3
Research on Conditions with Particular Relevance to Women
This chapter discusses women’s health research of the last 2 decades according to conditions.1 The committee limits its discussion according to its characterization of women’s health in Chapter 1—conditions that are specific to women; that are more common or serious in women; that have distinct causes, manifestations, outcomes, or treatments in women; or that have high morbidity or mortality in women. Appendix B summarizes the incidence, prevalence, and mortality data and trends that, in part, guided committee selections.
Given the impossibility of presenting all research on women’s health, the committee first discusses examples of successful research that contributed to progress in women’s health. The committee assessed progress on the basis of decreases in incidence or mortality or on the basis of scientific innovations that led to major transformations in approaching a condition. The committee then discusses conditions on which some progress has been made and those on which little progress has been made and about which heightened awareness and further research are needed. Although aware of comorbidities and cross-cutting issues, the committee organized the data for this chapter by condition to reflect of predominant models of research funding and publications.
The committee is aware that the conditions do not include all health conditions that are important to women; a number of conditions that affect many women’s quality of life—including arthritis, chronic fatigue syndrome, chronic pain, colorectal cancer, eating disorders, fibromyalgia, incontinence, irritable bowel syndrome, many pregnancy-related issues, melanoma, memory and cognitive changes associated with perimenopause, mental illness other than depression,
migraines, sexual dysfunction, stress-related disorders, thyroid disease, and type 2 diabetes—are not discussed here. Because of the volume of literature available, the committee could not discuss the research on all health conditions important to women and on some conditions there was little research to discuss. Absence of discussion does not indicate that the committee thought it unimportant. The committee highlighted conditions to provide examples of successes and examples of less progress on which overarching conclusions and recommendations can be based.
The diseases on which there has been substantial progress are breast cancer, cardiovascular disease, and cervical cancer. Conditions on which there has been some progress are depression, human immunodeficiency virus/acquired immune deficiency syndrome (HIV/AIDS), and osteoporosis. The committee discusses research on other conditions—unintended pregnancy,2 maternal mortality and morbidity, autoimmune diseases, alcohol and drug addiction, lung cancer, gynecologic cancers other than cervical cancer, non-malignant gynecological disorders, and dementia of the Alzheimer type (Alzheimer’s disease)—on which little progress has been made.
Each condition is discussed with regard to a brief evaluation of advances in research; its relevance to women’s health in terms of current incidence, prevalence, and mortality rates and trends therein; disparities in current incidence, prevalence, and mortality rates and trends therein among groups of women (see Box 3-1 for explanation of data on disparities); advances in research, particularly in relation to women’s health encompassing research on the understanding of the biology, prevention,3 and diagnosis of, screening, and treatment for it; research gaps; and lessons learned from the research and extent of progress. When discussing treatments, the committee focuses on conventional treatments and does not discuss complementary and alternative medicine (CAM) in detail. As discussed in a previous Institute of Medicine (IOM) report (2005), women are more likely than men to seek CAM therapies and, therefore, those therapies are important to consider when looking at women’s health, from the perspective of potential therapies as well as their potential toxicities and interactions with other medications. The reader is referred to the previous IOM report for further details on CAM research (IOM, 2005).
It is important to note that trends in incidence need to be interpreted in the context of changes in diagnostic criteria and technologies, which can result in the appearance of an increased incidence of a condition (see Box 3-2). This chapter addresses questions 2, 3, and 4 from Box 1-4, whether women’s health research is
|
2 |
The committee considered whether to discuss unintended pregnancy as a health outcome or a determinant of health. It decided to discuss it as an outcome, along with maternal mortality and morbidity, and discuss the determinants that increase the rate of unintended pregnancies in Chapter 2. |
|
3 |
Non-biological determinants of health are mentioned only briefly in this chapter. Details of research on them are discussed in Chapter 2. |
|
BOX 3-1 Data on Disparities Incidence, prevalence, and trend data across races and ethnicities are presented as available. For some conditions for which there is active surveillance, such as cancer, data are routinely collected and presented by race or ethnicity. For other conditions, data are available from the published literature. |
|
BOX 3-2 Interpretation of Changes in Incidence In looking at changes in incidence, it is important to consider whether an increase or a decrease in a rate is due to a real trend in occurrence or to a change in diagnostic criteria, sensitivity of diagnostic tests, screening programs, or another external factor that changes the likelihood of finding a case and might make it appear that incidence is changing (Devesa et al., 1984). For example, some increases seen in breast-cancer incidence have been attributed to more intensive screening programs increasing the ascertainment of cases and not an increase in the secular trend (Seigneurin et al., 2008). |
focused on the most appropriate and relevant conditions and end points, whether it is studying the most relevant groups of women, and whether the most appropriate research methods are being used.
CONDITIONS ON WHICH RESEARCH HAS CONTRIBUTED TO MAJOR PROGRESS
Breast Cancer
The committee considered a large and diverse body of scientific research on breast cancer to have contributed to major progress in understanding the basic biology of breast cancer and the identification of specific risk factors, which led to prevention efforts; in improvements in the detection and treatment of breast cancer; and ultimately in a decrease in mortality rates.
Incidence, Prevalence, and Mortality in Women
During the last 2 decades, there has been heavy investment in breast-cancer research owing in part to the lobbying efforts of breast-cancer survivors and
advocates (IOM, 2004a). One example is the authorization by Congress of a new funding mechanism for breast-cancer research through the Department of Defense, initially focused on pursuing interservice research on breast-cancer screening and diagnosis for military women and dependents of military men (IOM, 2004a). Increased funding was also made available from the National Cancer Institute, other government agencies (such as the Centers for Disease Control and Prevention [CDC] and the Agency for Healthcare Research and Quality [AHRQ]), and individual statewide programs (such as the California Breast Cancer Research Program, funded with tobacco-tax funds). In parallel, the private philanthropic community—such as the Susan G. Komen Foundation, the Breast Cancer Research Foundation, and Avon—raised awareness and money for research to improve treatment and quality of life of the growing number of breast-cancer survivors.
After remaining relatively steady from 1975 to 1990, the overall invasive– breast-cancer mortality in women in the United States began a steady fall in 1990 and continued to drop each year between 1998 and 2007 (NCI, 2010a). The age-adjusted mortality4 from invasive breast cancer dropped from 33.1 per 100,000 women in 1990 to 22.8 per 100,000 women in 2007 (NCI, 2010a). A consortium of investigators using 7 statistical models indicated that the portion of the reduction in mortality attributable to improved or increased screening varied from 28 to 65% (median, 46%), and the remainder was attributed to improved adjuvant therapies (Berry et al., 2005). Breast cancer, however, is still the second-leading cause of cancer deaths in women in the United States (ACS, 2009a; CDC, 2010).5
Despite many gains from research and regardless of the recent drop in mortality, the incidence of breast cancer in women is higher now than in 1975, and breast cancer is the most common non-skin cancer in women in the United States, estimated to account for about 28% of new cancer cases in 2010 (Jemal et al., 2010). The age-adjusted incidence of breast cancer was as high as 141.2 per 100,000 women in 1998 and 1999, and decreased to 124.7 per 100,000 women in 2007, up from about 100–105 per 100,000 women in 1975–1980 (NCI, 2010b). Much of the increase between 1980 and 1998 occurred during the 1980s and reflected increased detection of localized tumors through increased mammographic screening (Garfinkel et al., 1994; Miller et al., 1991; White et al., 1990). During those years, the incidence increased in every 4-year age group above 45 years. From 1999 to 2003, the age-specific incidence of breast cancer decreased in every age group over 45 years (Jemal et al., 2007). Jemal and colleagues (2007) con-
|
4 |
Data are from US Mortality Files, National Center for Health Statistics, Centers for Disease Control and Prevention. Rates are age-adjusted to the 2000 US Standardized Population (19 age groups—Census P25-1130). |
|
5 |
Lung cancer is the leading cause of cancer death in women; cardiovascular disease is the leading cause of death overall in women (see Appendix B for data). |
cluded that part of the decrease is “consistent with saturation in screening mammography.” The large decreases in invasive estrogen-positive breast cancers seen after July 2002 have been attributed to the identification, through the Women’s Health Initiative (WHI), of an increased risk of breast cancer associated with the use of menopausal hormone therapy and a precipitous decline in the number of hormone prescriptions filled after the rapid dissemination of that finding to women who were on hormone therapy (Chlebowski et al., 2009; Hausauer et al., 2009; Ravdin et al., 2007). Sharp decreases in breast cancer from 2002 to 2003 were seen in estrogen-positive tumors in women 50–69 years old (Jemal et al., 2007) and, in a study of white women, were largest in urban counties and counties that had low poverty rates (Hausauer et al., 2007).
Disparities Among Groups
Large disparities in breast-cancer incidence and mortality exist among different demographic groups (see Figure 3-1). Breast cancer is one of the few diseases whose incidence is higher in white women than in other ethnic groups; however, black women have higher mortality. Breast-cancer mortality increased in black women from 1975 to 1995—a period when breast cancer mortality in white women decreased (NCI, 2010b). Mortality in black women leveled off and began to decrease in 1995 (see Figure 3-1), but in 2005 mortality in black women (32.8 per 100,000) was still higher than in white women (23.3 per 100,000). The disparity is particularly high in black women under 50 years old (Baquet et al., 2008; DeSantis et al., 2008; Ghafoor et al., 2003; Grann et al., 2006). Both incidence and mortality are lower in Hispanic, Asian and Pacific Islander, and American Indian and Alaskan Native women than in white or black women (Ghafoor et al., 2003). Recently, Kinsey and colleagues (2008) examined breast-cancer mortality in black and white women in 1993–2001 as related to 4 levels of education. Mortality decreased by 1.4% in white women who had less than 12 years of education and by 4.3% in white women who had more than 16 years of education. In black women, a decrease (3.8%) was seen only in women who had more than 16 years of education; this shows an association of both race and education with breast-cancer mortality. American Indian and Alaskan Native women are also more likely to receive a diagnosis of late-stage disease than non-Hispanic white women (Wingo et al., 2008). Research has documented that Ashkenazi Jewish women have a genetic susceptibility to breast cancer (Rubinstein, 2004).
The high case-fatality rate from breast cancer in black women had been hypothesized as being due to differences in biologic factors and in access to timely screening and care (Ademuyiwa and Olopade, 2003; Shavers and Brown, 2002). The Carolina Breast Cancer Study showed that basal-like breast tumors were more prevalent among premenopausal African American women than among postmenopausal African American and non–African American women. That suggests a biologic cause of the excess mortality in young black women and leads
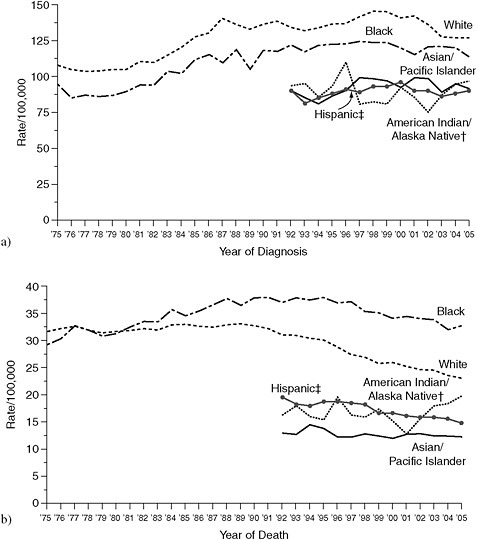
FIGURE 3-1 Annual breast cancer (a) incidence and (b) mortality in the United States by race or ethnicity. Incidence source: Surveillance, Epidemiology, and End Results (SEER) Program, National Cancer Institute (NCI)—1975–1991, SEER 9; 1992–2005, SEER 13. Mortality source: US Mortality Files, National Center for Health Statistics, Centers for Disease Control and Prevention. Rates age-adjusted to the 2000 US standard population (19 age groups—Census P25-1130).
†Rates for American Indians and Alaska Natives based on Contract Health Service Delivery Area counties.
‡Hispanics are not mutually exclusive from whites, blacks, Asians and Pacific Islanders, and American Indians and Alaskan Natives. Incidence data on Hispanics are based on the North American Association of Central Cancer Registries Hispanic Identification Algorithm and exclude cases from the Alaska Native Registry. Mortality data on Hispanics do not include cases from Connecticut, Maine, Maryland, Minnesota, New Hampshire, New York, North Dakota, Oklahoma, and Vermont.
SOURCE: http://www.cdc.gov/cancer/breast/statistics/race.htm (accessed May 3, 2010).
to the option of more aggressive therapies for this patient cohort (see below for discussion of treatment options) (Carey et al., 2006). Issues related to access to screening and care are discussed in Chapter 2; more details on the biology of breast cancer are discussed below.
Research Advances in Knowledge of Biology
Epidemiologic research has identified a variety of factors that are associated with changes in reproductive hormones that are also associated with breast cancer, such as age at first full-term pregnancy, number of full-term pregnancies, breastfeeding, and age at menarche and menopause. Through many types of studies, research has uncovered the role of estrogen in breast-cancer pathogenesis. It is known that estrogen binds to nuclear estrogen receptor α and, with the addition of cofactors, stimulates cell proliferation (Hall and McDonnell, 2005), and it is thus a risk factor for breast cancer. During the last decade, a second estrogen receptor, estrogen receptor β was identified (Kuiper et al., 1996; Mosselman et al., 1996). It is thought that estrogen mediates estrogen–receptor–signaling cross-talk with insulin-like growth-factor receptors to mediate breast-cancer pathogenesis (Clemons and Goss, 2001; Lee et al., 1999).
A family history of breast cancer is also a risk factor for breast cancer, and genetic research has provided an understanding of many of the mechanisms that underlie breast cancer (Hua et al., 2008; Olopade et al., 2008). Mutations in two tumor-suppressor genes—BRCA1 and BRCA2—are associated with breast cancer (Antoniou et al., 2008; Claus et al., 1998; Collins et al., 1995; Easton et al., 1993; Hall et al., 1990; Schubert et al., 1997) and responsible for 5–10% of breast cancers (ACS, 2010a). The germ-line BRCA1 and BRCA2 mutations are highly penetrant and greatly increase a person’s risk of breast cancer (Easton et al., 1993; Rowell et al., 1994). BRCA1 breast cancers are typically poorly differentiated, high-grade, infiltrating ductal carcinomas and are usually estrogen-receptor (ER)–negative, progesterone-receptor–negative, and Human Epidermal Receptor type 2 (HER2)/neu–negative (Bordeleau et al., 2010). BRCA2 breast cancer is characterized by early age of onset, bilaterality, and association with a risk of ovarian cancer (Frank et al., 1998; Krainer et al., 1997). Later research has identified other gene mutations that are associated with an increased risk of breast cancer, including TP53 (the human gene that encodes P53), PTEN, CASP8, FGFR2, MAP3K1, and LSP1 (see Garcia-Closas and Chanock, 2008, for review). Most of those mutations are low-penetrance variants, and much of the genetic component of breast cancer is not accounted for by known gene mutations.
The understanding of the cellular biology of breast cancer has improved over the past 2 decades, contributing to the development of a number of therapies directed toward interrupting pathways in breast-cancer cells for the prevention and treatment of breast cancer. In particular, the roles of a number of receptors in breast-cancer cells have been identified. Approximately two-thirds of breast cancers express ER. There are two types of estrogen receptors (α and β), but at
present only ERα has any known clinical significance. Withdrawal of estrogen (by oophorectomy) was shown to be an effective treatment for breast cancer in the 1890s (Beatson, 1896). Subsequently developed therapies directed toward interrupting the estrogen/ER pathway have been prime tools in treatment and prevention of breast cancer (see below).
HER2, also know as erbB2 and c-neu, is a member of the epidermal growth factor receptor family. Approximately 20–30% of breast cancers have amplified HER2 gene and/or over-express the protein (Cooke et al., 2001; Press et al., 1993; Slamon et al., 1987, 1989; Wolff et al., 2007; Zell et al., 2009). HER2 has been shown to be associated with poorer prognosis in women with newly diagnosed breast cancer, but it is also the target of specifically designed therapeutics directed toward it.
In addition to the ER and HER2 systems, several other important biologic pathways have been identified that have been shown or might serve as therapeutic targets in breast cancer. These include neo-angiogenesis, as mediated by the vascular endothelial growth factor (VEGF). This molecule is the target for bevacizumab, which has activity in the metastatic setting (Miller et al., 2007). Other investigational pathways include, but are not limited to, the insulin-like growth factors (IGFRs), mammalian target of rapamycin (M-TOR), AKT, PI3K, and MEK.
Research Advances in Prevention
Many factors and exposures that are associated with both increasing and decreasing risk of breast cancer can be addressed to help to decrease the incidence of breast cancer, including those presented in Box 3-3.
A major research finding from the WHI was the confirmation of an increased risk of breast cancer associated with the use of conjugated equine estrogen plus progestin (Prempro™) but not with estrogen alone (Premarin™) (Chlebwski et al., 2003; Writing Group for the Women’s Health Initiative Investigators, 2002). The dissemination of that finding resulted in a rapid decrease in the use of menopausal hormone therapy (Haas et al., 2004; Hersh et al., 2004) and a later decrease in breast cancer incidence (Krieger et al., 2010; Ravdin et al., 2007). That decline, however, was not seen equally across all socioeconomic, racial, and ethnic groups (Krieger et al., 2010).
Alcohol, even in moderate amounts, can increase the risk of breast cancer6 (see Suzuki et al., 2008, for meta-analysis), as can poor diet (see Norman et al., 2007, for review), and more specifically, obesity (Brown and Simpson, 2010; Schapira et al., 1994; Vainio and Bianchini, 2002a). Evidence suggests that the common mechanism whereby alcohol and obesity increase the risk of breast can-
|
6 |
Both the adverse and beneficial effects of alcohol consumption are discussed further in Chapter 2. |
|
BOX 3-3 Factors Associated with Breast Cancer Factors Associated with Increased Risk of Breast Cancer Hormone therapy Ionizing radiation Obesity Alcohol Genetic factors Factors Associated with Decreased Risk of Breast Cancer Exercise Early pregnancy Breastfeeding Treatments Associated with Risk of Breast Cancer Selective estrogen-receptor modulators Aromatase inhibitors or inactivators Prophylactic mastectomy SOURCE: National Cancer Institute. http://www.cancer.gov/cancertopics/pdq/prevention/breast/HealthProfessional (accessed August 3, 2010). |
cer is an increase in estrogen, which stimulates the proliferation of breast tissue (Brown and Simpson, 2010; Cleary et al., 2010; Ginsburg et al., 1996). The relationship between smoking and breast cancer is not clear. As reviewed by Coyle (2009), although data on deoxyribonucleic acid (DNA) adducts provide biological plausibility for an association between smoking and breast cancer, epidemiology studies have either shown no association or an inverse association. There is some evidence, however, that smoking during a first pregnancy (Innes and Byers, 2001) and secondhand-smoke exposure are associated with an increased risk of breast cancer (Cal EPA, 2005). The research on relevant behavioral factors is discussed in more detail in Chapter 2.
Exposure to ionizing radiation can increase the risk of breast cancer, especially if it occurs before the age of 20 years (Ronckers et al., 2005). Most studies that showed an increased risk of breast cancer in association with exposure to ionizing radiation looked at radiation levels higher than occur in mammography (Nelson et al., 2009a).
However, exercise, early pregnancy, and number of pregnancies predict a decrease in breast cancer, again with some evidence of a role of estrogen in the altered risk (Bernstein, 2008; Britt et al., 2007; Monninkhof et al., 2007; Pines, 2009).
Preventive measures apart from modifying risk factors have been developed for people at high risk for breast cancer. Recommendations for preventive options for breast cancer depend on a person’s risk (Guarneri and Conte, 2009; Sparano
et al., 2009). In very high-risk people—those who have a germ-line mutation in BRCA1 or BRCA2 with lobular carcinoma in situ and a strong family history of breast cancer—prophylactic mastectomy is an option to consider to reduce the risk of breast cancer (Bermejo-Pérez et al., 2007; Kaas et al., 2010; Nusbaum and Isaacs, 2007; Zakaria and Degnim, 2007), as is prophylactic oophorectomy (Metcalfe, 2009; Rebbeck et al., 2009). For people who have a high risk because of family history, chemoprevention is available. During the last 2 decades, 2 large sequential breast-cancer-prevention trials of healthy women at high risk for breast cancer demonstrated that treatment with tamoxifen (a selective estrogen receptor modulator) for 5 years could reduce the risk of invasive breast cancer by at least 50% (Fisher et al., 1998; Veronesi et al., 2007). Tamoxifen also reduced the risk of recurrent breast cancer after treatment in both younger and older women (Cuzick et al., 2003; Lewis, 2007; Schrag et al., 2000). In a study of postmenopausal women with a mean age of 58.5 years, tamoxifen and raloxifene (a selective estrogen receptor modulator approved for prevention of osteoporosis) had similar efficacy in reducing the risk of invasive breast cancer (Vogel et al., 2006). As a result of that research, older women at high risk for breast cancer now have two US Food and Drug Administration (FDA)–approved medications that reduce the risk of breast cancer and of osteoporosis. Side effects, however, contribute to low acceptance and use of tamoxifen (Fallowfield, 2005). In addition, identifying at-risk people can pose a problem.
Research Advances in Diagnosis
A number of diagnostic and screening methods—screen-film mammography, digital mammography, ultrasonography, magnetic resonance imaging (MRI), and biopsy—can identify breast cancer at earlier stages and facilitate early treatment.
The most widely used imaging technology for breast-cancer screening is mammography. Eight randomized trials evaluated the effectiveness of screening mammography in the United States (Shapiro, 1988; Shapiro et al., 1988), Sweden (Andersson and Janzon, 1997; Bjurstam et al., 2003; Frisell and Lidbrink, 1997; Nystrom et al., 2002; Tabar et al., 1995), Canada (Miller et al., 2000, 2002), and the United Kingdom (Alexander et al., 1999). Although criticisms of those trials have been published (Gotzsche and Olsen, 2000; Olsen and Gotzsche, 2001), independent review concluded that there was strong evidence of the effectiveness of mammography for women over 50 years old (Fletcher and Elmore, 2003; Health Council of the Netherlands, 2002; US Preventive Services Task Force, 2002; Vainio and Bianchini, 2002b). According to a meta-analysis that included all the trials, 15-year mortality from breast cancer in women 50–69 years old was decreased by 20–35%, and the reduction was statistically significant; it was reduced in women between 40–49 years old by about 20% (Fletcher and Elmore, 2003). Results of individual trials and another meta-analysis suggest statistically
significant reductions of 29–44% in the population 40–49 years old (Andersson and Janzon, 1997; Bjurstam et al., 2003; Hendrick et al., 1997). Some researchers have noted that screening mammography is associated with a high rate of false positives and overdiagnosis (that is, diagnosis of and treatment for some cancers that might not progress and cause morbidity or death) (Esserman et al., 2009). Such overdiagnosis results in patients being subjected to adverse effects of breast-cancer treatments and being labeled as having a “preexisting condition,” which can affect insurance coverage7 and raise emotional issues (Esserman et al., 2009). This points to the need to be able to differentiate between tumors that will progress and metastasize from those that will not. Recently, the US Preventive Services Task Force (2009) recommended “against routine screening mammography in women aged 40 to 49 years.” Instead, the task force said the decision to have mammography before the age of 50 years should be an individual choice and take into account individual risks and “the patient’s values regarding specific benefits and harms.” Those guidelines, however, are very controversial, “have had a polarizing effect in the breast-cancer community,” and have led to “confusion, fear, and anger on the part of patients with breast cancer, their families, and women’s health advocates” (Partridge and Winer, 2009). The communication of those guidelines is discussed further in Chapter 5.
The technology associated with mammography has improved substantially since the first studies of its efficacy. Major developments included more-sensitive high-resolution image intensifiers and film, low-absorption cassettes, and dedicated film processors, all of which contributed to radiation-dose reductions for women (Price and Butler, 1970). Changes in mammography tubes (for example, the use of molybdenum targets and filters with beryllium windows and smaller focal spots and the use of moving grids) improved image quality (Haus, 1990; Muntz and Logan, 1979).
Digital mammography was developed to overcome the limitations of screen-film mammography, such as difficulty visualizing low-contrast objects against dense backgrounds (Pisano and Yaffe, 2005; Shtern, 1992). Clinical trials (without death as an end point) have demonstrated that its diagnostic accuracy is equivalent to that of film mammography for the general population (Lewin et al., 2002; Pisano et al., 2005; Skaane et al., 2007; Vinnicombe et al., 2009), but that digital mammography has better accuracy than film in premenopausal and perimenopausal women, women who have dense breasts, and women less than 50 years old (Pisano et al., 2005). As of August 2010, 68.5% of accredited US mammography units were digital (FDA, 2010)—up from 36% in January 2008
(Karellas and Vedantham, 2008)—despite the high relative cost8 (Tosteson et al., 2008). Individualized screening strategies with such technologies as MRI and ultrasonography are being developed for women who are at high risk for breast cancer (Berg, 2009).
MRI provides three-dimensional images of the breast and outstanding soft-tissue contrast. Nine studies (Hagen et al., 2007; Hartman et al., 2004; Kriege et al., 2006; Kuhl et al., 2005; Leach et al., 2005; Lehman et al., 2005, 2007; Sardanelli et al., 2007; Warner et al., 2004) of women who were at very high risk for breast cancer collectively showed an increase in the detection of tumors by combining mammography and MRI for an overall sensitivity of 92.7% and greater detection of smaller, node-negative tumors (see Berg, 2009, for review; Kriege et al., 2004). Those results and others led the American Cancer Society to issue new guidelines for breast-cancer screening with MRI for women who have a 20% or greater lifetime risk of breast cancer9 (Saslow et al., 2007). Many women, however, cannot undergo MRI because of claustrophobia, obesity, renal insufficiency, or the presence of metallic implants (Berg, 2009). In addition, the false-positive MRI results that lead to unnecessary biopsy may limit its acceptability (Tillman et al., 2002).
Sonography is more available, better tolerated, and less expensive than is MRI as a supplemental tool to mammography (Berg, 2009). In high-risk women, mammography combined with sonography has a sensitivity of only 52% compared with 92.7% for mammography with MRI (Berg, 2009; Buchberger et al., 2000; Crystal et al., 2003; Gordon and Goldenberg, 1995; Hartman et al., 2004; Kaplan, 2001; Kelly et al., 2009; Kolb et al., 2002; Kuhl et al., 2005; Leconte et al., 2003; Lehman et al., 2005; Sardanelli et al., 2007; Warner et al., 2004). The supplemental breast-cancer detection rate of sonography in several studies has been consistently reported as 2.7–4.6 per 1,000 women screened (see Berg, 2009; Berg et al., 2008). Cancers found with screening sonography were almost always invasive and node-negative and had a median size of 9–11 mm (Buchberger et al., 2000; Corsetti et al., 2008; Crystal et al., 2003; Gordon and Goldenberg, 1995; Kaplan, 2001; Kolb et al., 2002; Leconte et al., 2003).
Newer technologies—such as tomosynthesis (Gur et al., 2009; Niklason et al., 1997; Poplack et al., 2007), digital subtraction mammography (Diekmann
et al., 2005; Dromain et al., 2006; Jong et al., 2003), dedicated breast computed tomography (Boone et al., 2001, 2006; Yang et al., 2007), positron-emission mammography (Berg et al., 2006), scintimammography (Khalkhali et al., 2000; Liberman et al., 2003), and magnetic resonance spectroscopy (Bartella et al., 2007; Huang et al., 2004; Meisamy et al., 2005)—have not yet been evaluated well enough in a screening setting to warrant their widespread adoption either adjunctively or as replacements for mammography.
It is important to note that all studies of screening with new technologies assess imaging end points, not mortality. The presupposition is that finding more cancers than are found with film mammography (at a less advanced stage) will lead to reduced mortality if implemented on a population-wide basis (Smith et al., 2004). As with other screening methods, there are adverse outcomes associated with false positives and overdiagnosis. In addition to stress and unnecessary biopsies conducted because of false positives, there is evidence that some breast tumors that would not progress to breast cancer are diagnosed as breast cancer through screening programs (Esserman et al., 2009).
Research Advances in Treatment
One of the earliest treatments for breast cancer, surgery with radical mastectomy and complete lymph-node removal, is disfiguring. A randomized clinical trial comparing 5-year survival after mastectomy, lumpectomy (tumor removal only), and lumpectomy with radiation showed that patients who underwent lumpectomy plus radiation had the same survival as those who underwent radical mastectomy (Komaki et al., 1990). Those results gave women options for breast cancer surgery.
Assessing breast-cancer metastases with sentinel lymph-node biopsy (SLNB) began in the middle 1990s, has replaced axillary lymph-node dissection (ALND) for determining the extent of spread of breast cancer, and has shown decreased posttreatment morbidity (Kell and Kerin, 2004; Lyman et al., 2005; Olson et al., 2008; Quan and McCready, 2009; Schrenk et al., 2000). A number of studies have demonstrated that SLNB is as accurate for staging breast cancer and is followed by similar short-term survival alone as in conjunction with ALND (Quan and McCready, 2009); large randomized controlled trials with longer followup are underway to evaluate SLNB further (Quan and McCready, 2009).
Surgery (lumpectomy or mastectomy) remains the primary treatment for breast cancer. Before and after surgery, adjuvant chemotherapy is an important component of breast-cancer treatment (NCI, 2009a). There has been development of many agents and combinations of agents and testing in clinical trials to assess survival and side effects in women who have breast cancer at different stages. The development of effective adjuvant therapies for early-stage breast cancer has greatly improved survival rates.
Several chemotherapeutic agents are available to treat patients with meta-
static breast cancer, resulting in substantial palliation and some survival benefits (Chia et al., 2007). More importantly, several trials have demonstrated that using chemotherapy to prevent recurrences in women with early-stage disease has had an enormous impact on mortality (Early Breast Cancer Trialists’ Collaborative Group, 2005). Early trials were principally focused on cyclophosphamide, methotrexate and 5-flourouracil (Buzdar et al., 1988). Later studies demonstrated that the addition of the anthracyclines (doxorubicin and epirubicin) and the taxanes (paclitaxel and docetaxel) improve outcomes even further (Early Breast Cancer Trialists’ Collaborative Group, 2005; Henderson et al., 2003; Martin et al., 2005).
As mentioned previously, therapies that interrupt the estrogen/ER pathway have been prime tools in treatment and prevention of breast cancer. Of these, the selective ER modulator (SERMs), tamoxifen, has been most influential in much of the decline in breast cancer mortality observed in the Western world over the last 25 years (Osborne, 1998). More recently, complete inhibition of estradiol synthesis in postmenopausal women has been affected by specific aromatase inhibitors (AIs), which are now known to be slightly more effective than tamoxifen in both the metastatic and adjuvant settings (Winer et al., 2005). Tamoxifen, and a similar SERM compound, raloxifene, have both been proven to prevent new ER-positive breast cancers in women at modestly high risk for the disease, and studies are underway to test the worth of AIs in this setting (Fisher et al., 1998; Vogel et al., 2006).
Trastuzumab is a monoclonal antibody that interferes with HER2 and has been shown to reduce mortality in both metastatic and adjuvant settings (Mariani et al., 2009). More recently, studies have demonstrated that a small molecular weight tyrosine kinase inhibitor, lapatinib, has activity in patients with HER2-positive metastatic breast cancer who have progressed on trastuzumab (Di Leo et al., 2008; Geyer et al., 2006). These two agents are now being compared, alone or in combination, in ongoing randomized adjuvant clinical trials.
Increased understanding of the genotypes and phenotypes of different breast cancers has allowed clinicians to individualize treatment in many ways. Studies in the 1970s and 1980s demonstrated that women with ER-positive breast cancer benefit from endocrine treatments, like tamoxifen and, therefore, those treatments should be used in those women (Early Breast Cancer Trialists’ Collaborative Group, 2005). In contrast, patients with HER2-negative breast cancers appear to gain little, if any, benefit from trastuzumab or lapatinib (Press et al., 2008; Slamon and Pegram, 2001). More recently, gene or protein expression assays combining ER, HER2, markers of proliferation, and other factors, have been shown to identify women who might forego chemotherapy or for whom chemotherapy might not work (Albain et al., 2010; Fong et al., 2009; van’t Veer et al., 2005). More recently, studies of tumors that do not express either ER or HER2 have demonstrated that another pathway, the poly (adenosine diphosphate [ADP]–ribose) polymerase pathway, is involved in DNA repair and, therefore, in tumor survival
and in resistance to chemotherapy. In early trials inhibitors of this pathway have been reported to have antitumor activity, and large prospective randomized trials to determine their clinical utility are now underway (Fong et al., 2009).
Genetic research has also helped to move toward personalized medicine for women who have breast cancer. Women with low cytochrome P450 2D6 activity do not effectively metabolize tamoxifen to its active metabolite, and identification of those poor metabolizers helps assess the benefits of tamoxifen in individual breast-cancer patients (Desta et al., 2004; Kiyotani et al., 2010; Rooney et al., 2004). In additional, gene-expression profiles can identify women who will or will not benefit from the use of anthracyclines and other therapies, thus avoiding exposure of women who would not benefit from that toxic class of drugs.
More recently, other agents—such as AIs, which interfere with postmenopausal women’s ability to produce the estrogen—have been shown in large-scale clinical trials to be superior to tamoxifen in extending survival in women who have metastatic disease and in preventing recurrence when used as primary adjuvant therapy (Sparano et al., 2009). In addition, treatment with AIs after a full course of tamoxifen continues to improve recurrence-free survival compared with cessation of hormone therapy (Goss et al., 2003; Winer et al., 2005), and they are approved to treat postmenopausal women for breast cancer (Winer et al., 2005).
Another significant advance was discontinuing the use of an ineffective treatment. Before 2000, bone-marrow transplantation was commonly used in combination with high-dose chemotherapy despite the absence of a randomized controlled trial that demonstrated its efficacy. A randomized controlled trial showed that the combined treatment did not improve survival in women who had metastatic breast cancer (Stadtmauer et al., 2000; Weiss, 1999); the finding was confirmed in other studies (Farquhar et al., 2003). The use of bone-marrow transplantation was abandoned in the late 1990s (Welch and Mogielnicki, 2002).
Knowledge Gaps
Scientific research, spurred by demands by and involvement of breast-cancer survivors and advocates, has improved survival of women who receive a diagnosis of breast cancer (IOM, 2006). There are now about 2.5 million women with a history of breast cancer either living disease free or undergoing treatments (ACS, 2010a). Although research has demonstrated that generally these women recover and lead relatively normal lives, some of the survivors may suffer serious sequelae, such as persistent fatigue, cognitive changes, musculoskeletal aches and pains, sexual difficulties, and secondary malignancies. Sequelae arise from the toxicity of therapies, from the psychological and emotional aspects following treatment (for example, mastectomies), and from concerns over recurrence. Leading-edge research is now focused on understanding the biopsychosocial mechanisms that underlie these persistent problems, and new therapies are being developed to help in their management with a goal of improving the survivors’
lives (Bower, 2008; IOM, 2006, 2008; Miller et al., 2008). Despite those gains, more than 40,000 US women died in 2009 from breast cancer (see Table B-2). In addition, the gains against breast cancer have not been seen among all demographics groups, and the reasons for the higher mortality in black women needs to be better understood and addressed. The ability to differentiate between tumors that will progress and metastasize from those that will not is needed.
Lessons Learned
Breast cancer is an example of a serious disease for which the risks, and consequences have been decreased through advances in scientific research. Research has led to improved overall prevention, detection, survival of, and treatments for breast cancer, but there is a need to focus research programs on quality-of-life issues as well as mortality. If one looks at the overall progress made in the health of women who have breast cancer and at the research findings, the successes can not be attributed to a single aspect of the research but rather to multi-pronged research, including molecular, cellular, and animal experiments; improving diagnostic techniques; implementation of widespread screening programs; observational studies; and clinical trials. The disparities that remain highlight the need to focus research on groups that have the highest risks and burdens of disease. In addition, the increased risk of breast cancer from the use of hormone therapy (conjugated equine estrogen plus progestin) and the lack of efficacy of bone-marrow transplantation point to the need to conduct clinical research to demonstrate the efficacy and safety of treatments before widespread public use.
Cardiovascular Disease
Cardiovascular disease—considered here as a group that includes heart disease and stroke—has seen major progress in women, as reflected in a decrease in mortality. Despite that progress and all that has been learned over the last 2 decades about cardiovascular disease in general, and about the potential differences in cardiovascular disease between women and men, in the United States cardiovascular disease is still the leading cause of death among women of almost all races and ethnicities,10 and it is a major contributor to morbidity and a decrease in quality of life in women (AHA, 2009).
|
10 |
Heart disease is the leading cause of death in white, black, and Hispanic women. It is the second-leading cause of death in Asian and Pacific Islander and in American Indian and Alaska Native women (see Appendix B). |
Incidence, Prevalence, and Mortality in Women
Cardiovascular disease in general used to be thought of more in relation to men than women, and most of the earlier cardiovascular research focused solely on men (AHRQ, 2009), which could be why some women underestimate their risk of cardiovascular disease and overstate their risk of breast cancer (Erblich et al., 2000). Statistics, however, show that cardiovascular disease has been the leading cause of mortality in US women since 1989 (see Appendix B). In 2006, one-third of US women had cardiovascular disease (Lloyd-Jones et al., 2009) and over 3 million women were discharged from short-stay hospitals with their first listed diagnosis as cardiovascular disease (AHA, 2009).
Since 1984, the non–age-adjusted number of deaths in women due to cardiovascular disease has exceeded the number in men; in 2005, nearly 0.5 million women died from cardiovascular disease—52.6% of all people who died from cardiovascular disease (AHA, 2009). Taking age into account, however, shows a different picture. The incidence is lower in women than in men in all age groups, and the prevalence is lower in women than men or the same in women as men between 20–79 years old, but higher in women 80 years old or older (Lloyd-Jones et al., 2009). Although cardiovascular disease in women remains a substantial problem, success can be seen in the recent decreases in mortality from cardiovascular disease. The age-adjusted rate fell by 48.9% in women (from 263.3 to 134.4 per 100,000) and by 50.8% in men (from 542.9 to 266.8 per 100,000) (Ford et al., 2007).
Cardiovascular disease can be classified as coronary heart disease,11 stroke, and non-ischemic heart disease (for example, mitral valve disease). Coronary heart disease and stroke are discussed here as examples of cardiovascular disease in women. The majority of cardiovascular disease in women is coronary heart disease, most of which is caused by atherosclerotic coronary disease or atherosclerosis. Coronary heart disease can manifest as angina (chest discomfort), ischemic heart disease (reduced blood supply to the heart) or acute myocardial infarction (heart attack). In 2006, about 8 million women in the United States were living with coronary heart disease (Lloyd-Jones et al., 2009). Data from the National Health and Nutrition Examination Survey (NHANES) indicate that the prevalence of acute myocardial infarction in women has increased over the last 2 decades but decreased in men (Towfighi et al., 2009). Of women 40 years old or older who have a recognized myocardial infarction, 23% die within a year compared with 18% of men (Lloyd-Jones et al., 2009). Younger, but not older, women have higher mortality during hospitalization after myocardial infarction than do men of the same age. The younger the patients, the higher is women’s mortality relative to men’s (Vaccarino et al., 1999).
Chronic coronary heart disease is a major contributor to heart failure in women. Almost 600,000 women are discharged from short-stay hospitals each year, and 2.5 million women are living with heart failure (AHA, 2009). The number of women living with chronic heart conditions is rising and is expected to continue doing so because of improved acute treatments for coronary heart disease, the aging of the population, and other advances in medical therapies.
Stroke, when considered separately from other cardiovascular conditions, is the third-leading cause of death among women, and about 4 million women survivors are estimated to be alive today (Lloyd-Jones et al., 2009). Each year, about 55,000 more women than men have strokes (Lloyd-Jones et al., 2009). Although it is attributable primarily to women’s longer life expectancy (Lloyd-Jones et al., 2009), more women have strokes even when compared with men in the same age group. One study analyzed NHANES data from 1999–2004 and found that self-reported stroke prevalence in women 45–54 years old was double that of men in the same age group (Towfighi et al., 2007).
Disparities Among Groups
With respect to cardiovascular disease as a group, 46.9% of black women 20 years old and older and 34.4% of white women had cardiovascular disease in 2006 (Lloyd-Jones et al., 2009).
Mortality from coronary heart disease was higher in black women than in white women (141 vs 110 per 100,000 age-adjusted population) in 2005. Black women in all age groups had a higher incidence of first heart attacks and overall heart attacks than white women. In 2006, the prevalence of coronary heart disease in women 20 years old or older was 6.9% in white women, 8.8% in black women, and 6.6% in Mexican American women (Lloyd-Jones et al., 2009).12
Data from 1998 indicate that the age-adjusted mortality rate from coronary heart disease and specifically from acute myocardial infarction is lower in Hispanic, Asian and Pacific Islander, and American Indian and Alaskan Native women than in either black or white women (CDC, 2001). Like mortality from most other cardiovascular diseases, mortality from stroke is substantially higher in black women than in white women (60.7 vs 44.0 per 100,000 age-adjusted population) (AHA, 2009). Low socioeconomic status is also related to higher mortality. Among women 60 years old or older with cardiovascular disease, those without a high school degree were twice as likely to die from their disease as were high school graduates (Lee et al., 2005). In one study that looked at age-adjusted death rates from cardiovascular disease in both black and white women, death rates are higher in those with less education and in those with less income (Pappas et al., 1993).
Research Advances in Knowledge of Biology
Research over the last 20 years has demonstrated that women have cardiovascular disease, and determining whether there are sex differences in cardiovascular disease and underlying biologic differences between women and men that could underlie the differences in disease is an active area of research (Rosenfeld, 2006; Shaw et al., 2009). A recent pooled analysis of data from 11 studies of acute coronary syndrome concluded that women have higher 30-day mortality; this may be largely explained by clinical differences on presentation (for example, women are older and have more comorbidities and risk factors than men) and differences in the severity of angiographically documented disease (Berger et al., 2009).
The Women’s Ischemia Syndrome Evaluation (WISE) study, sponsored by the National Heart, Lung, and Blood Institute, was conducted to evaluate diagnostic tests for heart disease in women and to determine whether evidence of myocardial ischemia occurs in the absence of obstructive coronary disease in women. Data from WISE highlight the role of microvascular dysfunction, subendocardial ischemia, inflammation, genetic predisposition, and neurohormonal imbalance in imparting risk in women (Bairey Merz et al., 2006; Quyyumi, 2006). Of the 7,603 women with symptoms screened, however, 936 (less than 5%) were enrolled in the study; women with a diagnosis of coronary artery disease on angiography and women with a previous coronary event (i.e., myocardial infarction, stroke, or revascularization) were excluded (Gulati et al., 2009). The WISE study only included females so sex-differences cannot be directly assessed. Differences in responses to ischemia are seen at the cellular level; different pathways trigger programmed cell death after ischemia in male and female rats and mice from birth (Bae and Zhang, 2005; Elsasser et al., 2000; Lang and McCullough, 2008; Vannucci et al., 2001). The clinical implications of those findings in humans and animals are unknown.
Research Advances in Prevention
Two-thirds of women who die suddenly from coronary heart disease had no previous symptoms (compared with half of men) (AHA, 2009). That suggests that primary prevention must be a key strategy to reduce the burden of coronary heart disease in women.
Smoking is the leading cause of cardiovascular disease, and the risk decreases quickly on smoking cessation. Hormone therapy (estrogen alone and estrogen plus progestin), as well as the selective estrogen-receptor modulators tamoxifen and raloxifene have been shown in a number of studies—the WHI, the Raloxifene Use for the Heart (RUTH) trial, and the Study of Tamoxifen and Raloxifene (STAR) trial—to increase the risk of stroke or fatal stroke in women (Nelson et al., 2009b; Stefanick, 2006; Writing Group for the Women’s Health Initiative Investigators, 2002). On the basis of the findings of the WHI, which was
designed to study the use of menopausal hormone therapy for primary prevention of cardiovascular disease, menopausal hormone therapy is not recommended to prevent cardiovascular disease (Wassertheil-Smoller et al., 2003; Writing Group for the Women’s Health Initiative Investigators, 2002). Behavioral factors (for example, smoking, eating habits, physical activity) that affect the risk of cardiovascular disease are discussed in Chapter 2.
An important sex difference in the prevention of stroke is the use of aspirin. Aspirin has been shown to prevent ischemic strokes in women (primarily among those over age 65) but not in men (Bailey et al., 2010; Ridker et al., 2005). The risk of hemorraghic stroke and gastrointestinal bleeding, however, may be increased by aspirin use, and because women often have uncontrolled blood pressure and stroke as they age, stroke and bleeding are particularly important health issues for older women (Bailey et al., 2010; Ridker et al., 2005). National guidelines recommend that those risks be weighed against the benefits of aspirin, and that age be taken into account in decisions about aspirin chemoprevention (Mosca et al., 2007).
In an early clinical trial, statins (3-hydroxy-3-methyl-glutaryl coenzyme A reductase inhibitors) were shown to be effective in lowering cholesterol in a Scottish trial in men (Shepherd et al., 1995). The absence of women in that and other statin trials led to questions about extrapolating the data to women and thus a delay in their use in women, even those who had previously had a coronary event. A meta-analysis of data from those trials later demonstrated the efficacy of statins in women (LaRosa et al., 1999). A large-scale trial (JUPITER) showed that primary prevention benefits from statins are similar in women 60 years old or older and men 50 years old or older (Mora et al., 2010), however, the number needed to treat (that is, the number of patients who would have to be treated to prevent a single outcome event) and side effects might be higher in women than men (Ridker et al., 2009). A 2009 meta-analysis showed benefits of statin therapy in both women and men at risk for cardiovascular disease (Brugts et al., 2009), and a meta-analysis of trials (not specifically in women) comparing early statin therapy after acute coronary syndrome with placebo or usual care at 1 and 4 months following showed no reduction in deaths, myocardial infarction, or stroke with statin therapy (Briel et al., 2006). A prospective cohort study showed an increased risk of cataracts, kidney failure, and liver dysfunction in both men and women with statin treatment (Hippisley-Cox and Coupland, 2010). Further research is needed to define the risk–benefit ratio of statins for primary prevention in diverse populations of women with varying risks of cardiovascular disease.
Patient, physician, health-system, and societal factors can all contribute to gender and racial disparities in cardiovascular-disease outcomes, but their relative contribution is not known. In a survey of 500 physicians (300 primary care physicians, 100 obstetricians/gynecologists, and 100 cardiologists), primary care physicians were significantly more likely to place women, who according to their Framingham risk score were in an intermediate-risk category, in a lower risk category than they did for men (Mosca et al., 2005). That rating affected the
recommendations the physicians provided for lifestyle and preventive pharmacotherapy. Earlier studies also indicated that physicians may manage women’s chest pain less aggressively, particularly black women (Schulman et al., 1999). Gender-based disparities in cardiovascular care have been documented in commercial health plans and the greatest disparity is present among those who had recent acute cardiac events (Chou et al., 2007a,b).
The American Heart Association (AHA) reviewed what is known about the use and effectiveness of percutaneous coronary interventions and adjunctive pharmacotherapy in men and women and concluded that invasive percutaneous coronary interventions are “performed less frequently and with greater delays in women” (Lansky et al., 2005). Rates of reperfusion therapy are also lower in women than in men, and “there is no evidence that the gap has narrowed in recent years” (Vaccarino et al., 2005). Greater complications and early mortality have been detected in women as compared to men following revascularization (bypass surgery and percutaneous coronary interventions) and, therefore, the lower number of procedures might be beneficial to women (Kim et al., 2007).
Studies have shown a higher risk of death or acute myocardial infarction in women who have unstable angina and an increase in non–ST-segment myocardial infarctions in women after invasive treatment than in women after conservative treatment.13 A meta-analysis of eight trials indicated that invasive strategies benefit high-risk women—that is, those who have increased concentrations of the biomarkers creatine kinase MB or troponin—but do not benefit and possibly increase risk in women who do not have increased concentrations of those biomarkers (O’Donoghue et al., 2008). Similarly, invasive treatment has been shown to benefit high-risk women who have acute coronary syndrome, but study results indicate no benefits of and even harm after invasive treatment in non–high-risk women who have acute coronary syndrome (Lansky et al., 2005).
AHA published sex-specific evidence-based guidelines for the prevention of cardiovascular disease in women in 2004 (Mosca et al., 2004), but the extent to which they have changed practice is not established. Most physicians are aware of the guidelines, but few state that they implement the guidelines (Mieres et al., 2005).
Research Advances in Diagnosis
Sex and gender differences in the presentation of cardiovascular disease have been studied over the last 2 decades, including studies looking for differences in clinical features (Canto et al., 2007; Correa-de-Araujo, 2006; Correa-de-Araujo and Clancy, 2006; DeCara, 2003; Dracup, 2007). The initial presentation of coronary heart disease is about 10 years later in women than in men (Mikhail, 2005). For myocardial infarction, the most common symptom for women is chest
pain, although myocardial infarction in women does occur in the absence of chest pain (AHA, 2010). Other symptoms of myocardial infarction include feeling out of breath; pain that runs along the neck, jaw, or upper back; nausea; vomiting or indigestion; unexplained sweating; sudden or overwhelming fatigue; and dizziness (AHA, 2010). Statistically, women who have myocardial infarctions are less likely than men to have coronary disease (Shaw et al., 2009). That chest pain is more common in men and is considered a “typical” symptom of heart disease may contribute to the finding that women are less likely to undergo diagnostic evaluation for symptoms and may have their conditions misdiagnosed (Brieger et al., 2004; Canto et al., 2007). Some evidence suggests that women and men experience cardiac pain differently, and that this affects the diagnosis of cardiovascular conditions and events (O’Keefe-McCarthy, 2008).
Women who have coronary heart disease are more likely to present with angina and fatigue, nausea and vomiting, and shortness of breath, whereas men are more likely to present with acute myocardial infarction or coronary heart disease death (DeCara, 2003). Women also have more atypical chest pain related to angina than men and more nausea, back pain, and jaw pain (Brieger et al., 2004; Canto et al., 2007; Kudenchuk et al., 1996; Milner et al., 1999).
Women and men vary in the predictive strength of risk factors, and this complicates the diagnosis of coronary arterial disease in women. Diabetes has been shown to be a stronger predictor of risk in women than in men (Scheidt-Nave et al., 1991). In a study of premenopausal women who underwent coronary angiography for suspected ischemia, diabetes was associated with hypothalamic hypoestrogenemia, increased prevalence and severity of angiographic coronary artery disease, and a slight increased risk of major adverse cardiovascular events (Ahmed et al., 2008). Isolated systolic hypertension is more common and more predictive in women than in men (Rich-Edwards et al., 1995), whereas a high concentration of low-density lipoproteins is more predictive of coronary arterial disease in men than in women (Rich-Edwards et al., 1995). An increase in triglycerides is a risk factor for cardiovascular disease in women (Evangelista and McLaughlin, 2009).
C-reactive protein has been suggested for use as a risk marker, particularly in women (Cook et al., 2006; Ridker et al., 2003). Research on the clinical relevance of C-reactive protein is ongoing. The US Preventative Task Force concluded that although data are convincing that C-reactive protein is associated with coronary heart disease, evidence that its use as a risk marker improves risk estimates is weak, and evidence that reducing C-reactive protein levels protects against coronary heart disease is lacking for either women or men (Buckley et al., 2009).
Sex differences have sometimes been reported in the sensitivity and specificity of diagnostic tests for cardiovascular disease. Kim and colleagues (2001) conducted a meta-analysis and found sex differences in the diagnostics. An AHA consensus statement in 2005 concluded that the present approach to diagnostic testing may require some variation when applied to women (Mieres et al., 2005). DeCara (2003) reviewed noninvasive cardiac testing in women and found a high
rate of false positives for coronary arterial disease with exercise electrocardiographic (ECG) stress testing in women (Hung et al., 1984; Mieres et al., 2005) and female-specific outcomes have been developed for exercise ECGs (Gulati et al., 2005). A systematic review for AHRQ, however, did not find sex differences in the accuracy of exercise myocardial perfusion imaging for diagnosis of coronary heart disease and found little difference in the accuracy of exercise myocardial perfusion imaging and exercise echocardiography for diagnosis of coronary heart disease in women (Grady et al., 2003).
Differences in the diagnosis of cardiovascular disease in women, if present, could bias the results of clinical trials. If trials are based on symptoms of coronary heart disease and cardiovascular disease that are more commonly seen in men than in women, and female cases will be missed.
Research Advances in Treatment
Research laid the groundwork for a number of important pharmacologic breakthroughs in treating patients with cardiovascular disease. The use of beta-blockers and aspirin as soon as possible after a myocardial infarction quickly became the standard of care for men; however, women were not receiving that care and it took a few years for the use of beta-blockers and aspirin in women to approach that in men (Berger et al., 2009). The use of stents in women lagged behnd their use in men because the size of the stent was based on male blood vessels, which are typically larger than in women (Lansky et al., 2005).
As summarized by Lansky and colleagues (2005), the current use of stents does not appear to differ between the sexes. In addition, the mortality associated with their use is similar in women and men unless confounding risk factors are present in women (Chauhan et al., 2005; Mehilli et al., 2000).
Research has demonstrated that adjunctive pharmacotherapy is beneficial in women who are undergoing percutaneous coronary intervention as secondary prevention, including the use of aspirin, ADP–receptor antagonist antiplatelet agents (clopidogrel and ticlopidine), glycoprotein (GP) IIb/IIa inhibitors, the antithrombin agents unfractionated heparin and low-molecular-weight heparin, and direct thrombin inhibitors (Antithrombotic Trialists Collaboration, 2002; Braunwald et al., 2002; Fernandes et al., 2002; Kong et al., 2002; Steinhubl et al., 1999; Stone et al., 2002; Wong et al., 2003; Yusuf et al., 2003). Because of women’s risk of bleeding complications at baseline (Lenderink et al., 2004), and because of risks of overdosing with GP IIb/IIIa inhibitors in women, care must be taken to avoid complications (Alexander et al., 2006).
Knowledge Gaps
Despite research advances in scientific knowledge related to diagnosis, risk factors, preventive interventions, and effective therapies for coronary heart disease in women, progress might have happened sooner if women had been better
represented in earlier studies of cardiovascular disease. More recently women have been enrolled in cardiovascular trials; however, a lack of knowledge about sex differences remains, in part because of a lack of sex-specific analysis and reporting of sex-specific results, with only 25% of trials of cardiovascular disease reporting on sex-specific results (Blauwet and Redberg, 2007; Blauwet et al., 2007; Grady et al., 2003). Because women have been consistently under-enrolled in cardiovascular-disease clinical trials (Grady et al., 2003; Kim and Menon, 2009; Sharpe, 2002), studies were not powered sufficiently to provide statistical significance on data for women, so meta-analyses have been used to determine whether the results obtained in men can be extrapolated to women (Grady et al., 2003). Because of their limitations, however, meta-analytic methods can not overcome a lack of enrollment or data on women, and they are not optimal for addressing the leading cause of mortality in women. A major limitation of evaluating data from eligible studies was that findings were often not stratified by sex, and little evidence was available to answer key questions about the disease in women (and in racial and ethnic minorities). Women continue to be inadequately represented in cardiovascular-disease clinical trials; this could be due in part to entry criteria that are based on symptoms seen more commonly in men (Grady et al., 2003). Cardiovascular disease may be a category in which sex-specific studies can be used to fill in the gaps (Lansky et al., 2005; O’Donoghue et al., 2008; Shaw et al., 2009).
Much also remains both to be learned about the biologic sex differences that underlie cardiovascular disease in men and to be done to use the information to develop sex-specific diagnostic, preventive, therapeutic, and rehabilitative approaches. Sex-specific diagnostic tools, especially for identifying subclinical disease, are needed as is a strategy for avoiding the delay in screening for, diagnosis of, and treatment of cardiovascular disease in women. Furthermore, the reasons for the disparities in mortality in different groups of women and how to address those disparities are also needed.
Although awareness of cardiovascular disease as the leading cause of death has nearly doubled among women since national educational programs, such as the Heart Truth and Red Dress campaigns, have been targeted to women—in 1997, only 30% of women recognized cardiovascular disease as the leading killer of women, significantly less than the 57% and 54% of women who recognized cardiovascular disease as the leading killer of women in 2006 and 2009, respectively (Mosca et al., 2010)—but awareness continues to lag among racial and ethnic minorities. Work is needed to raise awareness of the problem of cardiovascular diseases among women and their health-care providers, especially because awareness of cardiovascular-disease risk has been linked to the taking of preventive action (Christian et al., 2007; Mosca et al., 2006). Translation and communication issues are discussed in Chapter 5.
Lessons Learned
A major lesson was the recognition, not only by researchers but also by clinicians and the public, that cardiovascular disease is a major cause of morbidity and mortality in women. Part of the delay in recognizing that was lack of awareness, but other factors in the delay might be related to potential sex differences in the presentation (for example, age at presentation) of cardiovascular disease in women and men. That highlights the importance of recognizing the signs and symptoms of a disease in women, and ensuring that health practitioners and women are aware of those signs and symptoms. Sex-specific research on cardiovascular disease has shown potential sex-specific differences that affect everything from risk factors to diagnosis to treatment. Evidence indicates that behavioral factors are important for the prevention of cardiovascular disease (see Chapter 2 for a more detailed discussion), so research needs to go beyond the pathophysiology of the disease to the level of identifying effective interventions to modify people’s behaviors to prevent disease. Comorbidities with cardiovascular disease have also been seen and highlight the impact that comorbidities can have on diagnosis and treatment. Another major lesson is the importance of the translation of research findings into practice and policies to benefit all.
Cervical Cancer
The committee considered cervical cancer to be a disease where research findings have led to major advances in prevention and detection of the disease. There have been large decreases in incidence of and mortality from cervical cancer in the United States, mostly because of the use of the Papanicolaou (Pap) smear and the Bethesda rating system, both of which were developed before the period of our review. Cervical-cancer incidence and mortality continued to decrease during the last 20 years, and there are now improved treatment options for early-stage cervical cancer. Cervical-cancer research has also resulted in the development of a vaccine against human papilloma virus (HPV), an infectious agent that causes most cases of cervical cancer. The vaccine has the potential to protect women against cervical cancer, the leading cause of cancer death in women worldwide.
Incidence, Prevalence, and Mortality in Women
Cervical cancer was once one of the leading causes of cancer death in women in the United States, but its incidence and mortality in the United States decreased by about 74% from 1955 to 1992 and continues to decrease (ACS, 2009b). In the United States in 2010, it is estimated that 12,200 women will be diagnosed with cervical cancer, and 4,210 women will die from it (NCI, 2010c). The decreases in incidence and mortality are attributed mainly to regular cytology-based cervical-
cancer prevention programs, which were introduced in the United States in the 1960s (Wright, 2007; Zeferino and Derchain, 2006). In 2002–2007, the median age at diagnosis in the United States was 48 years; the median age at death was 57 years (NCI, 2010c).
Disparities Among Groups
Cervical-cancer incidence and mortality decreased in the United States from 1996 to 2005 in all races and ethnicities on which there are surveillance data (see Figure 3-2) (NCI, 2008). Disparities among races and ethnicities persist despite those gains (Barnholtz-Sloan et al., 2009). The age-adjusted average incidence (cases per 100,000) in 2001–2005 was highest, at 13.7, in Hispanic women, followed by 10.8 in black women and about 8 in white and Asian and
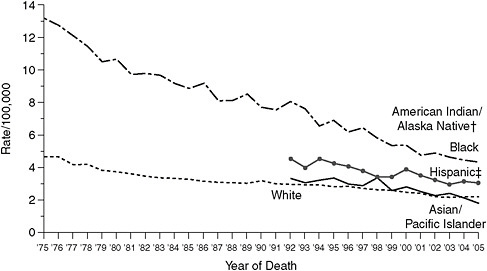
FIGURE 3-2 Cervical-cancer mortality in 1975–2005 by race.
Mortality source: US Mortality Files, National Center for Health Statistics, Centers for Disease Control and Prevention.
Rates per 100,000 and age-adjusted to 2000 US standard population (19 age groups—Census P25-1130).
†Rates for American Indians and Alaska Natives not displayed, because fewer than 16 cases were reported for at least 1 year within interval.
‡Hispanics not mutually exclusive from whites, blacks, Asians and Pacific Islanders, and American Indians and Alaska Natives. Mortality data on Hispanics do not include cases from Connecticut, Maine, Maryland, Minnesota, New Hampshire, New York, North Dakota, Oklahoma, and Vermont.
SOURCE: http://www.cdc.gov/cancer/cervical/statistics/race.htm (accessed May 5, 2010).
Pacific Islander women. In contrast, the average mortality (deaths per 100,000) in the same years was highest in black women at 4.7, followed by 3.2 in Hispanic women and 2.0 in white and Asian and Pacific Islander women. Possible reasons why advances are not translated to decreased mortality among all populations of women are discussed in Chapter 5. Incidence varies within racial and ethnic categories. For example, although overall Asian women are less likely to receive a diagnosis of cervical cancer than white women, within this subgroup Vietnamese American women are approximately 5 times more likely to be diagnosed with cervical cancer than white women (Taylor et al., 2004).
Research Advances in Knowledge of Biology
Epidemiologic, animal, and molecular studies have elucidated the role of HPV in cervical cancer and laid the groundwork for the novel diagnostic techniques and development of a vaccine discussed below and, ultimately, for the prevention of cervical cancer (Lehtinen and Paavonen, 2004; Olsson et al., 2009; zur Hausen, 2009).
The association between HPV type 16 and cervical cancer was found over 2 decades ago (Dürst et al., 1983, 1987). Research during the 1990s and early 2000s demonstrated that HPV is present in over 90% of premalignant cervical lesions and over 95% of cervical cancers and confirmed HPV as causal and necessary for cervical cancer (Bosch et al., 2002; Ferenczy and Franco, 2002; Franco et al., 2001; Muñoz, 2000; Muñoz et al., 2003; Walboomers et al., 1999). HPV 16 and 18 are responsible for cervical cancer (Schlecht et al., 2001; Woodman et al., 2007). Muñoz and colleagues (2003) demonstrated that HPV 16, 18, 31, 33, 35, 45, 52, and 58 account for 95% of squamous-cell carcinomas that are positive for HPV. Researchers also examined the structure of the virus particles; engineered proteins, called the L1 proteins, that reform to virus-like particles (or capsids); and demonstrated that the reformed L1 virus-like particles trigger an immune response in animals (Breitburd et al., 1995; Campo, 2002; Kirnbauer et al., 1992, 1993; Suzich et al., 1995; Zhou et al., 1991). The improved understanding of the relationship between HPV and cervical cancer laid the groundwork for the development of an HPV vaccine.
Research Advances in Prevention
It has been noted for centuries that cervical cancer is associated with sexual activity and sexual contacts (see Chapter 2 for discussion of sexual risk behavior) (zur Hausen, 2009), and limiting the number of sexual partners and using condoms have long been recommended for primary prevention of cervical cancer. Because sexual activity is a risk factor for cervical cancer, behaviors during teenage years and early adulthood can affect the risk of cervical cancer later in
life. There is recent evidence that condom use greatly reduces the risk of genital HPV infections (Winer et al., 2006).
The identification of the HPV virus as the causative agent in cervical cancer and the characterization of the virus and its components provided the basis of the development of a vaccine for the primary prevention of HPV infection and cervical cancer (Kulski et al., 1998). After trials to test safety (Harro et al., 2001) and antibody response (Carter et al., 2000; Petter et al., 2000), Koutsky and colleagues (2002) conducted a randomized controlled trial of an HPV 16 L1 virus-like particle vaccine in 2,392 women. A median of 17.4 months later, the vaccine had 100% efficacy in protecting against HPV 16 infection.14 Harper and colleagues (2004) conducted a randomized controlled trial of the efficacy of a bivalent HPV 16/18 virus-like particle vaccine against HPV 16 and HPV 18 infections in 1,113 women 15–25 years old. The vaccine was over 90% effective against incident and persistent infection with HPV 16/18. Relatively few minor and no serious adverse events were reported.
Gardasil®, a prophylactic vaccine containing the virus-like particle for HPV 6/11/16/18, has since been approved by FDA for girls and women 9–26 years old (FDA, 2008). Studies with the vaccine demonstrated safety; relatively few adverse events were reported. In one study the vaccine prevented 100% of cervical intraepithelial neoplasia (CIN), adenocarcinoma in situ or cancer, and vaginal, vulvar, perineal, and perianal intraepithelial lesions associated with vaccine-type HPV in women with no evidence of previous HPV infection (Garland et al., 2007). When the vaccine was administered to subjects who had no evidence of previous exposure to either HPV 16 or 18, the prophylactic HPV vaccine was 98% effective in preventing HPV 16– and 18–related CIN 2/3 and adenocarcinoma in situ (FUTURE II Study Group, 2007). In women with evidence of HPV infection, regardless of the type of HPV infection, the vaccine reduced the incidence of vulvar, vaginal, and perianal lesions by 34% and of cervical lesions by 20% (Garland et al., 2007). The efficacy of the vaccine in preventing HPV 16– or 18–related CIN2/3 and adenocarcinoma in situ was lower (44%) in women who had been previously exposed to HPV types 6/11/16/18. The estimated efficacy of the vaccine against all high-grade cervical lesions regardless of the causal HPV type was 17% (FUTURE II Study Group, 2007). In October 2009 FDA approved Cervarix™ an additional accine to prevent cervical cancer and precancerous lesions caused by HPV types 16 and 18 (FDA, 2009a).
In October 2009 FDA approved one form of the vaccine in boys and men aged 9–26 for prevention of genital warts (HHS, 2009). The approval did not address the prevention of transmission of HPV to girls and women, and there has been much debate on whether boys and men should be vaccinated to decrease the prevalence of HPV in girls and women (Campos-Outcalt, 2009; Cuschieri, 2009; Hibbitts, 2009; Hull and Caplan, 2009).
Despite the evidence indicating safety and efficacy, a number of controversial issues have arisen around the implementation of HPV vaccination programs. Those issues are discussed in Chapter 5.
Although HPV infection is the major risk factor for cervical cancer, other risk factors have been studied. In general, diet does not appear to modify risks greatly. Diets rich in beta-carotene, or high in vegetables, however, have been linked to lower risk in a few studies (Hirose et al., 1998; La Vecchia et al., 1988), and antioxidants, such as carotenoids, have been shown to decrease HPV persistence in women (Giuliano et al., 1997, 2003). Smoking is associated with an increased risk of cervical cancer, although the extent to which the association is independent of HPV is not known (CDC, 2002a). Some research studies have shown an increased risk of cervical cancer associated with exposure to second-hand smoke (Tay and Tay, 2004; Trimble et al., 2005), but most reviews have concluded that there is inadequate or only suggestive evidence of an association (Office of Environmental Health Hazard Assessment, 2005; Office of the Surgeon General, 2006).
Research Advances in Diagnosis
The consistent use of conventional Pap tests (in which cervical cells are smeared on a slide) for decades in the United States to screen for cervical cancer and precancerous lesions has drastically reduced the incidence of and mortality from cervical cancer (Jansson et al., 1998; Mathew and George, 2009; Schiffman and Castle, 2005; Sherman et al., 2005). Revised screening techniques have the goal of improving the sensitivity (false negatives), and specificity (false positives) of screening, which are suboptimal because of sampling, preparation, detection, and interpretation (Siebers et al., 2009). Liquid preparations allow cytologic screening, computer-assisted reading, and testing for HPV DNA using a single sample preparation (Sherman et al., 1997). In 1996, FDA approved the first liquid-based medium for gynecologic cytology, ThinPrep® (Guidos and Selvaggi, 1999). Early studies showed that liquid-based cytology was at least as good as the conventional Pap test in detecting low-grade squamous intraepithelial lesions and other epithelial-cell abnormalities (Austin and Ramzy, 1998; Bishop et al., 1998; Linder, 1998; Sherman et al., 1998; Vassilakos et al., 1998). Other studies, however, did not yield convincing evidence of improved results of liquid-based cytology, and systematic reviews cited a lack of well-designed comparative studies (that is, studies that used randomized design, biopsy-confirmed cervical lesions, blinded verification, and adequate power) to determine which method was superior (Arbyn et al., 2008; Davey et al., 2006; Klinkhamer et al., 2003). A recent randomized controlled trial that evaluated over 84,000 Dutch women showed that liquid-based cytology does not detect cervical-cancer precursors better than the conventional Pap test (Siebers et al., 2009). The American Congress of Obstetricians and Gynecologists (ACOG) concluded that liquid-based and conventional
methods of cervical cytology are both acceptable for use in testing (ACOG, 2003), and the American Cancer Society recommends the conventional Pap test or liquid-based cytology with different frequencies depending on age (ACS, 2009c). In recently released evidence-based guidelines, ACOG increased the recommended age at first screening and decreased the recommended frequency of cervical-cancer screening but not of pelvic examination (ACOG, 2009).
With the identification of the HPV types responsible for most cervical cancers, high-throughput DNA testing provides DNA diagnostic tests for HPV types associated with cervical cancer. Several FDA-approved HPV DNA diagnostic tests for high-risk types of HPV are effective in detecting HPV. Current guidelines for the management of women with abnormal cervical-cancer screening results have incorporated the use of HPV DNA testing as an adjunct to cytologic screening in their algorithms (Wright et al., 2007). Whether HPV DNA testing can replace cervical cytology remains to be seen.
Research Advances in Treatment
Precancerous lesions are often detected through routine Pap tests and affected cells are removed or destroyed before the development of cervical cancer (for example, with a loop electrosurgical excision procedure, cryosurgery, laser surgery, or cone biopsy).
Once a woman receives a diagnosis of cervical cancer, she can have surgery, radiation therapy, chemotherapy, or some combination of the three. The choice depends on the stage at diagnosis and on patient and physician preference. Clinical trials over the last 2 decades have provided information on the comparative efficacy and toxicity of different treatment regimens to allow evidence-based decisions on treatment protocols and improved quality of life after treatment for some stages of the disease.
Hysterectomy and radical hysterectomy, which were the standard treatments for early-stage (non-metastisized) cervical cancer, have good survival rates but were associated with major morbidity and eliminated the possibility of pregnancy (Chan and Naik, 2008). Over the last 2 decades, research has demonstrated the effectiveness of other surgical procedures or combination therapies that provide a better posttreatment quality of life than radical hysterectomy (Chan and Naik, 2008). For example, trachelectomy (removal of the cervix) is a fertility-sparing surgical option for younger women who have the disease, and laparoscopic radical hysterectomy has the potential of better postoperative recovery and cosmetics than traditional radical hysterectomy (Chan and Naik, 2008). Improvements in imaging also inform decision making for individual cases and allow more specific surgical treatments for localized advanced cancer (Sundar et al., 2005). Despite that progress, however, White (2008) noted that late effects of cervical-cancer treatments include bowel and bladder dysfunction and sexual difficulties.
Historically, radiation therapy (external radiation or brachytherapy where the
radioactive material is sealed in a container or wire and is placed directly into or near the tumor) was not given in combination with chemotherapy, but clinical trials demonstrated the advantage of administering cisplatin-based chemotherapy during radiation in some patients who had stage I or more advanced cervical cancer (Gibbons and Keys, 2000; Lukka et al., 2002; Morris et al., 1999; Peters et al., 2000; Rose et al., 1999). Increased toxicity, however, is seen with the addition of chemotherapy (Eifel, 2006; Loizzi et al., 2008).
Chemotherapeutic options for recurrent and metastatic cervical cancer have been reviewed (Moore, 2006; Tao et al., 2008). Cisplatin chemotherapy has been used since the 1980s against advanced cervical cancer in people who cannot be treated with surgery or radiation and against recurrent cervical cancer (Alberts and Mason-Liddil, 1989). The prognosis with that treatment is poor; its goal is to slow the progression of the disease (that is, palliation), and it has high toxicity. During the 1990s, other platin analogues and combination therapies that contained cisplatin and non–platin-related compounds (for example, mitolactol and ifosfamide) were used in clinical trials with the hope of improving survival and decreasing the toxicity associated with chemotherapy, but they had little success (Moore, 2006). Trials of combinations of other chemotherapeutic agents (such as paclitaxel and topotecan) with cisplatin in the 1990s and early 2000s showed promising results, including a decrease in toxicity and, in the case of topotecan, an increase in progression-free survival compared with cisplatin alone (Moore et al., 2004; Rose et al., 1999), but overall survival was unchanged (McQuellon et al., 2006; Moore et al., 2004). More recent research is investigating agents that target the vasculature of tumors and is developing drugs that target molecular pathways that are abnormal in cancer cells, including cervical cancer (Herrera et al., 2008). In addition, some HPV vaccines are showing potential as therapeutic agents by inducing HPV-specific antitumor immune responses in animal models (Hung et al., 2008).
Despite the research, the prognosis of late-stage cervical cancer is poor, and stage IVB cervical cancer is usually considered incurable (ACS, 2009b). As Moore (2006) pointed out, it is of note that the study by McGuire and colleagues (1996) used validated instruments to assess patient-reported quality of life and was the first randomized controlled study of palliative treatments for advanced cervical cancer to consider quality of life as an end point. Given the palliative nature of the treatments, the toxicity associated with the agents, and the importance of quality of life to women, quality of life is an important consideration in the development of future treatment, as it is in relation to other conditions and treatments. Greater attention to the impact of treatments on quality of life may help women make more informed choices among options for treatment and increase the effectiveness of the treatment they choose.
Knowledge Gaps
Despite the wide use of cytology-based screening, cervical cancer continues to be an important issue among women in the United States, especially among particular racial and ethnic groups. The biggest limitation of cervical cytology is its poor sensitivity, and liquid-based cytology has not improved sensitivity (Ronco et al., 2006).
Advances in treatments have decreased posttreatment morbidity associated with early-stage cervical cancer, but a lack of effective treatments for advanced and recurrent cervical cancer leaves 5-year survival in these cases extremely rare. In addition, research is needed to understand the factors that contribute to acceptance and use of the HPV vaccine, especially in miniority groups at the greatest risk for cervical cancer, since prevention of this disease is now possible (see Chapter 5 for further discussion).
Lessons Learned
The decreases in the incidence of and mortality from cervical cancer highlight the importance of the availability of appropriate diagnostic tests and screening for the prevention of or early treatment for some diseases. The combination of epidemiologic, animal, and cellular research on the etiology of cervical cancer, the role of HPV in cervical cancer, and an understanding of the disease and associated virus itself allowed the development of a vaccine against this cancer. The identification of quality of life as an important outcome in evaluating a therapeutic agent is another important lesson from the research on cervical cancer. The higher incidence and mortality still seen in some populations highlights the need to target translation and communication efforts to reach people who are at greatest risk for the disease.
CONDITIONS ON WHICH RESEARCH HAS CONTRIBUTED TO SOME PROGRESS
Depression
There has been some progress on the development of improved therapies for the treatment for depression over the last 2 decades. Shortcomings in diagnosis and in the use of those therapies, however, have limited the implementation of those treatments and diminished progress against the disease.
Incidence, Prevalence, and Mortality in Women
The prevalence of major depression in the United States is estimated to be 16% over a lifetime (Kessler et al., 2003). The rate in women is estimated to
be up to twice that in men (Kessler et al., 2003). The sex difference occurs in relation to the onset of depression (Marcus et al., 2005); chronicity and recurrence are similar between the sexes. Sex differences first emerge at the ages of 11–14 years and persist into adulthood (Angold et al., 1998). Pregnant women and nonpregnant controls have similar rates of depression (Gotlib et al., 1989), although the rates of major depression may increase during the postpartum period (Gotlib et al., 1989; Wisner et al., 1993). Women who have a strong family history of depression have a much higher risk of onset of first depression during the postpartum period (Sichel and Drischoll, 1999).
The etiology of gender differences in depression has been studied to help understand the greater vulnerability of women to depression. Stressful life events lead to an increased risk for depression (Mazure, 1998). Although it is not clear whether or not women experience more stressful life events than do men, women do experience more physical and sexual abuse than do men (Koss et al., 2003). Abuse is a risk factor for depression not only just after the abuse occurs, but throughout the lifetime following such abuse (Weiss et al., 1999). The lower social status of women compared with men is also implicated in rates of depression. Poverty is the chronic stressor most often associated with depression (Belle and Doucet, 2003). Women are more likely to live in poverty than are men. In addition, women appear more reactive to stressful life events than are men. Specifically, women are more likely to experience depression after a stressful life event than are men (Kessler and McLeod, 1984; Nazroo et al., 1997; Uhlenhuth and Paykel, 1973). Finally, two personality factors may predispose women to depression more than men. Women are more likely to score higher on measures on interpersonal dependency than do men, and such dependency is related to depression (Clark et al., 1992; Hammen, 1999; Mazure et al., 2000). Also, women tend to ruminate more in response to sadness than do men (Nolen-Hoeksema et al., 1999; Tamres et al., 2002). Those who ruminate are more likely to be diagnosed with major depression than are those who do not ruminate (Nolen-Hoeksema, 2004). In sum, women’s socioeconomic and social status may influence women’s increased likelihood of becoming depressed through abuse, chronic stress, and interpersonal dependency.
In both women and men, depression often coexists with other psychiatric disorders (such as anxiety disorders), with substance abuse, and with medical illnesses, such as cancer, HIV/AIDS, and diabetes. Depression can worsen the symptoms of those illnesses (NIMH, 2009).
Because of its high prevalence and its onset early in life, depression is estimated to be a leading cause of disease-related disability in women throughout the world (Murray and Lopez, 1997). The early onset contributes to problematic early-life decisions, poor parenting, lower work functioning, and intense personal pain in young women. Data on people who have early-onset major depression indicate that it adversely affects the educational attainment of women but not of men (Berndt et al., 2000). For example, “a randomly selected 21-year-old woman
who had early-onset major depression in 1995 could have expected to earn 12–18% less per year than a randomly selected woman whose major depression occurred after the age of 21 years” (Berndt et al., 2000).
Disparities Among Groups
Rates of depression differ by ethnicity. Rates are lower among Latinos, blacks, Asians, and American Indians than among whites; differences might be due, at least in part, to underdetection (Alegría et al., 2008; Beals et al., 2005; Takeuchi et al., 2007; Williams et al., 2007). Among Mexican Americans, rates of depression in immigrants are lower than in those born in the United States; rates of depression begin rising among immigrants after 13 years of living in the United States (Vega et al., 1998). Similarly, rates of depression are lower among blacks who immigrated to the United States from Africa or the Caribbean than among blacks born in the United States (Miranda et al., 2005). Despite the lower rates, evidence suggests that minority-group members who are depressed are less likely to get depression treatment than are white Americans (Williams et al., 2007). Furthermore, they are less likely to get high-quality care if they do get care when depressed. The lack of care has led to longer, more severe bouts of depression in blacks than in white Americans, who are much more likely to obtain care when it is needed. African Americans report greater stigma associated with treatment for mental health, which could be related to the decreased likelihood of treatment (Menke and Flynn, 2009).
Research Advances in Knowledge of Biology
For many decades, the role of the central monoaminergic system dominated pharmaceutical and academic research on depression. That focus followed findings in the 1950s that the monoamine oxidase inhibitors were effective antidepressants, later development of the tricyclic antidepressant imipramine, and the identification of their mechanisms of action and development of reserpine and amphetamine in the 1960s (Slattery et al., 2004). The delay seen in the action of antidepressants (it can take weeks for them to become effective) suggests that the acute effects of the agents on serotonin, dopamine, and other neurotransmitters are not directly mediating the antidepressive actions, and over the last 15 years animal models and studies in humans have investigated a number of novel targets for antidepressants. As discussed in review articles, there is evidence that disruption of neurotrophic factors—such as brain-derived neurotrophic factor, IGFR, the neuropeptide VGF, and VEGF—their receptors or alterations in neuronal structure, glial number, and other changes could be involved in depression (Elhwuegi, 2004; Kugaya and Sanacora, 2005; Malberg and Monteggia, 2008; Pav et al., 2008; Tanis and Duman, 2007; Warner-Schmidt and Duman, 2008). The hypothalamic–pituitary–adrenal axis and stress hormones could also play
a role (Cohen et al., 2007; Hindmarch, 2002; Pariante and Miller, 2001). The understanding of depression gained from those studies provides the potential for development of future pharmacologic treatments for depression.
Onset of depression in girls often occurs at the ages of 11–14 years, and this raises questions about the role of sex hormones in the increased risk of depression in women (Angold et al., 1998). Systematic reviews, however, consistently fail to find that rates of major depression are associated with other hormone-related events, including menopause, use of oral contraceptives, and hormone therapy (Kessler, 2003). Interestingly, perimenopause is associated with onset of depression (Schmidt, 2005) and there is some data to suggest the effectiveness of estradiol in perimenopausal depression.
Research Advances in Prevention
Cuijpers and colleagues (2008) reviewed available data on prevention of depression and conducted a meta-analysis of preventive programs. As discussed by the authors, although some studies have investigated treatment and intervention options, only a few have examined prevention of incident mental disorders, looking mostly at targeted populations or conditions (including postpartum depression, depression in school settings, and patients who have physical disorders).15 The meta-analysis indicated that some prevention programs decrease the incidence of depressive disorders (Cuijpers et al., 2008). Because gender differences in depression appear early on, those school programs targeted at preventing depression in adolescent girls are particularly important and worthy of further study and dissemination.
No therapeutic interventions have been identified for primary prevention of incident depression, although a number of the therapeutic agents discussed below are used to prevent the recurrence of depression.
Research Advances in Diagnosis
Recent evidence suggests there a need to revise current diagnostic criteria for depression. In a review of randomized, placebo-controlled trials, the value of antidepressant medications was found to increase with increasing severity of depression. The benefit of antidepressant medications for those who had mild or moderate symptoms was minimal or nonexistent whereas medications were substantially beneficial for those who had severe symptoms (Fournier et al., 2010).
That finding suggests that the Diagnostic and Statistical Manual of Mental Disorders (DSM) criteria for depression, which includes those with mild or moderate symptoms as well as severe symptoms, may not be appropriate for indicating who is likely to benefit from currently available medications.
Research Advances in Treatment
The development of animal models has guided the design of drugs to act on neurobiological targets recognized as involved in the pathophysiology of depression and has led to substantial advances in pharmacologic management of depression. The first of the targeted drugs are the selective serotonin reuptake inhibitors (SSRIs), followed more recently by norepinephrine uptake inhibitors and serotonin receptor antagonists (for example, trazodone and nefazodone). The newer dual-action antidepressants act by inhibiting the reuptake of both serotonin and norepinephrine. They appear to reduce depression and its associated symptoms with greater rapidity, although some trials have indicated similar efficacy and tolerability (Papakostas et al., 2007).
In the last 15 years, researchers have identified targeted nonpharmceutical therapies that are effective in treating for depression. Both interpersonal psychotherapy and cognitive–behavioral therapy can be as effective as medications in the acute treatment of depressed outpatients (Compton et al., 2004; DeRubeis et al., 1999; Frank et al., 2005; Hollon et al., 2005). Medication typically has a rapid and robust effect and can prevent symptom return for as long as it is continued or maintained, but it does little to reduce risk once its use is terminated (Hollon et al., 2005). Interpersonal psychotherapy may improve social functioning, and cognitive–behavioral therapy appears to have an enduring effect that reduces later risk (DeRubeis et al., 1999; Frank et al., 1990, 2005). Gender differences are seen in treatment-seeking behaviors (Dwight-Johnson et al., 2000).
Studies of programs to improve the effectiveness of treatment of depression in primary care suggest that collaborative care improves the quality of care and health outcomes of depressed patients (Katon et al., 1999; Vera et al., 2010); that more efficient methods, such as telephone case management, improve outcomes (Simon et al., 2000, 2004); that multimodal strategies that include case management tend to be more effective than improving clinician knowledge or skills alone (Cunningham and Zayas, 2002; Gensichen et al., 2006); that such programs can improve outcomes of adolescents (Asarnow et al., 2005) and elderly persons (Unutzer et al., 2002); and that active outreach strategies help to engage low-income and minority groups in such care (Revicki et al., 2005). The President’s New Freedom Commission on Mental Health, established by President George W. Bush in April 2002, recommended implementing high-quality improvement programs for depression (Hogan, 2003). Current systems do not often reimburse for telephone case management and, in general, improvement of care has not occurred (Netting and Williams, 1996).
The Sequenced Treatment Alternatives to Relieve Depression (STAR*D) study is a 7-year study of over 4,000 patients (64% of patients were women) designed to assess the effectiveness of depression treatments in patients diagnosed with major depressive disorder in both primary and specialty care settings (Rush et al., 2004). A large number of reports have been published from STAR*D. One study reported sex differences in response to citalopram; women were more responsive to the SSRI citalopram than men (Young et al., 2009).
Unfortunately, there are a number of side effects associated with antidepressants that can require careful monitoring of patients and that decrease adherence to medications. Side effects of antidepressants range in severity and depend upon the class of antidepressant, but can include sexual side effects, weight gain, and an increased risk of suicidal thinking and behavior in young adults (18–24 years old) (Khawam et al., 2006).
Knowledge Gaps
Delivering evidence-based care for depression is challenging owing to organizational and financial factors (such as limited coverage of psychotherapy and diversity in third-party management of services) (Goldman et al., 1999; Sharftsein, 1999), clinical features of depression (such as social withdrawal) (Kornstein and Schneider, 2001), societal factors (such as social stigma, limited public knowledge and language barriers) (Goldman et al., 1999; Lewis-Fernandez et al., 2005; Sirey et al., 2001), and clinician factors (such as limited knowledge and experience) (Gallo et al., 1999; Goldman et al., 1999; Meredith et al., 2001). For example, while antidepressant medication use rose over the last decade, many in need of depression care did not receive counseling, medications, or referral. There are many barriers to provision of evidence-based psychotherapy, such as lack of criteria for licensing providers in such treatments (Patel et al., 2006b). Outside of organized group practices, coordination between primary and mental health care is often difficult, given widespread use of “carve-out” behavioral health management companies that operate largely independently of health plans or medical practices (Patel et al., 2006b). Public-sector mental-health agencies prioritize clients with severe and persistent illness, while public-sector primarycare agencies are not organized or financed to support ongoing mental-health care (HHS, 1999; Patel et al., 2006b).
A major gap in depression research is the development of models of treatment that can be disseminated and sustained within current systems of care. A focus on getting care to underserved populations is particularly needed.
Although treatment for depression in women is generally similar to that in men, Grigoriadis and Robinson (2007) note the need for sex and gender issues to be evaluated. Pharmacotherapy for depression during pregnancy and during the postpartum period requires an assessment of the risks and benefits to the fetus (Ward and Zamorski, 2002). Studies of tricyclic antidepressants and SSRIs have
generally indicated that they are safe for pregnant women, but the evidence is far from conclusive (Källén, 2007; Way, 2007). FDA continues to rate most antide-pressants at level C, noting that the risk to the fetus cannot be ruled out (Hackley, 2010). Postpartum women who have depression can be treated similarly to those who have nonpuerperal depression, unless they are breastfeeding. Few data on antidepressants in breast milk are available (Birnbaum et al., 1999; Weissman et al., 2004). The American Academy of Pediatrics Committee on Drugs concluded that antidepressants are drugs whose effects on nursing infants are unknown but may be of concern (Birnbaum et al., 1999); the risks to the newborn from potential exposure to antidepressants in breast milk need to be weighed against the benefits of nursing and against the risks of the mother foregoing treatment with antidepressants.
In addition, comorbidities are often seen with depression (Huang et al., 2010). For example, associations have been seen between depression and obesity (Luppino et al., 2010), diabetes (Anderson et al., 2001), cancer (Goldberg, 1981), and some chronic diseases in old age (Huang et al., 2010).
Lessons Learned
Although there are effective interventions for depression, many people, particularly minority-group members, fail to get effective treatment. Effective treatment often requires either well-defined psychotherapies, medications, or both managed with careful followup. The more complex interventions are often difficult to disseminate and pay for within current health-care systems and therefore often do not reach women who need care. The occurrence of comorbidities with depression highlights the need to look for comorbidities when treating women. The sex differences in depression further highlight the broad range of sex differences in health outcomes to include neurological disorders. Furthermore, early intervention, particularly preventive interventions for adolescent girls, may well be an important factor in eliminating the high rates of depression among girls and women.
HIV/AIDS
Over the last 20 years, there have been major advances in the treatment of HIV/AIDS, including new treatments that have led to substantial reductions in mortality and morbidity and a major decline in maternal–fetal transmission of HIV. Although much of the initial HIV/AIDS research (particularly clinical trials) has been focused on men, and the proportion of women infected with HIV/AIDS has increased, especially in some groups, the advances have improved the treatment of HIV/AIDS in women as well as men. The committee therefore considers that on balance HIV/AIDS is a condition where scientific advances have led to progress in women’s health.
Incidence, Prevalence, and Mortality in Women
An estimated 1 million people live with HIV/AIDS in the United States (Hall et al., 2008; Kaiser Family Foundation, 2009). HIV/AIDS-related morbidity and mortality have decreased substantially in both men and women due to the development of effective combination antiretroviral therapies over the past 15 years (Bailey and Fisher, 2008; Palella et al., 1998). Although it is more prevalent in men, the rate of HIV/AIDS in women is increasing. In the US, women accounted for only 9% of AIDS cases in 1987 but account for over 27% today (Figure 3-3) (CDC, 2008a; Kaiser Family Foundation, 2009). As is discussed below, the increase is greatest in minority women.
Disparities Among Groups
In 2006, black women had an HIV incidence rate about 15 times that of white women, and Hispanic women had an HIV incidence rate about 4 times that of white women (CDC, 2008b). Among women living with HIV/AIDS, 64% are black and 15% are Hispanic (CDC, 2008c). In 2006, black and Hispanic women accounted for the majority of newly infected HIV women, 61% and 16%, respectively (CDC, 2008b). In 2006, the estimated HIV prevalence (per 100,000) among black women and Hispanic women was 1,122 and 263, respectively—almost 18 and 4 times that among white women (CDC, 2008d). According the CDC 2004 data (CDC, 2008c), HIV was the leading cause of death of black women 25–34 years old, the third-leading cause of death of black women 35–44 years old, and the fourth-leading cause of black women 45–54 years old. For Hispanic women, HIV was the fourth-leading cause of death of women 35–44 years old.
HIV/AIDS is increasingly a disease of lower socioeconomic groups and underserved minorities that have not uniformly accessed early or preventive care (CDC, 2003; IOM, 2001a). HIV prevalence and HIV-risk behaviors are disproportionately high in incarcerated women (De Groot, 2000; Dean-Gaitor and Fleming, 1999; Fogel and Belyea, 1999). The associations between mental-health disorders and HIV (Bing et al., 2001; Rosenberg et al., 2001; Stoskopf et al., 2001; Walkup et al., 1999), and between substance abuse and HIV, are also documented (Bing et al., 2001; CDC, 2002b; HHS, 2008).
Research Advances in Knowledge of Biology
In 1983, the first studies that suggested an association between the retrovirus HIV and AIDS were published (Barre-Sinoussi et al., 2004; Essex et al., 1983; Gallo et al., 1983; Gelmann et al., 1983). Work by Gallo and colleagues (1983) established HIV infection as causal for AIDS, and much research has characterized the life cycle of the virus (see De Clercq, 2007, for review). Over the last 20 years, basic research has further elucidated the etiology of and biology underlying
FIGURE 3-3 Estimated number and proportion* of all AIDS cases, in female adults and adolescents, 1985–2006.
NOTE: Data have been adjusted for reporting delay.
*Proportion of all cases that were diagnosed among women.
SOURCE: http://www.cdc.gov/hiv/topics/surveillance/resources/slides/women/slides/Women.ppt (accessed April 22, 2010).
AIDS. Women have benefited from that research, although much of it was conducted in men and there are some reports that progression from HIV to AIDS may differ by sex (Farzadegan et al., 1998; Nicastri et al., 2005). In addition, women are more likely than men to contract HIV during sexual intercourse (European Study Group on Heterosexual Transmission of HIV, 1992; Kaiser Family Foundation, 2009; Padian et al., 1991), and some sexually transmitted infections (STIs) in women have been associated with higher rates of HIV transmission and acquisition (Greenblatt et al., 1988; Plummer et al., 1991; Smith-McCune et al., 2010; Wasserheit, 1992). Two major studies—the HIV Epidemiology Research Study (HERS) and the Women’s Interagency Health Study (WIHS)—have increased our understanding of the natural history of HIV infection in women (Barkan et al., 1998; Harlow et al., 2000; Smith et al., 1997).
The interaction between HIV/AIDS and pregnancy has been studied. Pregnancy does not accelerate the decline in CD4 counts, and studies have not shown that pregnancy affects progression or survival in HIV-infected women, especially in developed countries (Alliegro et al., 1997; French and Brocklehurst, 1998; Minkoff et al., 2003). As the first generation of HIV-infected women reaches menopause, issues related to HIV and menopause assume greater relevance, and there is a paucity of data on the relationship. Differences have been seen between premenopausal and postmenopausal women who have AIDS. In some studies, postmenopausal women have lower CD4 lymphocyte counts (van Benthem et al., 2002), and maximum CD4-cell response to antiretroviral therapy decreases with age (Manfredi et al., 2003; Viard et al., 2001). Other studies did not show age-related differences in viral-load decreases (the study population was 33% women; no sex-specific analysis) (Tumbarello et al., 2003) or menopause-related immunologic and virologic responses (Patterson et al., 2009).
Research Advances in Prevention
Despite the increased understanding of the underlying virus, no vaccine or preventive treatment for HIV/AIDS is available (Flexner, 2007). Prevention focuses primarily on decreasing high-risk sexual behaviors that are associated with HIV/AIDS (see Chapter 2 for discussion). It is important to remember that many women are infected with HIV not because of their own high-risk sexual behaviors but because of the risk behavior of their partners (CDC, 2008c; Hader et al., 2001; Varghese et al., 2002). In part because of the lack of awareness of risk, those women often present for HIV testing with acute symptoms late in the course of infection.
Research Advances in Diagnosis
There has been no substantial improvement in the early diagnosis of HIV/AIDS in newly infected people (men and women) since the 1990s. In September
2006, the CDC recommended routine HIV testing for adults, adolescents, and pregnant women (13–64 years old) in health-care settings to “foster earlier detection of HIV infection; identify and counsel persons with unrecognized HIV infection and link them to clinical and prevention services; and further reduce perinatal transmission of HIV in the United States” (CDC, 2006).
Research Advances in Treatment
Major progress has been made in the treatment of HIV/AIDS over the last 15 years, and, as stated by Bailey and Fisher (2008), “antiretroviral therapy for HIV infection has transformed it from a terminal illness to a chronic manageable condition.” Detailed reviews of the development of HIV/AIDS drugs have been published (see Bailey and Fisher, 2008; De Clercq, 2007; Sturmer et al., 2009). In 1987, 4 years after the first studies suggested an association between the retrovirus HIV and AIDS (Barre-Sinoussi et al., 2004; Essex et al., 1983; Gallo et al., 1983; Gelmann et al., 1983), zidovudine (AZT), the first antiretroviral treatment, was approved for use in the United States (FDA, 2009b). Highly active antiretroviral therapy—a combination of nucleoside reverse transcriptase inhibitors (NRTIs), nonnucleoside reverse transcriptors (NNRTIs), and protease inhibitors—was in use by the middle 1990s (Bailey and Fisher, 2008). As of 2008, there are six classes of antiretroviral agents that work against HIV—viral reverse transcriptase enzyme inhibitors (NRTIs and NNRTIs), protease inhibitors, fusion inhibitors, entry inhibitors, and integrase inhibitors—and more than 20 drugs are available on the market (Bailey and Fisher, 2008). Although much of the research has been conducted in men, studies indicate that there are no major sex differences in immunologic and virologic response to effective combination antiretroviral therapy (Moore et al., 2003).
The underrepresentation of women in clinical trials has contributed to problems in addressing treatment toxicity. Most studies have not had adequate power to determine sex differences, but an overall review of the literature suggests that the severity and rate of toxicity of some antiretroviral drugs present unique challenges for women, including comorbidities and side effects associated with long-term HIV treatment, such as diabetes (Dube and Sattler, 2010; Nicastri et al., 2005), lipodystrophy (Sattler et al., 2001; Tien et al., 2007), metabolic syndrome (Sobieszczyk et al., 2008), and bone density (Dolan et al., 2004; Sobieszczyk et al., 2008). There is consistent evidence that the toxicity of NRTIs, such as potentially life-threatening lactic acidosis and acute pancreatitis, is more frequent in women (DeMeo et al., 2002; Moore et al., 2001) and that the risk of grade 4 anemia caused by AZT was significantly higher in women (Ssali et al., 2006). Other studies have found an increased prevalence of long-term metabolic abnormalities, such as lipodystrophy and dyslipidemia, in women (Bonfanti et al., 2003; Heath et al., 2002; Pernerstorfer-Schoen et al., 2001; Tien et al., 2003). The occurrence of skin rash (Antinori et al., 2001; Bersoff-Matcha et al., 2001; Mazhude et al., 2002; Wong et al., 2001) and hepatotoxicity (Sobieszczyk
et al., 2008) caused by nevirapine is more common in women. The underlying mechanisms of sex differences in the rate of adverse events are unknown, but it has been suggested that pharmacokinetic sex differences may play a major role (Modjtahedi et al., 2006).
The effects of HIV/AIDS treatments on contraception and pregnancy and the effects of pregnancy on the results of HIV/AIDS treatments have been studied. Treatments can decrease the effectiveness of some hormonal contraceptives, but the contraceptive medroxyprogesterone was not affected by nevirapine, efavirenz, or nelfinavir (Cohn et al., 2007). Deaths from hepatic failure have been reported in pregnant women receiving nevirapine in combination antiretroviral therapy (Hitti et al., 2004; Lyons et al., 2006), and there have been reports of maternal deaths due to lactic acidosis during exposure to ddI and d4T with other antiretroviral agents (CDC, 2002c). Women who have CD4 counts greater than 250/mm3 have greater rates of nevirapine-associated rash (Antinori et al., 2001; Bersoff-Matcha et al., 2001; Mazhude et al., 2002; Moore et al., 1996) and hepatic toxicity (Sanne et al., 2005). Drug-resistance rates appear similar in pregnant and nonpregnant HIV-infected women (Juethner et al., 2003; Palumbo et al., 2001).
Maternal–fetal transmission of HIV greatly affects a woman’s reproductive decisions, and advances in treatments reducing transmission are allowing women to bear children more safely. Where treatments and interventions are available, they have greatly decreased the likelihood that HIV infection will be transmitted to offspring. Research has identified risk factors16 for HIV transmission (Fawzi et al., 1998; Garcia et al., 1999; Groginsky et al., 1998; Landesman et al., 1996; Minkoff, 1997; Minkoff and Mofenson, 1994; Reinhardt et al., 1995) and interventions, such as elective cesarean section (Read, 2000), that effectively decrease the risks and increase women’s reproductive options. The transmission rate decreased from about 25% to 1–2% with advances in the care and treatment of pregnant HIV-infected women, including perinatal antiretroviral therapy (Cooper et al., 2002; The International Perinatal HIV Group, 1999; Mandelbrot et al., 1998; Read, 2000).
Knowledge Gaps
The recent failure in HIV-prevention strategies, including microbiocides and vaccines, in both women and men remains a serious problem in the control of HIV/AIDS (Buchbinder, 2009; Padian et al., 2007; Vandamme, 2008). Research
is needed on prevention strategies, especially in black and Latina women on viral, host, and immune factors that may influence HIV transmission and acquisition in women throughout their lives, on disease progression, mortality, comorbidities; and on efficacy, safety, pharmacology, and side effects that are associated with medications in HIV-infected women that are different from those in men.
Lessons Learned
HIV/AIDS provides an example of how women can benefit but also be at a disadvantage when research is conducted mainly in men. HIV and AIDS highlight disparities among women of different races and ethnicities and the need to assess the disparities to target effective prevention, diagnosis, and treatment activities. Those differences occur even for the side effects of antiretroviral agents. In addition, the complications that occur in pregnancy (and the potential complications for treatment of women and of the offspring) are important, especially given the stage of life at which infection with HIV often occurs.
Osteoporosis and Fragility Fractures
Falls by elderly people are a major cause of morbidity and greatly affect women because of their longevity (Francis, 2008), and osteoporosis makes women more susceptible to fractures following a fall (Gallagher, 2007). Over the last 20 years there have been advances in the knowledge of the basic science underlying osteoporosis and in the diagnosis and treatment of osteoporosis. The committee considers it to be a condition on which there has been some progress.
Incidence, Prevalence, and Mortality in Women
It is estimated that 10 million Americans have osteoporosis and another 34 million have low bone mass; 80% of them are women (NOF, 2002).17 Compared with women who have normal bone mineral density (BMD), women with osteoporosis and osteopenia had a rate ratio of sustaining a fragility fracture of 4.03 and 1.80 per 100 person-years, respectively (Siris et al., 2001). Only 18% of documented fragility fractures occurred among women who met BMD criteria for osteoporosis; 52% of women who had fractures had a BMD in the osteopenic range (T score, –1 to –2.5) in the preceding year (Siris et al., 2004).
Fragility fractures typically occur in the hips, wrists, and vertebrae and result in substantial morbidity and mortality (Cauley et al., 2000; Kado et al., 1999). Nearly one-fourth of those who fracture a hip die within 1 year (OTA, 1994), and elderly survivors of such fractures spend about 17% of their remaining life
in nursing facilities (Braithwaite et al., 2003). Hip fractures occur with higher frequency than spine fractures (Taylor et al., 2010).
The lifetime risk of osteoporosis and fragility fracture rises exponentially with age, with most fractures occurring in women 70 years old or older (Kiel et al., 1987; Melton et al., 1989). The risk increases linearly with the number of fractures that a person has had (Kado et al., 1999). The incidence of hip fractures increased during the 1990s, but incidence decreased more recently in some countries, including the United States and Canada (Brauer et al., 2009; Leslie et al., 2009).
Disparities Among Groups
Among whites and Asians over 50 years old, women have higher fracture risk than men. However, among Hispanics 50–59 years old, men have higher risk than women; after the age of 60 years, Hispanic women have a higher risk than men. Among blacks up to the age of 70 years, men have a higher fracture risk than women; after that women have a higher risk. The risk of fracture increases exponentially with age in all groups (Kanis, 2002).
Research Advances in Knowledge of Biology
Understanding of the pathophysiology of osteoporosis has improved over the last 2 decades (Close et al., 2006; Dennison et al., 2005; Sambrook and Cooper, 2006). That includes the identification of genes whose expression affects the risk of osteoporosis (Huang and Kung, 2006).
A number of factors that lead to an imbalance in bone-forming osteoblast and bone-resorbing osteoclast activity have been delineated (Ikeda, 2008). In mice, expression of the runx2 gene—which is necessary for osteoblast differentiation—decreases in bone tissue and osteoblasts with age; expression of peroxisome– proliferator–activated receptor γ2—which inhibits osteoblasts—increases with age (Moerman et al., 2004). In contrast, changes in osteoclast generation with age have not been established (Pietschmann et al., 2009). Decreasing estrogen also plays a role in bone loss, possibly through effects on cytokines and inflammation (Pietschmann et al., 2009).
Research Advances in Diagnosis
Osteoporosis is often undetected and undertreated. One study estimated that only one-third of women who are at high risk of fracture are told that they have osteoporosis, and only one-fourth receive appropriate treatment (Gehlbach et al., 2007). Another study funded by Merck, the National Osteoporosis Risk Assessment, followed over 200,000 community-dwelling postmenopausal women who had no known history of osteoporosis and found that 7.2% of them met criteria
for osteoporosis and 39.6% met criteria for osteopenia at study entry (Siris et al., 2001).
A number of new markers of bone remodeling are available, but identifying women at high risk of fracture to target effective treatments remains challenging (Blumsohn, 2003). Traditionally, fracture risk was based almost exclusively on BMD as measured with dual x-ray absorptionometry (DXA). There are not uniform standards for DXA machines, however, so results vary by machine and operator. In addition, DXA is insensitive because BMD is only one of several fracture risk factors—bone architecture is also important. Composite multivariable risk-prediction models (Cummings and Black, 1995; WHO, 2010) have enabled the identification of patients at higher risk for fracture on the basis of a combination of BMD, age, and other risk factors. For example, the FRAX model18 predicts the 10-year probability of hip fracture and other major osteoporotic fractures on the basis of sex, age, body-mass index, tobacco use, previous fracture, parental fracture, and glucocorticoid use. Although there is variability among models, the most important fracture risk factors appear to be previous fracture, advancing age, female sex, low BMD, prolonged use of glucocorticoids, and family history of fracture.
It is now routine to screen for BMD in women 65 years old or older and in younger women who have risk factors (Buencamino et al., 2009). As screening for osteoporosis with DXA has become more readily available, more women and their physicians face the dilemma of how to respond to DXA results that fall in the osteopenic range.
Explorations of new imaging techniques (such as high-resolution computed tomography) have not added substantially to the ability to identify women who are most likely to have fractures (Genant et al., 2008).
Appropriate clinical followup of women who begin pharmacologic treatment for osteoporosis is controversial. Repeat DXA scans have been routinely performed 2–3 years after treatment initiation, but the value of such testing has been called into question. A recent study found that among healthy older post-menopausal women, repeating BMD assessments for up to 8 years added little value to the initial BMD measurement (Hillier et al., 2007).
Research Advances in Prevention
One difficulty in preventing osteoporosis is the timeframe of the potential risk factors. Determinants of older women’s bone strength may date back to their adolescence. Bone mass typically increases until a woman is in her 40s, and the most rapid rise occurs during adolescence (Ensrud et al., 2000; Hannan et al., 2000). Dietary factors (such as high calcium consumption and adequate vitamin D) and doing weight-bearing exercise during adolescence and teenage years decrease the risk of osteoporosis, but teenage girls are not likely to be thinking
|
18 |
See http://www.shef.ac.uk/FRAX (accessed September 1, 2010). |
about their future bone health (Lloyd et al., 2000). In the first 1–2 years after menopause, bone loss accelerates (Garnero et al., 1996), and this is followed by an annual rate of loss of about 1% (Cooper and Melton, 1992). Low peak bone mass is thought to be associated with an increased risk of osteoporotic fracture, but the evidence is sparse (Cooper et al., 2006). Low body-mass index is a risk factor for fracture (De Laet et al., 2005).
There is controversy about the appropriate intake of vitamin D, including dosing levels and the possible role of calcium, for fracture prevention (Bischoff-Ferrari et al., 2007; Boonen et al., 2007; Cranney et al., 2007; Tang et al., 2007). There has never been a dose-ranging randomized controlled trial of vitamin D in susceptible populations. A meta-analysis of 12 double-blind trials (in which 89% of the participants were women) found that a high dose (over 400 IU/day) decreased the risk of nonverteral fractures (relative risk [RR], 0.80; 95% confidence interval [CI], 0.72–0.89) and the risk of hip fractures (RR, 0.82; 95% CI, 0.69–0.97). The effect was independent of calcium supplementation (Bischoff-Ferrari et al., 2009).
Several guidelines for prevention of and treatment for low BMD have emerged, most of which are consistent in their key treatment recommendations (Solomon et al., 2005). The guidelines have resulted in changes in clinical practice (Buencamino et al., 2009).
Research Advances in Treatment
Before the late 1990s, hormone therapy (estrogen or estrogen plus progestin) was the only treatment available to preserve bone density. There are now several types of treatment to choose from, including bisphosphonates, selective ER modulators (such as raloxifene), the parathyroid hormone analogue teriparatide, and calcitonin (Chesnut et al., 2000; Quattrocchi and Kourlas, 2004). Patients who have osteoporosis and demonstrate good long-term medication adherence have substantially lower risk of fracture (Emkey and Ettinger, 2006). Side effects, inconvenience, costs, and complexity of some dosing regimens have limited long-term adherence. Dosing intervals for bisphosphonates have been extended from a once-daily to a once-weekly, once-monthly, and even once-yearly schedule to improve adherence (Cramer and Silverman, 2006). Although that can improve adherence, the consequence of missing a dose or taking an incorrect dose is greater. Although they are considered rare, side effects of bisphosphonates, including severe musculoskeletal pain and osteonecrosis of the jaw, have been reported (Watts and Diab, 2010). In addition, there are some case reports of atypical subtrochanteric femur fractures while on bisphosphonates, but following a review of the data FDA concluded that bisphosphonates do not increase the risk of those fractures (HHS, 2010). Drug “holidays” are recommended depending on the fracture risk of patients (Watts and Diab, 2010).
The WHI found that postmenopausal women randomized to daily calcium
and vitamin D experienced a small but significant improvement in hip bone density. However, calcium and vitamin D did not significantly reduce hip fracture (Jackson et al., 2006). The mean followup of 7 years might have limited the ability to detect significant differences.
Nonpharmacologic treatments, such as bone-loading physical activity, could be used to prevent osteoporosis and later fractures in elderly people (Borer, 2005; Gregg et al., 2000; Wilkins and Birge, 2005). Exercise has also been shown to increase bone mass in rats (Iwamoto et al., 2005).
Relatively few women who have osteoporosis or osteopenia receive medications that have been proved to prevent new fractures. In one study, only 30% of 6,311 patients at high risk for fracture received medication for osteoporosis (Solomon et al., 2004). In addition to (male) sex, factors associated with nontreatment included age over 74 year or under 55 years, black race, having more than one comorbid condition, and (for women) being seen by a male physician.
Knowledge Gaps
Progress has been made in the prevention of and treatment for osteoporosis. Some organizations have developed position statements for treatment (Ettinger et al., 2006), but no formal treatment guidelines have been developed despite having been called for since the middle 1990s (Abbott et al., 1996; Goldhahn et al., 2008). The best approach for targeting young girls to decrease their risk of osteoporosis in the future is not known.
The most beneficial time to take osteoporosis drugs and the most appropriate screening (that is, whether hip or spine bone density is the best indicator of osteoporosis) (Dennison et al., 2005) are still not clear, and quality standards for bone-density scans are needed (Leslie and Manitoba Bone Density Program, 2006).
The details of the relationship between bone density and fall outcome remain unknown (Geusens et al., 2002), and some models indicate that factors other than bone density, such as age and sex, should also be taken into account in estimating the risk of bone fracture (Kanis et al., 2005).
Lessons Learned
The challenges around osteoporosis highlight the quality-of-life issues that are a problem particularly for women as a result of their longevity. In addition, the effects of lifestyle choices (such as eating habits) at an early age on the risk of osteoporosis at a later age emphasize the need to consider a woman’s full life span in evaluating risks of adverse health events.
CONDITIONS ON WHICH LITTLE PROGRESS HAS BEEN MADE
This section discusses a number of conditions on which little progress or improvement in health outcomes has been made. Numerous conditions could fit in this category. Instead of a comprehensive review of research on all conditions of concern to women, the committee discusses a few conditions as examples from which it draws broad conclusions.
First, the committee discusses two conditions related to pregnancy: unintended pregnancy and maternal morbidity and mortality. Other conditions considered to have little progress—alcohol and drug addiction, autoimmune diseases, dementia and Alzheimer’s disease, gynecologic cancers (other than cervical), gynecologic disorders (nonmalignant), and lung cancer—are then discussed.
Unintended Pregnancy
The committee considers that there has been no major progress in prevention of unintended pregnancy in light of the lack of decrease in rates over time and in comparison with rates in other countries. Pregnancy constitutes a condition with implications for the health of the pregnant woman as well as of the newborn. Whereas the management of the newborn has seen great progress resulting in a marked reduction of neonatal mortality, many obstetrical syndromes remain poorly understood and are mostly the subject of palliation rather than etiologic cure. Rates of unintended pregnancy have not declined over time and rates of unintended pregnancy remain substantially higher in the United States than in other countries. Unintended pregnancy is associated with increased morbidity for the mother as well as risks to the fetus. Although the committee has included unintended pregnancy and its potential health consequences with health outcomes, unintended pregnancy could also be considered a determinant of health. The ability to space and time children effectively benefits women, men, children, families, and society as a whole (IOM, 2004b). Although not all unintended pregnancies are unwanted, often unintended pregnancy is associated with increased morbidity in the mother and risks to infants and children born from unintended pregnancies, such as risks posed by delay in prenatal care, by tobacco use, by alcohol consumption, and by drug abuse. Infants and children born from unintended pregnancies are also at risk for lower birth weight, death in the first year of life, child abuse, and insufficient resources for a healthy environment. The mother is at risk for depression and physical abuse herself and for dissolution of her relationship with her partner. Both parents may experience economic hardship and failure to achieve their educational and career goals, and these are also challenges to maintaining a strong family unit (IOM, 2009).
Unintended pregnancy occurs for a variety of reasons, including lack of availability of and access to acceptable methods of contraception, failure of chosen contraceptive methods, less than optimal patterns of contraceptive use or
lack of use, and lack of adequate motivation to prevent pregnancy (IOM, 2009). Sexual risk behaviors and sexual assault are also associated with unintended pregnancy and are discussed in Chapter 2.
Incidence, Prevalence, and Mortality in Women
The rate of unintended pregnancies in the United States is among the highest in the developed world (Trussell and Wynn, 2008), despite the fact that decreasing the rate of unintended pregnancies is an objective of Healthy People 2010 (Klerman, 2000) and that family planning has been considered by CDC to be one of the 10 greatest public-health achievements of 20th century (CDC, 1999). About half (49%) of pregnancies are unintended, that is, unwanted or mistimed (Finer and Henshaw, 2006). Women are considered at risk for unintended pregnancy if they have sexual intercourse, are fertile (neither partner has been contraceptively sterilized or is infertile for any other reason), are not intentionally pregnant and have not been trying to conceive during any part of the year (IOM, 1995). Given those factors, of the 50 million sexually active women 18–44 years old, 28 million (56%) are at risk for unintended pregnancy (Frost et al., 2008). Some 74% of pregnancies among unmarried women in 2001 were unintended compared with 27% of those among married women (Finer and Henshaw, 2006).
Disparities Among Groups
The rate of unintended pregnancy is substantially higher among poor women (112 unintended pregnancies per 1,000 women with an income below the poverty level, or 62% of pregnancies) than among women who live at or above 200% of the poverty level (29 unintended pregnancies per 1,000 women or 38% of pregnancies) (Finer and Henshaw, 2006). Higher-income women were more likely to abort an unintended pregnancy than women below the poverty level, and the rate of unintended births in women below the poverty level (58 per 1,000 women) was higher than that of higher-income women (11 per 1,000 women at or above 200% of the poverty level). Women aged 18–19 years old (108 per 1,000 women) and 20–24 years old (104 per 1,000 women) are at highest risk (Finer and Henshaw, 2006). The percentage of pregnancies that are unintended was highest at earlier ages: 100% in those under 15 years old, 82% in those aged 15–19 years old, and 60% in those aged 20–24 years old (Finer and Henshaw, 2006). Unintended pregnancy is most likely to occur among women who are young, unmarried, of low income, or members of racial or ethnic minorities; black and Hispanic women have higher rates of unintended pregnancy than white women (Finer and Henshaw, 2006).
Research Advances in Prevention
Advances in social and behavioral interventions to prevent unintended pregnancy were discussed in Chapter 2 in relation to sexual risk behaviors. New contraceptive methods have become available over the last few decades, and the safety of oral contraceptives, which were first marketed in the United States in 1960, has improved over the same period as doses of the steroid hormones (estrogens and progestins) used in them have been continually reduced (Batur et al., 2003; Benagiano et al., 2007; Swica, 2007). Although there is evidence of an increased risk of venous thromboemoblism and pulmonary embolism in women taking oral contraceptives, especially in smokers (Chasan-Taber and Stampfer, 1998; Middeldorp, 2005), numerous studies have demonstrated the remarkable safety of current formulations of oral contraceptives, which for nearly all young, healthy women are safer than pregnancy (Beral et al., 1999; Chasan-Taber and Stampfer, 1998). New delivery systems for combined estrogen–progestin hormonal contraceptive preparations have been developed, including transdermal patches and vaginal rings. In addition, progestin-releasing implants and intrauterine devices that release progestin are now available (Tolaymat and Kaunitz, 2007). Another advance in contraceptive technology is the development of a female condom that, as do male condoms, has the potential to prevent not only pregnancy but also STIs, including HIV, when used consistently and correctly (Warren, 2000). Focus groups of girls and boys age 15–20 years, however, did not classify the female condom as “female controlled” (Latka et al., 2008).
Research Advances in Treatment
About half of unintended pregnancies end in abortion; therefore, contraception could reduce not only unintended pregnancy rates but also abortion rates and rates of STIs with some methods (IOM, 1995). The IOM report The Best Intentions: Unintended Pregnancy and the Well-Being of Children and Families discussed some of the potential medical and psychologic complications of surgical abortion. It stated that “whatever the risks associated with legal abortion in the United States, it remains a far less risky medical procedure for the woman than childbirth; over the 1979–1985 interval, for example, the mortality associated with childbirth was more than 10 times that of induced abortion” (IOM, 1995). It also stated that “abortion in the first trimester of pregnancy carries fewer risks to health than abortion in the second trimester of pregnancy and beyond.” As reported by IOM (1995), “data show a total complication rate of induced abortion of less than 1 percent (Hakim-Elahi et al., 1990).”
A previous IOM committee, the Committee on Antiprogestins: Assessing the Science, reviewed the literature on the use of mifepristone (RU486) and other antiprogestins as medical abortifacients and for other potential uses (IOM, 1993). At the time, RU-486 in combination with prostaglandin was approved for
early-pregnancy abortions in France, Sweden, and the United Kingdom, and the committee noted the “vast world literature with more than 60,000 patients treated in France” and stated that “repetition of large Phase 3 trials in the United States (to demonstrate efficacy and document side effects) does not appear necessary.” For second-trimester pregnancies, that committee recommended “conducting clinical trials in the United States to compare the established surgical procedure of dilation and evacuation (D&E) both to antiprogestins in combination with prostaglandins and to prostaglandins used alone” (IOM, 1993). FDA approved mifepristone as an abortifacient in 2000. Since that time four medical abortion regimens—mifepristone and misoprostol pills, mifepristone pills and vaginal misoprostol, methotrexate and vaginal misoprostol, and vaginal misoprostol alone—have received FDA approval for use in the United States. A number of studies have been conducted to compare the safety and efficacy of different forms of medical abortions (Creinin et al., 1997).
Knowledge Gaps
Substantial advances have been made in contraceptive technology, but rates of unintended pregnancy remain high in the United States. In 2001, the 49% of all pregnancies that were unintended resulted in 1.4 million births, 1.3 million induced abortions, and an estimated 400,000 miscarriages (Frost et al., 2008). The United States has higher rates of unintended pregnancy than other developed countries, such as France (33%) and Scotland (28%) (Trussell and Wynn, 2008). The rates are highest in black and Hispanic women. The reason for the discrepancy in rates between the United States and other developed countries is not well understood (IOM, 2009).
Despite the development of modern contraceptive methods over the last several decades, high rates of unintended pregnancy are unabated, and it is clear that there is a need not only to develop new and improved contraceptive methods, including non-hormonal contraceptives, but also to ensure that existing methods are more available and accessible and used more effectively. The IOM (2004b) has created a blueprint for action on contraceptive development that this committee endorses. The blueprint includes recommendations for setting priorities for research and other activities in three main categories: (1) identifying and validating novel contraceptive targets; (2) enhancing contraceptive drug discovery, development, and clinical testing; and (3) facilitating and coordinating implementation of contraceptive research and development. Development of new contraceptives remains slow even after the publication of that report, and decreased research and development activities at pharmaceutical companies provide little optimism that new contraceptives are on the horizon (Aitken et al., 2008). The IOM (2009) has also evaluated the Title X program, which is the only federal program in the United States that is devoted exclusively to providing family-planning services. In addition to the recommendations in the 2009 report, which the present com-
mittee endorses, the committee urges more research on how to strengthen services for the provision of contraceptive methods especially in women at greatest risk. Such health-services research should set priorities for the strengthening of service delivery systems not only through Title X, Medicaid, and the rest of the public sector, but also through the private sector. Important research gaps also remain in understanding the behavioral issues surrounding successful contraceptive use, and further behavioral-science research to address these gaps is warranted in both men and women.19 The work should include studies of the use of contraceptive methods for dual protection, that is, protection against both pregnancy and STIs, including HIV. Finally, although numerous epidemiologic investigations have demonstrated the safety of available contraceptives for healthy women, few studies of the safety of contraceptive use by women with medical disorders are available, and this gap should be addressed.
Lessons Learned
Unintended pregnancy is an example of a condition for which safe and effective prevention and treatment are available, but these are enmeshed in social and political controversies that are barriers to their use. A high proportion of pregnancies in the United States are unintended (unwanted or mistimed). Reducing that proportion will improve the health of women and children and will have important benefits to families and society in general. Progress in reducing unintended pregnancies will require not only making contraceptive methods more available, accessible, and acceptable through improved services, but also the development of new methods that meet additional needs.
Maternal Mortality and Morbidity
Although the US maternal mortality and morbidity rates have declined dramatically during the 20th century, they have decreased little over the past 2 decades. The committee, therefore, considers maternal mortality and morbidity to be a condition for which research over the past 2 decades has made little progress.
Incidence, Prevalence, and Mortality in Women
Although the United States experienced marked reductions in maternal mortality in the last century—from 607.9 deaths per 100,000 live births in 1915 to 12.1 in 2003 (Hoyert, 2007)—1 to 2 women die each day of pregnancy complications in the United States (CDC, 2008e). Recently maternal mortality in the
|
19 |
Some determinants of unintended pregnancy, including violence against women and sexual risk behavior, are discussed as determinants in Chapter 2. |
United States has been increasing—12.1 per 100,000 live births in 2003, 13.1 in 2004, and 15.1 in 2005—although it is unclear whether the increase is real or a result of improved reporting.20 As many as half of all pregnancy-related deaths in the United States are preventable (CDC, 2008e). The leading causes of pregnancy-related death in the United States are embolism, hemorrhage, and other medical conditions (Berg et al., 2003).
Of the nearly 4 million women in the United States who give birth each year, nearly 30% experience complications (Berg et al., 2009). Little progress is noted in achieving the Healthy People 2010 objective of no more than 24 deliveries with intrapartum complications per 100 deliveries. A study of maternal morbidity based on data from the National Hospital Discharge Survey for 1993–1997 and 2001–2005 concluded that the rate of obstetrical complications remained unchanged at 28.6% (Berg et al., 2009). In addition, surveys show increases in the rate of pregnancies complicated by preexisting medical conditions and in the rate of cesarean deliveries (from 21% of births during the mid-1990s to 32% in 2007) (Menacker and Hamilton, 2010).
Disparities Among Groups
Marked disparities in the risk of maternal death remain in the United States. In 2007, in non-Hispanic blacks there were 28.4 deaths per 100,000 live births compared to 10.5 among non-Hispanic whites and 8.9 among Hispanic women (Xu et al., 2010).
Knowledge Gaps
Every effort should be made to ensure that the outcome of each and every labor and delivery in the United States is a healthy newborn–mother tandem, and Healthy People 2010 strives for no more than 3.3 maternal deaths (defined as deaths or complications leading to death that occur within 42 days of birth or the termination of pregnancy) per 100,000 live births (HHS, 2000). That goal, and goals for maternal morbidity, remains unattained. Determining the best ways to reduce maternal mortality and morbidity should have high priority in research.
Alcohol- and Drug-Addiction Disorders
There has been some progress in understanding sex and gender differences in alcohol- and drug-addiction disorders over the last 20 years, but the prevalence of addiction in women has not decreased and in some groups has increased. There
is a dearth of well-designed studies on gender-specific approaches to addiction treatment, and knowledge gained from research has not been applied.
Incidence, Prevalence, and Mortality in Women
According to the Office of National Drug Control Policy (2006), there has been a dramatic increase in substance use and misuse, delinquency, and health problems in girls and women since 1992. Although illicit-drug abuse and alcohol-and drug-addiction disorders have traditionally been more prevalent in men than in women, the gap has narrowed over the last decades (Holdcraft and Iacono, 2004; Keyes et al., 2008). Women now make up 40% of cocaine users, and this reflects a sharp increase in the last decade (SAMHSA, 2005). Although generally women begin using substances later than men, women have consistently been found to have heightened vulnerability to the adverse effects of substance abuse (Brady et al., 2009), and they move more quickly to dependence (telescoping) and manifestation of the adverse health effects of alcohol (Diehl et al., 2007; Greenfield, 2002).
The prevalence of alcohol dependence or addiction is higher in men than in women; 5.42–10.4% and 2.32–4.9%, respectively (Grant et al., 2004; Tetrault et al., 2008). Grant and colleagues (2004) found an increase in dependence rates in black women 18–29 years old. With respect to drug abuse or addiction, 2007 data indicate that drug dependence or abuse is higher in men than in women (3.8% and 1.8%, respectively) (SAMHSA, 2008).
Disparities Among Groups
The prevalence of alcohol dependence and addiction in women varies with race and ethnicity. American Indian women have the highest rate, 4.49%, followed by 2.39, 2.37, 1.94, and 1.34% in black, white, Hispanic, and Asian women, respectively (Grant et al., 2004). Trends indicate a convergence in the male:female ratio for alcohol abuse and dependence in the younger cohorts that is seen in all races and ethnicities except blacks (Grant et al., 2004; Keyes et al., 2008). While there is no consistent evidence of racial/ethnic disparities in rates of drug abuse or dependence, the consequences (e.g., HIV/AIDS, incarceration) of drug abuse and dependence appear to be more severe among racial/ethnic minorities (HHS, 2003). Similarly, although the rates of alcohol abuse are lower among older populations, the risks for morbidity and mortality from alcohol abuse are greater in this group (Cummings et al., 2008).
Research Advances in Knowledge of Biology
A number of physiologic sex differences have been explored to explain those differences. Animal studies have shown the role of sex hormones in the brain
affecting, for example, the hippocampus, the amygdala, the anterior cingulate cortex, and the orbital frontal cortex (Goldstein et al., 2002). Human studies using functional imaging find differences in neural responses of men and women to rewards that are mediated, in part, by sex steroids and by different responses to drug cues between men and women with cocaine addictions (Kilts et al., 2004).
Neuroactive gonadal steroid hormones (such as estradiol, estriol, estrone, and dehydroepiandrosterone) also mediate effects in the brain. For example, evidence suggests that the extent of cocaine dependence (that is, the extent of cravings) in cocaine-dependent women correlates with the concentrations of progresterone and estradiol during the different phases of the menstrual cycle (Fox et al., 2008). Stress-induced relapses occur more frequently in women than in men, and the physiologic response to stress is different between cocaine-addicted men and women and between cocaine-addicted people and controls (Fox et al., 2005).
Sex differences have been found in pharmacokinetics and pharmacodynamics of alcohol. The difference is mainly due to sex differences in the activity of a key biotransformation enzyme (alcohol dehydrogenase) that is responsible for alcohol metabolism (Baraona et al., 2001). Women also have a slower transit time in the gastrointestinal tract (Baraona et al., 2001).
Research Advances in Prevention
Risk factors for drug abuse among women and girls include depression, posttraumatic stress disorder, self-destructive tendencies, delinquent predisposition, physical and mental health problems, family and social environment, having a partner who uses drugs, and childhood trauma and abuse (Becker and Grilo, 2006; Bukstein et al., 1992; Chatham et al., 1999; Hser et al., 1987; Langan and Pelissier, 2001).
Because alcohol use often co-occurs with use of other substances, many interventions are designed to prevent substance use and abuse in general. The Center for Substance Abuse Prevention funded 25 projects on substance-abuse prevention for adolescent girls. Qualitative data suggested that girls responded favorably to the programs, but significant changes in substance use were not found, possibly because of insufficient power and lack of specificity (Guthrie and Flinchbaugh, 2001). Currently the Substance Abuse and Mental Health Services Administration (SAMHSA) National Registry of Evidence-Based Programs and Practices lists only one program, the Athletes Targeting Healthy Exercise and Nutrition Alternatives (ATHENA) program, which is specifically designed for and targeted to girls. The school-based program targets middle- and high-school female athletes with interventions for substance abuse and other unhealthy behaviors, such as eating disorders. Reports to SAMHSA show that marijuana and alcohol use at 1-year followup were statistically lower in the schools with ATHENA than in those without it (HHS, 2007).
There is a paucity of research on gender differences in intervention effective-
ness (Blake et al., 2001). Existing substance-use prevention does not systematically address risk factors for girls and young women, or the etiological factors related to substance use and misuse, delinquency and related health problems by gender (Kumpfer et al., 2008). Adolescent girls have not been recruited for interventions at the same rate as boys and have had lower retention in prevention interventions (Guthrie and Flinchbaugh, 2001). The efficacy of prevention approaches may vary by gender. The ALERT Plus program for at risk adolescents was efficacious only for girls (Longshore et al., 2007), while a universal prevention educational curriculum showed lower benefits for girls than for boys (Vigna-Taglianti et al., 2009).
Research Advances in Diagnosis and Treatment
There have not been any major advances in the diagnosis or treatment of substance-use disorders in women. The same DSM criteria are used for both men and women. Gender differences in treatment need, use, and outcomes have been found but have not been translated into new treatments in women.
Although 10% of women of reproductive age need treatment for substance-use problems, 85% of those who need treatment do not receive or perceive the need for treatment. Not being ready to stop use, not being able to pay for treatment, and the stigma associated with addiction are the major reasons why women do not receive treatment (SAMHSA, 2008). A higher proportion of men than of women are referred into treatment through the criminal-justice system (Grella, 2008) and receive family support for entering treatment (Grella and Joshi, 1999). In contrast, more women access treatment by referrals from community agencies and by referrals associated with receiving a diagnosis of antisocial personality disorder, having engaged in sex work, or having initiated treatment oneself (Grella, 2008). Women rely more on public insurance for payment, and this makes them more vulnerable to public-policy changes that reduce eligibility and funding (Grella, 2008).
Although women enter treatment after fewer years of substance use than men, they tend to have more severe clinical profiles, particularly mood and anxiety disorders; more family conflict and parenting challenges; a much greater proportion of past and current physical and sexual abuse; and more problems related to lack of employment, education, and vocational skills (Brady et al., 1993; Stewart et al., 2003).
Minority-group women and men receive a lower level of care than non-Hispanic whites and are more likely to get such services via the criminal-justice system and informal sectors of substance-abuse treatment (Perron et al., 2009).
Women’s treatment adherence and retention has been associated with patient demographics (for example, marital status), psychosocial characteristics (for example, psychiatric comorbidity and history of trauma), and treatment-program characteristics (Greenfield and Pirard, 2009). Clinical studies that investigated the
possibility of sex differences in treatment retention and completion have shown mixed results, and population-based studies have reported few or no differences in treatment retention (Greenfield and Pirard, 2009).
There is little evidence that gender is an important predictor of treatment outcomes, but there is evidence of gender differences in the factors that predict treatment outcomes (Greenfield, 2002; Grella, 2008). Co-occurring mental-health disorders and history of abuse are two factors that might have more adverse effects on the prognosis of substance-abuse disorders in women (Greenfield and Pirard, 2009).
Women might benefit more from some services, for example receipt of mental-health services has a positive effect in posttreatment, reducing drug use by women but not by men (Marsh et al., 2004). A number of other gender differences have been reported in factors related to risk of relapse. Relapse in women is associated with not living with their children, depression, stressful marital relationships, and partners’ pressure to use substances (Grella, 2008).
Gender-specific addiction treatment has been developed and some studies suggest that women in programs that provide gender-specific treatment of addiction may stay in treatment longer and have better outcomes than those in non– gender-specific programs (Grella, 2008). Treatment completion and outcomes are better in women who receive residential services where they can be housed with their children, and in outpatient treatment that includes family therapy, family services, individual counseling, comprehensive support services, health and social services, and transportation (Grella, 2008).
Few studies have adequately assessed the effectiveness of gender-specific compared with mixed-gender treatments in women. Controlled randomized studies of treatment outcomes have not been conducted, only a small number of treatment outcomes have been studied, there is a lack of standardized measures and definitions of treatment factors and outcomes, and few studies have assessed long-term outcomes (Greenfield and Pirard, 2009; Grella, 2008).
Knowledge Gaps
There has been a dearth of research on the efficacy of gender-specific approaches for prevention of and treatment for addiction disorders, and there is a substantial gap in research on the efficacy of existing prevention and treatment approaches for racial and ethnic minorities populations. Social and cultural differences among subgroups of women are highly relevant for the course of addiction, its treatment, and the recovery process, and how to best address those differences is not known. Finally, there is a clear need to develop and test approaches that facilitate access to and increase use of treatment services by girls and women who need such services.
Lessons Learned
Research on drug addiction highlights the importance of co-occurring disorders and history of trauma in women, and of women’s social roles as mothers and their lower socioeconomic status in relation to vulnerability to addiction and to its treatment.
In addition, although the specific roles of sex differences in brain anatomy, neurobiology, pharmacokinetics and pharmacodynamics, menstrual cycle, gonadal steroid hormones, and stress response in addiction are not yet elucidated, these research avenues highlight how much can be learned about a disease and its pathophysiology by exploring sex differences at many diffent levels. The value of diverse studies—of cells, animals, and humans—is evident from the breadth and depth of information generated not only on addiction, but on sex differences and brain function in general.
Autoimmune Diseases
As a group, autoimmune diseases are the third-most common diseases in the United States, following heart disease and cancer. They affect 5–8% of the population,21 and over 78% of those affected are women (Fairweather et al., 2008). By the age of 50 years, 5–10% of women in the United States have autoimmunity. There have been advances in knowledge over the last 15–20 years about the etiology and pathophysiology of autoimmune diseases in women, but they have not yet led to effective treatments beyond treating for the symptoms. The diseases remain a major cause of morbidity in women. The committee discusses autoimmune diseases in general in this section, using examples when appropriate. The committee considers autoimmune diseases to be a category on which there has been little progress.
Incidence, Prevalence, and Mortality in Women
Many forms of autoimmunity affect women almost exclusively (see Table 3-1). Ulcerative colitis, diabetes mellitus, and myocarditis are exceptions; these occur in men either as often as or more often than in women (Rubtsov et al., 2010). Autoimmune diseases include many forms of arthritis that affect primarily
TABLE 3-1 Incidence Ratio of Autoimmunity Based on Sex
|
|
Female:Male |
|
Thyroid disease (Hashimoto’s) |
10:1 |
|
Graves disease |
7:1 |
|
Sjogren’s syndrome |
9:1 |
|
Lupus |
8:1 |
|
Rheumatoid arthritis |
2.5:1 |
|
Scleroderma |
3:1 |
|
Multiple sclerosis |
3:1 |
|
SOURCE: AARDA (2010). |
|
women—such as lupus, arthritis, fibromyalgia, and rheumatoid arthritis. Greater female vulnerability to autoimmunity is seen in animals. For instance, female mice are more prone to type 1 diabetes (in the nonobese diabetic [NOD] mouse model) and to Sjogren syndrome than male mice (Quintana and Cohen, 2001).
Autoimmune diseases affect women in the prime of their lives and can have a major impact on their quality of life (Gale and Gillespie, 2001); they are associated with high health-care costs and loss of productivity (Lau and Mak, 2009). The effects of autoimmune diseases were highlighted in a report to Congress that stated that “virtually all [autoimmune diseases] are debilitating, and require lifelong medical care. Although treatments exist for many autoimmune diseases, we do not yet have definitive cures for any of them. As a result, autoimmune diseases impose a heavy financial and emotional burden on patients and their families, and contribute significantly to our rising national health care costs” (HHS, 2005). There is evidence that the impact of rheumatoid arthritis on work disability is greater in women than in men (Wallenius et al., 2009).
Disparities Among Groups
Some autoimmune diseases are more common in black, American Indian, and Latina women than in white women (AARDA, 2010). For example, black women are 3 times more likely to have lupus than white women. In contrast, arthritis is most common in non-Hispanic white women (27.2%) followed by non-Hispanic black women (23.5%). Asian women (11.5%) had the lowest prevalence of arthritis (HRSA, 2008).
Research Advances in Knowledge of Biology
The 2001 IOM report Exploring the Biological Contributions to Human Health: Does Sex Matter? summarized potential environmental, hormonal, ge-
netic, and life-stage causes of autoimmunity and evaluated the evidence that those causes would differ between the sexes. The report concluded that although environmental factors (for example, drug-induced cases of lupus and toxin-induced scleroderma-like disease), hormones, and genetics all affect the immune system and could play a role in some of the differences, none can fully account for sex differences in the more common, idiopathic autoimmune diseases (IOM, 2001b). With respect to life-stage causes of autoimmunity, the report stated that “most diseases that are predominantly in females cluster in the young-adult years, whereas autoimmune diseases that affect younger or older patients are more evenly divided between the sexes” (IOM, 2001b). Since that report, further work has explored the pathophysiology of autoimmunity and the underlying causes of sex differences in autoimmunity.
Over the last 20 years, research on autoimmune diseases has taken advantage of novel molecular-biology techniques, including transgenic and knockout mice that are susceptible to autoimmune diseases, to explore the genetic components of autoimmunity (see Alarcon-Riquelme, 2007, for review). Research has examined complement-system–related genetic differences in autoimmune diseases, and some data suggest a partial role of the complement system in susceptibility and disease severity (Murray et al., 2007; Yu and Whitacre, 2004). A role for microchimerism22 has also been suggested (Ando and Davies, 2003; Sarkar and Miller, 2004). Because of the higher prevalence of autoimmune diseases in women, the role of the X chromosome in autoimmune diseases has been explored, especially its inactivation patterns (Invernizzi et al., 2008, 2009). No genetic difference has yet been identified to which any autoimmune disease or the predisposition of women to autoimmune diseases can be fully attributable. The etiology of autoimmune diseases is probably a complex interaction of more than one gene and environmental factors in the broad sense (Alarcon-Riquelme, 2007).
In both the NOD mouse model of autoimmunity and in humans who have Sjogren’s syndrome, there is dysregulation at the level of translation of the LMP2 proteasome protein (Yan et al., 1997). Without a fully functional proteasome there is interruption in T-cell education to self-proteins expressed in the major histocompatibility complex (MHC) class I surface protein and interruption of the critical regulators of life and death balance for T-cell selection (Faustman et al., 1991; Hayashi and Faustman, 1999).
The role of hormones in sex differences in autoimmunity, especially the role of the ER, has also been studied (Ackerman, 2006). Until the late 1990s, only a single type of ER, ER-α, was thought to exist. That receptor is localized to the reproductive organs, mammary glands, bone, cardiovascular system, and some brain regions (Couse et al., 1997). The gene for a second type of ER, ER-β, has been identified, and research has demonstrated that it is distributed in a wide
variety of tissues (Kuiper et al., 1996, 1997; Mosselman et al., 1996). With the discovery of that receptor has come the demonstration that estrogen can influence the development and function of CD4(+) T-helper immune cells (Pernis, 2007). Research in murine models of systemic lupus erythematosus (SLE) and other autoimmune diseases has also found differences in the ER between mice that get autoimmune diseases and mice that do not (Greenstein et al., 2001).
As reviewed by Invernizzi and colleagues (2008), research over the last 20 years has investigated whether X-chromosome inactivation could play a role in autoimmunity. For example, women who have primary billiary cirrhosis, systemic sclerosis, and autoimmune thyroid disease have enhanced X monosomy in blood lymphoctyes (Invernizzi et al., 2004, 2005). The potential role of X-chromosome inactivation, X-chromosome monosomy, and preferential loss of an X-chromosome in autoimmune diseases remains unclear (Invernizzi et al., 2009).
Differences have also been seen in risk of rejection of transplanted organs, depending on the sex of the donor and the sex of the recipient. Female recipients of male kidneys had greater risks of graft failure after transplantation, but not beyond the 10-year mark (Kim and Gill, 2009). H–Y antigen mismatch underlies the effect (Kim and Gill, 2009).
Research Advances in Prevention
Environment has always been considered a risk factor for autoimmunity, with gene–environment interactions thought to be potentially important (Cooper et al., 1999). For example, vitamin D has been proposed to play a role in autoimmune disease (Szodoray et al., 2008). People living in northern climates have a higher risk of autoimmunity, such as multiple sclerosis, than people of the same ethnicity living near the equator (Beretich and Beretich, 2009; Kulie et al., 2009; Kurtzke, 1975). Many have speculated that that could be related to vitamin D deficiencies in people who live in northern climates (Ascherio and Munger, 2007; Ebers, 2009). At the cellular level, vitamin D is obligatory for many steps in T-cell education, including steps related to the activation of NFkB, the main controlling protein for life–death decisions in T-cell education (Mahon et al., 2003).23 It has been speculated that the exposure of the immune system to a broader range of parasites and infections is beneficial in shaping the immune system to prevent self-reactivity (Kaufman, 2010). Thus, the hygiene theory postulates that the cleanliness of civilization may contribute to the increasing incidence of autoimmunity and allergies. There are also intestinal forms of autoimmunity, such as celiac disease, that are triggered by wheat in food but for wheat to trigger or exacerbate the disease, a person must have an altered set point and predisposition for autoimmunity. Many clustered cases of autoimmunity have been considered to
be secondary to viruses, although most of the evidence is circumstantial. Viruses can interfere with the MHC class I education pathway for self–non-self T-cell recognition (Cohn, 2008).
SLE is an autoimmune rheumatic disease that is often severe and is most commonly diagnosed in women in their childbearing years. Exposure of genetically predisposed people to one or more environmental triggers, such as Epstein-Barr virus, is thought to mediate the development of SLE (Simard and Costenbader, 2007). Stress caused by life events also may trigger or exacerbate autoimmunity, as is reported frequently in SLE patients (Karasawa et al., 2005).
Research Advances in Diagnosis
A survey commissioned by the American Autoimmune Related Diseases Association shows that it takes an average of 4 years for a correct diagnosis of an autoimmune disease to be made, an average of 4.04 physicians are seen before the diagnosis is made, and over 45% of the women who received a diagnosis of autoimmune diseases had been told that they were chronic complainers or were too concerned with their health (AARDA, 2010). Those statistics point to the need for more awareness of autoimmune diseases and for better training in medical school. There is no course syllabus for autoimmune diseases in general for physicians or nurses.
Research Advances in Treatment
Over the last 4 years, many new treatments for autoimmunity have emerged. Most of them work to suppress the immune system, because clinical therapies with more restricted targets have failed. However, lifelong and systemic immunosuppressive drugs are not only extremely expensive but associated with drug-induced complications—side effects on the normal parts of the immune system (Uetrecht, 2009).
Knowledge Gaps
Despite some progress in understanding the immune system and the pathophysiology of autoimmunity, there is no confirmed mechanism that explains why women are affected more than men by autoimmune disease, either for a single autoimmune disease or for them as a group. Furthermore, most medications used to treat people for these diseases are lifelong expensive treatments. Elucidation of the differences in the etiology and pathophysiology of these disorders is important because it might allow more to be done to protect women from autoimmune diseases, at least to the same degree as men.
Lessons Learned
The extensive morbidity and associated effects of autoimmune diseases on women’s quality of life highlight the need to look beyond mortality as an end point in researching women’s health. The importance of awareness among physicians of the signs, symptoms, and prevalence of diseases in women to improve the time between onset of symptoms and diagnosis is also evident from autoimmune disease.
Dementia of the Alzheimer Type and Other Dementias
Of all of the chronic, progressive age-associated diseases that threaten the lives, dignity, and independence of elderly women, perhaps none is more dreaded than dementia; dementia of the Alzheimer type (Alzheimer’s disease) is the leading type of dementia (Golanska et al., 2009).
Incidence, Prevalence, and Mortality in Women
About 4.8% of women 71–79 years old have dementia, a figure that rises to 27.8% between 80–89 years old and 34.7% at or after the age of 90 years (Plassman et al., 2007). The overall age-adjusted risk is about 50% higher in women than in men (Bachman et al., 1992; Canadian Study of Health and Aging, 1994; Launer et al., 1999), but the sex difference appears to lie principally in the overall increased average longevity of women (Hebert et al., 2001; Plassman et al., 2007). That increased longevity also means that women often outlive their partners and therefore have a high probability of having lost a member of their family support network by the time their Alzheimer’s disease is manifested. Research to delay or prevent dementia in aging women has high priority in women’s health research.
Research Advances in Knowledge of Biology
An active field of research in Alzheimer’s disease is the investigation of extracellular beta-amyloid plaques and neurofibrillary tangles—the pathologic findings used by Alzheimer over a century ago to characterize the disease (Lannfelt et al., 1995). Research includes their roles in the pathophysiology of Alzheimer’s disease and how to modulate the rate of production, polymerization, solubility, and clearance of the beta-amyloid protein that accumulates in the plaques in the extracellular brain environment (Lannfelt et al., 1995). In vitro and animal experiments (including genetically engineered animal models) have examined the contributions of genetics, enzyme activities, apoptotosis, hormones, metabolism, and such other forces as the oxidative stress and inflammation that occur with aging to the clinical manifestations of Alzheimer’s disease.
Researchers have investigated the role of sex hormones, especially estrogen, in women’s susceptibility to Alzheimer’s disease and dementia. Results of basic-science research, small clinical trials, and cohort studies of aging communities before the WHI suggested a potential role of post-menopausal estrogen therapy (estrogen or estrogen plus progestin) in attenuating the risk of cognitive dysfunction in older women (Ancelin and Ritchie, 2005). Therefore, a substudy of the WHI, the Women’s Health Initiative Memory Study (WHIMS), was conducted; it specifically examined the effect of hormone therapy—combined conjugated equine estrogen (CEE) plus medroxyprogesterone acetate or CEE alone in women with prior hysterectomy—in 4,532 WHI participants over 65 years old who did not have cognitive impairment at baseline (Shumaker et al., 2004). Annual Mini-Mental Status Examinations (MMSE) conducted over an average of 4.2 years revealed not only revealed no tendency toward stabilization of cognitive function in participants on hormone replacement but some greater declines in MMSE results in women who had lower baseline scores and received CEE. Some current research on this issue is being conducted in women closer to the menopausal transition, examining whether there is a critical window for hormone therapy (Sherwin, 2003), but some researchers have little enthusiasm for female-hormone replacement in the prevention of dementia and Alzheimer’s disease in aging postmenopausal women (Markou et al., 2005).
Studies of whether specific cognitive domains (for example, verbal abilities and memory) are differentially affected by sex have had mixed results. Genetic factors might confound the role of estrogen in Alzheimer’s disease (MacLusky, 2004). For example, there could be interactions between estrogen and the apolipoprotein e4 phenotype (either heterozygous or homozygous) (MacLusky, 2004). Depression, a disease that is also more common in women than in men, is frequently associated with Alzheimer’s disease, especially at its initial presentation and in early stages (Migliorelli et al., 1995). It is unknown, however, whether the etiology and pathophysiology of the two conditions are related.
Knowledge Gaps
There have been few major advances in the diagnosis and detection of or treatment for dementia and Alzheimer’s disease. Early onset dementia, however, is often misdiagnosed as Alzheimer’s disease rather than due to vascular dementias, frontotemporal lobar degenerations, traumatic head injury, or alcohol-related dementia, and that misdiagnosis can have adverse effects on outcomes (Mendez, 2006). A basic understanding of its pathogenesis is lacking, despite improved understanding of the role of plaques and tangles in Alzheimer’s disease. Much work remains to identify risk factors, clarify any potential roles of hormones, and improve treatments. To make progress in Alzheimer’s disease or dementia, a major scientific research thrust needs to occur, as has occurred for other diseases
where progress has been made. Research needs to provide evidence on the natural history of the disease, identification of risk factors to lead to prevention efforts, biomarkers for diagnosis, and measurement of the efficacy of new treatments.
Lessons Learned
The severely debilitating effects of Alzheimer’s disease and other dementias emphasize the importance of looking beyond mortality end points to improve quality of life and to consider health and prevention over the entire lifetime to identify ways earlier in life to prevent such conditions as Alzheimer’s later in life.
Gynecologic Cancers Other Than Cervical Cancer
The committee considers gynecologic cancers other than cervical cancer—corpus and uterine cancer, as well as ovarian cancer—to be a category of diseases in which there has been little progress over the last 20 years. Few gains have been made on improving survival rates.
Incidence, Prevalence, and Mortality in Women
Corpus and uterine cancer, most of which is endometrial cancer and will be referred to as such here, are the most common cancers of the female reproductive organs. It is estimated that there will be 42,160 new cases of endometrial cancer in the United States in 2009 (ACS, 2009d; NCI, 2009b). Most cases occur in women over 55 years old; the median age at diagnosis was 62 years and the median age at death was 72 years of age in 2002–2006 (NCI, 2009b). The American Cancer Society (ACS, 2009d) estimates that about 7,780 women in the United States will have died from endometrial cancer during 2009.
Ovarian cancer is the ninth-most common cancer in women (not counting skin cancer) and the fifth-leading cause of cancer death in women in the United States. It is estimated that there will have been 21,880 new cases of ovarian cancer and 13,850 deaths from it in the United States in 2010 (ACS, 2010b). Most women who have ovarian cancer receive the diagnosis when they are 60 years old or older (ACS, 2010b). Only modest gains have been made in survival of ovarian cancer over the last few decades (Cho and Shih Ie, 2009). Although the 5-year survival rate of women who receive the diagnosis in early stages (localized) of the disease is > 93%, women who are in an advanced stage (distant, cancer has metastasized) have a 5-year survival rate of < 28% (NCI, 2010d).
Disparities Among Groups
Endometrial cancer is more common in white women than black women (incidence, 24.4 per 100,000 in white women and 20.6 per 100,000 in black women), but black women are more likely to die from it (mortality, 7.2 per 100,000 and 3.9 per 100,000 for black and white women, respectively) (NCI, 2010e). The incidence of and deaths from endometrial cancer are lower in Asian and Pacific Islander women, American Indian and Alaskan Native women, and Hispanic women (incidence rates 17.3, 16.8, and 17.6 per 100,000 respectively; mortality, 2.5, 2.9, and 3.0 per 100,000, respectively) (NCI, 2010e).
Research Advances in Knowledge of Biology
Research on endometrial cancer has investigated biomarkers on endometrial-cancer cells, including the progesterone receptor, IGFR I, retinaldehyde dehydrogenase type II, and secreted frizzled-related protein 4 (Boruban et al., 2008). Such biomarkers have not yet been adequately characterized for use in diagnosis or for the development of treatments.
Evidence indicates that there are genetic factors in ovarian cancer; risk is higher in women who have a history of ovarian or breast cancer in a first-degree relative (Clarke-Pearson, 2009). Ovarian cancers are classified by type and degree of differentiation, but their management is not targeted toward specific classes of these cancers. Recent data indicate that the different types of tumors might have different underlying pathology that could be considered in the development of targeted treatments (Cho and Shih Ie, 2009). The development of rapid phenotypic analyses through proteomics could lead to improved diagnostic, screening and therapeutic tools (Faca et al., 2009).
Research Advances in Prevention
Physical activity is associated with reduced risk of endometrial cancer; the risk in physically active women is 20–40% lower than in inactive women (Friedenreich et al., 2010). The greatest reduction in risk is among those who have the highest levels of physical activity (Kramer and Wells, 1996). Several of the studies, however, did not adjust for the postmenopausal use of unopposed estrogen, which increases endometrial cancer risk (NCI, 2009b). Physical activity may reduce risk through effects on hormones (Kramer and Wells, 1996) or by lowering body fat (AICR, 2007); a few studies have examined how body weight and physical activity interact to affect the incidence of endometrial cancer, but the findings have been too inconsistent to support conclusions (NCI, 2009b). Morbidly obese women who have endometrial cancer have a greater likelihood of dying from their comorbidities or their cancers than leaner women who have endometrial cancer (Fader et al., 2009).
The BRCA2 gene mutation that greatly increases the risk of breast cancer also greatly increases the risk of ovarian cancer (Clarke-Pearson, 2009). Women who test positive for the BRCA2 gene mutation, especially those who have had breast cancer, have the option of a bilateral oophorectomy to prevent ovarian cancer.
Studies of the role of physical activity in the prevention of ovarian cancer have yielded inconsistent results, ranging from a reduced risk of ovarian cancer with activity to no association or a slight increase in risk with greater activity (Bertone et al., 2001; Biesma et al., 2006; Mink et al., 1996). One study found that sedentary behavior increases risk, but it did not decrease with light or moderate physical activity (Patel et al., 2006a). Physical activity helps in managing weight, which may be a risk factor for ovarian cancer, but how weight is related to the development of ovarian cancer is not well understood (AICR, 2007).
There is “limited suggestive evidence” of an association between intake of nonstarchy vegetables and a decrease in ovarian cancer (AICR, 2007; IOM, 2003). A pooled analysis of 12 prospective studies, however, found only a non-significant decrease in risk in women who had the highest intake of nonstarchy vegetables (Koushik et al., 2005). Another review of 7 cohort and 27 case–control studies found some evidence that vegetable but not fruit consumption has beneficial effects on the risk of ovarian cancer. So far, evidence of a role of other dietary exposures in ovarian-cancer risk is largely inconclusive (AICR, 2007). A possible increased risk caused by high meat consumption has been observed (for example, Schulz et al., 2004). In the WHI dietary-modification trial, women randomized to a low-fat diet had a statistically significant 40% reduction in risk of ovarian cancer compared with women on a usual diet looking at the last 4.1 years of a 8.1-year followup (Prentice et al., 2007). Results did not appear to be affected by the weight loss experienced by the dietary-change group or by a family history of ovarian cancer (Prentice et al., 2007). The total number of ovarian-cancer cases was relatively small, however. Having low lifetime exposure to estrogen—including low exposure because of late menarche, bearing children, and early menopause—also reduces risk (ACS, 2009e; Barber, 1987; Gates et al., 2010; Hankinson et al., 1995; Jordan et al., 2005). Epidemiologic studies found that oral contraceptive use is protective against both endometrial and ovarian cancer (Deligeoroglou et al., 2003).
Research Advances in Diagnosis
Endometrial cancer is diagnosed histologically. Pathologists are sometimes inconsistent in their diagnosis of endometrial cancer (Boruban et al., 2008; Zaino et al., 2006).
Ovarian cancer is also diagnosed histologically (Cho and Shih Ie, 2009). Over two-thirds of ovarian-cancer cases have progressed to stage 3 or 4, which involves the peritoneal cavity or other organs, when diagnosed (Clarke-Pearson,
2009). There is no routine screening for ovarian cancer. Large randomized trials, such as the Prostate, Lung, Colon, and Ovarian Cancer Screening Trial (PLCO), give promise of determining the efficacy of ovarian-cancer screening for reducing mortality (Clarke-Pearson, 2009). Serum CA125 assay, physical examination, and imaging examination (transvaginal ultrasonography) have been used with differing schedules for followup of asymptomatic women who had a prior ovarian-cancer diagnosis, with increased CA125 indicative of relapse in 56–94% of cases (Gadducci and Cosio, 2009). However, transvaginal ultrasonography and measurement of serum antigen CA125 are not specific for ovarian cancer (Roett and Evans, 2009).
Research Advances in Treatment
Surgery followed by radiotherapy is the main treatment for localized endometrial cancer, the most common presentation of the disease (Deeks, 2007). The combination of chemotherapy and hormone therapy is standard for advanced and recurrent endometrial cancer (Chaudhry and Asselin, 2009). The development of minimally invasive surgical techniques has decreased morbidity and improved quality of life in women after hysterectomy (Fiorelli et al., 2008). Hogberg (2008) reviewed data from a phase III trial conducted by the Gynecologic Oncology Group that demonstrated improved survival but increased toxicity, depending on the treatment regimen used. Adjuvants have been shown to reduce locoregional recurrence in stage 1 disease but not the risk of death (Deeks, 2007).
Stage and histologic grade of cancer at diagnosis, presence or absence of residual disease at the completion of initial surgery, functional stage and age, and use or nonuse of platinum-based chemotherapy are all important prognostic factors for ovarian cancer (Clarke-Pearson, 2009). New surgery and treatments with combinations of cytotoxic drugs have improved 5-year survival rates, but the overall cure rate remains around 30% (Bast et al., 2009).
Knowledge Gaps
Despite the increased efficacy and decreased toxicity of treatments, few gains have been made over the last 20 years in survival rates of people who have advanced endometrial cancer. Most women who receive a diagnosis of ovarian cancer present with advanced disease, and their long-term survival remains low.
Lessons Learned
The lack of progress on gynecologic cancers highlights the need for continued research on women’s health. The high mortality rate for some of these cancers that accompany a typical presentation with advanced-stage disease and poor
prognosis highlight the disadvantages of not having a diagnostic test that allows early detection at a point in disease progression when therapy can save lives.
Gynecologic Disorders (Nonmalignant)
Nonmalignant gynecologic disorders are common in women. They include uterine fibroids, endometriosis, chronic pelvic pain, pelvic floor disorders (including pelvic organ prolapse), polycystic ovarian syndrome, and STIs.24 These disorders cause morbidity in many women and greatly affect their quality of life. The committee considers there to have been little progress made against these conditions over the last 20 years, including in methods of detection and screening, as well as prognosis and therapy for those who develop the disease.
Incidence, Prevalence, and Mortality in Women
Uterine fibroids, or leiomyomas, are the most common solid pelvic tumors in women and cause symptoms in about 25% of women of reproductive age (Cramer and Patel, 1990). Data from several studies indicate that endometriosis affects up to 10% of women in the general population and up to 50% of premenopausal women, with a prevalence of 38% (range, 20–50%) in infertile women and in 71–87% in women who have chronic pelvic pain (Balasch et al., 1996; Carter, 1994; Koninckx et al., 1991; Ling, 1999). Of women of reproductive age, about 7% have hyperandrogenic chronic anovulation or polycystic ovarian syndrome (PCOS) (Azziz et al., 2004). An estimated one-third of all US women have some type of pelvic-floor disorder in their lifetimes. In a recent study, the weighted prevalence of at least one pelvic floor disorder was 23.7%, and 15.7% of these women experienced urinary incontinence (Nygaard et al., 2008). In a study of over 5,000 women 18–50 years old, about 15% had chronic pelvic pain in the last 3 months (Mathias et al., 1996). Finally, except for colds and influenza, STIs are the most common contagious diseases in the United States, and some are increasing in incidence, with millions of new cases of STIs each year (see Chapter 2).
Disparities Among Groups
Black women have an increased risk of uterine fibroids (Evans and Brunsell, 2007; Okolo, 2008) as well as a higher percentage of hysterectomies in women who are diagnosed with uterine fibroids (68% of black women diagnosed with uterine fibroids; 33% of white women) (Keshavarz et al., 2002). Black women have higher rates of chlamydiosis, gonorrhea, and primary and secondary syphilis than other racial or ethnic groups (CDC, 2008f). Those disparities are not ex-
plained by relationship and partner attributes (for example, marital history or sex with older, casual, and nonmonogamous partners (Harawa et al., 2003).
Research Advances in Knowledge of Biology
The primary cause and pathogenesis of uterine fibroids, and of racial disparities in fibroid biology, remain largely unknown (Andersen, 1998; Chen et al., 2001). Endometriosis is understood to constitute a complex interaction between endometrial genes and hormones, but many questions remain. The etiology of PCOS, pelvic-floor disorders, and pelvic pain remains poorly understood, and there has been little advance in understanding the underlying biologic mechanisms of these conditions. Researchers have decoded the genetic makeup of several common STIs, and these may lead to new pathways for treatment and diagnosis and to a vaccine strategy (Carlton et al., 2007).
Research Advances in Prevention
No substantial advances have been made in preventing nonmalignant gynecologic disorders.
Research Advances in Diagnosis
There has been little advance in the diagnosis of many nonmalignant gynecologic conditions over the last 15 years. A detailed history, physical examination, and imaging studies remain the mainstay of diagnosis of gynecologic disorders. Genetic testing for nonmalignant gynecologic conditions has not been used because of a lack of understanding of potential targets. However, polymerasechain-reaction testing has become more available for STIs and allow one-swab testing for multiple STIs.
Research Advances in Treatment
Although there has been very little advance in prevention, the treatment of gynecologic disorders has advanced. New medical and surgical treatments for uterine fibroids have evolved, although there is no defined gold standard on which to base practice (David and Ebert, 2005; Rougier-Chapman et al., 2001). A variety of grafts and meshes have recently been advocated for treatment for prolapse although a greater evidence base demonstrating the safety and efficacy of these products is needed (Silva and Karram, 2005). Pharmacologic and surgical treatment options are available for treatment for endometriosis, and new medical treatments—such as the levonorgestrel intrauterine device, selective progesterone-receptor modulators, angiogenesis inhibitors, and aromatase inhibitors—have shown some promise in the last decade. A few options are
available for treating PCOS and pelvic pain; treatment is largely symptom based and empirical (Chwalisz et al., 2004; Ferrero et al., 2009; Lockhat et al., 2004; Scarpellini et al., 2002).
Hysterectomy is an invasive procedure with complication rates (including infection, hemorrhage, thromboembolism, injury to adjacent organs, and death) of 17 to 23% (Makinen et al., 2001) and long-term effects on psychosexual health (McPherson et al., 2005). Despite those complications and new alternate options, about 600,000 hysterectomies are performed every year in the United States, making it the second most frequent major surgical procedure among reproductive-aged women (CDC, 2004); fibroid tumors, endometriosis, and uterine prolapse are the three conditions most associated with hysterectomies (CDC, 2004).
More progress has been made with STIs. Over the last decade, over 60 substances were identified as potential intravaginal or rectal microbicides, and about one-fourth of them have reached various stages of human testing (Fleck, 2004; Ramjee et al., 2000). A number of studies have examined the safety, efficacy, and tolerability of topical microbicides in preventing STIs, and new vaccine strategies have evolved (Harwell et al., 2003; Mayer et al., 2001).
Knowledge Gaps
Research on nonmalignant gynecologic conditions is sparse. Little is known about their underlying biology or prevention and treatment strategies. No methods controlled by women for preventing STIs are available. STI rates are increasing in high-risk populations, and a goal of eradication of STIs or a more aggressive targeting of STIs may be a potential public-health strategy.
Lung Cancer
Despite substantial research on lung and bronchial cancer,25 including studies that analyze data on women separately, little progress has been made in decreasing lung-cancer mortality in women. It remains the leading cause of cancer death in women in the United States, and there are few diagnostic, screening, and treatment options. The committee, therefore, considers lung cancer a disease where research has led to little progress against the disease.
Incidence, Prevalence, and Mortality in Women
Changing patterns of cigarette use led to greater increases in rates of lung cancer in women than in men; the rate in women increased by a factor of 4 from
the early 1960s to 2000 (Thomas et al., 2005).26 In 2007, about one-fifth of US women 18 years old or older were current smokers27 (CDC, 2008g). Lung cancer accounted for only 3% of female cancer deaths in 1950 and 25% in 2000 (HHS, 2001), and the estimate in 2008 is 26% (HRSA, 2008). Recent data indicate that death rates from lung cancer in women have leveled off since 2003 in some states and regions (Jemal et al., 2008).
The incidence of lung cancer is the second highest cancer incidence in women; there were an estimated 105,770 new cases in 2010, and the median age at diagnosis was 71. An estimated 71,080 women will die from lung cancer in the United States in 2010 at a median age of 72 years (NCI, 2010f).
Women and men with less than a high school education had a higher risk of lung cancer compared to college-educated women and men (Clegg et al., 2009). Similarly, women and men with an annual family income less than $12,500 had a significantly higher incidence of lung cancer compared to women and men with an annual family income of $50,000 or more (Clegg et al., 2009).
Disparities Among Groups
The incidence of lung cancer is similar in white and black women (54.7 per 100,000 and 54.8 per 100,000, respectively), but it is lower in American Indian and Alaskan Native women (39.7 per 100,000), Asian and Pacific Islander (28.1 per 100,000), and Hispanic women (25.4 per 100,000) (NCI, 2010f). Some evidence suggests that among cigarette-smokers, black and native Hawaiian women are more susceptible to lung cancer than white, Japanese American, and Latina women (Haiman et al., 2006). Lung-cancer incidence is also greater in those who have lower income (Clegg et al., 2009).
Many studies have demonstrated differences between blacks and whites in the lung-cancer treatment received (Farjah et al., 2009; Hardy et al., 2009). There were also great disparities in treatment between people in the highest socioeconomic quartile and older patients, women, and those in lower socioeconomic quartiles. The disparity (between blacks and whites) in treatment did not narrow much in the last 12 years. Factors other than doctors’ recommendations—such as distrust, beliefs and perceptions about lung cancer and treatment for it, and lower access to care despite insurance—might be involved in racial disparities (Farjah et al., 2009).
Socioeconomic status plays a large role in mortality from lung cancer in both men and women. Lower education levels are associated with higher lung-cancer
|
26 |
Lung-cancer deaths in men decreased over the same period; this reflects the decrease in men’s smoking, which in earlier periods had been much higher than women’s, but which has been declining faster than women’s (see Chapter 2 for discussion of smoking). |
|
27 |
Defined as “those who smoked more than 100 cigarettes in their lifetime and now smoke every day or some days” (CDC, 2008g). |
incidence and mortality (Alberg and Nonemaker, 2008; Clegg et al., 2009; Kinsey et al., 2008). Mortality from lung cancer decreased from 1993 to 2001 in people who had at least 16 years of education in every sex and race stratum except black women, whose mortality was stable; it increased in white women who had less than 12 years of education (Kinsey et al., 2008).
Research Advances in Understanding of Underlying Biology
It is unknown whether there are sex differences in the susceptibility to lung cancer (Planchard et al., 2009). A number of studies have found that women have a higher risk of lung cancer than men (Brownson et al., 1992; Harris et al., 1993; Henschke and Miettinen, 2004; Henschke et al., 2006; McDuffie et al., 1987; Osann et al., 1993; Risch et al., 1993; Zang and Wynder, 1996), but other studies have found no difference (Bain et al., 2004; Freedman et al., 2008; Kreuzer et al., 2000; Thun et al., 2002, 2008). A meta-analysis of data from the Nurses’ Health Study and the Health Professionals Follow-Up Study found no sex difference in lung-cancer rates in women and men with comparable smoking histories (Bain et al., 2004).
Research has looked at a number of genetic and cellular differences that might play a role in potential sex differences in the susceptibility to and survival of lung cancer. The data are not clear and seem to vary among lung-cancer types, but there is some evidence that oncogene activity could play a role (Ahrendt et al., 2001; D’Amico et al., 2000; Nelson et al., 1999). Research has also looked at differences in gastrin-releasing peptide receptor (Shriver et al., 2000), HER-2/neu (Negro et al., 2004; Vallbohmer et al., 2006), PTHrP (Montgrain et al., 2007), cytochrome p450 1A1 expression (Dresler et al., 2000; Mollerup et al., 1999), and DNA-repair capacity (Spitz et al., 2003; Wei et al., 2000). Estrogen could also be involved; there is avariant of ERα in lung cells (Mollerup et al., 2002), and non–small-cell lung carcinoma (NSCLC) responds to estrogen (Stabile et al., 2002).
Research Advances in Prevention
Smoking is estimated to cause about 95% of lung cancer cases, so understanding the reasons that women initiate and continue smoking is critical (see Chapter 2). Given the high incidence of lung cancer in women, the cases not related to smoking still constitute a substantial public-health problem. Little is known about other preventive measures for lung cancer. Some research has demonstrated that diet affects susceptibility to lung cancer, especially in women (Cranganu and Camporeale, 2009).
Research Advances in Diagnosis
Posttreatment survival of lung cancer is much higher if it is diagnosed at an earlier stage, when tumors are smaller (ACS, 2009f). Asymptomatic people are not screened routinely for lung cancer (Smith et al., 2009), and the data are mixed as to whether screening of them would be effective. Some data indicate that screening increases 10-year survival (with a survival rate of 88%) (Henschke et al., 2006); other data show an increase in the number of new cases of lung cancer and the number of resections in a trial of computer tomographic scanning but no concomitant decrease in advanced lung cancer or in lung-cancer deaths (Bach et al., 2007)—a suggestion of the importance of how a screening method is assessed (Black and Baron, 2007). Two large clinical trials—the National Lung Screening Trial and the PLCO Cancer Screening Trial—are underway and it is hoped that they will clarify whether screening results in decreased mortality from lung cancer. Computer-aided detection can detect about half the lesions that are overlooked by initial interpretation by a radiologist, so it holds promise for improving lung-cancer screening (White and Spitz, 1993).
Research Advances in Treatment
The rate of survival of lung cancer (measured as years of survival after diagnosis or treatment) is better in women than in men, and women typically live longer after treatment (Henschke et al., 2006; Ouellette et al., 1998). Some suggest that stage at diagnosis underlies the differences (Ferguson et al., 2000), but others have found better overall survival in women even if they have inoperable NSCLC (McGovern et al., 2009; Visbal et al., 2004). The longer survival in women does not appear to be treatment specific, having been seen after radiotherapy (Werner-Wasik et al., 1999), chemotherapy (Albain et al., 1991; Lord et al., 2002; Simon et al., 2008; Tsao et al., 2006; Wheatley-Price et al., 2009; Yamamoto et al., 2008), and surgery (Bouchardy et al., 1999; Keller et al., 2002). Although Wheatley-Price and colleagues (2009) and Yamamoto and colleagues (2008) observed better survival in women after chemotherapy, they also noted its greater toxicity in women, which would affect their quality of life. Prognostic markers may be sex specific (Dannenberg et al., 2003).
Knowledge Gaps
Despite a large amount of research, including research in women, lung cancer remains the leading cause of cancer deaths in women; the 5-year survival rate if it is detected at a late stage, which occurs in 45% of patients, is 15% (Pearman, 2008). Study results on what sex differences exist are at times contradictory.
Lessons Learned
The increase in survival observed after some treatments (for example, resection) and the contradictory results of screening programs highlight the importance of clinical trials of early detection of lung cancer. The fact that many cases of lung cancer are considered preventable, that is, they would not have occurred in the absence of smoking, highlights the need for effective public-health interventions.
CONCLUSIONS
-
For those conditions where substantial progress has been made in improving women’s health—breast cancer, cardiovascular disease, and cervical cancer—there is a large and diverse body of research from the last 20 years, including basic research at the molecular, cellular and organ level; animal studies; observational studies; and small and large randomized controlled clinical trials (for example, the WHI). That research led to advances in the understanding of the underlying biology and pathophysiology of the disease, which in turn led to advances in the prevention and diagnosis of and treatment for those conditions in women. Research has resulted in improved diagnostic tests and screening for diseases such as breast and cervical cancer; vaccines against a virus that causes cervical cancer, making prevention of cervical cancer possible; new treatments for cardiovascular disease; and targeted therapies for breast cancer. Although much work remains and the incidence and mortality for some of those diseases remains high, those advances have led to decreases in prevalence (for example, of cervical cancer) or in mortality (for example, from breast cancer and cardiovascular disease).
-
Fewer gains, however, have been made in many other conditions that affect women’s health.
-
Even for conditions where research has led to progress and improved health outcomes for women in general, disparities or inequities in that progress remain among groups of women, with some populations carrying a much greater burden of disease.
-
Even when great progress has been made through scientific advances, other factors in or determinants of health can present barriers to improving women’s health, such as the effects of societal beliefs or morals on the use of the vaccine for cervical cancer, social acceptance of and stigmas attached to depression, and use of and compliance with use of contraceptives to prevent unintended pregnancy.
-
Knowledge about differences in the manifestation of diseases is crucial for further studies to identify the underlying biology of disease in women vs men and to develop appropriate prevention, diagnosis, and treatment strategies for women.
REFERENCES
AARDA (American Autoimmune Related Diseases Association) 2010. Autoimmunity: A Major Women’s Health Issue. http://www.aarda.org/women_and_autoimmunity.php (accessed May 25, 2010).
Abbott, T. A., 3rd, B. J. Lawrence, and S. Wallach. 1996. Osteoporosis: The need for comprehensive treatment guidelines. Clinical Therapeutics 18(1):127–149; discussion 126.
Ackerman, L. S. 2006. Sex hormones and the genesis of autoimmunity. Archives of Dermatology 142(3):371–376.
ACOG (American Congress of Obstetricions and Gynecologists). 2003. Cervical Cancer Screening: Testing Can Start Later and Occur Less Often Under New ACOG Recommendations. http://www.acog.org/from_home/publications/press_releases/nr07-31-03-1.cfm (accessed November 15, 2009).
———. 2009. ACOG practice bulletin no. 109: Cervical cytology screening. Obstetrics and Gynecology 114(6):1409–1420.
ACS (American Cancer Society). 2009a. Detailed Guide: Breast Cancer What Are the Key Statistics for Breast Cancer? http://www.cancer.org/docroot/cri/content/cri_2_4_1x_what_are_the_key_statistics_for_breast_cancer_5.asp (accessed May 3, 2010).
———. 2009b. Detailed Guide: Cervical Cancer What Are the Key Statistics About Cervical Cancer? http://www.cancer.org/docroot/CRI/content/CRI_2_4_1X_What_are_the_key_statistics_for_cervical_cancer_8.asp (accessed November 19, 2009).
———. 2009c. American Cancer Society Guidelines for the Early Detection of Cancer. http://www.cancer.org/docroot/ped/content/ped_2_3x_acs_cancer_detection_guidelines_36.asp (accessed November 17, 2009).
———. 2009d. Overview: Endometrial Cancer: How Many Women Get Endometrial Cancer? http://www.cancer.org/docroot/CRI/content/CRI_2_2_1x_How_Many_Women_Get_Endometrial_Cancer.asp?sitearea= (accessed 2009).
———. 2009e. Detailed Guide: Ovarian Cancer. What Are the Risk Factors for Ovarian Cancer? http://www.cancer.org/docroot/CRI/content/CRI_2_4_2X_What_are_the_risk_factors_for_ovarian_cancer_33.asp (accessed May 25, 2010).
———. 2009f. Detailed Guide: Lung Cancer—Non-Small Cell How Is Non-Small Cell Lung Cancer Staged? http://www.cancer.org/docroot/CRI/content/CRI_2_4_3x_How_Is_Non-Small_Cell_Lung_Cancer_Staged.asp (accessed December 14, 2009).
———. 2010a. Breast Cancer Facts & Figures: 2009–2010. Atlanta, GA:ACS, Inc..
———. 2010b. Ovarian Cancer. http://www.cancer.org/Cancer/OvarianCancer/DetailedGuide/ovarian-cancer-key-statistics (accessed August 10, 2010).
Ademuyiwa, F. O., and O. I. Olopade. 2003. Racial differences in genetic factors associated with breast cancer. Cancer and Metastasis Reviews 22(1):47–53.
AHA (American Heart Association). 2009. Women and Cardiovascular Diseases—Statistics 2009. http://www.americanheart.org/downloadable/heart/1236184538758WOMEN.pdf (accessed December 1, 2009).
———. 2010. Heart Attack Symptoms and Warning Signs. http://www.americanheart.org/presenter.jhtml?identifier=4595 (accessed May 24, 2010).
Ahmed, B., C. N. Bairey Merz, B. D. Johnson, V. Bittner, S. L. Berga, G. D. Braunstein, T. K. Hodgson, K. Smith, L. Shaw, S. F. Kelsey, and G. Sopko. 2008. Diabetes mellitus, hypothalamic hypoestrogenemia, and coronary artery disease in premenopausal women (from the National Heart, Lung, and Blood Institute sponsored WISE study). American Journal of Cardiology 102(2):150–154.
Ahrendt, S. A., P. A. Decker, E. A. Alawi, Y. R. Zhu Yr, M. Sanchez-Cespedes, S. C. Yang, G. B. Haasler, A. Kajdacsy-Balla, M. J. Demeure, and D. Sidransky. 2001. Cigarette smoking is strongly associated with mutation of the k-ras gene in patients with primary adenocarcinoma of the lung. Cancer 92(6):1525–1530.
AHRQ (Agency for Healthcare Research and Quality). 2009. Cardiovascular Disease and Other Chronic Conditions in Women: Recent Findings. Rockville, MD: AHRQ.
AICR (American Institute for Cancer Research). 2007. Food, Nutrition, Physical Activity, and the Prevention of Cancer: A Global Perspective. Washington, DC: AICR.
Aitken, R. J., M. A. Baker, G. F. Doncel, M. M. Matzuk, C. K. Mauck, and M. J. Harper. 2008. As the world grows: Contraception in the 21st century. Journal of Clinical Investigation 118(4):1330–1343.
Alarcon-Riquelme, M. E. 2007. Recent advances in the genetics of autoimmune diseases. Annals of the New York Academy of Sciences 1110:1–9.
Albain, K. S., J. J. Crowley, M. LeBlanc, and R. B. Livingston. 1991. Survival determinants in extensive-stage non–small-cell lung cancer: The Southwest Oncology Group experience. Journal Clinical Oncology 9(9):1618–1626.
Albain, K. S., W. E. Barlow, S. Shak, G. N. Hortobagyi, R. B. Livingston, I. T. Yeh, P. Ravdin, R. Bugarini, F. L. Baehner, N. E. Davidson, G. W. Sledge, E. P. Winer, C. Hudis, J. N. Ingle, E. A. Perez, K. I. Pritchard, L. Shepherd, J. R. Gralow, C. Yoshizawa, D. C. Allred, C. K. Osborne, and D. F. Hayes. 2010. Prognostic and predictive value of the 21-gene recurrence score assay in postmenopausal women with node-positive, oestrogen-receptor-positive breast cancer on chemotherapy: A retrospective analysis of a randomised trial. Lancet Oncology 11(1):55–65.
Alberg, A. J., and J. Nonemaker. 2008. Who is at high risk for lung cancer? Population-level and individual-level perspectives. Seminars in Respiratory and Critical Care Medicine 29(3): 223–232.
Alberts, D. S., and N. Mason-Liddil. 1989. The role of cisplatin in the management of advanced squamous cell cancer of the cervix. Seminars in Oncology 16(4 Suppl. 6):66–78.
Alegría, M., G. Canino, P. E. Shrout, M. Woo, N. Duan, D. Vila, M. Torres, C. N. Chen, and X. L. Meng. 2008. Prevalence of mental illness in immigrant and non-immigrant US Latino groups. American Journal of Psychiatry 165(3):359–369.
Alexander, F. E., T. J. Anderson, H. K. Brown, A. P. Forrest, W. Hepburn, A. E. Kirkpatrick, B. B. Muir, R. J. Prescott, and A. Smith. 1999. 14 years of follow-up from the Edinburgh randomised trial of breast-cancer screening. Lancet 353(9168):1903–1908 .
Alexander, K. P., A. Y. Chen, L. K. Newby, J. B. Schwartz, R. F. Redberg, J. S. Hochman, M. T. Roe, W. B. Gibler, E. M. Ohman, and E. D. Peterson. 2006. Sex differences in major bleeding with glycoprotein IIB/IIIA inhibitors: Results from the CRUSADE (Can Rapid risk stratification of Unstable angina patients Suppress ADverse outcomes with Early implementation of the ACC/AHA guidelines) initiative. Circulation 114(13):1380–1387.
Alliegro, M. B., M. Dorrucci, A. N. Phillips, P. Pezzotti, S. Boros, M. Zaccarelli, R. Pristera, and G. Rezza. 1997. Incidence and consequences of pregnancy in women with known duration of HIV infection. Italian Seroconversion Study Group. Archives of Internal Medicine 157(22): 2585–2590.
Ancelin, M. L., and K. Ritchie. 2005. Lifelong endocrine fluctuations and related cognitive disorders. Current Pharmaceutical Design 11(32):4229–4252.
Andersen, J. 1998. Factors in fibroid growth. Baillière’s Clinical Obstetrics and Gynaecology 12(2): 225–243.
Anderson, R. J., K. E. Freedland, R. E. Clouse, and P. J. Lustman. 2001. The prevalence of comorbid depression in adults with diabetes: A meta-analysis. Diabetes Care 24(6):1069–1078.
Andersson, I., and L. Janzon. 1997. Reduced breast cancer mortality in women under age 50: Updated results from the Malmo Mammographic Screening Program. Journal of the National Cancer Institute Monographs (22):63–67.
Ando, T., and T. F. Davies. 2003. Postpartum autoimmune thyroid disease: The potential role of fetal microchimerism. Journal of Clinical Endocrinology and Metabolism 88(7):2965–2971.
Angold, A., E. J. Costello, and C. M. Worthman. 1998. Puberty and depression: The roles of age, pubertal status and pubertal timing. Psychological Medicine 28(1):51–61.
Antinori, A., F. Baldini, E. Girardi, A. Cingolani, M. Zaccarelli, S. Di Giambenedetto, A. Barracchini, P. De Longis, R. Murri, V. Tozzi, A. Ammassari, M. G. Rizzo, G. Ippolito, and A. De Luca. 2001. Female sex and the use of anti-allergic agents increase the risk of developing cutaneous rash associated with nevirapine therapy. AIDS 15(12):1579–1581.
Antithrombotic Trialists Collaboration. 2002. Collaborative meta-analysis of randomised trials of antiplatelet therapy for prevention of death, myocardial infarction, and stroke in high risk patients. British Medical Journal 324(7329):71–86.
Antoniou, A. C., A. P. Cunningham, J. Peto, D. G. Evans, F. Lalloo, S. A. Narod, H. A. Risch, J. E. Eyfjord, J. L. Hopper, M. C. Southey, H. Olsson, O. Johannsson, A. Borg, B. Pasini, P. Radice, S. Manoukian, D. M. Eccles, N. Tang, E. Olah, H. Anton-Culver, E. Warner, J. Lubinski, J. Gronwald, B. Gorski, L. Tryggvadottir, K. Syrjakoski, O. P. Kallioniemi, H. Eerola, H. Nevanlinna, P. D. Pharoah, and D. F. Easton. 2008. The BOADICEA model of genetic susceptibility to breast and ovarian cancers: Updates and extensions. British Journal of Cancer 98(8):1457–1466.
Arbyn, M., C. Bergeron, P. Klinkhamer, P. Martin-Hirsch, A. G. Siebers, and J. Bulten. 2008. Liquid compared with conventional cervical cytology: A systematic review and meta-analysis. Obstetrics and Gynecology 111(1):167–177.
Asarnow, J. R., L. H. Jaycox, N. Duan, A. P. LaBorde, M. M. Rea, P. Murray, M. Anderson, C. Landon, L. Tang, and K. B. Wells. 2005. Effectiveness of a quality improvement intervention for adolescent depression in primary care clinics: A randomized controlled trial. Journal of the American Medical Association 293(3):311–319.
Ascherio, A., and K. L. Munger. 2007. Environmental risk factors for multiple sclerosis. Part II: Noninfectious factors. Annals of Neurology 61(6):504–513.
Austin, R. M., and I. Ramzy. 1998. Increased detection of epithelial cell abnormalities by liquid-based gynecologic cytology preparations. A review of accumulated data. Acta Cytologica 42(1):178–184.
Azziz, R., K. S. Woods, R. Reyna, T. J. Key, E. S. Knochenhauer, and B. O. Yildiz. 2004. The prevalence and features of the polycystic ovary syndrome in an unselected population. Journal of Clinical Endocrinology and Metabolism 89(6):2745–2749.
Bach, P. B., J. R. Jett, U. Pastorino, M. S. Tockman, S. J. Swensen, and C. B. Begg. 2007. Computed tomography screening and lung cancer outcomes. Journal of the American Medical Association 297(9):953–961.
Bachman, D. L., P. A. Wolf, R. Linn, J. E. Knoefel, J. Cobb, A. Belanger, R. B. D’Agostino, and L. R. White. 1992. Prevalence of dementia and probable senile dementia of the Alzheimer type in the Framingham Study. Neurology 42(1):115–119.
Bae, S., and L. Zhang. 2005. Gender differences in cardioprotection against ischemia/reperfusion injury in adult rat hearts: Focus on Akt and protein kinase C signaling. Journal of Pharmacology and Experimental Therapeutics 315(3):1125–1135.
Bailey, A. C., and M. Fisher. 2008. Current use of antiretroviral treatment. British Medical Bulletin 87:175–192.
Bailey, A. L., C. L. Campbell, and S. S. Smyth. 2010. Aspirin for the primary prevention of cardiovascular disease in women. Current Cardiovascular Risk Reports 4:209–215.
Bain, C., D. Feskanich, F. E. Speizer, M. Thun, E. Hertzmark, B. A. Rosner, and G. A. Colditz. 2004. Lung cancer rates in men and women with comparable histories of smoking. Journal of the National Cancer Institute 96(11):826–834.
Bairey Merz, C. N., L. J. Shaw, S. E. Reis, V. Bittner, S. F. Kelsey, M. Olson, B. D. Johnson, C. J. Pepine, S. Mankad, B. L. Sharaf, W. J. Rogers, G. M. Pohost, A. Lerman, A. A. Quyyumi, and G. Sopko. 2006. Insights from the NHLBI-sponsored Women’s Ischemia Syndrome Evaluation (WISE) Study: Part II: Gender differences in presentation, diagnosis, and outcome with regard to gender-based pathophysiology of atherosclerosis and macrovascular and microvascular coronary disease. Journal of the American College of Cardiology 47(3 Suppl.):S21–S29.
Balasch, J., M. Creus, F. Fabregues, F. Carmona, J. Ordi, S. Martinez-Roman, and J. A. Vanrell. 1996. Visible and non-visible endometriosis at laparoscopy in fertile and infertile women and in patients with chronic pelvic pain: A prospective study. Human Reproduction 11(2):387–391.
Baquet, C. R., S. I. Mishra, P. Commiskey, G. L. Ellison, and M. DeShields. 2008. Breast cancer epidemiology in blacks and whites: Disparities in incidence, mortality, survival rates and histology. Journal of the National Medical Association 100(5):480–488.
Baraona, E., C. S. Abittan, K. Dohmen, M. Moretti, G. Pozzato, Z. W. Chayes, C. Schaefer, and C. S. Lieber. 2001. Gender differences in pharmacokinetics of alcohol. Alcoholism—Clinical and Experimental Research 25(4):502–507.
Barber, H. R. 1987. Ovarian cancer: Cause, diagnosis, and treatment. Comprehensive Therapy 13(6): 25–33.
Barkan, S. E., S. L. Melnick, S. Preston-Martin, K. Weber, L. A. Kalish, P. Miotti, M. Young, R. Greenblatt, H. Sacks, and J. Feldman. 1998. The Women’s Interagency HIV Study. Epidemiology 9(2):117–125.
Barnholtz-Sloan, J., N. Patel, D. Rollison, K. Kortepeter, J. MacKinnon, and A. Giuliano. 2009. Incidence trends of invasive cervical cancer in the United States by combined race and ethnicity. Cancer Causes and Control 20(7):1129–1138.
Barre-Sinoussi, F., J. C. Chermann, F. Rey, M. T. Nugeyre, S. Chamaret, J. Gruest, C. Dauguet, C. Axler-Blin, F. Vezinet-Brun, C. Rouzioux, W. Rozenbaum, and L. Montagnier. 2004. Isolation of a t-lymphotropic retrovirus from a patient at risk for acquired immune deficiency syndrome (AIDS). 1983. Revista de Investigación Clínica 56(2):126–129.
Bartella, L., S. B. Thakur, E. A. Morris, D. D. Dershaw, W. Huang, E. Chough, M. C. Cruz, and L. Liberman. 2007. Enhancing nonmass lesions in the breast: Evaluation with proton (1H) MR spectroscopy. Radiology 245(1):80–87.
Bast, R. C., Jr., B. Hennessy, and G. B. Mills. 2009. The biology of ovarian cancer: New opportunities for translation. Nature Reviews. Cancer 9(6):415–428.
Batur, P., J. Elder, and M. Mayer. 2003. Update on contraception: Benefits and risks of the new formulations. Cleveland Clinic Journal of Medicine 70(8):681–682, 685–686, 668–690 passim.
Beals, J., D. K. Novins, P. Spicer, C. M. Mitchell, and S. M. Manson. 2005. Prevalence of mental disorders and utilization of mental health services in two American Indian reservation populations: Mental health disparities in a national context. American Journal of Psychiatry 162:1723–1732.
Beatson, G. W. 1896. On the treatment of inoperable cases of carcinoma of the mamma: Suggestions for a new method of treatment with illustrative cases. Lancet 148(3802):104–107.
Becker, D. F., and C. M. Grilo. 2006. Prediction of drug and alcohol abuse in hospitalized adolescents: Comparisons by gender and substance type. Behaviour Research and Therapy 44(10): 1431–1440.
Belle, D., and J. Doucet. 2003. Poverty, inequality, and discrimination as sources of depression among US women. Psychology of Women Quarterly 27(2):101–113.
Benagiano, G., C. Bastianelli, and M. Farris. 2007. Hormonal contraception: State of the art and future perspectives. Minerva Ginecologica 59(3):241–270.
Beral, V., C. Hermon, C. Kay, P. Hannaford, S. Darby, and G. Reeves. 1999. Mortality associated with oral contraceptive use: 25 year follow up of cohort of 46 000 women from Royal College of General Practitioners’ Oral Contraception Study. British Medical Journal 318(7176):96–100.
Beretich, B. D., and T. M. Beretich. 2009. Explaining multiple sclerosis prevalence by ultraviolet exposure: A geospatial analysis. Multiple Sclerosis 15(8):891–898.
Berg, C. J., J. Chang, W. M. Callaghan, and S. J. Whitehead. 2003. Pregnancy-related mortality in the United States, 1991–1997. Obstetrics and Gynecology 101(2):289–296.
Berg, C. J., A. P. Mackay, C. Qin, and W. M. Callaghan. 2009. Overview of maternal morbidity during hospitalization for labor and delivery in the United States: 1993–1997 and 2001–2005. Obstetrics and Gynecology 113(5):1075–1081.
Berg, W. A. 2009. Tailored supplemental screening for breast cancer: What now and what next? American Journal of Roentgeneology 192(2):390–399.
Berg, W. A., I. N. Weinberg, D. Narayanan, M. E. Lobrano, E. Ross, L. Amodei, L. Tafra, L. P. Adler, J. Uddo, W. Stein, 3rd, and E. A. Levine. 2006. High-resolution fluorodeoxyglucose positron emission tomography with compression (“positron emission mammography”) is highly accurate in depicting primary breast cancer. Breast Journal 12(4):309–323.
Berg, W. A., J. D. Blume, J. B. Cormack, E. B. Mendelson, D. Lehrer, M. Bohm-Velez, E. D. Pisano, R. A. Jong, W. P. Evans, M. J. Morton, M. C. Mahoney, L. Hovanessian Larsen, R. G. Barr, D. M. Farria, H. S. Marques, K. Boparai, and for the ACRIN 6666 Investigators. 2008. Combined screening with ultrasound and mammography vs mammography alone in women at elevated risk of breast cancer. Journal of the American Medical Association 299(18):2151–2163.
Berger, J. S., M. J. Krantz, J. M. Kittelson, and W. R. Hiatt. 2009. Aspirin for the prevention of cardiovascular events in patients with peripheral artery disease: A meta-analysis of randomized trials. Journal of the American Medical Association 301(18):1909–1919.
Bermejo-Pérez, M. J., S. Márquez-Calderón, and A. Llanos-Méndez. 2007. Effectiveness of preventive interventions in BRCA1/2 gene mutation carriers: A systematic review. International Journal of Cancer 121(2):225–231.
Berndt, E. R., L. M. Koran, S. N. Finkelstein, A. J. Gelenberg, S. G. Kornstein, I. M. Miller, M. E. Thase, G. A. Trapp, and M. B. Keller. 2000. Lost human capital from early-onset chronic depression. American Journal of Psychiatry 157(6):940–947.
Bernstein, L. 2008. Identifying population-based approaches to lower breast cancer risk. Oncogene 27(S2):S3–S8.
Berry, D. A., E. S. Iversen, Jr., D. F. Gudbjartsson, E. H. Hiller, J. E. Garber, B. N. Peshkin, C. Lerman, P. Watson, H. T. Lynch, S. G. Hilsenbeck, W. S. Rubinstein, K. S. Hughes, and G. Parmigiani. 2002. BRCAPRO validation, sensitivity of genetic testing of BRCA1/BRCA2, and prevalence of other breast cancer susceptibility genes. Journal Clinical Oncology 20(11):2701–2712.
Berry, D. A., K. A. Cronin, S. K. Plevritis, D. G. Fryback, L. Clarke, M. Zelen, J. S. Mandelblatt, A. Y. Yakovlev, J. D. F. Habbema, E. J. Feuer, T. C. Intervention, and Surveillance Modeling Network Collaborators. 2005. Effect of screening and adjuvant therapy on mortality from breast cancer. New England Journal of Medicine 353(17):1784–1792.
Bersoff-Matcha, S. J., W. C. Miller, J. A. Aberg, C. van Der Horst, H. J. Hamrick, Jr., W. G. Powderly, and L. M. Mundy. 2001. Sex differences in nevirapine rash. Clinical Infectious Diseases 32(1):124–129.
Bertone, E. R., W. C. Willett, B. A. Rosner, D. J. Hunter, C. S. Fuchs, F. E. Speizer, G. A. Colditz, and S. E. Hankinson. 2001. Prospective study of recreational physical activity and ovarian cancer. Journal of the National Cancer Institute 93(12):942–948.
Biesma, R. G., L. J. Schouten, M. J. Dirx, R. A. Goldbohm, and P. A. van den Brandt. 2006. Physical activity and risk of ovarian cancer: Results from the Netherlands Cohort Study (The Netherlands). Cancer Causes & Control 17(1):109–115.
Bing, E. G., M. A. Burnam, D. Longshore, J. A. Fleishman, C. D. Sherbourne, A. S. London, B. J. Turner, F. Eggan, R. Beckman, B. Vitiello, S. C. Morton, M. Orlando, S. A. Bozzette, L. Ortiz-Barron, and M. Shapiro. 2001. Psychiatric disorders and drug use among human immunodeficiency virus-infected adults in the United States. Archives of General Psychiatry 58(8):721–728.
Birnbaum, C. S., L. S. Cohen, J. W. Bailey, L. R. Grush, L. M. Robertson, and Z. N. Stowe. 1999. Serum concentrations of antidepressants and benzodiazepines in nursing infants: A case series. Pediatrics 104(1):e11.
Bischoff-Ferrari, H. A., B. Dawson-Hughes, J. A. Baron, P. Burckhardt, R. Li, D. Spiegelman, B. Specker, J. E. Orav, J. B. Wong, H. B. Staehelin, E. O’Reilly, D. P. Kiel, and W. C. Willett. 2007. Calcium intake and hip fracture risk in men and women: A meta-analysis of prospective cohort studies and randomized controlled trials. American Journal of Clinical Nutrition 86(6): 1780–1790.
Bischoff-Ferrari, H. A., W. C. Willett, J. B. Wong, A. E. Stuck, H. B. Staehelin, E. J. Orav, A. Thoma, D. P. Kiel, and J. Henschkowski. 2009. Prevention of nonvertebral fractures with oral vitamin D and dose dependency: A meta-analysis of randomized controlled trials. Archives of Internal Medicine 169(6):551–561.
Bishop, J. W., S. H. Bigner, T. J. Colgan, M. Husain, L. P. Howell, K. M. McIntosh, D. A. Taylor, and M. H. Sadeghi. 1998. Multicenter masked evaluation of autocyte prep thin layers with matched conventional smears. Including initial biopsy results. Acta Cytologica 42(1):189–197.
Bjurstam, N., L. Bjorneld, J. Warwick, E. Sala, S. W. Duffy, L. Nystrom, N. Walker, E. Cahlin, O. Eriksson, L. O. Hafstrom, H. Lingaas, J. Mattsson, S. Persson, C. M. Rudenstam, H. Salander, J. Save-Soderbergh, and T. Wahlin. 2003. The Gothenburg Breast Screening Trial. Cancer 97(10):2387–2396.
Black, W. C., and J. A. Baron. 2007. CT screening for lung cancer: Spiraling into confusion? Journal of the American Medical Association 297(9):995–997.
Blake, S. M., H. Amaro, P. M. Schwartz, and L. J. Flinchbaugh. 2001. A review of substance abuse prevention interventions for young adolescent girls. Journal of Early Adolescence 21(3): 294–324.
Blauwet, L. A., and R. F. Redberg. 2007. The role of sex-specific results reporting in cardiovascular disease. Cardiology in Review 15(6):275–278.
Blauwet, L. A., S. N. Hayes, D. McManus, R. F. Redberg, and M. N. Walsh. 2007. Low rate of sex-specific result reporting in cardiovascular trials. Mayo Clinic Proceedings 82(2):166–170.
Blumsohn, A. 2003. Bone remodeling markers: Assessment of fracture risk and fracture risk reduction. Current Osteoporosis Reports 1(3):91–97.
Bonfanti, P., C. Gulisano, E. Ricci, L. Timillero, L. Valsecchi, S. Carradori, L. Pusterla, P. Fortuna, S. Miccolis, C. Magnani, A. Gabbuti, F. Parazzini, C. Martinelli, I. Faggion, S. Landonio, T. Quirino, and G. Vigevani. 2003. Risk factors for lipodystrophy in the CISAI cohort. Biomedicine and Pharmacotherapy 57(9):422–427.
Boone, J., T. R. Nelson, K. K. Lindfors, and J. A. Seibert. 2001. Dedicated breast CT: Radiation dose and image quality evaluation. Radiology 221(3):657–667.
Boone, J., A. Kwan, K. Yang, G. Burkett, K. Lindfors, and T. Nelson. 2006. Computed tomography for imaging the breast. Journal of Mammary Gland Biology and Neoplasia 11(2):103–111.
Boonen, S., P. Lips, R. Bouillon, H. A. Bischoff-Ferrari, D. Vanderschueren, and P. Haentjens. 2007. Need for additional calcium to reduce the risk of hip fracture with vitamin D supplementation: Evidence from a comparative metaanalysis of randomized controlled trials. Journal of Clinical Endocrinology and Metabolism 92(4):1415–1423.
Bordeleau, L., S. Panchal, and P. Goodwin. 2010. Prognosis of BRCA-associated breast cancer: A summary of evidence. Breast Cancer Research and Treatment 119(1):13–24.
Borer, K. T. 2005. Physical activity in the prevention and amelioration of osteoporosis in women—Interaction of mechanical, hormonal and dietary factors. Sports Medicine 35(9):779–830.
Boruban, M. C., K. Altundag, G. S. Kilic, and J. Blankstein. 2008. From endometrial hyperplasia to endometrial cancer: Insight into the biology and possible medical preventive measures. European Journal of Cancer Prevention 17(2):133–138.
Bosch, F. X., A. Lorincz, N. Munoz, C. J. Meijer, and K. V. Shah. 2002. The causal relation between human papillomavirus and cervical cancer. Journal of Clinical Pathology 55(4):244–265.
Bouchardy, C., G. Fioretta, M. De Perrot, M. Obradovic, and A. Spiliopoulos. 1999. Determinants of long term survival after surgery for cancer of the lung: A population-based study. Cancer 86(11):2229–2237.
Bower, J. E. 2008. Behavioral symptoms in patients with breast cancer and survivors. Journal Clinical Oncology 26(5):768–777.
Brady, K. T., D. E. Grice, L. Dustan, and C. Randall. 1993. Gender differences in substance use disorders. American Journal of Psychiatry 150(11):1707–1711.
Brady, K. T., S. E. Back, and S. F. Greenfield. 2009. Women and Addiction: A Comprehensive Handbook. New York: The Guilford Press.
Braithwaite, R. S., N. F. Col, and J. B. Wong. 2003. Estimating hip fracture morbidity, mortality and costs. Journal of the American Geriatrics Society 51(3):364–370.
Brauer, C. A., M. Coca-Perraillon, D. M. Cutler, and A. B. Rosen. 2009. Incidence and mortality of hip fractures in the United States. Journal of the American Medical Association 302(14): 1573–1579.
Braunwald, E., E. M. Antman, J. W. Beasley, R. M. Califf, M. D. Cheitlin, J. S. Hochman, R. H. Jones, D. Kereiakes, J. Kupersmith, T. N. Levin, C. J. Pepine, J. W. Schaeffer, E. E. Smith, III, D. E. Steward, P. Theroux, R. J. Gibbons, J. S. Alpert, D. P. Faxon, V. Fuster, G. Gregoratos, L. F. Hiratzka, A. K. Jacobs, and S. C. Smith, Jr. 2002. ACC/AHA 2002 guideline update for the management of patients with unstable angina and non-ST-segment elevation myocardial infarction—summary article: A report of the American College of Cardiology/American Heart Association task force on practice guidelines (Committee on the Management of Patients With Unstable Angina). Journal of the American College of Cardiology 40(7):1366–1374.
Breitburd, F., R. Kirnbauer, N. Hubbert, B. Nonnenmacher, C. Trin-Dinh-Desmarquet, G. Orth, J. Schiller, and D. Lowy. 1995. Immunization with viruslike particles from cottontail rabbit papillomavirus (CRPV) can protect against experimental CRPV infection. Journal of Virology 69(6):3959–3963.
Brieger, D., K. A. Eagle, S. G. Goodman, P. G. Steg, A. Budaj, K. White, and G. Montalescot. 2004. Acute coronary syndromes without chest pain, an underdiagnosed and undertreated high-risk group. Chest 126(2):461–469.
Briel, M., G. G. Schwartz, P. L. Thompson, J. A. de Lemos, M. A. Blazing, G. A. van Es, M. Kayikcioglu, H. R. Arntz, F. R. den Hartog, N. J. Veeger, F. Colivicchi, J. Dupuis, S. Okazaki, R. S. Wright, H. C. Bucher, and A. J. Nordmann. 2006. Effects of early treatment with statins on short-term clinical outcomes in acute coronary syndromes: A meta-analysis of randomized controlled trials. Journal of the American Medical Association 295(17):2046–2056.
Britt, K., A. Ashworth, and M. Smalley. 2007. Pregnancy and the risk of breast cancer. Endocrine-Related Cancer 14(4):907–933.
Brown, K. A., and E. R. Simpson. 2010. Obesity and breast cancer: Progress to understanding the relationship. Cancer Research 70(1):4–7.
Brownson, R. C., M. C. Alavanja, E. T. Hock, and T. S. Loy. 1992. Passive smoking and lung cancer in nonsmoking women. American Journal of Public Health 82(11):1525–1530.
Brugts, J. J., T. Yetgin, S. E. Hoeks, A. M. Gotto, J. Shepherd, R. G. Westendorp, A. J. de Craen, R. H. Knopp, H. Nakamura, P. Ridker, R. van Domburg, and J. W. Deckers. 2009. The benefits of statins in people without established cardiovascular disease but with cardiovascular risk factors: Meta-analysis of randomised controlled trials. British Medical Journal 338:b2376.
Buchberger, W., A. Niehoff, P. Obrist, P. DeKoekkoek-Doll, and M. Dunser. 2000. Clinically and mammographically occult breast lesions: Detection and classification with high-resolution sonography. Seminars in Ultrasound, CT and MRI 21(4):325–336.
Buchbinder, S. 2009. The epidemiology of new HIV infections and interventions to limit HIV transmission. Topics in HIV Medicine 17(2):37–43.
Buckley, D. I., R. Fu, M. Freeman, K. Rogers, and M. Helfand. 2009. C-reactive protein as a risk factor for coronary heart disease: A systematic review and meta-analyses for the US Preventive Services Task Force. Annals of Internal Medicine 151(7):483–495.
Buencamino, M. C., A. L. Sikon, A. Jain, and H. L. Thacker. 2009. An observational study on the adherence to treatment guidelines of osteopenia. Journal of Women’s Health 18(6):873–881.
Bukstein, O. G., L. J. Glancy, and Y. Kaminer. 1992. Patterns of affective comorbidity in a clinical population of dually diagnosed adolescent substance abusers. Journal of the American Academy of Child and Adolescent Psychiatry 31(6):1041–1045.
Buzdar, A. U., G. N. Hortobagyi, T. L. Smith, S. Kau, C. Marcus, F. A. Holmes, V. Hug, G. Fraschini, F. C. Ames, and R. G. Martin. 1988. Adjuvant therapy of breast cancer with or without additional treatment with alternate drugs. Cancer 62(10):2098–2104.
Cal EPA (California Environmental Protection Agency). 2005. Proposed Identification of Environmental Tobacco Smoke as a Toxic Air Contaminant. Part B: Health Effects. Sacramento: California Environmental Protection Agency.
Campo, M. S. 2002. Animal models of papillomavirus pathogenesis. Virus Research 89(2): 249–261.
Campos-Outcalt, D. 2009. The case for HPV immunization. Journal of Family Practice 58(12): 660–664.
Canadian Study of Health and Aging. 1994. Canadian Study of Health and Aging: Study methods and prevalence of dementia. Canadian Medical Association Journal 150(6):899–913.
Canto, J. G., R. J. Goldberg, M. M. Hand, R. O. Bonow, G. Sopko, C. J. Pepine, and T. Long. 2007. Symptom presentation of women with acute coronary syndromes: Myth vs reality. Archives of Internal Medicine 167(22):2405–2413.
Carey, L. A., C. M. Perou, C. A. Livasy, L. G. Dressler, D. Cowan, K. Conway, G. Karaca, M. A. Troester, C. K. Tse, S. Edmiston, S. L. Deming, J. Geradts, M. C. U. Cheang, T. O. Nielsen, P. G. Moorman, H. S. Earp, and R. C. Millikan. 2006. Race, breast cancer subtypes, and survival in the Carolina Breast Cancer Study. Journal of the American Medical Association 295(21):2492–2502.
Carlton, J. M., R. P. Hirt, J. C. Silva, A. L. Delcher, M. Schatz, Q. Zhao, J. R. Wortman, S. L. Bidwell, U. C. M. Alsmark, S. Besteiro, T. Sicheritz-Ponten, C. J. Noel, J. B. Dacks, P. G. Foster, C. Simillion, Y. Van de Peer, D. Miranda-Saavedra, G. J. Barton, G. D. Westrop, S. Muller, D. Dessi, P. L. Fiori, Q. Ren, I. Paulsen, H. Zhang, F. D. Bastida-Corcuera, A. Simoes-Barbosa, M. T. Brown, R. D. Hayes, M. Mukherjee, C. Y. Okumura, R. Schneider, A. J. Smith, S. Vanacova, M. Villalvazo, B. J. Haas, M. Pertea, T. V. Feldblyum, T. R. Utterback, C-L. Shu, K. Osoegawa, P. J. de Jong, I. Hrdy, L. Horvathova, Z. Zubacova, P. Dolezal, S-B. Malik, J. M. Logsdon, Jr., K. Henze, A. Gupta, C. C. Wang, R. L. Dunne, J. A. Upcroft, P. Upcroft, O. White, S. L. Salzberg, P. Tang, C-H. Chiu, Y-S. Lee, T. M. Embley, G. H. Coombs, J. C. Mottram, J. Tachezy, C. M. Fraser-Liggett, and P. J. Johnson. 2007. Draft genome sequence of the sexually transmitted pathogen trichomonas vaginalis. Science 315(5809):207–212.
Carter, J. E. 1994. Combined hysteroscopic and laparoscopic findings in patients with chronic pelvic pain. Journal of the American Association of Gynecologic Laparoscopists 2(1):43–47.
Carter, J. J., L. A. Koutsky, J. P. Hughes, S. K. Lee, J. Kuypers, N. Kiviat, and D. A. Galloway. 2000. Comparison of human papillomavirus types 16, 18, and 6 capsid antibody responses following incident infection. Journal of Infectious Diseases 181(6):1911–1919.
Cauley, J. A., D. E. Thompson, K. C. Ensrud, J. C. Scott, and D. Black. 2000. Risk of mortality following clinical fractures. Osteoporosis International 11(7):556–561.
CDC (Centers for Disease Control and Prevention). 1999. Ten great public health achievements—United States, 1900–1999. Morbidity and Mortality Weekly Report 48(12):241–243.
———. 2001. Mortality from coronary heart disease and acute myocardial infarction—United States, 1998. Morbidity and Mortality Weekly Report 50(06):90–93.
———. 2002a. Women and Smoking: A report of the Surgeon General. http://www.cdc.gov/mmwr/preview/mmwrhtml/rr5112a4.htm (accessed August 17, 2009).
———. 2002b. Drug-Associated HIV Transmission Continues in the United States. CDC. http://www.cdc.gov/hiv/resources/factsheets/idu.htm (accessed May 14, 2010).
———. 2002c. US Public Health Service Task Force recommendations for use of antiretroviral drugs in pregnant HIV-1–infected women for maternal health and interventions to reduce perinatal HIV-1 transmission in the United States. Morbidity and Mortality Weekly Report, http://www.cdc.gov/mmwr/preview/mmwrhtml/rr5118a1.htm (accessed May 17, 2010).
———. 2003. Late versus early testing of HIV—16 sites, United States, 2000–2003. Morbidity and Mortality Weekly Report 52(26):581–586.
———. 2004. Hysterectomy in the United States, 2000–2004. In Women’s Reproductive Health: Hysterectomy Fact Sheet. Atlanta, GA: CDC.
———. 2006. Revised recommendations for HIV testing of adults, adolescents, and pregnant women in health-care settings. Morbidity and Mortality Weekly Report 55(RR14), http://www.cdc.gov/mmwr/preview/mmwrhtml/rr5514a1.htm (accessed May 14, 2010).
———. 2008a. HIV/AIDS in the United States: An Update. Atlanta, GA:CDC.
———. 2008b. Subpopulation estimates from the HIV incidence surveillance system—United States, 2006. Morbidity and Mortality Weekly Report 57(36):985–989.
———. 2008c. CDC HIV/AIDS Fact Sheet: HIV/AIDS Among Women. http://www.cdc.gov/hiv/topics/women/resources/factsheets/pdf/women.pdf (accessed June 29, 2009).
———. 2008d. HIV prevalence estimates—United States, 2006. Morbidity and Mortality Weekly Report 57(39):1073–1076.
———. 2008e. Safe Motherhood Promoting Health for Women Before, During, and After Pregnancy. http://www.cdc.gov/nccdphp/publications/aag/pdf/drh.pdf (accessed May 3, 2010)
———. 2008f. Sexually Transmitted Disease Surveillance 2007. http://www.cdc.gov/std/stats07/Surv2007FINAL.pdf (accessed May 4, 2009).
———. 2008g. Current Smoking. http://www.cdc.gov/nchs/data/nhis/earlyrelease/200812_08.pdf (accessed August 17, 2009).
———. 2010. Breast Cancer Statistics. Division of Cancer Prevention and Control, National Center for Chronic Disease Prevention and Health Promotion, http://www.cdc.gov/cancer/breast/statistics/ (accessed May 3, 2010).
Chan, K. K., and R. Naik. 2008. Advances in surgical treatment of cervical cancer. Women’s Health 4(3):245–256.
Chasan-Taber, L., and M. J. Stampfer. 1998. Epidemiology of oral contraceptives and cardiovascular disease. Annals of Internal Medicine 128(6):467–477.
Chatham, L. R., M. L. Hiller, G. A. Rowan-Szal, G. W. Joe, and D. D. Simpson. 1999. Gender differences at admission and follow-up in a sample of methadone maintenance clients. Substance Use and Misuse 34(8):1137–1165.
Chaudhry, P., and E. Asselin. 2009. Resistance to chemotherapy and hormone therapy in endometrial cancer. Endocrine-Related Cancer 16(2):363–380.
Chauhan, M. S., K. K. Ho, D. S. Baim, R. E. Kuntz, and D. E. Cutlip. 2005. Effect of gender on inhospital and one-year outcomes after contemporary coronary artery stenting. American Journal of Cardiology 95(1):101–104.
Chen, C-R., G. M. Buck, N. G. Courey, K. M. Perez, and J. Wactawski-Wende.2001. Risk factors for uterine fibroids among women undergoing tubal sterilization. American Journal of Epidemiology 153(1):20–26.
Chesnut, C. H., 3rd, S. Silverman, K. Andriano, H. Genant, A. Gimona, S. Harris, D. Kiel, M. LeBoff, M. Maricic, P. Miller, C. Moniz, M. Peacock, P. Richardson, N. Watts, and D. Baylink. 2000. A randomized trial of nasal spray salmon calcitonin in postmenopausal women with established osteoporosis: The prevent recurrence of osteoporotic fractures study. PROOF study group. American Journal of Medicine 109(4):267–276.
Chia, S. K., C. H. Speers, Y. D’yachkova, A. Kang, S. Malfair-Taylor, J. Barnett, A. Coldman, K. A. Gelmon, S. E. O’Reilly, and I. A. Olivotto. 2007. The impact of new chemotherapeutic and hormone agents on survival in a population-based cohort of women with metastatic breast cancer. Cancer 110(5):973–979.
Chlebowski, R. T., S. L. Hendrix, R. D. Langer, M. L. Stefanick, M. Gass, D. Lane, R. J. Rodabough, M. A. Gilligan, M. G. Cyr, C. A. Thomson, J. Khandekar, H. Petrovitch, and A. McTiernan. 2003. Influence of estrogen plus progestin on breast cancer and mammography in healthy post-menopausal women: The Women’s Health Initiative Randomized Trial. Journal of the American Medical Association 289(24):3243–3253.
Chlebowski, R. T., L. H. Kuller, R. L. Prentice, M. L. Stefanick, J. E. Manson, M. Gass, A. K. Aragaki, J. K. Ockene, D. S. Lane, G. E. Sarto, A. Rajkovic, R. Schenken, S. L. Hendrix, P. M. Ravdin, T. E. Rohan, S. Yasmeen, G. Anderson, and the WHI Investigators. 2009. Breast cancer after use of estrogen plus progestin in postmenopausal women. New England Journal of Medicine 360(6):573–587.
Cho, K. R., and M. Shih Ie. 2009. Ovarian cancer. Annual Review of Pathology 4:287–313.
Chou, A. F., S. H. Scholle, C. S. Weisman, A. S. Bierman, R. Correa-de-Araujo, and L. Mosca. 2007a. Gender disparities in the quality of cardiovascular disease care in private managed care plans. Women’s Health Issues 17(3):120–130.
Chou, A. F., L. Wong, C. S. Weisman, S. Chan, A. S. Bierman, R. Correa-de-Araujo, and S. H. Scholle. 2007b. Gender disparities in cardiovascular disease care among commercial and medicare managed care plans. Women’s Health Issues 17(3):139–149.
Christian, A. H., W. Rosamond, A. R. White, and L. Mosca. 2007. Nine-year trends and racial and ethnic disparities in women’s awareness of heart disease and stroke: An American Heart Association national study. Journal of Women’s Health 16(1):68–81.
Chwalisz, K., L. Larsen, K. McCrary, and A. Edmonds. 2004. Effects of the novel selective progesterone receptor modulator (SPRM) asoprisnil on selected hormonal parameters in subjects with leiomyomata. Fertility and Sterility 82(Suppl. 2):S306.
Clark, D. A., A. T. Beck, and G. K. Brown. 1992. Sociotropy, autonomy, and life event perceptions in dysphoric and nondysphoric individuals. Cognitive Therapy and Research 16(6):635–652.
Clarke-Pearson, D. L. 2009. Clinical practice. Screening for ovarian cancer. New England Journal of Medicine 361(2):170–177.
Claus, E. B., J. Schildkraut, E. S. Iversen, Jr., D. Berry, and G. Parmigiani. 1998. Effect of BRCA1 and BRCA2 on the association between breast cancer risk and family history. Journal of the National Cancer Institute 90(23):1824–1829.
Cleary, M. P., M. E. Grossmann, and A. Ray. 2010. Effect of obesity on breast cancer development. Veterinary Pathology 47(2):202–213.
Clegg, L. X., M. E. Reichman, B. A. Miller, B. F. Hankey, G. K. Singh, Y. D. Lin, M. T. Goodman, C. F. Lynch, S. M. Schwartz, V. W. Chen, L. Bernstein, S. L. Gomez, J. J. Graff, C. C. Lin, N. J. Johnson, and B. K. Edwards. 2009. Impact of socioeconomic status on cancer incidence and stage at diagnosis: Selected findings from the Surveillance, Epidemiology, and End Results: National Longitudinal Mortality Study. Cancer Causes and Control 20(4):417–435.
Clemons, M., and P. Goss. 2001. Estrogen and the risk of breast cancer. New England Journal of Medicine 344(4):276–285.
Close, P., A. Neuprez, and J. Y. Reginster. 2006. Developments in the pharmacotherapeutic management of osteoporosis. Expert Opinion on Pharmacotherapy 7(12):1603–1615.
Cohen, S., D. Janicki-Deverts, and G. E. Miller. 2007. Psychological stress and disease. Journal of the American Medical Association 298(14):1685–1687.
Cohn, M. 2008. What roles do regulatory T cells play in the control of the adaptive immune response? International Immunology 20(9):11.
Cohn, S. E., J. G. Park, D. H. Watts, A. Stek, J. Hitti, P. A. Clax, S. Yu, J. J. Lertora, and ACTG A5093 Protocol Team. 2007. Depo-medroxyprogesterone in women on antiretroviral therapy: Effective contraception and lack of clinically significant interactions. Clinical Pharmacology and Therapeutics 81(2):222–227.
Collins, N., R. McManus, R. Wooster, J. Mangion, S. Seal, S. R. Lakhani, W. Ormiston, P. A. Daly, D. Ford, D. F. Easton, et al. 1995. Consistent loss of the wild type allele in breast cancers from a family linked to the BRCA2 gene on chromosome 13q12–13. Oncogene 10(8):1673–1675.
Compton, S. N., J. S. March, D. Brent, A. M. Albano, R. Weersing, and J. Curry. 2004. Cognitive-behavioral psychotherapy for anxiety and depressive disorders in children and adolescents: An evidence-based medicine review. Journal of the American Academy of Child and Adolescent Psychiatry 43(8):930–959.
Cook, N. R., J. E. Buring, and P. M. Ridker. 2006. The effect of including C-reactive protein in cardiovascular risk prediction models for women. Annals of Internal Medicine 145(1):21–29.
Cooke, T., J. Reeves, A. Lannigan, and P. Stanton. 2001. The value of the human epidermal growth factor receptor-2 (HER2) as a prognostic marker. European Journal of Cancer 37(Suppl. 1):3–10.
Cooper, C., and L. J. Melton, 3rd. 1992. Epidemiology of osteoporosis. Trends in Endocrinology and Metabolism 3(6):224–229.
Cooper, C., S. Westlake, N. Harvey, K. Javaid, E. Dennison, and M. Hanson. 2006. Review: Developmental origins of osteoporotic fracture. Osteoporosis International 17(3):337–347.
Cooper, E. R., M. Charurat, L. Mofenson, I. C. Hanson, J. Pitt, C. Diaz, K. Hayani, E. Handelsman, V. Smeriglio, R. Hoff, and W. Blattner. 2002. Combination antiretroviral strategies for the treatment of pregnant HIV-1-infected women and prevention of perinatal HIV-1 transmission. Journal of Acquired Immune Deficiency Syndromes 29(5):484–494.
Cooper, G. S., F. W. Miller, and J. P. Pandey. 1999. The role of genetic factors in autoimmune disease: Implications for environmental research. Environmental Health Perspectives 107 (Suppl. 5): 693–700.
Correa-de-Araujo, R. 2006. Serious gaps: How the lack of sex/gender-based research impairs health. Journal of Women’s Health 15(10):1116–1122.
Correa-de-Araujo, R., and C. M. Clancy. 2006. Catalyzing quality of care improvements for women. Women’s Health Issues 16(2):41–43.
Corsetti, V., N. Houssami, A. Ferrari, M. Ghirardi, S. Bellarosa, O. Angelini, C. Bani, P. Sardo, G. Remida, E. Galligioni, and S. Ciatto. 2008. Breast screening with ultrasound in women with mammography-negative dense breasts: Evidence on incremental cancer detection and false positives, and associated cost. European Journal of Cancer 44(4):539–544.
Couse, J. F., J. Lindzey, K. Grandien, J. A. Gustafsson, and K. S. Korach. 1997. Tissue distribution and quantitative analysis of estrogen receptor-alpha (ERALPHA) and estrogen receptor-beta (ERBETA) messenger ribonucleic acid in the wild-type and eralpha-knockout mouse. Endocrinology 138(11):4613–4621.
Coyle, Y. M. 2009. Lifestyle, genes, and cancer. Methods in Molecular Biology 472:25–56.
Cramer, J. A., and S. Silverman. 2006. Persistence with bisphosphonate treatment for osteoporosis: Finding the root of the problem. American Journal of Medicine 119(4 Suppl. 1):S12–S17.
Cramer, S. F., and A. Patel. 1990. The frequency of uterine leiomyomas. American Journal of Clinical Pathology 94(4):435–438.
Cranganu, A., and J. Camporeale. 2009. Nutrition aspects of lung cancer. Nutrition in Clinical Practice 24(6):688–700.
Cranney, A., T. Horsley, S. O’Donnell, H. A. Weiler, L. Puil, D. S. Ooi, S.A. Atkinson, L.M. Ward, D. Moher, D. A. Hanley, M. Fang, F. Yazd, C. Garritty, M. Sampson, N. Barrowman, A. Tsertsvadze, and V. Mamaladze. 2007. Effectiveness and Safety of Vitamin D in Relation to Bone Health. Evidence Report/Technology Assessment No. 158. Prepared by the University of Ottawa Evidence-Based Practice Center (UO-EPC) Under Contract No. 290-02-0021. AHRQ Publication No. 07-E013. Rockville, MD: Agency for Healthcare Research and Quality.
Creinin, M. D., E. Vittinghoff, E. Schaff, C. Klaisle, P. D. Darney, and C. Dean. 1997. Medical abortion with oral methotrexate and vaginal misoprostol. Obstetrics and Gynecology 90(4):611–616.
Crystal, P., S. D. Strano, S. Shcharynski, and M. J. Koretz. 2003. Using sonography to screen women with mammographically dense breasts. American Journal of Roentgeneology 181(1):177–182.
Cuijpers, P., A. van Straten, F. Smit, C. Mihalopoulos, and A. Beekman. 2008. Preventing the onset of depressive disorders: A meta-analytic review of psychological interventions. American Journal of Psychiatry 165(10):1272–1280.
Cummings, S. M., B. Bride, K. M. Cassie, and A. Rawlins-Shaw. 2008. Substance abuse. Journal of Gerontological Social Work 50(Suppl. 1):215–241.
Cummings, S. R., and D. Black. 1995. Bone mass measurements and risk of fracture in Caucasian women: A review of findings from prospective studies. American Journal of Medicine 98(2A):24S–28S.
Cunningham, M., and L. H. Zayas. 2002. Reducing depression in pregnancy: Designing multimodal interventions. Social Work 47:114–123.
Cuschieri, K. 2009. Should boys receive the human papillomavirus vaccine? No. British Medical Journal 339:b4921.
Cuzick, J., T. Powles, U. Veronesi, J. Forbes, R. Edwards, S. Ashley, and P. Boyle. 2003. Overview of the main outcomes in breast-cancer prevention trials. Lancet 361(9354):296–300.
D’Amico, T. A., T. A. Aloia, M-B. H. Moore, J. E. Herndon, II, K. R. Brooks, C. L. Lau, and D. H. Harpole, Jr. 2000. Molecular biologic substaging of stage I lung cancer according to gender and histology. Annals of Thoracic Surgery 69(3):882–886.
Dannenberg, H., P. Komminoth, W. N. M. Dinjens, E. J. M. Speel, and R. R. de Krijger. 2003. Molecular genetic alterations in adrenal and extra-adrenal pheochromocytomas and paragangliomas. Endocrine Pathology 14(4):329–350.
Davey, E., A. Barratt, L. Irwig, S. F. Chan, P. Macaskill, P. Mannes, and A. M. Saville. 2006. Effect of study design and quality on unsatisfactory rates, cytology classifications, and accuracy in liquid-based versus conventional cervical cytology: A systematic review. Lancet 367(9505):122–132.
David, M., and A. D. Ebert. 2005. Treatment of uterine fibroids by embolization: Advantages, disadvantages, and pitfalls. European Journal of Obstetrics, Gynecology, and Reproductive Biology 123(2):131–138.
De Clercq, E. 2007. The design of drugs for HIV and HCV. Nature Reviews Drug Discovery 6(12):1001–1018.
De Groot, A. S. 2000. HIV infection among incarcerated women: Epidemic behind bars. AIDS Reader 10(5):287–295.
De Laet, C., J. A. Kanis, A. Oden, H. Johanson, O. Johnell, P. Delmas, J. A. Eisman, H. Kroger, S. Fujiwara, P. Garnero, E. V. McCloskey, D. Mellstrom, L. J. Melton, 3rd, P. J. Meunier, H. A. Pols, J. Reeve, A. Silman, and A. Tenenhouse. 2005. Body mass index as a predictor of fracture risk: A meta-analysis. Osteoporosis International 16(11):1330–1338.
Dean-Gaitor, H. D., and P. L. Fleming. 1999. Epidemiology of AIDS in incarcerated persons in the United States, 1994–1996. AIDS 13(17):2429–2435.
DeCara, J. M. 2003. Noninvasive cardiac testing in women. Journal of the American Medical Women’s Association 58(4):254–263.
Deeks, E. 2007. Local therapy in endometrial cancer: Evidence based review. Current Opinion in Oncology 19(5):512–515.
Deligeoroglou, E., E. Michailidis, and G. Creatsas. 2003. Oral contraceptives and reproductive system cancer. In Women’s Health and Disease: Gynecologic and Reproductive Issues. Vol. 997, Annals of the New York Academy of Sciences, edited by G. Creatsas, G. Mastorakos and G. P. Chrousos. New York: New York Academy of Sciences. Pp. 199–208.
DeMeo, M. T., E. A. Mutlu, A. Keshavarzian, and M. C. Tobin. 2002. Intestinal permeation and gastrointestinal disease. Journal of Clinical Gastroenterology 34(4):385–396.
Dennison, E., Z. Cole, and C. Cooper. 2005. Diagnosis and epidemiology of osteoporosis. Current Opinion in Rheumatology 17(4):456–461.
DeRubeis, R. J., L. A. Gelfand, T. Z. Tang, and A. D. Simons. 1999. Medications versus cognitive behavior therapy for severely depressed outpatients: Mega-analysis of four randomized comparisons. American Journal of Psychiatry 156(7):1007–1013.
DeSantis, C., A. Jemal, E. Ward, and M. J. Thun. 2008. Temporal trends in breast cancer mortality by state and race. Cancer Causes and Control 19(5):537–545.
Desta, Z., B. A. Ward, N. V. Soukhova, and D. A. Flockhart. 2004. Comprehensive evaluation of tamoxifen sequential biotransformation by the human cytochrome P450 system in vitro: Prominent roles for CYP3A and CYP2D6. Journal of Pharmacology and Experimental Therapeutics 310(3):1062–1075.
Devesa, S. S., E. S. Pollack, and J. L. Young, Jr. 1984. Assessing the validity of observed cancer incidence trends. American Journal of Epidemiology 119(2):274–291.
Di Leo, A., H. L. Gomez, Z. Aziz, Z. Zvirbule, J. Bines, M. C. Arbushites, S. F. Guerrera, M. Koehler, C. Oliva, S. H. Stein, L. S. Williams, J. Dering, R. S. Finn, and M. F. Press. 2008. Phase III, double-blind, randomized study comparing lapatinib plus paclitaxel with placebo plus paclitaxel as first-line treatment for metastatic breast cancer. Journal Clinical Oncology 26(34):5544–5552.
Diehl, A., B. Croissant, A. Batra, G. Mundle, H. Nakovics, and K. Mann. 2007. Alcoholism in women: Is it different in onset and outcome compared to men? European Archives of Psychiatry and Clinical Neuroscience 257(6):344–351.
Diekmann, F., S. Diekmann, F. Jeunehomme, S. Muller, B. Hamm, and U. Bick. 2005. Digital mammography using iodine-based contrast media: Initial clinical experience with dynamic contrast medium enhancement. Investigative Radiology 40(7):397–404.
Dolan, S. E., J. S. Huang, K. M. Killilea, M. P. Sullivan, N. Aliabadi, and S. Grinspoon. 2004. Reduced bone density in HIV-infected women. AIDS 18(3):475–483.
Dracup, K. 2007. The challenge of women and heart disease. Archives of Internal Medicine 167(22): 2396.
Dresler, C. M., C. Fratelli, J. Babb, L. Everley, A. A. Evans, and M. L. Clapper. 2000. Gender differences in genetic susceptibility for lung cancer. Lung Cancer 30(3):153–160.
Dromain, C., C. Balleyguier, S. Muller, M-C. Mathieu, F. Rochard, P. Opolon, and R. Sigal. 2006. Evaluation of tumor angiogenesis of breast carcinoma using contrast-enhanced digital mammography. American Journal of Roentgeneology 187(5):W528–W537.
Dube, M. P., and F. R. Sattler. 2010. Inflammation and complications of HIV disease. Journal of Infectious Diseases 201(12):1783–1785.
Dürst, M., L. Gissmann, H. Ikenberg, and H. zur Hausen. 1983. A papillomavirus DNA from a cervical carcinoma and its prevalence in cancer biopsy samples from different geographic regions. Proceedings of the National Academy of Sciences of the United States of America 80(12):3812–3815.
Dürst, M., R. T. Dzarlieva-Petrusevska, P. Boukamp, N. E. Fusenig, and L. Gissmann. 1987. Molecular and cytogenetic analysis of immortalized human primary keratinocytes obtained after transfection with human papillomavirus type 16 DNA. Oncogene 1(3):251–256.
Dwight-Johnson, M., C. D. Sherbourne, D. Liao, and K. B. Wells. 2000. Treatment preferences among depressed primary care patients. Journal of General Internal Medicine 15(8):527–534.
Early Breast Cancer Trialists’ Collaborative Group. 2005. Effects of chemotherapy and hormonal therapy for early breast cancer on recurrence and 15-year survival: An overview of the randomised trials. Lancet 365(9472):1687–1717.
Easton, D. F., D. T. Bishop, D. Ford, and G. P. Crockford. 1993. Genetic linkage analysis in familial breast and ovarian cancer: Results from 214 families. The breast cancer linkage consortium. American Journal of Human Genetics 52(4):678–701.
Ebers, G. C. 2009. Editorial regarding “Explaining Multiple Sclerosis Prevalence by Ultraviolet Exposure: A Geospatial Analysis,” by Beretich and Beretich. Multiple Sclerosis 15(8):889–890.
Eifel, P. J. 2006. Concurrent chemotherapy and radiation therapy as the standard of care for cervical cancer. Nature Clinical Practice. Oncology 3(5):248–255.
Elhwuegi, A. S. 2004. Central monoamines and their role in major depression. Progress in Neuropsychopharmacology and Biolical Psychiatry 28(3):435–451.
Elsasser, A., K. Suzuki, and J. Schaper. 2000. Unresolved issues regarding the role of apoptosis in the pathogenesis of ischemic injury and heart failure. Journal of Molecular and Cellular Cardiology 32(5):711–724.
Emkey, R. D., and M. Ettinger. 2006. Improving compliance and persistence with bisphosphonate therapy for osteoporosis. American Journal of Medicine 119(4 Suppl. 1):S18–S24.
Ensrud, K. E., D. E. Thompson, J. A. Cauley, M. C. Nevitt, D. M. Kado, M. C. Hochberg, A. C. Santora, 2nd, and D. M. Black. 2000. Prevalent vertebral deformities predict mortality and hospitalization in older women with low bone mass. Fracture Intervention Trial Research Group. Journal of the American Geriatrics Society 48(3):241–249.
Erblich, J., D. H. Bovbjerg, C. Norman, H. B. Valdimarsdottir, and G. H. Montgomery. 2000. It won’t happen to me: Lower perception of heart disease risk among women with family histories of breast cancer. Preventive Medicine 31(6):714–721.
Esserman, L., Y. Shieh, and I. Thompson. 2009. Rethinking screening for breast cancer and prostate cancer. Journal of the American Medical Association 302(15):1685–1692.
Essex, M., M. F. McLane, T. H. Lee, N. Tachibana, J. I. Mullins, J. Kreiss, C. K. Kasper, M. C. Poon, A. Landay, S. F. Stein, D. P. Francis, C. Cabradilla, D. N. Lawrence, and B. L. Evatt. 1983. Antibodies to human T-cell leukemia virus membrane antigens (HTLV-MA) in hemophiliacs. Science 221(4615):1061–1064.
Ettinger, B., S. T. Harris, D. Kendler, B. Kessel, M. R. McClung, G. I. Gorodeski, M. L. Rothert, V. W. Henderson, M. K. Richardson, R. R. Freedman, J. C. Gallagher, S. R. Goldstein, J. V. Pinkerton, N. K. Reame, L. Speroff, C. A. Stuenkel, I. Schiff, W. H. Utian, I. D. Graham, P. K. Lammers, P. P. Boggs, and The North American Menopause Society. 2006. Management of osteoporosis in postmenopausal women: 2006 position statement of the North American Menopause Society. Menopause 13(3):340–367.
European Study Group on Heterosexual Transmission of HIV. 1992. Comparison of female to male and male to female transmission of HIV in 563 stable couples. European Study Group on Heterosexual Transmission of HIV. British Medical Journal 304(6830):809–813.
Evangelista, O., and M. A. McLaughlin. 2009. Review of cardiovascular risk factors in women. Gender Medicine 6(Pt. 1):17–36.
Evans, P., and S. Brunsell. 2007. Uterine fibroid tumors: Diagnosis and treatment. American Family Physician 75(10):1503–1508.
Faca, V., H. Wang, and S. Hanash. 2009. Proteomic global profiling for cancer biomarker discovery. Methods in Molecular Biology 492:309–320.
Fader, A. N., L. N. Arriba, H. E. Frasure, and V. E. von Gruenigen. 2009. Endometrial cancer and obesity: Epidemiology, biomarkers, prevention and survivorship. Gynecologic Oncology 114(1): 121–127.
Fairweather, D., S. Frisancho-Kiss, and N. R. Rose. 2008. Sex differences in autoimmune disease from a pathological perspective. American Journal of Pathology 173(3):600–609.
Fallowfield, L. 2005. Acceptance of adjuvant therapy and quality of life issues. Breast 14(6): 612–616.
Farjah, F., D. E. Wood, N. D. Yanez, 3rd, T. L. Vaughan, R. G. Symons, B. Krishnadasan, and D. R. Flum. 2009. Racial disparities among patients with lung cancer who were recommended operative therapy. Archives of Surgery 144(1):14–18.
Farquhar, C., R. Basser, S. Hetrick, A. Lethaby, and J. Marjoribanks. 2003. High dose chemotherapy and autologous bone marrow or stem cell transplantation versus conventional chemotherapy for women with metastatic breast cancer. Cochrane Database of Systematic Reviews 1: CD003142.
Farzadegan, H., D. R. Hoover, J. Astemborski, C. M. Lyles, J. B. Margolick, R. B. Markham, T. C. Quinn, and D. Vlahov. 1998. Sex differences in HIV-1 viral load and progression to AIDS. Lancet 352(9139):1510–1514.
Faustman, D., X. P. Li, H. Y. Lin, Y. E. Fu, G. Eisenbarth, J. Avruch, and J. Guo. 1991. Linkage of faulty major histocompatibility complex class I to autoimmune diabetes. Science 254(5039): 1756–1761.
Fawzi, W. W., G. I. Msamanga, D. Spiegelman, E. J. Urassa, N. McGrath, D. Mwakagile, G. Antelman, R. Mbise, G. Herrera, S. Kapiga, W. Willett, and D. J. Hunter. 1998. Randomised trial of effects of vitamin supplements on pregnancy outcomes and T cell counts in HIV-1-infected women in Tanzania. Lancet 351(9114):1477–1482.
FDA (US Food and Drug Administration). 2008. FDA Licenses New Vaccine for Prevention of Cervical Cancer and Other Diseases in Females Caused by Human Papillomavirus. http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/2006/ucm108666.htm (accessed November 16, 2009).
———. 2009a. FDA Approves New Vaccine for Prevention of Cervical Cancer. US Department of Health and Human Services. http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm187048.htm (accessed April 14, 2010).
———. 2009b. HIV/AIDS Historical Time Line 1981–1990. http://www.fda.gov/ForConsumers/ByAudience/ForPatientAdvocates/HIVandAIDSActivities/ucm151074.htm (accessed May 14, 2010).
———. 2010. MQSA National Statistics. http://www.fda.gov/Radiation-EmittingProducts/MammographyQualityStandardsActandProgram/FacilityScorecard/ucm113858.htm (accessed August 7, 2010).
Ferenczy, A., and E. Franco. 2002. Persistent human papillomavirus infection and cervical neoplasia. Lancet Oncology 3(1):11–16.
Ferguson, M. K., J. Wang, P. C. Hoffman, D. J. Haraf, J. Olak, G. A. Masters, and E. E. Vokes. 2000. Sex-associated differences in survival of patients undergoing resection for lung cancer. Annals of Thoracic Surgery 69(1):245–249; discussion 249–250.
Fernandes, L. S., J. E. Tcheng, J. C. O’Shea, B. Weiner, T. J. Lorenz, C. Pacchiana, L. G. Berdan, K. J. Maresh, D. Joseph, M. Madan, T. Mann, R. Kilaru, J. S. Hochman, N. S. Kleiman, and ESPRIT Investigators. 2002. Is glycoprotein IIB/IIIA antagonism as effective in women as in men following percutaneous coronary intervention?: Lessons from the ESPRIT study. Journal of the American College of Cardiology 40(6):1085–1091.
Ferrero, S., P. L. Venturini, N. Ragni, G. Camerini, and V. Remorgida. 2009. Pharmacological treatment of endometriosis: Experience with aromatase inhibitors. Drugs 69:943–952.
Finer, L. B., and S. K. Henshaw. 2006. Disparities in rates of unintended pregnancy in the United States, 1994 and 2001. Perspectives on Sexual and Reproductive Health 38(2):90–96.
Fiorelli, J. L., T. J. Herzog, and J. D. Wright. 2008. Current treatment strategies for endometrial cancer. Expert Review of Anticancer Therapy 8(7):1149–1157.
Fisher, B., J. P. Costantino, D. L. Wickerham, C. K. Redmond, M. Kavanah, W. M. Cronin, V. Vogel, A. Robidoux, N. Dimitrov, J. Atkins, M. Daly, S. Wieand, E. Tan-Chiu, L. Ford, and N. Wolmark. 1998. Tamoxifen for prevention of breast cancer: Report of the National Surgical Adjuvant Breast and Bowel Project P-1 Study. Journal of the National Cancer Institute 90(18):1371–1388.
Fleck, F. 2004. Microbicides preventing HIV infection could be available by 2010. Bulletin of the World Health Organization 82(5):393–394.
Fletcher, S. W., and J. G. Elmore. 2003. Mammographic screening for breast cancer. New England Journal of Medicine 348(17):1672–1680.
Flexner, C. 2007. HIV drug development: The next 25 years. Nature Reviews Drug Discovery 6(12):959–966.
Fogel, C. I., and M. Belyea. 1999. The lives of incarcerated women: Violence, substance abuse, and at risk for HIV. Journal of the Association of Nurses in AIDS Care 10(6):66–74.
Fong, P. C., D. S. Boss, T. A. Yap, A. Tutt, P. Wu, M. Mergui-Roelvink, P. Mortimer, H. Swaisland, A. Lau, M. J. O’Connor, A. Ashworth, J. Carmichael, S. B. Kaye, J. H. M. Schellens, and J. S. de Bono. 2009. Inhibition of poly(ADP-ribose) polymerase in tumors from BRCA mutation carriers. New England Journal of Medicine.
Ford, E. S., U. A. Ajani, J. B. Croft, J. A. Critchley, D. R. Labarthe, T. E. Kottke, W. H. Giles, and S. Capewell. 2007. Explaining the decrease in U. S. deaths from coronary disease, 1980–2000. New England Journal of Medicine 356(23):2388–2398.
Fournier, J. C., R. J. DeRubeis, S. D. Hollon, S. Dimidjian, J. D. Amsterdam, R. C. Shelton, and J. Fawcett. 2010. Antidepressant drug effects and depression severity: A patient-level meta-analysis. Journal of the American Medical Association 303(1):47–53.
Fox, H. C., M. Talih, R. Malison, G. M. Anderson, M. J. Kreek, and R. Sinha. 2005. Frequency of recent cocaine and alcohol use affects drug craving and associated responses to stress and drug-related cues. Psychoneuroendocrinology 30(9):880–891.
Fox, H. C., K. A. Hong, P. Paliwal, P. T. Morgan, and R. Sinha. 2008. Altered levels of sex and stress steroid hormones assessed daily over a 28-day cycle in early abstinent cocaine-dependent females. Psychopharmacology 195(4):527–536.
Francis, R. M. 2008. Fracture risk assessment. Current Orthopaedics 22(5):322–327.
Franco, E. L., E. Duarte-Franco, and A. Ferenczy. 2001. Cervical cancer: Epidemiology, prevention and the role of human papillomavirus infection. Canadian Medical Association Journal 164(7):1017–1025.
Frank, E., D. J. Kupfer, J. M. Perel, C. Cornes, D. B. Jarrett, A. G. Mallinger, M. E. Thase, A. B. McEachran, and V. J. Grochocinski. 1990. Three-year outcomes for maintenance therapies in recurrent depression. Archives of General Psychiatry 47(12):1093–1099.
Frank, E., D. Novick, and D. J. Kupfer. 2005. Antidepressants and psychotherapy: A clinical research review. Dialogues in Clinical Neuroscience 7(3):263–272.
Frank, T., S. Manley, O. Olopade, S. Cummings, J. Garber, B. Bernhardt, K. Antman, D. Russo, M. Wood, L. Mullineau, C. Isaacs, B. Peshkin, S. Buys, V. Venne, P. Rowley, S. Loader, K. Offit, M. Robson, H. Hampel, D. Brener, E. Winer, S. Clark, B. Weber, L. Strong, and A. Thomas. 1998. Sequence analysis of BRCA1 and BRCA2: Correlation of mutations with family history and ovarian cancer risk. Journal Clinical Oncology 16(7):2417–2425.
Freedman, N. D., M. F. Leitzmann, A. R. Hollenbeck, A. Schatzkin, and C. C. Abnet. 2008. Cigarette smoking and subsequent risk of lung cancer in men and women: Analysis of a prospective cohort study. Lancet Oncology 9(7):649–656.
French, R., and P. Brocklehurst. 1998. The effect of pregnancy on survival in women infected with HIV: A systematic review of the literature and meta-analysis. British Journal of Obstetrics and Gynaecology 105(8):827–835.
Friedenreich, C. M., L. S. Cook, A. M. Magliocco, M. A. Duggan, and K. S. Courneya. 2010. Case– control study of lifetime total physical activity and endometrial cancer risk. Cancer Causes and Control 21(7):1105–1116.
Frisell, J., and E. Lidbrink. 1997. The Stockholm mammographic screening trial: Risks and benefits in age group 40–49 years. Journal of the National Cancer Institute Monographs (22):49–51.
Frost, J. J., J. E. Darroch, and L. Remez. 2008. Improving contraceptive use in the United States. Issues Brief (Alan Guttmacher Institute) 1:1–8.
FUTURE II Study Group. 2007. Quadrivalent vaccine against human papillomavirus to prevent high-grade cervical lesions. New England Journal of Medicine 356(19):1915–1927.
Gadducci, A., and S. Cosio. 2009. Surveillance of patients after initial treatment of ovarian cancer. Critical Reviews in Oncology/Hematology 71(1):43–52.
Gale, E. A. M., and K. M. Gillespie. 2001. Diabetes and gender. Diabetologia 44(1):3–15.
Gallagher, J. C. 2007. Effect of early menopause on bone mineral density and fractures. Menopause—the Journal of the North American Menopause Society 14(3):567–571.
Gallo, J. J., S. D. Ryan, and D. E. Ford. 1999. Attitudes, knowledge, and behavior of family physicians regarding depression in late life. Archives of Family Medicine 8(3):249–256.
Gallo, R. C., P. S. Sarin, E. P. Gelmann, M. Robert-Guroff, E. Richardson, V. S. Kalyanaraman, D. Mann, G. D. Sidhu, R. E. Stahl, S. Zolla-Pazner, J. Leibowitch, and M. Popovic. 1983. Isolation of human t-cell leukemia virus in acquired immune deficiency syndrome (AIDS). Science 220(4599):865–867.
Garcia, P. M., L. A. Kalish, J. Pitt, H. Minkoff, T. C. Quinn, S. K. Burchett, J. Kornegay, B. Jackson, J. Moye, C. Hanson, C. Zorrilla, and J. F. Lew. 1999. Maternal levels of plasma human immunodeficiency virus type 1 RNA and the risk of perinatal transmission. Women and Infants Transmission Study group. New England Journal of Medicine 341(6):394–402.
Garcia-Closas, M., and S. Chanock. 2008. Genetic susceptibility loci for breast cancer by estrogen receptor status. Clinical Cancer Research 14(24):8000–8009.
Garfinkel, L., C. C. Boring, and C. W. Heath, Jr. 1994. Changing trends. An overview of breast cancer incidence and mortality. Cancer 74(1 Suppl):222–227.
Garland, S. M., M. Hernandez-Avila, C. M. Wheeler, G. Perez, D. M. Harper, S. Leodolter, G. W. K. Tang, D. G. Ferris, M. Steben, J. Bryan, F. J. Taddeo, R. Railkar, M. T. Esser, H. L. Sings, M. Nelson, J. Boslego, C. Sattler, E. Barr, L. A. Koutsky, and the Females United to Unilaterally Reduce Endo/Ectocervical Disease I Investigators. 2007. Quadrivalent vaccine against human papillomavirus to prevent anogenital diseases. New England Journal of Medicine 356(19): 1928–1943.
Garnero, P., E. Sornay-Rendu, M. C. Chapuy, and P. D. Delmas. 1996. Increased bone turnover in late postmenopausal women is a major determinant of osteoporosis. Journal of Bone and Mineral Research 11(3):337–349.
Gates, M. A., B. A. Rosner, J. L. Hecht, and S. S. Tworoger. 2010. Risk factors for epithelial ovarian cancer by histologic subtype. American Journal of Epidemiology 171(1):45–53.
Gehlbach, S. H., J. S. Avrunin, E. Puleo, and R. Spaeth. 2007. Fracture risk and antiresorptive medication use in older women in the USA. Osteoporosis International 18(6):805–810.
Gelmann, E. P., M. Popovic, D. Blayney, H. Masur, G. Sidhu, R. E. Stahl, and R. C. Gallo. 1983. Proviral DNA of a retrovirus, human T-cell leukemia virus, in two patients with AIDS. Science 220(4599):862–865.
Genant, H. K., K. Engelke, and S. Prevrhal. 2008. Advanced CT bone imaging in osteoporosis. Rheumatology 47 (Suppl. 4):iv9–iv16.
Gensichen, J., M. Beyerm, C. Muth, F. M. Gerlach, M. Von Koff, and J. Ormel. 2006. Case management to improve major depression in primary health care: A systematic review. Psychological Medicine 36(01):7–14.
Geusens, P., P. Autier, S. Boonen, J. Vanhoof, K. Declerck, and J. Raus. 2002. The relationship among history of falls, osteoporosis, and fractures in postmenopausal women. Archives of Physical Medicine and Rehabilitation 83(7):903–906.
Geyer, C. E., J. Forster, D. Lindquist, S. Chan, C. G. Romieu, T. Pienkowski, A. Jagiello-Gruszfeld, J. Crown, A. Chan, B. Kaufman, D. Skarlos, M. Campone, N. Davidson, M. Berger, C. Oliva, S. D. Rubin, S. Stein, and D. Cameron. 2006. Lapatinib plus capecitabine for HER2-positive advanced breast cancer. New England Journal of Medicine 355(26):2733–2743.
Ghafoor, A., A. Jemal, E. Ward, V. Cokkinides, R. Smith, and M. Thun. 2003. Trends in breast cancer by race and ethnicity. CA: A Cancer Journal for Clinicians 53(6):342–355.
Gibbons, S. K., and H. M. Keys. 2000. Advanced cervical cancer. Current Treatment Options in Oncology 1(2):157–160.
Ginsburg, E. S., N. K. Mello, J. H. Mendelson, R. L. Barbieri, S. K. Teoh, M. Rothman, X. Gao, and J. W. Sholar. 1996. Effects of alcohol ingestion on estrogens in postmenopausal women. Journal of the American Medical Association 276(21):1747–1751.
Giuliano, A. R., M. Papenfuss, M. Nour, L. M. Canfield, A. Schneider, and K. Hatch. 1997. Antioxidant nutrients: Associations with persistent human papillomavirus infection. Cancer Epidemiology Biomarkers & Prevention 6(11):917–923.
Giuliano, A. R., E. M. Siegel, D. J. Roe, S. Ferreira, M. L. Baggio, L. Galan, E. Duarte-Franco, L. L. Villa, T. E. Rohan, J. R. Marshall, and E. L. Franco. 2003. Dietary intake and risk of persistent human papillomavirus (HPV) infection: The Ludwig-Mcgill HPV Natural History Study. Journal of Infectious Diseases 188(10):1508–1516.
Golanska, E., K. Hulas-Bigoszewska, M. Sieruta, I. Zawlik, M. Witusik, S. M. Gresner, T. Sobow, M. Styczynska, B. Peplonska, M. Barcikowska, P. P. Liberski, and E. H. Corder. 2009. Earlier onset of Alzheimer’s disease: Risk polymorphisms within PRNP, PRND, CYP46, and APOE genes. Journal of Alzheimer’s Disease 17(2):359–368.
Goldberg, R. J. 1981. Management of depression in the patient with advanced cancer. Journal of the American Medical Association 246(4):373–376.
Goldhahn, J., B. Mitlak, P. Aspenberg, J. A. Kanis, R. Rizzoli, and J. Y. Reginster. 2008. Critical issues in translational and clinical research for the study of new technologies to enhance bone repair. Journal of Bone and Joint Surgery—Series A 90(Suppl. 1):43–47.
Goldman, L. S., N. H. Nielsen, and H. C. Champion. 1999. Awareness, diagnosis, and treatment of depression. Journal of General Internal Medicine 14(9):569–580.
Goldstein, J. M., L. J. Seidman, L. M. O’Brien, N. J. Horton, D. N. Kennedy, N. Makris, V. S. Caviness, Jr., S. V. Faraone, and M. T. Tsuang. 2002. Impact of normal sexual dimorphisms on sex differences in structural brain abnormalities in schizophrenia assessed by magnetic resonance imaging. Archives of General Psychiatry 59(2):154–164.
Gordon, P. B., and L. S. Goldenberg. 1995. Malignant breast masses detected only by ultrasound. A retrospective review. Cancer 76(4):626–630.
Goss, P. E., J. N. Ingle, S. Martino, N. J. Robert, H. B. Muss, M. J. Piccart, M. Castiglione, D. Tu, L. E. Shepherd, K. I. Pritchard, R. B. Livingston, N. E. Davidson, L. Norton, E. A. Perez, J. S. Abrams, P. Therasse, M. J. Palmer, and J. L. Pater. 2003. A randomized trial of letrozole in postmenopausal women after five years of tamoxifen therapy for early-stage breast cancer. New England Journal of Medicine 349(19):1793–1802.
Gotlib, I. H., V. E. Whiffen, J. H. Mount, K. Milne, and N. I. Cordy. 1989. Prevalence rates and demographic characteristics associated with depression in pregnancy and the postpartum. Journal of Consulting and Clinical Psychology 57(2):269–274.
Gotzsche, P. C., and O. Olsen. 2000. Is screening for breast cancer with mammography justifiable? Lancet 355(9198):129–134.
Grady, D., L. Chaput, and M. Kristof. 2003. Diagnosis and Treatment of Coronary Heart Disease in Women: Systematic Reviews of Evidence on Selected Topics. Evidence Report/Technology Assessment No. 81. (Prepared by the University of California, San Francisco-Stanford Evidence-based Practice Center under Contract No 290-97-0013.) AHRQ Publication No. 03-E037. Rockville, MD: Agency for Healthcare Research and Quality.
Grann, V., A. B. Troxel, N. Zojwalla, D. Hershman, S. A. Glied, and J. S. Jacobson. 2006. Regional and racial disparities in breast cancer-specific mortality. Social Science and Medicine 62(2): 337–347.
Grant, B. F., D. A. Dawson, F. S. Stinson, S. P. Chou, M. C. Dufour, and R. P. Pickering. 2004. The 12-month prevalence and trends in DSM-IV alcohol abuse and dependence: United States, 1991–1992 and 2001–2002. Drug and Alcohol Dependence 74(3):223–234.
Greenblatt, R. M., S. A. Lukehart, F. A. Plummer, T. C. Quinn, C. W. Critchlow, R. L. Ashley, L. J. D’Costa, J. O. Ndinya-Achola, L. Corey, A. R. Ronald, and K. K. Holmes. 1988. Genital ulceration as a risk factor for human immunodeficiency virus infection. AIDS 2(1):47–50.
Greenfield, S. F. 2002. Women and alcohol use disorders. Harvard Review of Psychiatry 10(2): 76–85.
Greenfield, S. F., and S. Pirard. 2009. Gender-specific treatment for women with substance use disorders. In Women and Addiction: A Comprehensive Handbook, edited by K. Brady, S. E. Back and S. F. Greenfield. New York: Guilford Press. Pp. 289–306.
Greenstein, B., R. Roa, Y. Dhaher, E. Nunn, A. Greenstein, M. Khamashta, and G. R. Hughes. 2001. Estrogen and progesterone receptors in murine models of systemic lupus erythematosus. International Immunopharmacology 1(6):1025–1035.
Gregg, E. W., M. A. Pereira, and C. J. Caspersen. 2000. Physical activity, falls, and fractures among older adults: A review of the epidemiologic evidence. Journal of the American Geriatrics Society 48(8):883–893.
Grella, C. E. 2008. From generic to gender-responsive treatment: Changes in social policies, treatment services, and outcomes of women in substance abuse treatment. Journal of Psychoactive Drugs (Suppl. 5):327–343.
Grella, C. E., and V. Joshi. 1999. Gender differences in drug treatment careers among clients in the National Drug Abuse Treatment Outcome Study. American Journal of Drug and Alcohol Abuse 25(3):385–406.
Grigoriadis, S., and G. E. Robinson. 2007. Gender issues in depression. Annals of Clinical Psychiatry 19(4):247–255.
Groginsky, E., N. Bowdler, and J. Yankowitz. 1998. Update on vertical HIV transmission. Journal of Reproductive Medicine 43(8):637–646.
Guarneri, V., and P. Conte. 2009. Metastatic breast cancer: Therapeutic options according to molecular subtypes and prior adjuvant therapy. Oncologist 14(7):645–656.
Guidos, B. J., and S. M. Selvaggi. 1999. Use of the thin prep pap test in clinical practice. Diagnostic Cytopathology 20(2):70–73.
Gulati, M., H. R. Black, L. J. Shaw, M. F. Arnsdorf, C. N. Merz, M. S. Lauer, T. H. Marwick, D. K. Pandey, R. H. Wicklund, and R. A. Thisted. 2005. The prognostic value of a nomogram for exercise capacity in women. New England Journal of Medicine 353(5):468–475.
Gulati, M., R. M. Cooper-DeHoff, C. McClure, B. D. Johnson, L. J. Shaw, E. M. Handberg, I. Zineh, S. F. Kelsey, M. F. Arnsdorf, H. R. Black, C. J. Pepine, and C. N. Merz. 2009. Adverse cardiovascular outcomes in women with nonobstructive coronary artery disease: A report from the Women’s Ischemia Syndrome Evaluation Study and the St. James Women Take Heart Project. Archives of Internal Medicine 169(9):843–850.
Gur, D., G. S. Abrams, D. M. Chough, M. A. Ganott, C. M. Hakim, R. L. Perrin, G. Y. Rathfon, J. H. Sumkin, M. L. Zuley, and A. I. Bandos. 2009. Digital breast tomosynthesis: Observer performance study. American Journal of Roentgeneology 193(2):586–591.
Guthrie, B. J., and L. J. Flinchbaugh. 2001. Gender-specific substance prevention programming: Going beyond just focusing on girls. Journal of Early Adolescence 21(3):354–372.
Haas, J., C. Kaplan, E. Gerstenberger, and K. Kerlikowske. 2004. Changes in the use of postmenopausal hormone therapy after the publication of clinical trial results. Annals of Internal Medicine 140:184–188.
Hackley, B. 2010. Antidepressant medication use in pregnancy: Safety of antidepressants in pregnancy. Journal of Midwifery and Women’s Health 55(2):90–100.
Hader, S. L., D. K. Smith, J. S. Moore, and S. D. Holmberg. 2001. HIV infection in women in the United States: Status at the millennium. Journal of the American Medical Association 285(9): 1186–1192.
Hagen, A. I., K. A. Kvistad, L. Maehle, M. M. Holmen, H. Aase, B. Styr, A. Vabo, J. Apold, P. Skaane, and P. Moller. 2007. Sensitivity of MRI versus conventional screening in the diagnosis of BRCA-associated breast cancer in a national prospective series. Breast Journal 16(4):367–374.
Haiman, C. A., D. O. Stram, L. R. Wilkens, M. C. Pike, L. N. Kolonel, B. E. Henderson, and L. Le Marchand. 2006. Ethnic and racial differences in the smoking-related risk of lung cancer. New England Journal of Medicine 354(4):333–342.
Hakim-Elahi, E., H. M. Tovell, and M. S. Burnhill. 1990. Complications of first-trimester abortion: A report of 170,000 cases. Obstetrics and Gynecology 76(1):129–135.
Hall, H. I., R. Song, P. Rhodes, J. Prejean, Q. An, L. M. Lee, J. Karon, R. Brookmeyer, E. H. Kaplan, M. T. McKenna, and R. S. Janssen. 2008. Estimation of HIV incidence in the United States. Journal of the American Medical Association 300(5):520–529.
Hall, J. M., and D. P. McDonnell. 2005. Coregulators in nuclear estrogen receptor action: From concept to therapeutic targeting. Molecular Interventions 5(6):343–357.
Hall, J. M., M. K. Lee, B. Newman, J. E. Morrow, L. A. Anderson, B. Huey, and M. C. King. 1990. Linkage of early-onset familial breast cancer to chromosome 17q21. Science 250(4988):1684–1689.
Hammen, C. 1999. The emergence of an interpersonal approach to depression. In The Interactional Nature of Depression: Advances In Interpersonal Approaches, edited by T. Joiner and J. Coyne. Washington, DC: American Psychological Association.
Hankinson, S. E., G. A. Colditz, D. J. Hunter, W. C. Willett, M. J. Stampfer, B. Rosner, C. H. Hennekens, and F. E. Speizer. 1995. A prospective study of reproductive factors and risk of epithelial ovarian cancer. Cancer 76(2):284–290.
Hannan, M. T., D. T. Felson, B. Dawson-Hughes, K. L. Tucker, L. A. Cupples, P. W. Wilson, and D. P. Kiel. 2000. Risk factors for longitudinal bone loss in elderly men and women: The Framingham Osteoporosis Study. Journal of Bone and Mineral Research 15(4):710–720.
Harawa, N. T., S. Greenland, S. D. Cochran, W. E. Cunningham, and B. Visscher. 2003. Do differences in relationship and partner attributes explain disparities in sexually transmitted disease among young white and black women? Journal of Adolescent Health 32(3):187–191.
Hardy, D., C. C. Liu, R. Xia, J. N. Cormier, W. Chan, A. White, K. Burau, and X. L. Du. 2009. Racial disparities and treatment trends in a large cohort of elderly black and white patients with nonsmall cell lung cancer. Cancer 115(10):2199–2211.
Harlow, S. D., P. Schuman, M. Cohen, S. E. Ohmit, S. Cu-Uvin, X. H. Lin, K. Anastos, D. Burns, R. Greenblatt, H. Minkoff, L. Muderspach, A. Rompalo, D. Warren, M. A. Young, and R. S. Klein. 2000. Effect of HIV infection on menstrual cycle length. Journal of Acquired Immune Deficiency Syndromes 24(1):68–75.
Harper, D. M., E. L. Franco, C. Wheeler, D. G. Ferris, D. Jenkins, A. Schuind, T. Zahaf, B. Innis, P. Naud, N. S. De Carvalho, C. M. Roteli-Martins, J. Teixeira, M. M. Blatter, A. P. Korn, W. Quint, and G. Dubin. 2004. Efficacy of a bivalent L1 virus-like particle vaccine in prevention of infection with human papillomavirus types 16 and 18 in young women: A randomised controlled trial. Lancet 364(9447):1757–1765.
Harris, R. E., E. A. Zang, J. I. Anderson, and E. L. Wynder. 1993. Race and sex differences in lung cancer risk associated with cigarette smoking. International Journal of Epidemiology 22(4):592–599.
Harro, C. D., Y. Y. Pang, R. B. Roden, A. Hildesheim, Z. Wang, M. J. Reynolds, T. C. Mast, R. Robinson, B. R. Murphy, R. A. Karron, J. Dillner, J. T. Schiller, and D. R. Lowy. 2001. Safety and immunogenicity trial in adult volunteers of a human papillomavirus 16 L1 virus-like particle vaccine. Journal of the National Cancer Institute 93(4):284–292.
Hartman, A. R., B. L. Daniel, A. W. Kurian, M. A. Mills, K. W. Nowels, F. M. Dirbas, K. E. Kingham, N. M. Chun, R. J. Herfkens, J. M. Ford, and S. K. Plevritis. 2004. Breast magnetic resonance image screening and ductal lavage in women at high genetic risk for breast carcinoma. Cancer 100(3):479–489.
Harwell, J. I., T. Moench, K. H. Mayer, S. Chapman, I. Rodriguez, and S. Cu-Uvin. 2003. A pilot study of treatment of bacterial vaginosis with a buffering vaginal microbicide. Journal of Women’s Health 12(3):255–259.
Haus, A. G. 1990. Technologic improvements in screen-film mammography. Radiology 174(3): 628–637.
Hausauer, A., T. Keegan, E. Chang, and C. Clarke. 2007. Recent breast cancer trends among Asian/Pacific Islander, Hispanic, and African–American women in the US: Changes by tumor subtype. Breast Cancer Research 9:R90.
Hausauer, A., T. Keegan, E. Chang, S. Glaser, H. Howe, and C. Clarke. 2009. Recent trends in breast cancer incidence in US white women by county-level urban/rural and poverty status. BMC Medicine 7(1):31.
Hayashi, T., and D. Faustman. 1999. NOD mice are defective in proteasome production and activation of NF-kappaB. Molecular Cell Biology 19(12):8646–8659.
Health Council of the Netherlands. 2002. The Benefit of Population Screening for Breast Cancer with Mammography. http://www.gezondheidsraad.nl/en/publications/benefit-population-screening-breast-cancer-mammography (accessed November 16, 2009).
Heath, K. V., K. J. Chan, J. Singer, M. V. O’Shaughnessy, J. S. Montaner, and R. S. Hogg. 2002. Incidence of morphological and lipid abnormalities: Gender and treatment differentials after initiation of first antiretroviral therapy. International Journal of Epidemiology 31(5):1016–1020.
Hebert, R., M. F. Dubois, C. Wolfson, L. Chambers, and C. Cohen. 2001. Factors associated with long-term institutionalization of older people with dementia: Data from the Canadian Study of Health and Aging. Journals of Gerontology. Series A, Biological Sciences and Medical Sciences 56(11):M693–M699.
Henderson, I. C., D. A. Berry, G. D. Demetri, C. T. Cirrincione, L. J. Goldstein, S. Martino, J. N. Ingle, M. R. Cooper, D. F. Hayes, K. H. Tkaczuk, G. Fleming, J. F. Holland, D. B. Duggan, J. T. Carpenter, E. Frei, III, R. L. Schilsky, W. C. Wood, H. B. Muss, and L. Norton. 2003. Improved outcomes from adding sequential Paclitaxel but not from escalating Doxorubicin dose in an adjuvant chemotherapy regimen for patients with node-positive primary breast cancer. Journal Clinical Oncology 21(6):976–983.
Hendrick, R. E., R. A. Smith, J. H. Rutledge, 3rd, and C. R. Smart. 1997. Benefit of screening mammography in women aged 40–49: A new meta-analysis of randomized controlled trials. Journal of the National Cancer Institute Monographs (22):87–92.
Henschke, C. I., and O. S. Miettinen. 2004. Women’s susceptibility to tobacco carcinogens. Lung Cancer 43(1):1–5.
Henschke C. I., D. F. Yankelevitz, D. M. Libby, M. W. Pasmantier, J. P. Smith, and O. S. Miettinen. 2006. Survival of patients with stage 1 lung cancer detected on CT screening. The New England Journal of Medicine 355(17):1763–1771.
Herrera, F. G., L. Vidal, A. Oza, M. Milosevic, and A. Fyles. 2008. Molecular targeted agents combined with chemo-radiation in the treatment of locally advanced cervix cancer. Reviews on Recent Clinical Trials 3:111–120.
Hersh, A. L., M. L. Stefanick, and R. S. Stafford. 2004. National use of postmenopausal hormone therapy: Annual trends and response to recent evidence. Journal of the American Medical Association 291(1):47–53.
HHS (US Department of Health and Human Services). 1999. Mental Health: A Report of the Surgeon General. Rockville, MD: US Department of Health and Human Services, Substance Abuse and Mental Health Services Administration, Center for Mental Health Services, National Institutes of Health, National Institute of Mental Health.
———. 2000. Healthy People 2010: Understanding and Improving Health. Washington, DC: US Government Printing Office.
———. 2001. Women and Smoking: A Report of the Surgeon General. http://www.surgeongeneral.gov/library/womenandtobacco/index.html (accessed January 13, 2010).
———. 2003. Drug Use Among Racial/Ethnic Minorities. Washington, DC: US Department of Health and Human Services.
———. 2005. Progress in Autoimmune Diseases Research. Washington, DC: US Department of Health and Human Services.
———. 2007. ATHENA (Athletes Targeting Healthy Exercise and Nutrition Alternatives). http://www.nrepp.samhsa.gov/listofprograms.asp?textsearch=alcohol&ShowHide=1&Sort=1&N1=1&A3=3&G2=2 (accessed February 22, 2010).
———. 2008. Substance Abuse and HIV/AIDS. US Department of Health and Human Services.
———. 2009. FDA Approves New Indication for Gardasil to Prevent Genital Warts in Men and Boys. http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm187003.htm (accessed June 12, 2010).
———. 2010. FDA Drug Safety Communication: Ongoing Safety Review of Oral Bisphosphonates and Atypical Subtrochanteric Femur Fractures. http://www.fda.gov/Drugs/DrugSafety/PostmarketDrugSafetyInformationforPatientsandProviders/ucm203891.htm (accessed July 7, 2010).
Hibbitts, S. 2009. Should boys receive the human papillomavirus vaccine? Yes. British Medical Journal 339:b4928.
Hillier, T. A., K. L. Stone, D. C. Bauer, J. H. Rizzo, K. L. Pedula, J. A. Cauley, K. E. Ensrud, M. C. Hochberg, and S. R. Cummings. 2007. Evaluating the value of repeat bone mineral density measurement and prediction of fractures in older women: The study of osteoporotic fractures. Archives of Internal Medicine 167(2):155–160.
Hindmarch, I. 2002. Beyond the monoamine hypothesis: Mechanisms, molecules and methods. European Psychiatry 17(Suppl. 3):294–299.
Hippisley-Cox, J., and C. Coupland. 2010. Individualising the risks of statins in men and women in England and Wales: Population-based cohort study. Heart 96(12):939–947.
Hirose, K., N. Hamajima, T. Takezaki, T. Kuroishi, K. Kuzuya, S. Sasaki, S. Tokudome, and K. Tajima. 1998. Smoking and dietary risk factors for cervical cancer at different age group in Japan. Journal of Epidemiology 8(1):6–14.
Hitti, J., L. M. Frenkel, A. M. Stek, S. A. Nachman, D. Baker, A. Gonzalez-Garcia, A. Provisor, E. M. Thorpe, M. E. Paul, M. Foca, J. Gandia, S. Huang, L. J. Wei, L. M. Stevens, D. H. Watts, and J. McNamara. 2004. Maternal toxicity with continuous nevirapine in pregnancy: Results from PACTG 1022. Journal of Acquired Immune Deficiency Syndromes 36(3):772–776.
Hogan, M. F. 2003. The President’s New Freedom Commission: Recommendations to transform mental health care in America. Psychiatric Services 54(11):1467–1474.
Hogberg, T. 2008. Adjuvant chemotherapy in endometrial carcinoma: Overview of randomised trials. Clinical Oncology 20(6):463–469.
Holdcraft, L. C., and W. G. Iacono. 2004. Cross-generational effects on gender differences in psychoactive drug abuse and dependence. Drug and Alcohol Dependence 74(2):147–158.
Hollon, S. D., R. B. Jarrett, A. A. Nierenberg, M. E. Thase, M. Trivedi, and A. J. Rush. 2005. Psychotherapy and medication in the treatment of adult and geriatric depression: Which monotherapy or combined treatment? Journal of Clinical Psychiatry 66(4):455–468.
Hoyert, D. L. 2007. Maternal mortality and related concepts. Vital Health Statistics 3(33):1–13.
HRSA (Health Resources and Services Agency). 2008. Women’s Health USA 2008. Rockville, MD: US Department of Health and Human Services.
Hser, Y. I., M. D. Anglin, and M. W. Booth. 1987. Sex differences in addict careers. 3. Addiction. American Journal of Drug and Alcohol Abuse 13(3):231–251.
Hua, S., C. B. Kallen, R. Dhar, M. T. Baquero, C. E. Mason, B. A. Russell, P. K. Shah, J. Liu, A. Khramtsov, M. S. Tretiakova, T. N. Krausz, O. I. Olopade, D. L. Rimm, and K. P. White. 2008. Genomic analysis of estrogen cascade reveals histone variant H2A.Z associated with breast cancer progression. Molecular Systems Biology 4:188.
Huang, C. Q., B. R. Dong, Z. C. Lu, J. R. Yue, and Q. X. Liu. 2010. Chronic diseases and risk for depression in old age: A meta-analysis of published literature. Ageing Research Reviews 9(2): 131–141.
Huang, Q. Y., and A. W. C. Kung. 2006. Genetics of osteoporosis. Molecular Genetics and Metabolism 88:295–306.
Huang, W., P. R. Fisher, K. Dulaimy, L. A. Tudorica, B. O’Shea, and T. M. Button. 2004. Detection of breast malignancy: Diagnostic MR protocol for improved specificity1. Radiology 232(2): 585–591.
Hull, S. C., and A. L. Caplan. 2009. The case for vaccinating boys against human papillomavirus. Public Health Genomics 12(5-6):362–367.
Hung, C-F., B. Ma, A. Monie, S-W. Tsen, and T-C. Wu. 2008. Therapeutic human papillomavirus vaccines: Current clinical trials and future directions. Expert Opinion on Biological Therapy 8(4):421–439.
Hung, J., B. Chaitman, J. Lam, J. Lesperance, G. Dupras, P. Fines, and M. Bourassa. 1984. Noninvasive diagnostic test choices for the evaluation of coronary artery disease in women: A multivariate comparison of cardiac fluoroscopy, exercise electrocardiography and exercise thallium myocardial perfusion scintigraphy. Journal of the American College of Cardiology 4(1):8–16.
Ikeda, K. 2008. Osteocytes in the pathogenesis of osteoporosis. Geriatrics and Gerontology International 8(4):213–217.
Innes, K. E., and T. E. Byers. 2001. Smoking during pregnancy and breast cancer risk in very young women (United States). Cancer Causes and Control 12(2):179–185.
The International Perinatal HIV Group. 1999. The mode of delivery and the risk of vertical transmission of human immunodeficiency virus type 1: A meta analysis of 15 prospective cohort studies. New England Journal of Medicine 340:977–987.
Invernizzi, P., M. Miozzo, P. M. Battezzati, I. Bianchi, F. R. Grati, G. Simoni, C. Selmi, M. Watnik, M. E. Gershwin, and M. Podda. 2004. Frequency of monosomy X in women with primary biliary cirrhosis. Lancet 363(9408):533–535.
Invernizzi, P., M. Miozzo, C. Selmi, L. Persani, P. M. Battezzati, M. Zuin, S. Lucchi, P. L. Meroni, B. Marasini, S. Zeni, M. Watnik, F. R. Grati, G. Simoni, M. E. Gershwin, and M. Podda. 2005. X chromosome monosomy: A common mechanism for autoimmune diseases. Journal of Immunology 175(1):575–578.
Invernizzi, P., S. Pasini, and M. Podda. 2008. X chromosome in autoimmune diseases. Expert Review of Clinical Immunology 4(5):591–597.
Invernizzi, P., S. Pasini, C. Selmi, M. E. Gershwin, and M. Podda. 2009. Female predominance and X chromosome defects in autoimmune diseases. Journal of Autoimmunity 33(1):12–16.
IOM. 1993. Clinical Applications of Mifepristone (RU486) and Other Antiprogestins: Assessing the Science and Recommending a Research Agenda. Washington, DC: National Academy Press.
———. 1995. The Best Intentions: Unintended Pregnancy and the Well-Being of Children and Families. Washington, DC: National Academy Press.
———. 2001a. No Time to Lose: Getting More from HIV Prevention. Washington, DC: National Academy Press.
———. 2001b. Exploring the Biological Contributions to Human Health: Does Sex Matter? Washington, DC: National Academy Press.
———. 2003. Fulfilling the Potential for Cancer Prevention and Early Detection. Washington, DC: The National Academies Press.
———. 2004a. Strategies to Leverage Research Funding: Guiding DoD’s Peer Reviewed Medical Research Programs. Washington, DC: The National Academies Press.
———. 2004b. Frontiers in Contraceptive Research. Washington, DC: The National Academies Press.
———. 2005. Complementary and Alternative Medicine in the United States. Washington, DC: National Academies Press.
———. 2006. From Cancer Patient to Cancer Survivor—Lost in Transition: An American Society of Clinical Oncology and Institute of Medicine Symposium. Edited by M. Hewitt and P. A. Ganz. Washington, DC: The National Academies Press.
———. 2008. Cancer Care for the Whole Patient: Meeting Psychosocial Health Needs. Edited by N. E. Adler and A. Page. Washington, DC: The National Academies Press.
———. 2009. A Review of the HHS Family Planning Program: Mission, Management, and Measurement of Results. Washington, DC: The National Academies Press.
Iwamoto, J., T. Takeda, and Y. Sato. 2005. Effect of treadmill exercise on bone mass in female rats. Experimental Animals 54(1):1–6.
Jackson, R. D., A. Z. LaCroix, M. Gass, R. B. Wallace, J. Robbins, C. E. Lewis, T. Bassford, S. A. Beresford, H. R. Black, P. Blanchette, D. E. Bonds, R. L. Brunner, R. G. Brzyski, B. Caan, J. A. Cauley, R. T. Chlebowski, S. R. Cummings, I. Granek, J. Hays, G. Heiss, S. L. Hendrix, B. V. Howard, J. Hsia, F. A. Hubbell, K. C. Johnson, H. Judd, J. M. Kotchen, L. H. Kuller, R. D. Langer, N. L. Lasser, M. C. Limacher, S. Ludlam, J. E. Manson, K. L. Margolis, J. McGowan, J. K. Ockene, M. J. O’Sullivan, L. Phillips, R. L. Prentice, G. E. Sarto, M. L. Stefanick, L. Van Horn, J. Wactawski-Wende, E. Whitlock, G. L. Anderson, A. R. Assaf, and D. Barad. 2006. Calcium plus vitamin D supplementation and the risk of fractures. New England Journal of Medicine 354(7):669–683.
Jansson, A., M. Gustafsson, and E. Wilander. 1998. Efficiency of cytological screening for detection of cervical squamous carcinoma. A study in the county of Uppsala 1991–1994. Upsala Journal of Medical Sciences 103(2):147–154.
Jemal, A., E. Ward, and M. J. Thun. 2007. Recent trends in breast cancer incidence rates by age and tumor characteristics among US women. Breast Cancer Research 9(3):R28.
Jemal, A., M. J. Thun, L. A. Ries, H. L. Howe, H. K. Weir, M. M. Center, E. Ward, X. C. Wu, C. Eheman, R. Anderson, U. A. Ajani, B. Kohler, and B. K. Edwards. 2008. Annual report to the nation on the status of cancer, 1975–2005, featuring trends in lung cancer, tobacco use, and tobacco control. Journal of the National Cancer Institute 100(23):1672–1694.
Jemal, A., R. Siegel, J. Xu, and E. Ward. 2010. Cancer Statistics, 2010. CA: A Cancer Journal for Clinicians 60(5):277–300.
Jong, R. A., M. J. Yaffe, M. Skarpathiotakis, R. S. Shumak, N. M. Danjoux, A. Gunesekara, and D. B. Plewes. 2003. Contrast-enhanced digital mammography: Initial clinical experience. Radiology 228(3):842–850.
Jordan, S. J., P. M. Webb, and A. C. Green. 2005. Height, age at menarche, and risk of epithelial ovarian cancer. Cancer Epidemiology Biomarkers and Prevention 14(8):2045–2048.
Juethner, S. N., C. Williamson, M. B. Ristig, P. Tebas, W. Seyfried, and J. A. Aberg. 2003. Nonnucleoside reverse transcriptase inhibitor resistance among antiretroviral-naive HIV-positive pregnant women. Journal of Acquired Immune Deficiency Syndromes 32(2):153–156.
Kaas, R., S. Verhoef, J. Wesseling, M. A. Rookus, H. S. A. Oldenburg, J. V. Peeters, and E. J. T. Rutgers. 2010. Prophylactic mastectomy in BRCA1 and BRCA2 mutation carriers: Very low risk for subsequent breast cancer. Annals of Surgery 251(3):488–492.
Kado, D. M., W. S. Browner, L. Palermo, M. C. Nevitt, H. K. Genant, and S. R. Cummings. 1999. Vertebral fractures and mortality in older women—A prospective study. Study of Osteoporotic Fractures Research Group. Archives of Internal Medicine 159(11):1215–1220.
Kaiser Family Foundation. 2009. HIV/AIDS Policy Fact Sheet. The HIV/AIDS Epidemic in the United States. Menlo Park, CA: Kaiser Family Foundation.
Källén, B. 2007. The safety of antidepressant drugs during pregnancy. Expert Opinion on Drug Safety 6(4):357–370.
Kanis, J. A. 2002. Diagnosis of osteoporosis and assessment of fracture risk. Lancet 359(9321): 1929–1936.
Kanis, J. A., F. Borgstrom, C. De Laet, H. Johansson, O. Johnell, B. Jonsson, A. Oden, N. Zethraeus, B. Pfleger, and N. Khaltaev. 2005. Assessment of fracture risk. Osteoporosis International 16(6):581–589.
Kaplan, S. S. 2001. Clinical utility of bilateral whole-breast US in the evaluation of women with dense breast tissue. Radiology 221(3):641–649.
Karasawa, R., S. Ozaki, K. Nishioka, and T. Kato. 2005. Autoantibodies to peroxiredoxin I and IV in patients with systemic autoimmune diseases. Microbiology and Immunology 49(1):57–65.
Karellas, A., and S. Vedantham. 2008. Breast cancer imaging: A perspective for the next decade. Medical Physics 35(11):4878–4897.
Katon, W., M. Von Korff, E. Lin, G. Simon, E. Walker, J. Unutzer, T. Bush, J. Russo, and E. Ludman. 1999. Stepped collaborative care for primary care patients with persistent symptoms of depression: A randomized trial. Archives of General Psychiatry 56(12):1109–1115.
Kaufman, J. 2010. Evolution and immunity. Immunology 130:459–462.
Kell, M. R., and M. J. Kerin. 2004. Sentinel lymph node biopsy. British Medical Journal 328(7452): 1330–1331.
Keller, S. M., M. G. Vangel, S. Adak, H. Wagner, J. H. Schiller, A. Herskovic, R. Komaki, M. C. Perry, R. S. Marks, R. B. Livingston, and D. H. Johnson. 2002. The influence of gender on survival and tumor recurrence following adjuvant therapy of completely resected stages II and IIIA non-small cell lung cancer. Lung Cancer 37(3):303–309.
Kelly, K., J. Dean, W. Comulada, and S-J. Lee. 2009. Breast cancer detection using automated whole breast ultrasound and mammography in radiographically dense breasts. European Radiology 20(3):734–742.
Keshavarz, H., S. D. Hillis, B. A. Kieke, and P. A. Marchbanks. 2002. Hysterectomy surveillance—United States, 1994–1999. Morbidity and Mortality Weekly Report 51(SS-5).
Kessler, R. C. 2003. Epidemiology of women and depression. Journal of Affective Disorders 74(1): 5–13.
Kessler, R. C., and J. McLeod. 1984. Sex differences in vulnerability to undesirable life events American Sociological Review 49:620–631.
Kessler, R. C., P. Berglund, O. Demler, R. Jin, D. Koretz, K. R. Merikangas, A. J. Rush, E. E. Walters, and P. S. Wang. 2003. The epidemiology of major depressive disorder: Results from the National Comorbidity Survey Replication (NCS-R). Journal of the American Medical Association 289(23):3095–3105.
Keyes, K. M., B. F. Grant, and D. S. Hasin. 2008. Evidence for a closing gender gap in alcohol use, abuse, and dependence in the United States population. Drug and Alcohol Dependence 93(1-2):21–29.
Khalkhali, I., J. Villanueva-Meyer, S. L. Edell, J. L. Connolly, S. J. Schnitt, J. K. Baum, M. J. Houlihan, R. M. Jenkins, and S. B. Haber. 2000. Diagnostic accuracy of 99mTc-sestamibi breast imaging: Multicenter trial results. Journal of Nuclear Medicine 41(12):1973–1979.
Khawam, E. A., G. Laurencic, and D. A. Malone, Jr. 2006. Side effects of antidepressants: An overview. Cleveland Clinic Journal of Medicine 73(4):351–353, 356–361.
Kiel, D. P., D. T. Felson, J. J. Anderson, P. W. Wilson, and M. A. Moskowitz. 1987. Hip fracture and the use of estrogens in postmenopausal women. The Framingham Study. New England Journal of Medicine 317(19):1169–1174.
Kilts, C. D., R. E. Gross, T. D. Ely, and K. P. G. Drexler. 2004. The neural correlates of cue-induced craving in cocaine-dependent women. American Journal of Psychiatry 161(2):233–241.
Kim, C., Y. S. Kwok, P. Heagerty, and R. Redberg. 2001. Pharmacologic stress testing for coronary disease diagnosis: A meta-analysis. American Heart Journal 142(6):934–944.
Kim, C., R. F. Redberg, T. Pavlic, and K. A. Eagle. 2007. A systematic review of gender differences in mortality after coronary artery bypass graft surgery and percutaneous coronary interventions. Clinical Cardiology 30(10):491–495.
Kim, E. S., and V. Menon. 2009. Status of women in cardiovascular clinical trials. Arteriosclerosis, Thrombosis, and Vascular Biology 29(3):279–283.
Kim, S. J., and J. S. Gill. 2009. H-Y incompatibility predicts short-term outcomes for kidney transplant recipients. Journal of the American Society of Nephrology 20(9):2025–2033.
Kinsey, T., A. Jemal, J. Liff, E. Ward, and M. Thun. 2008. Secular trends in mortality from common cancers in the United States by educational attainment, 1993–2001. Journal of the National Cancer Institute 100(14):1003–1012.
Kirnbauer, R., F. Booy, N. Cheng, D. R. Lowy, and J. T. Schiller. 1992. Papillomavirus L1 major capsid protein self-assembles into virus-like particles that are highly immunogenic. Proceedings of the National Academy of Sciences of the United States of America 89(24):12180–12184.
Kirnbauer, R., J. Taub, H. Greenstone, R. Roden, M. Durst, L. Gissmann, D. R. Lowy, and J. T. Schiller. 1993. Efficient self-assembly of human papillomavirus type 16 L1 and L1-l2 into virus-like particles. Journal of Virology 67(12):6929–6936.
Kiyotani, K., T. Mushiroda, C. K. Imamura, N. Hosono, T. Tsunoda, M. Kubo, Y. Tanigawara, D. A. Flockhart, Z. Desta, T. C. Skaar, F. Aki, K. Hirata, Y. Takatsuka, M. Okazaki, S. Ohsumi, T. Yamakawa, M. Sasa, Y. Nakamura, and H. Zembutsu. 2010. Significant effect of polymorphisms in CYP2D6 and ABCC2 on clinical outcomes of adjuvant tamoxifen therapy for breast cancer patients. Journal Clinical Oncology 28(8):1287–1293.
Klerman, L. V. 2000. The intendedness of pregnancy: A concept in transition. Maternal and Child Health Journal 4(3):155–162.
Klinkhamer, P. J., W. J. Meerding, P. F. Rosier, and A. G. Hanselaar. 2003. Liquid-based cervical cytology. Cancer 99(5):263–271.
Kojic, E. M., and S. Cu-Uvin. 2007. Special care issues of women living with HIV-AIDS. Infectious Disease Clinics of North America 21(1): ix, 133–148.
Kolb, T. M., J. Lichy, and J. H. Newhouse. 2002. Comparison of the performance of screening mammography, physical examination, and breast US and evaluation of factors that influence them: An analysis of 27,825 patient evaluations. Radiology 225(1):165–175.
Komaki, R., J. D. Cox, S. J. Kister, F. E. Gump, and A. Estabrook. 1990. Stage I and II breast carcinoma: Treatment with limited surgery and radiation therapy versus mastectomy. Radiology 174(1):255–257.
Kong, D. F., V. Hasselblad, D. E. Kandzari, L. K. Newby, and R. M. Califf. 2002. Seeking the optimal aspirin dose in acute coronary syndromes. American Journal of Cardiology 90(6):622–625.
Koninckx, P. R., C. Meuleman, S. Demeyere, E. Lesaffre, and F. J. Cornillie. 1991. Suggestive evidence that pelvic endometriosis is a progressive disease, whereas deeply infiltrating endometriosis is associated with pelvic pain. Fertility and Sterility 55(4):759–765.
Kornstein, S. G., and R. K. Schneider. 2001. Clinical features of treatment-resistant depression. Journal of Clinical Psychiatry 62 (Suppl. 16):18–25.
Koss, M. P., J. A. Bailey, N. P. Yuan, V. M. Herrera, and E. L. Lichter. 2003. Depression and PTSD in survivors of male violence: Research and training initiatives to facilitate recovery. Psychology of Women Quarterly 27(2):130–142.
Koushik, A., D. J. Hunter, D. Spiegelman, K. E. Anderson, A. A. Arslan, W. L. Beeson, P. A. van den Brandt, J. E. Buring, J. R. Cerhan, G. A. Colditz, G. E. Fraser, J. L. Freudenheim, J. M. Genkinger, R. A. Goldbohm, S. E. Hankinson, K. L. Koenig, S. C. Larsson, M. Leitzmann, M. L. McCullough, A. B. Miller, A. Patel, T. E. Rohan, A. Schatzkin, E. Smit, W. C. Willett, A. Wolk, S. M. Zhang, and S. A. Smith-Warner. 2005. Fruits and vegetables and ovarian cancer risk in a pooled analysis of 12 cohort studies. Cancer Epidemiology, Biomarkers and Prevention 14(9):2160–2167.
Koutsky, L. A., K. A. Ault, C. M. Wheeler, D. R. Brown, E. Barr, F. B. Alvarez, L. M. Chiacchierini, K. U. Jansen, and the Proof of Principle Study Investigators. 2002. A controlled trial of a human papillomavirus type 16 vaccine. New England Journal of Medicine 347(21):1645–1651.
Krainer, M., S. Silva-Arrieta, M. G. FitzGerald, A. Shimada, C. Ishioka, R. Kanamaru, D. J. MacDonald, H. Unsal, D. M. Finkelstein, A. Bowcock, K. J. Isselbacher, and D. A. Haber. 1997. Differential contributions of BRCA1 and BRCA2 to early-onset breast cancer. New England Journal of Medicine 336(20):1416–1421.
Kramer, M. M., and C. L. Wells. 1996. Does physical activity reduce risk of estrogen dependent cancer in women? Medicine & Science in Sports & Exercise 28(3):322–334.
Kreuzer, M., P. Boffetta, E. Whitley, W. Ahrens, V. Gaborieau, J. Heinrich, K. H. Jockel, L. Kreienbrock, S. Mallone, F. Merletti, F. Roesch, P. Zambon, and L. Simonato. 2000. Gender differences in lung cancer risk by smoking: A multicentre case–control study in Germany and Italy. British Journal of Cancer 82(1):227–233.
Kriege, M., C. T. Brekelmans, C. Boetes, P. E. Besnard, H. M. Zonderland, I. M. Obdeijn, R. A. Manoliu, T. Kok, H. Peterse, M. M. Tilanus-Linthorst, S. H. Muller, S. Meijer, J. C. Oosterwijk, L. V. Beex, R. A. Tollenaar, H. J. de Koning, E. J. Rutgers, J. G. Klijn, and the Magnetic Resonance Imaging Screening Study Group. 2004. Efficacy of MRI and mammography for breast-cancer screening in women with a familial or genetic predisposition. New England Journal of Medicine 351(5):427–437.
Kriege, M., C. T. Brekelmans, C. Boetes, S. H. Muller, H. M. Zonderland, I. M. Obdeijn, R. A. Manoliu, T. Kok, E. J. Rutgers, H. J. Koning, J. G. Klijn, and Dutch MRI Screening (MRISC) Study Group. 2006. Differences between first and subsequent rounds of the MRISC breast cancer screening program for women with a familial or genetic predisposition. Cancer 106(11):2318–2326.
Krieger, N., J. T. Chen, and P. D. Waterman. 2010. Decline in US breast cancer rates after the Women’s Health Initiative: Socioeconomic and racial/ethnic differentials. American Journal of Public Health 100(S1):S132–S139.
Kudenchuk, P. J., C. Maynard, J. S. Martin, M. Wirkus, and W. D. Weaver. 1996. Comparison of presentation, treatment, and outcome of acute myocardial infarction in men versus women (The Myocardial Infarction Triage and Intervention Registry). American Journal of Cardiology 78(1):9–14.
Kugaya, A., and G. Sanacora. 2005. Beyond monoamines: Glutamatergic function in mood disorders. CNS Spectrums 10(10):808–819.
Kuhl, C. K., S. Schrading, C. C. Leutner, N. Morakkabati-Spitz, E. Wardelmann, R. Fimmers, W. Kuhn, and H. H. Schild. 2005. Mammography, breast ultrasound, and magnetic resonance imaging for surveillance of women at high familial risk for breast cancer. Journal of Clinical Oncology 23(33):8469–8476.
Kuiper, G. G., E. Enmark, M. Pelto-Huikko, S. Nilsson, and J. A. Gustafsson. 1996. Cloning of a novel receptor expressed in rat prostate and ovary. Proceedings of the National Academy of Sciences of the United States of America 93(12):5925–5930.
Kuiper, G. G., B. Carlsson, K. Grandien, E. Enmark, J. Haggblad, S. Nilsson, and J. A. Gustafsson. 1997. Comparison of the ligand binding specificity and transcript tissue distribution of estrogen receptors alpha and beta. Endocrinology 138(3):863–870.
Kulie, T., A. Groff, J. Redmer, J. Hounshell, and S. Schrager. 2009. Vitamin D: An evidence-based review. Journal of the American Board of Family Medicine 22(6):698–706.
Kulski, J. K., J. W. Sadleir, S. R. Kelsall, M. S. Cicchini, G. Shellam, S. W. Peng, Y. M. Qi, D. A. Galloway, J. Zhou, and I. H. Frazer. 1998. Type specific and genotype cross reactive B epitopes of the L1 protein of HPV16 defined by a panel of monoclonal antibodies. Virology 243(2):275–282.
Kumpfer, K. L., P. Smith, and J. F. Summerhays. 2008. A wakeup call to the prevention field: Are prevention programs for substance use effective for girls? Substance Use and Misuse 43(8-9): 978–1001.
Kurtzke, J. F. 1975. A reassessment of the distribution of multiple sclerosis. Acta Neurologica Scandinavica 51(2):137–157.
La Vecchia, C., A. Decarli, M. Fasoli, F. Parazzini, S. Franceschi, A. Gentile, and E. Negri. 1988. Dietary vitamin A and the risk of intraepithelial and invasive cervical neoplasia. Gynecologic Oncology 30(2):187–195.
Landesman, S. H., L. A. Kalish, D. N. Burns, H. Minkoff, H. E. Fox, C. Zorrilla, P. Garcia, M. G. Fowler, L. Mofenson, and R. Tuomala. 1996. Obstetrical factors and the transmission of human immunodeficiency virus type 1 from mother to child. The Women and Infants Transmission Study. New England Journal of Medicine 334(25):1617–1623.
Lang, J., and L. McCullough. 2008. Pathways to ischemic neuronal cell death: Are sex differences relevant? Journal of Translational Medicine 6(1):33.
Langan, N. P., and B. M. M. Pelissier. 2001. Gender differences among prisoners in drug treatment. Journal of Substance Abuse 13(3):291–301.
Lannfelt, L., H. Basun, L. O. Wahlund, B. A. Rowe, and S. L. Wagner. 1995. Decreased alphasecretase-cleaved amyloid precursor protein as a diagnostic marker for Alzheimers disease. Nature Medicine 1(8):829–832.
Lansky, A. J., J. S. Hochman, P. A. Ward, G. S. Mintz, R. Fabunmi, P. B. Berger, G. New, C. L. Grines, C. G. Pietras, M. J. Kern, M. Ferrell, M. B. Leon, R. Mehran, C. White, J. H. Mieres, J. W. Moses, G. W. Stone, and A. K. Jacobs. 2005. Percutaneous coronary intervention and adjunctive pharmacotherapy in women: A statement for healthcare professionals from the American Heart Association. Circulation 111(7):940–953.
LaRosa, J. C., J. He, and S. Vupputuri. 1999. Effect of statins on risk of coronary disease: A metaanalysis of randomized controlled trials. Journal of the American Medical Association 282(24): 2340–2346.
Latka, M. H., F. Kapadia, and P. Fortin. 2008. The female condom: Effectiveness and convenience, not “female control,” valued by US urban adolescents. AIDs Education and Prevention 20(2): 160–170.
Lau, C. S., and A. Mak. 2009. The socioeconomic burden of SLE. Nature Reviews Rheumatology 5(7):400–404.
Launer, L. J., K. Andersen, M. E. Dewey, L. Letenneur, A. Ott, L. A. Amaducci, C. Brayne, J. R. Copeland, J. F. Dartigues, P. Kragh-Sorensen, A. Lobo, J. M. Martinez-Lage, T. Stijnen, and A. Hofman. 1999. Rates and risk factors for dementia and Alzheimer’s disease: Results from EURODEM pooled analyses. EURODEM Incidence Research Group and Work Groups. European Studies of Dementia. Neurology 52(1):78–84.
Leach, M. O., C. R. Boggis, A. K. Dixon, D. F. Easton, R. A. Eeles, D. G. Evans, F. J. Gilbert, I. Griebsch, R. J. Hoff, P. Kessar, S. R. Lakhani, S. M. Moss, A. Nerurkar, A. R. Padhani, L. J. Pointon, D. Thompson, and R. M. Warren. 2005. Screening with magnetic resonance imaging and mammography of a UK population at high familial risk of breast cancer: A prospective multicentre cohort study (MARIBS). Lancet 365(9473):1769–1778.
Leconte, I., C. Feger, C. Galant, M. Berliere, B. V. Berg, W. D’Hoore, and B. Maldague. 2003. Mammography and subsequent whole-breast sonography of nonpalpable breast cancers: The importance of radiologic breast density. American Journal of Roentgeneology 180(6):1675–1679.
Lee, A. V., J. G. Jackson, J. L. Gooch, S. G. Hilsenbeck, E. Coronado-Heinsohn, C. K. Osborne, and D. Yee. 1999. Enhancement of insulin-like growth factor signaling in human breast cancer: Estrogen regulation of insulin receptor substrate-1 expression in vitro and in vivo. Molecular Endocrinology 13(5):787–796.
Lee, J. R., F. Paultre, and L. Mosca. 2005. The association between educational level and risk of cardiovascular disease fatality among women with cardiovascular disease. Women’s Health Issues 15(2):80–88.
Lehman, C. D., J. D. Blume, P. Weatherall, D. Thickman, N. Hylton, E. Warner, E. Pisano, S. J. Schnitt, C. Gatsonis, M. Schnall, G. A. DeAngelis, P. Stomper, E. L. Rosen, M. O’Loughlin, S. Harms, and D. A. Bluemke. 2005. Screening women at high risk for breast cancer with mammography and magnetic resonance imaging. Cancer 103(9):1898–1905.
Lehman, C. D., C. Isaacs, M. D. Schnall, E. D. Pisano, S. M. Ascher, P. T. Weatherall, D. A. Bluemke, D. J. Bowen, P. K. Marcom, D. K. Armstrong, S. M. Domchek, G. Tomlinson, S. J. Skates, and C. Gatsonis. 2007. Cancer yield of mammography, MR, and US in high-risk women: Prospective multi-institution breast cancer screening study. Radiology 244(2):381–388.
Lehtinen, M., and J. Paavonen. 2004. Vaccination against human papillomaviruses shows great promise. Lancet 364(9447):1731–1732.
Lenderink, T., E. Boersma, W. Ruzyllo, P. Widimsky, E. M. Ohman, P. W. Armstrong, L. Wallentin, and M. L. Simoons. 2004. Bleeding events with abciximab in acute coronary syndromes without early revascularization: An analysis of GUSTO IV-ACS. American Heart Journal 147(5):865–873.
Leslie, W. D., and Manitoba Bone Density Program. 2006. The impact of bone area on short-term bone density precision. Journal of Clinical Densitometry 9(2):150–153.
Leslie, W. D., S. O’Donnell, S. Jean, C. Lagace, P. Walsh, C. Bancej, S. Morin, D. A. Hanley, and A. Papaioannou. 2009. Trends in hip fracture rates in Canada. Journal of the American Medical Association 302(8):883–889.
Lewin, J. M., C. J. D’Orsi, R. E. Hendrick, L. J. Moss, P. K. Isaacs, A. Karellas, and G. R. Cutter. 2002. Clinical comparison of full-field digital mammography and screen-film mammography for detection of breast cancer. American Journal of Roentgeneology 179(3):671–677.
Lewis, J. P. 2007. An interpretation of the EBCTCG data. Oncologist 12(5):505–509.
Lewis-Fernandez, R., A. K. Das, C. Alfonso, M. M. Weissman, and M. Olfson. 2005. Depression in US Hispanics: Diagnostic and management considerations in family practice. Journal of the American Board of Family Practice 18(4):282–296.
Liberman, M., F. Sampalis, D. S. Mulder, and J. S. Sampalis. 2003. Breast cancer diagnosis by scintimammography: A meta-analysis and review of the literature. Breast Cancer Research and Treatment 80(1):115–126.
Linder, J. 1998. Recent advances in thin-layer cytology. Diagnostic Cytopathology 18(1):24–32.
Ling, F. W. 1999. Randomized controlled trial of depot leuprolide in patients with chronic pelvic pain and clinically suspected endometriosis. Pelvic Pain Study Group. Obstetrics and Gynecology 93(1):51–58.
Lloyd, T., V. M. Chinchilli, N. Johnson-Rollings, K. Kieselhorst, D. F. Eggli, and R. Marcus. 2000. Adult female hip bone density reflects teenage sports-exercise patterns but not teenage calcium intake. Pediatrics 106(1):40–44.
Lloyd-Jones, D., R. Adams, M. Carnethon, G. De Simone, T. B. Ferguson, K. Flegal, E. Ford, K. Furie, A. Go, K. Greenlund, N. Haase, S. Hailpern, M. Ho, V. Howard, B. Kissela, S. Kittner, D. Lackland, L. Lisabeth, A. Marelli, M. McDermott, J. Meigs, D. Mozaffarian, G. Nichol, C. O’Donnell, V. Roger, W. Rosamond, R. Sacco, P. Sorlie, R. Stafford, J. Steinberger, T. Thom, S. Wasserthiel-Smoller, N. Wong, J. Wylie-Rosett, and Y. Hong. 2009. Heart disease and stroke statistics—2009 update: A report from the American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Circulation 119(3):480–486.
Lockhat, F. B., J. O. Emembolu, and J. C. Konje. 2004. The evaluation of the effectiveness of an intrauterine-administered progestogen (levonorgestrel) in the symptomatic treatment of endometriosis and in the staging of the disease. Human Reproduction 19(1):179–184.
Loizzi, V., G. Cormio, M. Vicino, and L. Selvaggi. 2008. Neoadjuvant chemotherapy: An alternative option of treatment for locally advanced cervical cancer. Gynecologic and Obstetric Investigation 65(2):96–103.
Longshore, D., P. L. Ellickson, D. F. McCaffrey, and P. A. St Clair. 2007. School-based drug prevention among at-risk adolescents: Effects of ALERT Plus. Health Education and Behavior 34(4):651–668.
Lord, R. V., J. Brabender, D. Gandara, V. Alberola, C. Camps, M. Domine, F. Cardenal, J. M. Sanchez, P. H. Gumerlock, M. Taron, J. J. Sanchez, K. D. Danenberg, P. V. Danenberg, and R. Rosell. 2002. Low ERCC1 expression correlates with prolonged survival after cisplatin plus gemcitabine chemotherapy in non-small cell lung cancer. Clinical Cancer Research 8(7):2286–2291.
Lukka, H., H. Hirte, A. Fyles, G. Thomas, L. Elit, M. Johnston, M. Fung Kee Fung, and G. Browman. 2002. Concurrent cisplatin-based chemotherapy plus radiotherapy for cervical cancer—A metaanalysis. Clinical Oncology 14(3):203–212.
Luppino, F. S., L. M. de Wit, P. F. Bouvy, T. Stijnen, P. Cuijpers, B. W. Penninx, and F. G. Zitman. 2010. Overweight, obesity, and depression: A systematic review and meta-analysis of longitudinal studies. Archives of General Psychiatry 67(3):220–229.
Lyman, G. H., A. E. Giuliano, M. R. Somerfield, A. B. Benson, 3rd, D. C. Bodurka, H. J. Burstein, A. J. Cochran, H. S. Cody, 3rd, S. B. Edge, S. Galper, J. A. Hayman, T. Y. Kim, C. L. Perkins, D. A. Podoloff, V. H. Sivasubramaniam, R. R. Turner, R. Wahl, D. L. Weaver, A. C. Wolff, and E. P. Winer. 2005. American Society of Clinical Oncology guideline recommendations for sentinel lymph node biopsy in early-stage breast cancer. Journal of Clinical Oncology 23(30):7703–7720.
Lyons, F., S. Hopkins, B. Kelleher, A. McGeary, G. Sheehan, J. Geoghegan, C. Bergin, F. M. Mulcahy, and P. A. McCormick. 2006. Maternal hepatotoxicity with nevirapine as part of combination antiretroviral therapy in pregnancy. HIV Medicine 7(4):255–260.
MacLusky, N. 2004. Estrogen and Alzheimer’s disease: The apolipoprotein connection. Endocrinology 145(7):3062–3064.
Mahon, B. D., A. Wittke, V. Weaver, and M. T. Cantorna. 2003. The targets of vitamin D depend on the differentiation and activation status of CD4 positive T cells. Journal of Cellular Biochemistry 89(5):922–932.
Makinen, J., J. Johansson, C. Tomas, E. Tomas, P. K. Heinonen, T. Laatikainen, M. Kauko, A. M. Heikkinen, and J. Sjoberg. 2001. Morbidity of 10 110 hysterectomies by type of approach. Human Reproduction 16(7):1473–1478.
Malberg, J. E., and L. M. Monteggia. 2008. VGF, a new player in antidepressant action? Science Signaling 1(18):pe19.
Mandelbrot, L., J. Le Chenadec, A. Berrebi, A. Bongain, J. L. Benifla, J. F. Delfraissy, S. Blanche, and M. J. Mayaux. 1998. Perinatal HIV-1 transmission: Interaction between zidovudine prophylaxis and mode of delivery in the French Perinatal Cohort. Journal of the American Medical Association 280(1):55–60.
Manfredi, R., L. Calza, D. Cocchi, and F. Chiodo. 2003. Antiretroviral treatment and advanced age: Epidemiologic, laboratory, and clinical features in the elderly. Journal of Acquired Immune Deficiency Syndromes 33(1):112–114.
Marcus, S. M., E. A. Young, K. B. Kerber, S. Kornstein, A. H. Farabaugh, J. Mitchell, S. R. Wisniewski, G. K. Balasubramani, M. H. Trivedi, and A. J. Rush. 2005. Gender differences in depression: Findings from the STAR*D study. Journal of Affective Disorders 87(2-3):141–150.
Mariani, G., A. Fasolo, E. De Benedictis, and L. Gianni. 2009. Trastuzumab as adjuvant systemic therapy for HER2-positive breast cancer. Nature Clinical Practice Oncology 6(2):93–104.
Markou, A., T. Duka, and G. M. Prelevic. 2005. Estrogens and brain function. Hormones 4(1): 9–17.
Marsh, J. C., D. Cao, and T. D’Aunno. 2004. Gender differences in the impact of comprehensive services in substance abuse treatment. Journal of Substance Abuse Treatment 27(4):289–300.
Martin, M., T. Pienkowski, J. Mackey, M. Pawlicki, J-P. Guastalla, C. Weaver, E. Tomiak, T. Al-Tweigeri, L. Chap, E. Juhos, R. Guevin, A. Howell, T. Fornander, J. Hainsworth, R. Coleman, J. Vinholes, M. Modiano, T. Pinter, S. C. Tang, B. Colwell, C. Prady, L. Provencher, D. Walde, A. Rodriguez-Lescure, J. Hugh, C. Loret, M. Rupin, S. Blitz, P. Jacobs, M. Murawsky, A. Riva, C. Vogel, and the Breast Cancer International Research Group 001 Investigators. 2005. Adjuvant docetaxel for node-positive breast cancer. New England Journal of Medicine 352(22):2302–2313.
Mathew, A., and P. S. George. 2009. Trends in incidence and mortality rates of squamous cell carcinoma and adenocarcinoma of cervix—worldwide. Asian Pacific Journal of Cancer Prevention 10(4):645–650.
Mathias, S. D., M. Kuppermann, R. F. Liberman, R. C. Lipschutz, and J. F. Steege. 1996. Chronic pelvic pain: Prevalence, health-related quality of life, and economic correlates. Obstetrics and Gynecology 87(3):321–327.
Mayer, K. H., J. Peipert, T. Fleming, A. Fullem, T. Moench, S. Cu-Uvin, M. Bentley, M. Chesney, and Z. Rosenberg. 2001. Safety and tolerability of buffergel, a novel vaginal microbicide, in women in the United States. Clinical Infectious Diseases 32(3):476–482.
Mazhude, C., S. Jones, S. Murad, C. Taylor, and P. Easterbrook. 2002. Female sex but not ethnicity is a strong predictor of non-nucleoside reverse transcriptase inhibitor-induced rash. AIDS 16(11):1566–1568.
Mazure, C. 1998. Life stressors as risk factors in depression. Clinical Psychology: Science and Practice 5:291–313.
Mazure, C. M., M. L. Bruce, P. K. Maciejewski, and S. C. Jacobs. 2000. Adverse life events and cognitive-personality characteristics in the prediction of major depression and antidepressant response. American Journal of Psychiatry 157(6):896–903.
McDuffie, H. H., D. J. Klaassen, and J. A. Dosman. 1987. Female-male differences in patients with primary lung cancer. Cancer 59(10):1825–1830.
McGovern, S. L., Z. Liao, M. K. Bucci, M. F. McAleer, M. D. Jeter, J. Y. Chang, M. S. O’Reilly, J. D. Cox, P. K. Allen, and R. Komaki. 2009. Is sex associated with the outcome of patients treated with radiation for nonsmall cell lung cancer? Cancer 115(14):3233–3242.
McGuire, W., J. Blessing, D. Moore, S. Lentz, and G. Photopulos. 1996. Paclitaxel has moderate activity in squamous cervix cancer. A Gynecologic Oncology Group Study. Journal Clinical Oncology 14(3):792–795.
McPherson, K., A. Herbert, A. Judge, A. Clarke, S. Bridgman, M. Maresh, and C. Overton. 2005. Psychosexual health 5 years after hysterectomy: Population-based comparison with endometrial ablation for dysfunctional uterine bleeding. Health Expectations 8(3):234–243.
McQuellon, R. P., H. T. Thaler, D. Cella, and D. H. Moore. 2006. Quality of life (QOL) outcomes from a randomized trial of cisplatin versus cisplatin plus paclitaxel in advanced cervical cancer: A Gynecologic Oncology Group Study. Gynecologic Oncology 101(2):296–304.
Mehilli, J., A. Kastrati, J. Dirschinger, H. Bollwein, F-J. Neumann, and A. Schomig. 2000. Differences in prognostic factors and outcomes between women and men undergoing coronary artery stenting. Journal of the American Medical Association 284(14):1799–1805.
Meisamy, S., P. J. Bolan, E. H. Baker, M. G. Pollema, C. T. Le, F. Kelcz, M. C. Lechner, B. A. Luikens, R. A. Carlson, K. R. Brandt, K. K. Amrami, M. T. Nelson, L. I. Everson, T. H. Emory, T. M. Tuttle, D. Yee, and M. Garwood. 2005. Adding in vivo quantitative 1H MR spectroscopy to improve diagnostic accuracy of breast MR imaging: Preliminary results of observer performance study at 4.0 T. Radiology 236(2):465–475.
Melton, L. J., 3rd, S. H. Kan, M. A. Frye, H. W. Wahner, W. M. O’Fallon, and B. L. Riggs. 1989. Epidemiology of vertebral fractures in women. American Journal of Epidemiology 129(5):1000–1011.
Melton, L. J., 3rd, E. A. Chrischilles, C. Cooper, A. W. Lane, and B. L. Riggs. 1992. Perspective. How many women have osteoporosis? Journal of Bone and Mineral Research 7(9):1005–1010.
Menacker, F., and B. E. Hamilton. 2010. Recent trends in cesarean delivery in the United States. NCHS Data Brief (35):1–8.
Mendez, M. F. 2006. The accurate diagnosis of early-onset dementia. International Journal of Psychiatry in Medicine 36(4):401–412.
Menke, R., and H. Flynn. 2009. Relationships between stigma, depression, and treatment in white and African American primary care patients. Journal of Nervous Mental Disorder 197(6): 407–411.
Meredith, L. S., M. Orlando, N. Humphrey, P. Camp, and C. D. Sherbourne. 2001. Are better ratings of the patient-provider relationship associated with higher quality care for depression? Medical Care 39(4):349–360.
Metcalfe, K. A. 2009. Oophorectomy for breast cancer prevention in women with BRCA1 or BRCA2 mutations. Women’s Health 5(1):63–68.
Middeldorp, S. 2005. Oral contraceptives and the risk of venous thromboembolism. Gender Medicine 2 (Suppl. A):S3–S9.
Mieres, J. H., L. J. Shaw, A. Arai, M. J. Budoff, S. D. Flamm, W. G. Hundley, T. H. Marwick, L. Mosca, A. R. Patel, M. A. Quinones, R. F. Redberg, K. A. Taubert, A. J. Taylor, G. S. Thomas, and N. K. Wenger. 2005. Role of noninvasive testing in the clinical evaluation of women with suspected coronary artery disease: Consensus statement from the Cardiac Imaging Committee, Council on Clinical Cardiology, and the Cardiovascular Imaging and Intervention Committee, Council on Cardiovascular Radiology and Intervention, American Heart Association. Circulation 111(5):682–696.
Migliorelli, R., A. Teson, L. Sabe, M. Petracchi, R. Leiguarda, and S. E. Starkstein. 1995. Prevalence and correlates of dysthymia and major depression among patients with Alzheimers disease. American Journal of Psychiatry 152(1):37–44.
Mikhail, G. W. 2005. Coronary heart disease in women. British Medical Journal 331(7515): 467–468.
Miller, A. B., T. To, C. J. Baines, and C. Wall. 2000. Canadian National Breast Screening Study-2: 13-year results of a randomized trial in women aged 50–59 years. Journal of the National Cancer Institute 92(18):1490–1499.
———. 2002. The Canadian National Breast Screening Study-1: Breast cancer mortality after 11 to 16 years of follow-up: A randomized screening trial of mammography in women age 40 to 49 years. Annals of Internal Medicine 137(5 Pt. 1):305–312.
Miller, A. H., S. Ancoli-Israel, J. E. Bower, L. Capuron, and M. R. Irwin. 2008. Neuroendocrine-immune mechanisms of behavioral comorbidities in patients with cancer. Journal Clinical Oncology 26(6):971–982.
Miller, B. A., E. J. Feuer, and B. F. Hankey. 1991. The increasing incidence of breast cancer since 1982: Relevance of early detection. Cancer Causes and Control 2(2):67–74.
Miller, K., M. Wang, J. Gralow, M. Dickler, M. Cobleigh, E. A. Perez, T. Shenkier, D. Cella, and N. E. Davidson. 2007. Paclitaxel plus bevacizumab versus paclitaxel alone for metastatic breast cancer. New England Journal of Medicine 357(26):2666–2676.
Milner, K. A., M. Funk, S. Richards, R. M. Wilmes, V. Vaccarino, and H. M. Krumholz. 1999. Gender differences in symptom presentation associated with coronary heart disease. American Journal of Cardiology 84(4):396–399.
Mink, P. J., A. R. Folsom, T. A. Sellers, and L. H. Kushi. 1996. Physical activity, waist-to-hip ratio, and other risk factors for ovarian cancer: A follow-up study of older women. Epidemiology 7(1):38–45.
Minkoff, H. 1997. HIV disease in pregnancy. Introduction. Obstetrics and Gynecology Clinics of North America 24(4):XI-XVII.
Minkoff, H., and L. M. Mofenson. 1994. The role of obstetric interventions in the prevention of pediatric human immunodeficiency virus infection. American Journal of Obstetrics and Gynecology 171(5):1167–1175.
Minkoff, H., R. Hershow, D. H. Watts, M. Frederick, I. Cheng, R. Tuomala, J. Pitt, C. D. Zorrilla, H. Hammill, S. K. Adeniyi-Jones, and B. Thompson. 2003. The relationship of pregnancy to human immunodeficiency virus disease progression. American Journal of Obstetrics and Gynecology 189(2):552–559.
Miranda, J., J. Siddique, T. R. Belin, and L. P. Kohn-Wood. 2005. Depression prevalence in disadvantaged young black women—African and Caribbean immigrants compared to US-born African Americans. Social Psychiatry Psychiatric Epidemiology 40(4):253–258.
Modjtahedi, B. S., S. P. Modjtahedi, and H. I. Maibach. 2006. Gender: A possible determinant in dosing of dermatologic drugs—An overview. Cutaneous and Ocular Toxicology 25(3):195–210.
Moerman, E. J., K. Teng, D. A. Lipschitz, and B. Lecka-Czernik. 2004. Aging activates adipogenic and suppresses osteogenic programs in mesenchymal marrow stroma/stem cells: The role of PPAR-gamma2 transcription factor and TGF-beta/BMP signaling pathways. Aging Cell 3(6):379–389.
Mollerup, S., D. Ryberg, A. Hewer, D. H. Phillips, and A. Haugen. 1999. Sex differences in lung cyp1a1 expression and DNA adduct levels among lung cancer patients. Cancer Research 59(14):3317–3320.
Mollerup, S., K. Jorgensen, G. Berge, and A. Haugen. 2002. Expression of estrogen receptors alpha and beta in human lung tissue and cell lines. Lung Cancer 37(2):153–159.
Monninkhof, E. M., S. G. Elias, F. A. Vlems, I. van der Tweel, A. J. Schuit, D. W. Voskuil, F. E. van Leeuwen, and TFPAC. 2007. Physical activity and breast cancer: A systematic review. Epidemiology 18(1):137–157.
Montgrain, P. R., R. Quintana, Y. Rascon, D. W. Burton, L. J. Deftos, A. Casillas, and R. H. Hastings. 2007. Parathyroid hormone-related protein varies with sex and androgen status in nonsmall cell lung cancer. Cancer 110(6):1313–1320.
Moore, A. L., O. Kirk, A. M. Johnson, C. Katlama, A. Blaxhult, M. Dietrich, R. Colebunders, A. Chiesi, J. D. Lungren, and A. N. Phillips. 2003. Virologic, immunologic, and clinical response to highly active antiretroviral therapy: The gender issue revisited. Journal of Acquired Immune Deficiency Syndromes 32(4):452–461.
Moore, D. H. 2006. Cervical cancer. Obstetrics and Gynecology 107(5):1152–1161.
Moore, D. H., J. A. Blessing, R. P. McQuellon, H. T. Thaler, D. Cella, J. Benda, D. S. Miller, G. Olt, S. King, J. F. Boggess, and T. F. Rocereto. 2004. Phase III study of cisplatin with or without paclitaxel in stage IVB, recurrent, or persistent squamous cell carcinoma of the cervix: A gynecologic oncology group study. Journal Clinical Oncology 22(15):3113–3119.
Moore, R. D., I. Fortgang, J. Keruly, and R. E. Chaisson. 1996. Adverse events from drug therapy for human immunodeficiency virus disease. American Journal of Medicine 101(1):34–40.
Moore, R. D., J. C. Keruly, and R. E. Chaisson. 2001. Incidence of pancreatitis in HIV-infected patients receiving nucleoside reverse transcriptase inhibitor drugs. AIDS 15(5):617–620.
Mora, S., R. J. Glynn, J. Hsia, J. G. MacFadyen, J. Genest, and P. M. Ridker. 2010. Statins for the primary prevention of cardiovascular events in women with elevated high-sensitivity C-reactive protein or dyslipidemia: Results from the justification for the use of statins in prevention: An intervention trial evaluating rosuvastatin (JUPITER) and meta-analysis of women from primary prevention trials. Circulation 121(9):1069–1077.
Morris, M., P. J. Eifel, J. Lu, P. W. Grigsby, C. Levenback, R. E. Stevens, M. Rotman, D. M. Gershenson, and D. G. Mutch. 1999. Pelvic radiation with concurrent chemotherapy compared with pelvic and para-aortic radiation for high-risk cervical cancer. New England Journal of Medicine 340(15):1137–1143.
Mosca, L., L. J. Appel, E. J. Benjamin, K. Berra, N. Chandra-Strobos, R. P. Fabunmi, D. Grady, C. K. Haan, S. N. Hayes, D. R. Judelson, N. L. Keenan, P. McBride, S. Oparil, P. Ouyang, M. C. Oz, M. E. Mendelsohn, R. C. Pasternak, V. W. Pinn, R. M. Robertson, K. Schenck-Gustafsson, C. A. Sila, S. C. Smith, Jr., G. Sopko, A. L. Taylor, B. W. Walsh, N. K. Wenger, and C. L. Williams. 2004. Evidence-based guidelines for cardiovascular disease prevention in women. Circulation 109(5):672–693.
Mosca, L., A. H. Linfante, E. J. Benjamin, K. Berra, S. N. Hayes, B. W. Walsh, R. P. Fabunmi, J. Kwan, T. Mills, and S. L. Simpson. 2005. National study of physician awareness and adherence to cardiovascular disease prevention guidelines. Circulation 111(4):499–510.
Mosca, L., H. Mochari, A. Christian, K. Berra, K. Taubert, T. Mills, K. A. Burdick, and S. L. Simpson. 2006. National study of women’s awareness, preventive action, and barriers to cardiovascular health. Circulation 113(4):525–534.
Mosca, L., C. L. Banka, E. J. Benjamin, K. Berra, C. Bushnell, R. J. Dolor, T. G. Ganiats, A. S. Gomes, H. L. Gornik, C. Gracia, M. Gulati, C. K. Haan, D. R. Judelson, N. Keenan, E. Kelepouris, E. D. Michos, L. K. Newby, S. Oparil, P. Ouyang, M. C. Oz, D. Petitti, V. W. Pinn, R. F. Redberg, R. Scott, K. Sherif, S. C. Smith, Jr., G. Sopko, R. H. Steinhorn, N. J. Stone, K. A. Taubert, B. A. Todd, E. Urbina, N. K. Wenger, Expert Panel/Writing Group; American Heart Association; American Academy of Family Physicians; American College of Obstetricians and Gynecologists; American College of Cardiology Foundation; Society of Thoracic Surgeons; American Medical Women’s Association; Centers for Disease Control and Prevention; Office of Research on Women’s Health; Association of Black Cardiologists; American College of Physicians; World Heart Federation; National Heart, Lung, and Blood Institute; American College of Nurse Practitioners. 2007. Evidence-based guidelines for cardiovascular disease prevention in women: 2007 update. Circulation 115(11):1481–1501. [erratum appears in circulation. 2007 Apr 17;115(15):E407].
Mosca, L., H. Mochari-Greenberger, R. J. Dolor, L. K. Newby, and K. J. Robb. 2010. Twelve-year follow-up of American women’s awareness of cardiovascular disease risk and barriers to heart health. Circulation 3(2):120–127.
Mosselman, S., J. Polman, and R. Dijkema. 1996. ER beta: Identification and characterization of a novel human estrogen receptor. FEBS Letters 392(1):49–53.
Muñoz, N. 2000. Human papillomavirus and cancer: The epidemiological evidence. Journal of Clinical Virology 19(1-2):1–5.
Muñoz, N., F. X. Bosch, S. de Sanjose, R. Herrero, X. Castellsague, K. V. Shah, P. J. F. Snijders, C. J. L. M. Meijer, and the International Agency for Research on Cancer Multicenter Cervical Cancer Study Group. 2003. Epidemiologic classification of human papillomavirus types associated with cervical cancer. New England Journal of Medicine 348(6):518–527.
Muntz, E., and W. Logan. 1979. Focal spot size and scatter suppression in magnification mammography. American Journal of Roentgeneology 133(3):453–459.
Murray, C. J. L., and A. D. Lopez. 1997. Alternative projections of mortality and disability by cause 1990–2020: Global burden of disease study. Lancet 349(9064):1498–1504.
Murray, J. A., S. B. Moore, C. T. Van Dyke, B. D. Lahr, R. A. Dierkhising, A. R. Zinsmeister, L. J. Melton, 3rd, C. M. Kroning, M. El-Yousseff, and A. J. Czaja. 2007. HLA DQ gene dosage and risk and severity of celiac disease. Clinical Gastroenterology and Hepatology 5(12):1406–1412.
Nazroo, J. Y., A. C. Edwards, and G. W. Brown. 1997. Gender differences in the onset of depression following a shared life event: A study of couples. Psychological Medicine 27(1):9–19.
NCI (National Cancer Institute). 2008. SEER Cancer Statistics Review, 1975–2005. http://seer.cancer.gov/csr/1975_2005/results_merged/sect_05_cervix_uteri.pdf (accessed August 20, 2009).
———. 2009a. Adjuvant and Neoadjuvant Therapy for Breast Cancer. http://www.cancer.gov/cancertopics/factsheet/therapy/adjuvant-breast (accessed April 8, 2010).
———. 2009b. SEER Stat Fact Sheets: Cancer of the Corpus and Uterus. www.seer.cancer.gov/statfacts/html/corp.html (accessed August 20, 2009).
———. 2010a. Fast Stats: Age-Adjusted U. S. Mortality Rates by Cancer Site All Ages, All Races, Female, 1975–2007. http://seer.cancer.gov/faststats/selections.php?#Output (accessed August 12, 2010).
———. 2010b. SEER Cancer Statistics Review, 1975–2007. http://seer.cancer.gov/csr/1975_2007/sections.html (accessed May 28, 2010).
———. 2010c. SEER Cancer Statistics Review, 1975–2007. http://seer.cancer.gov/statfacts/html/cervix.html#incidence-mortality (accessed August 20, 2010).
———. 2010d. SEER Stat Fact Sheets: Ovary. Bethesda, MD: National Cancer Institute.
———. 2010e. SEER Stat Fact Sheets: Corpus and Uterus. http://seer.cancer.gov/statfacts/html/corp. html (accessed August 10, 2010).
———. 2010f. SEER Fact Sheets: Cancer of the Lung and Bronchus. http://seer.cancer.gov/statfacts/html/lungb.html (accessed August 3, 2010).
Negro, A., B. K. Brar, and K. F. Lee. 2004. Essential roles of HER2/ERBB2 in cardiac development and function. Recent Progress in Hormone Research 59:1–12.
Nelson, H. D., K. Tyne, A. Naik, C. Bougatsos, B. K. Chan, and L. Humphrey. 2009a. Screening for breast cancer: An update for the US Preventive Services Task Force. Annals of Internal Medicine 151(10):727–737.
Nelson, H. D., R. Fu, J. C. Griffin, P. Nygren, M. E. Smith, and L. Humphrey. 2009b. Systematic review: Comparative effectiveness of medications to reduce risk for primary breast cancer. Annals of Internal Medicine 151(10):703–715.
Nelson, H. H., D. C. Christiani, E. J. Mark, J. K. Wiencke, J. C. Wain, and K. T. Kelsey. 1999. Implications and prognostic value of K-ras mutation for early-stage lung cancer in women. Journal of the National Cancer Institute 91(23):2032–2038.
Netting, E. E., and F. G. Williams. 1996. Case manager-physician collaboration: Implications for professional identity, roles, and relationships. Health and Social Work 21(3):216–216.
Nicastri, E., C. Angeletti, L. Palmisano, L. Sarmati, A. Chiesi, A. Geraci, M. Andreoni, and S. Vella. 2005. Gender differences in clinical progression of HIV-1-infected individuals during long-term highly active antiretroviral therapy. AIDS 19(6):577–583.
Niklason, L. T., B. T. Christian, L. E. Niklason, D. B. Kopans, D. E. Castleberry, B. H. OpsahlOng, C. E. Landberg, P. J. Slanetz, A. A. Giardino, R. Moore, D. Albagli, M. C. DeJule, P. F. Fitzgerald, D. F. Fobare, B. W. Giambattista, R. F. Kwasnick, J. Liu, S. J. Lubowski, G. E. Possin, J. F. Richotte, C. Y. Wei, and R. F. Wirth. 1997. Digital tomosynthesis in breast imaging. Radiology 205(2):399–406.
NIMH (National Institute of Mental Health). 2009. Women and Depression: Discovering Hope. http://www.nimh.nih.gov/health/publications/women-and-depression-discovering-hope/complete-index. shtml (accessed April 19, 2010).
NOF (National Osteoporosis Foundation). 2002. America’s Bone Health: The State of Osteoporosis and Low Bone Mass. http://www.nof.org/advocacy/prevalence/ (accessed June 3, 2010).
Nolen-Hoeksema, S. 2004. The response styles theory. In Depressive Rumination: Nature, Theroy, and Treatment, edited by C. Papageorgiou and A. Wells. New York: Wiley. Pp. 107–124.
Nolen-Hoeksema, S., J. Larson, and C. Grayson. 1999. Explaining the gender difference in depressive symptoms. Journal of Personality and Social Psychology 77(5):1061–1072.
Norman, S. A., S. L. Potashnik, M. L. Galantino, A. M. De Michele, L. House, and A. R. Localio. 2007. Modifiable risk factors for breast cancer recurrence: What can we tell survivors? Journal of Women’s Health 16(2):177–190.
Nusbaum, R., and C. Isaacs. 2007. Management updates for women with a BRCA1 or BRCA2 mutation. Molecular Diagnosis and Therapy 11(3):133–144.
Nygaard, I., M. D. Barber, K. L. Burgio, K. Kenton, S. Meikle, J. Schaffer, C. Spino, W. E. Whitehead, J. Wu, and D. J. Brody. 2008. Prevalence of symptomatic pelvic floor disorders in US women. Journal of the American Medical Association 300(11):1311–1316.
Nystrom, L., I. Andersson, N. Bjurstam, J. Frisell, B. Nordenskjold, and L. E. Rutqvist. 2002. Long-term effects of mammography screening: Updated overview of the Swedish randomised trials. Lancet 359(9310):909–919.
O’Donoghue, M., W. E. Boden, E. Braunwald, C. P. Cannon, T. C. Clayton, R. J. de Winter, K. A. Fox, B. Lagerqvist, P. A. McCullough, S. A. Murphy, R. Spacek, E. Swahn, L. Wallentin, F. Windhausen, and M. S. Sabatine. 2008. Early invasive vs conservative treatment strategies in women and men with unstable angina and non-ST-segment elevation myocardial infarction: A meta-analysis. Journal of the American Medical Association 300(1):71–80.
O’Keefe-McCarthy, S. 2008. Women’s experiences of cardiac pain: A review of the literature. Canadian Journal of Cardiovascular Nursing 18(3):18–25.
Office of Environmental Health Hazard Assessment. 2005. Proposed Identification of Environmental Tobacco Smoke as a Toxic Air Contaminant. http://www.oehha.org/air/environmental_tobacco/2005etsfinal.html (accessed August 12, 2010).
Office of National Drug Control Policy. 2006. Results from the 2006 National Survey on Drug Use and Health: National Findings. Edited by the US Department of Health and Human Services, Substance Abuse and Mental Health Services Administration Office of Applied Studies.
Office of the Surgeon General. 2006. Surgeon General’s Report—The Health Consequences of Involuntary Exposure to Tobacco Smoke. Atlanta, GA: US Department of Health and Human Services, Centers for Disease Control and Prevention, Coordinating Center for Health Promotion, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health.
Okolo, S. 2008. Incidence, aetiology and epidemiology of uterine fibroids. Best Practice and Research. Clinical Obstetrics and Gynaecology 22(4):571–588.
Olopade, O. I., T. A. Grushko, R. Nanda, and D. Huo. 2008. Advances in breast cancer: Pathways to personalized medicine. Clinical Cancer Research 14(24):7988–7999.
Olsen, O., and P. C. Gotzsche. 2001. Cochrane review on screening for breast cancer with mammog-raphy. Lancet 358(9290):1340–1342.
Olson, J. A., Jr., L. M. McCall, P. Beitsch, P. W. Whitworth, D. S. Reintgen, P. W. Blumencranz, A. M. Leitch, S. Saha, K. K. Hunt, and A. E. Giuliano. 2008. Impact of immediate versus delayed axillary node dissection on surgical outcomes in breast cancer patients with positive sentinel nodes: Results from American College of Surgeons oncology group trials Z0010 and Z0011. Journal of Clinical Oncology 26(21):3530–3535.
Olsson, S. E., S. K. Kjaer, K. Sigurdsson, O. E. Iversen, M. Hernandez-Avila, C. M. Wheeler, G. Perez, D. R. Brown, L. A. Koutsky, E. H. Tay, P. Garcia, K. A. Ault, S. M. Garland, S. Leodolter, G. W. Tang, D. G. Ferris, J. Paavonen, M. Lehtinen, M. Steben, F. X. Bosch, J. Dillner, E. A. Joura, S. Majewski, N. Munoz, E. R. Myers, L. L. Villa, F. J. Taddeo, C. Roberts, A. Tadesse, J. Bryan, R. Maansson, S. Vuocolo, T. M. Hesley, A. Saah, E. Barr, and R. M. Haupt. 2009. Evaluation of quadrivalent HPV 6/11/16/18 vaccine efficacy against cervical and anogenital disease in subjects with serological evidence of prior vaccine type HPV infection. Human Vaccines 5(10):696–704.
Osann, K. E., H. Anton-Culver, T. Kurosaki, and T. Taylor. 1993. Sex differences in lung-cancer risk associated with cigarette smoking. International Journal of Cancer 54(1):44–48.
Osborne, C. K. 1998. Tamoxifen in the treatment of breast cancer. New England Journal of Medicine 339(22):1609–1618.
OTA (US Congress Office of Technology Assessment). 1994. Hip Fracture Outcomes in People Age 50 and Over—Background Paper. OTA-BP-H-120. Washington, DC: US Government Printing Office.
Ouellette, D., G. Desbiens, C. Emond, and G. Beauchamp. 1998. Lung cancer in women compared with men: Stage, treatment, and survival. Annals of Thoracic Surgery 66(4):1140–1143; discussion 1143–1144.
Padian, N. S., S. C. Shiboski, and N. P. Jewell. 1991. Female-to-male transmission of human-immunodeficiency-virus. Journal of the American Medical Association 266(12):1664–1667.
Padian, N. S., A. van der Straten, G. Ramjee, T. Chipato, G. de Bruyn, K. Blanchard, S. Shiboski, E. T. Montgomery, H. Fancher, H. Cheng, M. Rosenblum, M. van der Laan, N. Jewell, and J. McIntyre. 2007. Diaphragm and lubricant gel for prevention of HIV acquisition in southern African women: A randomised controlled trial. Lancet 370(9583):251–261.
Palella, F. J., K. M. Delaney, A. C. Moorman, M. O. Loveless, J. Fuhrer, G. A. Satten, D. J. Aschman, S. D. Holmberg. 1998. Declining morbidity and mortality among patients with advanced human immunodeficiency virus infection. New England Journal of Medicine 338(13):853–860.
Palm, N. W., and R. Medzhitov. 2009. Pattern recognition receptors and control of adaptive immunity. Immunological Reviews 227:221–233.
Palumbo, P., B. Holland, T. Dobbs, C. P. Pau, C. C. Luo, E. J. Abrams, S. Nesheim, P. Vink, R. Respess, and M. Bulterys. 2001. Antiretroviral resistance mutations among pregnant human immunodeficiency virus type 1-infected women and their newborns in the United States: Vertical transmission and clades. Journal of Infectious Diseases 184(9):1120–1126.
Papakostas, G. I., M. E. Thase, M. Fava, J. C. Nelson, and R. C. Shelton. 2007. Are antidepressant drugs that combine serotonergic and noradrenergic mechanisms of action more effective than the selective serotonin reuptake inhibitors in treating major depressive disorder? A meta-analysis of studies of newer agents. Biological Psychiatry 62(11):1217–1227.
Pappas, G., S. Queen, W. Hadden, and G. Fisher. 1993. The increasing disparity in mortality between socioeconomic groups in the United States, 1960 and 1986. New England Journal of Medicine 329(2):103–109.
Pariante, C. M., and A. H. Miller. 2001. Glucocorticoid receptors in major depression: Relevance to pathophysiology and treatment. Biological Psychiatry 49(5):391–404.
Partridge, A. H., and E. P. Winer. 2009. On mammography—more agreement than disagreement. New England Journal of Medicine 361(26):2499–2501.
Patel, A. V., C. Rodriguez, A. L. Pavluck, M. J. Thun, and E. E. Calle. 2006a. Recreational physical activity and sedentary behavior in relation to ovarian cancer risk in a large cohort of U. S. women. American Journal of Epidemiology 163(8):709–716.
Patel, K. K., B. Butler, and K. B. Wells. 2006b. What is necessary to transform the quality of mental health care. Health Affairs 25(3):681–693.
Patterson, K. B., S. E. Cohn, J. Uyanik, M. Hughes, M. Smurzynski, and J. J. Eron. 2009. Treatment responses in antiretroviral treatment-naive premenopausal and postmenopausal HIV-infected-women: An analysis from AIDS Clinical Trials Group Studies. Clinical Infectious Diseases 49(3):473–476.
Pav, M., H. Kovaru, A. Fiserova, E. Havrdova, and V. Lisa. 2008. Neurobiological aspects of depressive disorder and antidepressant treatment: Role of glia. Physiological Research 57(2):151–164.
Pearman, T. 2008. Psychosocial factors in lung cancer: Quality of life, economic impact, and survivorship implications. Journal of Psychosocial Oncology 26(1):69–80.
Pernerstorfer-Schoen, H., B. Jilma, A. Perschler, S. Wichlas, K. Schindler, A. Schindl, A. Rieger, O. F. Wagner, and P. Quehenberger. 2001. Sex differences in HAART-associated dyslipidaemia. AIDS 15(6):725–734.
Pernis, A. B. 2007. Estrogen and CD4(+) T cells. Current Opinion in Rheumatology 19(5):414–420.
Perron, B. E., B. Alexander-Eitzman, D. Watkins, R. J. Taylor, R. Baser, H. W. Neighbors, and J. S. Jackson. 2009. Ethnic differences in delays to treatment for substance use disorders: African Americans, black Caribbeans and non-Hispanic whites. Journal of Psychoactive Drugs 41(4):369–377.
Peters, W. A., III, P. Y. Liu, R. J. Barrett, II, R. J. Stock, B. J. Monk, J. S. Berek, L. Souhami, P. Grigsby, W. Gordon, Jr., and D. S. Alberts. 2000. Concurrent chemotherapy and pelvic radiation therapy compared with pelvic radiation therapy alone as adjuvant therapy after radical surgery in high-risk early-stage cancer of the cervix. Journal Clinical Oncology 18(8):1606–1613.
Petter, A., K. Heim, M. Guger, A. Ciresa-Ko Nig, N. Christensen, M. Sarcletti, U. Wieland, H. Pfister, R. Zangerle, and R. Hopfl. 2000. Specific serum IgG, IgM and IgA antibodies to human papillomavirus types 6, 11, 16, 18 and 31 virus-like particles in human immunodeficiency virus-seropositive women. Journal of General Virology 81(3):701–708.
Pietschmann, P., M. Rauner, W. Sipos, and K. Kerschan-Schindl. 2009. Osteoporosis: An age-related and gender-specific disease—A mini-review. Gerontology 55(1):3–12.
Pines, A. 2009. Lifestyle and diet in postmenopausal women. Climacteric 12(Suppl. 1):62–65.
Pisano, E. D., and M. J. Yaffe. 2005. Digital mammography. Radiology 234(2):353–362.
Pisano, E. D., C. Gatsonis, E. Hendrick, M. Yaffe, J. K. Baum, S. Acharyya, E. F. Conant, L. L. Fajardo, L. Bassett, C. D’Orsi, R. Jong, M. Rebner, and the Digital Mammographic Imaging Screening Trial Investigators Group. 2005. Diagnostic performance of digital versus film mammography for breast-cancer screening. New England Journal of Medicine 353(17):1773–1783.
Planchard, D., Y. Loriot, A. Goubar, F. Commo, and J-C. Soria. 2009. Differential expression of biomarkers in men and women. Seminars in Oncology 36(6):553–565.
Plassman, B. L., K. M. Langa, G. G. Fisher, S. G. Heeringa, D. R. Weir, M. B. Ofstedal, J. R. Burke, M. D. Hurd, G. G. Potter, W. L. Rodgers, D. C. Steffens, R. J. Willis, and R. B. Wallace. 2007. Prevalence of dementia in the United States: The aging, demographics, and memory study. Neuroepidemiology 29(1-2):125–132.
Plummer, F. A., J. N. Simonsen, D. W. Cameron, J. O. Ndinya-Achola, J. K. Kreiss, M. N. Gakinya, P. Waiyaki, M. Cheang, P. Piot, A. R. Ronald, and E. N. Ngugi. 1991. Cofactors in male-female sexual transmission of human immunodeficiency virus type 1. Journal of Infectious Diseases 163(2):233–239.
Poplack, S. P., T. D. Tosteson, C. A. Kogel, and H. M. Nagy. 2007. Digital breast tomosynthesis: Initial experience in 98 women with abnormal digital screening mammography. American Journal of Roentgeneology 189(3):616–623.
Prentice, R. L., C. A. Thomson, B. Caan, F. A. Hubbell, G. L. Anderson, S. A. Beresford, M. Pettinger, D. S. Lane, L. Lessin, S. Yasmeen, B. Singh, J. Khandekar, J. M. Shikany, S. Satterfield, and R. T. Chlebowski. 2007. Low-fat dietary pattern and cancer incidence in the Women’s Health Initiative dietary modification randomized controlled trial. Journal of the National Cancer Institute 99(20):1534–1543.
Press, M. F., M. C. Pike, V. R. Chazin, G. Hung, J. A. Udove, M. Markowicz, J. Danyluk, W. Godolphin, M. Sliwkowski, R. Akita, M. C. Paterson, and D. J. Slamon. 1993. HER-2/NEU expression in node-negative breast cancer: Direct tissue quantitation by computerized image analysis and association of overexpression with increased risk of recurrent disease. Cancer Research 53(20):4960–4970.
Press, M. F., R. S. Finn, D. Cameron, A. Di Leo, C. E. Geyer, I. E. Villalobos, A. Santiago, R. Guzman, A. Gasparyan, Y. Ma, K. Danenberg, A. M. Martin, L. Williams, C. Oliva, S. Stein, R. Gagnon, M. Arbushites, and M. T. Koehler. 2008. HER-2 gene amplification, HER-2 and epidermal growth factor receptor MRNA and protein expression, and lapatinib efficacy in women with metastatic breast cancer. Clinical Cancer Research 14(23):7861–7870.
Price, J. L., and P. D. Butler. 1970. The reduction of radiation and exposure time in mammography. British Journal of Radiology 43(508):251–255.
Quan, M. L., and D. McCready. 2009. The evolution of lymph node assessment in breast cancer. Journal of Surgical Oncology 99(4):194–198.
Quattrocchi, E., and H. Kourlas. 2004. Teriparatide: A review. Clinical Therapeutics 26(6): 841–854.
Quintana, F. J., and I. R. Cohen. 2001. Autoantibody patterns in diabetes-prone NOD mice and in standard C57BL/6 mice. Journal of Autoimmunity 17(3):191–197.
Quyyumi, A. A. 2006. Women and ischemic heart disease: Pathophysiologic implications from the Women’s Ischemia Syndrome Evaluation (WISE) study and future research steps. Journal of the American College of Cardiology 47(3 Suppl.1):S66–S71.
Ramjee, G., N. S. Morar, M. Alary, L. Mukenge-Tshibaka, B. Vuylsteke, V. Ettiegne-Traore, V. Chandeying, S. A. Karim, and L. Van Damme. 2000. Challenges in the conduct of vaginal microbicide effectiveness trials in the developing world. AIDS 14(16):2553–2557.
Ravdin, P., K. Cronin, N. Howlader, C. Berg, R. Chlebowski, E. Feuer, B. Edwards, and D. Berry. 2007. The decrease in breast-cancer incidence in 2003 in the United States. New England Journal of Medicine 356:1670–1674.
Read, J. S. 2000. Cesarean section delivery to prevent vertical transmission of human immunodeficiency virus type 1 associated risks and other considerations. Annals of the New York Academy of Sciences 918:115–121.
Rebbeck, T. R., N. D. Kauff, and S. M. Domchek. 2009. Meta-analysis of risk reduction estimates associated with risk-reducing salpingo-oophorectomy in BRCA1 or BRCA2 mutation carriers. Journal of the National Cancer Institute 101(2):80–87.
Reinhardt, P. P., B. Reinhardt, J. L. Lathey, and S. A. Spector. 1995. Human cord blood mononuclear cells are preferentially infected by non-syncytium-inducing, macrophage-tropic human immunodeficiency virus type 1 isolates. Journal of Clinical Microbiology 33(2):292–297.
Revicki, D. A., J. Siddique, L. Frank, J. Y. Chung, B. L. Green, J. Krupnick, M. Prasad, and J. Miranda. 2005. Cost-effectiveness of evidence-based pharmacotherapy or cognitive behavior therapy compared with community referral for major depression in predominantly low-income minority women. Archives of General Psychiatry 62(8):868–875.
Rich-Edwards, J. W., J. E. Manson, C. H. Hennekens, and J. E. Buring. 1995. The primary prevention of coronary heart disease in women. New England Journal of Medicine 332(26):1758–1766.
Ridker, P. M., J. E. Buring, N. R. Cook, and N. Rifai. 2003. C-reactive protein, the metabolic syndrome, and risk of incident cardiovascular events: An 8-year follow-up of 14 719 initially healthy American women. Circulation 107(3):391–397.
Ridker, P. M., N. R. Cook, I. M. Lee, D. Gordon, J. M. Gaziano, J. E. Manson, C. H. Hennekens, and J. E. Buring. 2005. A randomized trial of low-dose aspirin in the primary prevention of cardiovascular disease in women. New England Journal of Medicine 352(13):1293–1304.
Ridker, P. M., J. G. MacFadyen, F. A. Fonseca, J. Genest, A. M. Gotto, J. J. Kastelein, W. Koenig, P. Libby, A. J. Lorenzatti, B. G. Nordestgaard, J. Shepherd, J. T. Willerson, and R. J. Glynn. 2009. Number needed to treat with rosuvastatin to prevent first cardiovascular events and death among men and women with low low-density lipoprotein cholesterol and elevated high-sensitivity C-reactive protein: Justification for the use of statins in prevention: An intervention trial evaluating rosuvastatin (JUPITER). Circulation. Cardiovascular Quality and Outcomes 2(6):616–623.
Risch, H. A., G. R. Howe, M. Jain, J. D. Burch, E. J. Holowaty, and A. B. Miller. 1993. Are female smokers at higher risk for lung cancer than male smokers?: A case–control analysis by histologic type. American Journal of Epidemiology 138(5):281–293.
Roett, M. A., and P. Evans. 2009. Ovarian cancer: An overview. American Family Physician 80(6): 609–616.
Ronckers, C. M., C. A. Erdmann, and C. E. Land. 2005. Radiation and breast cancer: A review of current evidence. Breast Cancer Research 7(1):21–32.
Ronco, G., N. Segnan, P. Giorgi-Rossi, M. Zappa, G. P. Casadei, F. Carozzi, P. D. Palma, A. Del Mistro, S. Folicaldi, A. Gillio-Tos, G. Nardo, C. Naldoni, P. Schincaglia, M. Zorzi, M. Confortini, and J. Cuzick. 2006. Human papillomavirus testing and liquid-based cytology: Results at recruitment from the new technologies for cervical cancer randomized controlled trial. Journal of the National Cancer Institute 98(11):765–774.
Rooney, P. H., C. Telfer, M. C. McFadyen, W. T. Melvin, and G. I. Murray. 2004. The role of cytochrome P450 in cytotoxic bioactivation: Future therapeutic directions. Current Cancer Drug Targets 4:257–265.
Rose, P. G., B. N. Bundy, E. B. Watkins, J. T. Thigpen, G. Deppe, M. A. Maiman, D. L. Clarke-Pearson, and S. Insalaco. 1999. Concurrent cisplatin-based radiotherapy and chemotherapy for locally advanced cervical cancer. New England Journal of Medicine 340(15):1144–1153.
Rosenberg, S., L. Goodman, F. Osher, M. Swartz, S. Essock, M. Butterfield, N. Constantine, G. Wolford, and M. Salyers. 2001. Prevalence of HIV, hepatitis B, and hepatitis C in people with severe mental illness. Journal of Public Health 91:31–37.
Rosenfeld, A. G. 2006. State of the heart: Building science to improve women’s cardiovascular health. American Journal of Critical Care 15(6):556–566.
Rougier-Chapman, D., S. Key, and J. Ryan. 2001. Uterine artery embolization for the treatment of symptomatic fibroid disease. Applied Radiology (September):11–17.
Rowell, S., B. Newman, J. Boyd, and M. C. King. 1994. Inherited predisposition to breast and ovarian cancer. American Journal of Human Genetics 55(5):861–865.
Rubinstein, W. S. 2004. Hereditary breast cancer in Jews. Familial Cancer 3(3-4):249–257.
Rubtsov, A. V., K. Rubtsova, J. W. Kappler, and P. Marrack. 2010. Genetic and hormonal factors in female-biased autoimmunity. Autoimmunity Reviews 9(7):494–498.
Rush, A. J., M. Fava, S. R. Wisniewski, P. W. Lavori, M. H. Trivedi, H. A. Sackeim, M. E. Thase, A. A. Nierenberg, F. M. Quitkin, T. M. Kashner, D. J. Kupfer, J. F. Rosenbaum, J. Alpert, J. W. Stewart, P. J. McGrath, M. M. Biggs, K. Shores-Wilson, B. D. Lebowitz, L. Ritz, and G. Niederehe. 2004. Sequenced treatment alternatives to relieve depression (STAR*D): Rationale and design. Controlled Clinical Trials 25(1):119–142.
Sambrook, P., and C. Cooper. 2006. Osteoporosis. Lancet 367:2010–2018.
SAMHSA (Substance Abuse and Mental Health Services Administration). 2005. Cocaine Use: 2002 and 2003. The NSDUH Report http://www.oas.samhsa.gov/2k5/cocaine/cocaine.htm (accessed August 12, 2010).
———. 2008. Treatment Episode Data Set (TEDS) Highlights—2006: National Admissions to Substance Abuse Treatment Services, Drug and Alcohol Services Information System Series. Rock-ville, MD: Office of Applied Studies.
Sanne, I., H. Mommeja-Marin, J. Hinkle, J. A. Bartlett, M. M. Lederman, G. Maartens, C. Wakeford, A. Shaw, J. Quinn, R. G. Gish, and F. Rousseau. 2005. Severe hepatotoxicity associated with nevirapine use in HIV-infected subjects. Journal of Infectious Diseases 191(6):825–829.
Sardanelli, F., F. Podo, G. D’Agnolo, A. Verdecchia, M. Santaquilani, R. Musumeci, G. Trecate, S. Manoukian, S. Morassut, C. de Giacomi, M. Federico, L. Cortesi, S. Corcione, S. Cirillo, V. Marra, A. Cilotti, C. Di Maggio, A. Fausto, L. Preda, C. Zuiani, A. Contegiacomo, A. Orlacchio, M. Calabrese, L. Bonomo, E. Di Cesare, M. Tonutti, P. Panizza, and A. Del Maschio. 2007. Multicenter comparative multimodality surveillance of women at genetic-familial high risk for breast cancer (HIBCRIT study): Interim results. Radiology 242(3):698–715.
Sarkar, K., and F. W. Miller. 2004. Possible roles and determinants of microchimerism in autoimmune and other disorders. Autoimmunity Reviews 3(6):454–463.
Saslow, D., C. Boetes, W. Burke, S. Harms, M. O. Leach, C. D. Lehman, E. Morris, E. Pisano, M. Schnall, S. Sener, R. A. Smith, E. Warner, M. Yaffe, K. S. Andrews, and C. A. Russell. 2007. American Cancer Society guidelines for breast screening with MRI as an adjunct to mammog-raphy. CA: A Cancer Journal for Clinicians 57(2):75–89.
Sattler, F. R., D. Qian, S. Louie, D. Johnson, W. Briggs, V. DeQuattro, and M. P. Dube. 2001. Elevated blood pressure in subjects with lipodystrophy. AIDS 15(15):2001–2010.
Scarpellini, F., M. Sbracia, S. Lecchini, and L. Scarpellini. 2002. Anti-angiogenesis treatment with thalidomide in endometriosis: A pilot study. Fertility and Sterility 78(Suppl. 1):S87.
Schapira, D. V., R. A. Clark, P. A. Wolff, A. R. Jarrett, N. B. Kumar, and N. M. Aziz. 1994. Visceral obesity and breast cancer risk. Cancer 74(2):632–639.
Scheidt-Nave, C., E. Barrett-Connor, D. L. Wingard, B. A. Cohn, and S. L. Edelstein. 1991. Sex differences in fasting glycemia as a risk factor for ischemic heart disease death. American Journal of Epidemiology 133(6):565–576.
Schiffman, M., and P. E. Castle. 2005. The promise of global cervical-cancer prevention. New England Journal of Medicine 353(20):2101–2104.
Schlecht, N. F., S. Kulaga, J. Robitaille, S. Ferreira, M. Santos, R. A. Miyamura, E. Duarte-Franco, T. E. Rohan, A. Ferenczy, L. L. Villa, and E. L. Franco. 2001. Persistent human papillomavirus infection as a predictor of cervical intraepithelial neoplasia. Journal of the American Medical Association 286(24):3106–3114.
Schmidt, P. J. 2005. Mood, depression, and reproductive hormones in the menopausal transition. American Journal of Medicine 118 (Suppl. 12B):54–58.
Schrag, D., K. M. Kuntz, J. E. Garber, and J. C. Weeks. 2000. Life expectancy gains from cancer prevention strategies for women with breast cancer and BRCA1 or BRCA2 mutations. Journal of the American Medical Association 283(5):617–624.
Schrenk, P., R. Rieger, A. Shamiyeh, and W. Wayand. 2000. Morbidity following sentinel lymph node biopsy versus axillary lymph node dissection for patients with breast carcinoma. Cancer 88(3):608–614.
Schubert, E. L., M. K. Lee, H. C. Mefford, R. H. Argonza, J. E. Morrow, J. Hull, J. L. Dann, and M. C. King. 1997. BRCA2 in American families with four or more cases of breast or ovarian cancer: Recurrent and novel mutations, variable expression, penetrance, and the possibility of families whose cancer is not attributable to BRCA1 or BRCA2. American Journal of Human Genetics 60(5):1031–1040.
Schulman, K. A., J. A. Berlin, W. Harless, J. F. Kerner, S. Sistrunk, B. J. Gersh, R. Dube, C. K. Taleghani, J. E. Burke, S. Williams, J. M. Eisenberg, and J. J. Escarce. 1999. The effect of race and sex on physicians’ recommendations for cardiac catheterization. New England Journal of Medicine 340(8):618–626.
Schulz, M., P. H. Lahmann, E. Riboli, and H. Boeing. 2004. Dietary determinants of epithelial ovarian cancer: A review of the epidemiologic literature. Nutrition and Cancer 50(2):120–140.
Seigneurin, A., M. Colonna, L. Remontet, P. Delafosse, and R. Ecochard. 2008. Artefact-free trends in breast cancer incidence over two decades in a whole French Département. Breast Journal 17(6):580–586.
Shapiro, S. 1988. Periodic Screening for Breast Cancer: The Health Insurance Plan Project and Its Sequelae, 1963–1986. Baltimore, MD: Johns Hopkins University Press.
Shapiro, S., W. Venet, and P. Strax. 1988. Current results of the breast cancer screening randomized trial: The Health Insurance Plan (HIP) of Greater New York Study. In Screening for Breast Cancer, edited by N. E. Day and A. B. Miller. Lewiston, NY: Hans Huber Publishers. Pp. 3–15.
Sharftsein, S. 1999. Healthcare policy and opportunities for psychotherapy and psychoanalysis. In The Challenage to Psychoanalysis and Psychotherapy: Soluations for the Future, edited by S. de Schill and S. Lebovich. Philidelphia, PA: Jessica Kingsley Publishers.
Sharpe, N. 2002. Clinical trials and the real world: Selection bias and generalisability of trial results. Cardiovascular Drugs and Therapy 16(1):75–77.
Shavers, V. L., and M. L. Brown. 2002. Racial and ethnic disparities in the receipt of cancer treatment. Journal of the National Cancer Institute 94(5):334–357.
Shaw, L. J., R. Bugiardini, and C. N. Merz. 2009. Women and ischemic heart disease: Evolving knowledge. Journal of the American College of Cardiology 54(17):1561–1575.
Shepherd, J., S. M. Cobbe, I. Ford, C. G. Isles, A. R. Lorimer, P. W. MacFarlane, J. H. McKillop, and C. J. Packard. 1995. Prevention of coronary heart disease with pravastatin in men with hypercholesterolemia. West of Scotland Coronary Prevention Study Group. New England Journal of Medicine 333(20):1301–1307.
Sherman, M. E., M. H. Schiffman, A. T. Lorincz, R. Herrero, M. L. Hutchinson, C. Bratti, D. Zahniser, J. Morales, A. Hildesheim, K. Helgesen, D. Kelly, M. Alfaro, F. Mena, I. Balmaceda, L. Mango, and M. Greenberg. 1997. Cervical specimens collected in liquid buffer are suitable for both cytologic screening and ancillary human papillomavirus testing. Cancer 81(2):89–97.
Sherman, M. E., M. Mendoza, K. R. Lee, R. Ashfaq, G. G. Birdsong, M. E. Corkill, K. M. McIntosh, S. L. Inhorn, D. J. Zahniser, G. Baber, C. Barber, and M. H. Stoler. 1998. Performance of liquid-based, thin-layer cervical cytology: Correlation with reference diagnoses and human papillomavirus testing. Modern Pathology 11(9):837–843.
Sherman, M. E., S. S. Wang, J. Carreon, and S. S. Devesa. 2005. Mortality trends for cervical squamous and adenocarcinoma in the United States. Relation to incidence and survival. Cancer 103(6):1258–1264.
Sherwin, B. B. 2003. Estrogen and cognitive functioning in women. Endocrine Reviews 24(2): 133–151.
Shriver, S. P., H. A. Bourdeau, C. T. Gubish, D. L. Tirpak, A. L. Davis, J. D. Luketich, and J. M. Siegfried. 2000. Sex-specific expression of gastrin-releasing peptide receptor: Relationship to smoking history and risk of lung cancer. Journal of the National Cancer Institute 92(1):24–33.
Shtern, F. 1992. Digital mammography and related technologies: A perspective from the National Cancer Institute. Radiology 183(3):629–630.
Shumaker, S. A., C. Legault, L. Kuller, S. R. Rapp, L. Thal, D. S. Lane, H. Fillit, M. L. Stefanick, S. L. Hendrix, C. E. Lewis, K. Masaki, and L. H. Coker. 2004. Conjugated equine estrogens and incidence of probable dementia and mild cognitive impairment in postmenopausal women: Women’s Health Initiative Memory Study. Journal of the American Medical Association 291(24): 2947–2958.
Sichel, D., and J. W. Drischoll. 1999. Women’s Moods, Women’s Minds: What Every Woman Must Know About Hormones, the Brain and Emotional Health. New York: Harper Collins Publishers, Inc.
Siebers, A. G., P. J. Klinkhamer, J. M. Grefte, L. F. Massuger, J. E. Vedder, A. Beijers-Broos, J. Bulten, and M. Arbyn. 2009. Comparison of liquid-based cytology with conventional cytology for detection of cervical cancer precursors: A randomized controlled trial. Journal of the American Medical Association 302(16):1757–1764.
Silva, W. A., and M. M. Karram. 2005. Scientific basis for use of grafts during vaginal reconstructive procedures. Current Opinions in Obstetrics and Gynecology 17(5):519–529.
Simard, J. F., and K. H. Costenbader. 2007. What can epidemiology tell us about systemic lupus erythematosus? International Journal of Clinical Practice 61(7):1170–1180.
Simon, G. E., M. VonKorff, C. Rutter, and E. Wagner. 2000. Randomised trial of monitoring, feedback, and management of care by telephone to improve treatment of depression in primary care. British Medical Journal 320(7234):550–554.
Simon, G. E., E. J. Ludman, S. Tutty, B. Operskalski, and M. VonKorff. 2004. Telephone psychotherapy and telephone care management for primary care patients starting antidepressant treatment: A randomized controlled trial. Journal of the American Medical Association 292(8):935–942.
Simon, G. R., M. Extermann, A. Chiappori, C. C. Williams, M. Begum, R. Kapoor, E. B. Haura, R. Ismail-Khan, M. J. Schell, S. J. Antonia, and G. Bepler. 2008. Phase 2 trial of docetaxel and gefitinib in the first-line treatment of patients with advanced nonsmall-cell lung cancer (NSCLC) who are 70 years of age or older. Cancer 112(9):2021–2029.
Sirey, J. A., M. L. Bruce, G. S. Alexopoulos, D. A. Perlick, P. Raue, S. J. Friedman, and B. S. Meyers. 2001. Perceived stigma as a predictor of treatment discontinuation in young and older outpatients with depression. American Journal of Psychiatry 158(3):479–481.
Siris, E. S., P. D. Miller, E. Barrett-Connor, K. G. Faulkner, L. E. Wehren, T. A. Abbott, M. L. Berger, A. C. Santora, and L. M. Sherwood. 2001. Identification and fracture outcomes of undiagnosed low bone mineral density in postmenopausal women: Results from the National Osteoporosis Risk Assessment. Journal of the American Medical Association 286(22):2815–2822.
Siris, E. S., Y. T. Chen, T. A. Abbott, E. Barrett-Connor, P. D. Miller, L. E. Wehren, and M. L. Berger. 2004. Bone mineral density thresholds for pharmacological intervention to prevent fractures. Archives of Internal Medicine 164(10):1108–1112.
Skaane, P., S. Hofvind, and A. Skjennald. 2007. Randomized trial of screen-film versus full-field digital mammography with soft-copy reading in population-based screening program: Follow-up and final results of Oslo II Study. Radiology 244(3):708–717.
Slamon, D., and M. Pegram. 2001. Rationale for trastuzumab (herceptin) in adjuvant breast cancer trials. Seminars in Oncology 28(Suppl. 3):13–19.
Slamon, D. J., G. M. Clark, S. G. Wong, W. J. Levin, A. Ullrich, and W. L. McGuire. 1987. Human breast cancer: Correlation of relapse and survival with amplification of the HER-2/NEU oncogene. Science 235(4785):177–182.
Slamon, D. J., W. Godolphin, L. A. Jones, J. A. Holt, S. G. Wong, D. E. Keith, W. J. Levin, S. G. Stuart, J. Udove, A. Ullrich, and M. F. Press. 1989. Studies of the HER-2/NEU proto-oncogene in human breast and ovarian cancer. Science 244(4905):707–712.
Slattery, D. A., A. L. Hudson, and D. J. Nutt. 2004. Invited review: The evolution of antidepressant mechanisms. Fundamental and Clinical Pharmacology 18(1):1–21.
Smith, D. K., D. L. Warren, D. Vlahov, P. Schuman, M. D. Stein, B. L. Greenberg, and S. D. Holmberg. 1997. Design and baseline participant characteristics of the Human Immunodeficiency Virus Epidemiology Research (HER) Study: A prospective cohort study of human immunodeficiency virus infection in US women. American Journal of Epidemiology 146(6):459–469.
Smith, R. A., S. W. Duffy, R. Gabe, L. Tabar, A. M. Yen, and T. H. Chen. 2004. The randomized trials of breast cancer screening: What have we learned? Radiologic Clinics of North America 42(5):793–806.
Smith, R. A., V. Cokkinides, and O. W. Brawley. 2009. Cancer screening in the United States, 2009: A review of current American Cancer Society guidelines and issues in cancer screening. CA: A Cancer Journal for Clinicians 59(1):27–41.
Smith-McCune, K. K., S. Shiboski, M. Z. Chirenje, T. Magure, J. Tuveson, Y. Ma, M. Da Costa, A. B. Moscicki, J. M. Palefsky, R. Makunike-Mutasa, T. Chipato, A. van der Straten, and G. F. Sawaya. 2010. Type-specific cervico-vaginal human papillomavirus infection increases risk of HIV acquisition independent of other sexually transmitted infections. PLoS One 5(4):e10094.
Sobieszczyk, M. E., D. R. Hoover, K. Anastos, K. Mulligan, T. Tan, Q. Shi, W. Gao, C. Hyman, M. H. Cohen, S. R. Cole, M. W. Plankey, A. M. Levine, and J. Justman. 2008. Prevalence and predictors of metabolic syndrome among HIV-infected and HIV-uninfected women in the Women’s Interagency HIV Study. Journal of Acquired Immune Deficiency Syndromes 48(3):272–280.
Solomon, D. H., M. A. Brookhart, T. K. Gandhi, A. Karson, S. Gharib, E. J. Orav, S. Shaykevich, A. Licari, D. Cabral, and D. W. Bates. 2004. Adherence with osteoporosis practice guidelines: A multilevel analysis of patient, physician, and practice setting characteristics. American Journal of Medicine 117(12):919–924.
Solomon, D. H., J. S. Finkelstein, P. S. Wang, and J. Avorn. 2005. Statin lipid-lowering drugs and bone mineral density. Pharmacoepidemiology and Drug Safety 14(4):219–226.
Sparano, J. A., L. J. Goldstein, B. H. Childs, S. Shak, D. Brassard, S. Badve, F. L. Baehner, R. Bugarini, S. Rowley, E. Perez, L. N. Shulman, S. Martino, N. E. Davidson, G. W. Sledge, and R. Gray. 2009. Relationship between topoisomerase 2A RNA expression and recurrence after adjuvant chemotherapy for breast cancer. Clinical Cancer Research 15(24):7693–7700.
Spitz, M. R., Q. Wei, Q. Dong, C. I. Amos, and X. Wu. 2003. Genetic susceptibility to lung cancer: The role of DNA damage and repair. Cancer Epidemiology, Biomarkers and Prevention 12(8):689–698.
Ssali, F., W. Stohr, P. Munderi, A. Reid, A. S. Walker, D. M. Gibb, P. Mugyenyi, C. Kityo, H. Grosskurth, J. Hakim, H. Byakwaga, E. Katabira, J. H. Darbyshire, and C. F. Gilks. 2006. Prevalence, incidence and predictors of severe anaemia with zidovudine-containing regimens in African adults with HIV infection within the DART Trial. Antiviral Therapy 11(6):741–749.
Stabile, L. P., A. L. Davis, C. T. Gubish, T. M. Hopkins, J. D. Luketich, N. Christie, S. Finkelstein, and J. M. Siegfried. 2002. Human non-small cell lung tumors and cells derived from normal lung express both estrogen receptor alpha and beta and show biological responses to estrogen. Cancer Research 62(7):2141–2150.
Stadtmauer, E. A., A. O’Neill, L. J. Goldstein, P. A. Crilley, K. F. Mangan, J. N. Ingle, I. Brodsky, S. Martino, H. M. Lazarus, J. K. Erban, C. Sickles, and J. H. Glick. 2000. Conventional-dose chemotherapy compared with high-dose chemotherapy plus autologous hematopoietic stem-cell transplantation for metastatic breast cancer. Philadelphia bone marrow transplant group. New England Journal of Medicine 342(15):1069–1076.
Stefanick, M. L. 2006. Risk-benefit profiles of raloxifene for women. New England Journal of Medicine 355(2):190–192.
Steinhubl, S. R., W. A. Tan, J. M. Foody, and E. J. Topol. 1999. Incidence and clinical course of thrombotic thrombocytopenic purpura due to ticlopidine following coronary stenting. Journal of the American Medical Association 281(9):806–810.
Stewart, D., M. Gossop, J. Marsden, T. Kidd, and S. Treacy. 2003. Similarities in outcomes for men and women after drug misuse treatment: Results from the National Treatment Outcome Research Study (NTORS). Drug and Alcohol Review 22(1):35–41.
Stone, G. W., C. L. Grines, D. A. Cox, E. Garcia, J. E. Tcheng, J. J. Griffin, G. Guagliumi, T. Stuckey, M. Turco, J. D. Carroll, B. D. Rutherford, and A. J. Lansky. 2002. Comparison of angioplasty with stenting, with or without abciximab, in acute myocardial infarction. New England Journal of Medicine 346(13):957–966.
Stoskopf, C., Y. Kim, and S. Glover. 2001. Dual diagnosis: HIV and mental illness, a population-based study. Community Mental Health Journal 37(6):469–479.
Sturmer, M., H. W. Doerr, and L. Gurtler. 2009. Human immunodeficiency virus: 25 years of diagnostic and therapeutic strategies and their impact on hepatitis B and C virus. Medical Microbiology Immunology 198(3):147–155.
Sundar, S. S., R. J. Gornall, and S. T. Kehoe. 2005. Advances in the management of cervical cancer. Menopause International 11(3):91–95.
Suzich, J. A., S. J. Ghim, F. J. Palmer-Hill, W. I. White, J. K. Tamura, J. A. Bell, J. A. Newsome, A. B. Jenson, and R. Schlegel. 1995. Systemic immunization with papillomavirus L1 protein completely prevents the development of viral mucosal papillomas. Proceedings of the National Academy of Sciences of the United States of America 92(25):11553–11557.
Suzuki, R., N. Orsini, L. Mignone, S. Saji, and A. Wolk. 2008. Alcohol intake and risk of breast cancer defined by estrogen and progesterone receptor status—A meta-analysis of epidemiological studies. International Journal of Cancer 122(8):1832–1841.
Swica, Y. 2007. The transdermal patch and the vaginal ring: Two novel methods of combined hormonal contraception. Obstetrics and Gynecology Clinics of North America 34(1): viii, 31–42.
Szodoray, P., B. Nakken, J. Gaal, R. Jonsson, A. Szegedi, E. Zold, G. Szegedi, J. G. Brun, R. Gesztelyi, M. Zeher, and E. Bodolay. 2008. The complex role of vitamin D in autoimmune diseases. Scandinavian Journal of Immunology 68(3):261–269.
Tabar, L., G. Fagerberg, H. H. Chen, S. W. Duffy, C. R. Smart, A. Gad, and R. A. Smith. 1995. Efficacy of breast cancer screening by age. New results from the swedish two-county trial. Cancer 75(10):2507–2517.
Takeuchi, D. T., M. Alegria, J. S. Jackson, and D. R. Williams. 2007. Immigration and mental health: Diverse findings in Asian, black, and Latino populations. American Journal of Public Health 97(1):11–12.
Tamres, L. K., D. Janicki, and V. S. Helgeson. 2002. Sex differences in coping behavior: A meta-analytic review and an examination of relative coping. Personality and Social Psychology Review 6(1):2–30.
Tang, B. M., G. D. Eslick, C. Nowson, C. Smith, and A. Bensoussan. 2007. Use of calcium or calcium in combination with vitamin D supplementation to prevent fractures and bone loss in people aged 50 years and older: A meta-analysis. Lancet 370(9588):657–666.
Tanis, K. Q., and R. S. Duman. 2007. Intracellular signaling pathways pave roads to recovery for mood disorders. Annals of Medicine 39(7):531–544.
Tao, X., W. Hu, P. T. Ramirez, and J. J. Kavanagh. 2008. Chemotherapy for recurrent and metastatic cervical cancer. Gynecologic Oncology 110(3 Suppl. 2):S67–S71.
Tay, S-K., and K-J. Tay. 2004. Passive cigarette smoking is a risk factor in cervical neoplasia. Gynecologic Oncology 93(1):116–120.
Taylor, A. J., L. C. Gary, T. Arora, D. J. Becker, J. R. Curtis, M. L. Kilgore, M. A. Morrisey, K. G. Saag, R. Matthews, H. Yun, W. Smith, and E. Delzell. 2010. Clinical and demographic factors associated with fractures among older Americans. Osteoporosis International Jun 18. [Epub ahead of print].
Taylor, V. M., Y. Yasui, N. Burke, T. Nguyen, E. Acorda, H. Thai, P. Qu, and J. C. Jackson. 2004. Pap testing adherence among Vietnamese American women. Cancer Epidemiology, Biomarkers and Prevention 13(4):613–619.
Tetrault, J. M., R. A. Desai, W. C. Becker, D. A. Fiellin, J. Concato, and L. E. Sullivan. 2008. Gender and non-medical use of prescription opioids: Results from a national US survey. Addiction 103(2):258–268.
Thomas, L., L. A. Doyle, and M. J. Edelman. 2005. Lung cancer in women: Emerging differences in epidemiology, biology, and therapy. Chest 128(1):370–381.
Thun, M. J., S. J. Henley, and E. E. Calle. 2002. Tobacco use and cancer: An epidemiologic perspective for geneticists. Oncogene 21(48 Review Issue 6):7307–7325.
Thun, M. J., L. M. Hannan, L. L. Adams-Campbell, P. Boffetta, J. E. Buring, D. Feskanich, W. D. Flanders, H. J. Sun, K. Katanoda, L. N. Kolonel, I. M. Lee, T. Marugame, J. R. Palmer, E. Riboli, T. Sobue, E. Avila-Tang, L. R. Wilkens, and J. M. Samet. 2008. Lung cancer occurrence in never-smokers: An analysis of 13 cohorts and 22 cancer registry studies. PLoS Medicine 5(9):1357–1371.
Tien, P. C., S. R. Cole, C. M. Williams, R. Li, J. E. Justman, M. H. Cohen, M. Young, N. Rubin, M. Augenbraun, and C. Grunfeld. 2003. Incidence of lipoatrophy and lipohypertrophy in the Women’s Interagency HIV Study. Journal of Acquired Immune Deficiency Syndromes 34(5):461–466.
Tien, P. C., Y. Barron, J. E. Justman, C. Hyman, M. H. Cohen, M. Young, A. Kovacs, and S. R. Cole. 2007. Antiretroviral therapies associated with lipoatrophy in HIV-infected women. AIDS Patient Care and STDs 21(5):297–305.
Tillman, G. F., S. G. Orel, M. D. Schnall, D. J. Schultz, J. E. Tan, and L. J. Solin. 2002. Effect of breast magnetic resonance imaging on the clinical management of women with early-stage breast carcinoma. Journal Clinical Oncology 20(16):3413–3423.
Tolaymat, L. L., and A. M. Kaunitz. 2007. Long-acting contraceptives in adolescents. Current Opinion in Obstetrics and Gynecology 19(5):453–460.
Tosteson, A. N., N. K. Stout, D. G. Fryback, S. Acharyya, B. A. Herman, L. G. Hannah, and E. D. Pisano. 2008. Cost-effectiveness of digital mammography breast cancer screening. Annals of Internal Medicine 148(1):1–10.
Towfighi, A., J. L. Saver, R. Engelhardt, and B. Ovbiagele. 2007. A midlife stroke surge among women in the United States. Neurology 69(20):1898–1904.
Towfighi, A., L. Zheng, and B. Ovbiagele. 2009. Sex-specific trends in midlife coronary heart disease risk and prevalence. Archives of Internal Medicine 169(19):1762–1766.
Trimble, C. L., J. M. Genkinger, A. E. Burke, S. C. Hoffman, K. J. Helzlsouer, M. Diener-West, G. W. Comstock, and A. J. Alberg. 2005. Active and passive cigarette smoking and the risk of cervical neoplasia. Obstetrics and Gynecology 105(1):174–181.
Trussell, J., and L. L. Wynn. 2008. Reducing unintended pregnancy in the United States. Contraception 77(1):1–5.
Tsao, A. S., D. Liu, J. J. Lee, M. Spitz, and W. K. Hong. 2006. Smoking affects treatment outcome in patients with advanced nonsmall cell lung cancer. Cancer 106(11):2428–2436.
Tumbarello, M., R. Rabagliati, K. De Gaetano Donati, S. Bertagnolio, E. Tamburrini, E. Tacconelli, and R. Cauda. 2003. Older HIV-positive patients in the era of highly active antiretroviral therapy: Changing of a scenario. AIDS 17(1):128–131.
Tyrer, J., S. W. Duffy, and J. Cuzick. 2004. A breast cancer prediction model incorporating familial and personal risk factors. Statistics in Medicine 23(7):1111–1130.
Uetrecht, J. 2009. Immune-mediated adverse drug reactions. Chemical Research in Toxicology 22(1): 24–34.
Uhlenhuth, E. H., and E. S. Paykel. 1973. Symptom intensity and life events. Archives of General Psychiatry 28(4):473–477.
Unutzer, J., W. Katon, C. M. Callahan, J. W. Williams, Jr., E. Hunkeler, L. Harpole, M. Hoffing, R. D. Della Penna, P. H. Noel, E. H. B. Lin, P. A. Arean, M. T. Hegel, L. Tang, T. R. Belin, S. Oishi, and C. Langston. 2002. Collaborative care management of late-life depression in the primary care setting: A randomized controlled trial. Journal of the American Medical Association 288(22):2836–2845.
US Preventive Services Task Force. 2002. Screening for breast cancer: Recommendations and rationale. Annals of Internal Medicine 137(5 Pt. 1):344–346.
———. 2009. Screening for breast cancer: US Preventive Services Task Force recommendation statement. Annals of Internal Medicine 151(10):716–726.
Vaccarino, V., L. Parsons, N. R. Every, H. V. Barron, and H. M. Krumholz. 1999. Sex-based differences in early mortality after myocardial infarction. National registry of myocardial infarction 2 participants. New England Journal of Medicine 341(4):217–225.
Vaccarino, V., S. S. Rathore, N. K. Wenger, P. D. Frederick, J. L. Abramson, H. V. Barron, A. Manhapra, S. Mallik, and H. M. Krumholz. 2005. Sex and racial differences in the management of acute myocardial infarction, 1994 through 2002. New England Journal of Medicine 353(7):671–682.
Vainio, H., and F. Bianchini. 2002a. IARC handbooks of cancer prevention. In Weight Control and Physical Activity. Vol. 6. Lyon, France: International Agency for Research on Cancer Press.
———. 2002b. Breast Cancer Screening, IARC Handbooks of Cancer Prevention, Vol. 7. Lyon, France: International Agency for Research on Cancer Press.
Vallbohmer, D., J. Brabender, D. Y. Yang, K. Danenberg, P. M. Schneider, R. Metzger, A. H. Holscher, and P. V. Danenberg. 2006. Sex differences in the predictive power of the molecular prognostic factor HER2/NEU in patients with non-small-cell lung cancer. Clinical Lung Cancer 7(5):332–337.
van Benthem, B. H., P. Vernazza, R. A. Coutinho, and M. Prins. 2002. The impact of pregnancy and menopause on CD4 lymphocyte counts in HIV-infected women. AIDS 16(6):919–924.
van’t Veer, L. J., S. Paik, and D. F. Hayes. 2005. Gene expression profiling of breast cancer: A new tumor marker. Journal Clinical Oncology23(8):1631–1635.
Vandamme, A. M. 2008. Cellulose sulfate for prevention of HIV infection. New England Journal of Medicine 359(19):2066–2067.
Vannucci, S. J., L. B. Willing, S. Goto, N. J. Alkayed, R. M. Brucklacher, T. L. Wood, J. Towfighi, P. D. Hurn, and I. A. Simpson. 2001. Experimental stroke in the female diabetic, DB/DB, mouse. Journal of Cerebral Blood Flow and Metabolism 21(1):52–60.
Varghese, B., J. E. Maher, T. A. Petermen, B. M. Branson, and R. W. Steketee. 2002. Reducing the risk of sexual HIV transmission: Quantifying the per-act risk for HIV on the basis of choice of partner, sex act, and condom use.Sexually Transmitted Diseases 29(1):38–43.
Vassilakos, P., F. de Marval, M. Munoz, G. Broquet, and A. Campana. 1998. Human papillomavirus (HPV) DNA assay as an adjunct to liquid-based pap test in the diagnostic triage of women with an abnormal pap smear. International Journal of Gynaecology and Obstetrics 61(1):45–50.
Vega, W. A., B. Kolody, S. Aguilar-Gaxiola, E. Alderete, R. Catalano, and J. Caraveo-Anduaga. 1998. Lifetime prevalence of DSM-III-R psychiatric disorders among urban and rural Mexican Americans in California. Archives of General Psychiatry 55(9):771–778.
Vera, M., C. Perez-Pedrogo, S. E. Huertas, M. L. Reyes-Rabanillo, D. Juarbe, A. Huertas, M. L. Reyes-Rodriguez, and W. Chaplin. 2010. Collaborative care for depressed patients with chronic medical conditions: A randomized trial in Puerto Rico. Psychiatric Services 61(2):144–150.
Veronesi, U., P. Maisonneuve, N. Rotmensz, B. Bonanni, P. Boyle, G. Viale, A. Costa, V. Sacchini, R. Travaglini, G. D’Aiuto, P. Oliviero, F. Lovison, G. Gucciardo, M. R. del Turco, M. G. Muraca, M. A. Pizzichetta, S. Conforti, and A. Decensi. 2007. Tamoxifen for the prevention of breast cancer: Late results of the Italian randomized tamoxifen prevention trial among women with hysterectomy. Journal of the National Cancer Institute 99(9):727–737.
Viard, J. P., A. Mocroft, A. Chiesi, O. Kirk, B. Roge, G. Panos, N. Vetter, J. N. Bruun, M. Johnson, and J. D. Lundgren. 2001. Influence of age on CD4 cell recovery in human immunodeficiency virus-infected patients receiving highly active antiretroviral therapy: Evidence from the Euro-SIDA study. Journal of Infectious Diseases 183(8):1290–1294.
Vigna-Taglianti, F., S. Vadrucci, F. Faggiano, G. Burkhart, R. Siliquini, and M. R. Galanti. 2009. Is universal prevention against youths’ substance misuse really universal? Gender-specific effects in the EU-Dap school-based prevention trial. Journal of Epidemiology and Community Health 63(9):722–728.
Vinnicombe, S., S. M. Pinto Pereira, V. A. McCormack, S. Shiel, N. Perry, and I. M. dos Santos Silva. 2009. Full-field digital versus screen-film mammography: Comparison within the UK breast screening program and systematic review of published data. Radiology 251(2):347–358.
Visbal, A. L., B. A. Williams, F. C. Nichols, 3rd, R. S. Marks, J. R. Jett, M. C. Aubry, E. S. Edell, J. A. Wampfler, J. R. Molina, and P. Yang. 2004. Gender differences in non-small-cell lung cancer survival: An analysis of 4,618 patients diagnosed between 1997 and 2002. Annals of Thoracic Surgery 78(1):209–215.
Vogel, V. G., J. P. Costantino, D. L. Wickerham, W. M. Cronin, R. S. Cecchini, J. N. Atkins, T. B. Bevers, L. Fehrenbacher, E. R. Pajon, Jr., J. L. Wade, III, A. Robidoux, R. G. Margolese, J. James, S. M. Lippman, C. D. Runowicz, P. A. Ganz, S. E. Reis, W. McCaskill-Stevens, L. G. Ford, V. C. Jordan, N. Wolmark, and the National Surgical Adjuvant Breast and Bowel Project (NSABP). 2006. Effects of tamoxifen vs raloxifene on the risk of developing invasive breast cancer and other disease outcomes: The NSABP study of tamoxifen and raloxifene (STAR) P-2 trial. Journal of the American Medical Association 295(23):2727–2741.
Walboomers, J. M., M. V. Jacobs, M. M. Manos, F. X. Bosch, J. A. Kummer, K. V. Shah, P. J. Snijders, J. Peto, C. J. Meijer, and N. Muñoz. 1999. Human papillomavirus is a necessary cause of invasive cervical cancer worldwide. Journal of Pathology 189(1):12–19.
Walkup, J., S. Crystal, and U. Sambamoorthi. 1999. Schizophrenia and major affective disorder among Medicaid recipients with HIV/AIDS in New Jersey. American Journal of Public Health 89(7):1101–1103.
Wallenius, M., J. F. Skomsvoll, W. Koldingsnes, E. Rodevand, K. Mikkelsen, C. Kaufmann, and T. K. Kvien. 2009. Comparison of work disability and health-related quality of life between males and females with rheumatoid arthritis below the age of 45 years. Scandinavian Journal of Rheumatology 38(3):178–183.
Ward, R. K., and M. A. Zamorski. 2002. Benefits and risks of psychiatric medications during pregnancy. American Family Physician 66(4):629–636.
Warner, E., D. B. Plewes, K. A. Hill, P. A. Causer, J. T. Zubovits, R. A. Jong, M. R. Cutrara, G. DeBoer, M. J. Yaffe, S. J. Messner, W. S. Meschino, C. A. Piron, and S. A. Narod. 2004. Surveillance of BRCA1 and BRCA2 mutation carriers with magnetic resonance imaging, ultrasound, mammography, and clinical breast examination. Journal of the American Medical Association 292(11):1317–1325.
Warner-Schmidt, J. L., and R. S. Duman. 2008. VEGF as a potential target for therapeutic intervention in depression. Current Opinion in Pharmacology 8(1):14–19.
Warren, M. 2000. The expanding role of the female condom. AIDS Analysis Africa 10(5):8–10.
Wasserheit, J. N. 1992. Epidemiological synergy. Interrelationships between human immunodeficiency virus infection and other sexually transmitted diseases. Sexually Transmitted Disease 19(2):61–77.
Wassertheil-Smoller, S., S. Hendrix, M. Limacher, G. Heiss, C. Kooperberg, A. Baird, T. Kotchen, J. D. Curb, H. Black, J. E. Rossouw, A. Aragaki, M. Safford, E. Stein, S. Laowattana, and W. J. Mysiw. 2003. Effect of estrogen plus progestin on stroke in postmenopausal women: The Women’s Health Initiative: A randomized trial. Journal of the American Medical Association 289(20):2673–2684.
Watts, N. B., and D. L. Diab. 2010. Long-term use of bisphosphonates in osteoporosis. Journal of Clinical Endocrinology and Metabolism 95(4):1555–1565.
Way, C. M. 2007. Safety of newer antidepressants in pregnancy. Pharmacotherapy 27(4):546–552.
Wei, Q., L. Cheng, C. I. Amos, L. E. Wang, Z. Guo, W. K. Hong, and M. R. Spitz. 2000. Repair of tobacco carcinogen-induced DNA adducts and lung cancer risk: A molecular epidemiologic study. Journal of the National Cancer Institute 92(21):1764–1772.
Weiss, E. L., J. G. Longhurst, and C. M. Mazure. 1999. Childhood sexual abuse as a risk factor for depression in women: Psychosocial and neurobiologica correlates. American Journal of Psychiatry 156:816–828.
Weiss, R. B. 1999. The randomized trials of dose-intensive therapy for breast cancer: What do they mean for patient care and where do we go from here? Oncologist 4(6):450–458.
Weissman, A. M., B. T. Levy, A. J. Hartz, S. Bentler, M. Donohue, V. L. Ellingrod, and K. L. Wisner. 2004. Pooled analysis of antidepressant levels in lactating mothers, breast milk, and nursing infants. American Journal of Psychiatry 161(6):1066–1078.
Welch, H. G., and J. Mogielnicki. 2002. Presumed benefit: Lessons from the American experience with marrow transplantation for breast cancer. British Medical Journal 324(7345):1088–1092.
Werner-Wasik, M., C. Scott, M. L. Graham, C. Smith, R. W. Byhardt, M. Roach, 3rd, and E. J. Andras. 1999. Interfraction interval does not affect survival of patients with non-small cell lung cancer treated with chemotherapy and/or hyperfractionated radiotherapy: A multivariate analysis of 1076 RTOG patients. International Journal of Radiation Oncology, Biology, Physics 44(2):327–331.
Wheatley-Price, P., C. Ma, L. F. Ashcroft, M. Nankivell, R. J. Stephens, S. C. White, P. Lorigan, N. Thatcher, F. H. Blackhall, and F. A. Shepherd. 2009. The strength of female sex as a prognostic factor in small-cell lung cancer: A pooled analysis of chemotherapy trials from the Manchester Lung Group and Medical Research Council Clinical Trials Unit. Annals of Oncology 21(2):232–237.
White, E., C. Y. Lee, and A. R. Kristal. 1990. Evaluation of the increase in breast cancer incidence in relation to mammography use. Journal of the National Cancer Institute 82(19):1546–1552.
White, I. D. 2008. The assessment and management of sexual difficulties after treatment of cervical and endometrial malignancies. Clinical Oncology 20(6):488–496.
White, L. N., and M. R. Spitz. 1993. Cancer risk and early detection assessment. Seminars in Oncology Nursing 9(3):188–197.
WHO (World Health Organization). 2010. WHO Fracture Risk Assessment Tool. http://www.sheffield. ac.uk/FRAX/ (accessed April 5, 2010).
Wilkins, C. H., and S. J. Birge. 2005. Prevention of osteoporotic fractures in the elderly. American Journal of Medicine 118(11):1190–1195.
Williams, D. R., H. M. Gonzalez, H. Neighbors, R. Nesse, J. M. Abelson, J. Sweetman, and J. S. Jackson. 2007. Prevalence and distribution of major depressive disorder in African Americans, Caribbean blacks, and non-Hispanic whites: Results from the National Survey of American Life. Archives of General Psychiatry 64(3):305–315.
Winer, E. P., C. Hudis, H. J. Burstein, A. C. Wolff, K. I. Pritchard, J. N. Ingle, R. T. Chlebowski, R. Gelber, S. B. Edge, J. Gralow, M. A. Cobleigh, E. P. Mamounas, L. J. Goldstein, T. J. Whelan, T. J. Powles, J. Bryant, C. Perkins, J. Perotti, S. Braun, A. S. Langer, G. P. Browman, and M. R. Somerfield. 2005. American Society of Clinical Oncology technology assessment on the use of aromatase inhibitors as adjuvant therapy for postmenopausal women with hormone receptor-positive breast cancer: Status report 2004. Journal Clinical Oncology 23(3):619–629.
Winer, R. L., J. P. Hughes, Q. Feng, S. O’Reilly, N. B. Kiviat, K. K. Holmes, and L. A. Koutsky. 2006. Condom use and the risk of genital human papillomavirus infection in young women. New England Journal of Medicine 354(25):2645–2654.
Wingo, P. A., J. King, J. Swan, S. S. Coughlin, J. S. Kaur, J. A. Erb-Alvarez, J. Jackson-Thompson, and T. G. Arambula Solomon. 2008. Breast cancer incidence among American Indian and Alaska native women: US, 1999–2004. Cancer 113(5 Suppl.):1191–1202.
Wisner, K. L., K. Peindl, and B. H. Hanusa. 1993. Relationship of psychiatric illness to childbearing status: A hospital-based epidemiologic study. Journal of Affective Disorders 28(1):39–50.
Wolff, A. C., M. E. Hammond, J. N. Schwartz, K. L. Hagerty, D. C. Allred, R. J. Cote, M. Dowsett, P. L. Fitzgibbons, W. M. Hanna, A. Langer, L. M. McShane, S. Paik, M. D. Pegram, E. A. Perez, M. F. Press, A. Rhodes, C. Sturgeon, S. E. Taube, R. Tubbs, G. H. Vance, M. van de Vijver, T. M. Wheeler, and D. F. Hayes. 2007. American Society of Clinical Oncology/College of American Pathologists guideline recommendations for human epidermal growth factor receptor 2 testing in breast cancer. Journal Clinical Oncology 25(1):118–145.
Wong, G. C., R. P. Giugliano, and E. M. Antman. 2003. Use of low-molecular-weight heparins in the management of acute coronary artery syndromes and percutaneous coronary intervention. Journal of the American Medical Association 289(3):331–342.
Wong, K. H., K. C. Chan, and S. S. Lee. 2001. Sex differences in nevirapine rash. Clinical Infectious Diseases 33(12):2096–2098.
Woodman, C. B. J., S. I. Collins, and L. S. Young. 2007. The natural history of cervical HPV infection: Unresolved issues. Nature Reviews: Cancer 7(1):11–22.
Wright, T. C. 2007. Cervical cancer screening in the 21st century: Is it time to retire the pap smear? Clinical Obstetrics and Gynecology 50(2):313–323.
Wright, T. C., Jr., L. S. Massad, C. J. Dunton, M. Spitzer, E. J. Wilkinson, and D. Solomon. 2007. 2006 consensus guidelines for the management of women with abnormal cervical cancer screening tests. American Journal of Obstetrics and Gynecology 197(4):346–355.
Writing Group for the Women’s Health Initiative Investigators. 2002. Risks and benefits of estrogen plus progestin in healthy postmenopausal women: Principal results from the Women’s Health Initiative Randomized Controlled Trial. Journal of the American Medical Association 288(3):321–333.
Xu, J., K. D. Kochanek, S. L. Muphy, and B. Tejada-Vera. 2010. National Vital Statistics Report. Deaths: Final Data for 2007. HHS (Department of Health and Human Services).
Yamamoto, H., I. Sekine, K. Yamada, H. Nokihara, N. Yamamoto, H. Kunitoh, Y. Ohe, and T. Tamura. 2008. Gender differences in treatment outcomes among patients with non-small cell lung cancer given a combination of carboplatin and paclitaxel. Oncology 75(3-4):169–174.
Yan, G., Y. Fu, and D. Faustman. 1997. Reduced expression of TAP1 and LMP2 antigen-processing genes in the nonobese diabetic (NOD) mouse due to a mutation in their shared bidirectional promoter. Journal of Immunology 159(6):3068–3080.
Yang, W. T., S. Carkaci, L. Chen, C-J. Lai, A. Sahin, G. J. Whitman, and C. C. Shaw. 2007. Dedicated cone-beam breast CT: Feasibility study with surgical mastectomy specimens. American Journal of Roentgeneology 189(6):1312–1315.
Young, E. A., S. G. Kornstein, S. M. Marcus, A. T. Harvey, D. Warden, S. R. Wisniewski, G. K. Balasubramani, M. Fava, M. H. Trivedi, and A. John Rush. 2009. Sex differences in response to citalopram: A STAR*D report. Journal of Psychiatric Research 43(5):503–511.
Xu J. Q., K. D. Kochanek, S. L. Murphy, B. Tejada-Vera. 2010. Deaths: Final data for 2007. National Vital Statistics Reports 58(19). Hyattsville, MD: National Center for Health Statistics. 2010.
Yusuf, S., S. R. Mehta, F. Zhao, B. J. Gersh, P. J. Commerford, M. Blumenthal, A. Budaj, T. Wittlinger, and K. A. A. Fox. 2003. Early and late effects of clopidogrel in patients with acute coronary syndromes. Circulation 107(7):966–972.
Zaino, R. J., J. Kauderer, C. L. Trimble, S. G. Silverberg, J. P. Curtin, P. C. Lim, and D. G. Gallup. 2006. Reproducibility of the diagnosis of atypical endometrial hyperplasia: A Gynecologic Oncology Group Study. Cancer 106(4):804–811.
Zakaria, S., and A. C. Degnim. 2007. Prophylactic mastectomy. Surgical Clinics of North America 87(2):317–331.
Zang, E. A., and E. L. Wynder. 1996. Differences in lung cancer risk between men and women: Examination of the evidence. Journal of the National Cancer Institute 88(3-4):183–192.
Zeferino, L. C., and S. F. Derchain. 2006. Cervical cancer in the developing world. Best Practice and Research, Clinical Obstetrics and Gynaecology 20(3):339–354.
Zell, J., W. Tsang, T. Taylor, R. Mehta, and H. Anton-Culver. 2009. Prognostic impact of human epidermal growth factor-like receptor 2 and hormone receptor status in inflammatory breast cancer (IBC): Analysis of 2,014 IBC patient cases from the California cancer registry. Breast Cancer Research 11(1):R9.
Zhou, J., X. Y. Sun, D. J. Stenzel, and I. H. Frazer. 1991. Expression of vaccinia recombinant HPV 16 L1 and L2 ORF proteins in epithelial cells is sufficient for assembly of HPV virion-like particles. Virology 185(1):251–257.
zur Hausen, H. 2009. Papillomaviruses in the causation of human cancers—A brief historical account. Virology 384(2):260–265.






























































































































