2
AMO Science and the Basic Laws of Nature
Atomic, molecular, and optical (AMO) experiments have reached such high levels of sophistication, precision, and accuracy that they are uniquely positioned to carry out the most demanding tests ever conducted of some of the most fundamental laws of nature. For example, AMO scientists aim at measuring subtle new effects due to a possible permanent electric dipole moment of an electron or an atom, which, if such exists, would require a dramatic extension of our theory of elementary particles. But this measurement proceeds via ultraprecise measurements in atomic physics rather than by using high-energy particle accelerators. Atomic physics experiments can also search for violations of the so-called charge, parity, and time reversal (CRT) symmetry, which states that matter and antimatter particles should have exactly the same mass and the same magnetism. Both of these symmetries are fundamental pillars of modern physics, so testing them is of the greatest significance. In yet another development, theories unifying gravity with the other fundamental interactions of nature suggest the possibility of spatial and temporal variations of physical “constants” such as the fine structure constant α of electromagnetism. In addition to being used to measure α to unprecedented precision as a test of quantum electrodynamics, techniques from AMO science can be used to place limits on the variation of α over cosmological timescales. And laser-based gravitational-wave detectors will characterize gravitational waves and unravel information about their violent origins and about the nature of gravity. These and other applications of AMO science for exploring the basic laws of nature are the topics of this chapter, along with some of the practical applications that result from this quest.
SPIN SCIENCE
One of the most interesting phenomena in quantum mechanics is that nature’s subatomic particles possess an intrinsic property called “spin.” First observed by Stern and Gerlach in 1922 (see Box 2–1), this property has no exact analogy in the classical world. For while planets may spin on their axes, the property is not an in-
|
BOX 2–1 The Discovery of the Intrinsic Spin of the Electron Two pieces of experimental evidence discovered in the 1920s, (1) the observation of so-called “fine structure splitting” of hydrogen spectral lines and (2) the Stern-Gerlach experiment, demonstrated that subatomic particles possess a spin angular momentum. The Stern-Gerlach experiment showed that a beam of silver atoms directed through an inhomogeneous magnetic field (see Figure 2–1–1) is split into two beams. This demonstrated that electrons possess an intrinsic angular momentum S of value S=ħ [(s(s+1)]½, where s=½ and ħ is Planck’s constant. This intrinsic angular momentum is a purely quantum mechanical property of the electron, the “electron spin.” It has an associated magnetic moment µs=–(e/2m) gS, where g is the so-called electron spin g-factor. Neither the spin nor the magnetic moment has a classical analogy. Like electrons, nuclei and their constituent particles, neutrons and protons, also possess intrinsic spins. 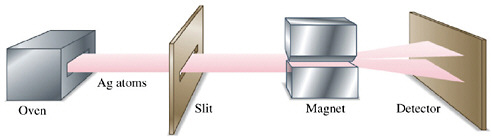
FIGURE 2–1–1 Silver atoms vaporized in an oven are shaped into a beam by the slit, and the beam is passed through a nonuniform magnetic field. The beam splits in two components that contain atoms with up and down spin. |
trinsic one—each planet in general does it in a different way. In the quantum world however, each elementary particle of a given type has exactly the same intrinsic spin angular momentum. This spin of the particle is also directly related to the intrinsic magnetism of the particle. In turn, that magnetism is one of the main ways in which the particle interacts with other particles and with its environment.
The magnetic forces on the spins of electrons and nuclei in atoms can be measured with remarkable precision because in many atoms the spins couple only weakly to other atoms in the environment. In fact, spin measurements are among the most precise in all of science. But sometimes it is the interactions with the environment itself that are interesting, as in magnetic resonance imaging (MRI), and atomic spin science is contributing to advances in that area. Other times, it is a possible new coupling of spins that is of interest. For example, it is predicted by the theory of supersymmetry that electrons and nuclei in atoms should possess a tiny offset between their center of electric charge and their center of mass, and that this offset should lie along the axis of spin. This shift is called a permanent electric
dipole moment (EDM). If it exists, it would be caused by a force that violates a cherished law in physics: time reversal (T) invariance (see Figure 2–1).
T-violating forces in the conventional Standard Model of elementary particle interactions generate an EDM far too small to be observed by any presently envisioned experiment, so the discovery of an EDM would reveal new forces and particles that lie outside the Standard Model. Such new forces and particles are absolutely needed, because the Standard Model has some glaring gaps. For example, it does not predict the overwhelming preponderance of matter over antimatter in the universe. But there is very little experimental evidence that would help establish any one of the alternative theories. According to some of the favorite contenders, such as supersymmetry, new forces should exist and should produce atomic EDMs at measurable levels. Figure 2–2 shows the ranges of some of the current predictions for EDMs arising from various dynamical models that extend the Standard Model of particle physics.
Since the energy of an atom with an EDM will depend upon the direction of the spin axis relative to an applied electric field, EDM experiments look for a tiny shift in the nuclear or electron spin resonance of an atom or molecule placed in a large electric field. Such spin resonance frequency measurements may provide our best opportunity to detect these tiniest effects of new fundamental forces that cannot yet be seen in experiments at high-energy accelerators. Atomic physics is taking up this challenge by creating ultrasharp spin resonances associated with long spin stability (hundreds to thousands of seconds in the case of nuclear spins in atomic gases or vapors). Experiments can now detect frequency shifts smaller than a nanohertz—less than one complete spin precession in 30 years. At such sensitivity,
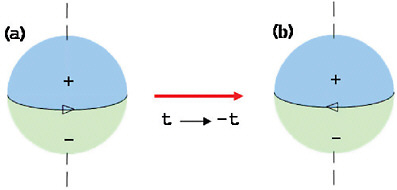
FIGURE 2–1 An electric dipole moment (EDM) of an atom (left) is a permanent separation between the centers of positive and negative charge along the axis of spin. Under time reversal (right), the spin direction is reversed but the charge separation is not. An observed EDM would have to be caused by forces that violate time reversal symmetry.
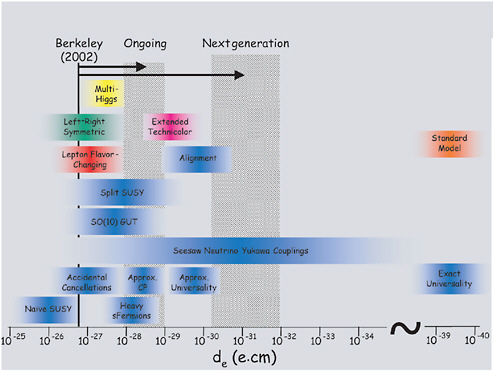
FIGURE 2–2 Scientific impact of current and next-generation electron EDM measurements. It is anticipated that next-generation measurement will reach the 10−31 e-cm level, equivalent to a dipole consisting of a positive and negative electronic charge separated by only 10−31 cm, which will test large classes of supersymmetric (SUSY) theories. Ongoing measurements of an atomic EDM due to nuclear spin and the EDM of the neutron have similar sensitivity to different SUSY parameters. Thus, atomic EDM experiments will be probing SUSY and other theories on a broad front. SOURCE: D.DeMille, Yale University.
subtle new effects due to EDMs could be seen if they are present (see Box 2–2). At the same time, such sensitivity to spins and to the effect of magnetic fields on spins is opening up new applications in medical research and diagnosis.
Magnetometry and Medical Imaging
Atomic spin magnetometers, developed in part for fundamental EDM and Lorentz experiments, are now surpassing low-temperature superconducting quantum interference devices (SQUIDs), formerly the industry standard for precise magnetometry. Atoms have higher sensitivity, higher spatial resolution, and no
|
BOX 2–2 Using Atoms to Probe the Particle Physics Frontier At first sight, it is remarkable that the study of atoms can tell us about forces that are normally explored at high-energy accelerators or large underground labs. Not only are atomic experiments carried out at ordinary energies, usually on a tabletop scale, but inside atoms the electrons are bound by well-understood electric and magnetic forces and move at very small energies compared with those reached at particle accelerators. Indeed, in much of atomic physics we can consider bound atomic electrons as point charges interacting with the electric and magnetic fields of the other charges in the atom. Close to an electron, however, there is a lot more happening. The electron envelops itself in a tiny cloud of emitted and reabsorbed virtual particles (Figure 2–2–1)—not only photons, but even the heavy particles studied at high-energy accelerators (similarly, the elementary constituents of the nucleus, the quarks, are surrounded by even more complicated clouds). The existence of these tiny clouds shows up in highly sensitive measurements of energy levels and other atomic properties, such as the exquisite measurements of the magnetism of the electron discussed in the next subsection, “Magnetometry and Medical Imaging.” Furthermore, the clouds will contain all virtual particles, even those too heavy to be produced by accelerators, so new physics beyond the current Standard Model might well show up first in atomic experiments. For example, it is widely believed that there are so-called supersymmetric particles that will eventually be found in high-energy experiments at new facilities such as the Large Hadron Collider (LHC). But for now the atomic EDM experiments are well positioned to discover supersymmetric particles if they do indeed exist. In fact, other atomic experiments have already shown how the small virtual clouds around each electron and quark endow atoms with remarkable properties that are of great interest to particle physics. One particle in such clouds, the 90 GeV Z0 boson, distinguishes left from right (that is, it violates parity) and causes an isolated atom to absorb right-handed, circularly polarized photons at a slightly different rate than left-handed photons. The effect is very small, but the measurements in the cesium atom are so precise and agree so well with the Standard Model as to show that a proposed heavier version of the Z0 must be at least eight times heavier than the known Z0, if it exists at all. Thus atomic physics, through parity violation measurements, has been able to give particle theorists information long before particle accelerators will have attained sufficient energy (700 GeV) to test for the existence of such a particle. |
need for cryogenic apparatus. Developments are under way that will allow measurements of the weak magnetic fields produced by the brain and the heart, which can provide valuable diagnostic and research information. These techniques are expected to help in diagnosis of epilepsy, cardiac arrhythmias, and other diseases. They also can be used in functional studies of the brain.
Advances in measuring nuclear spin resonances in the case of noble gases not only are helping in the searches for an EDM and Lorentz violation but are also opening up a new field in medical imaging. Recently, high-quality magnetic resonance images based on the gases 129Xe and 3He have begun to appear. For example,
as illustrated in Figure 2–3, the image of a human lung momentarily filled with 3He gas provides a high-resolution picture of the lung. Ordinary MRI techniques are not capable of producing such pictures. The clinical result is that lung disease can be diagnosed much earlier with these new instruments.
The secret to obtaining such images is to increase the fraction of nuclear spins that are aligned with each other, so that their magnetism adds up to a larger value. In ordinary MRI, this fraction is only a few parts per million. That is due to the trade-off between the aligning force exerted on the magnetic moments of the atoms by a strong magnetic field and the randomizing force the molecules exert on
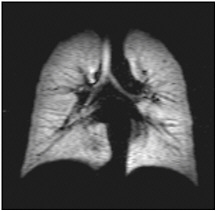
FIGURE 2–3 A coronal image of the lungs using magnetic resonance, when the lungs are momentarily filled with hyperpolarized atomic 3He gas. SOURCE: Center for In-vivo Hyperpolarized Gas MRI, Radiology Department, University of Virginia.
each other. But polarized laser light can pump angular momentum into the atoms very efficiently through a process of repeated optical absorption and fluorescence, called “optical pumping.” In this way, the alignment can be increased to several tens of percent. For such optically pumped, hyperpolarized atomic gases, the signal per nucleus is about 100,000 times larger than the signal per nucleus in normal MRI. This more than makes up for the smaller density of the nuclei in these gases compared with the density in human tissue.
Spin and Basic Forces
One effect of the cloud of virtual particles around the electron illustrated in Figure 2–2–1 is to modify the magnetism associated with the electron spin by about 0.1 percent. Using a single electron held in a magnetic trap, this tiny modification has been measured to astonishing accuracy: 10 decimal places. Equally astonishing, the theory of these radiative corrections has been used to calculate the effect to the same precision. The calculated result is expressed in terms of the fine structure constant α, which gives the strength of the electric coupling. It remains now for other atomic experiments, discussed in the subsection “Fine Structure Constant,” to measure a to equal precision; then it will be known whether or not the theory of quantum electrodynamics agrees with experiment to such unprecedented accuracy. In a more speculative vein, atomic experiments are so sensitive to the very fabric of space and time that they can measure its underlying symmetries as embodied in the principle of Lorentz invariance. This is the principle of relativity put forward by
Einstein in 1905. Atomic experiments can also search for violations of the so-called CPT symmetry, which states that matter and antimatter particles should have exactly the same mass and the same magnetism. Both of these symmetries are fundamental pillars of modern physics. A recently developed theoretical framework now exists for linking a possible Lorentz violation with CPT violation. Precise Lorentz tests include spin resonance experiments in which the spin precession frequency would depend on the orientation of the precession axis relative to a fixed direction in space, the vector bi in Figure 2–4. One of the most exact CPT tests is expected to result from a remarkable recent feat, the creation of antihydrogen in the laboratory. By comparing the spectral energies of antihydrogen atoms and ordinary hydrogen atoms, one can search for effects due to CPT violation.
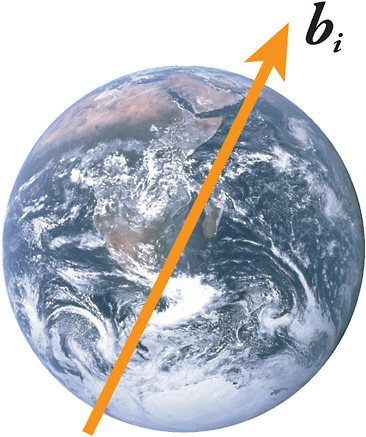
FIGURE 2–4 If there is a preferred direction bi in space in the vicinity of Earth, sensitive spin experiments will show a diurnal effect on Earth as it rotates. SOURCE: Image of Earth, National Aeronautics and Space Administration.
The great challenge in most EDM experiments and Lorentz symmetry tests is to detect the tiniest possible shifts in spin resonance frequency. The EDM sensitivity is mind-boggling. In the case of the mercury atom, if the atom were blown up to the size of Earth, the separation between centers of plus and minus charge would be less than one-hundredth of an angstrom!
ENERGY LEVELS, TIME, AND ATOMIC CLOCKS
From the discovery of the laws of planetary motion in the Renaissance to the development of quantum theory in the 20th century, precision timekeeping has historically accompanied advances in science. In particular, advances in AMO science have led to ever more accurate atomic clocks. Likewise, improvements in clocks have enabled revolutions in technology, starting with the determination of longitude in the 17th century and leading to the development of the modern global positioning satellite (see Figure 2–5 for one such system).
The measurement of frequency (or, equivalently, time duration) can be done with greater precision than any other measurement in physics. This precision stems from the ability to compare signals very accurately to natural periodic phenomena, such as planetary motion, the swinging of a pendulum, or the vibrations in a crystal of quartz. The pinnacle of frequency standards is the atomic clock, which relies for its precision on the generation of a highly accurate oscillating signal based on the vibrations in an isolated atom. This oscillation comes from the difference in energy ΔE between two states in the atom and the relationship of the energy difference to frequency, v, through Planck’s law, hv=ΔE, where h is Planck’s constant of quantum mechanics. The frequency of an atomic clock thus depends only on the properties of an isolated atom, regardless of whether that atom is located in Washington, D.C., or Beijing. The extraordinary advances in atomic clocks over the past 15 years have resulted mainly from isolating and cooling ions or neutral atoms, thereby eliminating Doppler shifts and other perturbations (see Box 2–3). The state-of-the-art accuracy of atomic clocks, which is approaching one second in 60 million years, involves the use of a small number of neutral atoms or a single ion in electric traps. Such accuracies are not merely academic—as indicated above, they are necessary for increasing the precision of navigation, from applications in space exploration to advanced architectures in GPS.
To attain such accuracy, the atoms in atomic clocks must be isolated from their environment, including from other nearby atoms. For this reason, accurate atomic clocks usually deal with very small numbers of atoms, in some cases just a single atom. However, extracting any useful information from such a small number of atoms requires collecting and averaging the weak signals they emit over very long periods of time. Even the most efficient detector will see at most one photon every

FIGURE 2–5 Schematic drawing of the Global Positioning System (GPS) constellation. The GPS uses 24 satellites (21 plus spares) at an altitude of about 11,000 miles, moving at about 7,000 mph. A GPS receiver on Earth synchronizes itself with the satellite code and measures the elapsed time since transmission by comparing the difference between the satellite code and the receiver code. The greater the difference, the greater the time since transmission. Knowing the time and the speed of light, the distance can be calculated. The time comes from four atomic clocks on each satellite. The clocks are accurate to within 0.003 seconds per 1,000 years. Using information from four or more satellites, the GPS receiver calculates latitude, longitude, and altitude. SOURCE: National Aeronautics and Space Administration.
time an atom decays, since only one photon is given off in the decay process. To find the decay rate, we have to count for a long time. This rate measurement is precisely the same kind of problem as measuring the odds for heads or tails in a coin toss. You have to play a long time to get beyond the small statistics of winning or losing streaks, to find the true average rate for tails.
Specifically, for the coin toss or any other random classical counting process, N trials are needed to produce a precision of 1/N½. This is known as the shot-noise limit. But shot noise is one of those seemingly immutable facts of nature that need to be rethought in the quantum realm of atoms. Indeed, one of the applications of
|
BOX 2–3 History of Laser-Cooled Atoms and Ions Over 30 years ago, in 1975, two pairs of West Coast scientists proposed the remarkable idea that a gas of atoms could be cooled by illuminating it with laser light. Only a few other scientists took much immediate note of this new idea, but by the late 1970s it had been successfully applied to cooling electrically charged and trapped ions to very cold temperatures, a thousandth of a degree above absolute zero. By the early 1980s other groups were trying to laser cool neutral atoms. Crucial support for this early work came from the Office of Naval Research (ONR), motivated by the hope that laser cooling would lead to better clocks, an essential element of modern precision navigation. Since that time, laser cooling has led to the realization of Bose-Einstein condensation in dilute gases and the achievement of the coldest temperatures known in the universe—less than a billionth of a degree above absolute zero. Eight atomic physicists have received Nobel prizes for work related to laser cooling and trapping, and hundreds of research groups are devoted to its study. Today, cold atoms represent a large fraction of all the research in AMO science. The frequency standards of the world’s major standards laboratories are laser-cooled atomic clocks (see Figure 2–3–1). International time ticks at a rate controlled by laser-cooled atoms. And the U.S. Naval Observatory, reaping the rewards of the ONR’s investment, is installing an ensemble of laser-cooled clocks to keep time for the nation’s military. Cold atom physics continues to bear fruit: Major research programs on precision inertial navigation systems are under way in the United States and abroad; quantum degenerate Bose and Fermi atomic gases are modeling condensed matter systems of interest for both basic science and practical devices (Chapter 3); and ions and atoms are being developed as the qubits for the new science of quantum information (see Chapter 7). 
FIGURE 2–3–1 Layout of the NIST-Boulder atomic fountain laser-cooled clock NIST F-1. This clock is now the most accurate clock in the world, ticking off time with an uncertainty of a few parts in 1016, about 1 second in 60 million years. SOURCE: J.Aarons, National Institute of Standards and Technology. |
the special feature of quantum mechanics known as “entanglement” or “quantum correlation” is to increase the amount of information that can be extracted from a single measurement of many atoms, effectively beating the shot-noise limit. By entangling N atoms in an atomic clock, it is possible for the inaccuracy in the averaged measurement to be improved to 1/N instead of 1/N½. This level of precision, the best allowed by quantum mechanics, is called the Heisenberg limit. For even a small number of atoms (say, N=1,000), this improvement is substantial. The recipe for operating an atomic clock in the Heisenberg limit is to create a collective quantum state of all of the atoms, sometimes called a “superatom,” that effectively speeds up the atomic clock by a factor of N.
New Clock Technologies and GPS
Precision oscillators and clocks have far-reaching, economically significant applications, including network synchronization, high-performance analog-to-digital conversion, and global positioning. It has been recognized for decades that specialized laser light sources have attributes that enable breakthrough performance for timing applications. These attributes include exceptional frequency stability and accuracy. However, these attributes could not be exploited for applications since it was not known how to transfer this stability and accuracy from the optical frequency range (1015 Hz) to frequencies accessible with traditional electronics (<1010 Hz). This long-standing problem, known as optical frequency counting, found a beautiful and technologically compelling solution by exploiting features associated with the output of femtosecond pulsed laser sources. The physicists who pioneered this breakthrough were awarded the 2005 Nobel prize in physics. As a result of efforts in many laboratories over the past 5 years, it is now possible to translate frequency stability and accuracy from the optical regime to the microwave regime and also to measure the ratio of optical frequencies with unprecedented precision. Very recent work indicates frequency counting methods may be extensible into the x-ray regime. This has direct, near-term technological impact. For example, the performance of high-end analog-to-digital converters—the key component in advanced radar systems—is currently limited by the frequency stability of cur rent-generation clocks. Next-generation optical oscillators enable clock jitter at the 1 femtosecond level, which is expected to improve the system performance in terms of both bandwidth and signal-to-noise ratio.
The atomic clock plays a central role in GPS architecture. Future advances in GPS performance will be enabled by high-stability clocks in the receiver and also in the satellite. Very recently, microfabrication techniques have been employed to create miniature (so-called “chip scale”) clocks with breakthrough performance characteristics for the receiver clock (Figure 2–6). Similar miniaturization efforts are

FIGURE 2–6 Left: Miniature atomic clock occupying less than 10 mm3. Right: The U.S. primary frequency standard, based on laser-cooled cesium atoms. SOURCE: National Institute of Standards and Technology.
being applied to produce compact magnetometers and gyroscopes. It is expected that these miniature atomic clocks will be widely deployed. Accurate atomic clocks also play important roles in submarine navigation systems.
Are the Constants of Nature Changing?
Our theoretical understanding of the universe and the known forces of nature incorporates a number of fundamental constants. Most famously, the speed of light, c, in a vacuum is constant to all observers. Other no less important physical constants are the fundamental quantum of angular momentum, known as Planck’s constant, h, and the charge e of the electron. From these three constants for charge, angular momentum, and speed we can form a dimensionless ratio (specifically, e2/ħc),1 known as the fine structure constant α. Although gravity was long thought to be truly constant over the lifetime of the universe, recent theories unifying it with the other physical interactions suggest the possibility of spatial and temporal variations of α and other physical “constants.”
Atomic physics comes into this picture because different atomic transitions depend differently on α so that comparing the rates of different atomic clocks over long periods of time allows one to put bounds on the local change of α with
time. Already there is a strong laboratory limit on the time variation of α: less than about one part in 1015. Recently, however, the accuracy achieved by atomic clocks has improved enough to test faint hints from cosmological and early Earth studies that α may have changed very slowly over the lifetime of the universe. Spectroscopic data provide some limited evidence that the value of α may indeed have evolved as the universe expanded. If this is confirmed, our understanding of quantum electrodynamics and its relation to the structure of spacetime would need to undergo major revision.
MEASURING DISTANCE AND MOTION USING INTERFEROMETERS
Interference is the term used in optical science for what happens when two (or more) optical beams that have traveled along different paths from the same source are brought back together. When the wave crests of two beams match up with each other, a bright spot is produced; when the wave crests of one beam match up with the wave troughs of the other, they cancel each other out and produce a dark spot. Because the wavelength of light is so short, about a micron or less, an interference meter (interferometer) is very sensitive to tiny relative changes in path length (or velocity) because of the Doppler effect. As the word “optical” in its name implies, AMO science continues to be at the forefront of the development of optical interferometers. The most celebrated practical application has been the ring laser gyroscope for navigation, which is the present industry standard. Exciting examples of the use of optical interferometry for fundamental physics and astronomy are at the ground-based Laser Interferometer Gravity-Wave Observatory (LIGO) and the proposed space-based Laser Interferometer Space Antenna (LISA), which are designed to detect directly gravitational waves coming from distant points in the universe (see subsection after next).
At the same time, AMO research is leading to new and even more sensitive interferometers based on the much shorter wavelengths associated with moving particles. This very active area is called de Broglie (or matter-wave) interferometry. New instruments based on this technique are expected to revolutionize the fields of inertial navigation and gravitational anomaly detection. There is also a remarkable application of matter-wave interferometry to measuring the fine structure constant α, which could help test the fundamental quantum theory of electricity and magnetism to unprecedented accuracy.
Optical Sensors for Navigation
Workhorse inertial sensors used widely in commercial and military navigation rely on ring-shaped lasers called ring laser gyroscopes (RLGs), which detect rota-
tion by comparing two light beams that travel around the same ring in opposite directions. The difference in light travel time in the two directions is proportional to the product of the rotation rate of the interferometer platform and the area enclosed by the interferometer.
A similar principle underlies the operation of fiber-optic gyroscopes (FOGs), in which the ring is replaced by loops of fiber. Advances in optical fiber and integrated optics technologies have resulted in significant improvements in the performance and cost of FOGs, to the extent that next-generation navigation systems will probably employ FOG technology rather than RLG. Developments in photonic band gap fiber technology may lead to another performance leap for these strategically vital systems. Photonic band gap fibers contain microstructured central cores that permit the light to travel mostly in air rather than glass.
Precision inertial navigation requires a detailed knowledge of Earth’s gravitational field (the acceleration due to gravity needs to be subtracted from the accelerometer outputs). Historically, this has resulted in close links between the navigation and geodesy communities. Advances in inertial technology have direct consequences for geodesy. For example, both GPS sensors and a large-area RLG detected minute changes in Earth’s rotation rate attributable to the tsunami in the Indian Ocean at the end of 2004. Laser-based gravimeters are being used worldwide to characterize Earth’s gravitational field. Recently they have been used for oil well logging and resource management.
Direct Detection of Gravitational Waves
Large-scale optical interferometric sensors form the cornerstone measurement technology for LIGO, the nation’s first great gravitational-wave observatory (see Figure 2–7), and for LISA, a future orbiting gravity-wave observatory. Gravitational waves are predicted to be tiny ripples in the otherwise smooth fabric of spacetime produced by violent events in the distant universe—for example, by the merging of two neutron stars or two black holes, or in the cores of supernova explosions. They have never been observed directly, but their influence on the orbital motion of the corotating binary pulsar PSR1913+16 has been confirmed by direct measurement. This observation was celebrated by the award of the Nobel prize in 1993.
Gravitational waves are emitted by accelerating masses much as electromagnetic waves are produced by accelerating charges. They travel to Earth at the speed of light, bringing with them information about their violent origins and about the nature of gravity. To detect gravity waves, which are very weak, requires a huge, highly precise optical interferometer. The LIGO design features two main interferometers, one in Hanford, Washington, and one in Livingston Parish, Louisiana. The interferometers are in the shape of an L with 4-km arms, each of which contains

FIGURE 2–7 Aerial view of one arm of the LIGO gravitational-wave detector in Livingston Parish, Louisiana. Two other large interferometers make up the second LIGO site at Hanford, Washington. SOURCE: LIGO Laboratory.
ultrastable laser beams that bounce back and forth millions of times between two freely hanging test masses fitted with mirrors, one at each free end of an arm. These assemblies are housed within high-vacuum tubes to eliminate light scattering from air. A third freely hanging mirrored test mass hangs at the vertex of the L. When a gravitational wave passes by, the distance between the test masses will change by a different amount in each arm (only about a hundred-millionth of the diameter of a hydrogen atom over the 4-km length of the arm). Such a tiny change can be detected by the change in relative phase between the laser beams in the two arms. A key challenge is to isolate the test masses from other disturbances such as seismic vibrations and air molecules. An upgrade to LIGO is expected to improve the instrument’s sensitivity by an order of magnitude.
Advanced measurement and instrumentation techniques developed for LIGO experiments—such as “squeezed light” sources/detectors and thermal noise miti-
gation strategies—may have practical impact in next-generation commercial and strategic sensors.
Matter-Wave Interferometry (de Broglie Wave Interference)
A completely different approach to improved inertial navigation might use matter waves instead of light waves. While the existing generation of navigation sensors perform at superb levels, advances in navigation technology are considered essential to support plans for broadly deployed autonomous defense and communications systems. Matter waves, known as de Broglie waves after the physicist who first proposed the wave-matter connection in quantum theory, might greatly improve inertial system accuracy, performance, and price. These sensors operate on wave-interference principles analogous to those underpinning the operation of the optical sensors described above. Were de Broglie wave sensors to reach the threshold of commercial viability, this could be considered one of the first great applications for ultracold atom physics, discussed in detail in Chapter 3.
Future gravimetry based on de Broglie wave interference will make possible long-distance airborne characterization of gravitational anomalies at unprecedented levels, for accurate detection of underground structures and tunnels as well as buried minerals and other natural resources. Widespread adoption of this technology requires significant progress in the development of supporting technologies, including robust, compact, and cost-effective laser and vacuum systems. Existing prototype systems require extensive vibration isolation in order to maintain the required benign environment for sensor operation. Development of interferometer configurations that mitigate sensitivity to platform motion while sustaining or improving sensor performance remains a significant scientific challenge.
The current generation of atomic sensors is based on laser-cooled ensembles of cold atoms in free space and pulses of laser light. The development of Bose-Einstein condensed/degenerate Fermi gas atomic sources and of de Broglie waveguide methods raises the interesting question whether these methods can be exploited to improve sensor performance. The central scientific challenge associated with this question is to understand how the inertial/gravimetric-phase information associated with the propagation of de Broglie waves is affected by interactions arising from, for example, atom-atom collisions, waveguide imperfections, atom-surface coupling, spurious magnetic and optical fields, and vibrations. Surprisingly, fermionic atoms now appear to be a viable alternative to bosons for such applications (see Chapter 3 for a discussion on the differences between cold bosons and fermions). An advantage of quantum-degenerate fermions for atom interferometry is that they do not suffer the kind of collisions that bosons are subject to, which can rapidly lead to decoherence and the destruction or random shift of an interference
pattern. This is a direct consequence of the Pauli exclusion principle, which forbids two fermions to occupy the same quantum state. This principle also renders fermionic interferometry somewhat analogous to white-light interferometry in conventional optics.
Fine Structure Constant
Matter-wave interference experiments can be used to make precision measurements of the value of the fine structure constant, α. The measurement of this constant is not just a curiosity but is an important component of critical tests of the quantum theory of electromagnetism, quantum electrodynamics (QED). To obtain α, a number of experiments that evolved from developments in different communities—laser cooling, de Broglie wave interferometry, and precise optical and mass spectroscopy—must be combined. The current goal is to measure α to a part in a billion accuracy. This value can then be used to calculate the spin magnetism of the electron, which—as discussed in the subsection “Optical Sensors for Navigation”—has already been measured to such accuracy. The result will be a test of our understanding of QED to a part per billion.
AMO PHYSICS IN THE STUDY OF THE DISTANT UNIVERSE
Early in the last century astronomers discovered that the light from distant stars and galaxies exhibits characteristic spectral features that can be accurately predicted using the atomic physics theory gleaned from experiments on Earth. That discovery provides direct evidence that the laws of physics apply across the distant reaches of the universe and from the earliest moments in time.
Atomic and molecular spectroscopy remains critical to our understanding of the cosmos, including some of the most exotic realms, from the dense plasmas on the surfaces of neutron stars to the cold dusty interiors of giant molecular clouds. Recent advances in space instrumentation have opened all ranges of the electromagnetic spectrum to astrophysical spectroscopy, which in turn has dramatically increased the need for precision atomic and molecular data. To address this need, AMO physicists carry out work in laboratory spectroscopy and study collision processes involving atoms, ions, molecules, and electrons, which often give rise to spectral lines. Theoretical calculations, benchmarked by laboratory experiments, provide essential input to the models used in the interpretation of astronomical observations.
The combination of astrophysical spectroscopy and precision atomic physics is proving to be an important testing ground for fundamental physics. As mentioned above, the spectra of distant quasars can be used to study the potential variation
of fundamental constants of nature. Precision spectroscopy also played a role in the recent discovery that the expansion of the universe is accelerating rather than decelerating, as had been expected. To explain this effect, physicists have posited the existence of a “dark energy”—the nature of which is not at all understood. The very existence of dark energy poses a challenge to the Standard Model of particle physics.
Gravity, the least understood of the four fundamental forces in nature, can be investigated with astrophysical observations, specifically in its strong-field regime, where it dramatically affects the nature of space and time. For instance, looking at the exotic environment of a massive black hole or a neutron star and using the spectra of highly ionized atoms of iron and other abundant elements as precision clocks can test the predictions of Einstein’s theory of general relativity in quantitative detail (see Figure 2–8).
However, the particular astrophysical conditions encountered in the vicinity of a black hole or neutron star can dramatically affect the spectra of atoms there. These environments are characterized by intense radiation fields, in which the energy density in the form of radiation can vastly exceed the thermal energy density of the gas. Atomic excitations are dominated by photoexcitation of the atoms and recombination cascades following photoionization. On the surfaces of neutron stars, gas

FIGURE 2–8 A simulation of the intensity pattern expected for the light emitted by a disk of material orbiting a black hole. The emission is asymmetric because the light emitted by atoms moving toward the observer is boosted in intensity, while the light from atoms moving away is diminished. The frequencies of spectral lines are similarly distorted by the intense gravitational field of the black hole. The figure also shows a strong distortion in the apparent shape of the disk—that is, the back of the disk looks like it is lifted up—due to the strong bending of light close to the black hole. SOURCE: C.Reynolds, University of Maryland.
is subjected to intense magnetic and electric fields that radically alter the atomic structure and, therefore, the spectra. Understanding such processes is a challenge for modern atomic physics. The challenges are being addressed with laboratory astrophysics experiments that use high-intensity lasers and/or particle accelerators to simulate such high-energy-density conditions. While the understanding of atomic physics under these conditions is still in its infancy, laser experiments can produce intense field regimes for short durations, as described in Chapter 4, which may be useful in this context.
The study of the early universe can yield clues to the fundamental laws of physics. Looking back in time, in the very earliest moments after the big bang the universe was compact and extremely hot. Similar conditions can be explored at accelerator facilities here on Earth using ultrarelativistic heavy ion collisions. A little later in the evolution of the cosmos, in the somewhat cooler instant of primordial nucleosynthesis, the positively charged nuclei of several chemical elements were produced—including those of hydrogen and helium together with small amounts of deuterium (HD) and lithium. The gas in the cosmos at these high temperatures was fully ionized and opaque so that it did not produce the spectral features that might be observed today.
As the universe expanded further and cooled, electrons and nuclei collided and recombined, producing neutral elements. As the universe cooled even further, molecular hydrogen and its isotope HD formed. Molecules can efficiently lose energy and allow a gas to cool under gravitational collapse, the first step in star formation. The study of molecular hydrogen formation in the early universe is an active area of study in AMO physics.
Beyond these earliest moments in the history of the cosmos, the universe only grew in complexity. Driven by the discovery of extrasolar planets and the exploration of the various moons circling planets in our solar system, astrophysicists and atomic and molecular spectroscopists are working to understand better the growth of complexity in the universe. The challenge to elucidate the evolutionary path from simple organic molecules—discovered in the interstellar medium and circumstellar shells—to simple proteins and ultimately to chemistries that give rise to life forms is exciting and compelling science. Spectroscopy is the tool with which the search for life in other planetary systems can be conducted, and AMO physics provides the backbone that supports this work.
There also remains a great deal to learn about the cosmic environment closer to home. For example, new tools enabled by AMO physics are improving our understanding of the comets in our solar system (see Box 2–4). Studying comets is expected to supply critical new information on the formation and transport of organic molecules in the solar system, as demonstrated by NASA’s celebrated comet sample return mission, Stardust.
|
BOX 2–4 X Rays from Comets In 1995, x-ray emission from a comet was first observed. As comets were thought to be visible because they reflect solar radiation from the cometary atmosphere, the source of the x rays was at first a mystery. Through the efforts of both atomic and molecular physicists and astronomers, the mechanism for x-ray production in comets has been identified as a “charge transfer” between highly ionized atoms in the solar wind1 and the neutral molecules in the comet’s atmosphere. Charge transfer occurs when an ion, such as seven-times-ionized oxygen, collides with a water molecule in the cometary atmosphere and captures an electron from the water, creating a highly excited state of six-times-ionized oxygen. This excited state decays by radiating light in the x-ray region of the spectrum (see Figure 2–4–1). Once this mechanism was postulated, particular x-ray spectral features could be associated with electron capture by a variety of different atomic ions in a range of ionization states. With increasing spectral resolution of x-ray telescopes in the future, coupled with further detailed calculations and measurements of charge transfer by atomic physicists, cometary x rays can be used as detailed diagnostics of the solar wind composition. |
AMO THEORY AND COMPUTATION CONNECTIONS TO ASTROPHYSICS AND ELEMENTARY PARTICLE PHYSICS
Hand in hand with the advances in experiments, there is a continuing advance in atomic theory with applications to astrophysics and elementary particle physics. Although the Hamiltonian describing the detailed interactions of electrons in atoms and molecules is known, the calculation of the structure and spectroscopy of many-electron systems to high accuracy continues to be a challenging problem. Atomic and molecular theorists have developed powerful numerical methods and extensive computer codes to calculate energy levels, wavefunctions, and spectral line strengths. For instance, the precise cesium parity violation experiment described above would not have been so valuable without equally precise calculations of the cesium wavefunctions needed to evaluate the signal expected from particle theory. Likewise, the experiments searching for an atomic or molecular EDM rely on the atomic codes to predict the expected size of T-violating effects in atoms and molecules. Similar codes are now being used to select atomic transitions to use in the search for a time change in α. In astrophysics many spectral lines are observed from species that are difficult to produce in the laboratory. Atomic and molecular theory provides critically important input to the models used in the interpretation of these astronomical observations.