2
METHODOLOGY
This chapter articulates the committee’s approach to its task. Of the dozens of pathogens known to exist in southwest and south-central Asia, the committee identified the ones that are known to cause long-term adverse health outcomes and infected at least one US veteran who served in southwest or south-central Asia in the period 1991-December 2005. The committee then oversaw a formal, comprehensive literature review that identified about 1,200 peer-reviewed studies about the late complications and latent and chronic infections that might be associated with primary infection by each of the pathogens. Those studies constituted the evidence from which the committee drew conclusions about the relationship between each primary infection and specific long-term adverse health outcomes in humans. Finally, the committee ranked the strength of the relationships through the five-category system presented at the end of this chapter.
IDENTIFYING THE INFECTIOUS DISEASES TO STUDY
Geographic Boundaries
As required by law, the committee considered infectious diseases that might have afflicted US troops who served in the 1991 Gulf War (PL 105-277 and PL 105-368). Additionally, in response to a request by the Department of Veterans’ Affairs, the committee considered infectious diseases that might have afflicted US troops during Operation Enduring Freedom (OEF) or Operation Iraqi Freedom (OIF). Thus, the committee’s preliminary deliberations covered infectious diseases known to occur specifically in Iraq, Kuwait, and Afghanistan and in the geographic region that includes the Arabian Peninsula, Syria, Lebanon, Israel, Iran, Qatar, Pakistan, Tajikistan, Kyrgyzstan, Uzbekistan, and Turkmenistan (Figure 2.1). The term southwest and south-central Asia refers to that region throughout this report.
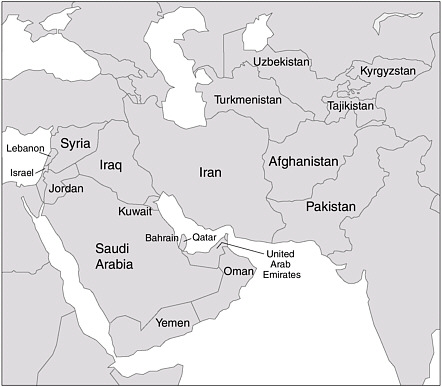
FIGURE 2.1 Southwest and South-Central Asia. The committee’s preliminary deliberations covered infectious diseases known to occur specifically in Iraq, Kuwait, and Afghanistan and in the geographic region that includes the Arabian Peninsula, Syria, Iran, Qatar, Pakistan, Tajikistan, Kyrgyzstan, Uzbekistan, and Turkmenistan.
SOURCE: The National Academies Press.
Infectious Diseases Endemic to Southwest and South-Central Asia That Have Long-Term Adverse Health Outcomes
The committee approached its task by first identifying infectious diseases that could have affected US troops deployed to southwest and south-central Asia. The committee members drew upon their collective knowledge of infectious diseases, which stems from both professional experience (Appendix A) and information gathered specifically for this study. The committee acquired information from numerous sources about illnesses diagnosed in troops deployed to southwest and south-central Asia, infectious diseases known to occur in that region, and conditions of special interest to veterans. The information came from peer-reviewed journal articles, surveillance and technical reports, presentations by physicians and scientists, and veterans and representatives of veterans’ groups.
Approximately 100 infectious diseases were identified for preliminary consideration (Table 2.1), including the four diseases specified in the legislation that directs the committee’s work (Box 2.1).
TABLE 2.1 Diseases and Etiologic Agents Considered by the Committee for Evaluation
|
Disease |
Etiologic Agent |
|
Bacterial diseases |
|
|
Acinetobacter infection |
Acinetobacter baumanii and other Acinetobacter species |
|
Actinomycosis |
Actinomyces spp. |
|
Anthrax |
Bacillus anthracis |
|
Bartonellosis |
Bartonella spp. |
|
Cat-scratch disease |
B. henselae |
|
Trench fever |
B. quintana |
|
Botulism |
Clostridium botulinum |
|
Brucellosis |
Brucella spp. |
|
Campylobacteriosis |
Campylobacter spp. |
|
Capnocytophaga infection |
Capnocytophaga spp. |
|
Chlamydia |
|
|
Genital infections |
Chlamydia trachomatis |
|
Pneumonia |
Chlamydia pneumoniae |
|
Cholera (including vibrio infections) |
Vibrio spp. |
|
Diphtheria |
Corynebacterium diphtheriae |
|
E. coli gastroenteritis |
Escherichia coli |
|
|
Enterotoxigenic E. coli |
|
|
Shiga toxin-producing E. coli |
|
|
Enteroaggregative E. coli |
|
|
Enteroinvasive E. coli |
|
|
Enterohemorrhagic E. coli |
|
|
Enteropathogenic E. coli |
|
Ehrlichioses |
Ehrlichia spp. |
|
Enteric fever |
|
|
Paratyphoid fever |
Salmonella enterica serovar Paratyphi A,B,C |
|
Typhoid fever |
Salmonella enterica serovar Typhi |
|
Enterococcal infection (vancomycin-resistant) |
Enterococcus spp. |
|
Gas gangrene |
Clostridium perfringens |
|
Hemophilus meningitis |
Haemophilus influenzae |
|
Helicobacter infection |
Helicobacter pylori |
|
Klebsiella infection |
Klebsiella spp. |
|
Legionnaire’s disease |
Legionella spp. |
|
Leptospirosis |
Leptospira spp. |
|
Listeriosis |
Listeria monocytogenes |
|
Lyme disease |
Borrelia burgdorferi |
|
Melioidosis |
Burkholderia pseudomallei |
|
Meningococcal infection |
Neisseria meningitidis |
|
Moraxella infection |
Moraxella catarrhalis |
|
Disease |
Etiologic Agent |
|
Mycoplasma infection |
Mycoplasma spp. |
|
Nocardiosis |
Nocardia spp. or aerobic actinomycetes |
|
Nontuberculous mycobacterial infection |
Mycobacteria spp. (except M. tuberculosis complex) |
|
Pasteurella infection |
Pasteurella spp. |
|
Pertussis (whooping cough) |
Bordetella pertussis |
|
Plague |
Yersinia pestis |
|
Plesiomonas shigelloides infection |
Plesiomonas shigelloides |
|
Pneumococcal disease |
Streptococcus pneumoniae |
|
Pseudomonas infection |
Pseudomonas aeruginosa |
|
Q fever |
Coxiella burnetii |
|
Rat bite fever |
Spirillum minus |
|
Relapsing fever |
Borrelia spp. |
|
Rickettsioses |
|
|
Boutonneuse fever |
Rickettsia conorii |
|
Louse-borne typhus |
Rickettsia prowazekii |
|
Marine typhus |
Rickettsia typhi |
|
Ehrlichiosis |
Ehrlichia chafeensis |
|
Anaplasmosis |
Anaplasma phagocytophilum |
|
Salmonellosis (non-typhoid) |
Salmonella spp. (except serovar Typhi) |
|
Shigellosis |
Shigella spp. |
|
Staphylococcal infection |
Staphylococci spp. |
|
Methicillin-resistant Staphylococcus aureus (MRSA) infection |
S. aureus (methicillin-resistant) |
|
Stenotrophomonas infection |
Stenotrophomonas maltophilia. |
|
Streptococcal infection (group A) |
Streptococcus pyogenes |
|
Tetanus |
Clostridium tetani |
|
Tuberculosis |
Mycobacterium tuberculosis |
|
Tularemia |
Francisella tularensis |
|
Yaws (nonvenereal treponemal infection) |
Treponema pertenue |
|
Yersiniosis |
Yersinia enterocolitica |
|
Fungal diseases |
|
|
Aspergillosis |
Aspergillus spp. |
|
Cryptococcus |
Cryptococcus spp. |
|
Histoplasmosis |
Histoplasmosis capsulatum |
|
Mucormycosis |
Fungi of the order Mucorales |
|
Helminthic diseases |
|
|
Ascariasis |
Ascaris lumbricoides |
|
Echinococcosis |
Echinococcus spp. |
|
Enterobiasis |
Enterobius vermicularis |
|
Filariasis |
Wuchereria bancrofti |
|
Hookworm disease |
Necator americanus and Ancylostoma duodenale |
|
Onchocerciasis |
Onchocerca volvulus |
|
Schistosomiasis |
Schistosoma mansoni and S. haematobium |
|
Disease |
Etiologic Agent |
|
Strongyloidiasis |
Strongyloides stercoralis |
|
Tapeworm disease (taeniasis) |
Taenia spp. and Diphyllobothrium latum |
|
Cysticercosis |
T. solium (Cysticercus cellulosae) |
|
Trichinosis |
Trichinella spiralis |
|
Trichuriasis |
Trichuris trichiura |
|
Protozoan diseases |
|
|
Amebiasis |
Entamoeba histolytica |
|
Cryptosporidiosis |
Cryptosporidium parvum |
|
Cyclosporiasis |
Cyclospora cayetanensis |
|
Giardiasis |
Giardia lamblia |
|
Isosporiasis |
Isospora bella |
|
Leishmaniasis |
Leishmania spp. |
|
Malaria |
Plasmodium spp. |
|
Microsporidiosis |
Microsporidia spp. |
|
Toxoplasmosis |
Toxoplasma gondii |
|
Viral diseases |
|
|
Adenoviral infection |
Adenovirus |
|
Avian influenza |
Influenza virus H5N1 |
|
Chickenpox (varicella) |
Human herpesvirus 3 (varicella-zoster virus) |
|
Crimean-Congo hemorrhagic fever |
Crimean-Congo hemorrhagic fever virus, genus Nairovirus |
|
Dengue |
Dengue virus, genus Flavivirus |
|
Dengue fever |
|
|
Dengue hemorrhagic fever, dengue shock syndrome |
|
|
Hantavirus hemorrhagic fever with renal syndrome and hantavirus pulmonary syndrome |
genus Hantavirus |
|
Influenza |
Influenza virus |
|
Rabies |
Rabies virus |
|
Retroviral diseases |
Human T-cell lymphotropic virus I (HTLV-I), HTLV-II, human immunodeficiency virus (HIV)-1 |
|
Rift Valley fever |
Rift Valley fever virus, genus Phlebovirus |
|
Sand fly fever |
Sand fly virus, genus Phlebovirus |
|
Sindbis virus disease |
Sindbis virus, genus Alphavirus |
|
Viral enteritis |
Various viruses |
|
Rotavirus infection |
group A Rotavirus |
|
Norovirus infection |
Norovirus |
|
Viral hepatitis |
Hepatitis viruses |
|
West Nile fever |
West Nile virus, genus Flavivirus |
|
Miscellaneous diseases |
|
|
Acute eosinophilic pneumonia |
Origin undetermined; not necessarily infectious |
|
Idiopathic enteropathy |
Origin undetermined; not necessarily infectious |
|
Madura foot (mycetoma) |
|
|
Actinomycetoma |
Various actinomycetes bacteria |
|
BOX 2.1 The Four Diseases Specified in PL 105-277 and PL 105-368 Leishmaniasis Sand fly fever Pathogenic Escherichia coli infection Shigellosis |
Though present in southwest or south-central Asia, some of the diseases on the committee’s preliminary list do not have long-term adverse health outcomes. The committee’s next step was to identify infectious diseases endemic in southwest and south-central Asia that have potential long-term adverse health outcomes, including secondary diseases or conditions (sequelae) caused by primary diseases, reactivation or recrudescence of diseases, and delayed presentation of diseases. Only diseases with known or possible long-term adverse health outcomes were selected from Table 2.1 for further evaluation.
The process began with the development of consensus on the meaning of long-term adverse health outcomes. Such health outcomes, the committee agreed, should have one or more of the following characteristics:
-
Substantial interruption of normal physical and mental functioning occurring outside the timeframe of acute infection.
-
Organ dysfunction or damage with a persistent effect.
-
Reproductive effects in military personnel, including birth defects in offspring of military personnel.
A long-term adverse health outcome may be reversible. The committee also considered the potential for secondary transmission of the pathogen.
The application of these criteria to the infectious diseases listed in Table 2.1 generated the infectious diseases contained in Box 2.2: infectious diseases that are endemic to southwest and south-central Asia and have long-term adverse health outcomes.
Direct Attribution to Military Service in Southwest and South-Central Asia
The committee examined the likelihood that the candidate infections would have occurred specifically during military deployment to southwest and south-central Asia during the
three operations in question. The risk of contracting a disease in the theater of operations must have been equal to or greater than the risk of contracting it in the United States. Moreover, given the natural history of the disease or infection, it must have been diagnosed in US troops in appropriate temporal relationship to deployment.
Chapter 4 comprises a review of infectious diseases that have been reported in US troops in close temporal relationship to the operations under study. On the basis of that review, the committee determined that many of the diseases in Box 2.2 have never been reported in US military personnel in close temporal relationship to deployment to southwest or south-central Asia during the Gulf War, OEF, or OIF. Nevertheless, it is impossible to prove that US troops did not contract any of the unreported diseases during deployment. Thus, the committee summarizes the acute and long-term characteristics of these unreported diseases in tabular form in Chapter 3 and excludes them from further analysis.
|
Bacterial diseases against which military personnel were immunized and for which vaccines are highly or fully protective Diphtheria Tetanus Bacterial diseases against which military personnel were immunized and for which vaccines are partly protective Meningococcal disease Pertussis (whooping cough) Bacterial diseases not more endemic in war theater than among US adult population Infections caused by Actinomyces Bartonella Capnocytophaga Chlamydia pneumoniae Clostridium botulinum Clostridium perfringens Francisella tularensis Legionella Listeria monocytogenes Moraxella catarrhalis Nocardia Non-cholera Vibrio Non-tuberculosis mycobacteria Pasteurella Plesiomonas shigelloides Staphylococcus Streptococcus Streptococcus pneumoniae Lyme disease Antibiotic-resistant or common nosocomial infections Infections caused by Acinetobacter (Multiple drug-resistant) Enterococcus (vancomycin-resistant) Klebsiella (multiple drug-resistant) Pseudomonas aeruginosa Staphylococcus aureus (methicillin-resistant) Stenotrophomonas maltophilia Of special concern to US troops Mycoplasma infection (atypical pneumonia) |
Of special concern to US troops Genital herpes Human immunodeficiency virus-1 Human T-cell lymphotropic virus infection (I) Human papillomavirus Viral disease not more endemic in war theater than among U.S. adult population Chickenpox (varicella) |
|
|
|
NOTE: The term infection refers to a primary infection that leads to disease. |
|||
Timing of Appearance of Long-Term Adverse Health Outcomes
Next, the committee determined the likelihood that a disease’s long-term adverse health outcomes would have been manifest and diagnosed during a person’s term of military service.
If the onset of adverse health outcomes typically occurs during the acute illness, the committee reasoned, the long-term effects would probably be detected, diagnosed, and reported during the patient’s term of military service. Likewise, diseases that have an acute phase— meaning that signs and symptoms become evident suddenly within hours or days of infection— are readily diagnosed during military service if the individual seeks medical care; in such cases, the physician would probably be cognizant of possible long-term adverse health outcomes. Finally, pre-existing conditions that were mild or latent before deployment but became reactivated or exacerbated during deployment also would probably be diagnosed during the patient’s military service. Therefore, infectious diseases with one or both of the following characteristics were not comprehensively evaluated in this study; instead, their acute and long-term health outcomes are summarized in tabular form in Chapter 3:
-
Onset of long-term adverse health outcomes typically occurs during the acute illness.
-
The disease is a preexisting infection that becomes reactivated or exacerbated during deployment.
In contrast, the committee determined, there are other infectious diseases whose long-term adverse health outcomes were unlikely to be diagnosed during military service in southwest and south-central Asia. Such diseases have the following characteristics:
-
The primary infection is subacute or the infected person is asymptomatic for days to years.
-
Long-term adverse health outcomes begin months to years after infection.
Because a subacute infection may go unnoticed and undiagnosed, the committee reasoned, the primary disease might not be reported. Furthermore, if long-term adverse health outcomes begin months or even years after infection, the condition would be more likely to be diagnosed after the person completes military service, and the diagnosing physician may not attribute the patient’s signs and symptoms to an infection acquired during deployment, particularly if the disease is uncommon in the United States, and thus more likely to be misdiagnosed. Infectious diseases that fit those criteria qualified for in-depth study.
The Infectious Diseases to Be Studied for Strength of Association with Long-Term Adverse Health Outcomes
The committee identified nine infectious diseases (Box 2.3) known to have long-term adverse health outcomes and to be potentially acquired by infected veterans during their military deployment to southwest and south-central Asia from 1991 to 2005. The committee conducted comprehensive reviews of the literature on the potential long-term adverse health outcomes of these diseases to determine the strength of association between the primary infection and the health outcome in humans.
|
BOX 2.3 Infectious Diseases with Long-Term Adverse Health Outcomes Studied for Strength of Association Brucellosis Campylobacteriosis Leishmaniasis Malaria Q fever Salmonellosis Shigellosis Tuberculosis West Nile fever |
Reasons for Excluding E. coli and Sand Fly Fever from In-Depth Study
Two of the infectious diseases named in PL 105-277 and PL 105-368—E. coli and sand fly fever—do not fulfill the above criteria for in-depth evaluation.
E. coli
Diarrheal infections were among of the most common ailments diagnosed in military personnel in regional theaters, and pathogenic E. coli is a well-recognized cause of diarrheal syndromes. The committee considered various infections related to pathogenic E. coli, focusing its attention on the role of pathogenic E. coli in diarrheal diseases. The unifying clinical syndrome associated with the various E. coli infections is a diarrheal illness that in healthy adults is usually transient without long-term adverse health outcomes. Therefore, the committee summarizes the health outcomes of E. coli infections in Chapter 3.
Sand Fly Fever
There are no published reports of sand fly fever in military personnel who served in the Gulf War; however, results from a search, requested by the Institute of Medicine (IOM), of a Department of Defense Gulf War hospitalization database identified five cases of this disease (the database and the search results are described in Chapter 4). As of December 2005, sand fly fever has not been found in military personnel serving in OIF and OEF. Sand fly fever is associated with a long-term adverse health outcome; however, the onset of the health outcome typically occurs during the acute illness. Therefore, the committee summarizes the health outcomes of sand fly fever in Chapter 3.
Comments on Diseases and Agents of Special Interest to Gulf War, OEF, and OIF Veterans
Several diseases and agents are of special interest to veterans of the Gulf War, OEF, and OIF. There is concern among Gulf War veterans that their symptoms might be connected to infection with Mycoplasma fermentans from contaminated vaccines (Nicolson et al. 2003) or exposure to biologic-warfare agents. In addition, during the Gulf War, troops stationed at Al Eskan Village, Saudi Arabia, developed respiratory illnesses at a high rate (Korenyi-Both et al. 1997; Korenyi-Both et al. 1992). The disease was termed Al Eskan disease and it has been hypothesized that a pathogen might be the cause. More recently, idiopathic acute eosinophilic pneumonia (IAEP) has been diagnosed in 18 military personnel serving in OIF or OEF (Shorr et al. 2004). IAEP is a syndrome characterized by febrile illness, diffuse pulmonary infiltrates, and pulmonary eosinophila (Allen et al. 1989; Badesch et al. 1989; Philit et al. 2002). There is also a
discussion of wound-associated infections (for example, infections caused by Acinetobacter baumannii) that appear to be more prevalent in OIF and OEF personnel than in civilian populations (Davis et al. 2005). Chapter 6 discusses Al Eskan disease, IAEP, wound-associated infections, mycoplasmas, and biologic-warfare agents.
REVIEW AND EVALUATION OF THE LITERATURE
Selection of the Literature
The committee adopted a policy of basing its conclusions primarily on peer-reviewed, published literature. Non-peer-reviewed publications provided additional information for the committee and raised issues that were researched further in the peer-reviewed literature.
Although the process of peer review by fellow professionals ensures high standards of quality, it does not guarantee the validity of a study or the generalizability of its results. Accordingly, committee members read each study critically and considered its relevance and quality.
Amassing the Literature
The committee oversaw a multistep process for amassing a robust collection of scientific literature about the long-term adverse health outcomes of the diseases listed in Box 2.3. The process began with a search of PubMed, a database created and managed by the National Library of Medicine that includes more than 15 million citations of biomedical publications from the 1950s to the present. The PubMed search focused on journal articles published through December 2005 that contain information about late complications, long-term sequelae, and latent infections related to the relevant infectious diseases and etiologic agents identified by the committee. Additional studies were identified from the reference lists of topical technical reports, textbooks, and other documents. Further PubMed searches identified pertinent articles on Al Eskan disease, IAEP, wound-associated infections, mycoplasmas, and biologic-warfare agents. Those initial searches generated about 20,000 articles.
Reviewing the Literature
On closer examination of that large body of literature, a subset of about 1,200 articles appeared to provide the types and quality of scientific evidence that the committee needed to accomplish its task. After securing the full text of those articles, the committee reviewed and assessed them for evidence of associations between primary infections by the etiologic agents of interest and specific long-term adverse health outcomes in humans.
The committee included several types of studies, including epidemiologic studies, case reports, and case series. Ideally, epidemiologic studies should have methodologic details, a control or reference group, reasonable adjustment for confounders, and statistical power to detect effects. Review articles, technical reports, and textbooks were used for background information.
The committee relied heavily on studies that focused on human adult populations. Because the IOM task concerns adults who may have become infected during military service, studies of children were generally excluded unless the studies dealt with reproductive outcomes. One exception is that studies of children were included if they provided information about adult
diseases; for example, a study on cerebral malaria in children might be reviewed if it provided information about the disease in adults.
CATEGORIES OF STRENGTH OF ASSOCIATION
The committee’s goal was to use the evidence in the medical and scientific literature to determine the relationships between the infectious diseases of interest and specific adverse health outcomes that might appear months to years after primary infections. Those relationships, presented in Chapter 5, are conceived in terms of the “strength of association” between a primary infection and a specific long-term adverse health outcome. The committee ranks strength of association qualitatively using a five-tier system, presented below in full.
Origin and Evolution of the Categories
A brief historical overview of the committee’s categories of association will elucidate their scientific roots. The International Agency for Research on Cancer (IARC), part of WHO, established criteria in 1971 to evaluate the human carcinogenic risk posed by chemicals (IARC 1998). First published in 1972, IARC’s evaluations are scientific, qualitative judgments by ad hoc working groups about the evidence of carcinogenicity or noncarcinogenicity provided by the available data. The working groups express their qualitative judgments in terms of five categories of the relative strength of the evidence that a substance or exposure is carcinogenic (IARC 1999a). Agencies in 57 countries use IARC’s published evaluations—a reflection of the widespread acceptance of the categorization scheme as it has been updated and applied to about 900 agents, mixtures, and exposures (IARC 1999b; IARC 2005).
In the early 1990s, an IOM committee adopted IARC’s categories in evaluating the adverse health outcomes of pertussis and rubella vaccines (IOM 1991). Later IOM committees used the categories, with some modifications, in evaluating the safety of childhood vaccines (IOM 1994a), the health outcomes of herbicides used in Vietnam (IOM 1994b; IOM 1996; IOM 1999; IOM 2001; IOM 2003b), and the relationship between indoor pollutants and asthma (IOM 2000a). The present committee’s predecessors also adapted and used the categories in evaluating the health effects of outcomes given to US troops and of chemical exposures that may have occurred during the Gulf War (IOM 2000b; IOM 2003a; IOM 2004; IOM 2005).
The five categories of strength of association used in this report are presented and defined below.
Sufficient Evidence of a Causal Relationship
Evidence from available studies is sufficient to conclude that there is a causal relationship between exposure to a specific agent and a specific health outcome in humans. The evidence includes supporting experimental data and fulfills the guidelines for sufficient evidence of an association (see next category). The association is biologically plausible, and the evidence satisfies several of the guidelines used to assess causality, such as strength of association, dose-response relationship, consistency of association, and a temporal relationship.
Sufficient Evidence of an Association
Evidence from available studies is sufficient to conclude that there is an association. A consistent association has been observed between exposure to a specific agent and a specific health outcome in several high-quality human studies in which chance and bias, including confounding, could be ruled out with reasonable confidence.
Limited or Suggestive Evidence of an Association
Evidence from available studies suggests an association between exposure to a specific agent and a specific health outcome, but the body of evidence is limited by the inability to rule out chance and bias, including confounding, with confidence. For example, at least one high-quality study that is sufficiently free of bias, including adequate control for confounding, reports an association, while other studies provide support for the association but are not sufficiently free of bias, including confounding. Alternatively, several studies of lesser quality are consistent in showing an association, and the results are probably not due to bias, including confounding.
Inadequate or Insufficient Evidence to Determine Whether an Association Exists
Evidence from available studies is of insufficient quantity, quality, or consistency to permit a conclusion regarding the existence of an association between exposure to a specific agent and a specific health outcome in humans.
Limited or Suggestive Evidence of No Association
Evidence from well-conducted studies is consistent in not showing an association between exposure of any magnitude to a specific agent and a specific health outcome in humans. A conclusion of no association is inevitably limited to the conditions, magnitudes of exposure, and length of observation in the available studies. The possibility of a very small increase in risk after the exposure studied cannot be excluded.
REFERENCES
Allen JN, Pacht ER, Gadek JE, Davis WB. 1989. Acute eosinophilic pneumonia as a reversible cause of noninfectious respiratory failure. New England Journal of Medicine 321(9):569-574.
Badesch DB, King TE Jr., Schwarz MI. 1989. Acute eosinophilic pneumonia: A hypersensitivity phenomenon? American Review of Respiratory Disease 139(1):249-252.
CDC (Centers for Disease Control and Prevention). 2005. Health Information for International Travel 2005-2006. Centers for Disease Control and Prevention. Atlanta, GA: US Department of Health and Human Services, Public Health Service.
Davis KA, Moran KA, McAllister CK, Gray PJ. 2005. Multidrug-resistant Acinetobacter extremity infections in soldiers. Emerging Infectious Diseases 11(8):1218-1224.
Heymann DL. 2004. Control of Communicable Diseases Manual. Washington, DC: American Public Health Association.
IARC (International Agency for Research on Cancer). 1998. Preamble to the IARC Monographs. [Online]. Available: http://www-cie.iarc.fr/monoeval/background.html [accessed July 19, 2005].
IARC. 1999a. Evaluation. Preamble to the IARC Monographs. [Online]. Available: http://www-cie.iarc.fr/monoeval/eval.html [accessed August 3, 2005].
IARC. 1999b. Objective and Scope. Preamble to the IARC Monographs. [Online]. Available: http://www-cie.iarc.fr/monoeval/objectives.html [accessed August 3, 2005].
IARC. 2005. Monographs on the Evaluation of Carcinogenic Risks to Humans. [Online]. Available: http://www-cie.iarc.fr/ [accessed August 3, 2005].
IOM (Institute of Medicine). 1991. Adverse Effects of Pertussis and Rubella Vaccines. Washington, DC: National Academy Press.
IOM. 1994a. Adverse Events Associated With Childhood Vaccines: Evidence Bearing on Causality. Washington, DC: National Academy Press.
IOM. 1994b. Veterans and Agent Orange: Health Effects of Herbicides Used in Vietnam. Washington, DC: National Academy Press.
IOM. 1996. Veterans and Agent Orange: Update 1996. Washington, DC: National Academy Press.
IOM. 1999. Veterans and Agent Orange: Update 1998. Washington, DC: National Academy Press.
IOM. 2000a. Clearing the Air: Asthma and Indoor Air Exposures. Washington, DC: National Academy Press.
IOM. 2000b. Gulf War and Health, Volume 1: Depleted Uranium, Sarin, Pyridostigmine Bromide, Vaccines. Washington, DC: National Academy Press.
IOM. 2001. Veterans and Agent Orange: Update 2000. Washington, DC: National Academy Press.
IOM. 2003a. Gulf War and Health, Volume 2: Insecticides and Solvents. Washington, DC: The National Academies Press.
IOM. 2003b. Veterans and Agent Orange: Update 2002. Washington, DC: The National Academies Press.
IOM. 2004. Gulf War and Health: Updated Literature Review of Sarin. Washington, DC: The National Academies Press.
IOM. 2005. Gulf War and Health, Volume 3: Fuels, Combustion Products, and Propellants. Washington, DC: The National Academies Press.
Korenyi-Both AL, Korenyi-Both AL, Molnar AC, Fidelus-Gort R. 1992. Al Eskan disease: Desert Storm pneumonitis. Military Medicine 157(9):452-462.
Korenyi-Both AL, Korenyi-Both AL, Juncer DJ. 1997. Al Eskan disease: Persian Gulf syndrome. Military Medicine 162(1):1-13.
Mandell GL, Bennett JE, Dolin R. 2005. Principles and Practice of Infectious Diseases. 6th ed. Philadelphia, PA: Elsevier Churchill Livingstone.
Nicolson GL, Nasralla MY, Nicolson NL, Haier J. 2003. High prevalence of mycoplasma infections in symptomatic (Chronic Fatigue Syndrome) family members of mycoplasma-positive Gulf War illness patients. Journal of the Chronic Fatigue Syndrome 11(2):21-36.
Philit F, Etienne-Mastroianni B, Parrot A, Guerin C, Robert D, Cordier JF. 2002. Idiopathic acute eosinophilic pneumonia: A study of 22 patients. American Journal of Respiratory and Critical Care Medicine 166(9):1235-1239.
Shorr AF, Scoville SL, Cersovsky SB, Shanks GD, Ockenhouse CF, Smoak BL, Carr WW, Petruccelli BP. 2004. Acute eosinophilic pneumonia among US Military personnel deployed in or near Iraq. Journal of the American Medical Association 292(24):2997-3005.
















