2
Sources of Funding for Biomedical Research
Biomedical research and development (R&D) is a large enterprise in the United States. In fiscal year (FY) 1999, the last year for which comprehensive survey data are available, federal spending on health R&D was $15.7 billion—21 percent of all federal expenditures on R&D that year (NIH, 2004a). Those figures are much larger in 2004, if only because the budget of the National Institutes of Health (NIH)—which supports roughly 83 percent of federally funded biomedical research—doubled between FY 1998 and FY 2003 and currently stands at more than $28.0 billion. The other major funders of biomedical research are the for-profit pharmaceutical, biotechnology, and medical equipment industries, which have outspent NIH in recent years. The Pharmaceutical Research and Manufacturers of America (PhRMA) reported that in 2003 its member companies (which include most of the large biotechnology companies as well as all the major pharmaceutical companies) spent $27.4 billion on R&D performed in the United States and another $5.8 billion on R&D performed abroad (PhRMA, 2004).
Other funders include venture capital funds; colleges and universities; nonprofit research institutions, foundations and other philanthropic and charitable organizations; and state governments. These sources of support are much smaller than the federal government and industry, and because of their small size, they often pursue a strategy of leveraging other resources to achieve their mission.
This chapter reviews the sources of funding that could potentially augment appropriated funds for the Congressionally Directed Medical Research Programs (CDMRP) of the Department of Defense (DOD) and briefly describes the types of R&D activities supported by these sources. Chapter 3 reviews examples of
collaborations between the federal and nonfederal funders of research described in this chapter.
THE FEDERAL GOVERNMENT
NIH last conducted a survey of federal support for health R&D in 1999 (NIH, 2004a).1 In that year, total federal spending on health R&D was approximately $15.7 billion, with NIH the top funder at $13.0 billion (83 percent). Other agencies in the Department of Health and Human Services (DHHS), including the Centers for Disease Control and Prevention (CDC) and the Food and Drug Administration, spent another $758.0 million. Non-DHHS federal agencies—primarily DOD, the Department of Veterans Affairs, and the Department of Energy—spent an additional $1.8 billion. Table 2-1 lists the agencies and their R&D budget obligations for FY 1997 through FY 1999.
Most of the federal funding of health R&D in 1999 was performed by nonfederal research institutions through extramural grants, cooperative agreements, and contracts, with extramural performers accounting for 76 percent of federal expenditures on health R&D. Of the total $11.9 billion spent on extramural health R&D in 1999, institutions of higher education received $8.3 billion, other nonprofits $2.3 billion, industry $0.9 billion, and state and local governments $141.0 million (NIH, 2004b). The NIH survey does not break out the amounts of these funds that go to basic versus applied research.
In 1999, 61 percent of federal expenditures in these fields were classified as basic research and 39 percent as applied research (the National Science Foundation [NSF] survey does not break out development expenditures by field). The emphasis on basic research was primarily due to NIH, which accounted for nearly 90 percent of all basic research funding in the three fields. NIH is the single largest funder of biomedical and behavioral research, with nearly two-thirds of its funding of research projects and centers supporting basic research. Most of the other agencies are mission oriented and place a greater emphasis on applied research and development. As a group, they spent 37 percent of their funding in these fields on basic research. DOD spends nearly three-quarters of its funding of biomedical and behavioral research on applied projects; basic research accounts for 26 percent.
The picture has no doubt changed since 1999, the first year of the five-year doubling of the NIH budget. For FY 2004, the NIH budget is $28.0 billion,2 and if the other agencies increased funding for health R&D by just 10 percent overall
|
1 |
A survey of federal agency funding of health R&D for the years FY 2000 through FY 2002 (actual) and FY 2003 through FY 2004 (estimated) was in the field while this report was being written. The QRC Division of Macro International Inc., is conducting the survey for NIH. |
|
2 |
See www4.od.nih.gov/officeofbudget/FY05pubs/MechanismTotal.pdf. |
TABLE 2-1 Federal Obligations for Health R&D by Source, FY 1997-FY 1999 (in millions of dollars)
|
|
1997 |
1998 |
1999 |
|
Department of Health and Human Services |
12,676.0 |
13,575.8 |
13,812.9 |
|
National Institutes of Health |
11,993.0 |
12,880.2 |
13,005.7 |
|
Centers for Disease Control and Prevention |
323.0 |
344.0 |
433.3 |
|
Food and Drug Administration |
158.6 |
142.9 |
130.3 |
|
Health Resources and Services Administration |
13.1 |
11.0 |
11.6 |
|
Office of the Assistant Secretary for Health/ Agency for Healthcare Research and Quality |
144.0 |
146.5 |
182.3 |
|
Other Health and Human Services Agencies |
44.3 |
51.2 |
49.7 |
|
Other Departments and Agencies |
2,404.8 |
2,712.6 |
1,845.7 |
|
Department of Agriculture |
127.2 |
140.1 |
148.7 |
|
Department of Defense* |
1,094.2 |
1,251.7 |
387.4 |
|
Department of Education |
24.1 |
29.1 |
0 |
|
Department of Energy |
318.8 |
367.6 |
372.2 |
|
Environmental Protection Agency |
123.5 |
155.5 |
141.6 |
|
Agency for International Development |
32.1 |
72.3 |
70.9 |
|
National Aeronautics and Space Administration |
179.4 |
174.9 |
186.4 |
|
National Science Foundation |
81.1 |
76.0 |
81.1 |
|
Department of Veterans Affairs |
259.9 |
294.4 |
294.4 |
|
All other departments and agencies |
164.6 |
151.0 |
162.9 |
|
Total |
15,080.8 |
16,288.4 |
15,658.6 |
|
*In FY 1999 DOD funds decreased substantially from estimated amounts because of reallocations. |
|||
since 1999, the total federal investment in health R&D would be nearly $31.0 billion. As already noted, most of this supports extramural awards to institutions of higher education. A small portion is spent in federal, industrial, and state laboratories.
Funding Through Foundations Established for Federal Agencies
One mechanism for expanding the federal government’s capacity to fund research is through the creation of foundations, such as the Foundation for the National Institutes of Health (FNIH) or the CDC Foundation (Box 2-1). These independent nonprofit enterprises were created to secure private funding for their agency’s activities and facilitate programmatic partnerships between their agency and other organizations—corporations, foundations, other nonprofits, and other government agencies—in efforts to improve public health and safety. They have supported initiatives such as fellowship programs, training, infrastructure, and basic and applied research.
|
BOX 2-1 The CDC Foundation was established in 1992 by Congress to forge partnerships with CDC to boost the agency’s programs. As an independent nonprofit organization, the foundation can accept funding and create programs that help donors and CDC scientists achieve common goals. It can find funding partners, negotiate deals, hire people, manage program budgets, identify experts, and report to donors. In 2002-2003 the foundation had revenues of $17.1 million and expenses of $10.2 million, of which $8.1 million were expended through cost-reimbursement agreements for programs. On its website, the foundation currently lists 37 corporations and 23 foundations that were supporting programs initiated as of July 2000 or that are currently active. Listed are 22 Global Health Programs, including: Asian Rotavirus Surveillance Program – Phase II. Partners: GlaxoSmithKline, PATH (Program for Appropriate Technology in Health) Development of Rapid Assessment Methods and Tools for Displaced Persons. Partner: Andrew W. Mellon Foundation. Joint Global Field Epidemiology and Laboratory Training Program–Kenya. Partner: Ellison Medical Foundation. Lilly International Laboratory Fellowships. Partner: Eli Lilly and Company. Violent Injury Surveillance and Prevention Program. Partners: John D. and Catherine T. MacArthur Foundation, World Health Organization. Also listed were eight Promoting Healthy Lifestyles programs, such as: Avon-CDC Foundation Mobile Access Program. Partner: Avon Foundation Price Fellowships for HIV Prevention Leadership. Partner: Price Foundation. Promoting Better Health for Young People Through Physical Activity and Sports. Partner: MetLife Foundation. The 22 Research and Education Programs included: Antimicrobial Resistant Bacteria Educational Program. Partners: AB Biodisk; Abbott Laboratories; Becton Dickinson and Company; bioMérieux, Inc.; Dade Microscan, Inc.; Merck & Co., Inc.; Ortho-McNeil Pharmaceutical, Inc.; Roche; GlaxoSmithKline. Applied Epidemiology Training Program for Medical Students. Partner: Pfizer Inc. Estimation of Prevalence of Erectile Dysfunction in the U.S. Partners: National Foundation for Sexual Health Medicine, Inc.; Pfizer Inc. |
PRIVATE INDUSTRY
Several sources of information are available on industry investment in health and medical research. PhRMA, which includes most of the large biotechnology companies as well as all of the major pharmaceutical companies, annually surveys its member companies on this topic. In 2003, member companies reported that they spent $27.4 billion on R&D performed in the United States and another $5.8 billion on R&D performed abroad (PhRMA, 2004). The total investment of $33.2 billion equaled 17.7 percent of the industry’s domestic sales on R&D—a higher percentage than was reported for any other U.S. industry.3 Like the pharmaceutical industry, the U.S. biotechnology industry is very research-intensive, spending $15.7 billion on research and development in 2001.4
The medical equipment and supplies industry and the health care services industry also invest their own funds in R&D ($3.7 billion and $0.5 billion, respectively, in 2000) (NSF, 2003a:Table A-8). Some of this is no doubt spent in areas of CDMRP programs, especially in cancer. In 2003, for example, 181 biotechnology and pharmaceutical companies had nearly 400 medicines for cancer in clinical trials, including 47 for breast cancer, 27 for ovarian cancer, 38 for leukemia, and 44 for prostate cancer (PhRMA, 2003). The only figure for spending by PhRMA members on cancer research is for 1997, when companies reported spending $1.4 billion on cancer R&D, approximately 7.4 percent of total pharmaceutical R&D (PhRMA, 1997). If cancer research continued to constitute 7.4 percent of pharmaceutical R&D (and it was probably more), it would have totaled nearly $2.5 billion in 2003.
The bulk of industrial R&D is conducted in-house on product development. In 2000, for example, the pharmaceuticals and medicines industry and medical equipment and supplies industry spent 84 percent of their R&D budgets in their own laboratories (NSF, 2003a:Table A-10), and most of the rest was contracted to other commercial firms. However, industry also lets contracts and awards grants for R&D at universities. Universities reported receiving $2.2 billion from all industry sectors in 2002 (it is not possible to break out specific industries, such as pharmaceuticals), which together constituted 6 percent of university expenditures on R&D (NSF, 2004b:Table 1).
The advocacy organization Research!America (R!A) generates an estimate of industry funding of health research based on data from PhRMA and the Biotechnology Industry Organization (BIO), adjusted to eliminate the overlap between the organizations’ memberships. R!A estimates that pharmaceutical and
|
3 |
|
|
4 |
See www.bio.org/speeches/pubs/er/statistics.asp. This total overlaps with the total reported by PhRMA, because of double counting of larger biotechnology companies in the two categories of biotechnology and pharmaceutical companies. |
biotechnology industries’ investments in R&D totaled $45.9 billion in 2001 and $49.9 billion in 2002 (R!A, 2003, 2004).
NSF conducts an annual survey of industrial R&D that reports data by type of industry. In 2000, the pharmaceuticals and medicines industry reported company-funded R&D expenditures of $12.9 billion, the medical equipment and supplies industry $3.8 billion, and health care services $0.6 billion, for a total of $17.2 billion (NSF, 2003a). Of this, approximately $2.7 billion was contracted to outside organizations, including universities. These numbers changed rather dramatically in 2001, when company-funded R&D expenditures totaled $10.1 billion for the pharmaceuticals and medicines industry (a drop of 21 percent), $5.9 billion for the medical equipment and supplies industry (up 57 percent), and $1.1 billion for the health care services industry (up 96 percent) (NSF, 2004a).
As would be expected, industry provides little funding directly to the federal government—approximately $16.1 million through FNIH and additional funding through the CDC Foundation.5 Industry also provides funding as cost sharing on grants and contracts to universities (the latter amount is unknown, although NSF reported making awards that included cost sharing totaling $534.0 million in FY 2001 (NSF, 2002). Industry collaboration with government is far more likely to occur when a company sees an opportunity to leverage the federal investment in long-term basic research by completing the applied work and development activities necessary to take a product to market.
Although industry invests heavily in biomedical research, it focuses mostly on short-term research and development projects with commercial promise, such as drug development and the creation of research tools and databases that can be used in applied research and development. Industry does, however, invest in some basic research. Most observers estimate that approximately 10 percent of biopharmaceutical industry investment is in basic research, which would amount to from $3 billion to $4 billion per year. If industry R&D investment in cancer research is $2.5 billion annually, 10 percent would amount to $250 million. However, hard, documented figures are not available.
VENTURE CAPITAL
Most venture capital is invested in start-up companies that have the potential to become economically successful—that is, companies that represent an opportunity for a high rate of return within five to seven years. It is an important source of equity for new, fast-growing companies (NVCA, 2004a). According to the MoneyTree survey for the first quarter of 2004, the life sciences sector—which includes companies in biotechnology and medical devices—continued to domi-
TABLE 2-2 Expenditures for Biomedical and Behavioral R&D at Universities and Colleges, FY 2001 (dollars in thousands)
|
Field |
Nonfederal |
Federal |
Total |
|
Biological Sciences |
2,071,446 |
3,872,150 |
5,943,596 |
|
Medical Sciences |
3,928,069 |
6,248,659 |
10,176,728 |
|
Psychology |
183,762 |
398,103 |
581,865 |
|
Total |
6,183,277 |
10,518,912 |
16,702,189 |
|
SOURCE: NSF, 2003b. |
|||
nate other industries in winning venture capital (NVCA, 2004b). During the quarter, 27 percent (or $1.3 billion) of total venture capital was invested in 71 biotechnology companies and 51 medical device companies. Proportionately, biotechnology and medical devices accounted for 20 percent and 7 percent, respectively, of all venture capital investments. In general, the goal of venture capital firms is to assist companies in the development of new products or services, not to invest in basic research.
ACADEMIA
NSF also conducts an annual survey of R&D expenditures by colleges and universities that includes data on the source of R&D funds in aggregate, not by field. In FY 2001, the most recent year for which data are available, colleges and universities spent $32.7 billion on R&D, about half of which ($16.7 billion) was spent on the biologic and medical sciences and psychology (NSF, 2003b). Table 2-2 separates these figures by field and by whether the funding was provided by federal or nonfederal sources. Overall, the federal government provided 59 percent ($19.2 billion) of R&D funding, and industry provided 7 percent ($2.2 billion). Colleges and universities reported funding 20 percent ($6.6 billion) from institutional funds. Institutional funds are:
funds, including related indirect costs, that an institution spends for R&D activities from the following unrestricted sources: general-purpose state or local government appropriations; general-purpose awards from industry, foundations, or other outside sources; tuition and fees; endowment income; gifts; and other institutional funds. (NSF, 2003b:266)
The rest came from state and local governments (7 percent) and other sources (7 percent).
A preliminary NSF report relying on FY 2002 data does not detail expenditures by field, but shows that the distribution of funding sources was about the same as in previous years, including 20 percent ($7.1 billion) from institutional sources (NSF, 2004b). Other data sources report that the federal government
accounted for 62 percent of the support awarded to U.S. universities, hospitals, and research institutes between 1991 and 2002 (AUTM, 2002). It is not known, however, how much of this funding was already cost shared or how much more might have been expended from an institution’s own funds—such as tuition and fees or endowment income—if required to increase cost sharing.
PHILANTHROPIC ORGANIZATIONS
It is estimated that philanthropic giving totaled about $240 billion in 2003, an increase over 2002 of 0.5 percent after inflation. Individuals by far contributed the largest portion of charitable dollars ($179 billion), most of which went to religious institutions. Corporations donated $13 billion to charity, foundations donated $26 billion, and charitable bequests totaled $22 billion (AAFRC, 2004).
Charitable giving from individuals, companies, foundations, and bequests has increased substantially (94.1 percent) since 1995, but most of this increase occurred before 2000. The compound rate of growth from 1995 to 2000 was 13.0 percent per year; from 2000 to 2003 it was 1.8 percent. Corporate giving has grown at a slower rate than the overall average. Among foundations, large independent (versus corporate, operating, and community) foundations do most of the giving, in 2001 contributing about $23.7 billion, or about 80 percent, of total foundation dollars.
Foundations and public charities such as the American Cancer Society have limited resources compared with the federal government—with foundations contributing $1 billion to $2 billion per year to health research—and most focus on funding activities that are not well supported by the federal government, such as public health, or activities that leverage federal funding, such as grants for exploratory research and new investigators. A few foundations, such as the Juvenile Diabetes Research Foundation International (JDRF), collaborate with federal agencies on projects of mutual interest, but the amounts of money they provide are modest relative to federal funding.
The amount of money provided by private foundations for national health research and development remained fairly constant from 1987 to 1996, but represented a small portion of overall funding. Foundations, voluntary agencies, and the Howard Hughes Medical Institute (HHMI) provided only about 9 percent of the total funding for academic health centers in 1997.
Although many boards of such private organizations will not donate money to a government agency for medical research, they will act as a partner in such efforts. The Burroughs Wellcome Fund, for example, partnered with DOD, the National Institute of Allergy and Infectious Diseases, and the Wellcome Trust on the malaria genome sequencing project. In this case, in addition to providing the dollars to sequence the genome, Burroughs Wellcome provided the top-down steering to bring the research community together on a recurring basis, which helped to provide the community with the tools it needed to accomplish the
effort. To be successful in this kind of endeavor—which leverages not only dollars but also expertise and organizational focus—at least one organization has to be the engine that moves the effort forward.
Health foundations that have been created from the proceeds of the sale of nonprofits that are going for-profit are a relatively new source of potential funding, with assets of about $8.0 billion and making grants totaling about $340.0 annually, generally in support of underserved populations. Although their grant making for the most part has not included medical research, these groups potentially could allocate a significant portion of their dollars to such efforts, particularly as related to underserved populations.
HHMI, with assets of close to $11.3 billion in 2003, is not classified as a foundation (it is a medical research organization), but it has a large cadre of elite investigators (who are employees not grantees of the institute), as well as a new research facility, and it provides grants to support life sciences education and research opportunities for K-12 and undergraduate students.
The Foundation Center collects data on patterns of giving by these organizations, with the latest figures included in the 2004 edition of its annual report, Foundation Giving Trends (Foundation Center, 2004). In 2002, total grant commitments by participating foundations were $15.9 billion. The largest number of grants and grant dollars were in education and health (Figure 2-1), with “health” defined in the report as including grants to hospitals for medical care.
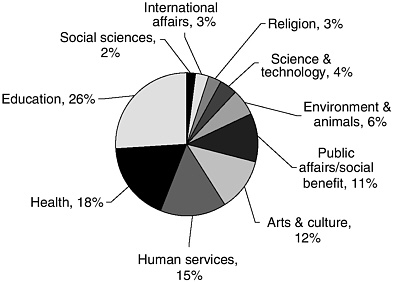
FIGURE 2-1 Foundation grant dollars by purpose, 2002. SOURCE: Foundation Center, 2004. Based on a sample of 1,005 larger foundations.
TABLE 2.3 The 12 Largest VHAs in Research Grant Expenditures, FY 2002
|
Association |
Grant expenditures (million $) |
Percentage of agency budget |
|
American Heart Association |
126.2 |
24 |
|
Juvenile Diabetes Research Foundation |
107.9 |
64 |
|
American Cancer Society |
95.7 |
11 |
|
Cystic Fibrosis Foundation |
51.6 |
36 |
|
The Leukemia & Lymphoma Society |
37.6 |
24 |
|
Multiple Sclerosis Foundation |
32.1 |
18 |
|
American Diabetes Association |
31.5 |
17 |
|
March of Dimes |
27.7 |
13 |
|
Muscular Dystrophy Association |
26.4 |
17 |
|
Arthritis Foundation |
25.7 |
20 |
|
Alzheimer’s Association |
17.5 |
— |
|
American Lung Association |
11.6 |
7 |
|
Total |
591.5 |
— |
|
SOURCE: Lichtman et al., 2004, Table 1. |
||
Most public charities that support medical research are voluntary health agencies (VHAs), which raise a substantial portion of dollars from the public, but may also seek foundation support. VHAs allocate substantial dollars for research; for example, the 12 largest health VHAs spent nearly $600.0 million on research in FY 2002 (Table 2-3). Overall, more than $500 million per year is contributed by some of the larger voluntary organizations to the medical research arena. Many of these VHAs focus their research efforts on a specific disease.
The National Health Council, a nonprofit whose mission is to advance the voluntary health movement and promote the importance of medical research, has more than 45 volunteer agencies as members,6 and in FY 2000 spent $506.9 million on research, ranging from $4,000 (Myositis Association of America) to $133,562,000 (American Heart Association) annually.
The amount of individual giving going to support medical research is not known, although press releases announcing some large awards and medical school annual reports indicate that a number of gifts are directed annually toward the support of health research—and the amount could be substantial (Bond et al., 1999).7 It must be remembered, though, that many of these are one-time gifts rather than continuing sources of collaboration and support.
There is no way to know how much collaboration exists between foundations and public charities and federal programs that fund biomedical research. How-
ever, such collaborations do occur, some of which are described in Chapters 3 and 4 and in Appendix A. (Box 2-2 illustrates an example of a foundation that regularly co-funds or coordinates funding of research projects research with NIH.)
Testimony at the committee’s April 2004 workshop indicated that philanthropic organizations are changing the focus of their research investment from basic research to clinical, translational, and behavioral research. Although the contribution of such organizations is small compared to federal and industry support, these organizations are always looking for opportunities to leverage their modest dollars, and these dollars are important because they can provide critical “venture capital.”8 Many efforts focus on exploratory research and new investigators, enabling these groups to gather the data needed to support an application for an R01 or similar grant from NIH. Philanthropic organizations often can move quickly to fill a gap, but sometimes they are limited in their ability to provide sustained research support. In addition, they must ensure that research investments are consistent with the charter or mandate of the organization, which might restrict funds geographically or institutionally (Bond et al., 1999).
STATE GOVERNMENTS
State funding for biomedical research has been on the rise since the late 1970s, although most states have had to dramatically cut budgets in recent years as fiscal constraints have worsened (McNichol and Harris, 2004). The National Conference of State Legislatures estimates that since FY 2002 the states have had to close a cumulative budget gap of more than $235 billion (NCSL, 2004:1). Higher education, a recipient of state funding for research and research infrastructure, has been especially hard hit, resulting in tuition increases in several states.
The states support R&D through various mechanisms, including direct appropriations to universities, R&D tax credits, the establishment of endowment funds, and the formation of nonprofit corporations. Although there is no complete database of state expenditures for health research, some studies of spending have been conducted by national organizations of both state support of health research or biotechnology and state use of tobacco settlement funds for research. Specific examples, such as Ohio’s Center for Stem Cell and Regenerative Medicine and the North Carolina Biotechnology Center, provide illustrations of the types of initiatives more likely to be undertaken and funded by states.
Most states also have economic development programs interested in funding medical research and biotechnology, with money that would not otherwise be spent on medical research (Battelle/SSTI, 2004). But states, like industry, are generally most interested in research that will have immediate commercial appli-
|
BOX 2-2 JDRF is the leading charitable funder and advocate of type 1 diabetes research worldwide. To date, JDRF has awarded more than $680 million for diabetes research. The foundation seeks to find a cure for diabetes and its complications by supporting multidisciplinary programs that bring together diabetes researchers from numerous institutions, private and public, and from diverse disciplines. JDRF also seeks to leverage its research impact by partnering with and stimulating increased research spending on the part of public and private medical organizations and other entities throughout the world. In FY 2003, JDRF provided $80.0 million, funding 500 centers, grants, and fellowships in 19 countries. Thirty-eight percent of its funding is spent outside the United States, often in partnerships with other governments. JDRF funding and leadership have been associated with most major scientific breakthroughs in type 1 diabetes research to date. In its collaborations with the federal government, JDRF has participated in numerous partnerships with NIH that have added value on both sides of the table, with NIH providing most of the funding and generally operating the programs. JDRF works to maximize the impact of its funding by complementing, not duplicating, NIH efforts. This year begins the second five-year term of earmarked money—$150.0 million per year for five years over and above the normal NIH appropriation for type 1 diabetes—which gives JDRF leverage in working with the agency on how the money is spent. One specific partnership with NIH, TrialNet, involves a consortium approach using cooperative agreements to conduct clinical trials for type 1 diabetes related to new onset, prevention of the disease, or amelioration of further damage. For this project, JDRF is providing funding to include four international sites in the consortium, an example of how foundation partnering can make a significant difference. Another JDRF partnership involves DOD, the National Aeronautics and Space |
cations, as this helps to build the state’s biotechnology industry and thus increase the number of jobs. States also are investing in the biomedical R&D capacity of their universities and other research institutions to enable them to compete more effectively for support from federal agencies—especially NIH—and industry and foundations. Most state economic development funds aim to leverage multiple federal dollars for each state dollar, not the other way around.
A few states, such as California and New Jersey, have peer reviewed research grant programs, but these are small relative to federal programs, and they concentrate on leveraging federal funds by supporting exploratory research projects and new investigators to help state medical researchers win R01 (traditional individual investigator-initiated) grants from NIH.
State revenues can be affected by the ups and downs of the business cycle, because they must balance their budgets each year. This makes it more difficult
|
Administration, and NIH working together to develop technologies for metabolic monitoring through the Technologies for Metabolic Monitoring and Julia Weaver Fund Research Program, managed by the U.S. Army Medical Research and Materiel Command. This year JDRF will spend about $3.5 million to fund this research, which is targeted at developing a means to understand, predict, and closely monitor metabolic products, with an emphasis on monitoring blood glucose levels. The scientific interests of DOD and JDRF come together in this effort, with DOD seeking a remote, noninvasive way to detect metabolic arrangements, while JDRF would like to have a glucose monitor. JDRF also partners with the Defense Advanced Research Projects Agency, which has a mutual interest in research on tissue regeneration and repair. JDRF plays an important and unique role in setting the global direction of diabetes research resources to ensure that they are used as effectively as possible. JDRF’s many international partnerships fund specific aspects of type 1 diabetes, while also bringing to the table the resources of the governments of each country involved, including their research councils. The international partnerships allow for effective leveraging of resources, as occurred, for example, when JDRF partnered with the Swedish Medical Research Council and the Knut & Alice Wallennberg Foundation to support a five-year program involving research networks in type 1 diabetes. Another project involves the Swedish Research Council, the Swedish Diabetes Association, and JDRF working together to support stem cell research. JDRF’s experience with these kinds of partnerships, coupled with its desire to promote stem cell research beyond the NIH guidelines, led to early talks with the Medical Research Council of the United Kingdom. In January 2002, the Council sponsored the formation of the International Stem Cell Forum. Currently, the Forum consists of 18 international funders of research with an interest in working together to further stem cell research. JDRF is the only nongovernment partner in this collaboration, one that provides an example of what can be accomplished through partnerships that provide not only money, but also scientific interest. |
for them to provide long-term support of research programs. Nevertheless, states do fund medical research and likely would be interested in partnering with federal programs such as CDMRP on projects of mutual interest. As mentioned previously, some states have established programs to support biomedical research, or all areas of research, with dedicated funding from the tobacco settlement or other sources of regular revenues, such as state tobacco taxes.
State Support for Health Research and Development
In 1998, Battelle and the State Science and Technology Institute (SSTI) conducted a survey of state funding of R&D for NSF. The study found that states spent $3.0 billion on R&D and R&D plants in FY 1995—$2.7 billion from their own sources and the rest from federal programs, industry, and foundations
(Battelle/SSTI, 1998). The states, which were asked to report spending by functional category and by scientific field, identified $278.0 million as health R&D, but $1.0 billion as biological, medical, and psychological research. Evidently, some states included funding of state university research in a separate category called “science and technology base.”
In 2001, McGeary and Smith conducted a study of state funding of health research for the Lasker Foundation, finding that data sources were scattered and incomplete (McGeary and Smith, 2001). But by extrapolating the 1995 survey data and adding tobacco settlement funding spent on research as well as other state programs established since 1995, the authors estimated that states were spending about $2.0 billion in 2001 on health research. Since 2001 tobacco settlement revenues have increased, but the fiscal crisis in many states has caused them to shift the revenue from programs to debt reduction. Much health research funding occurs through state colleges and universities and their medical centers, but this funding is probably already highly leveraged through cost sharing on federal grants and contracts.
According to a 2004 survey of state bioscience initiatives conducted for the Biotechnology Industry Organization, 40 states are investing in the development of bioscience and technology as a means of economic development and job creation (Battelle/SSTI, 2004:28). In general, the strategies are to encourage biotechnology firms to locate in the states, enhance the biomedical research capacity of state universities and other research institutions, foster greater industryuniversity interaction, build modern facilities with wet-lab space and specialized equipment to attract new firms, and create publicly supported commercialization, seed, and venture capital funds that can invest in bioscience-related companies (see Box 2-3 for an example).
Some states use state funds to support research projects that can leverage funding from merit-based federal science programs that need nonfederal investment in facilities or equipment to be competitive or that require nonfederal cost sharing. Firms can sometimes be induced to fund R&D in an area of mutual interest if the state will share the costs, and local foundations sometimes will contribute funds if they are interested in economic development as a means of expanding economic opportunities. Currently, 23 states have programs of biomedical research grants or centers of excellence, or both; 33 provide funding for biomedical research facilities; 9 have biomedical faculty development programs; 32 have university-related research parks (12 of them specifically for biotechnology enterprises); 33 offer R&D tax credits; and 17 have university-industry matching grant programs (Battelle/SSTI, 2004: Table 6).
State Expenditures of Tobacco Settlement Revenue
Many states planned on using funds from the 1998 Tobacco Master Settlement, when it was signed, for research purposes, particularly those addressing
|
BOX 2-3 In 1981, North Carolina’s General Assembly established the North Carolina Biotechnology Center (NCBC) as a private, nonprofit (close nonprofit) corporation that would provide economic benefit to the state through supporting biotechnology R&D and commercialization. NCBC works in partnership with the North Carolina Department of Commerce, the North Carolina Biosciences Organization, the Council for Entrepreneurial Development, the University of North Carolina system, the Community College system, private universities, the Small Business and Technology Development Center, Golden LEAF (tobacco settlement fund program), Chambers of Commerce, and many other groups. For the past 15 years, NCBC has received annually an average of $7.0 million from the state through bipartisan legislative support (NCBC, 1999). This funding comes mainly from state appropriations, but also from federal and private sector funds. The pharmaceutical partners helped build the center where NCBC is based. NCBC’s total annual budget is approximately $10.0 million, which is spent on research, workforce development and education programs, venture capital investment, and other activities (Alexandre, 2004). The amount spent on research depends on how much is allocated within the given year. For 2004, grant programs total approximately $3 to $4 million and are invested into various research programs. NCBC funds mostly transitional and applied research, rather than basic research, and has three core programs: Science and Technology Development, Business and Technology Development, and Education and Training. Other state-funded initiatives include the North Carolina Genomics and Bioinformatics Consortium, which promotes genomics, proteomics, and bioinformatics. |
tobacco-related illnesses. According to the report of the National Governors Association’s Center for Best Practices on tobacco settlement spending, 17 states (of the 46 that reported) allocated some portion of funding specifically to biomedical and health research, which includes research projects on cancer and tobacco-related diseases (NGA, 2001:3).
Since 2001, there has been a steady decline in the percentage of tobacco settlement funds used by states for health (this category includes medical research but not capital expenditures for research facilities). In 2003, 24 percent of the funding went to health (most of the funding went for deficit reduction), but only 17 percent is estimated for 2004. The states allocated about 36 percent of tobacco settlement funds to reduce budget shortfalls and are expected to allocate about 54 percent in 2004 (U.S. GAO, 2004). Thirteen states allocated tobacco settlement funds for health research in 2003—Arkansas, Florida, Georgia, Illinois, Louisiana, Maryland, Michigan, Nebraska, New Mexico, Ohio (see Box 2-4), Pennsylvania,
|
BOX 2-4 In 2003, Ohio Governor Bob Taft awarded a capital grant (through the state’s Wright Centers of Innovation Program) for $10.8 million to help build the Center for Stem Cell and Regenerative Medicine in Cleveland and another $8.6 million to finance research there. The center is a research collaboration among Case Western Reserve University, Cleveland Clinic, University Hospitals of Cleveland, Ohio State University, and seven industry partners (including Cleveland-based Athersys, Inc.). Funding for the center’s research comes from Ohio’s Biomedical Research and Technology Transfer Fund, which was created by the state’s share of tobacco settlement. The center is part of Ohio’s Third Frontier Project, a state initiative formed to create high-technology jobs by supporting research. |
Texas, and Utah. The percentages each state allocated for health research ranged from 0.2 percent in Texas to 28.7 percent in Maryland. The total, $278.9 million, was 2.3 percent of the $12.2 billion received by the states from the tobacco settlement in 2003.
SUMMARY
Several sources of nonfederal funding for medical R&D exist in the United States. The largest contributor is private industry, with the pharmaceutical industry alone providing over $32.0 billion in 2003. Most of the industrial investment is at the development end of the R&D spectrum; thus, interest in CDMRP programs would likely be focused primarily on ideas or projects that are ready for scale-up and production. Likewise, U.S. venture capital markets are more likely to seek opportunities where the likelihood of near-term payoff is greater. Although academia provides some funds and in-kind support for research, public universities in particular have been struggling with budget reductions caused by state fiscal constraints.
Philanthropic organizations, foundations, and public charities also make an important, but smaller, contribution to health research of approximately $1.0 billion annually. These groups tend to find ways to leverage the much larger federal and industrial investment and frequently support focused areas of research specific to one disease or set of disorders. Finally, states provide roughly $2.0 billion annually in support of bioscience-related R&D activities. Much of this investment is focused on employment and economic development.
REFERENCES
AAFRC (American Association of Fundraising Counsel). 2004. Americans Give $241 Billion to Charity in 2003 [press release]. [On-line.] Available: aafrc.org/press_releases/trustreleases/americansgive.html [accessed July 18, 2004].
Alexandre LM. 2004. Presentation by President and CEO of the North Carolina Biotechnology Center at workshop held by the Committee on Alternative Funding Strategies for DOD’s Peer Reviewed Medical Research Programs. National Academies, Washington, DC, April 27.
AUTM (Association of University Technology Managers). 2002. AUTM Licensing Survey: FY 2002: A Survey Summary of Technology Licensing (and Related) Performance for U.S. and Canadian Academic and Nonprofit Institutions, and Patent Management and Investment Firms. Northbrook, IL: AUTM [On-line.] Available: www.autm.net/index_ie.html [accessed July 18, 2004].
Battelle/SSTI (Science and Technology Institute). 1998. Survey of State Research and Development Expenditures, Fiscal Year 1995. Westerville, OH: Science and Technology Institute.
Battelle/SSTI. 2004. Laboratories of Innovation: State Bioscience Initiatives 2004. Prepared for BIO—Biotechnology Industry Organization, Washington, D.C. [On-line.] Available: www.bio.org/speeches/pubs/battelle2004.pdf [accessed June 14, 2004].
Bond EC, Peck MG, Scott M. 1999. The Future of Philanthropic Support for Medical/Health Research. [On-line.] Available: www.aaas.org/spp/dspp/cstc/fundscience/papers/bond.htm.
Foundation Center. 2004. Foundation Giving Trends, 2004. New York, NY: The Foundation Center. [Summary on-line.] Available: www.foundationcenter.org/research/trends_analy-sis/pdf/04fgthiltes.pdf [accessed July 18, 2004].
Lichtman MA, Hunter MD, Liders GJ. 2004. “Intellectual Property—The Dispute between Research Institutions and Voluntary Health Agencies.” Nature Biotechnology, 22(4):385-386.
McGeary M, Smith PM. 2001. State Support for Medical Research: Report on State Support for Health Research for Funding First. Washington, DC: McGeary and Smith, October 26. [Online.] Available: www.laskerfoundation.com/ffpages/reports/m1.htm [accessed July 18, 2004].
McNichol E, Harris M. 2004. Many States Cut Budgets as Fiscal Squeeze Continues. Washington, DC: Center on Budget and Policy Priorities, April 26. [On-line.] Available: www.cbpp.org/pubs/sfp.htm [accessed July 18, 2004].
NCBC (North Carolina Biotechnology Center). 1999. North Carolina Biotechnology Center: A Progress Report 1984-1999. [On-line.] Available: www.ncbiotech.org/pdffiles/progress.pdf [accessed July 20, 2004].
NCSL (National Conference of State Legislatures). 2004. State Budge & Tax Actions 2004: Preliminary Report. [On-line.] Available: www.ncsl.org/programs/fiscal/presbta04.htm#note4 [accessed July 12, 2004].
NGA (National Governors Association). 2001. 2001 State Tobacco Settlement Spending Initiatives (Issue Brief). Washington, DC: NGA Center for Best Practices. [On-line.] Available: www.nga.org/cda/files/IB20010411SPENDING.pdf [accessed July 18, 2004].
NIH (National Institutes of Health). 2004a. Federal Obligations for Health R&D, by Source or Performer, Fiscal Years 1985-2001. [On-line]. Available: grants.nih.gov/grants/award/research/sourfund.htm [accessed May 20, 2004].
NIH. 2004b. Federal Agencies Health R&D by Performer. [On-line]. Available: grants.nih.gov/grants/award/research/performr.htm [accessed June 29, 2004].
NSF (National Science Foundation). 2002. Memorandum, Christine C. Boesz, Inspector General, NSF, to Warren Washington, Chair, National Science Board, and Rita R. Colwell, Director, NSF, “Management Challenges for NSF in 2003.” Arlington, VA: NSF, December 23.
NSF. 2003a. Research and Development in Industry: 2000. NSF 03-318. Arlington, VA: NSF. [Online.] Available: www.nsf.gov/sbe/srs/nsf03318/start.htm [accessed July 18, 2004].
NSF. 2003b. Academic Research and Development Expenditures: Fiscal Year 2001. NSF 03-316. Arlington, VA: NSF. [On-line.] Available: www.nsf.gov/sbe/srs/nsf03316/start.htm [accessed July 18, 2004].
NSF. 2004a. Science and Engineering Indicators 2004. Two volumes: volume 1, NSB 04-1; volume 2, NSB 04-1A. Arlington, VA: National Science Board, NSF. [On-line.] Available: www.nsf.gov/sbe/srs/seind04/start.htm [accessed July 18, 2004].
NSF. 2004b. U.S. Academic R&D Continues to Grow as More Universities and Colleges Expand Their R&D Activities (NSF InfoBrief). NSF 04-319. Arlington, VA: NSF. [On-line.] Available: www.nsf.gov/sbe/srs/infbrief/nsf04319/start.htm [accessed July 18, 2004].
NVCA (National Venture Capital Association). 2004a. What Is Venture Capital? [Online.] Available: www.nvca.org/def.html [accessed July 12, 2004].
NVCA. 2004b. Venture Capital Stays the Course in Q1 2004 with $4.6 billion Invested (Press Release). [Online.] Available: www.nvca.org/pdf/Q104MTReleaseEmbargoFINAL.pdf [accessed July 12, 2004].
PhRMA (Pharmaceutical Research and Manufacturers of America). 1997. New Medicines in Development for Cancer: 1997 Survey. Washington, DC: PhRMA.
PhRMA. 2003. Medicines in Development for Cancer: 2003 Survey. Washington, DC: PhRMA, May. [On-line.] Available: www.phrma.org/newmedicines/resources/2003-05-12.109.pdf [accessed May 20, 2004].
PhRMA. 2004. Pharmaceutical Industry Profile 2004. Washington, DC: PhRMA. [On-line.] Available: www.phrma.org/publications/publications//2004-03-31.937.pdf [accessed May 20, 2004].
R!A (Research!America). 2003. 2001 Investment in U.S. Health Research [Issue Paper]. Alexandria, VA: R!A. Data supplied by Stacie Propst, Ph.D.
R!A. 2004. 2002 Investment in U.S. Health Research [Issue Paper]. Alexandria, VA: R!A. [On-line.] Available: www.researchamerica.org/publications/appropriations/healthdollar2002.pdf [accessed July 18 2004].
U.S. GAO (U.S. General Accounting Office). 2004. Tobacco Settlement: States’ Allocations of Fiscal Year 2003 and Expected Fiscal Year 2004 Payments. GAO-04-518. Washington, DC: GAO. [On-line.] Available: www.gao.gov/new.items/d04518.pdf [accessed July 18, 2004].


















