6
Uniquely Human Evolution of Sialic Acid Genetics and Biology
AJIT VARKI
Darwinian evolution of humans from our common ancestors with non-human primates involved many gene–environment interactions at the population level, and the resulting human-specific genetic changes must contribute to the “Human Condition.” Recent data indicate that the biology of sialic acids (which directly involves less than 60 genes) shows more than 10 uniquely human genetic changes in comparison with our closest evolutionary relatives. Known outcomes are tissue-specific changes in abundant cell-surface glycans, changes in specificity and/or expression of multiple proteins that recognize these glycans, and novel pathogen regimes. Specific events include Alu-mediated inactivation of the CMAH gene, resulting in loss of synthesis of the Sia N-glycolylneuraminic acid (Neu5Gc) and increase in expression of the precursor N-acetylneuraminic acid (Neu5Ac); increased expression of α2–6-linked Sias (likely because of changed expression of ST6GALI); and multiple changes in SIGLEC genes encoding Sia-recognizing Ig-like lectins (Siglecs). The last includes binding specificity changes (in Siglecs -5, -7, -9, -11, and -12); expression pattern changes (in Siglecs -1, -5, -6, and -11); gene conversion (SIGLEC11); and deletion or pseudogenization (SIGLEC13, SIGLEC14, and SIGLEC16). A nongenetic outcome of the CMAH mutation is human metabolic incorporation of foreign dietary Neu5Gc, in the face of circulat-
Center for Academic Research and Training in Anthropogeny, Glycobiology Research and Training Center, Departments of Medicine and Cellular and Molecular Medicine, University of California San Diego, La Jolla, CA 92093. E-mail: a1varki@ucsd.edu.
ing anti-Neu5Gc antibodies, generating a novel “xeno-autoantigen” situation. Taken together, these data suggest that both the genes associated with Sia biology and the related impacts of the environment comprise a relative “hot spot” of genetic and physiological changes in human evolution, with implications for uniquely human features both in health and disease.
The theory of evolution via descent by natural selection explains the diversity of life on Earth (Darwin, 1859). Huxley (1863) and Darwin (1871b) correctly predicted that the “great apes” (chimpanzees, bonobos, gorillas, and orangutans, i.e., nonhuman hominids, NHHs1) are our closest evolutionary cousins. Indeed, chimpanzees were once considered good models for human disease. However, there are major differences between humans and NHHs in the incidence and severity of various diseases, beyond those explained by anatomical reasons (Varki, 2000; Varki and Altheide, 2005; Finch, 2010).
Scholars of mathematical, physical, and chemical sciences sometimes ask why biology does not have the kinds of universal laws that underpin their disciplines. The reason is that although biological systems operate under mathematical, physical, and chemical principles, evolutionary mechanisms of random mutation and deterministic selection do not generate consistent or universal outcomes. Of course, a single origin of life combined with physical constraints resulted in some near-universals, such as the paradigm that nucleic acid sequences encode protein sequences (Crick, 1970). Another apparent biological universal is that all nucleated cells in nature are covered with a dense and complex coating of sugar chains (glycans) (Varki, 2006), which have numerous biological roles (Varki and Lowe, 2009). Thus, natural selection repeatedly recruited glycans as being the best molecules for decorating the cell surface. Here I focus on one aspect of cellular glycan coating that changed during human evolution, potentially explaining aspects of human uniqueness, in health and in disease.
SIALIC ACIDS DECORATE THE CANOPY OF THE CELL-SURFACE GLYCAN FOREST AND HAVE MULTIPLE BIOLOGICAL ROLES
In the Deuterostome lineage (vertebrates and so-called “higher” invertebrates) the outer ends of glycan chains are often capped by sialic acids (Sias) (Varki, 2007; Schauer, 2009). Biosynthetic pathways for these nine-carbon backbone molecules likely evolved from those for ancestral non-ulosonic acids (Lewis et al., 2009). Although Sias are rare in other taxa (with the exception of certain pathogenic/commensal bacteria, as discussed later) they are ubiquitous on all vertebrate cell surfaces and are essential for embryonic development (Schwarzkopf et al., 2002). Indeed, they mediate many critical endogenous functions by virtue of physical properties and via recognition by intrinsic receptors (Varki, 2007; Schauer, 2009). Also, cell-surface Sias are used by complement factor H (Pangburn et al., 2000) and by Sia-binding Ig-like lectins (Siglecs) (Varki and Angata, 2006; Crocker et al., 2007) as signals for “self” recognition in the vertebrate innate immune system. However, given their location and abundance (dozens to hundreds of millions of copies on each cell), Sias also are targets for extrinsic receptors of numerous pathogens (Varki, 2007). Meanwhile, Sias have been “reinvented” repeatedly via convergent evolution by microbes that interact with vertebrates (Vimr et al., 2004; Lewis et al., 2009). Such “molecular mimicry” allows microorganisms to use Sias not only to mask themselves from the complement and adaptive immune systems (Pangburn et al., 2000; Schauer, 2009) but also to engage the Siglecs (as discussed later), dampening the innate immune response (Carlin et al., 2009b). For all these reasons, Sias are at the nexus of an evolutionary arms race between vertebrate hosts and their pathogens, interactions characterized by many “Red Queen” processes (Varki, 2006; Varki and Angata, 2006). This competition may also explain why there are so many kinds of Sias, each presented in several different linkages to the underlying monosaccharide, on a variety of different types of glycans (Varki, 2007; Schauer, 2009).
“SERUM SICKNESS” AS A CLUE TO HUMAN UNIQUENESS
Given the considerations discussed in the previous section, it is not surprising that differences in Sia expression are common between different taxa, even closely related ones. However, on closer inspection, such differences tend to be relative rather than absolute (Zanetta et al., 2001). One classic exception was a difficulty in finding the Sia N-glycolylneuraminic acid (Neu5Gc) in human tissues (Gottschalk, 1960). Indeed, humans make antibody responses against Neu5Gc during “serum sickness reactions” induced by animal serum infusion, characterized by antibodies agglutinating animal red blood cells bearing Neu5Gc (Higashi et al., 1977; Merrick et
al., 1978; Malykh et al., 2001). However, Neu5Gc was detected in human cancers and fetal tissues (Malykh et al., 2001).
A SIA DIFFERENCE BETWEEN HUMANS AND NHHS
Besides Neu5Gc, the other major Sia on most mammalian cell types is N-acetylneuraminic acid (Neu5Ac). These molecules differ by one oxygen atom, which is added to CMP-Neu5Ac in the cytosol, in a reaction catalyzed by the enzyme cytidine monophosphate N-acetylneuraminic acid hydroxylase (CMAH) (Shaw and Schauer, 1989; Takematsu et al., 1994). Both CMP-Neu5Ac and CMP-Neu5Gc are transported into the Golgi, where they are donors for addition of these Sias to many glycoconjugates. Thus, most mammalian tissues contain both Sias. In contrast, Neu5Gc was claimed to be missing in normal human tissues (Gottschalk, 1960). We showed that whereas all NHHs had easily detectable Neu5Gc in erythrocytes and plasma proteins, it was indeed missing from normal human blood samples (Muchmore et al., 1998). This human-specific difference was explained by deletion of a critical 92-base-pair exon in the CMAH gene (Chou et al., 1998; Irie et al., 1998)2 encoding key amino acids required for enzymatic function. This single Alu-mediated mutation (Hayakawa et al., 2001) occurred in one ancestral hominin CMAH gene, an allele now universal to humans. Timing was estimated to be ~2–3 Mya (Chou et al., 2002), which is, interestingly, just before emergence of the genus Homo (Wood and Collard, 1999). Of course, any genomic signatures of selection are erased by such depths of evolutionary time.
HUMAN-SPECIFIC Neu5Gc LOSS AFFECTS PATHOGEN REGIMES
The loss of Neu5Gc and resulting excess of Neu5Ac (Fig. 6.1, step 1) would have affected relative efficacy of interactions of various pathogens with humans. Humans should be resistant to pathogens binding Neu5Gc (Kyogashima et al., 1989; Rolsma et al., 1998; Martin et al., 2005; Schwegmann-Wessels and Herrler, 2006; Campanero-Rhodes et al., 2007) and more susceptible to pathogens preferring to bind Neu5Ac. Particularly interesting is a difference in erythrocyte Sia-binding preference between malarial parasites of humans and African NHHs (Martin et al., 2005).
Indeed, we and others suggested that ancestral hominins escaped the prevailing NHH malaria by eliminating Neu5Gc production and that Plasmodium falciparum (today’s human “malignant malaria”) arose later, when a strain of the NHH malaria evolved to be able to bind preferentially to Neu5Ac-rich erythrocytes of humans (Rich et al., 2009; Varki and Gagneux, 2009). Further studies of Neu5Gc and Neu5Ac preferences of human and nonhuman pathogens are warranted.
DIFFERENTIAL EXPRESSION OF α2–6-LINKED SIAS BETWEEN HUMANS AND NHHS
Influenza viruses use Sias as binding targets, and strains infecting some other species do not easily “jump” into humans. However, this difference is not primarily explained by human Neu5Gc deficiency, because these viruses show only relative preferences for the two Sias (Suzuki et al., 2000). A bigger difference lies in the finding that although avian influenza viruses preferentially recognize Sias α2–3-linked to the underlying sugar chain, human viruses prefer the α2–6-linked variety (Daniels et al., 1984). This difference corroborates with α2–6-linked Sia expression on human upper airways (Baum and Paulson, 1990). Meanwhile, chimpanzees challenged with human influenza virus did not show severe infections (Snyder et al., 1986). In keeping with this finding, we found low expression of α2–6-linked Sias in upper airways of NHHs (i.e., more similar to their expression in mice and birds) (Gagneux et al., 2003). This difference likely results from preferential human up-regulation of the enzyme ST6Gal-I, which determines expression of α2–6-linked Sias (Appenheimer et al., 2003) in humans. One possibility is that malarial parasites that preferentially bind α2–3-linked Sias (Orlandi et al., 1992) could have selected for up-regulation of α2–6-linked Sias on ancestral hominin erythrocytes (Gagneux et al., 2003) and thus, secondarily, in other tissues (Fig. 6.1, step 1).
SIGLECS DIFFERENCES BETWEEN HUMANS AND NONHUMAN HOMINIDS
Siglecs are a family of Sia-binding proteins characterized by aminoterminal V-set Ig-like domains with a Sia-binding site (Varki and Angata, 2006; Crocker et al., 2007) followed by variable numbers of C2-set domains, a single transmembrane domain, and varying lengths of cytosolic tails that may or may not have signaling domains—typically immunoreceptor tyrosine-based inhibitory motifs (ITIMs), which can recruit the tyrosine phosphatases SHP-1 or SHP-2 and down-regulate cellular activation by antagonizing tyrosine kinase action (Varki and Angata, 2006; Crocker et al., 2007). Siglec homologs are present in most vertebrates (Cao et al.,
2009) and seem prominent in primates (Angata et al., 2004). One subclass called “CD33-related Siglecs” (CD33rSiglecs) is rapidly evolving via multiple genomic processes (Angata et al., 2004). Multispecies genomic BAC sequencing of the CD33-related Siglec gene cluster (Angata et al., 2004) followed by chimpanzee genome sequencing made it possible to clone and characterize what may be all 16 hominid Siglecs. Remarkably, as discussed later, human-specific differences from other NHHs have been found in many CD33rSiglecs.
HUMAN-SPECIFIC ADJUSTMENTS IN SIA RECOGNITION BY SIGLECS
The ancestral condition of hominid Siglecs appears to have been to recognize Neu5Gc preferentially (Sonnenburg et al., 2004). This supposition fits with the function of CD33rSiglecs to recognize Sias as “self” and send dampening signals to immune cells via cytosolic tail ITIMs (Varki and Angata, 2006; Crocker et al., 2007; Carlin et al., 2009b). Because no pathogen has been reported to synthesize Neu5Gc, and many can synthesize Neu5Ac, Neu5Gc should indeed be the preferred molecule for “self” recognition. Thus, when human ancestral hominins lost Neu5Gc, many CD33rSiglecs would have lost their preferred ligand (Fig. 6.1, step 2), likely causing excessive innate immune cell activation. Although this loss may even have been beneficial in short-term defense, it would be eventually
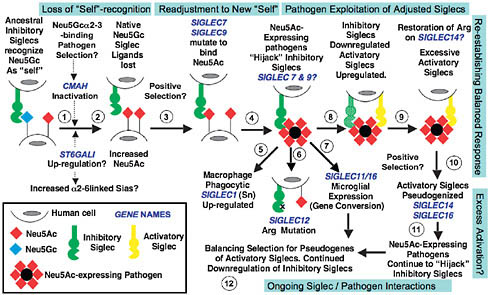
FIGURE 6.1 Proposed evolutionary scenario linking human-specific changes in Sia-related genes. It is impossible to conclusively prove evolutionary events and selection factors affecting Sia biology before the origin of modern humans. The speculative scenario presented here is based on available information and takes the parsimonious view that the events are related to one another. The first event may have been loss of Neu5Gc expression via CMAH inactivation and fixation (steps 1 and 2). A possible selection mechanism was a Neu5Gc-binding pathogen such as an NHH malaria, combined with genetic drift caused by ancestral demography. Because such organisms prefer binding α2–3-linked Sias, the increased human expression of α2–6-linked Sias may have been related. Human pathogen regimes would also have changed because of loss of Neu5Gc and excess of Neu5Ac. Some outcomes may have been positive (i.e., temporary escape from preexisting pathogens), and others may have been negative (e.g., increased susceptibility to Neu5Ac-binding pathogens and inability to modulate Neu5Gc/Neu5Ac ratios). Meanwhile, the loss of Neu5Gc should have resulted in loss of CD33rSiglec ligands needed for “self-recognition” (step 3). The likely hyperimmune state following Siglec ligand loss would have been followed by positive selection to allow multiple Siglecs (e.g., Siglec-9 and -7) to recognize Neu5Ac (step 4). Following this readjustment to the new “self,” a new risk would emerge. Although microbes appear incapable of synthesizing Neu5Gc, they have repeatedly reinvented Neu5Ac in multiple ways. Such pathogens would now be able to “hijack” inhibitory Siglecs such as Siglec-7 and -9, dampening the innate immune response of hominins (step 4). Indeed, several such organisms tend to be human-specific commensals. Notably, this proposed phase of pathogen exploitation of adjusted Siglecs is also the period of human evolution when newborns were becoming increasingly immature and more susceptible to these types of pathogens, especially those involved in brain invasion. Macrophage Siglec-1 might have then been up-regulated to enhance phagocytosis of Neu5Ac-expressing pathogens (step 5). Consequences of this proposed episode of pathogen exploitation of adjusted Siglecs could have been mutations of the Arg residue required for Sia recognition (Siglec-12, step 6) and the gene conversion event in Siglec-11 associated with recruitment to brain microglia (step 7). Eventually, immune cells would have down-regulated inhibitory Siglecs to escape the Neu5Ac-expressing pathogens while also up-regulating activatory Siglecs to respond to them (step 8). Perhaps this process explains why the critical Arg residue of the activatory Siglec-14 may have been restored in humans. This attempted reestablishment of a balanced response may have resulted in excessive activatory Siglecs (step 9), perhaps explaining the tendency of activatory Siglecs to be pseudogenized in modern humans (step 10). Of course, pathogens always evolve faster, and Neu5Ac-expressing pathogens are likely continuing to evolve to “hijack” our inhibitory Siglecs (step 11). Thus we likely have ongoing adjustments, with balancing selection for pseudogenes of the remaining activatory Siglecs and continued down-regulation of inhibitory Siglecs (step 12). It is also possible that these complex episodes of selection resulted in a changed profile of Siglec expression and function, not only in the innate immune system but also in other organs such as the placenta and the brain. Note that the human-specific changes in SIGLEC6 (placental trophoblast expression) and SIGLEC13 (deletion) are not incorporated into this model.
detrimental for reproductive fitness because of disease processes related to excessive immune responses. In keeping with this reasoning, human Siglecs studied show a preference for Neu5Ac over Neu5Gc (Sonnenburg et al., 2004). For this adjustment to occur, the V-set domain Sia-binding pockets of the CD33rSiglecs in ancestral hominins would have to be selected for multiple amino acid changes, switching either to specifically binding Neu5Ac or simply to accommodating it (Fig. 6.1, step 3). Indeed, sequence analyses indicate that this domain of CD33rSiglecs has undergone very rapid evolution in humans, even in comparison with relatively high rates in other taxa (Altheide et al., 2006). Taken together, the data suggest (Fig. 6.1) that lethality caused by Neu5Gc-binding pathogen (perhaps α2–3-linked Sia preferring) first selected the CMAH-null mutation, eliminating host Neu5Gc production. The resulting loss of “self” ligands for the CD33rSiglecs would have likely caused a hyperimmune state, perhaps with a temporary advantage. The next stage would have involved selection for amino acid changes to allow binding of Neu5Ac, restoring CD33rSiglec inhibitory function (Fig. 6.1, step 3).
MANY HUMAN PATHOGENS EXPRESS Neu5Ac, POTENTIALLY ENGAGING CD33rSIGLECS AND ATTENUATING INNATE IMMUNE RESPONSES
The switch of human Siglecs toward binding Neu5Ac (presumably selected to restore proper “self” recognition) would have exposed humans to pathogens that could “reinvent” Neu5Ac via convergent evolution, thus “hijacking” inhibitory Siglec function to dampen innate immune responses (Fig. 6.1, step 4, and later discussion). Indeed, many microorganisms that express Neu5Ac appear to be human-specific commensals, becoming pathogenic when circumstances allow (Vimr et al., 2004). For example, Group B Streptococcus expresses a Sia-containing capsule that engages human neutrophil Siglec-9, dampening responses (Carlin et al., 2009b). Other sialylated pathogens are recognized by Siglecs (Jones et al., 2003), likely with similar outcomes (Khatua et al., 2009). Notably, such pathogens would have been a strong selective force, because they often affect fetuses, infants, and young adults and frequently cause lethal brain infections (Vimr et al., 2004).
HUMAN-SPECIFIC CHANGES IN SIALOADHESIN ON MACROPHAGES
Sialoadhesin (Sialec-1, Sn) is a Siglec with 17 extracellular Ig-like domains, all conserved from mouse to human (Crocker et al., 1997). Also
conserved is the amino terminal V-set domain, which (even in the mouse) does not recognize Neu5Gc but binds only Neu5Ac, and only in α2–3 and α2–8 linkages (Crocker et al., 1997). Notably, Neu5Ac in α2–3 and α2–8 linkages are also the structures typically found on pathogens (Vimr et al., 2004). Furthermore, Sn is found primarily on macrophages, does not have a cytosolic signaling motif, and phagocytoses sialylated bacteria (Jones et al., 2003). Thus, although Sn has a role in modulating adaptive immunity (Oetke et al., 2006), a likely conserved function is to eliminate sialylated pathogens. Indeed, Sn in rodents is found at sites such as the sinuses of lymph nodes, spleen, and bone marrow that would first encounter bacteria invading extracellular fluids (Crocker et al., 1997) which filter blood or lymph-borne pathogens. In keeping with the human propensity for invasion by Neu5Ac-expressing pathogens, Sn is up-regulated in the human spleen compared with the chimpanzee (Brinkman-Van der Linden et al., 2000). In the chimpanzee, as in the rodent, only a subset of splenic macrophages is Sn positive, whereas in humans the distribution is more widespread (Brinkman-Van der Linden et al., 2000). Although more work is needed, current data suggest that Sn expression was up-regulated in humans, perhaps to deal with sialylated pathogens taking advantage of the Neu5Ac-preferring human CD33rSiglecs (Fig. 6.1, step 5). Interestingly, Sn is also up-regulated following inflammatory responses and in autoimmune diseases (Biesen et al., 2008) and has an additional role as a capture mechanism for certain viruses that have heavily sialylated envelope glycoproteins (Junt et al., 2007). In keeping with this notion, Sn-positive circulating monocytes may facilitate HIV entry into macrophages (Rempel et al., 2008), a viral invasion process prominent in humans.
HUMAN-SPECIFIC CHANGES IN A CONSERVED ARGININE RESIDUE REQUIRED FOR SIGLEC RECOGNITION OF SIAS
All Siglecs studied to date have a conserved arginine (Arg) residue in the V-set domain, essential for Sia binding (Varki and Angata, 2006; Crocker et al., 2007). This Arg residue underwent a human-specific mutation in Siglec-12, a CD33rSiglec found on macrophages and epithelial surfaces (Angata et al., 2001). Interestingly, restoration of the Arg residue regenerates binding with a preference for Neu5Gc (Angata et al., 2001), suggesting that this Siglec may have been “retired” following human loss of Neu5Gc (Fig. 6.1, step 6). In the second instance, as discussed later, the Arg residue of Siglec-5 and Siglec-14 appears to be mutated in all NHHs, but restored in humans (Angata et al., 2006).
HUMAN-SPECIFIC GENE CONVERSION INVOLVING SIGLEC-11
The gene encoding Siglec-11 is ~1 megabase away from the CD33rSiglec gene cluster on chromosome 19 (Angata et al., 2002) but has features of a CD33rSiglec, with a Sia-binding amino-terminal V-set domain and ITIMs in the cytosolic tail (Angata et al., 2002). The 5′ sequences of the SIGLEC11 encoding the first two Ig-like domains showed a >99% similarity to the corresponding 5′ end of a nearby Siglec pseudogene SIGLECP16 (Hayakawa et al., 2005). There is far less similarity in the rest of the sequences. Based on these and other data, we concluded that the SIGLEC11 gene underwent a gene conversion by the 5′ sequences of SIGLECP16, generating a protein with a human-specific amino acid sequence (Hayakawa et al., 2005). Indeed, this gene conversion is not seen in the NHH Siglec-11 orthologs (Hayakawa et al., 2005). Moreover, it is human universal, indicating possible selection following gene conversion (Fig. 6.1, step 7). One consequence is a change in binding specificity toward a preference for Neu5Ac over the ancestral preference for Neu5Gc. Another consequence is that, although Siglec-11 is expressed in both human and chimpanzee tissue macrophages, it is selectively expressed in brain microglia only in humans (Hayakawa et al., 2005). This unusual brain expression could be related to the propensity of sialylated pathogens to invade the human brain and/or the fact that microglia have multiple roles in the brain beyond innate immunity (Lu et al., 2005).
In some humans the pseudogene SIGLEC16P locus can instead encode the functional gene sequence SIGLEC16 (Cao et al., 2008), a molecule with potential activatory properties (as discussed later). Thus, some humans may have an activatory Siglec in brain microglia, and others may not. The population distribution of this segregating pseudo(gene) deserves further study. Consequences for microglia in human brain function and/or disease also need study.
HUMAN-SPECIFIC EXPRESSION OF SIGLEC-6 IN THE PLACENTAL TROPHOBLAST WITH UP-REGULATION IN PREECLAMPSIA
Siglec-6 is an inhibitory CD33rSiglec expressed on B cells of both humans and NHHs (Brinkman-Van der Linden et al., 2007). However, it also shows human-specific placental expression, not in immune cells but in the trophoblast (Brinkman-Van der Linden et al., 2007). Placental expression is maximal following human labor and delivery (Brinkman-Van der Linden et al., 2007), suggesting a possible role in modulating the
unusual tempo of human labor, which lasts much longer in humans than in the NHHs (Brinkman-Van der Linden et al., 2007).
Preeclampsia is a human-specific pregnancy complication of unknown cause, characterized by hypertension, proteinuria, and vascular abnormalities in the placenta leading to fetal dysfunction and early labor (Winn et al., 2009). In a microarray comparison of placental mRNAs, one of the genes showing the highest expression increase in preeclampsia was SIGLEC6 (Winn et al., 2009). It is interesting that both placental expression of Siglec-6 and preeclampsia itself are uniquely human phenomena. Many functional studies are needed, including analyses of placental Siglec-6 ligands (Brinkman-Van der Linden et al., 2007).
CD33rSIGLECS ARE EXPRESSED AT LOW LEVELS ON HUMAN T CELLS ASSOCIATED WITH OVERREACTIVE RESPONSES TO ACTIVATION
Although CD33rSiglecs are found on most human immune cells, essentially no expression was found on CD4+ T cells, and only low expression of Siglec-7 and -9 was found on CD8+ T cells (Ikehara et al., 2004; Varki and Angata, 2006; Crocker et al., 2007). In contrast, there was easily detectable expression of multiple CD33rSiglecs (particularly Siglec-5) on all NHH T cells examined (Nguyen et al., 2006). Thus, suppression of inhibitory CD33rSiglec expression is a human-specific condition, perhaps related to the need to escape Neu5Ac-expressing pathogens (Fig. 6.1, step 8). Regardless of the reason, we found that human T cells reacted more strongly to stimulation (Nguyen et al., 2006; Soto et al., 2010). Down-regulation of Siglec-5 on the chimpanzee T cells allowed more proliferation, and forced expression in human T cells dampened responses (Nguyen et al., 2006). Thus, the human T cell is in a relatively overreactive state, a least partly because of lack of Siglec-5 expression. In this regard, humans seem more prone to diseases involving T-cell activation, including AIDS (Rutjens et al., 2003), chronic hepatitis (Bettauer, 2010), rheumatoid arthritis, and bronchial asthma (Varki, 2000). This relative overreactivity may also explain T-cell activation and excessive release of cytokines (a “cytokine storm”) reported in human volunteers given a superactive anti-CD28 antibody (Stebbings et al., 2009) and the excessive human immune reactions in viral vector-based gene therapy trials (Mingozzi and High, 2007). More recently, we have found that human B cells are also relatively overreactive, compared with chimpanzee cells (Soto et al., 2010). Further studies are obviously needed, including any roles of activatory Siglecs (as discussed later).
HUMAN-SPECIFIC PSEUDOGENIZATION OF ACTIVATORY SIGLECS
Some primate Siglecs have a charged residue in the transmembrane domain and lack a major cytosolic tail. In at least two known instances (Siglec-14 and -16) (Angata et al., 2006; Cao et al., 2008) these molecules associate with the adaptor DAP-12, recruiting its immunoreceptor tyrosine-based activatory motifs (ITAMs) and effectively converting them into activatory Siglecs. Interestingly, Siglec-14 is undergoing repeated 5′-end gene conversions with Siglec-5, so that its Sia-binding specificity remains the same (Angata et al., 2006). This feature is also true of Siglec-16, because of gene conversion with Siglec-11 (Hayakawa et al., 2005). Analogous to “paired” inhibitory and activatory killer Ig-like receptors (KIR) (Parham, 2005), the most likely explanation is that activatory Siglecs were originally selected to respond against Sia-expressing pathogens that were using inhibitory Siglecs to suppress immune responses (Fig. 6.1, steps 8 and 9). Interestingly, both Siglec-14 and Siglec-16 are pseudogenized in some humans (Cao et al., 2008; Yamanaka et al., 2009). Additionally, Siglec-13 has potential for being an activatory Siglec and has been deleted in the human genome (Angata et al., 2004). Overall, there were apparently multiple human-unique pseudogenization events involving activatory Siglecs. Perhaps an evolutionary episode of excessive CD33Siglec-mediated activation resulted in the need to reestablish a balanced response (Fig. 6.1, step 10). Of course, pathogens are always ahead in an evolutionary arms race, and humans may still be in a period of ongoing adjustments, involving continued “hijacking” of inhibitory Siglecs (Fig. 6.1, step 11) and balancing selection for pseudogenization of activatory Siglecs (Fig. 6.1, step 12).
WAS SIA-RELATED BIOLOGY A “HOTSPOT” OF GENETIC AND PHYSIOLOGICAL CHANGES IN HUMAN EVOLUTION?
The high frequency of human-specific genetic changes associated with Sia biology is unexpected. Although some of these genes (e.g., Siglecs) are rapidly evolving in all taxa, the frequency of uniquely human changes seems unusually high compared with other species. For example, mouse and rat Siglecs appear nearly identical, and differences among NHHs and other Old World primates seem limited so far (Angata et al., 2004). Secondly, less than 60 genes are known to be directly involved in Sia biology (Altheide et al., 2006). Thus, one biochemical/biological pathway has almost 20% of its genes showing human-specific evolution. Overall, it is reasonable to suggest that Sia biology and Sia-related genes are a “hotspot” for genetic and physiological changes in human evolution. It is parsimonious to assume initially that all of these genetic changes are related to one another, as suggested in the scenario in Fig. 6.1. Although
several aspects are clearly speculative, the scenario is supported by available facts and includes testable concepts and hypotheses.
METABOLIC INCORPORATION OF Neu5Gc INTO HUMAN CELLS AND A DIETARY SOURCE OF Neu5Gc IN HUMAN TISSUES
We also discovered an unusual nongenetic consequence of CMAH loss (Fig. 6.2). Although Neu5Gc was reported in human cancers and fetal samples (suggesting an “oncofetal” antigen) (Malykh et al., 2001), the CMAH mutation damages the enzyme’s active site (Chou et al., 1998; Irie et al., 1998), which cannot be repaired. Also, a mouse with a human-like
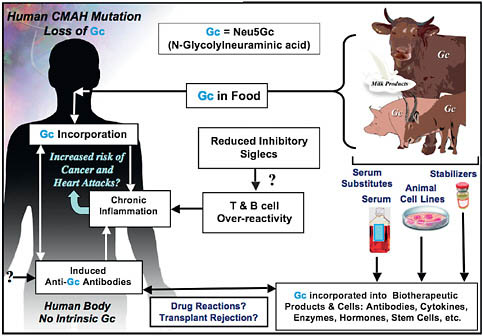
FIGURE 6.2 Two mechanisms for enhanced chronic inflammation and immune reactions in humans. Metabolic incorporation of dietary Neu5Gc (Gc) from mammalian foods in the face of circulating anti-Neu5Gc antibodies may contribute to chronic inflammation in endothelia lining blood vessels and in epithelia lining hollow organs, perhaps contributing to the increased risks of cardiovascular disease and carcinomas associated with these foods. The apparent T- and B-cell overreactivity of humans associated with decreased inhibitory Siglec expression may contribute further toward chronic inflammation. Also shown is that the fact that some molecular and cellular products of biotechnology are likely contaminated with Neu5Gc from multiple sources, potentially contributing to untoward reactions in some individuals.
Cmah mutation showed no endogenous Neu5Gc (Hedlund et al., 2007). Absent an alternate pathway for Neu5Gc synthesis, the sugar must enter from external sources. Indeed, cultured human cells express Neu5Gc because of uptake and metabolic incorporation from animal products in the medium (e.g., FCS) (Tangvoranuntakul et al., 2003; Bardor et al., 2005). This process involves macropinocytosis, delivery to the lysosome, and export of free Neu5Gc to the cytosol via the sialin transporter (Bardor et al., 2005). Once Neu5Gc reaches the human cytosol, it is a molecular “Trojan horse.” Differing by only one oxygen atom from endogenous Neu5Ac and having been eliminated only recently in evolutionary time, Neu5Gc is handled by human biochemical pathways as if it were native. Indeed, one can feed Neu5Gc to human cells and make them look like NHH cells (Bardor et al., 2005; Nguyen et al., 2005).
Classic studies showed that chickens generate a strong IgY antibody response against Neu5Gc (Malykh et al., 2001). Using a more specific version of such polyclonal antibodies and adding mass spectrometry to be certain (Hedlund et al., 2008), we confirmed the presence of Neu5Gc in human tumors and in fetal tissues (Tangvoranuntakul et al., 2003). Surprisingly, we also found smaller amounts in normal human tissues (Tangvoranuntakul et al., 2003). The likely explanation is a dietary origin. Voluntary Neu5Gc ingestion studies confirmed that humans could indeed take up Neu5Gc (Tangvoranuntakul et al., 2003).
ANTI-Neu5Gc ANTIBODIES IN HUMANS ARE OF BROAD AND HIGHLY VARIABLE SPECIFICITIES
Why should it matter that human tissues express small amounts of Neu5Gc derived from dietary sources? Although human biochemical pathways do not see Neu5Gc as foreign, it is detected as such by the immune system. Thus, contrary to prior work that used limited methodologies, we find anti-Neu5Gc antibodies circulating in all normal humans. In fact, some individuals have very high levels (Padler-Karavani et al., 2008), including complement-fixing IgGs capable of activating and/or killing cells expressing Neu5Gc (Nguyen et al., 2005). In this situation, a xeno-antigen can become metabolically incorporated into tissues, even while it is detected as being foreign by B cells. Thus, we call Neu5Gc a “xeno-autoantigen” in humans (Pham et al., 2009).
LIMITED DISTRIBUTION OF Neu5Gc IN FOODS AND DISEASE RISKS ASSOCIATED WITH RED MEAT
Because Sias are not found in plants, and Neu5Gc is not synthesized by microbes, the dietary source of Neu5Gc must be foods of animal
origin. Major sources appear to be red meats (i.e., lamb, pork, and beef) and, to a lesser extent, milk products (Tangvoranuntakul et al., 2003). In contrast, Neu5Gc is not found in poultry, and amounts in fish seem to be low (Tangvoranuntakul et al., 2003). Thus, within limits of current analyses, the primary source of human tissue Neu5Gc appears to be foods of mammalian origin. In this regard, many epidemiological studies have shown an association of red meat ingestion with increased risk for various diseases, including carcinomas (Fraser, 1999; Wiseman, 2008; Sinha et al., 2009), atherosclerosis (Fraser, 1999; Sinha et al., 2009), type-2 diabetes (Song et al., 2004), and age-dependent macular degeneration (Chong et al., 2009). Although there are other theories for how red meat consumption aggravates these diseases, most of these notions (other than the role of saturated fats in atherosclerosis) are unproven. We suggest that metabolic incorporation of dietary Neu5Gc in the face of anti-Neu5Gc antibodies contributes to red meat aggravation of diseases by stimulating chronic inflammation (Hedlund et al., 2008; Pham et al., 2009).
ANTI-Neu5Gc ANTIBODIES ENHANCE GROWTH OF Neu5Gc-POSITIVE TUMORS IN Neu5Gc-NULL MICE
Human carcinomas efficiently accumulate dietary Neu5Gc for multiple reasons, including up-regulation of lysosomal Sia transport by hypoxia (Yin et al., 2006) and enhanced macropinocytosis caused by growth factor activation. This accumulation occurs in the face of anti-Neu5Gc antibody responses, which are enhanced in such patients (Malykh et al., 2001). This combination suggests an immune reaction insufficient to kill the tumor that may, instead, stimulate it. Indeed, antibody-mediated inflammation is known to facilitate tumor progression by recruiting inflammatory cells, which stimulate angiogenesis and provide growth factors (Tan and Coussens, 2007). We mimicked the human situation using Neu5Gc-null mice bearing a syngeneic mouse tumor line that expresses low levels of Neu5Gc, similar to human tumors. Indeed, passively transferred anti-Neu5Gc immune serum from syngeneic Neu5Gc-null mice increased tumor growth rates associated with inflammation and angiogenesis (Hedlund et al., 2008), and these effects were blocked by a COX-2 inhibitor, a drug type that reduces human tumor incidence (Hedlund et al., 2008). Of course high levels of these antibodies may instead kill tumor cells, and it is possible that persons with very high anti-Neu5Gc antibodies are protected from some cancers. Indeed, can we harness human anti-Neu5Gc antibodies to target human cancers specifically?
SURPRISING DIFFERENCES BETWEEN HUMAN AND CHIMPANZEE HEART DISEASE
The commonest cause of death in both humans and captive chimpanzees is “heart disease,” manifested either as sudden “heart attacks” or as progressive heart failure (Lammey et al., 2008; Varki et al., 2009). However, early case reports suggested that the diseases in humans and chimpanzees are different, and recent studies have confirmed this notion (Lammey et al., 2008; Varki et al., 2009). Chimpanzees and other NHHs develop a progressive fibrotic replacement of the heart muscle (interstitial myocardial fibrosis), which can cause sudden death by altering heart rhythm or slower death by progressive cardiac failure. “Heart disease” in humans is different, caused by deposition of cholesterol in atherosclerotic plaques in the walls of large blood vessels, including coronary arteries (Pham et al., 2009; Varki et al., 2009). This deposition results in sudden or progressive loss of blood supply, explaining the common “heart attack” of humans (“myocardial infarction”) or progressive heart failure caused by “ischemic heart disease.” Although captive chimpanzees and others NHHs do have atherosclerosis (Varki et al., 2009), myocardial infarction and ischemic heart disease are rare, despite risk factors such as hypertension (Denton et al., 1995) and high levels of LDL cholesterol and lipoprotein(a) (Varki et al., 2009). Why do NHHs not often have the kind of heart disease common in humans? Conversely, why do humans not often suffer from the fibrotic heart disease so common in our closest evolutionary cousins?
HUMAN-SPECIFIC XENO-AUTOANTIBODY REACTION AGAINST ENDOTHELIUM: A CONTRIBUTING ROLE IN ATHEROSCLEROSIS?
For unclear reasons, accumulation of dietary Neu5Gc in human tissues is not uniform, and it tends to accumulate particularly in epithelial cells lining hollow organs (where carcinomas develop) or in the endothelium lining blood vessels (where atherosclerosis occurs). In fact, cultured endothelial cells fed with Neu5Gc (with Neu5Ac as a negative control) bind anti-Neu5Gc antibodies and deposit complement from human serum, resulting in cellular activation, expression of adhesion molecules, and binding of monocytes (Pham et al., 2009). Thus, although underlying mechanisms exist for many vascular diseases, we suggest that endothelial incorporation of Neu5Gc combines with circulating anti-Neu5Gc antibodies to aggravate processes such as atherosclerosis (Pham et al., 2009). Indeed, human atherosclerotic lesions show Neu5Gc accumulation not just in overlying endothelium but also inside the plaque (Pham et al., 2009). This Neu5Gc accumulation may facilitate production of anti-Neu5Gc
antibodies and further aggravate chronic inflammation in atherosclerosis progression. Thus, this xeno-autoantigen/autoantibody process may be an additional explanation for the increased atherosclerosis risk of consuming red meats and milk products.
A ROLE FOR Neu5Gc IN RED MEAT-RELATED FOOD POISONING?
Because Neu5Gc is present in some human cells, are we really resistant to Neu5Gc-binding pathogens? The typical low-affinity, high-avidity binding of pathogens to glycans seems unlikely to succeed when Neu5Gc molecules are rare on a human cell surface. An exception may arise when Neu5Gc is targeted by a multivalent toxin with relatively high affinity (Byres et al., 2008). Dietary Neu5Gc loads up epithelial and endothelial cells over time. Subsequent exposure to meat or milk products contaminated with SubAB toxin-expressing Escherichia coli would then allow the toxin to bind to gut epithelium, gain access to the bloodstream, and target the kidney endothelium, giving a hemolytic-uremic syndrome (Byres et al., 2008). The process may be facilitated by the fact that (unlike the cows in which this toxin is usually found) humans do not have circulating Neu5Gc-containing glycoproteins to act as natural toxin inhibitors (Byres et al., 2008). Thus, we speculate that individuals who consume large amounts of red meat and milk may not only increase their risk for this type of food poisoning but also prepare their tissues for attack by the toxin (Löfling et al., 2009).
WAS THE Neu5Gc XENO-AUTOANTIGEN PHENOMENON SIGNIFICANT IN HUMAN EVOLUTION?
Hunting and red meat consumption along with cooking very likely played a supporting role in the emergence of the genus Homo (Finch and Stanford, 2004; Carmody and Wrangham, 2009), and milk consumption was positively selected in some human civilizations (Tishkoff et al., 2007b). Indeed, these foods continue to be a vital source of important nutrients for currently undernourished populations. It should be noted that most diseases associated with red meat and/or milk consumption would not have affected natural selection in times past, because they are manifest primarily after the age of peak reproductive fitness. We now live much longer and have much greater access to red meat and milk, thus transforming these once beneficial foods into likely culprits for exacerbating diseases of older humans (Finch and Stanford, 2004).
POTENTIAL ROLES OF SIA-RELATED CHANGES IN UNIQUELY HUMAN DISEASE PROPENSITIES
We have here discussed multiple potential mechanisms by which uniquely human changes in Sia biology could contribute to such uniquely human disease phenotypes. Although many of the hypotheses are speculative and need further exploration, most are testable either by modeling in Neu5Gc-deficient and/or Siglec-modified mice or by studies in human subjects and human populations. Some of these issues are summarized in Fig. 6.2, along with reference to another area that deserves attention—the contamination of molecular and cellular biotherapeutic products by Neu5Gc derived from nonhuman sources.
FUTURE DIRECTIONS
This work has generated even more questions than answers. Apart from issues already discussed, some others are briefly discussed below.
Population Genetics and Polymorphisms of Siglecs
Siglec-12, -14, and -16 are partially pseudogenized (i.e., expressed as active and inactive alleles) in the human population (Angata et al., 2001; Cao et al., 2008; Yamanaka et al., 2009). Do any of these instances represent balanced polymorphisms, and are there more examples? Further studies must address allele distribution in various populations and consider associations with risk of diseases. Additional population-level studies of all Siglecs in NHHs are also warranted, not only to reaffirm that some changes are human specific but also to see whether additional differences and/or polymorphisms exist.
Siglecs in Bacterial Pathogenesis
Details of how Neu5Ac-expressing pathogens suppress immune responses via inhibitory Siglecs (Carlin et al., 2009b) are as yet unknown. Protein–protein interactions between bacteria and human Siglecs can also mediate similar processes (Carlin et al., 2009a). Meanwhile, the role of the activatory Siglecs in bacterial pathogenesis is postulated to be the opposite, but this notion needs proof. The potential role of Sn in clearing sialylated pathogens also needs further evaluation. We may well be looking at the “tip of the iceberg” regarding roles of Siglecs in bacterial pathogenesis.
What Is the Fate of Orally Ingested Neu5Gc?
We need to know mechanisms by which Neu5Gc is absorbed from the human gut and delivered to tissues. Early studies in rodents showed that the fate of ingested Neu5Gc may differ, based on the form in which it is presented (Nöhle and Schauer, 1984). We can now study these issues by feeding Cmah-null mice different forms of Neu5Gc and looking at its fate in the gut, body fluids, and tissues. At this time, we cannot assume that ingestion of a certain amount of Neu5Gc will deliver a corresponding amount to tissues. A related issue is the fate of Neu5Gc during food processing and cooking.
Mechanisms of Anti-Neu5Gc Antibody Induction
We are studying the tempo and mode of appearance of these highly variable antibodies in human samples and the potential mechanisms for their induction, using Cmah-null mice as a model. We also need to address whether Neu5Gc-containing glycans are truly T-cell-independent antigens, whether the antibody response involves a germline V-set domain, and if the antibody-binding pockets undergo affinity maturation. A related issue is whether these antibodies have any positive value (e.g., potentially protecting against enveloped viruses originating from other species).
Prognostic Value of Anti-Neu5Gc Antibodies
The highly variable anti-Neu5Gc antibody response of humans is further complicated because Neu5Gc itself is not the entire epitope recognized (i.e., the underlying glycan structures to which it is attached influences binding specificity). Thus, there are many possible Neu5Gc epitopes, and each human has a different response to each of them (Padler-Karavani et al., 2008). Because some of these epitopes are differentially expressed in different tissues, only some of the antibodies may have pathogenic roles, and the antibody subclasses may also make a difference. Perhaps one or more of these anti-Neu5Gc-antibodies will prove to be a predictive, prognostic, or diagnostic marker for one or more diseases. We are pursuing this possibility using a glycan microarray that contains matched Neu5Gc and Neu5Ac glycans as targets.
Complexity of the “Sialome” in the Cell Surface
The manner in which Sias are presented within the context of a complex cell-surface “landscape” can affect the way they interact with Siabinding proteins (Cohen et al., 2009). In other words, such proteins recognize not only linear glycan sequences but also more complex structures
presented on “clustered saccharide patches” (Varki, 1994) on cell surfaces, involving glycans of different types (Cohen et al., 2009). Thus, even specific epitopes in glycan arrays may not be representative of the “sialome” at the cell surface. These considerations apply not only to Siglecs but also to anti-Neu5Gc antibody epitopes. Another unexplored issue is whether loss of CMP-Neu5Gc in the Golgi has other consequences for competing biosynthetic pathways (e.g., we found an increase in Sia O-acetylation in the Cmah-null Neu5Gc-deficient mouse) (Hedlund et al., 2007). Finally, relative differences in biophysical properties between Neu5Gc and Neu5Ac could have consequences. Overall, the Sia biology changes in humans could alter more cell phenomena than we can currently imagine. One approach to exploring this issue is to feed different types of human cells with Neu5Gc (or Neu5Ac as a control) and then study interactions of anti-Neu5Gc antibodies or Siglecs, looking for differential binding by these proteins that cannot be explained by cell-surface glycan sequences.
Additional Phenotypes of Cmah-null Mice
The genomic lesion in our Cmah-null mice is almost identical to that of humans (Hedlund et al., 2007). The mice are viable and capable of reproduction, a situation that is not surprising, because the same is true of humans. Further studies of fertility are under way to look for any subtler differences. We have already reported that these mice show delayed wound healing and age-dependent hearing loss, similar to humans (Hedlund et al., 2007). We have preliminary evidence of metabolic differences that also deserve further study. Detailed neurobiological and cognitive studies are required to see if any known differences between human and NHH brains might be manifest. Of course, mice shared a common ancestor with primates more than 60 Mya, and the impact of this biochemical change in a rodent brain may not necessarily reflect what occurred in a hominid ancestor ~2–3 Mya. In this regard, it is fascinating that, even in animals with intact CMAH genes, the levels of brain Neu5Gc expression always seem very low (Gottschalk, 1960).
CONCLUSIONS AND PERSPECTIVES
The fact that so many genes related to Sia biology show human-specific differences from NHHs supports the notion that this system was a “hotspot” for evolutionary changes in the human lineage. Discussed here are some specific ways in which these changes would have impacted the immune system and human pathogen regimes. Although this discussion focuses on current human diseases, it also suggests a role for infectious diseases during human evolution. Of course, Sias and Siglecs are involved
in many other biological pathways. Thus, Sia-related differences between humans and NHHs are worthy of continued investigation.
This Sackler Symposium focused on understanding “The Human Condition” “In the Light of Evolution.” Since we reported these genetic differences between humans and NHHs (Chou et al., 1998), many others have been found (Varki and Nelson, 2007; Varki et al., 2008). Any explanation of human evolution and the human condition must take into account all the available data. Indeed, there are many approaches to anthropogeny (explaining the origin of humans) (Varki and Nelson, 2007; Varki et al., 2008), including studies of the fossil and archaeological record since our last common ancestors with other primates; exploring the impact of the environment (biological, physical, and cultural) on humans and other animals; comparisons of the ontogeny of each species; and, of course, species comparisons. All these approaches must be combined in a trans-disciplinary manner if we are eventually to explain human origins and human uniqueness.
ACKNOWLEDGMENTS
I gratefully acknowledge helpful comments from Miriam Cohen, Sandra Diaz, Jeff Esko, Pascal Gagneux, Chris Gregg, and Nissi Varki and Dr. Gagneux’s contributions to drawing the figures.






















