2
Opportunities and Challenges
There markable focus and unprecedented funding currently being directed toward providing affordable antiretrovirals (ARVs) to resource-poor countries and accelerating HIV/AIDS treatment and prevention programs worldwide reflect an admirable and ambitious shift in humanitarian, political, and economic perspectives on what is arguably the worst pandemic ever to hit humankind (see Box 2-1).
Many initiatives, organizations, and governments—including the Global Fund to Fight AIDS, Tuberculosis and Malaria (The Global Fund); U.S. funding for global HIV/AIDS efforts, in particular the President’s Emer-
|
BOX 2-1
SOURCES: Quinn and Chaisson, 2004; WHO, 2003a,b. |
gency Plan for AIDS Relief (PEPFAR); the World Health Organization’s (WHO) 3-by-5 campaign; the William J. Clinton Presidential Foundation; the World Bank’s Multi-Country HIV/AIDS Program for the Africa Region (MAP); and the courageous efforts of the afflicted countries themselves, such as ART scale-up in South Africa—have contributed to this historic opportunity to develop and implement the global scale-up of ARV therapy (ART). Each of these efforts is briefly reviewed in turn below. Issues related to coordinating these and other elements of the global response to the HIV/ AIDS pandemic are then examined. Next, the crucial issue of fiscal sustainability is addressed. The final section examines the ethical issues and challenges at both the local and international levels.
CURRENT EFFORTS
The Global Fund: Scale-Up of Antiretroviral Therapy in Action
The Global Fund to Fight AIDS, Tuberculosis, and Malaria, sponsored by the United Nations (UN), is a performance-based financing mechanism and network whose purpose is to provide countries with substantial resources that can be used at the national level and in a way that promotes country ownership. The goal is to help launch programs designed to combat these three devastating diseases. Although the Global Fund has been operating legally for nearly 2½ years, it has been functional for only about 2 years. Thus far the program has pledged $4.8 billion to fund 214 proposals among 121 countries through 2008; however, only $2.1 billion of these funds had been received from donors as of November 2003 (see Figure 2-1) (The Global Fund, 2003a). The proposals include specific plans for HIV/ AIDS, tuberculosis (TB), and malaria, as well as joint efforts (e.g., HIV/ AIDS–TB), and both country-specific and cross-border initiatives. Of these 214 proposals, 135 have moved forward in terms of having a specific work plan in place, and about $250 million had been dispersed as of January 2004 (Schwartlander et al., 2004) (see Box 2-2).
Global Fund monies are expected to provide enough ARVs to treat approximately 700,000 people over the course of the next several years. About 60 percent of Global Fund resources have gone or will go to sub-Saharan Africa, and about 60 percent toward HIV/AIDS (The Global Fund, 2003b). Of the amount dedicated to HIV/AIDS, a little less than half is dedicated to commodities, and about half of that amount to ARVs. The majority of project plans that have HIV/AIDS components include prevention elements, although the primary focus of most is treatment (i.e., ART).
After technical review and negotiations, the majority of funded HIV/ AIDS projects in African countries spend, on average, about US$400 per patient per year for ART (Schwartlander et al., 2004). Global Fund–sup-
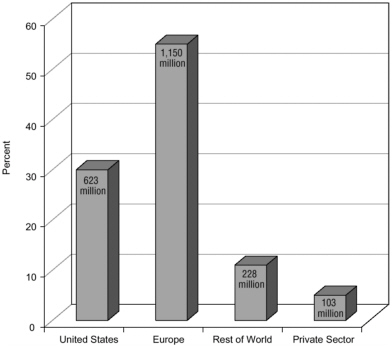
FIGURE 2-1 Contributions to the global fund to date.
SOURCE: The Global Fund, 2004c.
ported ARVs are a bit more expensive in Latin America, where income levels are slightly higher and resource constraints somewhat less pronounced, although negotiations are in progress to lower drug prices there as well. The drug prices are higher in Eastern Europe as well, where the annual cost of ART per patient can run as high as US$1,000. These figures include only commodities (e.g., drugs, very simple diagnostics) and not costs for health care workers and other basic infrastructure.
Many are concerned that the Global Fund monies have been slow to roll out and that, despite agreements being in place, some countries do not yet have the promised funds or ARVs. Total global funding budgeted for the fund in 2003 for HIV/AIDS was an estimated US$4.2 billion (including funding from all donor governments and UN agencies and disbursements from The World Bank, foundations, and major nongovernmental organizations [NGOs]). However, estimates from the Joint United Nations Program on HIV/AIDS (UNAIDS) indicate that actual spending in 2003 totaled only about US$3.6 billion (UNAIDS, 2003). It is believed that much of the discrepancy is due to the lag time between the receipt and disbursement of contributions to the Global Fund, as well as to differences between bud-
|
BOX 2-2 Created in 2002 by Kofi Annan, Secretary General of the United Nations, the Global Fund to Fight AIDS, Tuberculosis, and Malaria is an independent, nonprofit foundation. It is governed by an international board that is currently chaired by the U.S. Secretary of Health and Human Services. The foundation’s goal is to act quickly and effectively to mobilize resources for combating HIV/ AIDS, tuberculosis, and malaria. The foundation is governed by an international board that consists of representatives of donor and recipient governments, nongovernmental organizations, the private sector (including businesses and philanthropic organizations), and affected communities. In mobilizing resources, the Global Fund operates as a financial instrument to attract, manage, and disburse funding to fight HIV/AIDS, tuberculosis, and malaria worldwide through a competitive grant support mechanism. It seeks to fund projects that will form new or innovative alliances with governments, civil society, the private sector, and affected communities, which in turn will create local ownership of programs. The fund’s existence is based on strong public– private partnerships, results-based management, and a focus on local capacity building. Grant applications are evaluated by an independent technical review panel of disease-specific and cross-cutting health and development experts. In the evaluation process, funding priority is given to countries with the highest incidence of disease and the fewest financial resources and to programs that can coordinate and strengthen existing health systems. Additional weight is given to applications built on proven best practices that incorporate the principles of results-based management and strong mechanisms of accountability for both in-country and fund project managers. Funded projects cannot replace or reduce existing sources of funding. Since its inception, the Global Fund has attracted over US$5 billion in pledges from donor nations, foundations, corporations, and individuals; the total paid to the fund to date is just over US$2 billion. Since the review of three proposal rounds in April 2002 and January and October 2003, project grants total US$2 billion over 2 years to support 224 programs in 121 countries and 3 territories worldwide. With Global Fund resources, approved local programs will support an unprecedented scale-up of ART. Over 5 years, an estimated 700,000 people will receive ART, more than tripling current coverage in poor countries (including a 10-fold increase in Africa). The Global Fund will also reach 1 million children orphaned by AIDS; provide treatment for nearly 3 million people with infectious tuberculosis, tripling the coverage of treatment of multidrug-resistant tuberculosis; and finance 22 million combination drug treatments for resistant malaria. The Global Fund proves that with cooperation, goals can be achieved. SOURCES: The Global Fund, 2003a,b; 2004a,b; U.S. Department of State, 2004. |
geted amounts and actual spending by donor governments (Henry J. Kaiser Family Foundation, 2004a). This lag time points to the need to include funding mechanisms in all ART scale-up monitoring and evaluation programs so that any such problems can be identified and addressed. (See Chapter 5 for further discussion of monitoring and evaluation.)
If broad-based financing mechanisms for the procurement of ARVs are not in place (directly linked to some of the major donor sources), monies can often be slow to trickle in. Such financial uncertainty does not allow for the development or implementation of effective ART programming and places those who are relying on promised drugs and other essentials at risk for treatment failure. (See the discussion of fiscal sustainability later in this chapter.) Erratic provision of treatment caused by interruptions in the drug supply can lead to treatment failure regardless of how adherent patients may be or how experienced their health care providers are. Thus it is crucial that organizations providing global funding for HIV/AIDS act more expeditiously in getting the funds to countries so their programs can meet the needs of people on ART, as well as programmatic expectations and global expectations for scale-up.
The World Bank’s Multi-Country HIV/AIDS Program
In 2000, the World Bank began the US$500 million MAP to bolster existing national-level HIV/AIDS programs in sub-Saharan Africa. The MAP seeks to enhance prevention, care, and treatment programs, with particular emphasis on at-risk groups, including youth and women of childbearing age. Each aid package is multisectorial, providing money to both national and community-level programs and NGOs. In 2002, the Bank approved an additional US$500 million for MAPII. As of September 2003, US$848 million had been committed to African countries (see Table 2-1).
The following criteria must be met for MAP participation (The World Bank Group, 2001):
-
Satisfactory evidence of a strategic approach to HIV/AIDS, developed in a participatory manner
-
Existence of a high-level HIV/AIDS coordinating body, with broad representation of key stakeholders from all sectors, including people living with HIV/AIDS
-
Government commitment to quick implementation arrangements, including channeling grant funds for HIV/AIDS activities directly to communities, civil society, and the private sector
-
Agreement by the government to use multiple implementation agencies, especially NGOs/community based organizations
TABLE 2-1 World Bank HIV/AIDS Projects by Country
|
Country |
Commitment (US$millions) |
|
Benin |
23.0 |
|
Burkina Faso |
22.0 |
|
Burundi |
36.0 |
|
Cameroon |
50.0 |
|
Cape Verde |
9.0 |
|
Central Africa Republic |
17.0 |
|
Eritrea |
40.0 |
|
Ethiopia |
59.7 |
|
Gambia |
15.0 |
|
Ghana |
25.0 |
|
Guinea |
20.3 |
|
Kenya |
50.0 |
|
Madagascar |
20.0 |
|
Malawi |
35.0 |
|
Mauritania |
21.0 |
|
Mozambique |
55.0 |
|
Niger |
25.0 |
|
Nigeria |
90.3 |
U.S. Funding for Global HIV/AIDS Efforts
History of U.S. Funding
The United States has been funding international HIV/AIDS activities since 1986, when it made its first commitment of US$1.1 million. Funding levels increased slowly and steadily until 1999 ($219 million), at which point they increased substantially. The 1999 Leadership and Investment in Fighting an Epidemic (LIFE) Initiative under President Clinton and the 2001 creation of the Global Fund both significantly contributed to the increase in funding. By 2001, U.S. funding had more than tripled to $756 million, (a figure that included the first U.S. contribution to the Global Fund), and then rose again to $1.03 billion in 2002, $1.463 billion in 2003, and $2.3 billion in 2004 (Henry J. Kaiser Family Foundation, 2004b).
The money is channeled through both bilateral assistance and multilateral efforts. In 2003, for example, about 58 percent of the funding was allocated to bilateral programs, 24 percent to the Global Fund, and 18 percent to international HIV/AIDS research efforts at the National Institutes of Health (NIH) and the Centers for Disease Control and Prevention (CDC). Most of the bilateral assistance is administered by the U.S. Agency
for International Development (USAID). In 2004, USAID handled $550 million (i.e., 25 percent) of the total U.S. global HIV/AIDS funding. Other bilateral administrators have included CDC’s Global AIDS Program and the Departments of Commerce, Agriculture, Defense, and Labor. In 2005, the latter three departments will not be administering any U.S. funding for global HIV/AIDS; instead, the State Department will become the second-largest recipient and administrator of U.S. global HIV/AIDS funding (i.e., second to USAID).
Most of the U.S. multilateral assistance goes to the Global Fund (see above). UNAIDS, WHO, and The World Bank also receive U.S. multilateral support.
The President’s Emergency Plan for AIDS Relief
On May 27, 2003, President Bush signed Public Law 108-25, the United States Leadership against HIV/AIDS, Tuberculosis, and Malaria Act of 2003. Among other provisions, the act requires that the President establish a comprehensive, integrated 5-year strategy to combat global HIV/AIDS (Office of Legislative Policy and Analysis, 2004). Although the focus of past programs has been largely on prevention, the United States has begun broadening its response to provide care and treatment as well (Henry J. Kaiser Family Foundation, 2004b). Among other goals, the new funding is intended to provide ARVs to 2 million people (see Box 2-3).
In January 2004, Congress appropriated $2.4 billion to PEPFAR for fiscal year 2004, $488 million of which was new funding for the plan. On February 23, 2004, details of the $15 billion PEPFAR were released, along with the first $350 million (Office of the United States Global AIDS Coordinator, 2003), including $189 million in funding awards. The plan targets 15 countries: Botswana, Cote d’Ivoire, Ethiopia, Guyana, Haiti, Kenya, Mozambique, Namibia, Nigeria, Rwanda, South Africa, Tanzania, Uganda, Vietnam, and Zambia. The first round of funds is expected to provide treatment for 50,000 people.
The World Health Organization’s 3-by-5 Campaign: The Vision
The WHO 3-by-5 campaign, with the aim of providing ART to 3 million people by 2005, is a target, not a program. Established in 2002, 3-by-5 was based on an assessment of existing entry points in public health infrastructure (e.g., an estimated 500,000 persons needing therapy will be reached through already-functioning TB programs); an unprecedented global commitment to finally dealing with the growing enormity of a problem that had been, until then, underestimated; and accumulating evidence that the implementation of ART programs is feasible in resource-constrained
settings. As part of the campaign, WHO has established a set of simplified, standardized treatment guidelines (which are discussed in more detail in Chapter 4 and presented as Appendix C). As discussed in Chapter 4, several of the recommendations are understandably nonspecific, given the critical lack of knowledge and urgent need for research in some areas. It is important to note that the limitations of the guidelines do not reflect shortcomings in WHO’s 3-by-5 campaign, but rather the general state of knowledge about HIV/AIDS and the global inexperience with caring and treating for infected patients in resource-constrained settings. Box 2-4 provides more detail on the campaign.
The WHO campaign recognizes that almost all of the more than 40 million people now infected with HIV worldwide will eventually require access to therapy. Looking beyond 2005, WHO and its partners will be developing a new strategic approach to maintain the gains of 3-by-5 and to extend them, using sustainable financing and delivery mechanisms, so that ART becomes part of the primary health care package provided at every health center and clinic.
The William J. Clinton Presidential Foundation
The Clinton Foundation’s HIV/AIDS initiative has established effective strategies for the procurement of affordable, high-quality ARVs and diagnostics for several African and Caribbean countries (Mozambique, Rwanda, South Africa, Tanzania, the Bahamas, the Dominican Republic, and Haiti). In October 2003, the foundation announced an agreement with several generic suppliers, including suppliers of some of the most common first-line regimens, for a fixed-dose AZT, 3TC, and nevirapine combination at $239 per person per year and a fixed-dose D4T, 3TC, and nevirapine combination at $132 per person per year. Continued negotiations are also being directed toward a number of other drugs, including pediatric formulations. The procurement strategy of the foundation’s HIV/AIDS initiative is discussed in greater detail below in the section on fiscal sustainability.
Operational Plan for Comprehensive HIV and AIDS Care, Management, and Treatment for South Africa
Of the more than 5 million HIV-infected South Africans, 400,000 to 500,000 are eligible for ART based on the WHO guidelines, indicating one of the largest AIDS burdens in the world. Fully 12 percent of the general population is infected with HIV. Moreover, 21 percent of the population between the ages of 15 and 49 is infected. Although the introduction of nevirapine has decreased mother-to-child transmission over the past few years, 90,000 HIV-positive babies were at one point being born each year.
|
BOX 2-3 Overview of Goals
Focus
Target Areas
Management
|
Life expectancy in South Africa declined over the past decade to 50.7 years in 2002, largely because of HIV/AIDS.
Although South Africa is classified as a middle-income country, with a 2002 annual per capita gross domestic product (GDP) of approximately $2,408, huge disparities exist between the rich and poor, predicated mainly on race and stemming from a 46-year system of apartheid and a 300-year legacy of colonialism. About 10 percent of the population lives in abject poverty, and another 20 percent lives in poverty with incomes of less than US$2 a day. Most of the poverty exists in rural areas.
Since the country’s first democratic government came to power in 1994, national efforts have made remarkable headway through many socioeconomic, educational, and development interventions. Examples include providing clean water to 8.4 million new South Africans, providing access to electricity for 3.8 million households, building 1.46 million formal houses, increasing literacy, increasing the number of children in school, and increasing the number of people involved in an integrated nutrition program.
Also in the early 1990s, the South African government developed its first strategic plan for HIV/AIDS.
In terms of expenditures, the national response to the HIV epidemic was initially quite inadequate, with only US$4.4 million (2004 exchange rate) being spent in 1994. The budget gradually increased to reach some $50.7 million in 2002. Since then, expenditures have increased dramatically to approximately $444.6 million a year, and they are expected to reach $1.2 billion by 2008. This increase has been achieved during a period of modest economic growth (GDP growth was 0 percent from 1985 to 1994 and 2.8 percent from 1995 to 2002) and at a time when the government has been faced with a multitude of other health, educational, development, and socioeconomic challenges.
After acknowledging the need for an ART program in the public sector in 2002, the government established a joint health and treasury task team to investigate and determine the cost of such a program. The team delivered
|
BOX 2-4 On World AIDS Day 2003, WHO and UNAIDS released a detailed and concrete plan to reach the “3-by-5” target of providing ART to 3 million people living with AIDS in developing countries and those in transition by the end of 2005. The 3-by-5 campaign complements the groundbreaking commitment to addressing the global AIDS pandemic made by the U.S. PEPFAR initiative (see Box 2-3), the pathfinding work of NGOs, the efforts of pharmaceutical companies to reduce the prices of ARVs, the contributions of international foundations, the initiatives and hard work of many national and international agencies, and, critically, the courageous contributions of nations increasing access to AIDS treatment for their citizens. After 20 years of fighting the epidemic, it is now clear that a comprehensive approach to HIV/AIDS must include prevention, treatment, and care. WHO’s 3-by-5 campaign supports this comprehensive goal by using best-practice theories and technical reports to guide those involved in combating the HIV/AIDS pandemic. The technical briefs and reports provided focus on general HIV/AIDS management issues such as ART, patient follow-up, resistance surveillance, emergency scale-up of support personnel and community involvement, public health approaches to prevention, and guidelines for surveillance and monitoring and evaluation. NGOs, local communities, and governmental organizations are encouraged to use WHO’s comprehensive strategy and what is referred to as “the five pillars” to develop and promote programs linking treatment, prevention, care, and full social support for those affected by HIV/AIDS. The five pillars are as follows:
Ultimately, WHO’s success will rest on the ability of NGOs, affected communities, and public health officials to implement best practices and combat the disease effectively, economically, and in a timely fashion. With focused strategies and guiding principles; however, WHO has already taken a vital step toward the ultimate goal of providing universal access to AIDS treatment. SOURCES: Kim, 2004; WHO, 2003a. |
its report in August 2003, after which the Ministry of Health was requested to develop a plan for ART roll-out by September 2003. A task team was convened and, with the assistance of up to 40 technical consultants from the Clinton Foundation, an operational plan was written; this plan was endorsed by the Cabinet on November 19, 2003, without reservation. On January 23, 2004, the first patient received treatment.
The overarching goals of the plan are to provide comprehensive care and treatment for people living with HIV/AIDS and to facilitate the strengthening of the national health system in South Africa. It is important to note that, while the provision and effective management of ARVs will be an integral part of this effort, so, too, will preventative (particularly with regard to sexual behavior), nutritional, opportunistic infection, and non– health sector interventions.
More specifically, the goal is to provide ART to all South Africans in need of such treatment within 5 years. The care and treatment protocols for ARVs are rigorous and standardized, and the drugs will be delivered through provincial AIDS teams working in 53 different service points (one in each of South Africa’s 53 health districts) so as to maximize the equitable distribution of comprehensive care and treatment. The first-line ARV regimen is d4T/3TC/nevirapine (pregnant women) or efavirenz (all others); the second-line regimen is AZT/ddI/Kaletra (lopinavir and ritonavir).
As noted, ARVs are not the only component of the treatment plan. Ensuring access to prophylactic care and treatment for opportunistic infections is integral, as is providing nutrition-related interventions to those in need. The latter interventions are believed to be vital to slowing the progression of disease in HIV-infected persons. In recognition that some 80 percent of South Africans have recourse to traditional healers and take traditional medicines, the plan includes efforts to destigmatize traditional medicine and ensure that health professionals are aware of possible interactions and other issues surrounding the use of such medicines.
During the development of the plan, as with ART scale-up in general, human resource deficiencies were identified as the principal constraint (see Chapter 5 for further discussion), as there is approximately only one doctor for every 2,000 South Africans overall, with a remarkably skewed distribution between urban and rural areas. Thus the current plan includes forecasts of the growing numbers of trained health personnel that will be needed to implement ART effectively (see Table 2-2).
Furthermore, in February 2003, South Africa registered only one public-sector general physician per 4,829, one public-sector specialist physician per 10,403, and one public-sector professional nurse per 910 South Africans. Of most critical concern is the lack of a sufficient number of pharmacists, as there is only one public-sector pharmacist per 29,579 persons (South African National Treasury, 2003). Detailed and rigorous training manuals and processes for health professionals who will deliver HIV/AIDS interventions are being developed to extend the available expertise as much as possible. It is expected that the initiation of therapy will be by medical doctors, with nurses delivering much of the follow-up care.
A total estimated $696.5 million will be spent annually by 2007–2008, broken down as follows:
TABLE 2-2 Anticipated Personnel Needs for HIV/AIDS Care, Management, and Treatment in South Africa, by Category
|
Category |
Phase I |
Phase II |
Phase III |
|
Medical Officers |
110 |
271 |
628 |
|
Professional Nurses |
330 |
813 |
1,883 |
|
Enrolled Nurses |
220 |
542 |
1,255 |
|
Assistant Nurses |
220 |
542 |
1,255 |
|
Pharmacists |
110 |
271 |
314 |
|
Pharmacist Assistants |
110 |
271 |
314 |
|
Dieticians |
110 |
136 |
314 |
|
Counsellors/Community Health Workers |
1,100 |
2,710 |
6,275 |
|
Administrative Clerks |
220 |
542 |
1,255 |
|
SOURCE: Paper presented by Dr. Anthony Mbewu on South Africa plan. |
|||
-
$244.5 million to be spent on ARVs, if the prices negotiated by the Clinton Foundation can be implemented
-
$148 million for recruiting of new health professionals
-
$118.5 million for laboratory tests
-
$96.3 million for additional nutritional supplements and support
Based on this budget and according to the plan, 1.1 million individuals will be on ART by 2008 (i.e., all individuals in need of therapy at that time), at a cost of about R600 per year per patient. An important element of the plan is about R50 million per year for research. These funds will be channeled through the Medical Research Council and overseen by a research subcommittee convened by the Department of Health.
COORDINATING THE GLOBAL RESPONSE
The ambitious ART roll-out goals of the PEPFAR initiative, the South African government, WHO, and a considerable number of countries and organizations worldwide represent a desperately needed global response to a devastating human tragedy (see Table 2-3). No single group or nation has all the fiscal, human, or intellectual resources to address these challenges, nor will a single approach or regimen be suitable for all settings. An organized culture of learning by doing will be central to identifying and refining best practices for particular settings, and the sharing of knowledge, resources, and efforts will be critical to the rapid, effective, safe delivery of ARVs to the millions of people in immediate need of life-sustaining treatment.
Coordinating global HIV/AIDS efforts in a way that maximally ben-
TABLE 2-3 Bilateral HIV/AIDS Support from Selected Countries, 2002 (in US$ millions)
|
Country |
Projected 2002 Disbursements |
% of GDPa |
|
G8 Members |
||
|
United States |
$413 |
0.00410 |
|
United Kingdom |
300 |
0.02106 |
|
Japan |
85 |
0.00205 |
|
Germany |
55 |
0.00297 |
|
Canada |
39 |
0.00561 |
|
France |
25 |
0.00190 |
|
Italy (included in “other” below) |
||
|
Subtotal G8 Members |
$917 |
|
|
Other Countries |
||
|
Netherlands |
$55 |
0.01447 |
|
Norway |
35 |
0.02108 |
|
European Commission (EC) |
25 |
|
|
Other (including Italyb) |
190 |
|
|
Subtotal Other Countries |
$305 |
|
|
Total |
$1,222 |
|
|
aGDP percentages calculated from 2004 World in Figures, The Economist. bExact figures for Italy are not available. According to UNAIDS, estimated bilateral spending by Italy is <$25 million. NOTE: Amounts shown do not include funding for international HIV/AIDS research, and represent fiscal year (FY) 2002 budgeted/appropriated dollar amounts. Some countries are still finalizing FY 2002 figures. SOURCE: Henry J. Kaiser Family Foundation, 2004a,b. |
||
efits the affected countries and minimizes the diversion of resources from other critically important national health and social programs presents a great challenge. While recognizing the political complexities of a globally coordinated response of this magnitude, externally funded programs should make every effort to contribute to overall success rather than claiming organizational credit. It is imperative to maximize synergy and minimize competition among different initiatives and to ensure that the long-term potential to achieve sustainable results is not compromised by short-sighted ambitions and quick fixes. Equally imperative is the need to have a clear understanding of the local needs of affected communities and how they may differ from well-intentioned but sometimes misguided foreign perceptions.
A lack of coordination among donors and partners who enter countries with their various objectives and intentions can create confusion at the
country level and make it difficult to monitor and assess the progress and impact of the respective efforts. Monitoring and evaluation (discussed in more detail in Chapter 5) is vital to knowing whether programs are succeeding and if not, to identifying problems and areas in need of improvement. At a recent WHO/UNAIDS workshop on generating strategic information for use in ART scale-up, the need to standardize monitoring and evaluation processes was identified as an urgent priority (WHO, 2004a). Yet monitoring and evaluation can become highly complicated very quickly. Although defining and monitoring input variables (i.e., what goes into a program) may be relatively easy, objectively monitoring and evaluating what becomes of those input variables (i.e., how the program proceeds and what the outcomes are) can be much more difficult. Without internationally harmonized monitoring and evaluation procedures, countries may be at a loss as to how to proceed. Duplication of effort and generation of redundant data may create unnecessary additional work for those responsible for collecting and managing monitoring and evaluation data.
Role of National Governments
The role of the governments of affected countries will be crucial to a coordinated response, since NGOs and universities, which serve as valuable sources and concentrations of expertise, do not have the mandate to scale up. The sooner the national government assumes leadership and engages in a specific scale-up effort, the more likely it is that scale-up will proceed rapidly and efficiently. Moreover, as the national government defines the standard of care for its country, so, too, does it define the continuum of medical care and support services that its HIV/AIDS population should have. Thus it is important that country plans be in place to inform allocation decisions and that procurement processes be integrally linked to country plans. Accordingly, one of the explicit policies of the Global Fund is that recipient countries must develop their own plans, which means they must have the necessary systems in place for doing so, including monitoring systems to gather information demonstrating that the funds provided are being used appropriately (Schwartlander et al., 2004).
The need to respect country processes raises important questions about the readiness of countries to implement large-scale ART programs and effectively absorb the great influx of resources being made available. There must be visible and meaningful commitment at the national level, and there should be established country mechanisms for coordinating multiple donor efforts, particularly with regard to ARV procurement (to ensure a continuous, reliable drug supply). Budgets must be designed appropriately; priorities should be realistic, based on actual resource allocations, reflective of
cost-effectiveness and other value-driven considerations, and set by the countries rather than the donors; and the balance among various interventions should be founded in evidence-based knowledge, not arbitrary decisions. As important as these factors are to the short-term success and long-term sustainability of national HIV/AIDS strategic plans and global ART scale-up in general, all too often they do not receive sufficient consideration (Alban, 2002).
Assessment Tools
Quantitative assessment tools and costing analyses can be used to identify where the most absorptive capacity exists, where (e.g., in which regions, facilities) and how (e.g., which regimens) scale-up is likely to be most successful and cost-effective, and what steps need to be taken to scale up capacity in particular situations. A recent report on the cost and resource requirements associated with public-sector ART scale-up in Zambia illustrates the utility of such analyses for identifying key issues and exploring different approaches (e.g., alternative monitoring protocols and drug regimens) (Kombe and Smith, 2003).
Assessment tools may work best when the focus is on a specific geographic area. Otherwise, as the Clinton Foundation has learned from its experience conducting assessments of program readiness in several countries, the country’s needs overwhelm the ability to define the standard or continuum of care and services that are impacting or available to a specific population (Goosby, 2004). Focusing on a specific population in a particular geographic area allows for a more quantitative assessment of service points (e.g., clinics, drug storage facilities, and transportation to such facilities) that intersect with a given population, as well as the steps necessary to create such links where they do not exist. It is important to emphasize that the assessment is aimed not at creating a parallel system with new clinics, laboratories, and the like, but on evaluating the current system, identifying gaps in service, and determining steps that can be taken to fill those gaps. Box 2-5 describes one example of an assessment tool.
Of note, based on its experience assessing program readiness, the Clinton Foundation has repeatedly identified human resource capacity as the major limiting factor. Not only are most sites experiencing a serious shortage of trained, qualified health care workers, but the amount of time it takes to train such workers makes it impossible to solve the problem quickly by adding new workers to a system that already exists (as can be done with other, quickly available resources, such as the ARVs themselves). Human resource capacity is discussed in detail in Chapter 5.
|
BOX 2-5 In May 2003, DELIVER, a USAID-funded worldwide technical support contract implemented by John Snow, Inc., produced a 20-page guide for evaluating site capacity and readiness to initiate ART according to six domains: leadership and program model, services and clinical care, management and evaluation, staffing and experience, laboratory capacity, and drug management and procurement. An overall score determines which of five readiness stages characterizes a program and what steps can be taken to advance a program to a higher stage. The goal of the tool is to assess readiness based not on site type, size, or location, but rather on capacity, vision, and activities and services needed for the rational introduction and expansion of ART. The tool can also be used for site self-assessment and for the identification of areas in need of technical assistance and ways in which site programs can serve as resources for other programs. SOURCE: Hirschhorn et al., 2002. |
Importance of Engaging the Private Sector and Nongovernmental Organizations
Although the tendency when discussing and planning ART scale-up is to focus on the public sector, nongovernmental and private-sector programs may provide innovative, creative mechanisms for getting programs—including ART programs—off the ground more rapidly and operating more efficiently. While quality control is imperative, creative approaches that look beyond traditional entry points should be sought. Nontraditional entry points, such as mission hospitals, are increasingly being used to introduce and scale up ART programs. Consideration should also be given to other delivery mechanisms, such as franchising models that have shown substantial promise in reproductive health but have been explored less frequently for their potential to provide HIV/AIDS prevention and treatment (Montague, 2002; Smith, 2002; LaVake, 2003). Indeed, the number of nonpublic Global Fund recipients is growing, and only about half of the Global Fund resources are dedicated to public-sector recipients (Schwartlander, 2004).
While private programs can provide important innovations and capacity, it will be important as well for them to coordinate with public programs and to provide assurances and evaluation data related to the quality and consistency of the care they provide. Mechanisms will be needed to address the lack of sufficient training and access to updated regimens and protocol changes among private-sector practitioners, as well as the irregular and frequent movement of patients from provider to provider and the higher cost of private-sector ARVs (Brugha, 2003).
It must be recognized that the selection of particular locations and regions for the rapid implementation of scale-up, whether based on quantitative assessments or not, has important ethical implications. In particular, it is essential to ensure that the integration of ART programs into existing efforts does not further widen the gap between those who have access to health care (e.g., those living in urban areas and having access to hospitals and clinics) and those who do not (e.g., those living in remote, rural areas). To this end, efforts must be directed simultaneously at building the necessary infrastructure and capacity in areas where they do not exist. This and other human resource and infrastructure issues surrounding the management of ART scale-up are discussed in greater detail in Chapter 5 and in Appendix E.
Finally, as important as assessing readiness may be to introducing ART programs in the manner most likely to achieve maximal ARV coverage, it is equally important that scale-up efforts be able to proceed without having every detail in place.
Recommendation 2-1. Antiretroviral therapy scale-up in resource-constrained settings should proceed immediately through coordinated, aggressive action by national governments, donors, international agencies, and nongovernmental organizations. Donors must attempt to maximize the distribution of scarce resources—human, financial, and technical—for people in need within and among all resource-constrained countries and areas. To this end, multiple HIV/AIDS prevention and treatment initiatives need to be coordinated and integrated through national leadership and entitities that best meet the needs of their populations and of all individuals.
FISCAL SUSTAINABILITY
The promised funds and efforts of the U.S. government, WHO, the Global Fund, UNAIDS, and others represent a historic change in the global attitude and political will toward alleviating the burden of HIV/AIDS. In the absence of a cure, however, the provision of ART must be for life, and the funding for widespread ART must be sustainable in the long run and for an indefinite period of time. Moreover, all of the 40 million currently infected people will eventually require treatment, thus creating a very large, steady demand for ARVs for decades to come. Yet the global HIV/AIDS funding available today does not even meet the needs of the 6 million people currently eligible for ARVs according to WHO’s clinical criteria (Royal Danish Ministry for Foreign Affairs, 2000).
Based on 2001 calculations, the estimated annual cost of HIV/AIDS prevention and care for all people living with HIV/AIDS in 135 low- and middle-income countries is US$9.2 billion by 2005 (Schwartlander et al.,
2001). This figure includes $4.8 billion for prevention and $4.4 billion for care and support (i.e., ART, palliative care, prophylaxis and treatment for opportunistic infections, and support for orphans). More recently, UNAIDS has estimated the total need for HIV/AIDS funding to be $10.7 billion for 2005 and $14.9 billion by 2007 (Henry J. Kaiser Family Foundation, 2004a). These estimates cover prevention, care, and treatment delivery, but not research or infrastructure costs. By contrast, the total estimated funding for global HIV/AIDS in 2003 was US$4.2 billion, including $852 million in bilateral U.S. assistance, $1.163 billion in bilateral assistance from other countries (e.g., Canada, the European Commission, Germany, Japan, the Netherlands, the United Kingdom), $547 million from the Global Fund, $350 million from UN agencies, $120 million from The World Bank, $200 million from NGOs and foundations, and $1 billion from affected country governments (Henry J. Kaiser Family Foundation, 2004a). As already mentioned, the actual spending for 2003 was only $3.6 billion.
Achieving 3-by-5 and ultimately providing ARVs to all people in need will require substantially strengthening and upgrading the existing health system infrastructure. Otherwise, expanded ARV coverage may lead to increasing inefficiencies in resource allocation and higher costs per person for delivering interventions. A recent economic analysis of 83 countries showed that achieving high ARV coverage by 2015 will require additional annual spending of an estimated $US6.8 to 9.2 billion, including 25 percent for infrastructure (additional annual costs for prevention and care total more than an estimated $8.5 and $5.5 billion, respectively) (Kumaranayake et al., 2002). This same study showed that without infrastructure investments, ART will be able to expand by only about 10 percent.
As an example of how limited funds can impact affected countries, medications, counseling, and testing cost an estimated US$600 per patient per year in Botswana. Over 5 years, with 20,000 new patients added each year, the cumulative total cost will be $180 million. The Gates Foundation has committed $50 million over 5 years, with Merck matching that amount in ARVs. Concerns have been raised, however, about the sustainability of the program after the funding providing by the Gates Foundation and Merck ends (Rollnick, 2002; Attawell and Mundy, 2003).
As another example, in a recent study on the cost of comprehensive HIV/AIDS treatment in Mexico, ARVs account for an estimated 72.7 to 78.3 percent of the total annual cost of treatment per patient on ART (Bautista et al., 2003). It is interesting to note that, in contrast to what has been observed in Brazil (Ministry of Health [Brazil], 2003), the use of ARVs in Mexico is not cost-saving. Even though ART leads to a decline in hospital days, the decline is nowhere near large enough to offset the increase in ARV-related costs. Even a small reduction in the price of ARVs would have a measurable impact on the overall cost of therapy and make it easier for
the government to provide ARVs to the country’s estimated 64,000 people with symptomatic AIDS and another 116,000 to 177,000 infected with HIV. As is the case in so many other countries, access to HIV/AIDS treatment, including ARVs, varies considerably throughout Mexico; to meet the needs of the poor, the uninsured, and other HIV-infected people who cannot afford currently priced ARVs, the Minister of Health has committed to providing ARVs to all who need them by 2006.
In Zambia, a 2003 report on the per-patient costs and human resource requirements for scaling up ART in that country concluded that, at a perpatient annual cost of US$480, the current ART budget allows for provision of therapy to only about 10,000 people (Kombe and Smith, 2003). Given the uncertain future of the Global Fund and the unclear status of the $4.9 million already awarded to Zambia for HIV/AIDS interventions, however, the fiscal sustainability of treating even these initial 10,000 people, let alone the thousands of other infected people who need or will eventually need treatment, is uncertain. There are about 850,000 HIV-infected adults living in Zambia, including 100,000 who are already clinically eligible for ART. An estimated 85,000 to 110,000 new AIDS cases will likely continue to appear each year.
In Nigeria, about 10,000 patients receive therapy through the government program (at a heavily subsidized cost of US$8 per month) and 5,000 through state or private institutions, including NGOs (at a cost of about $90 to $100 per month). But even $8 is too much for many people, particularly in rural areas (Idoko, 2004). The added cost of basic laboratory monitoring—including CD4 counts, which are recommended at least every 6 months, as well as additional toxicity assays depending on the drug regimen and clinical symptoms—is prohibitively high for many patients. Since the Global Fund insists that drugs be free, it was not clear as of January 2004 how Nigeria would deal with the fact that its public program is based on heavily subsidized—not free—drugs. The program plan currently targets only 15,000 patients, yet there are about 4 to 5 million HIVinfected Nigerians (Idoko, 2004).
Procurement Strategy of the Clinton Foundation HIV/AIDS Initiative1
The Clinton Foundation’s established procurement agreements are with suppliers of both APIs (active pharmaceutical ingredients, which represent 60–75 percent of the cost of producing ARVs) and formulations, as well as with suppliers of diagnostic tests (including both CD4 and viral load tests). For example, under an agreement with Becton, Dickinson, and Company,
which was announced in January 2004, CD4 tests will be made available at US$8 per test. This figure includes the cost of reagents, an operational lease for the necessary instruments (including service, training, and controls), and consumables. This represents a substantial cost savings for what would otherwise be prohibitively expensive for many programs, as the retail price of the type of high-throughput instrument required for monitoring runs upwards of $100,000 to $130,000 per instrument, plus annual service costs are usually about 10 to 20 percent of the price of the instrument. The Clinton Foundation has also negotiated price reductions for CD4 assays with Beckman Coulter and for viral load assays with Roche Diagnostics, Bayer Health Care, and bioMerieux. The latter reductions make the price up to 80 percent lower as compared with the currently available lowest-price viral tests.
To illustrate the tremendous cost savings made possible by this type of combined agreement, the expected per-person cost of ART and CD4 and viral load monitoring in South Africa has been reduced from US$800 to less than $250, representing a savings of $500 million in a single year. In the Bahamas, procurement negotiations reduced the cost of generic ARVs per patient per year from approximately $3,800 to $500, a cost savings that allowed the Ministry of Health to increase the number of patients on ART from 350 to 1,200 without additional funding. Following the conclusion of another recent agreement, this annual cost is expected to be reduced even further, to about $300.
The increased buying power made possible by this type of procurement strategy not only is immediately beneficial to countries in need and to those living with HIV/AIDS, but also may lead to near- and long-term ARV price reductions. (For details on this strategy, see Box 5-3.) Given that there are only 300,000 people worldwide on ART, the current ARV marketplace is highly fragmented, with many small orders. For the ARV supplier, this situation translates into excess capacity, the production of small batches, little leverage with API suppliers, and relatively high prices (given that the APIs account for about 60–75 percent of the cost of drug production). Larger ARV orders should improve plant utilization at many levels and increase leverage with API suppliers, both of which should lead in turn to price reductions. A predictable future demand would also increase incentives to invest in research, development, and process improvement.
Although procuring the lowest possible price for ARVs is critically important to ensuring that as many people as possible receive the drugs, equally important is the need to ensure that all drugs being distributed are safe and of high quality. The Clinton Foundation uses WHO’s prequalifying Procurement Quality and Sourcing Project (WHO, 2004b) criteria as accepted by the South African Medicines Control Council (Medicines Control Council, 2004) and its own technical assistance to ensure that drug distri-
bution systems are secure and that inventory is properly maintained. Médicins sans Frontières, another organization that has made remarkable headway in procuring affordable ARVs for use in its pilot clinics in South Africa and elsewhere, has reportedly sought and received authorization from the South African Medicines Control Council for the use of Brazilian generic ARVs (Attawell and Mundy, 2003). Issues related to drug safety and quality are discussed in detail in Chapter 5.
Need for Continuous Funding
Not only will the sustainability and success of ART scale-up depend on long-term funding, but it is vitally important that such funding from external donors be continuous while upstream issues such as debt and trade rules are being addressed in ways that could reduce the long-term dependence of developing countries on this philanthropy. In some countries, uncertainties in funding are already putting ART programs at risk of failure by interrupting the procurement and timely delivery of drugs. In Nigeria, for example, where only 5,000 of more than 1 million symptomatic HIV-infected people are currently receiving ART through a national program initiated in 2002, promised Global Fund monies have been slow to arrive: although about $12 million is expected, only $1 to 1.2 million had arrived as of the end of January 2004 (Idoko, 2004). The result has been serious procurement problems and widespread treatment interruptions.
Self-Sustainability
While philanthropy is unquestionably an essential component of ART scale-up, particularly during the emergency phase of the next 20 years or so, the long-term sustainability of these efforts will ultimately depend on addressing the complex underlying socioeconomic and political factors that drive the HIV/AIDS pandemic, as they do other infectious disease epidemics, in resource-constrained settings. Infectious diseases have and will continue to persist, emerge, and be influenced by the backdrop of rapid and profound demographic, economic, social, and ecological change (Benatar, 2002). Addressing these underlying factors will require imaginative and creative new approaches that are beyond the scope of this report.
Recommendation 2-2. Donors should commit to continuous funding of antiretroviral therapy scale-up for decades to ensure the sustainability necessary to avert the medical hazard of interruptions in the continuity of treatment. Delays in donor funding after treatment programs are initiated will jeopardize the long-term durability of treatment regimens. Because it is estimated that 40 million people are currently infected
with HIV, and 5 million new infections occur each year, donors should plan now for increasing support in the future. To this end, innovative mechanisms should continue to be pursued by national governments and donors in partnership with industry to ensure the continuous procurement of quality drugs, diagnostics, and other commodities at the lowest possible cost. At the same time, national governments in countries severely affected by HIV/AIDS must begin to invest in and develop priorities for prevention and treatment programs to ensure those programs’ long-term sustainability and effectiveness.
ETHICAL ISSUES
As ART programs are scaled up throughout the developing world, many difficult decisions will be necessary, and in many cases, both local and institutional/international ethical issues will be faced. The public health ethical dimension of ART scale-up is just beginning to enter the arena of bioethical debate, and there are as yet few answers to the multitude of ethical questions raised by ART scale-up.
Local Issues
One of the most pressing questions is how to determine priorities for who should receive treatment. This question is crucial given that WHO’s ambitious 3-by-5 plan still targets only half of the 6 million people worldwide currently in need of life-sustaining treatment, and given that millions more infected but currently immunocompetent persons will need treatment in the future. There is no easy answer, no universally obvious single best or “right” ethical framework or theory of justice (e.g., egalitarian, utilitarian, libertarian) to apply. Even if there were agreement on a particular theory of justice, there would probably still be logistical and other practical challenges to address in translating that theory into health care rationing. Nevertheless, the question of how to select those to treat should not paralyze initiatives to provide life-sustaining ART.
Different organizations and countries are currently basing the selection of individuals for treatment on variable criteria, many of which are ethically defensible from one viewpoint or another. Although WHO and this report recommend using medical criteria, even this apparently objective measure involves certain judgments about providing the greatest good for the greatest number of individuals. An example would be recommending less-expensive regimens (in terms of both initial cost and the level of expensive laboratory monitoring) despite concern that some of these regimens appear to be considerably more susceptible to resistance in poor adherence settings, thus increasing the risk of long-term failure of scale-up. Box 2-6
|
BOX 2-6
SOURCE: Wasunna, 2004. |
presents some of the treatment selection criteria proposed for ART scale-up programs.
One proposed criterion is that patients be selected on the basis of provider predictions regarding who is most likely to adhere to lifetime treatment. As mentioned in Chapter 4, however, studies have shown that providers are rarely able to make such predictions accurately, and that all too often the process becomes discriminatory against poor, disadvantaged people. On the other hand, one study showed that, although screening for the selective treatment of patients more likely to adhere to their regimens may result in lower levels of resistance, it is also likely to lead to a higher incidence of drug-sensitive HIV and AIDS than would a policy of treating all HIV-infected individuals (Tchetgen et al., 2001).
In addition to patient selection, the reality that there is a limited supply of ARVs, as well as other resources needed to deliver them, raises important questions about where to begin ART scale-up. It may be necessary to start with relatively small programs in places where the capacity exists, and while these programs are running, to steadily increase the supporting infra-
structure needed for larger programs. Alternatively, starting small may create greater ethical challenges by resulting in even more injustice in societies already suffering enormous disparities in health and wealth, given that ART programs would be implemented where the capacity already exists and people already have access. Meanwhile, those without access to health care would need to wait until, with donor aid, the country had built enough infrastructure to treat them. Yet delaying the provision of treatment to those who can access it now because doing so would widen existing health disparities is not necessarily any more ethical. Is it not better to save any life than none?
That there are no easy resolutions for these and similar ethical issues can lead to endless philosophical controversy and raises important questions about what, if anything, global donors can do with respect to recommending criteria or specific decision-making strategies. One proposed approach to priority setting is described in Box 2-7.
Some experts believe that, because of the enormous variation in ethical perspectives among different cultures and communities, the decision-making process on such matters should remain entirely the responsibility of
|
BOX 2-7 In the absence of a satisfactory theory of justice that can be applied in every-day practice, American scholars Daniels and Sabin have proposed a process for priority setting that they call “accountability for reasonableness.” According to this framework, a fair priority-setting process meets four conditions. First, decisions must be made on the basis of the best available data by people who are fair minded and willing to make compromise good-faith decisions that are contextually relevant. Second, the rationale for how such decisions were reached must be made public. Third, allowance must be made for appeals so that decisions can be reconsidered in the light of new evidence or arguments. Fourth, there must be an enforcement process that facilitates the implementation of the previous three conditions. This process was developed in the context of the U.S. privately funded health care system, but it has also been found to be workable and acceptable within the Canadian publicly funded system. In the latter context, researchers identified the importance of seeking multiple perspectives on the problem under review, ensuring transparency and honesty in the decision-making process, identifying potential conflicts of interest, and achieving consensus. A recent detailed qualitative study of how the framework of accountability for reasonableness can be applied to access to intensive care units for neurosurgical patients provides evidence that such a process improves the fairness of priority setting. SOURCES: Daniels and Sabin, 1997; Martin et al., 2002, 2003. |
individual countries. Others believe a minimum set of criteria for patient selection in treatment programs, based on international human rights, should be established to help countries make these very difficult decisions, and that countries should have a structured process in place for implementing context-specific decisions about the distribution and administration of treatment.
The question then arises, however, of how such a national ethical decision-making process should be created. What can countries do to ensure that the process is fair? To avoid a first-come, first-served system open to corruption and abuse, countries need some sort of legitimate process, even an imperfect one, so they can at least begin ART rollout as they simultaneously begin working toward a more fair and just system. It has been suggested that each country create its own independent, properly constituted, legally recognized, transparent decision-making body and that the decisions made by this body be subject to monitoring and evaluation just as any component of ART scale-up should be. However, the difficulties of establishing an independent ethical review process within each country cannot be underestimated. Many resource-limited countries may not have the capacity to develop ethical guidelines. Nor can the pragmatic challenges of implementing ethically sound theory and policy be overestimated. Scientific progress takes place slowly and requires much human energy and skill, as well as abundant resources. Moral progress will likewise be slow, but it can be can be made if time, energy, and resources are devoted to this end. The decision-making process described above suggests the way forward. As such processes are gradually implemented, further lessons will be learned.
What is to be done, however, about dictator-ruled countries without the political will to establish such a process? How should such countries be treated by the international community? What are the ethical issues surrounding the provision of funds and resources to countries without ethical decision-making processes in place?
As with other aspects of ART scale-up, waiting until a perfect ethical decision-making process has been designed and implemented creates its own ethical challenge, as thousands of people continue to die each day from AIDS-related causes. As critically important as the ethical components of ART scale-up may be, providing treatment in the absence of detailed guidelines has itself become an ethical imperative.
Institutional and International Issues
Although AIDS is an epidemic of unprecedented proportions, people living in resource-constrained settings also suffer from a wide range of other life-threatening ailments, most notably TB and malaria. Not only do many of these people not have access to the medical and health resources
needed to treat, cure, or prevent these ailments, but they often do not have access to some of the most basic necessities of survival, such as safe drinking water. This situation is perhaps the greatest international ethical challenge (Benatar, 1998).
It is imperative that ART programs be strategically situated within existing health care systems so that funding for HIV/AIDS can contribute to building a sustainable health care infrastructure that can benefit all people in need, whether they suffer from HIV/AIDS, TB, malaria, or some other health complication. Otherwise, practical and public health considerations aside, it would be necessary to justify using ART to save the lives of HIV/ AIDS patients and not the lives of those in immediate need of other life-sustaining drugs to treat malaria and other life-threatening diseases. ART scale-up must be implemented in such a way that instead of exacerbating existing inequities, it helps rectify some of those inequities by instituting programs that help meet the life-sustaining demands of all people in need.
Clearly, the ethical challenges and implications of ART scale-up are extraordinarily complex—more so, some would argue, than the medical and logistical challenges outlined in detail throughout this report. Even if WHO’s 3-by-5 campaign and other efforts to accelerate the introduction of ARVs achieve the highest levels of success, insufficient drug supplies and the reality that not all infected individuals in need can receive treatment will necessitate making decisions about whom to treat. Yet this is by no means the only ethical problem that will arise. Indeed, we are living in the midst of an ethical dilemma even now as we watch patients die and children become orphaned while we struggle with valid technical questions of how to balance the risks and benefits associated with the new opportunities outlined earlier in this chapter. These and other ethical issues are described further in Appendix D.
The goal of ART scale-up should be to prolong survival while also reducing illness and the numbers of newly infected people. Therefore, success should be measured by survival, and drug regimens should be selected on the basis of providing therapy to as many people as possible. This is the case despite some concern that the use of more-expensive regimens than those currently being recommended by WHO might better serve to reduce the eventual emergence of resistance. The threat of resistance and the implications of its emergence are as yet unclear. Since the use of more-expensive regimens would reduce the number of people treated and since the goal of ART scale-up should be to reduce mortality now, the use of simpler, less-expensive regimens is recommended. As noted earlier, there are many valid ethical and cultural perspectives from which to judge whether a particular HIV/AIDS intervention or treatment program is ethical. The committee takes the perspective that the use of simple, standardized regimens that maximize not only the number of people treated but also the likelihood that
ART programs will be introduced into even the most remote, resourceconstrained areas currently provides the best balance between the ethical complications of deciding who should receive treatment and the ethical imperative to act now.
REFERENCES
Alban A. 2002. Priorities of AIDS Interventions in Africa: Principles and Practice in Five Countries. Copenhagen, Denmark: EASE International.
American Enterprise Institute. 2004. Remarks given by Ambassador Randall L. Tobias, U.S. Global HIV/AIDS Coordinator at the Department of State, in Washington, DC. [Online]. Available: http://www.state.gov/s/gac/rl/rm/2004/29181.htm [accessed August 24, 2004].
Attawell K, Mundy J. 2003. WHO, UK Department for International Development. Provision of Antiretroviral Therapy in Resource-Limited Settings: a Review of the Literature up to August 2003. [Online]. Available: http://www.who.int/3by5/publications/documents/en/ARTpaper_DFID_WHO.pdf [accessed August 24, 2004].
Bautista SA, Dmytraczenko T, Kombe G, Bertozzi SM. 2003 (June). Costing of HIV/AIDS Treatment in Mexico, Technical Report No. 020. The Partners for Health Reformplus Project, Abt Associates Inc., Bethesda, MD.
Benatar SR. 1998. Global disparities in health and human rights. American Journal of Public Health 88:295–300.
Benatar SR. 2002. The HIV/AIDS pandemic: A sign of instability in a complex global system. Journal of Medicine and Philosophy 27:163–177.
Brugha R. 2003. Antiretroviral treatment in developing countries: The peril of neglecting private providers. British Medical Journal 326:1382–1384.
Daniels N, Sabin J. 1997. Limits to health care: Fair procedures, democratic deliberation and the legitimacy problem for insurers. Philosophy & Public Affairs 26(4):303–350.
Goosby E. 2004 (January 28). Assessing the Readiness for ARV Program Implementation. Presentation at the Institute of Medicine Workshop on Antiretroviral Scale-up in Resource Constrained Settings, Washington, DC. Institute of Medicine Committee on Antiretroviral Drug Use in Resource-Constrained Settings.
Henry J. Kaiser Family Foundation. 2004a. Global Funding for HIV/AIDS in Resource Poor Settings (HIV/AIDS Policy Fact Sheet #6114). The Henry J. Kaiser Family Foundation. [Online]. Available: http://www.kff.org [accessed August 24, 2004].
Henry J. Kaiser Family Foundation. 2004b. U.S. Government Funding for HIV/AIDS in Resource Poor Settings (HIV/AIDS Policy Fact Sheet #6115). The Henry J. Kaiser Family Foundation. [Online]. Available: http://www.kff.org [accessed August 24, 2004].
Hirschhorn L, Fullern A, Shaw C, Prosser W, Noguera M. 2002. Tool to Assess Site Program Readiness for Initiating Antiretroviral Therapy (ART). [Online]. Available: http://www.deliver.jsi.com/2002/archives/hivaids/SofR_tool/index.cfm [accessed January 7, 2004].
Idoko J. 2004 (January 27). The 2003 WHO Guidelines for ARV use: Perspectives from a User. Presentation at the Institute of Medicine Workshop on Antiretroviral Scale-up in Resource Constrained Settings, Washington, DC. Institute of Medicine Committee on Antiretroviral Drug Use in Resource-Constrained Settings.
Kim J. 2004 (January 27). The Challenge of HIV in 2004 and the UNAIDS/WHO “3 by 5” Program. Presentation at the Institute of Medicine Workshop on Antiretroviral Scale-up in Resource Constrained Settings, Washington, DC. Institute of Medicine Committee on Antiretroviral Drug Use in Resource-Constrained Settings.
Kombe G, Smith O. 2003 (October). The Costs of Anti-retroviral treatment in Zambia. The Partners for Health Reformplus Project, Abt Associates Inc., Bethesda, MD.
Kumaranayake L, et al. 2002. Estimating the infrastructure requirements for an expanded response to HIV/AIDS in low and middle-income countries (Abstract TuPeE5185). Presented at the 14th International AIDS Conference, Barcelona, Spain. [Online]. Available: http://www.aids2002.com/Program/ViewAbstract.asp?id=/T-CMS_Content/Abstract/200206290751023109.xml [accessed July 1, 2004].
LaVake S. 2003. Applying social franchising techniques to youth reproductive health/HIV services. YouthNet Youth Issues Paper #2. Family Health International. [Online]. Available: http://www.fhi.org/NR/rdonlyres/eijal2kvvmtazwuqnkoqk4hbxmdazuanzwojv55tghflkma7sj2k2gusgfv6276emct5uxnwl6iamc/YI4.pdf [accessed July 1, 2004].
Margheiro L. 2004. Paper presented at the Institute of Medicine ARV Workshop, Washington, DC. Institute of Medicine Committee on Antiretroviral Drug Use in Resource-Constrained Settings.
Martin DK, Giacomini M, Singer PA. 2002. Fairness, accountability for reasonableness, and the views of priority setting decision-makers. Health Policy 61:279–290.
Martin DK, Singer PA, Bernstein M. 2003. Access to ICU beds for neurosurgery patients: A qualitative case study. Journal of Neurology, Neurosurgery and Psychiatry 74:1299–1303.
Medicines Control Council. Home page. [Online]. Available: http://www.mccza.com/ [accessed March 11, 2004].
Ministry of Health (Brazil). 2003. The Experience of the Brazilian AIDS Program. [Online]. Available: http://www.aids.gov.br/final/biblioteca/resposta/resp_ingles.pdf [accessed July 1, 2004].
Montague D. 2002. Franchising of health services in low-income countries. Health Policy and Planning 17(2):121–130.
Office of Legislative Policy and Analysis. 2004. Legislative Update, 108th Congress: United States Leadership Against Global HIV/AIDS, Tuberculosis, and Malaria Act of 2003, P.L. 108-25 (S. 250, H.R. 1298). [Online]. Available: http://olpa.od.nih.gov/legislation/108/publiclaws/hivtbmal.asp [accessed July 1, 2004].
Office of the United States Global AIDS Coordinator. 2003. The President’s Emergency Plan for AIDS Relief. 2003. The White House. [Online]. Available: http://www.state.gov/s/gac/ [accessed August 24, 2004].
Quinn TC, Chaisson RE. 2004. International Epidemiology of Human Immunodeficiency Virus in Infections Diseases. 3rd ed. Gorbach SL, Bartlett JG, and Blacklow NR, eds. Philadelphia, PA: WB Saunders Company.
Rollnick R. 2002. Botswana’s high stakes assault on AIDS. Africa Recovery 16(2). [Online]. Available: www.un.org/ecosocdev/geninfo/afrec/vol16no2/.
Royal Danish Ministry for Foreign Affairs. 2000. Building a global community: Globalization and the common good. Copenhagen: Royal Danish Ministry for Foreign Affairs.
Schwartlander B, Stover J, Walker N, Bollinger L, Gutierrez JP, McGreevey W, Opuni M, Forsythe S, Kumaranayake L, Watts C, Bertozzi S. 2001. AIDS. Resource needs for HIV/ AIDS. Science 292:2434–2436.
Schwartlander B, Stover J, Walker N, Bollinger L, Gutierrez JP, McGreevey W, Opuni M, Forsythe S, Kumaranayake L, Watts C, Bertozzi S. 2004 (January 28–29). The Global Fund’s Lessons Learned Regarding ARV Drug Scale-up in Resource-Constrained Settings. Paper presented at the Institute of Medicine ARV Workshop,Washington, DC. Institute of Medicine Committee on Antiretroviral Drug Use in Resource-Constrained Settings.
Smith E. 2002. Social franchising reproductive health services: Can it work? Marie Stopes International Working Paper #5. [Online]. Available: http://www.mariestopes.org.uk/pdf/working-paper-no5-social.pdf [accessed July 1, 2004].
South African National Treasury. 2003. Intergovernmental Fiscal Review. South African National Treasury Table 5.6, page 79. [Online]. Available: http://www.treasury.gov.za/documents/ifr/2003/default.htm [accessed July 1, 2004].
Tchetgen E, Kaplan EH, Friedland GH. 2001. Public health consequences of screening patients for adherence to highly active antiretroviral therapy. Journal of Acquired Immune Deficiency Syndrome 26:118–129.
The Foreign Press Center. 2003. Adapted from “O’Neill, Fauci Discuss President’s AIDS Initiatives,” Washington, DC on May 29, 2003. [Online]. Available: http://www.whitehouse.gov/news/releases/2003/05/print/20030529-8.html [accessed August 24, 2004].
The Global Fund. 2003a. The Global Fund Pledges and Contributions. [Online]. Available: http://www.theglobalfund.org/en/files/pledges&contributions.xls [accessed March 9, 2004].
The Global Fund. 2003b. Round 3 portfolio and programmatic analysis (unpublished). In: Summers T, Kates J, eds. U.S. Government Funding for HIV/AIDS in Resource Poor Settings, The Henry J. Kaiser Foundation.
The Global Fund. 2004a. The Global Fund Home Page. [Online]. Available: http://www.theglobalfund.org/en/ [accessed July 1, 2004].
The Global Fund. 2004b. The Global Fund to Fight AIDS, Tuberculosis and Malaria Progress Report. [Online]. Available: http://www.theglobalfund.org [accessed February 10, 2004].
The White House. 2003. The President’s Emergency Plan for AIDS Relief Fact Sheet. [Online]. Available: http://www.state.gov/g/oes/rls/fs/2003/22270.htm [accessed July 2, 2003].
The World Bank Group. 2001. The US $500 Million Multi-Country HIV/AIDS Program (MAP) for Africa Progress Review Mission—FY01. [Online]. Available: http://www.worldbank.org/afr/aids/map/prog_rpt_01.pdf.
UNAIDS (Joint United Nations Programme on HIV/AIDS). 2003. Report on the State of HIV/AIDS Financing. Geneva: UNAIDS.
U.S. Department of State. 2004. The President’s Emergency Plan for AIDS Relief, U.S. Five-Year Global HIV/AIDS Strategy. Washington, DC: U.S. Department of State.
Wasunna A. 2004 (January 28). Ethical Issues in the Scale-up of ARV Programs in Resource-Poor Settings. Paper presented at the Institute of Medicine Workshop on Antiretroviral Scale-up in Resource Constrained Settings, Washington, DC. Institute of Medicine Committee on Antiretroviral Drug Use in Resource-Constrained Settings.
WHO (World Health Organization). 2003a. Treating 3 Million by 2005. Making it Happen. The WHO Strategy . Geneva: WHO. [Online]. Available: http://www.who.int/3by5/en/ [accessed July 26, 2004].
WHO. 2003b. The World Health Report 2003: Shaping the Future. Geneva: WHO.
WHO. 2004a. WHO/UNAIDS Workshop on Strategic Information for Anti-Retroviral Therapy Programmes. [Online]. Available: http://www.who.int/hiv/strategic/mt300703/en/ [accessed March 11, 2004].
WHO. 2004b. World Health Organization Prequalification Project. [Online]. Available: http://mednet3.who.int/prequal/hiv/hivdefault.shtml [accessed March 11, 2004].
William J. Clinton Foundation. 2004. Foundation Programs. [Online]. Available: http://www.clintonpresidentialcenter.com/foundation_programs.html [accessed January 5, 2004].






























