects from the widest potential range of phenotypic effects.
Currently, there are two basic analytical approaches available to detect compositional changes in food. Targeted quantitative analysis is the traditional approach in which a method is established to quantify a predefined compound or class of compounds. In contrast, profiling methods involve the untargeted analysis of a complex mixture of compounds extracted from a biological sample with the objective of identifying and quantifying all compounds present in a sample. Advanced chemical and genetic profiling techniques—using molecular genetic, proteomic (analysis of complete complements of proteins), and metabolomic (global analysis of nonpeptide small molecules) approaches—are rapidly developing to produce technologies with the potential to provide an enormous amount of data for a given organism, tissue, or food product.
Despite these technological advances in analytical chemistry, our ability to interpret the consequences to human health of changes in food composition is limited. Compositional changes can be readily detected in food and the power of profiling methodologies is rapidly increasing our ability to demonstrate compositional differences among foods. The complexity of food composition challenges the ability of modern analytical chemistry and bioinformatics to chemically identify and determine the biological relevance of the many compositional changes that occur.
METHODS TO ASSESS THE POTENTIAL HUMAN CONSEQUENCES OF UNINTENDED COMPOSITIONAL CHANGES IN FOOD
The major challenges to predicting and assessing unintended adverse health effects of genetically modified (GM) foods—including those that are genetically engineered—are underscored by the severe imbalances between highly advanced analytical technologies and limited abilities to interpret their results and predict health effects that result from the consumption of food that is genetically modified, either by traditional or more modern technologies. The present state of knowledge requires that approaches for assessing the occurrence and significance
of unintended health effects encompass both targeted and profiling approaches, using a range of toxicological, metabolic, and epidemiological sciences. Encom-passing both of these approaches exploits what is known and increases the ability to prevent and assess unsuspected consequences.
Current safety assessments in the premarket period prior to commercialization focus on comparing the GE food with its conventional counterpart to identify uniquely different components. Typically, these comparisons are made on the basis of proximate analysis—an analytical determinant of major classes of food components—as well as nutritional components, toxins, toxicants, antinutrients, and any other characterizing components. The ideal comparator, in most cases, is a near-isogenic variety of food, genetically identical except for the presence of the novel trait, or a near-isogenic parental variety of food from which the GE variety was derived.
In addition to compositional comparisons, agronomic comparisons have been routinely conducted as part of the line selection phase in the development of GE crops. However, these comparisons of phenotypic expression tend to be superficial and could easily miss some varieties containing altered compositions that could impact adversely on human health.
Animal feeding trials are also used to compare the nutritional qualities of a GE crop with its conventional counterpart. Any adverse effects on the health of the animals indicate the possible existence of unexpected alterations in the GE crop that could adversely affect human health, if consumed.
Postmarketing surveillance is an approach to verify premarket screening for unanticipated adverse health consequences from the consumption of GE food. Although postmarketing surveillance has not been used to evaluate any of the GE crops that are currently on the market and there are challenges to its use, this approach holds promise in monitoring potential effects, anticipated and unanticipated, of GE foods that are not substantially equivalent to their conventional counterparts or that contain significantly altered nutritional and compositional profiles.
FRAMEWORK FOR IDENTIFYING AND ASSESSING UNINTENDED ADVERSE EFFECTS FROM GENETICALLY MODIFIED FOODS
The committee developed a framework for a model system based on methods to identify appropriate comparators; increase the knowledge of the determinants of compositional variability; increase the understanding of the biological effects of secondary metabolites in foods; develop more sensitive tools for assessing potential unintended effects from complex mixtures; and improve methods for tracing exposure to GM foods.
The framework, illustrated in a flowchart (Figure ES-2), was used to examine, identify, and evaluate systematically the unintended compositional changes and health effects of GM and, specifically, GE foods. By raising the appropriate questions in this systematic flowchart, the committee has provided a guide for
overall decision-making, providing alternative routes that can and should be taken according to the specific GM target. Further, the flow chart illustrates the need for appropriate tools to assess and utilize both pre- and postmarket approaches in the process of identifying unintended compositional changes and potential unintended adverse health effects. This model system for selecting and validating methods to detect and assess compositional changes in food serves as the basis for the committee’s recommendations to overcome limitations to current methods used to identify compositional differences and evaluate the health significance of new or altered compounds in GM foods.
Overall Findings and Recommendation
Findings
All new crop varieties, animal breeds (see the cloning subreport), and microbial strains carry modified DNA that differs from parental strains. Methods to genetically modify plants, animals, and microbes are mechanistically diverse and include both natural and human-mediated activities. Health outcomes could be associated with the presence or absence of specific substances added or deleted using genetic modification techniques, including genetic engineering, and with unintended compositional changes.
The likelihood that an unintended compositional change will occur can be placed on a continuum that is based on the method of genetic modification used (see Figure ES-1). The genetic modification method used, however, should not be the sole criterion for suspecting and subsequently evaluating possible health effects associated with unintended compositional changes.
All evidence evaluated to date indicates that unexpected and unintended compositional changes arise with all forms of genetic modification, including genetic engineering. Whether such compositional changes result in unintended health effects is dependent upon the nature of the substances altered and the biological consequences of the compounds. To date, no adverse health effects attributed to genetic engineering have been documented in the human population.
Recommendation 1
The committee recommends that compositional changes that result from all genetic modification in food, including genetic engineering, undergo an appropriate safety assessment. The extent of an appropriate safety assessment should be determined prior to commercialization. It should be based on the presence of novel compounds or substantial changes in the levels of naturally occurring substances, such as nutrients that are above or below the normal range for that species (see Chapter 3), taking into account the organism modified and the nature of the introduced trait.
Safety Assessment Tools for Assessing Unintended Effects Prior to Commercialization
Findings
Current voluntary and mandated safety assessment approaches focus primarily on intended and predictable effects of novel components of GE foods. Introduction of novel components into food through genetic engineering can pose unique problems in the selection of suitable comparators for the analytical procedures that are crucial to the identification of unintended compositional changes. Other jurisdictions, particularly the European Union, evaluate all GE food products prior to commercialization, but exempt from similar evaluation all other GM foods. As is discussed in Chapter 3, the policy to assess products based exclusively on their method of breeding is scientifically unjustified.
The most appropriate time for safety assessment of all new food is in the premarket period prior to commercialization, although verification of safety assessments may continue in the postmarket period, generally in cases when a potential problem has been identified or if there is elevated cause for concern. Examples of specific premarket assessments of newly introduced compositional changes to selected GE food are:
-
protein, fat, carbohydrate, fiber, ash, and water in a proximate analysis;
-
essential macro- and micronutrients in a nutritional analysis;
-
known endogenous toxicants and antinutrients in specific species;
-
endogenous allergens;
-
other naturally occurring, species-specific constituents of potential interest, such as isoflavones and phytoestrogens in soybean or alkaloids in tomato or potato;
-
gross agronomic characteristics;
-
data derived from domestic animal feeding trials to assess the nutritional quality of new crops; and
-
data derived from toxicological studies in animals.
Recommendation 2
The committee recommends that the appropriate federal agencies determine if evaluation of new GM foods for potential adverse health effects from both intended and unintended compositional changes is warranted by elevated concern, such as identification of a novel substance or levels of a naturally occurring substance that exceeds the range of recommended or tolerable intake.
Recommendation 3
For those foods warranting further evaluation, the committee recommends that a safety assessment should be conducted prior to commercialization and continued evaluation postmarket where safety concerns are present. Specifically, the committee recommends the following safety assessment actions.
-
Develop a paradigm for identifying appropriate comparators for GE food.
-
Collect and make publicly available key compositional information on essential nutrients, known toxicants, antinutrients, and allergens of commonly consumed varieties of food (see the Research Needs section, later in this chapter). These should include mean values and ranges that typically occur as a function of genetic makeup, differences in physiological state, and environmental variables.
-
Remove compositional information on GE foods from proprietary domains to improve public accessibility.
-
Continue appropriate safety assessments after commercialization to verify premarket evaluations, particularly if the novelty of the introduced substance or the level of a naturally occurring substance leads to increased safety concerns.
Analytical Methodologies
Findings
During the past decade, analytical methodologies for separating and quantifying messenger ribonucleic acids, proteins, and metabolites have improved markedly. Applying these methodologies to the targeted analysis of known nutrients and toxicants will improve the knowledge base for these food constituents. The broad application of targeted methods and continuing development of profiling methods will provide extensive information about food composition and further improve the knowledge base of defined chemical food constituents. The knowledge and understanding needed to relate such compositional information to potential unintended health effects is far from complete, however. Furthermore, currently available bioinformatics and predictive tools are inadequate for correlating compositional analyses with biological effects.
Analytical profiling techniques are appropriate for establishing compositional differences among genotypes, but they must also take into account modification of the profile obtained due to genotype-by-environmental interactions (the influence of the environment on expression of a particular genotype). The knowledge base required to interpret results of profiling methods, however, is insufficiently developed to predict or directly assess potential health effects associated with unintended compositional changes of GM food, as is the necessary associative information (e.g., proteomics, metabolomics, and signaling networks). Additionally, predictive tools to identify the expected behavior of complex and compound
structures are limited and require a priori knowledge of their chemical structure, their biological relevance, and their potential interactive targets.
Recommendation 4
The committee recommends the development and employment of standardized sampling methodologies, validation procedures, and performance-based techniques for targeted analyses and profiling of GM food performed in the manner outlined in the flow chart shown in Figure 7-1. Sampling methodology should include suitable comparisons to the near isogenic parental variety of a species, grown under a variety of environmental conditions, as well as ongoing assessment of commonly consumed commercial varieties of food. These include:
-
Reevaluation of current methodologies used to detect and assess the biological consequences of unintended changes in GM food, including better tools for toxicity assessment and a more robust knowledge base for determining which novel or increased naturally occurring components of food have a health impact.
-
Use of data collection programs, such as the Continuing Survey of Food Intakes by Individuals and the National Health and Nutrition Examination Survey (NHANES), to collect information, prior to commercial release of a new GM food, on current food and nutrient intakes and exposure to known toxins or toxicants through food consumption. The information collected should be used to identify food consumption patterns in the general population and susceptible population subgroups that indicate a potential for adverse reactions to novel substances or increased levels of naturally occurring compounds in GM food.
Additional Tools for Postcommercialization: Identification and Assessment of Unintended Effects
Findings
Postcommercialization or postmarket evaluation tools for verifying and validating premarket assessments of novel substances in food or detectable changes in diet composition, including tracking and epidemiological studies, are important components of the overall assessment of food safety. These tools provide a way to check the efficacy of premarket compositional and safety evaluations through a feedback process. In addition, information databases that result from postmarket studies can be valuable assets in the development of future premarket safety assessment tools.
Postmarket surveillance is a commonly accepted procedure, for example, with new pharmaceuticals and has been beneficial in the identification of harmful and unexpected side effects. As a result, pharmacologists accept postmarket surveillance as a part of the process to identify unexpected adverse outcomes from
their products. This example is especially pertinent to GE foods because of the unique ability of this process to introduce gene sequences to generate novel products into organisms intended for use as food and especially in situations where the novel products are introduced at levels that have the potential to alter dietary intake patterns (e.g., elevated levels of key nutrients).
Given the possibility that food with unintended changes may enter the marketplace despite premarket safety mechanisms, postmarket surveillance of exposures and effects is needed to validate premarket evaluations. On the other hand, there are many instances in which postmarket surveillance may not be warranted. For example, when compositional comparisons of a new GM crop or food (e.g., Roundup Ready soybeans) with its conventional counterpart indicate they are compositionally very similar; exposure to novel components remains very low. Thus the process of identifying unintended compositional changes in food is best served by combining premarket testing with postmarket surveillance, when compositional changes indicate that it is warranted, in a feedback loop that follows a new GM food or food product long term, from development through utilization (see Figure ES-2).
Recommendation 5
When warranted by changes such as altered levels of naturally occurring components above those found in the product’s unmodified counterpart, population-specific vulnerabilities, or unexplained clusters of adverse health effects, the committee recommends improving the tracking of potential health consequences from commercially available foods that are genetically modified, including those that are genetically engineered, by actions such as the following:
-
Improve the ability to identify populations that are susceptible to food allergens and develop databases relevant to tracking the prevalence of food allergies and intolerances in the general population, and in susceptible population subgroups.
-
Improve and include other postmarket resources for identifying and tracking unpredicted and unintended health effects from GM foods:
-
Improve the sensitivity of surveys and other analytical methodologies currently used to detect consumer trends in the purchase and use of GM foods after release into the marketplace.
-
Standardize methods for monitoring reports of allergenicity to new foods introduced into the marketplace and apply them to new GM foods.
-
Assure that current food labeling includes relevant nutritional attributes so that consumers can receive more complete information about the nutritional components in GM foods introduced into the marketplace.
-
Improve utilization of potential traceability technology, such as bar coding of animal carcasses and other relevant foods.
-
-
Develop a database of unique genetic sequences (DNA, polymerase chain reaction sequences) from GE foods entering the marketplace to enable their identification in post-market surveillance activities.
-
Utilize existing nationwide food intake and health assessment surveys, including NHANES, to:
-
Collect comparative information on diet and consumption patterns of the general population and ethnic subgroups in order to account for anthropological differences among population groups and geographic areas where GM foods may be consumed in skewed quantities, recognizing that this will be possible only under selected circumstances where intakes are not evenly distributed across population subgroups of interest and the relevant outcome data are available.
-
Provide better representation of the long-term nutritional and other health status information on a full range of children and ethnic groups whose intakes may differ significantly from those of the general population to determine whether changes in health status have occurred as a consequence of consuming novel substances or increased levels of naturally occurring compounds in GM foods released into the marketplace, recognizing again that this will be possible only under selected circumstances that allow one to assess associations between skewed eating patterns and specified health outcomes. Such associations would have to be followed up by other more controlled assessments.
-
Research Needs
Findings
There is a need, in the committee’s judgment, for a broad research and technology development agenda to improve methods for predicting, identifying, and assessing unintended health effects from the genetic modification of food. An additional benefit is that the tools and techniques developed can also be applied to safety assessment and monitoring of foods produced by all methods of genetic modification.
The tools and techniques already developed can be applied to the safety assessment and monitoring of foods produced by all methods of genetic modification. However, although current analytical methods can provide a detailed assessment of food composition, limitations exist in identifying specific differences in composition and interpreting their biological significance.
Recommendation 6
A significant research effort should be made to support analytical methods technology, bioinformatics, and epidemiology and dietary survey tools to detect
health changes in the population that could result from genetic modification and, specifically, genetic engineering of food. Specific recommendations to achieve this goal include:
-
Focusing research efforts on improving analytical methodology in the study of food composition to improve nutrient content databases and increase understanding of the relationships among chemical components in foods and their relevance to the safety of the food.
-
Conducting research to provide new information on chemical identification and metabolic profiles of new GM foods and proteomic profiles on individual compounds and complex mixtures in major food crops and use that information to develop and maintain publicly accessible databases.
-
Developing or expanding profiling databases for plants, animals, and microorganisms that are organized by genotype, maturity, growth history, and other relevant environmental variables to improve identification and enhance traceability of GMOs.
-
Developing improved bioinformatics tools to aid in the interpretation of food composition data derived from targeting and profiling methods.
Recommendation 7
Research also is needed to determine the relevance to human health of dietary constituents that arise from or are altered by genetic modification. This effort should include:
-
Focusing research efforts on developing new tools that can be used to assess potential unintended adverse health effects that result from genetic modification of foods. Such tools should include profiling techniques that relate metabolic components in food with altered gene expression in relevant animal models to specific adverse outcomes identified in GM animal models (animals genetically modified by contemporary biotechnology methods that are proposed to enter the food system).
-
Developing improved DNA-based immunological and biochemical tags for selected GM foods entering the marketplace that could be used as surrogate markers to rapidly identify the presence and relative level of specific foods for postmarket surveillance activities.
-
Developing improved techniques that enable toxicological evaluations of whole foods and complex mixtures, including:
-
microarray analysis,
-
proteomics, and
-
metabolomics.
-
CONCLUSION
In response to its charge, the committee has developed a framework to identify appropriate scientific questions and methods for determining unintended changes in the levels of nutrients, toxins, toxicants, allergens, or other compounds in foods from GMOs, in order to assess potential short- and long-term human health consequences of such changes. Although the array of analytical and epidemiological techniques available has increased, there remain sizeable gaps in our ability to identify compositional changes that result from genetic modification of organisms intended for food; to determine the biological relevance of such changes to human health; and to devise appropriate scientific methods to predict and assess unintended adverse effects on human health. The committee has identified and recommended pre- and postmarket approaches to guide assessment of unintended compositional changes that could result from genetic modification of foods and research avenues to fill the knowledge gaps.
The recommendations presented in this report reflect the committee’s application of its framework to questions of identification and assessment of unintended adverse health effects from foods produced by all forms of genetic modification, including genetic engineering, and they can serve as a guide for evaluation of future technologies.

















