9
Special Populations and Susceptibility
The goal of the U.S. Environmental Protection Agency’s (EPA) public health risk assessments is to protect all potentially affected populations, including subpopulations on the basis of gender, nutritional status, genetic predisposition, and life stages (e.g., childhood, pregnancy, old age) that might be more susceptible to toxic effects or that are highly or disproportionately exposed (e.g., children, ethnic groups) (EPA 2004). Children have been identified as a special population to consider in risk assessment because their health risks can differ from those of adults as a result of their immature physiology, metabolism, and different levels of exposure (EPA 1996, 2005b). Certain childhood cancers have been associated with exposures to solvents, including trichloroethylene, which is briefly discussed in this chapter.
Data on differential impacts to sensitive populations are frequently limited or absent, and the process for consideration of sensitive groups is poorly defined. As EPA stated, there is “not a single or exact method for examining potential susceptibility and associated risk” (EPA 2004, p. 43). In its draft risk assessment on trichloroethylene, EPA (2001b) relied on the evaluation by Pastino et al. (2000), who correctly stated that measures of susceptibility have not been incorporated into human risk assessment methods. Several papers have since been published that are relevant to this issue, particularly to pediatric risk and genetic susceptibility. These papers as well as relevant older studies and information on gender- and disease-based susceptibility are reviewed below.
CHILDHOOD CANCER
A sizable number of published scientific studies are relevant to the issue of parental occupational exposure to trichloroethylene and childhood cancer. The studies generally involve parental occupational exposures based on case-control studies. The committee did not have the time or resources to analyze all the studies, so it relied on a review paper (Colt and Blair 1998) and some new studies to illustrate the issues important in estimating the public health risk of parental exposure to occupational trichloroethylene and risk conveyed to their children. Chapter 2 provides guidelines for conducting a more rigorous review of the epidemiologic data.
The effects of parental occupational exposure on the risk of childhood cancer have been studied epidemiologically for more than 25 years. During that time, in most countries the nature of industry has changed in two important ways: through materials usage and levels of exposure. Specifically, trichloroethylene has been largely phased out as an industrial solvent. It is important to keep this in mind when reviewing the studies and their chronology. Colt and Blair (1998) reviewed information on parental exposure to solvents and the risk of childhood cancer. They summarized results from 48 published papers, virtually all of which were case-control studies. A few later papers have not clarified whether a relation exists (Shu et al. 1999; Schuz et al. 2000; McKinney et al. 2003; Infante-Rivard et al. 2005).
All studies used the case-control approach and therefore relied on questionnaire information, raising the usual methodological issues of the reliability of identifying specific exposures, selection of controls, and recall bias. Most studies relied on information about occupation and industry to infer exposures rather than questioning subjects about exposure to specific chemicals. Ages of the children studied varied from study to study. Often, only the mother was interviewed and asked about both her and her husband’s occupational history. It is very unlikely that women know about specific occupational exposures of their husbands, which is illustrated in two studies by Peters et al. (1981, 1985). In the first study, excess childhood brain cancer was associated with maternal and paternal exposure to chemicals, specifically to paint, and to work in the aerospace industry. An open-ended question addressed to the mother about specific chemicals to which either parent was exposed revealed little for the mothers and two mentions of trichloroethylene for the fathers. A follow-up study in which the fathers were interviewed revealed that five of them had exposure to trichloroethylene, whereas no control fathers did. Much like the community studies, the same occupations had other exposures to materials such as methylethylketone and other unspecified solvents. Following the approach of asking both parents about exposure to specific chemicals, Lowengart et al. (1987), in studying childhood leukemia, showed a risk associated with paternal exposure to
chlorinated hydrocarbons (most of which was trichloroethylene). Greater use was associated with higher risk. The previously cited study (Shu et al. 1999), using similar techniques in a larger study population, failed to show a risk associated with paternal exposures to trichloroethylene but the study period was considerably later when trichloroethylene was less likely to be used and when exposures were likely lower.
Theoretically, there are several ways that risk can be conveyed from parent to child. When the mother is exposed, her child might be exposed in utero, through breast milk, from contaminated clothing of the mother, or from germinal effects. For paternal exposure, bringing contaminated clothing home is possible or direct germinal effects to males transmitted during reproduction are possible. The latter is the most likely for trichloroethylene, given supportive animal toxicologic data (see Chapter 5).
Early studies on paternal occupational exposures suggest that exposure to trichloroethylene conveys a risk of leukemia and brain cancer developing in the offspring. Later studies do not show that risk. Over the time period of these studies, trichloroethylene was phased out as an industrial solvent and exposures in work settings have generally been reduced. Thus, the occupational studies provide data that suggest a relationship between parental exposure to trichloroethylene and risk of childhood cancers. The difficulties of studying rare diseases and the inability to measure exposure objectively limit any certainty about causality.
DEVELOPMENTAL ISSUES
Fetal and Pediatric Risk Assessment Concepts
Chemical-specific toxicokinetic and mode-of-action information are essential for fetal and pediatric risk assessment. As stated by a previous committee of the National Research Council, complete data would include the following (NRC 2000, p. 3):
-
the chemical’s toxicokinetics (i.e., its absorption, distribution, metabolism, and excretion) within the mother, fetus, and embryo;
-
the chemical’s toxicodynamics (i.e., how the chemical or a metabolite derived from it interacts with specific molecular components of developmental processes in the embryo and fetus or with maternal or extraembryonic components of processes supporting development);
-
the consequences of those interactions on cellular or developmental processes (also part of toxicodynamics); and
-
the consequence of the altered process for a developmental outcome, namely, the generation of a defect.
A framework evaluating developmental risk is similar to adult risk assessment in that it includes exposure assessment, toxicity assessment, and risk characterization. Each of these items involves evaluation of multiple parameters (Figure 9-1).
Tremendous advances have been made in recent years that facilitate the assembly of developmental toxicokinetic models. Such data include relevant ontogenic information about many of the phase I and phase II enzymes (Hines and McCarver 2002; McCarver and Hines 2002), which should be used for risk assessment (Dorne 2004). For the assessment of trichloroethylene, substantial data are available on toxicokinetics. Some, albeit less, information is available for items regarding developmental processes and their consequences (see Chapter 5). These advances, although still incomplete, can improve the precision of the risk assessment for trichloroethylene by narrowing uncertainty.
Approaches to Developmental Risk Assessment
Approaches defined under the new EPA guidelines for adult cancer risk assessment includes flexibility and the use of biologically based response models when appropriate (EPA 2005a). Similar flexibility and appropriate use of models are merited in evaluating toxicant risk for children under the ethical concept that children deserve, at a minimum, the same level of protection as adults. For any specific chemical, developmental susceptibility to
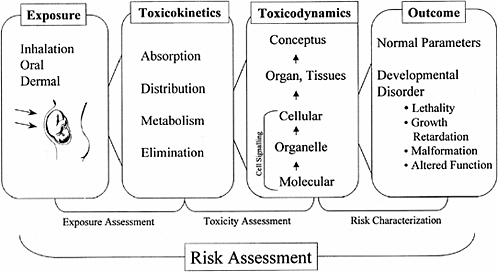
FIGURE 9-1 Overall framework to describe assessment of the effects of a toxicant on development. SOURCE: NRC 2000.
toxicants could be addressed in several ways depending on the information available. These include the following:
-
Using developmental physiologically based pharmacokinetic (PBPK) modeling with parameter estimates appropriate for children: Such models can improve the development of relative-risk information for a specific xenobiotic exposure (Ginsberg et al. 2004a). The PBPK approach has already been used to evaluate pediatric drug therapy (Ginsberg et al. 2004b) and to assess adult risks from trichloroethylene (see Chapter 10); a similar effort should be applied for risk assessment in children. To facilitate such modeling, relevant data sets of developmental physiologic variables have been assembled and published (Haddad et al. 2001). A review of developmental pharmacokinetic modeling suggests that the 3.16-fold default uncertainty value commonly used for interindividual pharmacokinetic variability might be insufficient for some chemicals (Fawer et al. 1979). In contrast, Pelekis et al. (2001) developed a pediatric PBPK model for multiple volatile organic compounds using parameter estimates for a 10-kg child (1-2 years old) that suggests such children might not need additional protection. However, it is important to recognize that their model has not been validated with any in vivo developmental data; potentially this could be done with observational data. In addition, the model addresses only the parent compound and not the fractional metabolic clearance of any putative toxic metabolites; it is also a single-age model that does not reflect every period of childhood. For example, toddlers typically have enhanced clearance compared with younger and older children, as well as adults, because of enhanced relative liver and kidney size. Thus, although this is a good start in utilizing the PBPK approach, additional work should be done to ensure that the observation is robust and to assess the degree to which it applies across development. As stated by Ginsberg et al. (2004a), multiple age-appropriate physiologic models are needed. Because available information about the ontogeny of pathways involved in trichloroethylene disposition is substantial (see below), this approach would markedly decrease uncertainty about trichloroethylene exposures for the fetus and the child. However, similar to adults, this approach will not assess toxicodynamic differences and, importantly, will not address end points that might be unique to the fetus and the child. To be optimal, the information must be further integrated using a serial approach that considers exposures serially from in utero to adulthood. Regardless of the limitations, this approach will significantly improve precision in risk estimates for children.
-
Using a developmental uncertainty factor: Such a factor could be added to the adult analysis to provide an arbitrary measure of added safety in consideration of a potential differential developmental susceptibility. This empiric approach has been used for risk assessment for childhood
-
susceptibility for multiple chemicals. However, the approach should be used with decreasing frequency or the precision of the uncertainty factors should improve as more data become available. For trichloroethylene, insufficient toxicodynamic data necessitates using an uncertainty factor for toxicodynamic effects, but the uncertainty of increased susceptibility from exposure might be eliminated by using developmental PBPK modeling.
-
Establishing that children do not need greater protection than adults: If the variability present in adults is such that a sufficient margin of safety is present, an empiric decision could be made that no additional protection is needed for children. Generally, in risk assessment, this strategy is deemed appropriate when the variability is small and any estimate that ignores it will not be far from the truth. For developmental risk assessment, such a decision requires substantial justification, including detailed documentation of no added toxicodynamic risk during development. For trichloroethylene, as well as most other compounds, insufficient information is available to justify this approach.
Human Disposition of Trichloroethylene
EPA has clearly recognized the wide variability in trichloroethylene disposition, assigning a 50-fold variation, and also correctly recognized that this variability might contribute to susceptibility. Among adults, trichloroethylene disposition varies at least 7-fold, and perhaps as much as 50-fold (Lipscomb et al. 1997; Fisher et al. 1998). Variation in all aspects of trichloroethylene disposition—including absorption, distribution, metabolism, and excretion—affects the degree of biologic exposure to trichloroethylene and its metabolites (Astrand 1975). For example, trichloroethylene is poorly water soluble and highly fat soluble. After the same dose, trichloroethylene blood concentrations and urinary excretion of metabolites are expected to be greater in obese than in slim individuals (Sato et al. 1991). Similarly, the blood concentration of trichloroethylene and the total trichloroethylene body burden are expected to be higher in women than in men (Sato et al. 1991). Physical exertion during exposure to trichloroethylene, which is associated with increased pulmonary ventilation and cardiac output, is associated with increased adsorption, blood concentrations, and metabolite excretion (Astrand 1975).
Trichloroethylene is metabolized to chloral, which spontaneously hydrates to form chloral hydrate (Byington and Leibman 1965). This initial metabolic step, which might involve the transient formation of an intermediate epoxide (Miller and Guengerich 1982), is rate limiting and is catalyzed predominantly by the cytochrome P-450 enzyme CYP2E1 (Ikeda et al. 1980; Nakajima et al. 1992a; Lipscomb et al. 1997). Of multiple cytochrome P-450 enzymes tested in vitro, trichloroethylene metabolism to chloral hy-
drate correlated only with immunologically detected CYP2E1, and chloral hydrate formation significantly correlated with the oxidation of known CYP2E1 substrates (Lipscomb et al. 1997). The metabolism of chloral hydrate depends on two forward pathways (oxidation to trichloroacetic acid and reduction to trichloroethanol) and one back reaction (forming chloral hydrate from trichloroethanol). The reduction of chloral hydrate to trichloroethanol is catalyzed by alcohol dehydrogenase (Friedman and Cooper 1960) and is NADH dependent, whereas the oxidation of chloral hydrate to trichloroacetic acid is mediated by aldehyde dehydrogenase (Cooper and Friedman 1958). The back reaction forming chloral hydrate from trichloroethanol appears to be catalyzed by CYP2E1 (Barton et al. 1996).
Trichloroethanol is also conjugated with glucuronate and the resulting water-soluble conjugate is excreted in the urine. Whether dichloroacetic acid is formed is controversial, with some investigators (Henderson et al. 1997) but not others documenting its presence in very low amounts in the blood of children given chloral hydrate for therapeutic indications. Brashear et al. (1997) found dichloroacetic acid at the limits of detection for mass spectrometry in adults exposed to trichloroethylene at 100 parts per million. Others have suggested that the presence of dichloroacetic acid is an analytic artifact and that dichloroacetic acid, if formed, is rapidly removed, precluding its measurement (Merdink et al. 1998). Further, no mechanism of dichloroacetic acid formation in humans has been described. The alternative to the initial CYP2E1-mediated trichloroethylene oxidation to chloral hydrate is direct conjugation of trichloroethylene with glutathione. This pathway accounts for less than 1% of the disposition of trichloroethylene and ultimately leads to the formation of cysteine conjugates or mercapturates (Green et al. 1997a). Although quantitatively a minor pathway, these metabolites have been associated with renal carcinogenesis and therefore are important in risk assessment (see Chapter 3).
Although intersubject differences in adsorption and distribution occur among adults, most of the variation in trichloroethylene disposition is secondary to differences in metabolism. In microsomal preparations from 23 human livers, CYP2E1-mediated trichloroethylene intrinsic clearance (Vmax/Km) to chloral hydrate varied about 6-fold (Lipscomb et al. 1997). Among healthy volunteers exposed to ambient trichloroethylene, the urinary excretion of trichloroacetic acid and trichloroethanol varied 6- to 7-fold (Fisher et al. 1998), whereas trichloroacetic acid formation after dosing with chloral hydrate varied almost 10-fold, with 5% to 47% of the dose excreted as urinary trichloroacetic acid (Marshall and Owens 1954). In contrast to this striking between-subject variability, within subject variability appeared to be less than 2-fold.
Developmental Toxicokinetic Information
Improved analysis of trichloroethylene developmental toxicokinetics is important because recent population studies suggest that 3% to 7% of children have measurable amounts of trichloroethylene in their blood (Sexton et al. 2005). Although less is known about trichloroethylene disposition in children than in adults, substantial information is available that merits consideration in risk analysis. This includes information about the developmental profiles of CYP2E1, alcohol dehydrogenase, and aldehyde dehydrogenase, as well as studies of the disposition of chloral hydrate in children. The developmental profile of CYP2E1, the enzyme responsible for the rate-limiting step, has been well characterized across fetal and pediatric age groups (Johnsrud et al. 2003). Further, substantial pharmacokinetic information is known about the disposition of the major proximate metabolite, chloral hydrate, a sedative commonly used in children. Dichloroacetic acid, which may or may not be a metabolite in humans, is used therapeutically in the relatively rare disorder congenital lactic acidosis, and some pharmacokinetic data are available in adults and children, so that age-related comparisons are possible.
Important Physiologic Changes
Many physiologic changes directly affect developmental toxicokinetics, whereas others constitute toxicodynamic differences or additional toxicodynamic targets not present in adults. The biologically relevant internal dose of trichloroethylene as well as that of its metabolites likely is altered by multiple physiologic developmental changes. These include not only overall growth but also changes in body composition, relative organ size, and hormonal changes. Of particular importance to trichloroethylene and other compounds that deposit into fat is the doubling of body fat during early infancy with a concomitant fall in the amount of total body water. The liver and kidney, the predominant organs of overall toxicant activation and deactivation, are severalfold larger relative to body weight in children than in adults; this effect is greatest among toddlers (Maxwell 1984). Growth hormone concentrations increase during the newborn period and again with puberty (Quattrin et al. 1990; Rose et al. 1991; Main et al. 1994). Such growth hormone changes are associated with differences in drug disposition and drug-metabolizing enzyme expression (Redmond et al. 1978; Lambert et al. 1986; Butler et al. 1989). Growth hormone changes are associated with alterations in developmental gene expression mediated by many pathways, including the early response genes c-fos and c-jun, and HNF-6, a hepatic transcription factor that activates a network involved in cytochrome P-450 and plasma protein gene regulation (Rastegar et al. 2000). In addition
to the differences in disposition that occur from growth and development, the fact that growth and development are occurring provides additional targets for derangement that are not present in adults. Thus, there are striking physiologic differences in children that affect xenobiotic disposition and likely alter their risk.
Developmental differences have been demonstrated in every aspect of pharmacokinetics; including absorption, distribution, metabolism, and excretion, using various therapeutic agents (see review by McCarver 2004). The impact of these differences on toxicokinetics has been less well evaluated. However, as stated above, in vitro and in vivo data have been generated that might facilitate PBPK modeling of trichloroethylene exposure in children. Oral and percutaneous absorption differences have been documented with many therapeutic agents (Heimann 1981; West et al. 1981; Rutter 1987; Barrett and Rutter 1994). Both routes are relevant for trichloroethylene exposure. Neither has been studied specifically for trichloroethylene; however, information generated for other compounds could be used to model the expected differences from these factors. Developmental variation in oral absorption is most marked in infancy and is due to differences in gastric pH, gastric emptying, pancreatic enzymes, and first-pass metabolism in the stomach, small intestine, and liver. Developmental differences in percutaneous absorption are due to differences in skin thickness, vascularization, and hydration. Distribution differs with age because of changes in body composition, protein, and tissue binding (Heimann 1981; Fisher et al. 1982; Nau et al. 1983; Lerman et al. 1989). An important distribution issue for the fetus is the ability of compounds to redistribute from amniotic fluid back to the fetus. In a rodent model, trichloroacetic acid was found to cycle from the fetus into the amniotic fluid and back into the fetus (Ghantous et al. 1986). Thus, amniotic fluid could act as a reservoir. Tissue drug binding, a more direct marker of the pharmacokinetic-pharmacodynamic interface than plasma values, might also be age dependent for some compounds (Park et al. 1982). Although the distribution of drugs across membranes recently has been shown to be influenced by a growing number of drug transporters, study of their ontogeny has just begun. It appears unlikely that a small, lipophilic, highly diffusible compound such as trichloroethylene would require a transporter, but this issue has not been addressed. In summary, although these processes are complex, sufficient information exists to support exploration of PBPK modeling for trichloroethylene pediatric exposure.
Ontogeny of Human Enzymes Involved in Trichloroethylene Disposition
The use of generalities about the direction and extent of differences in metabolism in children compared with adults in risk assessment cannot be supported by currently available data. The developmental expression pat-
tern varies by enzyme (Hines and McCarver 2002; McCarver and Hines 2002). Moreover, human and animal data are likely to differ, and such interspecies differences could be substantive. In contrast to rodents, human CYP isoform expression occurs relatively early, generally before birth or within the first several months of life (Hines and McCarver 2002). CYP2E1, the enzyme responsible for the rate-limiting step in trichloroethylene disposition, is expressed by week 8 of gestation in human fetal cephalic tissue at greater concentrations than in corresponding human fetal hepatic tissue (Brzezinski et al. 1999). In humans, hepatic expression occurs as early as the second trimester and rapidly increases after birth, with adult expression levels being reached by 3 months (Carpenter et al. 1996; Johnsrud et al. 2003). In contrast, rodent hepatic CYP2E1 expression begins postnatally (Keeter et al. 1990). Importantly, human CYP2E1 developmental expression data are sufficiently complete to support developmental PBPK modeling.
Differences in human alcohol dehydrogenase might be of equal or greater relevance to human trichloroethylene risk assessment compared with CYP2E1. Although CYP2E1 is the rate-limiting enzyme, the halogenated acetic acids trichloroacetic acid and dichloroacetic acid have been suggested as the proximate putative teratogenic species (Johnson et al. 1998a) as well as, perhaps, proximate nonrenal carcinogens (Herren-Freund et al. 1987; DeAngelo et al. 1989). As such, variability in alcohol dehydrogenase, as well as aldehyde dehydrogenase variability (discussed below), would influence the amount of chloral hydrate that is diverted to trichloroacetic acid, one of the putative toxicants. If alcohol-dehydrogenase-mediated conversion to trichloroethanol, a theoretically less toxic metabolite, is limited from immaturity or genetic factors, then more chloral hydrate would be available for conversion to trichloroacetic acid. However, this has not been directly confirmed in humans. Human alcohol dehydrogenase is a superfamily consisting of at least five classes encoded at seven genetic loci. Class I alcohol dehydrogenase includes three isoforms (α, β, and γ), and dimers of this class are the most effective at metabolizing ethanol to acetaldehyde. Thus, these isoforms have been well studied with ethanol (see below). In contrast, the relative capability of the various alcohol dehydrogenase isoforms in reducing chloral hydrate to trichloroethanol has not been published. As a general rule, for class I alcohol dehydrogenase, alcohols with bulky substituents are better substrates than ethanol. The ontogeny of alcohol dehydrogenase was described more than 30 years ago in seminal work by Smith et al. (1971), which was replicated and expanded by Estonius et al. (1996). In late gestation, fetal alcohol dehydrogenase activity is about 25% that of adult activity (Pikkarainen and Raiha 1967). Although expression is greatest in liver, class I alcohol dehydrogenase transcripts are widely distributed in all organs except fetal and adult brain, adult kidney, and placenta. In first-trimester human fetal liver samples, alcohol dehydrogenase α, encoded at ADH1A,
is the only detectable class I isoform. Beginning in the second trimester and continuing into the early third trimester, hepatic alcohol dehydrogenases α, β, and γ (encoded by ADH1A, ADH1B, and ADH1C, respectively) are all present, but alcohol dehydrogenases α and β predominate. By the late third trimester, human hepatic ADH1C expression has increased markedly, but ADH1B expression still predominates. In adult liver, ADH1A expression is not detected, and expression of ADH1B and ADH1C is equivalent. In lung, only ADH1B expression is detected, and it is similar in adult and fetal samples (Estonius et al. 1996). Alcohol dehydrogenase class III is expressed in virtually all tissues, including in the fetus; expression in fetal brain appears to be somewhat greater than in adult brain.
Aldehyde dehydrogenase is a superfamily of NAD(P)+-dependent enzymes whose characteristics and substrate specificity vary (Vasiliou et al. 2004). The aldehyde dehydrogenase isoforms are encoded at 17 genetic loci (at least), and the various forms are highly expressed in the microsomal, mitochondrial, and soluble fractions of the liver as well as at lower levels in other tissues (Koivula 1975). Which aldehyde dehydrogenase isoforms are capable of and most efficient at metabolizing chloral hydrate to trichloroacetic acid is unknown but needs to be determined. ALDH1A1, ALDH1A2, ALDH1A3, and ALDH8A1 are involved in the oxidation of retinaldehyde to retinoic acid, a critical factor in many developmental processes and signaling events. ALDH1A1, ALDH1B1, and ALDH2 are all involved in the oxidation of acetaldehyde, the proximate metabolite of ethanol. In addition, ALDH1L1 hydrolyzes 10-formyltetrahydrolate to tetrahydrofolate, another reaction critical to development (Krupenko et al. 1997). Multiple forms of aldehyde dehydrogenase participate in the metabolism of 4-hydroxynonenal and malondialdehyde, two predominant products of human lipid peroxidation, and others in the formation of glutamate and in γ-aminobutyric acid metabolism. Treating rats with trichloroethylene depressed aldehyde dehydrogenase activity for short-chain aliphatic aldehydes in the mitochondrial and cytosolic fractions but not in the microsomal fraction (Wang et al. 1999), and this activity appears to be due to chloral hydrate [median inhibitory concentration = 8 μM] (Wang et al. 1999; Poon et al. 2002). Thus, human aldehyde dehydrogenase theoretically is an important interaction point between trichloroethylene and multiple xenobiotics as well as multiple physiologic mechanisms. As such, more information on its role in trichloroethylene and chloral hydrate metabolism is needed.
Glutathione S-transferases (GSTs) are members of a family of enzymes that conjugate glutathione to electrophilic compounds. However, as noted above, glutathione conjugation represents the initial enzyme in the toxification pathway associated with renal carcinogenesis. The enzyme consists of two homodimeric subunits from one of five GST subunit classes (alpha, beta, mu, pi, theta, and zeta), and each enzyme is designated with a letter
indicating its class membership (A, B, M, P, T, and Z, respectively). Which GST is most efficient in conjugating trichloroethylene is unknown. GST ontogeny was recently reviewed (McCarver and Hines 2002; Ginsberg et al. 2004a). Briefly, the hepatic alpha isoforms, GSTA1 and GSTA2, are detected in the early fetal period and reach adult levels in the first 1-2 years of life (Strange et al. 1989). Fetal hepatic isoform mu expression is about 22% of adult expression and increases about 5-fold shortly after birth to adult levels (Strange et al. 1989). Isoform pi is the predominant class in early hepatic development, being expressed in the human fetus and young infant at levels that exceed adult concentrations by 500- and 200-fold, respectively (Strange et al. 1989). GSTA1 and GSTA2 are also expressed in the fetal kidney and renal expression increases in the first two postnatal years, whereas GSTM is lower in postnatal than in fetal samples (Beckett et al. 1990). GSTP1 has also been documented in early fetal renal collecting ducts (van Lieshout et al. 1998). Pulmonary developmental expression has also been evaluated with immunohistochemical and radioimmunoassays (Hiley et al. 1989). GSTP1 is expressed at high concentrations in early fetal pulmonary ductal columnar cells, but this expression decreases with gestation. In contrast, pulmonary expression of GSTM, GSTA1, and GSTA2 are relatively low, but consistent, across gestation.
Chloral Hydrate and Dichloroacetic Acid Studies in Children
The trichloroethylene metabolite chloral hydrate has been used as a sedative for more than 150 years and is prescribed frequently for children, usually as a single dose for procedural sedation. Adults are typically given 1 g, whereas children are given 50-75 mg/kg up to the adult dose. The sedative effect is believed to be due to the metabolite trichloroethanol (Marshall and Owens 1954), although this assumption has been controversial (Mayers et al. 1992). Concern about chloral hydrate use in children includes its competition with bilirubin glucuronidation in the newborn period (Lambert et al. 1990) and the increased risk of arrhythmias, particularly among patients with congenital heart disease (Hirsch and Zauder 1986).
Concern about the possible carcinogenic potential of chloral hydrate in children (Steinberg 1993) resulted in a series of developmental studies by the National Toxicology Program (NTP 2002a,b). In a 2-year study, B6C3F1 neonatal female mice under multiple chloral hydrate regimens, including groups given single or multiple doses for 2 years (both ranging from 0 to 100 mg/kg of body weight), the results were considered equivocal for carcinogenesis (NTP 2002a). The only positive result was an increased incidence of pituitary gland pars distalis adenomas at the highest dose (100 mg/kg for 24 months) without evidence of a dose response. Further, the incidence of pituitary adenomas in the high-dose group was similar to
historical but not concurrent controls. No single-dose regimen was associated with carcinogenesis. In the 2-year B6C3F1 male mouse study, increased incidence of hepatocellular adenoma or carcinoma (combined variable) was observed in mice fed ad libitum, and increased incidence of hepatocellular carcinoma was observed in dietary-controlled mice, which was associated with peroxisome proliferation (NTP 2002b). The concern over hepatocellular carcinogenesis was limited by the recognition that humans exhibited very weak liver peroxisome proliferative responses (Waxman 1999).
Although chloral hydrate is used clinically, the number of pharmacokinetic studies in children of various ages is limited. Critically ill infants and children (n = 22, ranging from 31 weeks postconceptional age to 13.6 years) participated in a pharmacokinetic study of chloral hydrate (Mayers et al. 1991). Patients were divided into three age groups, but it is unclear how many subjects were in each group. After a standard sedative oral dose (50 mg/kg, presumably to a maximum adult dose of 1 g), mean peak chloral hydrate plasma concentrations ranged from 3.89 ± 2.87 mg/L among toddlers and older children to 6.23 ± 2.28 mg/L among full-term infants to 8.01 ± 5.12 mg/mL among preterm newborns. After an initial rapid distribution phase, chloral hydrate terminal elimination occurred more slowly with a half-life of 1 to 10 hours. Apparent oral chloral hydrate clearance was about 5 L/hour/kg and did not differ by age. Trichloroethanol peak concentrations ranged from about 27 to 36 mg/L. Older children had a shorter trichloroethanol half-life of about 10 hours, whereas preterm and term infant groups had half-lives of about 40 and 28 hours, respectively. Because trichloroethylene is metabolized by glucuronidation, a pathway known to increase during the first few months of life, the observation of age-dependent trichloroethanol elimination was not surprising. Trichloroacetic acid increased to significant concentrations (10-20 mg/L) during the 7-day study and did not decline, so a half-life could be determined only in one patient. The authors noted that the dose of metric area under the curve for trichloroacetic acid during the first 24 hours was greater among older children. They speculated that an inability of infants to form the oxidative metabolite trichloroacetic acid was responsible, which suggests that aldehyde dehydrogenase was immature in these subjects. Unfortunately, the study did not include urinary metabolite data, which are necessary for determining the relative conversion to trichloroethanol and to trichloroacetic acid. Henderson et al. (1997) described chloral hydrate kinetics in three children treated with chloral hydrate (50 mg/kg) alone; with chloral hydrate (50 mg/kg) 15 minutes before a dose of [13C]dichloroacetic acid, or with two chloral hydrate doses before and after a dose of [13C]dichloroacetic acid. Details of the gas chromatography-mass spectrometry assay including linearity and reproducibility were not given. Chloral hydrate peak values were not given. The peak concentration of trichloroethylene was 115 mg/mL at 25
minutes. Peak values for dichloroacetic acid (22 mg/mL) and trichloroacetic acid (65 mg/mL) occurred much later, at 7.5 and 11.5 hours, respectively. Importantly, the time course of trichloroacetic acid does not match that in other reports. Henderson et al. (1997) further reported coadministration of dichloroacetic acid with chloral hydrate prolonged the half-life of dichloroacetic acid, suggesting that chloral hydrate inhibits the metabolism of dichloroacetic acid. The pharmacokinetics of dichloroacetic acid have been determined in a few studies (Fox et al. 1996; Barshop et al. 2004), one of which included mostly children (Barshop et al. 2004). Among healthy adult volunteers (Curry et al. 1991), oral and intravenous dichloroacetic acid bioavailability were similar, and no gender difference was observed. The apparent terminal half-life was noted to increase with chronic dosing by more than 20-fold in healthy adults (Curry et al. 1985, 1991) and by more than 10-fold in lactic acidosis patients (Barshop et al. 2004). Among adults treated for lactic acidosis with single doses of dichloroacetic acid varying from 30 to 100 mg/kg, dichloroacetic acid elimination was zero order at concentrations above 80-120 μg/mL and first order at lower concentrations with a terminal half-life of about 1.2 hours (Fox et al. 1996). The typical maximum concentration in both adult and pediatric lactic acidosis patients is 120-160 μg/mL after 50 mg/kg intravenously (Fox et al. 1996; Barshop et al. 2004). Among 37 patients, including 31 children (ages 7 months to 17.8 years) treated with either single doses (50 mg/kg/day, n = 6) or repeated doses (variable doses, n = 31) for congenital lactic acidosis, the terminal half-lives were 86 minutes and 11 hours, respectively (Barshop et al. 2004). Although studies in healthy adult volunteers are relevant to the general population, metabolic studies in children and adults with lactic acidosis might not be relevant as the underlying acid-base abnormalities could alter enzymatic activity. Cord blood samples from 52 women given chloral hydrate during labor indicated that chloral hydrate crosses the human placenta (Bernstine et al. 1954). Chloral hydrate, trichloroethanol, and trichloroacetic acid were the same or higher in 50%, 63%, and 57% of cord blood samples, respectively, compared with paired maternal blood samples. Concentrations in amniotic fluid samples were described as matching fetal samples. In contrast to information for chloral hydrate, data on the in vivo human disposition of low concentrations of trichloroethylene in pregnant women have not been reported but are needed. The human ontogeny of both dichloroacetic acid and chloral hydrate metabolism in vitro, as well as chloral hydrate metabolism in vivo, merits further definitive study so that the data could be used for improved PBPK modeling.
GENETIC SUSCEPTIBILITY
Currently, 114 human CYP2E1 single nucleotide polymorphisms have been reported to the National Center for Biotechnology Information database dbSNP. Of these, 65 have been validated. Of the validated single nucleotide polymorphisms, 40 occur in introns, 3 occur in the mRNA untranslated region, and 22 occur in the coding sequence or in the upstream sequence. For many single nucleotide polymorphisms, the functional significance, if any, is unknown. Three allelic variants, CYP2E1*2, *3, and *4 have been defined, each of which contains a single nucleotide polymorphisms leading to an amino acid change. However, two of these, CYP2E1*3, consisting of a guanine to adenine substitution at nucleotide (10023 G>A) (leading to a valine to isolencine change at amino acid 389 [V389I]), and CYP2E1*4, consisting of 4768 G>A (leading to V179I), were associated with normal in vitro CYP2E1 activity (Hu et al. 1997; Fairbrother et al. 1998). In contrast, 1168 G>A causes an arginine to histidine substitution (R76H), which was associated with about a two-thirds decrease in both enzyme expression and activity in vitro (Hu et al. 1997). However, the variant is rare, occurring in about 1% of Asian populations and not detected among Europeans. Another single nucleotide polymorphism, adenine to thymine at nucleotide 11112 (11112 A>T), is associated with a histidine to leucine substitution (H457L), but has not been given a haplotype designation, and its functional significance has not been reported in vitro or in vivo (Solus et al. 2004). Two haplotypes, CYP2E1*1B and CYP2E1*1D, contain genetic polymorphisms in the upstream regulatory region. CYP2E1*1D has been shown to be associated with increased CYP2E1 activity in vivo among individuals who are obese or who consume ethanol (McCarver et al. 1998). This insertion of 96 base pairs in the CYP2E1 regulatory regions occurs in about 25% of African Americans and a smaller percentage of Caucasians (McCarver et al. 1998); it represents the only known frequent, functional human CYP2E1 polymorphism described to date.
As noted above, it is unknown which human alcohol dehydrogenase isoform is most efficient at oxidizing chloral to trichloroethanol. The loci encoding two of the three class I alcohol dehydrogenase isoforms, ADH1B and ADH1C, exhibit functional genetic polymorphisms. Two ADH1B variants, ADH1B*2 and *3, have been associated with decreased susceptibility to fetal alcohol spectrum disorders (McCarver et al. 1997; Viljoen et al. 2001; Das et al. 2004; Warren and Li 2005), presumably from increased ethanol elimination by the enzymes these variants encode (Thomasson et al. 1995; Neumark et al. 2004). Similarly, alcohol dehydrogenase genetic polymorphisms have been associated with differences in the risk for alcohol dependence (Thomasson et al. 1991) and cancer in some studies (Yokoyama et al. 2001; Yang et al. 2002; Coutelle et al. 2004) but not others (Olshan
et al. 2001). The alcohol dehydrogenase variants are relatively common, occurring at allelic frequencies of 20% to 50%, depending on the ethnic group (Warren and Li 2005). Thus, it is plausible that the same alcohol dehydrogenase polymorphisms could be important factors for trichloroethylene teratogenic and carcinogenic susceptibility; studies directly addressing this hypothesis are needed.
Twenty-two aldehyde dehydrogenase genetic variants have been described, of which at least 11 appear to be functional (Vasiliou et al. 2004; Aldehyde Dehydrogenase Gene Superfamily Database1). Among the most studied variants is ALDH2*2, which encodes a low-activity variant of mitochondrial aldehyde dehydrogenase and occurs in about 30% of individuals of Asian descent (Thomasson et al. 1991). This genetic variant is associated with a lower frequency of alcohol dependence and inability to oxidize acetaldehyde, yielding a flushing reaction during ethanol consumption (Itoh et al. 1997). Monte Carlo analyses of the variability factors of other somewhat similar compounds, such as ethanol and toluene, suggest that the default pharmacokinetic uncertainty factor is not sufficient to account for the ALDH2*2 polymorphism (Ginsberg et al. 2002). The impact of aldehyde dehydrogenase variation on trichloroethylene disposition is unknown but is critical information for integrating genetic information into risk assessment.
GSTs are a family of phase II enzymes involved in the metabolism of many xenobiotics (Mannervik et al. 1992). Mammalian GSTs belong to three families of proteins that are expressed in cytosol, mitochondrial, and microsomal cellular fractions (Hayes et al. 2005). GSTs are catalytically active as hetero- or homodimers. These proteins are expressed in most human tissues, although numerous isoforms and subtypes are differentially expressed in cells and tissues (Strange et al. 1991). At least 16 cytosolic GSTs have been identified. These enzymes are named based on their amino acid sequences and immunologic characteristics (Board 1981; Mannervik 1985; Board et al. 1990). In general, mammalian cytosolic GSTs are divided into seven classes, which have been named alpha, mu, pi, sigma, theta, zeta, and omega. Many of the isoforms have known polymorphisms (Hayes et al. 2005). Examples include GSTM1*A (associated with normal protein levels and activity), GST1*B (associated with low protein levels), GSTM1*0 (a gene deletion that leads to a null phenotype), and GSTM1*1 × 2 (gene duplication). GSTM1*0 is seen in more than 50% of some populations (Board et al. 1990). The GSTT family has at least three known polymorphisms (Strange et al. 1984). Various classes of GSTs, especially their null mutants, have been associated with cancer (Strange et al. 1991; van Poppel et al. 1992).
|
1 |
See www.aldh.org. |
Whereas GSTs are generally considered to be important in the deactivation of electrophiles and oxidants, there are a few examples of bioactivation reactions. In these cases, the glutathione conjugate of a xenobiotic metabolite is more toxic than the xenobiotic or its metabolite alone. Such is the case for the glutathione conjugate of trichloroethylene known as S-1,2-dichlorovinyl-L-glutathione, which forms S-1,2-dichlorovinyl-L-cysteine in the presence of β-lyase (Dekant 1986). S-1,2-Dichlorovinyl-L-cysteine has been associated with kidney cancer (see Chapter 3), particularly in humans with a mutation in the von Hippel-Lindau gene. Populations with increased GSTT1 activity and a GSTM1 null allele have been found to be at particular risk for renal cell cancer (Brüning et al. 1997b).
Thus, depending on the specific GST isoform involved, a polymorphism in these enzymes can be expected to increase or decrease cancer risk. Predicting the human cancer or noncancer risks in humans depends on the specific gene and polymorphism expressed. Data for trichloroethylene are incomplete; which GST isoforms are most efficient in trichloroethylene disposition is unknown.
ACQUIRED STATES WITH POSSIBLE ALTERED SUSCEPTIBILITY
Multiple conditions, including ethanol ingestion, exposure to other solvents, fasting or starvation, obesity and diabetes, and consumption of some popular dietary items, such as green tea, have been shown to induce CYP2E1 (McCarver et al. 1998; Lieber 2004; Liangpunsakul et al. 2005; Yang and Raner 2005). Many conditions associated with CYP2E1 induction are quite common and therefore likely to occur concomitantly with trichloroethylene exposure. Alcoholism affects about 14 million Americans annually; it is estimated that about half of Americans over the age of 12, or about 110 million Americans, consume ethanol (SAMHSA 2003). Thus, a clear understanding of the interactions between trichloroethylene and ethanol and the effect on the trichloroethylene risk assessment is needed. The interaction between ethanol and trichloroethylene is complex and includes alterations in trichloroethylene kinetics and dynamics as well as those of its metabolites (for full discussion, see review by Pastino et al. [2000] and Chapter 10). Chronic heavy drinkers are likely to have enhanced trichloroethylene metabolism due to CYP2E1 induction. However, this is likely to be most relevant at high concentrations of trichloroethylene at which saturation would occur in the absence of enzyme induction. Chronic heavy drinkers who have progressed to cirrhotic liver damage could have decreased trichloroethylene metabolism. Individuals who have not recently consumed sufficient ethanol to induce CYP2E1 but who have acute, relatively concomitant exposure have decreased trichloroethylene metabolism from competitive inhibition (Muller et al. 1975). In addition to the effect on disposition, simultaneous
exposure to ethanol and chloral hydrate increases the sedative effects, perhaps from the ethanol-mediated shift in choral hydrate metabolism from oxidation to trichloroacetic acid to reduction to trichloroethanol (Watanabe et al. 1998). Obesity and diabetes induce CYP2E1 and both occur in millions of Americans (Harris 1995; Flegal et al. 2002). Although the impact of these common diseases would be expected to be similar to that from ethanol- or other solvent-mediated CYP2E1 induction, the relationships might be more complex. For example, increased body fat content affects trichloroethylene distribution so that urinary excretion rates are estimated to be greater in thin men than in obese men (Sato 1993). This effect might offset any enhanced CYP2E1-mediated metabolism in obesity. Notably, trichloroethylene-induced hepatotoxicity was not enhanced in a chemically induced rodent model of diabetes (Hanasono et al. 1975). Thus, direct study of the impact of these common states on trichloroethylene disposition and risk merits direct investigation.
GENDER
Simulated models suggest that, after the same dose of trichloroethylene, women have a slightly greater total body burden of trichloroethylene and its metabolites than men, reflected in a larger amount of total urinary metabolites (Sato et al. 1991). However, the size of this gender difference was relatively small (blood trichloroethylene concentrations 30% higher after 16 hours), and it was due to increased body fat content in women. In 16 volunteers (8 of each gender), equivalent ambient trichloroethylene exposure (100 parts per million for 4 hours) resulted in about 3.4-fold higher maximal concentrations and area-under-the-time-concentration curves of S-1,2-dichlorovinyl-L-glutathione in men than in women (Lash et al. 1999). This difference is intriguing because male rats were found to be efficient at trichloroethylene glutathione conjugation and male rat tubular cells were more susceptible to acute toxicity induced by S-1,2-dichlorovinyl-L-glutathione and S-1,2-dichlorovinyl-L-cysteine (Lash et al. 2001b). In contrast, one human case-control study of renal carcinogenesis showed an increased susceptibility for women (Dosemeci et al. 1999).
HUMAN VARIABILITY AND THE USE OF UNCERTAINTY FACTORS
Uncertainty factors are applied to risk estimates to account for variability in human populations as well as other factors. Following is a synopsis of EPA’s use of uncertainty factors in its draft health risk assessment for trichloroethylene.
The oral reference dose for trichloroethylene noncancer effects was calculated as 3 × 10-4 mg/kg-day, based on subchronic studies of rats and mice that showed effects at 1mg/kg-day. The uncertainty factors included the following:
-
A 50-fold uncertainty factor to account for differences between average and sensitive humans. This value was calculated by multiplying the value chosen for human pharmacokinetic variability (set at 15-20, see discussion below) by a 101/2-fold2 pharmacodynamic variation, which is the EPA default value.
-
A 101/2-fold default uncertainty factor for animal-to-human pharmacodynamic uncertainty. The previous value (15-20) was considered to account for animal-to-human pharmacokinetic uncertainty.
-
A 101/2-fold uncertainty for using subchronic instead of lifetime studies. EPA states that duration-response trends are not evident in animal studies, but some human studies indicate that prolonged exposure to trichloroethylene can increase the severity of effects. Therefore, the partial 101/2-fold uncertainty was used “until duration-response relationships are better characterized in humans.”
-
A 101/2-fold uncertainty factor for extrapolation from a lowest-observed-adverse-effect level (LOAEL) to a no-observed-adverse-effect level (NOAEL) because adverse effects were observed at the 1 mg/kg-day point of departure. EPA stated that the standard 10-fold default was not used because “1 mg/kg-day appears to be at the boundary where effects can begin to be observed.”
-
A 101/2-fold uncertainty factor to reflect background exposures to trichloroethylene and its metabolites to address cumulative risks.
In total, this generates a 5,000-fold uncertainty factor. Ultimately, however, a factor of 3,000 was used as the divisor because it is the largest divisor used by EPA in the presence of substantial uncertainty (EPA 2001b).
The trichloroethylene inhalation concentration of 4 × 10-2 mg/m3 was based on a subchronic exposure of 38 mg/m3 showing adverse effects on the central nervous system in human occupational studies. The uncertainty factors included the following:
-
A 10-fold default uncertainty factor for human variation,
-
A 10-fold default uncertainty factor for using subchronic instead of lifetime studies, and
-
A 10-fold default uncertainty factor for extrapolation from a LOAEL to a NOAEL uncertainty because the central nervous system and endocrine effects were LOAELs observed in occupational studies.
For cancer end points, EPA chose to use a range of slope factors in view of risk factors that can modify the effects of trichloroethylene in different populations. Of the cancer studies evaluated (including rodent and human studies), the highest and lowest cancer slope factors were not used and the range of the remaining studies was maintained. According to EPA “these remaining estimates constitute a middle range of risk estimates where confidence is greatest” (EPA 2001b). Recognizing differences in human responses and the potential for sensitive populations, EPA further states that “a single risk value is not appropriate to describe the differential effects of [trichloroethylene]” and “alternative slope factors have not been consolidated into a single estimate.”
For determining the oral reference dose, EPA used a method other than the default factor for considering human variability. Instead, a 15- to 20-fold factor was used based on the uncertainty of the potency for mouse liver tumor production. These uncertainty factors for potency were assumed to be the same as the uncertainty in the internal dose estimates defined as the area under the curve for trichloroacetic acid and dichloroacetic acid, respectively. This 15- to 20-fold factor was assumed to account for both human pharmacokinetic variability and the pharmacokinetic differences between animals and humans.3 The derivation of the 15- to 20-fold uncertainty factor was not thoroughly explained. EPA states: “A factor of 15-20 reflects the pharmacokinetic uncertainty in the liver between the 50th and 99th percentiles (see Table 9-1).” In the risk assessment, EPA supports this value with studies demonstrating that continuing exposure to trichloroethylene can increase the severity of effects. Somewhat paradoxically, the same studies were used to support the lesser 101/2- fold uncertainty factor for the oral dose.
Table 9-1, reproduced below, is adapted from Table 15 of Rhomberg (2000), which details the uncertainty in human risks of liver tumors based on an analysis of mouse liver tumors using trichloroacetic acid and dichloroacetic acid as an internal dose measure. The uncertainty for human potency in producing liver tumors is estimated by assuming the toxic equivalency of
|
3 |
From EPA (2001b, p. 4-7): Human variation: The NOAELs, LOAELs, and LED10s for adverse liver effects were estimated using a pharmacokinetic model. The parameter uncertainty in these modeled dose estimates (estimated between the 50th and 99th percentiles, see Table 9-1) is 15-fold if plasma TCA (trichloroacetic acid) is used as the dose metric and 20-fold if plasma DCA (dichloroacetic acid) is used. |
TABLE 9-1 Approximate Uncertainty Analysis Based on Log-Normal Error
|
Human potency based on |
Uncertainty in potencies |
||||||||||
|
Uncertainty in animal internal dose, GSDA |
Uncertainty in human internal dose, GSDH |
Uncertainty in human potency close, GSDPOT |
|||||||||
|
Mouse liver, TCA-auc |
2.1 |
|
|
|
2.4 |
|
|
|
3.2 |
|
|
|
Mouse liver, DCA-auc |
2.7 |
|
|
|
2.2 |
|
|
|
3.6 |
|
|
|
Rat kidney, thiol |
3.4 |
|
|
|
6.2 |
|
|
|
9.0 |
|
|
|
Mouse lung, CH-auc |
3 |
|
|
|
9 |
|
|
|
11.7 |
|
|
|
Mouse lung, CH-max |
3.5 |
|
|
|
9 |
|
|
|
12.5 |
|
|
|
|
Factor different from median estimate |
||||||||||
|
|
Percentile of potency uncertainty distribution |
||||||||||
|
Human potency based on |
1 |
2.5 |
5 |
10 |
25 |
50 |
75 |
90 |
95 |
97.5 |
99 |
|
Mouse liver, TCA-auc |
1/15 |
1/10 |
1/7 |
1/4.4 |
1/2.2 |
1 |
2.2 |
4.4 |
7 |
10 |
15 |
|
Mouse liver, DCA-auc |
1/20 |
1/12 |
1/8 |
1/5 |
1/2.4 |
1 |
5 |
5 |
8 |
12 |
20 |
|
Rat kidney, thiol |
1/170 |
1/74 |
1/37 |
1/17 |
1/4.4 |
1 |
4.4 |
17 |
37 |
74 |
170 |
|
Mouse lung, CH-auc |
1/300 |
1/120 |
1/56 |
1/23 |
1/5.2 |
1 |
5.2 |
23 |
56 |
120 |
300 |
|
Mouse lung, CH-max |
1/360 |
140 |
1/63 |
1/25 |
1/5.4 |
1 |
5.4 |
25 |
63 |
140 |
360 |
|
ABBREVIATIONS: CH,chloral hydrate; DCA, dichloroacetic acid; TCA, trichloroacetic acid. SOURCE: Rhomberg 2000. Reprinted with permission; copyright 2000, Environmental Health Perspectives. |
|||||||||||
internal dose (presented as the area under the curve) between humans and mice. The uncertainty distributions for the animal internal dose and human internal dose—which were derived from a Bayesian uncertainty analysis (Bois 2000a) using the Clewell et al. (2000) model—were mathematically combined to estimate the distribution for the uncertainty in human potency. As mentioned, EPA used the fold difference between the 50th and 99th percentile to estimate both the variability in human pharmacokinetics and the uncertainty regarding the extrapolation of pharmacokinetic parameters from animals to humans. Problems with considering this as a measure of human pharmacokinetic variability include its derivation from mouse data, use of the Clewell model compared with others, and use of an assessment of the variability in cancer potency for assigning the variability of noncancer effects.4
FINDINGS AND RECOMMENDATIONS
The scientifically appropriate inclusion of human variability into risk assessment is an ongoing challenge. EPA has attempted to account for human variability, particularly for vulnerable populations, with an array of uncertainty factors. EPA is encouraged to increase the precision of risk estimates used for fetuses and children with PBPK modeling approaches similar to that used for adults. Similar approaches also can be used to account for ethanol consumption and exposure to compounds with known metabolic interactions. Multiple suggestions are given below for additional data analysis and data generation, particularly to advance understanding of the role of genetic polymorphisms in trichloroethylene disposition as determinants of susceptibility. The committee questions whether the use of variability in animals to approximate human variability is appropriate.
Increased use of PBPK modeling in developmental risk assessment is essential for addressing health issues specific to children. This approach would increase the precision of risk assessment by enhancing the understanding of biologically relevant dosimetry related to fetal or pediatric exposures compared with adult exposures as well as, in some cases, allow for extrapolations across routes of exposure. In addition, available animal studies with blood concentrations could be used to model relevant target tissue concentrations (e.g., central nervous system or kidney) that are necessary to cause
specific end point effects with identified modes of action. If such modeled target tissue concentrations were available for both animals and humans, it would enhance the ability to determine whether a developmental risk is plausible based on relevant tissue dosimetry. Within the developmental PBPK modeling, it is important to recognize that children do not represent a single group and that several physiologic stages must be considered.
It is unknown which human enzymatic isoforms dispose of trichloroethylene and its metabolites most efficiently. This information is critical for determining the relevance of various common functional genetic polymorphisms already known among enzyme families involved in trichloroethylene disposition as well as those that might be identified in the near future. Knowing the relevance of these genetic polymorphisms to risk assessment could then be determined. Approaches include PBPK modeling such as that already performed for parathion and warfarin (Gentry et al. 2002).
PBPK models are needed, but do not address the well-recognized pharmacodynamic differences between children in adults. Intersubject variation in pharmacodynamic factors has not been well quantitated among adults and pharmacodynamic modeling of toxicant effects has not been performed. Further, modeling of long-term end points of toxicant effects is difficult. For this to be attempted, the critical end points must be defined and appropriate shorter term effect biomarkers of these end points validated.
Recommendations:
PBPK models for different physiologic stages of childhood development should be created for trichloroethylene. Research on children’s exposure to trichloroethylene will be required to support model development, including measurement of trichloroethylene metabolites in breast milk and biological matrices from children (e.g., cord blood, amniotic fluid, and meconium) in different age groups.
-
Improved information on dermal absorption and alterations in risk from developmental differences in skin thickness, as well as surface area and body weight determinations, is needed.
-
If interspecies differences are determined to be predominantly related to compound disposition, PBPK models that incorporate critical comparative biology and physiology can be used to extrapolate developmental studies in animals to humans.
-
More research is needed to understand which human enzymatic isoforms are most important in disposing trichloroethylene and its metabolites.
-
Better characterization is needed of the impact of physiologic conditions and disease states on trichloroethylene toxicity, par-
-
ticularly with low-dose chronic exposure. It is possible that existing data sets could be mined for pertinent information, particularly for common disorders or factors, such as diabetes, obesity, and alcohol consumption.
-
Additional data regarding intersubject variation in pharmacodynamic differences are needed across life stages and in various subpopulations before pharmacodynamic factors can be quantitated in risk assessment. Before such pharmacodynamic data can be generated, the critical targets and modes of action must be clarified from animal or in vitro studies.
























