5
Musculoskeletal Effects
This chapter evaluates the effects of fluoride exposure on the musculoskeletal system. Topics considered include the effects of fluoride on bone cells (both bone-forming and bone-resorbing cells), on the developing growth plate, and on articular cartilage as it may relate to arthritic changes. New data on the effects of fluoride on skeletal architecture, bone quality, and bone fracture are also considered. Information on bone cancer is provided in Chapter 10. Effects on tooth development and other issues of oral biology are discussed in Chapter 4.
CHEMISTRY OF FLUORIDE AS IT RELATES TO MINERALIZING TISSUES
Fluoride is the ionic form of the element fluorine. Greater than 99% of the fluoride in the body of mammals resides within bone, where it exists in two general forms. The first is a rapidly exchangeable form that associates with the surfaces of the hydroxyapatite crystals of the mineralized component of bone. Fluoride in this form may be readily available to move from a bone compartment to extracellular fluid. Bone resorption is not necessary for the release of fluoride in this form. However, the predominant form of fluoride in bone resides within the hydroxyapatite crystalline matrix.
Hydroxyapatite is the mature form of a calcium phosphate insoluble salt that is deposited in and around the collagen fibrils of skeletal tissues. The formula for pure hydroxyapatite is CA10(PO4)6OH2. It results from the maturation of initial precipitations of calcium and phosphate during the mineralization process. As the precipitate matures, it organizes into
hexagonal, terraced hydroxyapatite crystals. Recent analysis of bone mineral indicates that a significant proportion of the hydroxyapatite crystal is a form of carbonated apatite, where carbonyl groups (CO3−) replace some of the OH− groups. Carbonated apatite is more soluble than hydroxyapatite at acid pH. Fluoride incorporation into the crystalline structure of bone mineral occurs with the creation of a form of apatite known as fluoroapatite (or fluorapatite). The formula for this form of the crystal is Ca10(PO4)6F2 or Ca10(PO4)6OHF. These crystals also take on a hexagonal shape and are found in terraced layers but, depending on the extent of fluoride in the crystal, may be somewhat more elongated than pure hydroxyapatite. Because fluoroapatite is less soluble in acidic solutions than hydroxyapatite, it was expected that fluoride incorporation into bone might actually make the tissue stronger. However, this has proven not to be the case in human studies (see below).
Release of fluoride from bone when it is in the form of fluoroapatite requires osteoclastic bone resorption. Acidification of the mineral matrix by the osteoclast is sufficient to solubilize the fluoroapatite and allow free exchange with extracellular fluids. Once released, the effect of fluoride on bone cells may be evident; however, the form in which fluoride has its effect remains under debate. Some investigators contend that fluoride directly affects bone cells, but others claim that the effect must be mediated by fluoride while in a complex with aluminum.
Do fluroaluminate complexes exist in biological fluids? The answer to this question depends in large part on pH, protein concentration, and cell composition. However, in general, in the acid environment of the stomach much of the aluminum and fluoride exist in a complex of AlF3 or AlF4−. These forms (mostly AlF3) have been purported to cross the intestine and enter cells (Powell and Thompson 1993). Once inside a bone cell the AlFx form appears to activate a specific protein tyrosine kinase through a G protein and evoke downstream signals. A more complete discussion of this process is presented in a later section of this chapter.
The prolonged maintenance of fluoride in the bone requires that uptake of the element occurs at the same or greater rate than its clearance. This appears to be the case. (See Chapter 3 for more detailed discussion of the pharmacokinetic data on fluoride.) Turner et al. (1993) put forward a mathematical model that appears to fit the known pharmacokinetic data. This model assumes that fluoride influx into bone is a nonlinear function. This assumption is supported by pharmacokinetic data (Ekstrand et al. 1978; Kekki et al. 1982; Ekstrand and Spak 1990) and is required for the model to accurately predict fluoride movements. Another reasonable assumption is that the bulk of fluoride that moves between the skeleton and the extracellular fluid is due to bone remodeling. That is, most of the fluoride is either influxing or effluxing as a result of cellular activity. The outcome of the
Turner model predicts that (1) fluoride uptake is positively associated with the bone remodeling rate and (2) fluoride clearance from the skeleton takes at least four times longer than fluoride uptake. A key correlate to the first prediction is that the concentration of fluoride in bone does not decrease with reduced remodeling rates. Thus, it appears that fluoride enters the bone compartment easily, correlating with bone cell activity, but that it leaves the bone compartment slowly. The model assumes that efflux occurs by bone remodeling and that resorption is reduced at high concentrations of fluoride because of hydroxyapatite solubility. Hence, it is reasonable that 99% of the fluoride in humans resides in bone and the whole body half-life, once in bone, is approximately 20 years (see Chapter 3 for more discussion of pharmacokinetic models).
The effects of fluoride on bone quality are evident but are less well characterized than its effects on bone cells. Bone quality is an encompassing term that may mean different things to different investigators. However, in general it is a description of the material properties of the skeleton that are unrelated to skeletal density. In other words, bone quality is a measure of the strength of the tissue regardless of the mass of the specimen being tested. It includes parameters such as extent of mineralization, microarchitecture, protein composition, collagen cross linking, crystal size, crystal composition, sound transmission properties, ash content, and remodeling rate. It has been known for many years that fluoride exposure can change bone quality. Franke et al. (1975) published a study indicating that industrial fluoride exposure altered hydroxyapatite crystal size and shape. Although the measurements in their report were made with relatively crude x-ray diffraction analyses, they showed a shorter and more slender crystal in subjects who were aluminum workers and known to be exposed to high concentrations of fluoride. Other reports documenting the effects of fluoride on ultrasound velocities in bone, vertebral body strength, ash content, and stiffness have shown variable results (Lees and Hanson 1992; Antich et al. 1993; Richards et al. 1994; Zerwekh et al. 1997a; Søgaard et al. 1994, 1995, 1997); however, the general conclusion is that, although there may be an increase in skeletal density, there is no consistent increase in bone strength. A carefully performed comparison study between the effects of fluoride (2 mg/kg/day) and alendronate in minipigs likely points to the true effect: “in bone with higher volume, there was less strength per unit volume, that is, … there was a deterioration in bone quality” (Lafage et al. 1995).
EFFECT OF FLUORIDE ON CELL FUNCTION
Two key cell types are responsible for bone formation and bone resorption, the osteoblast and osteoclast, respectively. Osteoprogenitor cells give rise to osteoblasts. Osteoprogenitor cells are a self-renewing population of
cells that are committed to the osteoblast lineage. They originate from mesenchymal stem cells. Osteoblasts contain a single nucleus, line bone surfaces, possess active secretory machinery for matrix proteins, and produce very large amounts of type I collagen. Because they also produce and respond to factors that control bone formation as well as bone resorption, they play a critical role in the regulating skeletal mass. Osteoclasts are giant, multinucleated phagocytic cells that have the capability to erode mineralized bone matrix. They are derived from cells in the monocyte/macrophage lineage. Their characteristic ultrastructural features allow them to resorb bone efficiently by creating an extracellular lysosome where proteolytic enzymes, reactive oxygen species, and large numbers of protons are secreted. Osteoclastogenesis is controlled by local as well as systemic regulators.
Effect of Fluoride on Osteoblasts
Perhaps the single clearest effect of fluoride on the skeleton is its stimulation of osteoblast proliferation. The effect on osteoblasts was surmised from clinical trials in the early 1980s documenting an increase in vertebral bone mineral density that could not be ascribed to any effect of fluoride on bone resorption. Biopsy specimens confirmed the effect of fluoride on increasing osteoblast number in humans (Briancon and Meunier 1981; Harrison et al. 1981). Because fluoride stimulates osteoblast proliferation, there is a theoretical risk that it might induce a malignant change in the expanding cell population. This has raised concerns that fluoride exposure might be an independent risk factor for new osteosarcomas (see Chapter 10 for the committee’s assessment).
The demonstration of an effect of fluoride on osteoblast growth in vitro was first reported in 1983 in avian osteoblasts (Farley et al. 1983). This study showed that fluoride stimulated osteoblast proliferation in a biphasic fashion with the optimal mitogenic concentration being 10 µM. The finding that fluoride displayed a biphasic pattern of stimulation (achieving a maximal effect at a specific concentration and declining from there) suggests that multiple pathways might be activated. It is possible that low, subtoxic doses do stimulate proliferation, but at higher doses other pathways responsible for decreasing proliferation or increasing apoptosis might become activated. This thinking suggested that fluoride might have multiple effects on osteoblasts and that might be the reason for some paradoxical findings in the clinical literature (see below). Nevertheless, the characteristics of the fluoride effect point clearly to a direct skeletal effect. Some of these characteristics are as follows: (1) the effects of fluoride on osteoblasts occur at low concentrations in vivo and in vitro (Lau and Baylink 1998); (2) fluoride effects are, for the most part, skeletal specific (Farley et al. 1983; Wergedal et al. 1988); (3) fluoride effects may require the presence of a bone-active
growth factor (such as insulin-like-growth factor I or transforming growth factor β) for its action (Farley et al. 1988; Reed et al. 1993); and (4) fluoride affects predominantly osteoprogenitor cells as opposed to mature functioning osteoblasts (Bellows et al. 1990; Kassem et al. 1994).
Understanding the subcellular signaling mechanisms by which fluoride affects osteoblasts is of paramount importance. Information in this area has the potential to determine whether the fluoride effects are specific, whether toxicity is an issue, and what concentration may influence bone cell function. Moreover, as the pathways become more clearly defined, other targets might emerge. Two hypotheses in the literature describe the effect of fluoride. Both state that the concentration of tyrosine phosphorylated signal pathway intermediates is elevated after fluoride exposure. However, the means by which this occurs differs in the hypotheses. One view is that fluoride blocks or inhibits the activity of a phosphotyrosine phosphatase, thereby increasing the pool of tyrosine-phosphorylated proteins. The other view supports an action of fluoride (along with aluminum) on the stimulation of tyrosine phosphorylation that would also increase the pool of tyrosine-phosphorylated proteins. In the first hypothesis, growth factor activation of the Ras-Raf-MAP kinase pathway would involve stimulation of phosphotyrosine kinase activity. This is mediated by a family of cytosolic G proteins with guanosine triphosphate acting as the energy source. In the presence of fluoride, a sustained high concentration of tyrosine-phosphorylated proteins would be maintained because of the inability of the cell to dephosphorylate the proteins. This theory implicates the existence of a fluoride-sensitive tyrosine phosphatase in osteoblasts. Such an enzyme has been identified and purified. It appears to be a unique osteoblastic acid phosphatase-like enzyme that is inhibited by clinically relevant concentrations of fluoride (Lau et al. 1985, 1987, 1989; Wergedal and Lau 1992). The second hypothesis supports the belief that an AlFx complex activates tyrosine phosphorylation directly. Data from this viewpoint indicate that fluoride alone does not stimulate tyrosine phosphorylation but rather that it requires the presence of aluminum (Caverzasio et al. 1996). The purported mechanism is that the MAP kinase pathway is activated by AlFx, which triggers the proliferation response. A novel tyrosine kinase, Pyk2, has been identified that is known to be activated by AlFx through a G-protein-coupled response and might be responsible for this effect (Jeschke et al. 1998). Two key pieces of evidence that support a G-protein-regulated tyrosine kinase activation step in the fluoride effect are that the mitogenic effect of fluoride can be blocked by genistein (a protein tyrosine kinase inhibitor) and pertussis toxin (a specific inhibitor of heterotrimeric G proteins) (Caverzasio et al. 1997; Susa et al. 1997).
At least two other potential mechanisms deserve mention. Kawase and Suzuki (1989) suggested that fluoride activates protein kinase C (PKC),
and Farley et al. (1993) and Zerwekh et al. (1990) presented evidence that calcium influx into the cells might be a signal for the fluoride-mediated stimulation of proliferation.
In summary, the in vitro effects of fluoride on osteoblast proliferation appear to involve, at the least, a regulation of tyrosine-phosphorylated proteins. Whether this occurs through activation of MAP kinases, G proteins, phosphatases, PKC, or calcium (or a combination) remains to be determined. Whatever the mechanism, however, it is evident that fluoride has an anabolic activity on osteoblasts and their progenitors.
The effects of fluoride on osteoblast number and activity in in vivo studies and clinical trials essentially parallel the in vitro findings. Most reports document increased osteoblast number; however, some investigators have documented a complex and paradoxical effect of fluoride in patients with skeletal fluorosis. Boivin et al. (1989, 1990) reported that, in biopsy bone cores taken from 29 patients with skeletal fluorosis of various etiologies (0.79% ± 0.36% or 7,900 ± 3,600 milligrams per kilogram [mg/kg] of bone ash), there is an apparent increase in the production of osteoblasts with a concomitant increase in a toxic effect of fluoride at the cell level. They provided data to indicate that chronic exposure to fluoride in both endemic and industrially exposed subjects led to an increase in bone volume, an increase in cortical width, and an increase in porosity. However, there was no reduction in cortical bone mass. Osteoid parameters (unmineralized type I collagen) were also significantly increased in fluorotic patients. Interestingly, the fluorotic group had more osteoblasts than the control group, with a very high proportion of quiescent, flattened osteoblasts, but the mineral apposition rate was significantly decreased. It appeared as though the increased numbers of quiescent cells were in a prolonged inactive period. Thus, the conclusion drawn by these investigators was that fluoride exposure increased the birth rate of new osteoblasts, but at high concentrations there was an independent toxic effect on the cells that blocked the full manifestation for the increase in skeletal mass. Boivin et al. used a fluoride-specific electrode for measurements in acidified specimens of human bone. As a point of reference to the above findings, they found that normal control subjects (likely not to have lived in areas with water fluoridation) have mean fluoride content in bone ash (from iliac crest samples) ranging from 0.06% to 0.10% (600 to 1,000 mg/kg); untreated osteoporotic patients range from 0.05% to 0.08% (500 to 800 mg/kg); NaF-treated osteoporotic patients range from 0.24% to 0.67% (2,400 to 6,700 mg/kg) depending on duration of therapy; and skeletal fluorosis patients range from 0.56% to 1.33% (5,600 to 13,300 mg/kg) depending on the source and level of exposure (Boivin et al. 1988). All these ranges are of mean concentrations of fluoride and not individual measurements.
Effect of Fluoride on Osteoclasts
The effects of fluoride on osteoclast activity, and by extension the rate of bone resorption, are less well defined than its effects on osteoblasts. In general, there appears to be good evidence that fluoride decreases osteoclastogenesis and osteoclast activity in in vitro systems; however, its effect in in vivo systems is equivocal. This may be due, in part, to the systemic effects of fluoride in whole animals or humans. A further discussion on this point appears below.
Most reports in the literature studying the effect of fluoride on osteoclast function indicate an inhibition. In fact, the effect might be mediated through G-protein-coupled pathways as in the osteoblast. Moonga et al. (1993) showed that fluoride, in the form of AlF4− resulted in a marked concentration-dependent inhibition of bone resorption. In association with this inhibition, they found a marked increase in the secretion of tartrate-resistant acid phosphatase (TRAP). TRAP presumably originated from the osteoclast; however, its function as a secreted enzyme is not known. The fluoride effect was reproduced with cholera toxin, another Gs stimulator. This effect does not appear to be mediated solely by an AlFx complex because studies using NaF have reported similar findings (Taylor et al. 1989, 1990; Okuda et al. 1990).
Further evidence that fluoride might blunt osteoclastic bone resorption was reported in a study that investigated acid production as a critical feature of osteoclastic function. The pH within osteoclasts can be measured with the proton-sensitive dye acridine orange. Studies in which osteoclasts were observed found that parathyroid hormone induced osteoclast acidity but that calcitonin, cortisol, and NaF all blocked the effect. As acidification of the matrix is required for normal osteoclast function, fluoride, in this case, would act as an inhibitor to bone resorption (Anderson et al. 1986).
The effects of fluoride on bone resorption and osteoclast function in vivo present a complex picture. Some well-controlled animal studies document a decrease in osteoclast (as well as odontoclast) activity. In these studies, rodents and rabbits were exposed to doses of fluoride ranging from clinically relevant to high. Time courses ranged from days to weeks, and the findings indicated a statistically significant decrease in the number and activity of resorbing cells (Faccini 1967; Lindskog et al. 1989; Kameyama et al. 1994). Other studies documented little or no statistically significant effect of fluoride on osteoclast activity (Marie and Hott 1986; Huang 1987). Yet other work that utilized skeletal turnover and remodeling showed an increase in resorption after fluoride therapy (Kragstrup et al. 1984; Snow and Anderson 1986). These studies based their conclusions on the initiation of basic multicellular units (BMUs) and extent of remodeling surface. In the field of skeletal research, it has been accepted that adult bone remodels
itself through the generation of BMUs. This unit is a temporal description of remodeling starting with osteoclastic bone resorption and progressing through a coupled stimulation of bone formation. All BMU activity, thus, is initiated with the action of an osteoclast. An increase in remodeling surface also implies an increase in BMUs. Snow and Anderson (1986) and Kragstrup et al. (1984) demonstrated an increase in resorption under the influence of fluoride by measuring BMU numbers and remodeling surface, respectively. Because these data were derived from intact in vivo animal models, the investigators could not conclude that the effects of fluoride on osteoclastic bone resorption were direct.
It is interesting that only a single report has appeared that links fluoride exposure to the receptor activator of NF kappaB (RANK) ligand, RANK receptor, or osteoprotegerin (OPG) concentrations. These molecules have recently been characterized as end-stage regulators of osteoclast formation and activity (Lee and Kim 2003). RANK ligand is produced by a variety of cells, with osteoblasts being the most prominent. In its usual form, it is a membrane-associated factor that binds to the RANK receptor on preosteoclasts and induces their further differentiation. OPG is a decoy RANK receptor that is an endogenous inhibitor of bone resorption by virtue of its ability to bind RANK ligand. A clinical trial by von Tirpitz et al. (2003) showed that both fluoride and bisphosphonate therapy decreased OPG concentrations. If this were a direct effect of fluoride, one would expect to see an increase in bone resorption. Conversely, if fluoride blocked bone resorption, the decrease in OPG concentrations could be due to a compensatory feedback pathway. Unfortunately, there were not enough histologic or biochemical marker data in this report to determine whether the fluoride effect was direct or indirect.
EFFECTS OF FLUORIDE ON HUMAN SKELETAL METABOLISM
Bone Strength and Fracture
Cellular and Molecular Aspects
Inducing a permanent alteration of skeletal mass in an adult human (or experimental animal) is quite difficult, because bone, as an organ system, possesses an innate mechanism for self-correction. That is, rates of bone formation are controlled, for the most part, by rates of bone resorption. As osteoclastic bone resorption increases or decreases, there is a compensatory increase or decrease in the rate of osteoblastic bone formation. This coupling between the two cell activities was first described by Hattner et al. (1965), and is responsible for the maintenance of a steady-state skeletal mass in adults. These early results indicate that effective management of skeletal
mass would require controlling both cell processes. However, until recently, the only therapies approved by the U.S. Food and Drug Admnistration for treating osteoporosis in the United States targeted only osteoclastic bone resorption. They included molecules such as the bisphosphonates, estrogen and its analogs, and calcitonin derivatives. Currently, teraparitide is available as the only approved treatment that acts to stimulate osteoblastic bone formation. Fluoride falls into this category and that is the reason why there was such great interest in this ion as a potential therapy for osteoporosis. Unfortunately, fluoride did not prove to be an effective treatment for two major reasons. First, although it showed robust stimulation of bone mineral density (see below), its effects as an agent to reduce fractures have never been unequivocally documented. Second, because this naturally occurring element cannot be protected with a patent, the pharmaceutical industry has not been interested in investigating all its potential.
The first clinical trials of fluoride in humans were performed by Rich and Ensinck (1961). Since then many hundreds of reports have appeared in the medical literature. The overwhelming weight of evidence in these reports documents the effect of fluoride, at therapeutic doses, to be that of an increase in bone mineral density. The lowest dose of NaF to show a clear increase in bone mineral density was 30 mg/day, although there may be effects at lower doses (Hansson and Roos 1987; Kleerekoper and Balena 1991). Response was linear with time for at least 4 to 6 years (Riggs et al. 1990). This linear relationship was confirmed in another study lasting more than 10 years (Kleerekoper and Balena 1991). The observation that bone mineral density continues to increase with time is not surprising in and of itself; however, it differs from the action of the antiresorptive bisphosphonates. Whereas agents that depress bone resorption are most effective when the rate of bone remodeling is high, there appears to be no relationship between the rate of remodeling and the response to fluoride. Also, in contrast to the recent data demonstrating a persistence of bone density with the discontinuance of bisphosphonate therapy, discontinuance of fluoride therapy leads to immediate resumption of bone density loss (Talbot et al. 1996).
The dose and duration of fluoride exposure are critical components in determining the effects of the ingested ion on bone. In addition, approximately 30% of patients do not respond to fluoride at any dose (Kleerekoper and Mendlovic 1993). Moreover, there are wide variations in bioavailability among patients and fluoride preparations, and individual responses to the ion also vary widely (Boivin et al. 1993; Erlacher et al. 1995). Whereas the daily dose of fluoride in randomized therapeutic trials (20 to 34 mg/day) exceeds that for people drinking water with fluoride at 4 mg/L (4 to 8 mg/ day for 1 to 2 L/day), the latter may be exposed much longer, leading to comparable or higher cumulative doses and bone fluoride concentrations (see discussion later in this chapter.)
Allolio and Lehmann (1999)noted that the peak blood concentrations of fluoride after swallowing 8 oz of water (at 1.0 µg/L)all at once will reach 8.75 µg/L. If peak blood concentrations are proportional to water concentration,then consumption of 8 oz of water containing fluoride at 4 mg/L would produce peak concentrations below the threshold for effects on osteoblasts examined in vitro (95 ng/mL)(Ekstrand and Spak 1990). Assuming that the blood fluoride concentrations decline between each episode of water consumption of 8 oz or less, such exposures may not achieve a concentration of fluoride in the extracellular fluids sufficient to affect bone cells. A caveat to this analysis is that bone cells may be exposed to potentially higher (bbut unknown) concentrations because of their proximity to the mineralized bone compartment. There have been no direct measurements of the local fluoride concentration around a site of bone resorption. However, a calculation based on estimated rates of resorption,diffusion kinetics, and starting concentration indicates that bone cells and other cells in the immediate vicinity may experience high concentrations of fluoride.
The conditions for an estimate of the fluoride concentration as a function of distance from the osteoclast are as follows:
-
The bone being resorbed has a fluoride content of 3,000 mg per kg of bone ash.
-
Bone ash is assumed to include 65% of the volume of viable bone and the density of viable bone is 1.2 g/cm3. Thus,the concentration of fluoride in the bone compartment is approximately 5,500 µg/cm3.
-
An osteoclast resorbs bone at an average rate of about 30,000 µm3 in 2.5 weeks.
-
The osteoclast is delivering fluoride to the extracellular fluid space from a point source with a radius of 20 µm.
-
Diffusion occurs into a three-dimensional spherical space around the osteoclast.
-
The diffusion coefficient of fluoride in extracellular fluid is approximately 1.5 × 10−5 cm2/s.
Under these conditions, the following equation describes the concentration of fluoride as a function of time and distance from the site of bone resorption (Saltzman 2004):
where C is the concentration of fluoride as a function of distance and time, S is the delivery rate of fluoride from the resorption site, A is the radius of the point source from which the fluoride is delivered, D is the diffusion
coefficient of the fluoride, r is the distance from the resorption site, and t is the time after commencement of the resorption. A graphical representation of this function is presented in Figure 5-1.
An examination of the curves in Figure 5-1 indicates that the fluoride concentration around a site of bone resorption can be quite high immediately adjacent to the osteoclast. The theoretical maximum concentration at 20 µm from the site (at the surface of the osteoclast) would be about 5,500 µg/cm3. The concentration rapidly decays to zero in very short times at distances greater than 100 µm from the site. However, it appears that a sustained fluoride concentration is achieved in the range of hours and persists for the entire resorption process. Thus, by 2.5 weeks, the concentration of fluoride will be about 500 µg/cm3 at a distance of 250 µm from the resorption site.
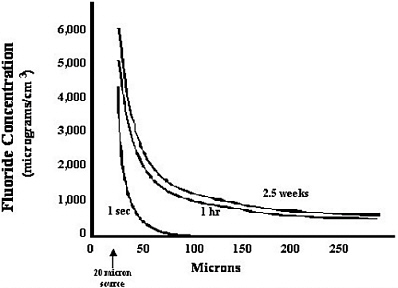
FIGURE 5-1 Concentration of fluoride plotted as a function of time and distance from the site of bone resorption. Release of fluoride from a site of bone resorption can achieve a near steady state concentration in a matter of hours. Twenty microns was defined as the radius of the point source from which fluoride was delivered to the extracellular fluid. Acknowledgement: Hani Awad, University of Rochester, Rochester, New York, assisted in this analysis.
The concentration of fluoride tends toward zero at longer distances. This modeling does not take into account any dissipation of fluoride due to flow of extracellular fluid through the bone marrow compartment. A more complete picture of the local concentration of fluoride around a resorption site should include this factor; however, there are no data on which to base this estimate. Thus, considering that within approximately 1 hour, the fluoride concentration achieves an equilibrium in the surrounding volume, it is likely that the actual fluoride concentration is less, but not substantially so.
Within 250 µm of a site of resorption, it is possible to encounter progenitor cells that give rise to bone, blood, and fat. Thus, one must assume that these cells would be exposed to high concentrations of fluoride. At this time, it is not possible to predict what effect this exposure would have on the functioning of skeletal elements, hematopoiesis, and adipose formation. It should also be pointed out that the number of resorbing sites in an adult skeleton at any point in time is quite small, on the order of 1 × 106 sites. That is, of the vast surface area of trabecular bone in a human skeleton, only about 1 million sites of bone resorption are occurring at any given moment. Whether these elevated concentrations of fluoride have a meaningful effect on bone metabolism can only be speculated at this time.
Some studies have measured the fluoride content of bone, but its effect on a direct measurement of bone strength in humans is not easy to determine. Animal studies have provided some clues. Some studies have reported a biphasic effect of fluoride on bone strength (Beary 1969; Rich and Feist 1970; Turner et al. 1992). For example, Turner et al. (1992) reported an increase in bone strength in rats with bone fluoride concentrations up to 1,200 mg/kg, but they found a decrease in strength back to that of untreated animals with concentrations around 6,000 to 7,000 mg/kg. Skeletal specimens with fluoride concentrations greater than this appeared to have less strength than control treated bone. A variable that may affect the analysis of bone strength is the age of the animal (see Chapter 3). Turner et al. (1995) performed another study in which they found little effect of fluoride on bone strength at any concentration in young rats but a significant effect in old rats. The predominant effect in the older animals clustered around bone fluoride concentrations of 6,000 to 8,000 mg/kg (Turner et al. 1995). Thus, whether fluoride has a biphasic effect on bone strength has not been firmly established.
Other reports in the literature suggesting that fluoride might diminish bone strength in animal models have appeared. Studies of rabbits by Turner et al. (1997) and Chachra et al. (1999) have put forward the point of view that fluoride exposure might decrease strength by altering the structural integrity of the bone microarchitecture. Turner et al. (1997) found no effects of fluoride on a number of bone serum markers, but an increase in bone formation and bone mass. However, this was accompanied by a decrease in
bone strength at multiple sites. In a subsequent paper, these authors suggest that the decrease in strength might be due to alterations in mineral crystal structure (Chachra et al. 1999). Whether these results occur in humans remains to be shown. A decrease in bone strength in a human population will definitely increase the risk of fracture and there have been case reports to document this, especially in subjects who may be highly susceptible to accumulating fluoride, such as those with renal failure (Gerster et al. 1983). A more complete discussion of the effects of fluoride in larger population studies follows.
The applicability of rat studies to quantitatively assess risk of bone fracture in humans is uncertain because of the physiological differences between the skeletons of the species. For example, fluoride uptake into bone occurs more readily in humans than in rats (see Chapter 3 and Appendix D). Rats do not undergo Haversian remodeling in their cortical bones as humans do. On the other hand, if fluoride affects bone properties through crystal structure and the mineral-collagen interface, changes in rat bone strength may provide a model for human bone strength (Turner et al. 1992). In addition, whereas the relationship between bone strength and fracture has been studied in rodents, no comparable data are available for humans. The committee therefore judges that the rat experiments provide qualitative support for an effect of fluoride on fractures in humans but cannot yet be used to make quantitative risk estimates for this end point.
The qualifications noted above for rats do not apply as strongly to the rabbit model. Rabbits undergo Haversian remodeling (i.e., osteoclast bone resorption within cortical bone) as do humans (T. Hirano et al. 1999), and the rabbit growth plate behaves more like a human than does a rat or mouse (Zaleske et al. 1982; Irie et al. 2005). Thus, the rabbit is a better model for studying bone effects than rats or mice.
Epidemiology Data
The committee reviewed epidemiologic data on the relationship between fluoride exposure and fractures from two sources: observational studies of exposure to fluoride in water and randomized clinical trials of the use of fluoride in treating osteoporosis. Table 5-1 summarizes studies of bone fracture in populations exposed to fluoride in drinking water. Most of these studies have compared fluoridated (1 mg/L) and nonfluoridated areas. A meta-analysis by McDonagh et al. (2000a, b) evaluated bone fractures in relation to water fluoridation. Consequently, they excluded data from areas with drinking water fluoridated above 1 mg/L, if data at 1 mg/L were available. Results for fractures were reported as evenly distributed around the null—no effect—but statistical testing showed significant heterogeneity among studies. Because the exposures evaluated in this paper did not spe-
TABLE 5-1 Studies on Bone Fracture in Populations Exposed to Fluoride in Drinking Water
|
Study Design |
Country |
Subjects |
Exposure |
|
Ecologic |
USA (national) |
Residents of fluoridated and nonfluoridated communities (age ≥ 65; n (fluoridated communities) = 40 million; n (nonfluoridated communities) = 30 million; n (cases) = 218,951) |
Fluoridated Nonfluoridated (concentrations not specified) |
|
Ecologic |
USA (national) |
Patients discharged with hip fracture in counties throughout the USA (n = 541,985) |
Fluoridated Nonfluoridated (concentrations not specified) |
|
Ecologic |
USA (national) |
5% of Medicare population (ages 65 to 89; n [cases] = 59,383) |
≤0.3 mg/L (natural) ≥0.7 mg/L (natural and artificial) |
|
Ecologic |
USA (national) |
Data from National Health Interview Surveys (ages ≥ 45; n = 44,031) |
≥0.7 mg/L (natural); groups assessed in terms of <20% or ≥80% of the population exposed to fluoridated water |
|
Prospective cohort |
USA (Oregon, Minnesota, Maryland, Pennsylvania) |
Women (ages ≥ 65; n = 5,781) |
Exposed to fluoridated or nonfluoridated (concentrations not specified) water for 20 years |
|
Ecologic |
USA (Minnesota) |
Participants in another epidemiology project (ages ≥ 50) |
10 years before and 10 years after fluoridation (1.1 mg/L) was implemented |
|
Prospective cohort |
USA (Pennsylvania) |
Women participating in osteoporotic fracture study (ages ≥ 65; n = 2,076) |
1.0 mg/L (artificial) 0.15 mg/L (natural) Number of years of exposure: 0, 1 to 10, 11 to 20, > 20 years |
|
Ecologic |
USA (Utah) |
Hip fracture patients (ages ≥ 65; n = 246) |
1 mg/L (artificial) <0.3 mg/L (natural) |
|
Observations |
Reference |
|
Relative risk (RR) of hip fracture in fluoridated communities was 1.08 (95% confidence interval [CI] 1.06 to 1.10) for women and 1.17 (95% CI 1.13 to 1.22) for men. Lack of dose-response relationship between hip fracture risk and duration of water fluoridation. Analyses of annual age-adjusted incidence rates by duration of county water fluoridation showed a pattern of lowest risk in nonfluoridated counties and highest risk in counties fluoridated for up to 5 years, but rates gradually declined for longer durations. |
Jacobsen et al. 1992 |
|
Weak positive association (before and after adjustment) between hip fracture incidence and percent of county residents who live in counties with fluoridated water. |
Jacobsen et al. 1990 |
|
RR of hip fracture in the fluoridated group was 1.00 (95% CI 0.92 to 1.09) for men and 1.01 (95% CI 0.96 to 1.06) for women. For ankle fracture, it was 1.01 (95% CI 0.87 to 1.16) for men and 1.00 (95% CI 0.92 to 1.08) for women. For fractures of the distal forearm and proximal humerus, a gender difference in risk was found. For women, there was no association between fluoridation and the two types of fractures. Men in fluoridated areas had a 23% higher risk of proximal humerus fracture (RR 1.23; 95% CI 1.06 to 1.43) and a 16% higher risk of distal forearm fracture (RR 1.16; 95% CI .02 to 1.33). |
Karagas et al. 1996 |
|
Rate of hip fracture hospitalization per 1,000 in the population with <20% exposed was 2.4 for women and 1.0 for men. For the group with ≥80% exposed, the rates were 2.2 for women and 1.1 for men. |
Madans et al. 1983 |
|
RR after multivariate adjustment was 0.96 (95% CI 0.83 to 1.10; P = 0.536) for nonvertebral fractures, 0.73 (95% CI 0.55 to 0.97; P = 0.033) for vertebral fractures, 0.69 (95% CI 0.50 to 0.96; P = 0.028) for hip fractures, 0.85 (95% CI 0.58 to 1.23; P = 0.378) for humerus fractures, and 1.32 (95% CI 1.00 to 1.71; P = 0.051) for wrist fractures. |
Phipps et al. 2000 |
|
Incidence of hip fracture was 484 per 100,000 residents before fluoridation and 450 per 100,000 residents after fluoridation. RR associated with fluoridation was 0.63 (95% CI 0.46 to 0.86). |
Jacobsen et al. 1993 |
|
Axial and appendicular bone mass was similar between women exposed to fluoride for >20 years and those exposed for ≤20 years. No significant association was found between fluoride exposure and wrist, spinal, nonspinal, osteoporotic, or hip fractures. |
Cauley et al. 1995 |
|
RR of hip fracture in the fluoridated population was 1.27 (90% CI 1.08 to 1.46) for women and 1.41 (95% CI 1.00 to 1.81) in men. |
Danielson et al. 1992 |
|
Study Design |
Country |
Subjects |
Exposure |
|
Prospective cohort |
USA (Iowa) |
Women from three communities with different concentrations of fluoride in water (ages 20-92, n = 1,300) |
1 mg/L (w/Ca at 60 mg/L) 1 mg/L (w/Ca at 375 mg/L) 4 mg/L (w/Ca at 15 mg/L) |
|
Prospective cohort |
USA (Iowa) |
Women from 3 communities with different concentrations of fluoride in water (ages 20-35 and 55-80; n = 158 [referents], n = 230 [high fluoride]) |
1 mg/L (w/Ca at 67 mg/L) 1 mg/L (w/Ca at 375 mg/L) 4 mg/L (w/Ca at 15 mg/L) |
|
Retrospective cohort |
USA (Iowa) |
Women from 3 communities with different concentrations of fluoride in water |
1 mg/L (w/Ca at 60 mg/L) 1 mg/L (w/Ca at 375 mg/L) 4 mg/L (w/Ca at 15 mg/L) |
|
Ecologic |
USA (Michigan) |
Female Medicaid recipients (ages ≥ 65) |
≥89% of the population receives fluoridated water (2 groups) <15% of the population receives fluoridated water |
|
Ecologic |
Canada |
Patients (ages 45 to 64, 65+) with hip fracture in two cities |
0.3 mg/L 1 mg/L |
|
Case-control |
United Kingdom |
Patients with hip fractures (ages ≥ 50; n [cases]) = 514; n [controls]= 527) |
<0.9 mg/L (artificial) ≥0.9 mg/L (natural) |
|
Ecologic |
England, Wales |
Patients discharged from hospital after hip fracture (ages ≥ 45; n = 20,393) |
0.005 to 0.93 mg/L (natural and artificial) |
|
Prospective cohort |
France |
Subjects enrolled in another study (ages ≥ 65; n = 3.216) |
0.05 to 0.11 mg/L 0.11 to 0.25 mg/L >0.25 mg/L |
|
Ecologic |
France |
Subjects enrolled in another study on aging (ages ≥ 65; n = 3,777) |
0.05 to 0.11 mg/L 0.11 to 1.83 mg/L |
|
Ecologic |
Germany |
Residents of fluoridated and nonfluoridated communities |
0.08 to 0.36 mg/L (natural) 0.77 to 1.20 mg/L (artificial) |
|
Observations |
Reference |
|
RR for osteoporotic fractures was 2.55 (P = 0.07) in the 4 mg/L group. Serum fluoride concentrations were not related to osteoporotic fractures or bone mineral density. |
Sowers et al. 2005 |
|
In the 4-mg/L group, RR of any fracture was 1.81 (95% CI 0.45 to 8.22) in premenopausal women and 2.11 (95% CI 1.01 to 4.43) in postmenopausal women. RR for fractures of the hip, wrist, or spine was 2.70 (95% CI 0.16 to 8.28) in premenopausal women and 2.20 (95% CI 1.07 to 4.69) in postmenopausal women. |
Sowers et al. 1991 |
|
Postmenopausal women in the 4 mg/L group reported significantly more fractures than the other two groups. |
Sowers et al. 1986 |
|
Long-bone fracture rates were 94.3 per 1,000 and 81.1 per 1,000 in the two populations that are ≥89% fluoridated. The rate was 78.8 per 1,000 in the population that was < 15% fluoridated. |
Avorn and Niessen 1986 |
|
For men, ages 45 to 64, standardized hospital admission rates were 0.59 and 0.55, respectively; for men over 65, rates were 5.09 and 4.52. For women, ages 45 to 64, corresponding rates were 0.60 and 0.71; and for ages over 65, they were 9.54 and 9.91. |
Suarez-Almazor et al. 1993 |
|
Estimated average lifetime exposure to fluoride in drinking water ranged from 0.15 to 1.79 mg/L. Odds ratio associated with an average lifetime exposure to ≥ 0.9 mg/L was 1.0 (94% CI 0.7 to 1.5). |
Hillier et al. 2000 |
|
Discharge rates ranged from 0.88 to 2.30. No correlation was found between discharge rates for patients with proximal femur fractures and water fluoride concentrations (r = 0.16, P = 0.34). Subsequent reanalysis of the data using a weighted least-squares technique showed a positive correlation between fluoride concentrations and hip fracture (r = 0.41, P = 0.009). |
Cooper et al. 1990, 1991 |
|
Odds ratio for hip fractures was 1, 3.25 (95% CI 1.66 to 6.38), and 2.43 (95% CI 1.11 to 5.33), respectively. Odds ratio for non-hip fractures was 1, 0.88 (95% CI 0.63 to 1.22), and 1.05 (95% CI 0.74 to 1.51). |
Jacqmin-Gadda et al. 1998 |
|
Odds ratio for hip fractures were 1 and 1.86 (90% CI 1.02 to 3.36), respectively. Odds ratio for non-hip fractures were 1 and 0.98 (95% CI 0.80 to 1.21), respectively. |
Jacqmin-Gadda et al. 1995 |
|
Mean annual incidence of hip fracture in the fluoridated community was 173.36 per 100,000 for women and 56.79 per 100,000 men. In the nonfluoridated group, it was 189.35 per 100,000 in women and 56.60 per 100,000 in men. |
Lehmann et al. 1998 |
|
Study Design |
Country |
Subjects |
Exposure |
|
Ecologic |
Italy |
Residents of two counties |
1.45 mg/L (natural) 0.05 mg/L (natural) |
|
Retrospective cohort |
Finland |
Residents of a rural location (n = 144,627) |
≤0.1 mg/L 0.11 to 0.30 mg/L (natural) 0.31 to 0.50 mg/L (natural) 0.51 to 1.00 mg/L (natural) 1.10 to 1.50 mg/L (natural) >1.50 mg/L (natural) |
|
Retrospective cohort |
Finland |
Premenopausal women in a province (ages 47 to 56; n = 3,222) |
<0.3 mg/L (natural) 1 to 1.2 mg/L (artificial) |
|
Ecologic |
Finland |
Patients with hip fracture (ages ≥ 50) |
<0.3 mg/L (natural) 1.0 to 1.2 mg/L (artificial) >1.5 mg/L (natural) |
|
Ecologic |
Finland |
Residents in two towns (n = 71,811 and n = 61,587) |
<0.1 mg/L 1 mg/L |
|
Retrospective cohort |
China |
Residents of rural communities exposed to various concentrations of fluoride in drinking water (ages ≥ 50; n = 8,266) |
0.25 to 0.34 mg/L (natural) 0.58 to 0.73 mg/L (natural) 1.00 to 1.06 mg/L (natural) 1.45 to 2.19 mg/L (natural) 2.62 to 3.56 mg/L (natural) 4.32 to 7.97 mg/L (natural |
|
Ecologic |
Mexico |
Children (ages 6-12 years) and adults (ages 13-60 years) |
ND to 1.5 mg/L (natural) 1.51 to 4.99 mg/L (natural) 5.0 to 8.49 mg/L (natural) 8.5 to 11.9 mg/L (natural) >12 mg/L (natural) |
|
Case-control |
USA |
Women participating in the Nurses’ Health Study (ages 30-55; n [hip fracture] = 53; n [forearm fracture] = 188; n [controls] = 241) |
Concentrations in toenails <2.00 ppm 2.00 to 3.35 ppm 3.36 to 5.50 ppm >5.50 ppm |
|
Observations |
Reference |
|
Significantly greater rate of fracture incidence, particularly femur fractures (RR for males 4.28 and for females 2.64), in the low-exposure community. |
Fabiani et al. 1999 |
|
Age-and area-adjusted RRs for men were 1.0, 1.05 (95% CI 0.90 to 1.22), 0.72 (95% CI 0.51 to 1.02), 1.03 (95% CI 0.81 to 1.32), 0.67 (95% CI 0.46 to 0.97), and 0.98 (95% CI 0.61 to 1.60). Corresponding values for women were 1.0, 0.93 (95% CI 0.84 to 1.02), 1.12 (95% CI 0.93 to 1.34), 1.12 (95% CI 0.96 to 1.31), 1.08 (95% CI 0.88 to 1.32), and 1.08 (95% CI 0.80 to 1.46). Among women aged 50 to 64 years, fluoride was associated with increased risk of hip fracture. Age- and area-adjusted rate ratio for this age group was 2.09 (95% CI 1.16 to 3.76) in the highest-exposure group (>1.5 mg/L) compared with the lowest-exposure group (≤0.1 mg/L). |
Kurttio et al. 1999 |
|
No significant difference in fracture incidence among the fluoridated (15.4%) and nonfluoridated group (13.4%) (P = 0.220). |
Kroger et al. 1994 |
|
No difference in incidence of hip fracture among exposure groups. Osteofluorosis was found in 22% of the high exposure group. Fluoride content of the bone was correlated with volumetric density of trabecular bone and osteoid-covered trabecular bone surface. |
Arnala et al. 1986 |
|
In the <0.1-mg/L exposure group, RR was 2.5 (95% CI 1.6 to 3.9) for men and 1.5 (95% CI 1.2 to 1.8) for women. In the group exposed to 1 mg/L, RR was 1.0 for men and women. |
Simonen and Laitinen 1985 |
|
Lowest prevalence of overall bone fracture was found in the 1.00 to 1.06 mg/L group and was significantly lower (P < 0.05) than that of the groups exposed to concentrations ≥4.32 and ≤0.34 mg/L. Prevalence of hip fracture was greatest in the in the 4.32 to 7.97 mg/L group and was significantly higher than the 1.0- to 1.06-mg/L group. |
Li et al. 2001 |
|
Increased bone fracture (bone types not specified) incidence was observed at concentrations ranging from 1.5 to 4.99 mg/L. A plot of the incidence of fractures in adults versus the average corresponding fluoride concentration by zone indicated a third-order polynomial correlation (R2 = 0.9995). Incidence in children was similar, except in one zone. Linear correlation between Dean index for dental fluorosis and the frequency of bone fracture in children (R2 = 0.94) and adults (R2 = 0.98). |
Alarcón-Herrera et al. 2001 |
|
Women with higher concentrations of toenail fluoride appeared to be at greater risk of forearm fracture but to have a lesser risk of hip fracture than women with toenail concentrations <2 ppm. Odds ratio of hip fracture in women with >5.50 ppm compared with those with <2.00 ppm was 0.8 (95% CI 0.2 to 4.0). Corresponding adjusted odds ratio for forearm fracture was 1.6 (95% CI 0.8 to 3.1). |
Feskanich et al. 1998 |
cifically address the committee’s charge, this meta-analysis and most of the studies on which it was based were not critically evaluated. The committee restricted its attention to the observational studies that most directly address the study charge: studies that examined long-term exposure to fluoride in the range of 2 to 4 mg/L or above in drinking water. Randomized clinical trials that exposed subjects to higher doses over shorter periods of time were also considered.
The committee considered a number of factors as it evaluated the available data, including the following:
-
The committee assumed that fluoride concentrations in bone are the most appropriate measure of exposure. Although difficult to measure in epidemiology studies, bone fluoride concentrations are positively associated with the amount of fluoride exposure, length of exposure, age, and certain diseases such as chronic renal insufficiency (see Chapter 3 for discussion of pharmacokinetic factors that affect fluoride uptake by bone). Use of other fluoride exposure measures is likely to cause measurement error. While exposure measurement error often biases results toward the null, there are many exceptions.
-
U.S. exposure estimates presented in Chapter 2 indicate that water will be the major route of exposure for Americans drinking or cooking with water containing fluoride at 4 mg/L but that other sources become more important at concentrations closer to 1 mg/L.
-
The incidence of fractures increases dramatically in old age. Minor or moderate traumas cause more fractures in the elderly than in healthy young adults. Other known or suspected risk factors include being female, being postmenopausal, diet (e.g., low calcium), physical inactivity, low body mass index, and use of certain drugs (e.g., corticosteroids) (Ross 1996; Woolf and Åkesson 2003). As a result, age is a very important covariate both as a potential confounder and as an effect modifier; control for age may need to be fairly detailed above age 50.
-
Self-reports of fractures are reasonably accurate, although vertebral fractures are typically underreported. Elderly women may overreport total fractures, but the percent of false positives may be lower for fractures of the wrist and hip (Nevitt et al. 1992; Honkanen et al. 1999). Thus, although epidemiological studies would be better if they confirm the presence or absence of fractures, self-reports may be adequate. For example, relative risk measures (risk and rate ratios, but not odds ratios) are unbiased if the outcome is nondifferentially underreported but false positives are negligible (Poole 1985). We might expect the degree of false-positive reporting and underreporting not to differ by fluoride water concentrations, thus tending to attenuate associations.
-
Fluoride may have different effects on fractures of different bones (as
-
suggested by Riggs et al. 1990). Consequently, epidemiologists need to be careful about the degree of aggregation of outcomes. If some bone sites are included that are not susceptible, then relative risk estimates will be biased toward the null; risk or rate differences would not.
-
Studies that measure outcome and covariates individually but exposure by group (e.g., by water concentration) use a partially ecologic or group-level design. This design greatly improves the ability to measure and control for covariates relative to pure ecologic studies; control of covariates is one of the major problems in purely ecologic studies. See Appendix C for a description of these design differences.
Below is a review of the available epidemiologic data for evaluating the adequacy of EPA’s maximum-contaminant-level goal (MCLG) for fluoride of 4 mg/L and secondary maximum contaminant level (SMCL) of 2 mg/L for protecting the public from bone fractures.
Studies Relevant to Assessing Risks at 4 mg/L
Observational Studies. The committee is aware of five published observational studies of fractures in subjects exposed to drinking water containing fluoride at 4 mg/L or higher (Sowers et al. 1986, 1991, 2005; Alarcón-Herrera et al. 2001; Li et al. 2001) and another (Kurttio et al. 1999) involving somewhat lower exposures that has some relevance. The first two Sowers papers examine the same cohort, one retrospectively (Sowers et al. 1986) and one prospectively (Sowers et al. 1991). Because the analysis in the 1986 paper is less detailed for fractures (particularly the discussion of potential confounders), it has been given less attention. Features of the key papers are highlighted in Table 5-2.
Sowers et al. (1991) directly assessed the risk of fracture from fluoride at 4 mg/L, reporting adjusted odds ratios (ORs) of 2.1 (95% CI = 1.0 to 4.4) for any fracture, and 2.2 (95% CI = 1.0 to 4.7) for fracture of the hip/wrist/spine in women 55 to 80 years of age at baseline (ORs were also elevated in younger women). The reference group was exposed to fluoride at 1 mg/L. This is a strong study, particularly because of its prospective cohort design. Although the 1993 National Research Council (NRC) report labeled it as ecologic, it is actually an individual-level study with an ecologic exposure measure (such designs are also called semi-individual; see Appendix C). Outcome and important covariates, including age, are measured at the individual level (control of covariates is particularly problematic in fully ecologic studies). This study has some weaknesses: confounding was assessed by using stepwise logistic regression (a common but less than optimal method for assessing confounding) and fractures were self-reported. Self-reports of fractures are often quite reliable (except for the spine, where
TABLE 5-2 Observational Studies of Bone Fractures in Populations Exposed to Fluoride Near 4 mg/L in Drinking Water
|
|
Li et al. (2001) |
Sowers et al. (1991) |
Alarcón-Herrera et al( 2001) |
||
|
Design |
Retrospective cohort with ecologic exposure measure |
Prospective cohort with ecologicexposure measure |
Ecologic |
||
|
Location |
China, 6 areas with fluoride ranging from 0.25 to 7.97 mg/L |
3 areas in Iowa (USA) with fluoride at 1or 4 mg/L |
Guadiana Valley, Mexico, with fluoride ranging from <1.5 to 16 mg/L |
||
|
No. subjects |
8,262 |
827 at baseline, good follow-up |
1,437 (333 less than 13 years old) |
||
|
Exposure assessment |
Ecologic; negligible sources other than water; very-long-term residents; very strong for this type of study |
Ecologic; other sources likely in low-exposure groups; long residence time |
Ecologic; inconsistent documentation (e.g., use of bottled water mentioned for only one area); permanent residents not defined |
||
|
Outcomes |
Self-reported fractures validated via x-ray, but lack of fracture not confirmed; recall bias seems unlikely; report all fractures since age 20 or 50, also hip since age 20; count number of subjects with fractures |
Self-reported fractures (spine fractures likely underreported) for 5 year followup; report all fractures, plus fractures of hip/wrist/spine; count number of subjects with fractures |
Self-reported fracture; any fracture “ever occurred without apparent cause, where a bone fracture would not normally be expected to occur”—highly subjective; counts multiple fractures per person? |
||
|
Confounding |
Very similar communities; many individual-level risk factors; imperfect method for covariate control (relying on significance tests) |
Similar communities; many individual-level risk factors; imperfect method for covariate control (relying on significance tests) |
No variables analyzed other than crude stratification by age (<13, ≥13); major weakness |
||
|
Results |
U-shaped results for all fractures, increasing trend for hip (age > 20): adjusted ORs (P values) versus 1 mg/L: |
Increased risk at 4 mg/L versus 1 mg/L Women 55 to 80 at baseline, adjusted ORs and (95% CI) versus 1 mg/L: |
Effect measures not presented; percent of fractures increases in adults from 3.1% (<1.5 mg/L) to 7.9% (1.51 to 4.99 mg/L), 8.9% (5 to 8.99 mg/L), but then decreases. P values for the two intermediate levels were 0.046 and 0.041. |
||
|
|
Fluoride, mg/L |
All sites |
Hip |
||
|
|
2.62 to 3.56 |
1.18 (0.35) |
1.73 (0.34) |
2.1 (1.0, 4.4) for any fracture |
|
|
|
4.32 to 7.97 |
1.47 (0.02) |
3.26 (0.02) |
2.2 (1.0, 4.7) hip/wrist/spine |
|
|
|
Total fractures since age 50 also provided |
|
|||
|
|
Li et al. (2001) |
Sowers et al. (1991) |
|
Alarcón-Herrera et al( 2001) |
|
|
Overall |
Strong study |
Strong study |
|
Weak study |
|
|
Additional comments |
|
|
|
Suggestive analysis of fracture versus dental fluorosis but insufficient detail |
|
|
|
Kurttio et al. 1999 |
Sowers et al. 2005 |
|||
|
Design |
Historical cohort |
Prospective cohort with both ecologic and individual-level exposure measures |
|||
|
Location |
Finland, rural communities nationwide |
Same three areas of Iowa as earlier study |
|||
|
No. subjects |
144,000+ |
1,300 women aged 20 to 92 (average, 55) |
|||
|
Exposure assessment |
Groundwater measurements of almost 9,000 wells Fluoride concentrations estimated for each residence by using weighted medians, smoothed interpolations. Categories: <0.1, 0.1 to 0.3, 0.3 to 0.5, 0.5 to 1.0, 1 to 1.5, and >1.5 mg/L. Highest category corresponded to sampled concentrations of less than detection level to approximately 6 mg/L. |
Ecologic (area of study) Individual (serum fluoride concentration) |
|||
|
Outcomes |
First recorded hip fracture |
Self-reported fracture, confirmed by medical records or x-ray copies, if available. Lack of fractures apparently not confirmed. Fractures separated into likely osteoporotic (hip, spine, wrist, ribs) and other. |
|||
|
Confounding |
Analyzed controlling for age and geographic sector. Age adjustment was conducted within broad strata of 50 to 64 and 65 to 80. No information on nutrition, alcohol use, or physical activity. |
Similar communities; many individual-level risk factors; imperfect method for covariate; control (relying on significance tests). Unclear if some covariates were included. |
|||
|
|
Kurttio et al. 1999 |
Sowers et al. 2005 |
|||
|
Results |
For comparisons between the >1.5 mg/L group and the <0.1 mg/L group (ages 50 to 65), adjusted RR = 2.09 (95% CI, 1.16 to 3.76) in women, RR = 0.87 (95% CI, 0.35 to 2.16) in men. For all ages combined, no associations apparent. For fluoride as a continuous variable, RR = 1.44 (95% CI, 1.12 to 1.86) for women below age 65 at start of follow-up, and RR = 0.75 (95% CI, 0.51 to 1.12) for men in same age stratum (age and region adjusted). Women ages 55 to 69 had the most elevated RR in the continuous-variable analysis. Among separate 5-year components of follow-up period, the results were inconsistent. |
Ecologic: 2.55-fold increased risk (P = 0.07) osteoporotic fracture at 4 mg/L versus 1 mg/L for all women (age breakdown not provided) after adjustment (including bone mineral density of femoral neck). Individual: RR = 1.16 (P = 0.66) for osteoporotic fracture versus log of serum fluoride for all women, after adjustment (including bone mineral density of femoral neck). |
|||
|
Overall |
Strong study |
Strong study |
|||
|
Additional comments |
Suggestive of hip fracture risk, with continuous gradient from lowest to highest exposures |
Weak association between bone density and serum fluoride (e.g., adjusted β = 0.011 ± 0.0073 (SE), P = 0.13 for femoral neck). Use of serum fluoride concentration may bias results toward null if there is nondifferential error relative to bone fluoride concentrations. Bone mineral density may be, in part, an intermediate variable. |
|||
underreporting is typical). Details about the interviewers (training or blinding to exposure) were not provided. The paper also examined fractures in a community with high calcium concentrations in water: the adjusted OR for fracture of the hip/wrist/spine was 1.6 (95% CI = 0.71 to 3.4) for the older women and 0.30 (95% CI = 0.04 to 3.4) for younger women (the ORs for all fractures were similar). The regression analysis comparing the high fluoride and the reference communities was adjusted for calcium intake, but it produced no change.
The newest study by Sowers et al. (2005) investigated bone fracture in relation to fluoride concentration in drinking water. The authors measured serum fluoride, providing a potentially improved exposure assessment. In this cohort study, fractures were assessed prospectively for 4 years. Fractures were self-reported and then confirmed with medical records or x-ray copies, if available; lack of fractures was apparently not confirmed. Sowers et al. (2005) collected individual-based information on people from the same regions as the 1986 and 1991 studies. They collected serum fluoride concentrations and bone mineral density of the hip, radius, and spine. The number of subjects was considerably expanded (n = 1,300) from the earlier studies. Although there may be overlap in specific subjects, all the fracture events were recent. The authors reported risk ratios of fractures in the high fluoride area that were similar to those in the previous studies (risk ratio = 2.55, P = 0.07, even when adjusting for bone mineral density, which could function as an intervening variable between water ingestion and fracture outcome). Use of ecologic exposure measures need not cause bias due to exposure measurement error (see Appendix C).
Serum fluoride concentration was higher in the community with fluoride at 4 mg/L in drinking water. Bone and serum concentrations are related but the latter have more noise—potentially much more, depending on how samples were collected. Serum fluoride concentrations can vary within individuals, returning to baseline within hours of exposure.
Fasting serum fluoride concentrations are considered a good (although not necessarily perfect) measure of long-term exposure and of bone fluoride concentrations (Ericsson et al. 1973; Parkins et al. 1974; Taves and Guy 1979; Waterhouse et al. 1980; Whitford 1994; Clarkson et al. 2000; see also Chapter 2 for a discussion of biomarkers and Chapter 3 on pharmacokinetics). Although methods for serum collection are not described in the paper, Sowers stated that fasting serum concentrations were taken “whenever possible” (M.F. Sowers, University of Michigan, personal commun., July 1, 2005). Measured serum fluoride concentration was not statistically associated with fracture incidence in the adjusted model, including bone density, a potential intermediate variable (measured serum fluoride was only weakly associated with bone mineral density). However, it is unclear whether serum fluoride was a useful surrogate for concentrations in bone
or chronic exposure here; random error would tend to bias results toward the null. Table 2 in the Sowers et al. (2005) paper indicated that long-term residency in the high-fluoride region was not associated with appreciably higher serum fluoride than short-term residency.
Besides differences in osteoporotic, but not other, fracture rates, these populations differed markedly with respect to smoking rates and hormone replacement (both lowest in the reference group) and physical activity (lowest in the high-fluoride group). It is unclear whether these factors were examined as potential confounders for fractures. Age subgroups were not presented in the new Sowers et al. study, so differences within age groups cannot be assessed and comparisons with the other observational studies on fractures cannot be made.
For all the Sowers studies, there is an unresolved question about whether the referent group (area with low fluoride and low calcium) might have a low fracture rate because of risk factors that are not controlled for in the studies, particularly as the high-calcium/low-fluoride region also showed increased fracture rates compared with the referent region. Potential bias due to such differences might be exacerbated by the use of an ecologic exposure measure (see Appendix C).
The study by Li et al. (2001) complements the Sowers studies in several ways, having a larger size and relatively strong exposure assessment for a partially ecologic study. It has a retrospective cohort design, increasing the potential for outcome and exposure misclassification, but these problems were addressed by the authors. Although exposure was assessed on the group level, exposure was finely categorized and other sources of fluoride exposure were estimated to be negligible. (Nonwater exposures to fluoride were presumably more important in the Sowers studies.) Communities were quite similar and individual-level risk factors were assessed. Fractures were self-reported; confirmation with x-rays showed very high validity (526 fractures confirmed among the 531 subjects reporting fractures). This study also has weaknesses. Confounding was assessed by statistical testing; the authors included a covariate in the logistic regression if they first found a statistically significant (P < 0.05) relationship between the variable and outcome analyzed bivariately. (Confounding should be judged by examining the effect measure, not statistical testing; see Rothman and Greenland 1998.) Absence of fractures was not confirmed, potentially biasing outcomes if false-positive reporting of fractures is expected to be more than an isolated occurrence. However, a limited number of sensitivity analyses of confounding performed by the committee did not explain the effect; recall bias seems an unlikely explanation for the U-shaped exposure-response curve (for all fractures since age 20), with the minimum fractures in the reference group of 1 mg/L. The dose-response curve for all fractures is plausible: some, but not all, animal studies suggest a biphasic relationship between bone fluoride
concentrations and bone strength (see discussion earlier in this section on cellular and molecular aspects).
The Li et al. study did not directly assess fluoride at 4 mg/L. However the exposure group just above 4 mg/L (4.32 to 7.97 mg/L) showed an increase in all fractures since age 20 (OR = 1.47, P = 0.01, estimated 95% CI = 1.10 to 1.97), all fractures since age 50 (OR = 1.59, P = 0.02, estimated 95% CI = 1.08 to 2.35), and hip fractures since age 20 (OR = 3.26, P = 0.02, estimated 95% CI = 1.21 to 9.81). The exposure group just below 4 mg/L (2.62 to 3.56 mg/L) showed the following: all fractures since age 20 (OR = 1.18, P = 0.35, estimated 95% CI = 0.83 to 1.67), all fractures since age 50 (OR = 1.04, P = 0.87, estimated 95% CI = 0.65 to 1.66), and hip fractures since age 20 (OR = 1.73, P = 0.34, estimated 95% CI = 0.56 to 5.33). CI values were estimated by the committee using the approach of Greenland (1998). Although the latter results are not statistically significant at the 0.05 level, they are consistent with an upward trend (increasing dose-response relationship), particularly the result for hip fracture. The inclusion of all fractures is likely to bias ORs toward the null.
Although the authors did not estimate trend, Figures 2 and 3 presented in the paper by Li et al. (2001) suggest that linear trends in proportions from the 1.00 to 1.06 mg/L category up would provide a reasonable fit in that range. Using a generalized linear model with the binomial distribution and the identity link, and midranges for the exposure categories, the committee estimated absolute increases in fractures of 1.3% (95% CI = 0.3 to 2.2, P = 0.01) for the increment from 1.00 to 4.00 mg/L for overall fractures since age 20, 0.4% (95% CI = 0.0 to 0.8, P = 0.04) for hip fractures since age 20, and 0.9% (95% = CI 0.2 to 1.7, P = 0.02) for overall fractures since age 50.
The U-shaped exposure-response curve for all fractures combined (but not hip fractures) for this population of individuals provides an interesting and potentially important finding. Whereas the trend for fractures appears to increase from 1.00 to 4.00 mg/L, it must be appreciated that the fracture rate in the 1.00 to 1.06 mg/L category was lower than the rate in the category with the lowest intake.
Estimated fluoride exposure in the Li study is higher than for the Sowers studies (see Table 5-4 later in this chapter). Assuming that exposure was predominantly due to water, the committee estimated that participants in the Li study consumed on average about 2.5 L per day for the 2.62- to 3.56-mg/L group and 2.3 L per day for 4.32- to 7.97-mg/L group (versus 0.9 to 1.2 L per day for the Sowers studies). These water consumption levels are in the 90th to 95th percentile for the United States (see Appendix B).
Alarcón-Herrera et al. (2001) is a much weaker ecologic study with little attention to covariates other than a rough stratification by age (see
Table 5-2 for a brief discussion). The results are qualitatively similar to the stronger studies.
In addition, a retrospective cohort study in Finland by Kurttio et al. (1999) is pertinent to the issue of fracture risk at 4 mg/L, even though relatively few wells in that study had drinking water with fluoride concentrations that high. Residents were grouped into exposure categories based on modeled fluoride concentrations in well water closest to their residence: ≤0.1, 0.11 to 0.30, 0.31 to 0.50, 0.51 to 1.00, 1.10 to 1.50, and >1.5 mg/L (ranging up to 2.4 mg/L). Fluoride monitoring results among water samples for the highest modeled group varied from below detection to about 6 mg/L. Hospital discharge registers were tracked between 1981 and 1994 for reports of hip fracture among the cohort. For all ages combined, no associations were found between fluoride content in drinking water and hip fracture. However, analysis of age strata (50 to 64 and 65 to 80) indicated an increased risk of hip fracture in women aged 50 to 64 exposed to fluoride at >1.5 mg/L (adjusted rate ratio of 2.09%; 95% CI, 1.16 to 3.76; based on 13 cases [3,908 person years] compared with those in the least exposed group (≤0.1 mg/L). Some covariates were adjusted by using ecologic measures, an imperfect technique.
Clinical Trials of Osteoporosis Treatment. Using the Cochrane Handbook methodology, Haguenauer et al. (2000) performed a meta-analysis of randomized clinical trials of fluoride in postmenopausal women with primary osteoporosis. Eleven studies met the inclusion criteria; analyses of specific end points included only a subset. The summary relative risk estimate for new vertebral fractures was slightly decreased: 0.87 (95% CI = 0.51 to 1.46) for 2 years of treatment (four trials) and 0.90 (95% CI = 0.71 to 1.14) for 4 years (five trials). The summary relative risk estimate for new nonvertebral fractures was increased: 1.20 (95% CI = 0.68 to 2.10) after 2 years (one trial) and 1.85 (95% CI = 1.36 to 2.50) after 4 years (four trials); the latter association was strongest in trials using high-dose, non-slow-release forms of fluoride. See Table 5-3 for the four studies included in the analysis of nonvertebral fractures after 4 years. All four studies were prospective, double-blinded, and placebo-controlled; all subjects received supplemental calcium. There was loss to follow-up, particularly in the study of Kleerekoper et al. (1991), but it was similar in frequency in treated and placebo groups.
Table 5-3 reports relative risks of nonvertebral fractures at 4 years. Rate ratios are also provided for several studies. Hip fracture results are reported only for Riggs et al. (1990); the number of hip fractures in the other trials was at most one per group. Riggs et al. reported both complete fractures and total fractures. Total fractures equal complete plus incomplete “stress” fractures; the latter were observed by roentgenography in participants re-
TABLE 5-3 Four Randomized Clinical Trials Examining Nonvertebral Fractures
|
|
Exposure |
Enrollment: Exposed and Placebo |
Participationa Exposed and Placebo |
Relative Risk (95% CI) Nonvertebral Fracturesb |
Rate Ratio (95% CI) Nonvertebral Fracturec |
|
Reginster et al. 1998 |
Fluoride at 20 mg/day as sodium monofluorophosphate, 4 years |
100, 100 |
84%, 81% |
1.1 (0.5, 2.4)d |
1.1 (0.5, 2.3) |
|
Pak et al. 1995 |
NaF at 50 mg/ day slow-release, 4 cycles: 12 months on, 2 months off |
54, 56 |
77%, 72% |
0.6 (0.2, 2.5)d |
|
|
Kleerekoper et al. 1991 |
NaF at 75 mg/ day, 4 years |
46, 38 |
60%, 61% |
1.5 (0.7, 3.5)d |
3.0 (2.0, 4.6) “hot spots”e |
|
Riggs et al. 1990 |
NaF at 75 mg/ day, 4 years |
101, 101 |
77%, 80% |
1.6 (1.0, 2.5) complete 2.3 (0.6, 8.8) complete, hip |
1.9 (1.1, 3.2) complete 3.1 (2.0, 5.0) totalf |
|
aParticipating person-time divided by total possible person-time. bRisks were computed by dividing the number of first incident fractures (at most one per subject) by the number of enrolled subjects. cRates were computed by dividing the number of incident fractures (possibly more than one per subject) by participating person-time. dThe numbers that appear to have been used in the meta-analysis of Haguenauer et al. (2000); see their Figure 5. eAreas of increased isotope uptake detected via radionuclide bone scan. fIn this study, total fractures = complete + incomplete “stress” fractures, the latter observed by roentgenography in participants reporting acute lower extremity pain syndrome. |
|||||
porting acute lower extremity pain syndrome (see Kleerekoper et al. 1991 for a different interpretation).
Comparison of Exposure in Randomized Clinical Trials and Observational Studies. Despite the methodological strengths of the randomized clinical trials, their use in this review has limitations. In particular, fluoride exposures in the trials were higher in magnitude (20 to 34 mg/day) than
in observational studies (5 to 10 mg/day for 4 mg/L) but shorter in time (years versus decades). One possibility is to compare studies using total fluoride exposure in absolute mass units. Because some biological effects (e.g., stimulation of osteoblasts) may occur only at high doses, results from clinical trials may not be directly comparable to risks due to long-term exposure to fluoride in water. On the other hand, the committee assumes that bone fluoride concentration is the most appropriate measure of exposure for examining fracture risk. Data permitting, it could be possible to compare the bone fluoride concentrations reached in the randomized clinical trials with those in the observational studies.
Of the four randomized clinical trails in the fracture meta-analysis, the committee was able to locate bone fluoride measurements for only one. Of the 202 postmenopausal women in the Riggs study, bone fluoride was measured before treatment and at 4 years in 43 treated and 35 placebo subjects (Lundy et al. 1995). Unfortunately, the data are presented only in a figure and in units of μmol of fluoride per mmol of calcium. The latter, however, can be approximately converted to mg/kg ash by using the following factors: 1 g of calcium per 7 g wet weight of bone (Mernagh et al. 1977) and 0.56 g of ash per g wet weight of bone (Rao et al. 1995). Using this conversion, the before-treatment bone ash fluoride concentrations were about 1,700 mg/kg in both the treated and the placebo groups. Taking the imprecision of the conversion factors into account, this value is consistent with reported concentrations for healthy, untreated persons living in areas without particularly high water fluoride concentrations and no other exceptional sources of fluoride intake (see Chapter 3). Four years later, bone ash concentrations were slightly higher in the placebo group and about 12,000 mg/kg in the treated group. The latter value should overestimate concentrations in the exposed group of the trial, because the average exposed subject in the Riggs study participated 3.1 years (Table 5-3).
Ideally, one would estimate bone concentrations in the other trials by using a pharmacokinetic model. Because the committee did not have an operational pharmacokinetic model, a regression model was used to estimate bone concentrations based on total fluoride exposure during clinical trials (see Chapter 3). Total exposures (Table 5-4) were estimated with the nominal daily dose and the average length of participation of the exposed group. The bone concentration for Riggs et al. estimated by this technique (7,400 mg/kg) is less than the value measured by Lundy et al. (roughly 12,000 mg/kg), but the latter examined a subset of subjects who had completed the full 4 years of the study. The regression model estimates 9,100 mg/kg in subjects completing 4 years. Although that estimate is still less than the measured concentration, Chapter 3 noted that the regression model may underestimate bone concentrations in fluoridated areas. Of the four clinical trials in Table 5-4, three were American. Fluoride exposure and concentra-
tions in bone may be overestimated for the Pak study because of the use of a slow-release, less bioavailable form of fluoride. In sum, average fluoride bone concentrations among treated trial participants appear to range from about 5,400 to 12,000 mg/kg.
Comparison of Results of Randomized Clinical Trials and Observational Studies. Table 5-4 also includes estimates of total exposure and average bone fluoride for two observational studies. The committee estimated average fluoride concentrations in bone in the study by Sowers et al. (1991) using the regression model developed for chronic water exposure in Chapter 3. This model predicts bone concentrations based on concentration of fluoride in water, length of exposure, and sex. The result is in the same range as the clinical trials. Since the regression model does not take water consumption rates into account, it should underpredict bone fluoride concentrations for people with high water consumption. The bone fluoride estimates for Li et al. (2001) are, therefore, probably underestimates. Estimates of bone fluoride concentrations could be improved through the use of pharmacokinetic models (see Chapter 3).
Table 5-4 summarizes fracture outcomes for the four clinical trials (nonvertebral) and observational studies. There are a number of differences in the way the outcome data were collected and analyzed. For example, Li et al. counted fractures occurring since age 20 (or age 50, not shown), a longer observation period than the other studies; Li et al. and Sowers et al. measured fractures in different bones than those studied in the clinical trials; if trials use subjects from fluoridated areas, the subjects receiving placebos are from areas with fluoride close to 1 mg/L. Although the comparison involves several assumptions and uncertainties, the estimated concentrations of fluoride in bone and results of the randomized clinical trials generally appear consistent with those of the observational studies.
Interpretation of Weight of Evidence of the Fracture Data on Fluoride at 4 mg/L. For making causal inferences, many epidemiologists prefer to formulate and test specific competing hypotheses (e.g., Rothman and Greenland 1998). Other epidemiologists find it useful to weigh the evidence in light of some traditional “criteria” (more properly, guidelines) for examining whether observed associations are likely to be causal (Hill 1965). The discussion below provides a perspective on how the committee evaluated and viewed the strength of the collective evidence on possible causal associations.
-
Consistency: Despite some design or data weaknesses, there is consistency among the results of all the observational studies using ecologic exposure measures. That is, none of the studies that included population ex-
TABLE 5-4 Estimated Bone Fluoride Concentrations and Bone Fracture Risks in Randomized Clinical Trials and Observational Studies
|
Reference |
Fluoride Exposure (mg/day) |
Average Length Exposure (years) |
Estimated Total Exposure (g) |
Estimated Bone Fluoride (mg/kg ash) |
Relative Risks (RR) or Odds Ratios (OR)a and (95% CI) |
|
Randomized clinical trials |
|||||
|
Reginster et al. 1998 (Belgium) |
20 |
3.4 |
24 |
5,400b |
1.1 (0.5, 2.4) RR nonvertebral, 4 years |
|
Pak et al. 1995 (USA) |
23 (slow-release) |
3.1 |
25 |
0.6 (0.2, 2.5) RR nonvertebral, 4.7 years |
|
|
Kleerekoper et al. 1991 (USA) |
34 |
2.4 |
30 |
6,200b |
1.5 (0.7, 3.5) RR nonvertebral, 4 years |
|
Riggs et al. 1990 (USA) |
34 |
3.1 |
38 |
7,400b |
1.6 (1.0, 2.5) RR complete nonvertebral, 4 years |
|
|
|
|
|
(12,000)d |
2.5 (1.7, 3.7) RR total nonvertebral, 4 years |
|
|
|
|
|
|
2.3 (0.6, 8.8) RR complete hip, 4 years |
|
Observational studies |
|||||
|
Sowers et al. 1991 |
4.88e |
35.9f |
64 |
7,200g |
2.1 (1.0, 4.4) OR any fracture, 5 years |
|
Baseline age 55 to 80 (4 mg/L area) |
|
|
|
|
2.2 (1.0, 4.7) OR hip/wrist/spine, 5 years |
|
Sowers et al. 2005 |
3.66 |
NAh |
NAh |
NAh |
2.55 (P = 0.07) OR osteoporotic, ecologic |
|
Age 20 to 92 (4 mg/L area) |
|
|
|
|
1.16 (P = 0.66) OR osteoporotic, log serum concentration |
|
Li et al. 2001 |
|
|
|
|
|
|
2.62 to 3.56 mg/L |
7.85i |
64j |
180 |
>6,200g |
1.18 (P = 0.35) OR, any site since age 20 |
|
|
|
|
|
|
1.73 (P = 0.34) OR, hip since age 20 |
|
4.32 to 7.97 mg/L |
14.1i |
61j |
320 |
>11,000g |
1.47 (P = 0.02) OR, any site since age 20 |
|
|
|
|
|
|
3.26 (P = 0.02) OR, hip since age 20 |
|
aWhen applied to cohort data, ORs overestimate RRs; the bias is small when odds are small as they are here. |
|||||
|
bEstimated using regression model for clinical trials (Chapter 3) based on total exposure. Bone concentrations for U.S. studies may be underestimated because of background exposure. cPossibly overestimated because of the use of a less bioavailable form of fluoride. dApproximate bone concentration measured in a subset exposed for 4 years. eAverage estimated fluoride intake for ages 55 to 80 in 4-mg/L area from Sowers et al. (1986). fAverage residence time from Sowers et al. 1986 (baseline) plus 5 years. gEstimated using regression model for water exposure (Chapter 3). Because of limitations in the regression model, these estimates do not take into account differences in water consumption. This may cause underestimation of bone fluoride concentrations for people with high water consumption rates, as estimated for participants in Li et al. (2001). hAverage length of exposure not available. Based on water fluoride concentrations alone, the average estimated bone concentrations are about 6,700 mg/kg ash (Chapter 3). iEstimated exposures for these groups are from Li et al. (2001). jAverage exposure length equals average age, based on lifetime exposure. |
-
posures above 4 mg/L found null or negative (inverse) associations between fluoride and bone fractures. There is probably minimal publishing bias here because of the intense interest on both sides of the fluoride controversy. Further, all the studies with exposure categories of approximately 2 mg/L and above in water showed elevated relative risks of fractures for these exposure estimates. However, the one study using an individual exposure measure found no association between fracture risk and serum fluoride. Because serum fluoride concentrations may not be a good measure of bone fluoride concentrations or long-term exposure, the ability to show an association might have been diminished.
-
Strength of association: Although weak associations (e.g., small relative risks) can be causal, it is harder to rule out undetected biases. As indicated in Table 5-2, relative risk estimates generally varied from about 1.5 to 2.2 for studies with ecologic measures of exposure.
-
Biologic plausibility/coherence: The weight of evidence of observational studies is increased when qualitative as well as quantitative; biochemical, physiological, and animal data suggest a biologically plausible mechanism by which a potential risk factor such as fluoride could cause adverse effects. In this instance, the type of physiological effect of fluoride on bone “quality” and the fractures observed in animal studies are consistent with the effects found in the observational studies. Furthermore, the results of the randomized clinical trials are consistent with an increased risk of nonvertebral fractures at fluoride concentrations in bone that may be reached by lifetime exposure to water at 4 mg/L.
-
Dose-response (biological gradient): For the most part, the observational studies discussed above observed higher fracture risk with higher exposure compared with 1 mg/L. The combined findings of Kurttio et al. (1999), Alarcón-Herrera et al. (2001), and Li et al. (2001) lend support to gradients of exposure and fracture risk between 1 and 4 mg/L.
The remaining traditional guidelines of Hill and others are not major issues here: time sequence of effect after exposure is fulfilled in all the observational studies and the clinical trials; none of those designs was cross-sectional and all were able to assess sequence. Specificity of effect or exposure is rarely germane in environmental epidemiology. Experiment (that is, effect of removal of exposure) does not apply in this instance.
When papers using different designs or studying disparate populations are evaluated, findings of consistency among these studies do not require that the doses, exposures, or relative risks be the same. (Such quantitative reconciliation is pertinent for efforts to establish unit risks for quantitative risk assessment, pooling studies, or meta-analyses, and assignment of specific potencies goes far beyond the charge or assessment by the committee.) Further, it is not necessary that there be exact quantitative correspon-
dence between animal and human data and physiologic, and epidemiologic exposures.
The weight of evidence supports the conclusion that lifetime exposure to fluoride at drinking water concentrations of 4 mg/L and higher is likely to increase fracture rates in the population, compared with exposure to fluoride at 1 mg/L, particularly in some susceptible demographic groups that are prone to accumulating fluoride into their bones.
Studies Relevant to Assessing Risks at 2 mg/L
The committee found four observational studies that involved exposures to fluoride around 2 mg/L (see Table 5-5). By far the strongest of those studies was by Kurttio et al. (1999). As described above, residents were grouped into exposure categories based on modeled fluoride concentrations in well water closest to their residence (≤0.1, 0.11 to 0.30, 0.31 to 0.50, 0.51 to 1.00, 1.10 to 1.50, and >1.5 mg/L [ranged up to 2.4 mg/L]) and hospital discharge registers were tracked for reports of hip fracture. Whereas no associations between fluoride content in drinking water and hip fracture were found for all ages combined, analysis of age strata (50 to 64 and 65 to 80 years) indicated an adjusted rate ratio of 2.09 (95% CI, 1.16 to 3.76) for hip fracture in women aged 50 to 64 exposed to fluoride at >1.5 mg/L.
Another study, performed in Finland, found no evidence of increased risk when hip fracture rates were compared in populations exposed to fluoride at ≤0.3, 1.0 to 1.2, and >1.5 mg/L (Arnala et al. 1986). However, this study had many weaknesses, including incomplete reporting methods, insufficient control of confounding, inability to assess cumulative exposure, and the possibility of nonsystematic or biased case ascertainment. It focused primarily on evaluating fluoride content and the histomorphometry of bone samples taken from the iliac crest of hip fracture patients and had the advantage of providing data on bone fluoride concentrations. Mean fluoride concentrations (± standard deviation) in bone were found to be 450 ± 190 mg/kg, 1,590 ± 690 mg/kg, and 3,720 ± 2,390 mg/kg in the low-, middle-, and high-exposure groups, respectively.
A study in France investigated fracture rates in relation to fluoride-using subjects enrolled in a different study on aging (Jacqmin-Gadda et al. 1995). Two fluoride exposure groups were compared: 0.05 to 0.11 mg/L and 0.11 to 1.83 mg/L. The odds ratio for hip fractures for the higher exposure group was 1.86 (95% CI, 1.02 to 3.36). The odds ratio for any fractures was 0.98 (95% CI, 0.80 to 1.21). These odds ratios were adjusted for age, gender, and Quetelet index for hip fractures and by age and gender for total fractures. (The authors selected confounders to include in their model on the basis of “statistical significance,” although a more appropriate approach would have been to select covariates based on how much they change the odds
TABLE 5-5 Studies Relevant to Assessing Bone Fracture Risks from Exposure to Fluoride at 2 mg/L in Drinking Water
|
|
Arnala et al. (1986) |
Jacqmin-Gadda et al. (1995) |
|
Design |
Semiecologic; individual outcome data and ecologic exposure measure |
Nested case control analysis drawn from cross-section study that was the first phase of a prospective cohort study. |
|
Location |
Finland, communities |
France |
|
No. subjects |
462 fractures among a population of unspecified size |
3,777 subjects age 65 and older from 75 civil parishes (mean residence time 41 years) |
|
Exposure assessment and categories |
Ecologic; exposure assignments drawn from a 1974 report by the National Board on Health on the fluoride content of drinking water in different communities Communities with fluoride at <0.3 mg/L, 1.0 to 1.2 mg/L, and >1.5 mg/L |
Two measurements were taken in 1991 and routinely thereafter (frequency not specified). Two exposure categories: 0.05 to 0.11 mg/L and 0.11 to 1.83 mg/L |
|
Outcomes |
Hip fractures among men and women combined, for age 50+. Factures due to severe trauma excluded. |
Hip fractures |
|
Effect measure |
Comparison of age-adjusted 10-year incidence of hip fracture for ages 50+ and component age decades. Binomial t test used to compare age-adjusted hip fracture rates. |
OR using multiple logistic regressions, controlling for confounders based on interview data. |
|
Chance |
No confidence intervals or P levels were provided. |
95% CI and P values given |
|
Confounding |
Age-adjustment only. No information on whether women were postmenopausal. No distinction between rates for males and females. |
Age, gender, Quetelet index (kg/ height2 in m), smoking, and sports activity |
|
Fabiani et al. (1999) |
Kurttio et al. (1999) |
|
Semiecologic; individual outcome data and ecologic exposure measures |
Historical cohort. |
|
Two regions of central Italy Avezzano (lower fluoride in water) and Bracciano(higher fluoride in water) |
Finland: rural communities nationwide |
|
935 in Avezanno 190 in Bracciano; subjects treated in a public hospital from each region |
144,000+ |
|
Drinking water sampled twice a year (years not specified), and one summary concentration was assigned to each region as a weighted mean. Avezanno (0.05 mg/L; range 0.040 to 0.058 mg/L; population of about 126,000) Bracciano (1.45 mg/L; range 0.15 to 3.40 mg/L; population of about 73,000) |
Groundwater measurements of almost 9,000 wells. Fluoride concentrations estimated for each residence by using weighted medians, smoothed interpolations. Categories: <0.1, 0.1 to 0.3, 0.3 to 0.5, 0.5 to 1.0, 1 to 1.5, and > 1.5 mg/L. Highest category corresponded to sampled concentrations of less than detection level to approximately 6 mg/L. |
|
Fractures at specific anatomical sites, reported by gender |
First recorded hip fracture |
|
Rates and 95% CI based on age-adjusted rates per 1,000 person years. |
Crude and adjusted rate ratios using Cox regression based on person years, compared with lowest exposure group. Age stratification based on age at start of follow-up period. Fluoride analyzed as categorical and continuous variable. |
|
95% CIs |
95% CI around the rate ratio. |
|
Authors relied on similarity of region to control for confounding. Analysis did not stratify or adjust for age, although mean ages of cases are shown (including whether the probabilities of their differences are P < 0.05). |
Analyzed controlling for age and geographic sector. Age adjustment was conducted within broad strata of 50 to 64 and 65 to 80 years. No information on nutrition, alcohol use, or physical activity. |
|
|
Arnala et al. (1986) |
Jacqmin-Gadda et al. (1995) |
|
Results |
Age-combined totals similar: 12.4/10,000 in low-fluoride, 11.9/10,000 in fluoridated, and 12.4/10,000 in high-fluoride areas. Component age groups generally similar to each other across exposure groups, except that age 80+ had lower incidence in the high-fluoride area. |
For higher versus lower fluoride exposures: OR = 1.86 (1.02 to 3.36), P = 0.04 for hip fractures; OR = 0.98 (0.80 to 1.21) for all fractures. ORs adjusted for variables associated with hip fractures (age, gender, Quetelet) or total fractures (age, gender). Calcium in water did not appear to be included in the model. |
|
Overall value of study regarding evaluation fracture risk at 2 mg/L |
Weak |
Weak |
|
Comments |
The paper was primarily devoted to histomorphology and bone fluoride concentrations in iliac crest. The results of that portion of the study are summarized in the accompanying text insofar as they bear on the incidence part of the paper. Incomplete reporting methods; insufficient control of confounding; inability to assess cumulative exposure; possibility of nonsystematic or biased case ascertainment/assignment; adjustment of group level covariate (region) rather than individual-level covariates. |
Paper was short (a letter to the editor) and did not have sufficient detail to assess the distribution of fluoride exposure with the higher category; lacked information on age subgroups and on genders; inability to assess cumulative exposure; referent group has very low exposure (<0.11 mg/L). |
|
Fabiani et al. (1999) |
Kurttio et al. (1999) |
|
Rates for low-fluoride area were statistically greater compared with Bracciano in the following categories: Females: femoral neck (hip), femur NOS (not otherwise specified), proximal humerus, nose, wrist Males: femoral neck (hip), femur NOS, nose, wrist Specifically for hip fracture (Avezanno/ Bracciano, rate per 1,000 person-years): males, 0.28/0.06, RR = 4.28 (95% CI, 4.16 to 4.40), average ages 70 and 52, respectively; females, 0.75/0.28, RR = 2.64 (95% CI 2.54 to 2.75), average ages 75 and 78, respectively. |
For comparisons between the >1.5-mg/L group and the <0.1-mg/L group (ages 50 to 65): Adjusted RR = 2.09 (95 CI, 1.16 to 3.76) in women, RR = 0.87 (95% CI, 0.35 to 2.16) in men. For all ages combined, no associations apparent. For fluoride as a continuous variable: RR = 1.44 (95% CI, 1.12 to 1.86) for women below age 65 at start of follow-up, and RR = 0.75 (95% CI, 0.51 to 1.12) for men in same age stratum (age and region adjusted). Women ages 55 to 69 had the most elevated RR in the continuous-variable analysis. Among separate 5-year components of followup period, the results were inconsistent. |
|
Weak |
Strong |
|
Serious design and analysis limitations. No data that would inform an assessment of a gradient. The dimension of the reported protective effect is not credible. |
Suggestive of hip fracture risk, with continuous gradient from lowest to highest exposures. |
ratio.) The committee found that because no data were presented on the distribution of fluoride exposure within the different groups, because data on gender and age were not reported separately, and because no parameters for assessing cumulative exposure were provided, reliable conclusions could not be drawn from this study.
Fabiani et al. (1999) conducted a study in two sociodemographically similar regions in central Italy. One region had fluoride concentrations in drinking water of 0.05 mg/L and the second region had fluoride at 1.45 mg/L. A significantly greater rate of fracture incidence, particularly femur fractures, were found in the low-exposure community. The relative risk was 4.28 (95% CI, 4.16 to 4.40) for males and 2.64 (95% CI, 2.54 to 2.75) for females. These risks were based on age-adjusted rates per 1,000 person-years. However, the number of cases was not provided and the mean age of cases in the two towns varied greatly in some instances. The investigators relied on similarity of regions to control for confounding, but it should be noted that the high-fluoride area included seven towns near Rome, whereas the lower-fluoride area included 35 towns further from Rome. Because of the serious design and analysis limitations of the study, the committee placed little weight on this study.
Overall, the committee finds that the available epidemiologic data for assessing bone fracture risk in relation to fluoride exposure around 2 mg/L is suggestive but inadequate for drawing firm conclusions about the risk or safety of exposures at that concentration. There is only one strong report to inform the evaluation, and, although that study (Kurttio et al. 1999) indicated an increased risk of fractures, it is not sufficient alone to base judgment of fracture risk for people exposed at 2 mg/L. It should be considered, however, that the Li et al. (2001) and Alarcón-Herrera et al. (2001) studies reported fracture increases (although imprecise with wide confidence intervals) between 1 and 4 mg/L, giving support to a continuous exposure-effect gradient in this range.
Skeletal Fluorosis
Excessive intake of fluoride will manifest itself in a musculoskeletal disease with a high morbidity. This pathology has generally been termed skeletal fluorosis. Four stages of this affliction have been defined, including a preclinical stage and three clinical stages that characterize the severity. The preclinical stage and clinical stage I are composed of two grades of increased skeletal density as judged by radiography, neither of which presents with significant clinical symptoms. Clinical stage II is associated with chronic joint pain, arthritic symptoms, calcification of ligaments, and osteosclerosis of cancellous bones. Stage III has been termed “crippling” skeletal fluorosis because mobility is significantly affected as a result of excessive calcifications
in joints, ligaments, and vertebral bodies. This stage may also be associated with muscle wasting and neurological deficits due to spinal cord compression. The current MCLG is based on induction of crippling skeletal fluorosis (50 Fed. Reg. 20164 [1985]). Because the symptoms associated with stage II skeletal fluorosis could affect mobility and are precursors to more serious mobility problems, the committee judges that stage II is more appropriately characterized as the first stage at which the condition is adverse to health. Thus, this stage of the affliction should also be considered in evaluating any proposed changes in drinking-water standards for fluoride.
Descriptions of skeletal fluorosis date back to the 1930s, when the pathology was first recognized in India in areas of endemic fluoride exposure (Shortt et al. 1937) and in occupationally exposed individuals in Denmark (Roholm 1937). From an epidemiological standpoint, few cases of clinical skeletal fluorosis have been documented in the United States. Stevenson and Watson (1957) performed a large retrospective study involving 170,000 radiologic examinations1 in people from Texas and Oklahoma, where many communities have fluoride water concentrations above 4 mg/L. They radiographically diagnosed only 23 cases of fluoride osteosclerosis in people consuming fluoride at 4 to 8 mg/L and no cases in people exposed to less (the number of people exposed in these categories was not provided). The cases (age 44 to 85) did not have unusual amounts of arthritis or back stiffness given their age (details not provided). Eleven had bone density of an extreme degree, and nine had more than minimal calcification of pelvic ligaments. The authors found no relationship between radiographic findings and clinical diagnosis or symptoms (details not provided). Cases were not classified as to the stage of the fluorosis (using the scheme discussed earlier). Based on the information in the paper, the committee could not determine whether stage II fluorosis was present. In a study of 253 subjects, Leone et al. (1955a) reported increased bone density and coarsened trabeculation in residents of a town with fluoride at 8 mg/L relative to another town with fluoride at 0.4 mg/L. Radiographic evidence of bone changes occurred in 10% to 15% of the exposed residents and was described as being slight and not associated with other physical findings except enamel mottling. The high-fluoride town was partially defluoridated in March 19522 (Maier 1953; Leone et al. 1954a,b; 1955b), a detail not mentioned in the radiographic study (Leone
et al. 1955a) but which could have affected its results and interpretation. Leone et al. (1954a,b; 1955b) state that “any significant physiological manifestations of prolonged exposure would not be expected to have regressed materially in the 18 months of partial defluoridation.” However, Likins et al. (1956) reported that urinary fluoride concentrations in males fell from means of 6.5 (children) and 7.7 (adults) mg/L before defluoridation to 4.9 and 5.1 mg/L, respectively, after 1 week, 3.5 and 3.4 mg/L, respectively, after 39 weeks, and 2.2 and 2.5 mg/L, respectively, after 113 weeks. These results indicate that, following defluoridation of the water supply, substantial changes in fluoride balance were occurring in the residents, including the apparent remobilization of fluoride from bone.
In patients with reduced renal function, the potential for fluoride accumulation in the skeleton is increased (see Chapter 3). It has been known for many years that people with renal insufficiency have elevated plasma fluoride concentrations compared with normal healthy persons (Hanhijärvi et al. 1972) and are at a higher risk of developing skeletal fluorosis (Juncos and Donadio 1972; Johnson et al. 1979). In cases in which renal disease and skeletal fluorosis were simultaneously present, it still took high concentrations of fluoride, such as from daily ingestion of 4 to 8 L of water containing fluoride at 2 to 3 mg/L (Sauerbrunn et al. 1965; Juncos and Donadio 1972), at least 3 L/day at 2 to 3 mg/L (Johnson et al. 1979), or 2 to 4 L/day at 8.5 mg/L (Lantz et al. 1987) to become symptomatic.
Most recently, the Institute of Medicine evaluated fluoride intake and skeletal fluorosis and was able to find only five reported cases of individuals with stage III skeletal fluorosis in the United States from approximately 1960 to 1997 (IOM 1997). Interestingly, however, a recent report has documented an advanced stage of skeletal fluorosis in a 52-year-old woman consuming 1 to 2 gal of double-strength instant tea per day throughout her adult life (Whyte et al. 2005). Her total fluoride intake was estimated at 37 to 74 mg/day from exposure to fluoride from well water (up to 2.8 mg/L) and instant tea. The report also documented the fluoride content of commercial instant teas and found substantial amounts in most brands. This illustrates the possibility that a combination of exposures can lead to higher than expected fluoride intake with associated musculoskeletal problems. Another case, documented by Felsenfeld and Roberts (1991), indicates the development of skeletal fluorosis from consumption of well water containing fluoride at 7 to 8 mg/L for 7 years. Renal insufficiency was not a factor in this case, but water consumption was considered likely to have been “increased” because of hot weather. Both cases mention joint stiffness or pain, suggesting at least stage II skeletal fluorosis.
From reports from the 1950s through the 1980s, it appears that preclinical bone changes and symptoms of clinical stages I and II may occur with bone concentrations between 3,500 and 12,900 mg/kg (Franke et al.
1975; Dominok et al. 1984; Krishnamachari 1986). The Public Health Service (PHS 1991) has reported that patients with preclinical skeletal fluorosis have fluoride concentrations between 3,500 and 5,500 mg/kg by ash weight. Clinical stage I patients have concentrations in the range of 6,000 to 7,000 mg/kg, stage II patients range from 7,500 to 9,000 mg/kg, and stage III patients have fluoride concentrations of 8,400 mg/kg and greater.3
However, a broader review of the literature on bone fluoride concentrations in patients with skeletal fluorosis revealed wider and overlapping ranges associated with different stages of the condition. Tables 5-6 and 5-7 show the reported concentrations of fluoride in bone ash and in bone (dry fat-free material) in cases of skeletal fluorosis. Most authors reported ash concentrations; others reported the dry weight concentrations or both types of results. Because ash contents (fraction of bone remaining in the ash) range widely,4 the committee did not convert dry weight concentrations to ash concentrations. As reported ranges for various bones in individuals can differ, the tables list the type of bone sampled, distinguishing between measurements of iliac crest or pelvis and other bones.
On the basis of data on fluoride in the iliac crest or pelvis, fluoride concentrations of 4,300 to 9,200 mg/kg in bone ash have been found in cases of stage II skeletal fluorosis, and concentrations of 4,200 to 12,700 mg/kg in bone ash have been reported in cases of stage III fluorosis. The overall ranges for other bones are similar. These ranges are much broader than those indicated by PHS (1991). Baud et al. (1978) showed an overlap in the fluoride content in iliac crest samples between their controls (mean 1,036 mg/kg, range <500 to >2,500) and their cases (mean 5,617 mg/kg, range <2,500 to >10,000). The above ranges overlap the measurements reported by Zipkin et al. (1958), for which no evidence of fluorosis was reported (4,496 ± 2015 and 6,870 ± 1629 mg/kg ash in iliac crest at 2.6 and 4 mg/L, respectively). The expected degree of skeletal fluorosis was not found in two small groups of patients dialyzed with fluoride-containing water, who accumulated average bone-ash fluoride concentrations of 5,000 mg/kg and 7,200 mg/kg (Erben et al. 1984). Some of the cases with the lowest values (e.g., Teotia and Teotia 1973; Pettifor et al. 1989) were known to have hypocalcemia or secondary hyperparathyroidism; many of the industrial case reports described no hypocalcemia. Thus, it appears that fluoride content in bone may be a marker of the risk of skeletal fluorosis. In other words, the likelihood and severity of clinical skeletal fluorosis increase with the
TABLE 5-6 Reported Concentrations of Fluoride in Bone Ash in Cases of Skeletal Fluorosis
|
|
Fluoride Concentration in Bone Ash, mg/kg in Bone Ash |
|
|
|
|
Stage of Skeletal Fluorosis |
Iliac Crest or Pelvis |
Other Bones |
Number of Individuals |
Reference |
|
Preclinical stage |
||||
|
Vague symptoms |
4,100 |
|
2 |
Franke and Auermann 1972 |
|
|
4300 |
|
|
|
|
Vague symptoms |
3,500 to 4,500 |
|
Authors’ summary |
Franke et al. 1975 |
|
Stage 0 to 1 |
||||
|
Stage 0 to I |
5,000 |
|
1 |
Franke and Auermann 1972 |
|
Stage 0 to I |
6,900 (mean) |
|
2 |
Schlegel 1974 |
|
Stage 0 to I |
5,000 to 5,500 |
|
Authors’ summary |
Franke et al. 1975 |
|
Stage 1 |
||||
|
Stage I |
6,000 |
|
2 |
Franke and Auermann 1972 |
|
|
6400 |
|
|
|
|
Stage I |
5,200 (mean) |
|
8 |
Schlegel 1974 |
|
Stage I |
6,000 to 7,000 |
|
Authors’ summary |
Franke et al. 1975 |
|
Stage 2 |
||||
|
Second phase |
9,200 |
3,100 to 9,900 |
1 |
Roholm 1937 |
|
Stage I to II |
8,700 |
|
1 |
Franke and Auermann 1972 |
|
Stage II |
7,700 |
|
2 |
Franke and Auermann 1972 |
|
|
7800 |
|
|
|
|
Stage II |
7,500 (mean) |
|
9 |
Schlegel 1974 |
|
Stage II |
7,500 to 9,000 |
|
Authors’ summary |
Franke et al. 1975 |
|
Stage II |
4,300 |
2,500 to 5,000 |
1 |
Dominok et al. 1984 |
|
|
4,700a |
|
|
|
|
Stage II |
8,800 |
4,900 to 11,100 |
1 |
Dominok et al. 1984 |
|
|
8900a |
|
|
|
|
Stage II |
|
2,900 to 4,400 |
1 |
Dominok et al. 1984 |
|
Stage 3 |
||||
|
Third phase |
|
7,600 to 13,100 |
1 |
Roholm 1937 |
|
Stage 3 |
|
6,300 |
1 |
Singh and Jolly 1961 |
|
Stage III |
|
11,500 |
1 |
Franke and Auermann 1972 |
|
Crippling fluorosis |
4,200 |
|
1 |
Teotia and Teotia 1973 |
|
Stage III |
8,400 |
|
1 |
Schlegel 1974 |
|
Stage III |
>10,000 |
|
Authors’ summary |
Franke et al. 1975 |
|
|
Fluoride Concentration in Bone Ash, mg/kg in Bone Ash |
|
|
|
|
Stage of Skeletal Fluorosis |
Iliac Crest or Pelvis |
Other Bones |
Number of Individuals |
Reference |
|
Stage III |
10,000 |
9,000 to 11,700 |
1 |
Dominok et al. 1984 |
|
Stage III |
9,100 |
4,200 to 11,000 |
1 |
Dominok et al. 1984 |
|
Stage III |
12,700 |
7,600 to 12,900 |
1 |
Dominok et al. 1984 |
|
Stage III |
8,600 |
8,500 to 12,400 |
1 |
Dominok et al. 1984 |
|
|
8,700a |
|
|
|
|
Stage not given, or range of stages |
||||
|
Skeletal fluorosis |
|
700 to 6,800b (mean, 3,430) |
10 |
Singh and Jolly 1961; see also Singh et al. 1961 |
|
Old fluorosis, 7 years without fluoride exposure |
3,000 |
|
1 |
Franke and Auermann 1972 |
|
Skeletal fluorosis |
2,650 |
|
4 |
Teotia and Teotia 1973 |
|
|
3,780 |
|
|
|
|
|
4,750 |
|
|
|
|
|
5,850 |
|
|
|
|
Industrial fluorosis |
5,617 (2,143)c |
|
43 (54 samples) |
Baud et al. 1978; Boillat et al. 1980 |
|
Endemic genu valgum |
|
7,283 (416)d |
20 (37 samples) |
Krishnamachari 1982 |
|
Skeletal fluorosis |
4,200 to 10,100 |
|
9 |
Boivin et al. 1986 |
|
Skeletal fluorosis |
13,300 (2,700)c |
|
6 |
Boivin et al. 1988 (summary of studiese) |
|
|
8,900 (3,400)c |
|
5 |
|
|
|
6,900 (1,900)c |
|
13 |
|
|
|
5,600 (2,100)c |
|
54 |
|
|
|
6,600 (2,700)c |
|
4 |
|
|
|
7,600 (4,800)c |
|
14 |
|
|
Skeletal fluorosis |
7,900 (3,600)c (range: 4,200 to 22,000) |
|
29 |
Boivin et al. 1989; 1990 f |
|
Admitted to hospital for skeletal pain or skeletal deformities |
5,580 (980)c (range: 4,430 to 6,790) |
|
7 |
Pettifor et al. 1989 |
|
aSamples from right and left sides in same individual. bTibia or iliac crest; includes 1 case of stage III fluorosis listed separately above. cIndicates mean and standard deviation. dIndicates mean and standard error. eIncludes some studies (or individuals from studies) listed separately above. fProbably includes individuals from other studies listed above. |
||||
TABLE 5-7 Reported Concentrations of Fluoride in Bone (Dry Fat-Free Material) in Cases of Skeletal Fluorosis
|
|
Fluoride Concentration in Bone, mg/kg in Dry Fat-Free Material |
|
|
|
|
Stage of Skeletal Fluorosis |
Iliac Crest or Pelvis |
Other Bones |
Number of Individuals |
Reference |
|
Preclinical stage |
||||
|
Vague symptoms |
1,700 and 2,100 |
|
2 |
Franke and Auermann 1972 |
|
Stage 0 to 1 |
||||
|
Stage 0 to I |
1,900 |
|
1 |
Franke and Auermann 1972 |
|
Stage 0 to I |
3,000 (mean) |
|
5 |
Schlegel 1974 |
|
Stage 1 |
||||
|
Early |
|
5,000 to 7,000 |
1 |
Wolff and Kerr 1938 (cited in Jackson and Weidmann 1958) |
|
Early |
|
6,260 and 7,200 |
2 |
Sankaran and Gadekar 1964 |
|
Stage I |
2,300 and 2,900 |
|
2 |
Franke and Auermann 1972 |
|
Stage I |
3,200 (mean) |
|
15 |
Schlegel 1974 |
|
Stage 2 |
||||
|
Moderate |
|
7,680 |
1 |
Sankaran and Gadekar 1964 |
|
Stage I to II |
4,300 |
|
1 |
Franke and Auermann 1972 |
|
Stage II |
4,100 and 4,600 |
|
2 |
Franke and Auermann 1972 |
|
Stage II |
3,000 (mean) |
|
18 |
Schlegel 1974 |
|
Stage 3 |
||||
|
Skeletal fluorosis |
|
8,600 |
1 |
Sankaran and Gadekar 1964 |
|
Advanced |
|
8,800 and 9,680 |
2 |
Sankaran and Gadekar 1964 |
|
Stage III |
3,600 (mean) |
|
4 |
Schlegel 1974 |
|
Stage not given |
||||
|
Old fluorosis, 7 years without fluoride exposure |
1,700 |
|
1 |
Franke and Auermann 1972 |
bone fluoride content, but a given concentration of bone fluoride does not necessarily correspond to a certain stage of skeletal fluorosis in all cases. Other factors (e.g., calcium intake) appear to influence fluorosis severity at different concentrations of bone fluoride.
Overall, the committee finds that the predicted bone fluoride concentrations that can be achieved from lifetime exposure to fluoride at 4 mg/L (10,000 to 12,000 mg/kg bone ash) fall within or exceed the ranges of concentrations that have been associated with stage II and stage III skeletal fluorosis. Based on the existing epidemiologic literature, stage III skeletal fluorosis appears to be a rare condition in the United States. As discussed above, the committee judges that stage II skeletal fluorosis is also an adverse health effect. However, the data are insufficient to provide a quantitative estimate of the risk of this stage of the affliction. The committee could not determine from the existing epidemiologic literature whether stage II skeletal fluorosis is occurring in U.S. residents who drink water with fluoride at 4 mg/L. The condition does not appear to have been systematically investigated in recent years in U.S. populations that have had long-term exposures to high concentrations of fluoride in drinking water. Thus, research is needed on clinical stage II and stage III skeletal fluorosis to clarify the relationship of fluoride ingestion, fluoride concentration in bone, and clinical symptoms.
EFFECT OF FLUORIDE ON CHONDROCYTE METABOLISM AND ARTHRITIS
The two key chondrocyte cell types that are susceptible to pathological changes are articular chondrocytes in the joint and growth plate chondrocytes in the developing physis. The medical literature on fluoride effects in these cells is sparse and in some cases conflicting.
From physical chemical considerations, it might be expected that mineral precipitates containing fluoride would occur in a joint if concentrations of fluoride and other cations (such as Ca2+) achieved a high enough concentration. A single case report by Bang et al. (1985) noted that a 74-year-old female who was on fluoride therapy for osteoporosis for 30 months had a layer of calcified cartilage containing 0.39% fluoride (or 3,900 mg/kg) by ash weight in her femoral head. The calcification was also visible on x-ray. Unfortunately, the limitation of this observation in a single patient is the lack of information on the preexistence of any calcified osteophytes. Nevertheless, it does indicate that at high therapeutic doses fluoride can be found in mineralizing nodules in articular cartilage.
Studies evaluating patient groups with a greater number of subjects found that the use of fluoride at therapeutic doses in rheumatoid patients showed a conflicting result. In one report (Duell and Chesnut 1991), fluoride exacerbated symptoms of rheumatoid arthritis, but, in another case
(Adachi et al. 1997), it was “well tolerated” with no evidence of worsening of the arthritis. No indications from either study implied that fluoride had a causal relationship with the rheumatoid arthritis. Perhaps the only study in the literature that attempts to link fluoride exposure to the induction of arthritis (osteoarthritis) is from Savas et al. (2001), who indicated that Turkish patients with demonstrated endemic fluorosis had a greater severity of osteoarthritic symptoms and osteophyte formation than age- and sex-matched controls.
The veterinary literature also contains a report indicating that, in 21 dairy herds consuming fluoride-containing feed and water, of the 100 cows examined and determined to have arthritic changes, the bone fluoride concentrations ranged from 2,000 to 8,000 mg/kg (Griffith-Jones 1977).
There are no data from which a dose-response relationship can be drawn regarding fluoride intake and arthritis in humans. However, in a rat study, Harbrow et al. (1992) showed articular changes with fluoride at 100 mg/L in drinking water but no effect at 10 mg/L. The changes with fluoride at 100 mg/L were a thickening of the articular surface (rather than a thinning as would be expected in arthritis) and there were no effects on patterns of collagen and proteoglycan staining. There are no comprehensive reports on the mechanism of fluoride effects in articular chondrocytes in vitro.
The effect of fluoride on growth plate chondrocytes is even less well studied than the effect on articular chondrocytes. It has been demonstrated that chronic renal insufficiency in a rat model can increase the fluoride content in the growth plate and other regions of bone (Mathias et al. 2000); however, this has not been known to occur in humans. Fluoride has also been shown to negatively influence the formation of mineral in matrix vesicles at high concentrations. Matrix vesicles are the ultrastructural particles responsible for initiating mineralization in the developing physis (Sauer et al. 1997). This effect could possibly account, in part, for the observation that fluoride may reduce the thickness of the developing growth plate (Mohr 1990).
In summary, the small number of studies and the conflicting results regarding the effects of fluoride on cartilage cells of the articular surface and growth plate indicate that there is likely to be only a small effect of fluoride at therapeutic doses and no effect at environmental doses.
FINDINGS
Fluoride is a biologically active ion with demonstrable effects on bone cells, both osteoblasts and osteoclasts. Its most profound effect is on osteoblast precursor cells where it stimulates proliferation both in vitro and in vivo. In some cases, this is manifested by increases in bone mass in vivo.
The signaling pathways by which this agent works are slowly becoming elucidated.
Life-long exposure to fluoride at the MCLG of 4 mg/L may have the potential to induce stage II or stage III skeletal fluorosis and may increase the risk of fracture. These adverse effects are discussed separately below.
The current MCLG was designed to protect against stage III skeletal fluorosis. As discussed above, the committee judges that stage II is also an adverse health effect, as it is associated with chronic joint pain, arthritic symptoms, slight calcification of ligaments, and osteosclerosis of cancellous bones. The committee found that bone fluoride concentrations estimated to be achieved from lifetime exposure to fluoride at 2 mg/L (4,000 to 5,000 mg/kg ash) or 4 mg/L (10,000 to 12,000 mg/kg ash) fall within or exceed the ranges historically associated with stage II and stage III skeletal fluorosis (4,300 to 9,200 mg/kg ash and 4,200 to 12,700 mg/kg ash, respectively). This suggests that fluoride at 2 or 4 mg/L might not protect all individuals from the adverse stages of the condition. However, this comparison alone is not sufficient evidence to conclude that individuals exposed to fluoride at those concentrations are at risk of stage II skeletal fluorosis. There is little information in the epidemiologic literature on the occurrence of stage II skeletal fluorosis in U.S. residents, and stage III skeletal fluorosis appears to be a rare condition in the United States. Therefore, more research is needed to clarify the relationship between fluoride ingestion, fluoride concentrations in bone, and stage of skeletal fluorosis before any firm conclusions can be drawn.
Although a small set of epidemiologic studies were useful for evaluating bone fracture risks from exposure to fluoride at 4 mg/L in drinking water, there was consistency among studies using ecologic exposure measures to suggest the potential for an increased risk. The one study using serum fluoride concentrations found no appreciable relationship to fractures. Because serum fluoride concentrations may not be a good measure of bone fluoride concentrations or long-term exposure, the ability to shown an association might have been diminished. Biochemical and physiological data indicate a biologically plausible mechanism by which fluoride could weaken bone. In this case, the physiological effect of fluoride on bone quality and risk of fracture observed in animal studies is consistent with the observational evidence. Furthermore, the results of the randomized clinical trials were consistent with the observational studies. In addition, a dose-response relationship is indicated. On the basis of this information, all members of the committee agreed that there is scientific evidence that under certain conditions fluoride can weaken bone and increase the risk of fractures. The majority of the committee concluded that lifetime exposure to fluoride at drinking-water concentrations of 4 mg/L or higher is likely to increase fracture rates in the population, compared with exposure at 1 mg/L, particularly in some
susceptible demographic groups that are more prone to accumulate fluoride in their bones. However, three of the 12 members judged that the evidence only supported a conclusion that the MCLG might not be protective against bone fracture. They judge that more evidence that bone fractures occur at an appreciable frequency in human populations exposed to fluoride at 4 mg/L is needed before drawing a conclusion that the MCLG is likely to be not protective.
Few studies have assessed fracture risk in populations exposed to fluoride at 2 mg/L in drinking water. The best available study was from Finland, which provided data that suggested an increased rate of hip fracture in populations exposed to fluoride at >1.5 mg/L. However, this study alone is not sufficient to determine the fracture risk for people exposed to fluoride at 2 mg/L in drinking water. Thus, the committee finds that the available epidemiologic data for assessing bone fracture risk in relation to fluoride exposure around 2 mg/L are inadequate for drawing firm conclusions about the risk or safety of exposures at that concentration.
RECOMMENDATIONS
-
A more complete analysis of communities consuming water with fluoride at 2 and 4 mg/L is necessary to assess the potential for fracture risk at those concentrations. These studies should use a quantitative measure of fracture such as radiological assessment of vertebral body collapse rather than self-reported fractures or hospital records. Moreover, if possible, bone fluoride concentrations should be measured in long-term residents.
-
The effects of fluoride exposure in bone cells in vivo depend on the local concentrations surrounding the cells. More data are needed on concentration gradients during active remodeling. A series of experiments aimed at quantifying the graded exposure of bone and marrow cells to fluoride released by osteoclastic activity would go a long way in estimating the skeletal effects of this agent.
-
A systematic study of stage II and stage III skeletal fluorosis should be conducted to clarify the relationship of fluoride ingestion, fluoride concentration in bone, and clinical symptoms. Such a study might be particularly valuable in populations in which predicted bone concentrations are high enough to suggest a risk of stage II skeletal fluorosis (e.g., areas with water concentrations of fluoride above 2 mg/L).
-
More research is needed on bone concentrations of fluoride in people with altered renal function, as well as other potentially sensitive populations (e.g., the elderly, postmenopausal women, people with altered acid-balance), to better understand the risks of musculoskeletal effects in these populations.


















































