5
Tier 1 International Agent-Only Processing Technologies
INTRODUCTION
In addition to the detonation-type technologies for munitions processing described in Chapter 4, the committee recognized that certain other types of processes could be used to destroy chemical agents, if not entire chemical munitions. The Non-Stockpile Chemical Materiel Project (NSCMP) already employs equipment that is based on one of these agent-only technologies. The rapid response system (RRS) and the single container agent neutralization system, described in Chapter 1, are good examples. These systems do not destroy complete munitions but use neutralization to destroy small amounts of agent contained in chemical agent identification sets. Several other technologies for agent-only destruction were identified and investigated. Of these, two(1) the Russian two-stage process of neutralization with addition of bitumen and (2) incinerationwere designated as Tier 1 technologies for agent-only destruction. These technologies were investigated more fully, and are described and evaluated in this chapter.
The Russian two-stage process employs neutralization (hydrolysis) as a first stage. In a second stage, the product of the neutralization step is added to bitumen, yielding a stable bitumen-salt product that can be landfilled or stored long term. In the United States, neither the non-stockpile program nor the stockpile program for chemical demilitarization employ technology similar to the Russian two-stage process.
On the other hand, incineration is employed on a large scale in the stockpile program. The U.S. Army’s baseline incineration system includes a process for reverse assembly (disassembly) of the stored munitions and collection of the contained agent. As such, it constitutes a complete munition processing system for munitions that have been properly stored. Agent drained from these munitions is sent to a liquid incinerator for destruction, while energetics and munition bodies are sent to separate furnaces for destruction or decontamination. This baseline system is now in use at four stockpile sites in the United States, where a variety of obsolete munition types containing nerve and mustard agents have been stored.
However, because of the poor condition of recovered non-stockpile items in general, it is not practical to employ a reverse assembly process for the munitions dealt with in non-stockpile operations. On the other hand, the agent destruction part of the baseline incineration process is applicable to recovered bulk agent or to agent that has been recovered from munitions. Three European countries currently employ incineration to destroy agent recovered from old and abandoned chemical weapons. However, the agent recovery methods used are considered out of date, and their future use is not recommended.
The committee designated additional technologies that destroy only agent, including acid digestion, biological treatment, the Defence Science and Technology Laboratory (DSTL) electric furnace, electrochemical treatment, photo-catalysis, and plasma arc technology, as Tier 2 technologies. They were investigated to a lesser extent and are covered in Chapter 6.
In this chapter, the committee briefly reviews some other applications of neutralization and hydrolysis elsewhere in the world to provide historical perspective for the technology being used in the United States to destroy recovered chemical weapons, as described in Chapter 1. Detailed descriptions and evaluations of the Russian two-stage neutralization/ bitumenization process and incineration processes follow that review.
USE OF NEUTRALIZATION AND HYDROLYSIS IN THE REST OF THE WORLD
Neutralization and hydrolysis1 are accepted and widely used technologies that have been used to treat a variety
|
1 |
As previously noted in Chapter 1, this report employs “neutralization” for the use of an organic reagent to destroy agent and “hydrolysis” for the use of an aqueous reagent to destroy agent. Neutralization and hydrolysis are often used interchangeably in the literature on chemical agent demilitarization. Hydrolysis is the more appropriate term from a chemical process perspective. Neutralization is more in keeping with the notion of “to neutralize and thereby render innocuous.” It may be found in the literature to refer to hydrolysis in aqueous or nonaqueous media. |
of chemical agents. Their use in the United States is summarized in Chapter 1. Other countries have also employed neutralization and hydrolysis. This brief review examines some instances where this technology has been used and describes some of the technical variations in how it has been applied.
Hydrolysis was used for the destruction of agent production facilities and chemical weapons in Iraq from 1991 to 1994 (Manley, 1997). Iraq had various weapons containing the nerve agents GA, GB, and a mixture of GF (O-cyclohexyl methylphosphonofluoridate) and GB. The latter mixture also contained up to 40 percent dichloromethane. First, all energetics were removed from the weapons. Several techniques were then employed to remove agent from the weapons, including removal of the filling plug when possible, drilling holes, and using shaped explosive charges to create holes. The drained weapons were rinsed, again using various techniques, and then burned. A former nerve agent production plant was converted into a hydrolysis facility, with a glass-lined, 3 cubic meter reactor used to carry out hydrolysis in batchwise fashion. A 15 weight percent solution of aqueous sodium hydroxide was used to treat 300-350 liters of agent per batch. Use of higher concentration caustic resulted in blockages when emptying the reactor. The reaction was considered complete when the agent concentration dropped below the detection limit of 1 ppm and the caustic concentration remained constant for two successive measurements. The hydrolysis products were pumped to a concrete-lined lagoon and allowed to evaporate to dryness. The dry residue was mixed with concrete and entombed in more concrete. The plant capacity was 1-1.5 metric tons per day. About 70 metric tons of GB and GB/GF mixture were destroyed. The same plant was used to destroy the GA recovered from 75 metric tons of impure product. The resulting hydrolysis product contained sodium cyanide and was placed in steel tanks that were sealed into large concrete storage bunkers.
Pearson and Magee (2002) state that neutralization is used at the Belgian chemical weapon dismantlement facility at Poelkapelle to destroy some of the approximately 60 combinations of agents used in World War I. The hydrolysis products are sent to the INDAVER commercial hazardous waste incinerator near Antwerp. Mustard agent is neutralized using an aqueous ethanolic caustic solution. Diphosgene, which sometimes contains phosgene in solution, and chloropicrin are treated with aqueous sodium hydroxide solution. Other agents are dissolved in butanol and incinerated without hydrolysis, although neutralization techniques for these were being investigated.
Hydrolysis with aqueous sodium hydroxide was used to destroy mustard agent, lewisite, and a mixture of mustard agent and lewisite contained in unearthed beer bottles in Samukawa, Japan (Kawataki et al., 2005). Several hundred bottles were involved. The tops were removed from the bottles, and the agent was removed and filtered. The agent was then fed to a stirred reactor containing caustic. The product from this first reactor was fed to a second reactor that contained an unspecified oxidation reagent. The product from the second reactor was analyzed to ensure that agent concentrations were below the threshold value of 0.05 mg/L for HD and 0.1 mg/L for lewisite. The liquid effluent was then passed to an industrial waste contractor; the ultimate fate was not reported. Some bottles contained solid wastes. These were treated to a U.S. 5X standard for decontamination by heating in an electric furnace.2 The electric furnace was also used to decontaminate the empty bottles and other solid waste to a 5X condition. Exhaust gases from both operations were treated before discharge using caustic scrubbing and adsorption on activated carbon.
A Russian procedure for destroying mustard agent/ lewisite mixtures and lewisite itself by caustic hydrolysis appears to be similar to that used at Samukawa. According to a summary in RIA Novosti, the destruction of the 241 tons of lewisite stored at Gorny was expected to be completed in December 2005 (Litovkin, 2005). A lewisite neutralization facility being built with German assistance at Kambarka was to go online in December 2005. There are 6,349 tons of lewisite stored there. The hydrolysate will be incinerated (GSN, 2005). It appears that the Gorny facility served as a pilot plant to demonstrate the technology.
Over 700 tons of mustard agent were destroyed by hydrolysis in Canada (Sutherland, 1997). The destruction was carried out at the Defence Research Establishment Suffield (DRES) in 1975-1976. Alkaline hydrolysis was employed and the products were incinerated. From 1989 to 1991, nerve agents (G and V classes) were removed from weapons using drill-and-drain methods and neutralized using 20 percent potassium hydroxide in methanol. The neutralization products were then incinerated. Lewisite was neutralized with an alkaline peroxide solution containing sodium or calcium chloride. The products included sodium or calcium arsenate, acetylene, and chloride salts.
The article by Sutherland also mentions the use in Russia of a combination of neutralization and incineration. The nerve agents GB and GD, and also mustard agent, were reacted with ethanolamine, and the products of these reactions were subsequently incinerated. VX was reacted with a 1:1 mixture of ethylene glycol and orthophosphoric acid, again with the products sent for incineration. Some 300,000 kg of toxic substances were destroyed in a facility that was too small to be considered industrial-scale.
A neutralization process using hydrogen peroxide to destroy agent is one of the technologies currently being used in Civitavecchia, Italy, at the NBC Joint Technical Logistics Center, which is known by its acronym, Ce.T.L.I. NBC. Demilitarization activities are focused on stockpiled munitions containing primarily Clark I agent but also a mixture of mustard agent and Clark I, chloropicrin, or adamsite (DM).3
An automated plant that went into full operation in 2003 has destroyed thousands of rounds ranging in size from 65 mm to 155 mm and larger. Rounds are identified using high-energy x-ray analyses (by means of a linear accelerator-type x-ray source capable of penetrating 10 cm of steel). The liquid agents are frozen in the bottom of the round by chilling the round in an upright position. The round is opened by unscrewing, drilling, or cutting; after the agent is liquefied by warming to ambient temperature, it is emptied into a container for neutralization. The emptied round is cleaned with concentrated nitric acid and bleach. The plant destroys 64 projectiles in an 8-hour work day and uses antiaerosol and activated carbon filters to mitigate air emissions.
The Clark I and the blister agent mixtures are destroyed using hydrogen peroxide, producing an acidic, arsenic-containing solution, which is neutralized with lime. In contrast, adamsite (in the form of hard, solid cylinders) is milled to form a powder. The products of blister agent neutralization and adamsite milling are mixed with sand, cement, and water. The mixtures are placed in containers made of reinforced concrete, and the resulting blocks are stored outside on platforms. The rainwater that collects below is checked periodically for the presence of arsenic.
Munitions recovered from excavation or retrieved from the sea are also destroyed at Civitavecchia if they are judged to have sufficient integrity to be safely transported. Badly damaged rounds that cannot be transported to Ce.T.L.I. NBC are handled on-site. A render-safe procedure is performed with a small shaped charge. Upon detonation, the shaped charge removes the fuze without initiating the burster and also opens an access hole for the agent fill. Technicians manually remove the agent from the munition and transport it to Civitavecchia for neutralization.
RUSSIAN TWO-STAGE PROCESS: NEUTRALIZATION WITH ADDITION OF BITUMEN
Description
The focus in Russia in recent years has been on a significantly different approach that involves neutralization and addition of the neutralent to bitumen to form a stabilized mass that can be safely stored in drums in a vault for some long period of time or landfilled.4 That approach is the focus of this section.
The chemical procedures involved have been developed for mustard agent, VX, VR (the Russian version of VX), GB, and GD. This technology was developed at the GOSNIIOCHT research institute in Russia (Krotovich, 1998). The procedures for VR, VX, GB, and GD were evaluated by a joint Russian/U.S. team (Bechtel, 1996).5 The joint evaluation involved experimental studies carried out at the Saratov Higher Military Engineering School of Chemical Defense in Russia and at the U.S. Chemical-Biological Defense Command in Edgewood, Maryland. At both sites, Russian and U.S. scientists worked together to carry out the chemical procedures involved, analyze the products of the reactions, and measure the toxicity of the final product.
VR is neutralized in Stage 1 of the process with a decontamination solution known as RD-4 (Pearson and Magee, 2002). This solution contains potassium isobutylate dissolved in isobutanol and N-methylpyrrolidinone. The VR and the RD-4 are reacted at atmospheric pressure at 90C-95C for 30 minutes to break the P-S bond, which causes the high toxicity of VR, and form diisobutyl methyl phosphonate and other products (see Figure 5-1).
In the second stage of the process, the reaction mass is added to bitumen at a starting temperature of 130C-140C that is subsequently increased to 180C under reduced pressure. This results in the formation of low volatility, low toxicity phosphorus-containing compounds and the vaporization of isobutanol and N-methylpyrrolidinone. The vapor stream is condensed to form a distillate that contains isobutanol and N-methylpyrrolidinone. Upon cooling, a solid bitumen-salt end product is formed. The joint evalu-
FIGURE 5-1 Reaction of Russian VX and potassium isobutylate. SOURCE: Bechtel, 1996.
FIGURE 5-2 Reaction of VX and potassium isobutylate. SOURCE: Bechtel, 1996.
ation report does not specifically indicate the fate of the diisobutyl methyl phosphonate in Stage 2 but does state that the phosphorus-containing products are tightly bound in the bitumen and would be impossible (or at the very least extremely difficult) to remove, thereby satisfying the CWC requirement for irreversible destruction.
VX is also reacted with RD-4 under similar conditions, again rupturing the P-S bond. The stage 1 reaction is shown in Figure 5-2. The Stage 2 bitumen addition is then carried out in the same fashion as for VR.
The destruction efficiencies (DEs) reported by Flamm and Pakhomov were not defined but were given as 99.9 percent
FIGURE 5-3 Notional reaction scheme for the addition of G-type agent to aqueous monoethanolamine (MEA). SOURCE: Bechtel, 1996.
for VR and VX for Stage 1 only and >99.999 percent for the overall process (Bechtel, 1996). VR and VX concentrations in the bitumen-salt end product were consistently below the gas chromatography detection limit of 1 × 10–4 mg/ml.
The joint evaluation report gives the results of toxicity testing on the final bitumen-salt products from the destruction of VR and VX. The measurement involved intragastric administration to rats, and the resulting LD50 was >5 × 103 mg/kg. This is a very low toxicity, placing the material in the Russian State category “slightly dangerous,” the same category as that of hexane, the solvent used in preparing the material administered to the rats.
About 3 kg of bitumen-salt product is produced for each kilogram of VX destroyed. About 1 kg of distillate is produced for each 2 kg of bitumen-salt product. The toxicity of the distillate was not measured.
Flamm and Pakhomov also describe the destruction of GB (sarin) and GD (soman) (Bechtel, 1996). The GB or GD is dissolved in industrial-grade ethanolamine containing about 20 percent water. The mixture is heated to 110C for 1 hour at atmospheric pressure (Figure 5-3). The reaction thus breaks the P-F bond, which is what causes the high toxicity of the G compounds. The products from this reaction are added to calcium hydroxide and bitumen, then heated to about 200C under reduced pressure for 1 hour. This treatment is expected to remove any last traces of GB and GD. Alcohols produced in the reactions and excess ethanolamine are distilled from the bitumenization mixture. The bitumen-salt mixture is drained from the reactor and allowed to solidify. About 5-7 kg of bitumen-salt product is produced for each kilogram of GB or GD destroyed. About 1 kg of distillate is produced for each 3 kg of bitumen-salt product. The toxicity of the distillate was not measured.
Again, the DEs reported by Flamm and Pakhomov were not defined but were given as 99.9 percent for GB and GD for Stage 1 only and >99.999 percent for the overall process (Bechtel, 1996). The GB and GD concentrations in the bitumen-salt product were generally below the gas chromatography limit of detection of 1 10–4 mg/ml.
The joint evaluation report gives the results of toxicity testing on the final bitumen-salt products from the destruction of GB and GD. The toxicity was again measured in rats, with LD50 results of >5 × 103 mg/kg. This is a low level of toxicity and puts the material in the Russian State category Level III, which makes it as toxic as the monoethanolamine solvent.
The Russian and U.S. scientists involved in the joint evaluation project concluded that destruction efficiencies were acceptably high, the reactions involved were irreversible, and the toxicity of the final product, the bitumen-salt product, was acceptably low. They also concluded that the distillates produced contained materials that could be recovered or recycled, thus reducing their contribution to overall waste production (Bechtel, 1996).
Although not presented in the joint evaluation report, reaction conditions have also been developed for the destruction of mustard agent. Mustard agent is preferably destroyed by neutralization with an equal weight of a reagent that is a 9:1 mixture of monoethanolamine and ethylene glycol (Pearson and Magee, 2002). The reaction proceeds for 1 hour at 100C-110C. The reaction mass is added to a “tough road bitumen” having a softening temperature of 65C-70C, with
calcium hydroxide added. The mixture is held at 170C for 45 minutes. This produces a bitumen mass with a softening temperature of 81.5C, enabling the mass to be formed into monolithic blocks that can be buried. Slightly different conditions are given by Krotovich (1998).
Country-by-Country Experience
Russia
A facility that will use neutralization and addition of the neutralent to bitumen technology is being built at Shchuch’ye in Russia (Ryan and McNelly, 2003). Construction was begun in March 2003. This facility will be used for destruction of much of the nerve agent now stored in Russia. Nerve agent (organophosphorous compounds) is said to account for 79.2 percent of Russia’s total chemical agent stockpile of about 40,000 tons (Petrov and Trubachev, 2000). Weapons and Stage 1 product will be shipped to Shchuch’ye from other sites. The plant is being constructed in stages. A pilot facility with a capacity of 500 metric tons per year will be built first. This facility will then be expanded to its full capacity of 1,200 metric tons per year.
The Russians had once planned to use the monoethanolamine and ethylene glycol process for destruction of their stockpile of 622 tons of mustard agent stored at Gorny (Saratov Oblast). They had planned to operate two process lines at Gorny: one for straight mustard agent using mono-ethanolamine and ethylene glycol, the other using aqueous caustic to destroy lewisite and mustard agent/lewisite mixtures (see above). To save money and to meet the CWC 1 percent destruction deadline, they used the lewisite line to destroy the mustard agent (Ember, 2003). The project was completed on November 14, 2003. They are now storing the 1,260 tons of hydrolysate in drums. It appears that the two-stage monoethanolamine/ethylene glycol neutralization followed by bitumenization will not be used in Russia for mustard agent because the country apparently has no more mustard agent apart from mustard agent/lewisite mixtures. Russia does not intend to employ the technology for destruction of arsenic-containing agents.
Evaluation Factors Analysis
Process Maturity
As noted above, the mustard agent version of the process of neutralization and addition of the neutralent to bitumen will not be used in Russia on a production scale. The nerve agent versions of the technology have not been used on a large scale but will be in the future. All three versions were tested extensively at the GOSNIIOCHT research institute.
Process Efficacy/Throughput
The process appears to completely destroy nerve and mustard agents. The Russians do not plan to apply the technology to arsenic-containing agents. As indicated above, they obtained nearly complete destruction of agent, generally down to a limit of detection of 1 10–4 mg/ml and a DE of >99.999 percent.
Process Safety
Pearson and Magee (2002) point out that the first step, the neutralization, is carried out at about 100C under atmospheric pressure and using monoethanolamine or the RD-4 solution; this step does not present any unusual safety hazards. The second step, bitumenization, is carried out at higher, but still modest, temperatures and under reduced pressure; it also presents no unusual safety hazards.
Public and Regulatory Acceptability in a U.S. Context
Neutralization processes to treat CWM have routinely achieved public and regulatory acceptability in the United States. As described in Chapter 1, the U.S. NSCMP’s explosive destruction system (EDS), RRS, and single container agent neutralization system employ neutralization to destroy recovered agent. Large-scale neutralization projects for stockpiled agent have been or will be carried out at the Newport, Pueblo, Aberdeen, and Blue Grass sites. While the method of disposing of the neutralent/hydrolysate can be a significant issue, the use of neutralization as the first step of the agent destruction project is generally readily accepted.
Secondary Waste Issues
A single significant waste, the bitumen-salt product, is produced by the Russian two-stage process. For VR destruction, the volume is about three times that of the starting VR. For GB and GD, the volume is four to seven times that of the starting agent. In both cases, this waste contains methylphosphonate products, which might leach from the waste over time. This possibility might impact disposal decisions. Petrov and Trubachev (2000) express concern about the possible eventual degradation of the bitumen-salt product and mention alternatives to the bitumenization step. As indicated above, the bitumen-salt products are not very toxic. No information was found on leaching as measured, for example, by the toxicity characteristic leaching procedure test that is used for compliance with RCRA regulations, and such testing would be needed before the product could be landfilled in the United States. Radilov (2004) presents toxicity data and then recommends that certain “sociohygienic” monitoring be done near destruction facilities in Russia. The bitumen-salt product will apparently be placed in permanent storage, although the literature is not entirely clear on this subject.
An earlier report (Khripunov and Parshall, 1999) indicates that the product will be placed in steel drums in aboveground vaults. A later reference (Ember, 2005) states only that the product will be placed in concrete vaults (polygons). The area is marshy, and the integrity of the storage facility is a concern. Clarification of the ultimate disposition of the bitumen-salt product is needed.
Process Costs
Some cost information for the Shchuch’ye facility is provided by Ryan and McNelly (2003). The United States has committed to funding the facility. The expected cost of construction, systemization, training, and start-up is given as $888 million. This cost appears to be very modest for a major facility. The area around Shchuch’ye is poor and lacking in infrastructure, such as regional roadways, water supply, gas supply, a hospital, and communications facilities.6 Russia and other countries, but not the United States, are funding the construction of this infrastructure. That cost is not included in the $888 million figure.
INCINERATION
Description
Incineration is the high-temperature oxidation of organic compounds. The carbon and hydrogen in the compounds are ideally converted completely to carbon dioxide and water. The chemical agents at issue here are organic compounds that also contain elements such as fluorine, chlorine, phosphorus, nitrogen, sulfur, and metals such as arsenic. This can result in the formation of inorganic compounds such as hydrogen fluoride, hydrogen chloride, phosphorus pentoxide, nitrogen dioxide or other nitrogen oxides, sulfur dioxide, and metal arsenic oxides during the incineration process (Pearson and Magee, 2002). Methods exist for the removal of these materials from the offgases from the process.
One issue with incineration technology is that organic compounds cannot generally be converted completely to carbon dioxide and water. Instead, trace amounts of compounds such as dioxins and furans, as well as other products of incomplete combustion, can be generated during the combustion process and must be controlled by scrubbing and cleaning the offgas using, for example, activated carbon adsorption. Even so, this characteristic of incineration processes has sometimes made it difficult for this technology to gain public acceptance, especially from stakeholders in local communities and environmental interest groups.
Incineration has been used for the destruction of both agents and various types of secondary waste. This report looks primarily at the destruction of agents. The U.S. Army and its contractors have developed very advanced and sophisticated incineration technology for the destruction of the U.S. chemical weapons stockpile. These are high-capacity, fixed facilities designed for destroying munitions in good condition. In general, they are not useful for the destruction of non-stockpile chemical weapons because the deteriorated condition of the weapons will not allow for their disassembly with the installed equipment. Moreover, these facilities are not cost-effective for destroying small quantities of munitions, nor are they readily adaptable for moving from site to site.
The baseline incineration system is shown schematically in Figure 5-4. It includes (1) the reverse assembly of munitions to separate agents, energetics, metal parts, and dunnage; (2) high-temperature combustion of agent in a liquid incinerator; (3) combustion of energetics in a deactivation furnace; and (4) decontamination of metal parts in a metal parts furnace. These three furnaces have secondary combustion chambers to promote complete oxidation of organic compounds. Uncontaminated dunnage is generally disposed of offsite. Each of the three furnaces has elaborate air pollution control systems to remove gaseous pollutants and particles. The unit operations in these systems include caustic scrubbers for acid gas removal, quench towers for gas cooling, venturi scrubbers for particulate removal, fabric filters for elimination of fine particulates and mist, induced draft fans for movement of the exhaust gases, and activated carbon adsorbers for final removal of organic compounds.
Country-by-Country Experience
United States
Before the baseline incineration system had been developed for destruction of the U.S. chemical weapon stockpile, incineration was used to destroy 2,800 metric tons (100 percent agent basis) of chemical weapons, corresponding to 60,000 munitions at the Rocky Mountain Arsenal and the Chemical Agent Munitions Disposal System at the Tooele Army Ammunition Plant, Utah (Sutherland, 1997). The latter installation was used primarily to develop the baseline process, which is now being used to destroy the U.S. stockpile of chemical agents and munitions at four of the eight storage sites in the continental United States. The first completely integrated baseline system was built on Johnston Atoll in the Pacific Ocean. It was successfully used to destroy the stockpiled munitions at that site and has now been closed. The four sites currently in operation are located at Tooele, Utah; Umatilla, Oregon; Anniston, Alabama; and Pine Bluff, Arkansas.
Germany
The German treatment facility at Munster is described by Martens (1997) and by Mihm (2000). It was toured by
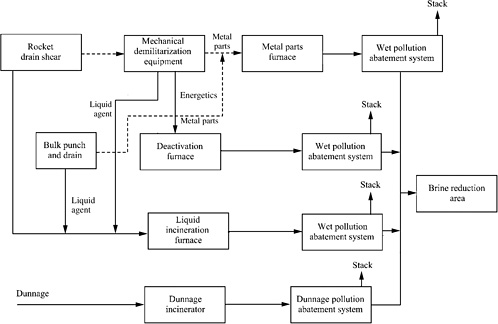
FIGURE 5-4 Block diagram of U.S. baseline incineration system. SOURCE: Pearson and Magee, 2002.
two of the committee members for this report. The facility has a capacity of about 70 metric tons per year, including contaminated waste from operations and metal scrap from munitions. Disassembly of munitions to access the agent involves the use of cutting equipment that requires intensive operator involvement. Solid wastes to be incinerated are placed in polyethylene containers, and liquids are placed in tin boxes. These are fed in a batch fashion to the incinerator. This incinerator has an evaporation chamber, where materials are held for 12 hours at 300C, and a burn-out chamber, where materials are held for 18 hours at 1000C. Offgases from these are burned in a main combustion chamber, with a residence time of 2 seconds and a temperature of 1200C. The flue gases are cleaned extensively before discharging. Solid waste, including arsenic products, is sent to a former salt mine. Aqueous effluent is sent to either a municipal wastewater treatment system or is evaporated.
United Kingdom
The United Kingdom facility at Porton Down employs a licensed hazardous waste incinerator for munitions destruction.7 This is again a fixed facility, not suitable for moving from site to site.
The weapons are initially stored in open-sided buildings. They are assessed using portable isotopic neutron spectroscopy and x rays to determine the nature of the fill and then placed in commercial freezing isocontainers at –15C to minimize the polymerization of mustard agent while in storage. Munitions containing phosgene or chlorine are drilled and vented to a caustic scrubbing system to destroy the phosgene or chlorine.
The frozen munitions containing mustard agent are sawed into two parts using either a band saw or a reciprocating saw. This is done in enclosed chambers, with the saws cutting into the cavities containing the mustard agent but above the level of the mustard agent. An operator enters the chamber and pours the agent into a glass container, which is in turn placed in a plastic bucket. The bucket also contains activated carbon to absorb the agent if the glass container leaks. The bucket is
sent to the incinerator for destruction of the mustard. Most of the mustard agent is the HT form.8
The incinerator has a continuously fed rotating kiln furnace and a batch-operated, fixed-hearth furnace. Offgases from the furnaces pass to a secondary combustion chamber operating at 1200C and a 2-second residence time. The furnaces and secondary combustion chambers are generally fired with waste oil. The gases then pass through a liquid quench system that drops the temperature to 200C. This system is designed to control dioxins and furans. The gases pass into a bag filter that has been precoated with lime or sodium bicarbonate to control acid gases and particulate, then to a caustic scrubber for more acid gas removal. The gases are analyzed continuously at the exit of the scrubber, and the feed to the incinerator is automatically stopped if limits are exceeded. The gases then pass through an induction fan and into a stack.
The bucket containing the mustard agent is placed in the fixed-hearth furnace for destruction. Fuzes and other small or low-energy parts containing energetics are placed in a steel bang box and detonated in the fixed-hearth furnace. Parts containing higher energy energetics are open-detonated. The metal bodies of the munitions are treated thermally in the incinerator and disposed of.
As of January 2006, about 850 chemical weapon rounds were in inventory at Porton Down, and it was expected that these would all be destroyed by April 2007.9
Canada
Chemical warfare agents, mainly mustard agent, were transported to DRES after World War II for destruction (Sutherland, 1997). Over 700 tons of mustard agent were destroyed in 1975 and 1976 using alkaline hydrolysis followed by incineration of the hydrolysate and landfilling of the residues.
Another campaign was initiated in 1989 aimed at destroying additional materials, including 12 tons of liquid or thickened mustard agent; 1.5 tons of lewisite; 0.3 tons of nerve agents (G and V classes); 300 tons of contaminated scrap, containers, and ordnance; 4 tons of contaminated waste such as decontaminating solution; and 40 tons of nonhazardous scrap. An incinerator was built for this purpose. Mustard agent was incinerated directly. The mustard-agent-filled munitions were punctured with shaped charges. The agent was drained from the munitions and poured into polyethylene-lined boxes. It was then frozen, transported to the incinerator, and fed through the solid waste feed system.10 The drained munitions were placed in sealed polyethylene-lined boxes and fed to the incinerator through the solid waste feed system. Nerve agents were neutralized with potassium hydroxide and the hydrolysate was incinerated. The incinerator had a combined liquid plus solid feed rate of 1.5 tons per hour. The incineration campaign took 3 months.
Iraq
An incinerator was built and operated at a desert site in Iraq to destroy mustard agent as part of the 1991-1994 program to destroy the agent production facilities and chemical weapons in Iraq following the Desert Storm military operation (Manley, 1997). All energetics were removed from the weapons. Several techniques were then employed to access the agent in the weapons, including removal of the filling plug when possible, drilling holes, and using shaped explosive charges to create holes. The drained weapons were rinsed, again using various techniques, and then burned. A single incinerator was constructed at the former agent production facility, using an existing tank farm to store agent and prepare it for incineration. Mustard agent was mixed with a particular petroleum fraction in which it was soluble, then with fuel oil. A large brick-lined furnace was constructed in a trench and two burner guns were installed. The combustion gases were diluted with ambient air, passed through a water quench tower, passed through a packed tower irrigated with a 5-10 percent caustic solution, passed through an induction fan, and discharged through a tall stack. The mustard agent feed rate was 300 kg/hr, and the overall campaign destroyed about 400 metric tons of agent.
Russia
A mobile neutralization and incineration system designed to destroy nerve agent was built and operated in Russia beginning in 1980 (Krotovich, 1998). This facility destroyed 300-350 metric tons of chemical agent in a 100-day period.
Evaluation Factors Analysis
Process Maturity
Pearson and Magee (2002) state that more chemical agent has been destroyed by incineration than by any other technology. Table 5-1 shows major chemical weapon destruction
TABLE 5-1 Destruction of Chemical Agents, 1958-1993
|
Site |
Agent |
Amounta (tonnes) |
Methodb |
Dates |
|
Rocky Mountain Arsenal, USA |
H |
2,786 |
I |
1969-1974 |
|
Rocky Mountain Arsenal, USA |
GB |
3,799 |
N |
1973-1976 |
|
Tooele (CAMDS),c USA |
GB |
34.5 |
I |
1981-1986 |
|
JACADS,d USA – OVT data |
GB |
36 |
I |
1991 |
|
Tooele (CAMDS),c USA |
VX |
7 |
I |
1984 |
|
JACADS,d USA – OVT data |
VX |
49 |
I |
1992 |
|
JACADS,d USA – OVT data |
HD |
51 |
I |
1992 |
|
DRES Canada |
H |
700 |
N/I |
1974-1976 |
|
DRES Canada |
H |
12 |
I |
1990-1991 |
|
DRES Canada |
VX, GA, GB |
0.3 |
N/I |
1990-1991 |
|
DRES Canada |
L |
1.5 |
N |
|
|
Munster, Germany |
H, etc. |
70/year |
I |
1980-present |
|
Shikhany, Russia |
GB, GD, H |
300 |
N/I |
1980-1990 |
|
Shikhany, Russia |
VRe |
30 |
N/I |
1980-1990 |
|
Porton Down, U.K. |
H |
20 |
I |
1970 |
|
Nancekuke, U.K. |
GB |
20 |
N |
1967-1968 |
|
Runcorn, U.K. |
H |
6,000 |
I |
1958-1960 |
|
Iraq (UNSCOM supervised) |
H |
500 |
I |
1992-1993 |
|
Iraq (UNSCOM supervised) |
GA |
30 |
N |
1992-1993 |
|
Iraq (UNSCOM supervised) |
GB, GB/GF |
70 |
N |
1992-1993 |
|
aU.S. figures based on 2,000-lb tons converted to 1,000-kg tonnes. bI, incineration; N, neutralization. cChemical Agent Munitions Disposal System experimental facility, Tooele Army Depot, Utah. dJohnston Atoll Chemical Agent Disposal System; data are from preoperational Operational Verification Tests (OVT). eVR is Russian VX. SOURCE: Adapted from Pearson and Magee, 2002. |
||||
projects worldwide from 1958 to 1993. The table lists projects involving incineration, neutralization (including hydrolysis), and a combination of neutralization and incineration.
Most of the projects were performed using either incineration or a combination of neutralization and incineration. Much of the U.S. stockpile has since been destroyed using incineration. This includes all of the 2,030 tons of agent that were in the Johnston Atoll stockpile and much of the 23,437 tons of agent that were at Anniston, Pine Bluff, Tooele, and Umatilla. Incineration of chemical weapons has been or is being used in Canada, Germany, Belgium, Russia, the United Kingdom, and Iraq. In all these countries, incineration was used solely or in part to directly destroy agent. The technology has been used to destroy a variety of agents and energetics in a variety of munitions. Systems of various sizes and degrees of sophistication have been built, ranging from the large, complex systems built for destruction of the U.S. stockpile to the small, considerably less complex system built for the Iraq application.
Process Efficacy/Throughput
Incineration is highly effective in destroying agents and energetics. At Johnston Atoll, the destruction and removal requirement (DRE) for GB and VX was 99.99999 percent and the required DRE for mustard agent was 99.99995 percent.11 DREs for energetics must be 99.99 percent or greater. The stockpile plants have experienced long periods of downtime, usually following an incident in the facility. Hundreds of workers are employed in the stockpile destruction plants, a large staff in the absolute sense but not unusually large for the tasks being performed; many of the workers are involved in operations peripheral to the incineration-related activities. The process has been used to destroy a wide variety of agents and energetics in a wide variety of munitions. Portableortable incinerators have been built and are technically feasible, but obtaining permits for operation would be difficult because of public and regulatory acceptance issues.
Process Safety
Dangerous materials are handled in these plants, solids handling operations are extensive, and operations and repairs inherently involve risk. These factors have led to incidents at the U.S. stockpile destruction plants, resulting in downtime. Incidents include releases of agent to the environment, but
their causes have been studied extensively and demilitarization facilities personnel have made a serious effort to learn from past incidents (NRC, 2002). The committee anticipates that such incidents will be less frequent in the newer plants (Pine Bluff, Umatilla, and Anniston). Historically, 12 years of operation of two destruction facilities prior to 2002 witnessed three confirmed agent releases to the environment involving the equivalent of a few small drops of agent in total. In comparison, several hundred leaks of stored agent occurred between 1990 and 2000, with the most serious resulting in release to the environment of 78 gallons of mustard agent. Thus, releases from storage of agent have been far more serious than releases from destruction facilities (NRC, 2002). Engineering controls have been put in place to mitigate recognized risks. Interlocks are used extensively. A major safety concern is having unburned agent or products of incomplete combustion exiting the stack. To minimize this risk, agent flow to the burner is stopped if the temperature in the primary combustion chamber drops below 1400C (Pearson and Magee, 2002).
Note that the risk associated with retrieving the munitions from the bunkers and moving them to the destruction facilities is approximately the same regardless of the destruction process employed, incineration, neutralization, or other.
Public and Regulatory Acceptability in a U.S. Context
Incineration of chemical weapons is applied extensively in the United States and in some foreign countries, such as the United Kingdom, Belgium, and Germany. It is used for both chemical agent and secondary waste, and, overall, it has been an effective technology. In addition, incineration was endorsed as a suitable technology for chemical agent and munitions destruction early on by the NRC (1984), and incinerators were built and operated at four of the Army’s stockpile sites. However, for several reasons, incineration has not been well received by public interest groups in the United States, and public pressure resulted in years of delays in obtaining permits, with attendant delays in schedule. This, in turn, caused the investment of millions of dollars in developing alternative technologies for destruction of chemical stockpiles, and such technologies have been selected for use at four of the Army’s chemical stockpile sites.
The opposition to the use of incineration is described in detail in a prior NRC report (NRC, 2002). Reasons for it include the perceived instability of the process, the potential for explosion, and the potential for unplanned releases of undesirable compounds. This public opposition to incineration is also evident in the U.S. Environmental Protection Agency’s Superfund program for cleanup of hazardous waste sites, where both on-site and off-site incineration were selected less frequently as treatment technologies as the years passed (EPA, 2004). For example, on-site incineration was selected four, seven, six, and four times in 1987, 1988, 1989, and 1990, respectively, for source control for remedial actions but not at all in 1998 through 2002. Similarly, off-site incineration was selected three, nine, nine, fifteen, and twelve times in 1987, 1988, 1989, 1990, and 1991 respectively, and three, two, six, one, and two times in 1998, 1999, 2000, 2001, and 2002, respectively.
The non-stockpile munitions disassembly procedures used in the United Kingdom, Germany, and Belgium require extensive operator involvement, with the attendant risk of exposure to agent. All of these countries consider these technologies to be out of date and are actively considering alternatives.
Secondary Waste Issues
Numerous secondary wastes are produced by incineration processes. Table 5-2 lists the liquid and solid wastes produced by the baseline incineration facility at Tooele (U.S. Army, 1999). This table also shows whether wastes generated are (1) incinerated within the facility, (2) sent to an off-site TSDF, or (3) placed in permitted on-site storage. Treated metal parts from the metal parts furnace (MPF) are described as a high-volume stream that contains the 5X-treated metal bodies of all munitions and agent ton containers.
The U.S. Army does not plan to incinerate the lewisite stored in 10 one-ton containers at the Tooele site. The method of destruction to be employed has not been finalized, but the leading candidate is treatment with sodium permanganate to precipitate a manganese oxide-arsenic solid (Ember, 2006). This solid would be encased in concrete and disposed of in a permitted hazardous waste landfill, with the remaining liquids treated further and injected into a deep well.
Process Costs
Incineration processes, especially as built and operated for the destruction of the U.S. chemical weapons stockpile, are complex and expensive. For example, the Johnston Atoll Chemical Agent Disposal System (JACADS), the first of the baseline plants, cost $254 million to build and $100 million for systemization (Sutherland, 1997). As of 1997, additional costs of over $500 million for operation were expected. It is not known how the costs of an incineration system to be used for non-stockpile chemical weapon destruction would compare with the JACADS costs.
COMPARATIVE EVALUATIONS OF TIER 1 AGENT-ONLY PROCESSING TECHNOLOGIES
Table 5-3 rates the Tier 1 agent-only processing technologies according to the evaluation factors defined in detail in Chapter 3. The ratings scheme is described in more detail in that chapter.
The committee also addressed the subfactors as given in Chapter 3 for each of the five main evaluation factors. The subfactor questions for the Tier 1 international tech-
TABLE 5-2 Tooele Chemical Agent Disposal Facility Waste Streams
|
Waste Stream |
Currently Treated by Incineration? |
|
Waste chemicals |
Yes, except for lewisite |
|
Brine reduction area baghouse residue |
No, offsite TSDF |
|
Brine salts |
No, offsite TSDF |
|
Brine tank sludge |
No, offsite TSDF |
|
Neutralization fluid |
Yes, except for laboratory operations |
|
Deactivation furnace system (DFS) cyclone residue |
No, offsite TSDF |
|
DFS heated discharge conveyor ash |
No, offsite TSDF |
|
DFS refractory |
No, offsite TSDF |
|
Demilitarization protective ensemble suits |
No, permitted long-term storage |
|
Laboratory liquid wastes |
No, offsite TSDF |
|
Laboratory solid wastes |
No, offsite TSDF |
|
Liquid incinerator refractory |
No, offsite TSDF |
|
Liquid incinerator slag |
No, offsite TSDF |
|
Miscellaneous metal parts |
Yes |
|
Mist eliminator filter/demister candles |
No, offsite TSDF |
|
Metal parts furnace (MPF) ash |
No, offsite TSDF |
|
MPF metal |
Yes |
|
MPF refractory |
No, offsite TSDF |
|
MPF residue |
No, offsite TSDF |
|
Pollution abatement system (PAS) brine |
No, offsite TSDF |
|
PAS brine sump sludge |
No, offsite TSDF |
|
PAS quench tower residue |
No, offsite TSDF |
|
Plastics |
No, permitted onsite storage |
|
Residue handling area baghouse residue |
No, offsite TSDF |
|
Spent activated carbon |
No, permitted onsite storage |
|
Spent hydraulic fluid |
Yes |
|
Spent silver fluoride pads |
No, offsite TSDF |
|
Spill cleanup material |
Yes |
|
Trash, debris, and protective clothing |
No, permitted on-site storage |
|
Waste oil |
Yes |
|
Wooden pallets |
No, permitted on-site storage |
|
SOURCE: Adapted from U.S. Army, 1999. |
|
TABLE 5-3 Evaluation Factor Rating Comparison of Tier 1 Agent-Only Processing Technologies with U.S. RRS/SCANS
|
Technology |
Evaluation Factors (Ratinga)b |
||||
|
Process Maturity |
Process Efficacy/ Throughput |
Process Safety |
Public and Regulatory Acceptability in a U.S. Context |
Secondary Waste Issues |
|
|
U.S. Neutralization/hydrolysis (RRS/SCANS) |
+ |
+ |
+ |
+ |
0 |
|
Russian two-stage process: neutralization with addition of bitumen |
? |
+ |
+ |
0 |
– |
|
Incineration |
+ |
+ |
+ |
– |
– |
|
aLegend: +, acceptable; 0, partially acceptable; , unacceptable; ?, inadequate information. |
|||||
nologies suitable for non-stockpile agent-only processing are addressed in Appendix C in Tables C-1 through C-5. These tables provide a convenient side-by-side means for comparing some specific aspects of these international technologies with the NSCMP equipment based on the available data and the expert judgment of the committee.
FINDINGS AND RECOMMENDATIONS
Finding 5-1. In the Russian context, the destruction efficiency for the Russian two-stage bitumen process is sufficiently high, the reactions involved are irreversible, and the toxicity of the bitumen-salt product from the Russian two-stage process is sufficiently low.
Finding 5-2. The Russian two-stage neutralization/ bitumenization process generates a large amount of secondary wastes that in the United States would require being land-filled and monitored indefinitely. The lack of information on the long-term stability of these wastes and the leaching of toxic materials from them (see “Secondary Waste Issues”) is a cause for concern.
Recommendation 5-1. The U.S. Army should not expend any resources on further evaluation of the Russian two-stage neutralization/bitumenization process for application in the United States.
Finding 5-3. Incineration is a highly developed and well-proven technology for the destruction of chemical agent and various types of secondary waste. Public acceptability of the technology is poor within the United States but adequate in most other countries.
Finding 5-4. The reverse assembly process used in the U.S. baseline incineration system is not applicable to non-stockpile chemical weapons because of the deteriorated conditions of the munitions.
Finding 5-5. Incineration of non-stockpile materials is used in the United Kingdom, Belgium, and Germany in conjunction with human handling and remotely controlled cutting of munitions. Such operations are labor intensive and could cause operators to be exposed to agent.
Finding 5-6. Although incineration is a robust, safe, and proven technology for the destruction of chemical agent, it is not the best technology for non-stockpile materials because access and acceptability are issues.
Recommendation 5-2. The U.S. Army should consider the difficulty of obtaining public acceptance when comparing incineration with other non-stockpile agent destruction technologies for use in the United States.
REFERENCES
Neutralization and Hydrolysis
GSN (NTI: Global Security Newswire). 2005. Russian CW Destruction Facility Nears Completion, October 27. Available online at <http://www.nti.org/d_newswire/issues/2005_10_27.html>. Last accessed March 6, 2006.
Kawataki, H., H. Shimoyama, Y. Yamane, and C. Furukawa. 2005. Destruction of OCW in Glass Beer Bottles at Samukawa. Available online at <http://www.dstl.gov.uk/news_events/conferences/cwd/2005/proceedings50.pdf>. Last accessed March 6, 2006.
Litovkin, V. 2005. Russia takes another step towards scrapping its chemical weapons. Available online at <http://en.rian.ru/analysis/20051201/42278210.htm>. Last accessed March 6, 2006.
Manley, R.G. 1997. “UNSCOM’s experience with chemical warfare agents and munitions in Iraq.” Pp. 241-254 in The Challenge of Old Chemical Munitions and Toxic Armament Wastes. T. Stock and K. Lohs, eds. Oxford, England: Oxford University Press.
Pearson, G.S., and R.S. Magee. 2002. “Critical evaluation of proven chemical weapon destruction technologies (IUPAC Technical Report).” Pure and Applied Chemistry 74(2): 187-316.
Sutherland, R.G. 1997. “The destruction of old and obsolete chemical weapons: Past experience.” Pp. 141-155 in The Challenge of Old Chemical Munitions and Toxic Armament Wastes. T. Stock and K. Lohs, eds. Oxford, England: Oxford University Press.
Russian Two-Stage Process
Bechtel (Bechtel International, Inc.). 1996. Joint Evaluation of the Russian Two-Stage Chemical Agent Destruction Process, Final Technical Report: Phases 1 & 2, Revised, July. Alexandria, Va.: Defense Special Weapons Agency.
Ember, L. 2003. “Ridding Russia of chemical weapons.” Chemical & Engineering News 81(48): 28-31.
Ember, L. 2005. “The Shchuch’ye dilemma.” Chemical & Engineering News 83(45): 19-23.
Khripunov, I., and G.W. Parshall. 1999. “U.S. assistance to Russian chemical weapons destruction: Identifying the next steps.” Available online at <http://cns.miis.edu/pubs/npr/vol06/64/krip64.pdf>. Last accessed March 6, 2006.
Krotovich, I.N. 1998. “Development of CW destruction technologies in Russia.” Pp. 89-93 in Proceedings of the International CW Destruction Symposium, Munster, Germany, March 22-25, 1998. V. Starrock and A. Krippendorf, eds. Munster, Germany: Munster Expo 2000 Committee.
Pearson, G.S., and R.S. Magee. 2002. “Critical evaluation of proven chemical weapon destruction technologies (IUPAC Technical Report).” Pure and Applied Chemistry 74(2): 187-316.
Petrov, V.G., and A.V. Trubachev. 2000. “On certain problems of technical and ecological safety during chemical weapons destruction in the Udmurt Republic.” Proceedings of Munster 2000, The 2nd International Symposium, Destruction of Chemical WeaponsTechnologies and Practical Aspects, July 30-August 3, 2000. Munster, Germany: Munster Expo 2000 Committee.
Radilov, A.S. 2004. “Toxicological and hygienic aspects of the problem of chemical detoxification of organophosphorus chemical warfare agents.” Available online at <http://www.dstl.gov.uk/news_events/conferences/cwd/2004/proceedings25.pdf>. Last accessed March 6, 2006.
Ryan, S., and P. McNelly. 2003. “Eliminating Russian nerve agent weapons.” Proceedings of the 2003 Chemical Weapons Demil Conference. Porton Down, England: Defence Science and Technology Laboratory.
Incineration
CNN (Cable News Network). 2001. WWI munitions transferred safely. Available online at <http://archives.cnn.com/2001/WORLD/europe/04/16/Vimy.munitions.02/index.html>. Last accessed March 6, 2006.
Ember, L.R. 2006. “Chemical weapons deadline at risk.” Chemical and Engineering News 84(16): 27-30.
EPA (U.S. Environmental Protection Agency). 2004. Treatment Technologies for Site Cleanup: Annual Status Report (11th Edition), EPA-542-R-03-009, February. Available online at <http://www.clu-in.org/download/remed/asr/11/asr.pdf>. Last accessed February 14, 2006.
Krotovich, I.N. 1998. “Development of CW destruction technologies in Russia.” Pp. 89-93 in Proceedings of the International CW Destruction Symposium, March 22-25. V. Starrock and A. Krippendorf, eds. Munster, Germany: Munster Expo 2000 Committee.
Manley, R.G. 1997. “UNSCOM’s experience with chemical warfare agents and munitions in Iraq.” Pp. 241-254 in The Challenge of Old Chemical Munitions and Toxic Armament Wastes. T. Stock and K. Lohs, eds. Oxford, England: Oxford University Press.
Martens, H. 1997. “The German programme for the disposal of old chemical weapons.” Pp. 166-178 in The Challenge of Old Chemical Munitions and Toxic Armament Wastes. T. Stock and K. Lohs, eds. Oxford, England: Oxford University Press.
Mihm, J. 2000. “Activities in the field of chemical weapons agent disposal in Germany.” Proceedings of Munster 2000, The 2nd International Symposium, Destruction of Chemical WeaponsTechnologies and Practical Aspects, July 30-August 3. Munster, Germany: Munster Expo 2000 Committee.
NRC (National Research Council). 1984. Disposal of Chemical Munitions and Agents. Washington, D.C.: National Academy Press.
NRC. 2002. Evaluation of Chemical Events at Army Chemical Agent Disposal Facilities. Washington, D.C.: The National Academies Press.
Pearson, G.S., and R.S. Magee. 2002. “Critical evaluation of proven chemical weapon destruction technologies (IUPAC Technical Report).” Pure and Applied Chemistry 74(2): 187-316.
Sutherland, R.G. 1997. “The destruction of old and obsolete chemical weapons: Past experience.” Pp. 141-155 in The Challenge of Old Chemical Munitions and Toxic Armament Wastes. T. Stock and K. Lohs, eds. Oxford, England: Oxford University Press.
U.S. Army. 1999. Incineration as a Land Disposal Restriction Treatment Technology for Chemical Agent Associated Waste. Available online at <http://www.hazardouswaste.utah.gov/CDS/OTHER/UCAR/UCAR_VOLUME_2/Background%20Document%20H.pdf>. Last accessed January 17, 2006.
Zanders, J.P. 1997. “The destruction of old chemical weapons in Belgium.” Pp. 197-230 in The Challenge of Old Chemical Munitions and Toxic Armament Wastes. T. Stock and K. Lohs, eds. Oxford, England: Oxford University Press.














