E
Fuels Research
The heat sink capacity of a fuel—hydrogen, for example—is due to its sensible heat (i.e., cp ΔT) and is thus proportional to the maximum temperature the fuel can achieve and the magnitude of its specific heat. Hydrogen has a much higher specific heat than hydrocarbon fuels, and assuming that ΔT is the same, is considered to have a higher heat capacity than hydrocarbon fuels; however, less hydrogen mass is required to achieve a given amount of heat release, so the hydrogen fuel flow rate is lower than that of a hydrocarbon. A more relevant measure of heat sink potential is the ratio of specific heat to the heat of combustion, which brings hydrocarbon fuels more in line with hydrogen in terms of heat capacity (within a factor of 2 or 3). Thus, it is not so much the specific heat of hydrogen that gives it better heat capacity than hydrocarbon fuels but rather its ability to sustain higher temperatures and thus increase the ΔT part of its heat capacity. In the absence of other considerations, conventional hydrocarbon fuels cannot be driven to as high a temperature as hydrogen, which is capable of sustaining temperatures up to the operating temperature of the engine.
In the case of an endothermic fuel like JP-7, another source of heat capacity is made available by cracking the fuel, a process of breaking its long-chain hydrocarbon molecules into lighter molecules that absorb heat (an endothermic process). In an endothermic fuel, the heat sink capability of the fuel is made up of its sensible heat plus any net endothermic capacity derived from high fuel dissociation reactions. The key is to find a fuel that has endothermic capability without degrading its exothermic capability. Typically, hydrocarbon decomposition processes are accompanied by carbon formation, or coking. Coking tends to foul heat transfer surfaces, which is undesirable. Thus, there are two parts to calculating the upper limit of a hydrocarbon fuel’s heat sink capability: the maximum temperature achievable without the system coking up, and the endothermic capacity of the cracking reactions that can occur.
Current U.S. state-of-the-art heat sink capability for an endothermic liquid hydrocarbon fuel under realistic conditions is roughly 1,500 Btu/lb, with a limit of 1300°F (Huang et al., 2002). Under operational conditions, a 1300°F temperature limit allows for a coking-limited lifetime of approximately 15 min. In this case, the total fuel heat sink capacity is a combination of 500 Btu/lb from endothermic reactions and roughly 1,000 Btu/lb from sensible heat. If the coking limit of a fuel could be raised by a few hundred degrees, its heat sink capability could be enhanced.
It is important to point out that the endothermic heat capacity of 500 Btu/lb is much less than the theoretical heat capacity achievable if the most desirable fuel reaction products could be obtained. Cracking JP-7 to 100 percent ethylene would absorb 1,500 Btu/lb, versus the 500 Btu/lb obtained from the product mix of methane, ethylene, ethane, etc. Thus, there is clearly room for increasing the endothermic capability of even our present candidate fuels. Because of the endothermic capability of our present hydrocarbon fuels, at the upper Mach number range for hypersonic air-breathing flight (approximately Mach >8), hydrogen is the fuel of choice; however, because of hydrogen’s difficulty of storage and its limited volumetric energy density, hydrocarbon fuels are more practical for lower Mach number applications. All of the hydrocarbon-based vehicles presently being contemplated anticipate using JP-7. As previously discussed, a robust fuels research and development (R&D) program could lead to higher Mach number limits for hydrocarbon fuels. The Russian AJAX (concept) vehicle, for example, increases heat sink capability by an interesting variation on conventional endothermic fuels. In the AJAX concept, water is added to the fuel to achieve steam reforming. In essence, steam reforming is fuel + water → CO + H2. This reaction absorbs (theoretically) 2,400 Btu/lb (versus 1,500 Btu/lb for cracking to ethylene). This is how the Russians theoretically obtain Mach 10 capability for AJAX, although the concept suffers reduced range owing to water consumption in the propulsion cycle.
GENESIS OF THE HYTECH/X-43C JP-7 FUEL “SYSTEM”
In the 1960s and early 1970s, the Air Force funded a considerable effort at Shell Research to develop endothermic methylcyclohexane (MCH), based on studies that identified the heat sink required for hydrocarbon-fueled hypersonic vehicles (Churchill et al., 1965; Lander and Nixon, 1971). Endothermic MCH required a supported platinum catalyst for dehydrogenation (to toluene and hydrogen) that was developed in pellet form (as is used industrially in fluidized beds for petroleum processing). After a long period of dormancy in the 1970s and 1980s, the work was restarted by Allied Signal (now Honeywell) and culminated in an expendable turbine-engine test, where the fuel was used to cool a hot air stream and then burned in an engine (Lipinski et al., 1992). There were two drawbacks to this technology: (1) regenerative cooling is best accomplished through a wall-mounted catalyst rather than pellets and (2) MCH is a relatively expensive specialty fuel.
Extensive discussions inside the Air Force Propulsion Directorate in the mid-1980s led to an effort to develop the endothermic potential of thermally/catalytically cracked liquid hydrocarbons using commercially available, wall-mounted zeolite catalysts (Spadaccini et al., 1993a). This task at the United Technologies Research Center was funded under program element 62203F/Project 3048 of the initial contract, F33615-87-C-2744, managed by Charlotte Eigel. This effort first reported the endothermic potential of JP-7 and JP-10, as well as JP-8 (Sobel and Spadaccini, 1994). The resulting endothermic hydrocarbon fuel capability contributed to selecting such fuels for the HyTech engine development program, which began in the mid-1990s (Spadaccini et al., 1993b). This separate (from HyTech) but coordinated fuel development effort has continued sporadically since HyTech funding became available. Recent tasks include extending the endothermic heat sink database to higher flow rate conditions and to other fuels (e.g., RP-1), as well as looking at relative combustion performance of the various alternative fuels (Huang and Sobel, 2002). The fuels research effort has also looked at the applicability of endothermic fuels to reusable aircraft and other applications (Lehrach et al., 1995).
A POSSIBLE FUELS RESEARCH PROGRAM
To provide some structure to a program that might be created by NAI, the committee consulted with Tim Edwards, a civilian fuels scientist at AFRL. Fuel is becoming the integrating factor of the
complete hypersonic vehicle system.1 It is difficult to give a reasonable estimate for a long-term endothermic fuels plan without bounding the problem, in that key drivers and constraints for fuel and fuel system development come from other nominally distinct disciplines—thermal management, combustion, and ground handling, to name a few. Thus, the fuel ultimately selected for a given hypersonic vehicle will probably be a compromise between cost, operability, and performance in a number of areas. NASA’s vision vehicle for air-breathing space access is fueled by liquid hydrogen, which has unmatched combustion performance and heat sink capability. Yet, liquid hydrogen’s low density (1/11th that of hydrocarbons) and high handling cost for maintaining it at approximately 20 K leave plenty of room for alternative fuels for many applications that are incompatible with large (hydrogen) vehicles and a “hard cryogen” infrastructure. A good, long-term fuels program would need to be coordinated with vehicle development under the umbrella of a national program such as NAI, since the fuel selection is driven by vehicle mission and operational envelope and would benefit from coordination with the development of rocket propellants. The three main vehicle categories—expendables (missiles), accelerators (launch vehicles), and cruise—place quite different constraints on the fuel. The program discussed below is an attempt to present programs that support hypersonics in general rather than a specific application.
As a baseline, assume that both hydrogen and hydrocarbon flight test vehicles have accomplished successful flight demonstrations. The engineering required to convert the hydrocarbon flight test vehicle to a successful standoff weapon for attacking time-critical targets is considered to be outside the scope of this research proposal. The state of the art appears to be Mach 6-7 for this type of expendable application and current endothermic fuel technology. This is not to minimize the difficulty and expense of successfully completing these flight tests—far from it! It is just a recognition that these vehicles are too far along to benefit much from fuel research at this point. This plan also does not include any further liquid hydrogen development, such as densification, gelation, and the like.
For the proposed endothermic fuel research, efforts are described that culminate in demonstrations, but also included is the development of modeling and simulation rules and tools for the effective use of the improved technology. A notional timeline is shown in Figure E-1 and discussed below.
-
Improved heat sink and coke reduction for conventional endothermic fuels. This task would address the development of improved catalysts, additives, and coke-resistant designs for the noctane/JP-7 kerosene class of hydrocarbons. (Rough order of magnitude [ROM] $500,000 per year for 5 years, with technologies demonstrated in long-duration panel tests in a radiant-heated AFRL facility)
-
Robust supersonic ignition and combustion of hydrocarbon fuels. This task would continue ongoing efforts to improve the combustion behavior of endothermic fuels, especially at the lower Mach numbers (including ignition). “Barbotage” fuel injection, fuel additives, ignition schemes, and high-performance piloting are options to improve endothermic fuel combustion and ignition performance. This is an expensive effort since it requires significant combustor testing under realistic conditions. (ROM $1 million per year for 7 years, culminating in a freejet test of an improved cold start/piloting system)
-
Improved endothermic hydrocarbon physical properties. It turns out that the physical properties of kerosene fuels are not well understood under high-temperature and high-pressure conditions. This work would be an extension of ongoing NIST work for NASA on RP-1. (ROM $200,000
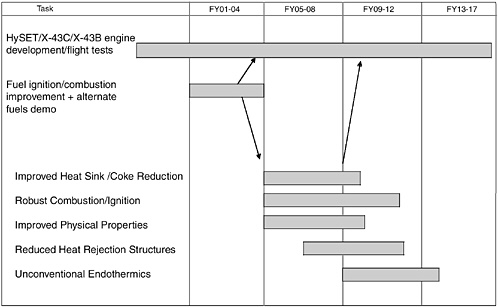
FIGURE E-1 Proposed endothermic fuel research timeline. SOURCE: Adapted from information provided by James T. Edwards, AFRL.
-
per year for 5 years, with the result being better physical property prediction tools for design and development)
-
Development of reduced-heat-rejection structures. To extend the Mach number capability of nonhydrogen-fueled hypersonic vehicles, one has to either improve the fuel heat sink capability or reduce the heat load from the vehicle into the fuel. The use of high-temperature ceramic engine structures appears to be capable of significantly reducing the required engine cooling (since the heat load into the fuel is proportional to the Tcombustion less the Tsurface driving force). Two key issues need to be addressed: (1) the structural manufacturability and durability of ceramic structures and (2) exposure of fuel to high-temperature surfaces and the impact on coking. (ROM $750,000 per year for 5 years, culminating in a direct-connect demonstration of a ceramic combustor)
-
Evaluation of unconventional endothermic fuels. This task would continue the development of steam reforming for increased heat sink (following up on current Russian work), as well as investigate other approaches to obtaining dramatically higher heat sinks than conventional endothermic fuels. For example, methanol reforming has been suggested as a promising alternative to steam reforming. This task would also look at more exotic alternatives, such as liquid metal decomposition and high-energy-density fuels. The fuel development would include both combustion and regenerative cooling research. (ROM $500,000 per year for 5 years)
REFERENCES
Published
Churchill, A.V., J.A. Hager, and A.E. Zengel. 1965. Fuels for Advanced Air-Breathing Weapon Systems. ASME Paper 65-0804. October. New York: American Society of Mechanical Engineers.
Huang, H., and D.R. Sobel. 2002. Endothermic Heat Sink of Jet Fuels. AFRL-PR-WP-TR-2002-2094. East Hartford, Conn.: United Technologies Research Center.
Huang, H., L. Spadaccini, and D. Sobel. 2002. Endothermic Heat-Sink of Jet Fuels for Scramjet Cooling. AIAA Paper 2002-3871. East Hartford, Conn.: United Technologies Research Center.
Lander, H., and A.C. Nixon. 1971. Endothermic fuels for hypersonic vehicles. Journal of Aircraft. 8(4):200-207.
Lehrach, R.P., L.J. Spadaccini, M.R. Glickstein, W.T. Fisher, and C.B. Graves. 1995. Applications Study of Endothermic Fuels. WL-TR-96-2122. East Hartford, Conn.: United Technologies Research Center.
Lipinski, J., R. Johnson, and C. White. 1992. Design, Fabrication, and Testing of a Lightweight Endothermic Methylcyclohexane Fuel Heat Exchanger/Reactor System. WL-TR-92-2076. East Hartford, Conn.: United Technologies Research Center.
Sobel, D.R., and L.J. Spadaccini. 1994. Development of Endothermic Potential of JP-8. WL-TR-94-2059. East Hartford, Conn.: United Technologies Research Center.
Spadaccini, L.J., M.B. Colket, P.J. Marteney, R. Roback, and M.R. Glickstein. 1993a. Endothermic Fuel/Catalyst Development and Evaluation Phase I. WRDC-TR-89-2141. East Hartford, Conn.: United Technologies Research Center.
Spadaccini, L.J., D.R. Sobel, M.B. Colket, P.J. Marteney, and M.R. Glickstein. 1993b. Endothermic Fuel/Catalyst Development and Evaluation Phases II, III, and IV. WL-TR-91-2126. East Hartford, Conn.: United Technologies Research Center.





