7
Using Surveillance Data for Action
The committee concluded that a coordinated surveillance system is needed to integrate and expand existing information across the multiple levels of decision making in order to generate actionable timely knowledge for stakeholders at the local, state or regional, and national levels. A robust surveillance system will help to monitor, evaluate, and improve policies, programs, and services; better direct the placement of resources; and provide a stronger basis for advocacy and education and a benchmark for clinicians. During its deliberations, the committee discussed whether the framework the committee was charged with developing should focus on chronic diseases in general or whether it should be oriented more specifically to cardiovascular and chronic lung diseases. The charge to the committee mentions both. The committee concluded that the focus, as stated in the charge, should be “primarily on cardiovascular and chronic lung disease.” An enlarged focus on chronic diseases would require an expanded committee, a lengthier study process, and additional resources that were not available. However, the committee resolved to work to ensure that the framework and infrastructure it recommends will, to the extent possible, be applicable to other chronic diseases.
The committee’s rationale for a nationwide cardiovascular and chronic lung diseases surveillance system is based on the recognition of major gaps in current monitoring approaches and on new opportunities provided by emerging technologies, data collection mechanisms, and healthcare reform. Rather than construct an entirely new surveillance system, however, the committee concluded that existing surveillance data collection efforts and cohort studies can and should be strengthened and integrated to provide the necessary surveillance information. In terms of gaps, chronic disease trends are currently monitored by different stakeholders through an incomplete patchwork of surveys (some standardized and many non-standardized), registries, cohort studies, and mortality vital statistics (German et al., 2001; Goff et al., 2007). Although national surveys remain a critically important source of information on behaviors, clinical preventive service use, and prevalence of diagnosed and undiagnosed conditions, how trends vary across localities is poorly understood. Furthermore, local institutions lack technical guidance, useful tools, and adequate resources to effectively monitor these outcomes in their own jurisdictions and patient populations. Detailed patient data on disease incidence, severity, treatment practices, and outcomes are gathered in many healthcare institutions and by most insurance companies without standardization or the means to disseminate more broadly or even compare the patterns and trends of their patient populations with benchmarks for the larger population. Key data sources such as Medicare and Medicaid are not easily accessed at the state and local levels, where targeted change is most likely to occur, nor are they readily linkable to other data sources. Underlying these weaknesses is the lack of national coordination and leadership for these disparate efforts.
In terms of opportunity, standardized initiatives to improve quality of care, the rapid expansion of electronic health record (EHR) systems and patient registries, and the recent passage of the Patient Protection and Affordable Care Act have all opened new avenues for the systematic collection, analysis, and dissemination of information on the incidence and severity of chronic disease in populations under care (Chassin et al., 2010). National leadership has been at the forefront of these recent developments, an example of the valuable opportunities that exist to extend that leadership to completing and unifying the fragmented components of chronic disease surveillance identified in this report.
Successful implementation of a framework for national surveillance of cardiovascular and chronic lung diseases requires a mechanism to coordinate, monitor, and support the multiple data collection systems that contribute to the surveillance system. Furthermore, the system must provide ways to ensure that the elements collected can evolve in step with new knowledge about emerging risk factors, advancing technologies, and new understanding of the basis for disease.
Given that the mission of the Department of Health and Human Services (HHS) is to protect the health and provide essential services to Americans,1 that HHS is already responsible for the funding and conduct of numerous surveillance efforts, and that it is in a position to bring together stakeholders from both the public and private sectors as well as from multiple geographic levels, the committee believes HHS is in the best position to lead the development and implementation of the recommended framework and system. Because the recommended framework is based upon existing data collection approaches, it is crucial that those organizations responsible for the conduct of those approaches be involved in determining the ways to use and integrate existing approaches. It should be recognized, however, that resources are rarely available to support all the desires of each of the stakeholders. As discussed in Chapter 6, trade-offs will have to be made in terms of what data are to be collected and the mechanisms for doing so. For example, cost constraints may result in sampling rather than a full population assessment or the use of self-report rather than biological examinations.
It is critical that those who represent their organizations or agencies on the committee have expertise in the prevention, diagnosis, treatment, and surveillance of CVD or COPD or have access to such expertise as they engage in their deliberations in order to address the problems and issues confronting them as they work to integrate and enhance surveillance for these conditions. For example, as discussed in Chapter 3, a number of difficult issues regarding collection of data for the surveillance of COPD remain to be resolved, and it is anticipated that the working group will play a major role in such resolution. The committee believes strongly that federal agencies should collaborate with the many state and local public agencies and national and state-level, nongovernmental organizations that conduct components of the proposed system.
The use of a coordinating body, as the committee recommends below, is in line with the approach taken by Canada in its developing Canadian Chronic Disease Surveillance System2 (CCDSS), which integrates a network of provincial and territorial surveillance systems. The CCDSS began with diabetes surveillance and, in 2009, expanded the system to track information on the incidence and prevalence of diagnosed hypertension. The intent is to expand the system over time to include surveillance of other chronic diseases. The Canadian system is guided by a Task Group on Surveillance of Chronic Disease and Injury. The membership of the Task Force is composed entirely of government agencies, however, and the committee strongly believes that developing an effective system for the United States requires the involvement of both public- and private-sector stakeholders.
Recommendation 1
The committee recommends that the Secretary of HHS establish and provide adequate resources for a standing national working group to oversee and coordinate cardiovascular and chronic pulmonary disease surveillance activity. This working group should include representatives from HHS (CDC, NIH, AHRQ, CMS, IHS, ONCHIT, FDA), other relevant federal agencies (e.g., VA and DOD), and tribal, state, and local public health agencies, as well as nongovernmental organizations with relevant roles in surveillance.
____________
1 See http://govinfo.library.unt.edu/npr/library/status/mission/mhhs.htm (accessed August 2, 2011).
2 See http://www.phac-aspc.gc.ca/cd-mc/cvd-mcv/ccdss-snsmc-2010/2-1-eng.php (accessed August 2, 2011).
BUILDING A FRAMEWORK
Effectiveness of a surveillance system depends on several factors. These factors are the quality of the data; the ongoing, systematic collection, analysis, and interpretation of the data; the ongoing use of the results to plan and implement prevention and control strategies; and the regular feedback from the end users to those organizing surveillance systems so that a dynamic system can evolve in a continuous manner. A surveillance system for cardiovascular and chronic lung diseases must provide data that can be used to understand the continuum of prevention, disease progression, treatment, and outcomes, and can be flexible enough to respond to new challenges and opportunities. Data are needed that can provide information on
- Incidence and prevalence of relevant conditions over time;
- Primary prevention, including both elimination of exposures in the physical and social environments that cause these diseases and reducing behavioral, clinical, and other risk factors (e.g., physical inactivity, poor diets, and smoking);
- Secondary prevention efforts (i.e., early detection and intervention);
- Tertiary prevention (i.e., management of symptomatic disease);
- Health outcomes, including quality of life;
- Costs, including both the direct medical costs and the indirect costs of lost productivity, earnings, and social burden; and
- Disparities in these factors by race or ethnicity, geographic region, and socioeconomic status. Furthermore, the system must be flexible enough to respond to new challenges and opportunities.
Recommendation 2
The committee recommends that the national working group place priorities for surveillance on data systems that can overtly:
- Track progress on nationally recognized goals and indicators regarding cardiovascular disease and chronic pulmonary disease incidence, prevalence, and prevention (e.g., Healthy People);
- Evaluate and inform national, state, and local efforts to control, reduce, and prevent these chronic diseases;
- Enable effective public health actions and policies;
- Improve treatment outcomes;
- Monitor and enhance quality of life; and
- Reduce disparities in risk and burden of these diseases.
Setting the Foundation
As noted in prior sections, the sources and types of data potentially applicable to surveillance can be cataloged and systematically combined to provide a diverse and rich resource. These resources can be used to generate information and knowledge about chronic diseases that are useful in tracking prevalence, guiding public health and prevention efforts, informing efforts to manage and treat the diseases, and developing policies that address disparities. Similarly, the users of surveillance data can be identified and assisted by prioritizing their data requirements, organizing data into measures and indicators that inform decisions, and developing dissemination strategies to make the data accessible and useful for them.
The data resources and the uses and users of those data are critical to refining the requirements for a surveillance framework. However, the data needed and the decisions to be made are complex, evolving, and interdependent. This requires the conceptual backbone of a framework that leverages knowledge we already have and provides durability and adaptability going forward.
Many chronic cardiovascular and lung conditions share common risk factors and follow a broadly similar natural history within patients and populations, which enabled the committee to adapt for its purposes a conceptual
framework for cancer surveillance developed by Wingo and colleagues (2005) and illustrated in Figure 7-1. The trajectory of a chronic disease usually begins at younger ages with a period of apparent good health, often with underlying risk factors present. Some risk factors may be genetic or congenital, others may be behavioral, and others may be found in the person’s social or physical environment. The risk factors may be ignored for a time, but eventually they are likely to lead to clinical signs or symptoms that motivate the person to consult health professionals. It is critical in the new surveillance system to collect data on these risk factors in order to identify precursors prior to or at the very earliest states of disease.
Alternatively, these early manifestations may be detected through screening. The person may then transition through diagnosis to treatment and an objectively changed stage of life. Once diagnosed, progression of symptoms may be controlled or may proceed at variable rates as individuals live with a chronic condition and its management. Either as a consequence of the initial condition or due to other intercurrent and complicating events, patients also experience other conditions and, eventually, the end of life. The stages of health (with risk factors), diagnosis, treatment, living with a chronic condition, and end of life are predictable in both likely occurrence and sequence.
Incorporating the logic and practices of primary, secondary, and tertiary prevention completes the crux of the framework. Prevention involves actions aimed at preventing or reducing the occurrence of a condition or minimizing the effects of a condition. Primary prevention is concerned with deterring the occurrence of disease in a population through strategies aimed at disease risk factors. Secondary prevention promotes the early detection of disease so that prompt treatment can be given in order to prevent further deterioration and early death. Screening services are major secondary prevention strategies. Tertiary prevention focuses on disease treatment and management to reduce the impact of disease (Last, 2001; Modeste, 1996).
Decision makers at all levels must recognize that prevention is relevant at all stages of a chronic health condition. Such recognition will aid in ensuring that policy, system design, and practice align to identify and deliver appropriate interventions throughout the course of a chronic condition.
Information about chronic conditions that evolve over the life course should include data on the timing and appropriateness of preventive and therapeutic interventions; identify patterns of incidence and prevalence;
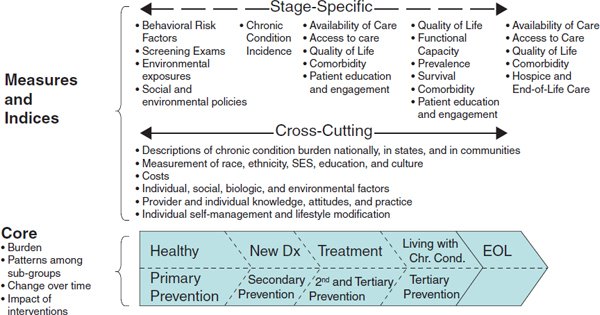
FIGURE 7-1 Framework for a national surveillance system for cardiovascular and chronic lung diseases.
SOURCE: Adapted from Wingo et al., 2005.
hypothesize about causation; track changes over time; and observe the impact of interventions on important health outcomes. Collecting and integrating data at each stage in the course of a chronic condition is important to a comprehensive surveillance system.
Measures and Indexes
Linking the information arising within the core of the framework to action occurs through a generation of measures and indexes. Within the core framework of progression of conditions and interventions over a life course, some measures and indexes will apply to specific stages of the condition sequence, while other measurements will apply to most if not all stages. Recognition and incorporation of both cross-cutting and stage-specific metrics is an important feature for fully supporting the range of decision makers. A full specification of a portfolio of measure and index definitions is beyond the scope of this report; however, examples of both cross-cutting and stage-specific measure concepts are included in Figure 7-1.
Recommendation 3
The committee recommends that HHS adopt the framework illustrated in Figure 7-1 as a guide for national surveillance of cardiovascular and chronic lung diseases.
The framework organizes data from traditional, evolving, and novel surveillance sources to reflect the development and progression of chronic conditions over a life course. The design also captures the impact of prevention as both a goal and an interventional intent. Information emerging from this core can be assembled into both cross-cutting and stage-specific metrics to inform the actions of decision makers in multiple roles and at the macro, meso, and micro levels of the health and healthcare systems. This general framework, while evolved specifically for chronic heart and lung diseases, is anticipated to be broadly applicable to other chronic health conditions, including the increasingly common occurrence of multiple chronic health conditions in the same individual.
Various data are needed to facilitate an effective surveillance system for cardiovascular disease and chronic lung disease. These data include information about incidence and prevalence of the conditions of interest as well as their risk factors, prevention efforts, treatments, and health outcomes. Chapter 4 describes the need to untangle the effects of environment, income, education, race, ethnicity, and genetics on cardiovascular disease (CVD) and chronic obstructive pulmonary disease (COPD) to foster the elimination of health disparities. Such efforts require more effective and efficient linkages of conventional surveillance data to these more contextually relevant data (e.g., socioeconomic status, birthplace, acculturation, geography, language, insurance, etc.).
Furthermore, an effective surveillance system must evolve to account for changing case definitions (e.g., those of myocardial infarction and COPD). It must also allow recognition of new disease entities and an understanding of how changes in public policy affect the disease being studied and how risk factors can have a major impact on incidence and prevalence of other diseases. For example, a reduction in heart disease deaths may lead to an increase in cancer prevalence as more individuals survive to older ages, when cancer becomes more common.
Incidence and prevalence of disease can be greatly affected by the presentation of patients into a node of the medical system. For instance, if a patient who suffers from chronic respiratory illness fails to seek medical care and hence does not receive a diagnosis of COPD, then it will be undercounted in survey data, which typically rely on questions such as “Did a doctor, nurse, or other health professional ever tell you that you have … ?” Similarly, patients who experience “silent” myocardial infarctions or who do not have cardiac enzymes drawn or electrocardiograms performed at the time of infarction will contribute to underreporting of the true incidence or prevalence of disease. Therefore it becomes essential to understand how a patient’s awareness of a condition, symptom, or disease can affect the seeking of medical care, and the apparent incidence or prevalence of disease as gathered by surveillance/reporting systems. Similarly, there are significant gender differences in the constellation of symptoms of cardiovascular disease for women as opposed to men. As awareness campaigns gain traction, women may seek more medical attention. This can lead to more diagnoses of CVD in women, and an apparent rise in incidence.
Cardiovascular and pulmonary diseases do not occur in isolation. Each exists on a backdrop of multiple other diseases whose risk factors, incidence, and prevalence are themselves changing. Because COPD contributes to an increased risk of myocardial infarction, it becomes increasingly imperative to consider the changing prevalence of COPD in surveillance of myocardial infarction. Similarly, the landscape of many risk factors for cardiovascular disease, diabetes, measures of glycemic control, hypertension, hypercholesterolemia, and other comorbities change over time. It is important to understand the fabric of change in which a disease and its risk factors are surveyed.
As discussed in Chapter 5, current surveillance efforts for CVD and COPD are incomplete. There are clear gaps in data collection of patient outcomes that are critical for surveillance. Functional capacity, quality of life, and patient engagement, and action measures are needed. Clear definitions and measurement techniques will need to be tested and implemented. Finally, a system is needed to overcome the many uncoordinated efforts that frequently produce inconsistent information (Goff et al., 2007; Yeh et al., 2010).
Existing data sources have complementary potential to provide surveillance information across the life span. Enhancing the use of current data sources requires coordination of data collection efforts, harmonization of some elements, expansion to include patient outcomes, and community-tailored items. Coordination of data collection efforts across federal, state, and local systems as well as healthcare delivery systems has great potential for future surveillance efforts. Such efforts should include, to the extent possible, standard definitions of key risk factors and outcomes, interventions, and a mechanism to link subjects and providers across the different data sources. The committee believes that serious consideration should be given to the scientific and cost considerations as well as the ethical and privacy issues associated with the use of a unique personal health identifier so that results can be compared within and across different geographical areas.
The increase in value of the multiple federal data sets that collect information from subjects, patients, healthcare providers, and healthcare insurers would be increased substantially if linkages across data sources were possible. The use of multiple informants to measure the burden of cardiovascular and chronic lung disease risk factors, behaviors, treatments, and outcomes could provide a comprehensive active surveillance system capable of providing information that could be used by multiple stakeholders to analyze, understand, and act effectively.
The committee has proposed a framework (Figure 7-1) for organizing surveillance efforts that is based on the core concepts of the life course and the role of prevention. This framework incorporates both traditional data sources such as surveys, registries, cohort studies, and vital statistics, as well as evolving or novel sources that include health services and patient-generated data as well as and environmental data (illustrated in Figure 7-2). It must be noted that integrating data from these multiple sources will not be an easy task. Multiple stakeholders must reach agreement about what needs to be collected and integrated, and mechanisms for doing so must be developed and implemented. Such efforts will require investments of both dollars and time and progress will likely occur in a series of steps taken over a number of years.
The recommended framework also provides for a system that is relevant to multiple users at various levels, for example:
- At the national level for developing policies, setting funding priorities, and identifying research needs;
- At the state or regional level to aid in planning and allocating resources for various programs, services, and educational and policy initiatives;
- At the county or other local level for developing and organizing public health and healthcare services, developing and advocating health promotion and disease prevention policies, and educating and mobilizing community leaders and members; and
- At the family and individual level for guiding personal changes in lifestyle and environment.
Recommendation 4
The committee recommends that the group that oversees and coordinates surveillance activity be charged with
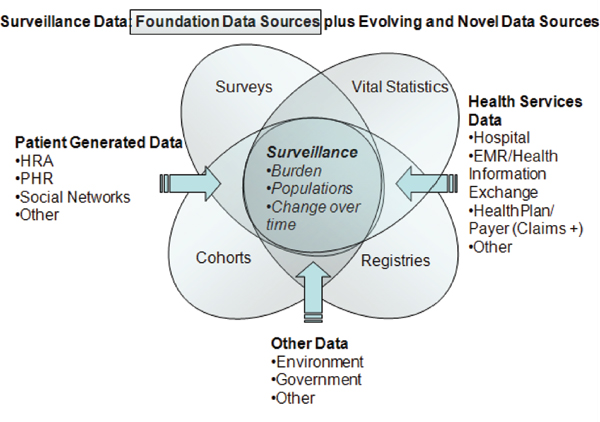
FIGURE 7-2 Traditional and evolving data sources for surveillance.
- Selecting surveillance indicators and, periodically, undertaking a review of the surveillance system in order to identify and incorporate necessary modifications;
- Improving collaboration and coordination among federal, tribal, state, and local agencies and nongovernmental organizations around the collection, compilation, and dissemination of surveillance information;
- Collecting and making available all types of surveillance data (survey, registry, EHR) at the most granular level consistent with protection of data privacy and confidentiality and, when feasible, linked with other data sources (i.e. clinical databases, public health data);
- Formation of public–private partnerships with the nongovernmental health sector; and
- Development of data sets for surveillance sources that can be made broadly accessible to a variety of users to support and guide action to improve health at the national, state, and local levels.
While the working group will provide direction as outlined above, a mechanism must be established to facilitate implementation of the enhanced and integrated system as it evolves. Several options exist for such a mechanism, two of which are described here. One option is the National Center for Health Statistics (NCHS) whose mission is “to provide statistical information that will guide actions and policies to improve the health of the American
people” (NCHS, 2009). The Health and Human Services Health Indicators Warehouse is operated by NCHS, and surveys and data systems in the NCHS purview include:
- National Ambulatory Medical Care Survey
- National Home and Hospice Care Survey
- National Hospital Ambulatory Medical Care Survey
- National Hospital Discharge Survey
- National Nursing Home Survey
- National Survey of Ambulatory Surgery
- National Survey of Residential Care Facilities
NCHS has a Board of Scientific Counselors that provides advice and makes recommendations about research and about new approaches for monitoring and evaluation of health-related policy changes. NCHS also facilitates the work of the Interagency Working Group on Summary Measures of Health (IAWG), which is composed of representatives from federal agencies and which is a forum for exchange of perspectives on summary measures of health.
Arguments in favor of designating NCHS as the office to coordinate the surveillance system include its portfolios of national surveys, which provide key national information on prevalence and distribution of disease and associated risk factors. NCHS also collects data on many chronic diseases and vital statistics, so it would be in a good position if the system were to expand to include chronic diseases in general. Other advantages of NCHS are that it is a federal statistical agency with independent judgment over its data because of its congressional designation, and it collects individual identifiers that enable linkage of data from its premier national survey, the National Health Interview Survey (NHIS), to Medicare data, national death records, and other federal data. Key NCHS surveys, including the NHIS, use multistage sampling designs, which enable them to sample states and other geographic areas. The NHIS is an in-person survey, which is entirely appropriate for a national benchmark survey. The National Health and Nutrition Examination Survey (NHANES) plays an important surveillance role because it does physical exams, collects biological specimens, and collects interview data. The State and Local Area Integrated Telephone Survey (SLAITS), an NCHS telephone survey that uses the sampling frame of the National Immunization Survey, could be adapted to be comprehensive and potentially could meet state and locally defined data needs. NCHS, together with the National Committee on Vital and Health Statistics and the HHS Data Council, clearly articulated a vision for health statistics consistent with the perspective and recommendations of this Committee (NCHS, 2002).
NCHS has many strengths but also some limitations. The main NCHS survey, the NHIS, relies on methods that produce representative samples nationally and at the state level, but these methods do not provide locally representative samples and data (e.g., at the substate or county level) to support local public health action to prevent and control chronic diseases. Coordination among the NCHS surveys and with other surveys conducted by CDC or other federal agencies needs to be strengthened to meet the goals of a nationally integrated system. Furthermore, NCHS does not encompass registry data, which are very important to measuring incidence and evaluating treatment of some chronic diseases; has limited capability in web-based query systems; and does not have established relationships with many stakeholders important to collecting and effectively disseminating surveillance data.
The National Heart, Lung, and Blood Institute (NHLBI) is another alternative mechanism that could be used to implement the decisions of the recommendation 1 working group. As the National Cancer Institute relates to the Surveillance Epidemiology and End Results (SEER) cancer surveillance system, so could NHLBI function in relation to a surveillance system for cardiovascular and chronic lung disease. The cancer-based SEER program collects information from population-based cancer registries that cover approximately 26 percent of the population. NCI staff are responsible for overseeing the quality of the system and work with registry staff to ensure data quality. They also implement the decisions of the Change Control Board (CCB), which is responsible for evaluating new features and potential changes to algorithms, database structure, and hardware infrastructure. The NCI also compiles and disseminates reports and findings regarding cancer and interacts, a function that NHLBI currently fulfills for heart and vascular diseases, lung diseases, blood diseases, and sleep disorders.
Arguments in favor of designating NHLBI include its expertise in cardiovascular and chronic lung disease and its history of funding data collection on these topics. Furthermore, it already works closely with many organiza-
tions implementing registries for CVD and could do so for organizations with registries developed for COPD, an important strength if the surveillance system is to meet the data needs of stakeholders nationally and at state and local levels.
Drawbacks include the fact that NHLBI would need to develop capacities it does not currently have or partner with others to collect and manage large data streams, conduct large surveys, marshal the resources necessary to generate data for state and local surveillance, and provide easy access to such data for a wide array of stakeholders. Furthermore, the institute is more focused on a particular set of diseases, so if the system were to expand to chronic disease more broadly, NHLBI would have difficulty expanding such a system without overlapping with other NIH institutes.
To further understand the basis and trajectories of cardiovascular and chronic lung diseases, the information collected by the system must be available and accessible to a variety of stakeholders as discussed in Chapter 6. While data from national surveys conducted by the federal, state, or local governments are usually readily available, private sources of data are frequently inaccessible or accessible only with great difficulty. A greater national investment is needed to ensure that chronic disease surveillance data are accessible to potential data users with a wide range of technical capacities. Federal, state, and local public agencies could play a stronger leadership role in making data accessible to all sectors of society. This role would be especially important to ensure the relevance and accessibility of such data for chronic disease surveillance and policy making at state and local levels as well as nationally.
Recommendation 5
The committee recommends that the Secretary of HHS designate a federal office with the following responsibilities:
- Producing and disseminating regular surveillance reports and key indicators of progress that support and stimulate action aimed at improving health and reducing disparities at the national, state, and local levels;
- Assuring that the surveillance data are accessible to a broad spectrum of users (e.g., public health agencies, health systems, researchers, policy makers, and advocacy groups) at all levels while protecting privacy and documenting the extent of that use; and
- Implementing the recommendations of the national working group recommended in Recommendation 1.
As discussed earlier in this chapter, the life-course perspective is important to understanding the trajectory of chronic diseases. Also needed are comparable data that enable analysis across different subpopulation groups and geographic levels and that can be linked across data sources. Existing data collection mechanisms provide valuable information that, with enhancements, can serve to meet the surveillance needs for CVD and chronic lung disease.
Recommendation 6
The committee recommends that HHS coordinate with voluntary bodies operating disease registries to promote collection and harmonization of data.
Recommendation 7
The committee recommends that governmental and nongovernmental organizations enhance existing national data sources in the following manner:
- Information on all elements of the recommended framework should be collected on the U.S. population across the life span, with special attention paid to collecting information on diverse and changing populations, including information on disparities.
- A minimum subset of actionable indicators as identified by the working group should be collected using comparable measures at the national, state, and local levels.
- Data should be increasingly linked across health domains and data sources.
Effective interventions to prevent CVD and chronic lung disease require tracking information at multiple geographic levels—local, state, and national. Likewise, federal healthcare reform legislation has established a national healthcare coverage and delivery policy, yet much of the implementation will occur at the state level. State and local policy makers, public health leaders, and health professionals need feedback afforded by surveillance systems to inform them of the magnitude of disease and disparities within their geographic areas compared to other areas, as well as the outcomes of their efforts.
Surveillance needs differ among communities. Community-tailored survey items will be necessary to understand the extent to which conditions vary by characteristics such as socioeconomic status, race/ethnicity, or geographic setting (e.g., urban versus rural). As discussed in Chapter 4, these factors are frequently associated with health disparities. To foster efforts to reduce these disparities, a surveillance system must be able to provide data for analysis of disparities not only at the national level but also at the regional, state, and local levels. Furthermore, the system will be most beneficial if comparisons can be made between and among various communities that require collection of comparable data.
Recommendation 8
The committee recommends that HHS develop a cardiovascular and chronic pulmonary disease survey question bank and technical support for use by tribal, state, and local agencies; nongovernmental organizations; and individual researchers for the purpose of enhancing the quality and comparability of population health surveys in order to identify trends in risk factors, diseases, treatments, and outcomes.
As discussed in Chapter 6, there is great potential for the use of electronic health records as sources of surveillance information. Currently, those records focus primarily on recording clinical information (e.g., diagnoses, laboratory work, and treatments). Chapters 2 and 3 described the importance of behavioral, social, and physical environmental risk factors in the development of cardiovascular and chronic lung disease. The Office of the National Coordinator for Health Information Technology (ONCHIT) has the responsibility to identify the minimum data to be collected for EHRs and is in a position to take action that would significantly enhance the surveillance information contained in EHRs.
Recommendation 9
The committee recommends that the Office of the National Coordinator for Health Information Technology expand the minimum data for electronic health records to include behavioral, social, and environmental risk factors for cardiovascular and chronic lung diseases in validated, interoperable ways in order to enhance the quality of surveillance data for these conditions.
Because EHRs are currently in use in only a minority of hospitals and practices, several interim steps are needed before their potential can be realized. Expansion of EHRs to the majority of clinical care settings will require significant investment in purchasing necessary equipment and software as well as staff training. Additional resources will need to be devoted to major issues such as interoperability of EHR systems and harmonization of data standards.
Many existing sources of surveillance information provide high-quality data that are critical to understanding the trajectory of cardiovascular and chronic lung diseases. However, those data lack standardization and cannot be linked across sources, and many of them are not readily accessible. Furthermore, there is a need for collection of data that facilitates analysis by various demographic variables, such as race/ethnicity, socioeconomic status, and geography. The committee believes the recommendations provided in this report lay the foundation or framework for the development of the complex, interdependent system needed.
REFERENCES
Chassin, M. R., J. M. Loeb, S. P. Schmaltz, and R. M. Wachter. 2010. Accountability measures—using measurement to promote quality improvement. New England Journal of Medicine 363(7):683-688.
German, R. R., L. M. Lee, J. M. Horan, R. L. Milstein, C. A. Pertowski, and M. N. Waller. 2001. Updated guidelines for evaluating public health surveillance systems: Recommendations from the guidelines working group. MMWR Recommendations and Reports 50(RR-13):1-35; quiz CE31-37.
Goff, D. C., Jr., L. Brass, L. T. Braun, J. B. Croft, J. D. Flesch, F. G. R. Fowkes, Y. Hong, V. Howard, S. Huston, S. F. Jencks, R. Luepker, T. Manolio, C. O’Donnell, R. Marie Robertson, W. Rosamond, J. Rumsfeld, S. Sidney, and Z. J. Zheng. 2007. Essential features of a surveillance system to support the prevention and management of heart disease and stroke: A scientific statement from the American Heart Association councils on epidemiology and prevention, stroke, and cardiovascular nursing and the interdisciplinary working groups on quality of care and outcomes research and atherosclerotic peripheral vascular disease. Circulation 115(1):127-155.
Last, J. M. 2001. A dictionary of epidemiology. 4th ed. New York: Oxford University Press.
Modeste, N. N. 1996. Dictionary of public health promotion and education: Terms and concepts. Thousand Oaks, CA: Sage Publications.
NCHS (National Center for Health Statistics). 2002. Shaping a health statistics vision for the 21st Century. Hyattsville, MD: Centers for Disease Control and Prevention.
NCHS. 2009. NCHS guidelines for ensuring the quality of information disseminated to the public. http://www.cdc.gov/nchs/about/policy/quality.htm (accessed August 2, 2011).
Wingo, P. A., H. L. Howe, M. J. Thun, R. Ballard-Barbash, E. Ward, M. L. Brown, J. Sylvester, G. H. Friedell, L. Alley, J. H. Rowland, and B. K. Edwards. 2005. A national framework for cancer surveillance in the United States. Cancer Causes and Control 16(2):151-170.
Yeh, R. W., and A. S. Go. 2010. Rethinking the epidemiology of acute myocardial infarction: Challenges and opportunities. Archives of Internal Medicine 170(9):759-764.












