1
Introduction
DOD’S CONGRESSIONALLY DIRECTED MEDICAL RESEARCH PROGRAMS
The U.S. Army, on behalf of the Department of Defense (DOD), administers a set of biomedical research programs that support basic, translational, and clinical research projects; research training; and research infrastructure for specific diseases identified by Congress. The programs consist of awards that are made to extramural investigators and that are selected through a two-stage external peer review process that includes scientists, clinicians, and consumer advocates. In recent years, Congress has earmarked approximately $350 million per year in total for these programs, called collectively the Congressionally Directed Medical Research Programs (CDMRP).
Origin of the Program
CDMRP was initiated in 1992 in response to several forces. One was the emergence of women’s health as an urgent public policy issue. In July 1991, for example, the New England Journal of Medicine published several studies showing that there was sex bias in the management of coronary heart disease. In addition, the National Institutes of Health (NIH) had recently launched a women’s health initiative and was requiring the inclusion of women in clinical trials.
At the same time, concern about breast cancer specifically was being mobilized by a new grassroots organization, the National Breast Cancer Coalition
(NBCC), which was created in May 1991 (Casamayou, 2001). In October 1991, NBCC’s efforts generated 600,000 letters to Congress and the White House that asked for increased spending on breast cancer research, and in February 1992, NBCC held research hearings during which leading breast cancer scientists identified research needs. On the basis of this meeting, NBCC began to campaign for “$300 million more” for cancer research and emphasized the need to fund research in ways that were different from those employed by traditional federal medical research agencies (Visco, 2004).
Lobbying by breast cancer groups had previously resulted in congressionally mandated funding increases for breast cancer research at DOD and the National Cancer Institute (NCI) in the fiscal year (FY) 1992 federal budget. The DOD budget earmarked $25 million for research on the screening and diagnosis of breast cancer among military medical beneficiaries and their dependents. Congress also directed NCI to increase its efforts in breast cancer research, as well as prostate and ovarian cancer research, by $100 million in FY 1992. However, the Budget Enforcement Act of 1990 imposed a strict cap on budget increases in domestic discretionary programs, and an increase of only $30 million was provided to NCI in FY 1992. Even with this increase, cuts in existing cancer research programs were necessary to accommodate the new mandates from Congress. This was the first time that cuts in ongoing cancer research programs were required to provide increases for new cancer research initiatives.
In addition, although this budget increase raised breast cancer research spending at NCI to $133 million in FY 1992, this was still substantially less than the $300 million urged by NBCC. As a result, many members of the cancer research and advocacy communities, spearheaded by NBCC, worked with Congress to identify a source of new funds for breast cancer research that would not further reduce the funding for existing cancer research programs.
One attractive source of funding at that time was DOD, which had approximately $29 billion in unobligated funds from prior years for the development of weapons systems planned before collapse of the Soviet Union in 1991.1 Those funds were put off limits by the Budget Enforcement Act of 1990, which established “firewalls” between the budgets for defense, foreign affairs, and domestic programs and imposed strict caps on funding increases in each of the three categories. A number of attempts were made to breach the firewalls by transferring defense funding to domestic programs, including two attempts in September 1992 that would have increased funding for breast cancer research specifically, but they all failed.2 Ultimately, Senator Tom Harkin put forward an amendment
to the FY 1993 defense budget to increase funding for breast cancer research within DOD (rather than at NIH) by $185 million, to bring the total breast cancer research program within DOD to $210 million (Watson, 1992). As a transfer within DOD’s research and development (R&D) budget, the amendment did not violate the budget agreement’s firewalls. The funds were to be taken from the Strategic Defense Initiative and thus would be above and beyond the battlefield medicine-oriented core program of the U.S. Army Medical Research and Materiel Command (USAMRMC),3 which was funded at $410 million in the Senate bill. The “Harkin Amendment for Breast Cancer” which passed in the Senate by a vote of 89 to 4, also stipulated that all projects funded by the resulting Breast Cancer Research Program (BCRP) would have to undergo peer review (Mervis, 1993).
During the debate on the amendment, Senator Harkin indicated his intent to have these funds made available to the cancer research community by the Army in collaboration with NCI. (NCI itself was slated to receive $220 million for breast cancer research in the Senate version of the FY 1993 appropriation act for NIH).4
Initial Establishment of the Program
Discussions between USAMRMC and NIH about participating in the setting of research priorities and the review of proposals broke down (Science, 1992; Washington Fax, 1992), and the USAMRMC contracted with the Institute of Medicine (IOM) to identify research priorities and funding mechanisms and design a peer review system (IOM, 1993).5 To manage the new breast cancer research program, USAMRMC established a new research area directorate (RAD), now known as the Office of the CDMRP.
|
|
bill over several years, including $170 million for breast cancer research in FY 1993 (Congressional Record, September 16, 1992, p. S13600). The amendment was defeated 62 to 36. The next day, Senator Alphonse D’Amato offered an amendment specifically to transfer $214 million for breast cancer research from DOD to NIH, which was defeated 53-43 (Congressional Record, September 17, 1992, p. S13700). |
At the time, the $210 million constituted a “one-time” appropriation that might or might not be continued in following years (IOM, 1997). The IOM report recommended that at least $151.5 million (72 percent) of the funding should be used for innovative interdisciplinary research projects in the form of new investigator, developmental and exploratory research, and investigator-initiated awards; that up to $27.0 million (13 percent) should be used for training and recruitment fellowships and programs; that up to $21.0 million (10 percent) should be used for infrastructure enhancement, such as banks of tumor samples, tissue, and cell lines and expanded cancer registries; and that $10.5 million (5 percent) should be used for administration (IOM, 1993:Ch. 2).
The report outlined three mechanisms of support for research projects: Individual Investigator Awards (similar to NIH’s R01 grants); New Investigator Awards (similar to NIH’s R29 FIRST grants); and Innovative Developmental and Exploratory (IDEA) Awards. IDEA Awards were a new mechanism intended to stimulate innovative but high-risk ideas of scientists already in or new to the field of cancer research. These scientists were not required to have the preliminary data that would be required by traditional individual investigator awards. The report recommended that the Army program use a two-tiered review system, in which the first tier would consist of peer review of scientific merit conducted by study sections (i.e., expert panels) and the second tier would consist of an assessment of programmatic relevance by an advisory council of 16 to 18 persons, 3 to 4 of whom would be consumer representatives or representatives of the public interest (IOM, 1993:Ch. 3). The Army adopted the IOM recommendations and engaged the American Institute of Biological Sciences to conduct the peer review process. The 2,668 proposals responding to the Broad Area Announcement issued by USAMRMC in late 1993 were reviewed and scored for scientific merit by 41 peer review panels and were referred to the advisory council recommended by IOM, which was (and still is) called the Integration Panel. The Integration Panel recommended 433 proposals for funding, and the awards were made by the end of September 1994 (Kaiser, 1994).6
Evolution of the Program
Few major changes have been made in the structure and administration of the program since the implementation of the FY 1993 appropriation. Consumers were added to the peer review panels in 1995. Additional programs were added, each with its own first tier peer review panels and second tier Integration Panel, following the BCRP model. CDMRP has over time expanded to include seven additional core research programs—neurofibromatosis in FY 1996; prostate cancer and ovarian cancer in FY 1997, biomedical issues directly relevant to military
health known as the Peer Reviewed Medical Research Program (PRMRP) in FY 1999; and tuberous sclerosis, chronic myelogenous leukemia, and prion disease in FY 2002—each established by Congress at the urging of advocacy groups.7 Each program has its own peer review panels and Integration Panel composed of scientists, clinicians, health care professionals, and consumer advocates who have extensive knowledge of or experience with the disease in question, or both.
In 1997, IOM was asked to review CDMRP’s BCRP. The report of the review noted that, beginning with the FY 1996 program announcements inviting applications, the program underwent a significant shift in program priorities and strategies:
While formerly oriented toward research on breast cancer prevention, detection, treatment, and quality of life, the mission of the BCRP explicitly shifted towards breast cancer eradication…the mandate to eradicate the disease was to be achieved by emphasis on innovation and new ideas, bringing new investigators into the field, focusing on under-represented areas, and fostering multidisciplinary approaches. (IOM, 1997:55)
The shift in priorities resulted in increased emphasis on IDEA and other award mechanisms for stimulating new ideas and innovative approaches both in basic and translational research. As will be documented below, the emphasis on exploratory research and new investigators has continued up to the present.
Contributions of the Program
IOM’s review in 1997 was conducted too early in the program to see research results. The report concluded that USAMRMC had “succeeded in establishing a fair peer review system and a broad-based research portfolio by stimulating scientist from a wide range of disciplines to participate as applicants, reviewers, and advisers” (IOM, 1997:11). It went on to say:
The [IOM] committee commends the Army for developing such a program under the serious time constraints and fluctuations in funding that have characterized the program to date. Moreover, the program fills a unique niche among public and private funding sources for cancer research. It is not duplicative of other programs and is a promising vehicle for forging new ideas and scientific breakthroughs in the nation’s fight against breast cancer.
The 1997 IOM committee identified a number of outstanding program features, including the flexibility of the annual priority setting process; the use of outside peer reviewers to evaluate proposals; the involvement of consumer advocates in the peer review process; and low administrative costs. It called for authorizing the program on a continuing rather than a year-to-year basis; introducing ongoing
planning and evaluation; and establishing an outside committee for oversight and program evaluation.
CDMRP had already evaluated the impact of including consumers on peer review panels, finding that technical experts as well as consumers considered consumer involvement to be beneficial (Andejeski et al., 2002a, 2002b). In addition, CDMRP has identified many of the program’s contributions in the areas of programmatic innovations, the building of infrastructure, and research advances.
Programmatic Innovations
Innovative program features include the participation of consumer advocates in all aspects of priority setting and peer review; a criteria-based scoring system for reviewing research proposals; and the development of the IDEA Award to foster innovation by supporting higher risk but potentially higher gain research ideas (Young-McCaughan et al., 2002; Rich et al., 1998).
Infrastructure Building
CDMRP has funded the development of shared research resources, such as registries; tissue, tumor, and cultured cell banks; transgenic animals; databases; and research centers and consortia. The cell banks were used in important telomerase studies and in the development of a BRCA1 mutant cell line. The program has also supported the development of new investigators and the involvement of experienced investigators from other fields of research. Through 2002, CDMRP had made more than 1,500 training and recruitment awards, including 35 institutional training grants, 1,156 pre- and postdoctoral fellowships, and 184 career development awards and sabbaticals (CDMRP, 2003:Table II-3).
Research Advances
DOD-funded investigators have made a number of advances. The most notable is probably the identification and understanding of antagonists to the overexpression of the HER2 gene, which led to the development of Herceptin, a breast cancer therapeutic agent based on an anti-HER2 antibody (CDMRP, 1999:Section 5). BCRP awarded Dennis Slamon two grants in 1993 totaling $1.7 million to establish a tumor tissue bank and study the biologic effects of HER2 overexpression and the impact of agonists and antagonists to the HER2 receptor in human breast tumor normal cell lines.8 Slamon had begun this work several
TABLE 1-1 CDMRP Award Results, FY 1993-FY 2003
|
|
BCRP |
PCRP |
OCRP |
NFRP |
|
Publications in Scientific Journals |
~6,200 |
~700 |
~100 |
~125 |
|
Abstracts/Presentations at Professional Meetings |
~4,200 |
~800 |
~120 |
~155 |
|
Patents/Licensures (including applications) |
~140 |
~35 |
~6 |
6 |
|
SOURCE: CDMRP, 2003. |
||||
years earlier with funding from the National Cancer Institute and Revlon, but researchers still lacked a regular source of breast tissue from women with breast cancer, women who were high risk (from biopsies), and normal women (from breast reduction surgery) (Stabiner, 1997:410).
Other investigators have studied the function of the hereditary breast cancer gene, BRCAl, and have developed a functional assay for BRCAl that can distinguish between normal and mutant copies of the gene. An early IDEA grant for $148,000 led to the development of FDA-approved devices for conducting ductal lavage, useful in studying cancer of the milk ducts of the breast. The grant recipient, Susan Love, could not obtain funding elsewhere, because she had never done research and her idea was unconventional (Haran, 2001). Prostate Cancer Research Program (PCRP)-funded researchers have discovered three genes implicated in hereditary prostate cancer and another gene that is frequently missing in nonhereditary metastatic prostate cancers, indicating that it might play an important role in suppressing the spread of prostate cancer (CDMRP, 2003:IV-12).
The CDMRP website includes a searchable database of award abstracts and publications associated with them.9 The number of publications in peer-reviewed journals, abstracts presented at professional meetings, and patents and licenses granted or applied for is in Table 1-1.
Since its inception through FY 2004, CDMRP has managed research programs that have totaled nearly $3 billion in congressional appropriations, and it has awarded a total of 4,910 grants and contracts, with appropriations made for peer reviewed research each year since 1992. Although CDMRP began as an artifact of the budget process in the early 1990s, it has grown to be well respected by the beneficiaries of its programs (advocacy groups and scientists), who view it as efficient, effective, responsive to science, and as a program with relatively low overhead costs (6 percent across core programs).
As new programs were added to CDMRP and the budget escalated—and the call for more earmarked funding grew louder—more organizations began to
compete for the same pool of funds. However, budgetary constraints have led Congress to question whether funding increases to these programs can continue within the resources available for military spending and whether there might be some way to leverage the investment in these programs through partnerships or collaborations with nonfederal sources.
IMPETUS FOR THIS REPORT
The conference report for DOD’s FY 2004 appropriations contained a provision entitled “Cost Sharing for Medical Research Programs,” in which it commended DOD “for its management of the peer reviewed medical research and cancer research programs,” but noted “with concern the challenge of funding increases to these programs within the resources available for military spending” (U.S. Congress, 2003). The conferees directed “the Assistant Secretary of Defense (Health Affairs), in consultation with the service Surgeons General and the Institute of Medicine, to investigate alternative funding sources, including private sector and non-Federal contributions that can best be used to leverage appropriated funds without biasing the peer review selection process.”
In response, the IOM Committee on Alternative Funding Strategies for DOD’s Peer Reviewed Biomedical Research Program was established to assess current and alternate funding mechanisms and funding sources, which include private sector and other nonfederal entities, for conducting biomedical research. Specifically, the committee was asked to carry out the following tasks:
-
Advise DOD on how these sources and mechanisms can be leveraged to augment appropriate funds.
-
Identify and advise DOD on new, possible future avenues of funding other than those described in task one.
-
Identify and advise DOD on:
-
Issues inherent in the federal procurement system that would impact grants and cooperative agreements
-
Regulations and policies should alternate funding strategies be used.
-
-
Identify risks and solutions regarding bioethics and peer review bias with respect to alternate funding.
To conduct its assessment in the short period of time allotted, the committee met three times between March and May and convened a two-day workshop April 26-27, 2004, where it heard numerous perspectives on the charge before it. Presenters included stakeholders from voluntary health agencies (VHAs) affiliated with the disease-specific programs of CDMRP, individuals representing
public-private partnerships, industrial and other private philanthropic and not-for-profit research sponsors, academic researchers and administrators, and representatives of state-run research programs (see Appendix C for the workshop agenda). In addition, the committee was briefed by CDMRP program managers. Much of the committee’s fact-gathering effort was focused on understanding the nature of the CDMRP research programs, which are described below. This understanding provided the backdrop against which any recommendations about alternative or supplemental sources of funding had to be considered.
OVERVIEW OF THE CURRENT CDMRP
CDMRP is a unit of USAMRMC—the medical research, development, logistics, and acquisition arm of the U.S. Army. USAMRMC operates six medical research laboratories and institutes that represent the core science and technology capability of the Command and that serve as centers of excellence in specific areas of biomedical research related to combat medicine.10 In addition to CDMRP, the Command is organized around four core RADs for infectious diseases, combat casualty care, military operational medicine, and medical chemical and biological defense. There is also a Telemedicine and Advanced Technology Research Center (TATRC).
In addition to the medical research programs planned by the Army, the Command administers programs added to the DOD budget by Congress, called Congressional Special Interest (CSI) programs, not all of which are administered by CDMRP. Some are assigned to the appropriate RAD or to TATRC. Current examples of CSI programs include Military Human Immunodeficiency Virus Research; Surgical Tissue Replacement and Repair; the Neurotoxin Exposure Treatment Program; the Osteoporosis and Bone Health Research Program; Epidermolysis Bullosa Research; and the Center for Innovative Minimally Invasive Therapy.
Partnerships: Consumers, the Scientific Community, and the Military
Most notably, CDMRP has been a pioneer in its work in consumer involvement in scientific priority setting (Rich et al., 1998). The participation of consumer reviewers, who are considered an integral part of the peer review and programmatic processes—and who act as equal partners with scientists—has helped assure that the human dimensions of a disease are incorporated into the scientific and program policy considerations, the investment strategy, and the research focus. Although limited in number, consumer reviewers are full voting members of the peer review panel. DOD works to achieve a broad ethnic and
|
10 |
See mrmc-www.army.mil/. |
cultural representation of consumer reviewers by disseminating information to minority consumer groups and by performing formal outreach activities targeted to these groups.
CDMRP makes every attempt to ensure that consumers have an equal voice in all processes, from the initial stakeholders’ meeting through programmatic and peer review. Survivors and their family members are well informed and offer a variety of perspectives, with some who focus on basic research, while others demonstrate more knowledge about diagnosis and treatment. Part of the reason that consumer representatives are so well informed is that advocacy groups, such as NBCC, provide training programs to teach consumers about science, as well as lobbying and advocacy.
Scientists and clinicians provide the needed subject matter expertise on peer review panels, and basic scientists and clinicians participate in vision setting and programmatic review, helping CDMRP support innovative, interdisciplinary approaches and collaborations in the scientific community that lead to uncovering the complex causes of disease and translating this knowledge into improvements in disease prevention, patient survival, and quality of life.
Military personnel, civilian, and contractor staff are responsible for executing the congressional directives, working together to implement each program. Several programs have a direct military focus, particularly that of improving the health of the military forces. The military continues to be a central partner in all aspects of CDMRP, through day-to-day coordination and administration, through programs that have a military focus, and through supporting research with the Small Business Innovation Research Program (SBIR).
Representatives of private industry sit on CDMRP Integration Panels, as do members of the public and other funding agencies, such as NCI, the Centers for Disease Control and Prevention (CDC), FDA, and the National Science Foundation, and they participate in discussions regarding effective business processes. In fact, before an Integration Panel is convened, the manager of a particular research program holds a meeting during which the various stakeholders who have an interest in that program can make recommendations. The Ovarian Cancer Research Program meeting, for example, would include representatives of pharmaceutical companies, biotechnology companies, different advocacy groups, NCI, and others who fund ovarian cancer research. These groups also help CDMRP programs establish their initial Integration Panels.
By most accounts, CDMRP has been efficiently managed, scientifically productive, and a valuable component of the nation’s health research enterprise, despite initial skepticism about its location in DOD and the participation of consumers in peer review as well as priority-setting processes. Some of the distinctive features of CDMRP are its inclusive program planning and priority-setting process, the rigorous peer review of proposals for scientific merit and program relevance by outside reviewers that includes consumers, the inclusion of VHAs representing patients and survivors in both the priority-setting and peer
review processes, the emphasis on exploratory high-risk/high-gain basic, translational, and clinical research projects and research capacity building, and the periodic sharing of results by investigators. Because of the size and focus of the programs, advocacy groups find CDMRP particularly accessible.
At the committee’s April workshop, Fran Visco, President of NBCC, said, “It is transparent in large part because it is focused on a particular issue and because of the program structure and the budget limitations. It is functional. It is responsive to real need.” Advocates also support the program’s strategy of fostering innovation by supporting novel ideas and new investigators. At the time of the establishment of BCRP, the original program, advocates for breast cancer research saw the program as a way to support new ideas about the causes of disease that could be translated into new treatments, instead of supporting research to extend and refine ideas that have been proven, which NIH was already doing with far greater resources (Marshall, 1993). In general, the emphasis on novel ideas and new investigators—whether the research is basic, clinical, or translational—has been adopted by all CDMRP programs.
Funding of CDMRP
In contrast to the individual institutes and centers of NIH, which are legislatively created standing entities that focus consistently on certain key diseases, CDMRP programs depend on yearly congressional appropriations for each program because they are not included in the President’s proposed budget for DOD. Congress adds the funds annually to the DOD appropriation to fund new programs or to augment existing DOD or Army programs. Thus, CDMRP originated and operates within an environment that required and fostered the development of novel approaches to its operation as a funding agency.
Planning occurs one year at a time, with no standing peer review panels. This allows CDMRP to create new research opportunities and to focus funding on the most recently recognized research gaps or controversies. The Integration Panel seeks a broad portfolio of grants across all disciplines and often gives preference to those proposals that involve interdisciplinary or collaborative research or that address a program priority, even if this results in funding proposals that may not have received the top score in peer review. Table 1-2 provides a summary of the funding history for each of the existing programs, from 1992 to 2004.11Figure 1-1 depicts the planning and funding process of CDMRP.
There are advantages to CDMRP being funded through annual congressional appropriations. Advocates who lobby Congress for funds are invested in the
|
11 |
After the report was drafted, Congress passed the FY 2005 appropriations bill for DOD, which continued the core programs at the FY 2004 level except for the Tuberous Sclerosis Complex Research Program ($3.2 million) and the National Prion Research Program ($1.5 million). See thomas.loc.gov/cgi-bin/cpquery/R?cp108:FLD010:@1(hr622). |
TABLE 1-2 Funding History of CDMRP Core Programs (in millions of dollars)
|
Program |
FY92 |
FY93 |
FY94 |
FY95 |
FY96 |
FY97 |
FY98 |
FY99 |
FY00 |
FY01 |
FY02 |
FY03 |
FY04 |
|
Breast Cancer |
25.0 |
210.0 |
30.0 |
150.0 |
75.0 |
108.3 |
135.0 |
136.8 |
176.3 |
177.4 |
151.5 |
152.2 |
150.0 |
|
Prostate Cancer |
|
|
|
|
|
45.0 |
40.0 |
50.0 |
75.0 |
100.0 |
85.0 |
85.0 |
85.0 |
|
Neurofibromatosis |
|
|
|
|
8.0 |
8.0 |
9.8 |
11.5 |
15.0 |
17.0 |
21.0 |
20.0 |
20.0 |
|
Ovarian Cancer |
|
|
|
|
|
7.5 |
10.0 |
10.0 |
12.0 |
12.0 |
10.2 |
10.0 |
10.0 |
|
Peer Reviewed Medical Research |
|
|
|
|
|
|
|
19.5 |
25.0 |
50.0 |
50.0 |
50.0 |
50.0 |
|
Chronic Myelogenous Leukemia |
|
|
|
|
|
|
|
|
|
|
5.0 |
4.3 |
4.3 |
|
Tuberous Sclerosis Complex |
|
|
|
|
|
|
|
|
|
|
1.0 |
2.0 |
3.0 |
|
National Prion |
|
|
|
|
|
|
|
|
|
|
42.5 |
0 |
0 |
|
Total |
25.0 |
210.0 |
30.0 |
150.0 |
83.0 |
168.8 |
194.8 |
227.8 |
303.3 |
356.4 |
366.2 |
323.5 |
322.3 |
|
SOURCE: Annual reports of CDMRP. |
|||||||||||||
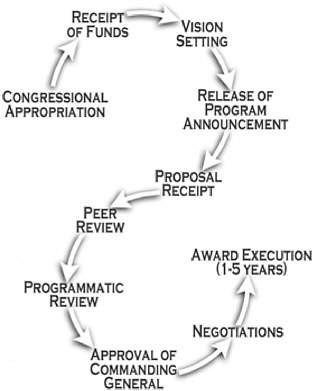
FIGURE 1-1 CDMRP budget and program planning and execution cycle.
SOURCE: CDMRP, 2003.
program at every level, and the year-to-year funding creates a sense of urgency to fund the best science as quickly as possible. In addition, although there is value in being part of the longer term budget process, the annual appropriations process means that earmarked money is less likely to be involved in a DOD funding rescission. Also, programs can make changes relatively quickly and responsively because they do not have long-term funding commitments. A disadvantage is that it is never known whether a particular program will exist from year to year. CDMRP has two years in which to obligate funds, however, and each award is fully funded up front, which enables the program to support projects of up to five years’ duration.
CDMRP Programs
Since its inception, CDMRP has managed 29 separate programs, 8 of which are considered core programs. Core programs have either received or have the
potential to receive multiple appropriations and are characterized by standing Integration Panels. The other programs managed by CDMRP have either received a one-time appropriation and/or are institutionally based. Each program emphasizes the specific needs of its research and advocacy communities. The core programs are described in Box 1-1.
|
BOX 1-1 The Breast Cancer Research Program (BCRP) is the second largest funder of extramural breast cancer research in the world, having managed approximately $1.52 billion in appropriations from FY 1992 to FY 2003. The awards supported through this program—which attempts to avoid duplicating NIH research funding approaches in this area through a complementary strategy—support innovative ideas, train future generations of scientists and clinicians, provide necessary research resources, bring bench research to the bedside, and emphasize the fostering of research in nontraditional areas for which pilot data may be lacking. Through FY 2002, BCRP has received more than 19,840 proposals and has made 3,671 awards. The Chronic Myelogenous Leukemia Research Program (CMLRP) was established in FY 2002 with a $5 million appropriation for research and was continued in FY 2003 and FY 2004 with congressional appropriations of $4.25 million each year. The National Prion Research Program (NPRP) was established by a one-time congressional appropriation of $42.5 million in FY 2002. The goal of NPRP is to develop a rapid, sensitive, and reproducible test for the detection of prions suitable for use both as an ante-mortem diagnostic test and a screening assay. In support of this goal, additional topics of interest include the prevention, transmission, and pathogenesis of transmissible spongiform encephalopathies as well as a better understanding of chronic wasting disease. A total of 38 awards have been made. The Neurofibromatosis Research Program (NFRP) has managed $110.3 million in congressional appropriations for FY 1996 through FY 2003. In the words of Brenda Duffy, President of Neurofibromatosis, Inc., at the April workshop, “Neurofibromatosis is a very small part of the DOD peer reviewed medical research program. The NF community cannot raise even a fraction of the money that goes to research as a result of the [CDMRP] program.” From FY 1996 to FY 2002, NFRP received 299 proposals, leading to 103 awards. The Ovarian Cancer Research Program (OCRP) appropriations for FY 1997 through FY 2003 have totaled $71.7 million. Since the program’s inception through |
CDMRP Program Planning and Execution
CDMRP has refined over the years a program execution and management strategy that allows it to adapt to the current needs of the research, clinical, and consumer communities. It uses a flexible seven-year execution and management cycle that spans all phases of program execution, from the development of a
|
September 2003, 575 proposals have been received and 63 awards have been made. Ann Kolker, Ovarian Cancer National Alliance, noted at the April workshop that this federal investment is significant given the scarcity of funds in the private sector, stating that, “given the very, very small amount of privately raised funds available for ovarian cancer research, it is frankly completely unrealistic to expect that charitable foundations and similar organizations could augment in any significant way the investment made by the Federal Government in ovarian cancer research.” The Peer Reviewed Medical Research Programs (PRMRPs) supports research on issues with direct relevance to military health. Appropriations for FY 1999 through FY 2002 have totaled $194.5 million. The program has built a research portfolio covering awards that span 32 topic areas relevant to military health. This program uses an advisory panel composed of representatives from the Army, Navy, Air Force, Marines, Department of Veterans Affairs, Office of the Assistant Secretary of Defense (Health Affairs), and U.S. Department of Health and Human Services to develop an investment strategy and conduct programmatic review. In the first four years of the program, 558 proposals have been received and 98 awards have been made. The Prostate Cancer Research Program (PCRP) was established in FY 1997. As a major funder of prostate cancer research, PCRP has been responsible for the management of $480 million in appropriations supporting innovative, multidisciplinary basic and clinical research relevant to prostate cancer. In addition, the program is committed to addressing the significant disparities in the incidence and mortality of prostate cancer that exist among different ethnic groups and has designed award mechanisms to stimulate research in these areas. For the first six years of this program, more than 3,400 proposals have been received, leading to 797 awards. The Tuberous Sclerosis Complex Research Program (TSCRP) was established by a $1 million appropriation in FY 2002 and was continued in FY 2003 and FY 2004 with congressional appropriations of $2 million and $3 million, respectively. TSCRP is supporting innovative research directed toward improving understanding of the role and function of proteins produced by the TSC1 and TSC2 tumor suppressor genes. |
vision through the awarding of research grants. Early in each fiscal year, after the congressional appropriation has been signed into law and funds have been received by the USAMRMC, each program’s Integration Panel meets to deliberate issues and concerns unique to that program and to establish a vision and investment strategy for the coming year. The investment strategy provides the framework and direction necessary to most effectively obligate each congressional appropriation, while avoiding unnecessary duplication with other funding agencies.
Award Mechanisms
A critical component of the CDMRP investment strategy is the development of specific award mechanisms that capture the current needs of both the research and advocacy communities. Separate announcements outlining the award mechanisms offered for each of the research programs managed by CDMRP are released each fiscal year. CDMRP has employed more than 40 types of award mechanisms, which fall into three categories: research; training and recruitment; and research resources. Awards are made in the form of grants, contracts, or cooperative agreements, with the research executed over one to five years, depending on the type of award mechanism.
Some of the research awards are designed to encourage innovative research by supporting ideas with little or none of the supporting data that the usual investigator-initiated applications must have. These awards have titles such as IDEA, Concept, or Exploratory and are equivalent to such NIH awards as R05 and R21 grants. The proportion of funding going to these awards to test novel ideas is high (more than half [57 percent] in FY 2002), although the percentage varies somewhat from program to program (see Figure 1-2).
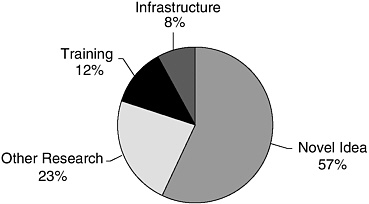
FIGURE 1-2 Distribution of extramural award funding among types of research support, FY 2002.
SOURCE: Calculated from CDMRP, 2003.
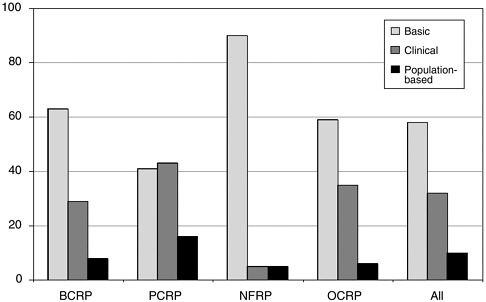
FIGURE 1-3 Distribution of extramural award funding among areas of research, FY 2002 (in percentages).
SOURCE: CDMRP, 2003.
Most of the mechanisms fund either basic or clinical research12 (e.g., IDEA Awards, New Investigator Awards, and Center Initiation Awards), while other mechanisms support clinical research exclusively (e.g., Clinical Translational Research Awards and Clinical Trial Awards). There also are training mechanisms (Pre- and Postdoctoral Fellowships and Career Development Awards), some of which provide support for investigators at Historically Black Colleges and Universities and Minority Institutions (e.g., Partnership Training Awards). More than half the funding goes to basic research, about a third to clinical research, and a tenth goes to population-based research. Again, the balance varies from program to program (see Figure 1-3). It should be noted that while the percentage of funding going to novel ideas (67 percent) and basic research (68
percent) is about the same, they are not congruent. IDEA and similar awards support clinical and translational research as well as basic research.
Each research program has a slightly different group of awards in its portfolio, with the mix depending on how other organizations and agencies are funding research in that area and on the amount of money Congress has appropriated to DOD for research on that disease. Although each award mechanism has different requirements, CDMRP works in all cases to promote innovative research by encouraging investigators to seek out novel, creative ideas, and solutions that will lead to disease-specific research in new directions.
The training and recruitment of new investigators, as well as the building of research resources in the form of centers of excellence, consortia, and imaging equipment, are major investments by CDMRP. For example, training awards are used to train the next generation of cancer investigators, while IDEA Awards, first instituted by BCRP in FY 1996, fund high-risk research projects that have the potential for large breakthroughs. Although IDEA Awards, which have now been adopted by other CDMRP programs, do not generally require the preliminary data that are typically required through other funding mechanisms, but proposals are still expected to be based on sound scientific principles. Thus, IDEA Awards are a critical source of funding for young investigators who lack research data accumulated over a longer career and for investigators who arrive at a novel approach for study.
Nonfederal Cost Sharing
According to CDMRP program announcements that invite applications for research funding, grantee institutions, whether commercial or nonprofit, are expected to “cost share” by providing the equipment needed to support proposed research. In rare cases that are individually negotiated, DOD will pay for 50 percent of the cost of the additional equipment needed to carry out a research project. DOD, however, takes into account the greater need for equipment at Historically Black Colleges and Universities and Minority Institutions.
Examples of Leveraging
There are many examples of CDMRP’s work in leveraging resources to facilitate and manage efforts to prevent and cure cancer and other diseases.
Matching Funds and Cost Sharing
In the early years of the prostate cancer program, matching funds were requested from other institutions. Currently, CDMRP has a group of contract specialists and grant managers who serve as technical advisors during award negotiations and work with the institutions to match costs in certain areas. Because
matching is not built into program announcements—except in the case of matching funds for equipment—a great deal of this activity takes place in the background. CDMRP also works to implement cost-sharing arrangements. IDEA Awards, for example, provide only $125,000 per year, which is not sufficient for conducting a clinical trial. Cost sharing helps make up the difference and takes place in the form of donations, which can be made for drugs, equipment, and some salaries. Cost sharing also may occur for research-related subject costs, such as blood tests and other components of a clinical trial or human research when these are not budgeted in CDMRP grants.
SBIR Program
CDMRP began participating in the SBIR program under DOD’s purview in FY 2000. After programmatic or peer review, if a significant gap remains in submissions or funding is insufficient for the accepted projects, CDMRP will offer an RFP for that area. For example, CDMRP has offered a SBIR RFP for ovarian cancer detection methods and has found this method of leveraging to be worthwhile, as it has recouped about 28 percent of the dollars spent. CDMRP has discussed the use of the SBIR model in some of its programs, particularly in looking at clinical and translation research in clinical trials, with the goal of providing more support in the drug development process, perhaps through Phase I or Phase II.
Examples of Partnerships
A central theme of almost all of the CDMRP award mechanisms is finding and funding the best and most innovative research aimed at eradicating disease. CDMRP programs look for untapped opportunities and underserved areas of research, and in the process they create partnerships and garner public trust. CDMRP investment strategies are re-evaluated annually, with success gauged in part by how many products are brought to the patient. CDMRP’s strategy of funding high-risk/high-payoff research means that many ideas do not pan out, but some have, leading quickly to Phase I clinical trials. In some cases, the clinical trials have involved partnerships with other organizations.
-
In 2004 CDMRP joined the Gynecological Cancer Foundation Allied Support Group, which includes eight major ovarian cancer funders and 14 advocacy organizations. CDMRP works to facilitate synergy among these organizations, which share the CDMRP goals of prevention, detection, and education among patients, laypersons, and physicians, particularly primary care physicians.
-
In an effort sponsored by BCRP, CDMRP is collaborating with the National Society of Nursing Oncologists to increase consumer participation in clinical trials, particularly those for gynecological diseases. The Era of Hope meeting
-
includes representatives from industry, public funding agencies, private funding agencies, and Historically Black Colleges and Universities and Minority Institutions, as well as survivors, scientists, clinicians, lawyers, and ethicists, to discuss not only DOD research findings, but also the use of innovation, invention, creativity, and collaboration to move findings from the bench to the bedside quickly.
-
CDMRP has co-funded grants with the National Cancer Institute, Office of the NIH Director, Department of Health and Human Services Office on Women’s Health, and California Breast Cancer Research Program.
-
PRMRP currently is holding a health research forum where program investigators and others instrumental in DOD-related research can discuss collaborations and strategies for progress.
-
NPRP has collaborated with numerous agencies throughout the country and will continue to hold meetings and form partnerships with others.
Communication with Other Funding Agencies and the Public
CDMRP’s use of the Common Scientific Outline (CSO) reflects its support for improved communication among funding agencies in the United States and abroad. This outline was initiated by NCI to categorize its funded research projects in a scientific and disease-related manner with the goal of reducing duplication and facilitating complementary research. CDMRP was invited to participate in this effort in 1997 and worked with NCI to develop a working model of the CSO. In subsequent years, additional cancer-funding organizations were asked to join the efforts of NCI and CDMRP in evaluating the utility of the CSO as a tool to facilitate the description of their respective portfolios and communication among funders. The CSO now includes 9 major cancer funders from the United States and 15 from the United Kingdom. In addition, CDMRP recently increased its effort to increase public awareness of its programs through advertising specific award mechanisms in national newspapers, distributing award information to consumer advocacy groups, and sponsoring funded investigators to attend scientific meetings.
More than 4,500 publications have resulted from CDMRP awards through FY 2001 and CDMRP staff has published articles and presented information at national scientific meetings. In addition, the CDMRP website disseminates up-to-date program information to the public and research community, and programs prepare and issue program announcements that provide details on individual award mechanisms, the application process, and requirements for submitting proposals.
THE COMMITTEE’S APPROACH TO ITS TASK
Over the course of its deliberations, the committee did not systematically evaluate CDMRP and its outcomes or compare its programs directly with those of NIH or any other federal funding agency. Not only was such an evaluation
outside its charge, but it also would have required far more time than was permitted. In general, the committee, based on testimony provided by advocacy groups and members of the scientific community at the April workshop, and committee members’ own knowledge of the program, supported the view that the CDRMP programs are of high quality and should be maintained. Rather than question those assumptions, it responded to its charge to identify potential sources of nonfederal funding, identify the range of mechanisms that could be used to leverage that nonfederal funding, and evaluate the impacts that the various potential mechanisms might have on the current program.
An important task of the committee was to define “leveraging.” In the federal grants world, the concept of leveraging retains the general idea that more is gained than invested (Feller, 1997:32-33). In one common type of leveraging, the agency is attempting to extend program funding to be able to make more research awards. This is typically achieved by imposing cost sharing or matching requirements on awardees. Another type of leveraging involves using federal awards to encourage other funders to collaborate in ways that achieve greater or faster results for society, for example, through the creation of synergies, critical mass, economies of scale or scope, or assembly of interdependent inputs (e.g., expertise, databases, research tools) required to tackle a problem. These benefits are the fundamental basis for research partnerships, alliances, and cooperative R&D ventures (Austin, 2000:8-10). The parties willingly collaborate to produce results that could not be reached if each acted alone—or at least not reached as quickly or easily. Companies, for example, might agree to jointly fund research that no one of them could afford to do alone and from which all could benefit.
The committee discussed another aspect of leveraging—that is, whether the nonfederal resources that are leveraged are new. New resources are those that otherwise would have been allocated to another purpose than research. Leveraging new resources increases the size of the funding pie, while leveraging funds that would have been spent on related research simply reallocates the resources included in the existing pie. Although a funding agency may claim it has increased funding for its research program, it may in fact have merely shifted funding from equivalent uses. Shifting funds in this way results in little or no net benefit, unless of course the recombined resources permit the attainment of an important result that could not otherwise be achieved.
The committee views leveraging that is intended to increase research results as preferable to leveraging that is intended simply to extend program funding, because the latter tends to shift funding without increasing social benefit. This position is consistent with recent federal policy changes regarding cost sharing, which will be discussed in Chapter 4. On the other hand, using CDMRP funds to induce collaborations that result in the whole becoming greater than the sum of the parts would clearly be beneficial and should be encouraged.
Although the committee was cognizant of the political pressures exerted on Congress to expand what is believed to be a successful program, it recognized
that funding decisions are congressional responsibilities and that citizens have the right to petition their government for federal support of such programs. The committee was not asked to comment on this dynamic, which was central to the creation and expansion of CDMRP. Although there is widespread support of CDMRP from advocacy groups and much of the scientific community, committee members are aware that some of their colleagues in the policy and scientific communities view the program with some degree of skepticism because of its location in the federal government outside NIH and the circumstances by which it was created.
During its deliberations the committee was cognizant of the changing funding environment for biomedical research. Since the creation of CDMRP, the NIH budget has doubled over a five-year period, and increasing demands have been placed on the overall DOD budget. Although these events were not central to its analysis, the committee considered them to be important trends when viewing CDMRP in a larger context.
In addition, the committee noted the importance of viewing the federal biomedical research investment in its entirety when making funding decisions, which means considering the breadth and depth of programs supported by other federal agencies, such as the Department of Veterans Affairs, NIH, and CDC. However, the committee was asked to focus more specifically on nonfederal sources of funding to augment CDMRP, rather than on similar federally funded programs where collaboration and leveraging also might be achieved.
One of the main tasks that faced the committee involved assessing whether and how changes made to bring private or nonfederal funds into the program would affect the positive contributions the program is currently making, a view that could be characterized as “first, do no harm.” This approach could also be characterized as an analysis of the tradeoffs between bringing more money into the program to fund a greater amount of research versus the cost of the changes that would be needed in order to gain those financial resources. Each potential source of alternative funding was assessed with these concerns in mind. The committee considered whether federal rules and regulations would have to be revised to address bioethical concerns or peer review biases that might result from collaborative funding mechanisms or from the infusion of nonfederal funds into the DOD programs.
Chapter 2 of this report focuses on sources of nonfederal funding, summarizing the statistics on funding of biomedical research by sector (industry, academia, other nonprofits, venture capital, and state government) and describing their different but complementary goals. This chapter reflects the factual basis for the committee’s findings and recommendations concerning sources of nonfederal funding.
Chapter 3 identifies and analyzes potential mechanisms for fund raising, based on a typology developed by the committee—that is, through partnerships, cost sharing or matching provisions, challenge grants, piggybacking arrange-
ments, jointly funded research subsidiaries, recoupment or return on investment provisions, or supplemental funding. This chapter describes the factual basis for the committee’s findings and recommendations concerning mechanisms of nonfederal funding.
Chapter 4 provides the committee’s analysis of the pros and cons of alternative mechanisms in terms of a common set of criteria or questions, including the three issues mentioned in the charge (federal rules and regulations that might have to be revised, bioethical concerns, and peer review bias) and additional criteria, such as impacts on the distinctive features of the program (consumer participation in priority setting and peer review, annual priority setting, program balance, and low overhead), and the added costs of fundraising.
Finally, Chapter 5 provides the committee’s conclusions and recommendations.
REFERENCES
Andejeski Y, Breslau ES, Hart E, Lythcott N, Alexander L, Rich I, Bisceglio I, Smith HS, Visco FM, and the US Army Medical Research and Materiel Command Fiscal Year 1995 Breast Cancer Research Program Integration Panel. 2002a. “Benefits and Drawbacks of Including Consumer Reviewers in the Scientific Merit Review of Breast Cancer Research.” Journal of Women’s Health & Gender-Based Medicine, 11(2):119-136.
Andejeski Y, Bisceglio IT, Dickerson K, Johnson JE, Robinson SI, Smith HS, Visco FM, Rich IM. 2002b. “Quantitative Impact of Including Consumers in the Scientific Review of Breast Cancer Research Proposals.” Journal of Women’s Health & Gender-Based Medicine, 11(4):379-388.
Austin JE. 2000. The Collaboration Challenge, How Nonprofits and Businesses Succeed through Strategic Alliances. San Francisco, CA: Jossey-Bass Publishers.
Casamayou MH. 2001. The Politics of Breast Cancer. Washington, DC: Georgetown University Press .
CDMRP (Congressionally Directed Medical Research Programs). 1999. FY99 CDMRP Annual Report. Fort Detrick, MD: CDMRP. [On-line.] Available: cdmrp.army.mil/annreports/1999annrep/ [accessed May 24, 2004].
CDMRP. 2003. Department of Defense Congressionally Directed Medical Research Programs: Annual Report, September 30, 2003. Fort Detrick, MD: Congressionally Directed Medical Research Programs. [On-line.] Available: cdmrp.army.mil/annreports/2003annrep/ [accessed May 24, 2004].
Feller I. 1997. Matching Fund and Cost-Sharing Experiences of U.S. Research Universities: Final Report to the National Science Foundation. Bethesda, MD: COSMOS Corporation.
Haran C. 2001. “Three’s Company: The Army, Women with Cancer and the Medical Community Have Joined Forces.” MAMM, 3:38-43,57. [On-line.] Available: cdmrp.army.mil/pubs/articles/threescompany.htm
IOM (Institute of Medicine). 1993. Strategies for Managing the Breast Cancer Research Program: A Report to the U.S. Medical Research and Development Command. Washington, DC: National Academy Press.
IOM. 1997. A Review of the Department of Defense’s Program for Breast Cancer Research. Washington, DC: National Academy Press.
Kaiser J. 1994. “Army Doles Out Its First $210 Million.” Science, 266(October 14):212.
Marshall E. 1993. “The Politics of Breast Cancer.” Science, 259(January 29):616-617.
Mervis J. 1993. “U.S. Army to Use Peer Review in Breast Cancer Programme.” Nature, 363(May 20):195.
Rich IM, Andejeski Y, Alciati MH, Bisceglio IC, Breslau ES, McCall L, Valadez A. 1998. “Perspective from the Department of Defense Breast Cancer Research Program.” Breast Disease, 10(5,6):33-45.
Schwartz SM, Slater DW, Heydrick FP, Woollett GR. 1995. “A Report of the AIBS Peer-review process for the U.S. Army’s 1994 Breast Cancer Initiative.” BioScience, 45(September):558-563.
Science. 1992. “Army Cancer Chief Gives NIH a Cold Shoulder.” Science, 258(December 4):1567.
Stabiner K. 1997. To Dance with the Devil: The New War on Breast Cancer: Politics, Power, and People. New York, NY: Delacorte Press.
U.S. Congress, House of Representatives, Committee on Appropriations. 2003. H. Rpt. 108-283 (September 24):336-337.
Visco F. 2004. Interview with Fran Visco by Michael McGeary and Rick Erdtmann, at the Institute of Medicine, Washington, DC, February 17.
Washington Fax. 1992. “Extramural Community Will Have the Opportunity to Participate in DOD $210 million Breast Cancer Research Program.” Washington DC, December 18. [On-line.] Available: www.washingtonfax.com/p1/1992/19921218.html.
Watson T. 1992. “Congress Supports Breast Cancer Research by Taking $210 Million from Military.” Nature, 359(October 8):471.
Young-McCaughan S, Rich IM, Lindsay GC, and Bertram KA 2002. “The Department of Defense Congressionally Directed Medical Research Program: Innovations in the Federal Funding of Biomedical Research.” Clinical Cancer Research, 8(4):957-962.
























