3
Improving Breast Cancer Screening Services
The controversy over mammography is often focused on whether or not it should be used as a screening tool. But another equally important issue, given its widespread use, is the optimization of mammography…. Considerable effort should, therefore, be devoted to determining how to make mammography as effective as it can be and to reduce the tremendous variation in interpretation and biopsy rates.38
Laura Esserman and colleagues
This chapter examines critical issues in providing high-quality breast screening services. The fundamental criterion for implementing a screening program for all women in a particular target group is that the screening tests should have an acceptable level of accuracy, cost effectiveness, and a favorable balance of benefits to harms. Although different programs might place relatively greater emphasis on detecting small tumors or on reducing the false-positive rate, there is little disagreement that achieving the highest practical balance between sensitivity and specificity is central to ongoing efforts to improve the quality of mammography services. This chapter reviews alternative approaches to the organization of breast screening services, ways that mammography could be improved, technologies that might augment or replace mammography in breast cancer screening, and the challenges in supporting and developing a well-trained workforce.
SCREENING OUTCOMES VARY BY COUNTRY
Nearly a dozen countries have national or regional screening programs in which personal invitations for regular mammograms are sent to all women over age 40 or 50, depending on the country (Table 3-1).74 There are international differences in breast cancer detection patterns and mortality. Examination of how these patterns are influenced by the organization of breast cancer screening should indicate ways to improve quality.
TABLE 3-1 Breast Screening Programs in Different Countries*
|
|
United States |
Canada |
|
Year screening program started |
1988 (Medicare) |
1988 (British Columbia was the 1st province) |
|
Age of women screened (target population) |
40 and older, generally until 75 |
50 to 69 |
|
Screening interval (years) |
1-2 |
2ii |
|
Percent of target population screened |
55-63%iv |
54%v |
|
Referral type |
Doctor or self-referral |
Doctor or self-referral |
|
Double readingx |
Somexi |
No |
|
Number of views |
2 |
2 |
|
Quality enforcement |
National law (MQSA)xiv |
Voluntary accreditationxv |
|
Quality assurance site visits |
Yes |
No |
|
Level of organization |
Medicare is national; otherwise based on state and private insurance provider policiesxx |
Provincexxi |
|
*Finland, Luxembourg, and Japan also have national mammography screening programs; Italy, Spain, and Norway have regional programs. iSwedish governments makes guidelines, but standards and practices are organized at the county level. iiThe Canadian Province of British Columbia offers annual mammograms. iiiScreening interval established individually by county. ivBased on year 2000 data from Behavioral Risk Factor Surveillance System Public Use Data Tape (CDC, 2001). Also National Health Interview Survey, 2000 (CDC 2002); women over 40, and mammograms in last year; ACS Breast Cancer Facts and Figures 2003-2004. vBased on 1996 data; Paquette et al. (2000); mammogram in past 2 years. viBased on 2001-2002 data; NHS Breast Screening Programme Annual Review 2003. viiBased on WE trial 1977-1979; attendence after first invitation; Lynge et al. viiiShould be range b/c age policies differ; WE trial data is old (1977) and small sample size, but all studies go back to this same ref. ixBased on 1990-1995 data; Facheboud et al., 1998, Int. J. Cancer. xISBN http://appliedresearch.cancer.gov/ibsn/data/double.html. Accessed February 4, 2004. xiThe United States does not require double reading of mammograms, but the practice is common. However, overall, double reading of screening mammograms is less common in the United States than other countries. |
||
|
United Kingdom |
Swedeni |
The Netherlands |
Australia |
|
1988 |
1986 |
1989 |
1994 |
|
50 to 70 |
Beginning at 40; ending at 64 or 74 |
50 to 75 |
40 to 79 |
|
3 |
1 1/2-2, depending on ageiii |
2 |
2 |
|
76%vi |
78%16 |
54 |
|
|
Invite |
Invite |
Invite |
Invite or self-referral |
|
No |
Yes |
Yes |
Yes |
|
2xii |
2 |
2xiii |
2 |
|
Voluntaryxvi |
National lawxvii |
National lawxvooo |
National accreditation requirementsxix |
|
Yes |
Yes |
Yes |
Yes |
|
National |
County (Swedish counties are comparable to stated in the U.S.) |
National |
National |
|
xiiInitial mammogram only; NHS reports as of December 2003 86% of local screening services are doing two-view mammographies. xiiiInitial only. xivThe Mammography Quality Standards Act ensures x-ray technical quality but does not review quality of interpretation. xvCanadian Association of Radiologists. xviRadiographic Quality Control Manual for Mammography. xviiNational Swedish law based on European Guidelines for Quality Assurance in Screening Mammography. xviiiDutch Technical Protocol for Quality Control. xixExternal quality assurance program must be established to receive funding. xxNo national policy. Contains aspects of both national centralized care (Medicare) and decentralized regional care (private). xxiCanadian government sets has national guidelines, but standards and service organization are set at the provincial level. |
|||
Most women who undergo biopsies will not have breast cancer. Although some might describe these biopsies as “needless,” in reality they reflect the lack of precision of current detection methods. Some of the imprecision is likely due to the quality of the mammographic interpretation, and some is due to the inherent limitations of the technology. Some solutions to the problem lie in organizational changes, such as adopting different procedures for interpreting mammograms, different standards, and different ways of organizing mammography services. Other solutions might lie in technological improvements.
Screening for breast cancer is organized differently in different countries. A close comparison between the different countries and the results offers some useful insights into strategies for reducing breast cancer mortality in the United States. Mortality is influenced by screening patterns, as well as patterns of care.
Screening programs can be compared according to a variety of measures, such as differences in breast cancer survival rates, rates of abnormal mammograms, or rates of false positives. But there are caveats to each of these measures.
A 2003 study reported that 5-year survival rates for all breast cancers are higher in the United States (89 percent) than in Europe (79 percent), but this was based on data from a heterogeneous group of countries including those with national or regional screening programs (Italy, Spain, The Netherlands, and the United Kingdom) and those without (Estonia and France).101 The study, which compared the United States Surveillance Epidemiology and End Results (SEER) data set with the comparable EUROCARE data set, revealed that breast cancer survival was higher for women in the United States than in Europe, at least for breast cancers diagnosed between 1990 and 1992.a (During the first decade of a service screening program, most breast cancer deaths will occur in women who were diagnosed before the program started which means that a reduction in breast cancer mortality will only emerge when most of the breast cancers in the target population have been screen detected.74) Five-year survival was 89 percent for women in the United States and 79 percent for women in Europe.101 Most of the difference in survival rates was due to the stage at which women were diagnosed. Forty percent of tumors in the SEER data set were early stage (T1N0M0) compared with only 30 percent in the EUROCARE set.b The authors attribute these differences to the availability
of diagnostic and treatment facilities and to the effectiveness of the different health care systems.102 The frequency with which different breast cancer treatments were used, such as the frequency of axillary node dissection, breast-conserving surgery, and the modified radical Halstead mastectomy, varied two- to three-fold among countries.100 Although the relative contributions of earlier detection and state-of-the-art therapy are difficult to quantify, a recent study concluded that early detection through screening had probably contributed more to the reduction of mortality rates than had improvements in therapy.115 The study, which was conducted in Sweden, compared the results of women who participated in screening with those who did not. Cancers detected in the women who participated in screening were detected at an earlier stage and were less likely to have invaded the lymph nodes, which gained them a prognostic advantage over women whose cancer was not screen-detected but was presumably detected through physical exam or development of symptoms.
There is substantial variation between countries, as well as within the United States, in the frequency that mammograms are identified as abnormal (Figure 3-1). A review of 32 studies showed that the screening programs with high rates of abnormal mammograms also tended to be those with lower positive predictive values for biopsies, suggesting that many of those biopsies could have been safely avoided.36
The data collected in the screening studies reviewed do not permit determination of the underlying causes of the variation in the percentage of mammograms that are judged to be abnormal and the predictive value of biopsies. Possible sources of variation include:
-
Characteristics of the population that was screened, including the age distribution, and the proportion of women being screened for the first time (prevalence screens) versus those who have been screened before (incidence screens).
-
Features of the mammography examinations (such as screening interval, number of views per breast, use of single versus double readings, and availability of prior films for comparison).
-
Features of physicians interpreting the mammogram (such as experience or comfort with ambiguity).
-
Features of the health care system (such as malpractice concerns, financial incentives, or national policies).
Finally, although this review emphasizes international variation, the considerable variation in performance within the United States is also worth noting, suggesting that international differences such as universal access to health care, more centralized health care systems, and high cost of malpractice litigation account for only part of the differences in screening program
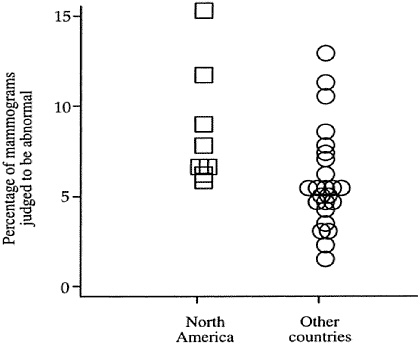
FIGURE 3-1 Frequency of abnormal mammograms in North America compared to other countries. These data are based on a review of 32 screening mammography studies.36 With the exception of one point that indicates a study from British Columbia, Canada, all points listed for North America are from studies conducted in the United States.
performance. Although on average there are fewer “excess” biopsies in European screening programs than in the United States (at least those programs that were reviewed in the study, which was conducted in the late 1980s and 1990s), there are also European programs that appear to be worse, at least by this measure.
In 1988, the United Kingdom had the highest breast cancer mortality rate in Europe. That same year, the national breast screening program was established, and it is now one of the most well-established, well-analyzed, and extensive screening programs in the world. Since then, the United Kingdom has had the greatest reduction in breast cancer mortality for Europe.19
There are several important differences in the delivery of breast screening services in the United States and other countries (Table 3-2). The main programmatic differences between the United States and Britain are:
-
All women in Britain receive invitations for screening mammograms
-
at regular intervals, whereas the large majority of women in the United States are referred by a health care provider or by themselves.
-
The British National Health Service pays for all screening mammograms in the United Kingdom, whereas mammograms in the United States are covered through a complex patchwork of medical payment systems that exclude millions of women.
-
The volume of mammographic interpretations required of radiologists to be eligible to read mammograms in the United States is about one-tenth that required in Britain.
-
The recommended interval for screening mammography is 12 months in the United States and 36 months in Britain.
-
Quality assurance standards concerning mammographic interpretation for the National Health Service Breast Screening Program are set nationally and are regularly monitored through a quality assurance network.
Although the threat of malpractice is frequently cited as an important reason for the difference in screening practices between the two countries, this is part of the larger context of the health care and can not be regulated through breast cancer screening programs. (Problems of malpractice in the United States are discussed later in this chapter in the section Breast Imagers Needed.)
Table 3-2 summarizes the different outcomes of the breast screening programs in the United States and Britain. The results in Table 3-2 are only valid for comparison within the same study which directly compares the two countries because of the similar methodology used in collecting the data; other studies with different methodology may result in different statistics. Overall, women in the United States are called back after screening mammograms about twice as often as women in Britain and significantly more of the surgical biopsies they undergo turn out to be negative. But this does not translate into improved rates of cancer detection, which are not significantly different between the countries. It could be argued that women in the United States are excessively subjected to unnecessary medical procedures.
Yet, the fact that breast cancer mortality rates in the United States are lower than they are in Britain must be considered. Although differences in treatment quality cannot be ruled out, there is a more immediate reason to expect higher breast cancer mortality in Britain. Breast cancers are detected at a later stage in Britain, and stage of detection is well established as a factor in survival. The three-fold difference in screening intervals between Britain and the United States is highly likely to be a significant contributor to the differences in mortality between the two countries. Longer screening intervals are associated with more false positives, as well as in increase in
TABLE 3-2 Comparison of Screening Mammography Outcomes in the United States and Britain
|
|
United States |
Britain |
Source |
Comments |
|
Sensitivity |
77% (>300/month) 70 % (≤ 300/month) |
79% (>300/month) |
Esserman et al., 2002 JNCIi |
Used enriched test set; U.S. figures are for high-volume radiologists |
|
Specificity |
88% (>300/month) |
88% |
|
Volume was not significantly correlated with specificity for any of the groups. |
|
Mammograms judged to be abnormal at 1st screen |
12.0% |
7.4% |
Smith-Bindman, 2003 JAMA |
U.S. value = median of estimates from two data sets (11.2-13.1) |
|
Mammograms judged to be abnormal at later screens |
7.4% |
3.6% |
|
U.S. value = median of estimates from two data sets (6.8-8.0) |
|
Mammograms judged to be abnormal |
6.9% |
4.9% |
Elmore et al., 2003 JNCI |
Values are medians of estimates for three or more studies; included both 1st and subsequent screening mammograms |
|
Women with abnormal mammograms later diagnosed with breast cancer |
7.6% |
12.3% |
|
Values are medians of estimates for three or more studies |
|
Negative biopsies |
73% |
40% |
|
Included all types of biopsy |
|
Biopsy rates/100 screening mammograms for later screens |
0.33 |
0.28 |
Smith-Bindman, 2003 JAMA |
Differences are not significant |
the frequency of late-stage cancers. Although there are many aspects of the British national screening program that should be considered for adoption in the United States, the 3-year screening interval is not one of them. Mammography services in both Sweden and several Canadian provinces also have high performance standards, but there are fewer published data and direct comparisons with services in the United States, so they are not reviewed here. The British Health Service monitors and tracks the outcomes of their breast cancer screening programs more thoroughly than do other countries.74
QUALITY ASSURANCE IMPROVES OUTCOMES
The National Health Service Breast Screening Programme (NHSBSP) in the United Kingdom has integrated quality assurance into all clinical aspects of its programs (Box 3-1).107 Ranges of acceptable performance for
|
BOX 3-1 The United Kingdom is divided into 11 National Health Service regions, each of which is supported by a quality assurance reference center that collects and collates data about the performance and outcomes of the breast screening program, organizes quality assurance visits, and provides support for the regional director of quality assurance and the professional coordinators. Each region has a quality assurance director for breast screening and a quality assurance reference center. Each regional quality assurance director is supported by a regional quality assurance team, which includes a professional coordinator from each of the professions that contribute to the breast screening program (radiology, radiography, pathology, surgery, breast care nursing, administration, and medical physics). Each professional coordinator meets regularly with colleagues in the region to review the performance and outcomes of the breast screening program, to share good practice, and to encourage continued improvements in the program. There is also a program of regular quality assurance visits to breast screening units. Regional quality assurance directors and professional coordinators meet regularly in a series of national coordinating committees. The committees produce guidance on good practice and set standards and targets for staff working in the breast screening program and for the technical performance of equipment. National standards and targets for the performance and outcomes of the program are also published. SOURCE: See http://www.cancerscreening.nhs.uk/breastscreen/quality-assurance.html. Accessed March 4, 2004. |
|
BOX 3-2 PERFORMS (PERsonal perFORmance in Mammographic Screening) is an integral part of quality assurance for breast cancer screening in the United Kingdom. It is a self-assessment program for mammogram interpretation, developed in 1991 and funded by the NHSBSP. As of 2003, it is the only system of its kind in the world.89 The PERFORMS program film set is released early each year. It contains 2 film sets, each with 60 two-view cases (mediolateral oblique and cranio-caudal). Up to 90 percent of U.K. radiologists use the PERFORMS system to assess their mammogram interpretation skills.124 PERFORMS results indicate the number of malignant cases a radiologist missed in the testing film set and whether they showed any patterns in the types of cases they missed, such as dense mammograms or mammograms with many microcalcifications. Pathology information is also provided where appropriate. Particular film sets allow the individual to see a large number of examples of one particular abnormality and have been shown to improve radiologists’ detection of these specific features.124 Additional advanced training sets are also available that concentrate on the types of cases that the radiologists were most likely to misinterpret. Analysis of the PERFORMS data can provide the participating radiologist with insight into how they perform in comparison with their anonymous colleagues. In addition, the program also can provide details concerning the specific cases that a radiologist incorrectly recalled for further assessment (false positives) or incorrectly identified as normal (false negative). Targeted training with the cases producing disagreement may achieve a higher level of consensus and reduce clinically important inconsistencies.10 An individual’s results are anonymous and are made available only to the radiologist who takes the test—although, for quality assurance purposes, the results can also be collated to provide anonymous regional or national results. |
recall, biopsy, and cancer detections rates have been established and an organized program operates at the local and national levels to monitor and achieve these targets. All screening programs in the United Kingdom receive data that enable a comparison of their recall and cancer detection rates with other programs. Both programs and individual radiologists below a minimum standard are subject to quality assurance. In contrast, the United States has only voluntary guidelines and there is no national organization to collect or monitor data to promote high levels of performance. Finally, an organized program of professional development in the United Kingdom specifically provides instruction related to mammography interpretation (Box 3-2). Although, a self-testing program exists in the United States, it is not widely used.
Strategies for skills improvement have been much discussed among breast imagers, advocates, and policymakers. The American College of Radiology (ACR) began offering a self-assessment program called Mammography Interpretive Skills Assessment in 1999, but there is no requirement for radiologists to use this voluntary program, and—as was the case with the voluntary Mammography Accreditation Program that preceded the Mammography Quality Standards Act (MQSA)—many do not use it. (The effectiveness of the ACR self-testing program has not been tested or compared with the PERFORMS program used in the United Kingdom.)
Experience with the MQSA demonstrates that a national quality assurance program could be successful in the United States. The MQSA led to nationwide improvements in the technical quality of mammography and it is reasonable to predict that a quality assurance program could be designed to improve the delivery of mammography services in the United States. This would include, but not be limited to, efforts to improve the quality of mammographic interpretation. Organization of services that are integrated and efficient—possibly through regionalization of certain tasks—are equally important and are discussed further in Chapter 7.
However, the MQSA is not without its critics. Even though there is little debate that the technical quality of mammography and consistency among practices has improved since its inception, compliance with MQSA regulations imposes a heavy tax on mammography facilities. In fact, the reauthorization of the MQSA in February 2004 included a mandates for the Institute of Medicine (IOM) to study ways that the MQSA could be improved both to improve the quality of mammographic interpretation and to ensure an adequate workforce.
Adopting Best Practices from Other Countries Can Save Lives
Mammographic screening services in the United States are typically separated from treatment, counseling, and other support services. Screening is poorly integrated into routine health care and tends to be more opportunistic than organized.108 In contrast, the European Code Against Cancer stresses that breast screening should be organized as part of integrated breast care centers.18
Another option, which has been shown to reduce the frequency of false positives, is to mandate second opinions by experts before biopsy is performed.81 Screening could take place in outlying centers with diagnosis and workup in centralized facilities. The Netherlands and Sweden have organized their service in this way, and they have thereby achieved very low rates of false positives.
Many features of European screening programs, if adopted in the United States, could improve screening. These features include invitations to screen-
ing, double reading of mammograms, and the organization of services in centralized high-volume facilities. Such services would include centralized facilities for interpreting mammograms and other screening data, whether it would be ultrasound, magnetic resonance imaging (MRI), or other new technologies once they are developed and validated. Centralization should not, however, involve reducing access to screening services. Consolidation of interpretation facilities does not need to coincide with consolidation of facilities that women attend for mammograms. In cases where traveling long distances might limit attendance at screening facilities, image acquisition and image interpretation could be conducted at separate locations.
Callback rates in mammography screening can be reduced when mammograms are read by breast imaging specialists at a central location, as opposed to having them dispersed among the sites where the mammography is done. By centralizing the reading, the mammography service reduced the overall callback rate by 2 percent, from 11 to 9 percent, which was statistically significant.76
On the other hand, the quality of a breast cancer screening program cannot be measured solely by the recall rate or the cancer detection rate, although these are important considerations. It is the rate of detection of small early stage, node-negative tumors that provides the greatest opportunity to save lives. Larger tumors are less often confused with normal breast structures and are less likely to be missed or to be false positives, and thus fewer women undergo unnecessary follow-up.109
Improving Screening Practices Can Reduce Health Care Costs
Aggregate costs of screening mammography in the United States are more than $3 billion, and cost savings in screening practices could have a significant impact.22 The average cost of a diagnostic workup following a false-positive mammogram is about $500 per case.29 About 40 million women in the United States are screened each year for breast cancer, which means that if the percentage of mammograms judged to be abnormal were reduced from 10 to 5 percent, 200,000 fewer women would be called back for follow-up work every year, which would translate into an annual savings of $100 million.
Equal Access Is a Component of Quality
In addition to considering how mammography should be organized to deliver optimal quality, it is essential to optimize access to services. Because access to health care in the United States is so uneven, it is important to consider not only the internal organization of a screening service, but also
how well it accommodates the financial, cultural, and educational situations of those it needs to serve.
Breast cancer mortality rates in the United States vary by race and ethnicity, and the gap between white and African American women is striking (Figure 3-2). Although the incidence of breast cancer among white females is higher than among other racial or ethnic groups, African-American women—particularly those older than age 64—have the highest risk for mortality from breast cancer.99 (The racial and ethnic categories included in this database were white, African American, Asian/Pacific Islander, American Indian/Alaskan Native, and Hispanic.) Similarly, 88 percent of white women survive at least 5 years after diagnosis, as compared with 73 percent of African-American women. Stage-specific survival rates do not differ significantly between the two groups.28
Group disparities in mammography rates may result from a broad combination of socioeconomic and cultural influences. Low socioeconomic status is characterized by low income, higher rates of poverty, lower levels of education, lack of private health insurance, lack of transportation, and lack of access to health care. Together, these factors are associated with lower rates of cancer screening, higher probability for later stage diagnosis, lack of breast health awareness, and mistrust and misunderstanding of the health care system.28,60,61,71,91,95 Higher poverty rates among African Americans are reflected in disproportionate numbers of women lacking adequate insurance, or any insurance at all.47,90 Insurance coverage is a significant predictor of whether or not a woman will receive a mammogram.57 Uninsured women and women with Medicaid are more likely to receive a breast cancer diagnosis at a late stage of disease, and are 30 to 50 percent more likely to die of their disease than women with private insurance.57
Yet when white, African-American, and Hispanic women were provided equal access to high-quality mammographic screening, all groups had similar rates of breast cancer survival regardless of age, stage of diagnosis, and socioeconomic status.123 These circumstances are, however, far from typical (Figure 3-3).
Social factors that restrict access to health care appear to contribute to racial and ethnic differences in breast cancer mortality. Biology may also play a role; even among women who have equal access to health care, fewer African Americans than whites are diagnosed with early stage breast disease, and more African Americans are diagnosed with advanced stage cancer.123 The peak age for incidence of breast cancer among African American women is 40 to 49, while among white women it is 50 to 59.90,91 The incidence of estrogen receptor-negative and progesterone receptor-negative tumors, which tend to be aggressive, is also significantly higher among African American women than among whites.61,77,90
Social injustice, in the form of social or institutional discrimination, can
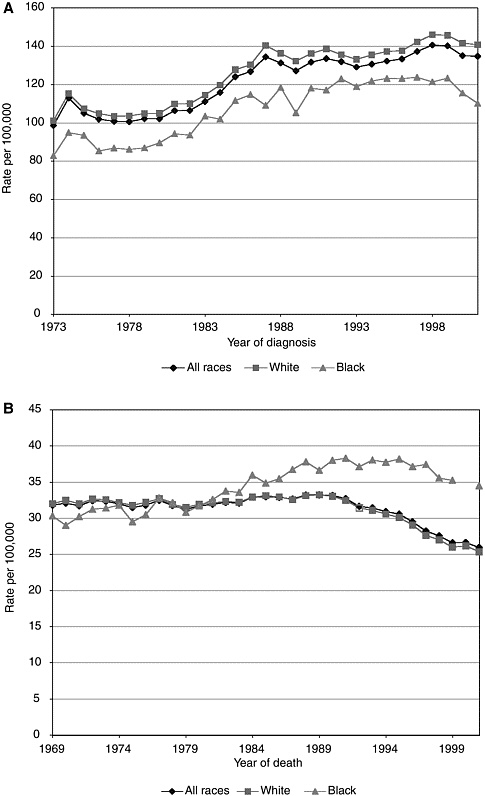
FIGURE 3-2 Breast cancer incidence (A) and mortality (B) in white and black women.114 Statistics were generated from malignant cases only.
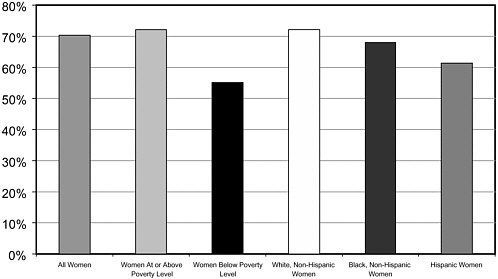
FIGURE 3-3 Poverty is a greater barrier to mammography than race or ethnicity.
SOURCE: National Center for Health Statistics, 2003.
frustrate screening attempts and create barriers for women seeking preventive screenings.45,50 African American women may also face cultural barriers to obtaining a mammogram, including false beliefs about cancer, traditions that discourage seeking medical care, and difficulties in communicating with their physicians.8,58,61,72,82,83
A number of programs have been initiated since 1990 to reduce the financial barriers to mammography rates in all groups. In 1991, Medicare began to cover part of the cost of screening mammograms, but screening rates failed to increase. In 1998, the co-payment and deductibles for the Medicare screening services were eliminated. All women aged 65 and older are eligible for Medicare and 94 percent of them choose the option. Yet, as noted earlier, these are the African-American women who suffer the greatest racial disparity in breast cancer mortality, suggesting that the lack of health insurance is not the predominant cause.
The Centers for Disease Control and Prevention’s (CDC’s) National Breast and Cervical Cancer Detection Program was launched in 1990 to provide screening services for uninsured women who were not eligible for Medicaid. Since then it has provided nearly 4 million screenings to 1.6 million women. In principle, this program should reduce the disparities in mortality that arise from lack of health insurance, although it has never been funded well enough to cover all, or even most, eligible women. For example, in 2002 approximately 400,000 women received at least one Pap test, mammogram, or clinical breast exam through the CDC program.26 By
comparison, approximately 9 million women between the ages of 40 and 65 lacked health insurance that year. (Women over 65 are eligible for Medicare and so would have little need for this program.)
Community-based programs, such as the North Carolina Breast Cancer Screening Program, also disseminate information about prevention and guide women to mammographic services.32
Equal access is a prerequisite for reducing the unequal burden of breast cancer, but other factors that contribute to equal use of health care services are also critical and must be taken into account.
IMPROVING MAMMOGRAPHY
Quality Assurance by Law
Mammography is possibly the most heavily legislated medical procedure in history. Between 1980 and 1994 alone, 43 state laws were passed concerning different aspects of screening for breast cancer.44 As a rule, state laws regulating mammography have been enacted before federal mammography laws. For example, 33 states had already passed laws supporting treatment and care following the detection of a breast malignancy when a similar federal law, the Breast and Cervical Cancer Prevention and Treatment Act (BCCPTA), was passed in 2000. Currently, more than 10 federal laws specifically address breast cancer screening, including laws governing quality and access (Box 3-3).
When Congress enacted the Medicare program as part of the Social Security Amendments in 1965, preventive services were explicitly excluded. (Although breast screening does not prevent the occurrence of breast cancer, it is considered secondary prevention because early detection can prevent deaths from breast cancer and, thus, mammography is considered a preventive service.) Since 1965, many preventive services have been added, but each addition requires a specific Act of Congress. Not every benefit recommended by experts has been added, and some have been added that were not recommended (such as bone density and prostate serum antigen [PSA] screening). Mammography benefits were initially included in the Medicare Catastrophic Act of 1988, which was repealed the following year, but subsequently included in the 1990 Budget Reconciliation Act. Women’s health and breast cancer advocacy groups were instrumental in the inclusion of mammography benefits.43
A Sweeping Act for Technical Quality
The MQSA was enacted in 1992 to ensure that all women have access to quality mammography for the detection of breast cancer in its earliest,
|
BOX 3-3 2003 Reauthorization of MQSA Legislation reauthorized for two years awaiting the findings of two Congressionally mandated reports. The first by the General Accounting Office to assess the States as Certifiers program and mammography access issues. The second by the Institute of Medicine on ways to improve physician recruitment, mammography interpretation, and mammography services. 2003 Medicare Prescription Drug and Modernization Act Created a prescription drug benefit for Medicare beneficiaries. The legislation also increased payments for mammography and provided an additional $10 million in funding for the National Breast and Cervical Cancer Early Detection Program, bringing the total to $220 million. The program is intended to provide 32,000 diagnostic and screening services to additional women who are hard to reach and have never been screened for these cancers. 2001 Native American Breast and Cervical Cancer Treatment Technical Amendment Act Amended Title XIX of the Social Security Act to clarify that Indian women with breast or cervical cancer who are eligible for health services provided under a medical care program of the Indian Health Service or of a tribal organization are included in the optional Medicaid eligibility category of breast or cervical cancer patients added by the BCCPTA of 2000. 2000 BCCPTA Gave States the option of providing medical assistance (Medicaid) for breast and cervical cancer-related treatment services to certain low-income women without creditable coverage who have already been screened for such cancers under the CDC breast and cervical cancer early detection program, and who need treatment. 1998 Reauthorization of MQSA
1997 Veterans’ Benefits Act Directed the Under Secretary for Health to develop a national policy for the Veterans Health Administration on mammography screening for veterans. 1997 Stamp Out Breast Cancer Act Authorized the U.S. Postal Service to issue a first-class breast cancer stamp. Seventy percent of the net amount raised is given to the National Institutes of Health |
|
and 30 percent is given to the Breast Cancer Research Program at the Department of Defense. 1993 Preventive Health Amendments Revised and authorized FY94-FY98 appropriations for a CDC program providing grants to states for breast and cervical cancer prevention programs, including mammography and Pap smear screening services. 1992 Indian Health Amendments
1992 MQSA
1990 Omnibus Budget Reconciliation Act Reauthorized Medicare payment and certification standards for screening mammography for women. 1990 Breast and Cervical Cancer Mortality Prevention Act Authorized FY91-FY93 appropriations for HHS grants to states for breast and cervical cancer screening, medical treatment referrals, and information development and dissemination; training of health professionals in breast and cervical cancer prevention and control; and mammography and cytological procedure quality assurance activities. 1988 Medicare Catastrophic Coverage Act Established Medicare payment and certification standards for screening mammography for women (repealed in 1989). |
|
BOX 3-4 Certification Bodies (issue approval to provide mammography services)
Accreditation Bodies (review quality of mammography facility)
Any state can apply to the FDA to become a mammography certification body and any state or nonprofit organization can apply to become an accreditation body; however, as of June 2004, only those listed above are currently approved by the FDA. In May of 2004 the State of California voluntarily withdrew its application seeking status as a mammography facility accreditation body. Most states have relied upon the ACR to set standards for mammography facilities. |
most treatable stages by establishing baseline quality standards for facilities performing mammography.118 It is the single most sweeping legislation affecting the early detection of breast cancer. The MQSA is responsible for the stringent set of regulations that govern clinical image quality, equipment, medical records, consumer complaint mechanisms, and personnel qualifications. As a result, mammography is unique among radiologic procedures for the requirements that outline every aspect of daily practice.
The initial push for MQSA legislation was sparked by public concern about the inconsistent quality of mammography and was spearheaded in a national effort led by women’s health organizations and breast cancer advocates. It was shepherded through the legislative process by Senator Barbara Mikulski.
The MQSA includes requirements that breast imaging facilities performing mammography must be certified by the Secretary of Health and Human Services and be accredited by an approved body (see Box 3-4). The
basic requirements that breast imaging facilities must meet for accreditation are established by the law which, in turn, directs the Food and Drug Administration (FDA) to set the standards for accreditation and to oversee enforcement of the MQSA.
In 1998, the first reauthorization of the MQSA added the requirement that women receive direct notification of their mammography results. Another reauthorization of the MQSA was passed by the Senate for reauthorization in February 2004, but to date the House has taken no action. Senate sponsors predict a major reauthorization of the MQSA in 2005. In the meantime, the Consolidated Appropriations Act for FY 2004, which was passed in January 2003 (Pub. L. No. 108-199), included a mandate for studies to be conducted by the U.S. General Accounting Office (GAO) and the IOM on how to further improve mammography quality and make appropriate adjustments to the MQSA. Included is a request for those studies to be completed in time for Congress to consider the studies’ results in the debate in the spring of 2005 for the MQSA reauthorization.
For the most part, the MQSA regulations are based on standards established by the ACR Mammography Accreditation Program. That program was set up as a voluntary means of technical quality improvement, and only about half of the mammography screening facilities participated. The ACR lacked the funds to conduct on-site visits and the legal authority to enforce compliance among imaging facilities.
After the federal enforcement of quality standards, the percentage of facilities with acceptable image quality increased significantly and site-to-site variations in radiation doses decreased.16 Along with the positive results of the legislation, considerable costs are associated with adherence to the standards. These costs include staff time, inspection fees, and the maintenance of paperwork for performance documentation.
The MQSA regulations set standards primarily for the technical quality of mammography, whereas quality standards for the interpretation of mammography are almost nonexistent. The only regulation relating to quality of interpretation requires that physicians who interpret mammograms must interpret a minimum of 960 mammograms every two years, an average of 480 per year (see following discussion under Variation in Mammographic Interpretation).
The MQSA requires every mammographic facility to keep track of all positive mammograms (BI-RADS® 4 and 5; Box 3-5), including follow-up correlation of pathology results with the interpreting physician’s mammography report. Facilities are audited every year to be sure the data have been collected and that each radiologist has seen his or her own results, but there is no further requirement for the use of the data, such as for skills improvement.
|
BOX 3-5 BI-RADS® was introduced by the ACR to provide a uniform system of assessing mammography results. Besides the categories shown in this box, BI-RADS® includes a detailed lexicon for standardized descriptions of lesions and other breast abnormalities. It is intended to guide radiologists and referring physicians in a decision-making process that facilitates the management of patients based on breast imaging. BI-RADS® is a useful and widely used tool for standardizing the interpretation of mammograms and for quantitative analysis. However, a serious limitation of quantitative analysis based on BI-RADS® is that the 6-point scale is not continuous and does not provide enough gradations of positive mammograms. It is also important to note that there inevitably will be some variability in how different readers assign mammograms to different categories. Although invaluable in communicating and quantifying results, it has not yet been demonstrated that use of BI-RADS® reduces variability among radiologists (reviewed by Elmore and colleagues, 2002).27,35 BI-RADS® system for standardization of mammogram interpretation and reporting. (Fourth Edition, 2003)
|
Variation in Mammographic Interpretation
Mammograms consist of shadowy outlines of fat and soft tissue in varying shades of gray. Interpreting them requires skill and experience, and, as for every type of imaging test, different radiologists may interpret the same mammograms differently. Many factors influence the accuracy of individual radiologists in recognizing clinically important abnormalities during screening mammography, but many other factors influence the consistency of mammographic interpretation (reviewed by Beam, Elmore, Sickles, and colleagues 7,15,35,41,105).
The accuracy of radiologists in interpreting mammograms depends on many factors, including case variation, practice variation, training and experience, and the type of screening program in which they practice. Box 3-4 summarizes those factors that have been reported in the peer-reviewed literature to be correlated with the interpretation of screening mammograms. It is important to keep in mind that estimates of performance are different for screening and diagnostic mammography. In screening, the central decision is whether or not to conduct additional workup (i.e., the callback decision). The goal of screening mammography is not to provide a definitive diagnosis or to recommend biopsy without further consideration. A true positive in screening occurs whenever a woman with breast cancer is given a recommendation for additional workups, whereas a true positive in diagnosis would be whenever breast cancer is detected.
Case Variation Influences Performance Measures
Individual characteristics such as breast density or history of breast cancer are known to increase the likelihood of both false-positive and false-negative results (Table 3-3).8,24,27 In addition, ambiguous mammograms such as those revealing possible microcalcifications—which are often difficult to interpret—increase the likelihood of disagreement among radiologists. A mammography practice that serves younger women is likely to have an overall lower sensitivity rating than an otherwise identical practice that serves older women. This is reflected in the observation by Beam and his colleagues that case-related differences accounted for more variation than individual differences among radiologists.10 For example, mammographic sensitivity increases with a woman’s age (Figure 3-4).
Also, approximately twice as many breast cancers are detected at first screens as compared to subsequent screens. This is because a cancer detected at a subsequent screen generally would have developed to the point where it can be seen on a mammogram only since the previous screen, whereas a cancer detected at a woman’s first screen could have been present for years. The result is not only a higher rate of cancers detected at first
TABLE 3-3 Factors That Affect the Quality of Screening Mammography
|
Primary Source of Variation |
Underlying Sources of Variation |
|
|
Case Variation |
||
|
Breast density |
Breasts that are mammographically dense are associated with more false positives and false negatives42,65
|
|
|
Breast cancer history |
Family history of breast cancer63 Previous biopsy |
|
|
Practice Variation |
||
|
Individual radiologists |
Subspecialty training in breast imaging Volume of mammograms read (but see text) Years since training |
|
|
Organization of mammography services |
|
|
|
|
Double-reading of mammograms by two radiologists improves accuracy51 |
|
|
|
Use of computer-assisted detection (CAD) by nonspecialists (but see text) |
|
|
|
Center designated as breast diagnostic and/or screening center or freestanding mammography center |
|
|
|
Availability of prior mammograms for comparison increases specificity |
|
|
Health care cystem |
False-positive rates have increased over time in the United States and are higher than in other countries. |
|
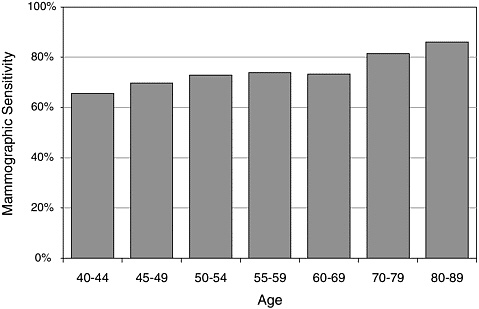
FIGURE 3-4 Mammographic sensitivity increases with a woman’s age.
screens, but a higher percentage of abnormal mammograms, biopsies, and false-positive findings. Therefore—if judged by rates of false positives—the apparent performance of a mammography service or individual radiologist would be influenced by the proportion of how many women are receiving their first mammogram.
Performance and Volume
A relationship between the volume of procedures performed and the outcome of those procedures has been established for many complex medical procedures, particularly in surgery and oncology.55 Many studies have suggested that the volume of mammograms read by a radiologist is correlated with accuracy, and mammography volume standards are mandated by federal law. However, relatively few studies have directly compared the number of mammograms read by a radiologist and the accuracy of their interpretations. The results of these are variable, and are shown in Table 3-4. The most comprehensive study to date was the analysis by Beam and his colleagues in 2003,12 which indicated that the volume of mammograms interpreted by a radiologist accounts for less than 2.5 percent of the variation. This means that more than 97 percent of inconsistency in interpretation is due to other factors.
TABLE 3-4 Relationship Between Volume of Mammograms Read and Accuracy of Interpretation Is Inconsistent
|
|
Comparison Groups (Average numbers of mammograms read per year for comparison groups) |
Outcome Measured |
Results |
|
Volume does not affect performance. |
|||
|
U.S. radiologists (n = 110) |
Analyzed on the basis of the total number of mammograms read in the past year (average number = 1,900; range =1-12,000) |
Accuracy at reading at reading a group of mammogram from a screening program. Also had more cancer cases than general population |
The number of mammograms read in the past year was not significantly associated with interpretive accuracy11 |
|
High-volume readers perform better. |
|||
|
U.S. radiologists (n = 59) |
< 1201 1201-3600 > 3600 |
Accuracy at reading PERFORMS2, which is a teaching set of mammograms enriched for cancer cases |
High-volume readers perform better Average sensitivity was equal for both low and medium volume groups (70%), but higher for the high volume group (77%)**37 |
|
High-volume readers do not perform better. |
|||
|
Canadian radiologists with >3 years experience (n = 35) |
< 2,000 2,000-2,999 3,000-3,999 4,000-5,199 |
Percent of screening mammograms interpreted as abnormal and cancer detection rate* |
The midvolume readers (2,000-3,999) detect fewer abnormal mammograms than the low- and high-volume groups. No significant difference in detection rates61 |
|
* This figure includes both true positives and false positives, but because the great majority of women screened do not have breast cancer, the percent of mammograms judged to be abnormal will depend more on the false-positive rate than on the true-positive rate. ** This reference is from a single study examining a group of radiologists that interpreted a common set of mammograms. Therefore, these sensitivity values should be compared only within this study and do not necessarily apply to population-based studies. |
|||
The volume of mammograms read by individual breast imagers is likely to be important—if not directly, perhaps as a proxy for other characteristics such as advanced training, specialization in breast imaging, or working in an organization with a mammography quality improvement program.
Specialists Are More Accurate Than Generalists
One study reported that compared to general radiologists, breast imaging specialists detect more cancers, recommend more biopsies, and have lower recall rates. In general, the specialists, who interpreted more than 5,000 mammograms per year, found two to three more cancers than general radiologists for every 1,000 mammograms.11
However, only 12 percent of radiologists interpreting mammograms are specialists,23 and most women do not see specialists. The false positive rates of community radiologists (those who work outside academic research centers) are quite variable. One study reported rates of false positives that ranged from 3 to 16 percent.35
Error Rates Depend on Context and Organizational Factors
Accuracy depends on context. Rates of false positives in the United States have increased over the years, and they vary among countries and health care systems. Defensive medicine is widely presumed to be prevalent in the United States, especially in mammography. Leonard Berlin testified on behalf of the ACR in Congress that malpractice suits in the United States are decided in favor of plaintiffs so often that many radiologists do not attempt to contest even seemingly frivolous cases.14
Rates of false positives in the Unites States nearly doubled from 1985 to 1993, from roughly 5 to 10 percent.35 This increase parallels the steadily increasing rates of malpractice suits related to failures to detect breast cancer through mammography, which is often proposed as a driving force in rates of false positives. Radiologists in the United States may be practicing more defensive medicine because they fear malpractice suits, which their counterparts in the United Kingdom face to a much lesser degree.34 Although British radiologists also report that they worry about malpractice, the scope of the problem is considerably less than it is in the United States. Only about 25 percent of British practices admitted to being sued over breast cancer, with nearly all cases being dropped.89 Many believe that concern over malpractice is an important factor in the relatively high rate of false-positive results in the United States, and anecdotal evidence supports this view. However, there are no reliable data to measure the extent of this problem, and such sensitive data would be difficult to obtain.
Options for Improving Mammographic Interpretation
As a rule, health care quality is less often improved by weeding out individual, low-performing “bad apples” than it is by organizational improvements. Health care depends more on the development of organizational structures and processes that support high-quality performance and that have built-in systems for continuous improvement and feedback about performance.54 The various options that have been proposed for improving the quality of mammographic interpretation in the United States are discussed below. They include increasing the required volume of mammograms, restricting the number of radiologists permitted to interpret mammograms, greater standardization of assessment categories, public reporting of performance, and better training for radiologists in mammographic interpretation.
Increase Volume Requirements
The MQSA standards for the minimum number of mammograms to be interpreted by certified radiologists are lower in the United States than for other countries with breast screening programs (Table 3-5). When the MQSA standards for mammography volume were established, the volume standards were designed to balance the perceived benefits of high volume against the need to avoid discouraging the already limited workforce from interpreting mammograms at all. Beam and his colleagues concluded that a 1 percent increase in accuracy would require 3,000 more mammograms to be read per radiologist per year.11 The Society of Breast Imaging “strongly disagrees with the implication that American radiologists recommend excessive workups and should be required to read 5,000 mammograms annually, as is required of British radiologists.”
TABLE 3-5 Mammography Volume Standards12
|
|
Required Number of Mammograms |
Average per Month |
|
United States |
960 over a 2-year period |
40 |
|
British Columbia, Canada |
2,500 per year |
>200 |
|
Sweden |
No target set, but only specialists interpret mammograms |
>1,000 |
|
United Kingdom |
5,000 per year |
>400 |
Restricting the Number of Radiologists Permitted to Interpret Mammograms
Beam and his colleagues predicted that health policy recommendations based on improving mammographic interpretations based solely on radiologist volume will be “misleading and ineffectual.”11
Beam and his colleagues estimated the effects of increasing median accuracy by prohibiting low-performing radiologists from interpreting mammograms.12 They calculated that, to achieve a 5 percent increase in the median accuracy among radiologists, the 2,200 radiologists at the bottom of the performance spectrum would need to be prohibited from interpreting mammograms. This would translate into a reduction in national service capacity of about 25 percent. The net effect of such a policy would be more likely to increase rather than decrease the number of women whose breast cancer escapes early detection. Further reducing a workforce that is already in short supply is deemed not the best option.
Greater Standardization
Use of ACR BI-RADS® could, in principle, lead to less variability among radiologists (see Box 3-5). However, recommendations for BI-RADS® categories have been shown to be applied inconsistently for mammographic abnormalities, suggesting that use of BI-RADS® by itself is not enough to reduce variability in mammography.116 For most BI-RADS® assessments, community radiologists make consistent recommendations, with an overall concordance of 97 percent. However, agreement is substantially lower when the assessments are “probably benign finding” (BI-RADS®), with a concordance of only 47 percent.69 This is the assessment most commonly associated with errors of interpretation, as well as malpractice cases. Agreement among radiologists is also lower for mammograms of women with dense breasts.69
Public Reporting of Performance
Publicly reporting mammogram interpretation performance results of radiologists has been resisted by radiologists. Quality conclusions based on performance might be misleading in view of differing risks, ages, or other characteristics of caseload among radiologists and facilities that could legitimately lead to differing results, as noted earlier. Although radiologists review nondiscoverable outcomes data for their positive cases on an annual basis, follow-up on negative mammograms is not possible. Also, it is feared that public reporting would provide targets for litigation and major professional and economic disincentives to radiologists entering or remaining in
the field of mammography, already a relatively unattractive and risky service with low reimbursement. In the United Kingdom, for example, individual performance results are provided only to the radiologist. Finally, in practice, publicly released health care performance data are rarely used by consumers even when available. In contrast, health care providers do use performance data results to improve the quality of their health care services. Marshall and colleagues reviewed studies on the public disclosure of health care performance data and concluded that78
…the use of public performance data by consumers and purchasers or for regulation purposes will remain relatively less important for the foreseeable future than use of the data as a catalyst to stimulate and promote internal [italics added] quality improvement mechanisms at the level of the organizational provider.
Better Training
Continuing medical education is required for radiologists in the United States, but the content is not uniformly organized and almost never targets recall or cancer detection rates. There is a view that continuing quality assurance through feedback of results could improve performance. In contrast, the United Kingdom has established a program that is voluntary but is used by more than 90 percent of radiologists who practice mammography.
OTHER TECHNOLOGY OPTIONS
As noted earlier, no breast cancer screening tool has better sensitivity and specificity than screen-film mammography, although it could be better. However, even with similar sensitivity and specificity, there may be ways to improve storage, transmission, cost, ease of use, and other characteristics of mammography that would add value. Certainly, any new technology or refinement should have equal success in accurately detecting abnormalities and an equal or better effect on health outcomes compared to current screening mammography. Reviewed briefly below are some leading technologies that are FDA approved and are being examined for their roles in breast cancer detection and diagnosis. Digital mammography and CAD have been proposed for use in screening of average risk women, whereas MRI is not expected to improve outcomes for average risk women but is being tested for use in certain groups of high-risk women.
Digital Mammography
For more than 10 years, researchers have been developing digital mammography devices in the hope that digitizing radiographic data will im-
prove on conventional imaging methods by allowing the different tasks of image acquisition, processing, and display to be separated and therefore refined, as well as allowing adjunct technologies such as CAD to be used more easily.59 But despite the introduction of full-field digital mammography units into the market, the question of whether digital actually improves cancer detection rates or workflow remains open.
Digital mammography systems offer better contrast and lower spatial resolution at a lower radiation dose than traditional screen film mammography.59 The relative diagnostic accuracy of digital mammography compared to traditional mammography is still undergoing study through the large Digital Mammographic Imaging Screening Trial (DMIST). As of this writing, digital mammography appears to improve specificity—possibly due to the flexibility of image display available to the interpreting radiologists.31 Digital units probably also improve workflow, allowing radiologists to view images in less than a minute, compared to the 8 to 10 minutes required from screen film systems.
Because digital mammography devices are more expensive than conventional devices, they will have to offer substantial advantages over film-screen mammography in order to be widely used. Research to date has not shown a dramatic difference between the two techniques. In a 2001 study, 4,945 women had both conventional and digital screening mammography exams. The conventional mammography device found a few more cancers than the digital unit; both devices missed cancers the other found. Overall, there were no major differences in cancer detection rates between the two techniques, although if the digital technique had been used alone, recall rates would have been lower.70 Similar results were reported in 2003 from a Norwegian study of 3,683 women.106 Each woman in the study had both digital and screen film mammography exams and they were independently interpreted. The cancer detection rates for the two imaging modalities were not significantly different, although the recall rate was slightly higher for digital mammography and the positive predictive value based on needle biopsy was slightly higher for screen film mammography.
In 2001, the National Cancer Institute (NCI) and the American College of Radiology Imaging Network (ACRIN) launched the multicenter DMIST study to compare digital mammography with standard mammography for the detection of breast cancer. The 49,520 women enrolled in the study will be followed for one year after receiving both digital and conventional mammograms. For further discussion of ACRIN and DMIST, see Chapter 6.
Computer-Aided Detection
Since 1989, technology developers have conducted experiments on the use of computer capability to aid in reading radiological images. Because
using mammography to detect cancer in normal breast tissue is fundamentally a signal-to-noise exercise, it is particularly suited to CAD technology.30 Because factors such as radiologist fatigue and distraction, the complexity of breast structure, and the subtle characteristics of early stage disease make interpreting mammograms challenging and contribute to both false-positive and false-negative results, the use of CAD with mammography becomes particularly attractive, offering experienced radiologists the option of a “double read.”
Basic CAD systems consist of a workstation with display and signal processing software. The CAD unit reads either manually digitized mammography films or directly digitized images and highlights areas of concern such as masses, calcifications, or architectural distortions, for the radiologist’s review. Images can be printed or displayed in soft copy on a monitor. CAD for mammography was formally introduced in 1998 when the FDA approved the first CAD device, ImageChecker M1000®, made by R2 Technology of Sunnyvale, California. In addition to ImageChecker, two other CAD devices cleared for use in the United States: (1) Second Look® by Nashua, New Jersey-based iCAD® and (2) MammoReader® by Intelligent Systems Software of Clearwater, Florida. ImageChecker and Second Look are also approved by the FDA for use with full-field digital mammography devices.
In a 2001 study, radiologists who interpreted mammograms, using both conventional mammography reading techniques as well as CAD technology, found nearly 20 percent more cancers with CAD than they did without, and the proportion of early stage malignancies detected increased from 73 to 77 percent. But they also found that the recall rate increased, from 6.5 percent when the radiologist interpreted the mammogram without CAD to 7.7 percent when CAD was used.46 This study analyzed only the ImageChecker M1000® system produced by R2 Technology and the results cannot be assumed to apply to every CAD system.
The reproducibility of CAD results has improved as the technology has been advanced. Bin Zheng and colleagues used 100 mammographic cases with four views each from a database of more than 1,000 digitized images and diagnostic results. The cases included 25 with microcalcification clusters and 75 with masses. Two-thirds of the cases had been confirmed malignant. Using ImageChecker®, Zheng scanned the images three times over a period of 3 weeks, checking for sensitivity, false-positive rates, and reproducibility of the results. The researchers found identical results in 213 of 400 images, for a reproducibility rate of 53 percent, an improvement from 38 percent found in a 2000 study based on an earlier version of the CAD system.125
The greatest clinical value in CAD probably does not lie in its ability to raise the performance level of all breast imagers, but rather in its potential
to bring the performance level of general radiologists to that of breast imaging specialists.88 As noted earlier, the great majority of screening mammography is done by general radiologists who tend to have lower sensitivity rates and higher false-positive rates than breast imaging specialists. In fact, a 2004 study reported that the use of CAD was not associated with statistically significant changes in recall or breast cancer detection rates.49 However, all radiologists in that study were considered breast imaging specialists, and the results of this study should not be extrapolated to use by community radiologists who vary widely in their proficiency.
An often overlooked challenge in establishing the value of CAD systems is that they are not all the same. There has been a series of peer-reviewed papers documenting the efficacy of the CAD systems produced by R2 Technology, but, to date, there are no peer-reviewed reports on the efficacy of any other commercially available CAD systems for breast imaging. Even for systems that have been analyzed in the peer-reviewed literature, when a manufacturer produces an “upgraded” system, the changes should be assessed in terms of diagnostic accuracy—as opposed to subjective evaluations of clarity of image, or other aspects of image processing that might appeal to the eye but not improve interpretation.
Reimbursement for this technology may be a key factor in the adoption of CAD, helping offset the cost of acquiring the technology and integrating the process into the existing organization of breast care services. The Center for Medicare & Medicaid Services (CMS) provides some support for the adoption of this new technology. In 2003, its reimbursement rate for CAD was $19.13 per exam. Also, in 2002 the agency expanded its coverage to include diagnostic exams and the use of CAD with digital mammography. CAD is treated as an add-on procedure to screening or diagnostic mammography. Ironically, although CAD offers a “second look” at a mammogram and is reimbursed by CMS (although not by all health care insurers and providers), actual double reading done by two radiologists is not reimbursed, even though the practice reportedly increases cancer detection by 5 to 15 percent.51
Whether the use of CAD technology affects a center’s workflow remains open to debate. In a facility that uses screen-film mammography devices, CAD can slow down workflow because of the extra time needed to digitize films. For many, CAD makes more sense in a digital environment, where the images can go directly from the mammography unit to the CAD device; yet even with direct digital images, image processing can be affected by the different algorithms used for image detection, digitization techniques, and methods of display each CAD system employs.
However, for those who do decide to incorporate CAD into their facility, the technology may provide an additional benefit beyond clinical ones. Because radiologists are the targets of litigation—especially for missed breast
cancer diagnoses—more than any other specialist, it may be worth the additional workflow impact or fiscal cost if the use of CAD reduces the likelihood of capricious judgments. If a radiologist and a CAD system both fail to detect an abnormal mammographic finding, then it becomes less likely that the missed cancer can be successfully argued to be due to negligence on the part of the interpreting radiologist.
Magnetic Resonance Imaging (MRI)
[In its current state,] MRI has nothing to do with the average woman undergoing screening for breast cancer. It’s a promising technology, but right now it’s overreaching to say it’s useful for the average woman.84
William J. Gradishar, M.D.
Northwestern University
Researchers have been exploring the use of MRI in breast cancer detection for more than 15 years.104 In 1991, the FDA cleared MRI for use as a diagnostic tool to evaluate breast tissue abnormalities found in other exams—but not as a screening tool. It has been suggested that MRI is useful in a number of clinical indications such as finding small breast lesions that are sometimes missed in mammograms, generating better images of dense or augmented breast tissue, revealing multifocality of breast cancer, and aiding in treatment staging and follow-up.103 There is also growing agreement in the clinical community that MRI could be a valuable tool in screening protocols for women at higher risk for breast cancer.
Results of the largest prospective, multicenter study of MRI screening to date were presented at the 2003 meeting of the American Society of Clinical Oncology. The study compared the findings of yearly x-ray mammography and MRI breast exams in more than 1,000 women at higher than average breast cancer risk. Over 2 years, 40 breast cancers were found. Sixteen percent of the cancers were identified by clinical exam, 36 percent by mammography exams, and 71 percent by MRI.67 However, MRI was less specific than mammography (88 versus 95 percent). In another 2003 study, researchers found that, for use as a tool in screening women with breast cancer gene mutations and helping diagnose disease earlier, MRI appeared to be superior to mammography and sonography (ultrasound).92
Likewise, mammography is also the primary technique for detecting ductal carcinoma in situ (DCIS). Recent research indicates that contrast-enhanced MRI might help detect otherwise occult foci (such as those that occur in DCIS), or in patients with small or dense breasts for whom mammography can be less reliable (Figure 3-5).53,121 A gadolinium-based con-
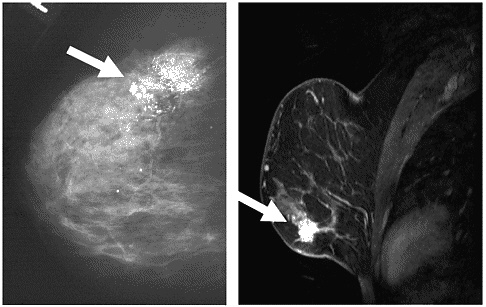
FIGURE 3-5 Examples of MRI and x-ray images of two different cases of DCIS. The case on the left is an x-ray mammogram of a breast showing DCIS in the upper portion of the breast. The case on the right is an MRI mammogram showing DCIS in the lower portion.
SOURCE: X-ray; UHrad.com. Women’s Imaging Teaching Files. http://www.uhrad.com/mamarc/mam016.htm [Accessed August 21, 2003]. MRI; Magnetic Resonance Science Center at University of California San Francisco. Breast MRI Website. http://www.mrsc.ucsf.edu/breast/dcis.html [Accessed August 21, 2003].
trast agent is administered intravenously to provide better soft-tissue contrast, and this helps differentiate cancer from benign surrounding tissue. The principle behind contrast-enhanced MRI relies, in part, on the assumption that cancerous lesions will have characteristic features. For example, compared to benign breast lesions, cancerous lesions tend to absorb the contrast agent faster and the gadolinium-based agents are washed out faster.
A prospective study of 51 patients with biopsy-proven DCIS who underwent contrast-enhanced MRI before surgical treatment indicated that contrast-enhanced MRI had significantly higher sensitivities and negative predictive values than mammography in the detection of residual disease, occult invasive cancer, and multicentric DCIS.53 Although contrast-enhanced MRI was statistically significantly more accurate than mammography for detecting multicentric DCIS, it was significantly less specific than mammography for detecting associated invasive disease.53
It is important to keep in mind that results for MRI based on high-risk
populations should not be extrapolated to the general screening population, because the positive predictive value of a screening testing depends on the prevalence of the condition being screened. By definition, breast cancer will be more prevalent in high-risk women. In addition, the lower specificity of MRI compared to mammography would translate into a substantially higher rate of false positives. According to Daniel Sullivan, Associate Director for the Cancer Imaging Program at the NCI, determining the value of MRI screening for the general population would require a study of more than 40,000 patients, a minimum of 3 years, and tens of millions of dollars.84 Unfortunately, the lack of evidence in support of MRI for breast cancer screening and the lack of FDA approval do not necessarily protect the public from misleading marketing (see Box 1-3 in Chapter 1).
The techniques for performing and interpreting breast MRI are not standardized. As a result, breast MRI performance has been variable, and it has been challenging to determine its clinical efficacy. The results of the 5-year International Breast MRI Consortium (Trial #6883), a study funded by the NCI and the National Institutes of Health (NIH) Office on Women’s Health, will soon be reported and will be a major step forward in clarifying and optimizing the clinical value of MRI. This multicenter study, conducted at 14 institutions, evaluated the diagnostic performance of breast MRI in women with suspicious mammographic or clinical findings. It is expected to yield definitive results about the diagnostic accuracy of breast MRI and should lead to recommendations for performing and interpreting breast MRI images. In addition, as of early 2004, an ongoing ACRIN trial testing the use of MRI to screen the contralateral, or noncancerous breast, in women with breast cancer is attempting to further define the role of MRI in detecting breast cancer in a high-risk population (Trial #6667).
Ultimately, more research on how MRI performs as a screening modality and exactly how high-risk women would benefit remains to be done.104 MRI is expensive, about 10 times the cost of conventional mammography, and because it will generate more false-positive results, it will become even more expensive with the added costs of additional biopsies and/or other diagnostic follow-up.
Ultrasound
Also known as sonography, ultrasound gained FDA approval in 1977 as a means to evaluate suspicious mammographic findings (see Appendix A). Ultrasound is especially useful in determining whether such findings depict benign cysts or solid lesions. When rigorous criteria are applied, the accuracy of ultrasound for diagnosing fluid-filled “simple” cysts—which are always benign and therefore require no further evaluation—has been found to be 98 to 100 percent.64 Ultrasound is also used to investigate
palpable abnormalities, of which 20 to 25 percent are simple cysts.6 It is often used instead of mammography to investigate palpable breast abnormalities in women who are pregnant, to avoid exposing the fetus to x-rays, and in women under age 30, for whom mammography is not as effective due to breast density (see Chapter 2).80 Unlike mammography, ultrasound is not affected by breast density.
Ultrasound can also determine whether a lesion located near the surface of the breast occurs within the skin, where it is unlikely to be malignant, or in the underlying breast tissue, necessitating further investigation.80 Because it can reveal various characteristics of suspicious lesions such as the likelihood of invasiveness, and spread within the ducts, ultrasound may in some cases inform the staging of breast cancer.68,80 Ultrasound can also contribute to attempts to distinguish between benign and malignant solid lesions, but criteria for assessing malignant features have yet to be established and validated in controlled multicenter clinical studies.6,80
The combination of screening mammography and ultrasound is highly sensitive for breast cancer in non-symptomatic women. For example, in the largest screening series of bilateral whole-breast ultrasound, which encompassed more than 13,000 examinations in women with dense breasts and previously negative mammograms, a “double screen” of mammography plus ultrasound detected 97 percent of breast cancers; other, smaller studies have produced similar results.6 As a competing technology, as opposed to a complementary technology, ultrasound falls short of mammography due to its relatively poor sensitivity for microcalcifications, the hallmark of DCIS.6 When ultrasound depicts microcalcifications, they are usually associated with a mass, and therefore indicative of invasive cancer (reviewed by Mehta, 2003).80 This raises the possibility that ultrasound actually finds the majority of DCIS that has become invasive6 (see also Chapter 2).
Some researchers have claimed, and published studies strongly suggest, that ultrasound can detect breast cancers that both mammography and clinical breast exams would miss (reviewed by Kopans, 2004).66 However, because breast screenings were not conducted independently in any of these studies, their results were potentially biased (for example, by researchers’ knowledge of a participant’s mammography findings prior to conducting ultrasound).66 Better information on the potential role of ultrasound in breast cancer screening should be forthcoming from a multicenter clinical trial that is currently being conducted by ACRIN with support from the Avon Foundation (see Chapter 6).6 The protocol, which includes blinded sonographic screening, aims to compare the sensitivity, specificity, and positive and negative predictive values of ultrasound and mammography combined with that of mammography alone. In order to make obvious any disparities that may exist among mammography, clinical breast examina-
tion, and ultrasound, the trial is limited to women with dense breasts who are at high risk for breast cancer.
Because the ACRIN trial will not measure death rates, but will rely on surrogate endpoints such as lesion size, nodal status, and diagnostic yield, it cannot directly determine whether the combination of ultrasound and mammographic screening has any effect on mortality from breast cancer (reviewed in Kopans 2004).66 Rather, it is intended as a preliminary, best-case scenario of ultrasound screening for breast cancer which, given promising results, would justify broader study.6 Moreover, even if the efficacy of screening ultrasound were established, several technical and practical limitations could hinder its adoption. Chief among these is the variability of results obtained in current clinical practice.6,68,80 Ultrasound tends to be more difficult in larger and fatty breasted women. However, a systematic study of factors that influence the performance of breast ultrasound—which may include breast size and shape, as well as lesion location—has yet to be conducted.6 Finally, as it is currently performed, ultrasound demands too much physician time to be a cost-effective screening method.6
BREAST IMAGERS NEEDED
Demand for breast imaging is rising as the U.S. population ages and as increasing numbers of women require routine screening.56,119 Improved screening methods and new technologies may help keep pace with these trends, but greater capacity in both personnel and imaging facilities will also be needed to ensure patient access. These resources are also crucial to the improvement of breast cancer detection, because a robust process of assessment, adoption, and dissemination of innovative technology and techniques by practitioners requires an equally robust workforce.
Despite these mounting demands, many in the breast imaging field point to stagnant growth, if not decline, in the availability of the services they provide. Over the long term, such a trend could threaten the advancement of breast cancer detection, which is already limited by the scarcity of radiologists conducting research in this area. The development of new technology also has the potential to solve some of the problems of demand by focusing mammographic services on populations who are most at risk, although these applications are probably at least 5 to 10 years in the future. The need for and difficulty in developing a large and well-trained workforce will likely help to push for research and adoption of technology that improves our ability to target mammographic screening to those who will benefit most. This section describes key factors influencing supply and demand for breast imaging and recommended measures to ensure the accessibility and advancement of breast cancer screening.
Access to Breast Cancer Screening Is Endangered
Recent increases in average waiting times for mammograms indicate that breast cancer screening facilities are operating at or near full capacity.56 In New York City, patients waited an average of more than 40 days in 2003 for first-time screening mammograms, as compared with 14 days in 1998; in parts of Florida, waiting times of three months are common.75 Such delays are likely to occur in locations where facilities have closed or where radiologic technologists (the individuals who actually perform mammograms) or radiologists who interpret mammograms—or both—are in short supply. Although no published studies document this trend, there is widespread consensus among breast imagers that workforce issues limit access to mammography and this problem is becoming more acute.
A 2002 report by the GAO found that women in some locations have problems obtaining timely mammography services and raised the prospect of future staff shortfalls, but concluded that the nation’s capacity was “generally adequate to meet the growing demand for these services.”119 Impressions of current capacity within the field of breast imaging are far less sanguine, and that community has criticized the GAO report on several counts.85 For example, they note that it does not distinguish among those radiologists who read some mammograms and those who are breast imaging subspecialists,c and therefore may have overestimated capacity for mammogram interpretation. Although approximately 20,000 radiologists interpret mammograms in the United States,12 only 2,000 of them are members of the subspecialty society, the Society of Breast Imaging. (There are also a small number of nonradiologists who meet the requirements for interpreting mammograms, but the great majority are radiologists, so for simplicity, this report refers simply to radiologists.)
In addition, the GAO may have underestimated the number of radiologic technologists required to meet national mammography needs. A 2000 GAO report found an 18 percent job vacancy rate for radiologic technologists, and a majority of participating hospitals reported greater difficulty in hiring technologists than in the previous year.119 Among radiologic technologists, the number of first-time examinees for mammography certificates has declined substantially each year from 1996 to 2000.85 Moreover, the most recent GAO projections do not take into account the time spent by technologists on activities other than screening, such as performing diagnostic mammograms, breast interventional procedures, and quality assur-
ance activities mandated by the MQSA.86 Finally, many radiologic technicians are involved in other radiology work and are not available to work full-time on mammography tasks.
Financial Woes Close Screening Facilities
A decline in the number of mammography screening facilities was reported by the ACR, which found that facility closures (979) outpaced openings (401) by more than two to one between April 2001 and February 2003 (Figure 3-6). Financial factors were the most frequently cited main reason for these closures. Although some facility closures probably reflect consolidation of services with more efficient service delivery, the scale of the closures suggests a serious decline in access.
For years, radiologists and health care administrators have asserted that reimbursement for mammography does not cover procedure costs, which include the soaring expense of malpractice liability insurance (see below) and the cost of compliance with MQSA regulations.56 Medicare now pays slightly more than $82 per mammogram; the ACR estimates that a screening mammogram costs about $87 to perform in a freestanding
FIGURE 3-6 Fewer mammography facilities each year. Fewer mammography facilities are being inspected by the FDA each year.87 Under the MQSA, which is enforced by the FDA, yearly inspections are required for accreditation. The year 2003 data were estimated from the initial result of the inspections in the first half of 2003 extrapolated using the trend from the previous 3 years.
clinic and more than $105 in a hospital.d For high volume settings, even relatively small shortfalls for a particular procedure can add up to large operating deficits. This is compounded in settings that are dedicated to providing a single procedure because there are no opportunities for cost recovery on other, profitable procedures.
A financial analysis of seven university-based breast programs reported that they all incurred losses in the professional component of mammography services.4 The driver of the loss was diagnostic mammograms.37 However, it should be noted that because women often seek out university-based programs for second opinions or difficult diagnoses, such programs are likely to conduct a larger proportion of diagnostic mammograms as compared with community facilities (Dieter Enzmann, personal communication).
Increasing Demand for Breast Imagers
The term “crisis” is routinely used in the radiology community to describe the shortage of breast imagers. Based on U.S. population estimates, 1.25 million additional women become age eligible for recommended mammography screening each year, while only about one to three dozen breast imaging subspecialists enter the profession each year (Priscilla Butler, ACR, personal communication). Although only about 12 percent of all mammograms are interpreted by breast imaging subspecialists (Barbara Monsees, personal communication), they spend more of their time doing breast imaging than other radiologists and tend to be more proficient. Thus increasing their ranks may not only help meet the growing demand for screening mammograms, but also improve the quality of breast image interpretation.
Mammography here is pretty awful. Five centers closed in 18 months and we’re working very hard, too hard. Two technologists put in their notice and the three of us part-time mammographers may have to leave because the insurance company is putting an extra charge to read mammography—i.e., my premium and hence, tail coverage will cost more than my husband, who is full time. Pretty crazy. It will take all of us leaving Florida mammography to make a change.
Anonymous Florida radiologist
January 2004
This is unlikely to happen soon, however, given the relative dearth of medical students entering radiology and, in particular, breast imaging. While the number of radiologists is growing by an estimated 2 percent per year, their workload is increasing by about 6 percent annually, and is likely to continue to outpace workforce expansion.2,112,113 Even more scarce than radiologists are those who choose to specialize in breast imaging. In a 2001 survey of radiology residents in accredited U.S. programs, 64 percent said they would not consider doing a fellowship in breast imaging.9 The same proportion of respondents said they did not want to spend more than 25 percent of their work time interpreting mammograms, due to reasons such as lack of interest, fear of lawsuits, high stress, low pay, and the fact that breast imaging is perceived as a female-dominated field. Even without a general shortage of radiologists for procedures other than mammography, there is great concern over the shortage of breast imagers.
Malpractice litigation is a particular concern for breast imagers due to the disproportionate number of claims against radiologists who interpret mammograms (see Box 3-6) and the comparatively high price they pay for malpractice insurance (Barbara Monsees, personal communication). Recent, precipitous increases in malpractice insurance premiums for all physicians reportedly have led significant numbers of physicians in high-risk specialties and/or locations to retire, relocate, or restrict their practices in order to avoid lawsuits.33,93 Such claims were only partially substantiated in a GAO assessment of the effects of rising malpractice premiums on access to health care, however.120 The ensuing report included an analysis of access to mammograms in five states that were cited by the American Medical Association (AMA) and other national health care provider organizations as facing “full blown liability crises.” The GAO concluded that access to mammograms had not been subject to “major reductions” in these states (unlike access to emergency surgery and newborn deliveries, which were found to be restricted in several, mostly rural, locations). The AMA disputed the GAO’s findings, claiming that the scope of the agency’s investigation was insufficient to support its conclusions.
To ensure adequate capacity over the long term for radiological services (and, more specifically, for breast imaging) the causes of the decline in workforce must be identified, prioritized, and addressed. Possible solutions to these larger issues include incentives to increase the number of breast imagers and radiologic technologists (such as by enriching the professional opportunities and resources for research), more efficient technology, better reimbursement for mammography, efforts to increase breast imagers’ productivity, and mechanisms to reduce the burden of malpractice insurance, such as tort reform.110
Problems relevant to tort reform are particularly intense in, but by no means limited to, mammography—or even, for that matter, limited to health
|
BOX 3-6 The threat of malpractice litigation disturbs breast imagers and radiology residents, and perhaps for good reason. Breast cancer in women leads to more malpractice claims than any other medical condition and is second only to the neurological impairment of newborns in the expense of paid claims, according to a 2002 report by the Physicians Insurers Association of America (PIAA) based on claims paid between January 1995 and June 2001.97 The latest PIAA study also found that an increasing percentage of claims regarding breast cancer are being filed against radiologists who interpret mammograms. Radiologists accounted for the largest proportion (33 percent) of paid claims involving breast cancer, up from 24 percent during the previous PIAA study based on claims paid between 1990 and 1995.96 Surprisingly, among breast cancer claims, obstetricians and gynecologists were the second largest group (23 percent). Other claims for missed breast cancer diagnosis, in descending order of percentage, named insurance corporations, surgical specialists, family and general practitioners, and internal medicine practitioners. The PIAA represents about 60 percent of physicians in private practice. Over the more than 16 years that the PIAA has collected data on medical malpractice claims related to breast cancer, an average of about 200 such claims per year have been reported by its member companies. Forty-one percent of these claims were successful and, by 2001, paid an average indemnity of more than $290,000. Focusing on the 450 paid claims involving the delayed diagnosis of breast cancer during the 2002 study period, the PIAA found that 74 percent of those claims involved pre- or peri-menopausal women. More than one-third of these claims involved women under age 40, who compose less than 5 percent of invasive breast cancer cases.3 This group accounted for 43 percent of the total cost of claims, and nearly half of all deaths, in malpractice cases involving delay of diagnosis of breast cancer. Successful malpractice claims for delayed diagnosis among younger women may be due to a combination of factors. Younger women tend to have more dense breasts, resulting in mammograms that are difficult to interpret (see Chapter 2) and thereby increasing the likelihood of a missed diagnosis. Yet because people tend to overestimate the benefits of mammography (see Chapter 4), particularly for younger women, jurors may not appreciate its limitations. As for the relatively high indemnities paid in such cases, awards to injured patients tend to reflect the severity of the damage sustained (an early death, in many of these cases), less than they reflect the degree of negligence attributed to the injury.13,20 Just as there are no simple cures for the malpractice liability crisis facing physicians across the United States110 (see Box 3-7), there is no straightforward solution to the particular legal vulnerability of radiologists who interpret mammograms. Instead of exploring risk management strategies that might reduce a radiologist’s exposure to lawsuits, this report recommends a variety of measures aimed at reducing the likelihood of a missed diagnosis. These could include the reorganization of breast cancer screening services to better serve young women at higher-than-average risk for breast cancer, improving the quality of mammographic interpretation, and developing more effective technologies to detect and diagnose breast cancer. |
|
BOX 3-7 For the first time in nearly 20 years, the United States is facing a broad-based crisis in the availability and affordability of malpractice liability insurance for physicians, hospitals, and other health care providers. Although liability is intended to provide a system of accountability, there is widespread agreement that the current system of tort liability is a poor way to prevent and redress injury resulting from medical error.17 The randomness and delay associated with the present pattern of accountability not only prevents severely injured patients from receiving prompt and fair compensation, but destabilizes liability insurance markets and attenuates the signal that liability is supposed to send health care providers regarding the need for quality improvement. The shortcomings of the current malpractice system come from three directions, all of which have contributed to the present crisis: inefficient and inequitable legal processes for resolving disputes, problematic responses by clinicians to the threat and cost of liability, and volatile markets for liability insurance. The best way to create a legal environment that fosters both high-quality patient care and relieves financial strain and administrative burden for health care providers is to replace tort liability with a system of patient-centered and safety-focused nonjudicial compensation—linking claims resolution to organization-based error disclosure and safety improvement processes. SOURCE: Committee on Rapid Advances Demonstration Projects: Health Care Finance and Delivery Systems, Fostering Rapid Advances in Health Care, IOM, 2003. |
care.79 The problem of malpractice is unlikely to be solved by individual medical specialties. The American College of Obstetrics and Gynecology worked hard to encourage the passage of federal legislation to regulate lawsuits for health care liability related to ob/gyn care, but in both 2003 and 2004 the bills they supported were defeated. It is not clear that the mammography field would have any more success. Moreover, this Committee was not comprised of experts in medical law and did not address the issue in depth, although a previous IOM Committee discussed medical malpractice and tort reform in its 2003 report on Fostering Rapid Advances in Health Care (see Box 3-7).52
More Radiologists Needed in Research
The erosion of access to mammography is not the only potential consequence of a shortage in radiologists who interpret mammograms. Radiologists, and particularly breast imaging subspecialists, are needed to refine,
test, and disseminate new technologies. Anticipated advances could enhance, redirect, or replace current breast imaging technologies used to detect and evaluate abnormalities, or they could take the form of new methods for the diagnosis, biopsy, treatment, and surveillance of treated patients. However, a decline in the number of breast imaging professionals, especially at academic institutions, is likely to limit scientific advancement in the field.
University clinicians play key roles in medical innovation—including the refinement, standardization, and evaluation of novel devices—and they make important contributions to the design of clinical trials, providing expertise in methodology and the measurement of outcomes such as quality of life, quality-adjusted survival, and cost-effectiveness.48 Radiologists appear to be in high demand in academia; a recent survey of radiology department chairs by the Radiological Society of North America found about 600 such job openings per year.1
Academic vacancies in radiology leave remaining radiology faculty with less time to engage in research. This is especially troubling given that radiologists’ research presence is relatively minor compared to most other medical specialists. Overall, about 3 percent of physicians in nonsurgical specialties cite research as their primary activity, whereas only 0.6 percent of radiologists do (Figure 3-7).94 In 2000, there were roughly 21,000 diagnostic radiologists in the United States, of whom only 127 cited research as their major professional activity. By comparison there were a similar number cardiologists, of whom 748 identified themselves primarily as researchers—nearly six times as many as their counterparts in radiology. The high service workload for breast imagers also takes a toll on research involvement. Many breast imagers who previously volunteered their time to serve as grant reviewers can no longer afford to take the time from their clinical practices (Barbara Monsees, personal communication). This is a loss not only because breast imagers are less available to contribute their perspectives to research on improving breast cancer detection, but they are also deprived of an opportunity to learn about new directions in research.
Radiology equipment is expensive and ever changing. Clinicians with research expertise play an important role in evaluating the impact of new technologies on health outcomes, but few radiologists who interpret mammograms receive any research training. While some radiologists serve as alpha or beta testers for commercial products, few undertake the sort of carefully designed clinical trials needed to demonstrate the medical value of new procedures or technologies. Better training of radiologists would enable more critical evaluation of equipment and techniques.
As C. Douglas Maynard, co-chair of the ACR’s Task Force on Human Resources commented, “We really need to be producing more research to take advantage of opportunities at the NIH, (but) in fact we are producing
FIGURE 3-7 The ranks of academic radiologists are thin compared to other specialties.
less.”1 In principle, research participation by radiologists could get a boost from the newest institute at NIH, the National Institute of Biomedical Imaging and Bioengineering (NIBIB). However, the NIBIB budget is small. At approximately $280 million, the 2003 budget for NIBIB was the second smallest of the NIH institutes and a mere 6 percent of the NCI budget. (The smallest budget was that of the National Institute of Nursing Research.) In addition, at least for the first round, fewer than one in 5 grant applications were from physician researchers; over 80 percent were from Ph.D. researchers (Barbara McNeil, NIBIB Council Member, personal communication).
Practitioners Drive Technology Adoption
The pending (or worsening) shortage in mammography personnel at all levels—from radiologist to technologist, in academia and in private practice—presents both barrier and impetus to innovation. Development may be driven by the need to improve process and efficiency to meet the growing demand for screening, but it may also be impeded by a lack of professionals who can assess and appropriately apply new technologies. The latter situation may occur if recruitment into the field of breast imaging fails to keep pace with the demand for screening, but it may also result from technical and regulatory demands that increase workload.
In today’s cost-conscious medical environment, even novel technologies that improve patient outcome present problems if they necessitate new investments in training and equipment.73 If such technologies expand—rather than replace—current methods for breast cancer detection and diagnosis, then innovation may further burden an overtaxed workforce.73 For example, as noted in Mammography and Beyond, current practice guidelines and standards of care for breast cancer detection, which are already complex, may be made more complicated with the incorporation of new technologies.
Breast imagers are essential not only in preserving access to breast cancer screening, but also in the evaluation of new technologies and the adoption of technological advances in breast cancer detection that will improve health outcomes in the future. The speed of adoption for new technologies and evidence-based practice guidelines for breast cancer detection is likely to vary according to region (urban versus rural) and clinical setting (community clinic versus academic health center). However, the rate of innovation ultimately is determined by the actions of individual physicians, who must be persuaded of the potential value of a new technique or technology if it is to become widely available. Appropriate research also has the potential to reduce the spread of inappropriate technologies such as those that increase costs but do not improve outcomes.
Righting the Balance of Supply and Demand
Where radiological services are in short supply, institutions have adopted a variety of short-term measures to meet their needs. They have used offsite “moonlighters” to take the place of attending physicians; called in retired radiologists; established cross-coverage agreements to share specialists among (sometimes competing) hospitals; and offered flexible schedules and the opportunity to work at home as a way to make a comparatively low-paying job more attractive.1 Many radiology facilities are increasingly relying on foreign radiologists.
Teleradiology has also been used in a variety of settings with limited local imaging capacity. The Air Force, anticipating the loss of half of its radiology staff between 2002 and 2005, developed a network that links radiologists at eight stateside hospitals operated by the U.S. Army, Air Force, or Navy with eight overseas hospitals (as of April 2003).21 According to an Air Force spokesperson, teleradiology “will not fix the shortage, but it will make maximum use of the radiologists we currently have.”117 Some U.S. hospitals have used similar arrangements to outsource interpretation—such as the reading of emergency scans performed during the night—to radiologists overseas.98 Although this scenario raises concerns about maintaining the quality of services, it presents a model that could be
adapted to establish centralized domestic facilities for mammographic interpretation, as described earlier in this chapter under the section Organization of Screening Services.
The supply of radiologists (and therefore of breast imagers) is unlikely to grow as quickly as demand for their expertise. Significant barriers exist to expanding training programs, and to permitting well-trained radiologists to immigrate to the United States.5 A freeze—at 1996 levels—on the number of house officers at institutions supported by CMS effectively limits the growth of residency programs, even for fields in such high demand as radiology. Visa restrictions, in addition to the burden of state and hospital licensure and ACR certification requirements, inhibit the immigration of highly qualified foreign radiologists. Given these circumstances, the committee focused on optimizing the productivity of the limited and increasingly precious supply of radiologists who interpret mammograms.
Expanding Capacity with Physician Extenders
Physician extenders such as physician assistants (PAs) and nurse practitioners have long been employed to increase capacity in a variety of medical specialties and settings, and their numbers are growing rapidly. Until recently, hospitals with residency training programs tended to rely on residents, whose costs were paid by CMS and who worked comparatively long hours, to perform duties that could have been performed by non-physicians.5 Today, with the number of residents capped and residents’ hours limited by new work rules, hospitals are hiring more physician extenders. Nonphysician provider training programs produced twice as many graduates in 1997 as they did in 1992, and several state legislatures have passed laws enabling them to practice.5 Until now, however, this trend has largely bypassed radiology, which employs less than 0.5 percent of all PAs.5
The position of radiology PA was introduced in the 1970s. At that time, candidates trained in baccalaureate programs at the University of Kentucky and at Duke and Brown Universities, but they have since closed due to lack of interest.5 In 1996, responding to a shortage of radiologists in the armed services, the Department of Defense (DoD) supported the launch of a baccalaureate training program at Weber State University in Utah for a distinct position known as the Radiology Practitioner Assistant. Although the DoD no longer funds this program, it continues to produce graduates, who are eligible for certification by their own board.
Most recently, the ACR and the American Society of Radiological Technologists (ASRT) have jointly defined a new physician extender with expertise in medical imaging, the radiological associate (RA).2 The RA offers a partial solution to the continuing chronic shortage of personnel in the field of radiology by simultaneously reducing radiologists’ workload
and establishing a career path by which to attract and retain radiation technologists (RTs), who are also in short supply, to the field. The RA’s duties are proposed to include patient management, certain routine procedures under direct supervision of a radiologist, and the communication of results to referring physicians; image interpretation, even on a preliminary basis, is expressly excluded. However, because individual states control licensing and regulation of nonphysician practitioners, an RA’s role and responsibilities may depend on his or her location.5,111 With ASRT funding, RA training programs catering to experienced RTs have been initiated at Loma Linda University, Midwestern State University, the University of North Carolina, and the University of Medicine and Dentistry in New Jersey, and are being developed at eight additional institutions.5
Should Nonphysicians Interpret Mammograms?
While RAs would enable breast imagers to focus on image interpretation and biopsies, physician extenders who interpret screening mammograms under the supervision of breast imaging specialists can further extend capacity. Evidence suggests that RTs could be specially trained to prescreen mammograms for the presence or absence of abnormalities25,111 or to double-read mammograms along with a radiologist.122 A series of studies supports the prospect for training and evaluation of physician extenders in mammographic interpretation.
Some of the most relevant experience of nonphysicians interpreting screening mammograms has occurred in the United Kingdom, where—despite a less burdensome malpractice environment and a different reimbursement structure—there is also a long-standing shortage of radiologists who interpret mammograms.40,122 One English “rapid access” breast clinic faced with unacceptably long wait times due to increased demand for mammograms coped with this problem by allowing mammograms of patients meeting specific criteria for concern to be interpreted initially by two specially trained RTs.25e (Candidate patients were not pregnant or lactating and were over 35, without clinically obvious cancer or axillary problems, but with worrisome symptoms such as breast pain, palpable masses, Paget’s disease, or nipple discharge.) Their findings were later reviewed by two radiologists, who decided whether the women should be recalled for additional studies. In 511 women thus screened and tracked for approximately two years, the sensitivity, specificity, and accuracy obtained by the RTs was comparable to that of radiologists. The women reportedly did not object when informed that an RT would provide the initial read on their exams.
In another study conducted in the United Kingdom, three RTs in a hospital that was otherwise unable to maintain double reading requirements for mammograms were trained to meet national certification standards.122 The trained radiographers performed well and were comparable in both accuracy and speed to that of the four participating radiologists.
A 6-week study of 33 experienced RTs conducted in a U.S. hospital found that even without training, the technologists could classify screening mammograms as to their need for additional workup (such as additional views, sonography, or biopsy) with “reasonable” accuracy.111 The technologists assigned each of the more than 3,000 women whose mammograms they reviewed to one of two categories: either she needed additional workup to explore possible abnormalities, or no workup was required. The RTs’ classification of more than 80 percent of these cases matched those of the hospital’s nine radiologists, and the RTs identified most of the cases that later proved to be malignant.
Each of the studies described above involved the interpretation of screening mammograms by RTs working under the supervision of a board-certified radiologist, and do not apply to diagnostic mammograms. The Committee does not suggest that RTs should interpret diagnostic mammograms or that screening mammograms should be interpreted solely by an RT; rather, they would work to expand the capacity of radiologists. RTs could also take on other tasks commonly performed by radiologists such as filling out forms, dictating, and hanging and taking down films. Having a physician extender support the work of breast imagers would add the cost of an extra salary, but this could result in overall reduced costs if they could take on other tasks commonly done by radiologists.
The most important requirement that would have to be met if nonphysicians were to interpret mammograms would be that the quality of the mammography service was shown to improve, or at least, did not decline. In fact, the use of physician extenders for double-reading has the potential to increase quality. Because double-reading by a second radiologist is not reimbursed by Medicare, few mammography facilities can afford to have two radiologists interpret each mammogram, even though this practice is known to improve sensitivity. Another requirement would be that mammograms interpreted by physician extenders should also be viewed by an interpreting radiologist.
Challenges to this proposal include the acceptance of the radiology profession and malpractice coverage. In May 2004 the ACR leadership council recently voted against a proposal to allow the interpretation of any imaging examination by nonphysicians. However, resistance to expanding professional boundaries has been overcome by other medical specialties, for example, the use of nurse practitioners and midwives in obstetrics and gynecology; these professionals have also dealt with similar malpractice
issues. Indeed, it is even conceivable that malpractice rates for radiologists could be lowered if they adopted the practice of double-reading made possible by supportive assistance from nonphysicians with specialized training in mammography.
The MQSA stipulates that mammograms are to be interpreted only by a physician specifically certified in mammography (Box 3-8). The Act does not, however, preclude other personnel from examining the mammograms that are also interpreted by certified physicians. Although not widely appreciated and rarely practiced, it would in fact be permissible within the provisions of the MQSA to have nonphysician personnel examine mammograms—as long as a certified physician signed the mammogram report indicating that he or she had interpreted it. This suggestion that physician
|
BOX 3-8 This includes only the initial requirements established by law in the MQSA. Further requirements are set by the FDA in the Code of Federal Regulations for Mammography, 21 CFR 900.12. Interpreting Physician—Interprets mammograms
Radiologic Technologist—Performs mammograms
Medical Physicist—Surveys mammography equipment and oversees quality assurance practices
|
extenders could be enlisted to help read mammograms would thus offer women a more thorough examination than is currently typical of most mammography facilities where mammograms are viewed only by a single breast imager. Physician extenders could potentially improve the overall accuracy of mammographic interpretation through double reading, as well as alleviate the burden on the breast imaging physicians by prescreening the mammograms to allow the interpreting physician to spend more time on the more problematic mammograms.
SUMMARY
Mammography is not a perfect screening technology, but it can reduce mortality from breast cancer. However, there is wide variation in performance among breast imaging facilities and individual breast imagers. Organizing breast screening services to increase the utilization of services as well as their quality and efficiency should thus be priority for health care payers and providers.
Approaches to improving mammography that need to be examined include organizational changes such as those implemented in some European countries including limiting interpretation to more expert and experienced breast imagers, and regionalization and reading at a central location. Although the evidence for how such changes might improve mammography in the United States is mixed, they have led to improvements in some European countries. Certainly, better accuracy and lower rates of callbacks and false positives should result in more cost-effective care.
Even without the single-payer, universal access health care system common to all other developed countries, screening services in the United States could adapt many features of those systems to regionally based programs. As pointedly noted by Harmon Eyre and his colleagues at the American Cancer Society:39
Screening under opportunistic condition rather than through a system is inefficient at both the individual level and population level; moreover, without a system, there is no readiness to implement any new early detection technology that could improve disease control.
A comprehensive system of early-detection potentially not only leads to high levels of participation but also insures that all the elements of a program of early detection an intervention are highly competent, interrelated, and inter-dependent. A system has the potential not only to increase quality but also to reduce the volume of small errors that contribute to incremental erosions of efficiency….
While there are many practical barriers that must be overcome to establish true population-based screening programs, a system of organized
screening holds the greatest potential to realize the benefits of reducing the incidence rate of advanced cancers.
Harmon J. Eyre, Robert A. Smith, Curtis J. Mettlin
Cancer Screening and Early Detection, 2003
State and federal legislators have taken an active role in exploring ways to improve breast cancer detection. The federal Mammography Quality Standards Act represents an unusual governmental intervention aimed at, and successful in, improving the technical quality of mammography. In the future, the MQSA may address the vexing problem of variation in radiologist interpretation of breast images. In spite of the impression left by widespread coverage in the national media,f differences among radiologists are not the largest component of the problem of inconsistency in interpretation. Other factors, notably organizational factors, have received much less attention, and are more difficult to control than individual factors, such as volume or training requirements.
New technologies, such as digital mammography, CAD, and MRI are being examined. Although they have advantages in some situations that may justify their use, they can add significant costs, and their value as improvements in sensitivity and specificity over screen-film mammography has not been established.
Organizational factors such as double reading by two radiologists improves accuracy; high volume centers on average have higher accuracy, above and beyond the increase attributable to reading volume of individual radiologists; and use of CAD can reduce the variability in mammographic interpretation among different readers.
As the U.S. population ages, demand for mammography will rise at a time when supply of personnel and facilities appears to be falling, increasingly threatening access. Among other things, low reimbursement and the unattractiveness of breast imaging as a subspecialty due to stress and malpractice litigation seem to be driving the impending shortages. Other problems include a dearth of radiologist researchers to conduct trials and investigate new approaches to breast cancer detection. To address the shortage of mammographers, expansion of responsibilities by nonphysicians to include preliminary interpretation of images should be considered. A key to improving mammographic interpretation is to reduce known and controllable sources of variability in quality, but at the same time to avoid adding to the burden of an already overextended workforce.
REFERENCES
1. The radiology shortage: will it continue? 2003. Radiological Society of North America News 13(2):10-11.
2. Radiology assistants will share workload in diagnostic imaging. 2004. Radiological Society of North America News 14(2):5-6.
3. American Cancer Society. 2003. Breast Cancer Facts and Figures 2003-2004. Atlanta, GA: American Cancer Society.
4. American College of Radiology. ACR Mammography Practice Cost Survey Executive Summary. Unpublished as of March 9, 2004.
5. American College of Radiology. Intersociety Conference 2003 Summary. Unpublished as of March 9, 2004.
6. American College of Radiology Imaging Network (ACRIN). Current Protocols: Screening Breast Ultrasound in High-Risk Women (ACRIN Protocol A6666-Open). Accessed May 12, 2004. Web Page. Available at: http://www.acrin.org/current_protocols.html#A6666.
7. Andrews FJ. 2001. Pain during mammography: implications for breast screening programmes. Australas Radiol 45(2):113-117.
8. Barroso J, McMillan S, Casey L, Gibson W, Kaminski G, Meyer J. 2000. Comparison between African-American and white women in their beliefs about breast cancer and their health locus of control. Cancer Nurs 23(4):268-276.
9. Bassett LW, Monsees BS, Smith RA, Wang L, Hooshi P, Farria DM, Sayre JW, Feig SA, Jackson VP. 2003. Survey of radiology residents: breast imaging training and attitudes. Radiology 227(3):862-869.
10. Beam CA, Conant EF, Sickles EA. 2002. Factors affecting radiologist inconsistency in screening mammography. Acad Radiol 9(5):531-540.
11. Beam CA, Conant EF, Sickles EA. 2003. Association of volume and volume-independent factors with accuracy in screening mammogram interpretation. J Natl Cancer Inst 95(4):282-290.
12. Beam CA, Conant EF, Sickles EA, Weinstein SP. 2003. Evaluation of proscriptive health care policy implementation in screening mammography. Radiology 229(2):534-540.
13. Berlin L. 2003. Missed mammographic abnormalities, malpractice, and expert wit-nesses: does majority rule in the courtroom? Radiology 229(1):288; author reply 289.
14. Berlin L. 2003. Testimony of Leonard Berlin, M.D. Regarding the Mammography Quality Standards Act Reauthorization. Senate Committee on Health, Education, Labor, and Pensions.
15. Betsill WL Jr, Rosen PP, Lieberman PH, Robbins GF. 1978. Intraductal carcinoma. Long-term follow-up after treatment by biopsy alone. JAMA 239(18):1863-1867.
16. Birdwell RL, Wilcox PA. 2001. The Mammography Quality Standards Act: Benefits and Burdens. Pisano ED, Editor. Breast Imaging. Amsterdam, Oxford: IOS Press.
17. Bovbjerg RR, Miller RH, Shapiro DW. 2001. Paths to reducing medical injury: professional liability and discipline vs. patient safety—and the need for a third way. J Law Med Ethics 29(3-4):369-380.
18. Boyle P, Autier P, Bartelink H, Baselga J, Boffetta P, Burn J, Burns HJ, Christensen L, Denis L, Dicato M, Diehl V, Doll R, Franceschi S, Gillis CR, Gray N, Griciute L, Hackshaw A, Kasler M, Kogevinas M, Kvinnsland S, La Vecchia C, Levi F, McVie JG, Maisonneuve P, Martin-Moreno JM, Bishop JN, Oleari F, Perrin P, Quinn M, Richards M, Ringborg U, Scully C, Siracka E, Storm H, Tubiana M, Tursz T, Veronesi U, Wald N, Weber W, Zaridze DG, Zatonski W, zur Hausen H. 2003. European Code Against Cancer and scientific justification: third version (2003). Ann Oncol 14(7):973-1005.
19. Boyle P, d’Onofrio A, Maisonneuve P, Severi G, Robertson C, Tubiana M, Veronesi U. 2003. Measuring progress against cancer in Europe: has the 15% decline targeted for 2000 come about? Ann Oncol 14(8):1312-1325.
20. Brennan TA, Sox CM, Burstin HR. 1996. Relation between negligent adverse events and the outcomes of medical-malpractice litigation. N Engl J Med 335(26):1963-1967.
21. Brewin B. 2003, February 12. DOD system brings medical expertise closer. Accessed April 11, 2003. Web Page. Available at: http://www.bio-itworld.com/news/021203_report2006.html.
22. Burnside E, Belkora J, Esserman L. 2001. The impact of alternative practices on the cost and quality of mammographic screening in the United States. Clin Breast Cancer 2(2):145-152.
23. Butler P (American College of Radiology). Personal communication, March 1, 2004.
24. Carney PA, Miglioretti DL, Yankaskas BC, Kerlikowske K, Rosenberg R, Rutter CM, Geller BM, Abraham LA, Taplin SH, Dignan M, Cutter G, Ballard-Barbash R. 2003. Individual and combined effects of age, breast density, and hormone replacement therapy use on the accuracy of screening mammography. Ann Intern Med 138(3):168-175.
25. Casey B. 2003, March 11. Breast center enlists radiographers for first look at mammograms. Accessed February 19, 2004. Web Page. Available at: http://www.auntminnie.com/default.asp?Sec=sup&Sub=wom&Pag=dis&ItemId=57614&stm=radiographers.
26. CDC (Centers for Disease Control and Prevention). 2003. The National Breast and Cervical Cancer Early Detection Program: Reducing Mortality Through Screening. Atlanta, GA: Centers for Disease Control and Prevention: Department of Health and Human Services.
27. Christiansen CL, Wang F, Barton MB, Kreuter W, Elmore JG, Gelfand AE, Fletcher SW. 2000. Predicting the cumulative risk of false-positive mammograms. J Natl Cancer Inst 92(20):1657-1666.
28. Chu KC, Lamar CA, Freeman HP. 2003. Racial disparities in breast carcinoma survival rates: separating factors that affect diagnosis from factors that affect treatment. Cancer 97(11):2853-2860.
29. Cyrlak D. 1988. Induced costs of low-cost screening mammography. Radiology 168(3):661-663.
30. D’Orsi CJ. 2001. Computer-aided detection: there is no free lunch. Radiology 221(3): 585-586.
31. Dakins DR. 2002. Mammography debate raises digital quality bar. Diagnostic Imaging. Com. Accessed March 3, 2002. Web Page. Available at: http://www.dimag.com/db_area/archives/2002/.
32. Earp JA, Eng E, O’Malley MS, Altpeter M, Rauscher G, Mayne L, Mathews HF, Lynch KS, Qaqish B. 2002. Increasing use of mammography among older, rural African American women: results from a community trial. Am J Public Health 92(4):646-654.
33. Eisenberg D, Sieger M, Tsiantar D, Berryman A, Cuadros P, Peltier M. 2003. The doctor won’t see you now. Time 161(23):46-56.
34. Elmore JG, Carney PA. 2002. Does practice make perfect when interpreting mammography? J Natl Cancer Inst 94(5):321-323.
35. Elmore JG, Miglioretti DL, Reisch LM, Barton MB, Kreuter W, Christiansen CL, Fletcher SW. 2002. Screening mammograms by community radiologists: variability in false-positive rates. J Natl Cancer Inst 94(18):1373-1380.
36. Elmore JG, Nakano CY, Koepsell TD, Desnick LM, D’Orsi CJ, Ransohoff DF. 2003. International variation in screening mammography interpretations in community-based programs. J Natl Cancer Inst 95(18):1384-1393.
37. Enzmann DAPHCVL. 2001. Providing professional mammography services: financial analysis. Health Policy Prac 219:467-473.
38. Esserman L, Cowley H, Eberle C, Kirkpatrick A, Chang S, Berbaum K, Gale A. 2002. Improving the accuracy of mammography: volume and outcome relationships. J Natl Cancer Inst 94(5):369-375.
39. Eyre HJ, Smith RA, Mettlin CJ. 2003. Cancer Screening and Early Detection. In: Holland JF, Frei E, Bast RC, Kufe DW, Pollack RE, Weichselbaum RR, Editors. Cancer Medicine. 6th ed. Ontario: BC Decker.
40. Field S. 1996. UK radiology workforce survey—breast imaging services. Royal College of Radiologists Newsletter 45:10-12.
41. Fisher B, Dignam J, Wolmark N, Mamounas E, Costantino J, Poller W, Fisher ER, Wickerham DL, Deutsch M, Margolese R, Dimitrov N, Kavanah M. 1998. Lumpectomy and radiation therapy for the treatment of intraductal breast cancer: findings from National Surgical Adjuvant Breast and Bowel Project B-17. J Clin Oncol 16(2):441-452.
42. Fletcher SW, Elmore JG. 2003. Clinical practice. Mammographic screening for breast cancer. N Engl J Med 348(17):1672-1680.
43. Foote SB, Blewett LA. 2003. Politics of prevention: expanding prevention benefits in the Medicare program. J Public Health Policy 24(1):26-40.
44. Fowler BA. 2000. Variability in mammography screening legislation across the states. J Womens Health Gender-Based Med 9(2):175-184.
45. Freeman H. 2004. Reducing Disparities in Cancer. Fulfilling the Potential of Cancer Prevention and Early Detection: An American Cancer Society and Institute of Medicine Symposium. Washington, DC: The National Academies Press.
46. Freer TW, Ulissey MJ. 2001. Screening mammography with computer-aided detection: prospective study of 12,860 patients in a community breast center. Radiology 220(3):781-786.
47. Fronstin P. 2000. Sources of Health Insurance and Characteristics of the Uninsured: Analysis of the March 2000 Current Population Survey. EBRI Issue Brief Number 228 ed. Washington, DC: Employee Benefits Research Institute.
48. Gelijns AC, Thier SO. 2002. Medical innovation and institutional interdependence: rethinking university-industry connections. JAMA 287(1):72-77.
49. Gur D, Sumkin JH, Rockette HE, Ganott M, Hakim C, Hardesty L, Poller WR, Shah R, Wallace L. 2004. Changes in breast cancer detection and mammography recall rates after the introduction of a computer-aided detection system. J Natl Cancer Inst 96(3):185-190.
50. Han B, Wells BL, Primas M. 2003. Comparison of mammography use by older black and white women. J Am Geriatr Soc 51(2):203-212.
51. Harvey SC, Geller B, Oppenheimer RG, Pinet M, Riddell L, Garra B. 2003. Increase in cancer detection and recall rates with independent double interpretation of screening mammography. Am J Roentgenol 180(5):1461-1467.
52. Hayes JC. 2002, December 3. Annual oration analyzes mammography controversies. Web Page. Available at: www.dimag.com/cgi-bin/webcast02/display_news.cgi?105.
53. Hwang ES, Kinkel K, Esserman LJ, Lu Y, Weidner N, Hylton NM. 2003. Magnetic resonance imaging in patients diagnosed with ductal carcinoma-in-situ: value in the diagnosis of residual disease, occult invasion, and multicentricity. Ann Surg Oncol 10(4):381-388.
54. Institute of Medicine. 2001. Crossing the Quality Chasm: A New Health System for the 21st Century. Washington, DC: National Academy Press.
55. Institute of Medicine. 2001. Interpreting the Volume-Outcome Relationship in the Context of Cancer Care. Washington DC: National Academy Press.
56. Institute of Medicine. 2001. Mammography and Beyond: Developing Technologies for the Early Detection of Breast Cancer. Washington, DC: National Academy Press.
57. Institute of Medicine. 2002. Care Without Coverage: Too Little, Too Late. Washington, DC: The National Academies Press.
58. Institute of Medicine. 2003. Fulfilling the Potential of Cancer Prevention and Early Detection. Washington DC: The National Academies Press.
59. James JJ. 2004. The current status of digital mammography. Clin Radiol 59(1):1-10.
60. Jones BA, Patterson EA, Calvocoressi L. 2003. Mammography screening in African American women: evaluating the research. Cancer 97(1 Suppl):258-272.
61. Joslyn SA, West MM. 2000. Racial differences in breast carcinoma survival. Cancer 88(1):1114-1123.
62. Kan L, Olivotto IA, Warren Burhenne LJ, Sickles EA, Coldman AJ. 2000. Standardized abnormal interpretation and cancer detection ratios to assess reading volume and reader performance in a breast screening program. Radiology 215(2):563-567.
63. Kerlikowske K, Carney PA, Geller B, Mandelson MT, Taplin SH, Malvin K, Ernster V, Urban N, Cutter G, Rosenberg R, Ballard-Barbash R. 2000. Performance of screening mammography among women with and without a first-degree relative with breast cancer. Ann Intern Med 133(11):855-863.
64. Kerlikowske K, Smith-Bindman R, Ljung BM, Grady D. 2003. Evaluation of abnormal mammography results and palpable breast abnormalities. Ann Intern Med 139(4):274-284.
65. Kolb TM, Lichy J, Newhouse JH. 2002. Comparison of the performance of screening mammography, physical examination, and breast US and evaluation of factors that influence them: an analysis of 27,825 patient evaluations. Radiology 225(1):165-175.
66. Kopans DB. 2004. Sonography should not be used for breast cancer screening until its efficacy has been proven scientifically. Am J Roentgenol 182(2):489-491.
67. Kriege M, Brekelmans CT, Boetes C, Rutgers EJ, Oosterwijk JC, Tollenaar RA, Manoliu RA, Holland R, de Koning HJ, Klijn JG. 2001. MRI screening for breast cancer in women with familial or genetic predisposition: design of the Dutch National Study (MRISC). Fam Cancer 1(3-4):163-168.
68. Kubota M, Inoue K, Koh S, Sato T, Sugita T. 2003. Role of ultrasonography in treatment selection. Breast Cancer 10(3):188-197.
69. Lehman C, Holt S, Peacock S, White E, Urban N. 2002. Use of the American College of Radiology BI-RADS guidelines by community radiologists: concordance of assessments and recommendations assigned to screening mammograms. Am J Roentgenol 179(1):15-20.
70. Lewin JM, Hendrick RE, D’Orsi CJ, Isaacs PK, Moss LJ, Karellas A, Sisney GA, Kuni CC, Cutter GR. 2001. Comparison of full-field digital mammography with screen-film mammography for cancer detection: results of 4,945 paired examinations. Radiology 218(3):873-880.
71. Li CI, Malone KE, Daling JR. 2003. Differences in breast cancer stage, treatment, and survival by race and ethnicity. Arch Intern Med 163(1):49-56.
72. Liang W, Burnett CB, Rowland JH, Meropol NJ, Eggert L, Hwang YT, Silliman RA, Weeks JC, Mandelblatt JS. 2002. Communication between physicians and older women with localized breast cancer: implications for treatment and patient satisfaction. J Clin Oncol 20(4):1008-1016.
73. Lohnberg A, van der Meulen B, Brown N, Nelis A, Rappert B, Webster A, Cabello C, Rosales M, Sanz-Menéndez L. 1999. Studying Innovation Strategies for Future Medical Technologies: Conceptual Framework and Methodologies for the FORMAKIN Project. European Commission Targeted Socio-Economic Research Programme.
74. Lynge E, Olsen AH, Fracheboud J, Patnick J. 2003. Reporting of performance indicators of mammography screening in Europe. Eur J Cancer Prev 12(3):213-222.
75. Maguire P. 2003. Is an access crisis on the horizon in mammography? ACP Observer.
76. Mainiero MB. 2003. Breast-Imaging Specialists Are More Efficient Than General Radiologists at Reading Mammograms. 103rd Annual American Roentgen Ray Society Meeting. San Diego, CA.
77. Marbella AM, Layde PM. 2001. Racial trends in age-specific breast cancer mortality rates in US women. Am J Public Health 91(1):118-121.
78. Marshall MN, Shekelle PG, Leatherman S, Brook RH. 2000. The public release of performance data: what do we expect to gain? A review of the evidence. JAMA 283(14):1866-1874.
79. Meadows S, Wingert P, Rosenberg D, Carmichael M, Johnson D, Childress S, Sinderbrand R, Breslau K, Shenfeld H. 2003. Civil Wars. Newsweek. Pp. 43-51.
80. Mehta TS. 2003. Current uses of ultrasound in the evaluation of the breast. Radiol Clin North Am 41(4):841-856.
81. Meyer JE, Eberlein TJ, Stomper PC, Sonnenfeld MR. 1990. Biopsy of occult breast lesions. Analysis of 1261 abnormalities. JAMA 263(17):2341-2343.
82. Miller AM, Champion VL. 1997. Attitudes about breast cancer and mammography: racial, income, and educational differences. Women Health 26(1):41-63.
83. Mitchell J, Lannin DR, Mathews HF, Swanson MS. 2002. Religious beliefs and breast cancer screening. J Womens Health (Larchmt) 11(10):907-915.
84. Mitka M. 2003. Researchers seek mammography alternatives. JAMA 290(4):450-451.
85. Monsees B. 2002. Is the GAO report on mammography correct? SBI News.
86. Monsees B. 2002. The breast imaging profession: take my job, please! SBI News.
87. Mourad WG. 2003, May 5. Mammography equipment evaluations and the annual survey—is your equipment up to the task? Accessed March 24, 2004. Web Page. Available at: http://www.fda.gov/cdrh/mammography/scorecard-article4.html.
88. National Cancer Institute. 2004. Fifth National Forum on Biomedical Imaging in Oncology Meeting Summary. Bethesda, MD.
89. National Health Service. Cancer Screening Programmes. 2003. NHS Breast Screening Programme Annual Review 2003.
90. Newman LA, Mason J, Cote D, Vin Y, Carolin K, Bouwman D, Colditz GA. 2002. African-American ethnicity, socioeconomic status, and breast cancer survival: a metaanalysis of 14 studies involving over 10,000 African-American and 40,000 White American patients with carcinoma of the breast. Cancer 94(11):2844-2854.
91. Oluwole SF, Ali AO, Adu A, Blane BP, Barlow B, Oropeza R, Freeman HP. 2003. Impact of a cancer screening program on breast cancer stage at diagnosis in a medically underserved urban community. J Am Coll Surg 196(2):180-188.
92. Pal S. 2003, November 30. Women with genetic history of breast cancer benefit from MR screening. Accessed February 19, 2004. Web Page. Available at: http://www.auntminnie.com/default.asp?Sec=rca&Sub=rsna_2003&pag=dis&ItemId=60243&stm=November+30%2C+2003+pal.
93. Pallarito K. 2003, February 27. Spike in malpractice premiums hurting access to care—survery. Accessed February 28, 2003. Web Page. Available at: http://www.auntminnie.com/default.asp?Sec=sup&Sub=imc&Pag=dis&ItemId=57516.
94. Pasko T, Seidman B (American Medical Association). 2002. Physician Characteristics and Distribution in the US, 2002-2003 Edition. Chicago, IL: AMA Press.
95. Philadelphia Black Women’s Health Project. 2002. Detection, Diagnosis, and Prevention of Breast Cancer. Accessed June 9, 2003. Web Page. Available at: http://www.blackwomenshealthproject.org/aabreastcancer.htm.
96. Physician Insurers Association of America. 1995. PIAA Breast Cancer Study. Rockville, MD: PIAA.
97. Physician Insurers Association of America. 2002. PIAA Breast Cancer Study. Rockville, MD: PIAA.
98. Pollack A. 2003, November 16. Who’s reading your x-ray? The New York Times. 3. P. 1.
99. Russell A, Langlois T, Johnson G, Trentham-Dietz A, Remington P. 1999. Increasing gap in breast cancer mortality between black and white women. WMJ 98(8):37-39.
100. Sant M. 2001. Differences in stage and therapy for breast cancer across Europe. Int J Cancer 93(6):894-901.
101. Sant M, Allemani C, Berrino F, Coleman MP, Aareleid T, Chaplain G, Coebergh JW, Colonna M, Crosignani P, Danzon A, Federico M, Gafa L, Grosclaude P, Hedelin G, Mace-Lesech J, Garcia CM, Moller H, Paci E, Raverdy N, Tretarre B, Williams EM. 2004. Breast carcinoma survival in Europe and the United States. Cancer 100(4):715-722.
102. Sant M, Capocaccia R, Coleman MP, Berrino F, Gatta G, Micheli A, Verdecchia A, Faivre J, Hakulinen T, Coebergh JW, Martinez-Garcia C, Forman D, Zappone A. 2001. Cancer survival increases in Europe, but international differences remain wide. Eur J Cancer 37(13):1659-1667.
103. Schnall MD. 2001. Application of magnetic resonance imaging to early detection of breast cancer. Breast Cancer Res 3(1):17-21.
104. Schnall MD. 2003. Breast MR imaging. Radiol Clin North Am 41(1):43-50.
105. Sickles EA, Wolverton DE, Dee KE. 2002. Performance parameters for screening and diagnostic mammography: specialist and general radiologists. Radiology 224(3):861-869.
106. Skaane P, Young K, Skjennald A. 2003. Population-based mammography screening: comparison of screen-film and full-field digital mammography with soft-copying reading—Oslo I study. Radiology 229(3):877-884.
107. Smith-Bindman R, Chu PW, Miglioretti DL, Sickles EA, Blanks R, Ballard-Barbash R, Bobo JK, Lee NC, Wallis MG, Patnick J, Kerlikowske K. 2003. Comparison of screening mammography in the United States and the United Kingdom. JAMA 290(16):2129-2137.
108. Smith RA, Wender RC. 2004. Cancer screening and the periodic health examination. Cancer 100(8):1553-1557.
109. Society of Breast Imaging. 2003, October 24. SBI response to JAMA article. Web Page. Available at: http://www.sbi-online.org/JAMA_Response.htm.
110. Studdert DM, Mello MM, Brennan TA. 2004. Medical malpractice. N Engl J Med 350(3):283-292.
111. Sumkin JH, Klaman HM, Graham M, Ruskauff T, Gennari RC, King JL, Klym AH, Ganott MA, Gur D. 2003. Prescreening mammography by technologists: a preliminary assessment. Am J Roentgenol 180(1):253-256.
112. Sunshine JH, Burkhardt JH. 2000. Radiology groups’ workload in relative value units and factors affecting it. Radiology 214(3):815-822.
113. Sunshine JH, Cypel YS, Schepps B. 2002. Diagnostic radiologists in 2000: basic characteristics, practices, and issues related to the radiologist shortage. Am J Roentgenol 178(2):291-301.
114. Surveillance Epidemiology and End Results (SEER) Program (www.seer.cancer.gov). 2003. SEER*Stat Database: Incidence—SEER 9 Registry Public-Use, Nov. 2002 Submission (1973-2000). National Cancer Institute, DCCPS, Surveillance Research Program, Cancer Statistics Branch.
115. Tabar L, Yen MF, Vitak B, Chen HH, Smith RA, Duffy SW. 2003. Mammography service screening and mortality in breast cancer patients: 20-year follow-up before and after introduction of screening. Lancet 361(9367):1405-1410.
116. Taplin SH, Ichikawa LE, Kerlikowske K, Ernster VL, Rosenberg RD, Yankaskas BC, Carney PA, Geller BM, Urban N, Dignan MB, Barlow WE, Ballard-Barbash R, Sickles EA. 2002. Concordance of breast imaging reporting and data system assessments and management recommendations in screening mammography. Radiology 222(2):529-535.
117. Trevino M. 2003. Air Force Teleradiology Project aims to alleviate staff shortages: plan could even out workflow and improve access to subspecialty reads. Accessed May 13, 2002. Web Page. Available at: http://www.diagnosticimaging.com/pacsweb/cover/cover05090202.shtml.
118. U.S. Food and Drug Administration. 2003, June 3. About Mammography Quality Standards Act (MQSA). Web Page. Available at: http://www.fda.gov/cdrh/mammography/mqsa-rev.html.
119. United States General Accounting Office. 2002. Mammography—Capacity Generally Exists to Deliver Services. Washington, DC: GAO.
120. United States General Accounting Office. 2003. Medical Malpractice: Implications of Rising Premiums on Access to Health Care. Washington, DC: GAO.
121. Viehweg P, Lampe D, Buchmann J, Heywang-Kobrunner SH. 2000. In situ and minimally invasive breast cancer: morphologic and kinetic features on contrast-enhanced MR imaging. MAGMA 11(3):129-137.
122. Wivell G, Denton ER, Eve CB, Inglis JC, Harvey I. 2003. Can radiographers read screening mammograms? Clin Radiol 58(1):63-67.
123. Wojcik BE, Spinks MK, Stein CR. 2003. Effects of screening mammography on the comparative survival rates of African American, white, and Hispanic beneficiaries of a comprehensive health care system. Breast J 9(3):175-183.
124. Wooding D. 2003, July 8. PERsonal perFORmance in Mammographic Screening. Accessed May 12, 2004. Web Page. Available at: http://ibs.derby.ac.uk/performs/index.shtml.
125. Zheng B, Hardesty LA, Poller WR, Sumkin JH, Golla S. 2003. Mammography with computer-aided detection: reproducibility assessment initial experience. Radiology 228(1):58-62.




























































