4
Targeted Radionuclide Therapy
Modern cancer therapy has proven partially successful in treating and prolonging the lives of patients with many common types of cancer. This limited success is due in part to the relative lack of specificity seen for many of the primary classes of anticancer agents and cytotoxic technologies in current medical practice. For most current cancer treatment options available today (e.g., conventional chemotherapy, external radiotherapy), the approach has been to destroy populations of cells that show uncontrolled growth. This focus on nonspecific cell division implies that the treatment will often be nonselective, damaging rapidly dividing nontumor cells, such as those in the gut. However, in recent years, there has been a much greater emphasis on “targeted therapies” that are designed to damage only the cancerous cells. There are currently hundreds of new pathway-targeted anticancer agents undergoing phase II and phase III clinical trials. Targeted radionuclide therapy is just one type within the category of “targeted therapies.” At present, effective targeted radiopharmaceutical therapeutics have been developed and validated for a few tumor types, such as malignant lymphoma; for most other tumor types, the older nonspecific types of cancer treatments are still the dominant form of therapy.
This chapter describes the unique promise of targeted radionuclide therapy (Sidebar 2.2) and highlights what is needed to facilitate the translation of new targeted radionuclide therapies into clinical practice. To obtain information needed for this chapter, the committee consulted with leaders in the fields of radiation oncology, nuclear medicine, oncology, and chemistry from both industry and academia to identify the most critical needs
for advancing targeted radionuclide therapy. The issues considered included the impact of deficiencies in radionuclide availability, trained personnel, and funding of the field.
The chapter is divided into the following sections:
-
Background (4.1),
-
Recommendations (4.5), and
-
Conclusions (4.6).
4.1
BACKGROUND
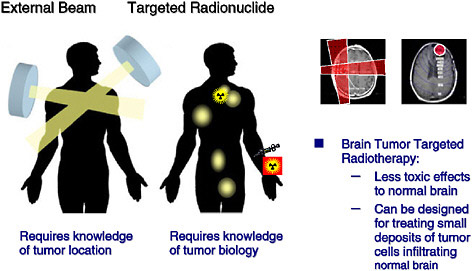
Radiation therapy uses ionizing radiation to kill cancer cells and shrink tumors by damaging the cells’ DNA, thereby stopping these cells from continuing to grow and divide. The most common way of exposing cancer patients to radiation is through external radiation therapy. With this approach, only a limited area of the body is irradiated by delivering a beam of high-energy x rays to the main tumor. Targeted radionuclide therapy, on the other hand, is like chemotherapy, because it is a systemic treatment; it uses a molecule labeled with a radionuclide to deliver a toxic level of radiation to disease sites. Unlike tumor-directed drugs and toxins, which kill only the directly targeted cells, a unique feature of radionuclides is that they can exert a “bystander” or “crossfire” effect (Figure 4.1), potentially destroying adjacent tumor cells even if they lack the specific tumor-associated antigen or receptor. In addition, a systemically administered targeted radiotherapeutic that combines the specificity of cancer cell targeting with the known antitumor effects of ionizing radiation has the potential to simultaneously eliminate both a primary tumor site and cancer that has spread throughout the body, including malignant cell populations undetectable by diagnostic imaging. Figure 4.2 illustrates and contrasts the differences between direct and bystander killing of tumors.
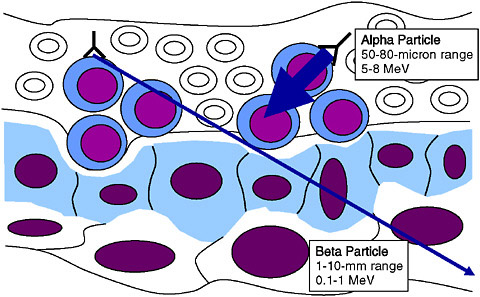
In targeted radionuclide therapy, the biological effect is obtained by energy absorbed from the radiation emitted by the radionuclide. Whereas the radionuclides used for nuclear medicine imaging emit gamma rays, which can penetrate deeply into the body, the radionuclides used for targeted radionuclide therapy must emit radiation with a relatively short path length. There are three types of particulate radiation of consequence for targeted radionuclide therapy—beta particles, alpha particles,1 and Auger
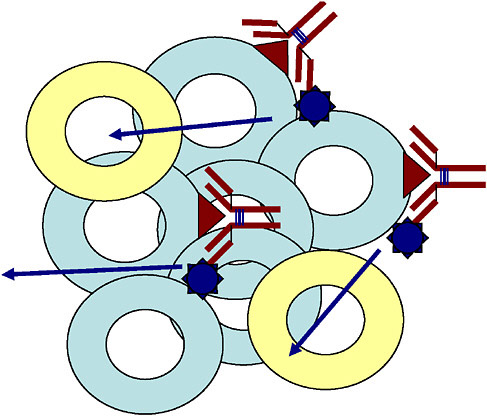
FIGURE 4.1 Schematic illustrating bystander effect for a labeled antibody (yellow circles = normal cells, light blue circles = tumor cells, the red triangles = antigen expressed on tumor cells, red Y-structure = antibody labeled with a radionuclide).
electrons2—which can irradiate tissue volumes with multicellular, cellular and subcellular dimensions (Figure 4.3), respectively. In some cases, mixed emitters are used to allow both imaging and therapy with the same radionuclide (e.g., the mixed beta/gamma emitter iodine-131). Moreover, within each of these categories, there are multiple radionuclides with a variety of tissue ranges, half-lives, and chemistries, offering the attractive possibility of tailor-making the properties of a targeted radionuclide therapeutic to the needs of an individual patient. The further development of this field is driven by the desire to move away from nonspecific toxic therapies commonly used in oncology and toward much less toxic targeted treatments, which impact only the targeted tissues.
|
SIDEBAR 4.1 Non-Hodgkin’s Lymphoma Lymphomas are a heterogeneous group of malignancies of the lymphatic system, which is a network of lymph vessels that is an integral part of the immune system. Other parts of the lymphatic system include the lymph nodes, tonsils, spleen, and thymus. Broadly, lymphomas are classified into two categories: Hodgkin’s lymphoma (also known as Hodgkin’s disease) and non-Hodgkin’s lymphoma. Non-Hodgkin’s lymphoma accounts for 85 percent of all lymphomas, and within this subgroup, there are many types of non-Hodgkin’s lymphoma that can be classified by cell type (B-cell versus T-cell) or by level of aggressiveness. |
At present, there are two commercially approved radioimmunotherapy agents, yttrium-90 ibritumomab tiuxetan (Zevalin®; Biogen-Idec Pharmaceuticals, San Diego, CA, approved by the Food and Drug Administration [FDA] in 2002) and iodine-131 tositumomab (BEXXAR®, GlaxoSmithKline, Philadelphia, PA, approved in 2003), both of which are used to treat indolent B-cell lymphoma (Sidebar 4.1) and related cancers. Both compounds target a B-cell3 restricted lineage protein (i.e., CD20 surface antigen) expressed on B-cells, which is observed in more than 95 percent of patients with B-cell malignancies, and produce excellent clinical results: on the order of 60—80 percent overall response and 20—40 percent complete response rates4 for patients with relapsed, recurrent, or refractory5 indolent6 B-cell lymphoma (Pohlman et al. 2006, Davies et al. 2004, Press 2003, Witzig et al. 2002). Although the radiobiologic principles and dosimetric requirements for the effective use of these two agents are still not fully understood, the clinical response shows that a single cycle of treatment with either of these two radiopharmaceuticals can result in essentially the same level of tumor response as multiple cycles of conventional chemotherapy, generally with a fraction of the toxicity (Macklis 2004).
In general, the use of both compounds involves a sequence of diagnostic and therapeutic sessions extending over about 7 to 10 days, where a com-
bination of medications is given and their biodistribution imaged. A patient treated with Zevalin® is first given a dose of nonradioactive antibody intravenously, followed by an infusion of a monoclonal antibody labeled with a gamma-emitting radionuclide (indium-111) as a tracer. The patient then undergoes an imaging study using a gamma camera (see Figure 4.4) that allows a physician to evaluate how the agent is distributed and cleared in the body. Finally, the patient is given a therapeutic dose of the monoclonal antibody radiolabeled with a beta-emitting radionuclide (yttrium-90) intravenously. Figure 4.5 illustrates how tumor response can be evaluated in a lymphoma patient treated with Zevalin® through the use of computed tomography (CT) and positron emission tomography (PET) scans.
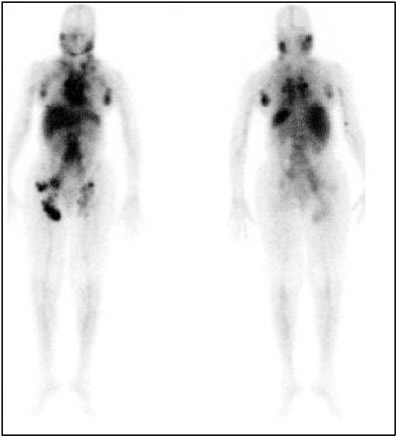
FIGURE 4.4 Initial radionuclide scan of patient infused with indium-111 labeled anti-CD20 antibody in preparation for a subsequent therapeutic infusion with an yttrium-90-labeled antibody (ibritumomab tiuxetan or Zevalin®) of the same specificity. SOURCE: Courtesy of Roger Macklis, Cleveland Clinic.
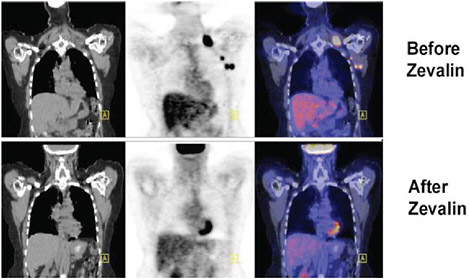
FIGURE 4.5 This set of “before and after” PET/CT images demonstrates the use of these nuclear imaging modalities to evaluate the clinical effects of radioimmunotherapy using radiopharmaceutical compounds such as yttrium-90 ibritumomab tiuxetan (Zevalin®) in the treatment of malignant lymphoma. SOURCE: Courtesy of Peter Conti, University of Southern California.
4.2
SIGNIFICANT DISCOVERIES
A number of critical observations and discoveries have emerged based on previous funding from the National Institutes of Health (NIH) and the Department of Energy (DOE) that have set the stage for advances such as those described below.
Two FDA-approved Labeled Antibodies for the Treatment of Lymphoma
Zevalin® and BEXXAR® are now in general clinical use with impressive response rates and comparatively limited and reversible toxicity. Acknowledged lymphoma experts have noted that the anti-CD20 radioimmunotherapy compounds represent the most active single agents ever developed for the treatment of indolent B-cell lymphoma.
Other Antibodies and Radionuclides in Pre-Clinical and Early Clinical Phases of Testing
Many of these classes of biologically targeted radiopharmaceuticals have shown clear objective responses with acceptable toxicity levels (DeNardo
2005, Sharkey and Goldenberg 2005). These newer classes of therapeutic radiopharmaceuticals include compounds incorporating different antibodies, different radionuclides, and different modes of use. Several groups have recently published results of preclinical and early clinical studies using small molecules or antibodies directed against more common cancers (e.g., lung, breast, colorectal, and brain cancers) and have demonstrated proof of principle. These radioimmunotherapy agents undergoing preclinical and early clinical testing include a variety of radionuclides (with alpha, beta, gamma, and mixed emission spectra), linker chemistries, and half-lives.
Translation of Alpha Particle-Emitting Radiotherapeutics from the Laboratory to the Clinical Setting
Basic chemical advances in labeling molecules at high levels of radioactivity have led to the ability to assess the therapeutic potential of alpha-emitting radionuclides in preclinical models of human malignancy. The predicted localized cytotoxicity of alpha particles has been demonstrated, providing compelling evidence for initiating clinical trials with monoclonal antibodies radiolabeled with an alpha-emitting radionuclide in patients with leukemia and brain tumors.
Therapeutic Benefit in Minimum Residual Disease7 Settings
Used in an adjuvant (i.e., postsurgical) setting, the clinical role of radioimmunotherapy would be to eradicate small nests of cells rather than large solid tumors for which much higher doses of radiation would be necessary.
4.3
CURRENT STATE OF THE FIELD AND EMERGING PRIORITIES
We are now entering an era of personalized medicine guided by new insights into basic biology and genetics that provide a better understanding of the steps that lead to cancer and other complex diseases. Medical practitioners now realize that tailoring treatment by taking into account an individual’s anatomy, physiology, and genetic background is often required, not only for judicious selection of the drug to be administered but also for determining the appropriate dose of the pharmaceutical. For example,
oncologists now are learning to use genetic signatures to determine which breast cancer patients might benefit most from various kinds of cytotoxic chemotherapy and which patients are not good candidates for standard treatments (O’Shaughnessy 2006).
Targeted radionuclide therapy has unique promise as a vehicle for personalized treatment of cancer, because both the targeting vehicle and the radionuclide can be tailored to the individual patient. Looking to the future, we can envision the following scenario:
-
Treatment planning will be based on anatomic and molecular characteristics determined by imaging and complemented by genetic evaluation. High-resolution anatomic imaging will provide information on tumor size, location, and multiplicity to guide in the selection of the radionuclide. Molecular imaging will identify appropriate therapeutic targets that are overexpressed on the tumor cells.
-
Sophisticated modeling and dosimetry software will be used to determine the best combination of radionuclide and targeting vehicle for attacking the tumor while avoiding harm to normal tissues. To ensure that all tumor cells are destroyed, it may be necessary to utilize “radiotherapeutic cocktails” formulated from radionuclides emitting different types of radiation, molecular carriers with different biological properties (antibodies, peptides, organic molecules) and binding to multiple tumor-associated targets. Monitoring of the distribution of the targeted radiotherapeutic agent or its surrogate by PET or single photon emission computed tomography will be done to plan subsequent dosing.
-
Evaluation of tumor response by molecular imaging will allow the oncologist to evaluate response to treatment and tailor the next treatment to the altered status of the tumor cells (a process known as adaptive radiotherapy). For example, a patient initially treated with a targeted radiotherapeutic with a high-energy beta-emitting radionuclide that reduces tumor volume may subsequently be treated for residual disease with a targeted radiotherapeutic with an alpha-emitting radionuclide that has much more focal radiation (Figure 4.3). Moreover, postirradiation response could also alter receptor target populations on the tumor cells, which would require altering the molecule to which the radionuclide is attached. It is this flexibility that makes this approach to cancer treatment attractive.
Implicit in the above scenario is the availability of truly effective targeted radiotherapeutic agents for solid tumors, such as breast, colon, prostate, and lung cancers, that are less radiosensitive and less accessible than lymphomas, where targeted radionuclide therapy has already demonstrated meaningful results. A number of broad emerging research priorities, described in the following section, show promise for achieving this goal and
are likely to have a considerable impact on targeted radionuclide therapy in the future.
4.3.1
Broad Emerging Research Priorities
The committee identified the following research priorities for targeted radionuclide therapy.
Optimal Radionuclides for Therapy
-
Alpha-Emitting Radionuclides. The range of alpha particles in tissue is only a few cell diameters, offering the exciting prospect of matching the cell-specific nature of molecular targeting with radiation of a similar range of action. Another attractive feature of alpha particles for targeted radionuclide therapy is that, as a consequence of their high linear energy transfer, they have greater biological effectiveness than either conventional external beam x-ray radiation or beta emitters. Studies performed in cell culture have demonstrated that human cancer cells can be killed even after being hit by only a few alpha particles (Akabani et al. 2006) and that unlike other types of radiation, where oxygen is necessary for free radicals to be generated, efficient cancer cell elimination can be achieved even in an hypoxic environment. Although the conceptual advantages of alpha particles have been appreciated for more than 25 years, clinical investigation of these promising targeted radiotherapeutics has only just begun. Phase I clinical trials (Sidebar 2.6) have been performed with bismuth-213- and astatine-211-labeled monoclonal antibodies in patients with leukemia and brain tumors (Couturier et al. 2005), respectively, and radium-223 is being evaluated in breast and prostate cancer patients with bone metastases (Nilsson et al. 2005). Even though these trials have not all been carried out at optimized dose levels, encouraging responses have been observed in some patients, with acceptable levels of toxicity in normal tissues. An important aspect of these trials is the demonstration that targeted radionuclide therapy with alpha particle emitters is now clinically and scientifically feasible due to advances in radiochemistry, providing further impetus for more extensive investigation of this promising therapeutic approach in cancer patients.
-
Beta-Emitting Radionuclides. Currently, the targeted radiotherapeutics approved by the FDA for human use are limited to four beta emitters: yttrium-90 and iodine-131, which are used in tandem with monoclonal antibodies to treat non-Hodgkin’s lymphoma, and samarium-153-EDTMP (Quadramet®) and strontium-89-chloride for palliation of bone metastases. However, the scope of preclinical and clinical research in the therapy field is much broader, involving at least eight additional beta-emitting radionu-
-
clides: lutetium-177, holmium-166, rhenium-186, rhenium-188, copper-67, promethium-149, gold-199, and rhodium-105 (Zalutsky 2003).
-
Auger Electron-Emitting Radionuclides. Auger electron emitters, such as bromine-77, indium-111, iodine-123, and iodine-125, are also being investigated. When used in concert with targeting vehicles that can localize these subcellular-range radiations in close proximity to cellular DNA, studies in cell culture have shown highly effective and specific tumor cell killing (Adelstein et al. 2003, P. Chen et al. 2006). The development of a matrix of targeted therapeutics offering multiple radionuclide—molecular carrier combinations can provide the tools to implement targeted radionuclide therapy regimens that are optimally tailored to the needs of individual patients.
Enhancing Target Concentration
The achievement of a therapeutically relevant level of radioactive drug in a tumor is critically dependent upon the concentration of the molecular target within the tumor. A number of strategies are being investigated for increasing the copy number and homogeneity of molecular targets on malignant cell populations (Mairs et al. 2000).
Management of Minimum Residual Disease
MRD settings can be difficult to treat by conventional means; however, targeted radionuclide therapy is likely to have an impact in treating MRD. Encouraging results from a phase II trial have been reported in the use of radiolabeled anti-carcinoembryonic antigen (CEA) antibody as an adjuvant in the treatment of colorectal cancer patients with liver metastases (Liersch et al. 2005), consistent with proof-of-principle studies in animal models (Koppe et al. 2006). These studies suggest that use of targeted radionuclide therapy as an adjuvant after surgical debulking could be a promising therapeutic strategy and provide evidence that targeted radionuclide therapy can be effective in solid tumors when applied in a setting of MRD. Follow-up studies are currently underway to investigate the clinical efficacy of the radiolabeled anti-CEA antibody.
Similarly, some encouraging results have been reported in the treatment of glioblastoma multiforme (GBM), a very aggressive type of brain tumor with poor prognosis. Conventional therapies such as chemotherapy and external beam radiation are largely ineffective because of dose-limiting toxicity to normal brain tissue. Because most GBM kill through local invasion and rarely metastasize outside the cranium, the clinical potential of loco-regionally applied targeted radionuclide therapy is being evaluated for the treatment of this malignancy. More than 300 patients have been treated worldwide with radiolabeled monoclonal antibodies injected directly into
the resection cavity created after surgical removal of visible tumor. Encouraging responses have been observed in many patients, with median survivals of up to 90 weeks seen in GBM patients treated with iodine-131-labeled anti-tenascin monoclonal antibody 81C6 compared with approximately 1-year survival for conventional combined-modality treatments (Reardon et al. 2006); furthermore, the toxic effects on normal tissues were low. Similar proof-of-principle studies involving other regionally confined targets have also been published (Koppe et al. 2006). As these examples illustrate, the strength of targeted radionuclide therapy is its ability to seek out microscopic, even subclinical, cancers and selectively deliver curative doses of radiation, and this should be vigorously explored.
Radiolabeled Small Molecules
Radiolabeled monoclonal antibodies have been the most widely pursued approach to targeted radionuclide therapy; however, smaller molecular carriers, such as peptides that regulate the endocrine system, have been found to offer advantages for certain applications. The advantages of these smaller molecules include rapid accumulation in tumor and clearance from most normal tissues, which make them well-suited to use in tandem with some of the most promising radionuclides for targeted radionuclide therapy such as astatine-211 and rhenium-188, which have half lives of less than 24 hours. A number of regulatory peptides and their corresponding receptors that are overexpressed on certain types of tumors are being evaluated for possible application of targeted radionuclide therapy. For example, the most clinically advanced example of this strategy is targeting of the somatostatin receptor. Somatostatin, which is a peptide hormone that regulates the endocrine system and its corresponding receptor, has been studied for targeted radionuclide therapy of medullary thyroid carcinomas and neuroendocrine tumors (Kwekkeboom et al. 2005). Labeled peptides that bind specifically to other regulatory peptide receptors are also being investigated (Matthay et al. 2006). Penetration of these exciting concepts into the clinical domain has been much slower in the United States than in Europe.
Pre-Targeting Strategies
One of the challenges of targeted radiopharmaceutical development has been achieving a balance between maximizing the absolute amount of radionuclide that can be delivered to the tumor and meeting the requirement that the tumor-to-normal organ dose ratios be as high as possible. The problem is that large molecules such as antibodies provide the highest tumor accumulation, while smaller molecules such as peptides provide the highest tumor-to-normal organ dose ratios. An intriguing solution would
be to break the treatment strategy into two steps, the first involving an unlabeled macromolecule followed later by administration of a radiolabeled small molecule that binds specifically to the protein. Exciting results utilizing bispecific antibodies have demonstrated proof-of-principle of this approach in animal models of human cancer, and early clinical trials are in progress (Goldenberg et al. 2006).
Radiobiological Factors
Conventional perspectives on the response of tissues to radiation may not adequately describe and predict the effects of targeted radiotherapeutics on tumor and normal tissues (Wiseman et al. 2003, Gokhale et al. 2005, Du et al. 2004). Because biologically targeted radiopharmaceutical therapy is generally characterized by low dose rates, the recently suggested hypersensitivity of mammalian cells to low dose radiation may play a role (Enns et al. 2004). The radiation-induced biological bystander effect (RIBBE) also could have a profound effect on targeted radionuclide therapy (Mothersill and Seymour 2004). In this process, cells not directly hit by radiation can be killed efficiently through an indirect but as yet unidentified mechanism. This is contrary to conventional radiation biological wisdom, which considers cell death to be a direct consequence of radiation traversal and energy deposition. These findings may have implications for targeted radionuclide therapy because if RIBBE could be harnessed, it could help compensate for variability in radiation dose deposition which is the bane of targeted radionuclide therapy (O’Donoghue et al. 2000). Although most work to date has been done with external beam radiation, investigations of low-dose hypersensitivity RIBBE with targeted radiopharmaceuticals are moving forward (Boyd et al. 2006) and could lead to novel strategies for cancer treatment. However, at present, both the radiobiology and the dosimetry of this field are topics of intense debate, and the implications for treatment are unclear.
4.3.2
Specific Research Priorities
For targeted radiopharmaceuticals to have a larger role in cancer treatment, the following key issues must be resolved:
-
labeling methodologies that circumvent problems caused by high radiation levels (i.e., radiolysis) that are reliable for preparing clinical doses of therapeutic radiopharmaceuticals;
-
more stable labeling methods for alpha emitters, particularly actinium-225 and astatine-211, to maximize the therapeutic potential of therapeutics labeled with these radionuclides;
-
improvements in specific activity of labeled drugs to exploit highly tumor-specific but low-abundance molecular targets for cancer therapy;
-
more universal methods for constructing targeted therapeutics that can be used with a variety of radionuclides;
-
identification of a biomarker to predict normal organ response (includes effects of prior therapies);
-
practical methods for calculating dose to tumor and normal tissues for radiation of short range and high potency (i.e., alpha particle and Auger electron emitters); and
-
strategies for reducing toxic effects to the kidneys from promising radiotherapeutics of lower molecular weight.
4.4
CURRENT IMPEDIMENTS TO FULL IMPLEMENTATION OF TARGETED RADIOPHARMACEUTICAL THERAPEUTICS
The committee solicited input from individuals working in academia and in industry to identify current obstacles to the advancement of targeted radionuclide therapy. The following two major impediments were identified.
-
Shortage of Radionuclides. Many of the most important radionuclides that are needed for determination of patient-individualized dosimetry and pharmacokinetics8 (iodine-124 and zirconium-89) or treatment (copper-67, bromine-77, and astatine-211) require production at an accelerator of higher energy and complexity than the small cyclotrons found in PET centers. The lack of a dedicated high-energy accelerator for the production of these and other radionuclides that form the basis for the future of targeted radionuclide therapy is a major barrier to progress in this field. Limitations in radionuclide availability restrict research and development in radiochemistry and radiobiology, assessment of efficacy, training, and clinical implementation. Of the five radionuclides identified as essential for therapeutic nuclear medicine (lutetium-177, astatine-211, yttrium-90, rhenium-186, and rhenium-188; see Chapter 5), only yttrium-90 is readily available in a form approved for use in humans. To allow individualized treatment, the armamentarium of radionuclides available in a form suitable for clinical use needs to be drastically increased.
-
Cumbersome Regulatory Requirements. There are three primary impediments to the efficient entry of promising new radiopharmaceutical compounds into clinical feasibility studies: (i) complex FDA toxicology and
-
other regulatory requirements (i.e., lack of regulatory pathways specifically for both diagnostic and therapeutic radiopharmaceuticals that take into account the unique properties of these agents); (ii) lack of specific guidelines from the FDA for good manufacturing practice of PET radiodiagnostics and other radiopharmaceuticals; and (iii) lack of a consensus for standardized image acquisition in nuclear medicine imaging procedures and protocols appropriate for multi-institutional clinical trials.
-
The costs associated with meeting the FDA toxicology requirements for evaluating a new radiotracer in humans are beyond the budgets of academic institutions and are a major regulatory impediment to radiopharmaceutical development and translation to clinical practice. Moreover, the level of evaluation required is beyond what is considered to be scientifically justified for a chemical compound typically administered once or twice at a tracer (i.e., not pharmacologically effective) level.9
4.5
RECOMMENDATIONS
RECOMMENDATION: Clarify and simplify regulatory requirements, including those for (A) toxicology and (B) current good manufacturing practices (cGMP) facilities.
Implementation Action A, Toxicology: The FDA should clarify and issue final guidelines for performing preinvestigational new drug evaluation for radiopharmaceuticals, particularly with regard to the recently added requirement for studies to determine late radiation effects for targeted radiotherapeutics.
Implementation Action B, cGMP: The FDA should issue final guidelines on cGMP for radiopharmaceuticals. These guidelines should be graded commensurate with the properties, applications, and potential risks of the radiopharmaceuticals. Instead of regulating minimal-risk compounds with the same degree of stringency as de novo compounds and new drugs that have pharmacologic effects.
Implementation Action C: To develop prototypes of standardized imaging protocols for multi-institutional clinical trials, members of the imaging community should meet with representatives of federal agen-
cies (e.g., DOE, NIH, FDA) to discuss standardization, validation, and pathways for establishing surrogate markers of clinical response.
4.6
CONCLUSIONS
As noted earlier, targeted radionuclide therapy has the promise to personalize treatment by tailoring the properties of the radionuclide and the targeting vehicle for each patient. BEXXAR® and Zevalin® demonstrate robust proof of principle that adding a radionuclide enhances the clinical efficacy when compared with treating the patient with the biologic agent (e.g., cold antibody) alone. In addition, targeted radiotherapeutics have the potential for treating patients at a lower cost and with less morbidity than more standard treatment procedures. For example, radiation synovectomy is an alternative to surgery for the treatment of rheumatoid arthritis that costs less and allows patients to return to normal life sooner. It is a relatively simple procedure that can be performed on an outpatient basis and that is under ongoing investigation in Europe, although the approach is relatively dormant in the United States.
Recent experience in Europe demonstrates the appeal of targeted radionuclide therapy to patients. Patients increasingly go to Europe to receive targeted radionuclide therapy treatments that are not available domestically, and the gap in technology is increasing. The French are constructing a consortium-funded, high-yield, and versatile cyclotron for radionuclide production that will become operational in 2008. Such a machine has been under discussion for more than 10 years in the United States, and if anything, we are further away than we were a decade ago from constructing this critical piece of infrastructure. For the United States to retain its status as a leader in the field, these hurdles will need to be addressed.