3
Framework to Characterize Biomarkers and Uses of Biomonitoring
RATIONALE FOR A FRAMEWORK TO CHARACTERIZE BIOMARKERS
The pace at which developments of biomarkers occur tends to exceed the biomonitoring community’s ability to cope with its obligations to ensure proper transmission of information on the meaning of the measurements. There are increasing anecdotal stories in the mass media about the concerns of people whose blood has been found to contain more than 100 chemicals. But the fact is that the numbers of studies, subjects, and substances determined in human biomonitoring are increasing (e.g., National Health and Nutrition Examination Survey), as is the awareness of the general public of those chemicals in our bodies (e.g., Sexton et al. 2004). Consequently, there is a need for clarification of what biomarkers can and cannot be used for. Despite proper warnings to the effect that “the measurement of an environmental chemical in a person’s blood or urine does not by itself mean the chemical causes disease” (CDC 2005), people who have almost unlimited access to good and bad information about chemicals no longer appear satisfied with such general words of caution. Not only is the general population entitled to know the advantages and limitations of biomonitoring, but those with public-health responsibilities also need to be adequately informed. The correct interpretation of biomarker measurements is therefore of utmost importance.
In many cases, data on biomarkers are obtained with the sole purpose of collecting information about their background concentrations in the
general population, not to assess health risks associated with the measurements. As will be discussed in later chapters, such studies are laudable in that they contribute to our knowledge about human exposure to environmental chemicals. But the limitations of that type of information are not necessarily recognized by all who need to know and understand them. The committee considered that clarification of the properties of the various biomarkers of exposure would be useful in helping to understand and clarify what can be said about a given measurement. A systematic framework to characterize the properties of biomarkers would help to inform scientists and the general population about biomarkers and their meaning when they are used in biomonitoring studies. It would also allow assessment of potential research gaps that need to be addressed to meet the requirements of specific uses of biomarkers. Detailed information about the interpretation of biomonitoring data is provided in Chapter 5. The present chapter focuses on the properties that characterize biomarkers of exposure in general. These properties are based on a weight-of-evidence approach that takes into account the specific context of a biomonitoring study under consideration.
TYPES OF BIOMARKERS
Chapter 1 describes the relationship between exposure to a toxic chemical and its clinically relevant health effects as a series of steps along a continuum. There often is no clear-cut distinction between some of the steps, and Figure 1-1 can help position three types of biomarkers: biomark-ers of exposure, of effect, and of susceptibility.
As the name implies, biomarkers of exposure allow assessment of exposure to a chemical on the basis of its measurement in a biologic matrix (NRC 1991). Typical examples are the measurement of dioxins in blood or blood lipids, mercury in hair, benzene in exhaled breath, and cadmium in urine. In itself, quantification of such a biomarker in a biologic matrix proves only that the chemical is in the organism. If the substance is not otherwise known to be endogenous, it can be concluded that there has been a transfer from the external environment to the individual organism. Any further interpretation of the concentration of a biomarker of exposure requires additional information about some of the relationships in the continuum. If the biomarker of exposure indicates that a chemical reached a critical target in the organism— for example, if it formed a DNA adduct—there is a greater likelihood of a potential link with a biological perturbation. As discussed in Chapter 1, this report focuses on biomarkers of exposure.
Biomarkers of effect are used to assess changes that have occurred in the biochemical or physiologic makeup of an individual. The further to the right in the continuum the biomarker is, the greater the clinical or health
relevance of its measurement. An abnormal value of a biomarker of effect near the center of the continuum may not signal detrimental effects on the health of an individual or group if, for example, the perturbation is reversible and steps are taken to ensure that the exposure that caused it ceases. However, the abnormal value can serve as a trigger for a remedial action. Examples of biomarkers of effect are plasma cholinesterase activity, urinary γ2-microglobulin, and packed red-cell volume (hematocrit).
The passage from one step to the next along the continuum will often depend on a person’s characteristics. A biomarker that allows the assessment of a person’s susceptibility to alter the progression along the continuum is called a biomarker of susceptibility. Examples are enzymatic genotypes and phenotypes, such as those seen with glutathione-S-transferase M, a phase II conjugation enzyme that often contributes to the detoxification of some electrophilic compounds (Perbellini et al. 2002).
KEY USES OF BIOMONITORING DATA
Biomonitoring has been a key tool in some landmark public-health actions. The incentive to reduce collective exposure to lead and thereby to protect the public in general and children, who are more sensitive to its deleterious neurotoxic effects, would probably have been less if we had not known how much lead was reaching our bodies and how much lead in our bodies was problematic. Just as important, the prospective followup and monitoring of blood lead clearly illustrated that our exposure-reduction strategies were successful in lowering it (Pirkle et al. 1995). The lead case is developed further in Chapter 5. The determination of aflatoxin-derived DNA adducts was also a key step in increasing our understanding of the carcinogenic effect of this naturally occurring toxin. It allowed us to derive a quantitative risk assessment of its hepatocarcinogenicity, which guided exposure reduction (Sharma and Farmer 2004). More recently, the measurement of biomarkers of exposure to methyl parathion was instrumental in understanding local epidemics of poisoning with this insecticide and in establishing remediation strategies (Rubin et al. 2002). The once unthinkable efforts to reduce exposure to second-hand smoke were proved successful through the Centers for Disease Control and Prevention (CDC) National Reports on Human Exposure to Environmental Chemicals. Indeed, from 1988 to 2002, a 68% reduction in urinary concentrations of cotinine (a specific biomarker of exposure to nicotine) was demonstrated in children 4-11 years old (Sinks 2005). Without specific measurements, the actual dose resulting from second-hand smoke would have been only inferred from expensive external measurements of airborne nicotine from cigarette smoke. The indirect assessment might also have been considered merely conjectural and unproved. But the unambiguous demonstration that it was
an important pathway of exposure to chemicals contained in tobacco smoke and that control policies reduced exposure was a key step in the success of the public-health strategy related to smoking (active and passive). Some argue strongly that public-health professionals and clinicians would benefit from access to a large pool of well-characterized biomarkers to guide both prevention of adverse health effects and health promotion (Jackson 2005).
Biomonitoring can also serve as a valuable tool in various public-health activities aimed at avoiding the deleterious effects associated with exposure to toxic substances. From a risk-assessment and risk-management perspective, the determination of markers of internal exposure may serve a number of purposes that can be situated along a continuum of risk-assessment and -management activities (e.g., Burke et al. 1992). Four broad categories are represented here because they pertain to activities that use biomonitoring: scoping, status and trends, exposure and health research, and risk assessment. Examples of types of activities included in each category are listed in Box 3-1. Scoping is a basic risk-management activity that may provide the first indication of a potential problem. The qualitative information gathered through scoping assists in addressing fundamental questions such as, is a chemical present in the biomonitoring sample (Burke et al. 1992)? Examples of scoping include screening, exploratory and source investigations,
|
BOX 3-1 Continuum of Risk-Assessment and -Management Activities Related to Exposure Biomonitoring
|
and societal hazard identification. Status and trends provide an assessment of the concentration of chemical in the population (for example, blood or urine) and whether the concentration of the chemical may be varying over time, across geographic regions, or within populations. Information on status and trends can assist with population surveillance and research. The biomonitoring data may be utilized to assess potential sources of exposure when combined with pathways research. Biomonitoring data can be used to support exposure and health research. Population-based biomonitoring data used in epidemiologic studies can identify highly exposed subpopulations and health endpoints of concern. This in turn can be applied to community and occupational studies. The data can also identify information needs for toxicologic, pharmacokinetic, and pharmacodynamic research. Biomonitoring data can be applied in clinical evaluations (that is, childhood lead) to assess individual risks or exposure.
All those activities have specific purposes and may be conducted alone (for example, for priority-setting among public-health actions) or as parts of a multitier strategy. Links between risk-assessment and -management activities (why biomonitoring is conducted) and the properties of the biomarkers (what characteristics our biomonitoring tools need to have for us to be able to conduct these activities) will be presented below.
PROPERTIES OF AND GROUPING FRAMEWORK FOR BIOMARKERS OF EXPOSURE
A simplified version of the relationships described in the continuum presented in Figure 1-1 shows the links between external dose,1 internal dose, and biologic effects as a triangle (Figure 3-1). The diagram illustrates the “operational” relationships between these elements, not the biologic relationships. Indeed, a systemic toxicant has to penetrate the organism before exerting its action. The direct link between external dose and biologic effect has no toxicologic meaning itself, but, as will be discussed below, more published information is available on the relationship between the external dose of toxicants and their biologic effects than between the internal dose—as assessed by a biomarker measurement—and biologic effects. The interpretation of biomonitoring data, and thus possible key uses, will depend on the body of knowledge about these links from animal or human studies.
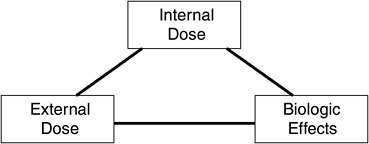
FIGURE 3-1 Operational relationships between internal dose, external dose, and biologic effects. Internal dose is measured through biomonitoring. Source: Adapted from Bernard and Lauwerys 1986.
Ultimately, a biomarker of exposure for which all the relationships in Figure 3-1 have been clearly delineated will probably be useful for risk-assessment purposes. The validation of biomarkers of exposure useful for risk-assessment purposes has been described (Schulte and Talaska 1995; Dor et al. 1999; WHO 2001). Chapter 5 provides case studies to illustrate how even partial knowledge of the relationships can guide various risk-assessment activities. But outside the complete risk-assessment framework, it should be stressed that even when only partial information is available, a biomarker can be useful from a public-health perspective for conducting some of the activities presented in Box 3-1. For example, observation in a prospective study that the concentration of a biomarker is increasing in the population might trigger complementary studies to understand the origin of exposure and its potential deleterious effects if these are unknown.
As indicated above, the body of knowledge about a given biomarker of exposure can be variable, and the interpretation of a biomarker concentration will depend on this body of knowledge. A framework (see Table 3-1) to characterize biomarkers was therefore developed to help the users of biomarkers to understand the advantages and the limits of interpretation of biomarker results. The framework links the potential uses of biomarkers to their properties. It is based on the simplified relationships between external dose, internal dose, and biologic effects (Figure 3-1). Table 3-1 consists of a matrix, the row headings are the properties of the biomarkers, and the column headings are the grouping categories (I through VII) to which specific biomarkers might belong, depending on their properties. Based on the properties of a biomarker for a specific grouping category, the biomarker provides information for the internal dose, external dose, or biological effects of a particular chemical, thus providing information on uses of the biomarker. The matrix indicates the minimal properties of a biomarker for inclusion in one of the groups indicated.
TABLE 3-1 Framework for Grouping Biomarkers of Exposurea
|
|
|
Biomarker Group |
||||||
|
Properties of Biomarkers |
I |
II |
III |
IV |
V |
VI |
VII |
|
|
Reproducible sampling and analytic method |
|
R |
R |
R |
R |
R |
R |
|
|
Known relationship of external dose to [BM] in animalsb |
|
|
R |
|
|
|
|
|
|
Known relationship of external dose to [BM] in humansb |
|
|
|
R |
|
R |
R |
|
|
Known relationship of [BM] to biologic effect in animals |
|
|
|
|
|
O |
|
|
|
Known relationship of [BM] to biologic effect in humans |
|
|
|
|
R |
|
R |
|
|
Known relationship of external dose to response in animals |
|
|
|
|
|
O |
|
|
|
Known relationship of external dose to response in humans |
|
|
|
|
|
O |
|
|
|
Biomarker informs about |
Internal dose |
|
Σ |
Σ |
Σ |
Σ |
Σ |
Σ |
|
External dosec |
|
|
Σ |
Σ |
|
Σ |
Σ |
|
|
Biologic effectsd |
|
|
|
|
Σ |
Σ |
Σ |
|
|
|
|
|
|
|
|
Potential for risk assessment |
||
|
aCheckmark in lower portion of table means that biomarkers in group can inform about stated elements of dose and effect. bImplies knowledge of pharmacokinetics of biomarker in relation to exposure to parent chemical. cThe relationship between external dose and internal dose may be influenced by metabolic polymorphisms and other factors, including socioeconomic status and racial and ethnic differences. dBiologic effects may include a wide range of observations, from very early biochemical perturbations to clinical signs of alteration of health. Abbreviations: [BM] = concentration of biomarker; R = required: O = optional (at least one of these is required). |
||||||||
Determining which properties are present in connection with a given biomarker should be based on a weight-of-evidence approach (Krimsky 2005) and will be peculiar to the context in which the biomarker is intended to be used. For example, there could be studies relating the concentration of a biomarker to toxic end points in workers exposed to relatively high doses of a toxicant. But the measured end point could be irrelevant for the general population exposed to doses that are much smaller (see “Adapting Occupational Reference Values to the General Population” in Chapter 5). In such a case, a toxicologist might consider that the biomarker does not possess the property “known relationship of [BM] to biologic effect relationship in humans” (where [BM] is the concentration of the biomarker) for the purpose of a study of the general population. In other words, professional judgment remains at the core of the application of the framework. Because each of the various risk-assessment and -management activities described in Box 3-1 may have its own specific purpose, a biomarker with a low group number may be just as valuable as one with a high number, given the objectives for which it is used.
The examples provided below of biomarkers in the various groups are presented for illustration purposes only and should not be considered as a final committee judgment on the group to which a given biomarker belongs. Readers interested in a more detailed discussion of some of these examples should consult Chapter 5.
Group I biomarkers correspond to substances that have been observed in bodily fluids but for which no relationships described in Figure 3-1 have yet been characterized. Even the analytic methods have not been shown to meet the basic criteria described in Chapter 4. Biomarkers in this category may be considered useless, but they might still find some utility in case of a sudden major accidental spill or intentional contamination with a given chemical. Indeed, suggestive evidence that a substance has the potential to cross the biologic barriers and find its way into the body may be important information. Any results with this type of biomarker must be viewed with caution because one may not be certain of chemical identity, quantitative accuracy, or biomarker specificity (for example, its presence in bodily fluids may be due to endogenous or other exogenous sources). Group I biomarkers can also include quantitatively important chromatographic peaks corresponding to uncharacterized substances and be found fortuitously during analyses of other well-characterized substances. The latter observations might trigger more research to identify the chemicals and understand their origin in the analyzed biologic sample. Various scoping activities can be pursued with such biomarkers. While this approach is not typically used in population biomonitoring studies, the finding of hundreds of unidentified peaks in human adipose tissue potentially of xenobiotic origin (Thornton et al. 2002), suggests that such an approach may be warranted. The field of
forensic toxicology provides examples where unknown peaks are investigated and shown to be related to human overdose or toxicity, with their identification leading to the development of important Group I (or higher) biomarkers (Broussard et al. 1997; Broussard et al. 2000).
Group II biomarkers also have not been linked to external exposure or to biologic effects, but sampling and analytic methods have been shown to yield reproducible and reliable results. Achieving this laboratory validation is a natural next step after the mere observation and identification of a chemical in human body fluids. It means that competent laboratories in different parts of a country or in different countries can obtain similar results. At the scoping stage of the risk-assessment and -management activities, for example, this implies that observations from different geographic locations—or samples collected at different times—can be reliably compared. From that perspective, status and trend activities can also be undertaken with such biomarkers. It should be noted, however, that the results still cannot be interpreted from a risk-assessment or clinical perspective. Moreover, because no link with external exposure to this substance or its parent compound has yet been established, this type of biomarker cannot guide pathway analyses or remedial actions. Polybrominated diphenyl ethers (PBDE) are illustrative of this group. Indeed, at the time of publication of this report, PBDEs could be measured reliably in human blood, but there is insufficient information to establish a relationship between the measurements and either external dose or toxicity. Developing such relationships is important, given the preliminary evidence of hormonal and developmental effects of PBDEs (Birnbaum and Staskal 2004).
With group III biomarkers, a reliable sampling and analytic technology is available, and a relationship between the external dose and the concentration of the biomarkers in laboratory animals has been demonstrated. The latter is important additional information in that it increases one’s confidence, when using information on such biomarkers, that the greater the concentration of the biomarker, the greater the dose received. However, because the information is available only from animal studies, an external dose to humans cannot be inferred from biomarker concentration information. Because no information is available on the human bioavailability of the parent chemical for various exposure routes, as with group II biomarkers, that limits the utility of group III biomarkers in guiding pathway analysis. Sometimes, good pharmacokinetic models are available to describe the fate of the substance in animals and some of the key parameters of the model are known for humans or can be inferred with reasonable confidence. Under such circumstances, the utility of group III biomarkers can expand to that described for the next group, group IV.
Group IV biomarkers also can be sampled and analyzed with reliable methods. In addition, the relationship between external dose and biomarker
concentration has been established in humans. That can stem, for example, from pharmacokinetic studies of human volunteers, from occupational studies of workers whose exposures have a much larger range than that encountered in the general population, from case studies of accidental poisoning, and from community studies in which multimedia exposure has been carefully analyzed. Ideally, the pharmacokinetic behavior of the parent chemical and of the biomarker is characterized on the basis of various exposure pathways, such as inhalation, ingestion, and dermal exposure. Such risk-assessment and -management activities as scoping, status and trends, and, to some degree, exposure and health research can be conducted using biomarkers in this group. Even without adequate knowledge about the toxicity of the substances involved, remedial actions can be taken to reduce exposure if the pathways that lead to the presence of the biomarker are well defined; however, this will typically not be available from human pharmacokinetic studies and would require robust exposure assessment as part of the establishment of a dose-biomarker relationship.
Because information is available about the toxicity of the parent chemicals of group V, VI, and VII biomarkers or of the biomarkers themselves, these biomarkers potentially have properties that allow their use for risk-assessment purposes.
For group V biomarkers, the relationship between concentration— internal dose—–and the toxicity of the parent chemical has been established and it is therefore usually possible to determine a biomarker concentration below which no toxicity is observed. Consequently, it is possible to predict the likelihood of toxicity associated with the parent chemical in a person or in a community. Of course, such factors as the specificity of the biomarker (whether it is peculiar to a given parent chemical or common to other substances) have to be taken into account when interpreting results. When test results obtained concomitantly indicate biologic anomalies or disease, group V biomarkers may help to establish whether there is a plausible causal relationship between the abnormal test results and exposure to the parent chemical. Most risk-assessment and -management activities along the continuum shown in Box 3-1 can be conducted, with augmentation if an exposure assessment showing major sources of human exposure to the chemical is available. That would enable apportionment of exposure and development of intervention measures if deemed necessary on the basis of the biomonitored concentrations found and the risks they imply. Group V is the first group in this framework that allows risk assessment to be performed on the basis of available data.
Group VI biomarkers have the same fundamental properties as those in group IV, so they also share basic uses. But group VI has another important attribute: toxicity information is available. It could be the dose-response relationship with the parent chemical in animals or in humans or the rela-
tionship between the biomarker concentration (the internal dose) and the toxicity that is known from animal studies. Such biomarkers can therefore inform the investigator about internal dose, external dose, and biologic effects (the three elements of the triangle presented in Figure 3-1). Uses of such biomarkers span all risk-assessment and -management activities. Many urinary phthalates probably belong to this group in that much is known about phthalate toxicology in animals and there is new information relating biomarker levels to possible effects in humans (Swan et al. 2005).
Group VII biomarkers can also be useful in risk assessment. Compared with group VI, the hallmark of group VII is that all operational relationships described in Figure 3-1 have been established in humans. The major improvement in group VII, compared with group V, is that one can now determine how changes in intake dose will affect biomarker concentrations; this can facilitate intervention decisions. However, as pointed out above, interventions are possible even without knowing the relationship between external dose and biomarker concentration as long as pathway analysis can document the major sources of chemical intake. There would probably be a consensus that blood lead belongs to group VII.
The committee believes that this framework will facilitate the dialogue between all interested and affected parties around biomonitoring, but competent professional judgment will remain at the core of the biomonitoring activities used in risk-assessment and -management. The depth of scientific knowledge can be very different between two biomarkers in the same group, but the framework assumes that enough is known to meet the qualifying properties shown in Table 3-1. And it is probably important to emphasize that with a given toxicologic database on any biomarker and its parent chemical, a weight-of-evidence approach will not necessarily lead all toxicologists to the same conclusion regarding the group to which a biomarker belongs. There would consequently not necessarily be a consensus on the interpretation of the biomonitoring results. However, it is hoped that the framework crystallizes the scientific debate over specific issues, enough to help in achieving such a consensus. Additional information, such as the half-life and other metabolic properties of a given biomarker, will determine the conditions under which the framework can be used for an intended purpose. The committee considers that biomarker selection should account for metabolism and be both biologically significant and relevant to the outcomes of concern. For example, a cross-sectional survey based on a single measurement of a rapidly metabolized chemical may provide limited or even misleading information concerning true population exposure levels. Although half-life, metabolic properties (which may be influenced by genetic polymorphisms or other lifestyle factors—see Chapter 4), and metabolites are key considerations, the committee intentionally did not factor metabolism into Table 3-1, as biomarkers with very different rates of metabolism may have similar applications.
The framework allows researchers and public-health authorities to select a biomarker on the basis of “standard” toxicologic properties depending on the objective of an investigation. It also helps to delineate the limits of some types of biomarkers and, we hope, to communicate information about biomarkers to the general public better. For example, a group II biomarker would be the quintessence of the CDC statement that the presence of a biomarker “does not by itself mean the chemical causes disease.” It is important to understand that a group VII biomarker is not necessarily better than a group IV biomarker, because the appropriate selection will rest on the intended purpose of its use along the continuum of risk-assessment and -management activities described in Box 3-1. Hence, there should be no a priori necessity to make all biomarkers group VII biomarkers.
It is outside the scope of this report to establish the group to which every existing biomarker of exposure belongs according to the framework presented. Furthermore, the group designation has to be contextualized to a study’s objectives, as stated previously. Some case studies will be presented in Chapter 5, however, to illustrate the potential usefulness of the framework.
CONCLUSIONS
-
A systematic framework to characterize the properties of biomarkers is a means of informing scientists and helps to inform the general population about biomarkers and their use in biomonitoring studies. It also allows an assessment of research gaps that need to be addressed to meet the requirements of some specific uses.
RECOMMENDATIONS
-
Investigators should use the proposed framework as a guide to characterize the biomarkers they intend to use in a biomonitoring study. The framework might serve as a guide in interpreting the results of the studies and in communicating the study objectives to various audiences.
-
The grouping framework should be used to specify the objectives of research projects in the development and validation of various biomarkers, that is, which properties of the biomarkers will be examined and how the studies may contribute in repositioning the biomarker in the framework.
-
Biomonitoring study design should consider absorption, distribution, metabolism, and elimination in selection of appropriate biomarkers to address the goals of research and surveillance.
REFERENCES
Bernard, A., and R. Lauwerys. 1986. Present status and trends in biological monitoring of exposure to industrial chemicals. J. Occup. Med. 28(8):558-562.
Birnbaum, L.S., and D.F. Staskal. 2004. Brominated flame retardants: Cause for concern? Environ Health Perspect. 112(1):9-17.
Broussard, L.A., T. Brustowicz, T. Pittman, K.D. Atkins, and L. Presley. 1997. Two traffic fatalities related to the use of difluoroethane. J. Forensic Sci. 42(6):1186-1187.
Broussard, L.A., A.K. Broussard, T.S. Pittman, and D.K. Lirette. 2000. Death due to inhalation of ethyl chloride. J Forensic Sci. 45(1):223-225.
Burke, T., H. Anderson, N. Beach, S. Colome, R.T. Drew, M. Firestone, F.S. Hauchman, T.O. Miller, D.K. Wagener, L. Zeise, and N. Tran. 1992. Role of exposure databases in risk management. Arch. Environ. Health 47(6):421-429.
CDC (Centers for Disease Control and Prevention). 2005. Third National Report on Human Exposure to Environmental Chemicals. U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, Atlanta, GA [online]. Available: http://www.cdc.gov/exposurereport/3rd/pdf/thirdreport.pdf [accessed Sept. 26, 2005].
Dor, F., W. Dab, P. Empereur-Bissonnet, and D. Zmirou. 1999. Validity of biomarkers in environmental health studies: The case of PAHs and benzene. Crit. Rev. Toxicol. 29(2): 129-168.
Jackson, R. 2005. Biomonitoring. Presentation at the Fourth Meeting on Human Biomonitoring of Environmental Toxicants, October 27, 2005, Irvine, CA.
Krimsky, S. 2005. The weight of scientific evidence in policy and law. Am. J. Public Health 95(Suppl. 1):S129-S136.
NRC (National Research Council). 1991. Monitoring Human Tissues for Toxic Substances. Washington, DC: National Academy Press.
Perbellini, L., N. Veronese, and A. Princivalle. 2002. Mercapturic acids in the biological monitoring of occupational exposure to chemicals. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 781(1-2):269-290.
Pirkle, J.L., L.L. Needham, and K. Sexton. 1995. Improving exposure assessment by monitoring human tissues for toxic chemicals. J. Expo. Anal. Environ. Epidemiol. 5(3):405-424.
Rubin, C., E. Esteban, R.H. Hill, Jr., and K. Pearce. 2002. Introduction—the methyl parathion story: A chronicle of misuse and preventable human exposure. Environ. Health Perspect. 110(Suppl. 6):1037-1040.
Schulte, P.A., and G. Talaska. 1995. Validity criteria for the use of biological markers of exposure to chemical agents in environmental epidemiology. Toxicology 101(1-2):73-88.
Sexton, K., L.L. Needham, and J.L. Pirkle. 2004. Human biomonitoring of environmental chemicals: Measuring chemicals in human tissue is the “gold standard” for assessing the people’s exposure to pollution. Am. Sci. 92(1):39-45.
Sharma, R.A., and P.B. Farmer. 2004. Biological relevance of adduct detection to the chemoprevention of cancer. Clin. Cancer Res. 10(15):4901-4912.
Sinks, T. 2005. Future Opportunities for Biomonitoring at CDC? Where we are and ? Where we go? Presentation at the Fourth Meeting on Human Biomonitoring of Environmental Toxicants, October 27, 2005, Irvine, CA.
Swan, S.H., K.M. Main, F. Liu, S.L. Stewart, R.L. Kruse, A.M. Calafat, C.S. Mao, J.B. Redmon, C.L. Ternand, S. Sullivan, and J.L. Teague. 2005. Decrease in anogenital distance among male infants with prenatal phthalate exposure. Environ. Health Perspect. 113(8):1056-1061.
Thornton, J.W., M. McCally, and J. Houlihan. 2002. Biomonitoring of industrial pollutants: Health and policy implications of the chemical body burden. Public Health Reports 117(4):315-323.
WHO (World Health Organization). 2001. Biomarkers in Risk Assessment: Validity and Validation. Environmental Health Criteria 222. Geneva: World Health Organization [online]. Available: http://www.inchem.org/documents/ehc/ehc/ehc222.htm [accessed Dec.1, 2005].













