2
The Incidence of Drug-Resistant TB in Southern Africa
|
Key Messages
|
Gerald Friedland, Yale School of Medicine, reviewed recent data on the incidence of drug-resistant TB worldwide, stressing that these data are inadequate and underestimate the reality of the situation. Friedland noted that knowledge of the true extent of the MDR/XDR TB epidemic is hampered by a lack of in-country laboratory capacity and the inadequacies of the existing health system infrastructure.
According to the World Health Organization (WHO), the number of new TB cases globally was 9.4 million in 2008 (WHO, 2009a). The disease killed 1.8 million people in that year, or 4,500 per day. While the highest number of cases of MDR TB (estimated annual numbers of new cases) is in Asia, the greatest per capita rates are in Africa (WHO, 2010a). No accurate and up-to-date data concerning the prevalence of MDR and XDR TB are available for most African countries. However, WHO estimates that more than 75,000 new MDR TB cases will occur in Africa in 2010 (Figure 2-1). By WHO estimates, 440,000 global cases of MDR TB arose in 2008; about 5.4 percent of these cases were XDR TB. About 15 percent of all TB cases, or 1.4 million, are associated with HIV infection. The 450,000 deaths caused by HIV-associated TB represent 26 percent of TB deaths and 23 percent of HIV deaths.
After briefly examining the relationship between reported incidence rates and the diagnostic tools used, this chapter summarizes information presented at the workshop on the incidence and burden of drug-resistant TB in South Africa. It then summarizes presentations and discussions that looked more closely at the outbreak of MDR and XDR TB in KwaZulu-Natal Province and that addressed coinfection with HIV/AIDS in southern Africa.
RELATIONSHIP BETWEEN REPORTED INCIDENCE AND DIAGNOSTIC TOOLS1
There is evidence that differences in the reported incidence of drug-resistant TB could be attributable, at least in part, to the diagnostic tools used in particular areas. Sturm reported that since 2005, the Church of Scotland Hospital (CoSH) in Tugela Ferry has been the only health care facility in KwaZulu-Natal Province to use culture diagnostics for all possible TB cases. The Msinga subdistrict, in which CoSH is located, is also the region in the province with the highest reported number of cases of XDR TB. Yet the incidence of drug-resistant TB in the Msinga subdistrict would not be expected to differ from that in the province’s other subdistricts and districts. The main difference appears to be that in the Msinga subdistrict, culture and drug susceptibility testing is done on all first contacts. Elsewhere, many patients die before a specimen is taken or the results of drug susceptibility testing become available.
Research also has found an association between the number of new cases of TB diagnosed by culture and the reported percentage of drug-resistant TB. In 2007, data on the presence of the F15/LAM4/KZN strain of
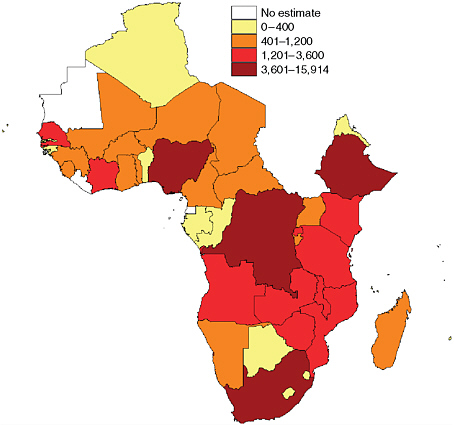
FIGURE 2-1 More than 75,000 new cases of MDR TB are estimated to have occurred in Africa in 2010.
SOURCE: Falzon et al., 2009.
M.tb. were published (Pillay and Sturm, 2007). This had been the dominant strain of MDR TB since 1994, and it had evolved into an XDR TB strain by 2001. The data suggest that the more new cases of TB are diagnosed by cultured specimens in a health district, the higher is the percentage of drug-resistant TB found in that district.2 In addition, the districts with the highest rates of MDR and XDR TB are those that house or have a strong link with academic centers, which send greater numbers of specimens for culture relative to other facilities.
For every patient who is diagnosed with and treated for MDR TB in southern Africa, it is likely that several more are diagnosed and not treated.
Further, an even larger number of those infected with MDR TB are never diagnosed because diagnostic capabilities are inadequate or because these people die without ever having been seen and diagnosed through the health care system. The metaphor Friedland used—which became a theme for the workshop—is that the number of cases of drug-resistant TB seen in southern Africa represents the “ears of the hippo,” a very small portion of a much larger entity that cannot be seen.
MDR AND XDR TB IN SOUTH AFRICA
According to several workshop presenters, drug-resistant TB imposes a tremendous health burden throughout southern Africa. Tomás Zimba of Mozambique’s Maputo Central Hospital described efforts to control and treat MDR TB patients in the country. Zimba explained that challenges in dealing with the co-epidemics of HIV and TB in Mozambique are similar to challenges experienced throughout the southern Africa region (see Box 2-1). Most of the discussion at the workshop focused on the experience in South Africa, however, where the incidence of MDR TB now exceeds 25 cases per 100,000 people in certain areas, according to Neel Gandhi of Tugela Ferry Care and Research Collaboration (TF CARES) and Albert Einstein College of Medicine.
To illustrate the burden of drug-resistant TB in South Africa, Martie van der Walt, Medical Research Council of South Africa (MRC), described the experience of King George V Hospital, the referral center for drug-resistant TB in KwaZulu-Natal Province. From February to August 2006, 211 patients with a positive culture on admission for drug-resistant pulmonary TB began their first course of therapy for MDR TB. Most of these patients were very ill at baseline, weighing below 50 kg (110 pounds) at the start of treatment, and mortality was common. Among these 211 patients, 4 of 12 with XDR TB and 16 of 199 with MDR TB died during the first 6 months of treatment (Kvasnovsky et al., 2008).
Richard Chaisson, Center for Tuberculosis Research, The Johns Hopkins University, noted that one of the United Nations’ Millennium Development Goals is to halve the amount of mortality and morbidity caused by infectious diseases relative to 1990 levels (UN, 2010). In large parts of Africa, this goal is becoming more distant for TB, according to Chaisson. In 1990, just 2 countries in Africa had a TB incidence greater than 300 per 100,000 population. This number had risen to 23 countries by 2005; in that same year, 6 countries had an incidence greater than 500 per 100,000 population, and 2 had an incidence greater than 1,000 per 100,000.
According to Chaisson, these figures translate into unmanageable numbers of patients presenting to the health care system. From 2005 to 2008,
|
BOX 2-1 Problems of Dealing with Drug-Resistant TB in Mozambiquea The experience of Mozambique illustrates the problems facing many areas of southern Africa in dealing with the epidemic of drug-resistant TB, according to Zimba. Almost constant warfare during the three decades from 1965 to 1994 created severe disruptions in the country’s health care system, just as political instability elsewhere in southern Africa has hampered health care delivery. Only 45 percent of the population has access to health care. Of Mozambique’s 22 million people, 12.5 percent of adults are estimated to be infected with HIV (UNAIDS, 2008) (see the discussion of TB and HIV coinfection later in this chapter). Southern Africa has adopted the WHO guidelines for diagnosis and treatment of TB, using microscopy as the standard for diagnosis and follow-up of TB cases. In Mozambique, the national TB control program was launched in 1977. The country has seen some success in expanding Directly Observed Treatment Short course (DOTS) to the community (community-based DOTS [C-DOTS]) and improving access for people in underserved areas (USAID, 2009). However, drug resistance has proven to be a great problem in the country. There are 255 laboratories in Mozambique that can diagnose TB using microscopy. But there is only one National Reference Laboratory that conducts culture and drug susceptibility testing for first-line anti-TB drugs. Samples for diagnosis from the district and provincial laboratories are transported to the National Reference Laboratory in the capital city of Maputo. Three months can pass before those laboratories receive feedback and the patient is informed of the diagnosis. A 1998–1999 nationwide survey of 709 culture-positive TB cases in Mozambique found that 3.4 percent of new cases were MDR (Mac-Arthur Jr et al., 2001). Drug resistance was more common among those with a history of prior treatment. A 2002–2003 report from the Mozambique national TB program similarly indicated that 3 percent of new TB cases were MDR (Nunes et al., 2005). In 111 samples from HIV-infected patients, 9 percent were found to have MDR TB. About 6 percent of new TB cases in patients infected with HIV, as well as 16 percent of TB cases among previously treated patients, were MDR. In 2009 there were an estimated 1,700 cases of MDR TB among notified pulmonary TB cases in Mozambique (WHO, Global Tuberculosis Control 2010; Annex 3). According to Zimba, however, it is known that the Mozambique national TB program has in the past seriously underestimated the numbers of new cases of MDR TB. The actual number of cases of primary MDR TB in Mozambique remains unknown. |
the largest hospital in South Africa, Chris Hani Baragwanath Hospital in Soweto, had between 6,500 and 6,800 new TB admissions per year. Inpatient mortality was 18 percent at 2 weeks, or about 1,200 deaths per year. Nine percent of those dying of TB had MDR TB, representing about 110 MDR TB deaths per year.
Estimating the contribution of TB to mortality in South Africa is difficult. This issue is addressed in Box 2-2.
Van der Walt presented data from the Preserving Effective TB Treatment with Second-line drugs (PETTS) study, which is a prospective cohort study of approximately 1,800 MDR TB patients in nine countries at 27 clinical sites and 17 laboratories. The objective of the study is to determine the frequency, timing, and risk factors for acquired resistance to second-line drugs in diverse MDR TB control programs. The study also is seeking to determine the effect of acquired second-line resistance on patient outcomes. Four provinces of South Africa, including KwaZulu-Natal, are participating in this study. A follow-up of the patients enrolled in the study has been performed every month to collect a culture and update a database used to monitor acquisition of resistance.
Van der Walt noted that cross-resistance3 to second-line drugs is especially problematic in South Africa. MDR TB patients in South Africa are three times more likely than their counterparts from other PETTS sites/countries to have cross-resistance to all the injectable drugs for TB at baseline. The PETTS study has shown that XDR TB was present at baseline in 10.3 percent of cases in 2005 (Kvasnovsky et al., 2009), while a 2005–2006 analysis of the PETTS cohort found that 1 percent of drug-resistant TB cases were TDR.4
OUTBREAK OF MDR AND XDR TB IN KWAZULU-NATAL PROVINCE5
KwaZulu-Natal Province, which comprises 11 health districts divided into a number of subdistricts, has a population of about 9.6 million people. The province has an enormous TB burden, and MDR and XDR TB strains are present throughout.
|
BOX 2-2 Estimating the Contribution of TB to Mortality in South Africaa According to Tuoane-Nkhasi, information on the contribution of TB to mortality in South Africa can be obtained from the country’s civil registration system based on registered deaths in the country. A medical practitioner must indicate the cause of death on the death notification form. Methods for analyzing cause-of-death data include the following:
About 85 percent of all deaths in the country are registered. The levels and trends of registered deaths from 1997 to 2007 in South Africa show that about 10 percent of deaths were among children aged 0–24 The majority of these deaths occurred during the first year in the postneonatal period. The annual number of child deaths increased consistently from 1997 to 2006, with a slight decrease in 2007. There were more male than female deaths among children, and more infant deaths occurred in KwaZulu-Natal and Gauteng than in the other provinces. In 2007, 11 death notification forms indicated that MDR or XDR TB was the underlying cause of death among children aged 0–44 Of the 10 leading underlying causes of death in that age group, TB was ranked sixth, accounting for 2.5 percent of deaths. Respiratory and cardiovascular disorders specific to the perinatal period were indicated as the leading cause of death. For both males and females aged 15–664,TB has been the leading underlying cause of death in South Africa since 1997. Since 2002, about 12 percent of all deaths in the country have been caused by respiratory TB, but the death notification forms usually do not specify whether this diagnosis was confirmed either bacteriologically or histologically. In 2006, |
|
the underlying cause of death among 607 people was identified as MDR TB; in 2007, this number decreased slightly to 597. There are a number of sources of uncertainty in these statistics:
Information from South Africa’s civil registration system is useful in providing numbers of deaths and their causes each year, but the statistics produced depend on the quality of the data input to the system. According to Tuoane-Nkhasi, concerted efforts of the public, the Department of Home Affairs, the Department of Health, and Statistics South Africa are required to improve these data. |
In 2006, a group led by Gandhi reported on 53 cases of XDR TB in Tugela Ferry in the Msinga subdistrict, which has a population of about 200,000 traditional Zulu people living largely in poverty (Gandhi et al., 2006). In June 2005, within a few months of the discovery of the first XDR TB case in Tugela Ferry, 5 other facilities in KwaZulu-Natal had cases. By December 2005, a further 18 facilities had reported cases; by June 2006, the total was 32 facilities; by December 2006, the total was 39; and by March 2007, it was 42. The latest estimates are that more than 60 facilities in KwaZulu-Natal have reported at least one case of XDR TB. In addition, every province in South Africa and all neighboring countries have reported cases of XDR TB.
MDR TB has undergone a similarly dramatic increase in KwaZulu-Natal Province. Gandhi noted that there were 2,654 cases of MDR TB in 2007 alone, representing a prevalence of 26 cases per 100,000 population.
According to Friedland, given the widespread distribution and continuing appearance of new cases of both MDR and XDR TB as of 2010, the situation is more appropriately defined as an epidemic than as a series of outbreaks.
The rapid increase in drug-resistant TB in KwaZulu-Natal Province was not consistent with the relatively small numbers of MDR TB cases among patients who had no prior history of treatment according to a 2001–2002 survey of drug-resistant TB in South Africa, the last survey conducted in the province prior to the outbreak of drug-resistant TB in 2006 (Weyer et al., 2004). That survey found, for example, that just 1.7 percent of patients with no history of prior TB treatment in KwaZulu-Natal Province had MDR TB. The outbreak of XDR TB in the province therefore raised the question of whether the 2001–2002 data underrepresented the extent of MDR TB infection.
The study reported by Gandhi, which received international attention, found that XDR TB in that population was highly fatal. All but one of the patients died (98 percent mortality), with a median survival of just 16 days from sputum collection.6 The study revealed high clustering rates, with 85 percent having genetically similar TB strains. The study also found that half of the patients had never been previously treated for TB, and more than 95 percent of patients with XDR TB had never been treated for MDR TB. Thus, these patients had not had the long-term exposure to second-line drugs that would be expected to lead to resistance. The study authors concluded that the transmission of XDR TB strains from person to person was likely.7
More recently, high levels of mortality have been confirmed in a much larger group of patients from Tugela Ferry with coinfection rates of HIV with MDR and XDR TB of 90 and 98 percent, respectively (Gandhi et al., 2010). MDR TB 1-year mortality was 71 percent, with a median survival of 60 days; XDR TB 1-year mortality was 83 percent, with a median survival of 28.5 days. Mortality was highest in the 30 days after sputum collection—prior to diagnosis of drug-resistant TB by conventional culture and drug susceptibility testing methods (Gandhi et al., 2010). Because these methods typically take 6–8 weeks to yield a result, the majority of drug-resistant TB patients in this high HIV-prevalence setting die before they are diagnosed
and treated.8 Gandhi suggested that a comprehensive response to the high mortality seen in MDR and XDR TB patients should include
-
prevention of the development of drug resistance through the expansion of DOTS and infection control strategies;
-
implementation of rapid diagnostics to reduce the time to diagnosis from 6–8 weeks to 1–10 days and use of intensified case finding to locate patients at earlier stages of disease; and
-
empiric use of second-line drugs in HIV-infected patients suspected of MDR or XDR TB, as well as integration of antiretroviral therapy into MDR and XDR TB treatment programs to facilitate early and widespread use.
Recent studies have demonstrated that survival decreases with an increasing degree of drug resistance. MDR TB patients with resistance to only isoniazid and rifampicin do better than those with additional first-line drug resistance. Likewise, XDR TB patients with resistance to only four or five drugs do better than those with resistance to all six drugs tested. Further more, resistance is increasing. At Tugela Ferry, the proportion of XDR TB isolates with resistance to six drugs increased steadily and consistently over a 2-year period, with a six-drug pattern being identified among more than 95 percent of patients by 2007 (Moll et al., 2007). When data from a different study performed in 2008–2009 are added, the six-drug phenotype constitutes 100 percent of the XDR TB strains. When the 2008–2009 group was tested for additional resistance to ethionamide and capreomycin, the vast majority of these XDR TB strains were found to be resistant not only to the six drugs previously identified but to all eight drugs tested. Further studies are required to understand the correlates of survival among those patients whose infection is resistant to eight drugs.
Other investigators have found high levels of resistance in the XDR TB seen in KwaZulu-Natal Province (Pillay and Sturm, 2007). Of 52 isolates of the F15/LAM4/KZN XDR TB strains, 100 percent were found to have mutations in the rss gene conferring resistance to streptomycin, kanamycin, amikacin, and capreomycin. In addition, 69 percent were found to have mutations in the pcnA gene conferring resistance to pyrazinamide. Most of the isolates were resistant to isoniazid, rifampicin, ethambutol, pyrazinamide, streptomycin, kanamycin, amikacin, capreomycin, ofloxacin, and ethionamide. That level of resistance leaves very few antibiotics that can be used to treat the disease.
TB AND HIV COINFECTION IN SOUTHERN AFRICA9
TB has become epidemic in the setting of a generalized HIV epidemic in southern Africa, and HIV infection has had a strong influence on the increased numbers of drug-resistant TB cases in the region. As several workshop participants emphasized, where there are high rates of HIV coinfection, the potential exists for the explosive spread of drug-resistant TB. Thus a theme of the workshop presentations and discussions was that understanding the dynamics of the TB epidemic in southern Africa requires knowledge of the ongoing HIV epidemic. Chaisson offered the following metaphor to illustrate the relationship between TB and HIV: if the TB cases seen in hospitals are the tip of an iceberg of TB infection, then the iceberg is floating in an ocean of HIV.
Drug-resistant TB in the context of southern Africa’s HIV epidemic raises many important clinical issues, including
-
the lack of diagnostic capacity to detect TB and perform drug susceptibility testing, with many patients dying before the extent of their drug resistance can be assessed;
-
infection control;
-
TB infections that test negative on sputum smears and extrapulmonary TB;
-
the availability of treatments, including second-line drugs;
-
drug toxicity;
-
treatment with antiretrovirals and the challenges of managing multiple medications and their interactions; and
-
immune reconstitution inflammatory syndrome (IRIS).10
Chaisson noted that default rates (the rate at which patients cease to continue prescribed treatments) in South Africa have been up to 20 percent of patients on second-line anti-TB treatment, with no difference between HIV-positive and HIV-negative groups. A retrospective study of 2,070 patients on standardized MDR TB treatment in nine provinces of South Africa between 2000 and 2004 revealed that patients coinfected with HIV were three times more likely to die than patients without HIV (van der Walt et al., 2007). According to Chaisson, it is not known whether the deaths were due to poor treatment outcomes, including the development of XDR TB, or the failure to receive appropriate therapy.
Looking at South Africa in particular, Chaisson noted that it has made remarkable progress in extending antiretroviral therapy among its population. The current challenge is to meet the global standard of care for providing antiretroviral therapy to TB patients coinfected with HIV, as it has been shown that survival is dramatically improved when antiretrovirals are given during rather than after TB therapy (Abdool Karim et al., 2010).












