Optical Imaging for In Vivo Assessment of Tissue Pathology
REBEKAH A. DREZEK, NAOMI J. HALAS, AND JENNIFER WEST
Rice University
Houston, Texas
For hundreds of years, optical imaging at both macroscopic and microscopic levels has been used as a tool to aid clinicians in establishing a diagnosis. Pathologists routinely use a simple compound microscope to examine stained and sectioned tissue at the microscopic level to determine a definitive diagnosis of cancer. At a macroscopic level, clinicians often rely on observed colors as indicators of physiologic status, for instance, yellow skin is associated with jaundice, blue or purple hues with cyanosis, and red with inflammation. In each of these examples, the human eye gathers qualitative information about a patient’s status based on either the gross visual appearance of tissue or a microscopic evaluation of stained tissue sections or cytologic samples.
Despite the clear importance of these qualitative optical approaches in current medical practice, these strategies are only sensitive to a highly limited subset of the wide array of optical events that occur when light interacts with biologic tissue. In fact, there is a compelling need for more quantitative optical imaging strategies that can probe tissue physiology in vivo in real time with high resolution at relatively low cost. In this talk, I describe emerging technologies for quantitative optical imaging and the use of these technologies to diagnose and monitor cancer. Particular emphasis is placed on how advances in nanobiotechnology are leading to new approaches to in vivo medical diagnostics. Because another talk in this session will consider luminescence-based nanomaterials (i.e., quantum dots), the discussion here is focused on nanomaterials, particularly gold-based materials, that provide a scatter-based or ab-
sorption-based optical signal. The general biocompatibility of gold, coupled with extensive prior medical applications of gold colloid, suggests a more straightforward regulatory path toward ultimate clinical use than for many other nanomaterials currently under development.
THE ROLE OF NANOTECHNOLOGY IN OPTICAL IMAGING OF CANCER
For more than 50 years, cancer was the second leading cause of death in the United States, accounting for more than 25 percent of deaths in the population. However, in the past two years, death from cancer has exceeded deaths from heart attacks, and cancer has become the primary cause of deaths in the United States. Early detection is recognized as a highly effective approach to reducing the morbidity and mortality associated with cancer. When diagnosed at an early stage when the cancer is still localized and risk for metastasis is low, most cancers are highly treatable and prognoses are favorable. However, if cancer is not diagnosed until metastasis to distant sites has already occurred, five-year survival is poor for a wide variety of organ sites (Table 1) (American Cancer Society, 2006). Thus, there is a significant clinical need for novel methods of early detection and treatment with improved sensitivity, specificity, and cost effectiveness.
In recent years, a number of groups have demonstrated that photonics-based technologies can be valuable in addressing this need. Optical technologies promise high-resolution, noninvasive functional imaging of tissue at competitive costs. However, in many cases, these technologies are limited by the inherently weak optical signals of endogenous chromophores and the subtle spectral differences between normal and diseased tissue.
In the past several years, there has been increasing interest in combining emerging optical technologies with novel exogenous contrast agents designed to probe the molecular-specific signatures of cancer to improve the detection limits and clinical effectiveness of optical imaging. For instance, Sokolov et al. (2003) recently demonstrated the use of gold colloid conjugated to antibodies to the epidermal growth factor receptor as a scattering contrast agent for biomolecular
TABLE 1 Cancer Survival at Five Years as a Function of Stage at Diagnosis
|
Organ |
Localized |
Regional |
Distant |
|
Prostate |
~100% |
>85% |
30% |
|
Oral |
>80% |
50% |
25% |
|
Breast |
>90% |
80% |
25% |
optical imaging of cervical cancer cells and tissue specimens. In addition, optical imaging applications of nanocrystal bioconjugates have been described by multiple groups, including Bruchez et al. (1998), Chan and Nie (1998), and Akerman et al. (2002). More recently, interest has developed in the creation of nanotechnology-based platform technologies that can couple molecular-specific early detection strategies with appropriate therapeutic intervention and monitoring capabilities.
METAL NANOSHELLS
Metal nanoshells are a new type of nanoparticle composed of a dielectric core, such as silica, coated with an ultrathin metallic layer, typically gold. Gold nanoshells have physical properties similar to gold colloid, particularly a strong optical absorption due to gold’s collective electronic response to light. The optical absorption of gold colloid yields a brilliant red color, which has been used effectively in consumer-related medical products, such as home pregnancy tests. In contrast, the optical response of gold nanoshells depends dramatically on the relative size of the nanoparticle core and the thickness of the gold shell.
By varying the relative thicknesses of the core and shell, the color of gold nanoshells can be varied across a broad range of the optical spectrum that spans the visible and near-infrared spectral regions (Brongersma, 2003; Oldenburg et al., 1998). Gold nanoshells can be made either to absorb or scatter light preferentially by varying the size of the particle relative to the wavelength of the light at their optical resonances. Figure 1 shows a Mie scattering plot of the nanoshell plasmon resonance wavelength shift as a function of nanoshell composition for a 60 nm core gold/silica nanoshell. In this figure, the core and shell of the nanoparticles are shown to relative scale directly beneath their corresponding optical resonances. Figure 2 shows a plot of the core/shell ratio versus resonance wavelength for a silica core/gold shell nanoparticle (Oldenburg et al., 1998). The extremely agile “tunability” of optical resonance is a property unique to nanoshells—in no other molecular or nanoparticle structure can the resonance of the optical absorption properties be “designed” as systematically.
Halas and colleagues have completed a comprehensive investigation of the optical properties of metal nanoshells (Averitt et al., 1997) and achieved quantitative agreement between Mie scattering theory and the experimentally observed optical-resonant properties. Based on this success, it is now possible to design gold nanoshells predictively with the desired optical-resonant properties and then to fabricate the nanoshell with the dimensions and nanoscale tolerances necessary to achieve these properties (Oldenburg et al., 1998). The synthetic protocol developed for the fabrication of gold nanoshells is very simple in concept:
-
Grow or obtain silica nanoparticles dispersed in solution.
-
Attach very small (1–2 nm) metal “seed” colloids to the surface of the
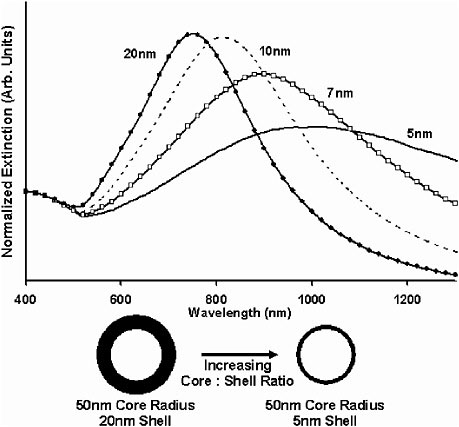
FIGURE 1 Optical resonances of gold-shell, silica-core nanoshells as a function of the core/shell ratio. Respective spectra correspond to the nanoparticles shown below them. Source: Loo et al., 2004. Reprinted with permission.
-
nanoparticles via molecular linkages; the seed colloids cover the dielectric nanoparticle surfaces with a discontinuous metal colloid layer.
-
Grow additional metal onto the “seed” metal colloid adsorbates via chemical reduction in solution.
This approach has been used successfully to grow both gold and silver metallic shells onto silica nanoparticles. Various stages in the growth of a gold metallic shell onto a functionalized silica nanoparticle are shown in Figure 3. Based on the core/shell ratios that can be achieved with this protocol, gold nanoshells with optical resonances extending from the visible region to approximately 3 µm in the infrared region can currently be fabricated. This spectral region includes the 800–1,300 nm “water window” of the near infrared, a region of high physiological transmissivity that has been demonstrated as the spectral region best suited for optical bioimaging and biosensing. The optical
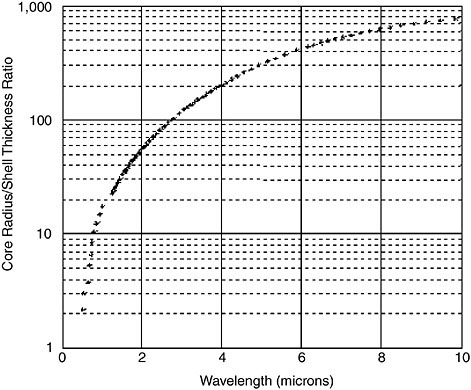
FIGURE 2 Core/shell ratio as a function of resonance wavelength for gold/silica nanoshells. Source: Loo et al., 2004. Reprinted with permission.

FIGURE 3 Transmission electron microscope images of gold/silica nanoshells during shell growth. Source: Loo et al., 2004. Reprinted with permission.
properties of gold nanoshells, coupled with their biocompatibility and ease of bioconjugation, render them highly suitable for targeted bioimaging and therapeutic applications. By controlling the physical parameters of the nanoshells, it is possible to engineer nanoshells that primarily scatter light, which is desirable for many imaging applications, or alternatively, to design nanoshells that are strong absorbers, which is desirable for photothermal-based therapy applications.
Because the same chemical reaction is used to grow the metal layer of gold nanoshells as is used to synthesize gold colloid, the surfaces of gold nanoshells are virtually chemically identical to the surfaces of the gold nanoparticles universally used in bioconjugate applications. Gold colloid was first used in biological applications in 1971 when Faulk and Taylor (1971) invented the immunogold staining procedure. Since then, the labeling of targeting molecules, especially proteins, with gold nanoparticles has revolutionized the visualization of cellular or tissue components by electron microscopy. The optical and electron-beam contrast qualities of gold colloid have provided excellent detection qualities for immunoblotting, flow cytometry, hybridization assays, and other techniques. Conjugation protocols exist for the labeling of a broad range of biomolecules with gold colloid, such as protein A, avidin, streptavidin, glucose oxidase, horseradish peroxidase, and IgG.
The vast prior history of gold-colloid-based materials has greatly facilitated the development of biomedical applications of newer gold-based nanoparticles. Figure 4 shows one example of the type of medical application enabled by using this class of material. The figure shows an in vitro proof-of-principle example of
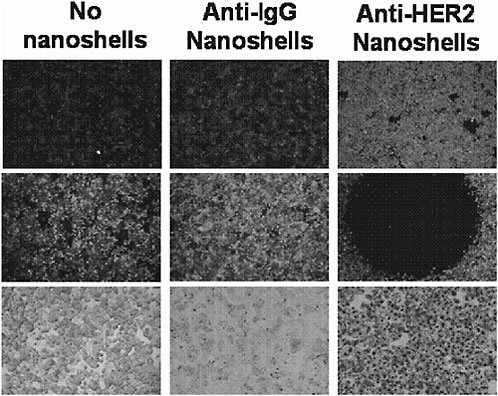
FIGURE 4 Dual imaging/therapy nanoshell bioconjugates. Source: Loo et al., 2005. Copyright 2005 American Chemical Society. Reprinted with permission.
gold nanoshells designed to simultaneously scatter (for imaging) and absorb (for photothermal therapy) near infrared light. Here, scattering and absorbing near infrared nanoshells are conjugated to an antibody for a common breast cancer surface marker. This enables both “lighting up” and, if desired, destroying cells that express this marker without harming other cells. In Figure 4, the top row shows scatter-based imaging of carcinoma cells. By increasing the laser power, it is possible to destroy cells selectively as shown in the middle row, which shows the viability of cells after laser irradiation. The left column shows a no-nanoshell (cells-only) control, and the middle column shows a nonspecific antibody control. The right column indicates successful imaging of cells followed by photothermal destruction (black circle = laser irradiation spot) based on the presence of a chosen marker.
Although in vitro demonstrations can be completed using simple microscopes, in vivo use of this type of nanomaterial requires coupling the development of appropriate materials with the development of optical devices that enable imaging of these materials in tissue. By careful design of these optical systems, it is possible to generate multiple order of magnitude improvements in optical contrast using nanomaterial imaging agents, which could potentially lead to the detection of much smaller lesions. In addition to examples from our own group, work is being done by other laboratories using a variety of other gold-based nanomaterials. In all cases, the move from in vitro cell-based demonstrations to in vivo clinical use is enabled by rapid developments in photonics-based strategies for real-time, low-cost in vivo imaging.
SUMMARY
Numerous research groups throughout the country are leveraging emerging techniques in optical imaging and nanotechnology to develop powerful new approaches for detecting molecular-specific signatures of precancers and early cancers. These groups are developing several classes of ultrabright contrast agents that strongly scatter and/or absorb at tunable wavelengths throughout the visible and near-infrared spectral bands, as well as methods of targeting these agents to molecular markers of neoplasia. They are demonstrating the efficacy of these agents in biological samples of progressively increasing complexity. These initial efforts will certainly be expanded in future studies.
Ultimately, the use of ultrabright contrast agents will extend the detection limits of optical technologies, increasing their sensitivity and specificity and promoting improved screening and detection of early lesions. We believe there is tremendous potential for synergy between the rapidly developing fields of biophotonics and nanotechnology. Combining the tools of these fields—together with the latest advances in understanding of the molecular origins of cancer—will offer a fundamentally new approach to the detection of cancer, a disease responsible for more than one-quarter of all deaths in the United States today.
REFERENCES
Akerman, M.E., W. Chan, P. Laakkonen, S.N. Bhatia, and E. Ruoslahti. 2002. Nanocrystal targeting in vivo. Proceedings of the National Academy of Sciences 99: 12617–12621.
American Cancer Society. 2006. Cancer Facts and Figures 2006. Available online at: http://www.cancer.org/docroot/STT/content/STT_1x_Cancer_Facts__Figures_2006.asp.
Averitt, R.D., D. Sarkar, and N.J. Halas. 1997. Plasmon resonance shifts of Au-coated Au2S nanoshells: insights into multicomponent nanoparticles growth. Physiology Review Letters 78: 4217–4220.
Brongersma, M.L. 2003. Nanoshells: gifts in a gold wrapper. Nature Materials 2: 296–297.
Bruchez, M., M. Moronne, P. Gin, S. Weiss, and A.P. Alivisatos. 1998. Semiconductor labels as fluorescent biological labels. Science 281: 2013–2016.
Chan, W.C.W., and S. Nie. 1998. Quantum dot bioconjugates for ultrasensitive nonisotopic detection. Science 281: 2016–2018.
Faulk, W.T., and G. Taylor. 1971. An immunocolloid method for the electron microscope. Immunochemistry 8: 1081–1083.
Loo, C., L. Hirsch, J. Barton, N. Halas, J. West, and R. Drezek. 2004. Nanoshell-enabled photonics-based cancer imaging and therapy. Technology in Cancer Research and Treatment 3: 33–40.
Loo, C., A. Lowery, N. Halas, J. West, and R. Drezek. 2005. Immunotargeted nanoshells for integrated imaging and therapy of cancer. Nano Letters 5: 709–711.
Oldenburg, S.J., R.D. Averitt, S.L. Westcott, and N.J. Halas. 1998. Nanoengineering of optical resonances. Chemical Physics Letters 288: 243–247.
Sokolov, K., M. Follen, J. Aaron, I. Pavlova, A. Malpica, R. Lotan, and R. Richards-Kortum. 2003. Real-time vital optical imaging of precancer using anti-epidermal growth factor receptor antibodies conjugated to gold nanoparticles. Cancer Research 63: 1999–2004.








