2
The Global Spread of Multidrug-Resistant and Extensively Drug-Resistant Tuberculosis
SCOPE OF THE PROBLEM
Paul Nunn of WHO summarized the available surveillance data on TB, MDR TB, and XDR TB. WHO, together with the International Union Against Tuberculosis and Lung Disease (IUATLD), regularly collects and analyzes global TB surveillance data. WHO has estimated that in 2006, the most recent year for which data are available, the total number of cases of TB worldwide was just still growing—from 9.1 million in 2005 to 9.2 million in 2006 (WHO, 2008a; see Figure 2-1), although the global incidence of TB per capita fell slightly, continuing the trend since 2003 (see Figure 2-2). The most dramatic reductions in per capita incidence appear to have occurred in Africa, apparently as a result of reductions in the prevalence of HIV (Figure 2-2). As noted in Chapter 1 and shown in Table 2-1, WHO estimated that of the approximately 9.2 million cases of TB in 2006, approximately 489,000 (95 percent confidence level, 455,093–614,215), or 4.8 percent, were MDR TB, and about 40,000 (or 0.4 percent) were XDR TB (Nunn, 2008).
Limitations of Global TB Estimates
Nunn stated that the WHO surveillance data on MDR and XDR TB have large confidence limits. To determine the percentages of MDR and XDR TB among all cases of TB, WHO first estimates the percentage of cases that are MDR, and then within those cases, the percentage that are XDR. A number of factors complicate the WHO estimates. First, no data
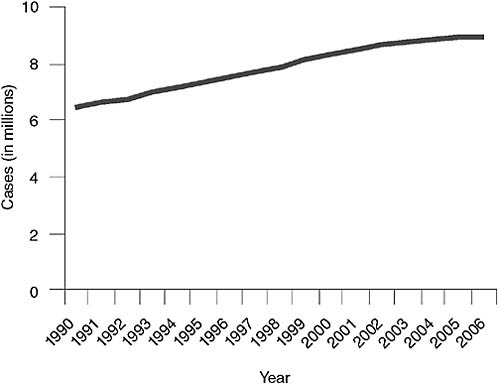
FIGURE 2-1 Global incidence of TB.
SOURCE: Nunn, 2008 (based on data from the WHO TB database, October 2008).
are available for many locations, particularly in sub-Saharan Africa. Second, in many countries, the availability of diagnostic laboratories is limited; nine African countries lack even a single reference laboratory capable of culturing TB and making a diagnosis. Finally, mortality data are unreliable because little is known about the long-term outcomes of the MDR cases that are reported. Megan Murray of Brigham and Women’s Hospital added that the WHO survey data represent surveillance samples of incident cases in which drug resistance was measured through drug sensitivity testing. The data come from two different sources: either newly presenting TB patients or retreatment cases. The data do not capture patients who develop MDR TB during the course of therapy, and therefore may yield considerable underestimates. The degree of underestimation will depend on when in the course of an epidemic the data are sampled. At the beginning of an epidemic, when many of those cases arise from people who fail therapy and amplify their drug resistance, the estimates will be especially low. Later, when transmission of drug resistance dominates most of those cases, the reported MDR TB rates will be higher.
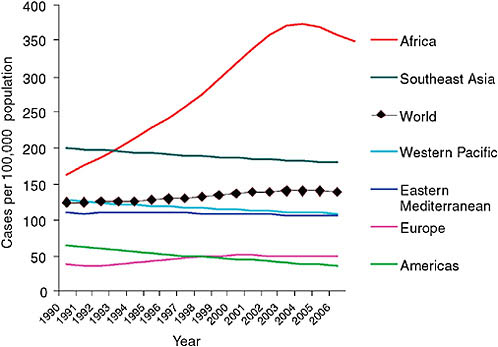
FIGURE 2-2 Per capita incidence of TB.
SOURCE: Nunn, 2008 (based on data from the WHO TB database, October 2008).
TABLE 2-1 Estimated Number of TB Cases and Number of Deaths, by Type, 2006
|
Form of TB |
Estimated Number of Cases |
Estimated Number of Deaths |
|
All forms |
9,200,000 |
1,650,000 |
|
Multidrug-resistant (MDR TB) |
489,000 |
120,000 |
|
Extensively drug-resistant (XDR TB) |
40,000 |
20,000 |
|
HIV-associated |
700,000 |
200,000 |
|
SOURCE: Nunn, 2008. (The data on total cases and deaths are from WHO, 2008a; the number of MDR TB cases is from WHO, 2008b; the deaths from MDR and XDR TB were estimated by Nunn’s team from published literature using the case numbers listed in the table; and the number of XDR TB cases [according to the revised October 2006 definition of XDR TB] was estimated from the MDR TB number listed in the table using the percentages from CDC, 2006.) |
||
Burden of MDR and XDR TB
Fifty-seven percent of the MDR TB burden is found in just three countries: as of 2006 China had about 131,000 cases per year, India about
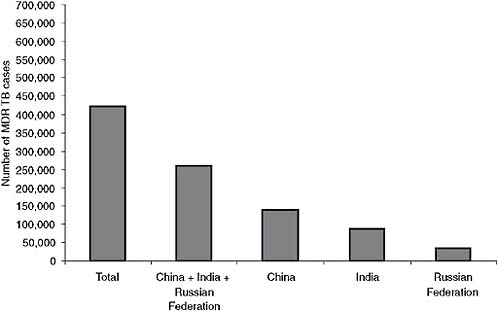
FIGURE 2-3 Two-thirds of the MDR TB burden is located in just three countries.
NOTE: As of 2006 China had about 131,000 cases per year, India about 110,000, and the Russian Federation about 36,000.
SOURCE: Nunn, 2008 (based on data from WHO, 2008b).
110,000, and the Russian Federation about 36,000 (see Figure 2-3). South Africa is a close fourth (WHO, 2008b). While a significant number of new TB cases are being diagnosed as MDR, most MDR TB occurs in previously treated patients. The incidence of MDR TB among previously treated patients is particularly high in Eastern Europe and in the eastern Mediterranean. In fact, the percentage of MDR TB among retreatment cases is approaching or exceeding 60 percent in three oblasts of the Russian Federation (Arkhangelsk, Tomsk, and Ivanovo) (Nunn, 2008). Nunn presented data comparing Estonia and Tomsk Oblasts following investments in TB and MDR TB control. Estonia’s notification rate for TB is decreasing, and the percentage of MDR TB is decreasing slightly. Although the TB notification rate in Tomsk is decreasing as well, the percentage of MDR TB among new cases is rising. These data demonstrate that investments in TB control alone will not be sufficient to combat the problem and that new drugs will be needed.
To investigate rates of XDR TB throughout the world and in a few selected regions, WHO and CDC surveyed the global Supranational Reference Laboratory (SRL) international network of laboratories using data for
2000–2004.1 The samples analyzed had been collected, tested for resistance to at least three second-line drugs, and stored. These data were compared with samples from the U.S. National Surveillance System (collected during 1993–2004); samples taken from a cohort of MDR TB patients in Latvia’s National MDR TB Registry during 2000–2002; and samples from South Korea’s National Reference Laboratory. With the exception of those from South Korea, the samples were not population based. The researchers found that, among the 49 countries included in the SRL network, 2 percent of cases were XDR TB; in the United States 4 percent were XDR TB, in Latvia 19 percent, and in South Korea 15 percent (CDC, 2006). Isolated incidents such as that which occurred in Tugela Ferry, South Africa (discussed below) demonstrate that pockets of dramatically higher rates are possible.
TB in the United States
Kenneth Castro of CDC discussed the status of TB in the United States. In 2007, 13,299 cases were reported in the United States. Until the mid-1980s, the incidence of TB in the United States had declined steadily—about 5–6 percent annually—for three decades. Concurrent with this decline in TB cases, categorical federal funds for TB control were progressively reduced, until they were eliminated in 1972 (IOM, 2000). From 1972 until 1980, federal funds were provided to the states in the form of block grants for control of communicable diseases, including TB. The result was the dismantling of many TB programs. An unprecedented resurgence of TB ensued, fueled by the association between TB and HIV and the occurrence of MDR TB. From 1985 to 1992, the incidence of TB increased by 20 percent in the United States. Because so many programs had been cut, the nation was ill prepared for this sudden resurgence. A federal TB task force was created in 1991 to coordinate the development of a national action plan to combat MDR TB, and Congress appropriated new funding for the effort. These new resources enabled many programs that had been dismantled to be reconstituted. As a result of this new investment, focused on rebuilding laboratory capacity, instituting infection control measures, and reinvigorating research capacity, the incidence of TB has again declined—by about 40 percent in the past 15 years. Unfortunately, according to Castro, renewed complacency has resulted as well. From 1993 to 2000, the rate of decline in the incidence of TB in the United States was 7.3 percent annually;
from 2000 to 2007, however, that figure fell to 3.8percent—an early sign of stagnation in the country’s progress against TB.
Among U.S.-born populations, the rate of TB in non-Hispanic blacks is eight times higher than in non-Hispanic whites. In 2007, fully 58 percent of U.S. cases were foreign born, reflecting the global nature of TB within the United States. Of these foreign-born cases, 24 percent were from Mexico, 12 percent from the Philippines, 8 percent from India, 7 percent from Vietnam, and 5 percent from China.
Fortunately, MDR TB has been under control in the United States since it was first recognized. In 1993, there were about 400 cases of MDR TB, which represented almost 3 percent of all culture-positive cases. That figure has since declined to about 100 cases of MDR TB—about 1percent—and has remained steady over the past 4–5 years.
Castro referred to the IOM report Ending Neglect (IOM, 2000), which offers five recommendations for addressing TB in the United States: (1) continue to maintain control by investing in the basic program activities that have yielded results; (2) accelerate the rate of decline, because the current rate is insufficient to achieve the goal of eliminating TB in the foreseeable future; (3) invest in research and development necessary to produce new tools (e.g., rapid and reliable diagnostics, new safe and effective drugs and vaccines); (4) invest in global control of TB because of its impact on the United States; and (5) advocate for and monitor progress toward the elimination of the disease. Castro also noted the imminent release of an article in the Journal of the American Medical Association describing the epidemiology of MDR TB (Shah et al., 2008).
Relationship Between HIV and TB
As noted, individuals who are HIV positive have compromised immune systems, and they are thus more susceptible to reactivation of their latent TB infection as their CD4 T lymphocyte counts fall. Although coinfection with HIV and MDR TB has received particular attention in Africa—especially in Tugela Ferry—it is also a threat in Eastern Europe. For example, Donetsk Province in the Ukraine has a high rate of MDR TB, and HIV was found to be a risk factor for MDR TB in a survey of all 4.7 million of its residents. Similar data exist for Latvia. Despite the prevalence of HIV and TB coinfection throughout the world, however, scientists have yet to determine conclusively whether there is a correlation between HIV status and transmission of TB (Escombe et al., 2008).
Lack of Adequate Treatments
Of even greater concern is that only about 10 percent of MDR TB patients are receiving any treatment, and only about 3 percent are being
treated under Green Light Committee (GLC) programs—according to WHO standards (WHO, 2008a).2 The remainder are probably being treated with drugs and programs of unknown quality, a particular concern in Eastern Europe.
Summary
Nunn summarized his remarks by noting that drug-resistant patients in 2004 were resistant to significantly more drugs than they were in 1994. MDR TB is increasing in several countries and decreasing in others. But MDR rates among new cases are approaching 25 percent in many Eastern European countries, and XDR TB is clearly emerging. Moreover, the overlap between TB and HIV infection is increasing the risk for MDR TB (WHO, 2008b).
UNDERREPORTING OF MDR TB IN AFRICA
Lack of Data
Yanis Ben Amor of the Earth Institute argued that the WHO data severely underestimate the problem of MDR TB in Africa. He stressed that WHO’s claim that rates in Africa are low is especially problematic because the TB community is willing to accept the WHO data, so appropriate measures to tackle the MDR TB threat in Africa are not taken. Although all of the African countries that have been surveyed, with the exception of Côte d’Ivoire, Mozambique, and Rwanda, show rates of MDR TB below 3 percent (see Figure 2-4), drug resistance surveys have never been conducted for more than half of Africa’s countries (25 of 46), so there are no data for these countries to support claims that MDR TB prevalence is low. Defenders of the WHO data justify these claims by arguing that national TB programs are operating effectively, and because of rifampicin’s later introduction to Africa relative to the rest of the world, there has been less time for resistance to develop.
Ben Amor compared the performance of national TB programs in the AFRO (African regional office) region with that of national TB programs in six countries of Eastern Europe that currently have the highest MDR TB rates (Estonia, Kazakhstan, Latvia, Lithuania, Russian Federation, and Uzbekistan). He reviewed data for the past 10 years and found that neither case detection nor treatment success rates offer strong evidence that the African programs have performed better than their counterparts in Eastern Europe, where MDR TB rates have already reached alarming levels (see
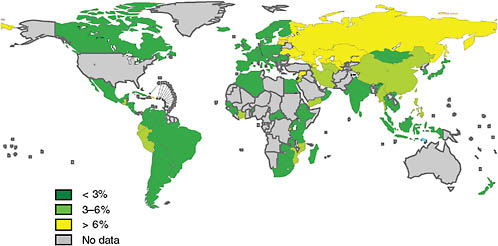
FIGURE 2-4 Percentage of MDR TB among new TB cases (1994–2007).
NOTE: Subnational averages were applied to China, Russia, and Indonesia. The boundaries and names shown and the designations used on this map do not imply the expression of any opinion whatsoever on the part of WHO concerning the legal status of any country, territory, city, or area or of its authorities, or concerning the delimitation of its frontiers or boundaries. Dotted lines on maps represent approximate border lines on which there may not yet be full agreement.
SOURCE: WHO, 2008b. Copyright 2008 WHO, reprinted with permission from WHO, 2008b.
Table 2-2). He suggested that the rifampicin argument is also invalid. For example, Mozambique introduced rifampicin roughly 10 years after South Africa, yet Mozambique’s MDR TB rate is already higher than South Africa’s. In addition, the MDR TB rate in Mozambique, which introduced rifampicin at the same time as Gambia, is 10 times higher than that in Gambia. Ben Amor further argued that countries identified as having the capability to conduct drug resistance surveys (WHO, 1997, 2000, 2004, 2008b) are more likely to have a well-functioning national TB program, laboratory structure, and transport network and therefore lower rates of MDR TB than those countries without these capabilities.
Old Data
Ben Amor discussed the credibility of the limited data that are being collected in Africa, arguing that there is compelling evidence that these data are misleading. The first and second maps of Africa shown in Figure 2-5 illustrate MDR TB data from the third WHO/IUATLD global report in 2004 and the fourth global report in 2008, respectively. The shading on the maps divides the countries into three categories on the basis of MDR TB rates (0 to 1.7 percent = low, 1.8 to 2.1 percent = moderate, and greater than 2.2 percent = high) (Ben Amor et al., 2008). In comparing these two maps, it is clear that over the past 4 years, all countries newly surveyed already had either moderate or high MDR TB levels, further suggesting that rates of MDR TB may not be as low as previously estimated. Furthermore, 14 of the 21 surveys illustrated on map 2 used data that were more than 5 years old. In the few settings where there were new surveys—for example, Botswana—MDR TB was found to be on the rise. According to Ben Amor, it is reasonable to assume that if new surveys were conducted in countries where the data are more than 5 years old, MDR TB rates would be higher. Using a formula developed by Zignol and colleagues (2006), map 3 shows
TABLE 2-2 Performance of National TB Programs
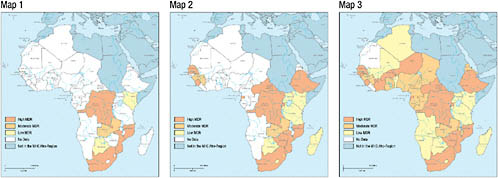
FIGURE 2-5 African countries with a known MDR TB rate.
NOTE: The first map represents data from the third global WHO/IUATLD report, published in 2004 (WHO, 2004); the second map represents data from the fourth WHO/IUATLD global report, published in 2008 (WHO, 2008b), as well as other sources; and the last map is based on a formula developed by Zignol et al., 2006, to estimate the MDR TB rate in countries in which a drug resistance survey was never conducted.
SOURCE: Updated from Ben Amor et al., 2008.
estimates of the MDR TB rates in those countries that have not yet conducted a drug resistance survey. This map shows a clustering of MDR TB in central Africa and South Africa. The formula used in map 3 suggests that, although Kenya’s last drug resistance survey was conducted in 1995 and the reported incidence was zero percent, a drug resistance survey conducted in Kenya today would not yield the same result.
Methodological Problems
The methodology used to conduct drug resistance surveys has a serious flaw that may result in underreporting. Current drug resistance surveys include only smear-positive TB cases, yet not all MDR TB cases are smear positive. In Latvia and the Ukraine, for example, where MDR TB rates are segregated on the basis of HIV status, it has been shown that there is a significant association between HIV positivity and MDR TB. Furthermore, HIV-positive TB patients are more likely than other TB patients to be smear negative. Given the high prevalence of HIV in many parts of Africa, current drug resistance protocols may substantially underestimate MDR TB levels.
Further underreporting may result from the fact that because MDR TB is underestimated in Africa, tools for its proper diagnosis in the region are not widely available. Even in settings where a national reference laboratory is in place to conduct drug resistance surveys, it is considered unethical to diagnose patients if second-line drugs capable of treating MDR TB are not available, which is the case in most countries where Ben Amor’s project, the Millennium Villages Project, has sites.
An Example: KwaZulu-Natal Province in Rural South Africa
Gerald Friedland of the Tugela Ferry Care and Research Collaboration provided a perspective on the MDR TB epidemic based on his experience in KwaZulu-Natal Province in rural South Africa, specifically Tugela Ferry. This rural area with a traditional Zulu population has an extraordinarily high incidence of TB—more than 1,000 per 100,000 population. The incidence of MDR TB, by Friedland’s calculation, is 100 per 100,000 population, much higher even than nonresistant TB in most parts of the world. Among MDR and XDR TB patients, the rate of HIV coinfection is greater than 90 percent. When this catastrophe was reported several years ago (Gandhi et al., 2006), many thought it was an outbreak that would dissipate over time. As indicated in Figure 2-6, however, from 2005 to 2007, both MDR and XDR TB cases continued to increase, and the ratio of XDR to MDR remained very high. In 2007, there were 102 MDR and 146 XDR cases. By comparison, in the United States, 124 cases of MDR
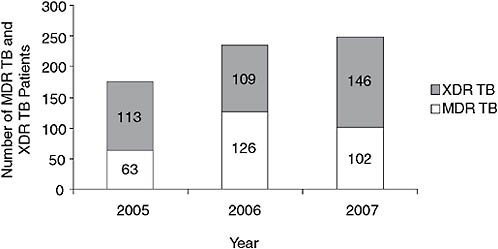
FIGURE 2-6 Numbers of MDR TB and XDR TB patients in Tugela Ferry, 2005–2007.
NOTE: The numbers of both MDR and XDR TB cases have continued to increase, as has the ratio of XDR to MDR.
SOURCE: Friedland, 2008.
TB were diagnosed in 2005, and from 1993 to 2005, 83 XDR cases were diagnosed.
Because of the data limitations discussed above, the full extent of the presence and consequences of MDR and XDR TB is unknown. According to Friedland, however, this outbreak is not limited to Tugela Ferry, but is much more widespread. By mid-2007, XDR TB had been reported by 60 health care facilities in KwaZulu-Natal; more than 4,700 cases of TB had been reported, 6 percent of which were XDR TB. The majority of the cases no longer came from Tugela Ferry. Neighboring countries—Botswana, Mozambique, Lesotho, Namibia, and possibly Zimbabwe—as well as all of the provinces in South Africa had reported cases as well.
Figure 2-7 demonstrates the limited knowledge of the extent of MDR TB in KwaZulu-Natal. At the beginning of the epidemic, the one facility in the province that had access to second-line drugs treated 686 patients for MDR TB, a mere 28 percent of the nearly 2,500 cases of MDR TB that were diagnosed. Behind these numbers, however, are an unknown number of patients who were seen and not diagnosed because of both the paucity of laboratory facilities and a policy of not doing cultures for individuals when they first present with TB, but only when they have failed treatment with first-line drugs. Finally, according to Friedland, the largest group is likely those who have never been diagnosed or treated because they have not visited a health care facility.
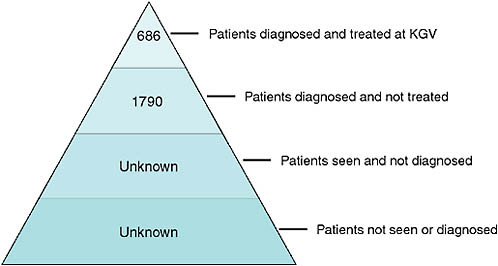
FIGURE 2-7 A representation of the limited knowledge of the extent of MDR TB in KwaZulu-Natal Province, 2006.
NOTE: KGV = King George V Hospital.
SOURCE: Friedland, 2008.
Impact of MDR and XDR TB on Mortality
Very high mortality is associated with the cases of MDR and XDR TB in Tugela Ferry. More than two-thirds of patients diagnosed with MDR TB and about 82 percent of patients with XDR TB die (Gandhi et al., 2009). Figure 2-8 shows the differences in mortality between MDR and XDR TB for Tugela Ferry; the top curve represents MDR TB mortality and the bottom curve XDR TB mortality. There is also a relationship between drug resistance and mortality. Rapid mortality is seen within the first 30 days; after 30 days, the rate of mortality increases with an increase in the number of drugs to which patients’ organisms are resistant.
Martie Van der Walt of the Medical Research Council South Africa addressed the impact of MDR and XDR TB on mortality in South Africa. She stated that while the problem of XDR TB in South Africa was quite grave in 2005, the Medical Research Council believes the situation has stabilized with regard to prevalence and early mortality. She presented preliminary results based on data gathered in the Eastern Cape Province of South Africa, just south of KwaZulu-Natal Province, demonstrating the progress that has been made since 2005. Data collection efforts involved two cohorts of XDR TB patients—the first started in October 2006 and the second in October 2007. Preliminary results indicated improvement in the survival rates of patients diagnosed with XDR TB between the 2006
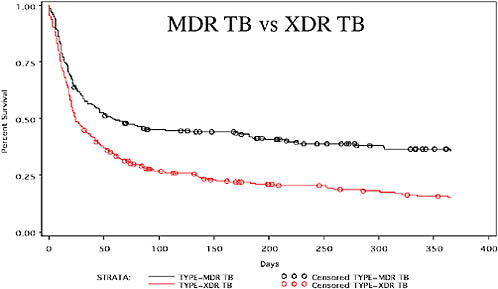
FIGURE 2-8 High mortality due to MDR and XDR TB in Tugela Ferry (2005–2007).
NOTE: The top curve represents MDR TB mortality (67 percent), and the bottom curve represents XDR TB mortality (82 percent).
SOURCE: Gandhi et al., 2009.
and 2007 cohorts. Van der Walt attributed these results to intensified case identification of and screening for drug resistance among treatment failures and patients with MDR TB. Despite this improvement, however, early mortality remains very high, perhaps because of baseline morbidity or additional drug resistance.
Using data from CDC’s Preserving Effectiveness of TB Treatment (PETT) study, Van der Walt examined the drug resistance of baseline isolates among MDR TB patients enrolled in 2005 and found that 1 percent of the 800 cases screened were resistant to all drugs tested. She explained that, just as the emergence of XDR TB strains is in some cases a natural consequence of treating MDR TB patients, fully resistant strains can be expected to be the next threat, and may be a cause of XDR TB patients not responding well to treatment for XDR TB. She hypothesized that some of the high mortality among XDR TB patients could be due to the emergence of fully resistant TB. She also cited as part of the problem patients’ lack of adherence to proper treatment protocols. As discussed later in this report, Neel Gandhi of the Tugela Ferry Care and Research Collaboration attributes this high mortality rate to high coinfection with HIV.
There are currently limited options for dealing with patients with drug-resistant disease in a country such as South Africa where the current
burden of MDR and XDR TB translates into limited bed capacity. Social and ethical issues arise with respect to keeping patients in lifelong isolation in hospitals, yet they cannot be sent home without treatment. Van der Walt stressed the importance of acting now to develop new drugs. Until new drugs are approved, she favors compassionate use of new drugs that are in development for the treatment of XDR or fully resistant TB.
THE THREAT OF TOTALLY DRUG-RESISTANT TB
Sarita Shah of the Tugela Ferry Care and Research Collaboration presented data based on ongoing surveillance for drug-resistant TB in Tugela Ferry demonstrating the steady progression of drug resistance in XDR TB patients over a 2-year period. Between 2005 and 2007, approximately 300 XDR TB cases were diagnosed. By 2007, the percentage of cases in South Africa resistant to all tested drugs—including all four of the first-line drugs and two second-line drugs—had risen from approximately 30 percent to approximately 95 percent.
Shah also presented data from a survey of the SRL network that first defined and quantified XDR TB. In this survey of more than 17,000 isolates, 234 that met the criteria for XDR TB were identified, and it was found that many of these XDR TB isolates had resistance to drugs beyond the minimum number required to be defined as XDR TB. Half of the XDR isolates were resistant to all four of the first-line drugs; many of these had additional second-line drug resistance, resulting in isolates with resistance to up to 10 drugs (Shah et al., 2007). This level of resistance severely limits treatment options based on the WHO guidelines for the programmatic management of drug-resistant TB, which require treatment with at least four effective drugs.
Alexander Sloutsky of the University of Massachusetts provided additional evidence for the potential emergence of totally resistant TB. Data from the Central Tuberculosis Research Institute in Moscow demonstrate the existence of one case of total drug resistance as early as 1997, with three additional cases in the following year. Sloutsky also showed data from Tomsk prison collected before the beginning of the Institute’s MDR TB treatment project, revealing that of 91MDR TB patients tested, 2 had a fully drug-resistant form of the disease (Sloutsky, 2008).
IMPORTANCE OF BETTER DATA
The true incidence and prevalence of drug-resistant TB are poorly characterized. Mark Harrington of the Treatment Action Group suggested that, while the data presented throughout the workshop suggest a crisis of significant magnitude, the paucity and limitations of those data are severe:
-
Most countries do not even report on their drug-resistant TB situation.
-
Data sets for most cohorts are incomplete, unpublished, or unvalidated.
-
There is no rigorous, randomized evidence for the standard of care for MDR TB.
Harrington noted that data are crucial to mounting the necessary response to the crisis. He cited data as one of the key elements that enabled activists to transform the struggle against HIV, saying, “We brought data to policy makers and scientists, and we used the data to demand money, services, and the nation’s attention.”
















