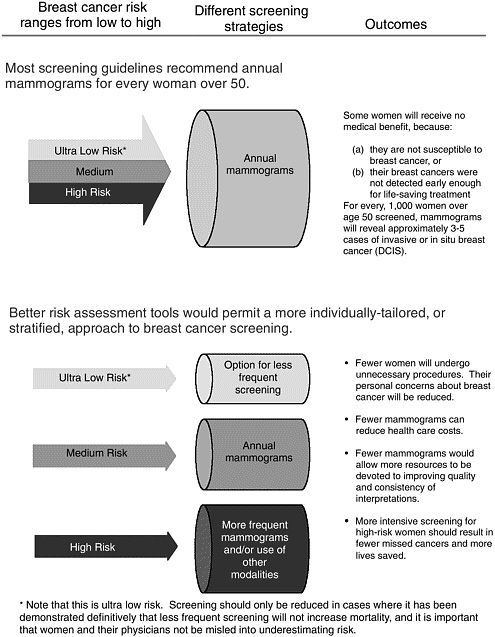
4
Understanding Breast Cancer Risk
Every woman is at some risk for breast cancer, but the degree ofrisk for individual women ranges from very low to very high. Understanding risk is important because it affects medical decisions—from whether a symptom-free woman should have a mammogram to how intensively to treat existing breast disease to how aggressively to pursue prevention strategies, such as the use of anti-estrogens or prophylactic mastectomy and removal of a woman’s ovaries.
If a screening technology existed that was so simple and so inexpensive that it could be used often enough to detect even fast-growing cancers, so reliable that no supplemental screening or diagnostic tools would be needed, and so convenient and comfortable that every woman would be willing and able to undergo frequent screening, then every woman could be screened and risk assessment would not be necessary. Unfortunately, not a single one of these conditions is met by current screening options for any type of cancer. Nor are there any tools on the horizon that promise to meet these conditions in the near term. Risk assessment is and will almost certainly remain an essential component of early detection of breast cancer.
Risk factors are identified (and new ones continue to be identified) through epidemiologic research studies, which typically measure the relative risk of the factors being studied (see Box 4-1). If a woman has a factor that is associated with a relative risk greater than 1, then—all other things being equal—her risk will be higher than the population average. If she does not have that factor, her risk will be lower.
Risk, or absolute risk, is a measure of the probability of developing cancer over a specified time interval. This is sometimes expressed as the
|
BOX 4-1 A relative risk compares the risk of disease among people with a particular risk factor to the risk among people without that risk factor. If the relative risk is above 1.0, then risk is higher among those with the risk factor than those without. Relative risks below 1.0 indicate a protective effect, or lower risk, associated with a particular factor. Relative risks are useful for comparisons, but they do not provide information about the absolute amount of additional risk experienced by the group with the risk factor in question. For example, current users of combination estrogen and progestin hormone replacement therapy (HRT) have a relative risk of 1.26, or a 26 percent increased risk. Although this increased risk may seem substantial, it proves to be less so in absolute terms because of the very low risk of breast cancer among young women in general. Among 10,000 women who have been using HRT for 5.2 years, 38 breast cancers would be expected to be diagnosed. Among 10,000 similar women who never used HRT, 30 cases would be expected over the same period. Therefore, the 26 percent increased relative risk results in an absolute risk of only 8 additional breast cancer cases per 10,000 women over a period greater than 5.2 years. Adapted from the American Cancer Society, Breast Cancer Facts and Figures 2003-2004.1 |
lifetime risk, or the risk to, say, age 70. Or the risk may be expressed as the probability that a woman of a given age will develop cancer in the next 10 years.
The statistic that one in eight women who survive to age 85 will develop some form of breast cancer in her lifetime is alarming, but this masks the important influence of age on risk (Table 4-1). Fewer than 5 percent of invasive breast cancers occur in women under age 40, whereas over three-quarters are in women over the age 50.
Numerous case-control and cohort studies over the past several decades have identified various factors, some of which have been shown to be consistently associated with risk, such as reproductive hormones, and others that are less consistent, such as dietary factors (Box 4-2, Table 4-2). Risk factors such as body mass index and dietary fat have been associated with specific types of breast cancer whose growth is stimulated by the sex hormones estrogen and progesterone.15 Family history increases risk although not as much as some women believe. Eighty-nine percent of women who develop breast cancer have no family history among their first-degree relatives (mother, daughter, or sister).16 The amount of increased risk depends on how close a relation the affected relative is, the age at which they developed breast cancer, and the number of relatives affected.
TABLE 4-1 Age-Specific Probabilities of Developing Breast Cancer1
|
If current age is |
Then the probability of developing breast cancer in the next 10 years is: |
or 1 in: |
|
20 |
0.05% |
2,152 |
|
30 |
0.40% |
251 |
|
40 |
1.45% |
69 |
|
50 |
2.78% |
36 |
|
60 |
3.81% |
26 |
|
70 |
4.31% |
23 |
|
BOX 4-2 Case-control studies are retrospective observational studies in which investigators identify one group of patients with a specified outcome (cases) and another group without the specified outcome (controls). Investigators then compare the histories of the cases and the controls to determine the extent to which each had the possible risk factor being investigated. Cohort studies are observational studies in which outcomes in a group of patients who possess the possible risk factor being tested (the cohort) are compared with outcomes in a control group of patients who do not possess the possible risk factor. For example, the occurrence of breast cancer would be compared between two groups of women neither of whom have breast cancer at the beginning of the study; one of the groups would possess the possible risk factor and the other group would not. The number of new cases of breast cancer in the two groups would be compared over time. |
Approximately 70 percent of women who develop breast cancer have the type of cancer called hormone receptor positive, which means that the cancerous tissue contains receptors for estrogen and/or progesterone. This association may, therefore, prove to be more relevant among women with elevated levels of these hormones, for example, premenopausal women or women using hormone replacement therapy.17,40,46 More research into risk profiles of such subtypes of breast cancers may elucidate a clearer connection between risk factors and the development of breast cancers. Although many factors that influence risk have been identified, it is still not possible to determine which women will develop breast cancer and which will not.
TABLE 4-2 Risk Factors for Breast Cancer
|
Risk Factor |
Relative Risk |
Category at Risk |
Comparison Category |
|
Germ-line mutation |
200? |
Heterozygous for BRCA1, age <40 |
Not heterozygous for BRCA1, age <40 |
|
|
15? |
Heterozygous for BRCA1, ages 60-69 |
Not heterozygous for BRCA1, ages 60-69 |
|
Cytological findings (fine-needle aspiration; nipple aspiration fluid) |
18.1 |
Proliferation with atypia and positive family history |
No abnormality detected |
|
4.9-5 |
Proliferation with atypia |
No abnormality detected |
|
|
|
2.5 |
Proliferation without atypia* |
No abnormality detected |
|
Other histologic findings |
17.3 |
Ductal carcinoma in situ |
No abnormality detected |
|
|
16.4 |
Lobular carcinoma in situ |
No abnormality detected |
|
Positive breast biopsy |
11 |
Hyperplasia with atypia and positive family history |
No hyperplasia, negative family history |
|
|
5.3 |
Hyperplasia with atypia |
No hyperplasia |
|
|
1.9 |
Hyperplasia without atypia |
No hyperplasia |
|
Past history of breast cancer |
6.8 |
Invasive breast carcinoma |
No history of invasive breast carcinoma |
|
Current age |
5.8 |
65 or older |
Less than 65 |
|
Radiation exposure |
5.2 |
Radiation therapy for Hodgkin’s disease |
No exposure |
|
|
1.6 |
Repeated fluoroscopy |
No exposure |
|
Risk Factor |
Relative Risk |
Category at Risk |
Comparison Category |
|
Breast density |
More than 75% of breast is mammographically dense |
Less than 25% of breast is mammographically dense |
|
|
Family history |
3.6 |
Two 1st-degree relatives with breast cancer |
No 1st- or 2nd-degree relative with breast cancer |
|
|
3.3 |
1st-degree relative with premenopausal breast cancer |
No 1st- or 2nd-degree relative with breast cancer |
|
|
1.8 |
1st-degree relative 50 years or older with postmenopausal breast cancer |
No 1st- or 2nd-degree relative with breast cancer |
|
|
1.5 |
2nd-degree relative with breast cancer |
No 1st- or 2nd-degree relative with breast cancer |
|
Age at first birth |
1.7-1.9 |
Nulliparous or 1st child after 30 |
1st child before 20 |
|
Late menopause |
1.2-1.5 |
Older than 55 years |
Younger than 45 |
|
2.70 |
Current user of estrogen and progestin |
Never used |
|
|
|
1.96 |
Current user of estrogen only |
Never used |
|
Early menarche |
1.3 |
Younger than 12 years |
Older than 15 years |
|
Alcohol intake |
1.2 2 |
drinks per day |
Nondrinker |
|
Body mass index |
1.2 |
80th percentile |
20th percentile |
|
*There is controversy over whether pathologic hyperplasia detected in breast biopsy samples is directly equivalent to cytologic hyperplasia detected in samples obtained through fine-needle aspiration or nipple aspiration. ?These relative risks are subject to ascertainment bias and may overestimate the true risk associated with germline mutations in BRCA genes.5 ≠The data for hormone replacement therapy was updated due to the release of a new study after the original risk of hormone replacement was presented by Singletary et al., 2003.64 SOURCE: Adapted from Singletary and colleagues.64 |
|||
BREAST DENSITY
Mammographic breast density may be the most undervalued and underused risk factor in studies investigating breast cancer.13,38,73 It is a heritable trait, although the contribution of breast density to increased risk is independent of the risk associated with BRCA1 and BRCA2 mutations.7 Despite the ethnic variation in breast density, breast cancer risk rises with increasing breast density for each of the ethnic groups recently analyzed by Ursin and colleagues; the groups they analyzed included African Americans, Asian Americans, and non-Latino whites.69 A 2002 study reported that the average relative risk of breast cancer for women in the highest category of percentage of dense tissue compared with those in the lowest category is about 4.7 Previous studies reported relative risk estimates ranging from 2 to 6, with the majority of those studies reporting a relative risk of 4 or more (reviewed by Boyd and colleagues, 2002).7 The genetic factors that determine breast density may also play a role in breast cancer.73
GENETIC RISK FACTORS
Before a cell becomes cancerous, it must accumulate a “critical mass” of molecular changes that alter key genes or their functions. The end result is a loss of the normal molecular controls on the cell’s growth and differentiation. Some of the cellular changes that make a woman susceptible to developing breast cancer can be inherited. Such germ-line mutations are believed to account for the striking incidence of breast cancer in certain families, especially breast cancer that develops in both a woman’s breasts and/or at a young age. But less than 10 percent of all breast cancer cases are thought to stem from inherited mutations, such as BRCA1 and BRCA2, that individually increase risk by a substantial amount.41
The majority of breast cancer cases are due to an accumulation of cellular (somatic) changes that occur during a patient’s lifetime. This is why age is such a significant factor in most cancers—because the longer a person lives, the more time there is for mutations to accumulate. These changes are not inherited, but rather stem from factors such as exposure to carcinogens in the external environment, or from excessive or untimely exposure to breast cancer-promoting substances within the body, such as circulating hormones, or simply because of random mutations that occur during cell division. Inherited genes can also influence genetic mutations that occur during a person’s lifetime if they increase the susceptibility of other genes to mutation. For example, the ability of a cell to correct mistakes in gene replication that occur during cell division is diminished when the genes that normally support DNA repair have mutated. As a result, mutations accumulate faster than they would otherwise.
BRCA Genes and the Shortcomings of Genetic Testing
Studies of families with an exceptionally large number of members with breast and ovarian cancer led to the discovery of the first two inherited breast cancer susceptibility genes. By searching for genetic markers shared by all affected family members (linkage analysis), researchers in the 1990s were able to pinpoint two breast cancer susceptibility genes, BRCA1 and BRCA2.41 Both genes are rare, but they confer very high risk. Both genes code for proteins that are thought to play a role in the repair of genetic defects, and therefore mutations that decrease their ability to repair or limit the proliferation of cells with genetic defects will increase the susceptibility to breast cancer.41
Initial studies suggested that women who tested positive for either mutation would have nearly a 90 percent chance of developing breast cancer by age 70.29 A recent study found that Ashkenazi Jewish women who carry one of the three BRCA1 and BRCA2 mutations associated with Ashkenazi ancestry and who reach age 80 have an 82 percent risk of developing breast cancer; those who reach age 60 have a 55 percent risk.45
These studies indicate that BRCA1 and BRCA2 tests would be a useful clinical tool to identify women at high risk for breast cancer, but the lifetime probability estimates for developing breast cancer among women who test positive for mutations of BRCA1 or BRCA2 (also called penetrance of the genes) is variable and often overestimated. Lifetime risks of breast cancer in women in the general population who test positive for BRCA1 (that is, women who are not preselected on the basis of a family history of breast cancer) could be as low as 45 percent, and 26 percent in such women who test positive for BRCA2 (reviewed by Begg, 2002).5 Other studies based on women from the general population produced higher penetrance estimates, but none was as high as those seen in women from high-risk families.
Overestimates of the penetrance of BRCA1 or BRCA2 result from sampling bias. Studies of women who have breast cancer and are known to have a family history of breast cancer will generate higher estimates of penetrance than studies that start with women in the general population and assess the overall percentage of women who test positive for BRCA mutations and develop breast cancer. Women with BRCA mutations who develop breast cancer usually have several other risk factors that are likely to be shared with their relatives. These relatives could be at somewhat greater than average risk of developing breast cancer, even if they do not test positive for BRCA mutations. Consequently, the percentage of these women who test positive and develop breast cancer is likely to be greater than that of women who test positive in lower-risk populations.5
Evidence shows that environmental factors also play a role in determin-
ing the penetrance of BRCA genes. Some studies find that a woman’s reproductive history can modify the penetrance of BRCA1 or BRCA2 (reviewed by Burke and Austin, 2002).10 Other studies find that cancer risk is relatively greater in younger women who test positive for BRCA mutations than in older women.10,45 Birth cohort and physical exercise also have been shown to partially mitigate the influence of BRCA1.45 Ashkenazi Jewish women born with one of the three mutations associated with Ashkenazi ancestry who were born before 1940 have an average lower likelihood of developing breast cancer than similar women born after 1940. In the same study, women with those mutations who had been physically active as teenagers and were not obese as young adults showed an approximate risk reduction of 10 years—that is, a 60-year-old woman who was not obese at age 21 and with a history of physical activity had approximately the same average risk as a 50-year old woman with a history of obesity and no physical activity. Such a change in penetrance over time is likely due to the influence of a changing environment.
As Wylie Burke and Melissa Austin summarize in an editorial in the Journal of the National Cancer Institute:
The most important implication of penetrance studies should perhaps be to temper our expectations for predictive genetic tests. Without a healthy respect for the many factors that may influence penetrance, we will continue to overestimate the risk conferred by BRCA 1 and BRCA 2 mutations alone and, thus, miss opportunities to develop truly effective prevention strategies for women who are genetically susceptible to breast cancer that are based on a broad understanding of causative factors.10
The wide range of penetrance estimates complicates decisions for preventive interventions like prophylactic mastectomy or tamoxifen chemoprevention, although even the lowest penetrance estimates might be high enough to suggest that women who test positive for BRCA mutations should be screened more aggressively. However, one study found that annual mammograms and biannual physical exams were less sensitive, and detected tumors at later stages in women with BRCA mutations than in women at greater than average risk for breast cancer who lack the mutations.9 Furthermore, studies have found that BRCA2-deficient cells are hypersensitive to the effects of radiation,54 so there is concern (but so far no evidence) that women, especially those with BRCA2 mutations, might be susceptible to radiation-induced genetic defects and cancer.
Another problem is that researchers have detected more than 2,000 mutations of BRCA1 or BRCA2,54 but the clinical significance of these is not yet known; some may not influence breast cancer risk. Consequently, more than 1 in 10 BRCA tests yields inconclusive results because the clinical significance of the specific mutations detected by the tests is unknown.2
Women also may test negative for mutations in BRCA1 or BRCA2 and still harbor a BRCA mutation that increases their risk of breast cancer because this mutation falls outside the range of mutations detected by current BRCA1 or BRCA2 tests.66 There is only one commercially available test for BRCA mutations. It costs about $450, and tests only for the three Ashkenazi mutations. A test for all of the known mutations in BRCA1 and BRCA2 genes would cost nearly $3,000 (Personal communication, W.A. Hockett III, Myriad Genetics, Inc., Vice President of Corporate Communications for Myriad Genetics, Inc., December 2, 2003). Testing negatively for BRCA mutations also does not rule out the possibility that a woman with a strong family history for breast cancer has inherited mutations in other genes that increase her breast cancer risk.63
Perhaps the biggest limitation is that less than one-quarter of 1 percent of women in the general population are believed to harbor BRCA mutations,18,32,57 and mutations in either of the BRCA genes account for only 2 to 3 percent of all breast cancers (reviewed by Wooster and Weber, 2003).71 Because more than 10 percent of women will develop breast cancer in their lifetimes, BRCA tests clearly will be a small piece in the puzzle of identifying individual risk.
Many more genetic risk factors have been published than have been verified. A literature review of epidemiological studies that assessed associations between polymorphisms and risk of cancer found that only a small proportion of the published studies were large and population-based.35 Because studies based on small samples sizes are prone to false-positive or false-negative findings, large and well-designed studies of genetic risk are essential. Studies that analyze multiple genes or polymorphisms would be especially useful in improving our understanding of breast cancer.
Polymorphisms
The search for other genetic markers that determine breast cancer susceptibility is ongoing and has focused on subtle DNA changes, known as polymorphisms, that are shared by many people, and that may affect susceptibility to carcinogens and cancer promoters in the environment or the body, or affect the body’s immune response to cancer cells. Each polymorphism probably increases or decreases breast cancer risk by only a small amount, perhaps a few percentage points. But because these polymorphisms are found in all people, their impact on breast cancer risk may be considerably greater than that of the relatively rare BRCA mutations,22 and the combined impact of several polymorphisms on breast cancer risk could be substantial.
A compilation of various polymorphisms might enable the stratification of some women into low- or high-risk breast cancer groups. However,
research on genetic polymorphisms that influence breast cancer susceptibility is in its infancy, and many more studies are needed before they are useful in stratifying women into breast cancer risk groups.22
Researchers seeking to discover polymorphisms that boost breast cancer risk have tended to focus their search on the most biologically plausible genes, such as those known to be involved in the metabolism of carcinogens, or the regulation of estrogen levels, or that are the normal variants (proto-oncogenes) of genes known to cause cancer (oncogenes). (Proto-oncogenes are involved in the regulation of normal cell growth and differentiation.) For the most part, reports of polymorphisms that affect susceptibility to breast cancer have been based on relatively small studies.
Table 4-3 presents the results of meta-analyses of studies on genetic polymorphisms that have been linked to breast cancer risk. Precise and validated estimations of the genetic risk associated with these polymorphisms will require large case-control studies. Of 35 polymorphisms in 19 different genes described in at least two breast cancer studies, only 13 polymorphisms in 10 genes showed an association with breast cancer. Only TNF-alpha and a variant of the HSP-70 protein show odds ratios higher than 3. Although an odds ratio of 3 or higher is a common benchmark of an important risk factor, this is still much lower than what is needed for screening tests, and would involve high false-positive or -negative rates, or both. Thus, although statistically significant at the population level, such a risk factor would not, by itself, be helpful in predicting individual risk. As of this writing, except for BRCA1 and BRCA2, no single genetic risk factor predicts the development of breast cancer well enough to be used on its own for individual risk stratification.
Relatively little research has been performed on combinations of polymorphisms which are addressed in only a few studies in breast cancer patients. Because the products of several genes interact (for example, nearly half of the genes reviewed by de Jong and colleagues play a role in estrogen metabolism), interactions between the genes are likely. Some investigators believe a whole genome screen would be the ideal method to detect new breast cancer susceptibility genes. This method, however, is still too expensive to carry out in large study populations.22 Until this is feasible, it would be useful to collect data on appropriately sized, well-described study populations.22 Analysis of several (or all) of the polymorphisms already known to be associated with breast cancer in the same population may increase our understanding of the etiology of breast cancer and permit better risk assessments (reviewed in 2001 by de Jong).22
TABLE 4-3 Genes Other Than BRCA1 and BRCA2 Involved in Breast Cancer Susceptibility22
|
Gene |
Description |
Effect on Breast Cancer Risk |
Odds Ratio* |
|
Rare genetic syndromes with increased breast cancer risk |
|||
|
Tp53 |
Mutation of this gene causes Li-Fraumeni syndrome and is characterized by an increased risk of several cancers. Expressed in three different variants. |
Associated with increased risk, particularly in white populations. Risk not shown in Hispanic, African-American, or Pakistani study participants. |
1.08 CI 0.88-1.13 |
|
ATM |
Mutation of this gene causes ataxia telangiectasia, a neurodegenerative disease characterized by lack of coordination, red lesions, and immune defects. |
Few patients survive to an age at which breast cancer occurs, but a role in increased risk is plausible and has been shown in some small studies. |
N/A |
|
PTEN |
Mutation of this gene causes Cowden syndrome, characterized by malformations resembling tumors composed of mature tissues, especially of the skin, mucous membranes, breast, and thyroid. |
Not likely to have an effect in the sense of classical heredity. Unknown if PTEN plays a role in sporadic breast cancer susceptibility. |
N/A |
|
LKB1 |
Mutation of this gene causes Peutz-Jeghers syndrome and is characterized by freckle-like spots on the lips, mouth and fingers and benign polyps in the intestines. |
Only likely to play a role in increased risk among those patients with Peutz-Jeghers syndrome. |
N/A |
|
Low penetrant cancer susceptibility genes: Proto-oncogenes |
|||
|
HRAS1 |
Protein product is a protein kinase that transmits signals from growth factor receptors. When mutated can result in abnormal cell cycle control. |
Moderately associated with increased risk. |
2.04 CI 1.73-2.41 |
|
Gene |
Description |
Effect on Breast Cancer Risk |
Odds Ratio* |
|
L-myc |
Protein product is a transcription factor that helps initiate cell division. When mutated can result in accelerated cell division and tissue growth. |
No association found. |
1.12 CI 0.77-1.63 |
|
TGFBR1*6a |
TGFBR1*6a is a variant of one of the receptors through which Transforming Growth Factor (TGF) exerts its actions. TGF-B is the most potent naturally occurring inhibitor of cell growth. |
A high-frequency, lowpenetrance allele that is moderately associated with increased risk.42 |
1.48 CI 1.11-1.9642 |
|
Low penetrant cancer susceptibility genes: Metabolic pathways |
|||
|
NAT1/NAT2 |
Protein product is an enzyme that can bioactivate several known carcinogens through acetylation. |
No association found. |
1.13 CI 0.91-1.39 |
|
GSTM1 |
Protein product is an enzyme responsible for the metabolism of a broad range of chemicals and carcinogens. |
Marginally significant increase in risk. |
1.13 CI 1.02-1.26 |
|
GSTP1 |
Protein product is an enzyme that plays an important role in detoxification. |
Appears to play a role in increased risk in a few small studies. |
1.19 CI 0.91-1.56 |
|
GSTT1 |
Protein product is an enzyme found in red blood cells; may detoxify some synthetic chemicals. |
No association found. |
1.04 CI 0.86-1.25 |
|
CYP1A1 |
Protein product is an enzyme responsible for metabolizing estrogens and polycyclic aromatic hydrocarbons. |
m1 polymorphism: Small increase in risk in the white population. |
0.99 CI 0.83-1.19 |
|
|
m2 polymorphism: Moderately increased risk in postmenopausal women. |
1.18 CI 0.94-1.48 |
|
|
Gene |
Description |
Effect on Breast Cancer Risk |
Odds Ratio* |
|
CYP1B1 |
Protein product is an enzyme responsible for metabolizing polycyclic aromatic hydrocarbons (3 variants codon 119, 432, and 453). |
No association found in pooled data from several studies. |
1.62 CI 1.15-2.29 |
|
CYP2D6 |
Protein product is an enzyme involved in the metabolism of commonly prescribed drugs including codeine. |
May play a role in increased risk. |
1.19 CI 0.97-1.45 |
|
Low penetrant cancer susceptibility genes: Estrogen pathway genes |
|||
|
CYP17 |
Protein product is an enzyme that mediates formation of estrogens, progesterones, and androgens. |
No association found in analysis of several studies. However, because age was not accounted for, increased risk for breast cancer in young women cannot be excluded. |
0.99 CI 0.88-1.11 |
|
CYP19 |
Protein product is an enzyme that converts androgens into estrogens and maintains the local level of estrogen. |
Might play a minor role. |
1.15 CI 0.72-1.85 |
|
ER |
Protein product is a receptor that binds and transfers estrogen to the nucleus; regulates the production of several transcription factors. |
Only five relatively small studies examined polymorphisms in the ER gene. Due to a small sample size, an association cannot be confirmed or excluded. |
0.84 CI 0.40-1.78 |
|
PR |
Protein product is a receptor that binds and transfers progesterone to the nucleus; regulates the production of several transcription factors. |
Results showed a decrease in risk. |
0.95 CI 0.78-1.16 |
|
Gene |
Description |
Effect on Breast Cancer Risk |
Odds Ratio* |
|
AR |
Protein product is a receptor that binds and transfers androgen to the nucleus; regulates the production of several transcription factors. |
Does not play a major role. |
N/A |
|
COMT |
Protein product is an enzyme that degrades catecholamine transmitters including estrogens, dopamine, epinephrine, and norepinephrine. |
No increase in risk. |
0.92 CI 0.76-1.10 |
|
UGT1A1 |
Protein product is an enzyme that helps maintain levels of estrogens and enhances the elimination of many synthetic chemicals. |
No association found. |
0.99 CI 0.80-1.24 |
|
Low penetrant cancer susceptibility genes: Immunomodulatory pathway genes |
|||
|
TNF-alpha |
Protein product is a cytokine that stimulates inflammation and immunological response to tumor cells. |
Association with increased risk shown in one small study. Additional data are required to define the precise association. |
3.49 CI 1.62-7.51 |
|
HSP70 |
Protein product is chaperone protein that regulates structure, localization, and turnover of cellular proteins. |
HSP70-hom: Increased risk associated. |
3.56 CI 1.26-10.01 |
|
|
HSP70-2: No association found. |
1.74 CI 0.55-5.52 |
|
|
Low penetrant cancer susceptibility genes: Iron metabolism genes |
|||
|
HFE/HH |
Mutation of this gene can cause iron accumulation resulting in cirrhosis of the liver, diabetes, abnormal skin pigmentation, and heart failure. |
Do not play major roles in increased risk. |
N/A |
|
Gene |
Description |
Effect on Breast Cancer Risk |
Odds Ratio* |
|
Other genes |
|||
|
VDR |
Protein product is a receptor that acts as a transcriptional regulatory factor and can stimulate cell differentiation. |
Study results are contradictory; association remains unclear. |
0.95 CI 0.74-1.20 |
|
APC |
Tumor suppressor gene that arrests the cell cycle and prevents further cell division and unregulated growth. When mutated the gene is associated with colorectal cancer. |
Probably does not play a role in increased risk of breast cancer. |
N/A |
|
*Odds ratios for only heterozygous genotype for most common variant alleles are listed (CI= 95% confidence interval). |
|||
MANAGING RISK
Individualized Risk Prediction
Understanding that women do not have uniform risk for breast cancer suggests the possibility that they could be stratified into high- or low-risk groups. In theory, such stratification should indicate which women are most likely to benefit from more intensive screening for breast cancer (Figure 4-1). For example, most women would gain no medical benefit from screening before age 40 or from twice-yearly screening, but a small minority could. Conversely, many women could safely be screened for breast cancer only every 2 years, or perhaps even at longer intervals. And even though men can develop breast cancer, it occurs too rarely to warrant mammography screening for men in the general population (Box 4-3). The goal of improving risk assessment is to stratify breast cancer detection strategies with the aim of increasing survival in high-risk women while decreasing cost and complications in low-risk women.23 The challenge lies in developing a more refined understanding of how to assess risk in individual women, and that depends on data from well-designed, large-scale epidemiological studies.
Mammography screening guidelines already take into account two of the most significant risk factors, gender and age. But we could do much
|
BOX 4-3 Breast cancer is not only a woman’s disease. Each year about 1,300 men in the United States are diagnosed with new cases of invasive breast cancer, and about 400 will die of breast cancer.1 The American Cancer Society reports that breast cancer is 100 times less common in men than women, and accounts for less than one-quarter of 1 percent of cancer deaths among men. The symptoms and types of breast cancers found in men are similar to those found in women, except for lobular cancers, which men do not develop. Many breast cancers among men are found only after the late onset of cancer symptoms are identified as malignant through biopsy without performing a mammogram. The prognosis for men with breast cancer was once thought to be worse than for women, but this is not true. Stage for stage, the survival rates are equal. With the exception of BRCA1 mutations and other gender-specific factors such as menarche and childbirth, breast cancer risk factors for men are similar to those in women, with older age and history of cancer being the predominant factors. However, the absolute risk of breast cancer is so low that screening mammography is not warranted in men. It is possible, but unlikely, that a risk factor for male breast cancer would be discovered that was so informative that it would identify a group of men whose risk of breast cancer was high enough to warrant screening, but current knowledge does not support the search for such a hypothetical factor. Mammography may, however, be useful in screening for recurrence or development of a new cancer in men who have already had breast cancer. And, clearly, physicians should be aware that although it is rare, male breast cancer does occur and any single signs or symptoms of breast cancer in a man should be investigated. |
better. Earlier attempts to base screening strategies on factors other than gender and age, such as family history or reproductive factors, have not been successful, largely because the relative risks of those factors are too low (reviewed by Smith, 1999;16 IOM, 200141). A risk factor that is used to stratify screening strategies must identify enough added risk that it is reliably linked to different outcomes.
Individualized risk prediction for breast cancer was first popularized with publication of the “Gail model.”33 In this model, five known risk factors are used to obtain risks of cancer over fixed time periods. The factors used are:
-
Age,
-
Age at menarche,
-
Age at first live birth,
-
Number of prior breast biopsies, and
-
Number of first-degree relatives with breast cancer.
The model was derived by Mitchell Gail and his colleagues who used a retrospective database obtained from the Breast Cancer Detection Demonstration Project study conducted in the 1970s to evaluate a variety of potential risk factors, including some that were not significantly associated with breast cancer risk, such as cigarette smoking or the use of oral contraceptives. The sample size included more than 200,000 women, which made it large enough to allow accurate prediction and internal validation of the predicted risks. The model has subsequently been validated in other datasets, and expanded to be relevant to women of different ethnic backgrounds (reviewed by Eva Singletary in 2003).64 The Gail model allows for simple tables that can be used to easily assess the risk for an individual woman while she is in the clinic for counseling, and has been used widely to stratify women in important ways. For example, eligibility for major breast cancer prevention trials, such as the National Surgical Adjuvant Breast and Bowel Project (NSABP) Study of Tamoxifen and Raloxifene (STAR) trial,a is based on the Gail risk score. Only those with a sufficiently high personal risk are eligible.
But the Gail model has some limitations. It has predictive value only for women over age 35 who have not previously been diagnosed with breast cancer,67 and it does not incorporate specific genetic risk factors. Although it is highly accurate at predicting the aggregate number of women within various age or other risk groups who will develop breast cancer within 5 years, its ability to predict which individual women will develop breast cancer is only slightly better than chance.60 It is, nonetheless, used to assist in determining whether individual women should engage in cancer prevention measures, because there are no better models that have been validated for individual risk prediction.
In summary, risk prediction based on easily obtained epidemiologic factors is currently accomplished widely using the Gail model. Genetic risk is predicted independently of this for women with BRCA mutations.29,45 An integrated approach to risk prediction is desirable and, in principle, risk models should also include the likelihood of adverse events. False positives and unnecessary treatment are adverse events, but they are not identifiable because it is not possible to predict which cases of breast cancer (including ductal carcinoma in situ, or DCIS) will become life-threatening without
treatment. In any event, it is a matter of debate as to whether the harms of breast cancer screening are severe enough to be included in risk models, and they might be more appropriately considered in the context of shared decision making (see discussion in Chapter 2 on harms of mammography).
A number of hurdles still need to be overcome, including the development of more encompassing tests to predict genetically based risk that would not limit the scope of breast cancer risk prediction to the influence of just one or two genes and their narrow range of known mutations, but instead would consider the effects of a wide array of genes and environmental factors that together determine breast cancer risk. Models that integrate risk information from two or more different types of assessments, such as BRCA test results and family history, are also needed. Certainly, there is no reason why risk calculated through methods similar to those used by Gail could not also be used to inform the intensity of the screening strategy.
The Committee believes that individual screening strategies are essential to improving the early detection of breast cancer, and risk assessment is an essential step in the development of individualized screening strategies (Box 4-4). In theory, assessment of individuals’ breast cancer risk could foster more accurate and less costly early breast cancer detection by determining screening strategies that are tailored to individual risks. However, the Committee emphasizes that even with individual risk assessment, at the current level of predictive accuracy, it is important to uphold the consensus guidelines for the minimum recommended use of mammography screening developed by nationally recognized organizations whose members are experts in the methodology of screening studies and who have carefully evaluated the evidence.
Caveats in Risk Stratification
The costs and benefits of increasing versus decreasing screening intensity are decidedly different. In considering a reduction in screening frequency, the benefits of lower costs and less inconvenience would be weighed against an added risk of dying from breast cancer. In contrast, a more aggressive strategy for high-risk women has the potential to save more lives. This, too, would require validation in appropriately designed clinical trials. Breast cancer is relatively rare in younger women, and the number of younger women who are at high risk would be even lower. But these relatively few high-risk women also tend to be those for whom standard mammography is less effective. Better methods of risk stratification could thus be of particular benefit to high-risk young women who would benefit from intensified screening—such as with more frequent screening or using technologies that compensate for the limitations of mammography.
Recommendations to begin mammography screening only after age 40
|
BOX 4-4 Imagine a blood test that could identify women whose risk of breast cancer is so low that they could safely forego regular mammogram screening. A recent study of post-menopausal women by Steven Cummings and colleagues suggests that such a test might someday be possible.19 Their study included more than 7,000 post-menopausal women whose average age was 66 years, and they found that the 4-year rate of breast cancer in women with undetectable serum estradiol levels was only 0.6 percent, compared to a rate of 3.0 percent for women whose estradiol levels were greater than 10 pmol/L (2.7 pg/mL), which translates into an approximately seven-fold difference between the two groups. For comparison, the average 10-year risk for a 40-year old woman is approximately 1.5 percent, which is at or below the threshold for recommending regular mammograms. It is important to note, however, that Cummings’ study reported 4-year rates, and these cannot be assumed to be the same as 10-year rates. But the comparison remains impressive—and suggests that some women over 60 might have a lower risk of breast cancer than average-risk 40-year-old women. It has been known for some time that the risk of developing breast cancer drops with declining levels of serum estradiol, which is the most active type of estrogen,37 but this study used a highly sensitive test to measure estradiol that allowed greater resolution of low estrogen levels than is possible in standard tests. In fact, this is one of the caveats of the study. The minute quantities of estradiol that differentiate between high- and low-risk women required more expensive and sensitive tests than are currently available for clinical use. Assays used in daily clinical practice measure estradiol levels in the range of 10 to 20 pg/mL, and are not sensitive enough to distinguish levels between 0 and 10 pg/ml, whereas the average level of estradiol in the study was about 3 pg/mL.48 Another caveat is that because the study consisted of a 4-year follow-up period, breast cancer incidence might only be delayed to a later point in time in post-menopausal women with ultralow estradiol levels. To date, there is no commercially available test. Research on the long-term accuracy and development of a clinically useful test will need to be completed before the test can be used to evaluate a woman’s risk of breast cancer. Other potential uses of highly sensitive estradiol measurements might be to identify women whose risk profiles make them candidates for preventive treatments, such as anti-estrogen. Such a test would be a welcome addition to the mix of risk assessment tools. |
are largely based on the fact that cancer incidence increases with age, but also because mammography is less sensitive in women younger than 40, because they tend to have dense breasts. More frequent mammography screening in younger women also has the downside of exposing a more radiation-sensitive breast to radiation, yet breast cancers in younger women tend to be more aggressive, suggesting younger women should be screened more often.
The fact that mammography is generally less sensitive in younger women and that younger women are more sensitive to radiation alters the balance of risk and benefit. Detection technologies that do not involve radiation would thus be likely to offer a relatively greater advantage to younger women.
Risk Perception Is Often Distorted
Women fear breast cancer more than any other disease,51,52 but their perception of risk is often distorted. In general, women in the United States and Canada tend to overestimate their risk of breast cancer, whereas women in the United Kingdom are more likely to underestimate their risk (reviewed by Hopwood in 2000).39 Many women are also unclear about risk factors. More than three-quarters of women in one large survey recognized family history as a major determining factor for developing breast cancer, but only 13 percent correctly identified old age as a risk factor.51 As a result, older women are more likely to underestimate their risk than younger women,20,36 who tend to overestimate their risk. One study reported that women in their forties overestimated their probability of dying of breast cancer within 10 years by more than 20-fold.6 The women in that study also overestimated the effectiveness of mammography. Considering the extreme bias in the media toward telling personal breast cancer stories of women in their thirties and the rarity of such stories of older women, these distorted perceptions are perhaps not surprising.11
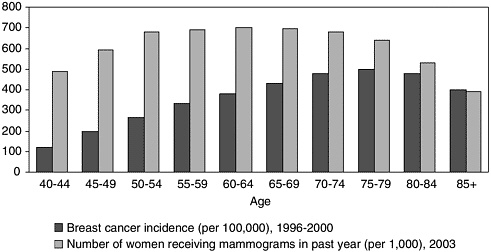
The likelihood that a woman will adhere to screening recommendations depends, in part, on her perceived risk of developing breast cancer.3,14 Despite the general validity of the Gail model in predicting risk, it does not predict risk perception or the inclination of a woman to follow mammography guidelines. A 1996 study in which more than 900 women were inter-viewed found a striking disparity between Gail model objective risk factors and the accuracy of women’s beliefs about their own risk and adherence to mammography screening guidelines.20 The observation that participation in screening mammography programs declines with age reflects this discordance between belief and behavior (Figure 4-2).
Reports of risk factors in the media as well as the scientific literature typically highlight relative risk rather than absolute risk which makes sense in the attempt to identify risk factors (see Box 4-1 for definitions), but it encourages exaggerated perceptions of personal risk. Most women whose mother had breast cancer are acutely aware that they are “at risk” for breast cancer, but few of them appreciate the moderate extent of their added risk. For example, the relative risk of developing breast cancer for a woman whose mother had breast cancer after age 50 is estimated to be 1.8 (see Table 4-2). If that woman is 40 years old, her underlying risk of
developing breast cancer within 10 years would be, on average, about 1.5 percent (Table 4-1). With her family history that risk is multiplied by 1.8 which gives her a 10-year breast cancer risk of 2.7 percent—higher than average, but still relatively low. Indeed, many women are surprised to learn than almost 90 percent of women who develop breast cancer have no close family history; that is, neither a mother, sister, or daughter with breast cancer.
Some women have gone to extreme measures to reduce their risk. For example, one study reported on 75 high-risk Canadian women who underwent bilateral mastectomy to avoid breast cancer, but the researchers found that on average the women had overestimated their lifetime risk of developing breast cancer before surgery three-fold.50 The women in the study with strong or limited family histories of breast cancer estimated their lifetime risk for breast cancer as approximately 75 percent, whereas their calculated risks were only 25 percent (for strong family histories) and 18 percent (for limited family histories). In contrast, the women with BRCA gene mutations estimated their lifetime risk as 80 percent, while the model used to calculated their risk (BRCAPRO) indicated a 65 percent lifetime risk—a difference that was not statistically significant. Of course, there is no way to be sure that the models are accurate for the individual women in this study because of uncertainty about the penetrance of the BRCA mutations in each woman.
Distorted risk perception includes perceptions about prognosis as well. Although the prognosis for DCIS is excellent, the prognosis for early inva-
TABLE 4-4 Percent of Women Who Rated Certain Outcomes “Likely” Was Not Significantly Different for Diagnoses, Despite Significantly Different Prognoses
|
|
Perceived Risk Among Women with Different Diagnoses* |
|
|
Possible Event |
DCIS |
Early Invasive Breast Cancer |
|
Developing a local recurrence |
53% |
45% |
|
Developing a distant recurrence |
36% |
39% |
|
Dying of breast cancer |
27% |
27% |
|
*None of the differences between diagnoses meet statistical significance. |
||
sive breast cancer is not. Ten years after a diagnosis of DCIS, 2 percent of women will have died of breast cancer compared to 11 percent of women diagnosed with early invasive breast cancer.28 Despite the different levels of risk, a study of 228 patients with either DCIS or early invasive breast cancer found no significant differences between the two groups in terms of perceived risk for recurrence or death from breast cancer (Table 4-4).58 In addition, both groups of women expressed similar levels of anxiety and depression: 56 percent of women with DCIS and 54 percent of women with early invasive breast cancer reported anxiety; 41 percent of women with DCIS and 48 percent of women with early invasive breast cancer reported depression.
Finally, not only do many women have distorted perceptions of their risks of developing breast cancer, but most women misunderstand or overestimate the benefits of mammography.6,24 A survey conduced in 1999 reported that a 57 percent majority of women in the United States believe that mammography affects their risk of developing breast cancer, compared to 37 percent who correctly responded that mammography does not influence breast cancer risk (Table 4-5).24 Women in the United Kingdom and Italy who were surveyed overestimated the benefits of mammography to an even greater extent, 69 and 81 percent, respectively. Likewise, most women in all countries surveyed overestimated the extent to which mammography can reduce mortality due to breast cancer.
Decisions and Uncertainty
When information is certain, decisions are simple. A 40-year-old woman with an invasive breast tumor that will metastasize within 5 years
TABLE 4-5 Most Women in the United States Overestimate the Benefits of Mammography
unless it is removed does not need a decision aid to take action. In contrast, a 65-year-old woman diagnosed with low-grade DCIS is likely to welcome a decision aid that allows her (and her physician) to integrate what is known about her personal risk factors with the likely benefits of different treatments. Likewise, a 75-year-old woman may want information that would assess her 10-year likelihood of death from other causes against the likelihood of dying from breast cancer in deciding whether to undergo screening.
Decision aids are tools that assist in choosing between complex alternatives such as determining optimal breast cancer screening strategies or choosing breast cancer treatment options. Sometimes these aids take the form of complex decision analyses, and sometimes they provide baseline probabilistic data in a variety of forms so that patients can better understand tradeoffs between risks and benefits. In the context of screening, formal decision analyses have been used by policymakers to evaluate the societal implications of varying strategies for a variety of tumors. From the perspective of an individual patient, these models can also be useful. However, information about probabilities at varying points in the screening and management process is sometimes more valuable. In the screening situation such information is useful because no screening test is 100 percent sensitive and specific. For example, a positive test for a BRCA1 or BRCA2 mutation
does not mean that it is certain a woman will get breast cancer, and conversely, a negative test does not mean she will not. Emerging data on genomic markers and circulating biomarkers suggest that genetic or chemical analyses may help stratify patients into individualized risk categories, but the results are in continual flux and will be difficult to interpret until appropriate longitudinal large-population studies are done. Thus, there is a need to transfer information as clearly as possible to patients as they face decisions about having one or more screening tests for breast cancer.
Effectiveness of Risk Communication and Decision Aids
A large body of research has shown that good communication and strong patient-provider relationships are linked to greater patient satisfaction, and positive health outcomes.43,55 Moreover, specific provider behaviors such as soliciting patients’ opinions, checking patient understanding, and encouraging patients to talk have been linked to reduction of malpractice claims.47 Poor communication, conversely, was associated with dissatisfaction, conflict, and worse outcomes. Studies suggested that dissatisfied patients tend to opt out of health plans,21 to change physicians,44,62 to initiate complaints against physicians,47 and to be noncompliant with medical recommendations.31,44
Women are more likely to get involved in decision making once they are given sufficient information about their medical options.30 These findings underscore the importance of educating women about the risks and benefits of various options. Studies show that without help, physicians are not consistently doing this well.8 Although there are some reputable decision aids available on the Internet, as well as risk information provided by the print lay press, there is also an abundance of misinformation to which women are exposed. Messages from direct-to-consumer advertisements about medical tests, procedures, or treatments can also be misleading (see Box 1-3 in Chapter 1). These advertisements tend to overemphasize breast cancer risks to women and the benefits that are likely to accrue if they pursue the medical options the ads publicize.34 The ads also tend to be fraught with misinformation, such as confusion of clinical benefits with laboratory accuracy.34,70
Individualized risk communication tends to improve women’s accuracy about their own risk, although different studies have reported that anywhere from 22 to 50 percent of the women studied still overestimate their risk.12 Edwards and colleagues reviewed 13 studies and concluded that individualized risk communication is also linked to increased participation in mammography screening programs.26,27 However, many studies have been based on the presumption that the goal of risk communication is to increase participation in screening services, whereas the more important
goal is to increase the number of women whose breast cancer is detected early enough to be effectively treated. Indeed, Edwards and his colleagues concluded that, based on the available data, increased use of mammography is not necessarily a consequence of more informed decision making.26,27,61
O’Connor and colleagues reviewed 200 decision aids, of which only 30 had been evaluated in methodologically valid clinical studies.56 Based on those 30 studies, they concluded that the decision aids improved subjects’ knowledge about their medical risks—although they did not necessarily influence their medical decisions. For example, the four breast cancer decision aids involved decisions about whether to undergo genetic testing for BRCA mutations. Those decision aids improved the test subjects’ understanding of their personal risk, but did not influence whether or not they intended to pursue genetic testing.
Although risk perception is often at odds with actual risk, numerous studies have shown that genetic risk counseling improves people’s understanding of their personal risk (reviewed by Hopwood in 2000).39 A systematic review of studies published from 1980 to 2001 on the effects of genetic counseling and testing for familial breast cancer on women’s perception of risk indicated that, overall, genetic counseling and testing appear to produce psychological benefits and to improve accuracy of risk perception, although 22 to 50 percent of the women in the studies reviewed continued to overestimate their risk.12
Even straightforward and accurate communication of risk can lead to unintended outcomes. For example, if people are asked to choose between an option that carries a 20 percent risk of dying versus an option that carries an 80 percent chance of survival, the overwhelming majority will opt for the survival option—even though the probable outcomes are identical. The differences in how the options are presented, or framed, are referred to as “loss framing” or “gain framing.” Women’s responses to information about the value of mammography are similarly affected by how the risks and benefits of mammography are framed.25 Communication of risk must ensure that women do not mistakenly identify themselves as being at such low risk that they make choices, such as foregoing mammograms entirely, that increase their risks of a preventable death from breast cancer.
SUMMARY
The ultimate purpose of this Institute of Medicine report is to identify better ways to reduce the burden of breast cancer through improving early detection and diagnosis. Because there is so much individual variation in susceptibility to breast cancer, it makes sense to develop more refined screening strategies that provide the greatest possible benefit for individual
women. Current screening strategies rely most heavily on age, followed by a history of breast disease. However, the development and progression of breast cancer is driven by biological factors such as genetic inheritance and mutations accumulated during a woman’s lifetime. Although much has been learned, research on the genetic risk factors for breast cancer is still in its infancy, but should, in time, increasingly yield the knowledge for individualized risk stratification.
The goal of improved risk assessment is not to increase the use of screening mammography, but rather to identify optimal strategies. For some women, that might mean fewer mammograms. For others, it might mean staying the course and getting annual mammograms after age 50. For still others, it might mean more frequent mammograms or the use of supplemental imaging technologies, such as magnetic resonance imaging or ultrasound, or eventually molecular imaging. The primary goal of national screening programs has been to maximize the number of women who receive regular mammograms, yet it is clear that not all women will benefit equally.
Risk assessment, however, is only the first step. The goal of revising screening strategies necessarily includes revising screening behaviors. Risk must be communicated to individual women (and understood by their physicians) in such a way that they can make informed decisions about screening and their lifestyle. Numerous studies have indicated that a physician’s or other provider’s referral is the single most important predictor of whether a woman will receive a mammogram. But as discussed earlier, this is correlated with a variety of other factors that influence access to mammography. One example is whether a women who receives a referral is already receiving regular health care and, in most cases, has health insurance, which is itself a major determinant of which women will receive regular mammograms (see section Equal Access in Chapter 3).
To date, the impact of risk communication on informed medical decision making is limited. Even for mammography, which has been the subject of much research on communicating risk, few data show that women are making informed decisions—even within programs to communicate individualized risk.27 This education is particularly relevant in enabling women to make appropriate decisions about their breast cancer screening because, as discussed earlier, a woman’s perception of her breast cancer risk often does not match her actual risk. Risk communication might increase participation in screening mammography for several reasons that are, in fact, contradictory to informed decisions. For example, a woman might be motivated to follow mammography guidelines, because she overestimates her personal risk, or because she overestimates the potential of mammography to reduce her risk.
Many women’s health and breast cancer advocates argue that women
must be enabled to make informed choices about screening,4,53,68 but that is not enough. Women and their physicians need better tools for assessing risk. Finally, communicators—physicians, professional societies, national health organizations, breast cancer advocates, and journalists—need a better understanding of how risk should be communicated.
REFERENCES
1. American Cancer Society. 2003. Breast Cancer Facts and Figures 2003-2004. Atlanta, GA: American Cancer Society.
2. American Medical Association, CME program publication. 2001. Identifying and Managing Hereditary Risk for Breast and Ovarian Cancer.
3. Aro AR, de Koning HJ, Absetz P, Schreck M. 1999. Psychosocial predictors of first attendance for organised mammography screening. J Med Screen 6(2):82-88.
4. Baines CJ. 2003. Mammography screening: are women really giving informed consent? J Natl Cancer Inst 95(20):1508-1511.
5. Begg CB. 2002. On the use of familial aggregation in population-based case probands for calculating penetrance. J Natl Cancer Inst 94(16):1221-1226.
6. Black WC, Nease RF Jr, Tosteson AN. 1995. Perceptions of breast cancer risk and screening effectiveness in women younger than 50 years of age. J Natl Cancer Inst 87(10):720-731.
7. Boyd NF, Dite GS, Stone J, Gunasekara A, English DR, McCredie MR, Giles GG, Tritchler D, Chiarelli A, Yaffe MJ, Hopper JL. 2002. Heritability of mammographic density, a risk factor for breast cancer. N Engl J Med 347(12):886-894.
8. Braddock CH 3rd, Edwards KA, Hasenberg NM, Laidley TL, Levinson W. 1999. Informed decision making in outpatient practice: time to get back to basics. JAMA 282(24):2313-2320.
9. Brekelmans CT, Seynaeve C, Bartels CC, Tilanus-Linthorst MM, Meijers-Heijboer EJ, Crepin CM, van Geel AA, Menke M, Verhoog LC, van den Ouweland A, Obdeijn IM, Klijn JG. 2001. Effectiveness of breast cancer surveillance in BRCA1/2 gene mutation carriers and women with high familial risk. J Clin Oncol 19(4):924-930.
10. Burke W, Austin MA. 2002. Genetic risk in context: calculating the penetrance of BRCA1 and BRCA2 mutations. J Natl Cancer Inst 94(16):1185-1187.
11. Burke W, Olsen AH, Pinsky LE, Reynolds SE, Press NA. 2001. Misleading presentation of breast cancer in popular magazines. Eff Clin Pract 4(2):58-64.
12. Butow PN, Lobb EA, Meiser B, Barratt A, Tucker KM. 2003. Psychological outcomes and risk perception after genetic testing and counselling in breast cancer: a systematic review. Med J Aust 178(2):77-81.
13. Carney PA, Miglioretti DL, Yankaskas BC, Kerlikowske K, Rosenberg R, Rutter CM, Geller BM, Abraham LA, Taplin SH, Dignan M, Cutter G, Ballard-Barbash R. 2003. Individual and combined effects of age, breast density, and hormone replacement therapy use on the accuracy of screening mammography. Ann Intern Med 138(3):168-175.
14. Cockburn J, Sutherland M, Cappiello M, Hevern M. 1997. Predictors of attendance at a relocatable mammography service for rural women. Aust N Z J Public Health 21(7):739-742.
15. Colditz GA, Rosner BA, Chen WY, Holmes MD, Hankinson SE. 2004. Risk factors for breast cancer according to estrogen and progesterone receptor status. J Natl Cancer Inst 96(3):218-228.
16. Collaborative Group on Hormonal Factors in Breast Cancer. 2001. Familial breast cancer: collaborative reanalysis of individual data from 52 epidemiological studies including 58,209 women with breast cancer and 101,986 women without the disease. Lancet 358(9291):1389-1399.
17. Cotterchio M, Kreiger N, Theis B, Sloan M, Bahl S. 2003. Hormonal factors and the risk of breast cancer according to estrogen- and progesterone-receptor subgroup. Cancer Epidemiol Biomarkers Prev 12(10):1053-1060.
18. Culver JB, Hull JL, Levy-Lahad E, Daly MB, Burke W. 2000, March 4. BRCA1 and BRCA2 Hereditary Breast/Ovarian Cancer. Accessed November 20, 2003. Web Page. Available at: http://www.geneclinics.org/servlet/access?db=geneclinics&site=gt&id=8888891&key=pxTA18SOqJ0uZ&gry=&fcn=y&fw=J1fF&filename=/profiles/brca1/index.html.
19. Cummings SR, Duong T, Kenyon E, Cauley JA, Whitehead M, Krueger KA. 2002. Serum estradiol level and risk of breast cancer during treatment with raloxifene. JAMA 287(2):216-220.
20. Daly MB, Lerman CL, Ross E, Schwartz MD, Sands CB, Masny A. 1996. Gail model breast cancer risk components are poor predictors of risk perception and screening behavior. Breast Cancer Res Treat 41(1):59-70.
21. Davies AR, Ware JE Jr, Brook RH, Peterson JR, Newhouse JP. 1986. Consumer acceptance of prepaid and fee-for-service medical care: results from a randomized controlled trial. Health Serv Res 21(3):429-452.
22. de Jong MM, Nolte IM, te Meerman GJ, van der Graaf WT, Oosterwijk JC, Kleibeuker JH, Schaapveld M, de Vries EG. 2002. Genes other than BRCA1 and BRCA2 involved in breast cancer susceptibility. J Med Genet 39(4):225-242.
23. Domchek SM, Eisen A, Calzone K, Stopfer J, Blackwood A, Weber BL. 2003. Application of breast cancer risk prediction models in clinical practice. J Clin Oncol 21(4):593-601.
24. Domenighetti G, D’Avanzo B, Egger M, Berrino F, Perneger T, Mosconi P, Zwahlen M. 2003. Women’s perception of the benefits of mammography screening: population-based survey in four countries. Int J Epidemiol 32(5):816-821.
25. Edwards A, Elwyn G, Covey J, Matthews E, Pill R. 2001. Presenting risk information—a review of the effects of “framing” and other manipulations on patient outcomes. J Health Commun 6(1):61-82.
26. Edwards A, Unigwe S, Elwyn G, Hood K. 2003. Personalised risk communication for informed decision making about entering screening programs. Cochrane Database Syst Rev (1):CD001865.
27. Edwards A, Unigwe S, Elwyn G, Hood K. 2003. Effects of communicating individual risks in screening programmes: Cochrane systematic review. BMJ 327(7417):703-709.
28. Ernster VL, Barclay J, Kerlikowske K, Wilkie H, Ballard-Barbash R. 2000. Mortality among women with ductal carcinoma in situ of the breast in the population-based surveillance, epidemiology and end results program. Arch Intern Med 160(7):953-958.
29. Ford D, Easton DF, Stratton M, Narod S, Goldgar D, Devilee P, Bishop DT, Weber B, Lenoir G, Chang-Claude J, Sobol H, Teare MD, Struewing J, Arason A, Scherneck S, Peto J, Rebbeck TR, Tonin P, Neuhausen S, Barkardottir R, Eyfjord J, Lynch H, Ponder BA, Gayther SA, Zelada-Hedman M, et al. 1998. Genetic heterogeneity and penetrance analysis of the BRCA1 and BRCA2 genes in breast cancer families. The Breast Cancer Linkage Consortium. Am J Hum Genet 62(3):676-689.
30. Ford S, Schofield T, Hope T. 2003. Are patients’ decision-making preferences being met? Health Expect 6(1):72-80.
31. Francis V, Korsch BM, Morris MJ. 1969. Gaps in doctor-patient communication. Patients’ response to medical advice. N Engl J Med 280(10):535-540.
32. Frank TS, Deffenbaugh AM, Reid JE, Hulick M, Ward BE, Lingenfelter B, Gumpper KL, Scholl T, Tavtigian SV, Pruss DR, Critchfield GC. 2002. Clinical characteristics of individuals with germline mutations in BRCA1 and BRCA2: analysis of 10,000 individuals. J Clin Oncol 20(6):1480-1490.
33. Gail MH, Brinton LA, Byar DP, Corle DK, Green SB, Schairer C, Mulvihill JJ. 1989. Projecting individualized probabilities of developing breast cancer for white females who are being examined annually. J Natl Cancer Inst 81(24):1879-1886.
34. Gollust SE, Hull SC, Wilfond BS. 2002. Limitations of direct-to-consumer advertising for clinical genetic testing. JAMA 288(14):1762-1767.
35. Goode EL, Ulrich CM, Potter JD. 2002. Polymorphisms in DNA repair genes and associations with cancer risk. Cancer Epidemiol Biomarkers Prev 11(12):1513-1530.
36. Grunfeld EA, Ramirez AJ, Hunter MS, Richards MA. 2002. Women’s knowledge and beliefs regarding breast cancer. Br J Cancer 86(9):1373-1378.
37. Hankinson SE, Willett WC, Manson JE, Colditz GA, Hunter DJ, Spiegelman D, Barbieri RL, Speizer FE. 1998. Plasma sex steroid hormone levels and risk of breast cancer in postmenopausal women. J Natl Cancer Inst 90(17):1292-1299.
38. Heine JJ, Malhotra P. 2002. Mammographic tissue, breast cancer risk, serial image analysis, and digital mammography. Part 2. Serial breast tissue change and related temporal influences. Acad Radiol 9(3):317-335.
39. Hopwood P. 2000. Breast cancer risk perception: what do we know and understand? Breast Cancer Res 2(6):387-391.
40. Huang WY, Newman B, Millikan RC, Schell MJ, Hulka BS, Moorman PG. 2000. Hormone-related factors and risk of breast cancer in relation to estrogen receptor and progesterone receptor status. Am J Epidemiol 151(7):703-714.
41. Institute of Medicine. 2001. Mammography and Beyond: Developing Technologies for the Early Detection of Breast Cancer. Washington, DC: National Academy Press.
42. Kaklamani VG, Hou N, Bian Y, Reich J, Offit K, Michel LS, Rubinstein WS, Rademaker A, Pasche B. 2003. TGFBR1*6A and cancer risk: a meta-analysis of seven case-control studies. J Clin Oncol 21(17):3236-3243.
43. Kaplan SH, Greenfield S, Ware JE Jr. 1989. Assessing the effects of physician-patient interactions on the outcomes of chronic disease. Med Care 27(3 Suppl):S110-S127.
44. Kasteler J, Kane RL, Olsen DM, Thetford C. 1976. Issues underlying prevalence of “doctor-shopping” behavior. J Health Soc Behav 17(4):329-339.
45. King MC, Marks JH, Mandell JB. 2003. Breast and ovarian cancer risks due to inherited mutations in BRCA1 and BRCA2. Science 302(5645):643-646.
46. Kushi LH, Potter JD, Bostick RM, Drinkard CR, Sellers TA, Gapstur SM, Cerhan JR, Folsom AR. 1995. Dietary fat and risk of breast cancer according to hormone receptor status. Cancer Epidemiol Biomarkers Prev 4(1):11-19.
47. Levinson W, Roter DL, Mullooly JP, Dull VT, Frankel RM. 1997. Physician-patient communication. The relationship with malpractice claims among primary care physicians and surgeons. JAMA 277(7):553-559.
48. McKiernan F, Wiley C. 2002. Measurement of serum estradiol. JAMA 287(12):1528.
49. Medstat. 2003. Mammography Use Declines When Breast Cancer Risk is Greatest. Accessed November 2003. Web Page. Available at: http://www.medstat.com/1news/093003.asp.
50. Metcalfe KA, Narod SA. 2002. Breast cancer risk perception among women who have undergone prophylactic bilateral mastectomy. J Natl Cancer Inst 94(20):1564-1569.
51. MORI Medicine & Science Research. 2002, October 17. Women See Family History Not Old Age as Greatest Breast Cancer Risk [MORI is an approved survey firm for the British National Health Service]. Accessed May 13, 2003. Web Page. Available at: http://www.mori.com/polls/2002/breakthrough.shtml.
52. Mosca L, Jones WK, King KB, Ouyang P, Redberg RF, Hill MN. 2000. Awareness, perception, and knowledge of heart disease risk and prevention among women in the United States. American Heart Association Women’s Heart Disease and Stroke Campaign Task Force. Arch Fam Med 9(6):506-515.
53. National Breast Cancer Coalition. 2003, March. Position Statement on Screening Mammography. Accessed November 25, 2003. Web Page. Available at: http://www.natlbcc.org.
54. National Cancer Institute. 2004, March 11. Genetics of Breast and Ovarian Cancer (PDQ(R)). Accessed April 21, 2004. Web Page. Available at: http://www.nci.nih.gov/cancerinfo/pdq/genetics/breast-and-ovarian#Section_1.
55. Nekhlyudov L, Partridge A. 2003. Breast cancer risk communication: challenges and future research directions: workshop report (United States). Cancer Causes Control 14(3):235-239.
56. O’Connor AM, Stacey D, Rovner D, Holmes-Rovner M, Tetroe J, Llewellyn-Thomas H, Entwistle V, Rostom A, Fiset V, Barry M, Jones J. 2003. Decision aids for people facing health treatment or screening decisions. Cochrane Database Syst Rev (3): CD001431.
57. Rahman N, Stratton MR. 1998. The genetics of breast cancer susceptibility. Annu Rev Genet 32:95-121.
58. Rakovitch E, Franssen E, Kim J, Ackerman I, Pignol JP, Paszat L, Pritchard KI, Ho C, Redelmeier DA. 2003. A comparison of risk perception and psychological morbidity in women with ductal carcinoma in situ and early invasive breast cancer. Breast Cancer Res Treat 77(3):285-293.
59. Ries LAG, Eisner MP, Kosary CL, Hankey BF, Miller BA, Clegg L, Mariotto A, Fay MP, Feuer EJ, Edwards BK, Editors. 2003. SEER Cancer Statistics Review, 1975-2000. Bethesda, MD: National Cancer Institute.
60. Rockhill B, Spiegelman D, Byrne C, Hunter DJ, Colditz GA. 2001. Validation of the Gail et al. model of breast cancer risk prediction and implications for chemoprevention. J Natl Cancer Inst 93(5):358-366.
61. Royak-Schaler R, Klabunde CN, Greene WF, Lannin DR, DeVellis B, Wilson KR, Cheuvront B. 2002. Communicating breast cancer risk: patient perceptions of provider discussions. Medscape Womens Health 7(2):2.
62. Rubin HR, Gandek B, Rogers WH, Kosinski M, McHorney CA, Ware JE Jr. 1993. Patients’ ratings of outpatient visits in different practice settings. Results from the Medical Outcomes Study. JAMA 270(7):835-840.
63. Rubin R. 2003, November 10. New agony over breast cancer. USA TODAY. P. 5.
64. Singletary SE. 2003. Rating the risk factors for breast cancer. Ann Surg 237(4):474-482.
65. Stahlberg C, Pedersen AT, Lynge E, Andersen ZJ, Keiding N, Hundrup YA, Obel EB, Ottesen B. 2004. Increased risk of breast cancer following different regimens of hormone replacement therapy frequently used in Europe. Int J Cancer 109(5):721-727.
66. Taylor MRG. 2001. Genetic testing for inherited breast and ovarian cancer syndromes: important concepts for the primary care physician. Postgrad Med J 77(903):11-15.
67. Theisen C. 2003. For patients, prediction of cancer risk can be worrisome. J Natl Cancer Inst 95(18):1360-1361.
68. Thornton H, Edwards A, Baum M. 2003. Women need better information about routine mammography. BMJ 327(7406):101-103.
69. Ursin G, Ma H, Wu AH, Bernstein L, Salane M, Parisky YR, Astrahan M, Siozon CC, Pike MC. 2003. Mammographic density and breast cancer in three ethnic groups. Cancer Epidemiol Biomarkers Prev 12(4):332-338.
70. Woloshin S, Schwartz LM, Tremmel J, Welch HG. 2001. Direct-to-consumer advertisements for prescription drugs: what are Americans being sold? Lancet 358(9288): 1141-1146.
71. Wooster R, Weber BL. 2003. Breast and ovarian cancer. N Engl J Med 348(23):2339-2347.
72. Yaffe MJ, Boyd NF, Byng JW, Jong RA, Fishell E, Lockwood GA, Little LE, Tritchler DL. 1998. Breast cancer risk and measured mammographic density. Eur J Cancer Prev 7(Suppl 1):S47-S55.
73. Ziv E, Shepherd J, Smith-Bindman R, Kerlikowske K. 2003. Mammographic breast density and family history of breast cancer. J Natl Cancer Inst 95(7):556-558.