Attachment E:
Fluidized Bed Steam Reforming
A bed of granular material can be made to exhibit fluid-like properties by passing a liquid or gas through it. This process is referred to as fluidization, and the apparatus that supports this process is referred to as a fluidized bed. Fluidization came to age during World War II, when the urgent demand for aviation gasoline led to the development and construction of the first fluid bed catalytic cracker. In addition to gasoline production, fluidization technology is broadly used in coal gasification and combustion, mineral processing, food processing, pharmaceuticals, soil washing, manufacturing of polymers, waste treatment, and environmental remediation. Its applications include several unit operations such as drying, heating/cooling, particle coating, and chemical reactions.
The Fluidized Bed Steam Reforming (FBSR) of nuclear waste is a relatively new technology, though the fluidization phenomenon and steam reforming are well established in the chemical engineering field. Steam reforming is a method for generating hydrogen by reacting fossil fuels with water. For example, for natural gas:
If coal is used as a carbon source, it first undergoes pyrolysis or devolatilization then the char (C) reacts with steam according to the following reaction:
The H2 is combined with O2 so that no excess H2 exists in the system at any one time. This combination is exothermic and provides energy in the form of heat for the autocatalytic operation of the FBSR.
The FBSR consists of two fluidized beds. The first one operates in a reducing environment and its function is to evaporate the liquid nuclear waste stream; destroy organics; reduce nitrates, nitrites, and nitric acid to nitrogen gas; and form a stable solid waste product. The first stage fluidized bed of the FBSR process is referred to as the Denitration and Mineralization Reformer, or DMR. The DMR uses superheated steam as the fluidizing media. The bed material consists of granular solid additives and co-reactant(s), such as carbon, clay, silica, and/or catalysts. Liquid waste is directly fed to the fluidized bed after minor pre-treatment (e.g., to concentrate or dilute solubles) except the addition of clay.
By analogy to the above steam reforming chemistry, the carbon fed to FBSR (coal in this instance) produces H2 and CO. For organic compounds in the waste stream which undergo pyrolysis to form various hydrocarbons, the reducing environment is generated by the following reaction:
Similarly, the nitrates contained in the liquid waste are reduced to
In the steam environment, the sodium oxide is transferred to sodium hydroxide:
yielding the overall reaction
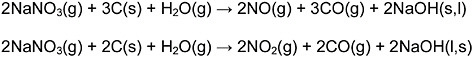
The NO and NO2 are further reduced to nitrogen gas by the reaction of CO, C, or H2 generated from the reaction of the organic material with steam as shown above. The nitrates can also be reduced by the addition of a catalyst or a metal. For example:
The second fluidized bed of the FBSR process operates in an oxidizing environment and is referred to as the Carbon Reduction Reformer, or CRR. The fluidizing gases are the off-gas from the first stage and added oxygen. Its function is to gasify carbon fines carried over in the process gases from the DMR, oxidize CO and H2 to CO2 and water, and convert trace acid gases to stable alkali compounds by reacting these acids with the bed media consisting of calcium carbonate and/or calcium silicate particles.
The addition of bulk aluminosilicates to the fluidized bed results in the production of anhydrous feldspathoid phases such as sodalite. The sodalite family of minerals (including nosean) are unique because they have cage-like structures formed of aluminosilicate tetrahedra. The remaining feldspathoid minerals, such as nepheline, have a silica “stuffed derivative” ring type structure. The cage structures are typical of sodalite and/or nosean phases where leach testing has indicated that the cavities in the cage structure retain anions and/or radionuclides which are ionically bonded to the aluminosilicate tetrahedra and to sodium cation.
Sodalite has the formula Na8[Al6Si6O24](Cl2). In sodalites and analogues with sodalite topologies, the cage is occupied by two sodium and two chlorine ions. When the 2NaCl are replaced by Na2SO4, the mineral phase is known as nosean, (Na6[Al6Si6O24](Na2SO4)). Since the Cl, SO4, and/or S2, are chemically bonded and physically restricted inside the sodalite cage structure, these species do not readily leach out of the respective FBSR waste form mineral phases. Thus, FBSR waste forms can be useful for immobilizing these species to prevent their leaching into groundwater.
Other minerals in the sodalite family, namely hauyne and lazurite which are also cage structured minerals, can accommodate either (SO4=) or (S=) depending on the REDOX of the sulfur during the steam reforming process. Sodalite minerals are known
to accommodate Be in place of Al and S2 in the cage structure along with Fe, Mn, and Zn, e.g., helvite (Mn4[Be3Si3O12]S), danalite (Fe4[Be3Si3O12]S), and genthelvite (Zn4[Be3Si3O12]S). These cage-structured sodalites were minor phases in HLW supercalcine waste forms and were found to retain Cs, Sr, and Mo into the cage-like structure, e.g., Mo as Na6[Al6Si6O24](NaMoO4)2. In addition, sodalite structures are known to retain B, Ge, I, and Br in the cage-like structures. Indeed, waste stabilization at Idaho National Laboratory currently uses a glass-bonded sodalite ceramic waste form (CWF) for disposal of electrorefiner wastes for sodium-bonded metallic spent nuclear fuel from the EBR II fast breeder reactor.



