2
Uses of Nanotechnology in Oncology and Cancer Research
One of the ways that scientists are working to overcome the shortcomings of current cancer diagnostics and treatments is through the use of nanotechnology, Dr. Barker explained. This chapter demonstrates current uses of nanotechnology in oncology and cancer research as presented by workshop speakers. In turn, diagnosis and monitoring, treatment, prevention, and clinical uses are discussed.
Genetic research has revealed that tumors are not only heterogeneous, but they continue to change with time, she said. For example, the brain tumor glioblastoma multiforme is treated as a single type of cancer, but recent research done by The Cancer Genome Atlas has revealed that there are at least four subtypes of this kind of cancer, and numerous subtypes are being discovered for ovarian and other cancers. The genetic expression of cancers also tends to change as they progress. “As important in understanding what the genome looks like, is how the genome is expressed in space over time as this is really important when you start thinking about delivering agents,” she said (see Figure 2).
Tumors also have numerous traits that make their effective treatment daunting, Dr. Barker pointed out. These traits include self-sufficiency in growth signals, the ability to evade programmed cell death and induce immunologic tolerance, limitless potential to replicate, and the ability to invade tissues and form metastases that can induce the growth of blood vessels to support them.
“If you understand what cancer fundamentally is, what you come to fairly quickly is that we are totally underpowered in terms of being
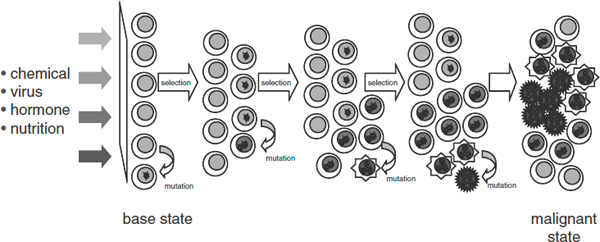
FIGURE 2 Cancer is a complex, evolving system involving chemical, viral, hormonal, and nutritional inputs. Over time, mutation and selection can lead to a malignant state, but there is insufficient biological understanding of these processes over time, according to Dr. Barker.
SOURCE: Barker presentation (July 12, 2010).
able to capture and deliver the kind of information [needed to effectively diagnose and treat cancer] in any of the technologies we currently use, including our chip technologies, because there is a lot of information being managed by cancer when it takes over a normal [biologic] process,” Dr. Barker said.
Nanotechnology has the capacity to deal with the complexity of cancer, she said, by providing tools that can help elucidate what drives cancer initiation and progression; providing tools that can help define the types and subtypes of cancer and combining measurement of cancer biomarkers that can diagnose cancer with therapies that target the specific disease identified by diagnostic measurements; capturing enough information to diagnose cancer at the earliest possible time; for established disease, defining therapeutic targets and directing agents to those target while sparing normal cells; monitoring the effectiveness of an intervention; and sensing pre-neoplastic changes that may benefit from preventive therapy.
“I see nanotechnology as an enabler of pretty much everything we want to do in terms of delivering information to cancer cells, getting information from cancer cells, and combining what we know about normal cells and what we know about cancer cells to be able to differentiate them,” Dr. Barker said.
Dr. Barker then elaborated, as did others, on what nanotechnology is doing or has the potential to do for the diagnosis, monitoring, treatment, and prevention of cancer.
DIAGNOSIS AND MONITORING
To diagnose cancer, physicians rely on imaging that reveals tumors or their linked tissue abnormalities. The detection limit for tumors depends, in part, on the selectivity and the signaling capacity of the contrast material that is used to make them apparent. Increasingly, cancer diagnosis also depends on molecular tests that can discern genes or proteins that are present in abnormal levels. Speakers at the workshop showed how nanotechnology has the potential for improving the diagnosis and monitoring of cancer by enabling high-throughput detection of complex molecular signatures and by enhancing imaging contrast.
Molecular Signatures
Much of modern cancer diagnostics that underlies the new “personalized medicine” approach being taken on the forefront of oncology depends on deciphering complex molecular signatures from blood or tumor samples. But, as Dr. Ferrari pointed out, detecting such cancer-linked molecular signatures in blood is like detecting a needle in a haystack because within a single drop of blood, there can be upward of a million different compounds. Adding to this challenge is the fact that enzymes in blood rapidly degrade the proteins present in a blood sample.
Dr. Ferrari then showed how this challenge is being met by various nanotechnologies, including one developed in his laboratory. In collaboration with Dr. Zhao, Dr. Ferrari has developed silicon chips that are engineered on the nanoscale to have a textured surface with micropores that can separate out proteins by size and charge (see Figure 3). Researchers can use these nanochips to do high-throughput separation of the low molecular weight components of blood proteins from other compounds in a blood sample. This not only enriches the less abundant but more diagnostically significant components of a blood sample, which can later be analyzed using mass spectroscopy, but it eliminates the enzymes that degrade the sample (Sakamoto et al., 2010).
“By taking out all of those compounds that you do not want, it is like taking the sun out of the sky; all of a sudden you can see the stars and it is very facile and quick—it literally takes seconds to perform,” Dr. Ferrari said.
Dr. Barker added that nanotechnology offers opportunities for unprecedented levels of sensitivity and breadth of information, with “bio-barcode” technologies able to detect as little as one molecule of interest in a drop of blood as well as to simultaneously measure hundreds of proteins (see Table 1). “This is an extraordinary leap forward for what we can do with diagnostics, in terms of the numbers of parameters we can
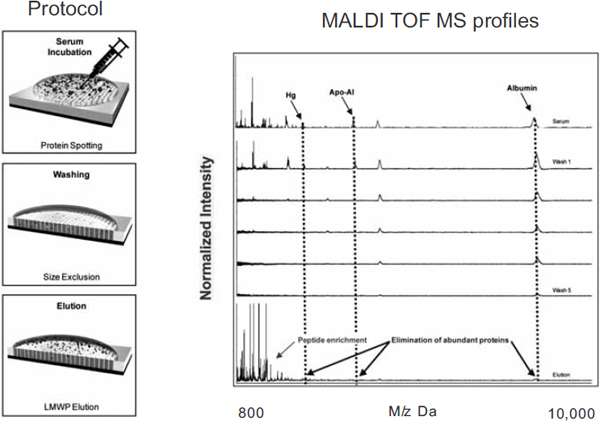
FIGURE 3 Nanotechonology can aid in the development of targeted diagnostics. For example, nanoporous silica films can aide in the identification of molecular signatures through high-throughput separation of low molecular weight components of blood proteins from other compounds in a blood sample.
NOTES: Apo-A1 = apolipoprotein A1; Hg = mercury; LMWP = low molecular weight peptide; MALDI TOF MS = matrix-assisted laser desorption/ionization time-of-flight mass spectrometry.
SOURCE: Ferrari presentation (July 12, 2010).
measure. It potentially gives us, for the first time, the chance to measure [protein] signatures, which is a really big step forward,” said Dr. Barker.
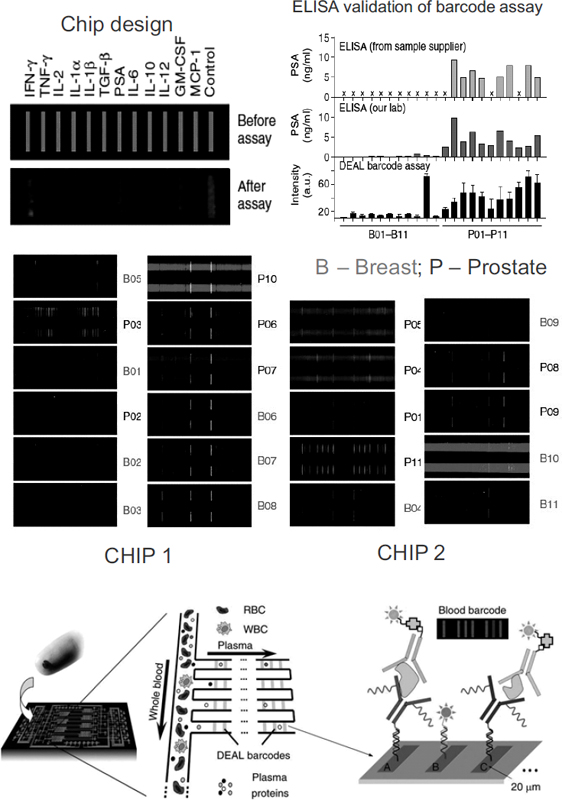
The barcode technologies sieve blood through nano-size channels on chips as small as four centimeters wide. The channels separate the plasma that contains cancer-linked proteins from the blood cells and let it flow down narrower channels that contain an array of bars coated with antibodies or other molecular probes. Each probe will bind to only a specific protein, and fluorescent tags for such binding cause a barcode to light up that indicates the blood’s protein signature (Heath et al., 2009) (see Figure 4).
Researchers tested the ability of this barcode to detect prostate-specific antigen (PSA), which is used to monitor prostate cancers, and found that it could detect minute changes in PSA that were not detected in standard PSA assays.
TABLE 1 Biomolecule Detection Technology
| Concentration | Molecules per Drop | Detection Methods and Targets/Diseases |
| 10−3 – Millimolar | Quadrillions |
Colorimetric/Enzymatic Chemistry Blood Sugar (Diabetes) |
| 10−6 – Micromolar 10−9 – Nanomolar 10−12 – Picomolar |
Trillions Billions Millions |
ELISA and Chemiluminescence Troponin, CK-MB, BNP, βhCG |
| 10−15 – Femtomolar 10−18 – Attomolar 10−21 – Zeptomolar |
Thousands Tens <1 |
Bio-barcode technologies Cancer: prostate, ovarian, breast Alzheimer disease, mad cow disease, pulmonary disease, cardiovascular disease |
NOTES: Nanotechnology offers opportunities for unprecedented levels of sensitivity for high content diagnostics. βhCG = β subunit of human chorionic gonadotropin; BNP = brain natriuretic peptide; CK-MB = creatine kinase MB fraction (the MB fraction is most specific to cardiac muscle); ELISA = enzyme-linked immunosorbent assay.
SOURCE: Barker presentation (July 12, 2010).
“These barcode technologies are really going to set the stage for early detection and are also driving the power of functional imaging of targets, which is one of the earlier wins in the clinic,” Dr. Barker said. “With this technology you can also think about prevention, which we haven’t been able to do in the past.” Dr. Ernie Hawk, vice president and division head for cancer prevention and population sciences at MD Anderson Cancer Center, added that “nanotechnology offers the potential to improve our ability to detect early-stage disease or to assay its progression,” but he noted that it remains to be seen whether nanotechnology screening devices will have the sensitivity and specificity to detect a small collection of cells on a neoplastic pathway.
Barcode technology is likely to be useful in monitoring response to cancer therapies. Dr. James Heath, Elizabeth W. Gilloon Professor and professor of chemistry at the California Institute of Technology, professor of molecular and medical pharmacology at the University of California, Los Angeles, and director of the NanoSystems Biology Cancer Center, showed how his barcode technology was able to reveal, over time, changes in key melanoma-linked proteins in patients undergoing T-cell immunotherapy. These patients just had to provide a pinprick of blood daily for the researchers to capture the change in the dynamic evolution of their protein signatures during the course of therapy.
Enhanced Contrast
Nanotechnology holds promise in improving diagnostic imaging by enhancing the contrast used to do the imaging. “Imaging is one of our earliest wins already,” said Dr. Barker. “We work with places like General Electric and other industries that are using nanotechnology to change everything they are doing about imaging. I think it is going to continue to … change imaging in the years to come.”
Dr. Lee Josephson, associate professor in the Department of Radiology at Harvard Medical School and associate professor with the Center for Translational Nuclear Medicine and Molecular Imaging at Massachusetts General Hospital, showed how magnetic iron-based nanoparticles with fluorescent tags can act as enhanced magnetic resonance (MR) contrast agents and be used for MR-based assays. These nanoparticles can be targeted to tumors by attaching probes for compounds linked to certain cancers, or they can target normal tissue by having probes for receptors found only on normal cells.
Magneto–fluorescent nanoparticles (MFNP) have two main advantages over standard MR contrast agents, according to Dr. Josephson. The magnetic and crystalline nature of these particles heightens their ability to be detected in MR scans. In addition, unlike many fluorescent chelates and dyes conventionally used for contrast, MFNP are internalized by cells
FIGURE 4 In vitro diagnosis and post-therapy monitoring using large-scale, multi-parameter protein analysis in microfluidic devices. (top) Multiplexed protein measurements of clinical patient sera for prostate and breast cancers. The integrated blood barcode chip (IBBC) is used to measure the cancer marker PSA and 11 cytokines from 22 cancer patient serum samples. B01–B11 are samples from breast cancer patients; P01–P11 are samples from prostate cancer patients. (bottom) The IBBC method: plasma is separated from a finger prick of blood using multiple DNA-encoded antibody barcode (DEAL) arrays patterned within microfluidic plasma-skimming channels for multiplex fluorescence detection.
NOTES: B = breast cancer; DEAL = DNA-encoded antibody barcode; GM-CSF = granulocyte-macrophage colony stimulating factor; IBBC = integrated blood barcode chip; IFN-γ = interferon-γ; IL-1α = interleukin-1α; IL-1β = interleukin-1β; IL-2 = interleukin-2; IL-6 = interleukin-6; IL-10 = interleukin-10; IL-12 = interleukin-12; MCP-1 = monocyte chemotactic protein-1; P = prostate cancer; PSA = prostate-specific antigen; RBC = red blood cell; TGF-β = transforming growth factor β; TNF-γ = tumor necrosis factor γ; WBC = white blood cell.
SOURCES: Barker presentation (July 12, 2010) and Fan et al. (2008). Adapted by permission from Macmillan Publishers Ltd: Nature Biotechnology 26(12):1373–1378, copyright 2008.
and are not rapidly metabolized, so they are retained long enough that they can be used for both pre-surgery imaging, as well as during surgery (intravital) to detect tumor margins (see Figure 5).
“If you inject MFNP 6 or even 12 hours prior to an operation, you will be able to see where they are intraoperatively,” Dr. Josephson said.
Dr. Kristen Kulinowski, senior faculty fellow in the Department of

FIGURE 5 Measurement of nanoparticle fluorescence to determine tumor margin. The Cy5.5-labeled nanoparticles were injected in the mouse model prior to surgery and were used as a contrast agent for magnetic resonance imaging. Images like the one above can then be taken intraoperatively for use in tumor border determination. This image compares tumor border determination using CLIO-Cy5.5 and green fluorescent protein (GFP). The tumor border was determined using signal intensity measurements.
NOTE: CLIO-Cy5.5 = cross-linked iron oxide–Cy5.5.
SOURCES: Josephson presentation (July 12, 2010) and Trehin et al. (2006). Reprinted, with permission, from Neoplasia, 2006. Copyright 2006 by Neoplasia Press.
Chemistry at Rice University and Director for External Affairs for the Center for Biological and Environmental Nanotechnology, added that researchers at Rice University have already started clinical trials using silica–gold nanoshells as real-time molecular probes for breast tissue that overexpresses the breast cancer biomarker HER2. The nanoshells are added to tissue slices removed during breast cancer excision surgery and within 5 minutes can be detected with an optical imaging system (Bickford et al., 2010). “All this could be done very rapidly while the patient was still on the operating table, rather than having to rely on postoperative follow-up and retreatment,” Dr. Kulinowski said. Dr. Josephson is currently exploring MFNP for measuring, during surgical removal of tumors, biomarkers that indicate the aggressiveness of cancer. “The idea is to look in aspirates of tumors for various biomarkers, such as growth factors, that would help the surgeon decide intraoperatively how aggressive is the cancer, instead of waiting for the report,” he said.
Dr. Steven Curley, professor of surgery, chief of gastrointestinal tumor surgery, and program director of multidisciplinary gastrointestinal care at MD Anderson Cancer Center, pointed out that gadolinium-loaded carbon nanostructures or gold-coated nanoparticles also can be used as contrast agents for MR and provide more detail than standard contrast agents. “The imaging characteristics and the ability to see things on a much finer scale will definitely be enhanced [with these nanoparticles],” he said.
Another advantage to gold nanoparticles is that they can be used to both diagnose and treat tumors. This is an example of theranostics, and it can be done by first using the particles to target the tumors, and then applying a selective energy source, such as a laser, that is readily absorbed by the gold nanoparticles but not by normal tissues. The heat created by that absorption kills the tumor cells.
In his studies, Dr. Curley found that even at very low concentrations, gold nanoparticles produce significant levels of heat when exposed to a very focused radiofrequency field. The production of heat by the exposed nanoparticles was not only concentration dependent, but also size dependent, with smaller nanoparticles leading to faster heating rates, given a constant volume fraction of gold, Dr. Curley reported. He was able to completely control the tumors in animal models using this nanotechnology, without damage to their normal tissues. “This has the potential to be a targeted therapy with few if any side effects,” he said. He added that investigators at Rice are currently using a similar thermal treatment using gold-coated nanoshells to treat oropharyngeal cancer in a clinical trial.
MR imaging using nanoparticles for contrast can also be done to do imaging assays of blood or other solutions. The advantage of this assay method stems from the penetrating radiofrequencies used in MR. “It allows us to have molecular readouts from solutions that are completely
refractory to light,” Dr. Josephson said. “You can’t do this with an ELISA or with fluorescent assays.”
Another advantage of doing MR assays is that the assay can be interrogated with different pulse sequences, so one can assay for more than one variable. MR assaying systems have been developed that are compact, inexpensive, and portable. One micro MR imager can image ten microliter wells simultaneously, Dr. Josephson reported, and is about the size of an old cell phone. No separations are needed to do the imaging, and it can detect all kinds of molecular targets and correct for unknown reagent concentrations and viscosity.
Dr. Josephson also discussed MR nanosensors. In collaboration with Michael Cima at MIT, Dr. Josephson developed a nanosensor for human chorionic gonadotrophin (hCG), which is produced by some tumors. The sensor detects the aggregation of particles caused by the binding of the hCG probe. The sensors have some of the same advantages as the MR assays—they can detect multiple compounds with simple instrumentation, and, particularly relevant to sensors, they emit penetrating radiofrequency radiation, but have no power supply.
“In other words, what is implanted in the animal has no battery. The energy comes from the external NMR,” Dr. Josephson said. This would enable the sensor to be implanted in an animal to detect substances released by tumors. He added that unlike blood tests that measure a cancer biomarker at a single moment in time, implantable sensors could measure the concentration of various biomarkers over time.
Quantum dots are another type of nanomaterial with versatile properties. Dr. Curley noted that quantum dots can function as optical imaging agents both for in vitro and in vivo blood testing, to track molecules, to show lymph node involvement for various cancers, and to image recurrent or residual infectious diseases.
TREATMENT
Several speakers showed how nanotechnology is likely to improve cancer treatment by improving its targeting precision. Many cancer drugs cause serious and sometimes fatal side effects because they are spread systemically throughout the body, where they do damage to healthy tissues. Such damage can be limited by more specific targeting to tumor cells.
The targeting can be passive and due to the physical properties of nanomaterials that enable them to penetrate tumor cells from the bloodstream, as previously described, or be active targeting due to being decorated with antibodies or other compounds that cause them to selectively bind to tumor cells. Selectivity can also be achieved by drugs encased in
nanoparticles that do not release their contents until they penetrate tumor cells.
Nanomedicines already on the market to treat breast or ovarian cancer do such specific targeting by encasing the conventional cytotoxic cancer drugs, such as Taxol, in albumin or liposomes, which are designed not to release their toxic contents until they enter tumor cells, thereby shielding healthy cells from their toxic effects. Dr. Neil Desai, senior vice president for global research and development at Abraxis Bioscience, reported that clinical trials of Abraxane, which is Taxol encased in albumin nanoparticles, found that that the maximum tolerated dose was about twice that for Taxol alone, and that breast cancer response rates of Abraxane were double that of Taxol. The drug was approved to treat breast cancer in 2005 and has since been shown in clinical trials to be an effective treatment for patients with pancreatic or lung cancers, or melanoma.
Researchers are also pursuing other nanoconstructs that shield healthy tissue from their toxic contents. Dr. DeSimone noted that his lab had created what he called “Trojan horse” nanoparticles that are pH-sensitive and chemically constructed to breakdown only in the intracellular environment. Such breakdown triggers the release of the drugs they carry.
Other nanoparticles have shown to be so selectively taken up by tumor cells, by both passive and active means, such that researchers can use higher and more effective doses of the cancer drugs they contain. For example, tumor necrosis factor (TNF) had been shown effective as a cancer treatment in limited limb perfusions, but had to be abandoned as a systemic treatment because of toxic reactions to the high enough doses needed to be effective. But with the advent of nanoparticles that are selectively taken up by tumor tissues, as opposed to healthy tissues, larger doses can now be safely used systemically, Dr. Steven Libutti, director of the Montefiore–Einstein Center for Cancer Care and professor and vice chair of surgery at Albert Einstein College of Medicine at Yeshiva University, reported. His research showed that he was able to safely administer tumor necrosis factor delivered via gold nanoparticles to melanoma patients at what was previously considered to be a lethal dose level of the compound (twice the LD50).
Consequently, reformulation of discontinued drugs is a growing area of nanomedicine development, Dr. McNeil noted. “Big pharma can produce tens of thousands, if not hundreds of thousands, of new chemical entities by medicinal chemistry,” he said. “By far, the majority of those have to be disqualified due to insolubility or toxicity and so forth. So something that has been postulated is that nanotechnology might be able to resurrect some of those drugs, because we can truly engineer properties into and out of that formulation.”
Nanomedicines have also been developed that not only specifically
target tumor cells, but the cancer-promoting genes they contain as well. Clinical trials of a nanomedicine that target specific RNAs are already underway, Dr. Barker pointed out. This nanoparticle contains silencing RNA that penetrates tumor cells via endocytosis (Davis et al., 2010).
Researchers can also create nanoparticles that have bigger payloads—multiple drugs, each with a different target, or drugs combined with agents that enhance their effectiveness. Nanoparticles can also have multiple functions. Some combine drugs with contrast agents, while others might someday be engineered to treat, monitor the effectiveness of treatment, and then re-treat if the treatment is not working, Dr. Barker noted. Dr. Li added that researchers also envision engineering “remote controlled drugs” that can be released or activated only when needed.
Nanotechnology has immense potential to further personalized medicine—defined as the use of new molecular technologies to get the right treatments to the right patients at the right time—many speakers noted. Dr. Ferrari pointed out that by using nanoparticles, researchers can personalize vectors not just to the patient but to the specific type of lesion the patient has, down to the subcellular level, in terms of which organelles it targets or which sections of RNA or DNA. This specialized targeting is “built into the physics and chemistry of the particles,” he said, which can also determine both where and when therapeutic drugs are released.
Dr. Barker noted that the multiplexing capabilities of nanomedicines offers the possibility of targeting the many and diverse genetic defects that underlie specific cancers, as well as combining lesion detection with drug delivery and monitoring of the drug’s effectiveness. “Personalizing means getting the bioactive molecules that you want at the right place at the right time, finding out whether they work pretty quickly, and engaging the biology into some sort of a natural healing process that is better than was present before the administration of the nano drug,” Dr. Ferrari added.
Dr. Barker concurred adding, “We’re developing a field that is actually looking at the interplay of whatever we’re administering with the cells that we’re interested in. And we’re doing that in ways we never did before. Why is that? It’s because we have the capability of nanostructures to do that. Right now we throw some small molecules into circulation and hope they get there, and generally they don’t. So I think this is an area where if you functionalize these particles and have the right delivery vehicle, you [can do better],” Dr. Barker said.
But Dr. Ferrari cautioned against overdecorating nanoparticles with compounds that target specific tumor cells or making their payloads too extensive because the more complex nanoparticles become, the less likely they may be to overcome the biological barriers that can prevent
them from reaching their targets. This concept is discussed further in the section Design Complexity of Nanomaterials for Medical Applications in Chapter 3.
PREVENTION
Many of the advantages nanotechnology provide for treatment and diagnostics are likely to be also relevant to the prevention of cancer, Dr. Hawk pointed out, including the ability to have larger payloads and deliver a combination of agents. Studies (McLaren et al., 2008; Meyskens et al., 2008) show that two compounds, such as DFMO (difluoromethylornithine) and Sulindac, can be more effective than either agent alone at reducing colon cancer recurrence, he said. “This and a great deal of other clinical work leads me to believe that using nanotechnology as a combinatorial platform will be as relevant to prevention as it is in therapy,” Dr. Hawk stressed.
But does the leakiness of tumor blood vessels, which enables passive transport of nanomaterials into cancer cells, occur in preinvasive lesions, and thus become relevant to prevention efforts using nanoparticles? This is not known yet, but is actively being explored, Dr. Hawk reported. Such selective targeting would be an advantage for compounds such as epigallocatechin-3-gallate (EGCG), which is found in green tea and appears to have some cancer-preventing properties, but has poor oral absorption, with few people consuming green tea in high enough quantities to reap the compound’s cancer-preventing benefits.
One research lab at the University of Wisconsin created a nanoparticle that could deliver high doses of EGCG. They found, in an animal model, that there was efficient uptake of the nano-delivered EGCG by prostate cancer cells, where it induced programmed cell death, inhibited the formation of blood vessels, and decreased tumor volume (Siddiqui et al., 2009). In addition, the nanoEGCG was as effective as a tenfold higher dose of EGCG delivered by standard means in a mouse xenograft model using prostate cancer cells (Siddiqui et al., 2009).
Researchers are also currently developing nano versions of nonsteroidal anti-inflammatory drugs (NSAIDs) that could mitigate the adverse effects of these drugs without compromising their protective properties, which include preventing gastric cancers of the gastrointestinal tract, Dr. Hawk added.
“Are we there yet in terms of nanotechnology impacting cancer prevention? I don’t think so. However, there are very important endeavors underway right now to try to expand the potential usefulness of this exciting technology in screening and prevention,” Dr. Hawk concluded.
NANOTECHNOLOGY IN THE CLINIC
Although many of the applications of nanomedicine described at the workshop are in preclinical stages, and some are still in proof-of-principle stages, several participants stressed that nanotechnology is already being applied to the clinic.
“Nanotechnology is a very real field. It is not science fiction, a ‘let’s see what happens in the future’ type of field,” Dr. Ferrari said. He pointed out that one of the first nanomedicines—the liposomal cancer medicine Doxil—has been used in the clinic for over 15 years, and in addition to the dozens of different nanotechnology approaches that are currently being tested, many clinical trials are testing agents that have already been approved, such as liposomes with doxorubicin in combination with other drugs.
During his presentations, Dr. Li showed a list of two dozen either approved nanotechnology cancer drugs or potential nanotechnology cancer drugs currently in clinical trials, which he said was just a partial list of all the nanomaterials being used in the clinic, and did not include Dr. Libutti’s nanoTNF, which is currently being tested in a clinical trial (see Table 2). In addition, Dr. Barker listed one nanotechnology imaging agent that has conditional FDA approval (iron oxide nanoparticles) and one in preclinical development (PAMAM dendrimers for MRI imaging).
TABLE 2 A Partial List of Nanotechnology Drugs Currently in Clinical Trials
| Compound | Name | Indication | Status |
| Liposomal doxorubicin | Myocet, Caelyx (Doxil) | Breast, ovarian, KS | Approved |
| Liposomal daunorubicin | Daunoxome | Kaposi sarcoma | Approved |
| Liposomal vincristine | Onco-TCS | Non-hodgkin lymphoma | Approved |
| Liposomal cisplatin | SPI-77 | Lung | Phase II |
| Liposomal lurtotecan | OSI-211 | Ovarian | Phase II |
| Cationic liposomal c-Raf AON | LErafAON | Various | Phase I/II |
| Cationic liposomal E1A pDNA | PLD-E1A | Breast, ovarian | Phase I/II |
| Thermosensitive liposomal doxorubicin | ThermoDox | Breast, liver | Phase I |
| Albumin-paclitaxel | Abraxane | Breast | Approved |
| Albumin-methotrexate | MTX-HSA | Kidney | Phase II |
| Dextran-doxorubicin | DOX-OXD | Various | Phase I |
| PEG-L-asparaginase | Oncaspar | Leukemia | Approved |
| PEG-IFN2a/-IFN2b | PegAsys/ PegIntron | Melanoma, leukemia | Phase I/II |
| PHPMA-doxorubicin | PK1 | Breast, lung, colon | Phase II |
| Galactosamine-targeted PK1 | PK2 | Liver | Phase I/II |
| PGA-paclitaxel | Xyotax | Lung, ovarian | Phase III |
| Paclitaxel-containing polymeric micelles | Genexol-PM | Breast, lung | Phase II |
| Cisplatin-containing polymeric micelles | Nanoplatin | Various | Phase I |
| Doxorubicin-containing polymeric micelles | NK911 | Various | Phase I |
| SN38-containing polymeric micelles | LE-SN38 | Colon, colorectal | Phase I |
| 90Yttrium-Ibritumomab tiuxetan (α-CD20) | Zevalin | Non-hodgkin lymphoma | Approved |
| DTA-IL2 fusion protein (α-CD25) | Ontak | T-cell lymphoma | Approved |
| Compound | Name | Indication | Status |
| Ozogamycin-gemtuzumab (α-CD33) | Mylotarg | Leukaemia | Approved |
| Doxorubicin-cBR96 (α-CD174) | SGN-15 | Lung, prostate, breast | Phase II |
NOTES: α-CD20 = anti-CD20, CD20 is cluster of differentiation 20, a cell surface protein; α-CD33 = anti-CD33, CD33 is cluster of differentiation 33; DOX-OXD = dextran conjugated doxorubicin; Doxorubicin-cBR96 (α-CD174) = doxorubicin conjugated to chimeric monoclonal antibody cBR96 (anti-CD174, CD174 is cluster of differentiation 174, a cell surface protein); DTA-IL2 fusion protein (α-CD25) = fusion protein of diphtheria toxin fragment A and interleukin 2 (this fusion protein targets CD25, a cell surface protein); Genexol-PM = Genexol–polymeric micelle; KS = Kaposi sarcoma; LE-SN38 = liposome-encapsulated 7-Ethyl-10-hydroxy-camptothecin; LErafAON = liposome encapsulated c-raf antisense oligonucleotide; MTX-HSA = human serum albumin–bound methotrexate; NK911 = polymeric micelle carrier system for doxorubicin; Onco-TCS = Onco-transmembrane carrier system, the drug vincristine; OSI-211 = liposomal lurtotecan drug manufactured by OSI Pharmaceuticals; PEG-IFNα2a/-IFNα2b = pegylated interferon α-2a/interferon α-2b; PEG-Lasparaginase = polyethylene glycol conjugated asparaginase; PGA-paclitaxel = polyglutamic acid conjugated paclitaxel; PHPMA-doxorubicin = poly(2-hydroxypropyl methacrylate) conjugated doxorubicin; PK1 = N-(2-hydroxypropyl)methacrylamide copolymer doxorubicin; PK2 = N-(2-hydroxypropyl)methacrylamide (HPMA) copolymer backbone and pendant doxorubicin (DOX) linked via a Gly-Phe-Leu-Gly peptide spacer; PLD-E1A = pegylated liposomal doxorubicin–linked E1A (an adenoviral oncogene) plasmid DNA; SGN-15 = cBR96-doxorubicin (see above) immunoconjugate, SGN stands for Seattle Genetics Inc.; SPI-77 = sterically stabilised liposomal cisplatin.
SOURCES: Li presentation (July 12, 2010) and Lammers et al. (2008). Reprinted by permission from Macmillan Publishers Ltd: British Journal of Cancer 99(3), copyright 2008.
















