2
Assessing Risk for Obesity in Young Children
GOAL: Assess, monitor, and track growth from birth to age 5.
This chapter examines the importance of growth monitoring from birth to assess the risk for obesity in young children. It also reviews prenatal factors that may influence that risk.
Infants and young children are weighed, and their length or height is recorded as part of routine well-child visits to the pediatrician or other health care provider. These visits offer the earliest opportunity to track children who are at risk of overweight or obesity and provide guidance to parents at an early stage of rapid weight gain so they can take preventive action. Weight-for-length or weight-for-height measurements need to be performed accurately, and updated guidelines should be used for the assessment. Updated guidelines from the Centers for Disease Control and Prevention (CDC) and the American Academy of Pediatrics (AAP) include using the World Health Organization (WHO) growth charts for children from birth to age 23 months and the CDC growth charts for ages 2 to 5 years; the CDC growth charts can be used to calculate and plot body mass index (BMI). The focus of a child’s visits to the health care provider should not be just
TABLE 2-1 Weight Statuses and Corresponding Percentiles
| Age (years) | At Risk for Overweight | Overweight | Obese |
| 0–2 (WHO charts) | 84.1st to 97.7th percentile | >97.7th percentile | N/A |
| 2–5 (CDC charts) | N/A | 85th to 95th percentile | >95th percentile |
| NOTE: N/A = not applicable. | |||
on healthy growth, but also on identifying excess weight-for-length or -height and monitoring the trajectory of change in weight-for-length, weight-for-height, or BMI-for-age. Health care providers should identify children less than 2 years of age as at risk for overweight if their growth measurements are between the 84.1st and 97.7th percentiles on the WHO charts, and as overweight if their measurements exceed the 97.7th percentile. Using the CDC charts, they should identify children aged 2–5 years as overweight if their measurements are between the 85th and 95th percentiles, and obese if their measurements exceed the 95th percentile (see Table 2-1).
Parents should be given appropriate information, suggestions, and referrals to identify behaviors putting the child at risk. In general, both health care practitioners and parents need to understand that “bigger is not always better.”
The training of pediatricians, nurses, and others who work with children needs to include basic obesity prevention. In the end, preventing childhood obesity requires early intervention. The environments in which children spend their time and information provided to parents on nutrition, activity, and sleep are critical to ensuring children’s well-being. When a child is at risk or already overweight or obese, the problem needs to be recognized in the early stages by all health care providers. Health care providers and programs that provide guidance to parents, such as the Special Supplemental Nutrition Program for Women, Infants, and Children (WIC), are best positioned to inform parents about obesity prevention for young children.
The Role of Health Care Professionals
Parents view pediatricians, primary care physicians, and health care providers as having the most authority in offering advice about childrearing (McLearn et al., 1998; Moseley et al., 2011). Parents interact with pediatricians and other health care providers in the early stages of child growth more than with any other pro-
viders (McLearn et al., 1998), except perhaps their daily child care provider. They seek advice on feeding, sleep, activity, and other aspects of early childhood behavior from health care providers, which creates the opportunity to inform parents about a range of factors that impact excess weight gain in the early years of life.
Well-child visits are standard visits at which the health care provider assesses and monitors the child’s health and growth. Usually eight visits occur at set intervals throughout the first 2 years of a child’s life. During these visits, children often are measured for length and weight, and this information is plotted on growth charts. This measurement should occur at every well-child visit. And while emphasis has historically been placed on identifying undernutrition or a lack of growth, equal attention needs to be given to excess weight-for-length, which is the measure of overweight in the first 2 years of life. After 2 years of age, children routinely visit health care providers for continual assessment of their growth. Although height and weight are almost always recorded during health maintenance visits, BMI calculations after age 2 are performed less consistently (Klein et al., 2010). To assess weight gain accurately, health care providers should consistently calculate BMI values and plot them on CDC’s gender-specific BMI-for-age charts.
In addition to monitoring the child’s growth, health care providers are in a position to observe and ask about the family environment. Observations of parental weight, discussions of childhood activities and family eating patterns, and clinical assessments of weight-for-length or -height can provide valuable information on the child’s health and the potential risk for later obesity.
Health care professionals and pediatricians are best positioned to identify excess weight in young children. The interaction between parents and health care providers gives parents an opportunity to become aware of their child’s excess weight early on to allow time for intervention and prevention.
Misperceptions of Excess Weight
Because parents and other caregivers have complete control over their young children’s food intake, it is important that parents understand the growth patterns and the significance of excessive weight gain during the first few years of life. However, studies show that many parents in fact do not understand the consequences of or are not concerned about early overweight or obesity in their children. In focus groups conducted with WIC mothers, some mothers expressed the belief that it was healthy for their babies to be overweight (Baughcum et al., 1998). The overweight mothers in the focus groups believed that their children were overweight because they were genetically prone to be so; therefore, the
extra weight was viewed as natural and not problematic (Baughcum et al., 1998). Mothers also tend to underestimate their children’s weight status, even when they correctly recognize overweight in themselves (Baughcum et al., 2000). Studies in Mexico and Greece showed that mothers who underestimate their children’s weight tend to have less income and education (Jimenez-Cruz et al., 2010; Manios et al., 2010); conversely, a similar study in The Netherlands revealed that this misperception exists regardless of mothers’ education level (Jansen and Brug, 2006). Of interest, Huang and colleagues (2007) found that parents are less likely to correctly identify overweight in their own children than in children who are unrelated to them. The researchers hypothesized that parents judge the weight of their own children using a different standard (Huang et al., 2007). These misperceptions can be corrected with the objective input of a health care professional. By helping parents understand the growth charts, health care professionals can give parents a tool with which to compare their children’s weight status and growth pattern objectively with those of a healthy reference population.
Updated Guidelines for Measuring Children
As noted above, the committee believes child growth and weight should be measured at every well-child visit. For this purpose, health care providers should use the updated CDC guidelines, which specify the WHO growth charts for the first 2 years of life and the CDC growth charts for ages 2–5. BMI should be calculated from the growth charts for children aged 2–5.
Recommendation 2-1: Health care providers should measure weight and length or height in a standardized way, plotted on World Health Organization growth charts (ages 0–23 months) or Centers for Disease Control and Prevention growth charts (ages 24–59 months), as part of every well-child visit.
Rationale
Until recently, growth data for children from birth to age 2, as well as for children over age 2, were plotted on 2000 CDC growth charts. The data used to generate the curves in CDC’s growth charts for ages 0–2 were collected from secondary sources and included information on infants raised in various health environments in the United States (Grummer-Strawn et al., 2010). On the other hand, the data for the WHO growth charts were collected from a large cohort of children from birth to age 2 living in various cultures who were raised in an optimal health
environment. These infants were breastfed for at least 12 months, introduced to complementary food around 6 months of age, and raised by mothers who did not smoke and in households of adequate income (de Onis et al., 2004). In addition, a cohort was measured longitudinally from birth to age 2 years, and weight and length velocity,1 along with weight, length, and BMI standard values, are available for this cohort (de Onis et al., 2004). Therefore, the WHO growth charts should be used for children aged 0–2.
On the other hand, the data collection for the growth curves for children over age 2 years was similar for both CDC and WHO. Therefore, the CDC growth charts should continue to be used for children 24-59 months of age (Grummer-Strawn et al., 2010). These standard or reference growth charts can be embedded in an electronic medical record and values of BMI (wt/ht2, kg/m2 [http://www.cdc.gov/nccdphp/dnpa/bmi/calc-bmi.htm]) and weight velocity calculated and plotted electronically along with the measured values. In addition to the growth charts that record weight-for-height data, children aged 2 and older should be assessed using the BMI calculator for children and teens. These data should be plotted on age- and gender-specific BMI charts to monitor growth.
A survey of primary care physicians and pediatricians conducted by the AAP found that while 99 percent of survey respondents measure and plot height and weight data for children during well-child visits, only 52 percent calculate BMI percentiles for children aged 2 years and older (Klein et al., 2010); only 45 percent calculate BMI percentiles at most or every well-child visit (Sesselberg et al., 2010). In a study of an academic pediatric practice, Hillman and colleagues (2009) found that only 59.7 percent of pediatric medical records contain CDC’s BMI-for-age growth charts and that resident physicians are more likely than attending physicians to document and plot BMI data for their patients. These statistics emphasize the need for physicians to be trained to consistently calculate BMI percentiles and plot them on growth charts to monitor children’s growth.
Major curves on the WHO charts are the 2.3rd, 15.9th, 50th, 84.1st, and 97.7th percentile curves. A child who is 2 years old or younger is considered overweight when he or she exceeds the 97.7th percentile on the WHO charts. The child should be identified as at risk of becoming overweight if his or her measurement crosses above the 84.1st percentile curve (Figure 2-1).
BMI calculations are not done for infants or children under the age of 2 years. In children aged 2 years and older, measurement of age- and gender-specific
_____________
1Defined as the rate of change in growth measurements over time.
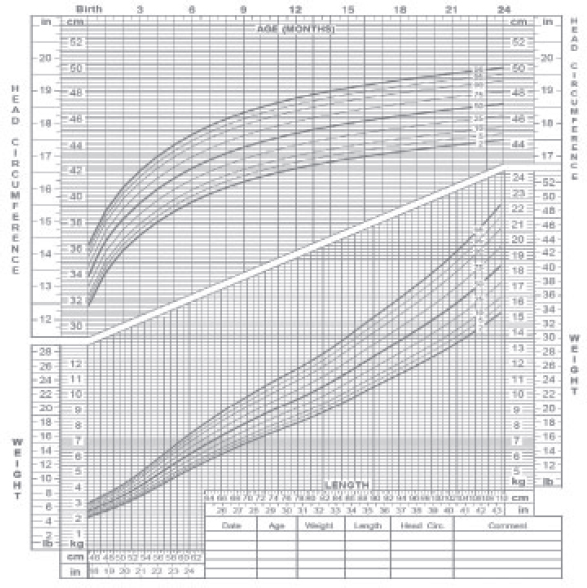
FIGURE 2-1 Weight-for-length growth chart for girls.
SOURCE: CDC, 2010.
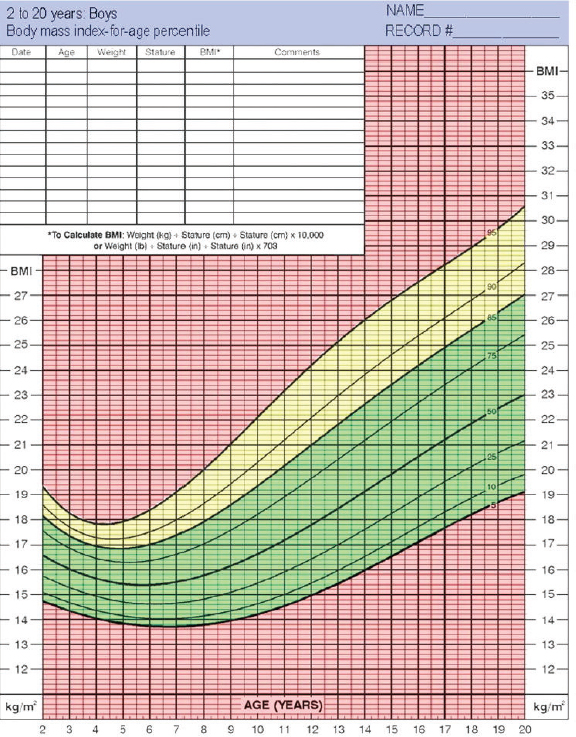
BMI can provide an indication of overweight or obesity. As shown in an example of the color-coded BMI chart (Figure 2-2), children whose age- and gender-specific BMI measurements fall in the green zone, which is between the 5th and 85th percentiles, are considered to be of healthy weight. Those whose measurements exceed the 95th percentile or fall below the 5th percentile, both red zones, are obese or underweight, respectively. And children whose BMI falls in the yellow area, between the 85th and 95th percentiles, are consid-
ered overweight. In all children, physicians and parents should begin preventive intervention when the growth measurements exceed the 84.1st or 85th percentile curve. This would require that physicians and parents understand and consistently use the growth charts.
Recommendation 2-2: Health care professionals should consider (1) children’s attained weight-for-length or body mass index at or above the 85th percentile, (2) children’s rate of weight gain, and (3) parental weight status as risk factors in assessing which young children are at highest risk of later obesity and its adverse consequences.
Rationale
Documenting and plotting BMI data on charts can help physicians see trends in children’s growth and detect early signs of overweight and obesity, allowing intervention before children become overweight or obese. Use of growth charts to plot BMI data is especially important as physicians can correctly identify the weight status of children visually only about half of the time (Huang et al., 2009). However, although many children are routinely measured for weight and length or height, there is often no follow-through in identifying those who are at risk of overweight or obesity once that information has been collected. A review of children’s medical records showed that even among those whose BMI categorized them as severely obese, only 76 percent were given this diagnosis by their physician; even fewer children were diagnosed if their BMI indicated that they were just overweight (10 percent) or obese (54 percent) (Benson et al., 2009). A review of outpatient preventive care visits by children with high BMIs likewise indicated that physicians severely underdiagnosed obesity in those patients (Patel et al., 2010). Although the lack of a diagnostic code is not necessarily the same as the lack of recognition and discussion of a condition, these studies indicate that physicians often miss the opportunity to identify children who are at risk of obesity before they become obese. Thus it is critical that physicians recognize the early signs of obesity. They should be trained to follow the guidelines of CDC and WHO, which indicate that children are overweight when their measurements exceed the two upper percentile curves on the growth charts. The committee believes it is important that the standard for risk be both a percentile and the amount of weight gain.
It can be argued that measurement and plotting of growth, and even a diagnosis of obesity, will not be sufficient if physicians fail to follow through on these results. In fact, a survey conducted by Jelalian and colleagues (2003) found that
one-fourth of physicians perceive themselves as being “not at all or only slightly competent” to treat obesity. Additionally, the collaborative community-based intervention and prevention programs that could be helpful to these physicians are not always available to primary care providers (Pomietto et al., 2009). Children grow rapidly in the early years of life. In the first year alone, most healthy infants triple their birth weight (Lightfoot et al., 2009). However, excessively rapid growth in weight-for-length or -height can indicate behaviors that put children at risk for obesity, even if the excess weight may not yet be apparent. Abundant epidemiologic evidence from the developed world now shows that more rapid weight gain during the first half of infancy predicts later obesity and cardio-metabolic risk (Gillman et al., 2008). Previous studies of both contemporary (Dennison et al., 2006; Hui et al., 2008; Taveras et al., 2009) and historical (Stettler et al., 2002) cohorts and two recent systematic reviews of infant growth and obesity (Baird et al., 2005; Monteiro and Victora, 2005) have concluded that infants at the highest end of the weight distribution and those who grow most rapidly (usually measured as weight gain) are more likely to be obese later in life. The review by Baird and colleagues (2005) found that infants who gain weight rapidly in the first year of life are 1.17 to 5.70 times more likely to develop later obesity than infants who do not gain weight rapidly.
Because health care professionals routinely document serial measures of weight and length and screen for abnormalities in weight status using published growth charts, practical tools based on the growth charts can be useful in assessing risky weight gain in infancy even before children reach the cutoffs for overweight or obesity.
In a study of height and weight data for more than 44,000 children from ages 1 to 24 months and at ages 5 and 10 years, Taveras and colleagues (in press) examined the association between upward crossing of major percentiles in weight-for-length in the first 2 years of life and prevalence of obesity at ages 5 and 10 years. “Major” percentile was defined as the 5th, 10th, 25th, 50th, 75th, 90th, and 95th percentiles on the CDC growth charts. The authors found that crossing upward of two or more major weight-for-length percentiles in the first 24 months of life was associated with later obesity. For example, the odds of obesity at age 5 were 2.08 (95 percent confidence interval [CI]: 1.84, 2.34) and at age 10 were 1.75 (95 percent CI: 1.53, 2.00) among children who had ever crossed upwards of two or more versus fewer than two major weight-for-length percentiles in the first 24 months of life. Additionally, upward crossing of two major weight-for-length percentiles in the first 6 months was associated with the highest prevalence of
obesity 5 and 10 years later. Thus, both total weight gain of 8.15 kg from 0 to 24 months and crossing upwards of two or more major weight-for-length percentiles in the first 24 months of life could be used to assess the risk of later obesity in pediatric primary care for infants and children under the age of 2 years.
Even if total weight gain or crossing of weight-for-length percentiles is used by health care professionals to identify excess weight gain in infancy, it is still necessary to identify modifiable determinants of excess gain in adiposity and determine what the proper response should be (Gillman, 2010). A robust literature has emerged regarding pre- and perinatal predictors of childhood adiposity (Oken et al., 2007, 2008; Taveras et al., 2006, 2008, 2009; Wright et al., 2009), but few studies have examined whether these factors also predict weight gain in early infancy. Furthermore, there is a need to examine trade-offs between more and less rapid weight gain for different outcomes. At least among infants born preterm, more rapid weight gain in early infancy predicts better neurocognitive outcomes in childhood (Casey et al., 2006; Ehrenkranz et al., 2006). Whether this holds true with term infants is less clear (Belfort et al., 2008). Thus, the amount of weight gain that optimizes both neurocognitive outcomes and obesity risk may differ by gestational age. Investigating these potential determinants of excess infant adiposity gain could lead to intervention strategies in clinical and public health settings to prevent childhood obesity and its consequences.
The family and physical environment in which children grow up can increase their obesity risk. Noting parental weight status can assist in assessing the risk of later obesity in children and should therefore be included in the routine health assessment of young children. Health care providers also can identify risk for obesity by asking about family history and assessing the immediate environment in which children spend their time.
Research shows that parental BMI is the strongest predictor of obesity in young adulthood (Maffeis et al., 1998; Whitaker et al., 1997). In children aged 1–5 years, having an obese mother increases the odds of developing obesity in their 20s by 3.6; having an obese father increases the odds by 2.9 (Whitaker et al., 1997). Having two obese parents increases the odds of later obesity by 13.6 in children aged 1–2 and by 15.3 in those aged 3–5 (Whitaker et al., 1997). Using multiple analyses, Maffeis and colleagues (1998) found that both mothers’ and fathers’ BMI status are the strongest predictor of obesity in their children. This finding has been confirmed by other studies on the influence of parental BMI on children’s risk of adult obesity (Abu-Rmeileh et al., 2008; Williams, 2001).
A number of plausible biological pathways link a mother’s prepregnancy and prenatal status to obesity in her offspring. Knowing more about a mother’s prepregnancy weight, gestational weight gain (GWG), diabetes and metabolism during pregnancy, and smoking during pregnancy can, along with growth monitoring and well-child visits, provide important hints in assessing a child’s risk of developing obesity. For each of these four factors, compelling health considerations other than their possible link to childhood obesity guide clinical recommendations and public health policy. And although uncertainty exists regarding the direct and independent causal influence of these factors on obesity in children, some varying level of evidence supports that possibility.
Prepregnancy Weight
Clear and consistent observational evidence indicates that women who are of higher weight at the onset of pregnancy have infants and children who are more likely to be obese (Oken, 2009; Whitaker, 2004; Whitaker and Dietz, 1998), with a strong dose-response gradient between the magnitude of the mother’s excess weight and that of her child (Whitaker, 2004). In the papers reviewed for a meta-analysis by Oken (2009), the magnitude of the association varied, but it tended to be quite strong (relative risks of 4 or greater) for the more extreme measures of childhood obesity and somewhat weaker for less extreme levels of childhood overweight. Maternal obesity was predictive of childhood obesity in general, most strongly for the more extreme levels of childhood obesity. The association was apparent for children in the age range of relevance to this report, under age 5, as well as for older children, adolescents, and adult offspring. A recent report on 9-year-old children from the Avon Longitudinal Study of Parents and Children (ALSPAC) cohort showed not just elevated BMI associated with elevated maternal weight prior to pregnancy, but also an array of indicators of elevated risk for cardiovascular disease, including changes in blood pressure, lipids, and inflammatory markers (Fraser et al., 2010).
To the committee’s knowledge, there is no direct evidence that interventions to produce more favorable maternal weight at the beginning of pregnancy have the expected and desired impact of reducing the child’s risk of overweight or obesity. Nonetheless, there are a number of possible reasons for the association between maternal prepregnancy obesity and childhood obesity, most but not all of which suggest that policies producing more favorable prepregnancy weight would reduce the risk of obesity in children. One pathway concerns the effect of mater-
nal obesity on the intrauterine environment, with the metabolic consequences of greater maternal weight potentially programming the fetus toward obesity (Levin, 2000; Vickers et al., 2007). This pathway is also considered below in the discussion of pathways linking maternal diabetes and related metabolic disorders to childhood obesity. A second possibility, not directly amenable to preventive interventions, is that the mother’s weight indicates a genetic predisposition to obesity that is transmitted to the child. Better weight maintenance by the mother would not alter her child’s genetic makeup, of course, but even under this scenario, the potential for the mother to manage her own weight despite her predisposition could well be beneficial in helping her better manage her infant’s weight. (A third, nonprenatal pathway might involve maternal behavior, with the mother’s own tendency toward an unfavorable balance of energy intake and expenditure being passed on to her infant and child through childrearing practices [Oliveria et al., 1992].)
As the prevalence of overweight and obesity has increased dramatically over the past few decades in the United States, so, too, has the proportion of women entering pregnancy above ideal weight (Chu et al., 2009; Yeh and Shelton, 2005). Mothers who are overweight or obese entering pregnancy (referred to in the remainder of this paragraph as “overweight”) are at high risk for pregnancy complications and make greater use of health care services and incur higher costs (Catalano, 2007; Chu et al., 2008; Heslehurst et al., 2008). In addition, maternal overweight is the strongest identified predictor for glucose intolerance and gestational diabetes mellitus (GDM), which themselves predict increased risk for pregnancy complications, as well as subsequent development of type 2 diabetes mellitus (American Diabetes Association, 2004; Bo et al., 2001; Crowther et al., 2005; Dabelea et al., 2005; Radesky et al., 2008). Infants born to overweight mothers are more likely to be born large for gestational age, are less likely to be breastfed, and are at higher risk for overweight and type 2 diabetes mellitus in later life (Gunderson, 2007; Li et al., 2003; Stuebe et al., 2005). Maternal overweight may be feeding into an intergenerational cycle in which offspring of heavier mothers are themselves more likely to weigh more and have impaired glucose tolerance entering their own pregnancies. Overweight mothers are most likely to gain excessive weight during pregnancy and to experience substantial weight retention postpartum, doubly disadvantaging themselves and their children.
Although the association between maternal and child weight and obesity is clear, there are compelling reasons to undertake studies to determine why that link exists. This is not merely a matter of academic interest but an opportunity to
understand the determinants of childhood obesity more fully, with implications for a wide range of potential interventions. The association between maternal and childhood obesity is a strong signal, and the opportunity exists to draw out its implications more fully. To the extent that the association indicates some biologically based fetal programming, it has important implications for gestational influences that include maternal diabetes, as well as chemicals, drugs, and microorganisms that have metabolic consequences. To the extent that the association is behavioral, the potential exists to incorporate particular educational components into prenatal care, using the marker of maternal obesity as an indication that the child may benefit from intervention. Ultimately, studies are needed that can experimentally improve prepregnancy weight and examine the consequences for obesity in offspring.
Gestational Weight Gain
At the time of the recent Institute of Medicine (IOM) report on guidelines for GWG (IOM, 2009), a small number of studies had been published that directly addressed the question of whether higher levels of GWG were associated with an increased risk of obesity in childhood and beyond. The literature was summarized by Oken (2009), who reported on seven studies, all of which identified an increased risk of elevated weight in offspring (children or adult) associated with higher GWG. Additional studies published since the time of Oken’s review have provided further support for the hypothesis that elevated GWG has an independent effect on the likelihood of obesity in children and adolescents (Oken, 2009).
Six recent studies examining the relationship of GWG to infant and childhood weight/obesity (Fraser et al., 2010; Margerison Zilko et al., 2010; Oken et al., 2009; Olson et al., 2009; Stuebe et al., 2009; Wrotniak et al., 2008) all provide supportive evidence for the hypothesis that elevated GWG leads to increased childhood weight or risk of obesity, although the degree of that support and specific results differ. Most directly relevant to the age range of interest for this report, Olson and colleagues (2009) considered the risk of overweight in children at age 3 and found that the risk was greater for children of mothers who gained more weight during pregnancy and that this association was stronger among overweight and obese mothers. Margerison Zilko and colleagues (2010) analyzed data from the National Longitudinal Survey of Youth and found that the children of women who gained more than the recommended amount during pregnancy had an increased risk of obesity at ages 2–20. Likewise, children born to mothers enrolled in the Collaborative Perinatal Project in the 1960s (Wrotniak et al.,
2008), when maternal obesity was rare, showed an association between GWG and odds of being overweight at age 7. The ALSPAC cohort in England was recently analyzed to address this question (Fraser et al., 2010). The analysis showed that 9-year-old children whose mothers exceeded the IOM recommendations for GWG had elevated BMIs, as well as increased waist size, fat mass, leptin, systolic blood pressure, and C-reactive protein and lower HDL (high-density lipoprotein) levels, all associated with cardiovascular disease in adults. Cumulatively, these results can be summarized as providing a preponderance of evidence supporting the association between GWG and an increase in average childhood weight, as well as an increase in the risk of children being classified as overweight or obese.
Some key methodological challenges in addressing this issue need to be noted. First, women who are overweight or obese prior to conception have a higher risk of having children who are obese and tend to have lower GWG than normal-weight or underweight women, an observation that calls for careful control of prepregnancy weight. Second, women who have higher GWG tend to have heavier babies (IOM, 2009), and infants who weigh more at birth are more likely to be obese as children and later in life (Freedman et al., 2005). However, controlling for birth weight is not necessarily appropriate insofar as birth weight is on the causal pathway linking elevated GWG to childhood obesity. There are other plausible pathways linking elevated GWG to childhood obesity, including a genetically shared maternal and infant tendency to gain weight, common dietary habits for the mother and her child, and a metabolic effect of maternal weight gain on infant appetite and glucose metabolism that supports weight gain. Regardless of the mechanism, however, the critical question is whether elevated GWG is causally related to childhood obesity such that improved compliance with GWG guidelines would reduce the occurrence of childhood obesity.
The guidelines for GWG recommended by the IOM were selected to balance and optimize maternal and infant health. Although there is legitimate debate regarding whether the levels are the precisely right ones, insufficient weight gain clearly is associated with adverse outcomes, notably fetal growth restriction and possibly preterm birth and its consequences, including infant mortality. There is also clear evidence that excessive GWG is associated with an increased risk of excessive weight retention and its sequelae in the mother following pregnancy, as well as with elevated infant weight and the resulting increased risk of Cesarean delivery. For the purposes of this discussion, the question is whether improved compliance with the GWG guidelines, particularly not exceeding the recommended weight gain, would have beneficial effects beyond the potential reduction in
obesity in young children. The answer is that it clearly would, reducing maternal obesity and infant macrosomia—both major health concerns—at a minimum.
The potential for negative effects of pushing more forcefully for compliance with the GWG recommendations also must be considered. Some health outcomes follow a continuum, so that lower GWG is predicted to increase the risk of small-for-gestational-age births (IOM, 2009; Viswanathan et al., 2008). The potential also exists for increasing maternal anxiety, given the substantial proportion of women who exceed the guidelines (IOM, 2009), with mothers becoming more fearful regarding the health of their child but not necessarily being able to take the actions necessary to address this concern.
Despite the consistent and growing observational data indicating that women with higher GWG have infants and children at greater risk of obesity, the question of causality remains unresolved. Research to determine directly the effect of GWG on childhood obesity is needed, notably randomized controlled trials (RCTs) of sufficient size to conclude whether more optimal GWG has the expected beneficial effect in reducing the risk of overweight and obesity in children.
The few published interventions to modify GWG have included relatively small numbers of participants and have had mixed results (Artal et al., 2007; Kinnunen et al., 2007; Olson et al., 2004; Polley et al., 2002; Wolff et al., 2008). A 2008 systematic review of diet and exercise interventions during and after pregnancy published in 1985–2007 identified only one high-quality prenatal intervention that included 120 women (Kuhlmann et al., 2008). Another systematic review of interventions to limit GWG among overweight and obese women similarly concluded that, given the limited information available, further evaluation through RCTs with adequate power is required (Dodd et al., 2008).
Maternal Diabetes During Pregnancy
As discussed in more detail below, evidence clearly shows that women who have insulin-dependent diabetes at the time of pregnancy have offspring at increased risk of obesity. However, the evidence regarding whether gestational diabetes, a far more common condition, has an independent effect on childhood obesity is much less certain. Furthermore, even when an association is observed, it is not clear whether that increased risk of childhood obesity is a by-product of the strong relationship between elevated prepregnancy BMI and risk of gestational diabetes, with the elevated maternal BMI rather than the gestational diabetes resulting in obesity in the offspring, or whether the association may reflect an independent effect of gestational diabetes. On the other hand, it is possible that at least part of
the effect of maternal obesity more generally is through the associated impaired glucose tolerance’s biological effect on the fetus. Shared genetic risks for diabetes and impaired glucose tolerance may also contribute to the association between maternal diabetes and childhood obesity.
Several studies have found an increased risk of obesity in the offspring of mothers with gestational diabetes (Silverman et al., 1991, 1995; Vohr et al., 1980), but Whitaker and colleagues (1998) note that all of these studies included a mix of mothers with gestational diabetes and with preexisting insulin-dependent diabetes. A study by Dabelea and colleagues (2000) examined pregnancy outcomes of women before and after their diagnosis with type 2 diabetes. The study results clearly indicated that children born after diagnosis had a greater risk of being obese than those born before diagnosis, a clear demonstration that genetics alone do not account for the association.
Results for gestational diabetes and obesity in offspring are less consistent. Whitaker and colleagues (1998) found no support for an increased risk of obesity in children born to mothers with either gestational diabetes or evidence of impaired glucose tolerance based on pregnancy screening. Wright and colleagues (2009) found an association between gestational diabetes and increased adiposity in 3-year-old children based on skinfold measures of adiposity but not based on BMI. Gillman and colleagues (2003) found a small, independent association between maternal gestational diabetes and adolescent obesity, not mediated by birth weight or confounded by prepregnancy BMI. The metabolic disturbances of gestational diabetes may be so much less severe than those of diagnosed type 2 diabetes that the consequences for offspring are more subtle. However, the markedly greater prevalence of gestational diabetes as opposed to type 2 diabetes in women of reproductive age makes the potential impact of gestational diabetes of great public health importance.
Evidence clearly demonstrates adverse effects of gestational diabetes on the health of offspring. These effects include the risk of macrosomia and its consequences in the form of Cesarean delivery and dystocia and physiologic changes at the time of birth (Catalano, 2007; Crowther et al., 2005). Improved control of diabetes during pregnancy has been shown to mitigate these risks (Crowther et al., 2005), whether through behavioral change (diet and exercise) or through medication. Thus there is a clear basis for supporting actions to improve the detection and control of diabetes in pregnancy and no apparent risk of harm, and such policies may also help reduce the risk of obesity in young children.
As part of the spectrum of concerns related to maternal prepregnancy obesity and GWG, the relationship between the maternal metabolic environment and infant eating, activity, and future weight gain needs to be more fully understood. Such research would help identify interventions with the potential to mitigate the adverse effects of maternal obesity, excess GWG, and diabetes, as well as provide more fundamental information on prenatal influences on the risk of childhood obesity. Programming for obesity in utero is quite plausible and a phenomenon worthy of elucidation.
Smoking During Pregnancy
A number of studies have documented an association between maternal smoking during pregnancy and the risk of obesity in offspring (Oken et al., 2005; Power and Jefferis, 2002; Von Kries et al., 2002). The literature generally has found positive associations that were reduced but not eliminated with adjustment for confounding and a stronger association with heavier smoking.
Although a statistical association between maternal smoking and childhood obesity appears to exist, the exact reasons for this association are not straightforward. Among the possible bases for the association, only some would predict that reductions in smoking would reduce the risk of early childhood obesity. The association may simply reflect confounding by socioeconomic status, attempts at statistical adjustment notwithstanding, given the strong relationship of lower education and income with both smoking during pregnancy and obesity in children. A direct causal effect of smoking on childhood obesity is plausible, but the pathway by which it might operate is uncertain (Chen and Morris, 2007). One paradoxical aspect of the association is that smoking clearly causes reduced fetal growth, and smaller size at birth is generally predictive of reduced risk of obesity. However, the impaired fetal growth may be followed by catch-up growth in infancy that is associated with obesity in childhood and beyond (Oken et al., 2008). There may also be a direct programming effect on the infant’s and child’s tendencies toward obesity, affecting appetite, activity levels, or metabolism.
The evidence that smoking during pregnancy has adverse effects on fetal growth, placental abruption, stillbirth, and infant mortality is compelling (Salihu and Wilson, 2007) and provides a clear basis for taking all possible measures to curtail or ideally eliminate the behavior. The only outcome of pregnancy that appears to benefit from maternal smoking is preeclampsia, with consistent evidence of reduced risk among smokers (England and Zhang, 2007), but the nega-
tive consequences of smoking during pregnancy more than offset this potential benefit.
The policy recommendations regarding smoking are already quite clear, so that research is not needed at this point to guide interventions. The main argument for a closer examination of the association between smoking during pregnancy and childhood obesity is to better understand pathways that may be relevant to other exposures that affect fetal development and metabolism. Elucidating the causal pathways suggested by an adverse effect of smoking on obesity could be beneficial in identifying and reducing other, analogous influences.
Abu-Rmeileh, N. M. E., C. L. Hart, A. McConnachie, M. N. Upton, M. E. J. Lean, and G. C. M. Watt. 2008. Contribution of midparental BMI and other determinants of obesity in adult offspring. Obesity 16(6):1388-1393.
American Diabetes Association. 2004. Gestational diabetes mellitus. Diabetes Care 27(90001):S88-S90.
Artal, R., R. B. Catanzaro, J. A. Gavard, D. J. Mostello, and J. C. Friganza. 2007. A lifestyle intervention of weight-gain restriction: Diet and exercise in obese women with gestational diabetes mellitus. Applied Physiology, Nutrition, and Metabolism 32(3):596-601.
Baird, J., D. Fisher, P. Lucas, J. Kleijnen, H. Roberts, and C. Law. 2005. Being big or growing fast: Systematic review of size and growth in infancy and later obesity. British Medical Journal 331(7522):929-931.
Baughcum, A. E., K. A. Burklow, C. M. Deeks, S. W. Powers, and R. C. Whitaker. 1998. Maternal feeding practices and childhood obesity: A focus group study of low-income mothers. Archives of Pediatrics and Adolescent Medicine 152(10):1010-1014.
Baughcum, A. E., L. A. Chamberlin, C. M. Deeks, S. W. Powers, and R. C. Whitaker. 2000. Maternal perceptions of overweight preschool children. Pediatrics 106(6):1380-1386.
Belfort, M. B., S. L. Rifas-Shiman, J. W. Rich-Edwards, K. P. Kleinman, E. Oken, and M. W. Gillman. 2008. Infant growth and child cognition at 3 years of age. Pediatrics 122(3):e689-695.
Benson, L., H. J. Baer, and D. C. Kaelber. 2009. Trends in the diagnosis of overweight and obesity in children and adolescents: 1999-2007. Pediatrics 123(1).
Bo, S., G. Menato, A. Lezo, A. Signorile, C. Bardelli, F. De Michieli, M. Massobrio, and G. Pagano. 2001. Dietary fat and gestational hyperglycaemia. Diabetologia 44(8):972-978.
Casey, P. H., L. Whiteside-Mansell, K. Barrett, R. H. Bradley, and R. Gargus. 2006. Impact of prenatal and/or postnatal growth problems in low birth weight preterm infants on school-age outcomes: An 8-year longitudinal evaluation. Pediatrics 118(3):1078-1086.
Catalano, P. M. 2007. Management of obesity in pregnancy. Obstetrics and Gynecology 109(2 Part 1):419-433.
CDC (Centers for Disease Control and Prevention). 2010. Growth Charts. http://www.cdc.gov/growthcharts/ (accessed March 11, 2011).
Chen, H., and M. J. Morris. 2007. Maternal smoking—a contributor to the obesity epidemic?Obesity Research and Clinical Practice 1(3):155-163.
Chu, S. Y., D. J. Bachman, W. M. Callaghan, E. P. Whitlock, P. M. Dietz, C. J. Berg, M. O’Keeffe-Rosetti, F. C. Bruce, and M. C. Hornbrook. 2008. Association between obesity during pregnancy and increased use of health care. New England Journal of Medicine 358(14):1444-1453.
Chu, S. Y., S. Y. Kim, and C. L. Bish. 2009. Prepregnancy obesity prevalence in the United States, 2004-2005. Maternal and Child Health Journal 13(5):614-620.
Crowther, C. A., J. E. Hiller, J. R. Moss, A. J. McPhee, W. S. Jeffries, and J. S. Robinson. 2005. Effect of treatment of gestational diabetes mellitus on pregnancy outcomes. New England Journal of Medicine 352(24):2477-2486.
Dabelea, D., R. L. Hanson, R. S. Lindsay, D. J. Pettitt, G. Imperatore, M. M. Gabir, J. Roumain, P. H. Bennett, and W. C. Knowler. 2000. Intrauterine exposure to diabetes conveys risks for type 2 diabetes and obesity: A study of discordant sibships. Diabetes 49(12):2208-2211.
Dabelea, D., J. K. Snell-Bergeon, C. L. Hartsfield, K. J. Bischoff, R. F. Hamman, and R. S. McDuffie. 2005. Increasing prevalence of gestational diabetes mellitus (GDM) over time and by birth cohort: Kaiser Permanente of Colorado GDM screening program. Diabetes Care 28(3):579-584.
de Onis, M., C. Garza, C. G. Victora, A. W. Onyango, E. A. Frongillo, and J. Martines. 2004. The WHO Multicentre Growth Reference Study: Planning, study design, and methodology. Food and Nutrition Bulletin 25(1 Suppl. 1).
Dennison, B. A., L. S. Edmunds, H. H. Stratton, and R. M. Pruzek. 2006. Rapid infant weight gain predicts childhood overweight. Obesity 14(3):491-499.
Dodd, J. M., C. A. Crowther, and J. S. Robinson. 2008. Dietary and lifestyle interventions to limit weight gain during pregnancy for obese or overweight women: A systematic review. Scandinavian Association of Obstetricians and Gynaecologists 87(7):702-706.
Ehrenkranz R. A., A. M. Dusick, B. R. Vohr, L. L. Wright, L. A. Wrage, and W. K. Poole. 2006. Growth in the neonatal intensive care unit influences neurodevelopmental and growth outcomes of extremely low birth weight infants. Pediatrics 117(4):1253-1261.
England, L., and J. Zhang. 2007. Smoking and risk of preeclampsia: A systematic review. Frontiers in Bioscience: A Journal and Virtual Library 12:2471-2483.
Fraser, A., K. Tilling, C. MacDonald-Wallis, N. Sattar, M. J. Brion, L. Benfield, A. Ness, J. Deanfield, A. Hingorani, S. M. Nelson, G. D. Smith, and D. A. Lawlor. 2010. Association of maternal weight gain in pregnancy with offspring obesity and metabolic and vascular traits in childhood. Circulation 121(23):2557-2564.
Freedman, D. S., L. K. Khan, M. K. Serdula, W. H. Dietz, S. R. Srinivasan, and G. S. Berenson. 2005. The relation of childhood BMI to adult adiposity: The Bogalusa Heart Study. Pediatrics 115(1):22-27.
Gillman, M. W. 2010. Early infancy as a critical period for development of obesity and related conditions. In Importance of Growth for Health and Development, Vol. 65, edited by A. Lucas, M. Makrides, and E. Ziegler. Boston, MA: Nestle Nutrition Institute Workshop. Pp. 13-24.
Gillman, M. W., S. Rifas-Shiman, C. S. Berkey, A. E. Field, and G. A. Colditz. 2003. Maternal gestational diabetes, birth weight, and adolescent obesity. Pediatrics 111(3).
Gillman, M. W., S. L. Rifas-Shiman, K. Kleinman, E. Oken, J. W. Rich-Edwards, and E. M. Taveras. 2008. Developmental origins of childhood overweight: Potential public health impact. Obesity 16(7):1651-1656.
Grummer-Strawn, L. M., C. Reinold, and N. F. Krebs. 2010. Use of World Health Organization and CDC growth charts for children aged 0-59 months in the United States. Morbidity and Mortality Weekly Report 59(RR-9):1-14.
Gunderson, E. P. 2007. Breastfeeding after gestational diabetes pregnancy: Subsequent obesity and type 2 diabetes in women and their offspring. Diabetes Care 30(Suppl. 2).
Heslehurst, N., H. Simpson, L. J. Ells, J. Rankin, J. Wilkinson, R. Lang, T. J. Brown, and C. D. Summerbell. 2008. The impact of maternal BMI status on pregnancy outcomes with immediate short-term obstetric resource implications: A meta-analysis. Obesity Reviews 9(6):635-683.
Hillman, J. B., S. D. Corathers, and S. E. Wilson. 2009. Pediatricians and screening for obesity with body mass index: Does level of training matter? Public Health Reports 124(4):561-567.
Huang, J. S., K. Becerra, T. Oda, E. Walker, R. Xu, M. Donohue, I. Chena, V. Curbelo, and A. Breslow. 2007. Parental ability to discriminate the weight status of children: Results of a survey. Pediatrics 120(1).
Huang, J. S., M. Donohue, G. Golnari, S. Fernandez, E. Walker-Gallego, K. Galvan, C. Briones, J. Tamai, and K. Becerra. 2009. Pediatricians’ weight assessment and obesity management practices. BMC Pediatrics 9(1).
Hui, L. L., C. M. Schooling, S. S. L. Leung, K. H. Mak, L. M. Ho, T. H. Lam, and G. M. Leung. 2008. Birth weight, infant growth, and childhood body mass index: Hong Kong’s children of 1997 birth cohort. Archives of Pediatrics and Adolescent Medicine 162(3):212-218.
IOM (Institute of Medicine). 2009. Weight Gain During Pregnancy: Reexamining the Guidelines. Washington, DC: The National Academies Press.
Jansen, W., and J. Brug. 2006. Parents often do not recognize overweight in their child, regardless of their socio-demographic background. European Journal of Public Health 16(6):645-647.
Jelalian, E., J. Boergers, C. S. Alday, and R. Frank. 2003. Survey of physician attitudes and practices related to pediatric obesity. Clinical Pediatrics 42(3):235-245.
Jimenez-Cruz, A., M. Bacardi-Gascon, O. Castillo-Ruiz, Z. Mandujano-Trujillo, and A. Pichardo-Osuna. 2010. Low income, Mexican mothers’ perception of their infants’ weight status and beliefs about their foods and physical activity. Child Psychiatry and Human Development 41(5):490-500.
Kinnunen, T. I., M. Pasanen, M. Aittasalo, M. Fogelholm, L. Hilakivi-Clarke, E. Weiderpass, and R. Luoto. 2007. Preventing excessive weight gain during pregnancy—a controlled trial in primary health care. European Journal of Clinical Nutrition 61(7):884-891.
Klein, J. D., T. S. Sesselberg, M. S. Johnson, K. G. O’Connor, S. Cook, M. Coon, C. Homer, N. Krebs, and R. Washington. 2010. Adoption of body mass index guidelines for screening and counseling in pediatric practice. Pediatrics 125(2):265-272.
Kuhlmann, A. K., P. M. Dietz, C. Galavotti, and L. J. England. 2008. Weight-management interventions for pregnant or postpartum women. American Journal of Preventive Medicine 34(6):523-528.
Levin, B. E. 2000. Metabolic imprinting on genetically predisposed neural circuits perpetuates obesity. Nutrition 16(10):909-915.
Li, R., S. Jewell, and L. Grummer-Strawn. 2003. Maternal obesity and breast-feeding practices. The American Journal of Clinical Nutrition 77(4):931-936.
Lightfoot, C., M. Cole, and S. R. Cole. 2009. The Development of Children, 6th ed. New York: Worth Publishers.
Maffeis, C., G. Talamini, and L. Tatò. 1998. Influence of diet, physical activity and parents’ obesity on children’s adiposity: A four-year longitudinal study. International Journal of Obesity 22(8):758-764.
Manios, Y., G. Moschonis, E. Grammatikaki, A. Anastasiadou, and T. Liarigkovinos. 2010. Determinants of childhood obesity and association with maternal perceptions of their children’s weight status: The “genesis” study. Journal of the American Dietetic Association 110(10):1527-1531.
Margerison Zilko, C. E., D. Rehkopf, and B. Abrams. 2010. Association of maternal gestational weight gain with short- and long-term maternal and child health outcomes. American Journal of Obstetrics and Gynecology 202(6).
McLearn, K. T., B. S. Zuckerman, S. Parker, M. Yellowitz, and M. Kaplan-Sanoff. 1998. Child development and pediatrics for the 21st century: The healthy steps approach. Journal of Urban Health 75(4):704-723.
Monteiro, P. O. A., and C. G. Victora. 2005. Rapid growth in infancy and childhood and obesity in later life—a systematic review. Obesity Reviews 6(2):143-154.
Moseley, K. L., G. L. Freed, and S. D. Goold. 2011. Which sources of child health advice do parents follow? Clinical Pediatrics 50(1):50-56.
Oken, E. 2009. Maternal and child obesity: The causal link. Obstetrics and Gynecology Clinics of North America 36(2):361-377.
Oken, E., S. Y. Huh, E. M. Taveras, J. W. Rich-Edwards, and M. W. Gillman. 2005. Associations of maternal prenatal smoking with child adiposity and blood pressure. Obesity Research 13(11):2021-2028.
Oken, E., E. M. Taveras, K. P. Kleinman, J. W. Rich-Edwards, and M. W. Gillman. 2007. Gestational weight gain and child adiposity at age 3 years. American Journal of Obstetrics and Gynecology 196(4).
Oken, E., E. B. Levitan, and M. W. Gillman. 2008. Maternal smoking during pregnancy and child overweight: Systematic review and meta-analysis. International Journal of Obesity 32(2):201-210.
Oken, E., K. P. Kleinman, M. B. Belfort, J. K. Hammitt, and M. W. Gillman. 2009. Associations of gestational weight gain with short- and longer-term maternal and child health outcomes. American Journal of Epidemiology 170(2):173-180.
Oliveria, S. A., R. C. Ellison, L. L. Moore, M. W. Gillman, E. J. Garrahie, and M. R. Singer. 1992. Parent-child relationships in nutrient intake: The Framingham Children’s Study. American Journal of Clinical Nutrition 56(3):593-598.
Olson, C. M., M. S. Strawderman, and R. G. Reed. 2004. Efficacy of an intervention to prevent excessive gestational weight gain. American Journal of Obstetrics and Gynecology 191(2):530-536.
Olson, C. M., M. S. Strawderman, and B. A. Dennison. 2009. Maternal weight gain during pregnancy and child weight at age 3 years. Maternal and Child Health Journal 13(6):839-846.
Patel, A. I., K. A. Madsen, J. H. Maselli, M. D. Cabana, R. S. Stafford, and A. L. Hersh. 2010. Underdiagnosis of pediatric obesity during outpatient preventive care visits. Academic Pediatrics 10(6):405-409.
Polley, B. A., R. R. Wing, and C. J. Sims. 2002. Randomized controlled trial to prevent excessive weight gain in pregnant women. International Journal of Obesity and Related Metabolic Disorders 26(11):1494-1502.
Pomietto, M., A. D. Docter, N. Van Borkulo, L. Alfonsi, J. Krieger, and L. L. Liu. 2009. Small steps to health: Building sustainable partnerships in pediatric obesity care. Pediatrics 123(Suppl. 5):S308-S316.
Power, C., and B. J. M. H. Jefferis. 2002. Fetal environment and subsequent obesity: A study of maternal smoking. International Journal of Epidemiology 31(2):413-419.
Radesky, J. S., E. Oken, S. L. Rifas-Shiman, K. P. Kleinman, J. W. Rich-Edwards, and M. W. Gillman. 2008. Diet during early pregnancy and development of gestational diabetes. Paediatric and Perinatal Epidemiology 22(1):47-59.
Salihu, H. M., and R. E. Wilson. 2007. Epidemiology of prenatal smoking and perinatal outcomes. Early Human Development 83(11):713-720.
Sesselberg, T. S., J. D. Klein, K. G. O’Connor, and M. S. Johnson. 2010. Screening and counseling for childhood obesity: Results from a national survey. Journal of the American Board of Family Medicine 23(3):334-342.
Silverman, B. L., T. Rizzo, O. C. Green, N. H. Cho, R. J. Winter, E. S. Ogata, G. E. Richards, and B. E. Metzger. 1991. Long-term prospective evaluation of offspring of diabetic mothers. Diabetes 40(Suppl. 2):121-125.
Silverman, B. L., B. E. Metzger, N. H. Cho, C. A. Loeb. 1995. Impaired glucose tolerance in adolescent offspring of diabetic mothers. Relationship to fetal hyperinsulinism. Diabetes Care 18:611-617.
Stettler, N., B. S. Zemel, S. Kumanyika, and V. A. Stallings. 2002. Infant weight gain and childhood overweight status in a multicenter, cohort study. Pediatrics 109(2I):194-199. Stuebe, A. M., J. W. Rich-Edwards, W. C. Willett, J. E. Manson, and K. B. Michels.
2005. Duration of lactation and incidence of type 2 diabetes. Journal of the American Medical Association 294(20):2601-2610.
Stuebe, A. M., M. R. Forman, and K. B. Michels. 2009. Maternal-recalled gestational weight gain, pre-pregnancy body mass index, and obesity in the daughter. International Journal of Obesity 33(7):743-752.
Taveras, E. M., S. L. Rifas-Shiman, K. S. Scanlon, L. M. Grummer-Strawn, B. Sherry, and M. W. Gillman. 2006. To what extent is the protective effect of breastfeeding on future overweight explained by decreased maternal feeding restriction? Pediatrics 118(6):2341-2348.
Taveras, E. M., S. L. Rifas-Shiman, E. Oken, E. P. Gunderson, and M. W. Gillman. 2008. Short sleep duration in infancy and risk of childhood overweight. Archives of Pediatrics and Adolescent Medicine 162(4):305-311.
Taveras, E. M., S. L. Rifas-Shiman, M. B. Belfort, K. P. Kleinman, E. Oken, and M. W. Gillman. 2009. Weight status in the first 6 months of life and obesity at 3 years of age. Pediatrics 123(4):1177-1183.
Taveras, E. M., S. L. Rifas-Shiman, B. Sherry, E. Oken, J. Haines, K. P. Kleinman, J. W. Rich-Edwards, and M. W. Gillman. In press. Crossing growth percentiles in infancy and risk of obesity in childhood. Archives of Pediatric and Adolescent Medicine.
Vickers, M. H., C. L. Cupido, and P. D. Gluckman. 2007. Developmental programming of obesity and type 2 diabetes. Fetal and Maternal Medicine Review 18(1):1-23.
Viswanathan, M., A. M. Siega-Riz, M. K. Moos, A. Deierlein, S. Mumford, J. Knaack, P. Thieda, L. J. Lux, and K. N. Lohr. 2008. Outcomes of maternal weight gain. Evidence Report/Technology Assessment 168:1-223.
Vohr, B. R., L. P. Lipsitt, and W. Oh. 1980. Somatic growth of children of diabetic mothers with reference to birth size. Journal of Pediatrics 97(2):196-199.
Von Kries, R., A. M. Toschke, B. Koletzko, and W. Slikker, Jr. 2002. Maternal smoking during pregnancy and childhood obesity. American Journal of Epidemiology 156(10):954-961.
Whitaker, R. C. 2004. Predicting preschooler obesity at birth: The role of maternal obesity in early pregnancy. Pediatrics 114(1).
Whitaker, R. C., and W. H. Dietz. 1998. Role of the prenatal environment in the development of obesity. Journal of Pediatrics 132(5):768-776.
Whitaker, R. C., J. A. Wright, M. S. Pepe, K. D. Seidel, and W. H. Dietz. 1997. Predicting obesity in young adulthood from childhood and parental obesity. New England Journal of Medicine 337(13):869-873.
Whitaker, R. C., M. S. Pepe, K. D. Seidel, J. A. Wright, and R. H. Knopp. 1998. Gestational diabetes and the risk of offspring obesity. Pediatrics 101(2).
Williams, S. 2001. Overweight at age 21: The association with body mass index in childhood and adolescence and parents’ body mass index. A cohort study of New Zealanders born in 1972-1973. International Journal of Obesity 25(2):158-163.
Wolff, S., J. Legarth, K. Vangsgaard, S. Toubro, and A. Astrup. 2008. A randomized trial of the effects of dietary counseling on gestational weight gain and glucose metabolism in obese pregnant women. International Journal of Obesity 32(3):495-501.
Wright, C. S., S. L. Rifas-Shiman, J. W. Rich-Edwards, E. M. Taveras, M. W. Gillman, and E. Oken. 2009. Intrauterine exposure to gestational diabetes, child adiposity, and blood pressure. American Journal of Hypertension 22(2):215-220.
Wrotniak, B. H., J. Shults, S. Butts, and N. Stettler. 2008. Gestational weight gain and risk of overweight in the offspring at age 7 y in a multicenter, multiethnic cohort study. American Journal of Clinical Nutrition 87(6):1818-1824.
Yeh, J., and J. A. Shelton. 2005. Increasing prepregnancy body mass index: Analysis of trends and contributing variables. American Journal of Obstetrics and Gynecology 193(6):1994-1998.