2
Toxicology
The basic toxicology of sarin and cyclosarin is discussed in Gulf War and Health: Volume 1, hereafter referred to as GW1 (IOM, 2000). That background information is reviewed briefly here, and the review is followed by a discussion of data published since the preparation of GW1, focusing on data that might be relevant to low-dose exposures to sarin. Because sarin and cyclosarin have the same mechanism of action and toxic effects, differing mainly in potency, data on the two compounds are discussed together.
PHYSICAL AND CHEMICAL PROPERTIES
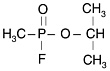
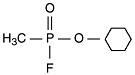
As discussed in GW1 (IOM, 2000), sarin (GB; o-isopropyl methylphosphonofluoridate) and cyclosarin (GF; cyclohexyl methylphosphonofluoridate) are potent neurotoxicant organophosphate esters. Their chemical structures and properties are presented in Table 2-1.
TOXICOKINETICS
Absorption and Metabolism
Organophosphorus (OP) compounds are absorbed rapidly and produce local and systemic effects. Exposure to sarin or cyclosarin can be fatal within minutes to hours. In vapor or liquid form, sarin can be, respectively, inhaled or absorbed through the skin, eyes, or mucous membranes (Stewart and Sullivan, 1992). Because of its extreme potency, sarin is lethal to 50% of exposed people at doses
TABLE 2-1 Physical and Chemical Properties of Sarin and Cyclosarin
|
Characteristic |
Sarin |
Cyclosarin |
|
Chemical name |
Isopropyl methylphosphonofluoridate |
O-Cyclohexyl-methylfluorophosphonate |
|
Synonyms |
Methylphosphonofluoridate, isopropyl ester |
Cyclohexyl methylphosphonofluoridate (CMPF) |
|
Chemical formula |
C4H10FO2P |
C7H14FO2P |
|
Chemical structure |
|
|
|
Molecular weight |
140.10 |
180.2 |
|
CAS Registry Number |
107-44-8 |
329-99-7 |
|
Physical state |
Colorless liquid |
Liquid |
|
Solubility in water, g/L |
Miscible with water |
0.37% (20°C); almost entirely insoluble in water |
|
Vapor pressure |
2.10 mm Hg at 20°C |
0.044 mm Hg at 25°C |
|
Data from DA, 1990. Table modified from NRC, 2003. |
||
of 100–500 mg through the skin, or 50–100 mg-min/m3 by inhalation (in a person who weighs about 70 kg) (Somani, 1992).
In the blood, sarin interacts with several esterases. Some, such as paraoxonase, hydrolyze sarin to inactive metabolites (Davies et al., 1996; Lotti, 2000). Two others—acetylcholinesterase (AChE) and butyrylcholinesterase (BuChE)—irreversibly bind to sarin. Those esterases in the blood are often described as false targets—by binding irreversibly to sarin, AChE and BuChE sequester sarin in the blood, thereby preventing some or all of it from reaching the central nervous system (CNS), depending on the dose (Spencer et al., 2000).
Distribution and Elimination
Animal data obtained by using radioactively labeled sarin indicate that sarin rapidly (within 1 min) distributes to the brain, lungs, heart, diaphragm, kidneys, liver, and plasma; the greatest concentrations are found in the last three (Little et al., 1986). The concentrations in all tissues decline rapidly; a decrease of 85% within 15 min was followed by a second, more gradual decline. The initial, rapid decline appears to be mediated by metabolism, not urinary elimination of the parent compound, inasmuch as about half the labeled sarin was associated within
the first minute with the major sarin metabolite isopropyl methylphosphonic acid (IMPA). A recent study in guinea pigs indicated that sarin stereoisomers reach the blood rapidly after nose-only exposures and then gradually decline (Spruit et al., 2000). The kidneys are the major route of elimination of sarin and its metabolites (Little et al., 1986). Urinary elimination of sarin is rapid (terminal elimination half-life, 3.7 ± 0.1 h); almost all the administered dose of sarin was retrieved from the urine in metabolite form after 2 days (Shih et al., 1994).
Minami et al. (1997) detected IMPA in urine of humans after a terrorist attack on the Tokyo subway system with sarin; peak concentrations were measured 10–18 h after exposure. Evidence of distribution of sarin to the human brain was found in 4 of the 12 people who died after exposure (Matsuda et al., 1998).
BIOMARKERS OF EXPOSURE
Inhibition of blood cholinesterases can be used as a biomarker of exposure to sarin. Although high doses of sarin inhibit both AChE and BuChE, at lower doses sarin preferentially inhibits AChE, making AChE inhibition a more sensitive biomarker of sarin exposure than BuChE inhibition (Sidell and Borak, 1992). Because inhibition of blood cholinesterases is a common feature of sarin, other OP compounds, and some other compounds, cholinesterase inhibition is not a specific biomarker of sarin exposure. Blood esterase activity returns to normal 1–3 months after exposure and this limits its utility as a biomarker to a short time after exposure (Grob, 1963). Fidder et al. (2002) developed a more specific biomarker of sarin poisoning by measuring organophosphate-inhibited BuChE in blood.
Sensitive methods for detecting methylphosphonic acids, which are metabolites of sarin, in blood or urine for use as a biomarker of sarin exposure have been developed (Shih et al., 1991; Black et al., 1994; Fredriksson et al., 1995; Tørnes et al., 1996; Black and Read, 1997, 1998) and used by Japanese researchers in the aftermath of the Tokyo terrorism incident (Minami et al., 1997, 1998; Noort et al., 1998). Those methods have the advantage of having more specificity than use of cholinesterase inhibition, however, they are limited by the fact that methylphosphonic acids are eliminated from the body within several days after exposure to sarin.
Researchers have also measured the amount of phosphyl moiety released upon reactivation of the phosphylated BuChE or AChE by fluoride ion or other treatments (Nagao et al., 1997; Polhuijs et al., 1997; Matsuda et al., 1998). Measurement of the phosphyl moiety allows the type and amount of the OP compound exposure to be determined, and can be used longer after a poisoning episode than by detection of sarin metabolites. More recently, Fidder et al. (2002) measured phosphylated nonapeptides created following pepsin digestion of inhibited BuChE.
MECHANISMS OF TOXICITY
Inhibition of Acetylcholinesterase
The principal mechanism of acute toxicity of sarin and cyclosarin, as of other OP compounds, is inhibition of AChE. AChE is responsible for the hydrolysis of acetylcholine (ACh) at the synapse, and inhibition of AChE leads to a rise in ACh and overstimulation at cholinergic synapses (Somani, 1992; Lotti, 2000; Spencer et al., 2000). At those synapses, the ACh binds and activates muscarinic and nicotinic receptors, the two major subtypes of ACh receptors. Sarin inhibits AChE by phosphorylating a serine hydroxyl on the ester portion of the active site of the enzyme. The phosphorylated enzyme is hydrolyzed very slowly, with a half-life of reactivation of hours to days (Gray, 1984). The phosphorylated enzyme can undergo a second process, called aging, by loss of an alkyl group (dealkylation). Aging occurs within about 5 h of sarin exposure (Sidell and Borak, 1992). After aging has occurred, the phosphorylated enzyme is resistant to cleavage or hydrolysis and can be considered irreversibly inhibited. Recovery of AChE function occurs only with synthesis of new enzyme. Most of the effects of sarin, including the acute cholinergic syndrome, are thought to be mediated by the excess ACh at the synapse.
Other Mechanisms
For decades, as discussed in GW1 (IOM, 2000), researchers observed puzzling relationships between the extent of neurobehavioral toxicity and the degree of inhibition of AChE. For example, sarin-induced tremor has a slight correlation with AChE inhibition in rat striatum, but chewing, hind-limb abduction, and convulsions have no clear correlation (Hoskins et al., 1986). Some sarin-treated rats with 90% inhibition of AChE in the striatum of the brain had no convulsions or hind-limb abduction, but rats with less enzyme inhibition exhibited both. On the basis of those findings, researchers have concluded that mechanisms other than the inhibition of AChE might also contribute to toxicity induced by sarin and other organophosphates. The difficulty, however, has been in differentiating among effects mediated directly by sarin and effects that are secondary to its inhibition of AChE.
Electrophysiologic experiments have indicated that sarin (in picomolar concentrations) can interact with one subtype of ACh receptors, the muscarinic ACh receptors (Rocha et al., 1998; Chebabo et al., 1999). That interaction appears to be direct and is not associated with the inhibition of AChE.
Several studies suggest that sarin can alter the concentrations of neurotransmitters other than ACh. In most of them, however, the neurotransmitter effects are seen in brain regions where there are cholinergic synapses and could be secondary to AChE inhibition (Dasheiff et al., 1977; Fernando et al., 1984;
Somani, 1992). Sarin-like agents have also been shown to alter second-messenger systems in rat brains, including activating phospholipase C gamma (Niijima et al., 1999), mitogen-activated protein kinase (MAPK), and c-Jun N-terminal kinase (JNK) protein activity (Niijima et al., 2000). The mechanisms underlying those effects are unknown. A recent study in rats (Abu-Qare and Abou-Donia, 2001) showed that concurrent exposure to sarin and pyridostigmine bromide produced biomarkers of oxidative stress (3-nitrotyrosine and 8-hydroxy-2′-deoxyguanosine in rats), raising the possibility that some effects in rats could be mediated by oxidative stress.
ACUTE HUMAN EXPOSURES TO ORGANOPHOSPHORUS COMPOUNDS
Immediate Effects
As discussed in GW2, clinical signs of toxicity associated with organophosphate-induced inhibition of AChE depend on dosage. Toxicity in humans and animals includes the signs associated with overstimulation of muscarinic receptors of the autonomic nervous system by ACh: SLUD (salivation and sweating, lacrimation, urination, and defecation), emesis, and bradycardia. AChE inhibition can also cause overstimulation (which can be followed by depression) of nicotinic receptors at neuromuscular junctions and autonomic ganglia and result in ataxia and fasciculations that, at higher dosages, can be followed by flaccid paralysis. Electromyographic changes can be observed after acute poisoning because nicotinic sites in muscles are affected; the changes include decreases in amplitude and increases in peak latencies in nerve conduction (Baker and Wilkinson, 1990; Gallo and Lawryk, 1991; Kaloianova and El Batawi, 1991). Stimulation of autonomic ganglia can also cause hypertension. As is the case at neuromuscular junctions, excess ACh in the CNS causes stimulation that can be followed by depression. Overstimulation can be manifested as nervousness, delirium, hallucinations, and psychoses. Obvious signs do not generally appear until nervous system AChE inhibition approaches 70%.
Not all exposed people show all signs, and signs can vary with the OP compound, dose, route of exposure, and species. Signs often appear within minutes or hours, but they might not appear for several days. Signs can last for minutes to weeks and can be followed by full recovery from obvious manifestations of cholinergic poisoning. If death occurs, it is due to respiratory failure, usually as a result of a combination of the autonomic effects mediated by the muscarinic and nicotinic ACh receptors and the effects of ACh at CNS receptors. Those effects can include excessive fluid in the respiratory tract, paralysis of the respiratory muscles, and depression of the respiratory centers of the CNS.
Delayed Effects
Intermediate Syndrome
Clinical manifestations of acute AChE inhibition in humans or animals are not generally long-lasting or delayed, but there are exceptions. An “intermediate syndrome” has been described after severe poisoning: muscle weakness that occurs about 16–120 h after exposure and 7–75 h after the onset of acute poisoning symptoms (Shailesh et al., 1994; He et al., 1998). Overstimulation of nicotinic receptors and then depression at neuromuscular junctions and muscle necrosis might be contributing factors. The muscle weakness can become severe and result in respiratory insufficiency. Recovery occurs, if respiration can be sustained, but it can take weeks. The intermediate syndrome has been reported in humans after exposure to malathion and diazinon (Gallo and Lawryk, 1991).
Organophosphorus-Induced Delayed Neuropathy
Another type of toxicity caused by a few OP compounds is a progressive, irreversible delayed neuropathy termed organophosphate-induced delayed neuropathy (OPIDN). OPIDN can occur in many species, including humans. Clinical manifestations of OPIDN include progressive ataxia that develops weeks to months after exposure. Lesions are found in peripheral nerves and the spinal cord (Ehrich and Jortner, 2001). OPIDN becomes manifest about 1–4 weeks after an acute exposure to some organophosphates; motor symptoms of ataxia and flaccid paralysis of the lower extremities are exhibited. Symptoms persist for up to a year and may be permanent in severe cases (De Bleecker et al., 1992).
OPIDN is thought to be mediated by effects on an enzyme known as neuropathy target esterase (NTE) (Somani, 1992; Moore, 1998; Lotti, 2000). OPIDN occurs only if OP compounds inhibit NTE sufficiently, and essentially irreversibly, within hours of exposure. Inhibition of NTE is not related to inhibition of AChE. OP compounds are tested for their potential to cause OPIDN before they are registered for use as insecticides, so most commercially available insecticides do not inhibit NTE.
Other Delayed Effects
As discussed in GW2, some studies have reported other persistent symptoms after poisoning with OP compounds or symptoms that appear 5–10 years after a poisoning episode, including neurologic and visual deficits, behavioral alterations, and impairment of cognition. Those effects, however, might be confounded by other factors or result from inappropriate study designs (see Baker and Wilkinson, 1990; Gallo and Lawryk, 1991; Kaloianova and El Batawi, 1991; Chambers and Levi, 1992; Ecobichon and Joy, 1994; Abou-Donia, 1995; Eyer, 1995; Jamal, 1997; Lotti, 2001 for reviews). Although some latent effects have
been noted in laboratory rats, the symptoms reported in people have been difficult to verify in animal studies partly because of difficulties in replication of exposures and extrapolation of end points from humans to animals (see Ballantyne and Marrs, 1992; Bushnell et al., 1993; Ecobichon and Joy, 1994; Gallo and Lawryk, 1991; Marrs et al., 1996; Mattsson et al., 1996; Maurissen et al., 2000 for reviews).
EXPERIMENTAL STUDIES
Most animal studies of sarin and cyclosarin examine the effects at lethal, near-lethal, or maximum tolerated doses (MTDs).1 Those high doses produce the acute cholinergic syndrome, in many cases necessitate pharmacologic intervention to prevent death, and are not useful in distinguishing between primary damage caused by the compound and secondary damage caused by hypoxic events after convulsions. There is no evidence that any Gulf War soldiers had the acute cholinergic syndrome, so studies of acute, high-dose exposure to sarin or cyclosarin are only briefly mentioned, and this section focuses more on studies—published since the preparation of GW2—of the long-term effects of low-dose exposures to compounds that are more relevant to the situation in the Gulf War. This section is organized by the end point studied, and also by studies that look at short-term effects and those that examine effects that persist for weeks or months after a single or short-term exposure. The studies investigating persistent effects are more relevant to the veterans’ situation.
Lethality Studies
In animals, sarin and cyclosarin in microgram quantities are acutely toxic and fatal in a matter of minutes. There is some variability, depending on the species and the route of administration. Table 2-2 outlines some of the doses and routes of administration that produce acute lethality (within 24 h) in animal species tested. The LD50 of cyclosarin in mice (243 μg/kg) is somewhat higher than that of sarin (170 μg/kg) (Clement, 1992). The immediate cause of death from sarin poisoning is respiratory arrest (Rickett et al., 1986); a study by Duncan et al. (2001) indicates that in swine it results from central respiratory failure.
Neurotoxicity
Short-Term Neurotoxicity
Sarin’s short-term behavioral effects are dose-dependent. Sarin has led to conditioned flavor aversion (at doses greater than 70 μg/kg) and to decreased
TABLE 2-2 Acute Lethality of Sarin Administered to Various Species
|
Species |
Routea |
LD50, μg/kg |
Reference |
|
Rat |
s.c. |
158–165 |
Landauer and Romano, 1984; Singer et al., 1987; Somani, 1992 |
|
Mouse |
s.c. |
160–170 |
Clement, 1991 |
|
Mouse |
i.m. |
179 |
Somani, 1992 |
|
Mouse |
i.v. |
109 |
Little et al., 1986; Tripathi and Dewey, 1989 |
|
Mouse |
Inhalation |
600 mg/min per m3 |
Husain et al., 1993 |
|
Guinea pig |
s.c. |
53 (divided doses) |
Fonnum and Sterri, 1981; Somani, 1992 |
|
Hen |
Oral |
561 |
Bucci et al., 1993 |
|
Hen |
s.c. |
16.5–16.7 |
Gordon et al., 1983 |
|
Cat |
s.c. |
30–35 |
Goldstein et al., 1987 |
|
ai.m. = intramuscular; i.v. = intravenous; s.c. = subcutaneous. |
|||
motor coordination in rats as measured by rotarod performance (at 98 μg/kg; Landauer and Romano, 1984). It has led to increased spontaneous locomotion at 61 μg/kg but decreased locomotor activity at higher doses immediately after treatment (Landauer and Romano, 1984); Nieminen et al. (1990) found 50 μg/kg, but not 12.5 μg/kg, to decrease locomotion until 6 h after intraperitoneal administration, and to decrease some behaviors 40–50 min after injection.
As discussed in GW1, short-term behavioral effects have been examined in the marmoset, a nonhuman primate. Doses at 33–55% of the LD50 disrupted the performance of animals’ food-reinforced visually guided reaching response. Performance returned to normal by 24 h after sarin administration (D’Mello and Duffy, 1985).
The only other studies of short-term behavioral consequences of low-dose exposures in nonhuman primates were carried out with soman, an OP nerve agent that also inhibits AChE. Hartgraves and Murphy (1992) studied the effects of different dosing regimens—which did not produce signs of acute toxicity—on equilibrium performance as measured on the primate equilibrium platform (PEP). This device requires the primate to manipulate a joystick to keep a rotating platform as level as possible. Doses below 2.0 μg/kg did not induce and doses above 2.75 μg/kg did induce decrements in PEP performance. Decrements were measured for 5 days after soman administration, but performance later returned to normal. Those findings, although not from sarin, are reported here because vestibular dysfunction has been reported as a long-term effect in humans after sarin exposure.
Since the preparation of GW1, Hulet et al. (2002) tested a functional observational battery and reported EEG results in guinea pigs after a single injection of sarin (0.3, 0.4, 0.5, or 0.6 times the LD50). Few changes were seen in the battery, but at 0.4LD50 and above, responses to an approaching pencil and to a rumptouch were different from controls, and they did not adjust to handling. No changes were seen up to 0.5LD50, but 0.6LD50 led to EEG evidence of seizures. Symptoms of the acute cholinergic syndrome were evident at 0.5LD50 and above.
Studies have looked at the effects of a single dose of sarin on glial markers. Damodaran et al. (2002) studied the effects of sarin (intramuscular injection at 50 μg/kg per milliliter vehicle) on mRNA expression of astroglial markers 1 and 2 h and 1, 3, and 7 days after treatment. Glial fibrillary acidic protein and vimentin were increased in the areas of the brain studied (cortex, midbrain, cerebellum, brainstem, and spinal cord); vimentin induction occurred sooner. Some effects on expression of both could still be detected 7 days after treatment.
Those data indicate that sarin exposure in animals can have effects on neurobehavioral and neurotoxic endpoints. No clear pattern of effects, however, emerges from those studies and their relevance to humans is unknown.
Persistent Neurotoxicity
As discussed in GW1 (IOM, 2000), long-term changes in the electroencephalogram (EEG) of rhesus monkeys have been seen after a single high dose of sarin (5 μg/kg) or a series of 10 small doses (1 μg/kg per week) (Burchfiel et al., 1976; Burchfiel and Duffy, 1982). The high dose was sufficient to produce the acute cholinergic syndrome, whereas each small dose produced few, if any, signs of acute poisoning. Changes persisted for a year after sarin administration, although they did not appear to have any behavioral or psychologic significance. In a later study in marmosets, no statistically significant changes in EEG were detected, but the increase in the beta 2 amplitude (22–40 Hz) approached statistical significance (p = 0.07) (Pearce et al., 1999). The dose did not produce a decrement in touchscreen-mediated discrimination tasks, which are indicators of cognitive functioning.
Since the preparation of GW1, research has been conducted in animals with sarin exposures designed to resemble those which might have occurred in the Gulf War. The studies were specifically designed to investigate possible effects of low-level exposure to sarin that persist for weeks or months after the exposure ends. Henderson et al. (2001; extended abstract encompassing other studies) studied locomotor activity and body temperature in rats exposed only intranasally to sarin (0.2 or 0.4 mg/m3 of air for 1 h/day for 1, 5, or 10 days) in the presence or absence of heat stress (32° C). The higher concentration (0.4 mg/m3) is one-tenth the lethal concentration (LCt50).2 The animals were monitored continually
for a month after exposure, and the data were grouped. No consistent effects were seen. Using the same treatment protocol, Henderson et al. (2002) looked at brain histopathologic effects in rats 30 days after exposure. Heat stress, but not sarin treatment, decreased weight gain and pulmonary function. No lesions or evidence of apoptosis were present. No effect was seen on total brain AChE measurements (from homogenates), but region-specific staining for AChE was decreased in the cerebral cortex, striatum, olfactory bulb, and CA1 region of the hippocampus. Thus, in general, the forebrain concentrations of AChE were most affected. Brain cytokine concentrations (interleukin (IL)-1β, IL-6, and tumor necrosis factor-α) were affected by both sarin treatment and heat stress; this is consistent with evidence of immunosuppression seen in other experiments (decreased anti-sheep RBC antibody forming cell response and suppression of T cell response) (Kalra et al., 2002).
Receptor density was measured for the M1, M2, and M3 subtypes of muscarinic receptors (Henderson et al., 2002). M1 receptors were decreased in a dose-dependent manner. No changes were seen on day 1 after 5 days of treatment, but a decrease in M1 receptors was seen in the olfactory tubercle 30 days after the highest dose. With heat stress, there were also dose-dependent decreases in M1 receptor density in the frontal cerebral cortex, olfactory tubercle, anterior olfactory nucleus, striatum, dentate gyrus, and CA1 region of the hippocampus 30 days after treatment. No changes were seen in M2 receptor densities with any treatments. Sarin did not affect M3 receptor density under normal conditions, but under heat stress there was an increase in the number of M3 receptors at day 1 and day 30 after 5 days of treatment in the frontal cortex, olfactory tubercle, and anterior olfactory nucleus and throughout the striatum. At 30 days, there was also an increase in M3 receptor density in the CA1 region of the hippocampus. Those studies provide the results most relevant to potential effects of exposures in the Gulf War. Although the results on receptor density are not such that they alter the conclusions of this committee regarding the strength of the association between exposure to sarin and neurologic health outcomes, they are suggestive of a potential mechanism through which sarin could cause long-term effects on the nervous system and indicate the desirability of future toxicologic and epidemiologic research. Further studies of concomitant exposure to stressors (e.g., heat) or other chemicals and sarin should also be conducted.
In addition to the studies by Henderson and colleagues, behavioral effects of single and repeated (three times in 1 week) doses of sarin (0, 0.8, 1.35, or 2.5 μg/L) have been investigated in an inhalation chamber since the preparation of GW1. The performance of rats in a T-maze was somewhat affected after exposure to sarin (Kassa et al., 2001a) or sarin plus oximes (Krejcova et al., 2002), as was performance of rats in a Y-maze after exposure to sarin (Kassa et al., 2001b) or to sarin plus oximes (Kassa et al., 2002). The performance of some of the controls, however, was also lower than expected in some of those studies with oximes, and many of the effects seen were reversed by 3 months.
In summary, some changes in EEG and histopathology persisted months to a year after exposure in animals. Those effects, however, did not appear to be associated with detectable behavioral changes or clinically-relevant effects.
Delayed Neurotoxicity
As discussed previously, exposure to some organophosphates produces a delayed neurotoxic syndrome known as organophosphate-induced delayed neuropathy. In some animal models, massive doses of sarin can cause delayed neurotoxicity, which is manifested in ataxia and paralysis days to weeks after a single high exposure or multiple lower exposures (Somani, 1992; Lotti, 2000; Spencer et al., 2000). The doses of most OP compounds capable of producing those neurotoxic effects in experimental animals are typically higher than the lethal dose. Therefore, to study delayed neurotoxicity, most species must be protected from death through pharmacologic and other interventions.
Table 2.3 summarizes findings of animal studies of OPIDN or other forms of delayed neurotoxicity after administration of sarin reviewed in GW1 (IOM, 2000). Sarin produced delayed neurotoxicity in six studies. In four of them, the doses were either the lethal dose or at least 30 times the lethal dose (Davies et al., 1960; Davies and Holland, 1972; Gordon et al., 1983; Willems et al., 1983). Animals displayed severe signs of acute cholinergic toxicity but were protected from death by administration of atropine and other agents. In two studies, however, sublethal doses were administered. Researchers administered sarin to mice (Husain et al., 1993) and white leghorn hens (Husain et al., 1995) for 10 days. At no time did sarin-exposed mice show signs of cholinergic toxicity, although AChE activity was inhibited by 27% (blood) and 19% (brain). No indication was provided on whether cholinergic symptoms were observed in the hens, but platelet AChE activity was inhibited by 72%. Animals developed muscular weakness of the limbs and slight ataxia within 14 days of the beginning of the study. NTE was inhibited in the brain, spinal cord, and platelets, and the spinal cord exhibited axonal degeneration. In several studies, however, sarin did not produce delayed neurotoxicity. Crowell et al. (1989) attributed the negative findings in hens to sarin’s inability to inhibit brain NTE substantially at nonlethal doses.
Taken together, the findings indicate that sarin can cause OPIDN in some animal species, particularly at doses that produce otherwise lethal effects.
Immunotoxicity
Kalra et al. (2002) studied T-cell responses to sarin in Fischer 344 rats. Nose-only exposure of rats to sarin (0, 0.2, or 0.4 mg/m3; 1 h/day for 1, 5, or 10 days) had some effects on T cells isolated from spleen of the rats 1 day after the final sarin treatment. Sarin at 0.2 or 0.4 mg/m3 for 5 or 10 days decreased antibody-forming cells and T cell proliferation, but the number and distribution of cells
were unchanged. Intracellular calcium responses and T cell proliferation were also affected by some treatments. Sarin decreased corticosterone (CORT) concentrations; this indicates that the effect was not mediated by the hypothalamic-pituitary-adrenal axis, and experiments with a ganglionic blocker suggested an autonomic effect.
Recent studies have investigated more long-term effects of sarin on the immune system. Kassa et al. (2000, 2001c) demonstrated modest and inconsistent effects on lymphocyte proliferation and production of N-oxides in rats 3 months after a single or repeated (three times in one week) 1-h inhalation-chamber exposure (0.8, 1.25, or 2.5 μg/L).
The effects of sarin on the immune system of animals, therefore, are inconsistent.
Genotoxicity
A study of the genotoxicity of sarin showed no evidence of genotoxicity (mutagenesis, chromosomal damage, unscheduled DNA synthesis, or sister chromatid exchange) (Goldman et al., 1988). In one study in rats, DNA synthesis was not changed, but an increase in unscheduled DNA repair was observed, although problems with controls and variability provide less confidence in those results (Klein et al., 1987). No studies on the genotoxicity of sarin have been published since GW1 (IOM, 2000).
Cancer
As discussed in GW1 (IOM, 2000) a standard subchronic (90-day) toxicology study of sarin was performed at the National Center for Toxicological Research (Bucci and Parker, 1992; Bucci et al., 1992). This subchronic study is discussed here because of some endpoints seen, but such a study is not adequate for determining the carcinogenicity of a chemical. A lack of tumours in such a study cannot be interpreted to indicate that the chemical is not a carcinogen. Rats were administered sarin in two formulations (type I stabilized with tributylamine and type II with diisopropylcarbodiimide) at three doses: the MTD, MTD/2, and MTD/4—corresponding to 300, 150, and 75 μg/kg per day, respectively—given by gavage. Both formulations produced profound inhibition of AChE and some deaths. No neoplastic lesions were detected after type I sarin, but nonneoplastic lesions (necrosis in the cerebrum related to hypoxia) were detected and were thought to be the cause of death in 3 of 36 female rats (1 at 75 μg/kg, and 2 at 300 μg/kg.). Type II sarin was associated with one neoplastic lesion, a lymphoma, in a male in the high-dose group.
No chronic animal studies have been conducted to determine the carcinogenic effects of exposure to sarin.
TABLE 2-3 Delayed Neurotoxicity of Sarin
|
Species |
Dose, μg/kg |
Route of Administrationa |
Frequency and/or Duration |
|
Chicken |
25 (LD50) |
i.m. |
1×/day for 26–28 days |
|
Hen |
500–2,500 |
i.m. (20% of total dose given) |
1×/day for 5 days |
|
Hen |
252 504–1,962 |
s.c. s.c. |
1× 1× |
|
Chicken |
70.2–281 23–94 |
Gavage Gavage |
1× 1×/week for 3 weeks |
|
Hen |
50 (LD50/10) |
s.c. |
1×/day for 10 days |
|
Hen |
600 900 1,500 900 1,200 |
i.m. |
1×/day for 2 days 1×/day for 3 days 1×/day for 5 days 1×/day for 1 day 1×/day for 1 day |
|
Rat |
75–300 |
Gavage |
5×/week for 13 weeks |
|
Mouse |
5 mg/m3 |
Inhalation |
20 min for 10 days |
|
Cat |
1,000 3.5 7 |
s.c. s.c. s.c. |
1× 1×/day for 10 days 1×/day for 5 days |
|
ai.m. = intramuscular; i.p. = intraperitoneal; i.v. = intravenous; s.c. = subcutaneous. bNA = not available; PAD = dodecyl iodide salt of P2S; P2S = pralidoxime mesylate, 2-hydroxyiminomethyl-N-methylpyri-dinium methyl methanesulfonate. cDN = delayed neuropathy. dNo hens were ataxic at 500 μg/kg. Figures not provided for doses higher than 1,000 μg/kg. |
|||
Reproductive or Developmental Toxicity
As discussed in GW1, sarin appears to have no reproductive effects in rats, rabbits, or dogs (LaBorde et al., 1996; Jacobson et al., 1959).
GENETIC SUSCEPTIBILITY
One of the mechanisms of sarin inactivation is hydrolysis by the enzyme paraoxonase (PON1), an esterase synthesized and secreted by the liver. It is
|
Protectionb |
Neurobehavioral Outcomesc |
Reference |
|
Atropine, P2S, PAD |
5/8 slight ataxia |
Davies and Holland, 1972 |
|
Atropine, P2S dose of 1,000 μg/kgd |
9/28 ataxia at minimal |
Davies et al., 1960 |
|
Physostigmine, atropine, P2S |
0/4 ataxia 12/12 ataxia to paralysis |
Gordon et al., 1983 |
|
Atropine |
None |
Bucci et al., 1993 |
|
Atropine |
None |
|
|
None |
Moderate ataxiae |
Husain et al., 1995 |
|
Atropine, Physostigmine, P2S |
0/4 DN 1/3 DN 8/9 DN 3/4 DN 4/4 DN |
Willems et al., 1983 |
|
NA |
None |
Bucci and Parker, 1992; Bucci et al., 1992 |
|
None |
Slight ataxiaf |
Husain et al., 1993 |
|
Physostigmine, atropine None None |
None Noneg Noneg |
Goldstein et al., 1987 |
|
eStudy does not report how many of five dosed animals developed moderate ataxia. fStudy does not report how many of six dosed animals developed slight ataxia. gNo behavioral signs of neurotoxicity, but sarin decreased conduction velocity of muscle spindle afferents and altered frequency response of primary and secondary nerve endings. |
||
found in humans in the brain and the blood. The human PON1 gene has polymorphisms at positions 192 (Arg/Gln) and 55 (Leu/Met) that affect serum PON1 activity (Furlong et al., 1993). Because human serum PON1 catalyzes the hydrolysis of OP insecticides and nerve gases such as sarin, those polymorphisms might substantially alter a person’s susceptibility to the toxicity of the chemicals. The polymorphism at position 192 accounts for three genotypes (QQ, RR, and QR) related to the catalytic properties of two forms of the PON1 enzyme (types R and Q allozymes), which hydrolyze some organophosphates at different rates.
The R allozyme (Arg192) hydrolyzes the organophosphate paraoxon at a high rate; however, it has a low activity against OP nerve agents such as sarin and soman (Davies et al., 1996). Lower activity means that more sarin would be bioavailable to exert its anticholinesterase effects. The Q allozyme has high activity against OP nerve agents and low activity against paraoxon. Thus, people with the Q allozyme (genotype QQ or QR) are expected to have greater hydrolysis of sarin than people homozygous for the R allele (genotype RR). Animal studies support the role of PON1 in protection against the toxicity of some OP compounds (Costa et al., 2003). The prevalence of the R allele is about 0.3 in Caucasian populations but 0.66 in the Japanese population (Yamasaki et al., 1997). Because that form is associated with low hydrolysis of sarin, the authors hypothesized that it could make the Japanese population more sensitive to the toxicity of sarin, which might contribute to their morbidity and mortality after the terrorist attacks in Japan. Yamada et al. (2001), however, reported that of 10 of the victims of the Tokyo attack, 7 expressed the PON1 Q allele (6 QR, 1 QQ). The genotype that confers high hydrolyzing activity toward sarin, therefore, did not appear to play a role in protecting those exposed against the toxicity of sarin.
The relationship between illness in Gulf War veterans and the PON1 genotype and serum AChE activity has been investigated by Haley et al. (1999). The enzyme activity, or ability to metabolize ACh, can be quantified in serum samples from the veterans. That activity is, in part, a function of the genotype of the veteran. Ill veterans (n = 25) were more likely than controls (n = 20) to possess the R allele (genotype RR or QR; OR, 3.50; CI, 0.26–2.80) and to exhibit lower PON1 type Q arylesterase activity. That study raises the possibility that the R allele represents a risk factor for illness in Gulf War veterans, but in a nested case–control study, Hotopf et al. (2003) did not find any differences in PON1 activity between symptomatic and asymptomatic Gulf War veterans. Those researchers studied symptomatic Gulf War veterans, healthy Gulf War veterans, symptomatic Bosnia peacekeeping veterans, and symptomatic nondeployed military controls. The main outcome measures were PON1 activity and genotype for PON1-55 and -192. The authors observed statistically significant differences in PON1 activity among the four groups, but the two gulf groups did not differ in PON1 activity. However, those deployed to the gulf had significantly lower PON1 activity than the non-Gulf War groups (median difference, 70.9; 95% CI, 20.2–121.5; p = 0.012); the differences were not explained by PON1 polymorphisms. PON1 activity was lower in Gulf War veterans than in military control groups. The effect is independent of ill health in Gulf War veterans.
Those studies do not entirely clarify the role of PON1 in Gulf War veterans. A study by Mackness et al. (2000) suggests that symptomatic Gulf War veterans have lower PON1 activity, but this is not explained by the various genotypes in Hotopf et al. (2003). Nonetheless, the decreased activity of PON1 would result in an increased susceptibility to OP insecticides and gases, such as sarin.
REFERENCES
Abou-Donia MB. 1995. Involvement of cytoskeletal proteins in the mechanisms of organophosphorus ester-induced delayed neurotoxicity. Clinical and Experimental Pharmacology and Physiology 22(5):358–359.
Abu-Qare AW, Abou-Donia MB. 2001. Combined exposure to sarin and pyridostigmine bromide increased levels of rat urinary 3-nitrotyrosine and 8-hydroxy-2'-deoxyguanosine, biomarkers of oxidative stress. Toxicology Letters 123(1):51–58.
Baker SR, Wilkinson CF. 1990. The Effect of Pesticides on Human Health. Princeton, NJ: Princeton Scientific Publishing Co., Inc.
Ballantyne B, Marrs TC. 1992. Clinical and Experimental Toxicology of Organophosphates and Carbamates. Oxford: Butterworth-Heinemann Ltd.
Black RM, Read RW. 1997. Application of liquid chromatography-atmospheric pressure chemical ionisation mass spectrometry, and tandem mass spectrometry, to the analysis and identification of degradation products of chemical warfare agents. Journal of Chromatography A 759:79–92.
Black RM, Read RW. 1998. Analysis of degradation products of organophosphorus chemical warfare agents and related compounds by liquid chromatography-mass spectrometry using electrospray and atmospheric pressure chemical ionisation. Journal of Chromatography A 794:233–244.
Black RM, Clarke RJ, Read RW, Reid MT. 1994. Application of gas chromatography-mass spectrometry and gas chromatography-tandem mass spectrometry to the analysis of chemical warfare samples, found to contain residues of the nerve agent sarin, sulphur mustard and their degradation products. Journal of Chromatography A 662(2):301–321.
Bucci TJ, Parker RM. 1992. Toxicity Studies on Agents GB and GD (Phase 2): 90-Day Subchronic Study of GB (Sarin, Type II) in CD-Rats. Available from the National Technical Information Service. NTIS/AD-A248 618/1.
Bucci TJ, Parker RM, Crowell JA, Thurman JD, Gosnell PA. 1992. Toxicity Studies on Agents GB and GD (Phase 2): 90-Day Subchronic Study of GB (Sarin, Type I) in CD-Rats. Available from the National Technical Information Service. NTIS/AD-A248 617/3.
Bucci TJ, Parker RM, Gosnell PA. 1993. Toxicity Studies on Agents GB and GD (Phase 2): Delayed Neuropathy Study of Sarin, Type II, in SPF White Leghorn Chickens. Available from the National Technical Information Service. NTIS/AD-A257357.
Burchfiel JL, Duffy FH. 1982. Organophosphate neurotoxicity: Chronic effects of sarin on the electroencephalogram of monkey and man. Neurobehavioral Toxicology and Teratology 4(6):767–778.
Burchfiel JL, Duffy FH, Van Sim M. 1976. Persistent effects of sarin and dieldrin upon the primate electroencephalogram. Toxicology and Applied Pharmacology 35(2):365–379.
Bushnell PJ, Pope CN, Padilla S. 1993. Behavioral and neurochemical effects of acute chlorpyrifos in rats: Tolerance to prolonged inhibition of cholinesterase. Journal of Pharmacology and Experimental Therapeutics 266(2):1007–1017.
Chambers JE, Levi PE, eds. 1992. Organophosphates, Chemistry, Fate, and Effects. San Diego: Academic Press.
Chebabo SR, Santos MD, Albuquerque EX. 1999. The organophosphate sarin, at low concentrations, inhibits the evoked release of GABA in rat hippocampal slices. Neurotoxicology 20(6):871–882.
Clement JG. 1991. Variability of sarin-induced hypothermia in mice: Investigation into incidence and mechanism. Biochemical Pharmocology 42(6):1316–1318.
Clement JG. 1992. Efficacy of various oximes against GF (cyclohexyl methylphosphonofluoridate) poisoning in mice. Archives of Toxicology 66(2):143–144.
Costa LG, Cole TB, Furlong CE. 2003. Polymorphisms of paraoxonase (PON1) and their significance in clinical toxicology of organophosphates. Journal of Toxicology 41:37–45.
Crowell JA, Parker RM, Bucci TJ, Dacre JC. 1989. Neuropathy target esterase in hens after sarin and soman. Journal of Biochemical Toxicology 4(1):15–20.
DA (Department of the Army). 1990. Occupational health guidelines for evaluation and control of occupational exposure to nerve agents GA, GB, GD and VX. DA Pam 40-8. US Department of the Army, Headquarters. Washington, DC.
Damodaran TV, Bilska MA, Rahman AA, Abou-Doni MB. 2002. Sarin causes early differential alteration and persistent overexpression in mRNAs coding for glial fibrillary acidic protein (GFAP) and vimentin genes in the central nervous system of rats. Neurochemical Research 27(5):407–415.
Dasheiff RM, Einberg E, Grenell RG. 1977. Sarin and adrenergic–cholinergic interaction in rat brain. Experimental Neurology 57(2):549–560.
Davies DR, Holland P. 1972. Effect of oximes and atropine upon the development of delayed neurotoxic signs in chickens following poisoning by DFP and sarin. Biochemical Pharmacology 21(23):3145–3151.
Davies DR, Holland P, Rumens MJ. 1960. The relationship between the chemical structure and neurotoxicity of alkyl organophosphorus compounds. British Journal of Pharmacology 15:271–278.
Davies HG, Richter RJ, Keifer M, Broomfield CA, Sowalla J, Furlong CE. 1996. The effect of the human serum paraoxonase polymorphism is reversed with diazoxon, soman and sarin. Nature Genetics 14(3):334–336.
De Bleecker JL, De Reuck JL, Willems JL. 1992. Neurological aspects of organophosphate poisoning. Clinical Neurology and Neurosurgery 94:93–103.
D’Mello GD, Duffy EA. 1985. The acute toxicity of sarin in marmosets (Callithrix jacchus): A behavioral analysis. Fundamentals in Applied Toxicology 5(6 Pt 2):S169–S174.
Duncan EJ, Conley JD, Grychowski KD, Conley SA, Lundy PM, Hamilton MG, Sawyer TW. 2001. A comparison of the effects of sarin and succinylcholine on respiratory parameters in anesthetized domestic swine. Military Medicine 166(4):322–327.
Ecobichon DJ, Joy RM, eds. 1994. Pesticides and Neurological Diseases. 2nd ed. Boca Raton, FL: CRC Press.
Ehrich M, Jortner BS. 2001. Organophosphorous-induced delayed neuropathy. In: Krieger RI, ed. Handbook of Pesticide Toxicology. Vol. 2, Agents. 2nd ed. San Diego: Academic Press. Pp. 997–1012.
Eyer P. 1995. Neuropsychopathological changes by organophosphorus compounds—a review. Human and Experimental Toxicology 14(11):857–864.
Fernando JC, Hoskins BH, Ho IK. 1984. A striatal serotonergic involvement in the behavioural effects of anticholinesterase organophosphates. European Journal Pharmacology 98(1):129–132.
Fidder A, Hulst AG, Noort D, de Ruiter R, van der Schans MJ, Benschop HP, Langenberg JP. 2002. Retrospective detection of exposure to organophosphorus anti-cholinesterases: Mass spectrometric analysis of phosphylated human butyrylcholinesterase. Chemical Research in Toxicology 15(4):582–590.
Fonnum F, Sterri SH. 1981. Factors modifying the toxicity of organophosphorous compounds including soman and sarin. Fundamentals in Applied Toxicology 1(2):143–147.
Fredriksson SÅ, Hammerström LG, Henriksson L, et al. 1995. Trace determination of alkyl methylphosphonic acids in environmental and biological samples using gas chromatography/negative-ion chemical ionization mass spectrometry and tandem mass spectrometry. Journal of Mass Spectrometry 30:1133–1143.
Furlong CE, Costa LG, Hassett C, Richter RJ, Sundstrom JA, Adler DA, Disteche CM, Omiecinski CJ, Chapline C, Crabb JW, Humbert R. 1993. Human and rabbit paraoxonases: Purification, cloning, sequencing, mapping and role of polymorphism in organophosphate detoxification. Chemical-Biological Interactions 87:35–48.
Gallo MA, Lawryk NJ. 1991. Organic phosphorus pesticides. In: Hayes WJ Jr, Laws ER Jr, eds. Handbook of Pesticide Toxicology. San Diego: Academic Press. Pp. 917–1123.
Goldman M, Klein AK, Kawakami TG, Rosenblatt LS. 1988. Toxicity Studies on Agents GB (Sarin, Types I and II) and GD (Soman). Available from the National Technical Information Service. NTIS AD-A187841.
Goldstein BD, Fincher DR, Searle JR. 1987. Electrophysiological changes in the primary sensory neuron following subchronic soman and sarin: Alterations in sensory receptor function. Toxicology and Applied Pharmacology 91(1):55–64.
Gordon J, Inns R, Johnson M, Leadbeater L, Maidment M, Upshall D, Cooper G, Rickard R. 1983. The delayed neuropathic effects of nerve agents and some other organophosphorus compounds. Archives of Toxicology 52(2):71–82.
Gray AP. 1984. Design and structure–activity relationships of antidotes to organophosphorus anticholinesterase agents. Drug Metabolism Reviews 15(3):557–589.
Grob D. 1963. Anticholinesterase intoxication in man and its treatment. Handbuch der Experimentellen Pharmakologie 15(Supplement, Chapter 22):989–1027.
Haley RW, Billecke S, La Du BN. 1999. Association of low PON1 type Q (type A) arylesterase activity with neurologic symptom complexes in Gulf War veterans. Toxicology and Applied Pharmacology 157(3):227–233.
Hartgraves SL, Murphy MR. 1992. Behavioral effects of low-dose nerve agents. In: Somani SM, ed. Chemical Warfare Agents. San Diego, CA: Academic Press. Pp. 125–154.
He F, Xu H, Qin F, Xu L, Huang J, He X. 1998. Intermediate myasthenia syndrome following acute organophosphates poisoning—an analysis of 21 cases. Human and Experimental Toxicology 17(1):40–45.
Henderson RF, Barr EB, Blackwell WB, Clark CR, Conn CA, Kalra R, March TH, Sopori ML, Tesfaigzi Y, Menache MG, Mash DC, Dokladny K, Kozak W, Kozak A, Wachulec M, Rudolph K, Kluger MJ, Singh SP, Razani-Boroujerdi S, Langley RJ. 2001. Response of F344 rats to inhalation of subclinical levels of sarin: Exploring potential causes of Gulf War illness. Toxicology and Industrial Health 17(5–10):294–297.
Henderson RF, Barr EB, Blackwell WB, Clark CR, Conn CA, Kalra R, March TH, Sopori ML, Tesfaigzi Y, Menache MG, Mash DC. 2002. Response of rats to low levels of sarin. Toxicology and Applied Pharmacology 184(2):67–76.
Hoskins B, Fernando JC, Dulaney MD, Lim DK, Liu DD, Watanabe HK, Ho IK. 1986. Relationship between the neurotoxicities of soman, sarin and tabun, and acetylcholinesterase inhibition. Toxicology Letters 30(2):121–129.
Hotopf M, Mackness MI, Nikolaou V, Collier DA, Curtis C, David A, Durrington P, Hull L, Ismail K, Peakman M, Unwin C, Wessely S, Mackness B. 2003. Paraoxonase in Persian Gulf War veterans. Journal of Occupational and Environmental Medicine 45(7):668–675.
Husain K, Vijayaraghavan R, Pant SC, Raza SK, Pandey KS. 1993. Delayed neurotoxic effect of sarin in mice after repeated inhalation exposure. Journal of Applied Toxicology 13(2):143–145.
Husain K, Pant SC, Raza SK, Singh R, Das Gupta S. 1995. A comparative study of delayed neurotoxicity in hens following repeated administration of organophosphorus compounds. Indian Journal of Physiology and Pharmacology 39(1):47–50.
Hulet SW, McDonough JH, Shih TM. 2002. The dose-response effects of repeated subacute sarin exposure on guinea pigs. Pharmacology, Biochemistry & Behavior 72(4):835–845.
IOM (Institute of Medicine). 2000. Gulf War and Health, Volume 1: Depleted Uranium, Pyridostigmine Bromide, Sarin, and Vaccines. Washington, DC: National Academy Press.
Jacobson KH, Christensen MK, DeArmon IA Jr, Oberst FW. 1959. Studies of chronic exposures of dogs to GB (isopropyl methylphosphonofluoridate) vapor. Archives of Industrial Health 19(1):5–10.
Jamal GA. 1997. Neurological syndromes of organophosphorus compounds. Adverse Drug Reactions and Toxicological Reviews 16(3):133–170.
Kaloianova FP, El Batawi MA. 1991. Human Toxicology of Pesticides. Boca Raton, FL: CRC Press.
Kalra R, Singh SP, Razani-Boroujerdi S, Langley RJ, Blackwell WB, Henderson RF, Sopori ML. 2002. Subclinical doses of the nerve gas sarin impair T cell responses through the autonomic nervous system. Toxicology and Applied Pharmacology 184(2):82–87.
Kassa J, Krocova Z, Vachek J. 2000. Long-term alteration of immune functions following low level exposure to sarin in rats. Acta Medica 43(3):91–94.
Kassa J, Koupilova M, Vachek J. 2001a. Long-term effects of low-level sarin inhalation exposure on the spatial memory of rats in a T-maze. Acta Medica 44(3):93–96.
Kassa J, Koupilova M, Vachek J. 2001b. The influence of low-level sarin inhalation exposure on spatial memory in rats. Pharmacology, Biochemistry & Behavior 70(1):175–179.
Kassa J, Pecka M, Tichy M, Bajgar J, Koupilova M, Herink J, Krocova Z. 2001c. Toxic effects of sarin in rats at three months following single or repeated low-level inhalation exposure. Pharmacology and Toxicology 88(4):209–212.
Kassa J, Krejcova G, Vachek J. 2002. The impairment of spatial memory following low-level sarin inhalation exposure and antidotal treatment in rats. Acta Medica (Hradec Kralove) 45(4):149–153.
Klein AK, Nasr ML, Goldman M. 1987. The effects of in vitro exposure to the neurotoxins sarin (GB) and soman (GD) on unscheduled DNA synthesis by rat hepatocytes. Toxicology Letters 38(3):239–249.
Krejcova G, Kassa J, Vachek J. 2002. Effect of atropine and the oxime HI-6 on low-level sarin-induced alteration of performance of rats in a T-maze. Acta Medica 45(3):107–110.
LaBorde JB, Bates HK, Dacre JC, Young JF. 1996. Developmental toxicity of sarin in rats and rabbits. Journal of Toxicology and Environmental Health 47(3):249–265.
Landauer MR, Romano JA. 1984. Acute behavioral toxicity of the organophosphate sarin in rats. Neurobehavioral Toxicology and Teratology 6(3):239–243.
Little PJ, Reynolds ML, Bowman ER, Martin BR. 1986. Tissue disposition of [3H]sarin and its metabolites in mice. Toxicology and Applied Pharmacology 83(3):412–419.
Lotti M. 2000. Organophosphorous compounds. In: Spencer P, Schaumburg H, Ludolph A, eds. Experimental and Clinical Neurotoxicology. 2nd edition. New York: Oxford University Press. Pp. 897–925.
Lotti M. 2001. Clinical toxicology of anticholinesterase agents in humans. In: Krieger RI, ed. Handbook of Pesticide Toxicology. Vol. 2, Agents. 2nd ed. San Diego, CA: Academic Press. Pp. 1043–1085.
Mackness B, Durrington PN, Mackness MI. 2000. Low paraoxonase in Persian Gulf War veterans self-reporting Gulf War Syndrome. Biochemical and Biophysical Research Communications 24:729–733.
Marrs TC, Maynard RL, Sidell FR. 1996. Chemical Warfare Agents: Toxicology and Treatment. New York: John Wiley & Sons.
Matsuda Y, Nagao M, Takatori T, Niijima H, Nakajima M, Iwase H, Kobayashi M, Iwadate K. 1998. Detection of the sarin hydrolysis product in formalin-fixed brain tissues of victims of the Tokyo subway terrorist attack. Toxicology and Applied Pharmacology 150(2):310–320.
Mattsson JL, Wilmer JW, Shankar MR, Berdasco NM, Crissman JW, Maurissen JP, Bond DM. 1996. Single-dose and 13-week repeated-dose neurotoxicity screening studies of chlorpyrifos insecticide. Food and Chemical Toxicology 34(4):393–405.
Maurissen JP, Shankar MR, Mattsson JL. 2000. Chlorpyrifos: Lack of cognitive effects in adult Long-Evans rats. Neurotoxicology and Teratology 22(2):237–246.
Minami M, Hui DM, Katsumata M, Inagaki H, Boulet CA. 1997. Method for the analysis of the methylphosphonic acid metabolites of sarin and its ethanol-substituted analogue in urine as applied to the victims of the Tokyo sarin disaster. Journal of Chromatography B Biomedical Sciences and Applications 695(2):237–244.
Minami M, Hui DM, Wang Z, Katsumata M, Inagaki H, Li Q, Inuzuka S, Mashiko K, Yamamoto Y, Ootsuka T, Boulet CA, Clement JG. 1998. Biological monitoring of metabolites of sarin and its by-products in human urine samples. Journal of Toxicologic Sciences 23(Suppl 2):250–254.
Moore DH. 1998. Long term health effects of low dose exposure to nerve agent. Journal of Physiology 92(3–4):325–328.
Nagao M, Takatori T, Matsuda Y, Nakajima M, Iwase H, Iwadate K. 1997. Definitive evidence for the acute sarin poisoning diagnosis in the Tokyo subway. Toxicology and Applied Pharmacology 144(1):198–203.
Nieminen SA, Lecklin A, Heikkinen O, Ylitalo P. 1990. Acute behavioural effects of the organophosphates sarin and soman in rats. Pharmacology and Toxicology 67(1):36–40.
Niijima H, Nagao M, Nakajima M, Takatori T, Matsuda Y, Iwase H, Kobayashi M. 1999. Sarin-like and soman-like organophosphorous agents activate PLC gamma in rat brains. Toxicology and Applied Pharmacology 156(1):64–69.
Niijima H, Nagao M, Nakajima M, Takatori T, Iwasa M, Maeno Y, Koyama H, Isobe I. 2000. The effects of sarin-like and soman-like organophosphorus agents on MAPK and JNK in rat brains. Forensic Science International 112(2–3):171–178.
Noort D, Hulst AG, Platenburg DH, Polhuijs M, Benschop HP. 1998. Quantitative analysis of O-isopropyl methylphosphonic acid in serum samples of Japanese citizens allegedly exposed to sarin: Estimation of internal dosage. Archives of Toxicology 72(10):671–675.
NRC (National Research Council). 2003. Acute Exposure Guideline Levels for Selected Airborne Chemicals: Volume 3. Washington DC: The National Academies Press.
Pearce PC, Crofts HS, Muggleton NG, Ridout D, Scott EAM. 1999. The effects of acutely administered low dose sarin on cognitive behaviour and the electroencephalogram in the common marmoset. Journal of Psychopharmacology 13(2):128–135.
Polhuijs M, Langenberg JP, Benschop HP. 1997. New method for retrospective detection of exposure to organophosphorus anticholinesterases: Application to alleged sarin victims of Japanese terrorists. Toxicology and Applied Pharmacology 146(1):156–161.
Rickett DL, Glenn JF, Beers ET. 1986. Central respiratory effects versus neuromuscular actions of nerve agents. Neurotoxicology 7(1):225–236.
Rocha ES, Chebabo SR, Santos MD, Aracava Y, Albuquerque EX. 1998. An analysis of low level doses of cholinesterase inhibitors in cultured neurons and hippocampal slices of rats. Drug and Chemical Toxicology 21 (Suppl 1):191–200.
Shailesh KK, Pais P, Vengamma B, Muthane U. 1994. Clinical and electrophysiological study of intermediate syndrome in patients with organophosphorous poisoning. Journal of the Association of Physicians of India 42(6):451–453.
Shih ML, Smith JR, McMonagle JD, Dolzine TW, Gresham VC. 1991. Detection of metabolites of toxic alkylmethylphosphonates in biological samples. Biological Mass Spectrometry 20(11):717–723.
Shih ML, McMonagle JD, Dolzine TW, Gresham VC. 1994. Metabolite pharmacokinetics of soman, sarin and GF in rats and biological monitoring of exposure to toxic organophosphorus agents. Journal of Applied Toxicology 14(3):195–199.
Sidell FR, Borak J. 1992. Chemical warfare agents: II. Nerve agents. Annals of Emergency Medicine 21(7):865–871.
Singer AW, Jaax NK, Graham JS, McLeod CG Jr. 1987. Cardiomyopathy in soman and sarin intoxicated rats. Toxicology Letters 36(3):243–249.
Somani SM. 1992. Chemical Warfare Agents. New York: Academic Press.
Spencer P, Wilson B, Albuquerque E. 2000. Sarin, other “nerve agents,” and their antidotes. In: Spencer P, Schaumburg H, Ludolph A, eds. Experimental and Clinical Neurotoxicology. 2nd edition. New York: Oxford University Press.
Spruit HE, Langenberg JP, Trap HC, van der Wiel HJ, Helmich RB, van Helden HP, Benschop HP. 2000. Intravenous and inhalation toxicokinetics of sarin stereoisomers in atropinized guinea pigs. Toxicology and Applied Pharmacology 169(3):249–254.
Stewart CE, Sullivan J Jr. 1992. Military munitions and antipersonnel agents. In: Sullivan JB Jr, Krieger G, eds. Hazardous Materials Toxicology: Clinical Principles of Environmental Health. Baltimore, MD: Williams & Wilkins. Pp. 986–1014.
Tørnes JA. 1996. Identification of alkyl methylphosphonic acids by thermospray tandem mass spectrometry. Rapid Communications in Mass Spectrometry 10:878–882.
Tripathi HL, Dewey WL. 1989. Comparison of the effects of diisopropylfluorophosphate, sarin, soman, and tabun on toxicity and brain acetylcholinesterase activity in mice. Journal of Toxicology and Environmental Health 26(4):437–446.
Willems JL, Palate BM, Vranken MA, De Bisschop HC. 1983. Proceedings of the International Symposium on Protection against Chemical Warfare Agents. Umea, Sweden: National Defense Research Institute. Pp. 95–100.
Yamada Y, Takatori T, Nagao M, Iwase H, Kuroda N, Yanagida J, Shinozuka T. 2001. Expression of paraoxonase isoform did not confer protection from acute sarin poisoning in the Tokyo subway terrorist attack. International Journal of Legal Medicine 115(2):82–84.
Yamasaki Y, Sakamoto K, Watada H, Kajimoto Y, Hori M. 1997. The Arg192 isoform of paraoxonase with low sarin-hydrolyzing activity is dominant in the Japanese. Human Genetics 101(1):67–68.