1
Introduction
Nearly 150 years ago, English chemist William Perkin set out to synthesize the alkaloid quinine from coal tar in an attempt to help treat malaria. He did not succeed in synthesizing quinine, but what he did do was create the beautiful purple dye—mauve—which would forever change everything from fundamental chemical synthesis and the design of the latest fashions, to the state of the physical environment and human health. It was the beginnings of using fossil fuel-derived hydrocarbons as the foundation of the twentieth century industrial revolution.
Today, modern society continues to depend on fossil fuel based hydrocarbons to make dyes and develop almost every chemical or material in the market place. Fossil fuels also serve as the main source of energy. But it is not clear this dependence can continue long into the future. One of the main issues with this dependence is that the combustion of fossil fuels produces carbon dioxide (CO2) and other waste products such as particulate matter, which end up in the atmosphere and have significant consequences for human health and the environment. It is generally accepted that the rise in concentrations in CO2 is causing global climate change1—warming the atmosphere, which in turn is causing sea level to rise. The CO2 absorbed by the oceans is in turn acidifying the water. Another significant issue involves reliability of global fossil fuel supplies.
There is a need to examine this situation and attempt to map a path
|
1 |
“Joint science academies’ statement: Global response to climate change,” http://nationalacademies.org/onpi/06072005.pdf |
forward that is sustainable—which allows humanity to meet current environmental, economic, and societal needs without compromising the progress and success of future generations.2 As the feedstock industry for modern economies, the chemical industry plays a major role in advancing the sciences and applications to support this—to work toward the design, creation, processing, use, and disposal of substances that better support the goals of sustainability.
SUSTAINABILITY AND CHEMICALS
According to the American Chemistry Council,3 “the business of chemistry [in the United States] is a $450 billion enterprise [about 26 percent of the global chemical production] and is a key element of the nation’s economy. It is the nation’s largest exporter, accounting for ten cents out of every dollar in U.S. exports. Chemistry companies invest more in research and development than any other business sector.” As a result of trends in fossil fuel supplies, as well as compliance with chemical regulatory policies, business drivers for the chemical industry have evolved significantly over the past 50 years. There is an increasingly competitive landscape. Once a major net exporter, the U.S. chemical industry is now essentially a net importer (trade went negative in 2000–2001).4 These forces combined with transparency requirements, liability risks, and health indicators make sustainability goals, along with innovation, increasingly integral components of a company’s ability to compete in the marketplace.5 Go to the web site of any global top 50 chemical companies6—from the top three, Dow Chemical, BASF, and DuPont who each have 2004 sales in the $30-40 billion range, to number 45 on the list Lyondell Chemical with 2004 sales of about $6 billion—and there will be a statement of commitment to achieving sustainability goals. For example, the following statement appears on the Lyondell web site:
We aim to achieve excellence in every aspect of our economic, social, and environmental performance. We are committed to operating our world-
wide business in a way that brings the greatest benefit to all of our stakeholders (employees, customers, investors, communities) and builds a sustainable future for generations to come,
One of the ways in which Lyondell says they work toward achieving these goals is by: “Investing in product and service innovations that use natural resources, as well as social and financial resources, in an efficient, effective, and economic manner over the long-term.” Achieving such goals in the business world, including the chemical industry, is often referred to as the “triple bottom line.”7 Many chemical companies are now even part of investment indexes such as FTSE4Good and the Dow Jones sustainability indexes that seek to raise business standards and investor awareness by tracking the financial performance of leading sustainability-driven companies.
However, the trend toward decreasing or at least flat research and development spending by the top 50 chemical companies and in industry as a whole8 (Figure 1.1) makes it difficult to advance the science and technology needed to support such sustainability goals. Going forward, the chemical industry is faced with a major conundrum—the need to be sustainable, a desire to support science and technological innovation, and a lacking investment in fundamental research and development to make it all possible.
Despite these overall conditions, many companies are making the extra effort to advance sustainability goals. Pharmaceutical companies in particular have been quite successful. In 2002, prescription and over the counter drug companies invested more than $32 billion in discovering and developing new medicines, marking the thirty-second straight year the industry has increased its investment in R&D.9 As part of this effort, many companies are turning to green chemistry—“the design, development, and implementation of chemical processes and manufactured products to reduce or eliminate substances hazardous to human health and the environment”10—and applying the twelve principles11 (Box 1.1) to redesign their active pharmaceutical ingredient (API) manufacturing processes. In this way, they have been able to dramatically reduce wastes generated. This success is highlight by the fact that five of the 52 winners
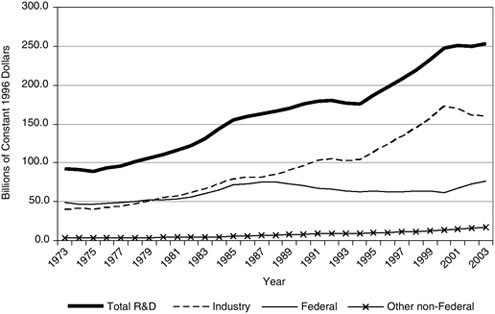
FIGURE 1.1 U.S. research and development funding, by source of funds: 1973–2003.
SOURCE: National Science Foundation, Division of Science Resources Statistics, National Patterns of R&D Resources, annual series, appendix tables B-2 and B-22.
of a U.S. EPA Presidential Green Chemistry Award12 are members of the pharmaceutical industry: Lilly (1999), Roche (2000), Pfizer (2002), Bristol Meyers Squibb (2004), and Merck (2005), which have all reported such kinds of improvements when green chemistry principles are applied.
Green chemistry thus offers a viable path for achieving sustainability goals across the chemical industry. That is, there is the potential to develop industrial technologies that could provide goods, products, and services in a way that does not reduce the supply chain of resources, harm the environment and human health, or limit the opportunities and choices for future generations.
More recently, a complimentary set of green engineering principles was developed (Box 1.2).13 Together with green chemistry, the application of these principles ideally provide one or more of the following benefits:
-
Lower costs of chemical processing
-
Require less energy
|
12 |
See EPA Presidental Green Chemistry Challenge web site: http://www.epa.gov/greenchemistry/presgcc.html |
|
13 |
Ritter, S. K. 2003. A Green Agenda for Engineering. Chemical and Engineering News. 81(29):30–32. |
|
BOX 1.1
|
|
BOX 1.2
|
-
Produce both basic and fine chemicals in a way that is less hazardous to both humans and ecosystems.
-
Be a source of chemical substitutes that improve quality of life.
-
Provide a way of ensuring prosperity and social wealth.
At the same time, there are barriers that exist in developing and implementing these green chemistry and engineering principles. A study by RAND14 identified a set of barriers which fall into four main areas:
-
Lack of research, technology development, and new process engineering;
-
Industrial infrastructure problems and integration barriers;
-
Up-front investments required; and
-
Lack of coordinated actions by means of regulations, incentives, and government purchasing.
Complementary barriers have been identified elsewhere,15,16 such as inadequacy of education and training of chemists and chemical engineers as well as managers who direct them, the difficultly with measuring progress in green chemistry, and the lack of tools for effectively comparing green chemistry with conventional approaches.17
Overcoming such barriers requires defining a “green” or “sustainable” agenda for the chemical industry. For example, the WTEC (World Technology Evaluation Center) Panel Report on Environmentally Benign Manufacturing18 identified the environmental research challenges facing the polymer processing industry, and the Vision2020 roadmap19 discusses environmental considerations for the CPI as a whole. In 2003, the UK Royal Society of Chemistry released the report Benign and Sustainable Chemical Technologies, which identified research opportunities to support sustainability in several areas, including: raw materials, agriculture and chemistry; new chemical routes (solvents and chemicals); catalysis, biocatalysis, and materials; new processes and process strategies; and environmental biotechnology.
STUDY AND REPORT
The purpose of this study was to assist the chemical industry in defining this sustainability agenda—that is, the necessary Grand Challenges and research objectives to enable the ongoing transition towards chemical products, processes, and systems that will help achieve the broader goals of sustainability. As a first step toward defining these objectives, a workshop was convened on February 7-8, 2005 that brought together a broad cross section of disciplines and organizations that make up the chemical industry (see Appendix C for the workshop agenda).
A detailed summary of the presentations and discussion from the workshop, a summary of the break-out sessions, and a list of participants appear in Appendixes D-G, respectively. Briefly, the workshop sessions are summarized below:
In the opening session, on Sustainability Science Literacy and Education to Enable the Adoption of More Sustainable Practices, speakers
|
15 |
Woodhouse, Edward. 2004. In Congressional Report. |
|
16 |
Adler, Robinson, and Rogers. 2002. In Sustainability Workshop AIChE Center for Waste Reduction Technologies. |
|
17 |
See presentation comments of Berkeley W. Cue in Appendix D, p. 116. |
|
18 |
International Technology Research Institute. 2001. Baltimore, MD. |
|
19 |
Vision2020 Technology Partnership. 1996. Technology Vision 2020—The U.S. Chemical Industry. |
Braden R. Allenby, Lauren Heine, and Mary Kirchhoff discussed opportunities to improve education at every level—from education of consumers and citizens, young people, scientists in government and business, to the employees of businesses that use and sell products. They addressed how to promote incorporation of sustainability concepts into educational curricula; develop educational materials, which include sustainability concepts as part of standardized testing programs; and provide professional development opportunities for faculty to learn about sustainability and advantageously incorporate it into research and educational efforts.
Next, speakers Berkeley W. Cue Jr., Richard Helling, and Robert J. Kavlock, talked about Enabling Technologies that Drive the Application of Green Chemistry and Engineering. They discussed the need for dedicated tools to evaluate and appreciate the numerous benefits, and potential impacts and consequences of sustainability efforts. The basic requirements, research tools, and enabling technologies needed to drive the application of green chemistry and engineering for sustainability in the chemical industry were provided.
In the third session, on New Chemistries and Processes that Lead to Commercially Viable Alternative Feedstocks to Fossil Fuels, speakers Stanley R. Bull, Mark T. Holtzapple, and Douglas C. Cameron highlighted the utilization of biomass, and other renewable and recyclable feedstocks for the production of current and future commodity chemicals. Consideration was also given to the impact of resources, materials used in the processing such as catalysts, recycling, water use, and waste generated.
Finally, Jeffrey J. Siirola, Glenn E. Nedwin, William J. Koros, and Klaus S. Lackner presented in the session on Reducing the Energy Intensity of the Chemical Process Industry, which focused on the high energy usage (intensity) of the chemical and allied industry, and the need for pursuing energy efficiency and renewable energy resources. They discussed improvements in energy efficient separation processes; utilization of enzyme catalysts for energy reduction and selectivity increases; improvements in energy efficiency for the production of biofuels and biofeedstocks; development of more effective lubricants; step change improvements in the use of solar energy and other renewable energy sources; and technological breakthroughs in CO2 separation, sequestration and use, were all addressed.
Based largely on the results of the two-day workshop, and the knowledge and experience of organizing committee members, this report identifies a set of overarching Grand Challenges for achieving sustainability in the chemistry industry, and makes recommendations about areas of research required to address those challenges. At the same time, this report is not inclusive of every research topic of relevance to sustainability, and it does not provide an in-depth analysis or an assessment of all that is needed to achieve sustainability in the chemical industry. This report is
meant as a starting point for further analysis, with a focus on those areas that present unique challenges and opportunities for the chemical industry that can also help address larger sustainability goals. Chapters 2-5 of the report provide more focused discussion on aspects of the four areas covered in the workshop. Based on these discussions, the set of recommended Grand Challenges and related research needs are provided in Chapter 6.









