1
Introductory Session1
During the opening session of the workshop, two representatives of sponsoring agencies—Paul M. Coates from the U.S. government and Peter Fischer from the Canadian government—described the goals of the Dietary Reference Intake Research Synthesis project and its potential uses. Then John W. Erdman, Jr., described the approach and framework of the Dietary Reference Intakes (DRIs). Dr. Erdman served as a member, vice-chair, and then chair of the Standing Committee on Scientific Evaluation of Dietary Reference Intakes that oversaw the 1994–2004 DRI process.
OVERVIEW
Presenters: Paul M. Coates and Peter Fischer
Goals of the Project
The main goal for the workshop was a discussion of expert perspectives on the research needs that have been identified in eight DRI reports (see Box 1-1). Taking part in the discussion were many of those who worked on the DRI reports, including some former chairs of DRI panels, other former committee and panel members, and many other experts (see Appendix B).
|
BOX 1-1 Dietary Reference Intake Reports
|
A federal steering committee, which has both U.S. and Canadian participants, was actively involved in planning for this project. The Federal DRI Steering Committee represents a wide range of interest in the
DRIs. (See Appendix B for a listing of committee members and the agencies they represent.) Dr. Coates, a member of the steering committee and cochairperson of its research synthesis subcommittee, expressed strong commitment on the part of the relevant U.S. federal agencies for the process of developing DRIs.
Because the DRI reports contain an enormous amount of information about research recommendations and knowledge gaps, the steering committee called on the Food and Nutrition Board to gather the DRI research recommendations, synthesize them in a meaningful and useful database, and seek input from interested parties in completing the effort. This collaborative effort among federal agencies from the two countries and the Food and Nutrition Board was designed to facilitate the identification and response to important research needs. In turn, this effort will inform future studies of human nutrient requirements.
The steering committee’s goals for the project include the following:
-
Gather the research recommendations from the eight DRI reports
-
Synthesize those recommendations in a searchable database
-
Identify gaps in knowledge—including progress that has been made and the gaps that still need to be filled
-
Stimulate research to fill gaps
-
Assist sponsors in setting priorities
-
Alert the research community to the gaps and priorities
-
Make future DRI recommendations even more meaningful
Dr. Coates asked researchers and other users of the data contained in the DRI reports to provide the Federal DRI Steering Committee with suggestions for improving and making the most use out of the database of DRI research recommendations.
Canadian Involvement
Canada has been involved in the DRI process from its inception in the mid-1990s. In particular, Canada has contributed financial support, representatives of Canadian government agencies are participants in the Federal DRI Steering Committee, Canadian scientists have served on each of the DRI subcommittees and panels, and the Canadian government currently is funding the production of a summary volume of the
DRIs. This summary will be published in both French and English—the two official languages of Canada.
The DRIs have become the cornerstone of Canadian nutrition policies. Currently, they are being used in Canada for policy development, the setting of standards and regulations, conducting risk assessments, and providing advice to the Canadian population related to healthy eating. The Institute of Nutrition, Metabolism and Diabetes of the Canadian Institutes for Health Research is one of the sponsors of this workshop. This sponsorship demonstrates commitment to this effort by the major health research funding organization in Canada.
Several of the research gaps are of special concern to Canada. For example, information is required on how geographical variables affect the vitamin D needs of Canadians who have limited exposure to sunshine in the northern latitudes and on vitamin D status for various levels of intake throughout the life span. Moreover, additional information is needed on vitamin B12 requirements for the elderly and how these can be met. In Canada, currently, synthetic vitamin B12 is not approved for use in fortified foods. Thus, the only form of vitamin B12 that many elderly individuals can absorb is available only in the form of supplements. In developing policies on the fortification of foods, Canada relies on the DRIs for children for modeling purposes, but there are huge information gaps related to the nutrient requirements of children.
APPROACH AND FRAMEWORK OF THE DIETARY REFERENCE INTAKES
Presenter: John W. Erdman, Jr.
A Brief History of Recommended Dietary Allowances
The development and publication of nutrient recommendations in the United States officially began in the early 1940s with the first edition of Recommended Dietary Allowances (NRC, 1943). That first report, which was requested by the U.S. Department of Defense, yielded just 10 dietary recommendations: those for energy, protein, 2 minerals, and 6 vitamins. The intent of Recommended Dietary Allowances (RDAs) was to ensure adequate nutrition, especially protection from deficiency diseases, and to set standards for public health programs. Two major avenues provided
the scientific basis of the RDAs: (1) observation of usual patterns that lead to healthy living, and (2) experimentally determined data on nutrient requirements.
The 10th (and final) edition of Recommended Dietary Allowances was published in 1989 (NRC, 1989b). By then, the scientific information base had expanded enough that the report contained recommendations for 7 minerals and 11 vitamins, in addition to protein and energy. In that report, Safe and Adequate Daily Dietary Intakes rather than RDAs were developed for an additional 2 vitamins and 5 minerals. These new nutrient values for 7 nutrients were ranges of the nutrients that were considered safe and adequate.
The users of RDAs included government, industry, academia, and health services. The uses of the RDAs, some of which were appropriate and some of which were not, included the following:
-
Guide for procuring food supplies for groups of healthy persons
-
Basis for planning meals for groups
-
Reference point for evaluating the dietary intake of population subgroups
-
Component of food and nutrition education programs
-
Reference point for the nutrition labeling of food and dietary supplements
Development of a New Framework
In 1989, the National Research Council (NRC) released Diet and Health (NRC, 1989a), which focused on nutrients and dietary patterns as related to the reduction of chronic disease risk. Then, in the early 1990s, the Food and Nutrition Board addressed a number of concerns and issues related to establishing nutrient requirements and recommendations. Among these were
-
a history of a lack of transparency for the process for establishing recommendations,
-
little published information on how the recommended values were determined,
-
new information on food components that had not been identified as nutrients (e.g., fiber and carotenoids),
-
an evolving concern about nutrients’ roles in protection against chronic disease,
-
a number of new statistical approaches for examining data, and
-
the use of the RDAs for some purposes that were considered to be inappropriate.
As a result, in 1993, the Food and Nutrition Board held a workshop called “How Should the Recommended Dietary Allowances Be Revised?” A report of the workshop was published the next year (IOM, 1994). That report proposed a new framework—the DRI framework. The framework included concepts of reduction of the risk of chronic diseases, a review of other dietary components, documentation of the rationale for the end points used, and recommendations to meet a variety of uses. It also included estimation of an upper intake level. Interest in setting upper levels grew, in part, because the new approach might increase recommendations for certain nutrients to alleviate chronic diseases, which, in turn, might move the intake values to a level that would be close to the upper level for one or more nutrients. Following release of the report of the workshop, the Food and Nutrition Board provided additional opportunities for input from scientists and users of nutrient recommendations.
Organizational Structure
The Food and Nutrition Board developed an organizational structure to carry out the DRI process, as shown in Figure 1-1. A standing committee oversaw the process, chaired for nearly seven years by the late Dr. Vernon Young. Two subcommittees were appointed to focus on specific aspects of the task—safe upper levels and applications of the DRIs. The Subcommittee on Upper Reference Levels of Nutrients provided input to the panels regarding safety and upper levels of intake, and the Subcommittee on Interpretation and Use of DRIs provided input to each panel concerning how to apply the DRIs for the nutrient group. Reflecting the joint nature of the project in the United States and Canada, Canadian and American scientists served on the committee, subcommittees, and panels.
Over time, six panels were appointed to address groups of nutrients. To some degree, nutrients were grouped together because of their relationship to chronic disease end points.
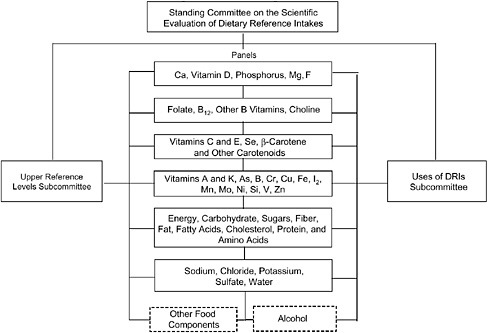
FIGURE 1-1 Organizational structure for the DRI process.
NOTE: Ca = calcium; Mg = magnesium; F = fluoride; Se = selenium; As = arsenic; B = Boron; Cr = chromium; Cu = copper; Fe = iron; I = iodine; Mn = manganese; Mo = molybdenum; Ni = nickel; Si = silicon; V = vanadium; Zn = zinc.
Types of DRI Values
The reports use four different types of DRI values:
-
Estimated Average Requirement (EAR)
-
Recommended Dietary Allowance (RDA)
-
Adequate Intake (AI)
-
Tolerable Upper Intake Level (UL)
With publication of the report on macronutrients, the Acceptable Micronutrient Distribution Range (AMDR) was added.
In Figure 1-2, three types of DRIs are placed on a hypothetical curve that depicts effects of increasing the observed level of intake. With a low level of intake, 100 percent of the population is deficient, based on some
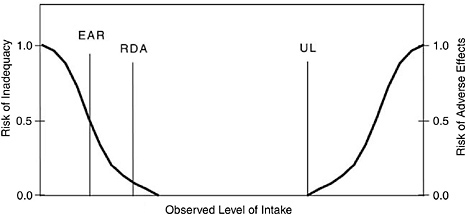
FIGURE 1-2 Relative positions of selected Dietary Reference Intake values on a curve showing intake versus risk.
NOTE: EAR = Estimated Average Requirement; RDA = Recommended Dietary Allowance; UL = Tolerable Upper Intake Level.
end-point criterion. With increasing intake, a point presumably would be reached at which 100 percent of the healthy population would have no deficiency or inadequacy based on that same end point.
The EAR is at the midpoint, where 50 percent of the population would meet the end-point criterion. The RDA, calculated as two standard deviations above the EAR,2 is the intake level at which, theoretically, 97.5 percent of the population would meet that end-point criterion. AIs are set when data are insufficient to establish an EAR. The AI for a nutrient is a recommended average daily intake level based on observed or experimentally determined approximations or estimates of nutrient intake by a group (or groups) of apparently healthy people and is assumed to be adequate.
Notably, a chronic disease end point was used as the basis for the EAR or AI only for fluoride, potassium, total fiber, and for the AMDRs. Dr. Erdman commented that in 1993 and 1994, it was anticipated that a larger number of nutrients would use chronic disease end points. A chronic disease end point was used for the UL for sodium and chloride.
For some nutrients, ULs were set. ULs are defined as the highest level of daily nutrient intake that is likely to pose no risk of adverse health effects for almost all individuals in the specified life-stage group.
The adverse effect identified as a basis for a UL ranged from diarrhea to much more serious conditions.
The AMDRs include ranges of intakes of energy nutrients (protein, carbohydrates, and fats). These are ranges of macronutrient intakes that are associated with reduced risk of chronic disease while providing adequate intakes of essential nutrients for a given age and gender group. For adults, for example, the AMDR for fat is 20 to 35 percent of energy; for carbohydrate, it is 45 to 65 percent; and for protein it is 10 to 35 percent.
The DRI Reports
Based on the work of these committees and panels, the Institute of Medicine has published eight major DRI reports, as shown earlier in Box 1-1. For each report, the box also includes an abbreviated title; these abbreviated titles are used for convenience in the remainder of this workshop summary.
Closing
To close the introductory session, Dr. Erdman expressed appreciation for the broad support that has made it possible to produce the DRI reports. He also highlighted the upcoming single volume summary of the DRI reports, Dietary Reference Intakes: The Essential Guide to Nutrient Requirements (IOM, 2006a), and extended thanks to Canada for making the summary report possible. The format of the summary report is based on input from the intended audience—dietitians, nutritionists, and other health professionals—and the report is expected to be very useful for practitioners and those who teach them.










