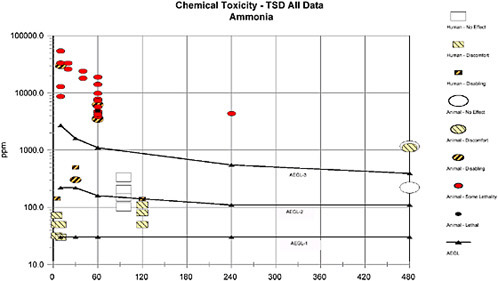
2
Ammonia1
Acute Exposure Guideline Levels
PREFACE
Under the authority of the Federal Advisory Committee Act (P.L. 92-463) of 1972, the National Advisory Committee for Acute Exposure Guideline Levels for Hazardous Substances has been established to identify, review, and interpret relevant toxicological and other scientific data and develop acute exposure guideline levels (AEGLs) for high-priority, acutely toxic chemicals.
AEGLs represent threshold exposure limits for the general public and are applicable to emergency exposure periods ranging from 10 minutes (min) to 8 hours (h). Three levels—AEGL-1, AEGL-2, and AEGL-3—are developed for each of five exposure periods (10 min, 30 min, 1 h, 4 h, and 8 h) and are distinguished by varying degrees of severity of toxic effects. The three AEGLs are defined as follows:
AEGL-1 is the airborne concentration (expressed as parts per million [ppm] or milligrams per cubic meter [mg/m3]) of a substance above which it is predicted that the general population, including susceptible individuals, could
experience notable discomfort, irritation, or certain asymptomatic nonsensory effects. However, the effects are not disabling and are transient and reversible upon cessation of exposure.
AEGL-2 is the airborne concentration (expressed as ppm or mg/m3) of a substance above which it is predicted that the general population, including susceptible individuals, could experience irreversible or other serious, long-lasting adverse health effects or an impaired ability to escape.
AEGL-3 is the airborne concentration (expressed as ppm or mg/m3) of a substance above which it is predicted that the general population, including susceptible individuals, could experience life-threatening health effects or death.
Airborne concentrations below the AEGL-1 represent exposure levels that can produce mild and progressively increasing but transient and nondisabling odor, taste, and sensory irritation or certain asymptomatic nonsensory effects. With increasing airborne concentrations above each AEGL, there is a progressive increase in the likelihood of occurrence and the severity of effects described for each corresponding AEGL. Although the AEGL values represent threshold levels for the general public, including susceptible subpopulations, such as infants, children, the elderly, persons with asthma, and those with other illnesses, it is recognized that individuals, subject to unique or idiosyncratic responses, could experience the effects described at concentrations below the corresponding AEGL.
SUMMARY
Ammonia is a colorless, corrosive, alkaline gas that has a very pungent odor. The odor detection level ranges from 5 to 53 ppm. Ammonia is used as a compressed gas and in aqueous solutions. It is also used in household cleaning products, in fertilizers, and as a refrigerant. Exposure to ammonia occurs as a result of accidents during highway and railway transportation, accidental releases at manufacturing facilities, and farming accidents.
Ammonia is very soluble in water. Because of its exothermic properties, ammonia forms ammonium hydroxide and produces heat when it contacts moist surfaces, such as mucous membranes. The corrosive and exothermic properties of ammonia can result in immediate damage (severe irritation and burns) to the eyes, skin, and mucous membranes of the oral cavity and respiratory tract. In addition, ammonia is effectively scrubbed in the nasopharyngeal region of the respiratory tract because of its high solubility in water.
The database for ammonia consisted primarily of case reports, human studies, and experimental studies on lethality and irritation in animals. The case reports were of limited use for quantitative evaluation, but the human and animal studies contained quantitative data useful for deriving AEGL values.
No reliable quantitative exposure data were available for humans dying as a result of accidental exposure to ammonia. One case report noted the death of
an individual exposed to a high unknown concentration of ammonia. Other case reports also contained no exposure estimates but showed that high concentrations of ammonia caused severe damage to the respiratory tract, particularly in the tracheobronchial and pulmonary regions. Death was most likely to occur when damage caused pulmonary edema. Nonlethal, irreversible, or long-term effects occurred when damage progressed to the tracheobronchial region, manifested by reduced performance on pulmonary function tests, bronchitis, bronchiolitis, emphysema, and bronchiectasis. Nondisabling reversible effects were manifested by irritation to the eyes, throat, and nasopharyngeal region of the respiratory tract. The odor of ammonia can be detected by humans at concentrations >5 ppm; the odor is highly penetrating at 50 ppm (10 min). Human volunteers exposed to ammonia showed slight irritation at 30 ppm (10 min); moderate irritation to the eyes, nose, throat, and chest at 50 ppm (10 min to 2 h); moderate to highly intense irritation at 80 ppm (30 min to 2 h); highly intense irritation at 110 ppm (30 min to 2 h); unbearable irritation at 140 ppm (30 min to 2 h), and excessive lacrimation and irritation at 500 ppm. Reflex glottis closure, a protective response to inhaling irritant vapors, occurred at 570 ppm for 21- to 30-year-old subjects, 1,000 ppm for 60-year-old subjects, and 1,790 ppm for 86- to 90-year-old subjects.
Acute lethality studies in animals showed that the lethal concentration in 50% (LC50) of the rats ranged from 40,300 ppm for a 10-min exposure to 7,338 and 16,600 ppm for 60-min exposures. For the mouse, LC50 values were 21,430 ppm for a 30-min exposure (almost all animals died in less than 13 min), 10,096 ppm for a 10-min exposure, and 4,230 and 4,837 ppm for 60-min exposures. Comparative data for the same exposure duration show that mice were more sensitive than rats to the acute exposure to ammonia (10-min LC50 values for mice and rats are 10,096 and 40,300 ppm, respectively). The lowest lethal concentration was 1,000 ppm for a cat exposed via an endotracheal tube, which probably exacerbated the effects in the tracheobronchial region (bronchopneumonia, bronchitis, bronchiolitis, and emphysema) by bypassing the scrubbing action of the nasopharyngeal region. Rats exposed by inhalation to lethal concentrations of ammonia showed signs of dyspnea, irritation to the eyes and nose, and hemorrhage in the lungs. Mice exposed to lethal concentrations of ammonia showed signs of irritation to the eyes and nose, along with tremors, ataxia, convulsions, seizures, and pathological lesions in the alveoli. Effects at nonlethal concentrations in mice and rats consisted of mild effects on the respiratory epithelium of the nasal cavity (mice and rats), reduction in the respiratory rate (mice), and evidence of eye irritation (rat). The RD50 (concentration causing a 50% reduction in respiratory rate) for the mouse was 300 ppm for a 30-min exposure.
The AEGL-1 value was based on a study in which 2/6 human subjects experienced faint irritation after exposure to ammonia at 30 ppm for 10 min (MacEwen et al. 1970). An interspecies uncertainty factor is not applied because human data are used to derive the AEGL-1. An intraspecies uncertainty factor of 1 was applied because ammonia is a contact irritant and is efficiently scrubbed
in the upper respiratory tract, particularly at the low AEGL-1 concentration. Irritation would be confined to the upper respiratory tract, and members of the population are not expected to respond differently. Atopic subjects, including asthmatics, responded similarly to nonatopics to brief nasal exposure to ammonia, and exercising subjects experienced only nonsignificant clinical changes in pulmonary function after exposure to ammonia. Asthmatic and exercising individuals are not expected to respond differently from nonasthmatic or resting individuals. Time scaling is not applied because upper respiratory tract irritation at low ammonia concentrations is not expected to become more severe with duration of exposure; adaptation may occur during prolonged exposure to ammonia. Therefore, the AEGL-1 value is 30 ppm for all exposure durations.
The AEGL-2 values were based on “offensive irritation” to the eyes and respiratory tract experienced by nonexpert human subjects (unfamiliar with the effects of ammonia or with laboratory studies) exposed to 110 ppm of ammonia for 2 h (Verberk 1977). The response of the nonexpert subjects ranged from “no sensation” to “offensive” eye irritation, cough, or discomfort and from “just perceptible” or “distinctly perceptible” to “offensive” throat irritation. However, AEGL-2 derivation was based on the response of the most sensitive nonexpert subjects. No residual effects were reported after termination of exposure, and pulmonary function was not affected by exposure. At the next higher concentration, some subjects reported the effects as unbearable and left the chamber after 30 min to 1 h; none remained for the full 2 h. An intraspecies uncertainty factor of 1 was selected because ammonia is a contact irritant, it is efficiently scrubbed in the upper respiratory tract, and any perceived irritation is not expected to be greater than that of the most sensitive nonexpert subject. The range of responses for this group is considered comparable to the range of responses that would be encountered in the general population, including asthmatics. Investigations have shown a link between nasal symptoms or allergic rhinitis and asthma, with rhinitis preceding the development of asthma, and studies have shown that atopic subjects, including asthmatics, and nonatopic subjects do not respond differently to a brief nasal exposure to ammonia. Exposure to exercising subjects showed only nonsignificant clinical changes in pulmonary function during exposure to ammonia at concentrations up to 336 ppm. In addition, a child experienced less severe effects than an adult exposed to very high concentrations of ammonia. The equation Cn × t = k, where n = 2, was used to extrapolate to 5-, 10-, and 30-min exposure durations. This equation was based on mouse and rat lethality data. The AEGL-2 values are 220, 220, 160, 110, and 110 ppm for exposure durations of 10 and 30 min and 1, 4, and 8 h, respectively. The value of 110 ppm was adopted for the 4- and 8-h values, because the maximum severity rating for irritation in the Verberk (1977) study changed very little between 30 min and 2 h and is not expected to change for exposures up to 8 h. The 30-min value was also adopted as the 10-min AEGL-2 value because time scaling would yield a 10-min AEGL-2 of 380 ppm, which might impair escape.
The AEGL-3 values were based on LC01 values of 3,317 and 3,374 ppm derived by probit analysis of mouse lethality data reported by Kapeghian et al.
(1982) and MacEwen and Vernot (1972), respectively. An interspecies uncertainty factor of 1 was applied to the mouse data because the mouse was the most sensitive species among mammals and the mouse is considered unusually sensitive to respiratory irritants. An uncertainty factor of 3 was applied to account for intraspecies variability because concentrations of ammonia that are life threatening cause severe tracheobronchial and pulmonary damage and these effects are not expected to be more severe in asthmatics than in nonasthmatics, in children than adults, or in exercising than nonexercising individuals (see rationale for AEGL-2), but tracheobronchial and pulmonary effects may occur at a lower concentration in the elderly. Investigations showed that reflex glottis closure (protective mechanism) is 3-fold less sensitive in the elderly than in young subjects; this mechanism may be applicable only when concentrations of ammonia exceed 570 ppm. In addition, a larger interspecies or intraspecies uncertainty factor would lower the 30-min AEGL-3 to approximately 500 ppm, which was tolerated by humans without lethal or long-term consequences. ten Berge’s equation (Cn × t = k) was used to extrapolate to the relevant exposure durations. The value of n was calculated from the regression coefficients (b1/b2) for the mouse lethality data reported by ten Berge et al. (1986). The 5-min AEGL value was requested by the ammonia industry. The AEGL values and toxicity end points are summarized in Table 2-1.
1.
INTRODUCTION
Ammonia is a colorless, corrosive, alkaline gas that has a very pungent odor, detectable by humans at concentrations >5 ppm. It can be liquefied under pressure. Ammonia is very soluble in water; it forms ammonium hydroxide when it contacts moist surfaces, producing heat because of its exothermic prop-
TABLE 2-1 Summary of AEGL Values for Ammonia
|
Classification |
10 min |
30 min |
1 h |
4 h |
8 h |
End Point (Reference) |
|
AEGL-1 (nondisabling) |
30 ppm (21 mg/m3) |
30 ppm (21 mg/m3) |
30 ppm (21 mg/m3) |
30 ppm (21 mg/m3) |
30 ppm (21 mg/m3) |
Mild irritation (MacEwen et al. 1970) |
|
AEGL-2 (disabling) |
220 ppm (154 mg/m3) |
220 ppm (154 mg/m3) |
160 ppm (112 mg/m3) |
110 ppm (77 mg/m3 |
110 ppm (77 mg/m3) |
Irritation: eyes and throat; urge to cough (Verberk 1977) |
|
AEGL-3 (lethal) |
2,700 ppm (1,888 mg/m3) |
1,600 ppm (1,119 mg/m3) |
1,100 ppm (769 mg/m3) |
550 ppm (385 mg/m3) |
390 ppm (273 mg/m3) |
Lethality (Kapeghian et al. 1982; MacEwen and Vernot 1972) |
erty. Ammonia and air will explode when ignited under some conditions (not otherwise described). Although it is generally regarded as nonflammable, ammonia is classified as a flammable gas by the National Fire Protection Association (Budavari et al. 1989; Lewis 1993; Pierce 1994). Table 2-2 summarizes the physical and chemical properties of ammonia.
TABLE 2-2 Physical and Chemical Data
|
Property |
Descriptor or Value |
Reference |
|
Chemical name |
Ammonia |
|
|
Synonyms |
Anhydrous ammonia, ammonia gas, AM-Fol, nitro-sil, R 717, spirit of hartshorn, UN1005 (DOT) |
|
|
CAS registry no. |
7664-41-7 |
|
|
Chemical formula |
NH3 |
Weast et al 1984 |
|
Molecular weight |
17.03 |
Weast et al 1984 |
|
Physical state |
colorless gas (or liquid) |
Lewis 1993 |
|
Vapor pressure |
8.5 atm at 20°C |
Lewis 1993 |
|
Density (liquid) |
0.6818 at 33.35°C, 1 atm 0.6585 at 15°C, 2.332 atm 0.6386 at 0°C, 4.238 atm 0.6175 at 15°C, 7.188 atm 0.5875 at 35°C, 13.321 atm |
O’Neil et al. 2001 |
|
Specific volume |
22.7 ft3/lb at 70°C |
Lewis 1993 |
|
Critical temperature |
132.9°C |
Pierce 1994 |
|
Pressure at critical temperature |
111.5 atm |
Pierce 1994 |
|
Solubility |
89.9 g/100 mL cold water |
Weast et al. 1984 |
|
Boiling/freezing point |
−33.5°C/−77°C |
Lewis 1993 |
|
Autoignition temperature |
650°C (1,204°F) |
Lewis 1993 |
|
Explosive limit |
16-25% by volume in air |
Pierce 1994 |
|
Ionization constants |
Kb 1.774 × 10−5 Ka 5.637 × 10−10 at 25°C |
Pierce 1994 |
|
Alkalinity |
1% solution, pH = 11.7 |
Pierce 1994 |
|
Conversion |
1 ppm = 0.7 mg/m3 at 25°C, 1 atm 1 mg/m3 = 1.43 ppm |
Pierce 1994 |
Ammonia is produced commercially by a modified Haber reduction process using atmospheric nitrogen and a hydrogen source. Ammonia is used as a compressed gas, as an aqueous solution (28%) called aquammonia, and as a household cleaning product (10%). It is widely used as a fertilizer, where the anhydrous gas or aqueous solution is injected directly into the soil. Ammonia is also used as a refrigerant in commercial installations, and it is used in the manufacture of other chemicals (Pierce 1994).
Ammonia is transported on highways (in tanker trucks), by railways, in pipelines, and on barges. Exposure to the general public can occur from accidents during transportation on highways and railways, during transfer between transportation vessels and storage vessels, by accidental releases at manufacturing facilities, and from farming accidents during soil application.
The data evaluated for AEGL derivation were obtained from case studies of accident victims exposed to high concentrations of ammonia, experimental studies in humans exposed to lower but irritating concentrations of ammonia, and experimental studies on lethality and irritation in animals. Additional data are available on long-term exposure to ammonia in the agricultural industry (feeding lots and poultry houses) but are not considered relevant for deriving acute exposure values for ammonia.
2.
HUMAN TOXICITY DATA
2.1.
Human Lethality
Quantitative exposure estimates of acute lethality of ammonia in humans are not well documented. In one case study the exposure concentration was estimated, but the duration was not. Another study reconstructs the exposure due to an accidental spill resulting in deaths. The remaining studies document the types of effects encountered when humans are acutely exposed to lethal concentrations of ammonia.
A worker was exposed to a very high concentration of ammonia vapor, estimated as 10,000 ppm. Duration of exposure was not reported, but it could have been a few minutes; nevertheless, the worker continued to perform his duties for an additional 3 h after the exposure. He experienced coughing, dyspnea, and vomiting soon after exposure. Three hours after initial exposure, his face was “red and swollen,” his mouth and throat were “red and raw,” his tongue was swollen, his speech was difficult, and he had conjunctivitis. He died of cardiac arrest 6 h after exposure. An autopsy revealed marked respiratory irritation, denudation of the tracheal epithelium, and pulmonary edema (Mulder and Van der Zalm 1967).
Caplin (1941) reported on 47 persons accidentally exposed to ammonia in an enclosed area (air raid shelter). The patients were divided into three groups depending on the degree to which they were affected: mildly, moderately, or severely. No deaths occurred among the nine mildly affected patients. Three of
27 moderately affected patients showed signs and symptoms similar to pulmonary edema and died within 36 h. Nine moderately affected patients developed bronchopneumonia within 2-3 days, and three died 2 days after the onset. The mortality rate for the moderately affected patients was 22% (6/27). The 11 severely affected patients developed pulmonary edema; seven died within 48 h. The mortality rate for the severely affected patients was 63% (7/11). Walton (1973) reported on the death of one of seven workers exposed to ammonia in an industrial accident. The autopsy report noted marked laryngeal edema, acute congestion, pulmonary edema, and denudation of the bronchial epithelium. These studies show that individuals who develop pulmonary edema (evidence of damage to alveolar region) after inhaling ammonia are more likely to die than those who do not.
Individuals who are acutely exposed to high concentrations of ammonia and survive the immediate effects may die weeks to months later, probably due to secondary effects of exposure. A 25-year-old man died 60 days after exposure to a high concentration of ammonia in a farming accident (Sobonya 1977). The autopsy report noted damage to the bronchial epithelium, bronchiectasis, mucus and mural thickening of the smallest bronchi and bronchioles, fibrous obliteration of small airways, and a purulent cavitary pneumonia characterized by large numbers of Nocardia asteroides (nocardial pneumonia). Three co-workers exposed in the accident died immediately. Hoeffler et al. (1982) reported on the case of a 30-year-old woman who died 3 years after exposure to ammonia during an accident involving a tanker truck carrying anhydrous ammonia (Houston accident). Her injuries resulted in severe immediate respiratory effects, including pulmonary edema. She required mechanically assisted respiration throughout her remaining life. Bronchiectasis was detected 2 years after exposure and confirmed on autopsy. The autopsy examination also showed bronchopneumonia and cor pulmonale (heart disease secondary to pulmonary disease). According to the authors, the bronchiectasis may have been due to bacterial bronchitis or to the chemical injury.
In the Houston accident, the crash of a tanker truck released 17.2 tonnes of pressurized anhydrous ammonia. The chemical cloud extended 1,500 m downwind and was 550 m wide. Five people were killed, 178 were injured, some with permanent disabling injuries (not otherwise described). The fatalities and disabling injuries occurred within about 70 m of the accident (NTSB 1979). The Potchefstroom, South Africa accident involved a pressurized ammonia storage tank that failed and instantaneously released 38 tonnes of anhydrous ammonia into the atmosphere. Eighteen people died and an unknown number were injured (Lonsdale 1975). A visible cloud extended about 300 m wide and about 450 m downwind; all deaths occurred within 200 m of the release point (Pedersen and Selig 1989). Pedersen and Selig used the WHAZAN gas dispersion model, which incorporated meteorological data and physicochemical data for ammonia to predict the concentration isopleths for ammonia released during both the Houston and Potchefstroom accidents. For the Houston accident, a 10,000-ppm isopleth extended 600 m long and 350 m wide, the 5,000-ppm isopleth was
835 m long and 430 m wide, the 2,500-ppm isopleth was 875 m long and 420 m wide, and the 1,200-ppm isopleth was to 1,130 m long and 400 m wide. The investigators reported that their model overestimated the distance to zero deaths (200m) by 2.9 times for the Houston accident and by 2.5 times for the Potchefstroom accident. Pederson and Selig estimated the risk due to a few minutes, exposure to ammonia as very high for the general population at 10,000 ppm, as high for risk of fatalities among the general population and as very high for the vulnerable population (elderly people, children, and people with respiratory or heart disorders) at 5,000 ppm, and as some risk to the general population and high risk to the vulnerable population at 2,500 ppm.
Pedersen and Selig estimated the LC50 for a 30-min exposure to the general population to be 11,500 ppm. They did not report their actual LC50 estimate for the vulnerable population, but it would be lower than that estimated for the general population.
Mudan and Mitchell (1996) used the HGSYSTEM gas dispersion model to estimate atmospheric ammonia concentrations generated at the time of the ammonia accident in Potchefstroom. They provided upper-bound (wind speed = 1 m/s) and lower-bound (wind speed = 2 m/s) estimates of ammonia concentration based on distance from the release point and the time after release. Instantaneous concentrations were estimated to be in excess of 500,000 ppm (upper bound) within 50 m of the release point. The model predicted rapidly decreasing concentrations, such that, by 1 min after the release, concentrations would fall below 100,000 ppm. Mudan and Mitchell estimated that personnel were exposed to ammonia concentrations exceeding 50,000 ppm for the first 2 min, decreasing to 10,000 ppm during the next 3-4 min. The charts provided by Mudan and Mitchell of the South Africa accident showed that 10 workers were in Zone 1 (50 m of the release point) at the time of release; seven died (100% mortality for workers exposed outside). All survivors in Zone 1 remained sheltered inside buildings and therefore would not have experienced the outside atmospheric ammonia concentrations predicted by the model. Five deaths occurred in Zone 2 (50-100 m). Workers in Zone 2 who were upwind and outside at the time of the release survived, as did those who escaped in an upwind direction. Workers in Zone 2 who were downwind and outside at the time of release or attempted to escape downwind did not survive (except for one worker who escaped downwind; 83% mortality of workers exposed). All Zone 2 victims who died were outside; whereas individuals who were inside buildings survived. Five deaths occurred in Zone 3 (100 to ~200 m). Four victims were found downwind and >150 m from the release point, and another victim was found <150 m from the release point and in a crosswind location. The charts did not show the location or number of any survivors downwind and inside or outside buildings in Zone 3 (i.e., no data were available from the charts to determine if there were individuals who remained outside buildings in Zone 3 and survived). Therefore, the mortality rate cannot be calculated for Zone 3. It appears that within 150 m of the release point, individuals downwind of the ammonia cloud and outside a building were not likely to survive, but individuals downwind and sheltered indoors
or those upwind whether or not they were sheltered indoors were likely to survive. Thus, the lack of data on survivors in the path of the plume precludes estimating ammonia concentrations associated with zero mortality. RAM TRAC (1996) used the results of the HGSYSTEM gas dispersion model to predict 5-min ammonia concentrations of 87,479 ppm for 60% mortality, 73,347 ppm for 26% mortality, and 33,737 ppm for zero mortality for the Potchefstroom accident. RAM TRAC estimated a 5-min LC50 of 83,322 ppm. See Section 7.1 for details of the evaluation of dose reconstruction models.
Henderson and Haggard (1943) reported that, exposure to ammonia at concentrations >2,500 ppm for durations ≥30 min is dangerous to humans. They noted that concentrations ≥5,000 ppm are rapidly fatal to humans.
2.2.
Nonlethal Toxicity
2.2.1.
Experimental Studies, Case Reports, and Anecdotal Data
The available literature detailing the disabling, long-term, or irreversible effects of inhaling ammonia gas or vapor is quite extensive. However, none of the studies contain quantitative exposure data. The acute effects of inhaling high nonlethal concentrations of ammonia include burns to the eyes and oral cavity and damage to the nasopharyngeal and tracheobronchial regions of the respiratory tract. Manifestations of damage include conjunctivitis, corneal burns, visual impairment, pain in the pharynx and chest, cough, dyspnea, hoarseness, aphonia, rales, wheezing, rhonchi, hyperemia and edema of the pharynx and larynx, tracheitis, bronchiolitis, and purulent bronchial secretions (Levy et al. 1964; Walton, 1973; Hatton et al. 1979; Montague and Macneil 1980; Flury et al. 1983; O’Kane 1983). Cyanosis, tachycardia, convulsions, and abnormal electroencephalograms also have been described for some patients (Kass et al. 1972; Walton 1973; Hatton et al. 1979; Montague and Macneil 1980). Pulmonary edema occurred in some patients who survived (Caplin 1941) but is most often seen in fatal cases. A few case studies are described below to document some of the disabling or irreversible injuries seen in individuals who inhaled high concentrations of ammonia. Some of the injuries would probably have resulted in death without rescue and medical treatment. The duration of exposure is reported when known.
Short-term recovery from serious injury due to inhaling ammonia is exhibited by three children and a 17-year-old female exposed to high but unknown concentrations of ammonia in the Houston accident (Hatton et al. 1979). These patients suffered second- or third-degree burns to the body, damage to the eyes, burns to the oral mucosa, upper-airway obstruction (probably due to damage to the laryngeal and tracheobronchial regions), and some pulmonary damage. All four patients recovered within 7-32 days. Nine of 14 patients exposed to an unknown concentration ammonia by inhalation for only a few seconds or few min-
utes showed moderate symptoms of chest abnormalities or airway obstruction and recovered within 6.3 days (average) (Montague and Macneil 1980).
Two young women accidentally exposed to anhydrous ammonia fumes (concentration unknown) for 30 or 90 min continued to show effects more than 2 years after exposure (Kass et al. 1972). One woman was found unconscious 90 min after the accident, and the other woman was exposed when she went outdoors for 30 min after the accident. The accident in which these two women were injured involved a railroad tanker car carrying 33,000 gal of anhydrous ammonia; 8 people died and 70 were injured. A heavy fog kept the ammonia vapors close to the ground for a long period of time after the accident. Damage to the eyes caused marked visual deterioration. Bronchiectasis was detected 2 years after exposure, and pulmonary function tests showed abnormalities indicative of small-airway obstruction. Various tests and examinations showed areas of atelectasis and emphysema in the lungs, thickened alveolar walls with histiocytic infiltration into the alveolar spaces, and mucous and desquamated cells in the bronchiolar lumen. Some of these effects may be secondary to the damage caused by ammonia. The woman exposed for 90 min was carrying her 1-year-old child, who was exposed at the same time. The child became “quite ill” but recovered completely except for a chemical scar on his abdomen (Kass et al. 1972).
In another accident, four patients (three farm workers and one refrigeration technician) who had been struck in the face and upper body with liquid ammonia had damage to their tracheobronchial regions, causing upper-airway obstruction and injury to the respiratory tract persisting for 2 years after the accident (Levy et al. 1964). A man splashed with liquid ammonia during a refrigeration accident showed evidence of peripheral (possibly bronchiolitis) and central airway obstruction 5 years after the accident (Flury et al. 1983). Tubular bronchiectasis was detected 8 years after exposure of a 28-year-old man to a high concentration of anhydrous ammonia in an industrial accident. Twelve years after exposure, the man continued to have a productive cough, frequent bronchial infections, dyspnea upon exertion, and severe airflow obstruction (62% reduction in forced expiratory volume at 1 s, FEV1; Leduc et al. 1992). O’Kane (1983) described several patients who had been exposed to ammonia vapor by inhalation for 5 min. One developed necrotizing pneumonia and was “left with chronic infective lung disease”, one had persistent hoarseness and a productive cough for several months, and a third was left with a diffusion defect that was 75% of normal. Finally, Shimkin et al. (1954) described a man who developed epidermoid carcinoma 6 months after ammonia was splashed on his upper lip and nose. The authors postulated that the carcinoma was due to a single-exposure chemical trauma that exteriorized a latent cutaneous carcinoma. There was no evidence that ammonia caused the carcinoma.
Nondisabling and reversible effects of inhaling ammonia have been documented in several experimental studies of human subjects exposed to ammonia at various concentrations and durations. These studies are summarized below.
Five or six laboratory workers inhaled the exhaust fumes generated in an exposure chamber for an inhalation study and noted that the disagreeable odor and respiratory distress would prevent a person from voluntarily remaining in an atmosphere containing 170 ppm of ammonia (average concentration, 140-200 ppm) for an appreciable length of time (Weatherby 1952).
Henderson and Haggard (1943) reported that, based on observations of human responses to ammonia, the lowest concentration (or threshold) to cause coughing is 1,720 ppm, the lowest concentration to cause eye irritation is 698 ppm, and the lowest concentration to cause throat irritation is 408 ppm. They reported the least detectable odor to be 53 ppm. Pierce (1994) reported the odor threshold as 5-53 ppm.
McLean et al. (1979) examined the effect of ammonia on nasal airway resistance (NAR) in atopic and nonatopic human subjects. Ammonia (100 ppm at a pressure of 9 newtons/cm2) was introduced into each nostril for 5, 10, 15, 20, or 30 seconds (s). NAR was measured every minute for 5 min and then every 2 min for 10 min (total of 10 measurements over a 15-min period) using a pneumotachograph attached to a face mask. The same subjects were used for each successive ammonia exposure, which immediately followed the NAR measurements. The nonatopic subjects were screened based on strict criteria that included a questionnaire, physical examination, spirometry, nasal smear for eosinophils, and a battery of 19 prick and six intracutaneous tests. Nonatopic subjects could have no personal or immediate family history of atopic disease (allergic rhinitis, asthma, or atopic dermatitis), could have no more than 5% eosinophils in their nasal smears, and had to have a negative prick test reaction. Atopic subjects were screened based on a characteristic history of allergic rhinitis and at least one 3+ or 4+ prick test reaction. Some of the atopic subjects had a history of asthma. All subjects had been symptom-free for several weeks before the study, and none were taking medications that would influence skin or mucosal tests. Baseline NAR measurements were made for a 15-min period before introducing the ammonia. Additional tests included introducing 0.1 mL of aerosolized phosphate-buffered saline, 0.1 mL atropine, or 0.1 mL chlorpheniramine maleate into the nostrils, each followed by ammonia for 20 s.
The NAR after ammonia exposure to nonatopic and atopic subjects increased significantly with time of exposure from 5 to 20 s. Only a small further increase was noted for subjects exposed for 30 s compared with 20 s. The percent increase for atopic compared with nonatopic subjects was similar, and there was no difference between the allergic rhinitis subjects with or without a history of asthma. Atropine inhibited the response to ammonia in atopic and nonatopic subjects by up to 89%, whereas chlorpheniramine had no effect on the NAR induced by ammonia. The study’s authors noted that the results of atropine and chlorpheniramine administration suggest that ammonia irritancy is mediated primarily by a parasympathetic reflex on the nasal vasculature and not via histamine release (McLean et al. 1979).
The Industrial Bio-Test Laboratories (1973) determined the irritation threshold in 10 human volunteers exposed to ammonia at four different concen-
trations (32, 50, 72, or 143 ppm) for 5 min. Irritation was defined as any annoyance to the nose, throat, eyes, mouth, or chest. The results are summarized in Table 2-3. The subjects showed dose-related responses for dryness of the nose and also eye, throat, nasal, and chest irritation. The severity of the effects was not noted.
MacEwen et al. (1970) studied six human volunteers exposed head only to ammonia at concentrations of 30 and 50 ppm for 10 min. The scale for intensity/description of irritation to the nose and eyes was as follows: 0, no irritation/not detectable; 1, faint/just perceptible, not painful; 2, moderate/moderate irritation; 3, strong/discomforting, painful, but may be endured; and 4, intolerable/exceedingly painful, cannot be endured. The scale for odor intensity/description was as follows: 0, no odor/no detectable odor; 1, very faint/minimum but positively perceptible odor; 2, faint/weak odor, readily perceptible; 3, easily noticeable/moderate intensity; 4, strong/highly penetrating; and 5, very strong/intense. At 30 ppm, two subjects reported irritation as faint (grade = 1) and three as not detectable (grade = 0); one gave no response. Also at 30 ppm, the odor was strong or highly penetrating for three subjects (grade = 4) and easily noticeable or moderate (grade = 3) for two subjects; no response was given by one subject. At 50 ppm, four subjects reported the irritation as moderate (grade = 2), faint or just perceptible (grade = 1) for one, and not detectable (grade = 0) for another. The odor was strong or highly penetrating (grade = 4) for all six subjects inhaling 50 ppm of ammonia. This study showed a concentration-related increase in the intensity of the response to ammonia at concentrations of 30 and 50 ppm.
Silverman et al. (1949) studied seven male subjects exposed to 500 ppm of anhydrous ammonia by means of a nose and mouth mask; six subjects were exposed for 30 min and one for 15 min. The inspired ammonia concentration was calculated, and the expired ammonia concentration was analyzed in grab samples taken every 3 min. The analytical technique consisted of a modified Nessler’s reagent using a Klett photoelectric colorimeter. The sensitivity of the technique was 0.5 µg of ammonia. Respiratory rate and minute volume were
TABLE 2-3 Effect of Ammonia Inhalation on Human Volunteers Exposed for 5 Min
|
Effects |
32 ppm |
50 ppm |
72 ppm |
134 ppm |
|
Dryness of the nose |
+ (1)a |
+ (2) |
— |
— |
|
Nasal irritation |
— |
— |
+ (2) |
+ (7) |
|
Eye irritation |
— |
— |
+ (3) |
+ (5) |
|
Lacrimation |
— |
— |
— |
+ (5) |
|
Throat irritation |
— |
— |
+ (3) |
+ (8) |
|
Chest irritation |
— |
— |
— |
+ (1) |
|
aNumber of volunteers showing a response out of a total of 10 participating. Source: Data from Industrial Bio-Test Laboratories 1973, as cited in NIOSH 1974. |
||||
measured for each subject. Throat irritation was reported by two subjects. Nasal irritation with stuffiness similar to that of a cold or nasal dryness was reported by six subjects. The stuffiness lasted for about 24 h. Only two subjects were able to continue nasal breathing for the full 30 min, the others changing to mouth breathing on account of nasal dryness and irritation. Hypoesthesia (decreased sensitivity) of the skin around the nose and mouth was experienced by all subjects, and excessive lacrimation was reported by two. Hyperventilation (increases in the respiratory rates and minute volumes) occurred in all subjects. Hyperventilation occurred immediately in three subjects, was delayed for 10-30 min in the remaining four, and fluctuated with a 25% decrease at 4- to 7-min intervals. The increase in the minute volume was 141-289%. No coughing was reported; the authors noted that 1,000 ppm caused immediate coughing. This study showed that irritation of the upper respiratory tract and throat occurred in subjects inhaling 500 ppm of anhydrous ammonia for 15-30 min. There was no difference in the effects noted in the subject inhaling ammonia for 15 min and those inhaling ammonia for 30 min.
Verberk (1977) examined the effects of ammonia on respiratory function and recorded the subjective responses of two groups of subjects. One group consisted of eight individuals familiar with the effects of ammonia and who had no previous exposure (expert group, 29-53 years old); the other group consisted of eight university students unfamiliar with the effects of ammonia or with experiments in laboratory situations (nonexpert group, 18-30 years old). The subjects were paid for their participation and were informed that the study involved subjective effects and posed no danger to their health at the concentrations used. The subjects had the opportunity to leave the chamber before the test was completed. Four members of each group were smokers. Each group was exposed to ammonia at concentrations of 50, 80, 110, and 140 ppm for up to 2 h. Subjective responses (e.g., smell, eye irritation, throat irritation, cough) were recorded every 15 min and parameters of respiratory function (vital capacity, forced expiratory volume (FEV1 s), forced inspiratory volume (FIV1 s) were measured before exposure and after the 2-h exposure. Subjective responses were rated on a scale of 0-5 (0 = no sensation; 1 = just perceptible; 2 = distinctly perceptible; 3 = nuisance; 4 = offensive; and 5 = unbearable). Chamber concentrations were monitored instantaneously using an infrared spectrometer. There was no effect on respiratory function in either group inhaling any concentration of ammonia.
Table 2-4 summarizes the average and range of responses for both groups. Generally, the expert group scored responses lower than those of the nonexpert group. Four nonexpert subjects exposed to 140 ppm left the exposure chamber between 30 min and 1 h, and none remained in the chamber for the full 2 h. The greatest difference in responses between the expert and nonexpert groups was in general discomfort. The expert group perceived no general discomfort even after exposure to the highest concentration for 2 h, whereas the four nonexpert subjects perceived their general discomfort to range from “distinctly perceptible” to “unbearable” after 1 h. This study showed dose- and duration-response relation-
TABLE 2-4 Average (Range) Scores of Subjective Responses of Expert and Nonexpert Subjects Exposed to Ammoniaa
|
Response |
50 ppm |
80 ppm |
110 ppm |
140 ppmc |
||||
|
Expert |
Nonexpert |
Expert |
Nonexpert |
Expert |
Nonexpert |
Expert |
Nonexpertc |
|
|
Smell |
|
|
|
|
|
|
|
|
|
½ h |
2.0 (1-3)b |
2.5 (2-3) |
2.0 (1-3) |
3.0 (2-4.5) |
2.0 (2-3) |
3.0 (2-4) |
2.0 (1-3) |
4.0 (2-4.5) |
|
1 h |
2.0 (1-3) |
2.5 (1-4) |
2.0 (1-3) |
3.0 (2-4) |
2.0 (2-3) |
3.0 (2-4) |
2.0 (1-3) |
4.0 (3.5-4.5) |
|
2 h |
2.0 (0.5-3) |
3.0 (2-4) |
1.5 (0.5-3) |
3.0 (2-4) |
2.0 (1.5-3) |
3.0 (2-4) |
2.0 (1-3) |
WDc |
|
Eye irritation |
|
|
|
|
|
|
|
|
|
½ h |
1.5 (0-3) |
0.8 (0-3) |
1.5 (1-2) |
1.5 (0-4) |
2.5 (1-3) |
2.5 (0-4) |
3.0 (1.5-3.5) |
3.0 (1-4.8) |
|
1 h |
1.5 (0-3) |
0.8 (0-3) |
2.0 (0-3) |
1.5 (0-3) |
2.5 (2-3.5) |
2.5 (0-4) |
2.0 (2-3) |
3.5 (1-5) |
|
2 h |
1.0 (0-2) |
1.2 (0-3) |
1.5(0-2) |
2.0 (0-4) |
2.0 (0.3-3) |
2.5 (0-4) |
2.5 (1-3) |
WD |
|
Throat irritation |
|
|
|
|
|
|
|
|
|
½ h |
0.4 (0-2) |
0.4 (0-1) |
0.8 (0-2) |
1.0 (0-3 |
1.5 (0-3.5) |
2.0 (0-4) |
1.0 (0-2) |
3.7 (3.5-5) |
|
1 h |
0.4 (0-3) |
0.5 (0-3) |
1.0 (0-3) |
1.4 (1-3) |
1.4 (0-3) |
2.5 (1-4) |
1.5 (0-2) |
4.5 (2-4) |
|
2 h |
0.7 (0-3) |
1.5 (0.3) |
0.8 (0-2) |
2.0 (0-4) |
1.0 (0-2) |
3.0 (2-4) |
1.0 (0-3.7) |
WD |
|
Urge to cough |
|
|
|
|
|
|
|
|
|
½ h |
0.2 (0-1.2) |
0.2 (0-1) |
0.3 (0-1) |
0.5 (0-2) |
0.8 (0-2) |
1.5 (0-2) |
0.5 (0-2) |
2.0 (0-5) |
|
1 h |
0.3 (0-2) |
0.2 (0-2) |
0.5 (0-2) |
1.0 (0-2) |
0.5 (0-3.5) |
1.7 (0-3) |
0.6 (0-2.5) |
1.7 (0-3) |
|
2 h |
0.3 (0-2) |
0.4 (0-2) |
0.4 (0-2) |
0.3 (0-4) |
0.3 (0-2.5) |
1.7 (0-4) |
0.4 (0-2.3) |
WD |
|
General discomfort |
|
|
|
|
|
|
|
|
|
½ h |
0 |
0.1 (0-1) |
0 |
1.0 (0-3) |
0.2 (0-2) |
1.0 (0-3) |
0 |
2.2 (0-4) |
|
1 h |
0 |
0.2 (0-1) |
0 |
1.2 (0-3) |
0.2 (0-1) |
1.2 (0-3) |
0 |
3.3 (0-4.7) |
|
2 h |
0 |
1.0 (0-2) |
0 |
1.3 (0-3) |
0.3 (0-1) |
1.5 (0-4) |
0 |
WD |
|
Irritation to chest |
Similar to urge to cough, but scores tended to be a little lower. |
|||||||
|
aExpert subjects: individuals who were familiar with the effects of ammonia and who had no previous exposure; nonexperts students were unfamiliar with the effects of ammonia or with experiments in laboratory situations. bBased ona scale of 1-5: 0 = no sensation; 1 = just perceptible; 2 = distinctly perceptible; 3 = nuisance; 4 = offensive; and 5 = unbearable. cOnly four of the nonexpert subjects tolerated the ammonia for 1 h; none of the nonexpert subjects tolerated the ammonia for 2h. Source: Adapted fromVerberk 1977. |
||||||||
ships for the effects of ammonia, particularly for the nonexpert subjects. This study also showed that general knowledge about the chemical may help alleviate the concern about exposure and the intensity of the symptoms experienced during exposure.
Cole et al. (1977) studied the effects of exercise on 18 servicemen who inhaled ammonia at concentrations of 71, 106, 144, or 235 mg/m3 (102, 152, 206, or 336 ppm). The subjects were exposed for durations of between 95 and 120 min while cycling under a load of 20 watts increased up to 180 watts in 20-watt increments (based on assumptions of “zero time” and extrapolation from figures of Cole et al.). The same subjects served as their own controls. Measurements of respiratory parameters (respiratory rate, minute volume, tidal volume, and oxygen uptake) and cardiac frequency were taken under control conditions when the subjects inhaled air only and during the experimental conditions when the subjects inhaled ammonia. During exposure to ammonia, the subjects noted only a sensation in the nose and a slight dryness of the mouth. Minute volume was decreased by 8%, 10%, and 6% at 152, 206, and 336 ppm, respectively, compared with control measurements; statistical significance was achieved for all three concentrations. However, no clear dose-related trend was observed relative to the control measurements. The tidal volume was significantly decreased (9 and 8%, respectively) and respiratory frequency was increased (10 and 8% respectively) at 206 and 336 ppm compared with the control values, but there was no clear dose-response relationship. The small changes in tidal volume and respiratory frequency are unlikely to be clinically significant.
Sundblad et al. (2004) studied the acute effects of repeated low-level ammonia exposures of human subjects at rest and performing ergometric exercise. Twelve healthy atopic adults (seven females and five males, 21-28 years old, with a mean age of 25) with no reported present or past symptoms of allergy or airway disease were exposed in a 20-m3 stainless steel chamber to ammonia at 0, 5, and, 25 ppm for 3 h on three separate occasions separated by at least 7 days in which subjects did not undergo experimental ammonia exposures. Exposure concentrations were monitored by infrared spectrophotometry. During each 3-h exposure period, 1.5 h was spent at seated rest and 1.5 h was spent exercising at 50 watts on a bicycle ergometer; activity was changed every 30 min. At specific times during exposure and 1.5 h postexposure, the subjects rated their level of discomfort related to odor, eyes, and airway symptoms and general symptoms (such as headache, dizziness, nausea, “feeling of intoxication”) on a scale of 0-100. The general symptoms were characterized by Sundblad and co-workers as central nervous system (CNS) effects. Sundblad et al. (2004) performed no neurophysiological measurements or studies showing systemic uptake of ammonia.
Subjective symptom rankings by questionnaire exhibited a dose-response relationship. Based on examination of questionnaire results, Sundblad et al. (2004) noted a tendency of sensory adaptation to “solvent smell” among those exposed to 5 ppm but not those exposed to 25 ppm. Ratings of symptoms related to eye and respiratory irritation and general symptoms were significantly greater in the 25-ppm exposure group than those of controls, while about half of the
symptoms experienced by the 5-ppm exposure group exhibited higher rankings than in the control group. Average rating of irritation and the CNS symptoms did not exceed “rather” (rating of 48). All symptomatic effects were transient.
Sundblad et al. (2004) collected pretrial and posttrial measurements to characterize lung function, methacholine challenge, cell composition in nasal lavage fluids, total and differential peripheral leukocyte counts, complement factor C3b, exhaled nitric oxide, body temperature, and peak expiratory flow. Under the Sundblad et al. experimental protocol, ammonia at 5 or 25 ppm did not induce detectable changes in pulmonary function or total cell concentration in nasal lavage fluid or induce an exposure-related bronchial response to methacholine, an increase in exhaled nitric oxide, an increase in the total or differential leukocyte, or a change in complement factor C3b.
Ferguson et al. (1977) reported that workers in their company in 1972 did not voluntarily use gas masks until ammonia concentrations reached 400 or 500 ppm. They also reported that before 1951 workers were subjected to continuous concentrations ranging from 150 to 200 ppm. To establish the bounds for controlled exposure studies, they conducted two reconnaissance experiments. In the first experiment they reported that four male subjects were able to tolerate “continued exposure” of 130-150 ppm (duration not reported) after exposure to lower concentrations for <2 h. In the second experiment they noted that in the bicarbonate plant, after 30 min of acclimation at 100 ppm, a 30-s exposure at 300 ppm was just barely tolerable.
In the controlled exposure study, Ferguson et al. assessed the effect of ammonia on six (three groups of two) human volunteers (industrial workers) exposed to concentrations of 25, 50, or 100 ppm after exposure to the same concentrations during a 1-week practice period. The subjects were exposed at a sodium bicarbonate plant in areas where concentrations of 25 and 50 ppm were achieved; the subjects were exposed to 100 ppm in an exposure chamber. Ammonia concentrations were monitored each half hour using detector tubes certified by the National Institute for Occupational Safety and Health (NIOSH) that had an overall accuracy of ± 10%. Exposure periods ranged from 2 to 6 h/day for 5 weeks. There was no adverse effect on respiratory function and no increase in the frequency of eye, nose, and throat irritation with increasing concentrations. The only complaints were lacrimation and nasal dryness during brief excursions above 150 ppm. There was no interference with performance of work duties and no effect on pulse rate or respiratory function during exercise (i.e., no effect on physical or mental ability to perform work duties) that was consistent with concentration or duration. Definite redness of the nasal mucosa occurred in one subject exposed to 100 ppm with excursion up to 200 ppm, but the effect cleared by the next morning (i.e., no lasting effects occurred). Four of the six subjects were exposed to different concentrations, making it difficult to establish trends related to exposure concentration or duration.
Erskine et al. (1993) measured the threshold concentration of ammonia required to elicit reflex glottis closure, which is a protective response stimulated by inhaling irritant or noxious vapors at concentrations too low to produce
cough. It is accompanied by a brief pause in inspiration. The investigators measured glottis closure in 102 healthy nonsmoking subjects, ranging from 17 to 96 years old, after single intermittent breaths of ammonia vapor using an inspiratory pneumotachograph. The results showed a strong positive correlation coefficient of .85 between age and the threshold concentration. The younger subjects were more sensitive, with the reflex response occurring at 571±41.5 ppm (± standard error) in subjects 21-30 years old compared with 1,791±52 ppm (± standard error) in subjects 86 to 95 years old. The threshold was about 1,000 ppm for 60-year-old subjects. The data showed that younger people are about three times more sensitive to the induction of this protective mechanism (glottis closure) by ammonia than the elderly.
2.2.2.
Epidemiologic Studies
Holness et al. (1989) compared the respiratory effects in a group of 58 workers (51 production and six maintenance workers at Allied Chemical Canada, Ltd.) exposed to ammonia during the production of soda ash with 31 control workers from stores and offices. The exposed group had worked in soda ash production for an average of 12.2 years. The workers were assessed at the beginning of a workweek and at the end of the workweek. They were assessed based on a questionnaire, sense of smell, and pulmonary function. The time-weighted average ammonia concentration was 9.2±1.4 ppm (mean ± standard deviation) for the exposed workers compared with 0.3±0.1 ppm for a control group assessed over one workweek. The investigators reported essentially no differences in the parameters assessed comparing the first and last days of the workweek and no differences based on level or length of exposure to ammonia. There were no differences between the two groups.
Minor pulmonary function deficits have been observed in swine workers exposed to ammonia, in combination with dust and endotoxin (Reynolds et al. 1996). While ammonia levels as high as 200 ppm have been reported (Carlile 1984), mean exposure levels of 4-7 ppm are more typical for workers (Reynolds et al. 1996; Donham et al. 1995). Confounding due to exposure to multiple agents and lack of information on clinical symptoms limit the usefulness of these data.
2.3.
Summary
Numerous case studies describing disabling, irreversible, or long-term effects on humans inhaling ammonia at high concentrations were available in the literature. However, measured concentrations were not available for any of these studies.
Dose reconstruction has been conducted using WHAZAN and HGSYSTEM models to predict atmospheric ammonia concentrations produced dur-
ing the Houston and Potchefstroom accidents. LC50 values were estimated from results of each model. An evaluation of these models is presented in Section 7.1.
Sensitive individuals include children, elderly people, and people with respiratory or heart disorders. For very brief (<1 min) high-level exposures, decreased sensitivity of reflex glottis closure in elderly people implies a loss of protective reflexes, which could increase the risk of damage to the lower respiratory tract from the effects from inhaled ammonia in the elderly.
Ammonia causes severe irritation and burning to the skin, eyes, oral cavity, and respiratory tract, particularly mucous surfaces immediately upon contact due to the rapid conversion of ammonia to the very caustic ammonium hydroxide. Therefore, acute exposure to very high concentrations of ammonia severely damages the pulmonary region (bronchiolar and alveolar) of the respiratory tract, with permanent injury or death likely, even with prompt medical attention. Pulmonary edema, in particular, signals a poor prognosis for recovery in the short term, and secondary effects such as bronchiectasis, bronchopneumonia, and emphysema have occurred in individuals who survived for several days or sometimes several years. The damage caused by ammonia is progressive down the respiratory tract, starting with irritation of the nasopharyngeal region, extending to the tracheobronchial region, and finally the bronchiolar and alveolar regions.
Humans who have inhaled ammonia at concentrations high enough to experience disabling effects without causing death usually experience severe damage to the eyes, oral cavity, and respiratory tract involving the tracheobronchial region. Severe damage to the eyes can cause permanent visual deterioration or blindness. Damage to the pharynx and/or tracheobronchial regions may cause airway obstruction that could lead to death if medical help is not available. Damage to the lungs (particularly the bronchioles) may be manifested by bronchopneumonia. Chronic effects of acute exposure to ammonia (manifested years after exposure) have included bronchiectasis, bronchiolitis, atelectasis, emphysema, chronic bronchitis, and reduced performance in pulmonary function tests. The long-term effects are considered to be secondary to the initial damage caused by ammonia.
Nondisabling and reversible effects of ammonia are summarized in Table 2-5.
3.
ANIMAL TOXICITY DATA
3.1.
Acute Lethality
3.1.1.
Rats
Groups of 10 male CFE rats were exposed to 0, 6,210, 7,820, or 9,840 ppm (0, 4,343, 5,468, or 6,881 mg/m3, respectively) of ammonia for 1 h; surviving
TABLE 2-5 Summary of Nondisabling and Reversible Effects of Inhaled Ammonia in Humans
|
Concentration |
Duration of Exposure |
Effecta |
Reference |
|
5 ppm |
3 h, with rest and exercise for 1.5 h each |
Subjective rating of eye discomfort and smell, headache, dizziness, and “feeling of intoxication” significantly greater than of controls; sensory adaptation to odor; no exposure-related change in pulmonary function, increase in nasal cells, no increase in exhaled NO, and no alteration in bronchial response to methacholine. |
Sundblad et al. 2004 |
|
25 ppm |
3 h, with rest and exercise for 1.5 h each |
Subjective rating of eye, upper respiratory, and throat irritation, smell, headache, dizziness, and “feeling of intoxication” significantly greater than of controls; no sensory. Adaptation to odor; no exposure-related change in pulmonary function, increase in nasal cells, no increase in inhaled NO, and no alteration in bronchial response to methacholine. |
Sundblad et al. 2004 |
|
30 ppm |
10 min |
Odor was moderately intense to highly penetrating; irritation was faint or not detectable. |
MacEwen et al. 1970 |
|
32 ppm |
5 min |
Nasal dryness. |
Industrial Bio-Test Laboratories 1972 |
|
35 ppm |
5 min |
Nasal dryness. |
Industrial Bio-Test Laboratoris 1973 |
|
50 ppm |
10 min |
Highly penetrating odor; moderate irritation. |
MacEwen et al. 1970 |
|
50 ppm |
30 min |
Moderately intense odor; moderate irritation to eyes and nose; mild irritation to throat and chest; slight urge to cough; slight general discomfort. |
Verberk 1977 |
|
50 ppm |
1 h |
Highly intense odor; moderate irritation to eyes, nose, throat, and chest; mild urge to cough; slight general discomfort. |
Verberk 1977 |
|
50 ppm |
2 h |
Offensive odor; moderate irritation to eyes, nose, throat, and chest; mild urge to cough; mild general discomfort |
Verberk 1977 |
|
72 ppm |
5 min |
Nasal, eye, and throat irritation. |
Industrial Bio-Test Laboratories 1973 |
|
80 ppm |
30 min |
Highly intense odor; highly intense eye and nose irritation; moderate throat and chest irritation; mild urge to cough; moderate general discomfort. |
Verberk 1977 |
|
80 ppm |
1 h |
Highly intense odor; moderate eye, nose, throat, and chest irritation; mild urge to cough; moderate general discomfort. |
Verberk 1977 |
|
Concentration |
Duration of Exposure |
Effecta |
Reference |
|
80 ppm |
2 h |
Highly intense odor; highly intense eye, nose, throat, and chest irritation; highly intense urge to cough; and moderate general discomfort. |
Verberk, 1977 |
|
100 ppm |
5-30 s |
Significant increase in nasal airway resistance, but atopic subjects, including asthmatics, responded similarly to the nonatopic subjects. |
McLean et al. 1979 |
|
100 ppm |
2-6 h/day, 5 weeks |
No adverse effects on respiratory function and no increase in frequency of eye, nose, or throat irritation. |
Ferguson et al. 1977 |
|
110 ppm |
30 min |
Highly intense odor; highly intense eye, nose, throat, and chest irritation, mild urge to cough, and moderate general discomfort. |
Verberk 1977 |
|
110 ppm |
1 h |
Highly intense odor; highly intense eye, nose, throat, and chest irritation; moderate urge to cough; moderate general discomfort. |
Verberk 1977 |
|
110 ppm |
2 h |
Highly intense odor; highly intense eye, nose, throat, chest irritation; urge to cough; general discomfort. |
Verberk 1977 |
|
140 ppm |
30 min |
Highly intense odor; unbearable eye, nose, throat, and chest irritation; mild urge to cough; moderate general discomfort. |
Verberk 1977 |
|
140 ppm |
1 h |
Highly intense odor; unbearable eye, nose, throat, and chest irritation; moderate urge to cough; moderate general discomfort. |
Verberk 1977 |
|
140 ppm |
2 h |
Highly intense odor; unbearable eye and nose irritation; highly intense throat and chest irritation; highly intense urge to cough; unbearable general discomfort. |
Verberk 1977 |
|
143 ppm |
5 min |
Nose, eye, throat, and chest irritation; lacrimation. |
Industrial Bio-Test Laboratories 1973 |
|
500 ppm |
15-30 min |
Nose and throat irritation; nasal dryness and stuffiness; excessive lacrimation; hyperventilation; unbearable. |
Silverman et al. 1949 |
|
570 ppm |
Single breath |
Threshold for reflex glottis closure, 21 to 30-year-old subjects. |
Erskine et al. 1993 |
|
1,000 ppm |
Single breath |
Threshold for reflex glottis closure, 60-year-old subjects. |
Erskine et al. 1993 |
|
1,000 ppm |
NR |
Immediate urge to cough. |
Silverman et al. 1949 |
|
1,790 ppm |
Single breath |
Threshold for reflex glottis closure, 86 to 90-year-old subjects. |
Erskine et al. 1993 |
|
aThe categories from Verberk (1977) have been recategorized as follows: just perceptible = slight; distinctly perceptible = mild; nuisance = moderate; offensive= highly intense; unbearable = unbearable. NR = not reported. |
|||
animals were observed for 14 days (MacEwen and Vernot 1972). Signs of eye and nasal irritation were seen immediately, followed by labored breathing and gasping. Surviving animals exposed to the low concentration weighed less than controls on day 14, and gross examination showed mottling of the liver and fatty changes at the two highest concentrations. All rats exposed to 6,210 ppm survived, and eight exposed to 7,820 ppm and nine exposed to 9,840 ppm died. The LC50 was 7,338 ppm (95% confidence interval = 6,822-7,893 ppm).
Appelman et al. (1982) calculated LC50 values for 7- to 8-week-old male and female Wistar rats exposed to ammonia by inhalation. Five animals of each sex per group were exposed to ammonia at concentrations ranging from 9,870 to 37,820 mg/m3 (14,114-54,083 ppm) for 10, 20, 40, or 60 min and observed for 14 days. Clinical signs of toxicity during exposure included restlessness, closing of the eyes, signs of eye irritation (particularly for 60-min exposures), eye discharge (after 30 min), wet noses, and nasal discharge. Mouth breathing and signs of dyspnea also were observed; the signs of dyspnea disappeared within 24 h after exposure terminated. Gross findings included hemorrhagic lungs in animals dying early and those killed at termination. The lowest concentrations causing death were 23,389 mg/m3 (33,446 ppm) for a 10-min exposure to males, 18,290 mg/m3 (26,155 ppm) for a 20-min exposure (30% mortality) to males, 12,620 mg/m3 (18,047 ppm) for a 40-min exposure to males, and 9,870 mg/m3 (14,114 ppm) for a 60-min exposure to males and females. The LC50 values and mortality rates for male, female, and male and female rats combined as reported by Appelman et al. (1982) are summarized in Table 2-6. The data showed that the LC50 values were significantly higher in male rats than in females for the 20-, 40-, and 60-min exposures.
Coon et al. (1970) exposed male and female Sprague-Dawley or Long-Evans rats repeatedly or continuously to ammonia for various durations. No clinical signs of toxicity or gross pathologic findings were reported for 15 rats exposed to 222 ppm (155 mg/m3) 8 h/day for 6 weeks. No deaths or clinical signs of toxicity were reported for 15 rats similarly exposed to 1,101 ppm (770 mg/m3); nonspecific inflammatory changes, which were slightly more severe than in controls, were observed in the lungs. Continuous exposure of 15 rats to 57 ppm (40 mg/m3) for 114 days resulted in no clinical signs of toxicity or other clinically significant effects compared with the controls. Continuous exposure of 48 rats to ammonia for 90 days resulted in no clinical signs of toxicity or other effects at 182 ppm (127 mg/m3). Mild nasal discharge observed in about 25% of 49 rats was the only clinical sign attributed to the 90-day continuous exposure to 375 ppm (262 mg/m3). Mild signs of dyspnea, nasal irritation, and 98% mortality occurred among 51 rats exposed to 651 ppm (455 mg/m3) continuously for 65 days (exposure terminated early); histopathologic examinations were not conducted on these animals. Thirteen of 15 rats (87%) died during a 90-day continuous exposure to 672 ppm (470 mg/m3). Histopathologic lesions included focal or diffuse interstitial pneumonitis in the lungs of all animals examined and renal tubular calcification, bronchial epithelial calcification, renal tubular epi-
TABLE 2-6 Acute Lethality Data for Male and Female Rats Exposed to Ammonia
|
Experimental Concentration (ppm) |
Exposure Time (min) |
Mortality Rate |
LC50 (ppm) |
||
|
29,959 |
10 |
0/5 |
0/5 |
0/10 |
|
|
33,433 |
|
1/5 |
0/5 |
1/10 |
|
|
37,766 |
|
5/5 |
1/5 |
6/10 |
37,094 (male) |
|
38,925 |
|
5/5 |
0/5 |
5/10 |
44,945 (female) |
|
54,083 |
|
5/5 |
4/5 |
9/10 |
40,300 (male and female) |
|
26,155 |
20 |
3/5 |
0/5 |
3/10 |
|
|
27,213 |
|
1/5 |
0/5 |
1/10 |
|
|
28,814 |
|
5/5 |
2/5 |
7/10 |
25,511 (male) |
|
29,201 |
|
3/5 |
3/5 |
6/10 |
32,661 (female) |
|
33,176 |
|
5/0 |
4/5 |
9/10 |
28,595 (male and female) |
|
18,047 |
40 |
2/5 |
0/5 |
2/10 |
|
|
19,176 |
|
4/5 |
1/5 |
5/10 |
|
|
22,694 |
|
4/5 |
1/5 |
5/10 |
17,532 (male) |
|
23,295 |
|
5/5 |
3/5 |
8/10 |
23,724 (female) |
|
24,081 |
|
5/5 |
2/5 |
7/10 |
20,300 (male and female) |
|
14,114 |
60 |
2/5 |
1/5 |
3/10 |
|
|
14,629 |
|
4/5 |
0/5 |
4/10 |
|
|
16,159 |
|
5/5 |
0/5 |
5/10 |
14,086 (male) |
|
17,875 |
|
5/5 |
1/5 |
6/10 |
19,691 (female) |
|
18,933 |
|
5/5 |
2/5 |
7/10 |
6,600 (male and female) |
|
Source: Appelman et al. 1982. Reprinted with permission; copyright 1982, American Industrial Hygiene Association Journal. |
|||||
thelial cell proliferation, myocardial fibrosis, and fatty changes in the liver of several animals. These effects also occurred in control animals, but the severity was greater in the exposed animals.
3.1.2.
Mice
Silver and McGrath (1948) calculated the LC50 value for mice exposed to ammonia (6.1-9.0 mg/L or 8,723-12,870 ppm) by inhalation for 10 min and observed for 10 days. The concentrations of ammonia in the exposure chamber were measured analytically. Each group consisted of 20 mice (sex and strain not specified). During exposure the mice closed their eyes, exhibited great excitement initially but soon became quiet, gasped, pawed, scratched their noses, and convulsed before dying. At the lowest concentration of 8,723 ppm, 25% of the animals died, and 80% died at the highest concentration of 12,870 ppm. Overall 90/180 mice died during the second 5-min of exposure and another eight died during the observation period. The other animals surviving exposure recovered rapidly. The LC50 for the 10-min exposure was 7.06 mg/L (10,096 ppm).
Groups of 10 male CF1 mice were exposed to ammonia at analytically measured concentrations of 0, 3,600, 4,550, or 5,720 ppm (0, 2,520, 3,185, 4,004 mg/m3) for 1 h (MacEwen and Vernot 1972). Immediately upon exposure, the animals showed signs of nasal and eye irritation, followed by labored breathing and gasping. Animals surviving the low and intermediate concentrations lost weight during the 14-day observation period. Gross examination of surviving mice showed mild congestion of the liver at the intermediate and high concentrations. Three mice exposed to 4,500 ppm died, and nine exposed to 5,720 ppm died, but none exposed to 3,600 ppm died. The LC50 was 4,837 ppm (95% confidence interval = 4,409-5,305 ppm).
In a study by Hilado et al. (1977), four Swiss mice per group were exposed to 7,143-28,571 ppm of ammonia for 30 min. Exposure concentrations were calculated rather than measured analytically. One mouse died at 19,048 ppm, two at 21,429 ppm, three at 23,810 ppm, and four each at 26,190 and 28,571 ppm. All deaths occurred during exposure except the death at the lowest concentration, which occurred 1 day after exposure. No deaths occurred after exposure to concentrations of 14,286 ppm or lower. The LC50 value was reported as 21,000 ppm for the 30-min exposure. In 1978, Hilado et al. reported the LC50 as 21,430 ppm for the 30-min exposure; the previous value was probably rounded to two significant figures.
Kapeghian et al. (1982) determined the LC50 value for male albino ICR mice (12/group) exposed to 1,190-4,860 ppm of ammonia for 1 h. Concentrations of ammonia in the exposure chambers were measured analytically. The animals were observed for 14 days following exposure. A control group exposed to air only was included for comparison. Clinical signs, which were noted immediately and lasted 5-10 min, included excitation/escape behavior, rapid vigorous tail revolution, blinking and scratching (eye and nose irritation), and dyspnea. As signs of irritation decreased, the animals became less active and other signs of toxicity were noted, including tremors, ataxia, clonic convulsions, frothing, coma, final tonic extensor seizure, and death. At the higher concentrations, almost all deaths (90%) occurred during the first 15-20 min of exposure and as late as 45 min at the lower concentrations. Additional deaths occurred during the first 3 days following exposure. All deaths occurred at concentrations ≥3,950 ppm (25 to 100% mortality). The mortality response was 22/24 at 4,860 ppm; 8/12 at 4,490 ppm; 5/12 at 4,220 ppm; 3/12 at 3,950 ppm; and 0/12 at 3,440, 2,130, 1,340, and 1,190 ppm. The LC50 was 4,230 ppm for the 1-h inhalation exposure to ammonia. Other effects observed during the 14-day observation period included lethargy, dyspnea, weight loss, and a “humped back” appearance. The pathologic lesions occurring in mice that died during exposure included acute vascular congestion, intra-alveolar hemorrhage, disruption of alveolar septal continuity, and acute congestion of hepatic sinusoids and blood vessels. In animals surviving the 14-day observation period, pathologic lesions included mild to moderate pneumonitis (dose-related severity), focal atelectasia in the lungs (4,860 ppm), and degenerative hepatic lesions (dose-related sever-
ity, 3,440 to 4,860 ppm). The author did not discuss specific effects in animals exposed to concentrations less than 3,440 ppm.
Groups of 12 male albino ICR mice were exposed to ammonia at concentrations of 0, 1,350, or 4,380 ppm for 4 h and the effects of ammonia on hexobarbital-induced latency to hypnosis (time to loss of righting reflex) and sleeping time were assessed 1 h after exposure terminated (Kapeghian et al. 1985). All mice exposed to 1,350 ppm survived; three mice exposed to 4,380 ppm died during exposure and one died during hexobarbital hypnosis. Latency to hypnosis was significantly reduced in animals exposed to both concentrations compared with controls exposed to air only. Hexobarbital sleeping time was significantly increased in animals exposed to 4,380 ppm of ammonia. The hexabarbital effects were not attributed directly to exposure to ammonia.
3.2.
Nonlethal Toxicity
3.2.1.
Rats
Dalhamn (1956) studied the effect of inhaling ammonia on tracheal ciliary activity in male Wistar rats. Two or three rats per group were exposed to 0, 3, 6.5, 10, 20, 45, or 90 ppm of ammonia for 10 min. No effects were observed in rats exposed to air. In rats exposed to ammonia, ciliary activity ceased in 7-8 min with 3 ppm, 150 s with 6.5 ppm, 20 s with 20 ppm, 10 s with 45 ppm, and 5 s with 90 ppm. Thus, the time required for ciliary activity to cease showed a concentration-response relationship. Within 20-30 s after exposure was terminated, ciliary activity resumed.
The behavioral activity (wheel running) was assessed in three male Long-Evans rats exposed sequentially to the following concentrations of ammonia: 100, 300, 300, or 100 ppm for 6 h for each session with 2 days separating each session (Tepper et al. 1985). The activity of the rats on the running wheel was recorded during exposure and the time between exposures. The rats had previously been exposed to ozone in a similar experiment that was terminated 2 weeks before starting the experiment with ammonia. Controls were not described, but the performance of treated animals was compared to control performances, probably conducted before exposure to ozone. Exposure to 100 ppm of ammonia resulted in an immediate 61% reduction in activity compared with control activity; activity on the wheel ceased almost completely throughout exposure at 300 ppm. After termination of exposure to either 100 or 300 ppm, the activity of the rats steadily increased to 154% and 185%, respectively, compared with that of controls during the first 4 h postexposure.
Groups of eight male rats (Crl:COBS CD[SD]) were exposed to ammonia at concentrations of 15, 32, 310, or 1,157 ppm for 24 h (Schaerdel et al. 1983). No behavioral changes or evidence of irritation to the eyes or mucous membranes were observed. Blood gases (pO2 and pCO2) and pH were measured at 0, 8, 12, and 24 h; no changes were noted for pCO2 and pH. Small changes within
the normal range for rats occurred for pO2. Groups of seven rats were also exposed continuously to ammonia at concentrations of 0, 4, 24, 44, 165, or 714 ppm for 3 or 7 days. Minimal lesions were seen in the respiratory epithelium of the nasal cavity in animals exposed for 7 days (the authors did not indicate which concentrations of ammonia caused the lesions).
Pinson et al. (1986) showed that respiratory mycoplasmosis is exacerbated by exposure to ammonia. Groups of F344/N rats infected with Mycoplasma pulmonis or uninfected were exposed continuously to 100 ppm of ammonia for 3, 5, 7, and 9 days after inoculation to assess the histopathologic effect on the respiratory tract. Ammonia caused hyperplasia and degenerative lesions in the respiratory epithelium of the anterior nasal cavity. Submucosal inflammatory lesions were minimal in uninfected animals exposed to ammonia; these lesions were prominent in infected animals and more severe in infected animals exposed to ammonia. There were inconsistencies in the write-up of this report.
Groups of five female Wistar rats were exposed to gaseous ammonia at concentrations of 0, 25, or 300 ppm for 6 h/day for 5, 10, or 15 days (Manninen et al. 1988). Clinical signs of toxicity were not described. Gross lesions included large hemorrhages on the surfaces of the lungs in several exposed rats (exposure group not reported) and a few control rats, suggesting that the effect may not be treatment related. There were no signs of tracheobronchial or alveolar damage or histopathological effects in the respiratory tract. The liver and kidneys were normal in appearance.
3.2.2
Mice
Barrow et al. (1978) calculated RD50 values for ammonia, based on its sensory irritant effects on the upper respiratory tract of the mouse. The RD50 is the concentration expected to elicit a 50% reduction in respiratory rate. Barrow et al. predicted that the RD50 concentration would elicit intense sensory irritation and is expected to be rapidly incapacitating to humans. Groups of four outbred male Swiss Webster mice were exposed to ammonia by inhalation for 30 min. The authors did not report the concentration of ammonia inhaled by the mice, but judging by the graphic representations, the concentrations were 100, 200, 400, and 800 ppm. The maximum depression in respiratory rate was achieved within the initial 2 min of exposure, after which the response diminished. The RD50 was 303 ppm (95% confidence limits = 188-490 ppm) for a 30-min inhalation exposure to ammonia. There was no microscopic examination of the respiratory tract.
In a follow-up study, Buckley et al. (1984) assessed the histopathologic effects of repeated exposures to ammonia at the RD50 concentration of 303 ppm. Groups of 16-24 male Swiss-Webster mice were exposed to 303 ppm of ammonia for 6 h/day for 5 days; an unexposed group served as the control. The respiratory tract was examined in one-half the animals killed immediately after terminating exposure and in the other half killed 3 days later. The authors did not
describe any clinical signs of toxicity. Histopathological findings, which were confined to the respiratory epithelium of the nasal cavity, included minimal exfoliation, erosion, ulceration and necrosis; moderate inflammatory changes; and slight squamous metaplasia. No lesions were seen in the tracheobronchial or pulmonary regions.
In a similar study, Zissu (1995) exposed groups of 10 male Swiss OF1 mice to ammonia at analytically measured concentrations of 0.3 × RD50 (78.0 ppm), RD50 (257 ppm), or 3 × RD50 (711 ppm) for 6 h/day for 4, 9, or 14 days. The three target concentrations were 90.9, 303, and 909 ppm. Control mice were exposed to filtered air. The entire respiratory tract was examined microscopically. No clinical signs of toxicity were noted for mice exposed to ammonia. Pathologic lesions including rhinitis with metaplasia and necrosis were seen only in the respiratory epithelium of the nasal cavity of mice inhaling 711 ppm (3 × RD50); the severity of the lesions increased with duration of exposure, ranging from moderate on day 4, to severe on day 9, and very severe on day 14. No lesions were seen in the controls or in mice inhaling the lower concentrations of ammonia. In contrast to the study conducted by Buckley et al. (1984), this study showed no lesions in the nasal cavity of mice exposed to 257 ppm, which is near the RD50 of 303 ppm.
Behavioral activity (wheel running) was assessed in 6 male Swiss mice exposed sequentially to ammonia at 100, 300, 300, or 100 ppm for 6 h each session with 2 days separating each session (Tepper et al. 1985). The activity of the mice on the running wheel was recorded during each 6-h exposure and for 2 days after each exposure. These mice had been previously exposed to ozone in a similar experiment terminated 2 weeks before starting the experiment using ammonia. Controls were not described, but the performance of treated animals was compared to control performances, probably conducted before exposure to ozone. At 100 ppm, activity showed an initial increase during the first hour, followed by a marked decrease during the third and fourth hours, and an increase exceeding control activity during the fifth and sixth hours. At 300 ppm, activity was suppressed throughout exposure; it returned to control levels after exposure was terminated. The results suggest that the mice adapted to inhaling 100 ppm of ammonia but not to 300 ppm. The authors attributed the decreased activity to the sensory irritant property of ammonia.
3.2.3.
Cats
Four groups of five stray mixed-breed cats were fitted with cuffed endotracheal tubes and subjected to a battery of pulmonary function tests (baseline results) followed by exposure to 1,000 ppm of ammonia gas for 10 min to evaluate the effect of ammonia on pulmonary function and lung pathology. Two unexposed cats were housed with the experimental cats for pathologic comparison (Dodd and Gross 1980). On days 1, 7, 21, and 35 following exposure, a group of cats was given pulmonary function tests, killed, and examined for gross
and microscopic lesions in the lungs. Signs of toxicity included poor general condition, severe dyspnea, anorexia, dehydration, bronchial breath sounds, sonorous and sibilant rhonchi, and coarse rales. Pulmonary function tests showed evidence of airway damage throughout the experiment and central lung damage on day 21. Gross examination of the lungs showed congestion, hemorrhage, edema, and evidence of interstitial emphysema and collapse. Bronchopneumonia, which caused the death of one animal, was commonly seen after day 7. Microscopic examination showed necrosis and sloughing of the bronchial epithelium accompanied by acute inflammation on day 1; no notable findings occurred in the bronchiolar or alveolar regions. Healing of the mucosal epithelium of the bronchi was noted on day 7, and varying degrees of bronchitis, bronchiolitis, bronchopneumonia, and bulbous emphysema were seen on days 21 and 35. The authors attributed the effects on days 21 and 35 to opportunistic bacteria or viruses. They suggested that the effects of ammonia are biphasic, consisting of an acute phase, which could cause death, and a secondary phase, which could cause debilitating chronic respiratory dysfunction.
3.2.4.
Other Species
Boyd et al. (1944) exposed groups of healthy rabbits to ammonia at 10,010 ppm (range 5,005 to 12,441 ppm) [7,000 mg/m3, range 3,500-8,700 mg/m3] for 1 h before or after intratracheal cannulation, which was inserted to collect respiratory tracheal fluid. The mean survival time was 33 h for rabbits exposed before cannulation and 18 h for rabbits exposed after cannulation. Signs of toxicity included marked excitation during the early stages of exposure followed by a “curare-like paralysis.” The major effects of exposure occurred in the respiratory tract at both concentrations, the tracheobronchial and pulmonary regions of animals exposed to ammonia after cannulation and the pulmonary region of animals exposed before cannulation. Microscopically, the trachea and bronchi appeared normal in rabbits exposed before cannulation but were severely damaged in animals exposed after cannulation. Bronchiolar (damage to epithelial lining) and alveolar effects (congestion, edema, atelectasis, hemorrhage, and emphysema) were similar in both groups.
Groups of three rabbits, 15 guinea pigs, two dogs, and three monkeys were exposed to ammonia 8 h/day for 6 weeks at concentrations of 222 or 1,101 ppm (155 or 770 mg/m3; Coon et al. 1970). No clinical signs of toxicity or other clinically significant effects occurred in animals exposed to 222 ppm except for focal pneumonitis in one monkey. The only effects observed at 1,101 ppm were mild to moderate lacrimation and dyspnea in the dogs and rabbits during the first week of exposure only and nonspecific inflammatory changes in the lungs of guinea pigs. The same number of animals of each species was exposed continuously to 57 or 672 ppm (40 or 470 mg/m3) for 90 days; no clinical signs of toxicity or other clinically significant effects were observed at 57 ppm. At 672 ppm, marked eye irritation (heavy lacrimation) and nasal discharge were seen in dogs
and erythema, discharge, and corneal opacity were seen in the rabbits. Hemorrhagic lesions occurred in the lungs of one dog and moderate lung congestion in two rabbits. Focal or diffuse interstitial pneumonitis was seen in all animals; renal tubular calcification, bronchial epithelial calcification, renal tubular epithelial cell proliferation, myocardial fibrosis, and fatty changes in the liver were observed in several animals of each species. Similar lesions were seen in control animals but were more severe in the treated animals. Four guinea pigs died during the experiment (Coon et al. 1970).
3.3.
Summary
The LC50 values for mice and rats are presented in Table 2-7. The LC50 for rats ranged from 7,338 and 16,600 ppm for 60-min exposures to 40,300 ppm for a 10-min exposure. The LC50 for mice ranged from 4,230 ppm for a 60-min exposure to 10,096 ppm for a 10-min exposure. The lowest experimental concentrations associated with lethality are summarized in Table 2-8.
Rats exposed to lethal concentrations of ammonia showed signs of dyspnea and irritation to the eyes and nose and hemorrhage in the lungs (Appelman et al. 1982). Mice exposed to lethal concentrations of ammonia showed signs of irritation to the eyes and nose, labored breathing, and gasping, along with tremors, ataxia, convulsions, and seizures; pathologic lesions occurred in the alveoli (Silver and McGrath 1948; MacEwen and Vernot 1972; Kapeghian et al. 1982).
Nondisabling reversible effects in laboratory animals were mild after single exposures and transient after repeated exposures (subchronic duration), suggesting that adaptation occurred. Rats showed a decrease in tracheal ciliary activity during exposure to 3-90 ppm for 10 min (Dalhamn 1956), a decrease in motor activity (wheel running) during exposure to 100 ppm for 6 h, and a complete cessation of motor activity during exposure to 300 ppm for 6 h (Tepper et al. 1985). Mice exposed to the same concentrations of ammonia showed responses similar to those of the rats. Another study in rats exposed to concentrations of ammonia ranging from 15 to 1,157 ppm for 24 h did not show any behavioral changes or irritation to the eyes or mucous membranes; only minimal effects on the respiratory epithelium of the upper respiratory tract were seen after continuous exposure to concentrations up to 714 ppm for several days (Schaerdel et al. 1983). A 50% reduction in the respiration rate (RD50) was noted in mice exposed to about 300 ppm for 30 min (Barrow et al. 1978). Repeated exposures of the mice to the RD50 for 6 h/day for 3 or 7 days did not cause pathologic lesions in the respiratory epithelium (Buckley et al. 1984), but exposure to approximately three times the RD50 (711 ppm) resulted in slight to moderate exfoliation, erosion, ulceration, and necrosis of the respiratory epithelium of the nasal cavity; no lower respiratory tract lesions were produced (Zissu 1995). The RD50 is considered to be incapacitating to humans (Barrow et al. 1978). There was no evidence of pulmonary lesions in mice or rats exposed to a single nonlethal concentration of ammonia.
TABLE 2-7 Comparison of Acute Lethality (LC50) Data in Different Species
|
Species |
LC50 |
Exposure Time (min) |
Reference |
|
|
mg/m3 |
ppm |
|||
|
Rat |
28,130 |
40,300 |
10 |
Appelman et al. 1982 |
|
Mouse |
7,060 |
10,096 |
10 |
Silver and McGrath 1948 |
|
Rat |
19,960 |
28,595 |
20 |
Appelman et al. 1982 |
|
Mouse |
14,986 |
21,430 |
30 |
Hilado et al. 1978 |
|
Rat |
14,170 |
20,300 |
40 |
Appelman et al. 1982 |
|
Rat |
5,131 |
7,338 |
60 |
MacEwen and Vernot 1972 |
|
Rat |
11,592 |
16,600 |
60 |
Appelman et al. 1982 |
|
Mouse |
3,383 |
4,837 |
60 |
MacEwen and Vernot 1972 |
|
Mouse |
2,858 |
4,230 |
60 |
Kapeghian et al. 1982 |
TABLE 2-8 Lowest Experimental Concentrations Causing Death
|
Species |
Concentration (ppm) |
Exposure Time (min) |
% Mortality |
Reference |
|
Mouse |
8,723 |
10 |
25 |
Silver and McGrath 1948 |
|
Mouse |
19,048 |
30 |
25 |
Hilado et al. 1977 |
|
Mouse |
3,950 |
60 |
25 |
Kapeghian et al. 1982 |
|
Mouse |
4,550 |
60 |
30 |
MacEwen and Vernot 1972 |
|
Mouse |
4,380 |
240a |
25 |
Kapeghian et al. 1985 |
|
Rat |
33,433 |
10 |
10 |
Appelman et al. 1982 |
|
Rat |
26,155 |
20 |
30 |
Appelman et al. 1982 |
|
Rat |
18,047 |
40 |
20 |
Appelman et al. 1982 |
|
Rat |
14,114 |
60 |
30 |
Appelman et al. 1982 |
|
Cat |
1,000 |
10 |
5 |
Dodd and Gross 1980 |
|
aNo observation period after exposure. |
||||
Signs of eye and respiratory irritation were observed in several species exposed continuously or repeatedly to ammonia for 6 weeks to 114 days (Coon et al. 1970). Except for nonspecific inflammation of the lungs at 1,101 ppm, repeated daily exposures to rats of 57 ppm for 114 days or 222 or 1,101 ppm for 6 weeks (8 h/day) produced no effects. Almost all rats died after continuous exposure to 651 or 672 ppm for 65 days. Repeated exposures to 1,101 ppm for 6 weeks (8 h/day) produced transient dyspnea and lacrimation in dogs and rabbits, whereas continuous exposure to 672 ppm for 90 days resulted in signs of irritation to the eyes and nose and pathologic lesions in the lungs of dogs and rabbits and pneumonitis in several species (dog, rabbit, guinea pig, and monkey). Studies on repeated exposures showed that mice are more sensitive than other species; for example, mice exposed to 771 ppm for only a few days showed pathologic effects, whereas other species required higher concentrations or longer exposure durations to produce pathologic or clinical effects (Coon et al. 1970).
4.
SPECIAL CONSIDERATIONS
4.1.
Metabolism, Disposition, and Kinetics
Ammonia is a product of amino acid and protein metabolism; therefore, it is found naturally in the body. The normal concentration of ammonia in the blood of humans is about 1 µg/mL. The liver rapidly detoxifies ammonia to urea in order to maintain the isotonic system (Visek 1972; Pierce 1994).
The concentration of ammonia in blood remains stable (not altered significantly) after inhalation exposure of humans to very high concentrations of ammonia gas, indicating a lack of appreciable absorption from the respiratory tract or rapid detoxification. Leduc et al. (1992) reported normal concentrations of ammonia in the blood of a 28-year-old man exposed to an unknown concentration of ammonia gas that was sufficient to cause severe tracheobronchial injury. Swotinsky and Chase (1990) presented no supporting data but stated that individuals with impaired liver function could have elevated levels of ammonia in blood after inhalation exposure.
Silverman et al. (1949) reported no changes in blood or urine ammonia, urea, or nonprotein nitrogen in seven human subjects exposed to ammonia at concentrations of 350-500 ppm for 30 min. The concentration of ammonia in expired air remained stable at 350-400 ppm after 10-27 min of exposure, suggesting an equilibrium with the concentration in inhaled air. Within 3-8 min following exposure, the concentration of ammonia in expired air decreased to preexposure levels. The calculations of Silverman et al. indicated that, if all the retained ammonia was absorbed into the blood, there would have been no significant change in blood or urine urea, ammonia, or nonprotein nitrogen.
Animal studies have shown, however, that blood ammonia levels may be altered following inhalation exposure. Schaerdel et al. (1983) measured ammonia levels in the blood of rats 8, 12, and 24 h after inhalation exposures to 15, 32, 310, or 1,157 ppm of ammonia for 24 h. The values were corrected by preexposure concentrations (control). The blood ammonia concentrations 8 and 12 h after exposure to 15 and 32 ppm and 24 h after exposure to 15 ppm were slightly below those of the controls. The concentrations of ammonia in the blood of rats exposed to 310 and 1,157 ppm exceeded control levels and showed a peak at 8 h and time-related decreases at 12 and 24 h postexposure. Mayan and Merilan (1976) found no significant increase in the blood ammonia levels in male Holstein calves after exposure to 50 or 100 ppm of ammonia for 2.5 h compared with the preexposure levels. Blood urea nitrogen and pH were not significantly altered after exposure to ammonia. Adult female rabbits exposed to 50 or 100 ppm of ammonia for 2.5-3 h showed a significant increase in blood urea nitrogen at 100 ppm and no significant increase in blood pH (Mayan and Merilan 1972).
During exposure, ammonia is efficiently retained (scrubbed) in the nasopharyngeal region of the respiratory tract, thus protecting the lower regions from damage. However, the work by Silverman et al. (1949) indicated that scrubbing
of ammonia in the nasopharyngeal area is concentration and time dependent. Landahl and Herrmann (1950) showed that 91-93% of ammonia [concentrations = 40, 200, or 300 mg/m3 (57, 286, or 429 ppm); flow rate = 18 L/min] inhaled by human subjects was retained in the respiratory tract during a single inspiration. At the same flow rate, 83% of inhaled ammonia was retained in the nose.
Mongrel dogs exposed to 150-500 mg/m3 (215-715 ppm) of ammonia vapor retained 74-83% of the inhaled ammonia in the entire respiratory tract; 76-80% of inhaled ammonia can be retained in the upper respiratory tract (Egle 1973). The duration of exposure was not reported. Ventilatory rate and tidal volume had no effect on retention. Other experiments showed 78-80% retention in the lower respiratory tract and 88% retention in the upper respiratory tract when mechanical devices were used to bypass the upper and lower respiratory tracts.
4.2.
Mechanism of Toxicity
Ammonia is an irritant gas that produces effects immediately on contact with moist mucous membranes of the eyes, mouth, and respiratory tract via the formation of ammonium hydroxide (a corrosive alkali) or the production of heat (Wong 1995). Because of its irritant properties, individuals coming into contact with ammonia vapor (or gas) will try to escape as quickly as possible (Swotinsky and Chase 1990). The odor threshold for ammonia is lower than its irritancy effect and serves as a warning of its presence.
4.3.
Structure-Activity Relationship
Ammonia is an alkaline substance, and its corrosiveness is not different from that of other corrosive agents such as calcium, sodium, potassium hydroxide, and calcium oxide. Aerosols or vapors and fumes are very caustic on contact with moist mucous membranes, causing injury of the respiratory tract and eyes (Pierce 1994).
4.4.
Other Relevant Information
4.4.1.
Odor
The odor threshold for ammonia is between 5 and 53 ppm (Pierce 1994), suggesting that it has adequate warning properties. Ferguson et al. (1977) reported the odor threshold for ammonia in the presence of mixed odors as 10-20 ppm. The odor of ammonia at 30 ppm described as moderately intense by 2/6 subjects and highly penetrating by 3/6, indicating that the odor threshold was clearly exceeded at 30 ppm (MacEwen et al. 1970). A group of nonexpert and
expert subjects judged the odor of 50 ppm of ammonia to be just perceptible to nuisance during the first 30 min of exposure and just perceptible to offensive after 2 h.
Ferguson et al. (1977) conducted a study showing adaptation to concentrations up to 150 ppm of ammonia, with excursions up to 200 ppm, in individuals acclimated to 25-100 ppm for 1 week. More details of this study are described in Section 2.2.1.
4.4.2.
Species Variability
ten Berge et al. (1986) found that mice are usually more sensitive (to irritants) than other mammals. The most direct comparison of mice and rats to inhalation exposure to ammonia can be found in Table 2-7 of this chapter. These data show that the mouse is 2.7 to 4 times more sensitive to inhalation exposure to ammonia than the rat.
4.4.3.
Susceptible Populations
Erskine et al. (1993) showed that the glottis of elderly people (86-90 years old) is less responsive to inhalation exposure to ammonia than younger people (21-30 years old); the two age groups differed by a factor of 3. McLean et al. (1979) showed that nonatopic and atopic subjects, some of whom had a history of asthma, responded similarly in a nasal airway resistance (NAR) test to 100 ppm of ammonia introduced into each nostril under pressure for up to 30 s. The increased NAR was attributed to parasympathetic reflex and not to histamine release. Ammonia is water soluble and efficiently scrubbed in the nasopharyngeal regions; ammonia would not reach the tracheobronchial and pulmonary regions of the respiratory tract until the scrubbing action has been saturated. It is unlikely that concentrations detected only by odor or irritation to the nasal cavity or eyes would reach the tracheobronchial and pulmonary regions and have a differential effect on asthmatic individuals.
4.4.4.
Concentration-Exposure Duration Relationship
Appelman et al. (1982) used multiple linear weighted regression to show the general correlation between concentration, time, and mortality expressed as probit. They derived the following equation:
where a, b, and q are the regression parameters; c is the concentration (mg/m3 or ppm); and t is the time of exposure. The values for regression parameters for the
combined sexes were as follows: a = 4.62, b = 2.30, and q = 47.8. The quotient for b/a is equal to n. Converting the above equation to
shows that the relationship of any concentration and time corresponding to a mortality rate can be expressed as Cn × t = k, where n = 2.02. ten Berge et al. (1986) reported an n value of 2 and confidence intervals of 1.6 and 2.4 for ammonia. ten Berge et al. (1986) also noted that the value of the exponent n should be derived empirically.
5.
DATA ANALYSIS FOR AEGL-1
5.1.
Human Data Relevant to AEGL-1
Human data relevant for deriving the AEGL-1 value are summarized in Section 2.3, Table 2-5. Faint or no detectable irritation was reported for exposure to 30 ppm for 10 min (MacEwen et al. 1970), and moderate irritation was reported for exposure to 50 ppm for 10 min to 2 h (MacEwen et al. 1970; Verberk 1977). Moderate irritation also was reported for exposure to 80 ppm for up to 1 h. No adverse effect on respiratory function has been reported for exposure to ammonia at concentrations of 140 ppm for 2 h or up to 500 ppm for 30 min (Verberk 1977; Silverman et al. 1949).
5.2.
Animal Data Relevant to AEGL-1
Animal studies were available, but none was judged to be adequate for deriving AEGL-1 values in view of the available human data.
5.3.
Derivation of AEGL-1
Humans experience either faint or no irritation after exposure to ammonia at 30 ppm for 10 min (MacEwen et al. 1970); therefore, 30 ppm was used to derive AEGL-1 values. An interspecies uncertainty factor is not applied to these data because the AEGL value is based on human data. An intraspecies uncertainty factor of 1 was selected because ammonia is efficiently scrubbed in the upper respiratory tract, and if irritation occurs, it would be confined to the nasal cavity (and possibly the eyes). Nonatopic and atopic subjects, including asthmatics, responded similarly in a nasal airway resistance test when 100 ppm of ammonia was introduced into each nostril for up to 30 s (McLean et al. 1979); therefore, asthmatic individuals are not expected to respond differently than nonasthmatic individuals. Exercising subjects showed only a clinically nonsig-
nificant decrease in pulmonary function after exposure to higher concentrations of ammonia (Cole et al. 1977); therefore, exercise is not expected to cause an appreciable difference in effects experienced during exposure to AEGL-1 concentrations. The same value is proposed for 5, 30, 60, 240, and 480 min, because any effects that occur are not expected to become more severe with duration of exposure because adaptation occurs during prolonged exposure. AEGL-1 values are summarized in Table 2-9. The AEGL-1 value of 30 ppm for all time points is supported by observations that humans reported similar intensities of response after exposure to 50 ppm for 10 min to 2 h (MacEwen et al. 1970; Verberk 1977).
6.
DATA ANALYSIS FOR AEGL-2
6.1.
Human Data Relevant to AEGL-2
Data detailing disabling, irreversible, or long-term effects of ammonia were discussed in Section 2.2.1. The immediate response of individuals exposed to severe irritating concentrations of ammonia is to escape. Therefore, only those people who are incapacitated and unable to escape or those who are not rescued by others would remain in an atmosphere containing highly irritating concentrations of ammonia. They would be in danger with prolonged continuous exposure. The case studies on irreversible or long-term effects of ammonia did not report exposure concentrations and cannot be used to derive AEGL values. Several studies showing reversible irritation in humans had quantitative exposure data judged suitable for deriving AEGL-2 levels. These studies are summarized in Table 2-10. The subjects in the Verberk (1977) study were exposed to concentrations ranging from 50 to 140 ppm for durations ranging up to 2 h, and this study established exposure concentrations and durations of exposure considered to be offensive and unbearable but reversible. Other studies provide additional data to support to the Verberk study. Silverman et al. (1949) exposed subjects to 500 ppm of ammonia for 30 min by means of a half mask; there was no direct contact of the eyes with the ammonia. Ferguson et al. (1977) reported no adverse effects on respiratory function in human volunteers exposed for 2-6 h/day for 5 days to ammonia levels as high as 100 ppm.
There are difficulties in determining the ammonia concentrations associated with irreversible effects for the longer exposure times (4 or 8 h). Reversible
TABLE 2-9 AEGL-1 Values for Ammonia
|
5 min |
10 min |
30 min |
1 h |
4 h |
8 h |
|
30 ppm (21 mg/m3) |
30 ppm (21 mg/m3) |
30 ppm (21 mg/m3) |
30 ppm (21 mg/m3) |
30 ppm (21 mg/m3) |
30 ppm (21 mg/m3) |
TABLE 2-10 Nonlethal Effects of Ammonia on Humans and Experimental Animals
|
Species |
Concentration (ppm) |
Exposure Time (min) |
Effect |
Reference |
|
Human |
50 |
10 |
Moderate irritation (NOS). |
MacEwen et al. 1970 |
|
Human |
110 |
120 |
Irritation: eyes, nose, throat, chest. |
Verberk 1977 |
|
Human |
140 |
30 |
Irritation: eyes, nose, throat, chest; urge to cough. |
Verberk 1977 |
|
Human |
140 |
120 |
Nuisance irritation: eyes, throat; urge to cough. |
Verberk 1977 |
|
Human |
143 |
5 |
Irritation: eyes, mouth, nose, throat, chest. |
Ind. Bio.-Test Lab. 1973 |
|
Human |
571 |
One breath |
Threshold for glottis closure in young males. |
Erskine et al. 1993 |
|
Human |
500 |
30 |
Only 2 of 7 subjects tolerated ammonia via nose breathing; irritation effects: nose and throat; lacrimation, hyperventilation, decreased respiratory function. |
Silverman et al. 1949 |
|
Mouse |
303 |
30 |
RD50 (50% depression in respiratory rate) |
Barrow et al. 1978 |
|
aBased on C2 × t = k. Abbreviations: NA, not applicable; NOS, not otherwise specified. |
||||
effects may become irreversible and irreversible effects may become lethal due to delays in medical treatment as well as to continued exposure. Furthermore, exposure concentrations were not measured for the cases in which severe but reversible damage occurred in the respiratory tract. Therefore, AEGL-2 levels for ammonia can be determined from studies reporting “unbearable” upper respiratory tract irritation, which could potentially impair the ability to escape, rather than the threshold for irreversible or long-term effects. The unbearable concentrations are much lower than those that would be associated with the threshold for irreversible damage to the respiratory tract.
6.2.
Animal Data Relevant to AEGL-2
The RD50 (30-min exposure) of 303 ppm for the mouse (Barrow et al. 1978), which is predicted to cause intense sensory irritation and rapid incapacitation in humans, produced histopathological lesions in the nasal cavity but not
in the tracheobronchial or pulmonary regions in mice exposed repeatedly for 5 days (Buckley et al. 1984). Tepper et al. (1985) showed that mice exposed to 300 ppm ceased motor activity (wheel running) during the entire 6-h exposure period. These mice had prior ozone exposure that may have affected the outcome of the study.
6.3.
Derivation of AEGL-2
The AEGL-2 values were based on “offensive” irritation to the eyes and respiratory tract experienced by nonexpert human subjects (unfamiliar with the effects of ammonia or with laboratory studies) exposed to 110 ppm of ammonia for 2 h (Verberk 1977). The responses of the nonexpert subjects ranged from “no sensation” to “offensive” for eye irritation, cough, or discomfort and from “just perceptible” or “distinctly perceptible” to “offensive” for throat irritation. No residual or irreversible effects were reported after termination of exposure, and pulmonary function was not affected by exposure. At the next higher concentration of 140 ppm, some subjects reported the effects to be unbearable and left the chamber between 30 min and 1 h; none remained for the full 2 h. Some irritation to the eyes, nose, throat, and chest along with a disagreeable odor are expected at the AEGL-2 level. An interspecies uncertainty factor is not applied to these data because the AEGL values are based on human data. An intraspecies uncertainty factor of 1 was selected because ammonia is a contact irritant, it is efficiently scrubbed in the upper respiratory tract, and any perceived irritation is not expected to be greater than that of the most sensitive nonexpert subject. The range of responses for this group is considered comparable to the range of responses that would be encountered in the general population, including asthmatics. Investigations have shown a link between nasal symptoms or allergic rhinitis and asthma, with rhinitis preceding the development of asthma (Corren 1997), and studies have shown that atopic subjects, including asthmatics, and nonatopic subjects respond similarly to a brief nasal exposure to ammonia (McLean et al. 1979). Exposure to exercising subjects showed only clinically nonsignificant changes in pulmonary function during exposure to ammonia at concentrations up to 336 ppm (Cole et al. 1977). In addition, a child experienced less severe effects than an adult exposed to very high concentrations of ammonia (Kass et al. 1972).
Time scaling across the pertinent timeframes was based on the ten Berge et al. (1986) equation (Cn × t = k, where C = concentration, n = 2, and k is a constant). The value of n was derived from mouse and rat lethality data and was reported by Appelman et al. (1982) and ten Berge et al. (1986). The value of 110 ppm was adopted as the 4- and 8-h values, because the maximum severity rating for irritation in the Verberk (1977) study changed very little between 30 min and 2 h and is not expected to change for exposures up to 8 h. The 30-min value was also adopted as the 10-min AEGL-2 value because time scaling would yield a
10-min value (380 ppm) that might impair escape. The AEGL-2 values are summarized in Table 2-11.
The AEGL values are supported by other studies showing that exposures up to 100 ppm were tolerated by human subjects for 2-6 h without causing serious effects (Ferguson et al. 1977). The data of Cole et al. (1977) and Silverman et al. (1949) showed no serious irreversible effects at 336 or 500 ppm, respectively.
7.
DATA ANALYSIS FOR AEGL-3
7.1.
Human Data Relevant to AEGL-3
Although numerous case studies describing lethal and potentially life-threatening exposures to ammonia resulting from various accidental releases were found in the literature, the lack of definitive information on actual exposure concentrations limits the usefulness of these studies for establishing AEGL-3 values. Substantial uncertainties are associated with the values derived from the gas dispersion models (WHAZAN and HGSYSTEM). In both cases, estimates for atmospheric ammonia concentrations were used as surrogates for exposure concentration. For the South Africa ammonia accident, the HGSYSTEM dispersion model did not address exposure estimates for the survivors sheltered inside buildings or the people located upwind from the release. This fact alone renders any analysis derived from the WHAZAN or RAM TRAC, which includes individuals sheltered inside buildings, inadequate for estimating human survival levels. The HGSYSTEM model of the South Africa accident may be unable to address releases from multiple sources, unable to model a delayed transport scenario or puff expansion in calm wind followed by wind transport, and is limited by the complex meteorological conditions (Mazzola 1996). In a more detailed analysis of the HGSYSTEM model and dose reconstruction models in general, Mazzola (1997) noted that (1) the absence of real-time meteorological data during and subsequent to the release would significantly limit the confidence in using HGSYSTEM modeling results; (2) the HGSYSTEM may be unable to accurately simulate the complex thermodynamics of anhydrous ammonia releases; (3) the HGSYSTEM is unable to address indoor concentrations; and (4) the Benign Bubble hypothesis cannot be proven in the absence of three-dimensional wind field data. Mazzola also noted other sources of uncertainties in the HGSYSTEM model of the Potchefstroom, South Africa, ammonia accident as reported by Mudan and Mitchell (1996); as the levels of uncertainty
TABLE 2-11 AEGL-2 Values for Ammonia
|
10 min |
30 min |
1 h |
4 h |
8 h |
|
220 ppm (154 mg/m3) |
220 ppm (154 mg/m3) |
160 ppm (112 mg/m3) |
110 ppm (77 mg/m3) |
110 ppm (77 mg/m3) |
accumulate and become very large, confidence in the final results diminishes. Therefore, atmospheric ammonia concentrations generated by the HGSYSTEM model cannot serve as a surrogate for exposure and should not be used to derive AEGL values.
Because of the inability to estimate the response variable, the inability to estimate concentrations to individuals sheltered inside buildings, and the uncertainties associated with accident dose reconstruction as surrogates for exposure, animal data are preferred for deriving AEGL-3 values. Although there is an inherent weakness in extrapolating from experimental animal concentrations to human exposure, animal studies are strengthened by having measured exposure concentrations and known response data. Therefore, an approach using experimental animal data, where exposure estimates are more reliable, is recommended for deriving AEGL-3 values.
7.2.
Animal Data Relevant to AEGL-3
Data from the rat studies reported by Appelman et al. (1982) and MacEwen and Vernot (1972) and the mouse studies reported by Silver and McGrath (1948), MacEwen and Vernot (1972), and Kapeghian et al. (1982) were considered relevant to deriving AEGL-3 values. The rat study by Appelman et al. and the mouse studies by Kapeghian et al. and MacEwen and Vernot were well conducted. However, the results of the Appelman et al. study were based on four different exposure durations, whereas only one exposure duration was used in the mouse studies by Kapeghian et al. and MacEwen and Vernot. ten Berge et al. (1986) noted that mice are more sensitive to respiratory irritants than other mammalian species. The two mouse studies, however, produced similar LC50 values (4,230 and 4,837 ppm), which increases the confidence in using the mouse data to derive the AEGL-3 values. In addition, probit analysis of the rat data reported by MacEwen and Vernot (1972) produced an LC50 value of 7,338 ppm for a 60-min exposure; this value is less than one-half the LC50 of 16,600 ppm derived by Appelman et al. The discrepancy in the two studies increases the uncertainty of using the rat data to derive AEGL-3 values. A study in the cat provided the lowest lethal concentration of 1,000 ppm (Dodd and Gross 1980). Lower respiratory tract lesions produced in cats exposed to ammonia are similar to those described for humans. However, the cats were exposed using a cuffed endotracheal tube, which bypassed the nasopharyngeal region where a significant amount of scrubbing occurs. This method of exposure could produce more severe tracheobronchial lesions than would occur from nose breathing. It should be noted that the cat study used only one ammonia concentration and one exposure duration; it was not designed to evaluate exposure-related effects. Because of inconsistencies in the results of the rat studies and the exposure method used for cat the study, the mouse studies are considered the most suitable for deriving AEGL-3 values.
7.3.
Derivation of AEGL-3
LC01 values derived from the mouse and rat studies are presented in Table 2-12. AEGL-3 values are derived using the data for mice reported by Kapeghian et al. (1982) and MacEwen and Vernot (1972). The 60-min LC01 derived by the probit analysis of Kapeghian et al. is 3,317 ± 195 ppm (± standard error), and the 60-min LC01 derived by probit analysis of the MacEwen and Vernot data is 3,374 ± 376 ppm. The LC01 values from the two mouse studies are similar and both have small standard errors. These values compare closely with the 2,932 ppm 60-min LC01 derived from use of regression coefficients from the combined mouse datasets of Kapeghian et al. (1982) and Silver and McGrath (1948) as presented by ten Berge et al. (1986) (see Table 2-12). For comparison, LC01 values using the rat data reported by Appelman et al. (1982) and MacEwen and Vernot (1972) also are presented. The Benchmark Dose approach was applied to the Kapeghian et al. and MacEwen and Vernot mouse data; the resulting BMDL05 values derived from the probit model are 3,278 and 3,219 ppm, respectively.
The mouse is unusually sensitive to exposure to respiratory irritants, including ammonia (ten Berge et al. 1986); therefore, an interspecies uncertainty factor of 1 was applied to the LC01 for the mouse. An uncertainty factor of 3 was applied to account for intraspecies variability because concentrations of ammonia that are life threatening cause severe tracheobronchial and pulmonary damage and these effects are not expected to be more severe in asthmatics than in nonasthmatics (McLean et al. 1979), more severe in children than adults (Kass et al. 1972), or more severe in exercising than in nonexercising individuals (Cole et al. 1977; see rationale for AEGL-2), but tracheobranchial and pulmonary effects may occur at a lower concentration in the elderly. Investigations showed that reflex glottis closure (protective mechanism) is 3-fold less sensitive in the elderly than in young subjects (Erskine et al. 1993); this mechanism may be
TABLE 2-12 LC01 Estimates for Ammonia Derived from Animal Data
|
Exposure Time (min) |
Concentration (ppm) |
|||||
|
Mousea |
Mouseb |
Mousec |
Moused |
Rata |
Ratb |
|
|
5 |
9,800 |
11,688 |
11,487 |
6,031 |
34,356 |
17,899 |
|
30 |
4,104 |
4,772 |
4,690 |
2,462 |
14,134 |
7,307 |
|
60 |
2,932 |
3,374 |
3,317 |
1,741 |
10,024 |
5,167 |
|
240 |
1,494 |
1,687 |
1,658 |
871 |
5,042 |
2,584 |
|
480 |
1,067 |
1,193 |
1,172 |
616 |
3,575 |
1,827 |
|
aConcentrations derived using Appelman et al. (1982) regression coefficients b0 = 47.8, b1 = 4.64, and b2 = 2.30 for the rat and ten Berge et al. (1986) regression coefficients b0 = 54.5, b1 = 5.95, and b2 = 2.89 for the mouse. bDerived from data reported by MacEwen and Vernot 1972; n = 2. cDerived from data reported by Kapeghian et al. 1982; n = 2. dDerived from data reported by Silver and McGrath 1948; n = 2. |
||||||
applicable only when concentrations of ammonia exceed 570 ppm. A larger interspecies or intraspecies uncertainty factor would lower the 30-min AEGL-3 value to approximately 500 ppm, which was tolerated by humans without lethal or long-term consequences (Silverman et al. 1949). Therefore, applying a total uncertainty factor of 3 to the LC01 values of 3,317 or 3,374 ppm results in an AEGL-3 value of 1,100 ppm for the 1-h duration. ten Berge’s equation was used to extrapolate to the relevant exposure durations. The value of n was calculated from the regression coefficients (b1/b2) for mouse data reported by ten Berge et al. (1986). The AEGL-3 values for 10, 30, 60, 240, and 480 min are presented in Table 2-13.
No verified lethal concentrations for ammonia in humans were found in the available literature. However, Silverman et al. (1949) reported that 1,000 ppm induced an immediate urge to cough. Legters (1980) noted that coughing may indicate that the adsorptive (scrubbing) capacity of the upper respiratory tract has been exceeded and that ammonia is penetrating the lower respiratory passages. Data presented in Section 2.1 show that death in humans exposed to ammonia is associated with damage to the lower respiratory tract, and data presented in Section 2.2.1 showed effects caused by ammonia on the lower respiratory tract that would be lethal without prompt medical attention. Therefore, concentrations of ammonia that exceed the scrubbing capacity of the upper respiratory tract and cause coughing, which indicates lower respiratory effects, have potentially serious effects. Although no experimental studies were available for exposures to ammonia for durations longer than 1 h, there is a need to derive AEGL-3 values for 4- and 8-h exposures. Kass et al. (1972) showed that the ammonia cloud formed after an accident does not always dissipate rapidly. In the accident with the railroad car, a heavy fog kept the ammonia cloud close to the ground for a prolonged period of time.
The AEGL-3 value for 8 h is supported by studies in rats, rabbits, guinea pigs, dogs, and monkeys showing that daily 8-h exposures to 1,101 ppm for 6 weeks caused no deaths (Coon et al. 1970). The only effects observed were nonspecific inflammation (rats and guinea pigs), lacrimation (dogs and rabbits), and dyspnea (dogs and rabbits).
8.
SUMMARY OF AEGLs
8.1.
AEGL Values and Toxicity End Points
The AEGL values are summarized in Table 2-14. Ammonia is irritating upon immediate contact with mucous surfaces of the eyes, mouth, and respiratory tract. The following factors were taken into account in proposing the AEGL values. Inhaling low concentrations of ammonia causes mild irritation to the eyes, nose, and throat, which is reversible upon termination of exposure. Individuals will attempt to escape immediately from atmospheres containing ammo-
TABLE 2-13 AEGL-3 Values for Ammonia
|
10 min |
30 min |
1 h |
4 h |
8 h |
|
2,700 ppm (1,888 mgm3) |
1,600 ppm (1,119 mgm3) |
1,100 ppm (769 mgm3) |
550 ppm (385 mgm3) |
390 ppm (273 mgm3) |
TABLE 2-14 AEGL Values for Ammonia
|
Classification |
10 min |
30 min |
1 h |
4 h |
8 h |
End Point (Primary References) |
|
AEGL-1 (nondisabling) |
220 ppm (154 mgm3) |
30 ppm (21 mgm3) |
30 ppm (21 mgm3) |
30 ppm (21 mgm3) |
30 ppm (21 mgm3) |
Mild irritation (MacEwen et al. 1970) |
|
AEGL-2 (disabling) |
220 ppm (154 mgm3) |
220 ppm (154 mgm3) |
160 ppm (112 mgm3) |
110 ppm (77 mgm3) |
110 ppm (77 mgm3) |
Irritation: eyes and respiratory tract, urge to cough (Verberk 1977) |
|
AEGL-3 (lethal) |
2,700 ppm (1,888 mgm3) |
1,600 ppm (1,119 mgm3) |
1,100 ppm (769 mgm3) |
550 ppm (385 mgm3) |
390 ppm (273 mgm3) |
Threshold for lethality (LC01) (Kapeghian et al. 1982; MacEwen and Vernot 1972) |
nia at concentrations considered highly irritating or intolerable. Reflex glottis closure and nasopharyngeal scrubbing may protect the lower respiratory tract from potential injury during brief exposures. When the scrubbing capacity of the nasopharyngeal region is exceeded, the potential for damage to the lower regions of the respiratory tract increases. Most deaths occur when damage causes pulmonary edema or airway obstruction. However, recovery from airway obstruction is usually assured with medical treatment, whereas pulmonary edema may lead to death even with medical treatment.
8.2.
Comparison of AEGLs with Other Standards and Criteria
Table 2-15 summarizes standards and guidelines established by various agencies and organizations. The AEGL values are similar to the values recommended by other organizations and agencies. The 1-h ERPG-3 (750 ppm) is slightly less than the proposed AEGL-3 value of 1,100 ppm, the ERPG-2 value (150 ppm) is slightly less than the AEGL-2 value of 110 ppm, and ERPG-1 value (25 ppm) is the same as the AEGL-1 value. NIOSH’s IDLH is slightly
TABLE 2-15 Extant Standards and Guidelines for Ammonia
|
Guideline |
10 min |
30 min |
1 h |
4 h |
8 h |
|
AEGL-1 |
30 ppm |
30 ppm |
30 ppm |
30 ppm |
30 ppm |
|
AEGL-2 |
220 ppm |
220 ppm |
160 ppm |
110 ppm |
110 ppm |
|
AEGL-3 |
2,700 ppm |
1,600 ppm |
1,100 ppm |
550 ppm |
390 ppm |
|
ERPG-1 (AIHA)a |
|
|
25 ppm |
|
|
|
ERPG-2 (AIHA) |
|
|
150 ppm |
|
|
|
ERPG-3 (AIHA) |
|
|
750 ppm |
|
|
|
EEGL (NRC)b |
|
|
100 ppm |
|
100 ppm (24 h) |
|
PEL-TWA (OSHA)c |
|
|
|
|
50 ppm |
|
IDLH (NIOSH)d |
|
300 ppm |
|
|
|
|
REL-TWA (NIOSH)e |
|
|
|
|
25 ppm |
|
REL-STEL (NIOSH)f |
35 ppm (15 min) |
|
|
|
|
|
TLV-TWA (ACGIH)g |
|
|
|
|
25 |
|
TLV-STEL (ACGIH)h |
35 ppm (15 min) |
|
|
|
|
|
MAK (Germany)i |
|
|
|
|
20 |
|
MAK Peak Limit (Germany)j |
|
|
|
|
|
|
OELV (Sweden)l (Dutch) |
50 ppm (15 min) |
|
|
|
25 ppm |
|
SMACm |
|
|
20 ppm |
|
14 ppm (24 h) |
|
aERPG (emergency response planning guideline, American Industrial Hygiene Association) (AIHA 2000). The ERPG-1 is the maximum airborne concentration below which it is believed nearly all individuals could be exposed for up to 1 h without experiencing other than mild, transient adverse health effects or without perceiving a clearly defined objectionable odor. The ERPG-1 for ammonia is based on a concentration associated with a mild odor perception or mild irritation. The ERPG-2 is the maximum airborne concentration below which it is believed nearly all individuals could be exposed for up to 1 h without experiencing or developing irreversible or other serious health effects or symptoms that could impair an individual’s ability to take protective action. At the ERPG-2 level, ammonia will likely have a strong odor and cause some eye and upper respiratory irritation in susceptible populations, but serious effects are unlikely. The ERPG-3 is the maximum airborne concentration below which it is believed nearly all individuals could be exposed for up to 1 h without experiencing or developing life-threatening health effects. The ERPG-3 for ammonia is based on the median lethal concentrations of 7,340-16,600 ppm for the rat and 4,230-4,840 ppm for the mouse. This concentration may cause respiratory distress and severe eye and nasal irritation. bEEGL (Emergency exposure guidance level, National Research Council) (NRC 1987). The EEGL is the concentration of contaminants that can cause discomfort or other evidence of irritation or intoxication in or around the workplace but avoids death, other se |
|||||
higher than the value for the AEGL-2 30-min exposure. Mahlum and Sasser (1991) determined maximum exposure levels for operators in nuclear reactor control rooms. The recommended 2-min exposure limit was 300 ppm, which would allow a person to perform their task, don protective clothing, and suffer no long-lasting effects.
8.3.
Data Adequacy and Research Needs
A large body of data was available for deriving AEGL values for ammonia. The studies on lethal or irreversible effects in humans did not have quantitative exposure estimates. However, human studies on upper respiratory tract irritation with quantitative exposure were available. In the human studies available, subjects were exposed to ammonia at concentrations that ranged from odor detection levels to concentrations causing “unbearable” irritation to the respiratory tract and eyes. Human studies using concentrations of ammonia higher than those reported in this document have the potential for causing more severe irritation and are not necessary for further documenting of exposure-response relationships in humans. The available human data were considered adequate for deriving AEGL-1 and -2 values. Lethality data were available for two animal species, and these data were considered adequate for deriving AEGL-3 values. The only data deficiency of note was the lack of lethal data for rodents for exposure periods longer than 1 h.
9.
REFERENCES
ACGIH (American Conference of Governmental Hygienists). 2001. Threshold Limit Values for Chemical Substances and Physical Agents and Biological Exposure Indices. American Conference of Governmental Hygienists, Cincinnati, OH.
AIHA (American Industrial Hygiene Association). 2000. Emergency Response Planning Guidelines for Ammonia. AIHA Emergency Response Planning Guideline Committee, Fairfax, VA.
Appelman, L.M., W.F. ten Berge, and P.G. Reuzel. 1982. Acute inhalation toxicity study of ammonia in rats with variable exposure periods. Am. Ind. Hyg. Assoc. J. 43(9):662-665.
Barrow, C.S., Y. Alarie, and M.F. Stock. 1978. Sensory irritation and incapacitation evoked by thermal decomposition products of polymers and comparisons with known sensory irritants. Arch. Environ. Health 33(2):79-88.
Boyd, E.M., M. MacLachlan, and W.F. Perry. 1944. Experimental ammonia gas poisoning in rabbits and cats. J. Ind. Hyg. Toxicol. 26(1):29-34.
Buckley, L.A., X.Z. Jiang, R.A. James, K.T. Morgan, and C.S. Barrow. 1984. Respiratory tract lesions induced by sensory irritants at the RD50 concentration. Toxicol. Appl. Pharmacol. 74(3):417-429.
Budavari, S., M.J. O’Neil, A. Smith, and P.E. Heckelman, eds. 1989. Ammonia. P. 81 in The Merck Index: An Encyclopedia of Chemicals, Drugs, and Biologicals, 11th Ed. Rahway, NJ: Merck.
Caplin, M. 1941. Ammonia-gas poisoning- Forty-seven cases in a London shelter. Lancet 241(July 26):95-96.
Carlile, F.S. 1984. Ammonia in poultry houses: A literature review. World Poult. Sci. 40(2):99-113.
Cole, T.J., J.E. Cotes, G.R. Johnson, H.D. Martin, J.W. Reed, and M.J. Saunders. 1977. Ventilation, cardiac frequency and pattern of breathing during exercise in men exposed to o-chlorobenzylidene malonitrile (CS) and ammonia gas in low concentrations. Q. J. Exp. Physiol. Cogn. Med. Sci. 62(4):341-351.
Coon, R.A., R.A. Jones, L.F. Jenkins, Jr., and J. Siegel. 1970. Animal inhalation studies on ammonia, ethylene glycol, formaldehyde, dimethylamine, and ethanol. Toxicol. Appl. Pharmacol. 16(3):646-655.
Corren, J. 1997. Allergic rhinitis and asthma: How important is the link? J. Allergy Clin. Immunol. 99(2):S781-S786.
Dalhamn, T. 1956. Mucous flow and ciliary activity in the trachea of healthy rats and rats exposed to respiratory irritant gases (S02, H3N, HCHO). A functional and morphologic (light microscopic and electron microscopic) study, with special reference to technique. Acta Physiol. Scand. 36 (Suppl. 123):1-161.
DFG (Deutsche Forschungsgemeinschaft [German Research Association]). 2000. List of MAK and BAT Values 2000. Maximum Concentrations and Biological Tolerance Values at the Workplace. Commission for the Investigation of Health Hazards of Chemical Compounds in the Work Area Report No. 36. Weinheim, Federal Republic of Germany: Wiley-VCH.
Dodd, K.T., and D.R. Gross. 1980. Ammonia inhalation toxicity in cats: A study of acute and chronic respiratory dysfunction. Arch. Environ. Health 35(1):6-14.
Donham, K., S.J. Reynolds, P. Whitten, J.A. Merchant, L. Burmeister, and W.J. Popendorf. 1995. Respiratory dysfunction in swine production facility workers: Dose-response relationships of environmental exposures and pulmonary function. Am. J. Ind. Med. 27(3):405-418.
Egle, J.L., Jr. 1973. Retention of inhaled acetone and ammonia in the dog. Am. Ind. Hyg. Assoc. J. 34(12):533-539.
EPA (U.S. Environmental Protection Agency). 1994. Methods for Derivation of Inhalation Reference Concentrations and Application of Inhalation Dosimetry. EPA/600/8-90/066F. Environmental Criteria and Assessment Office, Office of Health and Environmental Assessment, Office of Research and Development, U.S. Environmental Protection Agency, Research Triangle Park, NC.
Erskine, R.J., P.J. Murphy, J.A. Langston, and G. Smith. 1993. Effect of age on the sensitivity of upper airways reflexes. Br. J. Anaesth. 70(5):574-575.
Ferguson, W.S., W.C. Koch, L.B. Webster, and J.R. Gould. 1977. Human physiological response and adaption to ammonia. J. Occup. Med. 19(5):319-326.
Flury, K.E., D.E. Dines, J.R. Rodarte, and R. Rodgers. 1983. Airway obstruction due to inhalation of ammonia. Mayo Clin. Proc. 58(6):389-393.
Hatton, D.V., C.S. Leach, A.L. Beaudet, R.O. Dillman, and N. Di Ferrante. 1979. Collagen breakdown and ammonia inhalation. Arch. Environ. Health 34(2):83-87.
Henderson, Y., and H.W. Haggard. 1943. Special characteristics of various irritant gases: Subgroup A 1: Ammonia gas. Pp. 125-126 in Noxious Gases. New York: Reinhold.
Hilado, C.J., C.J. Casey, and A. Furst. 1977. Effect of ammonia on Swiss albino mice. J. Combust. Toxicol. 4:385-388.
Hilado, C.J., H.G. Cumming, A.M. Machado, C.J. Casey, and A. Furst. 1978. Effect of individual gaseous toxicants on mice. Proc. West. Pharmacol. Soc. 21:159-160.
Hoeffler, H.B., H.I. Schweppe, and S.D. Greenberg. 1982. Bronchiectasis following pulmonary ammonia burn. Arch. Pathol. Lab. Med. 106(13):686-687.
Holness, D.L., J.T. Purdham, and J.R. Nethercott. 1989. Acute and chronic respiratory effects of occupational exposure to ammonia. Am. Ind. Hyg. Assoc. J. 50(12):646-650.
Industrial Bio-Test Laboratories, Inc. 1973. Irritation Threshold Evaluation Study with Ammonia. Publication IBT 663-03161. Report to International Institute of Ammonia Refrigeration by Industrial Bio-Test Laboratories, Inc. March 23, 1973 (as cited in NIOSH 1974).
Kapeghian, J.C., H.H. Mincer, and A.B. Jones, A.J. Verlangieri, and I.W. Waters. 1982. Acute inhalation toxicity of ammonia in mice. Bull. Environ. Contam. Toxicol. 29(3):371-378.
Kapeghian, J.C., A.B. Jones, and I.W. Waters. 1985. Effects of ammonia on selected hepatic microsomal enzyme activity in mice. Bull. Environ. Contam. Toxicol. 35(1):15-22.
Kass, I., N. Zamel, C.A. Dobry, and Holzer. 1972. Bronchiectasis following ammonia burns of the respiratory tract: A review of two cases. Chest 62(3):282-285.
Landahl, H.D., and R.G. Herrmann. 1950. Retention of vapors and gases in the human nose and lung. Arch. Ind. Hyg. Occup. Med. 1(1):36-45.
Leduc, D., P. Gris, P. Lheureux, P.A. Gevenois, P. de Vuyst, and J.C. Yernault. 1992. Acute and long term respiratory damage following inhalation of ammonia. Thorax 47(9):755-757.
Legters, L. 1980. Biological Effects of Short High-Level Exposure to Gases: Ammonia. Phase Report, May 1979-May 1980. DAMD 17-79-C-9086. AD A094501. Prepared for U.S. Army Medical Research and Development Command, Fort Detrick, Frederick, MD, by Environ Control, Inc, Rockville, MD.
Levy, D.M., M.B. Divertie, T.J. Litzow, and J.W. Henderson. 1964. Ammonia burns of the face and respiratory tract. J. Am. Med. Assoc. 190:873-876.
Lewis, R.J., Sr., ed. 1993. Hawley’s Condensed Chemical Dictionary, 12th Ed. New York: Van Nostrand Reinhold. .
Lonsdale, H. 1975. Ammonia Tank Failure-South Africa. American Institute of Chemical Engineers, Ammonia Plant Safety 17:126-131 (as cited by RAM TRAC 1996).
MacEwen, J.D., and E.H. Vernot. 1972. Toxic Hazards Research Unit Annual Technical Report: 1972. AMRL-TR-72-62. NTIS AD-755 358. Aerospace Medical Research Laboratory, Wright-Patterson Air Force Base, OH.
MacEwen, J.D., J. Theodore, and E.H. Vernot. 1970. Human exposure to EEL concentrations of monomethylhydrazine. Pp. 355-363 in Proceedings of the 1st Annual Conference Environmental Toxicology, September 9-11, 1970, Wright-Patterson Air Force Base, OH. AMRL-TR-70-102, Paper No 23. Aerospace Medical Research Laboratory, Wright-Patterson Air Force Base, OH.
Mahlum, D.D., and L.B. Sasser. 1991. Evaluation of Exposure Limits to Toxic Gases for Nuclear Reactor Control Room Operators. NUREG/CR-5669. PNL -7522. Prepared by Pacific Northwest Laboratory, Richland, WA, for the Nuclear Regulatory Commission, Office of Nuclear Regulatory Research, Division of Safety Issue Resolution, Washington, DC.
Manninen, A., S. Anttila, and H. Savolainen. 1988. Rat metabolic adaptation to ammonia inhalation. Proc. Soc. Exp. Biol. Med. 187(3):278-281.
Markham, R.S. 1986. A Review of Damage from Ammonia Spills. Paper presented at the 1986 Ammonia Symposium, Safety in Ammonia Plants and Related Facilities,
American Institute of Chemical Engineers Boston, MA, August 1986 (as cited in Pedersen and Selig 1989).
Mayan, M.H., and C.P. Merilan. 1972. Effects of ammonia inhalation on respiration rate of rabbits. J. Anim. Sci. 34(3):448-452.
Mayan, M.H., and C.P. Merilan. 1976. Effects of ammonia inhalation on young cattle. N.Z. Vet. J. 24(10):221-224.
Mazzola, C. 1996. Inherent Uncertainties in Dose Reconstructions Using Dispersion Models. Presented to the National Advisory Committee for Acute Exposure Guideline Levels for Hazardous Substances, December 16, 1996, Washington, D.C.
Mazzola, C. 1997. Potchefstroom Dose Reconstruction: Inherent Uncertainties that Significantly Limit Effective Application to Human Health Standards Process. Prepared for the National Advisory Committee for Acute Exposure Guideline Levels for Hazardous Substances (NAC/AEGL), by Stone & Webster Engineering Corporation. May, 1997.
McLean, J.A., K.P. Mathews, W.R. Solomon, P.R. Brayton, and N.K. Baynel. 1979. Effect of ammonia on nasal resistance in atopic and nonatopic subjects. Ann. Otol. Rhinol. Laryngol. 88(2 Pt.1):228-234.
Michaels, R.A. 1998. Emergency planning: Critical evaluation of proposed AEGLs for ammonia. Process Saf. Prog. 17(2): 134-137.
Montague, T.J., and A.R. Macneil. 1980. Mass ammonia inhalation. Chest 77(4):496-498.
Mudan, K., and K. Mitchell. 1996. Report on the Potchefstroom, South Africa Ammonia Incident. Four Elements, Inc, Columbus, OH. 14 pp.
Mulder, J.S., and H.O. Van der Zalm. 1967. A fatal case of ammonia poisoning [in Dutch]. Tijdsch. Soc. Geneeskd. 45:458-460 (as cited in NIOSH 1974).
NIOSH (National Institute for Occupational Safety and Health). 1974. Criteria for a Recommended Standard Occupational Exposure to Ammonia. HEW74-136. National Institute for Occupational Safety and Health, Public Health Service, U.S. Department of Health, Education and Welfare, Cincinnati, OH.
NIOSH (National Institute for Occupational Safety and Health). 1997. NIOSH Pocket Guide to Chemical Hazards. DHHS (NIOSH) 97-140. National Institute for Occupational Safety and Health, Public Health Service, U.S. Department of Health, Education and Welfare, Cincinnati, OH.
NRC (National Research Council). 1987. Emergency and Continuous Exposure Guidance Levels for Selected Airborne Contaminants, Vol. 7. Ammonia, Hydrogen Chloride, Lithium Bromide, and Toluene. Washington, DC: National Academy Press.
NRC (National Research Council). 1993. Guidance for Developing Community Emergency Exposure Levels for Hazardous Substances. Washington, DC: National Academy Press.
NRC (National Research Council). 2000. Spacecraft Maximum Allowable Concentrations for Selected Airborne Contaminants, Vol. 4. Washington, DC: National Academy Press.
NRC (National Research Council). 2001. Standing Operating Procedures for Developing Acute Exposure Guideline for Airborne Chemicals. Washington, DC: National Academy Press.
NTSB (National Transportation Safety Board). 1979. Survival in Hazardous Materials Transportation Accidents. NTSC-HZM-79-4. National Transportation Safety Board (as cited in Pedersen and Selig 1989).
O’Kane, G.J. 1983. Inhalation of ammonia vapor: A report on the management of eight patients during the acute stages. Anesthesia 38(12):1208-1213.
O’Neil, M.J., A. Smith, P.E. Heckelman, eds. 2001. Ammonia. Pp. 87 in The Merck Index: A Encyclopedia of Chemicals, Drugs, and Biologicals, 13th Ed. Whitehouse Station, NJ: Merck & Co.
Pedersen, F., and R.S. Selig. 1989. Predicting the consequences of short-term exposure to high concentrations of gaseous ammonia. J. Hazard. Mater. 21(2):143-159.
Pierce, J.O. 1994. Ammonia. Pp. 756-782 in Patty’s Industrial Hygiene and Toxicology, 4th Ed., Vol. II, Pt. A Toxicology, G.D. Clayton, and F.E. Clayton, eds. New York: John Wiley & Sons.
Pinson, D.M., T.R. Schoeb, J.R. Lindsey, and J.K. Davis. 1986. Evaluation of scoring and computerized morphometry of lesions of early Mycoplasma pulmonis infection and ammonia exposure in F344/N rats. Vet. Pathol. 23(5):550-555.
RAM TRAC. 1996. Acute Inhalation Risk Potentially Posed by Anhydrous Ammonia. RAM TRAC Corporation, Schenectady, NY. 99pp.
Reynolds, S., K.J. Donham, P. Whitten, J.A. Merchant, L.F. Burmeister, and W.J. Popendorf. 1996. Longitudinal evaluation of dose-response relationships for environmental exposures and pulmonary function in swine production workers. Am. J. Ind. Med. 29(1):33-40.
Schaerdel, A.D., W.J. White, C.M. Lang, B.H. Dvorchik, and K. Bohner. 1983. Localized and systemic effects of environmental ammonia in rats. Lab. Anim. Sci. 33(1):40-45.
Shimkin, M.B., A.A. de Lorimer, J.R. Mitchell, and T.P. Burroughs. 1954. Appearance of carcinoma following single exposure to a refrigeration ammonia-oil mixture. Arch. Ind. Hyg. Occup. Med. 9(3):186-193.
Silver, S.D., and F.P. McGrath. 1948. A comparison of acute toxicities of ethylene imine and ammonia to mice. J. Ind. Hyg. Toxicol. 30(1):7-9.
Silverman, L., J.L. Whittenberger, and J. Muller. 1949. Physiological response of man to ammonia in low concentrations. J. Ind. Hyg. Toxicol. 31(2):74-78.
Sobonya, R. 1977. Fatal anhydrous ammonia inhalation. Hum. Pathol. 8(3):293-299.
Sundblad, B.M., B.M. Larsson, F. Acevedo, L. Ernstgard, G. Johanson, K. Larsson, and L. Palmberg. 2004. Acute respiratory effects of exposure to ammonia on healthy persons. Scand. J. Work Environ. Health 30(4):313-321.
Swedish National Board of Occupational Safety and Health. 1996. Ordinance of the Swedish National Board of Occupational Safety and Health Containing Provisions on Occupational Exposure Limit Values. Adopted August 28th, 1996.
Swotinsky, R.B., and K.H. Chase. 1990. Health effects of exposure to ammonia: Scant information. Am. J. Ind. Med. 17(4):515-521.
ten Berge, W.F., A. Zwart, and L.M. Appelman. 1986. Concentration-time mortality response relationship of irritant and systemically acting vapours and gases. J. Hazard. Mater. 13(3):301-309.
Tepper, J.S., B. Weiss, and R.W. Wood. 1985. Alterations in behavior produced by inhaled ozone or ammonia. Fundam. Appl. Toxicol. 5(6 Pt. 1):1110-1118.
Verberk, M.M. 1977. Effects of ammonia on volunteers. Int. Arch. Occup. Environ. Health 39(2):73-81.
Visek, W.J. 1972. Effects of urea hydrolysis on cell life-span and metabolism. Fed. Proc. 31(3):1178-1193.
Walton, M. 1973. Industrial ammonia gassing. Br. J. Ind. Med. 30(1):78-86.
Weast, R.C., M.J. Astle, and W.H. Beyer, eds. 1984. CRC Handbook of Chemistry and Physics, 65th Ed. Boca Raton, FL: CRC Press.
Weatherby, J.H. 1952. Chronic toxicity of ammonia fumes by inhalation. Proc. Soc. Exp. Biol. Med. 81(1):300-301.
Wong, K.L. 1994. Ammonia. Pp. 39-59 in Spacecraft Maximum Allowable Concentrations for Selected Airborne Contaminants, Vol. 1. Washington, DC: National Academy Press.
Zissu, D. 1995. Histopathological changes in the respiratory tract of mice exposed to ten families of airborne chemicals. J. Appl. Toxicol. 15(3):207-213.
APPENDIX A
Derivation of AEGL-1 Values
|
Key study: |
MacEwen et al. 1970 |
|
Toxicity end point: |
Faint or irritation (humans) |
|
Time scaling: |
None |
|
Uncertainty factors: |
Interspecies: NA Intraspecies: 1 |
|
Calculations: |
|
|
10-min: AEGL-1: |
30 ppm/UF = 30 ppm/L = 30 ppm |
|
30-min, 1-, 4-, and 8-h: |
AEGL-1: Same as AEGL-1: 30 ppm |
Derivation of AEGL-2 Values
|
Key study: |
Verberk 1977 |
|
Toxicity end point: |
Irritation: eyes and upper respiratory tract in humans |
|
Time scaling: |
Cn × t = k; n = 2 (ten Berge et al. 1986) |
|
Uncertainty factors: |
1 for intraspecies variability; not applicable for interspecies sensitivity |
|
Calculations: |
|
|
Point of departure: |
110 ppm for 2 h |
|
10-min AEGL: |
Same as the 30-min value = 220 ppm |
|
30-min AEGL-2: |
Cn × t = k; C = 110 ppm, t = 120 min, n = 2 C = (k/t)1/2 = (1.45 × 106 ppm•min/30 min)1/2 C = 220 ppm |
|
1-h AEGL-2: |
C = (k/t)1/2 = (1.45 × 106 ppm•min/30 min)1/2 C = 160 ppm |
|
4-h AEGL-2: |
C = 110 ppm, same as the POD |
|
8-h AEGL-2: |
C = 110 ppm, same as the POD |
Derivation of AEGL-3 Values
|
Key study: |
Kapeghian et al. 1982; MacEwen and Vernot 1972 |
|
Toxicity end point: |
Lethality: the LC50 for the two sets of mouse data were extrapolated to an LC01 |
|
Time scaling: |
Cn × t = k; n = 2 (ten Berge et al. 1986) |
|
Uncertainty factors: |
Three for intraspecies variability; one for interspecies sensitivity |
|
Calculations: |
|
|
1-h AEGL-3: |
C = 3,317 ppm/3 (uncertainty factor) = 1,106 ppm C = 3,374 ppm/3 (uncertainty factor) = 1,125 ppm |
|
Kapeghian et al. 1982 |
Cn × t = k; C = 1,106 ppm, t = 60 min, n = 2, k = 7.335 107 ppm•min C = (k/t)1/2 = (7.335 × 107 ppm•min/60 min)1/2 C = 1,106 ppm = 1,100 ppm |
|
MacEwen and Vernot 1972 |
Cn × t = k; C = 1,125 ppm, t = 60 min, n = 2, k = 7.59 × 107 ppm•min C = (k/t)1/2 = (7.59 × 107 ppm•min/60 min)1/2 C = 1,125 ppm = 1,100 ppm |
|
10-min AEGL -3: |
C = (k/t)1/2 = (7.335 × 107 ppm•min/10 min)1/2 C = 2,708 ppm = 2,700 ppm C = (k/t)1/2 = (7.59 × 107 ppm•min/10 min)1/2 C = 2,755 ppm = 2,700 ppm |
|
30-min AEGL-3: |
C = (k/t)1/2 = (7.335 × 107 ppm•min/30 min)1/2 C = 1,564 ppm = 1,600 ppm C = (k/t)1/2 = (7.59 × 107 ppm•min/30 min)1/2 C = 1,591 ppm = 1,600 ppm |
|
4-h AEGL-3: |
C = (k/t)1/2 = (7.335 × 107 ppm•min/240 min)1/2 C = 553 ppm = 550 ppm C = (k/t)1/2 = (7.59 × 107 ppm•min/240 min)1/2 C = 562 ppm = 560 ppm |
|
8-h AEGL-3: |
C = (k/t)1/2 = (7.335 × 107 ppm•min/480 min)1/2 C = 391 ppm = 390 ppm C = (k/t)1/2 = (7.59 × 107 ppm•min/480 min)1/2 C = 398 ppm = 400 ppm |
APPENDIX B
Acute Exposure Guideline Levels for Ammonia
Derivation Summary for Ammonia AEGLS
AEGL-1 VALUES
|
10 min |
30 min |
1 h |
4 h |
8 h |
|
30 ppm |
30 ppm |
30 ppm |
30 ppm |
30 ppm |
|
Reference: MacEwen, J.D.; J. Theodore, and E. H. Vernot. 1970. Human exposure to EEL concentrations of monomethylhydrazine, AMRL-TR-70-102, Paper No. 23. In: Proc. 1st Ann. Conf. Environ. Toxicol., September 9-11, 1970, Wright-Patterson AFB, OH. Pp. 355-363. |
||||
|
Test species/Strain/Sex/Number: Humans. |
||||
|
Exposure route/Concentrations/Durations: Inhalation. |
||||
|
Effects: 30 ppm for 10 min: 2/6 subjects reported faint irritation; 3/6 reported no irritation; 1/6 provided no response. |
||||
|
End point/Concentration/Rationale: Faint irritation in human subjects exposed to 30 ppm of ammonia for 10 min. The responses by all subjects exposed to 30 ppm of ammonia were consistent with the definition of AEGL-1 or below the definition of AEGL-1. |
||||
|
Uncertainty factors/Rationale: Total uncertainty factor: 1. Interspecies: Not applicable. |
||||
|
Intraspecies: 1; Ammonia is a contact irritant and is efficiently scrubbed in the upper respiratory tract, particularly at the low AEGL-1 concentration; therefore, members of the population are not expected to respond differently to effects confined to the upper respiratory tract. Atopics, including asthmatics, and nonatopics responded similarly to a brief nasal exposure to ammonia. Exercising subjects showed only a clinically nonsignificant decrease in pulmonary function after exposure to ammonia. |
||||
|
10 min |
30 min |
1 h |
4 h |
8 h |
|
30 ppm |
30 ppm |
30 ppm |
30 ppm |
30 ppm |
|
Modifying factor: 1. |
||||
|
Animal to human dosimetric adjustment: Not applicable. |
||||
|
Time scaling: The severity of upper respiratory tract irritation is not expected to increase with duration of exposure to low concentrations of ammonia; therefore, the same value is applied to all AEGL-1 exposure duration. |
||||
|
Data adequacy: Upper respiratory tract irritation at 30 ppm and above is well documented in the literature. Therefore, sufficient data were available to document the irritation threshold. |
||||
AEGL-2 VALUES
|
10 min |
30 min |
1 h |
4 h |
8 h |
|
220 ppm |
220 ppm |
160 ppm |
110 ppm |
110 ppm |
|
Reference: Verberk, M.M. 1977. Effects of ammonia on volunteers. Int. Arch. Occup. Environ. Health 39:73-81. |
||||
|
Test species/Strain/Sex/Number: Humans, mixed sex; 8 expert and 8 nonexpert subjects. |
||||
|
Exposure route/Concentrations/Durations: Inhalation; 50, 80, 110, or 140 ppm for durations up to 2 h. |
||||
|
Effects: 50 ppm: just perceptible to offensive odor; no sensation to nuisance eye, nose, and throat irritation; no sensation to distinctly perceptible urge to cough, chest irritation, or general discomfort. 80 ppm: just perceptible to offensive odor; no sensation to offensive eye, nose, throat, and chest irritation and urge to cough; no sensation to nuisance general discomfort; 110 ppm: distinctly perceptible to offensive odor; no sensation to offensive eye, nose, throat, and chest irritation, urge to cough, or general discomfort; 140 ppm: just perceptible to offensive odor; just perceptible to unbearable eye irritation; no sensation to offensive nose, throat, and chest irritation, urge to cough, or general discomfort; severity ratings: 0 = no sensation, 1 = just perceptible, 2 = distinctly perceptible, 3 = nuisance, 4 = offensive, and 5 = unbearable. |
||||
|
End point/Concentration/Rationale: 110 ppm for 2 h; respiratory tract and eye irritation and urge to cough ranged from “no sensation” to “offensive” during the 2-h exposure of the nonexpert subjects. The AEGL-2 derivation was based on the response (offensive irritation) of the most sensitive nonexpert subjects. The responses changed very little between 30 min and 2 h. The nonexperts considered the effects to be near the maximum response (offensive), whereas the expert responses were always of a lesser degree. |
||||
|
10 min |
30 min |
1 h |
4 h |
8 h |
|
220 ppm |
220 ppm |
160 ppm |
110 ppm |
110 ppm |
|
Uncertainty factors/Rationale: Total uncertainty factor: 1. Interspecies: Not applicable. Intraspecies: 1; Ammonia is a contact irritant and is efficiently scrubbed in the upper respiratory tract, and any perceived irritation experienced by the general public including sensitive individuals at low AEGL-2 concentrations is not expected to be greater than that of the most sensitive nonexpert subject. Atopics, including asthmatics, and nonatopics responded similarly to a brief nasal exposure to ammonia; a child experienced less severe effects than that of an adult exposed to high concentrations of ammonia; and exercising subjects showed only a nonclinically significant decrease in pulmonary function after exposure to ammonia. |
||||
|
Modifying factor: 1; POD was from a controlled exposure study on human subjects. |
||||
|
Animal to human dosimetric adjustment: Not applicable. |
||||
|
Time scaling: Cn × t = k, where n = 2 based on an analysis of empirical mouse and rat lethality data in which the times of exposure ranged from 10 to 60 min (ten Berge et al. 1986). Values for 4 and 8 h are the same as the POD because the responses of the subjects did not change considerably between 30 min and 2 h and are not expected to change for exposures up to 8 h. The 10-min AEGL-2 is the same as the 30-min AEGL-2 because the time-scaled value of 380 ppm might impair escape. |
||||
|
Data adequacy: The AEGL-2 values were based on a study using human subjects exposed to ammonia for 2 h; the responses of the subjects ranged from “no sensation” to “offensive,” which is expected to be comparable to the range of responses in the general public, including sensitive individuals. Case reports of long-term or irreversible effects in humans with exposure estimates were not available in the literature. |
||||
AEGL-3 VALUES
|
10 min |
30 min |
1 h |
4 h |
8 h |
|
2,700 ppm |
1,600 ppm |
1,100 ppm |
550 ppm |
390 ppm |
|
References: MacEwen, J.D., and E.H. Vernot. 1972. Toxic Hazards Research Unit Annual Technical Report. SysteMed Report No. W-72003, AMRL-TR-72-62. Sponsor: Aerospace Medical Research Laboratory, Wright-Patterson AFB, OH. (I); Kapeghian, J.C., H.H. Mincer, and A.B. Hones et al. 1982. Acute inhalation toxicity of ammonia in mice. Bull. Environ. Contam. Toxicol. 29:371-378. (II) |
||||
|
Test species/Strain/Number: CF1 or ICR male mice, 10 or 12 per group. |
||||
|
10 min |
30 min |
1 h |
4 h |
8 h |
|
2,700 ppm |
1,600 ppm |
1,100 ppm |
550 ppm |
390 ppm |
|
Exposure route/Concentrations/Durations: Inhalation: 0, 3,600, 4,550, or 5,700 ppm for 1 h (I). Inhalation: 0; 1,190; 1,340; 2,130; 3,400; 3,950; 4,220; 4,860 ppm for 1 h (II). |
||||
|
Effects: (I): Clinical signs: nasal and eye irritation, labored breathing, gasping, convulsions, and low body weight gain. Mortality: 3,600 ppm (0/10), 4,500 ppm (3/10), and 5,720 ppm (9/10); LC01: 3,374 ppm. (II): Clinical signs: eye and nasal irritation, hypoactivity, labored breathing, ataxia, convulsions, weight loss. (III): Mortality: ≤3,440 ppm (0/12), 3,950 ppm (3/12), 4,220 ppm (5/12), 4,490 ppm (8/12), and 4,860 ppm (12/12); LC01: 3317 ppm. |
||||
|
End point/Concentration/Rationale: Lethality; LC01 = 3,374 ppm (I) and 3,317 ppm (II) for 1 h are the estimated thresholds for lethality derived by probit analysis of the data. Both numbers when divided by an uncertainty factor of 3 give the same result when the AEGL value is expressed to two significant figures. Uncertainty factors/Rationale: Total uncertainty factor: 3 Interspecies: 1, The mouse was unusually sensitive to ammonia compared with other mammalian species. An UF of 3 would yield a 30 min AEGL-3 value below a level that humans can tolerate (500 ppm) for 30 min. Intraspecies: 3, Life-threatening concentrations of ammonia cause severe tracheobronchial and pulmonary effects and these effects, are not expected to be more severe in asthmatics than in nonasthmatic individuals, more severe in children than in adults, or more severe in exercising than resting individuals, but tracheobronchial and pulmonary effects may occur at a lower concentration in the elderly than in young adults. Reflex glottis closure (protective mechanism) is 3-fold less sensitive in the elderly than in young subjects; this mechanism may only be applicable when concentrations of ammonia exceed 570 ppm. |
||||
|
Modifying factor: 1. |
||||
|
Animal to human dosimetric adjustment: 1. |
||||
|
Time scaling: Cn × t = k where n = 2 based on an empirical analysis of mouse and rat lethality data in which the durations of exposure ranged from 10 to 60 min (ten Berge et al. 1986). |
||||
|
Data adequacy: No quantitative exposure data were available for humans who died from exposure to ammonia. Lethality data were available for two animal species—mice and rats. The AEGL-3 values were based on two mouse studies that were in close agreement, although they were conducted 12 years apart by two different laboratories. |
||||