4 Evaluating Hazards and Assessing Risks in the Laboratory
4.B.1 Chemical Hygiene Plan (CHP)
4.B.2 Material Safety Data Sheets (MSDSs)
4.B.3 Globally Harmonized System for Hazard Communication
4.B.4 Laboratory Chemical Safety Summaries
4.B.6 Additional Sources of Information
4.B.7.1 The National Library of Medicine Databases
4.B.7.2 Chemical Abstracts Databases
4.C TOXIC EFFECTS OF LABORATORY CHEMICALS
4.C.1.1 Dose-Response Relationships
4.C.1.2 Duration and Frequency of Exposure
4.C.2 Assessing Risks of Exposure to Toxic Laboratory Chemicals
4.C.3.1 Irritants, Corrosive Substances, Allergens, and Sensitizers
4.C.3.4 Reproductive and Developmental Toxins
4.C.3.5 Toxins Affecting Other Target Organs
4.D FLAMMABLE, REACTIVE, AND EXPLOSIVE HAZARDS
4.D.1.2 Flammability Characteristics
4.D.1.3 Classes of Flammability
A key element of planning an experiment is assessing the hazards and potential risks associated with the chemicals and laboratory operations to be used. This chapter provides a practical guide for the trained laboratory personnel engaged in these activities. Section 4.B introduces the sources of information for data on toxic, flammable, reactive, and explosive chemical substances. Section 4.C discusses the toxic effects of laboratory chemicals by first presenting the basic principles that form the foundation for evaluating hazards for toxic substances. The remainder of this section describes how trained laboratory personnel can use this understanding and the sources of information to assess the risks associated with potential hazards of chemical substances and then to select the appropriate level of laboratory practice as discussed in Chapter 4. Sections 4.D and 4.E present guidelines for evaluating hazards associated with the use of flammable, reactive, and explosive substances and physical hazards, respectively. Finally, nanomaterials, biohazards, and radioactivity hazards are discussed briefly in sections 4.F and 4.G, respectively.
The primary responsibility for proper hazard evaluations and risk assessments lies with the person performing the experiment. That being said, the responsibility is shared by the laboratory supervisor. The actual evaluations and assessments may be performed by trained laboratory personnel, but these should be checked and authorized by the supervisor. The supervisor is also responsible for ensuring that everyone involved in an experiment and those nearby understand the evaluations and assessments. For example, depending on the level of training and experience, the immediate laboratory supervisor may be involved in the experimental work itself. In addition, some organizations have environmental health and safety (EHS) offices, with industrial hygiene specialists to advise trained laboratory personnel and their supervisors in risk assessment. When required by federal regulation, Chemical Hygiene Officers (CHOs) play similar roles in many organizations. As part of a culture of safety, all of these groups work cooperatively to create a safe environment and to ensure that hazards are appropriately identified and assessed prior to beginning work.
4.B.1 Chemical Hygiene Plan (CHP)
Beginning in 1991, every laboratory in which hazardous chemicals are used has been required by federal regulations (Occupational Safety and Health Administration [OSHA] Occupational Exposure to Hazardous Chemicals in Laboratories, 29 CFR § 1910.1450) to have a written CHP, which includes provisions capable of protecting personnel from the ‘’health hazards presented by hazardous chemicals used in that particular workplace.” All laboratory personnel should be familiar with and have ready access to their institution’s CHP. In some laboratories, CHPs include standard operating procedures for work with specific chemical substances, and the CHP may be sufficient as the primary source of information used for risk assessment and experiment planning. However, most CHPs provide only general procedures for handling chemicals, and prudent experiment planning requires that laboratory personnel consult additional sources for information on the properties of the substances that will be encountered in the proposed experiment. Many laboratories require documentation of specific hazards and controls for a proposed experiment.
4.B.2 Material Safety Data Sheets (MSDSs)
Federal regulations (OSHA Hazard Communication Standard 29 CFR § 1910.1200) require that manufacturers and distributors of hazardous chemicals provide users with material safety data sheets (MSDSs),1 which are designed to provide the information needed to protect users from any hazards that may be associated with the product. MSDSs have become the primary vehicle through which the potential hazards of materials obtained from commercial sources are communicated to trained laboratory personnel. Institutions are required by law (OSHA Hazard Communication Standard) to retain and make readily available the MSDSs provided by chemical suppliers. The MSDSs themselves may be electronic or on paper, as long as employees have unrestricted access to the documents. Be aware that some laboratories have been asked by local emergency personnel to print paper copies in the event of an emergency.
As the first step in risk assessment, trained laboratory personnel should examine any plan for a proposed experiment and identify the chemicals with toxicological properties they are not familiar with from previous experience. The MSDS for each unfamiliar chemical should be examined. Procedures for accessing MSDS files vary from institution to institution. In some cases, MSDS files are present in each laboratory, but often complete files of MSDSs are maintained only in a central location, such as the institution’s EHS office. Many laboratories are able to access MSDSs electroni-
_______________
1In the Globally Harmonized System for Hazard Communication, the term “material safety data sheet” has been shortened to “safety data sheet (SDS).” This book will continue to use the term MSDS as it is more recognizable at the time of writing than SDS.
cally, either from CD-ROM disks, via the internet, or from other computer networks. Laboratory personnel can always contact the chemical supplier directly and request that an MSDS be sent by mail.
MSDSs are technical documents, several pages long, typically beginning with a compilation of data on the physical, chemical, and toxicological properties of the substance and providing concise suggestions for handling, storage, and disposal. Finally, emergency and first-aid procedures are usually outlined. At present, there is no required format for an MSDS; however, OSHA recommends the general 16-part format created by the American National Standards Institute (ANSI Z400.1). The information typically found in an MSDS follows:
1. Supplier (with address and phone number) and date MSDS was prepared or revised. Toxicity data and exposure limits sometimes undergo revision, and for this reason MSDSs should be reviewed periodically to check that they contain up-to-date information. Phone numbers are provided so that, if necessary, users can contact the supplier to obtain additional information on hazards and emergency procedures.
2. Chemical. For products that are mixtures, this section may include the identity of most but not every ingredient. Hazardous chemicals must be identified. Common synonyms are usually listed.
3. Physical and chemical properties. Data such as melting point, boiling point, and molecular weight are included here.
4. Physical hazards. This section provides data related to flammability, reactivity, and explosion hazards.
5. Toxicity data. OSHA, the National Institute for Occupational Safety and Health (NIOSH), and the American Conference of Governmental Industrial Hygienists (ACGIH) exposure limits (as discussed in section 4.C.2.1) are listed. Many MSDSs provide lengthy and comprehensive compilations of toxicity data and even references to applicable federal standards and regulations.
6. Health hazards. Acute and chronic health hazards are listed, together with the signs and symptoms of exposure. The primary routes of entry of the substance into the body are also described. In addition, potential carcinogens are explicitly identified. In some MSDSs, this list of toxic effects is quite lengthy and includes every possible harmful effect the substance has under the conditions of every conceivable use.
7. Storage and handling procedures. This section usually consists of a list of precautions to be taken in handling and storing the material. Particular attention is devoted to listing appropriate control measures, such as the use of engineering controls and personal protective equipment necessary to prevent harmful exposures. Because an MSDS is written to address the largest scale at which the material could conceivably be used, the procedures recommended may involve more stringent precautions than are necessary in the context of laboratory use.
8. Emergency and first-aid procedures. This section usually includes recommendations for firefighting procedures, first-aid treatment, and steps to be taken if the material is released or spilled. Again, the measures outlined here are chosen to encompass worst-case scenarios, including accidents on a larger scale than are likely to occur in a laboratory.
9. Disposal considerations. Some MSDSs provide guidelines for the proper disposal of waste material. Others direct the users to dispose of the material in accordance with federal, state, and local guidelines.
10. Transportation information. This chapter only evaluates the hazards and assesses the risks associated with chemicals in the context of laboratory use. MSDSs, in contrast, must address the hazards associated with chemicals in all possible situations, including industrial manufacturing operations and large-scale transportation accidents. For this reason, some of the information in an MSDS may not be relevant to the handling and use of that chemical in a laboratory. For example, most MSDSs stipulate that self-contained breathing apparatus and heavy rubber gloves and boots be worn in cleaning up spills, even of relatively nontoxic materials such as acetone. Such precautions, however, might be unnecessary in laboratory-scale spills of acetone and other substances of low toxicity.
Originally, the principal audience for MSDSs was constituted of health and safety professionals (who are responsible for formulating safe workplace practices), medical personnel (who direct medical surveillance programs and treat exposed workers), and emergency responders (e.g., fire department personnel). With the promulgation of federal regulations such as the OSHA Hazard Communication Standard (29 CFR § 1910.1200) and the OSHA Laboratory Standard (29 CFR § 1910.1450), the audience for MSDSs has expanded to include trained laboratory personnel in industrial and academic laboratories. However, not
all MSDSs are written to meet the requirements of this new audience effectively.
In summary, among the currently available resources, MSDSs remain the best single source of information for the purpose of evaluating the hazards and assessing the risks of chemical substances. However, laboratory personnel should recognize the limitations of MSDSs as applied to laboratory-scale operations. If MSDSs are not adequate, specific laboratory operating procedures should be available for the specific laboratory manipulations to be employed:
1. The quality of MSDSs produced by different chemical suppliers varies widely. The utility of some MSDSs is compromised by vague and unqualified generalizations and internal inconsistencies.
2. Unique morphology of solid hazardous chemicals may not be addressed in MSDSs; for example, an MSDS for nano-size titanium dioxide may not present the unique toxicity considerations for these ultrafine particulates.
3. MSDSs must describe control measures and precautions for work on a variety of scales, ranging from microscale laboratory experiments to large manufacturing operations. Some procedures outlined in an MSDS may therefore be unnecessary or inappropriate for laboratory-scale work. An unfortunate consequence of this problem is that it tends to breed a lack of confidence in the relevance of the MSDS to laboratory-scale work.
4. Many MSDSs comprehensively list all conceivable health hazards associated with a substance without differentiating which are most significant and which are most likely to actually be encountered. As a result, trained laboratory personnel may not distinguish highly hazardous materials from moderately hazardous and relatively harmless ones.
4.B.3 Globally Harmonized System (GHS) for Hazard Communication
The GHS of Classification and Labeling of Chemicals is an internationally recognized system for hazard classification and communication. (Available at http://www.unece.org.) It was developed with support from the International Labour Organization (ILO), the Organisation for Economic Co-operation and Development, and the United Nations Sub-Committee of Experts on the Transport of Dangerous Goods with the goal of standardizing hazard communication to improve the safety of international trade and commerce. Within the United States, the responsibility for implementing the GHS falls to four agencies: OSHA, the Department of Transportation, the EPA, and the Consumer Product Safety Commission. At the time this book was written, the agencies had not yet provided final guidance on use of GHS. The revised Hazard Communication Standard (29 CFR § 1910.1200) is expected to be issued by OSHA in the near future.
GHS classifies substances by the physical, health, and environmental hazards that they pose, and provides signal words (e.g., Danger), hazard statements (e.g., may cause fire or explosion), and standard pictogram-based labels to indicate the hazards and their severity. When transporting hazardous chemicals, use the pictograms specified in the UN Recommendations on the Transport of Dangerous Goods, Model Regulations. For other purposes, the pictograms in Figure 4.1 should be used. Container labels should have a product identifier with hazardous ingredient disclosure, supplier information, a hazard pictogram, a signal word, a hazard statement, first-aid information, and supplemental information. Three of these elements—the pictograms, signal word, and hazard statements—are standardized under GHS. The signal words, either “Danger” or “Warning,” reflect the severity of hazard posed. Hazard statements are standard phrases that describe the nature of the hazard posed by the material (e.g., heating may cause explosion).
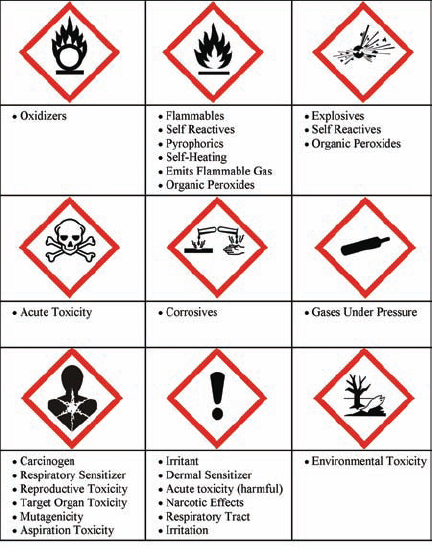
FIGURE 4.1 GHS placards for labeling containers of hazardous chemicals.
GHS recognizes 16 types of physical hazards, 10 types of health hazard, and an environmental hazard.
Physical hazards include
• explosives;
• flammable gases;
• flammable aerosols;
• oxidizing gases;
• gases under pressure;
• flammable liquids;
• flammable solids;
• self-reactive substances;
• pyrophoric liquids;
• pyrophoric solids;
• self-heating substances;
• substances which, in contact with water, emit flammable gases;
• oxidizing liquids;
• oxidizing solids;
• organic peroxides; and
• corrosive to metals.
Health hazards include
• acute toxicity,
• skin corrosion or irritation,
• serious eye damage or eye irritation,
• respiratory or skin sensitization,
• germ cell mutagenicity,
• carcinogenicity,
• reproductive toxicology,
• target organ systemic toxicity—single exposure,
• target organ systemic toxicity—repeated exposure, and
• aspiration hazard.
Environmental hazard includes
• Hazardous to the aquatic environment:
ο acute aquatic toxicity or
ο chronic aquatic toxicity with
• bioaccumulation potential
• rapid degradability.
In addition to the labeling requirements, GHS requires a standard format for Safety Data Sheets (SDS) that accompany hazardous chemicals. Note the change in terminology from MSDS. SDSs must contain a minimum of 16 elements:
1. identification,
2. hazard(s) identification,
3. composition/information on ingredients,
4. first-aid measures,
5. firefighting measures,
6. accidental release measures,
7. handling and storage,
8. exposure controls/personal protection,
9. physical and chemical properties,
10. stability and reactivity,
11. toxicological information,
12. ecological information,
13. disposal considerations,
14. transport information,
15. regulatory information, and
16. other information.
As with current MSDSs, these sheets are intended to inform employers and personnel of the hazards associated with the chemicals they are handling, and to act as a resource for management of the chemicals. Trained personnel should evaluate the information and use it to develop safety and emergency response policies, protocols, and procedures that are tailored to the workplace or laboratory.
4.B.4 Laboratory Chemical Safety Summaries (LCSSs)
As discussed above, although MSDSs are invaluable resources, they suffer some limitations as applied to risk assessment in the specific context of the laboratory. Committee-generated LCSSs, which are tailored to trained laboratory personnel, are on the CD accompanying this book. As indicated in their name, LCSSs provide information on chemicals in the context of laboratory use. These documents are summaries and are not intended to be comprehensive or to fulfill the needs of all conceivable users of a chemical. In conjunction with the guidelines described in this chapter, the LCSS gives essential information required to assess the risks associated with the use of a particular chemical in the laboratory.
The format, organization, and contents of LCSSs are described in detail in the introduction on the CD. Included in an LCSS are the key physical, chemical, and toxicological data necessary to evaluate the relative degree of hazard posed by a substance. LCSSs also contain a concise critical discussion, presented in a style readily understandable to trained laboratory personnel, of the toxicity, flammability, reactivity, and explosivity of the chemical; recommendations for the handling, storage, and disposal of the title substance; and first-aid and emergency response procedures.
The CD contains LCSSs for 91 chemical substances. Several criteria were used in selecting these chemicals, the most important consideration being whether the
substance is commonly used in laboratories. Preference was also given to materials that pose relatively serious hazards. Finally, an effort was made to select chemicals representing a variety of classes of substances, so as to provide models for the future development of additional LCSSs. A blank copy of the form is provided for development of laboratory-specific LCSSs.
4.B.5 Labels
Commercial suppliers are required by law (OSHA Hazard Communication Standard) to provide their chemicals in containers with precautionary labels. Labels usually present concise and nontechnical summaries of the principal hazards associated with their contents. Note that precautionary labels do not replace MSDSs and LCSSs as the primary sources of information for risk assessment in the laboratory. However, labels serve as valuable reminders of the key hazards associated with the substance. As with the MSDS, the quality of information presented on a label can be inconsistent. Additionally, labeling is not always required for chemicals transferred between laboratories within the same building.
4.B.6 Additional Sources of Information
The resources described above provide the foundation for risk assessment of chemicals in the laboratory. This section highlights the sources that should be consulted for additional information on specific harmful effects of chemical substances. Although MSDSs and LCSSs include information on toxic effects, in some situations laboratory personnel should seek additional more detailed information. This step is particularly important when laboratory personnel are planning to use chemicals that have a high degree of acute or chronic toxicity or when it is anticipated that work will be conducted with a particular toxic substance frequently or over an extended period of time. Institutional CHPs include the requirement for CHOs, who are capable of providing information on hazards and controls. CHOs can assist laboratory personnel in obtaining and interpreting hazard information and in ensuring the availability of training and information for all laboratory personnel.
Sections 4.B.2 and 4.B.4 of this chapter provide explicit guidelines on how laboratory personnel use the information in an MSDS or LCSS, respectively, to recognize when it is necessary to seek such additional information.
The following annotated list provides references on the hazardous properties of chemicals and which are useful for assessing risks in the laboratory.
1. International Chemical Safety Cards from the International Programme on Chemical Safety (IPCS, 2009). The IPCS is a joint activity of the ILO, the United Nations Environment Programme, and the World Health Organization. The cards contain hazard and exposure information from recognized sources and undergo international peer review. They are designed to be understandable to employers and employees in factories, agriculture, industrial shops, and other areas, and can be considered complements to MSDSs. They are available in 18 languages and can be found online through the NIOSH Web site, www.cdc.gov/niosh, or through the ILO Web site, www.ilo.org.
2. NIOSH Pocket Guide to Chemical Hazards (HHS/CDC/NIOSH, 2007). This volume is updated regularly and is found on the NIOSH Web site (http://www.cdc.gov/niosh). These charts are quick guides to chemical properties, reactivities, exposure routes and limits, and first-aid measures.
3. A Comprehensive Guide to the Hazardous Properties of Chemical Substances, 3rd edition (Patnaik, 2007). This particularly valuable guide is written at a level appropriate for typical laboratory personnel. It covers more than 1,500 substances; sections in each entry include uses and exposure risk, physical properties, health hazards, exposure limits, fire and explosion hazards, and disposal or destruction. Entries are organized into chapters according to functional group classes, and each chapter begins with a general discussion of the properties and hazards of the class.
4. 2009 TLVs and BEIs: Based on the Documentation of the Threshold Limit Values for Chemical Substances and Physical Agents and Biological Exposure Indices (ACGIH, 2009). A handy booklet listing ACGIH threshold limit values (TLVs) and short-term exposure limits (STELs). These values are under continuous review, and this booklet is updated annually. The multivolume publication Documentation of the Threshold Limit Values and Biological Exposure Indices (ACGIH, 2008b) reviews the data (with reference to literature sources) that were used to establish the TLVs. (For more information about TLVs, see section 4.C.2.1 of this chapter.)
5. Fire Protection for Laboratories Using Chemicals (NFPA, 2004). This is the national fire safety code pertaining to laboratory use of chemicals. It describes the basic requirements for fire protection of life and property in the laboratory. For example, the document outlines technical
requirements for equipment such as fire suppression systems and ventilation systems for flammables and defines the maximum allowable quantities for flammable materials within the laboratory.
6. Fire Protection Guide to Hazardous Materials, 13th edition (NFPA, 2001). This resource contains hazard data on hundreds of chemicals and guidance on handling and storage of, and emergency procedures for, those chemicals.
7. Bretherick’s Handbook of Reactive Chemical Hazards, 7th edition (Urben, 2007). This handbook is a comprehensive compilation of examples of violent reactions, fires, and explosions due to unstable chemicals, as well as reports on known incompatibility between reactive chemicals.
8. Hazardous Chemicals Handbook, 2nd edition (Carson and Mumford, 2002). This book is geared toward an industrial audience. It provides basic information about chemical hazards and synthesizes technical guidance from a number of authorities in chemical safety. The chapters are organized by hazard (e.g., “Toxic Chemicals,” “Reactive Chemicals,” and “Cryogens”).
9. Sax’s Dangerous Properties of Industrial Materials, 11th edition, three volumes (Lewis, 2004). Also available on CD, this compilation of data for more than 26,000 chemical substances contains much of the information found in a typical MSDS, including physical and chemical properties; data on toxicity, flammability, reactivity, and explosivity; and a concise safety profile describing symptoms of exposure. It also contains immediately dangerous to life or health (IDLH) levels for approximately 1,000 chemicals, and for laboratory personnel it is a useful reference for checking the accuracy of an MSDS and a valuable resource in preparing a laboratory’s own LCSSs.
10. Patty’s Industrial Toxicology, 5th edition (Bingham et al., 2001). Also available on CD, this authoritative reference on the toxicology of different classes of organic and inorganic compounds focuses on health effects; hazards due to flammability, reactivity, and explosivity are not covered.
11. Proctor and Hughes’ Chemical Hazards of the Workplace, 5th edition (Hathaway and Proctor, 2004). This resource provides an excellent summary of the toxicology of more than 600 chemicals. Most entries are one to two pages and include signs and symptoms of exposure with reference to specific clinical reports.
12. Sittig’s Handbook of Toxic and Hazardous Chemicals and Carcinogens, 5th edition, two volumes (Pohanish, 2008). This very good reference, which is written with the industrial hygienists and first responder in mind, covers 2,100 substances.
13. Clinical Toxicology, 1st edition (Ford et al., 2001). This book is designed for clinicians and other health care providers. It describes the symptoms and treatment of poisoning from various sources.
14. Casarett and Doull’s Toxicology: The Basic Science of Poisons, 7th edition (Klaassen, 2007). This complete and readable overview of toxicology is a good textbook but is not arranged as a ready reference for handling laboratory emergencies.
15. Catalog of Teratogenic Agents, 11th edition (Shepard and Lemire, 2004). This catalog is one of the best references available on the subject of reproductive and developmental toxins.
16. Wiley Guide to Chemical Incompatibilities, 2nd edition (Pohanish and Greene, 2003). Simple-to-use reference listing the incompatibilities of more than 11,000 chemicals. Includes information about chemical incompatibility, conditions that favor undesirable reactions, and corrosivity data.
17. Occupational Health Guidelines for Chemical Hazards (HHS/CDC/NIOSH, 1981) and a supplement (HHS/CDC/NIOSH, 1995). The guidelines currently cover more than 400 substances and are based on the information assembled under the Standards Completion Program, which served as the basis for the promulgation of federal occupational health regulations (“substance-specific standards”). Typically five pages long and written clearly at a level readily understood by trained laboratory personnel, each set of guidelines includes information on physical, chemical, and toxicological properties; signs and symptoms of exposure; and considerable detail on control measures, medical surveillance practices, and emergency first-aid procedures. However, some guidelines date back to 1978 and may not be current, particularly with regard to chronic toxic effects. These guidelines are available on the NIOSH Web site (http://www.cdc.gov/niosh/).
A number of Web-based resources also exist. Some of these are NIOSH Databases and Information Resources (www.cdc.gov/niosh) and TOXNET through the National Library of Medicine (NLM; www.nlm.nih.gov).
4.B.7 Computer Services
In addition to computerized MSDSs, a number of computer databases are available that supply data for creating or supplementing MSDSs, for example, the NLM and Chemical Abstracts (CA) databases. These
and other such databases are accessible through various online computer data services; also, most of this information is available as CD and computer updates. Many of these services can be accessed for up-to-date toxicity information.
Governmental sources of EHS information include
• NIOSH (www.cdc.gov/niosh),
• OSHA (www.osha.gov),
• Environmental Protection Agency (EPA; www.epa.gov).
4.B.7.1 The National Library of Medicine Databases
The databases supplied by NLM are easy to use and free to access via the Web. TOXNET is an online collection of toxicological and environmental health databases. TOXLINE, for example, is an online database that accesses journals and other resources for current toxicological information on drugs and chemicals. It covers data published from 1900 to the present. Databases accessible through TOXNET include the Hazardous Substance Data Base (HSDB) Carcinogenic Potency Database (CPDB), the Developmental and Reproductive Toxicology Database (DART), the Genetic Toxicology Data Bank (GENE-TOX), the Integrated Risk Information System (IRIS), the Chemical Carcinogenesis Research Information System (CCRIS), and the International Toxicity Estimates for Risk (ITER). Other databases supplied by NLM that provide access to toxicological information are PubMed, which includes access to MEDLINE, PubChem, and ChemIDPlus. Free text searching is available on most of the databases.
4.B.7.2 Chemical Abstracts Databases
Another source of toxicity data is Chemical Abstracts Service (CAS). In addition to the NLM, several services provide CAS, including DIALOG, ORBIT, STN, and SciFinder. Searching procedures for CAS depend on the various services supplying the database. Searching costs are considerably higher than for NLM databases because CAS royalties must be paid. Telephone numbers for the above suppliers are as follows:
DIALOG, 800-334-2564;
Questel, 800-456-7248;
STN, 800-734-4227;
SciFinder, 800-753-4227.
Additional information can be found on the CAS Web site, www.cas.org.
Specialized databases also exist. One example is the ECOTOX database from EPA (www.epa.gov/ecotox). This database provides information on toxicity of chemicals to aquatic life, terrestrial plants, and wildlife.
Searching any database listed above is best done using the CAS registry number for the particular chemical.
4.B.7.3 Informal Forums
The “Letters to the Editor” column of Chemical & Engineering News (C&EN), published weekly by the American Chemical Society (ACS), was for many years an informal but widely accepted forum for reporting anecdotal information on chemical reactivity hazards and other safety-related information. Although less frequently updated, the ACS maintains an archive of all safety-related letters submitted to C&EN on the Web site of the Division of Chemical Health and Safety (CHAS) of ACS. CHAS also publishes the Journal of Chemical Health and Safety. Additional resources include the annual safety editorial called “Safety Notables: Information from the Literature” in the Organic Process Research and Development and community Listservs relating to laboratory safety.
4.B.8 Training
One important source of information for laboratory personnel is training sessions, and the critical place it holds in creating a safe environment should not be underestimated. Facts are only as useful as one’s ability to interpret and apply them to a given problem, and training provides context for their use. Hands-on, scenario-based training is ideal because it provides the participants with the chance to practice activities and behaviors in a safe way. Such training is especially useful for learning emergency response procedures. Another effective tool, particularly when trying to build awareness of a given safety concern, is case studies. Prior to beginning any laboratory activity, it is important to ensure that personnel have enough training to safely perform required tasks. If new equipment, materials, or techniques are to be used, a risk assessment should be performed, and any knowledge gaps should be filled before beginning work. (More information about training programs can be found in Chapter 2, section 2.G.)
4.C TOXIC EFFECTS OF LABORATORY CHEMICALS
4.C.1 Basic Principles
The chemicals encountered in the laboratory have a broad spectrum of physical, chemical, and toxicological properties and physiological effects. The risks associated with chemicals must be well understood
prior to their use in an experiment. The risk of toxic effects is related to both the extent of exposure and the inherent toxicity of a chemical. As discussed in detail below, extent of exposure is determined by the dose, the duration and frequency of exposure, and the route of exposure. Exposure to even large doses of chemicals with little inherent toxicity, such as phosphate buffer, presents low risk. In contrast, even small quantities of chemicals with high inherent toxicity or corrosivity may cause significant adverse effects. The duration and frequency of exposure are also critical factors in determining whether a chemical will produce harmful effects. A single exposure to some chemicals is sufficient to produce an adverse health effect; for other chemicals repeated exposure is required to produce toxic effects. For most substances, the route of exposure (through the skin, the eyes, the gastrointestinal tract, or the respiratory tract) is also an important consideration in risk assessment. For chemicals that are systemic toxicants, the internal dose to the target organ is a critical factor. Exposure to acute toxicants can be guided by well-defined toxicity parameters based on animal studies and often human exposure from accidental poisoning. The analogous quantitative data needed to make decisions about the neurotoxicity and immunogenicity of various chemicals is often unavailable.
When considering possible toxicity hazards while planning an experiment, recognizing that the combination of the toxic effects of two substances may be significantly greater than the toxic effect of either substance alone is important. Because most chemical reactions produce mixtures of substances with combined toxicities that have never been evaluated, it is prudent to assume that mixtures of different substances (i.e., chemical reaction mixtures) will be more toxic than their most toxic ingredient. Furthermore, chemical reactions involving two or more substances may form reaction products that are significantly more toxic than the starting reactants. This possibility of generating toxic reaction products may not be anticipated by trained laboratory personnel in cases where the reactants are mixed unintentionally. For example, inadvertent mixing of formaldehyde (a common tissue fixative) and hydrogen chloride results in the generation of bis(chloromethyl)ether, a potent human carcinogen.
All laboratory personnel must understand certain basic principles of toxicology and recognize the major classes of toxic and corrosive chemicals. The next sections of this chapter summarize the key concepts involved in assessing the risks associated with the use of toxic chemicals in the laboratory. (Also see Chapter 6, section 6.D.) Box 4.1 provides a quick guide for performing a toxicity-based risk assessment for laboratory chemicals.
4.C.1.1 Dose-Response Relationships
Toxicology is the study of the adverse effects of chemicals on living systems. The basic tenets of toxicology are that no substance is entirely safe and that all chemicals result in some toxic effects if a high enough amount (dose) of the substance comes in contact with a living system. As mentioned in Chapter 2, Paracelsus noted that the dose makes the poison and is perhaps the most important concept for all trained laboratory personnel to know. For example, water, a vital substance for life, results in death if a sufficiently large amount (i.e., gallons) is ingested at one time. On the other hand, sodium cyanide, a highly lethal chemical, produces no permanent (acute) effects if a living system is exposed to a sufficiently low dose. The single most important factor that determines whether a substance is harmful (or, conversely, safe) to an individual is the relationship between the amount (and concentration) of the chemical reaching the target organ, and the toxic effect it produces. For all chemicals, there is a range of concentrations that result in a graded effect between the extremes of no effect and death. In toxicology, this range is referred to as the dose-response relationship for the chemical. The dose is the amount of the chemical and the response is the effect of the chemical. This relationship is unique for each chemical, although for similar types of chemicals, the dose-response relationships are often similar. (See Figure 4.2.) Among the thousands of laboratory chemicals, a wide spectrum of doses exists that are required to produce toxic effects and even death. For most chemicals, a threshold dose has been established (by rule or by consensus) below which a chemical is not considered to be harmful to most individuals.
In these curves, dosage is plotted against the percent of the population affected by the dosage. Curve A represents a compound that has an effect on some percent of the population even at small doses. Curve B represents a compound that has an effect only above a dosage threshold.
Some chemicals (e.g., dioxin) produce death in laboratory animals exposed to microgram doses and therefore are extremely toxic. Other substances, however, have no harmful effects following doses in excess of several grams. One way to evaluate the acute toxicity (i.e., the toxicity occurring after a single exposure) of laboratory chemicals involves their lethal dose 50 (LD50) or lethal concentration 50 (LC50) value. The LD50 is defined as the amount of a chemical that when ingested, injected, or applied to the skin of a test animal under controlled laboratory conditions kills one-half (50%) of the animals. The LD50 is usually expressed in milligrams or grams per kilogram of body weight. For volatile chemicals (i.e., chemicals with sufficient
BOX 4.1
Quick Guide for Toxicity Risk Assessment of Chemicals
The following outline provides a summary of the steps discussed in this chapter that trained laboratory personnel should use to assess the risks of handling toxic chemicals. Note that if a laboratory chemical safety summary (LCSS) is not already available, this enables a worker to prepare his or her own LCSS.
1. Identify chemicals to be used and circumstances of use. Identify the chemicals involved in the proposed experiment and determine the amounts that will be used. Is the experiment to be done once, or will the chemicals be handled repeatedly? Will the experiment be conducted in an open laboratory, in an enclosed apparatus, or in a chemical fume hood? Is it possible that new or unknown substances will be generated in the experiment? Are any of the trained laboratory personnel involved in the experiment pregnant or likely to become pregnant? Do they have any known sensitivities to specific chemicals?
2. Consult sources of information. Consult an up-to-date LCSS for each chemical involved in the planned experiment or examine an up-to-date material safety data sheet (MSDS) if an LCSS is not available. In cases where substances with significant or unusual potential hazards are involved, consult more detailed references such as Patnaik (2007), Bingham et al. (200 1), and other sources discussed in section 4.B. Depending on the laboratory personnel’s level of experience and the degree of potential hazard associated with the proposed experiment, obtain the assistance of supervisors and safety professionals before proceeding with risk assessment.
3. Evaluate type of toxicity. Use the above sources of information to determine the type of toxicity associated with each chemical involved in the proposed experiment. Are any of the chemicals to be used acutely toxic or corrosive? Are any of the chemicals to be used irritants or sensitizers? Will any select carcinogens or possibly carcinogenic substances be encountered? Consult the listings of the resources described in section C.4.6 of this chapter to identify chemical similarities to known carcinogens. Are any chemicals involved in the proposed experiment suspected to be reproductive or developmental toxins or neurotoxins?
4. Consider possible routes of exposure. Determine the potential routes of exposure for each chemical. Are the chemicals gases, or are they volatile enough to present a significant risk of exposure through inhalation? If liquid, can the substances be absorbed through the skin? Is it possible that dusts or aerosols will be formed in the experiment? Does the experiment involve a significant risk of inadvertent ingestion or injection of chemicals?
5. Evaluate quantitative information on toxicity. Consult the information sources to determine the LD50, discussed in section 4.C.1.1 of this chapter, for each chemical via the relevant routes of exposure. Determine the acute toxicity hazard level for each substance, classifying each chemical as highly toxic, moderately toxic, slightly toxic, and so forth. For substances that pose inhalation hazards, take note of the threshold limit value-time weighted average (TLV-TWA), short-term exposure limit, and permissible exposure limit values. (See section 4.C.2.1)
6. Select appropriate procedures to minimize exposure. Use the basic prudent practices for handling chemicals, which are discussed in Chapter 6, section 6.C for all work with chemicals in the laboratory. In addition, determine whether any of the chemicals to be handled in the planned experiment meet the definition of a particularly hazardous substance due to high acute toxicity, carcinogenicity, and/or reproductive toxicity. If so, consider the total amount of the substance that will be used, the expected frequency of use, the chemical’s routes of exposure, and the circumstances of its use in the proposed experiment. As discussed in this chapter, use this information to determine whether it is appropriate to apply the additional procedures for work with highly toxic substances and whether additional consultation with safety professionals is warranted (see Chapter 6, section 6.D).
7. Prepare for contingencies. Note the signs and symptoms of exposure to the chemicals to be used in the proposed experiment. Note appropriate measures to be taken in the event of exposure or accidental release of any of the chemicals, including first aid or containment actions.
Note: See Box 4.2 for a quick guide for assessing the physical, flammable, explosive, and reactive hazards in the laboratory and Box 4.3 for a quick guide for assessing biological hazards in the laboratory.
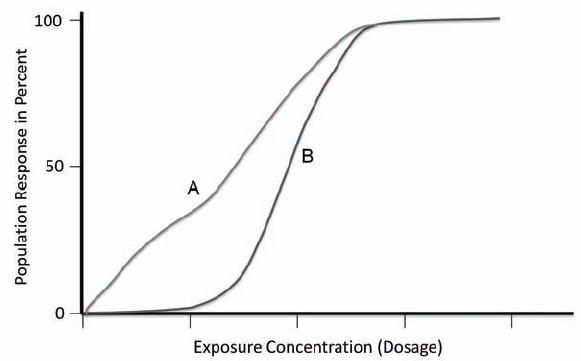
FIGURE 4.2 A simple representation of possible dose-response curves.
vapor pressure that inhalation is an important route of chemical entry into the body), the LC50 is often reported instead of the LD50. The LC50 is the concentration of the chemical in air that will kill 50% of the test animals exposed to it. The LC50 is given in parts per million, milligrams per liter, or milligrams per cubic meter. Also reported are LCLO and LDLO values, which are defined as the lowest concentration or dose that causes the death of test animals. In general, the larger the LD50 or LC50, the more chemical it takes to kill the test animals and, therefore, the lower the toxicity of the chemical. Although lethal dose values may vary among animal species and between animals and humans, chemicals that are highly toxic to animals are generally highly toxic to humans.
4.C.1.2 Duration and Frequency of Exposure
Toxic effects of chemicals occur after single (acute), intermittent (repeated), or long-term repeated (chronic) exposure. An acutely toxic substance causes damage as the result of a single short-duration exposure. Hydrogen cyanide, hydrogen sulfide, and nitrogen dioxide are examples of acute toxins. In contrast, a chronically toxic substance causes damage after repeated or long-duration exposure or causes damage that becomes evident only after a long latency period. Chronic toxins include all carcinogens, reproductive toxins, and certain heavy metals and their compounds. Many chronic toxins are extremely dangerous because of their long latency periods: the cumulative effect of low exposures to such substances may not become apparent for many years. Many chemicals may be hazardous both acutely and chronically depending on exposure level and duration.
In a general sense, the longer the duration of exposure, that is, the longer the body (or tissues in the body) is in contact with a chemical, the greater the opportunity for toxic effects to occur. Frequency of exposure also has an important influence on the nature and extent of toxicity. The total amount of a chemical required to produce a toxic effect is generally less for a single exposure than for intermittent or repeated exposures because many chemicals are eliminated from the body over time, because injuries are often repaired, and because tissues may adapt in response to repeated low-dose exposures. Some toxic effects occur only after long-term exposure because sufficient amounts of chemical cannot be attained in the tissue by a single exposure. Sometimes a chemical has to be present in a tissue for a considerable time to produce injury. For example, the neurotoxic and carcinogenic effects from exposure to heavy metals usually require long-term, repeated exposure.
The time between exposure to a chemical and onset of toxic effects varies depending on the chemical and the exposure. For example, the toxic effects of carbon monoxide, sodium cyanide, and carbon disulfide are evident within minutes. The chemical reaches the target organ rapidly and the organ responds rapidly. For many chemicals, the toxic effect is most severe between one and a few days after exposure. However, some chemicals produce delayed toxicity; in fact, the neurotoxicity produced by some chemicals is not observed until a few weeks after exposure. Delayed toxic effects are produced by chemical carcinogens and some organ toxins that produce progressive diseases such as pulmonary fibrosis and emphysema: in humans, it usually takes 10 to 30 years between exposure to a known human carcinogen and the detection of a tumor, and pulmonary fibrosis may take 10 or more years to result in symptoms.
4.C.1.3 Routes of Exposure
Exposure to chemicals in the laboratory occurs by several routes: (1) inhalation, (2) contact with skin or eyes, (3) ingestion, and (4) injection. Important features of these different pathways are detailed below.
4.C.1.3.1 Inhalation
Toxic materials that enter the body via inhalation include gases, the vapors of volatile liquids, mists and sprays of both volatile and nonvolatile liquid substances, and solid chemicals in the form of particles, fibers, and dusts. Inhalation of toxic gases and vapors produces poisoning by absorption through the mucous membranes of the mouth, throat, and lungs and also damages these tissues seriously by local action. Inhaled gases and vapors pass into the capillaries of the lungs and are carried into the circulatory system, where absorption is extremely rapid. Because of the large surface area of the lungs in humans (approximately 75 m2), they are the main site for absorption of many toxic materials.
The factors governing the absorption of gases and vapors from the respiratory tract differ significantly from those that govern the absorption of particulate substances. Factors controlling the absorption of inhaled gases and vapors include the solubility of the gas in body fluids and the reactivity of the gas with tissues and the fluid lining the respiratory tract. Gases or vapors that are highly water soluble, such as methanol, acetone, hydrogen chloride, and ammonia, dissolve predominantly in the lining of the nose and windpipe (trachea) and therefore tend to be absorbed from those regions. These sites of absorption are also potential sites of toxicity. Formaldehyde is an example of a reactive highly water-soluble vapor for which the nose is a major site of deposition. In contrast to water-soluble gases, reactive gases with low water solubility, such as ozone, phosgene, and nitrogen dioxide, penetrate farther into the respiratory tract and thus come into contact with the smaller tubes of the airways. Gases and vapors that are not water soluble but are more fat soluble, such as benzene, methylene chloride, and trichloroethylene, are not completely removed by interaction with the surfaces of the nose, trachea, and small airways. As a result, these gases penetrate the airways down into the deep lung, where they can diffuse across the thin alveoli lung tissue into the blood. The more soluble a gas is in the blood, the more it will be dissolved and transported to other organs.
For inhaled solid chemicals, an important factor in determining if and where a particle will be deposited in the respiratory tract is its size. One generalization is that the largest particles (>5 µm) are deposited primarily in the nose, smaller particles (1 to 5 µm) in the trachea and small airways, and the smallest particles in the alveoli region of the lungs. Thus, depending on the size of an inhaled particle, it will be deposited in different sections of the respiratory tract, and the location affects the local toxicity and the absorption of the material. In general, particles that are water soluble dissolve within minutes or days, and chemicals that are not water soluble but have a moderate degree of fat solubility also clear rapidly into the blood. Those that are not water soluble or highly fat soluble do not dissolve and are retained in the lungs for long periods of time. Metal oxides, asbestos, fiberglass, and silica are examples of water-insoluble inorganic particles that are retained in the lungs for years.
A number of factors affect the airborne concentrations of chemicals, but vapor pressure (the tendency of molecules to escape from the liquid or solid phase into the gaseous phase) is the most important characteristic. The higher the vapor pressure is, the greater the potential concentration of the chemical in the air. For example, acetone (with a vapor pressure of 180 mmHg at 20 °C) reaches an equilibrium concentration in air of 240,000 ppm, or approximately 24%. Fortunately, the ventilation system in most laboratories prevents an equilibrium concentration from developing in the breathing zone of laboratory personnel.
Even very low vapor pressure chemicals are dangerous if the material is highly toxic. A classic example is elemental mercury. Although the vapor pressure of mercury at room temperature is only 0.0012 mmHg, the resulting equilibrium concentration of mercury vapor is 1.58 ppm, or approximately 13 mg/m3. The TLV for mercury is 0.05 mg/m3, more than two orders of magnitude lower.
The vapor pressure of a chemical increases with temperature; therefore, heating solvents or reaction mixtures increases the potential for high airborne concentrations. Also, a spilled volatile chemical evaporates very quickly because of its large surface area, creating a significant exposure potential. Clearly, careful handling of volatile chemicals is very important; keeping containers tightly closed or covered and using volatiles in laboratory chemical hoods help avoid unnecessary exposure to inhaled chemicals.
Certain types of particulate materials also present potential for airborne exposure. If a material has a very low density or a very small particle size, it tends to remain airborne for a considerable time. For example, the very fine dust cloud generated by emptying a low-density particulate (e.g., vermiculite or nanomaterials) into a transfer vessel takes a long time to settle. Such operations should therefore be carried out in a laboratory chemical hood or in a glovebox.
Operations that generate aerosols (suspensions of microscopic droplets in air), such as vigorous boiling,
high-speed blending, or bubbling gas through a liquid, increase the potential for exposure via inhalation. Consequently, these and other such operations on toxic chemicals should also be carried out in a laboratory chemical hood.
4.C.1.3.2 Contact with Skin or Eyes
Chemical contact with the skin is a frequent mode of injury in the laboratory. Many chemicals injure the skin directly by causing skin irritation and allergic skin reactions. Corrosive chemicals cause severe burns. In addition to causing local toxic effects, many chemicals are absorbed through the skin in sufficient quantity to produce systemic toxicity. The main avenues by which chemicals enter the body through the skin are the hair follicles, sebaceous glands, sweat glands, and cuts or abrasions of the outer layer. Absorption of chemicals through the skin depends on a number of factors, including chemical concentration, chemical reactivity, and the solubility of the chemical in fat and water. Absorption is also dependent on the condition of the skin, the part of the body exposed, and duration of contact. Differences in skin structure affect the degree to which chemicals are absorbed. In general, toxicants cross membranes and thin skin (e.g., scrotum) much more easily than thick skin (e.g., palms). Although an acid burn on the skin is felt immediately, an alkaline burn takes time to be felt and its damage goes deeper than the acid. When skin is damaged, penetration of chemicals increases. Acids and alkalis injure the skin and increase its permeability. Burns and skin diseases are the most common examples of skin damage that increase penetration. Also, hydrated skin absorbs chemicals better than dehydrated skin. Some chemicals such as dimethyl sulfoxide actually increase the penetration of other chemicals through the skin by increasing its permeability.
Contact of chemicals with the eyes is of particular concern because the eyes are sensitive to irritants. Few substances are innocuous in contact with the eyes; most are painful and irritating, and a considerable number are capable of causing burns and loss of vision. Alkaline materials, phenols, and acids are particularly corrosive and can cause permanent loss of vision. Because the eyes contain many blood vessels, they also are a route for the rapid absorption of many chemicals.
4.C.1.3.3 Ingestion
Many of the chemicals used in the laboratory are extremely hazardous if they enter the mouth and are swallowed. The gastrointestinal tract, which consists of the mouth, esophagus, stomach, and small and large intestines, can be thought of as a tube of variable diameter (approximately 5 m long) with a large surface area (approximately 200 m2) for absorption. Toxicants that enter the gastrointestinal tract must be absorbed into the blood to produce a systemic injury, although some chemicals are caustic or irritating to the gastrointestinal tract tissue itself. Absorption of toxicants takes place along the entire gastrointestinal tract, even in the mouth, and depends on many factors, including the physical properties of the chemical and the speed at which it dissolves. Absorption increases with surface area, permeability, and residence time in various segments of the tract. Some chemicals increase intestinal permeability and thus increase the rate of absorption. More chemical will be absorbed if the chemical remains in the intestine for a long time. If a chemical is in a relatively insoluble solid form, it will have limited contact with gastrointestinal tissue, and its rate of absorption will be low. If it is an organic acid or base, it will be absorbed in that part of the gastrointestinal tract where it is most fat soluble. Fat-soluble chemicals are absorbed more rapidly and extensively than water-soluble chemicals.
4.C.1.3.4 Injection
Exposure to toxic chemicals by injection does not occur frequently in the laboratory, but it occurs inadvertently through mechanical injury from sharp objects such as glass or metal contaminated with chemicals or syringes used for handling chemicals. The intravenous route of administration is especially dangerous because it introduces the toxicant directly into the bloodstream, eliminating the process of absorption. Nonlaboratory personnel, such as custodial workers or waste handlers, must be protected from exposure by placing sharp objects in special trash containers and not ordinary scrap baskets. Hypodermic needles with blunt ends are available for laboratory use.
4.C.2 Assessing Risks of Exposure to Toxic Laboratory Chemicals
Exposure to a harmful chemical results in local toxic effects, systemic toxic effects, or both. Local effects involve injury at the site of first contact; the eyes, the skin, the nose and lungs, and the digestive tract are typical sites of local reactions. Examples of local effects include (1) inhalation of hazardous materials causing toxic effects in the nose and lungs; (2) contact with harmful materials on the skin or eyes leading to effects ranging from mild irritation to severe tissue damage; and (3) ingestion of caustic substances causing burns and ulcers in the mouth, esophagus, stomach, and intestines. Systemic effects, by contrast, occur after the toxicant has been absorbed from the site of contact into the bloodstream and distributed throughout the body. Some chemicals produce adverse effects on all tissues of the body, but others tend to selectively injure a par-
ticular tissue or organ without affecting others. The affected organ (e.g., liver, lungs, kidney, and central nervous system) is referred to as the target organ of toxicity, although it is not necessarily the organ where the highest concentration of the chemical is found. Hundreds of systemic toxic effects of chemicals are known; they result from single (acute) exposures or from repeated or long-duration (chronic) exposures that become evident only after a long latency period.
Toxic effects are classified as either reversible or irreversible. Reversible toxicity is possible when tissues have the capacity to repair toxic damage, and the damage disappears after cessation of exposure. Irreversible damage, in contrast, persists after cessation of exposure. Recovery from a burn is a good example of reversible toxicity; cancer is considered irreversible, although appropriate treatment may reduce the effects in this case.
Laboratory chemicals are grouped into several classes of toxic substances, and many chemicals display more than one type of toxicity. The first step in assessing the risks associated with a planned laboratory experiment involves identifying which chemicals in the proposed experiment are potentially hazardous substances. The OSHA Laboratory Standard (29 CFR § 1910.1450) defines a hazardous substance as a chemical for which there is statistically significant evidence based on at least one study conducted in accordance with established scientific principles that acute or chronic health effects may occur in exposed employees. The term “health hazard” includes chemicals that are carcinogens, toxic or highly toxic agents, reproductive toxins, irritants, corrosives, sensitizers, hepatotoxins, nephrotoxins, neurotoxins, agents that act on the hematopoietic systems, and agents that damage the lungs, skin, eyes, or mucous membranes.
The OSHA Laboratory Standard further requires that certain chemicals be identified as particularly hazardous substances (commonly known as PHSs) and handled using special additional procedures. PHSs include chemicals that are select carcinogens (those strongly implicated as a potential cause of cancer in humans), reproductive toxins, and compounds with a high degree of acute toxicity. When working with these substances for the first time, it is prudent to consult with a safety professional prior to beginning work. This will provide a second set of trained eyes to review the safety protocols in place and will help ensure that any special emergency response requirements can be met in the event of exposure of personnel to the material or accidental release.
Highly flammable and explosive substances make up another category of hazardous compounds, and the assessment of risk for these classes of chemicals is discussed in section 4.D. This section considers the assessment of risks associated with specific classes of toxic chemicals, including those that pose hazards due to acute toxicity and chronic toxicity.
The following are the most common classes of toxic substances encountered in laboratories.
4.C.2.1 Acute Toxicants
Acute toxicity is the ability of a chemical to cause a harmful effect after a single exposure. Acutely toxic agents cause local toxic effects, systemic toxic effects, or both, and this class of toxicants includes corrosive chemicals, irritants, and allergens (sensitizers).
In assessing the risks associated with acute toxicants, it is useful to classify a substance according to the acute toxicity hazard level as shown in Table 4.1. LD50 values can be found in the LCSS or MSDS for a given substance, and in references such as Sax’s Dangerous Properties of Industrial Materials (Lewis, 2004), A Comprehensive Guide to the Hazardous Properties of Chemical Substances, 3rd Edition (Patnaik, 2007), and the Registry of Toxic Effects of Chemical Substances (RTECS) (NIOSH). Table 4.2 relates test animal LD50 values expressed as milligrams or grams per kilogram of body weight to the probable human lethal dose, expressed in easily understood units, for a 70-kg person.
Special attention is given to any substance classified according to the above criteria as having a high level of acute toxicity hazard. Chemicals with a high level of acute toxicity make up one of the categories of PHSs defined by the OSHA Laboratory Standard. Any compound rated as highly toxic in Table 4.1 meets the OSHA criteria for handling as a PHS.
Table 4.3 lists some of the most common chemicals with a high level of acute toxicity that are encountered in the laboratory. These compounds are handled using the additional procedures outlined in Chapter 6,
TABLE 4.1 Acute Toxicity Hazard Level
| Hazard Level | Toxicity Rating | Oral LD50 (rats, per kg) | Skin Contact LD50 (rabbits, per kg) | Inhalation LC50 (rats, ppm for 1 h) | Inhalation LC50 (rats, mg/m3 for 1 h) |
| High | Highly toxic | <50 mg | <200 mg | <200 | <2,000 |
| Medium | Moderately toxic | 50 to 500 mg | 200 mg to 1 g | 200 to 2,000 | 2,000 to 20,000 |
| Low | Slightly toxic | 500 mg to 5 g | 1 to 5 g | 2,000 to 20,000 | 20,000 to 200,000 |
TABLE 4.2 Probable Lethal Dose for Humans
|
|
||
|
Toxicity Rating |
Animal LD50(per kg) |
Lethal Dose When Ingested by 70-kg (150-lb) Human |
|
|
||
|
Extremely toxic |
<5 mg |
A taste (<7 drops) |
|
Highly toxic |
5 to 50 mg |
Between 7 drops and 1 tsp |
|
Moderately toxic |
50 to 500 mg |
Between 1 tsp and 1 oz |
|
Slightly toxic |
500 mg to 5 g |
Between 1 oz and 1 pint |
|
Practically nontoxic |
>5 g |
>1 pint |
|
|
||
SOURCE: Modified, by permission, from Gosselin et al. (1984); reprinted by permission from Lippincott Williams and Wilkins, http://lww.com.
section 6.D. In some circumstances, all these special precautions may not be necessary, such as when the total amount of an acutely toxic substance is a small fraction of the harmful dose. An essential part of prudent experiment planning is to determine whether a chemical with a high degree of acute toxicity should be treated as a PHS in the context of a specific planned use. This determination not only involves consideration of the total amount of the substance to be used but also requires a review of the physical properties of the substance (e.g., Is it volatile? Does it tend to form dusts?), its potential routes of exposure (e.g., Is it readily absorbed through the skin?), and the circumstances of its use in the proposed experiment (e.g., Will the substance be heated? Is there likelihood that aerosols may be generated?). Depending on the laboratory personnel’s level of experience and the degree of potential hazard, this determination may require consultation with supervisors and safety professionals.
Because the greatest risk of exposure to many laboratory chemicals is by inhalation, trained laboratory personnel must understand the use of exposure limits that have been established by agencies such as OSHA and NIOSH and by an organization such as ACGIH.
The TLV assigned by the ACGIH, defines the concentration of a chemical in air to which nearly all individuals can be exposed without adverse effects. These limits reflect a view of an informed scientific community and are not legal standards. They are designed to be an aid to industrial hygienists. The TLV time-weighted average (TWA) refers to the concentration safe for exposure during an entire 8-hour workday; the TLV-STEL is a higher concentration to which workers may be exposed safely for a 15-minute period up to four times during an 8-hour shift and at least 60 minutes between these periods. TLVs are intended for use by professionals after they have read and understood the documentation of the TLV for the chemical or physical agent under study.
TABLE 4.3 Examples of Compounds with a High Level of Acute Toxicity
|
|
|
|
Acrolein |
Methyl fluorosulfonate |
|
Arsine |
Nickel carbonyl |
|
Chlorine |
Nitrogen dioxide |
|
Diazomethane |
Osmium tetroxide |
|
Diborane (gas) |
Ozone |
|
Dimethyl mercury |
Phosgene |
|
Hydrogen cyanide |
Sodium azide |
|
Hydrogen fluoride |
Sodium cyanide (and other cyanide salts) |
|
|
|
OSHA defines the permissible exposure limit (PEL) analogously to the ACGIH values, with corresponding 8-hour TWA and ceiling limits based on either a 15-minute TWA or an instantaneous reading, whichever is possible. In some cases, OSHA also defines a maximum peak concentration that cannot be exceeded beyond a given duration. Compliance with PELs is required, and the limits are enforceable by OSHA. PEL values allow trained laboratory personnel to quickly determine the relative inhalation hazards of chemicals. In general, substances with 8-hour TWA PELs of less than 50 ppm should be handled in a laboratory chemical hood. Comparison of these values to the odor threshold for a given substance often indicates whether the odor of the chemical provides sufficient warning of possible hazard. However, individual differences in ability to detect some odors as well as anosmia for ethylene oxide or olfactory fatigue for hydrogen sulfide can limit the usefulness of odors as warning signs of overexposure. LCSSs contain information on odor threshold ranges and whether a substance is known to cause olfactory fatigue.
Recommended exposure limits (RELs) are occupational exposure limits recommended by NIOSH to protect the health and safety of individuals over a working lifetime. Compliance with RELs is not required by law. RELs may also be expressed as a ceiling limit that should never be exceeded over a given time period, but the limit is usually expressed as a TWA exposure for up to 10 hours per day during a 40-hour workweek. As with TLVs, RELs are also expressed as STELs. One should not exceed the STEL for longer than 15 minutes at anytime throughout a workday.
A variety of devices are available for measuring the concentration of chemicals in laboratory air, so that the degree of hazard associated with the use of a chemical is assessed directly. Industrial hygiene offices of many institutions assist trained laboratory personnel in measuring the air concentrations of chemicals.
4.C.3 Types of Toxins
4.C.3.1 Irritants, Corrosive Substances, Allergens, and Sensitizers
Lethal dose and other quantitative toxicological parameters generally provide little guidance in assessing the risks associated with corrosives, irritants, allergens, and sensitizers because these toxic substances exert
their harmful effects locally. It would be very useful for the chemical research community if a quantitative measure for such effects were developed. When planning an experiment that involves corrosive substances, basic prudent handling practices should be reviewed to ensure that the skin, face, and eyes are protected adequately by the proper choice of corrosion-resistant gloves and protective clothing and eyewear, including, in some cases, face shields. Similarly, LD50 and LC50 data are not indicators of the irritant effects of chemicals, and therefore special attention should be paid to the identification of irritant chemicals by consulting LCSSs, MSDSs, and other sources of information. Allergens and sensitizers are another class of acute toxicants with effects that are not included in LD50 or LC50 data.
4.C.3.1.1 Irritants
Irritants are noncorrosive chemicals that cause reversible inflammatory effects (swelling and redness) on living tissue by chemical action at the site of contact. A wide variety of organic and inorganic chemicals are irritants, and consequently, skin and eye contact with all reagent chemicals in the laboratory should be minimized. Examples include formaldehyde, iodine, and benzoyl chloride.
4.C.3.1.2 Corrosive Substances
Corrosive substances are those that cause destruction of living tissue by chemical action at the site of contact and are solids, liquids, or gases. Corrosive effects occur not only on the skin and eyes but also in the respiratory tract and, in the case of ingestion, in the gastrointestinal tract as well. Corrosive materials are probably the most common toxic substances encountered in the laboratory. Corrosive liquids are especially dangerous because their effect on tissue is rapid. Bromine, sulfuric acid, aqueous sodium hydroxide solution, and hydrogen peroxide are examples of highly corrosive liquids. Corrosive gases are also frequently encountered. Gases such as chlorine, ammonia, chloramine, and nitrogen dioxide damage the lining of the lungs, leading, after a delay of several hours, to the fatal buildup of fluid known as pulmonary edema. Finally, a number of solid chemicals have corrosive effects on living tissue. Examples of common corrosive solids include sodium hydroxide, phosphorus, and phenol. If dust from corrosive solids is inhaled, it causes serious damage to the respiratory tract.
There are several major classes of corrosive substances. Strong acids such as nitric, sulfuric, and hydrochloric acid cause serious damage to the skin and eyes. Hydrofluoric acid is particularly dangerous and produces slow-healing painful burns (see Chapter 6, section 6.G.6). Strong bases, such as metal hydroxides and ammonia, are another class of corrosive chemicals. Strong dehydrating agents, such as phosphorus pentoxide and calcium oxide, have a powerful affinity for water and cause serious burns on contact with the skin. Finally, strong oxidizing agents, such as concentrated solutions of hydrogen peroxide, also have serious corrosive effects and should never come into contact with the skin or eyes.
4.C.3.1.3 Allergens and Sensitizers
A chemical allergy is an adverse reaction by the immune system to a chemical. Such allergic reactions result from previous sensitization to that chemical or a structurally similar chemical. Once sensitization occurs, allergic reactions result from exposure to extremely low doses of the chemical. Some allergic reactions are immediate, occurring within a few minutes after exposure. Anaphylactic shock is a severe immediate allergic reaction that results in death if not treated quickly. Delayed allergic reactions take hours or even days to develop, the skin is the usual site of such delayed reactions, becoming red, swollen, and itchy. Delayed chemical allergy occurs even after the chemical has been removed; contact with poison ivy is a familiar example of an exposure that causes a delayed allergic reaction due to uroshiol. Also, just as people vary widely in their susceptibility to sensitization by environmental allergens such as dust and pollen, individuals also exhibit wide differences in their sensitivity to laboratory chemicals.
Because individuals differ widely in their tendency to become sensitized to allergens, compounds with a proven ability to cause sensitization should be classified as highly toxic agents within the institution’s CHP. When working with chemicals known to cause allergic sensitization, follow institutional policy on handling and containment of allergens and highly toxic agents. Once a person has become sensitized to an allergen, subsequent contact often leads to immediate or delayed allergic reactions.
Because an allergic response is triggered in a sensitized individual by an extremely small quantity of the allergen, it may occur despite personal protection measures that are adequate to protect against the acute effects of chemicals. Laboratory personnel should be alert for signs of allergic responses to chemicals. Examples of chemical substances that cause allergic reactions in some individuals include diazomethane; dicyclohexylcarbodiimide; formaldehyde and phenol derivatives; various isocyanates (e.g., methylene diphenyl diisocyanate (MDI) or toluene diisocyanate (TDI), used in adhesives, elastomers, and coatings); benzylic and allylic halides; metals including nickel, beryllium, platinum, cobalt, tin, and chromium; and acid anhydrides such as acetic anhydrides.
4.C.3.2 Asphyxiants
Asphyxiants are substances that interfere with the transport of an adequate supply of oxygen to vital organs of the body. The brain is the organ most easily affected by oxygen starvation, and exposure to asphyxiants leads to rapid collapse and death. Simple asphyxiants are substances that displace oxygen from the air being breathed to such an extent that adverse effects result. Acetylene, carbon dioxide, argon, helium, ethane, nitrogen, and methane are common asphyxiants. Certain other chemicals have the ability to combine with hemoglobin, thus reducing the capacity of the blood to transport oxygen. Carbon monoxide, hydrogen cyanide, and certain organic and inorganic cyanides are examples of such substances.
4.C.3.3 Neurotoxins
Neurotoxic chemicals induce an adverse effect on the structure or function of the central or peripheral nervous system, which can be permanent or reversible. The detection of neurotoxic effects may require specialized laboratory techniques, but often they are inferred from behavior such as slurred speech and staggered gait. Many neurotoxins are chronically toxic substances with adverse effects that are not immediately apparent. Some chemical neurotoxins that may be found in the laboratory are mercury (inorganic and organic), organophosphate pesticides, carbon disulfide, xylene, tricholoroethylene, and n-hexane. (For information about reducing the presence of mercury in laboratories, see Chapter 5, section 5.B.8.)
4.C.3.4 Reproductive and Developmental Toxins
Reproductive toxins are defined by the OSHA Laboratory Standard as substances that cause chromosomal damage (mutagens) and substances with lethal or teratogenic (malformation) effects on fetuses. These substances have adverse effects on various aspects of reproduction, including fertility, gestation, lactation, and general reproductive performance, and can affect both men and women. Many reproductive toxins are chronic toxins that cause damage after repeated or long-duration exposures with effects that become evident only after long latency periods. Developmental toxins act during pregnancy and cause adverse effects on the fetus; these effects include embryo lethality (death of the fertilized egg, embryo, or fetus), teratogenic effects, and postnatal functional defects. Male reproductive toxins in some cases lead to sterility.
When a pregnant woman is exposed to a chemical, generally the fetus is exposed as well because the placenta is an extremely poor barrier to chemicals. Embryotoxins have the greatest impact during the first trimester of pregnancy. Because a woman often does not know that she is pregnant during this period of high susceptibility, women of childbearing potential are advised to be especially cautious when working with chemicals, especially those rapidly absorbed through the skin (e.g., formamide). Pregnant women and women intending to become pregnant should seek advice from knowledgeable sources before working with substances that are suspected to be reproductive toxins. As minimal precautions, the general procedures outlined in Chapter 6, section 6.D, should be followed, though in some cases it will be appropriate to handle the compounds as PHSs.
For example, among the numerous reproductive hazards to female laboratory scientists, gestational exposure to organic solvents should be of concern (HHS/CDC/NIOSH, 1999; Khattak et al., 1999). Some common solvents in high doses have been shown to be teratogenic in laboratory animals, resulting in developmental defects. Although retrospective studies of the teratogenic risk in women of childbearing age of occupational exposure to common solvents have reached mixed conclusions, at least one such study of exposure during pregnancy to multiple solvents detected increased fetal malformations. Thus, inhalation exposure to organic solvents should be minimized during pregnancy. Also, exposure to lead or to anticancer drugs, such as methotrexate, or to ionizing radiation can cause infertility, miscarriage, birth defects, and low birth weight. Certain ethylene glycol ethers such as 2-ethoxyethanol and 2-methoxyethanol can cause miscarriages. Carbon disulfide can cause menstrual cycle changes. One cannot assume that any given substance is safe if no data on gestational exposure are available.
Specific hazards of chemical exposure are associated with the male reproductive system, including suppression of sperm production and survival, alteration in sperm shape and motility, and changes in sexual drive and performance. Various reproductive hazards have been noted in males following exposure to halogenated hydrocarbons, nitro aromatics, arylamines, ethylene glycol derivatives, mercury, bromine, carbon disulfide, and other chemical reagents (HHS/CDC/NIOSH, 1996).
Information on reproductive toxins can be obtained from LCSSs, MSDSs, and by consulting safety professionals in the environmental safety department, industrial hygiene office, or medical department. Literature sources of information on reproductive and developmental toxins include the Catalog of Teratogenic Agents (Shepard and Lemire, 2007), Reproductively Active Chemicals: A Reference Guide (Lewis, 1991), and “What Every Chemist Should Know About Teratogens” in the Journal of Chemical Education (Beyler and Meyers, 1982). The State of California maintains a list of chemicals it
considers reproductive toxins, and additional information can be found through the NLM TOXNET system. The study of reproductive toxins is an active area of research, and laboratory personnel should consult resources that are updated regularly for information.
4.C.3.5 Toxins Affecting Other Target Organs
Target organs outside the reproductive and neurological systems are also affected by toxic substances in the laboratory. Most of the chlorinated hydrocarbons, benzene, other aromatic hydrocarbons, some metals, carbon monoxide, and cyanides, among others, produce one or more effects in target organs. Such an effect may be the most probable result of exposure to the particular chemical. Although this chapter does not include specific sections on liver, kidney, lung, or blood toxins, many of the LCSSs mention those effects in the toxicology section.
4.C.3.6 Carcinogens
A carcinogen is a substance capable of causing cancer. Cancer, in the simplest sense, is the uncontrolled growth of cells and can occur in any organ. The mechanism by which cancer develops is not well understood, but the current thinking is that some chemicals interact directly with DNA, the genetic material in all cells, to result in permanent alterations. Other chemical carcinogens modify DNA indirectly by changing the way cells grow. Carcinogens are chronically toxic substances; that is, they cause damage after repeated or long-duration exposure, and their effects may become evident only after a long latency period. Carcinogens are particularly insidious toxins because they may have no immediate apparent harmful effects.
Because cancer is a widespread cause of human mortality, and because exposure to chemicals may play a significant role in the onset of cancer, a great deal of attention has been focused on evaluation of the carcinogenic potential of chemicals. However, a vast majority of substances involved in research, especially in laboratories concerned primarily with the synthesis of novel compounds, have not been tested for carcinogenicity. Compounds that are known to pose the greatest carcinogenic hazard are referred to as select carcinogens, and they constitute another category of substances that must be handled as PHSs according to the OSHA Laboratory Standard. A select carcinogen is defined in the OSHA Laboratory Standard as a substance that meets one of the following criteria:
1. It is regulated by OSHA as a carcinogen.
2. It is listed as known to be a carcinogen in the latest Annual Report on Carcinogens issued by the National Toxicology Program (NTP) (HHS/CDC/NTP, 2005).
3. It is listed under Group 1 (carcinogenic to humans) by the International Agency for Research on Cancer (IARC).
4. It is listed under IARC Group 2A (probably carcinogenic to humans) or 2B (possibly carcinogenic to humans), or under the category “reasonably anticipated to be a carcinogen by the NTP,” and causes statistically significant tumor incidence in experimental animals in accordance with any of the following criteria: (a) after inhalation exposure of 6 to 7 hours per day, 5 days per week, for a significant portion of a lifetime to dosages of less than 10 mg/m3; (b) after repeated skin application of less than 300 mg/kg of body weight per week; or (c) after oral dosages of less than 50 mg/kg of body weight per day.
Chemicals that meet the criteria of a select carcinogen are classified as PHSs and should be handled using the basic prudent practices given in Chapter 6, section 6.C, supplemented by the additional special practices outlined in Chapter 6, section 6.D. Work with compounds that are possible human carcinogens may or may not require the additional precautions given in section 6.D. For these compounds, the LCSS should indicate whether the substance meets the additional criteria listed in category 4 and must therefore be treated as a select carcinogen. If an LCSS is not available, consultation with a safety professional such as a CHO may be necessary to determine whether a substance should be classified as PHS. Lists of known human carcinogens and compounds that are “reasonably anticipated to be carcinogens” based on animal tests can be found in the 11th Report on Carcinogens (HHS/CDC/NTP, 2005). This report is updated periodically. Check the NTP Web site (ntp.niehs.nih.gov) for the most recent edition. Additional information can be found on the OSHA and IARC Web sites (www.osha.gov and www.iarc.fr).
In the laboratory many chemical substances are encountered for which there is no animal test or human epidemiological data on carcinogenicity. In these cases, trained laboratory personnel must evaluate the potential risk that the chemical in question is a carcinogenic substance. This determination is sometimes made on the basis of knowledge of the specific classes of compounds and functional group types that have previously been correlated with carcinogenic activity. For example, chloromethyl methyl ether is a known human carcinogen and therefore is regarded as an OSHA select carcinogen requiring the handling procedures outlined in section 6.D. On the other hand, the carcinogenicity of ethyl chloromethyl ether and certain other alkyl chloromethyl ethers is not established, and
these substances do not necessarily have to be treated as select carcinogens. However, because of the chemical similarity of these compounds to chloromethyl methyl ether, these substances may have comparable carcinogenicity, and it is prudent to regard them as select carcinogens requiring the special handling procedures outlined in section 6.D.
Whether a suspected carcinogenic chemical is treated as a PHS in the context of a specific laboratory use is affected by the scale and circumstances associated with the intended experiment. Trained laboratory personnel must decide whether the amount and frequency of use, as well as other circumstances, require additional precautions beyond the basic prudent practices of section 6.C. For example, the large-scale or recurring use of such a chemical might suggest that the special precautions of section 6.D be followed to control exposure, whereas adequate protection from a single use of a small amount of such a substance may be obtained through the use of the basic procedures in section 6.C.
When evaluating the carcinogenic potential of chemicals, note that exposure to certain combinations of compounds (not necessarily simultaneously) causes cancer even at exposure levels where neither of the individual compounds would have been carcinogenic. 1,8,9-Trihydroxyanthracene and certain phorbol esters are examples of tumor promoters that are not carcinogenic themselves but dramatically amplify the carcinogenicity of other compounds. Understand that the response of an organism to a toxicant typically increases with the dose given, but the relationship is not always a linear one. Some carcinogenic alkylating agents exhibit a dose threshold above which the tendency to cause mutations increases markedly. At lower doses, natural protective systems prevent genetic damage, but when the capacity of these systems is overwhelmed, the organism becomes much more sensitive to the toxicant. However, individuals have differences in the levels of protection against genetic damage as well as in other defense systems. These differences are determined in part by genetic factors and in part by the aggregate exposure of the individual to all chemicals within and outside the laboratory.
4.C.3.7 Control Banding
Control banding is a qualitative risk assessment and management approach to assist in determining the appropriate handling of materials without occupational exposure limits (OELs) and to minimize the exposure of personnel to hazardous material.2 It is not intended to be a replacement for OELs but as an additional tool. The system uses a range of exposure and hazard “bands” that, when mapped for a given material and application, help the user determine the appropriate safety controls that should be in place. The approach is built on two major premises: (1) there are a limited number of control approaches and (2) that many problems have been encountered and solved before. Control banding uses the solutions that experts have developed previously to control occupational chemical exposures and applies those solutions to other tasks with similar exposure concerns.
By considering the physical and chemical characteristics and hazards posed by the material (e.g., toxicity), the quantity used, the intended use or application, and the mode of exposure (e.g., inhalation), a graduated scale of controls can be applied, from general ventilation requirements to requiring containment of the material to recommending that the user seek expert advice. Because this approach is expected to provide simplified guidance for assessing hazards and applying controls, it is anticipated that control banding will have utility for small- and medium-size nonchemical businesses; however, larger companies may also find it useful for prioritizing chemical hazards and hazard communication.
Note that a number of control banding models exist, each with its own level of complexity and applicability to a variety of scenarios. Within the United States, questions about the utility of control banding for workplaces initiated a review by NIOSH on the critical issues and potential applications of the system. The resulting report, Qualitative Risk Characterization and Management of Occupational Hazards: Control Banding (CB) (HHS/CDC/NIOSH, 2009b), can be found on the NIOSH Web site. It provides an overview of the major concepts and methodologies and presents a critical analysis of control banding.
Control banding is of interest internationally, and variations on the methodology can be found in many countries. More information about control banding can be found by consulting these Web sites and articles.
• (UK Health and Safety Executive) Control of Substances Hazardous to Health Regulations, www.coshh-essentials.org.uk/
• ILO Programme on Safety and Health at Work and the Environment (SafeWork), www.ilo.org/
• NIOSH, www.cdc.gov/niosh/
• “Training Health and Safety Committees to Use Control Banding: Lessons Learned and Opportunities for the United States” (Bracker et al., 2009)
• “Evaluation of COSHH Essentials: Methylene Chloride, Isopropanol, and Acetone Exposures in a Small Printing Plant” (Lee et al., 2009)
_______________
2For information on how OELs are determined, see Alaimo (2001).
• “Application of a Pilot Control Banding Tool for Risk Level Assessment and Control of Nanoparticle Exposures” (Paik et al., 2008)
• “‘Stoffenmanager,’ a Web-Based Control Banding Tool Using an Exposure Process Model” (Marquart et al., 2008)
• “History and Evolution of Control Banding: A Review” (Zalk and Nelson, 2008)
• “Control Banding: Issues and Opportunities.” A Report of the ACGIH Exposure Control Banding Task Force (ACGIH, 2008a)
• “Evaluation of the Control Banding Method—Comparison with Measurement-Based Comprehensive Risk Assessment” (Hashimoto et al., 2007)
• Guidance for Conducting Control Banding Analyses (American Industrial Hygiene Association, 2008)
4.D FLAMMABLE, REACTIVE, AND EXPLOSIVE HAZARDS
In addition to the hazards due to the toxic effects of chemicals, hazards due to flammability, explosivity, and reactivity need to be considered in risk assessment. These hazards are described in detail in the following sections. Further information can be found in Bretherick’s Handbook of Reactive Chemical Hazards (Urben, 2007), an extensive compendium that is the basis for lists of incompatible chemicals included in other reference works. The handbook describes computational protocols that consider thermodynamic and kinetic parameters of a system to arrive at quantitative measures such as the reaction hazard index. Reactive hazards arise when the release of energy from a chemical reaction occurs in quantities or at rates too great for the energy to be absorbed by the immediate environment of the reacting system, and material damage results. An additional resource is the Hazardous Chemical Handbook (Carson and Mumford, 2002). The book is geared toward an industrial audience and contains basic descriptions of chemical hazards along with technical guidance.
Box 4.2 is a quick guide for assisting in the assessment of the physical, flammable, explosive, and reactive hazards in the laboratory.
4.D.1 Flammable Hazards
4.D.1.1 Flammable Substances
Flammable substances, those that readily catch fire and burn in air, may be solid, liquid, or gaseous. The most common fire hazard in the laboratory is a flammable liquid or the vapor produced from such a liquid. An additional hazard is that a compound can enflame so rapidly that it produces an explosion. Proper use of substances that cause fire requires knowledge of their tendencies to vaporize, ignite, or burn under the variety of conditions in the laboratory.
For a fire to occur, three conditions must exist simultaneously: an atmosphere containing oxygen, usually air; a fuel, such as a concentration of flammable gas or vapor that is within the flammable limits of the substance; and a source of ignition (see Figure 4.3). Prevention of the coexistence of flammable vapors and an ignition source is the optimal way to deal with the hazard. When the vapors of a flammable liquid cannot always be controlled, strict control of ignition sources is the principal approach to reduce the risk of flammability. The rates at which different liquids produce flammable vapors depend on their vapor pressures, which increase with increasing temperature. The degree of fire hazard of a substance depends also on its ability to form combustible or explosive mixtures with air and on the ease of ignition of these mixtures. Also important are the relative density and solubility of a liquid with respect to water and of a gas with respect to air. These characteristics can be evaluated and compared in terms of the following specific properties.
4.D.1.2 Flammability Characteristics
4.D.1.2.1 Flash Point
The flash point is the lowest temperature at which a liquid has a sufficient vapor pressure to form an ignitable mixture with air near the surface of the liquid. Note that many common organic liquids have a flash point below room temperature: for example, acetone (–18 °C), benzene (–11.1 °C), diethyl ether (–45 °C), and methyl alcohol (11.1 °C). The degree of hazard associated with a flammable liquid also depends on other properties, such as its ignition point and boiling point. Commercially obtained chemicals are clearly labeled as to flammability and flash point. Consider the example of acetone given in section 4.C.1.3.1. At ambient pressure and temperature, an acetone spill produces a concentration as high as 23.7% acetone in air. Although it is not particularly toxic, with a flash point of -18 °C and upper and lower flammable limits of 2.6% and 12.8% acetone in air, respectively (see Table 4.4), clearly an acetone spill produces an extreme fire hazard. Thus the major hazard given for acetone in the LCSS is flammability.
4.D.1.2.2 Ignition Temperature
The ignition temperature (autoignition temperature) of a substance, whether solid, liquid, or gaseous, is the minimum temperature required to initiate or cause self-sustained combustion independent of the heat source. The lower the ignition temperature, the greater the potential for a fire started by typical laboratory
The following outline provides a summary of the steps discussed in this chapter that laboratory personnel should use to assess the risks of managing physical hazards in the laboratory.
1. Identify chemicals to be used and circumstances of use. Identify the chemicals involved in the proposed experiment and determine the amounts that will be used. Is the experiment to be done once, or will the chemicals be handled repeatedly? Will the experiment be conducted in an open laboratory, in an enclosed apparatus, or in a chemical fume hood? Is it possible that new or unknown substances will be generated in the experiment?
2. Consult sources of information. Consult an up-to-date laboratory chemical safety summary, material safety data sheet, or NIOSH Pocket Guide to Chemical Hazards (HHS/CDC/NIOSH, 2007). In cases where substances with significant or unusual potential physical hazards are involved, consult more detailed references such as Urben (2007) and other sources discussed in section 4.B of this chapter. Depending on the laboratory personnel’s level of experience and the degree of potential hazard associated with the proposed experiment, obtain the assistance of supervisors and safety professionals before proceeding with risk assessment.
3. Evaluate type of physical, flammable, explosive, or reactive hazard(s) posed by the chemicals. Use the above sources of information to determine the hazards associated with each chemical involved in the proposed experiment. Examples of questions that could be asked are: Are any of the chemicals to be used corrosive? Are any of the chemicals to be used flammable or explosive? Are any of the chemicals strong oxidizers? Will the reaction result in a large release of gases? Are any cryogens required?
4. Evaluate the hazards posed by chemical changes over the course of the experiment. Consider the changes in pressure, heat, flammability, and explosivity during the experiment. If the experiment is a scale-up of a reaction, consider how changes in gas production and temperature may change the engineering controls required for safety.
5. Evaluate type of physical hazard(s) posed by the equipment required. Review procedures for correct use of all equipment. Identify heat and ignition sources and consider appropriate placement for them in the laboratory. Are any pieces of equipment going to be under high or low pressure (e.g., a vacuum line)? Is the glassware free of cracks and chips?
6. Select appropriate procedures to minimize risk. Use the basic prudent practices for handling chemicals, which are discussed in Chapter 6, section 6.C for all work with chemicals in the laboratory and the guidance on using laboratory equipment in Chapter 7. Determine if the chemicals or equipment in use pose physical hazards to laboratory personnel. If so, consider methods to minimize the risk posed by the hazard through substitution with another chemical, if possible, or use of engineering controls (e.g., chemical fume hoods, grounding cables, inert atmospheres) and personal protective equipment if not. Use information in this chapter and Chapter 7 to determine whether it is appropriate to apply the additional procedures before continuing work and whether additional consultation with safety professionals is warranted (see section 4.D).
7. Prepare for contingencies. Be aware of institutional procedures in the event of emergencies and accidents. Some questions to consider are: What are appropriate measures to take in the event of fire, explosion, or injury? What are the appropriate first-aid procedures in the event of heat, cold, or chemical burns? What is the procedure in the event of an emergency or non-emergency spill of the chemicals?
NOTE: For a quick guide for assessing the toxicity hazards associated with laboratory chemicals, see Box 4.1. For a quick guide for assessing biological hazards in the laboratory, see Box 4.3.
equipment. A spark is not necessary for ignition when the flammable vapor reaches its autoignition temperature. For instance, carbon disulfide has an ignition temperature of 90 °C, and it can be set off by a steam line or a glowing light bulb. Diethyl ether has an ignition temperature of 160 °C and can be ignited by a hot plate.
4.D.1.2.3 Limits of Flammability
Each flammable gas and liquid (as a vapor) has two fairly definite limits of flammability defining the range of concentrations in mixtures with air that will propagate a flame and cause an explosion. At the low extreme, the mixture is oxygen rich but contains insufficient fuel. The lower flammable limit (lower explosive
limit [LEL]) is the minimum concentration (percent by volume) of the fuel (vapor) in air at which a flame is propagated when an ignition source is present. The upper flammable limit (upper explosive limit [UEL]) is the maximum concentration (percent by volume) of the vapor in air above which a flame is not propagated. The flammable range (explosive range) consists of all concentrations between the LEL and the UEL. This range becomes wider with increasing temperature and in oxygen-rich atmospheres and also changes depending on the presence of other components. The limitations of the flammability range, however, provide little margin of safety from the practical point of view because, when a solvent is spilled in the presence of an energy source, the LEL is reached very quickly and a fire or explosion ensues before the UEL is reached.
4.D.1.3 Classes of Flammability
Several systems are in use for classifying the flammability of materials. Some (e.g., Class I—flammable
| NFPA Flammability Ratinga | Flash Point (ºC) | Boiling Point (ºC) | Ignition Temperature (ºC) | Flammable Limits (% by volume) | ||
| Lower | Upper | |||||
| Acetaldehyde | 4 | −39 | 21 | 175 | 4 | 60 |
| Acetic acid (glacial) | 2 | 39 | 118 | 463 | 4 | 19.9 |
| Acetone | 3 | −20 | 56 | 465 | 2.5 | 12.8 |
| Acetonitrile | 3 | 6 | 82 | 524 | 3 | 16 |
| Carbon disulfide | 4 | −30 | 46 | 90 | 1.3 | 50 |
| Cyclohexane | 3 | −20 | 82 | 245 | 1.3 | 8 |
| Diethylamine | 3 | −23 | 57 | 312 | 1.8 | 10.1 |
| Diethyl ether | 4 | −45 | 35 | 180 | 1.9 | 36 |
| Dimethyl sulfoxide | 2 | 95 | 189 | 215 | 2.6 | 42 |
| Ethyl alcohol | 3 | 13 | 78 | 363 | 3.3 | 19 |
| Heptane | 3 | −4 | 98 | 204 | 1.05 | 6.7 |
| Hexane | 3 | −22 | 69 | 225 | 1.1 | 7.5 |
| Hydrogen | 4 | −252 | 500 | 4 | 75 | |
| Isopropyl alcohol | 3 | 12 | 83 | 399 | 2 | 12.7 @ 200 (93) |
| Methyl alcohol | 3 | 11 | 64 | 464 | 6 | 36 |
| Methyl ethyl ketone | 3 | −9 | 80 | 404 | 1.4 @ 200 (93) | 11.4 @ 200 (93) |
| Pentane | 4 | <−40 | 36 | 260 | 1.5 | 7.8 |
| Styrene | 3 | 31 | 146 | 490 | 0.9 | 6.8 |
| Tetrahydrofuran | 3 | −14 | 66 | 321 | 2 | 11.8 |
| Toluene | 3 | 4 | 111 | 480 | 1.1 | 7.1 |
| p-Xylene | 3 | 25 | 138 | 528 | 1.1 | 7 |
a0, will not burn under typical fire conditions; 1, must be preheated to burn, liquids with FP ≥ 93.4 °C (200 °F); 2, ignitable when moderately heated, liquids with FP between 37.8 °C (100 °F) and 93.4 °C (200 °F); 3, ignitable at ambient temperature, liquids with FP < 22.8 °C (73 °F), bp ≥ 37.8 °C (100 °F) or FP between 22.8 °C and 37.8 °C (100 °F); 4, extremely flammable, readily dispersed in air, and burns readily, liquids with FP < 22.8 °C (73 °F), bp < 37.8 °C (100 °F).
SOURCE: Adapted with permission from Fire Guide to Hazardous Materials (13th Edition), Copyright © 2001, National Fire Protection Association.
liquid, see Chapter 5, section 5.E.5, Table 5.2) apply to storage or transportation considerations. Another (Class A, B, C—paper, liquid, electrical fire) specifies the type of fire extinguisher to be used (see Chapter 7, section 7.F.2 on emergency equipment). To assess risk quickly, the most direct indicator is the NFPA system, which classifies flammables according to the severity of the fire hazard with numbers 0 to 4 in order of increasing hazard: 0, will not burn; 1, must be preheated to burn; 2, ignites when moderately heated; 3, ignites at normal temperature; 4, extremely flammable (Figure 4.4). Substances rated 3 or 4 under this system require particularly careful handling and storage in the laboratory. Some vendors include the NFPA hazard diamond on the labels of chemicals. The Fire Protection Guide on Hazardous Materials (NFPA, 2001) contains a comprehensive listing of flammability data and ratings. Note that other symbols may be found in the Special Hazard quadrant of the diamond. These symbols (see Table 4.5) are not endorsed by NFPA.
The NFPA fire hazard ratings, flash points, boiling points, ignition temperatures, and flammability limits of a number of common laboratory chemicals are given in Table 4.4 and in the LCSSs (see accompanying CD). The data illustrate the range of flammability for liquids commonly used in laboratories. Dimethyl sulfoxide and glacial acetic acid (NFPA fire hazard ratings of 1 and 2, respectively) are handled in the laboratory without great concern about their fire hazards. By contrast, both acetone (NFPA rating 3) and diethyl ether (NFPA rating 4) have flash points well below room temperature.
Note that tabulations of properties of flammable substances are based on standard test methods, which have very different conditions from those encountered in practical laboratory use. Large safety factors should be applied. For example, the published flammability limits of vapors are for uniform mixtures with air. In a real situation, local concentrations that are much higher than the average may exist. Thus, it is good practice to set the maximum allowable concentration for safe working conditions at some fraction of the tabulated LEL; 10% is a commonly accepted value.
Among the most hazardous liquids are those that have flash points near or below 38 °C (100 °F) according to OSHA (29 CFR § 1910.106) and below 60.5 °C (140.9 °F) according to the U.S. Department of Transportation (49 CFR § 173.120). These materials can be hazardous in the common laboratory environment. There is particular risk if their range of flammability is broad. Note that some commonly used substances are potentially very hazardous, even under relatively
FIGURE 4.4 National Fire Protection Association (NFPA) system for classification of hazards.
SOURCE: Reproduced with permission from NFPA 704-2007. System for the Identification of the Hazards of Materials for Emergency Response, Copyright © 2007 National Fire Protection Association. This symbol is for illustrative purposes only. The NFPA does not classify individual chemicals and is not responsible in any way for the numerical values assigned to any chemical.
TABLE 4.5 Additional Symbols Seen in the NFPA Diamond
|
|
|
|
Symbol |
Meaning |
|
|
|
|
ACID |
Acid |
|
ALK |
Alkali |
|
CORR |
Corrosive |
|
Radioactive |
|
|
|
|
cool conditions (see Table 4.4). Some flammable liquids maintain their flammability even at concentrations of 10% by weight in water. Methanol and isopropyl alcohol have flash points below 38 °C (100 °F) at concentrations as low as 30% by weight in water. High-performance liquid chromatography users generate acetonitrile–water mixtures that contain from 15–30% acetonitrile in water, a waste that is considered toxic and flammable and thus cannot be added to a sewer.
Because of its extreme flammability and tendency for peroxide formation, diethyl ether is available for laboratory use only in metal containers. Carbon disulfide is almost as hazardous.
4.D.1.4 Causes of Ignition
4.D.1.4.1 Spontaneous Combustion
Spontaneous ignition (autoignition) or combustion takes place when a substance reaches its ignition temperature without the application of external heat. The possibility of spontaneous combustion should always be considered, especially when storing or disposing of materials. Examples of materials susceptible to spontaneous combustion include oily rags, dust accumulations, organic materials mixed with strong oxidizing agents (e.g., nitric acid, chlorates, permanganates, peroxides, and persulfates), alkali metals (e.g., sodium and potassium), finely divided pyrophoric metals, and phosphorus.
4.D.1.4.2 Ignition Sources
Potential ignition sources in the laboratory include the obvious torch and Bunsen burner, as well as a number of less obvious electrically powered sources ranging from refrigerators, stirring motors, and heat guns to microwave ovens (see Chapter 7, section 7.C). Whenever possible, open flames should be replaced by electrical heating. Because the vapors of most flammable liquids are heavier than air and capable of traveling considerable distances, special note should be taken of ignition sources situated at a lower level than that at which the substance is being used. Flammable vapors from massive sources such as spills have been known to descend into stairwells and elevator shafts and ignite on a lower story. If the path of vapor within the flammable range is continuous, as along a floor or benchtop, the flame propagates itself from the point of ignition back to its source.
Metal lines and vessels discharging flammable substances should be bonded and grounded properly to discharge static electricity. There are many sources of static electricity, particularly in cold dry atmospheres, and caution should be exercised.
4.D.1.4.3 Oxidants Other Than Oxygen
The most familiar fire involves a combustible material burning in air. However, the oxidant driving a fire or explosion need not be oxygen itself, depending on the nature of the reducing agent. All oxidants have the ability to accept electrons, and fuels are reducing agents or electron donors [see Young (1991)].
Examples of nonoxygen oxidants are shown in Table 4.6. When potassium ignites on addition to water, the metal is the reducing agent and water is the oxidant. If the hydrogen produced ignites, it becomes the fuel for a conventional fire, with oxygen as the oxidant. In ammonium nitrate explosions, the ammonium cation is oxidized by the nitrate anion. These hazardous combinations are treated further in section 4.D.2. (See Chapter 6, section 6.F, for a more detailed discussion on flammable substances.)
4.D.1.5 Special Hazards
Compressed or liquefied gases present fire hazards because the heat causes the pressure to increase and the container may rupture (Yaws and Braker, 2001). Leakage or escape of flammable gases produces an explosive atmosphere in the laboratory; acetylene, hydrogen, ammonia, hydrogen sulfide, propane, and carbon monoxide are especially hazardous.
Even if not under pressure, a liquefied gas is more concentrated than in the vapor phase and evaporates rapidly. Oxygen is an extreme hazard and liquefied air is almost as dangerous because nitrogen boils away first, leaving an increasing concentration of oxygen. Liquid nitrogen standing for a period of time may have condensed enough oxygen to require careful handling. When a liquefied gas is used in a closed system, pres-
TABLE 4.6 Examples of Oxidants
|
|
|
Gases: chlorine, fluorine, nitrous oxide, oxygen, ozone, steam |
|
Liquids: bromide, hydrogen peroxide, nitric acid, perchloric acid, sulfuric acid |
|
Solids: bromates, chlorates, chlorites, chromates, dichromates, hypochlorites, iodates, nitrates, nitrites, perchlorates, peroxides, permanganates, picrates |
|
|
sure may build up and adequate venting is required. If the liquid is flammable (e.g., hydrogen and methane), explosive concentrations may develop without warning unless an odorant has been added. Flammability, toxicity, and pressure buildup become more serious on exposure of gases to heat.
(Also see Chapter 6, section 6.G.2.5, for more information.)
4.D.2 Reactive Hazards
4.D.2.1 Water Reactives
Water-reactive materials are those that react violently with water. Alkali metals (e.g., lithium, sodium, and potassium), many organometallic compounds, and some hydrides react with water to produce heat and flammable hydrogen gas, which ignites or combines explosively with atmospheric oxygen. Some anhydrous metal halides (e.g., aluminum bromide), oxides (e.g., calcium oxide), and nonmetal oxides (e.g., sulfur trioxide), and halides (e.g., phosphorus pentachloride) react exothermically with water, resulting in a violent reaction if there is insufficient coolant water to dissipate the heat produced.
(See Chapter 6, section 6.G, for further information.)
4.D.2.2 Pyrophorics
For pyrophoric materials, oxidation of the compound by oxygen or moisture in air proceeds so rapidly that ignition occurs. Many finely divided metals are pyrophoric, and their degree of reactivity depends on particle size, as well as factors such as the presence of moisture and the thermodynamics of metal oxide or metal nitride formation. Other reducing agents, such as metal hydrides, alloys of reactive metals, low-valent metal salts, and iron sulfides, are also pyrophoric.
4.D.2.3 Incompatible Chemicals
Accidental contact of incompatible substances results in a serious explosion or the formation of substances that are highly toxic or flammable or both. Although trained laboratory personnel question the necessity of following storage compatibility guidelines, the reasons for such guidelines are obvious after reading descriptions of laboratories following California earthquakes in recent decades [see Pine (1994)]. Those who do not live in seismically active zones should take these accounts to heart, as well. Other natural disasters and chemical explosions themselves can set off shock waves that empty chemical shelves and result in inadvertent mixing of chemicals.
Some compounds pose either a reactive or a toxic hazard, depending on the conditions. Thus, hydro-cyanic acid (HCN), when used as a pure liquid or gas in industrial applications, is incompatible with bases because it is stabilized against (violent) polymerization by the addition of acid inhibitor. HCN can also be formed when cyanide salt is mixed with an acid. In this case, the toxicity of HCN gas, rather than the instability of the liquid, is the characteristic of concern.
Some general guidelines lessen the risks involved with these substances. Concentrated oxidizing agents are incompatible with concentrated reducing agents. Indeed, either may pose a reactive hazard even with chemicals that are not strongly oxidizing or reducing. For example, sodium or potassium, strong reducing agents frequently used to dry organic solvents, are extremely reactive toward halocarbon solvents (which are not strong oxidizing agents). Strong oxidizing agents are frequently used to clean glassware, but they should be used only on the last traces of contaminating material. Because the magnitude of risk depends on quantities, chemical incompatibilities will not usually pose much, if any, risk if the quantity of the substance is small (a solution in an NMR tube or a microscale synthesis). However, storage of commercially obtained chemicals (e.g., in 500-g jars or 1-L bottles) should be carefully managed from the standpoint of chemical compatibility.
(For more information about compatible and incompatible chemicals, see Chapter 5, section 5.E.2.)
4.D.3 Explosive Hazards
4.D.3.1 Explosives
An explosive is any chemical compound or mechanical mixture that, when subjected to heat, impact, friction, detonation, or other suitable initiation, undergoes rapid chemical change, evolving large volumes of gases that exert pressure on the surrounding medium. The term applies to materials that either detonate or deflagrate. Heat, light, mechanical shock, and certain catalysts initiate explosive reactions. Hydrogen and chlorine react explosively in the presence of light. Acids, bases, and other substances catalyze the explosive polymerization of acrolein, and many metal ions can catalyze the violent decomposition of hydrogen peroxide. Shock-sensitive materials include acetylides, azides, nitrogen triiodide, organic nitrates, nitro compounds, perchlorate salts (especially those of heavy metals such as ruthenium and osmium), many organic peroxides, and compounds containing diazo, halamine, nitroso, and ozonide functional groups.
Table 4.7 lists a number of explosive compounds. Some are set off by the action of a metal spatula on the solid; some are so sensitive that they are set off by the action of their own crystal formation. Diazomethane (CH2N2) and organic azides, for example, may decom-
TABLE 4.7 Functional Groups in Some Explosive Compounds
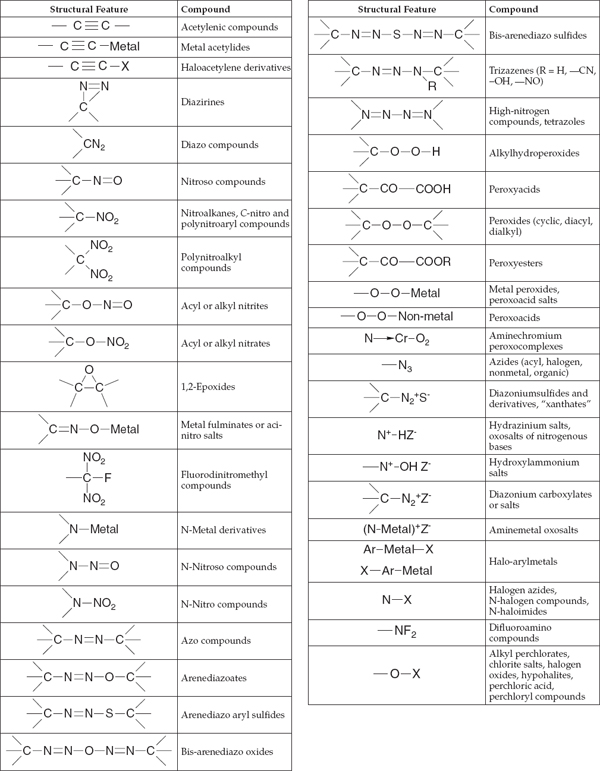
SOURCE: Carson and Mumford (2002). Reprinted from Hazardous Chemicals Handbook (Second Edition), Carson, P. and Mumford, C. “Reactive Chemicals”, p. 228, Copyright 2002, with permission from Elsevier.
pose explosively when exposed to a ground glass joint or other sharp surfaces (Organic Syntheses, 1973, 1961).
4.D.3.2 Azos, Peroxides, and Peroxidizables
Organic azo compounds and peroxides are among the most hazardous substances handled in the chemical laboratory but are also common reagents that often are used as free radical sources and oxidants. They are generally low-power explosives that are sensitive to shock, sparks, or other accidental ignition. They are far more shock sensitive than most primary explosives such as TNT. Inventories of these chemicals should be limited and subject to routine inspection. Many require refrigerated storage. Liquids or solutions of these compounds should not be cooled to the point at which the material freezes or crystallizes from solution, however, because this significantly increases the risk of explosion. Refrigerators and freezers storing such compounds should have a backup power supply in the event of electricity loss. Users should be familiar with the hazards of these materials and trained in their proper handling.
Certain common laboratory chemicals form peroxides on exposure to oxygen in air (see Tables 4.8 and 4.9). Over time, some chemicals continue to build peroxides to potentially dangerous levels, whereas others accumulate a relatively low equilibrium concentration of peroxide, which becomes dangerous only after being concentrated by evaporation or distillation. (See Chapter 6, section 6.G.3.) The peroxide becomes concentrated because it is less volatile than the parent chemical. A related class of compounds includes inhibitor-free monomers prone to free radical polymerization that on exposure to air can form peroxides or other free radical sources capable of initiating violent polymerization. Note that care must be taken when storing and using these monomers—most of the inhibitors used to stabilize these compounds require the presence of oxygen to function properly, as described below. Always refer to the MSDS and supplier instructions for proper use and storage of polymerizable monomers.
Essentially all compounds containing C—H bonds pose the risk of peroxide formation if contaminated with various radical initiators, photosensitizers, or catalysts. For instance, secondary alcohols such as isopropanol form peroxides when exposed to normal fluorescent lighting and contaminated with photosensitizers, such as benzophenone. Acetaldehyde, under normal conditions, autoxidizes to form acetic acid. Although this autoxidation proceeds through a peroxy acid intermediate, the steady-state concentrations of that intermediate are extremely low and pose no hazard. However, in the presence of catalysts (Co2+) and under the proper conditions of ultraviolet light, temperature, and oxygen concentration, high concentrations of an explosive peroxide can be formed. The chemicals described in Table 4.9 represent only those materials that form peroxides in the absence of such contaminants or otherwise atypical circumstances.
TABLE 4.8 Classes of Chemicals That Can Form Peroxides
|
|
|
|
Class A: Chemicals that form explosive levels of peroxides without concentration |
|
|
Isopropyl ether |
Sodium amide (sodamide) |
|
Butadiene |
Tetrafluoroethylene |
|
Chlorobutadiene (chloroprene) |
Divinyl acetylene |
|
Potassium amide |
Vinylidene chloride |
|
Potassium metal |
|
|
Class B: These chemicals are a peroxide hazard on concentration (distillation/evaporation). A test for peroxide should be performed if concentration is intended or suspected.* (See Chapter 6, section 6.C.3) |
|
|
Acetal |
Dioxane (p-dioxane) |
|
Cumene |
Ethylene glycol dimethyl |
|
Cyclohexene |
ether (glyme) |
|
Cyclooctene |
Furan |
|
Cyclopentene |
Methyl acetylene |
|
Diaacetylene |
Methyl cyclopentane |
|
Dicyclopentadiene |
Methyl-isobutyl ketone |
|
Diethylene glycol dimethyl |
Tetrahydrofuran |
|
ether (diglyme) |
Tetrahydronaphthalene |
|
Diethyl ether |
Vinyl ethers |
|
Class C: Unsaturated monomers that may autopolymerize as a result of peroxide accumulation if inhibitors have been removed or are depleteda |
|
|
Acrylic acid |
Styrene |
|
Butadiene |
Vinyl acetate |
|
Chlorotrifluoroethylene |
Vinyl chloride |
|
Ethyl acrylate |
Vinyl pyridine |
|
Methyl methacrylate |
|
|
|
|
*These lists are illustrative, not comprehensive.
SOURCES: Jackson et al. (1970) and Kelly (1996).
Although not a requirement, it is prudent to discard old samples of organic compounds of unknown origin or history, or those prone to peroxidation if contaminated; secondary alcohols are a specific example.
Class A compounds are especially dangerous when
TABLE 4.9 Types of Compounds Known to Autoxidize to Form Peroxides
|
|
|
Ethers containing primary and secondary alkyl groups (never distill an ether before it has been shown to be free of peroxide) |
|
Compounds containing benzylic hydrogens |
|
Compounds containing allylic hydrogens (C=C—CH) |
|
Compounds containing a tertiary C—H group (e.g., decalin and 2,5-dimethylhexane |
|
Compounds containing conjugated, polyunsaturated alkenes and alkynes (e.g., 1,3-butadiene, vinyl acetylene) |
|
Compounds containing secondary or tertiary C—H groups adjacent to an amide (e.g., 1-methyl-2-pyrrolidinone) |
|
|
peroxidized and should not be stored for long periods in the laboratory. Good practice requires they be discarded within 3 months of receipt. Inventories of Class B and C materials should be kept to a minimum and managed on a first-in, first-out basis. Class B and C materials should be stored in dark locations. If stored in glass bottles, the glass should be amber. Containers should be marked with their opening date and inspected every 6 months thereafter.
Class B materials are often sold with autoxidation inhibitors. If the inhibitor is removed, or if inhibitor-free material is purchased, particular care must be taken in their long-term storage because of the enhanced probability of peroxide formation. Purging the container headspace with nitrogen is recommended. Several procedures, including test strips, are available to check Class B materials for peroxide contamination. (For information about testing for peroxides, see Chapter 6, section 6.G.3.2) No special disposal precautions are required for peroxide-contaminated Class B materials.
In most cases, commercial samples of Class C materials are provided with polymerization inhibitors that require the presence of oxygen to function and therefore are not to be stored under inert atmosphere. Inhibitor-free samples of Class C compounds (i.e., the compound has been synthesized in the laboratory or the inhibitor has been removed from the commercial sample) should be kept in the smallest quantities required and under inert atmosphere. Unused material should be properly disposed of immediately, or if long-term storage is necessary, an appropriate inhibitor should be added.
(For more information about handling of peroxides, see Chapter 6, section 6.G.3.)
4.D.3.3 Other Oxidizers
Oxidizing agents may react violently when they come into contact with reducing materials and sometimes with ordinary combustibles. Such oxidizing agents include halogens, oxyhalogens and organic peroxyhalogens, chromates, and persulfates as well as peroxides. Inorganic peroxides are generally stable. However, they may generate organic peroxides and hydroperoxides in contact with organic compounds, react violently with water (alkali metal peroxides), and form superoxides and ozonides (alkali metal peroxides). Perchloric acid is a powerful oxidizing agent with organic compounds and other reducing agents. Perchlorate salts are explosive and should be treated as potentially hazardous compounds.
Baths to clean glassware generally contain strong oxidizers and should be handled with care. For many years, sulfuric acid–dichromate mixtures were used to clean glassware. These solutions are corrosive and toxic and present difficulties for disposal. Their use should be avoided if at all possible. A common substitute is a sulfuric acid–peroxydisulfate solution, and commercial cleaning solutions that contain no chromium are readily available. Confusion about appropriate cleaning bath solutions has led to explosions due to mixing of incompatible chemicals such as potassium permanganate with sulfuric acid or nitric acid with alcohols. For information about how to clean glassware appropriately, consider contacting the manufacturer of the equipment.
4.D.3.4 Powders and Dusts
Suspensions of oxidizable particles (e.g., flour, coal dust, magnesium powder, zinc dust, carbon powder, and flowers of sulfur) in the air constitute a powerful explosive mixture. These materials should be used with adequate ventilation and should not be exposed to ignition sources. Some solid materials, when finely divided, spontaneously combust if allowed to dry while exposed to air. These materials include zirconium, titanium, Raney nickel, finely divided lead (such as prepared by pyrolysis of lead tartrate), and catalysts such as activated carbon containing active metals and hydrogen.
4.D.3.5 Explosive Boiling
Not all explosions result from chemical reactions; some are caused physically. A dangerous explosion can occur if a hot liquid or a collection of very hot particles comes into sudden contact with a lower boiling-point material. Sudden boiling eruptions occur when a nucleating agent (e.g., charcoal, “boiling chips”) is added to a liquid heated above its boiling point. Even if the material does not explode directly, the sudden formation of a mass of explosive or flammable vapor can be very dangerous.
4.D.3.6 Other Considerations
The hazards of running a new reaction should be considered especially carefully if the chemical species involved contain functional groups associated with explosions or are unstable near the reaction or work-up temperature, if the reaction is subject to an induction period, or if gases are byproducts. Modern analytical techniques (see Chapter 6, section 6.G) can be used to determine reaction exothermicity under suitable conditions.
Even a small sample may be dangerous. Furthermore, the hazard is associated not with the total energy released but with the remarkably high rate of a detonation reaction. A high-order explosion of even milligram
quantities can drive small fragments of glass or other matter deep into the body; therefore, use minimum amounts of these hazardous materials with adequate shielding and personal protection. A compound is apt to be explosive if its heat of formation is more than 100 cal/g less than the sum of the heats of formation of its products. In making this calculation, a reasonable reaction should be used to yield the most exothermic products.
Scaling up reactions introduces several hazards. Unfortunately, the current use of microscale teaching methods in undergraduate laboratories increases the likelihood that graduate students and others are unprepared for problems that arise when a reaction is run on a larger scale. These problems include heat buildup and the serious hazard of explosion from incompatible materials. The rate of heat input and production must be weighed against that of heat removal. Bumping the solution or a runaway reaction can result when heat builds up too rapidly.
Exothermic reactions can run away if the heat evolved is not dissipated. When scaling up experiments, sufficient cooling and surface for heat exchange should be provided, and mixing and stirring rates should be considered. Detailed guidelines for circumstances that require a systematic hazard evaluation and thermal analysis are given in Chapter 6, section 6.G.
Another situation that can lead to problems is a reaction susceptible to an induction period; particular care must be given to the rate of reagent addition versus its rate of consumption. Finally, the hazards of exothermic reactions or unstable or reactive chemicals are exacerbated under extreme conditions, such as high temperature or high pressure used for hydrogenations, oxygenations, or work with supercritical fluids.
4.E.1 Compressed Gases
Compressed gases can expose the trained laboratory personnel to both mechanical and chemical hazards, depending on the gas. Hazards can result from the flammability, reactivity, or toxicity of the gas; from the possibility of asphyxiation; and from the gas compression itself, which could lead to a rupture of the tank or valve. (See Chapter 7, section 7.D.)
4.E.2 Nonflammable Cryogens
Nonflammable cryogens (chiefly liquid nitrogen) can cause tissue damage from extreme cold because of contact with either liquid or boil-off gases. In poorly ventilated areas, inhalation of gas due to boil off or spills can result in asphyxiation. Another hazard is explosion from liquid oxygen condensation in vacuum traps or from ice plug formation or lack of functioning vent valves in storage Dewars. Because 1 volume of liquid nitrogen at atmospheric pressure vaporizes to 694 volumes of nitrogen gas at 20 °C, the warming of such a cryogenic liquid in a sealed container produces enormous pressure, which can rupture the vessel. (See Chapter 6, section 6.G.4, and Chapter 7, section 7.E.2, for detailed discussion.)
4.E.3 High-Pressure Reactions
Experiments that generate high pressures or are carried out at pressures above 1 atm can lead to explosion from equipment failure. For example, hydrogenation reactions are frequently carried out at elevated pressures, and a potential hazard is the formation of explosive O2/H2 mixtures and the reactivity/pyrophoricity of the catalyst (see section 6.G.5). High pressures can also be associated with the use of supercritical fluids.
When evaluating whether a reaction generates high pressures, it is important to consider not just the initial reaction conditions, but the kinetics and thermodynamics of the reaction as a whole. Is any stage of the reaction exothermic? What are the characteristics of the reactants, products, intermediates, and synthetic byproducts (explosive, gaseous, etc.)? What are the temperature and pressure requirements for equipment used during the reaction? If scaling up a reaction, carefully calculate the expected temperatures and pressures that will be generated and the rates at which any pressures will be generated. Be sure to choose laboratory equipment that is appropriate for every stage of the reaction, and consult with the manufacturer if there are any questions or concerns about whether a given reactor or piece of equipment is appropriate for high-pressure work. (For more information about using high-pressure equipment, see Chapter 7, section 7.E.)
In many cases, barricading is not necessary if the appropriate reaction vessel, fittings, and other equipment are used. However, the laboratory environment must be designed to accommodate the failure of the equipment: ventilation must be adequate to handle discharge from a high-pressure reaction to prevent asphyxiation, laboratory personnel may require hearing protection to guard against the sound of a rupture disc failure, and barricades are necessary if catastrophic failure could result in injury or death of laboratory personnel. For specific information regarding barricade design, see Porter et al. (1956); Smith (1964); and the Handbook of Chemical Health and Safety (Alaimo, 2001).
4.E.4 Vacuum Work
Precautions to be taken when working with vacuum lines and other glassware used at subambient pressure are mainly concerned with the substantial danger of
injury in the event of glass breakage. The degree of hazard does not depend significantly on the magnitude of the vacuum because the external pressure leading to implosion is always 1 atmosphere. Thus, evacuated systems using aspirators merit as much respect as high-vacuum systems. Injury due to flying glass is not the only hazard in vacuum work. Additional dangers can result from the possible toxicity of the chemicals contained in the vacuum system, as well as from fire following breakage of a flask (e.g., of a solvent stored over sodium or potassium). (For more information about working with equipment under vacuum, see Chapter 7, section 7.E.)
Because vacuum lines typically require cold traps (generally liquid nitrogen) between the pumps and the vacuum line, precautions regarding the use of cryogens should be observed also. Health hazards associated with vacuum gauges have been reviewed (Peacock, 1993). The hazards include the toxicity of mercury used in manometers and McLeod gauges, overpressure and underpressure situations arising with thermal conductivity gauges, electric shock with hot cathode ionization systems, and the radioactivity of the thorium dioxide used in some cathodes. (For information about reducing the presence of mercury in laboratories, see Chapter 5, section 5.B.8.)
4.E.5 Ultraviolet, Visible, and Near-Infrared Radiation
Ultraviolet, visible, and infrared radiation from lamps and lasers in the laboratory can produce a number of hazards. Medium-pressure Hanovia 450 Hg lamps are commonly used for ultraviolet irradiation in photochemical experiments. Ultraviolet lights used in biosafety cabinets, as decontamination devices, or in light boxes to visualize DNA can cause serious skin and corneal burns. Powerful arc lamps can cause eye damage and blindness within seconds. Some compounds (e.g., chlorine dioxide) are explosively photosensitive.
When incorrectly used, the light from lasers poses a hazard to the eyes of the operators and other people present in the room and is also a potential fire hazard. Depending on the type of laser, the associated hazards can include mutagenic, carcinogenic, or otherwise toxic laser dyes and solvents; flammable solvents; ultraviolet or visible radiation from the pump lamps; and electric shock from lamp power supplies.
At the time of this publication, two systems for classifying lasers are in use. Before 2002, lasers were classified as I, II, IIIA, IIIB, and IV. From 2002 forward, a revised system is being phased in which classifies lasers as 1, 1M, 2, 2M, 3R, 3B, and 4. Although they have different designations, both systems classify lasers based on their ability to cause damage to individuals. The older designation is given in the text with the new designation in parentheses. Class I (1) lasers are either completely enclosed or have such a low output of power that even a direct beam in the eye could not cause damage. Class II (2) lasers, can be a hazard if a person stares into the beam and resists the natural reaction to blink or turn away. Class IIIA (1M, 2M, or 3R, depending on power output) lasers can present an eye hazard if a person stares into the beam and resists the natural reaction to blink or turn away or views the beam with focusing optical instruments. Class IIIB (3B) lasers can produce eye injuries instantly from both direct and specularly reflected beams, although diffuse reflections are not hazardous. The highest class of lasers, Class IV (4), presents all the hazards of Class III (3B) lasers but because of their higher power output may also produce eye or skin damage from diffuse scattered light. In addition to these skin and eye hazards, Class IV (4) lasers are a potential fire hazard.
Select protective eyewear with the proper optical density for the specific type of laser in use. Dark lenses can be hazardous because of the risk of looking over the top of the glasses. Leave laser safety glasses in a bin outside the laboratory so that people entering use the appropriate laser safety glasses. When operating or adjusting a laser, remove or cover any reflective objects on hands and wrists to reduce the chance of reflections. Consider using beam blocks and containment walls to reduce the chance of stray reflections in the laboratory. When using a laser-based microscope, consider using a camera and computer display to view the sample rather than direct viewing through the eyepiece. Anyone who is not the authorized operator of a laser system should never enter a posted laser-controlled laboratory if the laser is in use. Visitors may be present when a laser is in use, but they must be authorized by the laboratory supervisor. Visitors must not operate the equipment and should be under the direct supervision of an approved operator.
4.E.6 Radio Frequency and Microwave Hazards
Radio frequency (rf) and microwaves occur within the range 10 kHz to 300,000 MHz and are used in rf ovens and furnaces, induction heaters, and microwave ovens. Extreme overexposure to microwaves can result in the development of cataracts or sterility or both. Microwave ovens are increasingly being used in laboratories for organic synthesis and digestion of analytical samples. Only microwave ovens designed for laboratory or industrial use should be used in a laboratory. Use of metal in microwave ovens can result in arcing and, if a flammable solvent is present, in fire or explosion. Superheating of liquids can occur. Capping of vials and other containers used in the oven can result
in explosion from pressure buildup within the vial. Inappropriately selected plastic containers may melt.
4.E.7 Electrical Hazards
The electrocution hazards of electrically powered instruments, tools, and other equipment are almost eliminated by taking reasonable precautions, and the presence of electrically powered equipment in the laboratory need not pose a significant risk. Many electrically powered devices are used in homes and workplaces in the United States, often with little awareness of the safety features incorporated in their design and construction. But, in the laboratory these safety features should not be defeated by thoughtless or ill-informed modification. The possibility of serious injury or death by electrocution is very real if careful attention is not paid to engineering, maintenance, and personal work practices. Equipment malfunctions can lead to electrical fires. If there is a need to build, repair, or modify electrical equipment, the work should ideally be performed or, at a minimum, inspected by a trained and licensed electrician or electrical expert. All laboratory personnel should know the location of electrical shutoff switches and circuit breaker switches and should know how to turn off power to burning equipment by using these switches. Laboratory equipment should be correctly bonded and grounded to reduce the chances of electric shock if a fault occurs.
Some special concerns arise in laboratory settings. The insulation on wires can be eroded by corrosive chemicals, organic solvent vapors, or ozone (from ultraviolet lights, copying machines, and so forth). Eroded insulation on electrical equipment in wet locations such as cold rooms or cooling baths must be repaired immediately. In addition, sparks from electrical equipment can serve as an ignition source in the presence of flammable vapor. Operation of certain equipment (e.g., lasers, electrophoresis equipment) may involve high voltages and stored electrical energy. The large capacitors used in many flash lamps and other systems are capable of storing lethal amounts of electrical energy and should be regarded as live even if the power source has been disconnected.
Loss of electrical power can produce extremely hazardous situations. Flammable or toxic vapors may be released from freezers and refrigerators as chemicals stored there warm up; certain reactive materials may decompose energetically on warming. Laboratory chemical hoods may cease to function. Stirring (motor or magnetic) required for safe reagent mixing may cease. Return of power to an area containing flammable vapors may ignite them.
4.E.8 Magnetic Fields
Increasingly, instruments that generate large static magnetic fields (e.g., NMR spectrometers) are present in research laboratories. Such magnets typically have fields of 14,000 to 235,000 G (1.4 to 23.5 T), far above that of Earth’s magnetic field, which is approximately 0.5 G. The magnitude of these large static magnetic fields falls off rapidly with distance. Many instruments now have internal shielding, which reduces the strength of the magnetic field outside of the instrument (see Chapter 7, Table 7.1). Strong attraction occurs when the magnetic field is greater than 50 to 100 G and increases by the seventh power as the separation is reduced. However, this highly nonlinear falloff of magnetic field with distance results in an insidious hazard. Objects made of ferromagnetic materials such as ordinary steel may be scarcely affected beyond a certain distance, but at a slightly shorter distance may experience a significant attraction to the field. If the object is able to move closer, the attraction force increases rapidly, and the object can become a projectile aimed at the magnet. Objects ranging from scissors, knives, wrenches, and other tools, keys, steel gas cylinders, buffing machines, and wheelchairs have been pulled from a considerable distance to the magnet itself.
Superconducting magnets use liquid nitrogen and liquid helium coolants. Thus, the hazards associated with cryogenic liquids (see section 4.E.2) are of concern, as well.
The health effects of exposure to static magnetic fields is an area of active research. Currently, there is no clear evidence of a negative health impact from exposure to static magnetic fields, although biological effects have been observed (Schenck, 2000), and recently, guidelines on limits of exposure to static magnetic fields have been issued by the International Commission on Non-ionizing Radiation (ICNIRP, 2009), which is a collaborating organization with the World Health Organization’s International Electromagnetic Field Project.
(For more information about magnetic fields, see Chapter 7, section 7.C.8.41.)
4.E.9 Sharp Edges
Among the most common injuries in laboratories are cuts from broken glass. Cuts can be minimized by the use of correct procedures (e.g., the procedure for inserting glass tubing into rubber stoppers and tubing, which is taught in introductory laboratories), through the appropriate use of protective equipment, and by careful attention to manipulation. Glassware should
always be checked for chips and cracks before use and discarded if any are found. Never dispose of glass in the general laboratory trash. It should only be placed in specific glassware disposal bins. This will reduce the chance of anyone changing the trash receiving a cut.
Other cut hazards include razors, box cutters, knives, wire cutters, and any other sharp-edged tool. When working with these tools, it is important to wear appropriate eye protection and cut-resistant gloves. Follow basic safety procedures when using a cutting tool:
• Inspect the tool prior to use. Do not use it if it is damaged.
• When cutting, always use a tool with a sharp edge. Dull edges are more likely to slip and cause harm.
• Keep hands out of the line of the cut.
• Stand off-line from the direction of the cut.
• If using a box cutter or other tool with a mounted blade, ensure that the blade is well seated before use.
• Never use a cutting tool for a task for which it was not designed, for example, as a screwdriver or lever for opening a container.
• Never submerge a sharp object in soapy or dirty water. It can be difficult to see and poses a risk to the dishwasher.
4.E.10 Slips, Trips, and Falls
Other common injuries in the laboratory arise from slipping, tripping, or improper lifting. Spills resulting from dropping chemicals not stored in protective rubber buckets or laboratory carts can be serious because the laboratory worker can fall or slip into the spilled chemical, thereby risking injury from both the fall and exposure to the chemical. Chemical spills resulting from tripping over bottles of chemicals stored on laboratory floors are part of a general pattern of bad housekeeping that can also lead to serious accidents. Wet floors around ice, dry ice, or liquid nitrogen dispensers can be slippery if the areas are not carpeted and if drops or small puddles are not wiped up as soon as they form.
Attempts to retrieve 5-gallon bottles of distilled water, jars of bulk chemicals, and rarely used equipment stored on high shelves often lead to back injuries in laboratory environments. Careful planning of where to store difficult-to-handle equipment and containers (because of weight, shape, or overall size) reduces the incidence of back injuries.
4.E.11 Ergonomic Hazards in the Laboratory
General workplace hazards also apply in the laboratory. For example, laboratory personnel are often involved in actions such as pipetting and computer work that can result in repetitive-motion injuries. Working at a bench or at a microscope without considering posture can result in back strain, and some instruments require additional in-room ventilation that may raise the background noise level to uncomfortable or hazardous levels. With these and other issues such as high or low room temperatures and exposure to vibrations, it is important to be aware of and to control such issues to reduce occupational injuries. For example, microscope users may find that using a camera to view images on a screen, rather than direct viewing through the eyepiece, reduces back and eye strain.
The Centers for Disease Control and Prevention (CDC) and the National Institutes of Health have information on their Web sites (www.cdc.gov and www.nih.gov, respectively) describing specific ergonomic concerns for laboratories and proposed solutions. The CDC provides a downloadable self-assessment form to aid in evaluating these hazards. NIOSH (www.cdc.gov/niosh) and OSHA (www.osha.gov) provide information about vibration, noise levels, and other workplace hazards.
Nanoscale materials are of considerable scientific interest because some chemical and physical properties can change at this scale. (See definition of engineered nanomaterials below.) These changes challenge the researcher’s, manager’s, and safety professional’s understanding of hazards, and their ability to anticipate, recognize, evaluate, and control potential health, safety, and environmental risks. Essentially any solid may be formed in the nano size range, and in general, the term “nanomaterials” has been broadly accepted as including a number of nanometer-scale objects, including: nanoplates, nanofibers (including nanotubes); and nanoparticles. In addition to the conventional hazards posed by the material, hazard properties may also change.
Nanoparticles are dispersible particles that are between 1 and 100 nm in size that may or may not exhibit a size-related intensive property. The U.S. Department of Energy (DOE, 2008, 2009) states that engineered nanomaterials are intentionally created, in contrast with natural or incidentally formed, and engineered to be between 1 and 100 nm. This definition
FIGURE 4.5 U.S. Department of Energy graded exposure risk for nanomaterials. This figure assumes that no disruptive force (e.g., sonication, grinding, burning) is applied to the matrix. SOURCE: Adapted from Karn (2008).
excludes biomolecules (proteins, nucleic acids, and carbohydrates).3 Incidentally formed nanoparticles are often called “ultrafine” particles.
As with hazardous chemicals, exposures to these materials may occur through inhalation, dermal contact, accidental injection, and ingestion, and the risk increases with duration of exposure and the concentration of nanoparticles in the sample or air. Inhalation presents the greatest exposure hazard. Nanomaterials suspended in a solution or slurry pose a lesser hazard, but because the solutions can dry into a powder, they should be handled with care. Nanomaterials suspended in a solution or slurry present a hazard whenever mechanical energy is imparted to the suspension of slurry. Sonication, shaking, stirring, pouring, or spraying of a suspended nanomaterial can result in an inhalation exposure. Suspensions also represent a dermal exposure potential. Nanoparticles that are fixed within a matrix pose the least hazard as long as no mechanical disruption, such as grinding, cutting, or burning, occurs. (See Figure 4.5.)
Nanoparticles can enter the laboratory in a variety of ways. For example, the materials may be imported into the lab for characterizations or be incorporated into a study. Alternatively, they could be created (synthesized) in the lab as part of an experiment. In either case, it is important for laboratory personnel to know about the presence and physical state of the nanomaterial (i.e., powder, in solution, on a solid matrix, or in solid matrix) so they can manage the hazards accordingly.
Nanoparticles have significantly greater relative surface areas than larger particles of an equivalent mass, and animal studies have demonstrated a correlation between biological effects (toxic response) and surface area. Thus, nanoparticles represent a greater toxic hazard than an equivalent mass of the same material in larger form. In addition, the number of particles per unit mass is far greater than the number of particles in bulk material per unit mass, resulting in significantly different inhalational hazards between the two forms. Because of their size, nanoparticles can penetrate deep into the lungs, and with a large number of particles in a small volume, can overwhelm the organ and disrupt normal clearance processes. The greater surface reactivity also plays a role in this disruption. Once inside the lungs, nanoparticles may translocate to other organs via pathways not demonstrated in studies with larger particles. In addition, at the interface of the nanoparticle and human cell surface, bioactivity may occur. For example, nanometal particles have been demonstrated to produce reactive oxygen species, implicating the presence of free radicals, and causing the biological effects of inflammation and fibrosis.
The nanoparticulate forms of some materials show unusually high reactivity, especially for fire, explosion, and catalytic reactions. Engineered nanoparticles and nanostructured porous materials have been used effectively for many years as catalysts for increasing the rate of reactions or decreasing the temperature needed for reactions in liquids and gases. Depending on their composition and structure, some nanomaterials initiate catalytic reactions that would not otherwise be anticipated from their chemical composition. Note also that nanomaterials may be attached to the surface of larger particles. In those cases, the larger material may take on the higher reactivity features of the engineered nanoscale material, even though it is not in the form of a particle in the 1- to 100-nm size range.
As noted above, because material properties can change at the nanoscale, nanomaterials should not be assumed to present only those hazards known to be associated with bulk forms of material having the same composition. Instead, they must be handled as though toxic and reactive until credible evidence eliminates uncertainty. Hazard information is available on a limited number of nano-size materials. For example, NIOSH has proposed special exposure limits for nano-size titanium dioxide that are significantly more restrictive than for larger particles of titanium
_______________
3Note that this definition is slightly different from the definition of the International Organization for Standardization, where “nano-object is defined as material with one, two, or three external dimensions in the size range of approximately 1–100 nm. Sub categories of nano-object are (1) nanoplate, a nano-object with one external dimension at the nanoscale; (2) nanofber, a nano-object with two external dimensions at the nano scale with a nanotube defined as a hollow nanofber and a nanorod as a solid nanofber; and (3) nanoparticle, a nano-object with all three external dimensions at the nanoscale. Nano-objects are commonly incorporated in a larger matrix or substrate referred to as a nanomaterial” (HHS/CDC/NIOSH, 2009a).
dioxide. Determination of EHS issues is an ongoing effort. The CHO assisting with protection from the EHS hazards will need special education and training to adequately assist in risk assessment and control of nanomaterial risks. Specialized monitoring equipment is required to evaluate potential exposures or release of nanomaterials.
Although there is limited specific guidance on evaluation and control of risks posed by nanomaterials, preliminary research suggests that a well-designed ventilation system with high-efficiency filtration is effective at capturing nanoparticles. However, recent studies (Ellenbecker and Tsai, 2008) have demonstrated that conventional laboratory chemical hoods may create turbulence that can push the materials back into the laboratory space. Lower flow hoods with less turbulence may be more appropriate. (For more information about engineering controls for handling of nanoparticles, see Chapter 9, section 9.E.5. For further information on transportation, see Chapter 5, section 5.F.2 and Chapter 6, section 6.J for information about working with nanoparticles.)
Biohazards are a concern in laboratories in which microorganisms, or material contaminated with them, are handled. Anyone who is likely to come in contact with blood or potentially infectious materials at work is covered under OSHA’s Bloodborne Pathogen Standard, 29 CFR § 1910.1030. These hazards are usually present in clinical and infectious disease research laboratories but may also be present in any laboratory in which bodily fluids, tissues, or primary or immortalized cell lines of human or animal origin are handled. Biohazards are also present in any laboratory that uses microorganisms, including replication-deficient viral vectors, for protein expression or other in vitro applications. Occasionally, biohazards are present in testing and quality control laboratories, particularly those associated with water and sewage treatment plants and facilities involved in the production of biological products and disinfectants. Teaching laboratories may introduce low-risk infectious agents as part of a course of study in microbiology.
Synthetic biology makes it possible to synthesize microorganisms from basic chemical building blocks, and these microorganisms may have different hazards from their naturally occurring relatives. If a microorganism identical or very similar to one found in nature is synthesized, the risks are assumed to be similar to those of the naturally occurring microorganism. If a novel microorganism is synthesized, however, extra caution must be used until the characteristics of the agent are well understood.
Risk assessment for biohazardous materials can be complicated because of the number of factors that must be considered. The things that must be accounted for are the organism being manipulated, any alterations made to the organism, and the activities that will be performed with the organism. Risk assessment for biological toxins is similar to that for chemical agents and is based primarily on the potency of the toxin, the amount used, and the procedures in which the toxin is used. An example of a risk assessment for a material with unknown biological risks can be found in Backus et al. (2001). See Box 4.3 for a quick guide to assessing risks from biohazards in the laboratory.
Certain biological toxins and agents are classified as select agents under 42 CFR Part 73 and have additional regulatory and security requirements that must be considered when receiving and working with these agents. For detailed information on risk assessment of biohazards, consult the fifth and most recent edition of Biosafety in Microbiological and Biomedical Laboratories (BMBL; HHS/CDC/NIH, 2007a) and the NIH Guidelines for Research Involving Recombinant DNA Molecules (NIH, 2009). BMBL is considered the consensus code of practice for identifying and controlling biohazards and was first produced by the CDC and the National Institutes of Health in 1984. (Also see Chapter 6, section 6.E, and Chapter 11.)
4.H HAZARDS FROM RADIOACTIVITY
This section provides a brief primer on the potential hazards arising from the use of radioactivity in a laboratory setting. A comprehensive treatment of this topic is given in Radiation Protection: A Guide for Scientists, Regulators, and Physicians (Shapiro, 2002). For an introduction to health physics, see Cember and Johnson (2008). Note that the receipt, possession, use, transfer, and disposal of most radioactive materials is strictly regulated by the U.S. Nuclear Regulatory Commission (USNRC; see 10 CFR Part 20, Standards for Protection Against Radiation) and/or by state agencies who have “agreements” with the USNRC to regulate the users within their own states. Radioactive materials may be used only for purposes specifically described in licenses issued by this agency to licensees. Individuals working with radioactive materials should thus be aware of the restrictions and requirements of these licenses. Consult your radiation safety officer or other designated EHS professional for training, policies, and procedures specific to uses at your institution.
Unstable atomic nuclei eventually achieve a more stable form by emission of some type of radiation. These nuclei or isotopes are termed radioactive. The emitted radiation may be characterized as particulate (α, β, proton, or neutron) or electromagnetic (γ rays
BOX 4.3
Quick Guide to Risk Assessment for Biological Hazards in the Laboratory
The following steps are provided to assist trained laboratory personnel in performing a risk assessment of activities involving biohazardous materials. This is not intended as a flowchart; rather, all questions should be considered before arriving at the final risk assessment.
1. Identify the risk group of the parent organism (the American Biological Safety Association has a database of risk groups from various sources online at http://absa.org).
2. This Risk Group assignment is only a starting point; the actual biosafety level at which the work is performed may be higher or lower depending on the remainder of the risk assessment.
3. Refer to the Agent Summary Statement in Biosafety in Microbiological and Biomedical Laboratories (BMBL; HHS/CDC/NIH, 2007a) if available. This will provide a recommended biosafety level as well as personal protective and containment equipment to use while handling the organism. It will also summarize the frequency and route of laboratory-acquired infections.
4. Identify the natural route of transmission for the parent organism. This will indicate the most likely route of laboratory-acquired infections. Be aware that at the volumes and concentrations used in the laboratory, however, organisms can often be transmitted in ways other than their normal route of exposure (e.g., aerosol transmission of agents normally only transmitted via mosquito bite).
5. Consider any modifications made to the organism. Has the host range been modified? Have virulence factors been inserted or removed? Has the organism been rendered replication-defective? If so, is there any possibility of recombination events with wild-type organisms to restore replication competency?
6. Consider the transgenes expressed by the organism. Think about the effect of aberrant expression of that protein if personnel were accidentally exposed to the organism. Is the organism now expressing oncogenes or toxins, or knocking down expression of a tumor suppressor?
7. What volume and concentration of agent are handled at any one time? In general, higher volumes or concentrations call for stricter safety precautions. Recombinant organisms in volumes greater than 10 L in a single vessel have additional regulatory requirements as well.
8. Will research animals be used in any procedures? If so, will they be restrained or anesthetized? What is the immune status of the research animals? Is there a possibility that research animals will excrete potentially infectious or otherwise harmful substances?
9. Are sharps utilized in any procedures? Use of sharps should be minimized whenever possible. If sharps must be used with potentially infectious materials, use caution.
10. Will any manipulations generate aerosols (e.g., vortexing, centrifuging)? If so, perform these operations in a biosafety cabinet or other appropriate containment equipment whenever possible.
11. Are vaccinations or treatments available for the agents in question? Consult with an occupational health provider to determine the necessity for vaccination or postexposure management.
12. Are the organisms used particularly hazardous for certain groups of people (e.g., pregnant women, immunocompromised individuals)? Notify any personnel who will work with or around the organism(s) of these special concerns.
NOTE: For a quick guide for assessing the toxicity hazards associated with laboratory chemicals, see Box 4.1. For a quick guide for assessing physical flammable, explosive, and reactive hazards in the laboratory, see Box 4.2.
or X rays). Particulate radiations have both mass and electromagnetic radiations, which are sometimes referred to as photons. Radiation that has enough energy to ionize atoms and create ion pairs is referred to as ionizing radiation. Ionizing radiation not only comes from unstable nuclei, but can also be produced by machines such as particle accelerators, cyclotrons, and X-ray machines.
Alpha particles are charged particles containing two protons and two neutrons and are emitted from certain heavy atoms such as uranium and thorium. These particles are relatively large, slow, heavy, and easily stopped by a sheet of paper, a glove, a layer of clothing or even a dead layer of skin cells, and thus present virtually no external exposure hazard to people. However, because of the very large number of ionizations that α particles produce in short distances, α emitters can present a serious hazard when they come in contact
with internal living cells and tissues. Special precautions are thus taken to ensure that α emitters are not inhaled, ingested, or injected. Care must be taken with unsealed α-emitting sources to control contamination and minimize the potential for internal uptakes.
A β particle (see Table 4.10) is an electron emitted from the nucleus of a radioactive atom. Positively charged counterparts of β particles are called positrons. Beta particles are much less massive and less charged than α particles and interact less intensely with atoms in the materials through which they pass, which gives them a longer range than α particles. Examples of β emitters commonly used in biological research are hydrogen-3 (tritium) (3H), carbon-14 (14C), phosphorus-32 (32C), phosphorus-33 (33P), and sulfur-35 (35P). Although low-energy β particles are usually stopped by the dead layer of skin, higher energy β particles can penetrate more deeply and cause high exposures to the skin and eyes. The energy level of the β particle thus determines if shielding and exposure monitoring is required when working with these materials, as well as how contamination surveys are performed. Table 4.10 provides typical examples of high-energy, low-energy, and extremely low-energy β-particle handling precautions. When shielding is used to reduce external exposures from β emitters, a low-density shielding material such as Plexiglas, Lucite, or acrylic works best.
Gamma rays, x rays, and photon radiations have no mass or charge. Gamma rays are generally emitted from the nucleus during nuclear decay, and x rays are emitted from the electron shells. Extremely dense material such as lead typically makes the best shields for these electromagnetic forms of radiation. Iodine-125 (125I), indium-111 (111In), and chromium-51 (51Cr) are a few examples of radionuclides sometimes used in research laboratories.
Neutrons are emitted from the nucleus during decay, have no electrical charge, and are one-fourth the mass of an α particle. Exposure to neutrons can be hazardous because the interaction of neutrons with molecules in the body can cause disruption to molecules and atoms. Because of its lack of charge, the neutron is difficult to shield, can penetrate deeply into tissues, and can travel hundreds of yards in air depending on the kinetic energy of the neutron. A neutron is slowed when it collides with the nucleus of other atoms. This transfers kinetic energy of the neutron to the nucleus of the atom. As the mass of the nucleus approaches the mass of the neutron, this reaction becomes more effective in slowing the neutron. Therefore water and other hydrogen-rich materials, such as paraffin or concrete, are often used as shielding material.
TABLE 4.10 Examples of β Emitters
|
|
|||
|
High-Energy Emitters |
Low-Energy Emitters |
Extremely Low Energy Emitters |
|
|
|
|||
|
Examples |
Cl-35 P-32 Sr-90 |
C-14 S-35 P-33 |
H-3 |
|
Shielding |
Shielding required (Plexiglas) |
Not required |
Not required |
|
Contamination survey type |
Survey meter |
Survey meter |
Wipe sample |
|
Exposure dosimetry |
Recommended |
Not required |
Not required |
|
|
|||
Radioactive decay rates are reported in curies (1 curie [Ci] = 3.7 × 1010 disintegrations per second [dps]) or in the International System of Units (SI) in becquerels (1 Bq = 1 dps). The decay rate provides a characterization of a given source but is not an absolute guide to the hazard of the material. The hazard depends on the nature, as well as the rate of production, of the ionizing radiation. In characterizing human exposure to ionizing radiation, it is assumed that the damage is proportional to the energy absorbed. The radiation absorbed dose (rad) is defined in terms of energy absorbed per unit mass: 1 rad = 100 ergs/g (SI: 1 Gy = 1 J/kg = 100 rads). For electromagnetic energy, the roentgen (R) produces 1.61 × 1012 ion pairs per gram of air (SI: 1 C/kg = 3.876 R).
Acceptable limits for occupational exposure to ionizing radiation are set by the USNRC based on the potential amount of tissue damage that can be caused by the exposure. This damage is expressed as a dose equivalent; the common unit for dose equivalent is the roentgen equivalent man (rem). The dose equivalent is determined by the rad multiplied by a weighting factor, called a quality factor, to account for the differences in the nature of the ionizing radiation from different types of radiation. Table 4.11 shows the quality factors for different types of radiation. For γ rays and X rays, rad and rem are virtually equivalent.
Damage may occur directly as a result of the radiation interacting with a part of the cell or indirectly by the formation of toxic substances within the cell. The extent of damage incurred depends on many factors, including the dose rate, the size of the dose, and the site of exposure. Effects may be short term or long term. Acute short-term effects associated with large
TABLE 4.11 Radiation Quality Factors
|
|
|
|
Type of Radiation |
Quality Factor (Q) |
|
|
|
|
x, γ, or β radiation |
1 |
|
α particles |
20 |
|
Neutrons of unknown energy |
10 |
|
|
|
doses and high dose rates—for example, 100,000 mrad (100 rad) in less than 1 week—may include nausea, diarrhea, fatigue, hair loss, sterility, and easy bruising. In appropriately managed workplaces, such exposures are impossible unless various barriers, alarms, and other safety systems are deliberately destroyed or bypassed. Single-dose exposures higher than 500 rem are probably fatal. A single dose of ~100 rem may cause a person to experience nausea or skin reddening, although recovery is likely. However, if these doses are cumulative over a period of time rather than a single dose, the effects are less severe. Long-term effects, which develop years after a high-dose exposure, are primarily cancer. Exposure of the fetus in utero to radiation is of concern, and the risk of damage to the fetus increases significantly when doses exceed 15,000 mrem. The USNRC has set limits for whole-body occupational exposure at 5,000 mrem/year, with minors and declared pregnant workers allowed only 500 mrem/year (or 9-month gestation period), and members of the public allowed only 100 mrem/year (see Table 4.12). Exposure limits are lower in facilities operated by the U.S. Department of Energy and other agencies. Note that properly managed work with radioactive materials in the vast majority of laboratory research settings can be performed without any increase in a worker’s exposure to radiation.
As with all laboratory work, protection of laboratory personnel against the hazard consists of good facility design, operation, and monitoring, as well as good work practices. The ALARA (as low as reasonably achievable) exposure philosophy is central to both levels of protection. The amount of radiation or radioactive material used should be minimized. Exposures should be minimized by shielding radiation sources, laboratory personnel, and visitors and by use of emergency alarm and evacuation procedures. The amount of time spent working with radioactive materials should be minimized. Physical distance between personnel and radiation sources should be maximized, and whenever possible, robotic or other remote operations should be used to reduce exposure of personnel. (Also see Chapter 6, section 6.E.)
TABLE 4.12 U.S. Nuclear Dose Limits Regulatory Commission
|
|
||
|
Area of Dose |
Occupational Dose Limits (mrem/year) |
Public Dose Limits (mrem/year) |
|
|
||
|
Total effective dose equivalent (or whole body: external + internal) |
5,000 |
100* |
|
Committed dose equivalent (or any organ dose) |
50,000 |
NA |
|
Eye dose equivalent (or lens of the eye) |
15,000 |
NA |
|
Shallow dose equivalent (or skin dose) |
50,000 |
NA |
|
Extremity dose (or shallow dose to any extremity) |
50,000 |
NA |
|
Minor (less than 18 years of age) |
10% of occupational limits for adults |
NA |
|
Embryo/fetus of declared pregnant woman (limit taken over time of pregnancy) |
500 |
NA |
|
Personnel dosimetry is required if occupational dose is likely to exceed 10% of the limit (for embryo/fetus, it is required if worker’s dose is likely to exceed 100 mrem during gestation period) |
||
|
|
||
*NOTE: For 10 CFR § 35.75 patient release, limit is 500 mrem.






































