7
Iron Pentacarbonyl1
Acute Exposure Guideline Levels
PREFACE
Under the authority of the Federal Advisory Committee Act (P.L. 92-463) of 1972, the National Advisory Committee for Acute Exposure Guideline Levels for Hazardous Substances has been established to identify, review, and interpret relevant toxicologic and other scientific data and develop acute exposure guideline levels (AEGLs) for high-priority, acutely toxic chemicals.
AEGLs represent threshold exposure limits for the general public and are applicable to emergency exposure periods ranging from 10 minutes (min) to 8 hours (h). AEGL-2 and AEGL-3 levels, and AEGL-1 levels as appropriate, will be developed for each of five exposure periods (10 min, 30 min, 1 h, 4 h, and 8 h) and will be distinguished by varying degrees of severity of toxic effects. It is believed that the recommended exposure levels are applicable to the general population, including infants and children and other individuals who may be sensitive and susceptible. The three AEGLs have been defined as follows:
AEGL-1 is the airborne concentration (expressed as parts per million [ppm] or milligrams per cubic meter [mg/m3]) of a substance above which it is predicted that the general population, including susceptible individuals, could experience notable discomfort, irritation, or certain asymptomatic nonsensory effects. However, the effects are not disabling and are transient and reversible upon cessation of exposure.
AEGL-2 is the airborne concentration (expressed as ppm or mg/m3) of a substance above which it is predicted that the general population, including sus-
ceptible individuals, could experience irreversible or other serious, long-lasting adverse health effects or an impaired ability to escape.
AEGL-3 is the airborne concentration (expressed as ppm or mg/m3) of a substance above which it is predicted that the general population, including susceptible individuals, could experience life-threatening health effects or death.
Airborne concentrations below the AEGL-1 represent exposure levels that can produce mild and progressively increasing but transient and nondisabling odor, taste, and sensory irritation or certain asymptomatic nonsensory effects. With increasing airborne concentrations above each AEGL, there is a progressive increase in the likelihood of occurrence and the severity of effects described for each corresponding AEGL. Although the AEGL values represent threshold levels for the general public, including sensitive subpopulations, such as infants, children, the elderly, persons with asthma, and those with other illnesses, it is recognized that certain individuals, subject to unique or idiosyncratic responses, could experience the effects described at concentrations below the corresponding AEGL.
SUMMARY
Iron pentacarbonyl is one of several iron carbonyls. It is formed by the interaction of carbon monoxide with finely divided iron. Iron pentacarbonyl is used in the manufacture of powdered iron cores for electronic components, as a catalyst and reagent in organic reactions, and as an antiknock agent in gasoline. Iron pentacarbonyl is pyrophoric in air (50 C auto ignition point), burning to ferric oxide.
Quantitative toxicity data and odor detection data for humans are unavailable. Qualitative descriptions of the signs and symptoms of iron pentacarbonyl exposure include giddiness and headache and occasionally dyspnea and vomiting. With the exception of dyspnea, these signs and symptoms are alleviated upon removal from exposure, but fever, cyanosis, and coughing may occur 12-36 h after exposure. This information could not be validated, and additional details were unavailable.
Animal data are limited to lethality findings in rats, mice, and rabbits. Based on the limited data available, the rat appears to be the most sensitive species as determined by the 30-min LC50 of 118 ppm and a 4-h LC50 of 10 ppm relative to the 30-min LC50 of 285 ppm for the mouse. For mice a 1.35-fold increase in the LC50 exposure concentration resulted in near 100% mortality for the same exposure duration, suggesting a steep exposure-response relationship for this species above the lethality threshold. Similarly, a 2.8-fold increase in exposure concentration (from 86 to 244 ppm) resulted in an increased mortality rate in rats from 4/12 to 11/12. No reproductive/developmental toxicity, genotoxicity, or carcinogenicity data are available for iron pentacarbonyl.
Data were unavailable for determining the exponent n. The concentration-exposure time relationship for many irritant and systemically acting vapors and gases may be described by Cn × t = k, where the exponent n ranges from 1 to 3.5 (ten Berge et al. 1986). In the absence of chemical-specific data, temporal scaling was performed using n = 3 when extrapolating to shorter time points and n = 1 when extrapolating to longer time points.
Data consistent with AEGL-1 effects were limited to labored breathing and signs of irritation in rats exposed to 5.2 ppm for 4 h and no observable effects in rats exposed for 6 h/day to 1 ppm for 28 days. However, analysis of the overall dataset for iron pentacarbonyl indicated a very steep exposure-response curve with little margin between exposures producing no observable effects and those resulting in lethality. No AEGL-1 values were recommended.
Limited data in rats revealed that there is only a small margin between exposures causing little or no toxicity and those causing more severe effects and death. No effect was observed following exposure of rats to 1 ppm, for 6 h/day for up to 28 days, while a single exposure to 2.91 ppm for 6 h/day caused notable signs of toxicity and 10% mortality. The occurrence of deaths in laboratory species several days following cessation of exposure was considered in the derivation of the AEGL-2 values. In the absence of exposure-response data for serious and/or possibly irreversible effects, AEGL-2 values were developed by a 3-fold reduction in the AEGL-3 values. This 3-fold reduction was justified by the steep exposure-response relationship in rats, where there appears to be about a 3-fold difference between exposures that produce no lethality and those resulting in 50-100% lethality. The AEGL-2 values also reflect the application of an uncertainty factor of 3 for both interspecies variability and intraspecies variability as described for the development of AEGL-3 values.
Animal data consistent with the definition of AEGL-3 were limited to 30-min LC50 values for rats (118 ppm) and mice (285 ppm), a 45.5-min LClo value for rabbits (250 ppm), and a 4-h LC50 in rats (10 ppm). In addition to a 4-h LC50 value for rats, Biodynamics (1988) provided a 4-h LC16 estimate of 6.99 ppm and an estimated lethality threshold (4 h) of 5.2 ppm for male and female rats. Data from a study by BASF (1995), however, showed that a single 6-h exposure to 2.91 ppm resulted in 10% (1/10 rats) mortality and that a second exposure resulted in 50% mortality. Remaining rats, however, survived an additional 26 six-h exposures, while rats exposed to 1.0 ppm exhibited no clinical signs of toxicity. Using benchmark dose (BMD) analysis of the BASF data, a 6-h exposure to 1.0 ppm was selected as the point of departure for AEGL-3 derivation. A total uncertainty factor of 10 was applied. An uncertainty factor of 3 was applied to account for interspecies variability and is justified by the 2- to 3-fold variance observed for rats and mice and uncertainties in extrapolating to humans. An additional factor of 3 was applied to account for uncertainties regarding individual variability in the toxic response to iron pentacarbonyl. Additionally, iron pentacarbonyl exhibits a steep exposure-response relationship with little margin between minimal and lethal effects and little individual variability in the response of test animals. The AEGL values for iron pentacarbonyl are presented in Table 7-1.
The AEGL-3 values are defensible when compared to the absence of a toxic response in rats following multiple exposures (6 h/day at 1 ppm for 28 days).
Neither quantitative nor qualitative data are available regarding the potential carcinogenicity of iron pentacarbonyl by any route of exposure. Therefore, a quantitative assessment of potential risk is not possible. Genotoxicity tests in several strains of Salmonella typhimurium were negative.
1.
INTRODUCTION
Iron pentacarbonyl is one of several iron carbonyls. It is formed by the interaction of carbon monoxide with finely divided iron, as shown below (Brief et al. 1967):
The reaction rate is proportional to the square of the carbon monoxide partial pressure. The presence of oxygen, carbon dioxide, and oxidizing gases retards the formation of iron pentacarbonyl.
TABLE 7-1 Summary of AEGL Values for Iron Pentacarbonyl
Iron pentacarbonyl is used in the manufacture of powdered iron cores for electronic components, as a catalyst and reagent in organic reactions, and as an antiknock agent in gasoline (Sunderman et al. 1959; Budavari et al. 1989). Iron pentacarbonyl is pyrophoric in air (−15°C flashpoint; 50°C autoignition temperature), burning to ferric oxide (ACGIH 2001). It is also light sensitive, decomposing to iron nonacarbonyl and carbon monoxide (ACGIH 1991). Exposure of the general population to iron pentacarbonyl probably would be limited to pressurized releases at manufacturing sites utilizing this chemical intermediate.
Information regarding odor threshold is unavailable. Chemical and physical data for iron pentacarbonyl are shown in Table 7-2.
2.
HUMAN TOXICITY DATA
2.1.
Acute Lethality
Information regarding the lethal toxicity of iron pentacarbonyl is limited to statements by Stokinger (1994) that death may occur 4-11 days following exposure (exposure terms not provided). Pathologic findings may include pulmonary hepatization, vascular injury, and degeneration of the central nervous system. No further details are available regarding these qualitative descriptions.
TABLE 7-2 Chemical and Physical Data
|
Property |
Descriptor or Value |
Reference |
|
Synonyms |
Iron carbonyl; pentacarbonyl iron |
|
|
Common name |
Iron pentacarbonyl |
ACGIH 2001; Budavari et al. 1989 |
|
Chemical formula |
Fe(CO)5 |
Budavari et al. 1998 |
|
Molecular weight |
195.90 |
Budavari et al. 1998 |
|
CAS Registry No. |
13463-40-6 |
Budavari et al. 1998 |
|
Physical state |
Liquid |
Budavari et al. 1998 |
|
Solubility |
Insoluble in water and dilute acids, soluble in most organic solvents |
ACGIH 2001; Budavari et al. 1998 |
|
Vapor pressure |
35 torr at 25°C 40 mm Hg at 30.3°C |
ACGIH 2001; Brief et al. 1967 |
|
Density |
1.46-1.52 at 20°C |
ACGIH 2001 |
|
Boiling/melting point |
103°C/−20°C |
ACGIH 2001 |
|
Conversion factors in air |
1 mg/m3 = 0.13 ppm 1 ppm = 8.0 mg/m3 |
ACGIH 2001 |
2.2.
Nonlethal Toxicity
Information on the nonlethal toxicity of iron pentacarbonyl in humans is limited to qualitative descriptions provided by Stokinger (1994). Stokinger noted that the signs and symptoms of iron pentacarbonyl exposure are similar to those for nickel carbonyl and include giddiness and headache and occasionally dyspnea and vomiting, these effects being similar to those associated with metal fume fever. With the exception of dyspnea, these signs and symptoms are alleviated upon removal from exposure, but fever, cyanosis, and coughing may occur 12-36 h after exposure. No source was provided for validation of this information, and no further details were available.
2.2.1.
Epidemiologic Studies
No epidemiologic studies of iron pentacarbonyl toxicity are available.
2.3.
Reproductive/Developmental Toxicity
Data regarding the reproductive/developmental toxicity of iron pentacarbonyl in humans are not available.
2.4.
Genotoxicity
No human genotoxicity data for iron pentacarbonyl are currently available.
2.5.
Carcinogenicity
Information regarding the potential carcinogenicity of iron pentacarbonyl in humans is not available.
2.6.
Summary
Information regarding the toxicity of iron pentacarbonyl in humans is limited to unverifiable qualitative statements regarding signs and symptoms of exposure. Exposure terms relating to lethal or nonlethal effects are not available.
3.
ANIMAL TOXICITY DATA
3.1.
Acute Lethality
Acute lethality data are available for several laboratory species but are lim-
ited to only a few older studies. These studies tend to lack details on the analytical techniques used for determining exposure concentrations.
3.1.1.
Rats
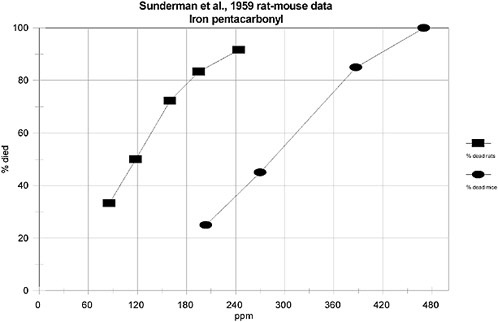
Sunderman et al. (1959) conducted toxicity studies in which male and female Wistar rats (130-180 g) were exposed for 30 min to iron pentacarbonyl at concentrations of 1.57, 2.08, 2.98, or 3.62 mg/L (equivalent to 204, 270, 387, or 470 ppm). There was no description of analytical methods regarding measurement of exposure atmospheres. A total of 20 rats were used in each exposure; three or four rats were exposed at a time. The exposure chamber consisted of an 11-L desiccator. Iron pentacarbonyl was dissolved in ethyl ether and injected into the desiccator via a Vigreux column and a motor-driven syringe. Airflow was set at 11 L/min. Mortality ratios were determined at 3 and 5 days postexposure and are shown in Table 7-3. The investigators estimated the 30-min LC50 as 0.91 mg/L (118 ppm) with a 95% confidence interval of 0.73-1.14 mg/L (95-148 ppm). There was no mention of test animal deaths occurring during or immediately following exposure.
Gage (1970) reported the results of inhalation studies on groups of four male and four female rats exposed to iron pentacarbonyl. Two 5.5-h exposures (on consecutive days) to 15 ppm produced lethargy, respiratory difficulty, 0.2-0.4% carboxyhemoglobin, and four deaths 3-4 days following exposure. Necropsy revealed pulmonary edema and congestion in the dead rats. One 5.5-h exposure to 33 ppm resulted in lethargy, respiratory difficulty, 4% carboxyhemoglobin, and three deaths one day following exposure. Necropsy findings again included pulmonary edema and congestion. The experiments utilized dynamic atmospheres (i.e., continuously generated and passed through the exposure chamber). The iron pentacarbonyl atmosphere was generated by injecting liquid test article (in petroleum ether) at a known rate into a metered stream of air. The iron pentacarbonyl test atmospheres were not verified by analytical techniques.
In an acute inhalation study conducted by Biodynamics for International Specialty Products, groups of 10 CD Sprague-Dawley rats (five/sex/exposure) were exposed to iron pentacarbonyl (0,5.2, 17, 28, or 60 ppm; 99.5% purity) for 4 h (Biodynamics 1988). A group of 10 rats exposed to clean air served as controls. Impinger samples of chamber air were taken every hour for 1 min and analyzed colorimetrically. For each exposure group, chamber concentrations varied (see Table 7-4), but the response was 100% lethality at analytical concentrations at or above 14 ppm and no lethality at or below 6.6 ppm. The results of this experiment are summarized in Table 7-4. The investigators calculated a 4-h LC50 of 10 ppm (both sexes combined, 95% confidence interval of 8.5-13 ppm). No deaths occurred at 5.2 ppm, but 100% mortality was observed for the remaining exposure groups. Deaths occurred at 1-8 days postexposure. For the 60-, 28-,
TABLE 7-3 Lethal Toxicity of Iron Pentacarbonyl in Rats Exposed for 30 min
|
Exposure Concentration (ppm) |
Mortality at 3 Days |
Mortality at 5 Days |
|
244 |
11/12 |
11/12 |
|
195 |
12/18 |
15/18 |
|
160 |
12/18 |
13/18 |
|
118 |
3/12 |
6/12 |
|
86 |
1/12 |
4/12 |
|
Source: Sunderman et al. 1959. Reprinted with permission; copyright 1959, American Medical Association. |
||
TABLE 7-4 Mortality in Rats Exposed for 4 h to Iron Pentacarbonyl
|
Concentration (ppm) |
Mortality (Number Dead/Number Exposed) |
|||
|
Nominal |
Analytical (range) |
Males |
Females |
Combined |
|
Control |
— |
0/5 |
0/5 |
0/10 |
|
7.5 |
5.2 (4.1-6.6) |
0/5 |
0/5 |
0/10 |
|
24 |
17 (14-20) |
5/5 |
5/5 |
10/10 |
|
38 |
28 (19-38) |
5/5 |
5/5 |
10/10 |
|
80 |
60 (55-64) |
5/5 |
5/5 |
10/10 |
|
Source: Biodynamics 1988. |
||||
17-, and 5.2-ppm groups, the respective mortality ratios at postexposure day 5 were 6/10, 7/10, 9/10, and 0/10. Carboxyhemoglobin increased in a dose-related fashion (up to 11.6% increase in the high-dose group relative to controls) but was unaffected in the low-dose group. A lethality versus concentration plot provided in the study indicated that the 5.2-ppm concentration was near a lethality threshold. Gross pathology of rats that died spontaneously revealed red discoloration in several tissues (not specified) and pulmonary edema. The study authors indicated that this was consistent with animals that are not exsanguinated upon death and, therefore, could not be unequivocally considered treatment related.
A 28-day exposure study (consistent with good laboratory practice [GLP] and Organisation for Economic Co-operation and Development [OECD] guidelines) was conducted by BASF Aktiengesellschaft (BASF 1995) in which male and female Wistar rats (five/sex/exposure group) were exposed for 6 h/day, 5 days/week, to iron pentacarbonyl (99.5% purity) at concentrations of 0.1, 0.3, 1, 3, or 10 ppm (0, 0.1, 0.3, 1, 2.91, and 9.85 ppm analytical). A group of 10 rats exposed to clean air only served as controls. All of the rats in the 10-ppm exposure group died within 4 days of the first and only 6-h exposure (see Table 7-5). Prior to death, these rats exhibited clinical signs of piloerection, lassitude, red discharge (confirmed as blood) around the nostrils, and labored respiration. Five of 10 rats in the 3-ppm group were dead within 4 days after only two 6-h ex-
TABLE 7-5 Mortality in Rats Exposed to Iron Pentacarbonyl for 6 h/Day for Up to 28 Days
|
Concentration (ppm) |
Results |
||
|
Test Groupa |
Analytical |
Mortality (Number Dead/Number Exposed) |
Comments |
|
0 control |
– |
0/10 |
No clinical signs |
|
4 |
0.1 (0.1 ± 0.01) |
0/10 |
No clinical signs |
|
E |
0.3 (0.3 ± 0.01) |
0/10 |
No clinical signs |
|
1 |
1 (1.00 ± 0.02) |
0/10 |
No clinical signs |
|
2 |
3 (2.91 ± 0.01) |
5/10 |
One death after first exposure; 50% after two exposures; death occurred within 4 days |
|
3 |
10 (9.85) |
10/10 |
Dead or terminated in extremis after one exposure; deaths occurred within 3 days |
|
aGroup designators as reported in BASF 1995. |
|||
posures. On days 4 and 5 the surviving rats exhibited piloerection and accelerated respiration, and on days 6 through 9 they still exhibited accelerated respiration. From day 10 to the end of the study, the rats exhibited no abnormal clinical signs. Moribund animals also exhibited impaired respiration and bloody discharge from the nostrils. Necropsy of these animals revealed severe pulmonary damage as well as damage to the spleen. None of the rats in the 0.1-, 0.3-, or 1-ppm groups exhibited any clinical signs even after 4 weeks of exposure, although some rats in the 1.0-ppm group were found (upon necropsy) to have increased absolute and relative lung weights. The investigators stated that this could possibly be treatment related. The mortality and exposure-response reported in this study are consistent with those of the previously described acute inhalation study by Biodynamics. The data from these studies suggest a steep exposure-response relationship and a lethality threshold of ~3-5 ppm for exposures of 4-6 h in duration.
3.1.2.
Mice
Sunderman et al. (1959) also conducted lethality studies using Swiss albino mice (18-20 g; gender not specified) using the same exposure system as described for the rat studies (se Section 3.1.1). Exposure concentrations over the 30-min exposure period were 1.57, 2.08, 2.98, and 3.62 mg/L (204, 270, 387, and 470 ppm). The investigators estimated the 30-min LC50 as 2.19 mg/L (285 ppm) with a 95% confidence interval of 1.91-2.51 mg/L (248-326 ppm). Results of this experiment are shown in Table 7-6.
TABLE 7-6 Lethal Toxicity of Iron Pentacarbonyl in Mice Exposed for 30 min
|
Exposure Concentration (ppm) |
Mortality at 3 Days |
Mortality at 5 Days |
|
470 |
16/20 |
20/20 |
|
387 |
15/20 |
17/20 |
|
270 |
8/20 |
9/20 |
|
204 |
5/20 |
5/20 |
|
Source: Sunderman et al. 1959. Reprinted with permission; copyright 1959, American Medical Association. |
||
In addition to the experiments conducted to estimate an LC50, experiments were conducted to assess the effectiveness of antidotes (dithiocarb, dimercaprol, penicillamine, and CaNa2EDTA). For these experiments, groups of 10 albino Swiss mice (gender not specified) were exposed to 3.0 mg of iron pentacarbonyl/L (390 ppm) for 30 min. Lethality was assessed at 3 and 5 days postexposure. At 3 days postexposure, mice not receiving an antidote exhibited 50-90% mortality (Table 7-5). At 5 days postexposure, mice not given any antidote and exposed for 30 min to 390 ppm, exhibited 70-100% lethality (see Table 7-7). These data and the data from the LC50 experiments suggest a steep exposure-response curve (≈1.35-fold increase in the LC50 produces near 100% mortality) for this strain of mouse. Preliminary data indicated that CaNa2EDTA may have provided some protection against iron pentacarbonyl-induced toxicity.
3.1.3.
Rabbits
Armit (1908) reported that a 45.4-min exposure of rabbits (age, number, strain, and gender not specified) to 0.025 volume percent (≈250 ppm) of iron pentacarbonyl resulted in fatality. No further information is available regarding this finding. Stokinger (1981) cited an oral LD50 of 18 mg/kg and a dermal LD50 of 240 mg/kg for rabbits.
TABLE 7-7 Lethal Toxicity of Iron Pentacarbonyl (390 ppm) in Six Groups of Mice Exposed for 30 min
|
Mortality at 3 Days |
Mortality at 5 Days |
|
8/10 |
9/10 |
|
5/10 |
7/10 |
|
10/10 |
10/10 |
|
9/10 |
10/10 |
|
9/10 |
9/10 |
|
9/10 |
9/10 |
|
Source: Sunderman et al. 1959. Reprinted with permission; copyright 1959, American Medical Association. |
|
3.2.
Nonlethal Toxicity
Data on the nonlethal toxicity of iron pentacarbonyl in animals are limited to two unpublished studies regarding pathology in dead rats.
3.2.1.
Rats
Gage (1970) reported the results of inhalation studies on groups of four male and four female rats exposed to iron pentacarbonyl. One group receiving eighteen 5.5-h exposures to 7 ppm exhibited no overt signs of toxicity and necropsy findings were unremarkable. Exposures to higher concentrations (15 and 33 ppm) were lethal. As previously described, the experiments utilized dynamic atmospheres (i.e., continuously generated and passed through the exposure chamber) generated by injecting liquid test article (in petroleum ether) at a known rate into a metered stream of air. Iron pentacarbonyl concentrations were not verified by analytical techniques.
In a 4-h inhalation study reported by Biodynamics (1988), groups of five male and five female Sprague-Dawley CD rats were exposed (whole body) to iron pentacarbonyl at concentrations of 5.2, 17, 28, or 60 ppm (analytical concentration). A control group was exposed to clean air. With the exception of the 5.2-ppm group, all exposures resulted in 100% lethality within 9 days after exposure (mortality ratios at day 5 are noted in Section 3.1.1). Clinical signs during exposure were limited to decreased activity, closing of the eyes, and labored breathing. At 1-2 h postexposure, however, clinical observations for all treatment groups included labored breathing (not for the 5.2-ppm group), lacrimation, mucoid and bloody nasal discharge, salivation, hypothermia (60-ppm group only), and ano-genital staining. A slight exposure-related increase in carboxyhemoglobin levels was observed in males, especially at 1 h into the exposure, but tended to return to normal by the end of the exposure. Even rats in the 5.2-ppm group exhibited a slight increase in carboxyhemoglobin relative to unexposed controls [males: 3.7% (1 h) and 3.5% (4 h) versus 3.2% (1 h) and 3.3% (4 h) for controls; females: 3.0% (1 h) and 2.5% (4 h) versus 2.6% (1 h) and 3.2% (4 h) for controls]. Neurological examinations (gait, muscle tone, reflexes) revealed no findings in the 5.2-ppm group rats. At 1-2 h postexposure, rats in the 5.2-ppm group exhibited lacrimation, nasal discharge, and salivation at incidences similar to those of unexposed controls. Although gross pathology findings at terminal necropsy (postexposure day 15) of rats in the 5.2-ppm group revealed red lungs in a few rats and red turbinates in one male, the study authors indicated the treatment relationship of these findings to be equivocal.
In a 28-day inhalation exposure study (BASF 1995), groups of SPF-Wistar rats (five males and five females per group) were exposed (whole body) to iron pentacarbonyl (0, 0.1, 0.3, 1, 3, or 10 ppm), 6 h/day, for up to 28 days (analytical concentrations were 0, 0.1, 0.3, 1, 2.91, and 9.85 ppm). Although 50% and 100% lethality occurred in the 3-ppm and 10-ppm groups, respectively, no rats
died in the lower (0.1-, 0.3-, and 1.0-ppm) groups, nor were there any clinical signs or findings reported for the 0.1-, 0.3-, and 1-ppm groups over the treatment period.
3.3.
Developmental/Reproductive Toxicity
No data are available regarding the developmental/reproductive toxicity of iron pentacarbonyl in animals.
3.4.
Genotoxicity
BASF (1988) conducted mutagenicity (Ames test) studies using Salmonella typhimurium strains TA1535, TA100, TA1537, and TA98 and iron pentacarbonyl concentrations of 20-5,000µg/plate. There was no evidence of genotoxicity (with or without S9) and no bacteriotoxic effects.
3.5.
Carcinogenicity
No data are available regarding the carcinogenic potential of iron pentacarbonyl in animals.
3.6.
Summary of Toxicity Data in Animals
Limited data are available indicating that 4-h exposure of rats to 5.2 ppm was without serious toxicologic effect, but analysis of lethality data suggests that this exposure may be approaching a lethality threshold. Exposure of rats to 1 ppm for 6 h/day for 28 days was without notable toxicity, as were eighteen 5.5-h exposures to 7 ppm. Lethal toxicity data are available for rats, mice, and rabbits. Based on the limited data available, the rat appears to be the most sensitive species as determined by the 30-min LC50 of 118 ppm relative to the 30-min LC50 of 285 ppm for the mouse. Additionally, a single 5.5-h exposure to 33 ppm or two 5.5-h exposures to 17 ppm resulted in 19% and 25% mortality, respectively, within 1-2 days. A 45.4-min exposure to 250 ppm caused death in rabbits, and a 4-h LC50 of 10 ppm was also reported for rats. For mice a 1.35-fold increase in the LC50 resulted in near 100% mortality for the same exposure duration, suggesting a steep exposure-response relationship for this species (see Section 3.1.2). Similarly, a 2.8-fold increase in exposure concentration (from 86 to 244 ppm) increased the mortality rate in rats from 4/12 to 11/12 (5 days postexposure; see Table 7-3). In calculating a 4-h LC50 of 10 ppm for rats, Biodynamics (1988) estimated a lethality threshold of ~5.2 ppm for 4 h, and BASF (1995) reported 50% mortality in rats following only two 6-h exposures to 3 ppm.
Data regarding the nonlethal effects of iron pentacarbonyl in animals are limited to data from rat studies showing inconsequential effects (similar to responses observed for control groups) or evidence of pulmonary involvement (congestion and edema) at exposure also associated with up to 25% mortality (Gage 1970; Biodynamics 1988; BASF 1995).
4.
SPECIAL CONSIDERATIONS
4.1.
Metabolism and Disposition
Data on the metabolism and disposition of iron pentacarbonyl are not currently available.
4.2.
Mechanism of Toxicity
The specific mechanism of toxicity for iron carbonyl following inhalation exposure has not been elucidated. Data from inhalation studies with animals suggest that the chemical acts as a pulmonary and an airway irritant and that portal-of-entry effects are the most notable. Data from these studies indicate that lethality likely results from pulmonary damage.
4.3.
Structure-Activity Relationships
Toxicity data for other iron carbonyls are not available. Limited data suggest that nickel carbonyl is more toxic than iron pentacarbonyl (Armit 1908; Sunderman et al. 1959) and that dimercaprol and sodium diethyldithiocarbamate, both of which are effective antidotes for nickel carbonyl, are ineffective for iron pentacarbonyl (Sunderman et al. 1959).
Iron pentacarbonyl should not be confused with carbonyl iron, which is particulate iron (CAS No. 7439-89-6) formed by heating gaseous iron pentacarbonyl (Fe[CO]5). This process deposits metallic iron as submicroscopic crystals that form microscopic spheres (Huebers et al. 1986). In the case of carbonyl iron, the “carbonyl” refers to the process and not the composition of the material (Huebers et al. 1986). Studies are available that investigated the toxicity of inhaled carbonyl iron (Warheit et al. 1991) in rats and orally administered carbonyl iron in humans (Gordeuk et al. 1987; Devasthali et al. 1991). In both studies, carbonyl iron was not found to be especially toxic.
4.4.
Other Relevant Information
4.4.1.
Species Variability
The limited lethality data suggest minor species variability as shown by the somewhat lower 30-min LC50 value for rats relative to mice and rabbits.
4.4.2.
Concurrent Exposure Issues
No concurrent exposure issues have been identified that would directly impact the derivation of AEGL values for iron pentacarbonyl.
5.
DATA ANALYSIS FOR AEGL-1
5.1.
Summary of Human Data Relevant to AEGL-1
Quantitative data regarding AEGL-1 level effects of inhaled iron pentacarbonyl in humans are not available. Qualitative data are limited to nonverifiable descriptions of signs and symptoms of exposure. Specifically, Stokinger (1981) reported giddiness and headache as signs and symptoms of iron pentacarbonyl exposure but provided no exposure terms or data references with which to verify these findings. Information regarding odor detection is not available.
5.2.
Summary of Animal Data Relevant to AEGL-1
Findings consistent with AEGL-1 end points were available from a 4-h inhalation study reported by Biodynamics (1988) in which groups of five male and five female Sprague-Dawley CD rats were exposed (whole body) to iron pentacarbonyl at concentrations of 5.2, 17, 28, or 60 ppm (analytical concentration). A control group was exposed to clean air. Although all rats in the 17-, 28-, and 60-ppm groups died, rats in the 5.2-ppm group exhibited only labored breathing and a slight increase in carboxyhemoglobin relative to unexposed controls. Neurological examinations (gait, muscle tone, reflexes) revealed no findings in the 5.2-ppm rats. At 1-2 h postexposure, rats in the 5.2-ppm group exhibited lacrimation, nasal discharges, and salivation at incidences similar to those of unexposed controls. Data from a 28-day inhalation exposure study (BASF 1995) in rats showed that exposure (6 h/day) to a concentration of 1 ppm resulted in no significant clinical signs or findings over the treatment duration.
5.3.
Derivation of AEGL-1
Data consistent with AEGL-1 end points are limited to two studies in rats demonstrating that acute exposure to 5.2 ppm for 4 h (Biodynamics 1988) or exposure to 1 ppm for 6 h/day for 28 days (BASF 1995) produced minor irritation or no observable effects, respectively. However, analysis of lethality data by Biodynamics (1988) suggests that the 5.2 ppm exposure may be near a lethality threshold (see Section 7.3), and therefore this exposure would be inappropriate for development of AEGL-1 values. The findings from the BASF study indicated that even a 28-day exposure to 1 ppm for 6 h/day was without discernable
effect in rats. Although this exposure represents a no-adverse-effect-level (NOAEL), the utility and validity for AEGL-1 derivation are questionable. This exposure, representing a no-effect level, does not meet the criteria for AEGL-1 category effects. Based on the available data from laboratory species, it is difficult to identify an exposure causing notable irritation that does not approach an exposure causing severe effects or death. Therefore, AEGL-1 values are not recommended (see Table 7-8).
6.
DATA ANALYSIS FOR AEGL-2
6.1.
Summary of Human Data Relevant to AEGL-2
Quantitative data consistent with AEGL-2 level effects in humans following exposure to iron pentacarbonyl are unavailable. Qualitative data are limited to nonverifiable descriptions of signs and symptoms of exposure; no quantitative exposure terms are available. Stokinger (1981) reported dyspnea, fever, and cyanosis as signs and symptoms of iron pentacarbonyl exposure but provided no exposure values or verification.
6.2.
Summary of Animal Data Relevant to AEGL-2
Quantitative data consistent with AEGL-2 level effects in animals following exposure to iron pentacarbonyl are unavailable. The reports by Biodynamics (1988) and BASF (1995) provide some information regarding nonlethal effects (i.e., lassitude, pulmonary and airway irritation, labored respiration, carboxyhemoglobin formation, gross pathology and histopathology findings in the lungs) in rats exposed to iron pentacarbonyl, but the effects were not consistent with AEGL-2 severity (i.e., they were not of great severity and were not irreversible). Signs of more severe effects (e.g., pulmonary damage, hypothermia) were associated with single 6-h exposure of rats to 3 ppm (2.91 ppm analytical; BASF 1995), but this exposure also resulted in lethality (10% after one exposure, 50% after only two 6-h exposures). Although these data demonstrate effects that could be considered consistent with AEGL-2 effects, the 6-h exposure to 3 ppm
TABLE 7-8 AEGL-1 Values for Iron Pentacarbonyl
|
AEGL Level |
1 min |
30 min |
1 h |
4 h |
8 h |
|
AEGL-1 (nondisabling) |
NR |
NR |
NR |
NR |
NR |
|
NR: not recommended. Numerical values for AEGL-1 are not recommended (1) because of the lack of available data and (2) because an inadequate margin of safety exists between the derived AEGL-1 and the AEGL-2. Absence of an AEGL-1 does not imply that exposure below the AEGL-2 is without adverse effects. |
|||||
appears to also represent a threshold for lethality. A definitive estimate of the lethality threshold determination is complicated by the fact that lethality may be delayed up to several days following exposure (Sunderman et al. 1959; Biodynamics 1988; BASF 1995).
6.3.
Derivation of AEGL-2
Data from the BASF (1995) study were considered for the derivation of AEGL-2 values. Exposure of rats to a concentration of 1 ppm for 6 h/day for up to 28 days caused no significant effects, and therefore this exposure is not appropriate for deriving AEGL-2 values. Exposure of rats to 2.91 ppm for 6 h/day caused notable signs of toxicity and 10% (1 of 10 rats) mortality after only one exposure and significant mortality (50%) after an additional exposure. In a study reported by Gage (1970), multiple 5.5-h exposures of rats to 7 ppm produced no toxic effects, whereas two 5.5-h exposures to 15 ppm resulted in 100% mortality 3-4 days following exposure. These findings indicate that for rats exposed to iron pentacarbonyl there is only a small margin between exposures causing little or no toxicity and those causing more severe effects and death. The deaths in laboratory species several days after cessation of exposure are also considered in the derivation of AEGL-2 values.
In the absence of exposure-response data consistent with AEGL-2 effects, the AEGL-2 values were derived by a 3-fold reduction in the AEGL-3 values. Such an approach results in values that are clearly below the threshold for lethality or severe toxic responses and is justified for a chemical exhibiting a steep exposure-response curve. The resulting AEGL-2 values are shown in Table 7-9 and Appendix A.
7.
DATA ANALYSIS FOR AEGL-3
7.1.
Summary of Human Data Relevant to AEGL-3
Quantitative data consistent with AEGL-3 level effects in humans following exposure to iron pentacarbonyl are unavailable. Qualitative data are limited to nonverifiable descriptions of signs and symptoms of exposure. Although Stokinger (1981) reported that death may occur 4-11 days after a lethal exposure, no exposure concentration terms or reference sources were provided.
7.2.
Summary of Animal Data Relevant to AEGL-3
Animal data consistent with the AEGL-3 definition are limited to a 30-min LC50 of 118 ppm for rats, a 4-h LC50 of 10 ppm for rats, a 30-min LC50 of 285
TABLE 7-9 AEGL-2 Values for Iron Pentacarbonyla
ppm for mice, and a 45.5-min lethality value (250 ppm) for rabbits. The 30-min LC50 values for rats and mice come from well-conducted experiments reported by Sunderman et al. (1959), but the report lacks details regarding analytical techniques for measuring the exposure concentrations used in the experiments. The 4-h LC50 of 10 ppm for rats comes from a well-conducted study by Biodynamics (1988) with analytically determined exposure concentrations. Additionally, this report provided a 4-h LC16 of 6.99 ppm and analysis of lethality data that indicated 5.2 ppm to be an estimate of the lethality threshold. In a study reported by Gage (1970), two 5.5-h exposures of rats to 15 ppm resulted in 100% mortality at 3-4 days following exposure, and a single 5.5-h exposure to 33 ppm killed three of four rats 1 day postexposure. Lethality data were also available from a 28-day study by BASF (1995), showing 100% lethality in rats following a single 6-h exposure to 10 ppm (9.85 ppm analytical) and 50% lethality in rats following two 6-h exposures to 3 ppm (2.91 ppm analytical).
7.3.
Derivation of AEGL-3
Because of the availability of analytically determined exposure concentrations and overall study quality, data from the BASF (1995) study were used to derive AEGL-3 values. In this study a single 6-h exposure to 2.91 ppm resulted in the death of one of 10 rats, while a second exposure produced 50% mortality. The remaining five rats survived 26 additional 6-h exposures. Exposure of rats to 1.0 ppm for 6 h/day for 28 days resulted in no clinical signs of toxicity.
Because a latency period is associated with iron pentacarbonyl-induced lethality, it was not possible to determine whether the four additional animals that died would have done so from only the first exposure or if the second exposure was necessary. Given this uncertainty, a log-probit benchmark dose analysis was performed (U.S. Environmental Protection Agency [EPA] software V 1.3.1) for two different possibilities, and the results are presented in Table 7-10. In one case it was assumed that only one of 10 animals would have died from one exposure, and in the other case it was assumed that five of 10 animals would have died from a single exposure.
Because the data do not permit any distinction among the hypotheses that one exposure would have killed one, two, three, four, or five animals, the worst-
TABLE 7-10 Log-Probit Benchmark Dose Analysis of BASF (1995) Rat Data Using EPA Software V 1.3.1
|
Benchmark |
Number of Animals Dying at 2.91 ppm for 6 h |
|
|
1 of 10 |
5 of 10 |
|
|
MLE LC01 |
2.4 ppm |
1.9 ppm |
|
BMCL LC05 |
1.7 ppm |
0.80 ppm |
case scenario that one exposure would have killed five animals was assumed. The benchmark dose analysis of this scenario provided an maximum likelihood estimate (MLE) LC01 of 1.9 ppm and a BMCL LC05 of 0.80 ppm. Due to insufficient data differentiating the MLE LC01 from the BMCL LC05, the more conservative lower one-sided confidence limit on the benchmark concentration (BMCL) LC05, value of 0.80 ppm would normally have been selected as the point of departure for the AEGL-3 estimation. However, because no deaths resulted from a 28-day exposure to 1 ppm, 1 ppm was considered a more reasonable point of departure than 0.8 ppm.
In the absence of human data, and because there is some variability among the laboratory species tested, some uncertainty exists regarding species variability. However, Sunderman et al. (1959) provided data for rats and mice tested in a comparable manner (see Tables 7-4 and 7-5). Generally, the rat appears to be about two to three times more sensitive than the mouse (see Figure 7-1).
Because the most sensitive species was used (rat) and conservative experimental results were used for the AEGL-3 point of departure, an interspecies uncertainty factor (UF) of 3 is supportable. The uncertainty adjustment for intraspecies variability (UF of 3) was supported by several points. The available toxicity data indicate that acute inhalation exposure to iron pentacarbonyl results in portal-of-entry effects (i.e., airway and lungs) rather than systemic effects, and therefore variability in response due to dosimetric factors may be limited. Additionally, lethality in rats following acute inhalation exposure to iron pentacarbonyl exhibits a steep exposure-response relationship with little margin between minimal and lethal effects and little individual variability in the response of test animals (Biodynamics 1988). Finally, the total UF of 10 resulted in AEGL-3 values that were consistent with the acute exposure data and the data from multiple-exposure animal studies.
The available data for rats and mice suggest that the exposure-response curve for iron pentacarbonyl is steep. Exposure-response data for the same toxicity end point over multiple time periods are limited (30-min LC50 and 4-h LC50) for iron pentacarbonyl. Data were unavailable for a definitive mathematical determination of the time scaling factor, n, for the equation Cn × t = k (see Appendix C). In the absence of chemical-specific data, temporal scaling was performed using n = 3 when extrapolating to shorter time points and n = 1 when extrapolating to longer time points.
It was considered reasonable to extrapolate the data back to 10 min based on a benchmark dose analysis of the Sunderman et al. (1959) data on rats exposed for 30 min. One caveat to the Sunderman data is that the animals were observed for only 5 days; however, the data are probably reasonable because in the BASF (1995) study rats died within 4 days of exposure at the lowest dose. These data were analyzed using a log probit model (EPA Benchmark Dose software V 1.31). The 30-min LC01 MLE was calculated to be 45 ppm, and the lower 95% confidence limit of the LC05 was 17 ppm. Choosing the lower value of 17 ppm and applying a total UF of 10, the 30-min value of 1.7 ppm calculated from the Sunderman et al.data affirms the protectiveness of the extrapolated AEGL-3 value of 0.23 ppm. Because the point-of-departure for AEGL-3 derivation was of 6-h duration, the 30-min AEGL-3 value was adopted as the 10-min value (NRC 2001).
These values are reasonable when viewed against all of the data on iron pentacarbonyl and well below any lethal concentrations in animals (see Appendix D). Use of a larger total UF would drive AEGL-3 values far below any observed levels of concern.
The draft AEGL-3 values for iron pentacarbonyl are shown in Table 7-11, and their derivation is presented in Appendix A.
8.
SUMMARY OF AEGL VALUES
8.1.
AEGL Values and Toxicity End Points
Due to the lack of exposure-response data consistent with AEGL-1 end points and the subsequent inability to define realistic exposures for such end points, AEGL-1 values are not recommended. AEGL-2 values are based on a 3-fold reduction in the AEGL-3 values. The point of departure for deriving AEGL-3 values was an estimated lethality threshold of 1.0 ppm for a 6-h exposure as determined by BMC analysis of rat lethality data. Available data for rodents suggest there is little margin between exposures void of notable effects and those causing death. Uncertainty factors were applied to account for this observation. The relationships of the AEGL values to one another and to available data are shown in the category plot in Appendix D.
8.2.
Comparison with Other Standards and Guidelines
Other standards and guidelines for iron pentacarbonyl are presented in Table 7-12. No other values are currently available.
8.3.
Data Adequacy and Research Needs
The animal lethality data were sufficient for the development of AEGL-3 values. The findings from these lethality studies also suggested an exposure-
TABLE 7-11 AEGL-3 Values for Iron Pentacarbonyla
TABLE 7-12 Extant Standards and Guidelines for Iron Pentacarbonyl
|
Guideline |
Exposure Duration |
||||
|
10 min |
30 min |
1 h |
4 h |
8 h |
|
|
AEGL-1 |
NR |
NR |
NR |
NR |
NR |
|
AEGL-2 |
0.077 ppm |
0.077 ppm |
0.060 ppm |
0.037 ppm |
0.025 ppm |
|
AEGL-3 |
0.23 ppm |
0.23 ppm |
0.18 ppm |
0.11 ppm |
0.075 ppm |
|
ERPG-1 (AIHA)a |
— |
— |
— |
— |
— |
|
ERPG-2 (AIHA)a |
— |
— |
— |
— |
— |
|
ERPG-3 (AIHA)a |
— |
— |
— |
— |
— |
|
EEGL (NRC)b |
— |
— |
— |
— |
— |
|
PEL-TWA (OSHA)c |
— |
— |
— |
— |
0.1 ppm |
|
PEL-STEL (OSHA)d |
— |
— |
— |
— |
0.2 ppm |
|
IDLH (NIOSH)e |
— |
— |
— |
— |
— |
|
REL-TWA (NIOSH)f |
— |
— |
— |
— |
0.1 ppm |
|
REL-STEL (NIOSH)g |
— |
— |
0.2 ppm |
— |
— |
|
TLV-TWA (ACGIH)h |
— |
— |
— |
— |
0.1 ppm |
|
STEL/ceiling (ACGIH)i |
— |
— |
— |
— |
0.2 ppm |
|
MAK (Germany) |
— |
— |
— |
— |
0.1 ppm |
|
MAK Spitzenbegrenzung (Germany)k |
— |
— |
— |
— |
1 ppm |
|
Einstaztolermanzwert (Germany)l |
— |
— |
— |
— |
— |
|
aERPG (Emergency Response Planning Guidelines, American Industrial Hygiene Association) (AIHA 1994). The ERPG-1 is the maximum airborne concentration below which it is believed nearly all individuals could be exposed for up to 1 h without experiencing other than mild, transient adverse health effects or without perceiving a clearly defined objectionable odor. The ERPG-2 is the maximum airborne concentration below which it is believed nearly all individuals could be exposed for up to 1 h without experiencing or developing irreversible or other serious health effects or symptoms that could impair an individual’s ability to take protective action. The ERPG-3 is the maximum airborne concentration below which it is believed nearly all individuals could be exposed for up to 1 h without experiencing or developing life-threatening health effects. bEEGL (Emergency Exposure Guidance Levels, National Research Council) (NRC 1985). The EEGL is the concentration of contaminants that can cause discomfort or other evidence of irritation or intoxication in or around the workplace but avoids death, other severe acute effects, and long-term or chronic injury. |
|||||
response relationship for which there appeared to be little margin between exposures producing little or no toxicity and those resulting in lethal responses. The available studies also showed that the respiratory tract may be a primary target for the lethality of this chemical following inhalation exposure. Information re-
garding the human experience was limited to inadequately validated qualitative descriptions of nonspecific responses.
The most notable research need is to provide definitive exposure-response data for nonlethal effects, thereby allowing for a more precise description of the exposure-response profile for iron pentacarbonyl, particularly in terms of AEGL-2 effects. AEGL-1 are not recommended due to the absence of data specific to response end points consistent with the AEGL-1 definition. Furthermore, available data suggest that there is little margin between these levels, thereby rendering development of AEGL-1 values tenuous and of questionable utility. Although LC50 data are available for two species, the overall database is insufficient to definitively determine the magnitude of species variability in the lethal response to inhaled iron pentacarbonyl.
9.
REFERENCES
ACGIH (American Conference of Governmental Industrial Hygienists). 1991. Iron pentacarbonyl. Pp. 806-807 in Documentation of the Threshold Limit Values and Biological Exposure Indices, 6th Ed. American Conference of Governmental Industrial Hygienists, Cincinnatti, OH.
ACGIH (American Conference of Governmental Hygienists). 2001. Documentation of the Threshold Limit Values for Chemical Substances and Physical Agents and Biological Exposure Indices, 7th Ed. American Conference of Governmental Hygienists, Cincinnatti, OH.
AIHA (American Industrial Hygiene Association). 1999. The AIHA Emergency Response Planning Guideline and Workplace Environmental Exposure Level Guides Handbook. American Industrial Hygiene Association, Fairfax, VA.
Armit, H.W. 1908. The toxicology of nickel carbonyl, Part II. J. Hyg. 8:565-600.
BASF. 1988. Report on the Study of Eisenpentacarbonyl in the Ames Test. BASF Department of Toxicology. EPA/OTS Doc # 0529732.
BASF. 1995. Study on the Inhalation Toxicity of Eisenpentacarbonyl as a Vapor in Rats -28 Day Test. BASF Department of Toxicology. EPA/OTS Doc # 89-950000244.
Biodynamics. 1988. An Acute Inhalation Toxicity Study of Iron Pentacarbonyl in the Rat. Final Report. EPA/OTS Doc ID 88-920001300.
Brief, R.S., R.S. Ajemian, and R.G. Confer. 1967. Iron pentacarbonyl: Its toxicity, detection, and potential for formation. Am. Ind. Hyg. Assoc. J. 28(1):21-30.
Budavari, S., M.J. O'Neil, A. Smith, and P.E. Heckelman, eds. 1989. Iron pentacarbonyl. P. 806 in The Merck Index: An Encyclopedia of Chemicals, Drugs, and Biologicals, 11th Ed. Rahway, NJ: Merck.
Budavari S, O'Neil, M.J., Smith, A., Heckelman, P.E., Kennedy, J.F., Eds. 1998. Iron pentacarbonyl. The Merck Index. 11th ed. Merck and Co., Whitehouse, NJ. p. 874.
Devasthali, S.D., V.R. Gordeuk, G.M. Brittenham, J.R. Bravo, M.A. Hughs, and L.J. Keating. 1991. Bioavailability of carbonyl iron: A randomized, double-blind study. Eur. J. Hematol. 46(5):272-278.
DFG (Deutsche Forschungsgemeinschaft). 1999. List of MAK and BAT Values 1999: Maximum Concentrations and Biological Tolerance Values at the Workplace. Commission for the Investigation of Health Hazards of Chemical Compounds in
the Work Area. Report No. 35. Weinheim, Federal Republic of Germany: Wiley-VCH.
EPA (U.S. Environmental Protection Agency). 2002. Benchmark Dose Software Version 1.3.1. National Center for Environmental Assessment, U.S. Environmental Protection Agency.
Gage, J.C. 1970. The subacute inhalation toxicity of 109 industrial chemicals. Br. J. Ind. Med. 27(1):1-18.
Gordeuk, V.R., G.M. Brittenham, M. Hughes, L.J. Keating, and J.J. Opplt. 1987. High-dose carbonyl iron for iron deficiency anemia: A randomized double-blind trial. Am. J. Clin. Nutr. 46(6):1029-1034.
Haber, F. 1924. Zur Geschichte des Gaskrieges. Pp. 76-92 in Fünf Vorträge aus den Jahren 1920-1923. Berlin: J. Springer.
Huebers, H.A., G.M. Brittenham, E. Csiba, and C.A. Finch. 1986. Absorption of carbonyl iron. J. Lab. Clin. Med. 108(5):473-478.
NIOSH (National Institute for Occupational Safety and Health). 2004. Iron pentacarbonyl. In NIOSH Pocket Guide to Chemical Hazards. Publication No. 97-140. National Institute for Occupational Safety and Health, Public Health Service, U.S. Department of Health, Education and Welfare, Cincinnati, OH [online]. Available: http://www.setonresourcecenter.com/MSDS_Hazcom/NPG/npgd0345.html [accessed July 27, 2007].
NRC (National Research Council). 1985. Emergency and Continuous Exposure Guidance Levels for Selected Airborne Contaminants, Vol. 5. Washington, DC: National Academy Press.
NRC (National Research Council). 1993. Guidelines for Developing Community Emergency Exposure Levels for Hazardous Substances. Washington, DC: National Academy Press.
NRC (National Research Council). 2001. Standing Operating Procedures for Developing Acute Exposure Guideline Levels for Hazardous Chemicals. Washington, DC: National Academy Press.
Rinehart, W.E., and T. Hatch. 1964. Concentration-time product (CT) as an expression of dose in sublethal exposures to phosgene. Am. Ind. Hyg. Assoc. J. 25:545-553.
Stokinger, H.E. 1981. Metal carbonyls Mex(CO)y. Pp. 1792-1807 in Patty’s Industrial Hygiene and Toxicology, Vol. IIA. Toxicology, G.D. Clayton, and F.E. Clayton, eds. New York: John Wiley & Sons.
Stokinger, H.E. 1994. Metals. In: Clayton, G.D., Clayton, F.E., Eds., Patty’s Industrial Hygiene and Toxicology. John Wiley & Sons, New York. Pp. 1792-1807.
Sunderman, F.W., B. West, and J.F. Kincaid. 1959. A toxicity study of iron pentacarbonyl. AMA Arch. Ind. Health 19(1):11-13.
ten Berge, W.F., A. Zwart, and L.M. Appelman. 1986. Concentration-time mortality response relationship of irritant and systemically acting vapours and gases. J. Hazard. Mater. 13: 301-309.
Warheit, D.B., M.C. Carakostas, M.A. Hartsky, and J.F. Hansen. 1991. Development of a short-term inhalation bioassay to assess pulmonary toxicity of inhaled particles: Comparisons of pulmonary responses to carbonyl iron and silica. Toxicol. Appl. Pharmacol. 107(2):350-368.
APPENDIX A
Derivation of AEGL-1 Values
Quantitative data regarding responses consistent with the AEGL-1 definition were not available for acute inhalation exposure of humans or test animals to iron pentacarbonyl. Because of the lack of appropriate data, reliable AEGL-1 values could not be determined. Additionally, the exposure-response relationship and apparent extreme toxicity of iron pentacarbonyl following inhalation exposure in animals suggest little margin between exposures with little or no apparent effect and those causing lethality. Therefore, AEGL-1 values are not recommended. Absence of an AEGL-1 value does not imply that exposure below the AEGL-2 concentration is without adverse effects.
Derivation of AEGL-2 Values
|
Key study: |
AEGL-2 values were derived by a 3-fold reduction in the AEGL-3 and therefore are also based on the data reported by BASF (1995). |
|
Toxicity end point: |
The AEGL-3 values were reduced by a factor of 3 as a threshold estimate for serious and/or irreversible effects. Comparison of the resulting values with available animal data affirms that the resulting values would be below those causing a lethal response and that they are consistent with the steep exposure-response relationship indicated by the animal data. |
|
Scaling: |
As per AEGL-3 development. |
|
Uncertainty factors: |
3 for uncertainties regarding interspecies variability as per AEGL-3 development. |
|
|
3 for intraspecies variability as per AEGL-3 development. |
Derivation of AEGL-3 Values
|
Key study: |
BASF 1995. |
|
Toxicity end point: |
10% lethality following a single 6-h exposure of male and female rats to 2.91 ppm; 50% mortality following |
|
|
two 6-h exposures to 2.91 ppm. Data from an independent study (Biodynamics 1988) provided a 4-h LC50 of 10 ppm, a 4-h LC16 of 6.99 ppm, and an estimated 4-h lethality threshold of 5.2 ppm. The AEGL-3 point of departure (NOAEL for lethality) was estimated to be 1.0 ppm (6-h exposure) based on BMD analysis and evaluation of the available data (see Section 7.3). |
|
Scaling: |
Data were unavailable for determining the exponent “n.” The concentration-exposure time relationship for many irritant and systemically acting vapors and gases may be described by Cn × t = k, where the exponent n ranges from 1 to 3.5 (ten Berge et al. 1986). In the absence of chemical-specific data, temporal scaling was performed using n = 3 when extrapolating to shorter time points and n = 1 when extrapolating to longer time points. (1.0 ppm)1 × 6 h = 6 ppm•h (1.0 ppm)3 × 6 h = 6 ppm•h |
|
Uncertainty factors: |
3 for uncertainties regarding interspecies variability. Lethality data suggest some variability (approximately 2 to 3 fold based on data from Sunderman et al. 1959) among the laboratory species tested. Definitive data regarding a lethality threshold for humans exposed to iron pentacarbonyl are not available. |
|
|
3 for intraspecies variability. The adjustment for this area of uncertainty was limited to 3 because the available toxicity data indicate that acute inhalation exposure to iron pentacarbonyl results in port-of-entry effects (i.e., airway and lungs) rather than systemic effects, and therefore variability in response due to dosimetric factors may be limited. Additionally, lethality in rats following acute inhalation exposure to iron pentacarbonyl exhibits a steep exposure-response relationship with little margin between minimal and lethal effects and little individual variability in the response of test animals. |
|
10-min AEGL-3: |
Due to uncertainties in extrapolating from a 6-h experimental time point to a 10-min AEGL-specific du |
|
|
ration, the 30-min AEGL-3 has been adopted as the 10-min AEGL-3. |
|
|
10-min AEGL-3 = 0.23 ppm (1.8 mg/m3) |
|
30-min AEGL-3: |
C3 × 0.5 h = 6.0 ppm•h C = 2.28 ppm 30-min AEGL-3 = 2.28 ppm/10 = 0.23 ppm (1.8 mg/m3) |
|
1-h AEGL-3: |
C3 × 1 h = 6.0 ppm•h C = 1.82 ppm 1-h AEGL-3 = 1.82 ppm/10 = 0.18 ppm (1.4 mg/m3) |
|
4-h AEGL-3: |
C3 × 4 h = 6.0 ppm•h C = 1.14 ppm 4-h AEGL-3 = 1.14 ppm/10 = 0.11 ppm (0.88 mg/m3) |
|
8-h AEGL-3: |
C × 8 h = 6.0 ppm•h C = 0.75 ppm 8-h AEGL-3 = 0.75 ppm/10 = 0.075 ppm (0.60 mg/m3) |
APPENDIX B
Time Scaling for Iron Pentacarbonyl AEGLs
The relationship between dose and time for any given chemical is a function of the physical and chemical properties of the substance and its unique toxicological and pharmacological properties. Historically, the relationship according to Haber (1924), commonly called Haber’s law or Haber’s rule (i.e., C × t = k, where C = exposure concentration, t = exposure duration, and k = a constant) has been used to relate exposure concentration and duration to effect (Rinehart and Hatch 1964). This concept states that exposure concentration and exposure duration may be reciprocally adjusted to maintain a cumulative exposure constant (k) and that this cumulative exposure constant will always reflect a specific quantitative and qualitative response. This inverse relationship of concentration and time may be valid when the toxic response to a chemical is equally dependent on the concentration and the exposure duration. However, an assessment by ten Berge et al. (1986) of LC50 data for certain chemicals revealed chemical-
specific relationships between exposure concentration and exposure duration that were often exponential. This relationship can be expressed by the equation Cn × t = k, where n represents a chemical-specific, and even a toxic end point-specific, exponent. The relationship described by this equation is basically the form of a linear regression analysis of the log-log transformation of a plot of C versus t. ten Berge et al. (1986) examined the airborne concentration (C) and short-term exposure duration (t) relationship relative to death for approximately 20 chemicals and found that the empirically derived value of n ranged from 0.8 to 3.5 among this group of chemicals. Hence, the value of the exponent (n) in the equation Cn × t = k quantitatively defines the relationship between exposure concentration and exposure duration for a given chemical and for a specific health effect end point. Haber’s rule is the special case where n = 1. As the value of n increases, the plot of concentration versus time yields a progressive decrease in the slope of the curve.
Data were not available to derive an exposure concentration-exposure duration relationship (n) for propargyl alcohol. In the absence of chemical-specific data, temporal scaling was performed using n = 3 when extrapolating to shorter time points and n = 1 when extrapolating to longer time points using the Cn × t = k equation (NRC 2001).
Although exposure-response data for the same toxicity end point over multiple time periods were limited to several LC50 values, these data suggested a near-linear relationship. Therefore, the value of n was set at unity for the exponential temporal scaling equation, C1 × t = k.
APPENDIX C
ACUTE EXPOSURE GUIDELINES FOR IRON PENTACARBONYL
Derivation Summary for Iron Pentacarbonyl AEGLS
AEGL-1 VALUES
|
10 min |
30 min |
1 h |
4 h |
8 h |
|
Not recommended |
Not recommended |
Not recommended |
Not recommended |
Not recommended |
|
Key reference: Not applicable. |
||||
|
Test species/Strain/Number: Not applicable. |
||||
|
Exposure route/Concentrations/Durations: Not applicable. |
||||
|
Toxicity end point: Data unavailable for defining AEGL-1-specific end points. |
||||
|
Time scaling: Not applicable. |
||||
|
Concentration/Time selection/Rationale: Not applicable. |
||||
|
Uncertainty factors/Rationale: Not applicable. |
||||
|
10 min |
30 min |
1 h |
4 h |
8 h |
|
Not recommended |
Not recommended |
Not recommended |
Not recommended |
Not recommended |
|
Modifying factor: Not applicable. |
||||
|
Animal-to-human dosimetric adjustments: Not applicable. |
||||
|
Comments: NR: not recommended. Numerical values for AEGL-1 are not recommended (1) because of the lack of available data, and (2) because an inadequate margin of safety exists between the derived AEGL-1 and the AEGL-2 values. Absence of an AEGL-1 value does not imply that exposure below the AEGL-2 concentration is without adverse effects. |
||||
AEGL-2 VALUES
|
10 min |
30 min |
1 h |
4 h |
8 h |
|
0.077 ppm |
0.077 ppm |
0.060 ppm |
0.037 ppm |
0.025 ppm |
|
Key reference: Not applicable; see AEGL-3. |
||||
|
Test species/Strain/Number: Rat/Wistar/5 males and 5 females per exposure group. |
||||
|
Exposure route/Concentrations/Durations: Not applicable; see AEGL-3. |
||||
|
Toxicity end point: 3-fold reduction in AEGL-3 values. |
||||
|
Time scaling: Cn × t = k, where n =1 or 3; as per AEGL-3 values. |
||||
|
Concentration/Time selection/Rationale: See procedure/rationale for AEGL-3. |
||||
|
Uncertainty factors/Rationale Total Uncertainty Factor: 10 (as per AEGL-3 values). |
||||
|
Modifying factor: None applied |
||||
|
Animal-to-human dosimetric adjustments: None. |
||||
|
Data adequacy: Although definitive data were unavailable that described effects consistent with the AEGL-2 definition, a 3-fold reduction in AEGL-3 values was considered appropriate for development of AEGL-2 values. This approach is consistent with the available data demonstrating a steep exposure-response curve. Under ambient atmospheric conditions, iron pentacarbonyl may undergo photochemical decomposition to iron nonacarbonyl and carbon monoxide or burn to ferric oxide. |
||||
AEGL-3 VALUES
|
10 min |
30 min |
1 h |
4 h |
8 h |
|
0.23 ppm |
0.23 ppm |
0.18 ppm |
0.11 ppm |
0.075 ppm |
|
Key reference: BASF. 1995. Study on the inhalation toxicity of eisenpentacarbonyl as a vapor in rats—28 day test. BASF Department of Toxicology. EPA/OTS Doc # 89-950000244. |
||||
|
10 min |
30 min |
1 h |
4 h |
8 h |
|
0.23 ppm |
0.23 ppm |
0.18 ppm |
0.11 ppm |
0.075 ppm |
|
Test species/Strain/Number: Rat/Wistar/5 males and 5 females per exposure group. |
||||
|
Exposure route/Concentrations/Durations: 6-h inhalation exposure |
||||
|
Test Group |
Exposure Concentration (ppm analytical) |
|||
|
0 |
clean air control |
|||
|
4 |
0.1 (0.1 ± 0.01) |
|||
|
E |
0.1 (0.1 ± 0.01) |
|||
|
1 |
1 (1.00 ± 0.02) |
|||
|
2 |
3 (2.91 ± 0.01) |
|||
|
3 |
10 (9.85) |
|||
|
Toxicity end point: 10% mortality after one 6-h exposure to 2.91 ppm; 50% mortality following two 6-h exposures. A benchmark dose analysis of the BASF (1995) data provided an MLE LC01 of 1.9 ppm and a BMDL LC05 of 0.80 ppm. |
||||
|
Time scaling: Cn × t = k, where n =1 or 3. The concentration-exposure time relationship for many irritant and systemically acting vapors and gases may be described by Cn × t = k, where the exponent n ranges from 1 to 3.5 (ten Berge et al. 1986). In the absence of chemical-specific data, temporal scaling was performed using n = 3 when extrapolating to shorter time points and n = 1 when extrapolating to longer time points. Due to uncertainties in extrapolating from the 6-h point of departure to 10 min, the 30-min AEGL-3 was adopted as the 10-min value. |
||||
|
Concentration/Time selection/Rationale: A benchmark dose analysis of the BASF (1995) data provided an MLE LC01 of 1.9 ppm and a BMCL LC05 of 0.80 ppm. Due to insufficient data differentiating the MLE LC01 from the BMCL LC05, the more conservative BMCL LC05 value of 0.80 ppm would normally have been selected as the point of departure for the AEGL-3 estimation. However, because no deaths resulted from a 28-day exposure to 1 ppm, 1 ppm was considered a more reasonable point of departure than 0.8. |
||||
|
Uncertainty factors/Rationale: Total uncertainty factor: 10 |
||||
|
Interspecies: |
3 to account for data deficiencies in species variability in the toxic response to iron carbonyl and for possible variability in toxicodynamics; exposures causing lethality in rats and mice varied by 2- to 3-fold. |
|||
|
Intraspecies: |
3 to account for possible individual variability in the sensitivity to iron pentacarbonyl-induced toxicity. Adjustment of the AEGL-3 values by application of greater uncertainty was not considered necessary because the total uncertainty factor of 10 resulted in AEGL-3 values that were reasonable compared to the available acute exposure data and data from multiple-exposure |
|||
|
10 min |
30 min |
1 h |
4 h |
8 h |
|
0.23 ppm |
0.23 ppm |
0.18 ppm |
0.11 ppm |
0.075 ppm |
|
|
animal studies. Additionally, lethality of rats following acute inhalation exposure to iron pentacarbonyl exhibits a steep exposure-response relationship with little margin between minimal and lethal effects and little individual variability in the response (Biodynamics 1988). |
|||
|
Modifying factor: None. |
||||
|
Animal-to-human dosimetric adjustments: None. |
||||
|
Data adequacy: The AEGL-3 values have been developed based on an estimate of the lethality threshold as determined by data available from a well-conducted GLP study. Under ambient atmospheric conditions, iron pentacarbonyl may undergo photochemical decomposition to iron nonacarbonyl and carbon monoxide or burn to ferric oxide. |
||||
APPENDIX D
Category Plot for Iron Pentacarbonyl AEGLS
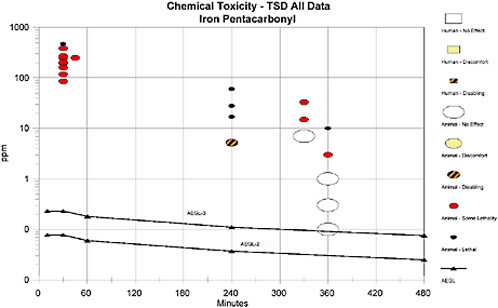
FIGURE 7-2 Category plot of animal toxicity data compared to AEGL values. Note that the above plot includes multiple exposure studies for which a single exposure was input into the plot (5.5 h/d for two days in rats (Gage 1970); 6h/d, 5d/wk for 28 days in rats (BASF 1995). No effect = No effect or mild discomfort. Discomfort = Notable transient discomfort/irritation consistent with AEGL-1 level effects. Disabling = Irreversible/long-lasting effects or an impaired ability to escape. Some lethality = Some, but not all, exposed animals died. Lethal = All exposed animals died.