




Below is the uncorrected machine-read text of this chapter, intended to provide our own search engines and external engines with highly rich, chapter-representative searchable text of each book. Because it is UNCORRECTED material, please consider the following text as a useful but insufficient proxy for the authoritative book pages.
Appendix D Standards for Reporting Meta-Analyses of Clinical Trials and Observational Studies: QUOROM and MOOSE QUOROM CHECKLIST Improving the quality of reports of meta-analyses of randomised controlled trials (RCTs): The QUOROM statement checklist Heading Subheading Descriptor Title Identify the report as a meta-analysis [or systematic review] of RCTs Abstract Use a structured format Describe Objectives The clinical question explicitly Data sources The databases (i.e., list) and other information sources Review The selection criteria (i.e., population, intervention, methods outcome, and study design); methods for validity assessment, data abstraction, and study characteristics, and quantitative data synthesis in sufficient detail to permit replication Results Characteristics of the RCTs included and excluded; qualitative and quantitative findings (i.e., point estimates and confidence intervals); and subgroup analyses Conclusion The main results Describe Introduction The explicit clinical problem, biological rationale for the intervention and rationale for review 199
200 KNOWING WHAT WORKS IN HEALTH CARE Methods Searching The information sources, in detail (e.g., databases, registers, personal files, expert informants, agencies, hand-searching), and any restrictions (years considered, publication status, language of publication) Selection The inclusion and exclusion criteria (defining population, intervention, principal outcomes, and study design) Validity The criteria and process used (e.g., masked conditions, assessment quality assessment, and their findings) Data The process or processes used (e.g., completed abstraction independently, in duplicate) Study The type of study design, participantsâ characteristics, characteristics details of intervention, outcome definitions, &c, and how clinical heterogeneity was assessed Quantitative The principal measures of effect (e.g., relative risk), data method of combining results (statistical testing and Synthesis confidence intervals), handling of missing data; how statistical heterogeneity was assessed; a rationale for any a-priori sensitivity and subgroup analyses; and any assessment of publication bias Results Trial flow Provide a meta-analysis profile summarizing trial flow (see figure) Study Present descriptive data for each trial (e.g., age, sample characteristics size, intervention, dose, duration, follow-up period) Quantitative Report agreement on the selection and validity data synthesis assessment; present simple summary results (for each treatment group in each trial, for each primary outcome); present data needed to calculate effect sizes and confidence intervals in intention-to-treat analyses (e.g. 2Ã2 tables of counts, means and SDs (standard deviations), proportions) Discussion Summarize key findings; discuss clinical inferences based on internal and external validity; interpret the results in light of the totality of available evidence; describe potential biases in the review process (e.g., publication bias); and suggest a future research agenda Quality of reporting of meta-analyses Reprinted from Lancet, Vol 354, Moher, D., D. J. Cook, S. Eastwood, I. Olkin, D. Rennie, D. F. Stroup, and the QUOROM Group. Improving the quality of reports of meta-analyses of randomised controlled trials: The QUOROM statement, 1896-1900, Copyright 1999, with permission from Elsevier.
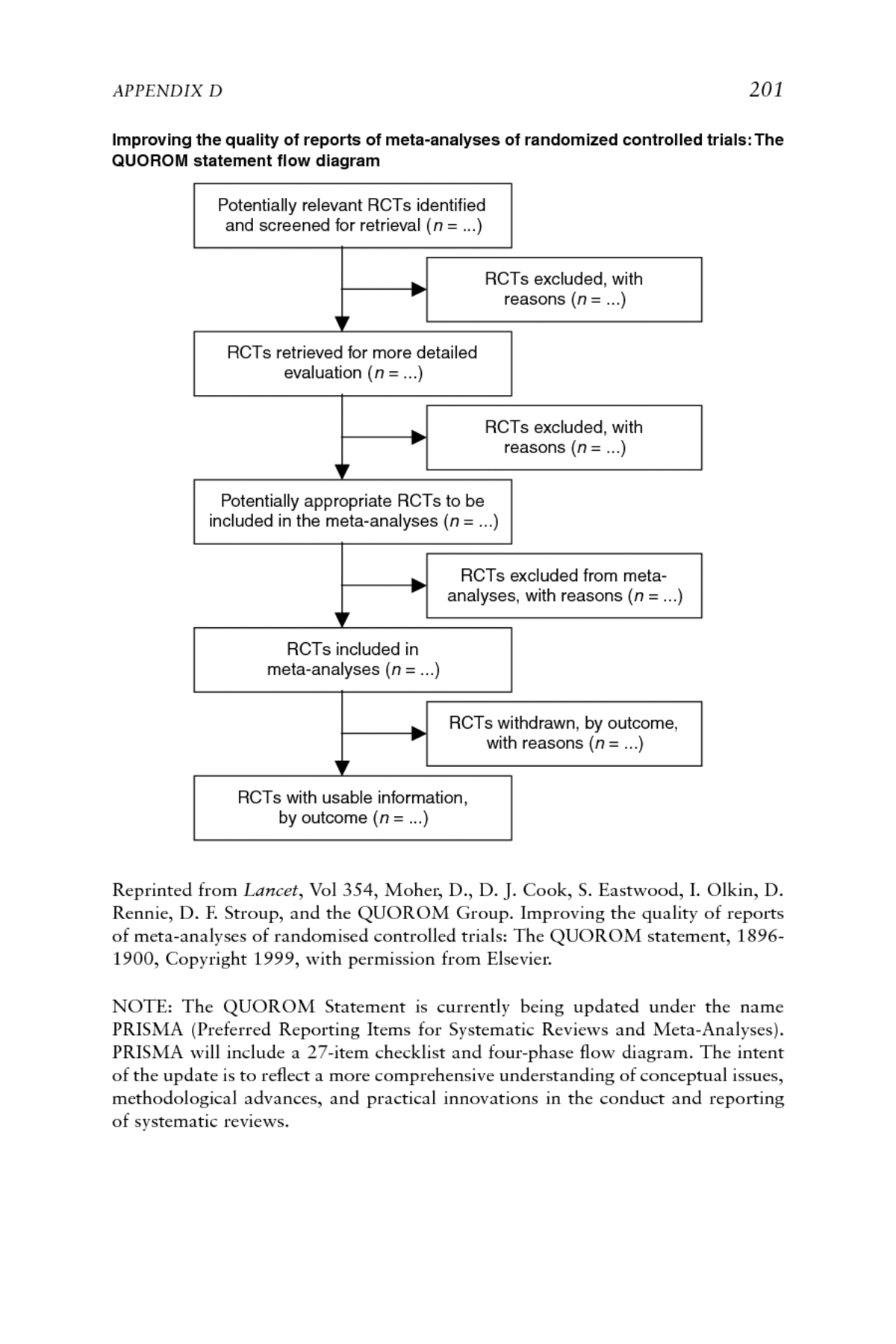
APPENDIX D 201 Improving the quality of reports of meta-analyses of randomized controlled trials: The QUOROM statement flow diagram Potentially relevant RCTs identified and screened for retrieval (n = ...) RCTs excluded, with reasons (n = ...) RCTs retrieved for more detailed evaluation (n = ...) RCTs excluded, with reasons (n = ...) Potentially appropriate RCTs to be included in the meta-analyses (n = ...) RCTs excluded from meta- analyses, with reasons (n = ...) RCTs included in meta-analyses (n = ...) RCTs withdrawn, by outcome, with reasons (n = ...) RCTs with usable information, by outcome (n = ...) Reprinted from Lancet, Vol 354, Moher, D., D. J. Cook, S. Eastwood, I. Olkin, D. Rennie, D. F. Stroup, and the QUOROM Group. Improving the quality of reports of meta-analyses of randomised controlled trials: The QUOROM statement, 1896- Appendix D 1900, Copyright 1999, with permission from Elsevier. NOTE: The QUOROM Statement is currently being updated under the name PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses). PRISMA will include a 27-item checklist and four-phase flow diagram. The intent of the update is to reflect a more comprehensive understanding of conceptual issues, methodological advances, and practical innovations in the conduct and reporting of systematic reviews.
202 KNOWING WHAT WORKS IN HEALTH CARE MOOSE CHECKLIST A Proposed Reporting Checklist for Authors, Editors, and Reviews of Meta-analyses Of Observational Studies Reporting of background should include Problem definition Hypothesis statement Description of study outcome(s) Type of exposure of intervention used Type of study designs used Study population Reporting of search strategy should include Qualifications of searchers (e.g., librarians and investigators) Search strategy, including time period included in the synthesis and keywords Effort to include all available studies, including contact with authors Databases and registries searched Search software used, name and version, including special features used (eg, explosion) Use of hand searching (e.g., reference lists of obtained articles) List of citations located and those excluded, including justification Method of addressing articles published in languages other than English Method of handling abstracts and unpublished studies Description of any contact with authors Reporting of methods should include Description of relevance or appropriateness of studies assembled for assessing the hypothesis to be tested Rationale for the selection and coding of data (e.g., sound clinical principles or convenience) Documentation of how data were classified and coded (e.g., multiple raters, blinding, and interrater reliability) Assessment of confounding (e.g., comparability of cases and controls in studies where appropriate) Assessment of study quality, including blinding of quality assessors; stratification or regression on possible predictors of study results Assessment of heterogeneity Description of statistical methods (e.g., complete description of fixed or random effects models, justification of whether the chosen models account for predictors of study results, dose-response models, or cumulative meta-analyses) in sufficient detail to be replicated Provision of appropriate tables and graphics Reporting of results should include Graphic summarizing individual study estimates and overall estimate Table giving descriptive information for each study included Results of sensitivity testing (e.g., subgroup analysis) Indication of statistical uncertainty findings Reporting of discussion should include Quantitative assessment of bias (e.g., publication bias) Justification for exclusion (e.g., exclusion of non-English-language citations) Assessment of quality of included studies Reporting of conclusions include Consideration of alternative explanations for observed results
APPENDIX D 203 Generalization of the conclusions (i.e., appropriate for the data presented and within the domain of the literature review) Guidelines for future research Disclosure of funding source Reprinted, with permission, from JAMA 2000, 283:2008-2012. Copyright 2000 by American Medical Association. All rights reserved. REFERENCES Cochrane Collaboration. 2006. Revising the QUOROM Statement. Cochrane News 37 http:// www.cochrane.org/newslett/CochraneNews37lores.pdf (accessed September 12, 2007). Moher, D., D. J. Cook, S. Eastwood, I. Olkin, D. Rennie, D. F. Stroup, and the QUOROM Group. 1999. Improving the quality of reports of meta-analyses of randomised controlled trials: The QUOROM statement. Lancet 354:1896-1900. PLoS editors. 2007. Many reviews are systematic but some are more transparent and com- pletely reported than others. PLoS Medicine 4(3):e147. Stroup, D. F., J. A. Berlin, S. C. Morton, I. Olkin, G. D. Williamson, D. Rennie, D. Moher, B. J. Becker, T. A. Sipe, S. B. Thacker, and the Meta-analysis Of Observational Studies in Epidemiology (MOOSE) Group. 2000. Meta-analysis of observational studies in epide- miology: A proposal for reporting. JAMA 283(15):2008-2012.
