8
Chemistry-Based Identification for Detect-to-Warn Applications
The characterization technologies discussed in this chapter are those that respond to the chemical composition of the target molecules rather than to their three-dimensional structure or biological activity. The discussion covers several distinct classes of molecules present in biological agent cells, including the following:
-
Proteins. Many studies have evaluated the feasibility of identifying microorganisms by detecting the proteins present within or on the surface of the organisms. Peptides1 and combinations of peptides may be unique to certain classes of microorganisms and can also be detected.
-
Lipids. The membranes that surround cells contain high concentrations of lipids, usually closely associated with proteins. The lipid and protein composition varies from one type of microorganism to another.
-
Carbohydrates. Chains of differing carbohydrate molecules, termed polysaccharides, are also found on the surface of cells and may be used to identify distinct varieties of microorganisms.
-
Metabolites. These smaller molecules serve as intermediates in metabolic pathways, energy sources, communicators within a cell and between cells, and regulators of activity, and are involved in many other crucial metabolic functions. Rapid sensing of metabolites could potentially be used to determine that a biological threat is present. Although metabolites are typically not unique to any specific type of cell and are therefore not useful for identifying a particular class of microorganism, one exceptional small metabolite—dipicolinic acid—is diagnostic of some spore-forming microorganisms such as Bacillus anthracis, the bacterium that causes anthrax.
-
Nucleic acids. DNA is the blueprint for all biological activity within most organisms. The presence of DNA and/or RNA can be used to indicate the presence of biological versus nonbiological materials. One prevalent type of RNA, messenger RNA (mRNA), is used as a template for protein synthesis within cells. This type of RNA is rapidly synthesized and degraded within cells, with a turnover rate of only 2 or 3 minutes in microorganisms.2 The presence of high levels of mRNA can therefore be used as an indicator of cell activity and viability. Dormant cells, such as spores, would not have high levels of mRNA but would still contain other types of nucleic acids such as ribosomal RNA (rRNA) and DNA.
Chemical methods of identifying these target molecules include techniques based on molecular mass, composition, functional groups, relative affinity for various surfaces, or other chemical properties. These methods may be complementary to alternative techniques for identifying the same classes of molecules discussed in other chapters (e.g., immunoassays for proteins, discussed in Chapter 7).
Several centuries of effort by many tens of thousands of chemists have gone into developing the equipment in a modern analytical chemistry laboratory. While the full complement of technologies could be useful for bioagent detection or identification, a review of the techniques most often used indicates that only two or three technologies stand out as the most promising candidates for routine rapid bioagent detection. They are discussed below.
MASS SPECTROMETRY
Perhaps the most fundamental characteristic of a chemical component is its intrinsic molecular weight or mass. Mass spectrometers characterize molecules by taking advantage of differences in their mass. A simple metabolite may have a mass of several hundred daltons, while some of the large proteins have a mass well over a million daltons. Mass spectrometers can be among the most complex instruments in a chemical laboratory, yet they are highly versatile and are heavily used for many chemical analyses. (See Box 8.1 for a brief primer on mass spectrometers.) Over the past decade, many investigators have explored the application of mass spectrometry to the identification of biothreat agents.
Among the most definitive molecular discriminators of potential biothreat agents are proteins and distinctive lipid, peptide, and polysaccharide components of bacterial membranes and cell walls. Of the several thousand different proteins in a typical bacterium, some may be unique to a particular strain or subspecies and would therefore have excellent potential for the identification of the strain. Other proteins may be common to multiple species of dangerous microorganisms and thus serve as useful flags for the presence of these organisms.
Because of their relatively high cost and complexity, mass spectrometers are better suited to the identification of biological agents rather than their detection. Several different approaches have been taken to the identification of proteins and other complex cell components by mass spectrometry. One, often termed "biomarker fingerprinting," involves generating a mass spectrum of a prepared sample and attempting to match it against a previously collected library of mass spectra of known organisms. This method is the one most often proposed for bioagent identification by mass spectrometry. In this approach, it is not necessary to know the identity or function of the proteins or other complex molecules that are responsible for the signal. Like a fingerprint, it is the pattern of signals that is identified and compared with a list of potential suspects.
|
Box 8.1 The separation of molecules based on their mass is, in principle at least, fairly straightforward. If each molecule in a mixture is given a push in the same direction, the lighter ones will move faster and the heavier ones slower, effecting a separation in space. Alternatively, a force can be applied to a mixture of moving molecules. The lighter ones will be more strongly affected by the force and the heavier ones less affected, due to their inertia, again resulting in a separation. Mass spectrometers of all varieties function on these simple principles and operate in a vacuum to avoid collisions of the target molecules with the nitrogen and oxygen molecules in air. Mass spectrometers initially put an electric charge on the molecules to form ions. These ions can then be manipulated by an electric or magnetic field, providing the force needed to accelerate them and steer them into a desired path. In a time-of-flight (TOF) mass spectrometer, the same charge is put on all the molecules, and they are accelerated in a straight line so that they fly down an evacuated tube. By measuring how long they take to reach a detector at a fixed position, and taking into account the length of the flight tube and the charge on the ions, one can obtain a mass spectrum that plots the |
|
variation in mass versus the number of ions detected at each mass. Other varieties of mass spectrometers are frequently used in chemistry and physics laboratories. Some separate ions by injecting ions into a curved electric field. Varying the field in a well-defined way can force unwanted ions out of the pathway to a detector, effectively allowing the detection of only ions of a specific desired mass. Major classes of mass spectrometers that operate in this fashion are termed quadrupole mass spectrometers and ion-trap mass spectrometers. Several varieties of hybrid mass spectrometers exist. One example is a combination of a quadrupole mass spectrometer with a time-of-flight spectrometer. One of the reasons for the popularity of these systems is that the first mass spectrometer can be used to select a small range of masses, rejecting the bulk of other masses that could interfere with subsequent detection. The selected ions are then separated with high resolution by the second mass spectrometer. These systems are also designed to break larger ions into smaller ones, thus providing information about the structure of the original molecule. This two-spectrometer technique is known as tandem mass spectrometry and also as MS/MS. Any mass spectrometer requires a method of forming ions from the molecules in the sample. Two primary ionization methods are in common use for proteins and other large molecules that might be diagnostic of biothreat agents. One is termed matrix-assisted laser desorption and ionization, usually referred to as MALDI. In MALDI, the sample—perhaps a protein mixture in the case of biothreat agents—is mixed with a larger quantity of an organic molecule (the matrix). The matrix is selected for its ability to efficiently absorb radiation from a laser. When the dried sample-matrix mixture is exposed to a laser beam, the matrix absorbs the laser energy and transfers it to the sample, typically forming positive ions with a single charge. These charged molecules are separated and detected in a mass spectrometer. MALDI experiments most frequently use a TOF mass spectrometer in a system usually known as MALDI-TOF. A second method of ionizing the sample is known as electrospray ionization. In this method, a liquid sample is continuously aerosolized into a fine spray near a needle maintained at high electrical potential. The droplets take on a charge from the electric field. As the charged droplets evaporate, the charge is transferred to proteins or other organic molecules in the sample, forming ions that can be separated by a mass spectrometer. Unlike MALDI, which produces mostly ions with a single charge, electrospray ionization results in ions with many charges on them. This characteristic has both advantages and disadvantages. One advantage is that mass spectrometers separate ions by their mass-to-charge ratio rather than by their mass only. This means that large, heavy, multiply-charged ions are detected at the same mass as small singly-charged ions. Many types of mass spectrometers are easier to design and build for these lower mass ions and have better sensitivities than they do for high-mass ions. The disadvantage is that a single parent molecule in an electrospray system may be present in many different charge states, resulting in a complex spectrum. While modern software is effective in dealing with this complexity and calculating the mass of the parent molecule, this is still a characteristic that one would rather not deal with for the rapid detection of bioagents, particularly in a complex environmental sample. Electrospray applications to whole proteins derived from lysed cellular components have been limited. As a result, the vast majority of protein applications first use enzymes to break the protein down into small peptides, which are ionized efficiently. Liquid chromatography equipment is then used to remove compounds that might interfere in the analysis and to separate the resulting peptide mixture. These steps add time and complexity to the sample preparation and probably explain why electrospray ionization is rarely used for the mass spectrometry of biothreat microorganisms. Well-organized tutorials on mass spectrometry can be found on the Web sites of the American Society of Mass Spectrometrya and the British Mass Spectrometry Society.b
|
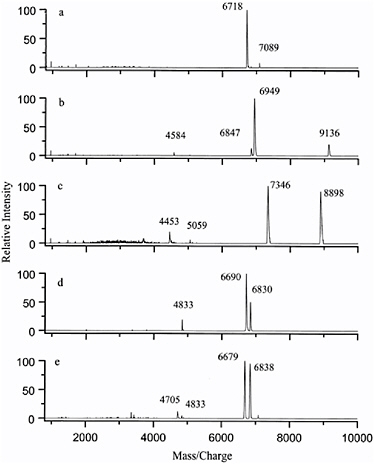
FIGURE 8.1 Comparion of mass spectra from various Bacillus species: (a) B. thuringiensis, (b) B. subtilis, (c) B. globigii, (d) B. cereus, and (e) B. anthracis Sterne.
Research into the mass spectrometric methods for the characterization of microorganisms is an active field today, with several reviews available.3Figure 8.1 illustrates mass spectra of 5 different species of Bacillus bacteria. The first four species are not normally considered to be harmful, and the last is the Bacillus anthracis species responsible for anthrax. Note first that each mass spectrum is different. While the first four are very different from one another and might be expected to be easily differentiated, spectra (d) and (e) are similar but still different in detail. It is interesting that spectrum (d) is of Bacillus cereus, a bacterium that is known to be very similar to Bacillus anthracis from a microbiology standpoint but that does not have the lethal effects of B. anthracis. It should also be noted that no attempt was made to identify the molecular source of the signals in the mass spectra. With additional effort it is possible to identify many of the biomarker components,4 but this is not necessary for fingerprint matching.
Bacterial cells also contain characteristic lipids that vary in composition among species. These lipids may be rapidly converted to volatile forms that can be ionized for mass spectrometry characterization. This method has been shown to differentiate successfully among species and strains within species if pure bacterial samples are used.5 However, it is not likely to be selective enough to identify a hostile organism in a mixture with benign background organisms. This limitation probably precludes its use for rapid bioagent identification. For the biomarker fingerprinting approach, the mathematical procedures used to match the observed spectra with the library of candidate microorganism spectra are every bit as important as the mass spectrometry itself, and this is an area of active research.6
|
3 |
B.L.M. van Baar. 2000. Characterisation of bacteria by matrix-assisted laser desorption/ionization and electrospray mass spectrometry. FEMS Microbiology Reviews 24:193-219. C. Fenselau and P.A. Demirev. 2001. Characterization of intact microorganisms by MALDI mass spectrometry. Mass Spect. Rev. 20:157-171 J.O. Lay. 2000. MALDI-TOF mass spectrometry and bacterial taxonomy. Trends Anal. Chem. 19:507-516. |
|
4 |
R.D. Holland, C.R. Duffy, F. Rafii, J.B. Sutherland, T.M. Heinze, C.L. Holder, K.J. Voorhees, J.O. Lay. 1999. Identification of bacterial proteins observed in MALDI TOF mass spectra from whole cells. Anal. Chem. 71(15):3226-3230. P.A Demirev, J. Ramirez, and C. Fenselau. 2001. Tandem mass spectrometry of intact proteins from characterization of biomarkers from Bacillus cereus T spores. Anal. Chem. 73(23):5725-5731. B. Amiri-Eliasi and C. Fenselau. 2001. Characterization of protein biomarkers desorbed by MALDI from whole fungal cells. Anal. Chem. 73(23):5228-5231. |
|
5 |
F. Basile, M.B. Beverly, C. Abbas-Hawks, C.D. Mowry, and K.J. Voorhees. 1998. Direct mass spectrometric analysis of in situ thermally hydrolyzed and methylated lipids from whole bacterial cells. Anal. Chem. 70:1555-1562. |
|
6 |
Fenselau and Demirev, 2001. See note 3 above. P.A. Demirev, J.S. Lin, F.J. Pineda, and C. Fenselau. 2001. Bioinformetrics and mass spectrometry for microorganism |
A second approach to mass spectrometric identification is to target specific proteins known to be present in biothreat agents. A prime example might be the detection of botulinum toxin, a biothreat agent that can be released as a relatively pure protein rather than as an infectious organism.7 In this case the detector is focused on a specific molecule, and its detection is an immediate warning that the threat agent is present. The disadvantage of this approach is that the protein may be difficult to detect rapidly in the presence of other proteins that might be present in the environment. A variety of chemical purification methods are available for this circumstance, but using them increases detection time and instrumental complexity. These factors probably exclude this approach from consideration for rapid identification.
In matrix-assisted laser desorption and ionization time-of-flight (MALDI-TOF) mass spectrometry (see Box 8.1), one can optimize the system and procedures to partially classify whole proteins. Typically, only a few of the many proteins in a sample are detected. Alternatively, the microorganism can be exposed to an enzyme that cuts the proteins into smaller peptides in a somewhat reproducible and predictable manner. These peptides, usually in the mass range of 1,000 to 5,000 daltons, are often ionized and detected with MALDI. The pattern of detected masses is then used in a fingerprinting mode for identification of many of the proteins in the sample. A characteristic of this latter approach is that it requires multiple and sometimes complex sample preparation steps before a sample is suitable for analysis. The time required for an approach that includes a protein digestion step (typically more than 15 minutes) probably excludes it from use in detect-to-warn applications.
Early MALDI work with microorganisms involved isolation of a particular species from a carefully prepared bacterial culture, chemical breakdown of the cell membranes to expose the internal cell structures and molecules, and analysis of the resulting complex solution in the mass spectrometer. However, more recent work has shown that much of this effort can be avoided by directly analyzing the intact bacterial cells. The cells are mixed with the MALDI matrix, dried, and analyzed immediately without intervening complex chemical procedures.8
While the number of cellular constituents detected by this method may be small in comparison with the total number of molecules in the cell, the spectral fingerprints obtained have been demonstrated to be sufficient for the discrimination among similar bacterial and viral species and even among different strains of Bacillus anthracis spores9 as long as the target organism is analyzed in the absence of major concentrations of background organisms. Most of the current efforts in the use of mass spectrometry for bioagent detection are taking this approach.
The currently fielded chemical biological mass spectrometer (CBMS) Block III, a component of the Biological Integrated Detection System (BIDS), uses a fingerprint approach. Rather than characterizing intact organisms with MALDI, however, this system rapidly pyrolyzes (vaporizes with high temperature) cellular components from a collected aerosol, ionizes them with an electron impact device, and generates
|
|
identification: Proteome-wide post-translational modifications and database search algorithms for characterization on intact H. pylori. Anal. Chem. 73:4566-4573. R.J. Arnold and J.P. Reilly. 1998. Fingerprint matching of E. coli strains with matrix-assisted laser desorption/ionization time-of-flight mass spectrometry of whole cells using a modified correlation approach. Rapid Commun. Mass. Spectrom. 12:630-636. K.H. Jarman, S.T. Cebula, A.J. Saenz, C.E. Petersen, N.B. Valentine, M.T. Kingsley, and K.L. Wahl. 2000. An algorithm for automated bacterial identification using matrix-assisted laser desorption/ionization mass spectrometry. Anal. Chem. 72:1217-1223. J.J. Bright, M.A. Claydon, M. Soufian, and D.B. Gordon. 2002. Rapid typing of bacteria using matrix-assisted laser desorption ionization time-of-flight mass spectrometry and pattern recognition software. J. Microbiol. Meth. 48:127-138. W.A. Bryden, Johns Hopkins University. Presentation to the committee on September 25, 2002. |
a mass spectral fingerprint in an ion trap mass spectrometer. While potentially a useful rapid screening tool for biological agents on the battlefield, it is unlikely that this system will have the specificity needed for the extremely low false alarm rates required of a rapid identifier in a detect-to-warn architecture. The pyrolysis fragmentation pattern is a function of many variables, and it has not been demonstrated that these signatures offer sufficient discrimination in a complex background to support a 10-6 false alarm rate.
Challenges for Rapid, Simple-to-Use Mass Spectrometry Identification Systems
MALDI-TOF mass spectrometers are used routinely in laboratories to identify proteins. It should be recognized, however, that these samples have been previously purified such that they contain only a few distinct proteins. Even with the very best-performing laboratory MALDI mass spectrometers, it is difficult to achieve identification of unknown proteins when more than a few proteins are present in the sample. Typical indoor aerosol samples could contain hundreds of different species of microorganisms (see Chapter 3), including both viable and nonviable bacteria, spores, algae, fungi, and viruses that are associated with particles in the 1- to 30-micrometer range. While at any one time only about 10 to 20 different species may be identifiable by techniques requiring cell culturing, many more species are expected to be present that are not culturable by standard microbiological methods. Mass spectrometry approaches could be used to detect the proteins of all organisms, both culturable and nonculturable. Preliminary investigations into identification of mixtures containing a few different well-known species show some progress,10 but the number of organisms used for these studies is very small in comparison with the number that may be found in real-world samples. The committee is aware of recent work by DARPA assessing the performance of MALDI for agent identification in samples containing significant concentrations of background organisms. Combined efforts are being applied in areas of sample preparation, the MALDI ionization procedures, and detection algorithms. Summary data are expected to be available by the time this report is published.
Several reviewers have noted that use of MALDI for microorganism identification is made more difficult by spectral complexities and by loss of signal if inorganic salts are present in the sample.11 Many of the locations in which detect-to-warn identification systems might be deployed are in coastal areas likely to contain large concentrations of salts in the sampled aerosol. However, simple washing can be used to wash away most of the interfering salts and leave the organisms and proteins on the sample holder. Other sample preparation processes could also be implemented to remove salts.12 For detect-to-warn applications, it would be necessary to develop methods for sample washing/processing that are extremely rapid (accomplished in seconds rather than minutes).
An additional challenge is the degree of reproducibility required for the fingerprints in order to achieve accurate identification. A pioneer in intact microorganism identification with MALDI reports that it is often difficult to obtain reproducible spectra, even from the same organism.13 Other investigators have reported that selected mass spectrometer signals can be identified that are reproducible in different laboratories.14 One assessment using a commercial microorganism MALDI detection system for pure
|
10 |
Jarman et al., 2000. See note 6 above. K.L. Wahl, S.C. Wunschel, K.H. Jarman, N.B. Valentine, C.E. Petersen, M.T. Kingley, K.A. Zartalas, and A.J. Saenz. 2002. Analysis of microbial mixtures by matrix-assisted laser desorption/ionization time-of-flight mass spectrometry. Anal. Chem. 74:6191-6199. |
|
11 |
J.O. Lay. 2000. MALDI-TOF mass spectrometry and bacterial taxonomy. Trends in Anal. Chem. 19:507-516. B.L.M. van Baar, TNO Prins Maurits Laboratory. Presentation to the committee on September 25, 2002. |
|
12 |
M. Kussman, E. Nordhoff, H. Rahbek-Nielsen, S. Haebel, M. Rossel-Larsen, L. Jokobsen, J. Gobom, E. Mirgorodskaya, A. Kroll-Kristensen, L. Palm, and P. Roepstorff. 1997. Matrix-assisted laser desorption/ionization mass spectrometry sample preparation techniques designed from various peptide and protein analytes. J. Mass Spectrometry 32:593-601. |
|
13 |
J. Lay, 2000. See note 11 above. |
|
14 |
Z. Wang, K. Dunlop, S.R. Long, and L. Li. 2002. Mass spectrometric methods for generation of protein mass database used for bacterial identification. Anal. Chem. 74:3174-3182. |
microorganisms under tightly controlled conditions stated that MALDI could not properly identify a quarter of the samples separately identified by the gold-standard PCR sequence method.15
In comparing the direct MALDI analysis of intact microorganisms with more complex methods using MALDI or electrospray ionization mass spectrometry after a chemical separation, it has been found that the direct MALDI method is more prone to interference from other components in the sample and provides many fewer fingerprint components from which to identify the microorganism.16 However, progress has been made in using the MALDI mass spectrometer (MS) for the direct identification of bacterial species in mixtures of up to five organisms,17 and the differentiation between two strains of Staphylococcus aureus has also been demonstrated using MALDI-MS.18 In spite of this recent progress in developing MALDI-MS methods for bacterial identification, the characteristics and conditions that produce reproducible fingerprints from intact microorganisms using MALDI, the best procedures for matching the fingerprints with libraries of fingerprints of known organisms, and the level of bacterial identification (e.g., species, strain) that can be achieved in complex environmental samples are not well understood at the present time.
It has been suggested that since the individual biothreat agents in aerosols are separated into discrete particles that can be characterized individually, a system could be devised that first detects individual biological particles in a moving aerosol stream by optical methods19 and feeds only selected individual particles into the MALDI spectrometer. It is claimed that in this way, potential interference resulting from the presence of many different organisms in a sample for MALDI mass spectrometry may be avoided.20 While this suggestion is indeed interesting, the committee is aware of only preliminary data indicating that this approach might be successful. A major challenge will be the sensitivity of detection. Laboratory-based MALDI-TOF systems typically require on the order of 10 to 100 separate detection events to achieve sufficient signal to allow molecule identification. The direct aerosol sampling approach would necessitate identification using only a single detection event. While this could potentially be a powerful tool to obtain near-instantaneous identification of organisms, further laboratory work is needed to determine if direct MALDI-MS analysis of individual aerosol particles is a viable approach for rapid pathogen identification.
An additional challenge for continuous-use detect-to-warn applications is the instrumentation complexity. While mass spectrometry is certainly one of the mainstays of today's chemistry and biotechnology laboratories, current laboratory MALDI-TOF mass spectrometers do not come in small easy-to-use packages. They typically are benchtop- or floor-mounted systems at least the size of several three-drawer file cabinets. The MALDI-TOF contains a significant amount of hardware: a moderate-power laser, an extensive vacuum system, multiple power supplies running high voltages, and very high speed electronics. With this mechanical and electronic complexity comes cost. A typical laboratory system having performance appropriate for peptide mass fingerprinting sells for around $250,000, although several groups have reported results for intact protein application using more compact, less expensive systems.21 Modern MALDI-MS systems that are commercially available are very reliable but—as with any analytical instrumentation—they require a regular maintenance schedule for reliable operation.
|
15 |
Z. Du, R. Yang, Z. Guo, Y. Song, and J. Wang. 2002. Identification of Staphylococcus and determination of its methicillin resistance by matrix-assisted laser desorption/ionization time-of-flight mass spectrometry. Anal. Chem. 74:5487-5491. |
|
16 |
Wang et al., 2002. See note 14 above. |
|
17 |
Wahl et al., 2002. See note 10 above. |
|
18 |
K. Bernardo, N. Pakulat, M. Macht, O. Krut, H. Seifert, S. Fleer, F. Hunger, and M. Kronke. 2002. Identification and discrimination of Staphylococcus aureus strains using matrix-assisted laser desorption/ionization-time of flight mass spectrometry. Proteomics 2:747-753. |
|
19 |
M.A. Stowers, A.L. van Wuijckhuijse, J.C.M. Marijnissen, B. Scarlett, B.L.M. van Baar, and Ch.E. Keintz. 2000. Application of matrix-assisted laser desorption/ionization to on-line aerosol time-of-flight mass spectrometry. Rapid Commun. Mass Spectrom. 14:829-833. |
|
20 |
van Baar, 2002. See note 11 above. |
|
21 |
See, for example, P.A. Demirev, Y.P. Ho, V. Ryzhov, and C. Fenselau. 1999. Microorganism identification by mass spectrometry and protein database searches. Anal. Chem. 71:2732-2738. |
Of course, the challenge of reliable operation is much greater if the intended use of the mass spectrometer is for detect-to-warn applications that require constant operation in an unattended fashion. Achieving this level of reliability with the complexity of a mass spectrometer would require additional instrument engineering.
Active efforts are under way to scale down and simplify MALDI-TOF systems for bioagent detection applications.22 Efforts have also been directed toward miniaturizing the components of mass spectrometers.23 However, the scaling laws for mass spectrometry dictate that there will be a reduction in performance as the spectrometer dimensions are reduced. Thus, levels of performance achieved in laboratory systems are not likely to be reproduced in these smaller packages. This is exemplified by the demonstration of a mass resolution of about 440 (mass/delta mass) at a mass of 1,675 daltons for a well-known miniaturized system.24 This mass resolution is about 30 times poorer than that shown for a similar mass by a conventional laboratory MALDI mass spectrometer.25 Poorer mass resolution means poorer accuracy in the identification of microorganisms by their biomarker fingerprint. While the development of compact mass spectrometers for bioagent detection could provide valuable insights into future directions, the committee believes that a higher priority for practical detect-to-warn scenarios would be to demonstrate that the best available laboratory equipment is capable of accurately identifying important bioagents in the presence of naturally occurring microorganisms found in real-world environments. Once the basic capability is established in the laboratory, it will be clearer what performance trade-offs might be acceptable in simpler, more compact systems.
GAS- AND LIQUID-PHASE SEPARATIONS FOR PATHOGEN DETECTION
While mass spectrometers are widely used in chemical laboratories, methods for separating molecules in flowing vapor or liquid streams are nearly ubiquitous. In these methods the sample streams flow through a column packed with material that interacts reversibly with molecules in the sample. The molecules that interact strongly with the packing move more slowly through the column than those that have weak interactions. This process, called chromatography, enables the separation of complex chemical mixtures. Both gas chromatography (separation of volatile molecules in the gas phase) and liquid chromatography (separation in the liquid phase) are used for a wide variety of chemicals and may be applied to some classes of bioagents. Electrophoresis, in which liquid-phase separations are driven by electric fields, is another frequently used method.
Protein toxins such as botulinum toxin or ricin—a highly toxic protein extracted from the castor bean plant—can be separated from many other proteins by liquid chromatography or electrophoresis. In some complex mixtures of proteins with similar chemical characteristics, however, the separation is not as clean. To address these cases, one can combine two or more different separation methods to provide sufficient selectivity. A major effort in liquid separations taking this approach is a handheld system known as μChemLab.26 A recent version is capable of detecting a lethal dose of ricin starting with an aerosol sample, and new developments are under way that will increase the sensitivity by another factor of 100 needed to detect botulinum toxin in lethal doses.27 Other groups are also investigating the use of new fluorescent dyes to achieve sensitive detection of proteins after separation using microfluidic systems.28
|
22 |
T.J. Cornish and W. A. Bryden. 1999. Miniature time-of-flight mass spectrometer for a field-portable biodetection system. Johns Hopkins APL Technical Digest. 20:335-342. W.A. Bryden, Johns Hopkins University. Presentation to the committee on September 25, 2002. R.D. English and R.J. Cotter. 2003. A miniaturized matrix-assisted laser desorption/ionization time of flight mass spectrometer with mass-correlated acceleration focusing. J. Mass Spectrom. 38:296-304. |
|
23 |
E.R. Badman and R.G. Cooks. 2000. Miniature mass analyzers. J. Mass Spectrom. 35:659-671. |
|
24 |
English and Cotter, 2003. See note 22 above. |
|
25 |
R.J. Cotter. 1999. The new time-of-flight mass spectrometry. Anal. Chem. 71:445A-451A. |
|
26 |
D. Lindner. 2001. The μChemLab Project: Micro Total Analysis System R&D at Sandia National Laboratories. Lab on a Chip 1, 15N-19N. |
|
27 |
D. Lindner, Sandia National Laboratories. Presentation to the committee on September 25, 2002. |
|
28 |
Y.J. Liu, R.S. Foote, S.C. Jacobson, R.S. Ramsey, and J.M. Ramsey. 2000. Electrophoretic separation of proteins on a microchip |
Capillary electrophoretic separation, fluorescent labeling, and detection are currently achieved in 5 minutes.29 Aerosol sampling will require some additional time, and very dirty aerosol samples may require additional processing steps to achieve distinguishable detection peaks for toxin identification. Additional work is required to determine the reliability of this method for toxin identification in environmental aerosol samples. This method has the potential to be very rapid (2 minutes or less) for the detection of toxins in relatively simple samples such as aerosols that require minimal sample processing prior to separation, labeling, and detection of toxins.
A gas-phase version of μChemLab has been under development for a number of years. While this effort has been primarily directed toward the detection of chemical agents, recent work has evaluated the detection of cellular fatty acid components after chemical treatment to make them sufficiently volatile to be separated by gas chromatography. Gas chromatography of volatilized fatty acids for bacterial identification is well known, but these procedures are nearly always performed on pure samples of the microorganism isolated during lengthy cell culturing procedures that are not practical for rapid bioagent detection. Thus, while the achievement of sensitive protein separations in a handheld system represented by the μChemLab program is impressive, it is still an open research question whether it will be able to perform protein fingerprinting of viruses and bacteria in real-world samples.
A system that combines aerosol pyrolyses with gas chromatography and ion mobility spectrometry has been developed into a fieldable unit.30 Ion mobility, like mass spectrometry, segregates ions by their velocity, but in a molecular gas rather than in a vacuum. While it does not have the selectivity of mass spectrometry, it is may be somewhat simpler in design. Still, complexity and reliability issues for systems operated on a continuous basis without routine maintenance are potential limitations. Further, performance of these systems in environments containing large numbers of naturally occurring microorganisms has not been demonstrated. Therefore, as with MALDI-MS identification of bioagents, work is needed to determine the suitability of this approach for the identification of bioagents in the presence of naturally occurring microorganisms and other aerosols found in the environments that are being protected.
CHEMICAL SENSORS
As discussed in the introduction, biological organisms and molecules display chemical characteristics that can be exploited to detect and identify them through a variety of chemical sensors. Chemical sensors are versatile and available in many configurations that can be small in size, rapid in reporting information, and low in cost. If such sensors could be made reliable enough and cheap enough, it might be possible to use them as biological “smoke detectors” and distribute them widely throughout a building or other target that needs to be protected. When highly distributed, they are likely to be closer to any potential release and hence experience a still larger concentration of agent, thereby possibly lessening the stringent demands on discriminating an agent from the ambient background. It is this interplay of highly distributed and cheaper but simpler sensors that offers an intriguing trade space.
Chemical sensors consist of two essential components: a chemically selective layer that binds the target molecule and a transducer that converts the binding event into a measurable electrical signal that can be monitored, displayed, and used for process control. Box 8.2 summarizes several common types of chemical sensor transducers and offers some considerations for the design of chemical sensor systems. More detailed information on chemical sensors can be obtained from several review articles.31
|
|
with noncovalent, postcolumn labeling. Anal. Chem. 72(19):4608-4613. |
|
29 |
Lindner, 2001. See note 26 above. |
|
30 |
A.P. Snyder, W.M. Maswadeh, J.A. Parsons, A. Tripathi, H.L.C. Meuzelaar, J.P. Dworzanski, and M.G. Kim. 1999. Field detection of bacillus spore aerosols with stand-alone pyrolysis-gas chromatography-ion mobility spectrometry. Field Anal. Chem. and Tech. 3(4-5):315-326. A.P. Snyder, W.M. Maswadeh, A. Tripathi, and J.P. Dworzanski. 2000. Detection of gram-negative Erwinia herbicola outdoor aerosols with pyrolysis-gas chromatography/ion-mobility spectrometry. Field Analytical Chemistry and Technology 4(2-3):111-126. |
|
31 |
J. Janata, M. Josowicz, P. Vanysek, and D.M. DeVaney. 1998. Chemical sensors. Anal. Chem. 70:179R-208R. |
|
Box 8.2 Chemical sensors can include a wide variety of chemically interactive surfaces and transducers for detecting the binding of chemicals to the interactive surfaces. The chemically interactive layers can be designed to be selective to specific chemicals. Examples include sensors based on antibody-antigen interactions and sensors based on sequence-specific DNA hybridization. Sensors can also be designed to include semiselective films—i.e., those that will interact to varying degrees with many chemicals. In this case, greater selectivity can be achieved by using a collection of sensors with different chemically interactive surfaces. This type of sensor array is analogous to our sense of smell. The olfactory system contains many different receptors that are not highly selective; each receptor will respond to many vapors, and a given vapor will elicit a response from many different receptors. In the same way, sensor arrays have been developed for vapor analysis in which each sensor includes a different cross-reactive layer and the fingerprint of the response from the sensor array is used to identify the vapors present. One potential advantage of a semiselective sensor array approach is that by understanding how the sensor elements interact with chemicals, it may be possible to obtain useful information about unknown samples, in order to classify the unknown and determine the potential risk it may pose. Another approach to obtaining chemical selectivity using semiselective sensors is to combine information from sensors that operate using different transduction mechanisms. These are often called hybrid sensor array systems.a In all sensor array systems, one tries to make the information reported from each of the sensors as independent as possible (perhaps using different operating principles or different modes of interactions with chemicals); otherwise, selectivity is correspondingly reduced. A complete listing of all transduction methods, chemically interactive surfaces, and combinations of sensor elements into arrays for chemical sensing is beyond the scope of this report. However, below is a brief overview of several common chemical sensor transduction principles and some important considerations for designing sensors for pathogen detection. More detailed information on chemical sensors can be obtained in review articles focused on specific aspects of chemical sensing.b Transduction Principles Three of the most common transduction principles for chemical sensing involve mass, electrochemical, and optical detection. Mass sensors can produce a signal based upon the mass of chemicals that interact with the sensing film. Acoustic wave devices are the most common sensors in this class. They are made of piezoelectric materials that bend when a voltage is applied to the crystal or that generate a voltage when they are bent. Acoustic wave sensors are typically operated by applying an oscillating voltage at the resonant frequency of the crystal and then measuring the change in resonant frequency when chemicals interact with the sensing surface. In addition to mass, other properties such as surface viscosity can also affect the resonant frequency and must be considered when interpreting sensor response data. Two common types of acoustic wave devices are thickness shear mode devices (TSMs, also called quartz crystal microbalances, QCMs) and surface acoustic wave devices (SAWs). In TSM devices, the acoustic wave propagates through the bulk of the crystal, perpendicular to the sensor surface, while in SAW devices, |
|
|
K. Haupt and K. Mosbach. 2000. Molecularly imprinted polymers and their use in biomimetic sensors. Chemical Reviews 100:2495-2504. D.T. McQuade, A.E. Pullen, and T.M. Swager. 2000. Conjugated polymer-based chemical sensors. Chemical Reviews 100:2537-2574. J.W. Grate. 2000. Acoustic wave microsensor arrays for vapor sensing. Chem. Rev. 100:2627-2648. K.J. Albert, N.S. Lewis, C.L. Schauer, G.A. Sotzing, S.E. Stitzel, T.P. Vaid, and D.R. Walt. 2000. Cross-reactive chemical sensor arrays. Chem. Rev. 100:2595-2626. P.C Jurs, G.A. Bakken, and H.E. McClelland. 2000. Computational methods for the analysis of chemical sensor array data from volatile analytes. Chem. Rev. 100:2649-2678. O.S. Wolfbeis. 2002. Fiber-optic chemical sensors and biosensors. Anal. Chem. 74:2663-2677. |
|
the acoustic wave propagates along the surface of the crystal. Electrochemical sensors involve using electrodes to measure the electrochemical changes that occur when chemicals interact with a sensing surface. The electrical changes can be based upon a change in the measured voltage between the electrodes (potentiometric), a change in the measured current at a given applied voltage (amperometric), or a change in the ability of the sensing material to transport charge (conductometric). Some of the most common types of electrochemical sensors are ion-selective electrodes (ISE) such as pH electrodes. These potentiometric sensors include a membrane that selectively passes certain ions, so that the potential drop across the membrane (and therefore the measured electrochemical potential) is dependent upon the concentration of ions in the sample. Optical sensors often employ optical fibers or planar waveguides to direct the light to the sensing film. Evanescent waves propagating from waveguides can be used to optically probe only the sensing film, and not the bulk of the sample solution, in order to decrease the optical background signal from the sample. The measured optical signals can include absorbance, fluorescence, chemiluminescence, surface plasmon resonance (to probe refractive index), or changes in light reflectivity. The most sensitive optical sensors are based upon fluorescence measurements, and with sophisticated optical equipment, it is even possible to measure fluorescence from a single molecule.c One practical limitation, however, is the limited lifetime of fluorescent molecules due to photobleaching over time. One example of an optical sensor array is a device developed by Walt and coworkersd that includes a collection of hundreds of optical fibers, each with individual polymer beads containing embedded indicator dyes attached to the end. Detection Formats An important consideration in the development of a chemical sensor is the method by which the analyte interacts with the sensor to generate the signal.e In a direct binding approach, the transducer can directly detect the analyte molecules, or the analyte displaces a reporter tag to decrease the sensor signal, or the analyte results in a change in the interactive film that is then measured by the transducer. In another format, a sandwich assay, a detector molecule such as an optical tag is added after the addition of the analyte and is designed to bind to the analyte, sandwiching the analyte between the chemically interactive surface and the optical tag. Many different direct binding and sandwich assays can be designed for use in combination with acoustic wave, electrochemical, and optical devices. Direct binding assays require only one binding step, so they are typically faster than sandwich assays, which require at least two binding steps. The use of additional tags, however, provides a means for designing a very sensitive sensor system. For instance, the tag can have a very large mass or include an immobilized enzyme that catalyzes a reaction to generate a multitude of fluorescent molecules that deposit onto an optical sensor surface for very high sensitivity. A highly desirable sensor platform for both rapid response and sensitive detection would therefore consist of a direct binding assay that generates a very large signal. This could be achieved by developing a very sensitive detector, or by designing selective films that are dramatically affected by the presence of the analyte, such that a small amount of analyte binding to the sensing surface generates a large sensor response. Sensor Reversibility and Surface Fouling Two important considerations for extended sensor operation and environmental monitoring are reversibility of the binding interaction and surface fouling. For a reversible binding interaction, a sensor response will be observed when the analyte is present but will rapidly return to baseline when the analyte is removed. Many vapor-polymer interactions used in sensor arrays for vapor analysis are reversible within seconds under normal operating conditions.f Other interactions such as antibody-antigen binding or DNA hybridization require harsh conditions (e.g., high temperatures, low pH) to remove the bound analytes. The harsh conditions can affect the quality of the interactive surface and therefore limit the lifetime of the sensor. Some sensors are designed to be used until a positive signal is |
|
obtained and then thrown away, rather than cleaned for continued use. Reuse of sensor surfaces is desirable to decrease reagent use and operation cost. Preliminary work will be needed to establish the number of times a sensor can be reused before it needs to be regenerated or replaced due to accumulation of nonspecifically bound compounds. Surface fouling can also limit the lifetime of chemical sensors. Surface fouling is the nonspecific binding of chemicals in the sample matrix onto the sensor surface. Surface fouling can result in false positive signals or completely destroy the operation of the sensor by interfering with analyte-sensing surface interactions, giving false negative signals. In some cases, it is possible to clean sensor surfaces periodically to remove contamination. If chemical cleaning is used to remove analytes and nonspecifically bound molecules, one must ensure that the sensor is not compromised. A method for automated cleaning and storage of cleaning materials within the sensor system may need to be developed prior to deployment of a sensor system. If continuous or periodic monitoring over extended periods of time is desired, it is often necessary to engineer the sensor system to produce a new sensing surface automatically for each measurement, or to replace the sensing surface periodically as surface fouling occurs. This can be achieved by using many different engineering approaches. For example, a moving tape is being used for collecting each aerosol sample within the MALDI-TOF mass spectrometry system under development at Johns Hopkins Applied Physics Laboratory.g This same approach could be used to produce a moving interactive chemical sensor surface for chemical sensing. Another approach that has been used to mitigate problems with sensor reversibility and surface fouling is the use of beads as interactive surfaces within a sensor system. Fresh interactive beads can be fluidically delivered to the transducer before each measurement and removed from the sensor system after each measurement.h The beads are therefore disposed of after each measurement (or periodically after surface fouling buildup), and only pristine interactive surfaces are used for detection. Problems with limited sensor lifetime and surface fouling can sometimes also be minimized by adding sample preparation steps that remove chemicals that cause problems, before they have a chance to damage the sensing surface or interfere with the sensor response. This overview of chemical sensors only touches the surface regarding the multitude of chemically interactive films, chemical transducers, sensor arrays, and other considerations that must be taken into account when developing chemical sensor systems. In order to develop effective chemical sensors, it is critical to first understand the operating requirements and sample type, and to consider the complete system, from sample acquisition through detection and data analysis, since these requirements can dramatically change the selection of the chemical sensor system and sensing strategy.
|
Many of the chemical sensors that have been investigated for pathogen detection are based upon the sequence-based recognition of DNA, structural recognition of pathogens or pathogen biomarkers, or cell-based function. These are discussed in Chapters 6, 7, and 9, respectively. This chapter focuses on chemical sensors for pathogen detection that respond to the chemical nature of pathogen biomarkers.
One of the challenges in the development of chemistry-based sensors for pathogen detection is the selection of pathogen biomarkers. Most chemical biomarkers discussed in this chapter are semiselective—that is, the biomarker may distinguish between general classes of microorganisms but will not be able to identify the specific species or strain of organism. For example, calcium dipicolinate is a unique component of endospores. Therefore, dipicolinic acid can be used to indicate the presence of endospores but will not be able to distinguish between dangerous Bacillus anthracis spores and other nontoxic Bacillus spores.32 An even more general indicator, such as the presence of DNA, will be useful for determining that an unknown material is biological in nature but will not be able to discriminate the source of the biological material (unless sequence-based analysis is used, as discussed in Chapter 6). Cell metabolites will also be very difficult to use as specific markers for pathogens. Metabolites are generally common to many different cell types and therefore difficult to use for discrimination between specific microorganisms. In addition, the concentration and distribution of metabolites often depend upon the nutrients available, and so can vary even for the same cell types.
In spite of the inherent limitations of semiselective pathogen detection, this approach could be useful if it is used to trigger more selective pathogen detection systems, which typically require more time and expense for each analysis. Assuming the false alarm rate can be made sufficiently low, semiselective pathogen detection could also be used to trigger initial low-regret responses—such as alterations in building ventilation—while waiting for confirmation using more selective pathogen identification systems.
It may be possible to decrease the number of false alarms by developing sensor arrays that detect multiple pathogen biomarkers simultaneously. This concept of sensor arrays has been widely used in the field of vapor analysis. In this approach, each element of the array is designed to respond to different general properties of vapors. The specific identification of the vapors present therefore relies on the use of statistical methods to identify the vapor from the fingerprint of the response from all array elements. A similar approach could be used for pathogen detection, in which each element of an array responds to different properties (e.g., pathogen biomarkers) of an organism. Such an approach would require a well-characterized environmental background signal and research to determine the fingerprints that would constitute a positive signal. If this approach is successful, it is expected that the first generations of such bio smoke detectors would be more expensive than conventional smoke alarms, so that these sensors might need to be distributed more sparsely than smoke alarms. A cost-benefit analysis will be needed to determine whether and under what conditions such a sparse network makes sense. If the analysis shows that such a network can be cost-effective and can address a significant portion of the threat space, methods will be needed for determining the optimum placement of the sensors and how to extract information from a sparse detection network.
The use of chemical sensors for semiselective pathogen detection is an area that has not been extensively investigated. Below are a few examples of chemical sensors that are being developed.
Dipicolinic Acid Analysis
An endospore (spore) is a dormant state that is exhibited by some gram-positive bacteria (e.g., Bacillus anthracis or Clostridium perfringens) when they are under stress or deprived of nutrients. Spores are particularly resistant to chemical and physical damage and can survive for decades. When the appropriate nutrients and conditions are present, endospores germinate into vegetative cells. Often the conditions within other biological systems (e.g., humans) are suitable for spore germination. For these reasons, spores represent one class of bioagents that are of special concern. As mentioned previously, one chemical marker for the presence of spores is calcium dipicolinate (dpa). Endospores typically
contain 2 to 15 percent dpa by dry weight, so this marker has the potential for sensitive detection of the presence of spores.33
In addition to pyrolysis followed by gas chromatography (GC)-MS or GC-ion mobility spectrometry, described above, several chemical sensing approaches have been reported for the detection of dpa.34 Photoluminescence of a terbium-dipicolinate complex is the most sensitive method that has been reported. This method involves first extracting the dipicolinic acid from the cells and then adding excess terbium to form a terbium-dipicolinate complex that is detected. Photoluminescence is generated by irradiating the sample at the wavelength absorbed by dpa (280 nanometers). Energy transfer from dpa to the terbium excited state dramatically enhances the terbium luminescence. Terbium luminescence contains several characteristic narrow emission peaks between 490 and 695 nanometers and has an unusually long fluorescence lifetime, so that the luminescence can be distinguished from interfering fluorescence in biological samples.35 Scientists at the U.S. Army Research Laboratory have demonstrated a limit of detection of 1,000 cfu per milliliter in a total analysis time of 5 to 7 minutes.36 This analysis time included 3 minutes for a rapid chemical extraction procedure to release virtually all of the dpa from the spores and all additional reagent additions prior to photoluminescence detection. Some dpa is released even without the extraction procedure; therefore, high concentrations of spores can currently be detected in a liquid sample in only 2 to 4 minutes. The use of microfluidic reaction chambers could further shorten this analysis time. Research has also been conducted to determine sample constituents that will interfere with the photoluminescence detection, and a method has been developed to mitigate the interference caused by the presence of phosphate ions.37 Further challenges include automating and miniaturizing the reagent additions and sample processing steps.
The terbium-dpa photoluminescence method for spore detection is well suited for implementation in a wet chemistry lab. However, additional research and development would be required in the areas of microfluidics, reagent storage, and optical sensor systems to develop a small, integrated sensor system that could automatically analyze liquid samples generated from an aerosol sampler. As with the other dpa detection methods discussed previously in this chapter, this semiselective assay will only detect the presence of spores, so for general pathogen detection, strategies must be developed to combine this assay with additional sensors that detect other pathogens of concern. If the detection of a wide range of
|
33 |
Dipicolinic acid is only present in the interior of the spore and is not part of the cell wall; thus, dpa must somehow be extracted from the cells to be tested. |
|
34 |
P.M. Pellegrino, N.F. Fell, and J.B. Gillespie. 2002. Enhanced spore detection using dipicolinate extraction techniques. Analytica Chimica Acta 455:167-177. D.C. White, C.A. Lytle, Y.D.M. Gan, Y.M. Piceno, M.H. Wimpee, A.D. Peacock, and C.A. Smith. 2002. Flash detection/identification of pathogens, bacterial spores and bioterrorism agent biomarkers from clinical and environmental matrices. Journal of Microbiological Methods 48(2-3):139-147. A.P. Snyder, W.M. Maswadeh, J.A. Parsons, A. Tripathi, H.L.C. Meuzelaar, J.P. Dworzanski, and M.G. Kim. 1999. Field detection of bacillus spore aerosols with stand-alone pyrolysis-gas chromatography-ion mobility spectrometry. Field Analytical Chemistry and Technology 3(4-5):315-326. R. Goodacre, B. Shann, R.J. Gilbert, E.M. Timmins, A.C. McGovern, B.K. Alsberg, D.B. Kell, and N.A. Logan. 2000. Detection of the dipicolinic acid biomarker in Bacillus spores using Curie-point pyrolysis mass spectrometry and Fourier transform infrared spectroscopy. Anal. Chem. 72:119-127. R. Nudelman, B.V. Bronk, and S. Efrima. 2000. Fluorescence emission derived from dipicolinic acid, its sodium, and its calcium salts. Applied Spectroscopy 54:445-449. A.A. Hindle and E.A.H. Hall. 1999. Dipicolinic acid (DPA) assay revisited and appraised for spore detection. Analyst 124:1599-1604. D. Helm and D. Naumann. 1995. Identification of some bacterial-cell components by FTIR spectroscopy. FEMS Microbiology Letters 126:75-79. S.S. Iqbal, M.W. Mayo, J.G. Bruno, B.V. Bronk, C.A. Batt, and J.P. Chambers. 2000. A review of molecular recognition technologies for detection of biological threat agents. Biosensors and Bioelectronics 15:549-578. |
|
35 |
N.F. Fell, P.M. Pellegrino, and J.B. Gillespie. 2001. Mitigating phosphate interference in bacterial endospore detection by Tb dipicolinate photoluminescence. Analytica Chimica Acta 426:43-50. P.M. Pellegrino, N.F. Fell, and J.B. Gillespie. 2002. Enhanced spore detection using dipicolinate extraction techniques. Analytica Chimica Acta 455:167-177. |
|
36 |
Pellegrino et al., 2002. See note 35 above. |
|
37 |
Fell et al., 2001. See note 35 above. |
pathogens is required, a more generalized detection methodology and platform that can include an array of sensors or other multiplexed assay would be useful.
DIRECT LABELING OF PATHOGENS FOR DETECTION
The use of direct labeling of pathogens followed by rapid detection has been demonstrated using detection formats such as flow cytometry and optical detection on a planar sensor. The general advantage of this approach is that only one binding step is used for detection, and sample processing (other than aerosol collection) was not used for the implementations described below.
Flow Cytometry
Flow cytometry has been investigated as an approach for the detection and classification of organisms based upon their light-scattering properties (size) and direct labeling of their DNA using fluorescent dyes.38 Flow cytometry has also been used in combination with structural-based recognition (discussed in Chapter 7) for selective pathogen identification by, for example, labeling cells with fluorescently labeled antibodies. While structural-based recognition can be very selective, chemistry-based analysis described in this section is semiselective, and the analysis of multiple cross-correlated parameters is required to improve selectivity.
Flow cytometry has been used to differentiate organisms from background particles by labeling their DNA. A 100 microliter liquid sample was mixed with 100 microliters of a micromolar concentration of fluorescent dye that labels DNA and incubated for 3 minutes. After dilution with a buffer solution, the particles were analyzed using a flow cytometer, which optically analyzes the light scattering and fluorescence of individual particles (e.g., cells). A total analysis time of less than 5 minutes was needed (not including aerosol sampling), with a detection limit of 1,000 cfu per milliliter. Due to the analysis of two parameters—particle size and DNA content—there was some ability to discriminate between organisms and background particles and also some ability to identify classes of organisms due to their differences in size and shape. This example highlights the utility of collecting additional sample parameters when using semiselective pathogen detection approaches.
Planar Sensors
Semiselective biodetection can also be achieved by directly labeling components of pathogens upon adsorption to a sensor surface. Because many types of planar chemical sensors can be miniaturized, strengthened, and cheapened, this general approach, with further development, has the potential to be used as a biological smoke detector that could be distributed throughout facilities. In one approach, scientists are developing a pathogen detection system that includes fluorescent dyes immobilized onto a quartz substrate or optical fiber.39 The system is designed so that the fluorescence signal is enhanced when analytes of interest bind to fluorescent dye molecules that are immobilized onto the sensor surface. For example, semiselective detection of bacterial spores is achieved by immobilizing calcein dye on the sensor surface, which binds to the calcium ions associated with the spores. The spore detection limit was 1,000-2,000 spores on the sensor surface, and spores could be accumulated either from microdroplets of solution40 or by direct aerosol deposition in a humid environment.41 Data collection was typically accomplished in 10 to 30 minutes; however, positive signals could be seen in 2 minutes, indicating that
|
38 |
S.A. Sincock, H. Kulaga, M. Cain, P. Anderson, and P.J. Stopa. 1999. Applications of flow cytometry for the detection and characterization of biological aerosols. Field Anal. Chem. Technol. 3:291-306. |
|
39 |
L.C. Taylor, M.B. Tabacco, and J.B. Gillespie. 2001. Sensors for detection of calcium associated with bacterial endospore suspensions. Analytica Chimica Acta 435:239-246. H. Chuang, P. Macuch, and M.B. Tabacco. 2001. Optical sensors for detection of bacteria. 1. General concepts and initial development. Anal. Chem. 73:462-466. |
|
40 |
Taylor et al., 2001. See note 39 above. |
|
41 |
M.B. Tabacco, Echo Technologies. Presentation to the committee on September 26, 2002. |
this approach has the potential for rapid detection if 1,000 to 2,000 spores can be rapidly delivered to the sensor surface.
Achieving pathogen selectivity using this approach is a challenge. Substitution of bacteria or pollen at the same concentration as the bacterial spores resulted in a signal that was about 20 percent of the spore signal. On the other hand, fungal spores resulted in a decrease in signal of about 20 percent.42 These data suggest that the response from a single sensor would be impossible to interpret. Current work is proceeding to combine data from multiple sensors in order to improve the discrimination of the sensor system.
Other dyes under investigation include nucleic acid dyes to detect primarily bacteria and viruses43 and protein dyes to detect toxins. The DNA dyes will stain all nucleic acids, and the protein dye binds electrostatically to COOH and OH groups, so complete characterization of the sensors in environmental samples will be required to determine the utility of these semiselective sensors. The detection limit for DNA-based detection of organisms is currently 1,000 organisms bound to the sensor surface,44 and the detection limit for cholera toxin is currently 120 nanomoles.45 Encouraging data have also been obtained that indicate that binding of the fluorescent dye molecules to the many identical binding sites on dendrimers can increase the optical signal and improve the stability of a fluorescent dye used for DNA staining. This work also points out one challenge in the comparison of the performance of different sensor systems: It is very difficult to compare the performance of systems when the measurements are obtained and reported in different ways (e.g., solution concentration vs. amount of analyte bound to sensor surface, milligrams per milliliter vs. moles per liter vs. molecules per milliliter). Common guidelines regarding the reporting of the testing and performance of pathogen detection systems would aid in the comparison and evaluation of various sensor systems.
Direct fluorescent labeling of pathogen components bound to the sensor surface offers the potential for very rapid response for air monitoring applications since aerosols can be deposited directly on the sensor surface for analysis. In general, it is desirable to minimize the number of processing steps if very fast sensor response (less than 5 minutes) is required. In addition, the optics required (488 nanometer excitation, visible wavelength detection) can be small and relatively inexpensive. Some limitations of this approach are the sensitivity of the system to baseline instability and the effect of environmental sample matrices on the baseline signal and sensor response when relying only on an enhanced fluorescence signal upon binding (rather than a wavelength shift, for example, which is more easily distinguished). In addition, from current data it is not clear that the detection limit of the direct detection method will be suitable in the 2-minute time frame owing to the time required for mass transport to enable dye to interact with the pathogen materials (reported detection limits are obtained after data accumulation times typically ranging from 10 to 30 minutes). However, for high concentrations of agent, a 2-minute analysis time may be possible.
The current systems under development are promising for rapid semiselective monitoring but are not yet ready for deployment. Since this is a dry assay, material will accumulate on the sensor surface after each use, and additional work is needed to develop a method for automatically renewing the sensor surface (see Box 8.2). Extensive testing of an array of several different types of semiselective sensors is required to characterize environmental backgrounds, quantify the number of false positives, and determine the detection limit under actual operating conditions of interest. The ability to detect single fluorescent-labeled organisms in microfluidics systems46 suggests that the detection sensitivity of inexpensive, portable sensor systems can also be improved in the future. Other sensor platforms (see
|
42 |
Taylor et al., 2001. See note 39 above. |
|
43 |
Chuang et al., 2001. See note 39 above. |
|
44 |
Taylor et al., 2001. See note 39 above. A.C. Chang, J.B. Gillespie, and M.B. Tabacco. 2001. Enhanced detection of live bacteria using a dendrimer thin film in an optical biosensor. Anal. Chem. 73:467-470. |
|
45 |
Tabacco, 2002. See note 41 above. |
|
46 |
M.A. McClain, C.T. Culbertson, S.C. Jacobson, and J.M. Ramsey. 2001. Flow cytometry of Escherichia coli on microfluidic devices. Anal. Chem. 73:5334-5338. |
Box 8.2) may also be suitable approaches for semiselective detection based on Ca++, DNA, and protein detection.
VAPOR ANALYSIS OF CELL METABOLITES
Standard sensors for environmental monitoring (e.g., particle, CO2, temperature, vapor, or humidity sensors) may be useful additions to a sensor array to detect situations that are abnormal and therefore trigger additional analysis using more specific sensor systems for pathogen detection.
Some work has been done to investigate the use of vapor sensor arrays, often termed "electronic noses," for bacterial monitoring and detection. This approach does not involve direct detection of the bacteria themselves but of volatile compounds emitted by the bacteria or bacterial preparations. In general, relatively high cell concentrations in solution or on cell culture plates are required in order to obtain detectable levels of vapor for bacterial identification by vapor analysis of the air in close proximity to the bacterial sample. However, this approach may be useful as a trigger to initiate analysis by more selective sensor systems. The response times of vapor sensor arrays are on the order of seconds, and the vapor interactions are reversible, so that rapid air monitoring can be done over extended periods of time with a single sensor array. A rugged vapor sensor system will have to be designed to prefilter particles that will foul the sensor surface, or the surface will have to be renewed periodically when surface fouling occurs. The committee is not aware of any studies investigating vapors emitted from spores. However, it is expected that vapors emitted from spores would be derived from materials associated with the spore coat and related to the method of spore preparation and aerosol formation.
This approach has been applied successfully to situations such as bioprocess monitoring, medical diagnosis,47 or the detection of food spoilage.48 Some work has also been done to use the signatures obtained using vapor sensor arrays to distinguish between several bacteria in mixtures,49 but detection levels do not currently look promising for facility protection. For example, in one study, 5 × 108 cells per milliliter was sufficient to detect organisms and distinguish between Enterobacter aerogenes and Escherichia coli; however, different E. coli strains could not be distinguished under any conditions.50
Microscale Monitoring of Cell Metabolites
With progress in microfluidic systems and improvements in the sensitivity of chemical sensors, there is ongoing research in monitoring the environment and metabolites of very small volumes either within cells or in the surrounding medium. For example, optical nanosensors are being developed for monitoring analytes within and around individual cells,51 and methods are under development for manipulating and analyzing single cells on microfluidic chips.52 While these approaches may not be suitable for environmental monitoring for pathogen detection owing to practical issues such as organism sampling,
|
47 |
T. Bachinger and C.F. Mandenius. 2000. Searching for process information in the aroma of cell cultures. Trends in Biotechnology 18:494-500. T.D. Gibson, O. Prosser, J.N. Hulbert, R.W. Marshall, P. Corcoran, P. Lowery, E.A. Ruck-Keene, and S. Heron. 1997. Detection and simultaneous identification of microorganisms from headspace samples using an electronic nose. Sensors and Actuators B-Chemical 44:413-422. |
|
48 |
Y. Blixt and E. Borch. 1999. Using an electronic nose for determining the spoilage of vacuum-packaged beef. International Journal of Food Microbiology 46:123-134. |
|
49 |
Gibson et al., 1997. See note 47 above. M. Holmberg, F. Gustafsson, E.G. Hornsten, F. Winquist, L.E. Nilsson, L. Ljung, and I. Lundstrom. 1998. Bacteria classification based on feature extraction from sensor data. Biotechnology Techniques 12:319-324. C.M. McEntegart, W.R. Penrose, S. Strathmann, and J.R. Stetter. 2000. Detection and discrimination of coliform bacteria with gas sensor arrays. Sensors and Actuators B-Chemical 70:170-176. |
|
50 |
McEntgart et al., 2000. See note 49 above. |
|
51 |
J.Z. Lu and Z. Rosenzweig. 2000. Nanoscale fluorescent sensors for intracellular analysis. Fresenius Journal of Anal. Chem. 366:569-575. H.A. Clark, M. Hoyer, M.A. Philbert, and R. Kopelman. 1999. Optical nanosensors for chemical analysis inside single living cells. 1. Fabrication, characterization, and methods for intracellular delivery of PEBBLE sensors. Anal. Chem. 71:4831-4836. |
|
52 |
P.C.H. Li and D.J. Garrison. 1997. Transport, manipulation, and reaction of biological cells on-chip using electrokinetic effects. Anal. Chem. 69:1564-1568. |
nanosensor handling, and system clogging, developments in this area could someday lead to the future realization of smaller, cheaper, and more sensitive chemical sensors and the identification of additional biomarkers and methodologies for semiselective biodetection.
FINDINGS AND RECOMMENDATIONS
Finding 8-1: Nearly all encouraging results in the use of mass spectrometry for microorganism identification have been achieved under conditions in which the target organism is present in much higher concentrations than other potentially interfering background organisms. Limited work has been conducted to determine the potential of this approach to accurately identify bioagents in circumstances where the agent organism is present with equal or greater numbers of naturally occurring organisms and other background components. It may be expected, however, that several ongoing efforts in this area may show increasing promise as new data become available.
Recommendation 8-1: Further research in the rapid identification of bioagents using mass spectrometry should be focused on determining the capabilities (e.g., detection limit, selectivity, reproducibility, and total analysis time) of this approach for bioagent identification in complex environmental backgrounds that include the concentration, diversity, and variability of naturally occurring microorganisms and inorganic particles that are expected to be present in deployment circumstances. These activities should be pursued using the best-performing laboratory mass spectrometers. To the extent that recent MALDI mass spectrometry activities show improved detection performance and discrimination in the presence of interfering backgrounds, spectrometers in this class may deserve a higher priority.
Finding 8-2a: Detection of chemicals that are markers for semiselective biodetection can potentially be accomplished in less than 1 minute using small, relatively low-cost sensors, but very little work has been done in this area, and off-the-shelf solutions are not available.
Finding 8-2b: An unacceptable number of false positives is expected if only one semiselective sensor is used. The most promising approach is likely to be the development of sensor arrays that consist of combinations of semiselective sensors and the use of statistical methods to determine if the responses of the sensors in combination indicate that further analysis or action is required. Further research is needed to determine the selectivity that can be obtained by using semiselective biodetection arrays to measure pathogen releases of concern.
Finding 8-2c: Continuous monitoring at 1- to 2-minute intervals will require rapid, reversible detection chemistry or a means to renew the sensing surface between analyses.
Recommendation 8-2: Further research is needed on semiselective sensors in the following areas:
-
Identification of detectable biomarkers and/or classes of biomarkers that can be used to improve selectivity, and development of methods for analyzing information obtained from multiple rapid sensors (sensor arrays).
-
Rigorous characterization of the signal/noise (environmental plus instrument noise) of the sensor arrays under operating conditions in the environment. If the signal/noise is not suitable for detection, research should be conducted to improve the sensitivity of transducers and to develop better labels and detection methods that are not affected by the environmental background signal.
-
Development of reporter tags and transduction methods that minimize the number of steps for binding and detection.
-
Methods for efficient integration of the sensors with aerosol collectors.
-
Methods for enhancing mass transport of analytes to the chemically interactive sensing surface.
-
Development of reversible detection chemistries or methods for renewing the sensing surface between analyses.
Finding 8-3: It is very difficult to compare the performance of sensor systems when the measurements are obtained and reported in different ways (e.g., solution concentration vs. amount of analyte bound to sensor surface; milligrams per milliliter vs. moles per liter vs. molecules per milliliter).
Recommendation 8-3: Common guidelines for reporting of the testing and performance of pathogen detection systems should be developed. This would aid in the comparison and evaluation of various sensor systems.



















