8
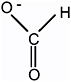
Formate
Hector D. Garcia, Ph.D. NASA-Johnson Space Center Toxicology Group Habitability and Environmental Factors Division Houston, Texas
TABLE 8-1 Physical and Chemical Properties
|
Formula |
HCOO− |
|
|
|
Chemical Name |
Formate |
||
|
Synonyms |
Methanoate, carboxylate |
||
|
CAS registry no. |
64-19-7 (formic acid) |
||
|
|
544-17-2 (calcium formate) |
||
|
|
6150-82-9 (magnesium formate) |
||
|
Molecular weight |
45 |
||
|
Solubility |
The calcium and magnesium salts are highly soluble in water |
||
|
Formate is the anion of formic acid, the simplest organic acid. |
|||
|
Source: Data from HSDB 2006. |
|||
OCCURRENCE AND USE
Formic acid is found in the venom of ants, from which it got its name (Latin formica), as well as in the venoms of other insects and a variety of plants. Formate ions (COOH−) are generated in mammalian tissues during normal metabolism of glyoxylate. Formate ions are also generated as an intermediate during mammalian metabolism of methanol and formaldehyde to carbon dioxide (CO2). In industry, formic acid is used as a food preservative, in a variety of chemical syntheses, in tanning leather, as a decalcifying agent, and in the preservation of silage (Liesivuori 1986; Dorland 1988; Thompson 1992). Endogenous concentrations of formate in human blood are in the range of 0.1-0.2 micromoles per milliliter (µmole/mL) (4.5-9.0 micrograms per milliliter [µg/mL]) (Medinsky and Dorman 1995). See Table 8-1 for the physicalchemical properties.
Formate is added to potable water on the International Space Station (ISS) in the process of adding minerals to pure water to make it more palatable. Highly pure water is supplemented with calcium and magnesium as the formate and acetate salts by adding 100 mL of a stock containing formate at 37.8 g/L to 44 L of pure (fuel-cell generated) water to yield about 86 mg/L (0.52 millimoles per liter [mmol/L]). Normally, the room-temperature water-dispensing port on the ISS is supplied with this U.S. formate-containing water, sometimes diluted with non-formate-containing water from other sources, while heated water (without formate) comes from Russian-supplied sources (either regenerated or natural water from Russia). If all the drinking water were formate-containing U.S. water, an astronaut would ingest 241 mg per day (d), at a consumption rate of 2.8 L/d of water.
TOXICOKINETICS AND METABOLISM
The distribution, metabolism, and elimination of formate have been studied in humans, nonhuman primates, rodents, and pigs.
Absorption
Gastrointestinal (GI) absorption of formate is almost total, as seen in cases of accidental and intentional self poisoning with formic acid solutions (Naik et al. 1980). No data were found on the rate of absorption of formate from the GI tract.
Distribution
Absorbed formate is distributed in the body water compartment, which in humans, is about 0.5 L per kilogram (kg) (Liesivuori and Savolainen 1991). No data were found on tissue-specific differences in the distribution of formate.
Metabolism
The major route of formate metabolism in the rat, monkey, and presumably in humans is oxidation to CO2 via the folate biochemical path-
way. Formate enters the pathway by combining with tetrahydrofolate. In a tetrahydrofolate-dependent reaction, formic acid is slowly metabolized to CO2 in primates but is metabolized about twice as rapidly in nonprimates (McMartin et al. 1977).
Formic acid is produced during metabolism of methanol in humans and animals, and the delayed toxicity of ingested methanol in humans appears to be due to the toxicity of formate. The susceptibility of various species to methanol toxicity is inversely related to the rate of tetrahydrofolate-dependent oxidation of formate to CO2 (Black et al. 1985). Two mechanisms may be operative in explaining low formate oxidation in species susceptible to methanol toxicity: low hepatic tetrahydrofolate concentrations and reduced hepatic 10-formyltetrahydrofolate dehydrogenase activity (Johlin et al. 1987). The activity of 10-formyltetrahydrofolate dehydrogenase, the enzyme that catalyzes the final step of formate oxidation to CO2, has been reported to be markedly (four- to fivefold) reduced in both monkey and human liver compared to rodent liver, consistent with lower formate oxidation rates in primates. Both of these mechanisms should apply to ingested formate (for example, in spacecraft drinking water) as well as formate generated by the metabolism of methanol.
Excretion
The majority of ingested formate is excreted as its metabolite, CO2. Renal elimination of formate appears to be minimal (Kerns et al. 2002), being 5% in humans (Lund 1948). In a later study, when sodium formate was given orally to humans, 2.1% of the dose was detected in urine collected over 24 hours (h) (Malorny 1969).
In monkeys, whose sensitivity to the ocular toxicity of methanol is similar to that of humans, the half-life of formic acid in the blood is about 31-51 minutes (min) for intravenous (iv) doses of 50-470 mg/kg (Clay et al. 1975). At low doses (≤100 mg/kg), iv-infused formic acid is cleared from monkeys with a half-life of 31 min (Liesivuori and Savolainen 1991). The half-life increases with increasing formate dose, indicating a nonlinearity in formate disposition. Data from animal studies (Clay et al. 1975; McMartin et al. 1977) suggest that this represents an unusual dose-dependent metabolic clearance that is the result of depletion of the co-substrate (tetrahydrofolate) for the 10-formyl tetrahydrofolate dehydrogenase reaction. Under this kinetic condition, the elimination of formate will appear first-order with increasing doses, but the apparent elimination rate constant will decline with increasing doses
(Galinsky and Levy 1981). This behavior is only an approximation of more-complex formate elimination kinetics that will occur with a dynamic state of depletion and repletion of the co-substrate as formate is introduced into the body and then eliminated. However, at every dose studied, formate elimination in monkeys appears to behave as a pseudo-first-order process over a period of time when approximately 75-87% of the dose had been eliminated (Clay et al. 1975). In humans who had ingested toxic doses of methanol and had been treated with fomepizole and folate supplementation, the endogenous elimination half-life of formate was reported to be 3.4 h (Kerns et al. 2002). Interpretation of formate half-lives calculated using data from methanol intoxications, however, is complicated by the simultaneous production of new formate because of continuing methanol metabolism. The observed formate half-life could be formation-rate limited and not elimination-rate limited. The true formate half-life could be less than or equal to that observed after methanol ingestion. Thus, the 3.4-h half-life value may not be relevant when the exposure to formate is not from the ingestion of methanol.
The formate concentrations in the blood of a methanol-intoxicated 21-year (y)-old patient remained relatively constant until blood pH was normalized by bicarbonate treatment, after which formate concentrations declined, suggesting that formate elimination may be pH dependent (Jacobsen et al. 1988).
TOXICITY SUMMARY
Formic acid is highly irritating and is caustic to the skin.
The main toxic effects associated with the ingestion of methanol (a metabolic precursor of formate) are metabolic acidosis and blindness from optic nerve injury. The visual toxicity has been shown to be independent of pH (Martin-Amat et al. 1978). The mechanism of cytotoxicity is believed to involve histotoxic hypoxia (the reduced ability of cells or tissue to accept and use oxygen from the blood, such as in cyanide poisoning) in aerobic cells caused by the impeding of oxygen metabolism (Liesivuori 1986). Optic nerve cells have relatively few mitochondria and thus have a high sensitivity to histotoxic hypoxia (Jacobsen and McMartin 1986). Formate acts as a reversible inhibitor of the mitochondrial cytochrome c oxidase complex (Nicholls 1975, 1976; Erecinska and Wilson 1980) with an apparent inhibition constant (Ki) between 1 and 30 millimolar (mM) (45-1,350 µg/mL) at pH 7.4, 30C (Nicholls 1975, 1976), depending on the reduction state of the system. Inhibition of aero-
bic respiration is known to stimulate anaerobic glycolysis, resulting in increased lactate production and intra- and extracellular lactic acidosis (Seisjo 1992). Metabolic and lactic acidosis are both hallmark features of severe human methanol intoxication (Erlanson et al. 1965; Koivusalo 1970; Jacobsen and McMartin 1986). The acidosis causes a decreased production of adenosine triphosphate (ATP) (Liesivuori and Savolainen 1991).
Interpretation of toxicity data from experiments involving exposures to methanol is complicated by interspecies as well as nutritional status-dependent differences in the rate of metabolism of methanol. Although reports from Finland (Liesivuori et al. 1987; Liesivuori and Savolainen 1991) and the United States (Martin-Amat et al. 1977, 1978; McMartin et al. 1977, 1980; Jacobsen et al. 1988; Johlin et al. 1989; Tephly 1991; Eells et al. 1996) implicate formate as the toxic agent in methanol poisoning, dissenting investigators from Japan (Hayasaka et al. 2001) assert that formaldehyde is the toxic agent in methanol-induced eye toxicity.
Acute and Short-Term Exposures (≤10 d)
Moore et al. (1994) reported that iv folinic acid and furosemide and urinary alkalinization resulted in the survival after attempted suicide of a woman who ingested 110 g of formic acid. They state that ingestion of over 60 g of formic acid is potentially fatal. A successful suicide reported by Westphal et al. (2001) was attributed to massive acidosis, hemolysis, bleeding complications, and hepatic and renal failure after ingestion of an estimated 44-88 mL of a decalcifying agent containing 60% volume per volume (v/v) formic acid. Westphal’s review of the literature indicated that formic acid concentrations in the blood of healthy humans range from 3.2-56 µg/mL and that ingestion of less than 30 g of formic acid was never fatal. Westphal et al.’s reported value of formate at 56 µg/mL blood in unexposed humans appears to be a typographical error, however, based on examination of his cited sources for this value. The highest value found in any of the articles cited by Westphal et al. was 5.6 ± 4.5 µg/mL (Baumann and Angerer 1979).
No reports were found in the literature of ocular toxicity in humans who had ingested formic acid or formate, whereas ocular toxicity is commonly seen in cases of human exposure to methanol. McMartin et al. (1980) reported severe metabolic acidosis and optic disc edema in two patients whose blood formate concentrations were 11.1 and 26.0 mil-
liequivalents (mEq) per milliliter when measured after hospitalization for methanol intoxication. The patient with the higher blood formate concentration died despite aggressive treatment (bicarbonate, iv 10% ethanol, and hemodialysis) of both patients.
Histotoxicity (manifested as extracellular calcium casts) has been reported in several tissues examined 1 h after the fifth daily dosing in male New Zealand rabbits (3,070 ± 220 g) given daily iv 1 mL doses of formate at 100 mg/kg (Liesivuori et al. 1987). If we assume a body water volume to body mass ratio of about 700 mL water per kilogram, the rabbits had about 2,200 mL body water, and the formate concentration immediately after the injection would have been 307 mg/2,200 mL = 140 µg/mL body water. Measured blood formate concentrations were 0.7 µmole/g (29 µg/mL) at 1 h after the fifth daily dose, 0.5 µmole/g (21 µg/mL) at 2 h, and 0.2 µmole/g (8.4 µg/mL) at 20 h. Thus, it appears that 80% of the administered dose of formate had disappeared from the blood by 1 h postdosing, 5% more during the second hour postdosing, and 6% during the following 18 h. This is consistent with a half-life of formate in rabbits of about 26 min. The reduction of blood formate concentrations could be caused by either rapid sequestration of formate into other tissues or by rapid metabolism of the formate. No mention was made in this report of any ocular toxicity in the rabbits.
In humans, permanent visual damage has been associated with prolonged exposures (>24 h) to blood formate concentrations > 7 mM (315 µg/mL) produced during methanol intoxications (Hayreh et al. 1977, 1980; Jacobsen and McMartin 1986). The formate concentration in these patients (315 µg/mL) was higher than the initial concentration calculated for the rabbits (140 µg/mL) in Leisivuori’s study. Also, the fact that formate was being constantly produced by metabolism of ingested methanol means that elevated blood concentrations of formate were maintained for a prolonged period in methanol-intoxicated humans as opposed to rapidly declining to near background levels in the formate-treated rabbits, presumably from metabolism. Clinical observations in methanol-intoxicated humans have shown that in its initial stages, developing ocular toxicity can be reversed, even >24 h after methanol ingestion, by treatments such as bicarbonate, fomepizole or ethanol, iv folinic acid, or dialysis, which reduce blood formate concentrations and metabolic acidosis (Barceloux et al. 2002). Such reversibility is consistent with a mechanism of formate toxicity involving gradual optic nerve histotoxicity due to prolonged metabolic hypoxia and with the reversibility of formate inhibition of cytochrome c oxidase (Nicholls 1975).
Martin-Amat et al. (1978) determined that the ocular toxicity (edema of the optic disc and loss of pupillary response) of methanol could be reproduced in male rhesus monkeys by iv infusion of sodium formate at 0.5 M, even when bicarbonate was administered to prevent acidosis. Infusion at a rate of about 3.1 mEq/kg/h (formate at 140 mg/kg/h) after a loading dose of 1.25 mmole/kg (formate at 56 mg/kg) was calculated so as to maintain 10-30 mEq/L (450-1,350 µg/mL) formate in the blood over a period of 25-39 h. Note that because a quasi-steady state was achieved, the infusion rate is also an estimate of the rate of removal of formate from the blood by metabolism and excretion. In most of the treated animals, no pupillary response to light was observed at between 24 and 48 h after the onset of formate infusion, but in one of the four monkeys tested, normal pupillary reflexes and only moderate optic disc edema were observed at 25 h after initiation of treatment. In this monkey, the maximum blood concentration of formate achieved was 540 µg/mL at 25 h postinitiation of treatment, compared to 900-1,530 µg/mL for the other three monkeys, as measured at later times (39-50 h post-initiation of treatment). The onset of ocular toxicity generally occurred more rapidly in the formate-treated animals than in monkeys that had received methanol in previous studies.
A more sensitive test for ocular toxicity (measurement of the reductions in the a and b waves of electroretinograms, [ERGs]) was used by Eells et al. (1996) to demonstrate that intraperitoneal (ip) injections of methanol in a regimen that maintained blood formate concentrations at 4-6 mM (180-270 µg/mL) for 60 h showed evidence of causing retinal dysfunction in the absence of retinal histopathology in nitrous oxide (N2O)-treated rats. The retinal dysfunction was not correlated with any clinical signs. The rats in this experiment were made to more closely resemble humans and nonhuman primates in their methanol sensitivity by using subanesthetic concentrations of N2O to selectively inhibit formate oxidation by inactivating the enzyme methionine synthetase, thereby reducing the production of tetrahydrofolate (Eells et al. 1996). The 4-6 mM (180-270 µg/mL) concentrations of formate in blood were maintained in this experiment by ip injections of methanol at 2 g/kg in 12 h intervals. Significant reductions in ERG a-wave and b-wave amplitude were not observed until 60 h after methanol administration, although blood formate concentrations reached the plateau level of about 180 µg/mL by 12 h, the earliest time of measurement. Another group of rats in this experiment was treated with higher doses of methanol so that blood formate concentrations increased almost linearly from a baseline of 0.8 mM (36 µg/mL) to 7 mM (315 µg/mL) at 12 h to 15 mM (675 µg/mL) at 60 h after methanol administration. ERGs of these high-dose rats showed reduc-
tions in the b wave as early as 24 h after methanol administration and profound attenuation or complete elimination of the b wave by 48-60 h after methanol administration. Significant but less pronounced reductions in the a wave were seen in the high-dose animals. Histopathologic changes including edema in the outer nuclear layer and vacuolization in the photoreceptors and the bases of retinal pigment epithelium cells were seen in the high-dose but not the low-dose rats at 48 h after methanol administration.
The ocular toxicities of 1% methanol, 0.1% or 1% formaldehyde, or 1% formate were compared in rabbits after a single intravitreal injection of 100 µL (Hayasaka et al. 2001). The eyes were examined ophthalmoscopically at 1 d, 2 d, 1 week (wk), 2 wk, and 1 month (mo) after treatment. Mild inflammation was seen at 1 d and 2 d for all chemicals, but by 1 wk, the eyes treated with methanol and formate appeared nearly normal while those treated with 0.1% formaldehyde showed retinal vessel dilation and those treated with 1% formaldehyde showed mild posterior subcapsular cataract, retinal vessel dilation, and retinal hemorrhages. At 1 mo, animals who received 0.1% or 1% formaldehyde showed mild subcapsular cataract, vessel dilation, and juxtapapillary retinal hemorrhages. Histopathologic study of the eyes showed nearly normal retinas in animals that received vehicle, methanol, or formate, but disorganized ganglion cell layer and outer nuclear layer in eyes that received 0.1% formaldehyde, and markedly disorganized retina in eyes that received 1% formaldehyde. Similar differences were seen in the optic nerves, with formaldehyde-treated eyes showing vacuolizations. The calculated intravitreal concentrations of the treatment chemicals were methanol at 700 µg/mL, formate at 700 µg/mL, and formaldehyde at 70 µg/mL and 700 µg/mL. The report did not consider the potential for dilution of intravitreal chemicals into the systemic circulation. Equally importantly, because these experiments involved a single dose of formate and methanol, the resulting reversible intracellular hypoxia may not have been prolonged enough to produce lasting injury, whereas the high reactivity of formaldehyde would be expected to cause immediate local injury.
From the above observations, it appears that the production of ocular toxicity by formate requires that the formate blood concentration remain elevated (≥180 µg/mL) for at least 24 h. There are not sufficient data, however, to predict with confidence the effects of repeated low doses of formate such that blood concentrations oscillate between toxic and nontoxic concentrations.
The amount of formate needed to achieve a blood formate concentration (180 µg/mL) in humans that has been reported (Eells et al. 1996)
to cause electrophysiologic toxicity (but no histopathology) in retinas of N2O-treated rats for exposures of up to 60 h would require ingestion of a bolus of formate at about 9.1 g. This value was calculated (180 mg/L × 70 kg × 0.72 L/kg = 9,070 mg) assuming that formate is distributed throughout the water that makes up 72% (Lentner 1981) of a 70 kg body, assuming 100% uptake of ingested formate and no metabolism or excretion so that peak concentrations could be achieved and maintained.
Subchronic and Chronic Exposures (≥10 d)
No reports were found describing exposures to formate with durations of 10 d or more.
Genotoxicity
In tests by the National Toxicology Program (NTP), formic acid was found not to be mutagenic to Salmonella typhimurium, with or without metabolic activation (Thompson 1992). No genotoxicity studies on mammalian cells were found.
Reproductive Toxicity
No reports were found describing the reproductive toxicity of formate.
Developmental and Fetal Toxicity
No reports were found describing the developmental or fetal toxicity of formate.
Summary
The published literature on the toxic effects of formate in humans and animals is summarized in Table 8-2. Probably because it is not a common contaminant and because it is a normal product of metabolism, no organizations have set drinking water exposure limits for formate.
TABLE 8-2 Toxicity Summary
|
Dose and Route |
Exposure Duration |
Species and Strain |
Effects |
Reference |
|
Pre-exposure |
||||
|
0.1-0.2 µmole/mL (4.5-9.0 µg/mL) |
Endogenous in blood |
Human |
Endogenous formate concentration in human blood |
Medinsky and Dorman 1995 |
|
Acute and Short Term Exposures |
(≤10 d) |
|
|
|
|
Effects on vision: |
||||
|
0.5 M sodium formate buffer (Na:H=10:1) 140 mg/kg/h iv infusion |
24-48 h |
Maccaca mulatta monkey, male; n = 3/4 |
No pupillary reflexes in response to light |
Martin-Amat et al. 1978 |
|
0.5 M sodium formate buffer (Na:H=10:1) 140 mg/kg/h iv infusion |
39-50 h |
Maccaca mulatta monkey, male; n = 4/4 |
Optic disc edema with normal vascular bed and intracellular edema with intraaxonal swelling |
Martin-Amat et al. 1978 |
|
180-270 µg/mL blood pre- exposed to 1:1 N2O/O2 |
48 h |
Long-Evans rats, male |
NOAEL for retinal dysfunction (ERG changes) with no clinical signs |
Eells et al. 1996 |
|
180-270 µg/mL blood pre- exposed to 1:1 N2O/O2 |
60 h |
Long-Evans rats, male |
LOAEL for retinal dysfunction (ERG changes), but no metabolic acidosis or retinal histopathology |
Eells et al. 1996 |
|
315-675 µg/mL blood pre- exposed to 1:1 N2O/O2 |
48 h |
Long-Evans rats, male |
LOAEL for retinal dysfunction (ERG changes), metabolic acidosis, and retinal histopathology |
Eells et al. 1996 |
|
Dose and Route |
Exposure Duration |
Species and Strain |
Effects |
Reference |
|
Histotoxic hypoxia: |
||||
|
100 mg/kg/d; iv for 5 d |
1/d × 5 d |
New Zealand rabbit, male |
Histotoxicity including calcium aggregates in the kidneys, liver, heart, and brain, and an increased number of myelin figures in cardiac cells |
Liesivuori et al. 1987 |
|
Lethality: |
||||
|
348 µg/mL in blood at admission |
Unknown |
Human |
Shock, metabolic acidosis, hemolysis. |
Verstraete et al. 1989 |
|
1.2 mg/mL in blood at 8 h posthospitalization |
Unknown |
Human |
Coma; severe metabolic acidosis; optic disc edema |
McMartin et al. 1980 |
|
Abbreviations: ERG, electroretinogram; LOAEL, lowest-observed-adverse-effect level; NOAEL, no-observed-adverse-effect level. |
||||
RATIONALE
Table 8-3 lists formate SWEGs for 1-, 10-, 100-, and 1,000-d durations, that were calculated as follows. Acceptable concentrations (ACs) were determined following the guidelines of the National Research Council (NRC 2000). ACs were calculated assuming consumption of 2.8 L of water per day. This includes an average of 800 mL/d of water used to prepare and reconstitute food in addition to 2 L/d for drinking. A value of 70 kg was used as the mass of an average astronaut. A number of conservative assumptions were made in calculating the spacecraft water exposure guidelines (SWEG) values for formate based on the retinal toxicity data reported by Eells et al. (1996). First of all, the experiments reported by Eells et al. involved an artificial-exposure scenario (ip methanol and inhaled N2O) designed as a mechanistic model rather than as a means of extrapolating toxic exposures. Also, although the weight of evidence in both humans and animals, as presented above in the toxicity sections, strongly indicates that retinal toxicity requires 24-60 h of continuous blood formate concentrations > 315 µg/mL, SWEG values were calculated to avoid even brief, transient blood formate concentrations ≥180 µg/mL. A 1-h blood half-life, twice that reported for monkeys (whose sensitivity to formate is similar to humans), iv infused at ≤100 mg/kg, was assumed in calculating SWEG values. It is assumed that an iv-infused bolus of formate at 100 mg/kg would be equivalent to a concentration of 100 mg/kg × 70 kg ÷ 670 mL = 10,448 µg/mL in 670 mL of drinking water. The only reported value found for the half-life of formate in humans was 3.4 h, as seen in methanol-intoxicated individuals (Kerns et al. 2002). This clinical half-life is almost certainly greater than the true half-life of formate, because of continuing production of large amounts of new formate from methanol in the intoxicated individuals.
Spaceflight Effects
None of the reported adverse effects of formate exposures are known to be affected by spaceflight.
1-d SWEG
Because one of the sources of ISS water is supplemented with multiple formate salts that result in a formate concentration of 86 mg/L
TABLE 8-3 Spacecraft Water Exposure Guidelines for Formate
|
Duration |
mg/L |
Target Toxicity |
|
1 d |
10,000 |
Ocular effects (abnormal ERG) |
|
10 d |
2,500 |
Ocular effects (abnormal ERG) |
|
100 d |
2,500 |
Ocular effects (abnormal ERG) |
|
1,000 d |
2,500 |
Ocular effects (abnormal ERG) |
(86 µg/mL) in that source of drinking water, daily consumption of formate from ISS water is ≤ 2.8 L/d × 86 mg/L ≈ ≤ 240 mg/d. No data were found on the rate of absorption of formate from the gut; therefore, the most conservative assumption is instantaneous 100% absorption and distribution into a body water volume of 0.72 L/kg × 70 kg = 50.4 L. The weight of evidence suggests that humans can metabolize formate at about the same rate (half-life = 0.5 h) as monkeys, because they have comparable hepatic concentrations of tetrahydrofolate (Tephly 1991), but the only published half-life for formate in humans is 3.5 h as reported for methanol-intoxicated humans treated with folinic acid (Kerns et al. 2002). Use of such a prolonged half-life in calculations of acceptable (that is, low) formate concentrations, however, would be excessively conservative. To allow for interindividual differences in metabolic rates and temporal fluctuations in hepatic tetrahydrofolate concentrations, it should be sufficient to assume a maximum half-life of 1 h for individuals exposed to formate (not derived as a product of methanol metabolism). We can mathematically model the kinetics of blood formate concentrations by making the following assumptions:
-
The background blood formate concentrations in humans are ≤10 µg/mL. This value represents the mean +2 standard deviations for the highest value reported by Baumann and Angerer (1979) for unexposed healthy humans and agrees well with the higher of the endogenous blood formate concentrations (4.5-9.0 µg/mL) reported by Medinski and Dorman (1995).
-
The half-life of formate in the blood of humans is ≤1 h unless the blood formate is a product of metabolism of ingested methanol.
-
The 2.8 L/d average water consumption is divided into 5 × 400 mL beverages (3 with meals + 1 midafternoon + 1 before bedtime) and 3 × 270 mL for food hydration.
For a one-compartment model, the concentration of formate in the
blood resulting from ingestion of water containing formate can be calculated using the following equation:
µg formate after ingestion per mL blood = µg formate before ingestion per mL blood + (µg formate per mL in water × mL of water ingested)/mL blood.
The amount of formate in the blood at t h after ingestion, assuming a 1-h half-life, can be calculated using the following equation:
Figure 8-1 illustrates the predicted kinetics of formate concentrations in the blood over a >5-d period in crewmembers ingesting 2.8 L/d of U.S.-provided ISS water (containing formate at 86 µg/mL) fractionated over a
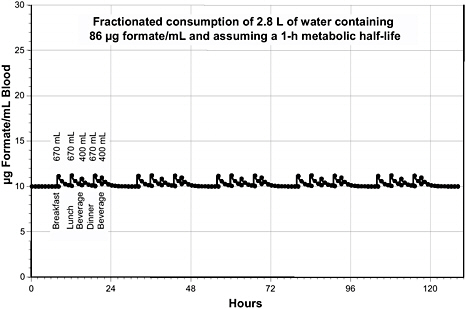
FIGURE 8-1 Calculated blood formate kinetics for a 70 kg person ingesting water containing formate at 86 µg/mL. The daily pattern of ingestion was assumed to be 670 mL at 8 h (for example, 8:00 a.m.), 670 mL at 12 h, 400 mL at 15 h, 670 mL at 19 h, and 400 mL at 21 h. The metabolic half-life was assumed to be 1 h.
conservatively low number of portions (that is, high volume of water per portion). Consumption of up to 670 mL of this water increases the peak blood concentrations by ~1 µg/mL, but the concentrations return to baseline between ingested portions. Baseline concentrations of blood formate have been reported by various authors to range from 4.9 to 10.3 µg/mL (Bouchard et al. 2001); thus, a transient increase of ~1 µg/mL would be highly unlikely to cause any adverse effects.
To estimate an AC of formate in drinking water for a 1-d exposure duration, one can select a concentration that is below the lowest concentration reported to cause minimal effects. As noted earlier, visual-system damage in humans has been associated with prolonged exposures (>24 h) to blood formate concentrations > 315 µg/mL. Eells et al. (1996) reported that blood formate concentrations of 180-279 µg/mL in rats (treated with N2O to make their response to formate similar to that of humans and repeatedly ip injected with methanol) were a NOAEL for retinal dysfunction, histopathology, and metabolic acidosis for exposures of 48 h and a LOAEL for retinal dysfunction, still without histopathology or metabolic acidosis, for exposures of 60 h. ERGs of high-dose (315 µg/mL at 12 h up to 675 µg/mL at 60 h) rats showed reductions in the b wave as early as 24 h after methanol administration and profound attenuation or complete elimination of the b wave by 48-60 h after methanol administration. Thus, the sensitivity of rats in Eells et al.’s 1996 study to formate-induced ocular toxicity is similar to that of humans, and concentrations of formate in drinking water that do not increase blood formate concentrations above 180 µg/mL should be acceptable for humans. Figure 8-2 shows that drinking water containing formate at 10,000 µg/mL (10,000 ppm or 10 g/L) meets this goal because peak blood formate concentrations are about 150 µg/mL. After ingestion of each portion of water, the blood formate concentration decreases rapidly, returning to baseline within 20 h after the first ingestion.
The weight of evidence strongly indicates that the ocular toxicity of formate is caused by prolonged cellular hypoxia and that mild ocular symptoms (for example, blurred vision) can be reversed if the blood concentration of formate is promptly reduced. Thus, a 1-d exposure to formate concentrations that transiently approach (but do not reach) concentrations that are minimally toxic to the retina when maintained for 60 h would not be expected to cause any ocular toxicity.
As illustrated in Figure 8-2, continued exposure to formate at 10,000 µg/mL drinking water for several days results in a repetition each day of the same pattern of blood formate concentrations seen on the first day and of formate concentrations returning to baseline values before the
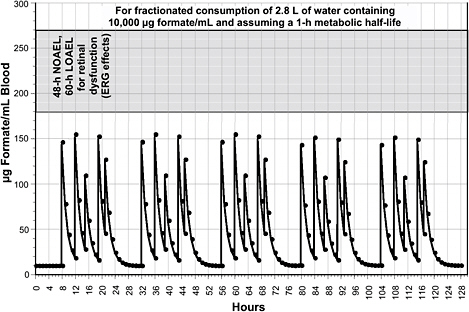
FIGURE 8-2 Calculated blood formate kinetics for a 70-kg person ingesting water containing formate at 10,000 µg/mL. The pattern of ingestion was assumed to be 670 mL at 8 h (for example, 8:00 a.m.), 670 mL at 12 h, 400 mL at 15 h, 670 mL at 19 h, and 400 mL at 21 h. The metabolic half-life was assumed to be 1 h.
first exposure of each 24-h period. At 10,000 µg/mL in the water, the maximum blood formate concentration achieved is about 150 µg/mL, which is below the lower borderline of the concentration range reported by Eells et al. (1996) to be a LOAEL for ocular toxicity for a continuous 60-h exposure.
10-d, 100-d, and 1,000-d SWEGs
Experimental data on which one could base the calculation of a safe concentration of formate in drinking water for exposure durations of ≥ 10 d are unavailable, so one must extrapolate from the available data on shorter-term exposures based on reasonable assumptions. Although the kinetics of the ocular toxicity of formate is, to some extent, dependent on both the concentration of formate in the blood and the duration of exposure, the weight of evidence indicates that there is an effective threshold concentration of formate below which ocular toxicity will not occur re-
gardless of the duration of exposure. This can also be inferred from the fact that formate is present at concentrations up to about 10 µg/mL in normal blood as a product of metabolism.
One approach to establishing longer-term SWEGs is to determine a concentration of formate that would cause only minimal inhibition of the mitochondrial cytochrome c oxidase whose inactivity leads to the retinal edema responsible for clinical ocular toxicity. Based on the lower limit of the Ki range (45-1,350 µg/mL) for formate determined by Nicholls (1976), an intracellular formate concentration as low as 45 µg/mL can reversibly produce about 50% inhibition of the enzymatic activity of the cytochrome c oxidase complex. The formate concentration in drinking water that would produce a blood formate concentration of 45 µg/mL can be calculated, assuming a drinking water volume of 670 mL and a body water volume of 50.4 L as follows:
Peak concentrations of 45 µg/mL blood, such as would be produced by the ingestion of water containing formate at 2,500 µg/mL, would be transient, however (see Figure 8-3). Thus, the level of inhibition of cytochrome c oxidase would peak at about 50% and decline to near baseline before the next exposure (drink). Such a pattern of brief, partial inhibition of cytochrome c oxidase is not expected to result in any clinical or even histologic adverse effects. Therefore, the ACs and SWEGs for all exposure durations longer than 1 d can be set at 2,500 µg/mL in drinking water (see Table 8-4 for a summary of ACs and SWEGs):
10-d SWEG = 2,500 µg/mL
100-d SWEG = 2,500 µg/mL
1,000-d SWEG = 2,500 µg/mL
RECOMMENDATIONS
To refine the data on which the longer-term SWEGs are based, studies should be performed to determine the minimum level of inhibition of cytochrome c oxidase that leads to detectable ocular toxicity and the concentration of formate that will produce that level of inhibition.
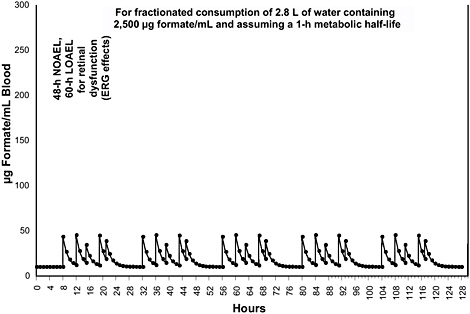
FIGURE 8-3 Calculated blood formate kinetics for a 70-kg person ingesting water containing formate at 2,500 µg/mL. The pattern of ingestion was assumed to be 670 mL at 8 h (for example, 8:00 a.m.), 670 mL at 12 h, 400 mL at 15 h, 670 mL at 19 h, and 400 mL at 21 h. The metabolic half-life was assumed to be 1 h.
TABLE 8-4 Acceptable Concentrations (ACs)
|
End Point |
Exposure Data |
Species and Reference |
Uncertainty Factors |
Acceptable Concentrations (mg/L) |
|||||||
|
Inter-individual |
To NOAEL |
Inter-species |
Exposure Time |
Space-flight |
1 d |
10 d |
100 d |
1,000 d |
|||
|
48-h NOAEL for ERG effects |
Injections (ip) of methanol to produce blood concentrations of 180-270 µg/mL |
Rat, male (Eells 1996) |
1 |
1 |
1 |
1 |
1 |
10,000 |
— |
— |
— |
|
60-h LOAEL for ERG effects |
Injections (ip) of methanol to produce blood concentrations of 180-270 µg/mL |
Rat, male (Eells 1996) |
1 |
1 |
1 |
1 |
1 |
— |
2,500 |
2,500 |
2,500 |
|
SWEG |
|
|
|
|
|
|
|
10,000 |
2,500 |
2,500 |
2,500 |
REFERENCES
Barceloux, D.G., G.R. Bond, E.P. Krenzelok, H. Cooper, J.A. Vale, and American Academy of Clinical Toxicology Ad Hoc Committee on the Treatment Guidelines for Methanol Poisoning. 2002. American Academy of Clinical Toxicology practice guidelines on the treatment of methanol poisoning. J. Toxicol. Clin. Toxicol. 40(4):415-446.
Baumann, K., and J. Angerer. 1979. Occupational chronic exposure to organic solvents, VI. Formic acid concentration in blood and urine as an indicator of methanol exposure. Int. Arch. Occup. Environ. Health 42:241-249.
Black, K.A., J.T. Eells, P.E. Noker, C.A. Hawtrey, and T.R. Tephly. 1985. Role of hepatic tetrahydrofolate in the species difference in methanol toxicity. Proc. Nat. Acad. Sci. U.S.A. 82(11):3854-3858.
Bouchard, M., R.C. Brunet, P.O. Droz, and G. Carrier. 2001. A biologically based dynamic model for predicting the disposition of methanol and its metabolites in animals and humans. Toxicol. Sci. 64(2):169-184.
Clay, K.L., R.C. Murphy, and W.D. Watkins. 1975. Experimental methanol toxicity in the primate. Toxicol. Appl. Pharmacol. 34(1):49-61.
Dorland, W.A.N. 1988. Dorland's Illustrated Medical Dictionary. Philadelphia: W.B. Saunders Co.
Eells, J.T., M.M. Salzmann, M.F. Lewandowski, and T.G. Murray. 1996. Formate-induced alterations in retinal function in methanol-intoxicated rats. Toxicol. Appl. Pharmacol. 140(1):58-69.
Erecinska, M., and D. Wilson. 1980. Inhibitors of cytochrome c oxidase. Pharmacol. Ther. 8:1-20.
Erlanson, P., H. Fritz, K. Hagstam, B. Liljenberg, N. Tryding, and G. Voigt. 1965. Severe methanol intoxication. Acta Med. Scand. 17:393-408.
Galinsky, R.E., and G. Levy. 1981. Dose- and time-dependent elimination of acetaminophen in rats: Pharmacokinetic implications of cosubstrate depletion. J. Pharmacol. Exp. Ther. 219(1):14-20.
Hayasaka, Y., S. Hayasaka, and Y. Nagaki. 2001. Ocular changes after intravitreal injection of methanol, formaldehyde, or formate in rabbits. Pharmacol. Toxicol. 89(2):74-78.
Hayreh, M.S., S.S. Hayreh, G. Baumbach, P. Cancilla, G. Martin-Amat, and T. R. Tephly. 1980. Ocular toxicity of methanol: An experimental study. Pp. 35-53 in Neurotoxicity of the Visual System, W. Merigan, and B. Weiss, eds. New York: Raven Press.
Hayreh, M.S., S.S. Hayreh, G.L. Baumbach, P. Cancilla, G. Martin-Amat, T.R. Tephly, K.E. McMartin, and A.B. Makar. 1977. Methyl alcohol poisoning. III. Ocular toxicity. Arch. Ophthalmol. 95:1851-1858.
HSDB (Hazardous Substances Data Bank). 2006. Formic Acid. U.S. National Library of Medicine. [Online]. Available at: http://toxnet.nlm.nih.gov/cgi-bin/sis/search/f?./temp/~Vl8xGe:2 [access September 5, 2006].
Jacobsen, D., and K.E. McMartin. 1986. Methanol and ethylene glycol poison-
ings. Mechanism of toxicity, clinical course, diagnosis and treatment. Med. Toxicol. 1(5):309-334.
Jacobsen, D., R. Webb, T.D. Collins, and K.E. McMartin. 1988. Methanol and formate kinetics in late diagnosed methanol intoxication. Med. Toxicol. Adverse Drug Exp. 3(5):418-423.
Johlin, F.C., C.S. Fortman, D.D. Nghiem, and T.R. Tephly. 1987. Studies on the role of folic acid and folate-dependent enzymes in human methanol poisoning. Mol. Pharmacol. 31(5):557-561.
Johlin, F.C., E. Swain, C. Smith, and T.R. Tephly. 1989. Studies on the mechanism of methanol poisoning: Purification and comparison of rat and human liver 10-formyltetrahydrofolate dehydrogenase. Mol. Pharmacol. 35(6):745-750.
Kerns, W.N., C. Tomaszieski, K.E. McMartin, M. Ford, J. Brent; META Study Group. Methylpyrazole for Toxic Alcohols. 2002. Formate kinetics in methanol poisoning. J. Toxicol. Clin. Toxicol. 40(2):137-143.
Koivusalo, M. 1970. Methanol. Pp. 564-605 in International Encyclopedia of Pharmacology and Therapeutics, J. Tremolieres, ed. New York: Pergamon Press.
Lentner, C., ed. 1981. Geigy Scientific Tables. West Caldwell, NJ: Ciba-Geigy Corporation.
Liesivuori, J. 1986. Slow urinary elimination of formic acid in occupationally exposed farmers. Ann. Occup. Hyg. 30(3):329-333.
Liesivuori, J., V.M. Kosma, A. Naukkarinen, and H. Savolainen. 1987. Kinetics and toxic effects of repeated intravenous dosage of formic acid in rabbits. Br. J. Exp. Pathol. 68(6):853-861.
Liesivuori, J., and H. Savolainen. 1991. Methanol and formic acid toxicity: Biochemical mechanisms. Pharmacol. Toxicol. 69(3):157-163.
Lund, A. 1948. Excretion of methanol and formic acid in man after methanol consumption. Acta Pharmacol. Toxicol. (Copenh.) 4:205-212.
Malorny, G. 1969. Low-dose exposure of humans to sodium formate and formic acid [in German]. Z. Ernahrungswiss. 9:340-348.
Martin-Amat, G., K.E. McMartin, S.S. Hayreh, M.S. Hayreh, and T.R. Tephly. 1978. Methanol poisoning: Ocular toxicity produced by formate. Toxicol. Appl. Pharmacol. 45(1):201-208.
Martin-Amat, G., T.R. Tephly, K.E. McMartin, A.B. Makar, M.S. Hayreh, S.S. Hayreh, G. Baumbach, and P. Cancilla. 1977. Methyl alcohol poisoning. II. Development of a model for ocular toxicity in methyl alcohol poisoning using the rhesus monkey. Arch. Ophthalmol. 95(10):1847-1850.
McMartin, K.E., J.J. Ambre, and T.R. Tephly. 1980. Methanol poisoning in human subjects. Role for formic acid accumulation in the metabolic acidosis. Am. J. Med. 68(3):414-418.
McMartin, K.E., G. Martin-Amat, A.B. Makar, and T.R. Tephly. 1977. Methanol poisoning. V. Role of formate metabolism in the monkey. J. Pharmcol. Exp. Therap. 201:564-572.
Medinsky, M.A., and D.C. Dorman. 1995. Recent developments in methanol toxicity. Toxicol. Lett. 82-83:707-711.
Moore, D.F., A.M. Bentley, S. Dawling, A.M. Hoare, and J.A. Henry. 1994. Folinic acid and enhanced renal elimination in formic acid intoxication. J. Toxicol. Clin. Toxicol. 32(2):199-204.
Naik, R.B., W.P. Stephens, D.J. Wilson, A. Walker, and H.A. Lee. 1980. Ingestion of formic acid-containing agents—report of three fatal cases. Postgrad. Med. J. 56:451-456.
Nicholls, P. 1975. Formate as an inhibitor of cytochrome c oxidase. Biochem. Biophys. Res. Commun. 67:610-616.
Nicholls, P. 1976. The effects of formate on cytochrome aa3 and on electron transport in the intact respiratory chain. Biochem. Biophys. Acta 430:13-29.
NRC (National Research Council). 2000. Methods for Developing Spacecraft Water Exposure Guidelines. Washington, DC: National Academy Press.
Seisjo, B. 1992. Pathophysiology and treatment of focal cerebral ischemia. Part I: Pathophysiology. J. Neurosurg. 77:169-184.
Tephly, T.R. 1991. The toxicity of methanol. Life Sci. 48(11):1031-1041.
Thompson, M. 1992. NTP technical report on the toxicity studies of formic acid (CAS No. 64-18-6) administered by inhalation to F344/N rats and B6C3F1 mice. Toxic. Rep. Ser. 19:1-D3.
Verstraete, A.G., D.P. Vogelaers, J.F. van den Bogaerde, F.A. Colardyn, C.M. Ackerman, and W.A. Buylaert. 1989. Formic acid poisoning: Case report and in vitro study of the hemolytic activity. Am. J. Emerg. Med. 7(3):286-290.
Westphal, F., G. Rochholz, S. Ritz-Timme, N. Bilzer, H. W. Schutz, and H. J. Kaatsch. 2001. Fatal intoxication with a decalcifying agent containing formic acid. Int. J. Legal Med. 114(3):181-185.