11
Oxygen
This chapter summarizes relevant epidemiologic and biomedical studies associated with exposures to low-oxygen atmospheres. Selected chemical and physical properties and pathophysiologic and mechanistic data are also presented. The subcommittee considered all of that information in its evaluation of the Navy’s current and proposed 1-hour (h), 24-h, and 90-day guidance levels for oxygen. The subcommittee’s recommendations for minimal oxygen levels are provided at the conclusion of this chapter along with a discussion of the adequacy of the data for defining those levels and the research needed to fill the remaining data gaps.
PHYSICAL AND CHEMICAL PROPERTIES
Oxygen is a colorless, odorless, tasteless gas that supports combustion (Budavari et al. 1989). Selected physical and chemical properties are listed in Table 11-1.
OCCURRENCE AND USE
Oxygen is a highly combustible gas that is necessary to sustain life (Patty 1963; Ebbing and Wrighton 1996). The principal uses of oxygen stem from its strong oxidizing and life-sustaining properties. It is used in medicine for therapeutic purposes (Ebbing and Wrighton 1996). Oxygen is used in steel production, copper smelting, and coal gasification (Hawley 1977). It also is used in the synthesis of methanol, acetylene, and other chemicals and as an oxidizer for liquid rocket propellants (Hawley 1977).
TABLE 11-1 Physical and Chemical Properties of Oxygena
Ambient air is composed of 20.9% oxygen, 78.1% nitrogen, 0.03% carbon dioxide, and less than 1% other gases (Lide 1991). Data collected on nine nuclear-powered ballistic missile submarines indicate an average partial pressure of oxygen (PO2, the product of the barometric pressure and the percentage of oxygen in the ambient atmosphere) of 148 millimeters of mercury (mmHg) and a range of 123-188 mmHg; data collected on 10 nuclear-powered attack submarines indicate an average PO2 of 149 mmHg and a range of 118-180 mmHg (Hagar 2003). See Box 11-1 for descriptions of terms related to gas pressures and oxygen physiology.
SUMMARY OF TOXICITY AND ADVERSE RESPONSES ASSOCIATED WITH LOW-OXYGEN ENVIRONMENTS
Oxygen is a highly combustible gas that is necessary to sustain animal life. Excessive amounts of oxygen in the system (hyperoxia) can be detrimental to human health. Oxygen toxicity can occur with exposure to hyperoxic conditions at ambient pressure or in hyperbaric environments. Oxygen toxicity is characterized by pulmonary toxicity, neurotoxicity, and
|
BOX 11-1 Terms Related to Gas Pressures and Oxygen Physiology Terms related to the pressure of the atmosphere or the partial pressure of oxygen Normobaric: Denoting a barometric pressure equivalent to sea-level pressure (760 mmHg). Hyperbaric: Pertaining to pressure of ambient gases above sea-level normal (>760 mmHg). Hypobaric: Pertaining to pressure of ambient gases below sea-level normal (<760 mmHg). Partial pressure of oxygen (PO2): The partial pressure of oxygen is determined by the barometric pressure. At sea level, the atmospheric pressure is 760 mmHg, and oxygen makes up 20.946% of inspired air. Thus, at sea level, oxygen exerts a partial pressure of about 159 mmHg (760 × 0.20946). Fraction of inspired oxygen (FiO2): The percent of oxygen in the inspired gas. Terms related to the partial pressure of oxygen within different sites of the respiratory tract Partial pressure of inspired oxygen (PiO2): The partial pressure of oxygen in inspired air is not equivalent to that found in the atmosphere. Water vapor humidifies the inspired air and dilutes the amount of oxygen by reducing the partial pressure by the saturated vapor pressure (47 mmHg). The PiO2 at sea level is 149 mmHg ([760 47] × 0.2094). Alveolar partial pressure of oxygen (PAO2): The partial pressure of oxygen in the alveolus is controlled by the rate of oxygen absorption into the blood and the rate at which oxygen is delivered by ventilatory processes. The PAO2 at sea level is about 104 mmHg. Terms that define oxygen transport within the blood Arterial oxygen tension (PaO2): Equivalent to the partial pressure of oxygen in the plasma phase of arterial blood or the amount of dissolved oxygen in the plasma phase. The PaO2 is determined by PAO2 and the interface between the alveolus and the capillaries. Measured by an electrode that senses dissolved oxygen molecules, such as a co-oximeter. Alveolar-arterial PO2difference: The difference between measured PaO2 and calculated PAO2. |
|
Oxygen saturation of hemoglobin (SaO2): The percentage of all the available heme binding sites saturated with oxygen is the hemoglobin oxygen saturation or the SaO2. SaO2 is determined mainly by PaO2 and the relationship between the two variables is the oxygen dissociation curve. An SaO2 of 97% means that for every 100 hemoglobin binding sites, 97 are occupied with an oxygen molecule, and the other three are either bound to something else or are unbound. Oxygen saturation curve: The oxyhemoglobin dissociation curve mathematically equates SaO2 to PaO2. Hyperoxia: An excess of oxygen in the system resulting from exposure to high oxygen concentrations, especially at hyperbaric pressures of oxygen. Hypoxia: A concentration of oxygen in arterial blood that is less than normal. Anoxia refers to complete lack of oxygen. |
ocular toxicity (Carraway and Piantadosi 1999). Oxygen toxicity in humans is dependent on both the exposure concentration and the duration of exposure. For example, studies in human volunteers have shown that adverse pulmonary effects can occur following 12-16 h of exposure to oxygen at 1.0 absolute atmospheres (ATA), following 8-14 h of exposure at 1.5 ATA, or following 3-6 h of exposure at 2.0 ATA (Clark and Lambertsen 1971; Clark et al. 1999). Oxygen-induced pulmonary toxicity is characterized by substernal and tracheal irritation and cough consistent with tracheobronchitis (Carraway and Piantadosi 1999).
Exposure to abnormally high oxygen concentrations (for example, PO2 above 1.5 to 2.0 ATA) is also associated with seizures, dizziness, nausea, tunnel vision, blindness, fatigue, anxiety, confusion, ataxia, and other signs of neurotoxicity (Carraway and Piantadosi 1999). Retrolental fibroplasia is a common ophthalmologic effect seen in newborn children exposed to high levels of oxygen under normal barometric (normobaric) conditions (Weinberger et al. 2002). Oxygen-induced retinopathies have been reported to occur under ambient conditions in adults (Nichols and Lambertsen 1969; Kobayashi and Murakami 1972); however, the incidence of that disease is quite low (Carraway and Piantadosi 1999). Some sailors with specialized job duties, such as divers, potentially are at risk for oxygen toxicity; however, oxygen toxicity is not anticipated to be a significant concern among submariners, because submarines are often kept oxygen deficient to decrease the risk of onboard fires. Therefore, this review focuses on hypoxic, rather than hyperoxic, environmental conditions.
Hypoxia may be defined as “any state in which the oxygen in the lung, blood and/or tissues is abnormally low compared with that of normal resting man breathing air at sea level” (Bartels et al. 1973). Environmental hypoxia, as occurs on board submarines, exists when the barometric pressure or the PO2 is low (Bartels et al. 1973). Exposures to hypoxic conditions can trigger adaptive and adverse responses. Physiologic changes involve the cardiovascular, pulmonary, and hematopoietic systems and include hyperventilation, tachycardia, pulmonary hypertension, cerebral vasoconstriction, systemic vasodilation, hypocapnia, respiratory alkalosis, erythropoietin synthesis, enhanced red blood cell production, and an increased hematocrit. Other changes include altered moods and impaired cognitive or motor performance. People who have not acclimated to hypoxic conditions can develop headaches, fatigue, shortness of breath, nausea, anorexia, sleep disturbances, or vomiting. Severe hypoxia can also result in pulmonary edema, cerebral edema, or retinal hemorrhage, although these lesions are most commonly seen at high altitudes.
Effects in Humans
Environmental Exposures to Low-Oxygen Environments
Although oxygen makes up about 20.9% of the atmosphere at all elevations, PO2 decreases as elevation increases, which means that there is less oxygen available for respiration at high altitudes. At sea level, the barometric pressure is about 760 mmHg, and the PO2 in dry air is about 160 mmHg (760 mmHg × 0.209). Inspired air is humidified, thus reducing the PO2 to 149 mmHg. In a normal human, the PO2 in the alveolus is similar to that in blood (about 100 mmHg in alveolus vs about 94 mmHg in the arterial blood, where oxygen is carried by hemoglobin). At 5,500 m, the barometric pressure is about 380 mmHg, and the PO2 is only 80 mmHg (380 × 0.209). As one ascends in altitude, the partial pressure of inspired oxygen (PiO2, PO2 corrected for the water vapor that humidifies the inspired air and dilutes the amount of oxygen), the arterial oxygen tension (PaO2, a measure of the partial pressure of oxygen in the plasma phase of arterial blood), and the arterial oxygen saturation (SaO2, the percentage of heme binding sites saturated with oxygen) also decrease (see Table 11-2).
Environmental hypoxia is a common occurrence since a significant portion of the human population lives at high altitude (≥2,500 meters [m]). Responses observed in people due to high altitude (hypobaric hypoxia)
TABLE 11-2 Arterial Blood Gas Values Associated with Different Altitudes
|
Altitude (m) |
Barometric Pressure (mmHg) |
Partial Pressure of Inspired Oxygen (PiO2) (mmHg) |
Arterial Oxygen (PaO2) (%) |
Arterial oxygen Saturation (SaO2) (%) |
|
0 |
760 |
149 |
94 |
97 |
|
1,500 |
630 |
122 |
66 |
92 |
|
2,500 |
564 |
108 |
60 |
89 |
|
3,000 |
523 |
100 |
53 |
85 |
|
3,600 |
483 |
91 |
52 |
83 |
|
4,600 |
412 |
76 |
44 |
75 |
|
5,500 |
379 |
69 |
40 |
71 |
|
6,100 |
349 |
63 |
38 |
65 |
|
7,300 |
280 |
52 |
34 |
50 |
|
Abbreviations: m, meters; mmHg, millimeters of mercury. Source: Hultgren 1997. Reprinted with permission; copyright 1997, Hultgren Publications. |
||||
have been extensively studied and may be relevant for submariners. At altitudes above 2,500 m, most people exhibit decreased SaO2 (Moore 2000). People can live at high altitudes because they acclimate to the decreased PO2 associated with reduced atmospheric pressure. Physiologic changes in response to high altitudes predominantly involve the cardiovascular, pulmonary, and hematopoietic systems (Hultgren 1997; Moore 2000). A decrease in PaO2 is a potent stimulus to the carotid and aortic chemoreceptors and results in hyperventilation and tachycardia. Decreased PaO2 (hypoxia) is also a powerful stimulus for increased cerebral blood flow and vasodilation. However, hypocapnia caused by hypoxic hyperventilation may cause cerebral vasoconstriction, thus offsetting vasodilatory effects of hypoxia. Hypoxic hyperventilation also can result in a respiratory alkalosis that causes a shift of the oxyhemoglobin curve to the left.
Increased ventilation and enhanced maximum oxygen extraction are additional aspects of immediate and chronic high-altitude acclimatization (Moore 2000). Longer-term responses include elevated renal erythropoietin synthesis, which results in enhanced red blood cell production and increased hematocrit. Genetic differences have been recognized in some high-altitude natives, such as Quechaus and Sherpas. Some high-altitude natives demonstrate blunted hypoxic ventilatory responses, which are mediated by the carotid body oxygen sensor; reduced hypoxic pulmonary vasoconstrictor responses; increased blood volumes; elevated red blood cell mass;
and additional modifications to striatal muscle metabolism (Hochachka et al. 1999; Moore 2000). Many of these changes are also found in people adapted for endurance performance (Hochachka et al. 1999).
People who have not acclimated to high altitude before engaging in physical activities can develop acute high-altitude illness, which also is referred to as acute mountain sickness (AMS). Headache is the most common AMS symptom and is often most intense during the night and shortly after arising in the morning. This phenomenon is usually attributed to increased hypoxemia caused by altitude-induced periods of sleep apnea. Other symptoms may include fatigue, shortness of breath, nausea, anorexia, sleep disturbances, or vomiting (Hackett and Roach 2001; Basnyat and Murdoch 2003). Symptoms generally develop within 6-18 h after arrival at high altitude. The incidence of AMS among participants attending North American conferences in locations at altitudes of 1,920-2,956 m was about 25% (Montgomery et al. 1989; Honigman et al. 1993). Rapid ascent to altitudes above 3,600 m causes AMS in many people (34-68%) (Sonna 2002). Rapid ascent to altitudes at or above 5,333 m causes severe, incapacitating symptoms in almost all individuals. Although AMS is usually self-limiting, in rare cases individuals can develop life-threatening high-altitude pulmonary edema (HAPE) or high-altitude cerebral edema (HACE). The incidence of HAPE or HACE is altitude-dependent and is reportedly 0.1-8% (Sonna 2002; Basnyat and Murdoch 2003). Risk factors for the development of AMS include the rate of ascent, final altitude, exertion, and individual susceptibility factors, including age, presence of lung disease, and physical condition (Honigman et al. 1993; Basnyat and Murdoch 2003).
Experimental studies have confirmed the development of symptoms compatible with AMS in people exposed to hypobaric hypoxic conditions. For example, Shukitt-Hale et al. (1998) exposed 23 U.S. Army personnel (18-29 years of age) to an environment mimicking low altitude and two environments mimicking moderately high altitude, equivalent to 550, 4,200, and 4,700 m, respectively, for 4.5 h. The participants were evaluated using symptom, mood, and cognitive and motor performance measures. At 4,700 m, significant effects were exhibited in 7 of 9 symptom measures, 12 of 16 mood measures, and 7 of 10 cognitive and motor performance measures. Cognitive and motor performance was affected on relatively simple tasks, such as simple and choice reaction time and addition tests. At 4,200 m, significant effects were exhibited in 1 of 9 symptom measures, 4 of 16 mood measures, and 4 of 10 cognitive and motor performance measures. The authors concluded that exposure to a simulated altitude of 4,200 m was associated with fewer AMS symptoms compared with incidences seen
following exposures to the equivalent of 4,700 m, suggesting that even relatively small changes in altitude can have a dramatic effect on symptom reporting and performance. Shukitt and Banderet (1988) showed that acute human exposures to environmental conditions equivalent to an altitude of 1,600 m results in mood changes. Friendliness, clear thinking, dizziness, sleepiness, and unhappiness were increasingly reported at 4,300 m, whereas only sleepiness was over-reported at 1,600 m. At 4,300 m, the altered moods differed from baseline on the day of arrival (within 1-4 h), differed even more after 1 day (18-28 h), and returned to baseline by day 2 (42-52 h). Morning and evening values were similar at each altitude. Therefore, changes in mood states at higher altitudes have a distinct and measurable time course. Studies performed by Li et al. (2000a) in healthy young college students (17-18 years of age) showed that 1-h exposures to simulated high altitudes at or above 2,800 m were associated with decreased SaO2 and adverse effects on mood state. Reduced performances on a visual four-choice reaction-time test were observed following a 1-h exposure to simulated high altitudes at or above 3,600 m (Li et al. 2000b).
The pathophysiology of high-altitude diseases has been reviewed (Roach and Hackett 2001; Basnyat and Murdoch 2003). It is thought that environmental hypoxia triggers several changes that precede AMS development, including impaired gas exchange, increased sympathetic activity, fluid retention and redistribution, increased cerebral blood flow, altered blood-brain barrier permeability, and ultimately cerebral edema (vasogenic edema) in the most severe cases. Development of HACE represents a severe end-stage manifestation of this vasogenic response. Rapid ascent to high altitude is also associated with the development of noncardiogenic pulmonary edema. HAPE is thought to be caused by the combination of hypoxia-induced pulmonary hypertension and increased permeability of the pulmonary capillary endothelium (Basnyat and Murdoch 2003). HAPE is further characterized by elevated pulmonary artery pressure, normal left atrial filling pressure, and normal ventricular function (Basnyat and Murdoch 2003).
Clinical manifestations of HAPE include a nonproductive cough, rales, dyspnea on exertion, fever, fatigue, weakness, resting tachycardia and tachypnea, and cyanosis. Left untreated, HAPE can be rapidly fatal. Risk factors for HAPE include moderate to severe exertion, exposure to cold temperatures, anxiety, young age, male gender, and possibly obesity. HAPE usually begins within the first 2-4 days after rapid ascent to high altitudes, and onset commonly occurs during the second night of sleep at high altitude. Most physicians have maintained that HAPE is unknown or rare at
elevations below 2,440 m (Raymond 2003). However, Gabry et al. (2003) described 52 lowlanders who developed HAPE after skiing at 1,400-2,400 m. Although this retrospective study could not rule out possible confounding etiologies for pulmonary edema, such as infection or illicit drug use, it provides intriguing evidence that some individuals might develop HAPE at lower altitudes than previously recognized (Raymond 2003). The barometric pressure at 1,500 m is 630 mmHg, and the PO2 is about 132 mmHg. Those conditions are analogous to breathing air containing about 17.3% oxygen at sea level. A retrospective study by Cremona et al. (2002) further supports Gabry’s observations that lung disease could occur in the absence of frank signs of HAPE. Cremona showed that at 4,559 m about 75% of 262 climbers who were not diagnosed with HAPE had an increase in closing volume suggestive of subclinical pulmonary edema. Forty (15%) of the participants also had radiologic or physical diagnostic evidence—rales or pulmonary edema on chest radiographs—indicating increased pulmonary extravascular lung volumes. Only one participant had clinical features consistent with full-blown HAPE.
Experimental Studies
Multiple human chamber studies have evaluated physiologic responses, clinical signs and symptom reporting, and effects on cardiovascular and cognitive performance in people exposed to low-oxygen environments. One issue that needs to be considered is whether responses to environmental hypoxia are influenced by barometric pressure. There is some evidence in sheep that pulmonary responses observed under normobaric hypoxic conditions and hypobaric hypoxic conditions are not equivalent; indeed, clinical effects are often more severe under hypobaric conditions (Levine et al. 1988). However, Sheedy et al. (1996) failed to demonstrate that normobaric hypoxic conditions and hypobaric hypoxic conditions resulted in different cardiopulmonary responses in rats. Roach et al. (1996) exposed nine healthy men (ages not specified) to a simulated high-altitude environment (equivalent to 4,564 m; barometric pressure = 432 mmHg; PiO2 = 80 mmHg), a normobaric hypoxic environment (barometric pressure = 760 mmHg, PO2 = 80 mmHg), or a normoxic hypobaric environment (barometric pressure = 432 mmHg; PiO2 = 115 mmHg), which was achieved by adding supplemental oxygen to the chamber, for 9 h. Symptoms associated with AMS were more prevalent in the simulated high-
altitude environment (five of nine subjects developed AMS) than in the normobaric hypoxic environment (two of nine subjects developed AMS) despite the presence of similar SaO2 values (about 83%) in the two treatment groups. A relative lack of AMS symptoms following exposures to normobaric hypoxic conditions was reported by Meehan (1986). It is unknown why combined environmental hypoxia and hypobaria exacerbates AMS symptoms in subjects. Tucker et al. (1983) reported greater pulmonary ventilation during normobaric hypoxic conditions (equivalent to 14% oxygen) than during a high-altitude (similar to 4,570 m) exposure in six resting subjects exposed for 2-h to the same PO2. Studies by Loeppky et al. (1996, 1997) have largely substantiated those results. Collectively, these studies suggest that normobaric hypoxic conditions result in fewer adverse effects when compared with findings seen in people at equivalent hypoxic high-altitude (hypobaric) conditions. Because of the possible differences between the responses observed under normobaric and hypobaric conditions, the majority of the subcommittee’s review focuses on experimental studies conducted under conditions that would be found on board submarines (that is, normobaric hypoxic atmospheres).
Exposure to environmental hypoxia under normobaric conditions can impair physical performance. For example, Taylor and Bronks (1996) reported that in healthy young male subjects (average age of 20.9 years; n = 14), exercise times were reduced about 25% when performing moderate exercise (30 to 60 watts) on a cycle ergometer and breathing an oxygen-deficient atmosphere (fraction of inspired oxygen [FiO2] = 0.135) compared with exercise times recorded for the same subjects under normoxic conditions (FiO2 = 0.2093). The SaO2 values at exhaustion under hypoxic conditions were lower than those observed under normoxic conditions (69.9% vs 93.4%). Exercise under hypoxic conditions also was associated with increased heart rates, reduced maximal oxygen uptake, and increased plasma lactate and ammonia concentrations.
Piehl Aulin et al. (1998) examined whether normobaric hypoxic conditions affect erythropoiesis, blood pressure, physical performance, or mood. Healthy young male (n = 17) and female (n = 3) endurance athletes (20 to 32 years of age) were housed under normoxic, mild hypoxic (16.2% oxygen), or moderate hypoxic (14.9% oxygen) conditions intermittently (12 h hypoxic, 12 h normoxic) for 10 consecutive days followed by exposure to normoxic conditions for 7 days. PaO2 and SaO2 were significantly decreased during the 10-day intermittent hypoxic exposure. Living at normobaric hypoxia corresponding to an altitude of 2,000 m for 12 h per day was
associated with increased production of erythropoietin and stimulation of erythropoiesis within 2 to 5 days of initial exposure. Increased erythropoietin production and concomitant increases in red blood cell production also were observed by Rodriguez et al. (2000) in people exposed to hypobaric hypoxic conditions for 90 minutes (min) and by Berglund et al. (2002) in people (n = 7) exposed for 10 days to moderately hypoxic (reported PO2 = 14 kilopascals, equivalent to 105 mmHg), normobaric conditions. Piehl Aulin et al. (1998) further report that submaximal and maximal oxygen uptakes, blood pressures at rest and during exercise, and profile of mood states (POMS) tests did not change during their study.
Hodkinson et al. (2003) examined whether normobaric hypoxia causes activation of coagulation and might therefore increase the risk of venous thromboembolism. These investigators exposed six healthy male volunteers to either dry air or a hypoxic gas mixture composed of 12.8% oxygen in nitrogen (equivalent to breathing air at 3,660 m) for 3 h. The volunteers were seated during exposure. Hodkinson et al. (2003) did not observe significant differences in hemostatic or endothelial markers between the control and hypoxic groups even though platelet and leukocyte counts were significantly higher in the hypoxic group. There were increases in fibrinogen and von Willebrand factor as well as rheological changes, but these changes were not significantly different from those exhibited in controls.
Altitudes as low as 1,500 m (PO2 about 127.5 mmHg, equivalent to an atmosphere containing about 17.9% oxygen at sea level) have been associated with reduced cognitive and motor performance (Gustafsson et al. 1997). Chamber studies, including several experiments designed to replicate conditions found on board submarines, have shown that exposures to normobaric hypoxic conditions also might result in reduced cognitive performance. Karlin and Curtis (1945) demonstrated reduced physical performance and mental efficiency in submariners exposed to an atmosphere containing 17% oxygen and 3% carbon dioxide for 50 h. Shukitt et al. (1988) reported that short-term (15-day) exposure to normobaric atmospheres containing 13% oxygen can result in decreased cognitive function, altered mood states, moderate AMS symptoms, and impaired fine motor control. For 5 consecutive days, Cymerman et al. (2002) exposed seven submariners (31.8 ± 6.1 years of age) to environmental conditions that would be encountered on board a disabled submarine (environmental hypoxia, 16.75% oxygen; hypercapnia, 2.5% carbon dioxide; and cold temperatures, 4°C). Within 2 days, subjects reported cold stress and muscle discomfort that lasted throughout the rest of the study. Participants also developed decreased postural control after 66 h of exposure. Loss of bal-
ance was especially apparent when the testing was performed with the subject’s eyes closed. The submariners did not report a significant increase in AMS symptoms despite the hypoxic conditions within the chamber. The study authors concluded that the observed effects would not impair submariners’ abilities to perform their duties when participating in a rescue effort.
Gustafsson et al. (1997) and Linde et al. (1997) simulated normal work shifts on board Swedish and U.S. submarines under three normobaric hypoxic conditions. Young male volunteers (20-28 years of age, n = 22) participated in the experiments. The men were exposed to intermittent to nearly continuous hypoxic conditions and were tested using a battery of cognitive performance measures. Prolonged (10-day) continuous exposures to moderately hypoxic conditions (PO2 = 105 mmHg) were associated with symptoms compatible with AMS. Symptoms of AMS were also exhibited during more intermittent exposures (for example, 24-h) at a PO2 of 97.5 mmHg. Performance on one test of motor function (finger tapping) was adversely affected by intermittent exposures at a PO2 of 97.5 mmHg or 10-day exposures at a PO2 of 105 mmHg. Linde et al. (1997) reported that effects on cognitive performance were found to be small and were prevented when the PO2 was maintained above 97.5 mmHg.
Physical exertion may further exacerbate the cognitive effects of normobaric hypoxic environmental conditions. Knight et al. (1990a) exposed 13 healthy male subjects (average age, 24 ± 6) to 13%, 17%, or 21% oxygen atmospheres for 15 days. Chamber carbon dioxide levels were raised to 0.9% (ambient air contains about 0.03%) to mimic conditions on board submarines. Subjects were tested at rest and under submaximal work rates designed to achieve 35% or 65% of the maximum rate of oxygen uptake. A significant reduction in SaO2 (83-85% under hypoxic conditions vs 94% at ambient conditions) was observed during exercise in atmospheres containing 13% and 17% oxygen. Despite decreases in SaO2, Knight et al. (1990a) did not demonstrate a significant decrement in the volunteers’ abilities to solve computational problems during exposures to moderate hypoxic conditions even when subjects performed moderate exercise. Knight et al. (1990b) reported symptoms in 13 healthy male subjects (average age, 24 ± 6) exposed to both normoxic and hypoxic conditions for 15 days. Exposures were conducted under conditions that simulated atmospheres found on board submarines. The exposure scenarios were as follows: 21% oxygen for 3 days, 17% oxygen for 3 days, 21% oxygen for 3 days, 13% oxygen for 3 days, and 21% oxygen for 3 days. Because of the similarity in the subjects’ physical measures (age, weight, height), the subcommittee presumes that the cohorts used in the two Knight studies
were identical. Significant changes in AMS scores were observed in volunteers when exposed to atmospheres containing 17% oxygen at hypobaric pressure (PO2 = 98 mmHg) or when exposed to atmospheres containing 13% oxygen at normobaric pressure (PO2 = 99 mmHg). Many of the subjects (42%) displayed symptoms compatible with AMS after 1 day of exposure to an atmosphere containing 13% oxygen. Fewer of the subjects (8%) exposed at 13% oxygen continued to report symptoms of AMS for the next 2 days. Symptoms compatible with AMS were observed in 3 of 11 of the subjects when they were exposed to an atmosphere containing 17% oxygen for 3 days and only when the barometric pressure in the chamber was reduced to 576 mmHg for the final 7 h of exposure (Knight et al. 1990a).
Cerebral hypoxia may produce slowing of the alpha rhythm, increases in the slow-wave components, and other changes in the human electroencephalogram (EEG). Van der Worp et al. (1991) used quantitative EEGs to investigate cerebral hypoxia in humans exposed to normobaric hypoxic conditions. Middle cerebral blood flow velocity and cortical EEGs were collected in a group of healthy young male subjects (20-27 years of age, n = 10) before and during exposures to environmental hypoxia when SaO2 values were 80%, 70%, and 60%. Environmental hypoxia associated with an SaO2 of 60% caused an increase in EEG slow-wave activity. Lesser degrees of environmental hypoxia caused only minimal EEG changes. Schellart and Reits (2001) demonstrated that acute exposures of healthy volunteers (18-52 years of age, n = 14) to normobaric hypoxic conditions (equivalent to an atmosphere containing about 10% oxygen) caused a rapid increase (within 20 min) in the amplitude of all bands. The volunteers also had significantly reduced SaO2 (mean was 67.2 ± 6.0% after 20 min), and some individuals demonstrated sleepiness or reduced attentiveness when their SaO2 dropped below 55%. Kraaier et al. (1988) showed that normobaric hypoxic conditions equivalent to conditions at 6,096 m caused increases in slow-wave activity and decreases in alpha-wave activity. Ozaki et al. (1995) observed similar EEG changes in people exposed to hypobaric hypoxic conditions equivalent to those at altitudes of 3,000-6,000 m. Changes in the alpha rhythm (10-11 hertz), the most sensitive EEG parameter examined, occurred at hypobaric hypoxic conditions equivalent to those found at 3,000 m. Increased environmental hypoxia further depressed that wave component. Changes in theta rhythm were also observed when conditions equivalent to those at an altitude of 5,000 m were reached.
An acute decrease in PaO2 is a strong stimulus for increased cerebral blood flow. Van der Worp et al. (1991) showed that exposures to normo-
baric hypoxic conditions resulted in increased blood flow velocity. Huang et al. (1987) likewise showed that transient increases in cerebral blood flow occur in people following initial exposures to high-altitude conditions. Buck et al. (1998) used positron emission tomography (PET) with [15O]H2O to assess cerebral blood flow in eight healthy volunteers (average age, 28 ± 5 years) exposed to normobaric hypoxic conditions that mimicked those at 3,000 or 4,500 m of elevation. PaO2 values observed following exposures to simulated altitudes of either 3,000 or 4,500 m were 57.2% ± 6.5 and 40.9% ± 5.1, respectively. Increased brain blood flow was observed following exposures to hypoxic conditions associated with an altitude of 4,500 m. Changes were most pronounced in the hypothalamus (32.8% increase), thalamus (19.2% increase), and cerebellum (17.6% increase). Increases in other brain regions varied from 9.1% to 13.8%.
Occupational and Epidemiologic Studies
Few occupational studies have explored whether chronic hypoxia is associated with any adverse health effects. Basnyat and Litch (1997) assessed the incidence of illness among porters and trekkers in the Himalayan mountains. A cohort of 155 members of commercial trekking groups (102 Nepali porters, 31 Nepali trek staff, and 22 Western trekkers) were observed for 22 days as they ascended elevations ranging from 487 m to 5,100 m. Medical problems occurred in 45% of subjects. High-altitude pharyngitis and bronchitis (12%), AMS (8%), and gastroenteritis (6%) were the most common illnesses reported. West (1999) discussed how supplemental environmental oxygen can improve sleep quality, mental performance, productivity, and general well-being in high-altitude workers.
Airline personnel is another group that routinely works under hypoxic conditions. Commercial jets operate under reduced atmospheric pressure, low humidity, and mild hypoxic conditions. Nicholas et al. (1998) reported that U.S. pilots and navigators have exhibited significantly increased mortality due to cancer of the kidneys and renal pelvis, motor neuron diseases, and external causes. In addition, increased mortality due to prostate cancer, brain cancer, colon cancer, and cancers of the lip, buccal cavity, and pharynx was suggested. These results must be considered cautiously, because other environmental factors associated with high-altitude work, such as increased cosmic radiation, might also be implicated in these findings.
Effects in Animals
Because hypoxia has been well studied in humans, the subcommittee’s review of the experimental animal literature focused on less-studied clinical end points, including histologic changes in organs following acute to chronic hypoxia. The other literature cited further describes the effects of severe hypoxia.
Acute Toxicity
Kleinsasser et al. (2003) reported that pigs can develop radiographic and clinical features compatible with early HAPE after 48-h exposures to normobaric hypoxic conditions (10% oxygen). There is some experimental evidence that prior respiratory-tract infection might increase the risk of HAPE. Carpenter et al. (1998) showed that weanling rats infected with Sendai virus that were exposed to normobaric hypoxic conditions (PO2 = 76 mmHg) for 24 h exhibited the early stages of pulmonary edema.
Repeated Exposures, Subchronic Toxicity, and Chronic Toxicity
Subchronic (21-day) or chronic (>90-day) exposures to moderate to severe environmental hypoxia (for example, atmospheric oxygen <10-12%) in rats are associated with multiple pathologic changes, including reduced body weight and body-weight gain (Sheedy et al. 1996; Cervos Navarro et al. 1999; Lorente et al. 2002), adrenal gland and spleen atrophy (Wolman et al. 1993; Lorente et al. 2002), carotid body hypertrophy (Clarke et al. 2000), hypertension (Schweda et al. 2000), increased numbers of myocardial mitochondria (Cervos Navarro et al. 1999), right ventricular hypertrophy (Sheedy et al. 1996; Schweda et al. 2000), increased lung growth (Sekhon and Thurlbeck 1996), multi-organ lipidosis (Wolman et al. 1993), remodeling of the brain and lung microvasculature (LaManna et al. 1992; Aguirre et al. 2000), and hippocampal neuronal loss (Shukitt-Hale et al. 1996). Cervos-Navarro et al. (1991) found that cats exposed to decreasing amounts of oxygen (21%, 15%, 10%, 8%, 7%, and 5%) over a period of 320 days developed microvascular proliferation and decreased Purkinje cell numbers.
Reproductive Toxicity in Males
Hypoxia is hypothesized to reduce fertility (Vitzthum and Wiley 2003); however, few studies have been conducted to confirm that hypothesis. Saxena (1995) reported that rhesus monkeys exposed to a simulated altitude of 4,411 m (barometric pressure = 428.8 mmHg) for 6 h per day for 21 days developed decreased semen volumes, sperm counts, and sperm motility as well as elevations in pH and in fructose concentrations. Those changes were associated with degeneration of the germinal epithelium and spermatogenic arrest observed at the end of the exposure; monkeys had not recovered 3 weeks after the exposure. Gonzales et al. (1990) reported that rats exposed to conditions associated with an altitude of 4,340 m for 4 days developed pyknotic primary spermatocytes and spermatids, necrosis of numerous cells, and sloughing of primary spermatocytes. Gosney (1984) showed that young adult male Wistar albino rats exposed to hypobaric hypoxic conditions at a barometric pressure of 380 mmHg for 28 days had smaller testes compared with controls kept in ambient conditions. Histologic examinations of the testes revealed degeneration and sloughing of spermatogenic cells and changes in Leydig cell numbers in the hypoxic rats. Fahim et al. (1980) showed that chronic exposures to a simulated 6,000-m environment were associated with reduced plasma testosterone concentrations and vacuolization and pyknosis of spermatogenic tissues.
Immunotoxicity
There is very limited data available concerning immunologic effects associated with hypoxia. Klokker et al. (1993) exposed eight healthy male volunteers (22-28 years of age) to room air at a simulated altitude of 5,486 m for 20 min in a hypobaric chamber. Exposures to severe hypobaric hypoxic conditions resulted in transient increases in leukocyte concentrations and natural killer-cell numbers and activity.
Genotoxicity
No data could be found indicating that hypoxia results in genotoxicity.
Carcinogenicity
No data could be found indicating that hypoxia results in carcinogenicity. Airline pilots reportedly exhibit increased mortality due to cancer of the kidneys and renal pelvis (Nicholas et al. 1998). Those findings were not considered by the subcommittee, because airline personnel are exposed to increased cosmic radiation and other environmental factors that are absent on board submarines.
PHYSIOLOGIC, TOXICOKINETIC, AND MECHANISTIC CONSIDERATIONS
The mammalian alveolus is designed to absorb oxygen from the air and subsequently deliver it to the blood via a concentration gradient. The oxygen dissociation curve shows the percent saturation of hemoglobin at various PO2 (Figure 11-1). The sigmoid shape of the oxygen dissociation
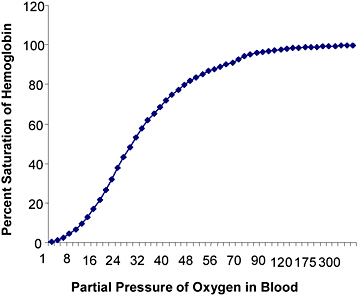
FIGURE 11-1 Hypothetical human blood oxygen dissociation curve at 37°C, pH = 7.4.
curve is a result of the cooperative binding of oxygen to the four polypeptide chains. Thus, hemoglobin is most attracted to oxygen when three of the four polypeptide chains are bound to oxygen. Increased body temperature, increased partial pressure of carbon dioxide, decreased blood pH, and increased 2,3-diphosphoglycerate favor the off-loading of oxygen from the hemoglobin molecule.
Under normal conditions, the plateau of the oxyhemoglobin dissociation curve occurs at a PO2 of 70 mmHg. At high PO2, usually in the lungs, hemoglobin binds to oxygen to form oxyhemoglobin. When the blood is fully saturated, all the erythrocytes are in the form of oxyhemoglobin. PO2 below 60 mmHg have increasingly negative effects on the oxygen saturation of hemoglobin.
Oxygen circulates in the blood and diffuses to the tissues, via a concentration gradient, to be used in cellular metabolism. As the erythrocytes travel to tissues deprived of oxygen, the PaO2 decreases. Consequently, oxyhemoglobin releases oxygen to form hemoglobin. The amount of oxygen available to tissues is dependent on the amount of oxygen entering the lungs, the efficiency of the pulmonary gas exchange, the blood flow to the tissues, and the ability of the blood to carry oxygen. Hypoxia is a relative deficiency of oxygen in the tissues and may be caused by a reduction in PO2, inadequate oxygen transport, or the inability of the tissues to use oxygen.
SUBCOMMITTEE RECOMMENDATIONS
The subcommittee’s recommendation for EEGL and CEGL values for oxygen are summarized in Table 11-3. The current U.S. Navy values are provided for comparison.
1-Hour EEGL
Multiple chamber studies have exposed people to atmospheres containing ≤105 mmHg oxygen for several days (Gustafsson et al. 1997; Linde et al. 1997; Knight et al. 1990 a,b; Roach et al. 1996; Berglund et al. 2002). People exposed to those hypoxic conditions developed significant reductions in SaO2 and clinical effects, including headaches and other symptoms of AMS, increased cerebral blood flow, reduced exercise endurance, increased heart rates, reduced maximal oxygen uptake, and
TABLE 11-3 Emergency and Continuous Exposure Guidance Levels for Oxygen (mmHg)
|
Exposure Level |
Current U.S. Navy Values |
NRC Recommended |
||
|
Maximum |
Minimum |
Minimum Values |
||
|
EEGL |
|
|
|
|
|
|
1 h |
220 |
130 |
105 |
|
|
24 h |
160 |
130 |
127 |
|
CEGL |
|
|
|
|
|
|
90 days |
160 |
130 |
140 |
|
Abbreviations: CEGL, continuous exposure guidance level; EEGL, emergency exposure guidance level, h, hour; mmHg, millimeters of mercury; NRC, National Research Council. |
||||
increased plasma lactate and ammonia concentrations (Knight et al. 1990a,b; Taylor and Bronks 1996; Buck et al. 1998). Despite the decreases in SaO2, Knight et al. (1990b) did not demonstrate significant decrements in the volunteers’ abilities to solve computational problems during moderate hypoxia, even while volunteers performed moderate exercise. The weight of evidence of the available chamber studies indicates that in an emergency, a submarine crew should be able to tolerate a 1-h exposure to air containing a PO2 of 105 mmHg without developing adverse clinical effects.
24-Hour EEGL
Several studies have examined the responses of healthy men to hypoxic conditions mimicking those found on board U.S. submarines. In 1945, Naval researchers demonstrated reduced physical performance and mental efficiency among submariners exposed to 17% oxygen (129 mmHg) and 3% carbon dioxide for 50 h (Karlin and Curtis 1945). Cymerman et al. (2002) exposed seven submariners (31.8 ± 6.1 years of age) to environmental conditions that would be encountered on board a disabled submarine (127 mmHg oxygen, 2.5% carbon dioxide, 4°C) for 5 consecutive days. Within 2 days, subjects reported cold stress and muscle discomfort that lasted throughout the rest of the study. Participants also developed decreased postural control after 66 h of exposure. Loss of balance was especially apparent when the testing was performed with the subject’s eyes closed. The submariners did not report significant increases in AMS symptoms despite the hypoxic conditions within the chamber. The authors of this
study concluded that the effects of the study conditions would not impair submariners’ abilities to perform their duties when participating in a rescue effort. The weight of evidence indicates that in an emergency, a submarine crew should be able to tolerate a 24-h exposure to air containing 127 mmHg oxygen without developing adverse clinical effects. Commercial airplanes offer a useful comparison. They operate at cabin pressures equivalent to altitudes of 1,525-2,000 m (Hultgren 1997). The barometric pressure at 1,500 m is 630 mmHg, and the PO2 is about 132 mmHg.
90-Day CEGL
Short-term (<2-3 days) experimental exposures to air with a PO2 of about 132 mmHg have been associated with mood changes and reduced cognitive and motor performance (Shukitt and Banderet 1988). Those changes would not be acceptable during a 90-day deployment. The weight of evidence suggests that prolonged exposures to atmospheres containing about 140 mmHg would not cause adverse effects in healthy young adult males. The subcommittee anticipates that many crewmen will undergo physiologic adaptations in response to this mildly hypoxic environment, although others might experience drowsiness and other symptoms during the beginning of 90-day deployments. These effects are not expected to degrade performance of the crew or result in other adverse effects. Comparisons to naturally occurring conditions is noteworthy. The recommended 90-day CEGL for minimum levels of oxygen, 140 mmHg, is encountered at altitudes roughly equivalent to 1,000 m. Moreover, the U.S. Occupational Safety and Health Administration (OSHA) standard for workers in oxygen-deficient atmospheres requires air to be at least 19.5% oxygen. OSHA has classified air containing less than 19.5% oxygen to be immediately dangerous to life or health (IDLH) (29 CFR 1910.146(b).
DATA ADEQUACY AND RESEARCH NEEDS
Additional studies are needed to evaluate the appropriateness of the 90-day CEGL. The subcommittee could not find any studies examining the effect of subchronic exposure to mild hypoxia on mood state or cognitive performance. The subcommittee suggests the Navy perform prospective studies to evaluate submariners for complaints of headaches, fatigue, and
other symptoms that might be associated with the mild hypoxic environment often encountered on board submarines.
REFERENCES
Aguirre, J.I., N.W. Morrell, L. Long, P. Clift, P.D. Upton, J.M. Polak, and M.R. Wilkins. 2000. Vascular remodeling and ET-1 expression in rat strains with different responses to chronic hypoxia. Am. J. Physiol. Lung Cell Mol. Physiol. 278(5):L981-L987.
Bartels, H., P. Dejours, R.H. Kellogg, and J. Mead. 1973. Glossary on respiration and gas exchange. J. Appl. Physiol. 34(4):549-558.
Basnyat, B., and J.A. Litch. 1997. Medical problems of porters and trekkers in the Nepal Himalaya. Wilderness Environ. Med. 8(2):78-81.
Basnyat, B., and D.R. Murdoch. 2003. High-altitude illness. Lancet 361(9373):1967-1974.
Berglund, B., M. Gennser, H. Ornhagen, C. Ostberg, and L. Wide. 2002. Erythropoietin concentrations during 10 days of normobaric hypoxia under controlled environmental circumstances. Acta. Physiol. Scand. 174(3):225-229.
Buck, A., C. Schirlo, V. Jasinksy, B. Weber, C. Burger, G.K. von Schulthess, E.A. Koller, and V. Pavlicek. 1998. Changes of cerebral blood flow during shortterm exposure to normobaric hypoxia. J. Cereb. Blood Flow Metab. 18(8):90-6-910.
Budavari, S., M.J. O'Neil, A. Smith, and P.E. Heckelman, eds. 1989. Oxygen. Pp.1101 in the Merck Index: An Encyclopedia of Chemicals, Drugs, and Biologicals, 11th Ed. Rahway, NJ: Merck and Co., Inc.
Carpenter, T.C., J.T. Reeves, and A.G. Durmowicz. 1998. Viral respiratory infection increases susceptibility of young rats to hypoxia-induced pulmonary edema. J. Appl. Physiol. 84(3):1048-1054.
Carraway, M.S., and C.A. Piantadosi. 1999. Oxygen toxicity. Respir. Care Clin. N. Am. 5(2):265-295.
Cervos-Navarro, J., S. Sampaolo, and G. Hamdorf. 1991. Brain changes in experimental chronic hypoxia. Exp. Pathol. 42(4):205-212.
Cervos Navarro, J., R.C. Kunas, S. Sampaolo, and U. Mansmann. 1999. Heart mitochondria in rats submitted to chronic hypoxia. Histol. Histopathol. 14(4):1045-1052.
Clark, J.M., and C.J. Lambertsen. 1971. Rate of development of pulmonary O2 toxicity in man during O2 breathing at 2.0 Ata. J. Appl. Physiol. 30(5):739-752.
Clark, J.M., C.J. Lambertsen, R. Gelfand, N.D. Flores, J.B. Pisarello, M.D. Rossman, and J.A. Elias. 1999. Effects of prolonged oxygen exposure at 1.5, 2.0, or 2.5 ATA on pulmonary function in men (predictive studies V). J. Appl. Physiol. 86(1):243-259.
Clarke J.A., M.B. Daly, J.M. Marshall, H.W. Ead, and E.M. Hennessy. 2000. Quantitative studies of the vasculature of the carotid body in the chronically hypoxic rat. Braz. J. Med. Biol. Res. 33(3):331-340.
Cremona, G., R. Asnaghi, P. Baderna, A. Brunetto, T. Brutsaert, C. Cavallaro, T.M. Clark, A. Cogo, R. Donis, P. Lanfranchi, A. Luks, N. Novello, S. Panzetta, L. Perini, M. Putnam, L. Spagnolatti, H. Wagner, and P.D. Wagner. 2002. Pulmonary extravascular fluid accumulation in recreational climbers: A prospective study. Lancet 359(9303):303-309.
Cymerman, A., A.J. Young, T.J. Francis, D.D. Wray, D.T. Ditzler, D. Stulz, M. Bovill, and S.R. Muza. 2002. Subjective symptoms and postural control during a disabled submarine simulation. Undersea Hyperb. Med. 29(3):204-215.
Ebbing, D.D., and M.S. Wrighton, eds. 1996. Pp. 957 in General Chemistry, 5th Ed. Boston, MA: Houghton Mifflin.
Fahim, M.S., F.S. Messiha, and S.M. Girgis. 1980. Effect of acute and chronic simulated high altitude on male reproduction and testosterone level. Arch. Androl. 4(3):217-219.
Gabry, A.L., X. Ledoux, M. Mozziconacci, and C. Martin. 2003. High-altitude pulmonary edema at moderate altitude (<2,400 m; 7,870 feet): A series of 52 patients. Chest 123(1):49-53.
Gonzales, G.F., L. Rodriguez, J. Valera, E. Sandoval, and M. Garcia-Hjarles. 1990. Prevention of high altitude-induced testicular disturbances by previous treatment with cyproheptadine in male rats. Arch. Androl. 24(2):201-205.
Gosney, J.R. 1984. Effects of hypobaric hypoxia on the Leydig cell population of the testis of the rat. J. Endocrinol. 103(1):59-62.
Gustafsson, C., M. Gennser, H. Ornhagen, and G. Derefeldt. 1997. Effects of normobaric hypoxic confinement on visual and motor performance. Aviat. Space Environ. Med. 68(11):985-992.
Hackett, P.H., and R.C. Roach. 2001. High-altitude illness. N. Engl. J. Med. 345(2):107-114.
Hagar, R. 2003. Submarine Atmosphere Control and Monitoring Brief for the COT Committee. Presentation at the First Meeting on Emergency and Continuous Exposure Guidance Levels for Selected Submarine Contaminants, January 23, 2003, Washington, DC.
Hawley, G.G. 1977. Oxygen. Pp. 643-644 in The Condensed Chemical Dictionary, 9th Ed. New York: Van Nostrand Reinhold Co.
Hochachka, P.W., J.L. Rupert, and C. Monge. 1999. Adaptation and conservation of physiological systems in the evolution of human hypoxia tolerance. Comp. Biochem. Physiol. A. Mol. Integr. Physiol. 124(1):1-17.
Hodkinson, P.D., B.J. Hunt, K. Parmar, and J. Ernsting. 2003. Is mild normobaric hypoxia a risk factor for venous thromboembolism? J. Thromb. Haemost. 1(10):2131-2133.
Honigman, B., M.K. Theis, J. Koziol-McLain, R. Roach, R. Yip, C. Houston, L.G. Moore, and P. Pearce. 1993. Acute mountain sickness in a general tourist
population at moderate altitudes. Ann. Intern. Med. 118(8):587-592.
HSDB (Hazardous Substances Data Bank). 2004. Oxygen (CASRN 7782-44-7) TOXNET, Specialized Information Services, U.S. National Library of Medicine, Bethesda, MD. [Online]. Available: http://toxnet.nlm.nih.gov/cgibin/sis/htmlgen?HSDB [accessed January 22, 2004].
Huang, S.Y., L.G. Moore, R.E. McCullough, R.G. McCullough, A.J. Micco, C. Fulco, A. Cymerman, M. Manco-Johnson, J.V. Weil, and J.T. Reeves. 1987. Internal carotid and vertebral arterial flow velocity in men at high altitude. J. Appl. Physiol. 63(1):395-400.
Hultgren, H. 1997. Physiological effects of high altitude. Pp. 1-11 in High Altitude Medicine. San Francisco, CA: Hultgren Publications.
Karlin, J.E., and J.F. Curtis. 1945. Observations on Efficiency of Submarine Personnel during Prolonged Submergence When the Atmospheric Oxygen is Maintained at 17% and the Carbon Dioxide at 3%. AD 639625. Naval Submarine Medical Research Lab, New London, CT.
Kleinsasser, A., D.L. Levin, A. Loeckinger, and S.R. Hopkins. 2003. A pig model of high altitude pulmonary edema. High Alt. Med. Biol. 4(4):465-474.
Klokker, M., A. Kharazmi, H. Galbo, I. Bygbjerg, and B.K. Pedersen. 1993. Influence of in vivo hypobaric hypoxia on function of lymphocytes, neutrocytes, natural killer cells, and cytokines. J. Appl. Physiol. 74(3):1100-1106.
Knight, D.R., A. Cymerman, J.A. Devine, R.L. Burse, C.S. Fulco, P.B. Rock, D.V. Tappan, A.A. Messier, and H. Carhart. 1990a. Symptomatology during hypoxic exposure to flame-retardant chamber atmospheres. Undersea Biomed. Res. 17(1):33-44.
Knight, D.R., C.L. Schlichting, C.S. Fulco, and A. Cymerman. 1990b. Mental performance during submaximal exercise in 13 and 17% oxygen. Undersea Biomed. Res. 17(3):223-230.
Kobayashi, T., and S. Murakami. 1972. Blindness of an adult caused by oxygen. JAMA 219(6):741-742.
Kraaier, V., A.C. Van Huffelen, and G.H. Wieneke. 1988. Quantitative EEG changes due to hypobaric hypoxia in normal subjects. Electrencephalogr. Clin. Neurophysiol. 69(4):303-312.
LaManna, J.C., L.M. Vendel, and R.M. Farrell. 1992. Brain adaptation to chronic hypobaric hypoxia in rats. J. Appl. Physiol. 72(6):2238-2243.
Levine, B.D., K. Kubo, T. Kobayashi, M. Fukushima, T. Shibamoto, and G. Ueda. 1988. Role of barometric pressure in pulmonary fluid balance and oxygen transport. J. Appl. Physiol. 64(1):419-428.
Li , X.Y., X.Y. Wu, C. Fu, X.F. Shen, Y.H. Wu, and T. Wang. 2000a. Effects of acute mild and moderate hypoxia on human mood state. Space Med. Med. Eng. (Beijing) 13(1):1-5.
Li, X.Y., X.Y. Wu, C. Fu, X.F. Shen, C.B. Yang, and Y.H. Wu. 2000b. Effects of acute exposure to mild or moderate hypoxia on human psychomotor performance and visual-reaction time. Space Med. Med. Eng. (Beijing) 13(4):235-239.
Lide, D R., ed. 1991. Pp. 14-19 in CRC’s Handbook of Chemistry and Physics, 72nd Ed. Boca Raton, FL: CRC Press.
Linde, L., C. Gustafsson, and H. Ornhagen. 1997. Effects of reduced oxygen partial pressure on cognitive performance in confined spaces. Mil. Psychol. 9(2): 151-168.
Loeppky, J.A., P. Scotto, and R.C. Roach. 1996. Acute ventilatory response to simulated altitude, normobaric hypoxia, and hypobaria. Aviat. Space Environ. Med. 67(11):1019-1022.
Loeppky, J.A., M. Icenogle, P. Scotto, R. Robergs, H. Hinghofer-Szalkay, and R.C. Roach. 1997. Ventilation during simulated altitude, normobaric hypoxia and normoxic hypobaria. Respir. Physiol. 107(3):231-239.
Lorente, M., R.M. Mirapeix, M. Miguel, W. Longmei, D. Volk, and J. CervosNavarro. 2002. Chronic hypoxia induced ultrastructural changes in the rat adrenal zona glomerulosa. Histol. Histopathol. 17(1):185-190.
Meehan, R.T. 1986. Renin, aldosterone, and vasopressin responses to hypoxia during 6 hours of mild exercise. Aviat. Space Environ. Med. 57(10 Pt 1):960-965.
Montgomery, A.B., J. Mills, and J.M. Luce. 1989. Incidence of acute mountain sickness at intermediate altitude. JAMA 261(5):732-734.
Moore, L.G. 2000. Comparative human ventilatory adaptation to high altitude. Respir. Physiol. 121(2-3):257-276.
Nicholas, J.S., D.T. Lackland, M. Dosemeci, L.C. Mohr Jr., J.B. Dunbar, B. Grosche, and D.G. Hoel. 1998. Mortality among US commercial pilots and navigators. J. Occup. Environ. Med. 40(11):980-985.
Nichols, C.W., and C. Lambertsen. 1969. Effects of high oxygen pressures on the eye. N. Engl. J. Med. 281(1):25-30.
Ozaki, H., S. Watanabe, and H. Suzuki. 1995. Topographic EEG changes due to hypobaric hypoxia at simulated high altitude. Electroencephalogr. Clin. Neurophysiol. 94(5):349-356.
Patty, F.A. 1963. Inorganic compounds of oxygen, nitrogen, and carbon: Oxygen. Pp. 911-917 in Patty’s Industrial Hygiene and Toxicology, Vol. 2. Toxicology, 2nd Rev. Ed., D.W. Fassett, and D.D. Irish, eds. New York: John Wiley & Sons.
Piehl Aulin, K., J. Svedenhag, L. Wide, B. Berglund, and B. Saltin. 1998. Shortterm intermittent normobaric hypoxia—haematological, physiological and mental effects. Scand. J. Med. Sci. Sports. 8(3):132-137.
Raymond, L.W. 2003. Altitude pulmonary edema below 8,000 feet: What are we missing? Chest 123(1):5-7.
Roach, R.C., and P.H. Hackett. 2001. Frontiers of hypoxia research: Acute mountain sickness. J. Exp. Biol. 204(Pt. 18):3161-3170.
Roach, R.C., J.A. Loeppky, and M.V. Icenogle. 1996. Acute mountain sickness: Increased severity during simulated altitude compared with normobaric hypoxia. J. Appl. Physiol. 81(5):1908-1910.
Rodriguez, F.A., J.L. Ventura, M. Casas, H. Casas, T. Pages, R. Rama, A. Ricart,
L. Palacios, and G. Viscor. 2000. Erythropoietin acute reaction and haematological adaptations to short, intermittent hypobaric hypoxia. Eur. J. Appl. Physiol. 82(3):170-177.
Saxena, D.K. 1995. Effect of hypoxia by intermittent altitude exposure on semen characteristics and testicular morphology of male rhesus monkeys. Int. J. Biometeorol. 38(3):137-140.
Schellart, N.A., and D. Reits. 2001. Transient and maintained changes of the spontaneous occipital EEG during acute systemic hypoxia. Aviat. Space Environ. Med. 72(5):462-470.
Schweda, F., F.C. Blumberg, A. Schweda, M. Kammerl, S.R. Holmer, G.A. Riegger, M. Pfeifer, and B.K. Kramer. 2000. Effects of chronic hypoxia on renal renin gene expression in rats. Nephrol. Dial. Transplant. 15(1):11-15.
Sekhon, H.S., and W.M. Thurlbeck. 1996. Lung morphometric changes after exposure to hypobaria and/or hypoxia and undernutrition. Respir. Physiol. 106(1):99-107.
Sheedy, W., J.S. Thompson, and A.H. Morice. 1996. A comparison of pathophysiological changes during hypobaric and normobaric hypoxia in rats. Respiration 63(4):217-222.
Shukitt, B.L., and L.E. Banderet. 1988. Mood states at 1600 and 4300 meters terrestrial altitude. Aviat. Space Environ. Med. 59(6):530-532.
Shukitt, B.L., R.L. Burse, L.E. Banderet, et al. 1988. Cognitive Performance, Mood States, and Altitude Symptomatology in 13-21% Oxygen Environments. Technical Report No. T18-88. U.S. Army Research Institute of Environmental Medicine, Natick, MA (as cited in Gustafsson et al. 1997).
Shukitt-Hale, B., T. Kadar, B.E. Marlowe, M.J. Stillman, R.L. Galli, A. Levy, J.A. Devine, and H.R. Lieberman. 1996. Morphological alterations in the hippocampus following hypobaric hypoxia. Hum. Exp. Toxicol. 15(4):312-319.
Shukitt-Hale, B., L.E. Banderet, and H.R. Lieberman. 1998. Elevation-dependent symptom, mood, and performance changes produced by exposure to hypobaric hypoxia. Int. J. Aviat. Psychol. 8(4):319-334.
Sonna, L.A. 2002. Pulmonary oedema at moderately high altitudes. Lancet 359(9303):276-277.
Taylor, A.D., and R. Bronks. 1996. Effect of acute normobaric hypoxia on quadriceps integrated electromyogram and blood metabolites during incremental exercise to exhaustion. Eur. J. Appl. Physiol. Occup. Physiol. 73(1-2):121-129.
Tucker, A., J.T. Reeves, D. Robertshaw, and R.F. Grover. 1983. Cardiopulmonary response to acute altitude exposure: Water loading and denitrogenation. Respir. Physiol. 54(3):363-380.
Van der Worp, H.B., V. Kraaier, G.H. Wieneke, and A.C. Van Huffelen. 1991. Quantitative EEG during progressive hypocarbia and hypoxia. Hyperventilation-induced EEG changes reconsidered. Electroencephalogr. Clin. Neurophysiol. 79(5):335-341.
Vitzthum, V.J., and A.S. Wiley. 2003. The proximate determinants of fertility in
populations exposed to chronic hypoxia. High Alt. Med. Biol. 4(2):125-139.
Weinberger, B., D.L. Laskin, D.E. Heck, and J.D. Laskin. 2002. Oxygen toxicity in premature infants. Toxicol. Appl. Pharmacol. 181(1):60-67.
West, J.B. 1999. Oxygen enrichment of room air to improve well-being and productivity at high altitude. Int. J. Occup. Environ. Health 5(3):187-193.
Wolman, M., J. Cervos-Navarro, S. Sampaolo, and A. Cardesa. 1993. Pathological changes in organs of rats chronically exposed to hypoxia. Development of pulmonary lipidosis. Histol. Histopathol. 8(2):247-255.



























